KR20100109293A - Novel organic electroluminescent compounds and organic electroluminescent device using the same - Google Patents
Novel organic electroluminescent compounds and organic electroluminescent device using the same Download PDFInfo
- Publication number
- KR20100109293A KR20100109293A KR1020090027846A KR20090027846A KR20100109293A KR 20100109293 A KR20100109293 A KR 20100109293A KR 1020090027846 A KR1020090027846 A KR 1020090027846A KR 20090027846 A KR20090027846 A KR 20090027846A KR 20100109293 A KR20100109293 A KR 20100109293A
- Authority
- KR
- South Korea
- Prior art keywords
- substituted
- unsubstituted
- alkyl
- organic
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent materials, e.g. electroluminescent or chemiluminescent
- C09K11/06—Luminescent materials, e.g. electroluminescent or chemiluminescent containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/82—Carbazoles; Hydrogenated carbazoles
- C07D209/86—Carbazoles; Hydrogenated carbazoles with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
- C07D213/16—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom containing only one pyridine ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/02—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with only hydrogen atoms or radicals containing only carbon and hydrogen atoms, directly attached to carbon atoms of the nitrogen-containing ring; Alkylene-bis-isoquinolines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/36—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems
- C07D241/38—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems with only hydrogen or carbon atoms directly attached to the ring nitrogen atoms
- C07D241/40—Benzopyrazines
- C07D241/42—Benzopyrazines with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/52—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings condensed with carbocyclic rings or ring systems
- C07D263/54—Benzoxazoles; Hydrogenated benzoxazoles
- C07D263/56—Benzoxazoles; Hydrogenated benzoxazoles with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached in position 2
- C07D263/57—Aryl or substituted aryl radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D265/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one oxygen atom as the only ring hetero atoms
- C07D265/28—1,4-Oxazines; Hydrogenated 1,4-oxazines
- C07D265/34—1,4-Oxazines; Hydrogenated 1,4-oxazines condensed with carbocyclic rings
- C07D265/38—[b, e]-condensed with two six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/60—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings condensed with carbocyclic rings or ring systems
- C07D277/62—Benzothiazoles
- C07D277/64—Benzothiazoles with only hydrocarbon or substituted hydrocarbon radicals attached in position 2
- C07D277/66—Benzothiazoles with only hydrocarbon or substituted hydrocarbon radicals attached in position 2 with aromatic rings or ring systems directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/22—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/91—Dibenzofurans; Hydrogenated dibenzofurans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/76—Dibenzothiophenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0816—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring comprising Si as a ring atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0834—Compounds having one or more O-Si linkage
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1033—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1037—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with sulfur
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/104—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with other heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1059—Heterocyclic compounds characterised by ligands containing three nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1092—Heterocyclic compounds characterised by ligands containing sulfur as the only heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K71/00—Manufacture or treatment specially adapted for the organic devices covered by this subclass
- H10K71/10—Deposition of organic active material
- H10K71/16—Deposition of organic active material using physical vapour deposition [PVD], e.g. vacuum deposition or sputtering
- H10K71/164—Deposition of organic active material using physical vapour deposition [PVD], e.g. vacuum deposition or sputtering using vacuum deposition
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Quinoline Compounds (AREA)
- Indole Compounds (AREA)
Abstract
본 발명은 신규한 유기 발광 화합물 및 이를 포함하고 있는 유기 전계 발광 소자에 관한 것으로, 상세하게는 본 발명에 따른 유기 발광 화합물은 하기 화학식 1로 표시되는 것을 특징으로 한다.The present invention relates to a novel organic light emitting compound and an organic electroluminescent device comprising the same, in detail, the organic light emitting compound according to the invention is characterized in that represented by the formula (1).
[화학식 1][Formula 1]
본 발명에 따른 유기 발광 화합물은 발광효율이 좋고 재료의 색순도 및 수명특성이 뛰어나 구동수명이 매우 우수한 OLED 소자를 제조할 수 있는 장점이 있다.The organic light emitting compound according to the present invention has the advantage of being able to manufacture an OLED device having a good luminous efficiency and excellent color purity and life characteristics of the material and excellent driving life.
Description
본 발명은 신규한 유기 발광 화합물 및 이를 채용하고 있는 유기 전계 발광 소자에 관한 것이다. 구체적으로는 전계 발광 특성을 갖는 신규한 안트라센 유도체 및 이를 발광 물질로서 이용한 유기 전계 발광 소자에 관한 것이다.The present invention relates to a novel organic light emitting compound and an organic electroluminescent device employing the same. Specifically, the present invention relates to a novel anthracene derivative having electroluminescent properties and an organic electroluminescent device using the same as a light emitting material.
표시 소자 중, 전기 발광 소자(electroluminescence device: EL device)는 자체 발광형 표시 소자로서 시야각이 넓고 콘트라스트가 우수할 뿐만 아니라 응답속도가 빠르다는 장점을 가지고 있으며, 1987년 이스트만 코닥(Eastman Kodak)사에서는 발광층 형성용 재료로서 저분자인 방향족 디아민과 알루미늄 착물을 이용하고 있는 유기 EL 소자를 처음으로 개발하였다[Appl. Phys. Lett. 51, 913, 1987].Among the display elements, an electroluminescence device (EL device) is a self-luminous display element that has a wide viewing angle, excellent contrast, and high response speed.Eastman Kodak Co., Ltd. in 1987 An organic EL device using a low molecular aromatic diamine and an aluminum complex as a light emitting layer formation material was first developed [Appl. Phys. Lett. 51, 913, 1987].
유기 EL 소자는 전자 주입 전극(음극) 과 정공 주입 전극(양극) 사이에 형성된 유기막에 전하를 주입하면 전자와 정공이 쌍을 이룬 후 소멸하면서 빛을 내는 소자이다. 플라스틱 같은 휠 수 있는(flexible) 투명 기판 위에도 소자를 형성할 수 있을 뿐 아니라, 플라즈마 디스플레이 패널(Plasma Display Panel)이나 무기 EL 디스플레이에 비해 낮은 전압에서 (10V이하) 구동이 가능하고, 또한 전력 소모가 비교적 적으며, 색감이 뛰어나다는 장점이 있다. 또한 유기 EL 소자는 녹색, 청색, 적색의 3가지 색을 나타낼 수가 있어 차세대 풍부한 색 디스플레이 소자로 많은 사람들의 많은 관심의 대상이 되고 있다. An organic EL device is a device that emits light when an electron is injected into an organic film formed between an electron injection electrode (cathode) and a hole injection electrode (anode) and then disappears after pairing electrons and holes. The device can be formed on a flexible transparent substrate such as plastic, and can be driven at a lower voltage (less than 10V) compared to a plasma display panel or an inorganic EL display, and also consumes power. It is relatively small and has the advantage of excellent color. In addition, the organic EL device can display three colors of green, blue, and red, and thus, has become a subject of much interest as a next-generation rich color display device.
유기 EL 소자에서 발광 효율, 수명 등의 성능을 결정하는 가장 중요한 요인은 발광 재료로서, 이러한 발광 재료에 요구되는 몇 가지 특성으로는 고체상태에서 발광 양자 수율이 커야하고, 전자와 정공의 이동도가 높아야 하며, 진공 증착시 쉽게 분해되지 않아야 하고, 균일한 박막을 형성, 안정해야한다. The most important factor that determines the performance of light emission efficiency, lifespan, etc. in the organic EL device is the light emitting material. Some characteristics required for such a light emitting material include high light emission quantum yield in the solid state, and mobility of electrons and holes. It should be high, not easily decomposed during vacuum deposition, and should form and stabilize a uniform thin film.
유기EL소자는 보통 anode/HIL/HTL/EML/ETL/EIL/cathode로 구성되는데 발광층(EML)을 어떻게 형성하느냐에 따라 청색, 녹색, 적색의 유기 전기 발광 소자를 각각 구현할 수가 있다. The organic EL device is usually composed of anode / HIL / HTL / EML / ETL / EIL / cathode. The organic electroluminescent device of blue, green, and red can be realized depending on how the emission layer (EML) is formed.
발광재료는 기능적인 측면에서 호스트 재료와 도판트 재료로 구분될 수 있는데 일반적으로 EL 특성이 가장 우수한 소자 구조로는 호스트에 도판트를 도핑하여 발광층을 만드는 것으로 알려져 있다. 최근에 고효율, 장수명 유기 EL 소자의 개발이 시급한 과제로 대두되고 있으며, 특히 중대형 OLED 패널에서 요구하고 있는 EL 특성 수준을 고려해 볼 때 기존의 발광재료에 비해 매우 우수한 재료의 개발이 시급한 실정이다.The light emitting material can be classified into a host material and a dopant material in terms of its function. In general, a device structure having excellent EL characteristics is known to make a light emitting layer by doping a host with a dopant. Recently, the development of high efficiency and long life organic EL devices has emerged as an urgent task, and considering the level of EL characteristics required in medium and large OLED panels, it is urgent to develop materials that are much superior to existing light emitting materials.
한편, 녹색 형광 재료로는 Alq를 호스트로 하여, 도판트로는 쿠마린 유도체(화합물 d, C545T), 퀴나크리돈 유도체(화합물 e), DPT(화합물 f) 등을 수 내지 십수 % 정도로 도핑을 하는 시스템이 개발되어 널리 쓰이고 있다. 그러나, 이들 종래 의 발광재료는 초기 발광효율의 경우, 상용화 가능한 수준의 성능을 보이나, 초기 효율 저하가 두드러지며 수명 측면에서 상당한 문제점을 보이고 있어, 대화면의 고성능 패널에서는 채택하기가 힘든 한계를 보이고 있다.On the other hand, as a green fluorescent material, Alq is used as a host, and as a dopant, doping coumarin derivatives (compounds d and C545T), quinacridone derivatives (compound e), DPT (compound f) and the like are about 10 to 10%. It is developed and widely used. However, these conventional light emitting materials show a commercially available level of performance in the case of the initial luminous efficiency, but the initial efficiency is markedly deteriorated and shows considerable problems in terms of lifespan, which is difficult to adopt in large screen high performance panels. .
또한, OLED 소자에서의 수명 측면에서도 결코 만족할만한 수준이 되질 못하여 더욱 안정되고, 더욱 성능이 뛰어난 호스트 재료의 개발이 요구되고 있다.In addition, in terms of lifespan in OLED devices, they are never satisfactory, and development of a more stable and more excellent host material is required.
본 발명의 목적은 상기와 같은 종래 기술의 문제점을 해결하기 위한 것으로, 기존의 호스트 재료보다 발광 효율 및 소자 수명이 좋으며, 적절한 색좌표를 갖는 우수한 골격의 유기 발광 화합물 및 이를 발광재료로 채용하는 고효율 및 장수명의 유기 전계 발광 소자를 제공하는 것이다. An object of the present invention is to solve the problems of the prior art as described above, the luminous efficiency and device life is better than the conventional host material, an organic light emitting compound having an excellent skeleton having a suitable color coordinate and a high efficiency and employing it as a light emitting material and It is to provide a long life organic electroluminescent device.
상기 목적을 달성하기 위하여 본 발명에서는 신규한 유기 발광 화합물을 제조하였으며, 이러한 화합물들이 유기 전계 발광 소자에서 발광 물질로 사용될 수 있고, 특히 발광 호스트, 바람직하게는 녹색 호스트로 사용됨으로써, 유기 전계 발광 소자의 효율 상승, 구동전압 하강 및 안정성 상승효과를 제공한다.In order to achieve the above object, in the present invention, a novel organic light emitting compound has been prepared, and these compounds can be used as a light emitting material in an organic electroluminescent device, and in particular, as a light emitting host, preferably a green host, thereby providing an organic electroluminescent device. Increase efficiency, drive voltage drop and stability increase effect.
본 발명은 하기 화학식 1의 유기 발광 화합물을 제공한다.The present invention provides an organic light emitting compound of the formula (1).
[화학식 1][Formula 1]
[상기 화학식 1에서,[In Formula 1,
L1 및 L2는 서로 독립적으로 화학결합이거나, 치환 또는 비치환된 (C6-C30)아릴렌, 치환 또는 비치환된 (C3-C30)헤테로아릴렌, 치환 또는 비치환된 5원 내지 7원의 헤테로시클로알킬렌, 치환 또는 비치환된 방향족고리가 하나이상 융합된 5원 내지 7원의 헤테로시클로알킬렌, 치환 또는 비치환된(C3-C30)시클로알킬렌, 치환 또는 비치환된 방향족고리가 하나이상 융합된 (C3-C30)시클로알킬렌, 치환 또는 비치환된 아다만틸렌, 치환 또는 비치환된 (C7-C30)바이시클로알킬렌, 치환 또는 비치환된(C2-C30)알케닐렌, 치환 또는 비치환된(C2-C30)알키닐렌, 치환 또는 비치환된(C6-C30)아르(C1-C30)알킬렌, 치환 또는 비치환된(C1-C30)알킬렌티오, 치환 또는 비치환된(C1-C30)알킬렌옥시, 치환 또는 비치환된(C6-C30)아릴렌옥시, 치환 또는 비치환된(C6-C30)아릴렌티오, -O- 또는 -S-이고;L 1 and L 2 are each independently a chemical bond or a substituted or unsubstituted (C6-C30) arylene, a substituted or unsubstituted (C3-C30) heteroarylene, a substituted or unsubstituted 5 to 7 member Heterocycloalkylene, 5- to 7-membered heterocycloalkylene fused with one or more substituted or unsubstituted aromatic rings, Substituted or unsubstituted (C3-C30) cycloalkylene, substituted or unsubstituted aromatic ring (C3-C30) cycloalkylene, substituted or unsubstituted adamantylene, substituted or unsubstituted ( C7-C30) bicycloalkylene, substituted or unsubstituted (C2-C30) alkenylene, substituted or unsubstituted (C2-C30) alkynylene, substituted or unsubstituted (C6-C30) ar (C1-C30) ) Alkylene, substituted or unsubstituted (C1-C30) alkylenethio, substituted or unsubstituted (C1-C30) alkyleneoxy, substituted or unsubstituted (C6-C30) aryleneoxy, substituted or unsubstituted (C6-C30) arylenethio, -O- or -S-;
R1 내지 R4, Ar1은 각각 독립적으로 수소, 중수소, 할로겐, 치환 또는 비치환된(C1-C30)알킬, 치환 또는 비치환된(C6-C30)아릴, 치환 또는 비치환된 (C3-C30)시클로알킬이 하나이상 융합된 치환 또는 비치환된(C6-C30)아릴, 치환 또는 비치환된(C3-C30)헤테로아릴, 치환 또는 비치환된 5원 내지 7원의 헤테로시클로알킬, 치환 또는 비치환된 방향족고리가 하나이상 융합된 5원 내지 7원의 헤테로시클로알킬, 치환 또는 비치환된(C3-C30)시클로알킬, 치환 또는 비치환된 방향족고리가 하나이상 융합된 (C3-C30)시클로알킬, 치환 또는 비치환된 아다만틸, 치환 또는 비치환된(C7-C30)바이시클로알킬, 시아노, NR11R12, BR13R14, PR15R16, P(=O)R17R18[R11 내지 R18은 서로 독립적으로 치환 또는 비치환된(C1-C30)알킬, 치환 또는 비치환된(C6-C30)아릴 또는 치환 또는 비치환된(C3-C30)헤테로아릴이다.], 치환 또는 비치환된 트리(C1-C30)알킬실릴, 치환 또는 비치환된 디(C1-C30)알킬(C6-C30)아릴실릴, 치환 또는 비치환된 트리(C6-C30)아릴실릴, 치환 또는 비치환된 (C6-C30)아르(C1-C30)알킬, 치환 또는 비치환된(C1-C30)알킬옥시, 치환 또는 비치환된(C1-C30)알킬티오, 치환 또는 비치환된(C6-C30)아릴옥시, 치환 또는 비치환된(C6-C30)아릴티오, 치환 또는 비치환된(C1-C30)알콕시카보닐, 치환 또는 비치환된(C1-C30)알킬카보닐, 치환 또는 비치환된(C6-C30)아릴카보닐, 치환 또는 비치환된(C2-C30)알케닐, 치환 또는 비치환된(C2-C30)알키닐, 치환 또는 비치환된(C6-C30)아릴옥시카보닐, 치환 또는 비치환된(C1-C30)알콕시카보닐옥시, 치환 또는 비치환된(C1-C30)알킬카보닐옥시, 치환 또는 비치환된(C6-C30)아릴카보닐옥시, 치환 또는 비치환된(C6-C30)아릴옥시카보닐옥시, 카르복실, 나이트로, , 또는 하이드록시이거나 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있으며;R 1 to R 4 , Ar 1 are each independently Substituted or unsubstituted fused with one or more hydrogen, deuterium, halogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C3-C30) cycloalkyl Substituted (C6-C30) aryl, substituted or unsubstituted (C3-C30) heteroaryl, substituted or unsubstituted 5- to 7-membered heterocycloalkyl, substituted or unsubstituted aromatic ring 5 1-7 membered heterocycloalkyl, Substituted or unsubstituted (C3-C30) cycloalkyl, substituted or unsubstituted aromatic ring (C3-C30) cycloalkyl, substituted or unsubstituted adamantyl, substituted or unsubstituted (C7- C30) bicycloalkyl, cyano, NR 11 R 12 , BR 13 R 14 , PR 15 R 16 , P (═O) R 17 R 18 [R 11 to R 18 are each independently substituted or unsubstituted (C 1 -C30) alkyl, substituted or unsubstituted (C6-C30) aryl or substituted or unsubstituted (C3-C30) heteroaryl], substituted or unsubstituted tri (C1-C30) alkylsilyl, substituted or unsubstituted Substituted di (C1-C30) alkyl (C6-C30) arylsilyl, substituted or unsubstituted tri (C6-C30) arylsilyl, substituted or unsubstituted (C6-C30) ar (C1-C30) alkyl, substituted Or unsubstituted (C1-C30) alkyloxy, substituted or unsubstituted (C1-C30) alkylthio, substituted or unsubstituted (C6-C30) aryloxy, substituted or unsubstituted (C6-C30) arylthio , Substituted or unsubstituted (C1-C30) alkoxycarbonyl, substituted or non- Substituted (C1-C30) alkylcarbonyl, substituted or unsubstituted (C6-C30) arylcarbonyl, substituted or unsubstituted (C2-C30) alkenyl, substituted or unsubstituted (C2-C30) alkynyl, Substituted or unsubstituted (C6-C30) aryloxycarbonyl, substituted or unsubstituted (C1-C30) alkoxycarbonyloxy, substituted or unsubstituted (C1-C30) alkylcarbonyloxy, substituted or unsubstituted (C6-C30) arylcarbonyloxy, substituted or unsubstituted (C6-C30) aryloxycarbonyloxy, carboxyl, nitro, , Or may be linked with (C3-C30) alkylene or (C3-C30) alkenylene, which may or may not be hydroxy or contain a fused ring with adjacent substituents to form an alicyclic ring and a monocyclic or polycyclic aromatic ring;
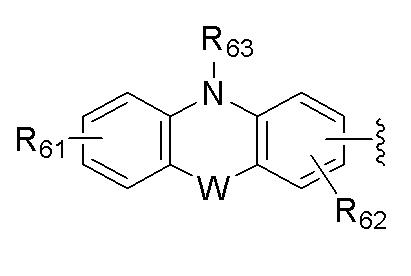
W는 -(CR51R52)m-, -(R51)C=C(R52)-, -N(R53)-, -S-, -O-, -Si(R54)(R55)-, -P(R56)-, -P(=O)(R57)-, -C(=O)- 또는 -B(R58)-이고, R51 내지 R58 및 R61 내지 R63은 상기 R1 내지 R4에서의 정의와 동일하고;W is-(CR 51 R 52 ) m -,-(R 51 ) C = C (R 52 )-, -N (R 53 )-, -S-, -O-, -Si (R 54 ) (R 55 )-, -P (R 56 )-, -P (= 0) (R 57 )-, -C (= 0)-or -B (R 58 )-, and R 51 to R 58 and R 61 to R 63 is as defined in R 1 to R 4 above;
상기 헤테로시클로알킬 및 헤테로아릴은 B, N, O, S, P(=O), Si 및 P로부터 선택된 하나 이상의 헤테로원자를 포함하며; The heterocycloalkyl and heteroaryl include one or more heteroatoms selected from B, N, O, S, P (= 0), Si and P;
m은 1 또는 2의 정수이다.] m is an integer of 1 or 2.]
본 발명에 기재된 “알킬”, “알콕시” 및 그 외 “알킬”부분을 포함하는 치환체는 직쇄 또는 분쇄 형태를 모두 포함한다. Substituents comprising the "alkyl", "alkoxy" and other "alkyl" moieties described herein include both straight and pulverized forms.
본 발명에 기재된 「아릴」은 하나의 수소 제거에 의해서 방향족 탄화수소로부터 유도된 유기 라디칼로, 각 고리에 적절하게는 4 내지 7개, 바람직하게는 5 또는 6개의 고리원자를 포함하는 단일 또는 융합고리계를 포함하며, 다수개의 아릴이 단일결합으로 연결되어 있는 형태까지 포함한다. 구체적인 예로 페닐, 나프틸, 비페닐, 안트릴, 인데닐(indenyl), 플루오레닐, 페난트릴, 트리페닐레닐, 피렌일, 페릴렌일, 크라이세닐, 나프타세닐, 플루오란텐일 등을 포함하지만, 이에 한정되지 않는다. 상기 나프틸은 1-나프틸 및 2-나프틸을 포함하며, 안트릴은 1-안트릴, 2-안트릴 및 9-안트릴을 포함하며, 플루오레닐은 1-플루오레닐, 2-플루오레닐, 3-플루오레닐, 4-플루오레닐 및 9-플루오레닐을 모두 포함한다. "Aryl" described in the present invention is an organic radical derived from an aromatic hydrocarbon by one hydrogen removal, and a single or fused ring containing 4 to 7, preferably 5 or 6 ring atoms in each ring as appropriate. It includes a system, including a form in which a plurality of aryl is connected by a single bond. Specific examples include phenyl, naphthyl, biphenyl, anthryl, indenyl, fluorenyl, phenanthryl, triphenylenyl, pyrenyl, peryleneyl, chrysenyl, naphthasenyl, fluoranthenyl, and the like. It is not limited to this. Said naphthyl includes 1-naphthyl and 2-naphthyl, anthryl includes 1-anthryl, 2-anthryl and 9-anthryl, and fluorenyl is 1-fluorenyl, 2- Fluorenyl, 3-fluorenyl, 4-fluorenyl and 9-fluorenyl.
본 발명에 기재된 「헤테로아릴」은 방향족 고리 골격 원자로서 B, N, O, S, P(=O), Si 및 P로부터 선택되는 1 내지 4개의 헤테로원자를 포함하고, 나머지 방향족 고리 골격 원자가 탄소인 아릴 그룹을 의미하는 것으로, 5 내지 6원 단환 헤테 로아릴, 및 하나 이상의 벤젠환과 축합된 다환식 헤테로아릴이며, 부분적으로 포화될 수도 있다. 또한, 본 발명에서의 헤테로아릴은 하나 이상의 헤테로아릴이 단일결합으로 연결된 형태도 포함한다. 상기 헤테로아릴기는 고리 내 헤테로원자가 산화되거나 사원화되어, 예를 들어 N-옥사이드 또는 4차 염을 형성하는 2가 아릴 그룹을 포함한다. 구체적인 예로 퓨릴, 티오펜일, 피롤릴, 이미다졸릴, 피라졸릴, 티아졸릴, 티아디아졸릴, 이소티아졸릴, 이속사졸릴, 옥사졸릴, 옥사디아졸릴, 트리아진일, 테트라진일, 트리아졸릴, 테트라졸릴, 퓨라잔일, 피리딜, 피라진일, 피리미딘일, 피리다진일 등의 단환 헤테로아릴, 벤조퓨란일, 벤조티오펜일, 이소벤조퓨란일, 벤조이미다졸릴, 벤조티아졸릴, 벤조이소티아졸릴, 벤조이속사졸릴, 벤조옥사졸릴, 이소인돌릴, 인돌릴, 인다졸릴, 벤조티아디아졸릴, 퀴놀릴, 이소퀴놀릴, 신놀리닐, 퀴나졸리닐, 퀴녹살리닐, 카바졸릴, 페난트리딘일, 벤조디옥솔릴 등의 다환식 헤테로아릴 및 이들의 상응하는 N-옥사이드(예를 들어, 피리딜 N-옥사이드, 퀴놀릴 N-옥사이드), 이들의 4차 염 등을 포함하지만, 이에 한정되지 않는다.The "heteroaryl" described in the present invention contains 1 to 4 heteroatoms selected from B, N, O, S, P (= O), Si, and P as aromatic ring skeleton atoms, and the remaining aromatic ring skeleton atoms are carbon. Meaning an aryl group which is 5 to 6 membered monocyclic heteroaryl, and polycyclic heteroaryl condensed with one or more benzene rings, and may be partially saturated. In addition, heteroaryl in the present invention also includes a form in which one or more heteroaryl is connected by a single bond. Such heteroaryl groups include divalent aryl groups in which heteroatoms in the ring are oxidized or quaternized to form, for example, N-oxides or quaternary salts. Specific examples include furyl, thiophenyl, pyrrolyl, imidazolyl, pyrazolyl, thiazolyl, thiadiazolyl, isothiazolyl, isoxazolyl, oxazolyl, oxadiazolyl, triazinyl, tetrazinyl, triazolyl, tetra Monocyclic heteroaryl such as zolyl, furazanyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, benzofuranyl, benzothiophenyl, isobenzofuranyl, benzoimidazolyl, benzothiazolyl, benzoisothia Zolyl, Benzoisoxazolyl, Benzoxazolyl, Isoindoleyl, Indolyl, Indazolyl, Benzothiadiazolyl, Quinolyl, Isoquinolyl, Cinolinyl, Quinazolinyl, Quinoxalinyl, Carbazolyl, Phenantridinyl , Polycyclic heteroaryls such as benzodioxolyl and the like, and their corresponding N-oxides (eg, pyridyl N-oxides, quinolyl N-oxides), quaternary salts thereof, and the like. .
또한, 본 발명에 기재되어 있는 “(C1-C30)알킬, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, (C6-C30)아르(C1-C30)알킬, (C1-C30)알킬옥시, (C1-C30)알킬티오, (C1-C30)알킬옥시카보닐, (C1-C30)알킬카보닐, (C1-C30)알킬옥시카보닐옥시, (C1-C30)알킬카보닐옥시” 등의 알킬은 탄소수 1 내지 20개로 제한될 수 있고, 탄소수 1 내지 10개로 제한될 수 있다. “(C6-C30)아릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, (C6-C30)아르(C1-C30)알킬, (C6-C30)아릴옥시, (C6-C30)아릴티오, (C6-C30)아릴카보닐, (C6-C30)아릴옥시카보닐, (C6- C30)아릴카보닐옥시, (C6-C30)아릴옥시카보닐옥시”등의 아릴은 탄소수 6 내지 20개로 제한될 수 있고, 탄소수 6 내지 12개로 제한될 수 있다. “(C3-C30)헤테로아릴”의 헤테로아릴은 탄소수 4 내지 20개로 제한될 수 있고, 탄소수 4 내지 12개로 제한될 수 있다. “(C3-C30)시클로알킬”의 헤테로아릴은 탄소수 3 내지 20개로 제한될 수 있고, 탄소수 3 내지 7개로 제한될 수 있다. “(C2-C30)알케닐 또는 알키닐”의 알케닐 또는 알키닐은 탄소수 2 내지 20개로 제한될 수 있고, 탄소수 2 내지 10개로 제한될 수 있다.Also described herein are “(C1-C30) alkyl, tri (C1-C30) alkylsilyl, di (C1-C30) alkyl (C6-C30) arylsilyl, (C6-C30) ar (C1-C30) ) Alkyl, (C1-C30) alkyloxy, (C1-C30) alkylthio, (C1-C30) alkyloxycarbonyl, (C1-C30) alkylcarbonyl, (C1-C30) alkyloxycarbonyloxy, ( Alkyl, such as "C1-C30) alkylcarbonyloxy", may be limited to 1 to 20 carbon atoms, and may be limited to 1 to 10 carbon atoms. “(C6-C30) aryl, di (C1-C30) alkyl (C6-C30) arylsilyl, tri (C6-C30) arylsilyl, (C6-C30) ar (C1-C30) alkyl, (C6-C30) Aryloxy, (C6-C30) arylthio, (C6-C30) arylcarbonyl, (C6-C30) aryloxycarbonyl, (C6-C30) arylcarbonyloxy, (C6-C30) aryloxycarbonyloxy Aryl such as ”may be limited to 6 to 20 carbon atoms, and may be limited to 6 to 12 carbon atoms. Heteroaryl of "(C3-C30) heteroaryl" may be limited to 4 to 20 carbon atoms, it may be limited to 4 to 12 carbon atoms. Heteroaryl of "(C3-C30) cycloalkyl" may be limited to 3 to 20 carbon atoms, it may be limited to 3 to 7 carbon atoms. Alkenyl or alkynyl of "(C2-C30) alkenyl or alkynyl" may be limited to 2 to 20 carbon atoms, and may be limited to 2 to 10 carbon atoms.
또한 본 발명에 기재되어 있는 “치환 또는 비치환”이라는 기재는 각각 독립적으로 중수소, 할로겐, 할로겐이 치환 또는 비치환된 (C1-C30)알킬, (C6-C30)아릴, (C6-C30)아릴이 치환 또는 비치환된 (C3-C30)헤테로아릴, B, N, O, S, P(=O), Si 및 P로부터 선택된 하나 이상을 포함하는 5원 내지 7원의 헤테로시클로알킬, 방향족고리가 하나이상 융합된 5원 내지 7원의 헤테로시클로알킬, (C3-C30)시클로알킬, 방향족고리가 하나이상 융합된 (C6-C30)시클로알킬, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, (C7-C30)바이시클로알킬, (C2-C30)알케닐, (C2-C30)알키닐, 시아노, 카바졸릴, NR21R22, BR23R24, PR25R26, P(=O)R27R28[R21 내지 R28은 서로 독립적으로 (C1-C30)알킬, (C6-C30)아릴 또는 (C3-C30)헤테로아릴이다.], (C6-C30)아르(C1-C30)알킬, (C1-C30)알킬(C6-C30)아릴, (C1-C30)알킬옥시, (C1-C30)알킬티오, (C6-C30)아릴옥시, (C6-C30)아릴티오, (C1-C30)알콕시카보닐, (C1-C30)알킬카보닐, (C6-C30)아릴카보닐, (C6-C30)아릴옥 시카보닐, (C1-C30)알콕시카보닐옥시, (C1-C30)알킬카보닐옥시, (C6-C30)아릴카보닐옥시, (C6-C30)아릴옥시카보닐옥시, 카르복실, 나이트로 또는 하이드록시로 이루어진 군으로부터 선택된 하나 이상으로 더 치환되거나, 서로 인접한 치환체가 연결되어 고리를 형성하는 것을 의미한다.In addition, the description of "substituted or unsubstituted" described in the present invention is each independently deuterium, halogen, (C1-C30) alkyl, (C6-C30) aryl, (C6-C30) aryl substituted or unsubstituted 5- to 7-membered heterocycloalkyl, aromatic rings containing one or more selected from these substituted or unsubstituted (C3-C30) heteroaryl, B, N, O, S, P (= 0), Si and P 5- to 7-membered heterocycloalkyl, at least one fused, (C3-C30) cycloalkyl, at least one fused (C6-C30) cycloalkyl, tri (C1-C30) alkylsilyl, di (C1) -C30) alkyl (C6-C30) arylsilyl, tri (C6-C30) arylsilyl, adamantyl, (C7-C30) bicycloalkyl, (C2-C30) alkenyl, (C2-C30) alkynyl, Cyano, carbazolyl, NR 21 R 22 , BR 23 R 24 , PR 25 R 26 , P (═O) R 27 R 28 [R 21 to R 28 are independently of each other (C1-C30) alkyl, (C6- C30) aryl or (C3-C30) heteroaryl], (C6-C30) ar (C1-C30) alkyl, (C1-C30) alkyl (C6) -C30) aryl, (C1-C30) alkyloxy, (C1-C30) alkylthio, (C6-C30) aryloxy, (C6-C30) arylthio, (C1-C30) alkoxycarbonyl, (C1-C30 ) Alkylcarbonyl, (C6-C30) arylcarbonyl, (C6-C30) aryloxycarbonyl, (C1-C30) alkoxycarbonyloxy, (C1-C30) alkylcarbonyloxy, (C6-C30) aryl Carbonyloxy, (C6-C30) aryloxycarbonyloxy, carboxyl, nitro or hydroxy is further substituted with one or more selected from the group consisting of, or means that adjacent substituents are connected to form a ring.
상기 L1 및 L2는 서로 독립적으로 화학결합이거나, 페닐렌, 나프틸렌, 안트라세닐렌, 비페닐렌, 플루오레닐렌, 트리페닐레닐렌, 플루오란테닐렌, 크리세닐렌, 터페닐렌, 페난트릴렌, 피레닐렌, 퍼릴레닐렌 등의 아릴렌, 피리디닐렌, 피라지닐렌, 퓨릴렌, 티에닐렌, 셀레노페닐렌, 퀴놀리닐렌, 퀴녹살리닐렌, 페난트롤리닐렌 등의 헤테로아릴렌으로부터 선택되어지나, 이에 한정되는 것은 아니며, 상기 화학식 1에서와 같이 더 치환될 수 있다.L 1 and L 2 are each independently a chemical bond, phenylene, naphthylene, anthracenylene, biphenylene, fluorenylene, triphenylenylene, fluoranthrenylene, chryrenylene, terphenylene, Heteroarylene, such as arylene, pyridinylene, pyrazinylene, furylene, thienylene, selenophenylene, quinolinylene, quinoxalinylene, phenanthrolinylene, such as phenanthrylene, pyrenylene, and peryleneylene It is selected from, but is not limited thereto, and may be further substituted as in the formula (1).
또한, 상기 R1 내지 R4, Ar1은 서로 독립적으로 페닐, 나프틸, 안트릴, 비페닐, 플루오레닐, 페난트릴, 피레닐, 퍼릴레닐 등의 아릴, 피리디닐, 피라지닐, 퓨릴, 티에닐, 셀레노페닐, 퀴놀리닐, 퀴녹살리닐, 페난트롤리닐, 카바졸릴, 벤조피레리디닐, 등의 헤테로아릴, 테트라하이드로나프틸 등의 시클로알킬이 융합된 아릴, 벤조피페리디노, 디벤조모폴리노, 디벤조아제피노 등의 하나 이상의 방향족고리가 융합된 헤테로시클로알킬, NR11R12, BR13R14, PR15R16, 또는 P(=O)R17R18[R11 내지 R18은 서로 독립적으로 치환 또는 비치환된(C1-C30)알킬, 치환 또는 비치환된(C6-C30)아릴 또는 치환 또는 비치환된(C3-C30)헤테로아릴이다.]로부터 선택되어지나, 이에 한정되는 것은 아니며, 상기 화학식 1에서와 같이 더 치환될 수 있다.In addition, the R 1 to R 4 , Ar 1 are independently of each other aryl such as phenyl, naphthyl, anthryl, biphenyl, fluorenyl, phenanthryl, pyrenyl, perylenyl, pyridinyl, pyrazinyl, furyl Heteroaryl, such as thienyl, selenophenyl, quinolinyl, quinoxalinyl, phenanthrolinyl, carbazolyl, benzopyreridinyl, and aryl, benzopiperidino fused with cycloalkyl such as tetrahydronaphthyl Heterocycloalkyl fused with one or more aromatic rings such as dibenzomorpholino, dibenzoazino, NR 11 R 12 , BR 13 R 14 , PR 15 R 16 , or P (═O) R 17 R 18 [R 11 to R 18 are independently substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6- C30) aryl or substituted or unsubstituted (C3-C30) heteroaryl.], But is not limited thereto, and may be further substituted as in Chemical Formula 1.
또한, 상기 또는 는 구체적으로 하기 구조로 예시될 수 있다.In addition, or Specifically, it can be illustrated by the following structure.
[R51 내지 R58은 서로 독립적으로 치환 또는 비치환된 (C1-C30)알킬, 치환 또는 비치환된 (C6-C30)아릴 또는 치환 또는 비치환된(C3-C30)헤테로아릴이거나, 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있다.][R 51 to R 58 are independently of each other substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl or substituted or unsubstituted (C3-C30) heteroaryl, or an adjacent substituent. And (C3-C30) alkylene or (C3-C30) alkenylene with or without a fused ring to form an alicyclic ring and a monocyclic or polycyclic aromatic ring.]
본 발명에 따른 유기 발광 화합물은 보다 구체적으로 하기의 화합물로서 예시될 수 있으나, 하기 화합물이 본 발명을 한정하는 것은 아니다.The organic light emitting compound according to the present invention may be more specifically exemplified as the following compound, but the following compound does not limit the present invention.
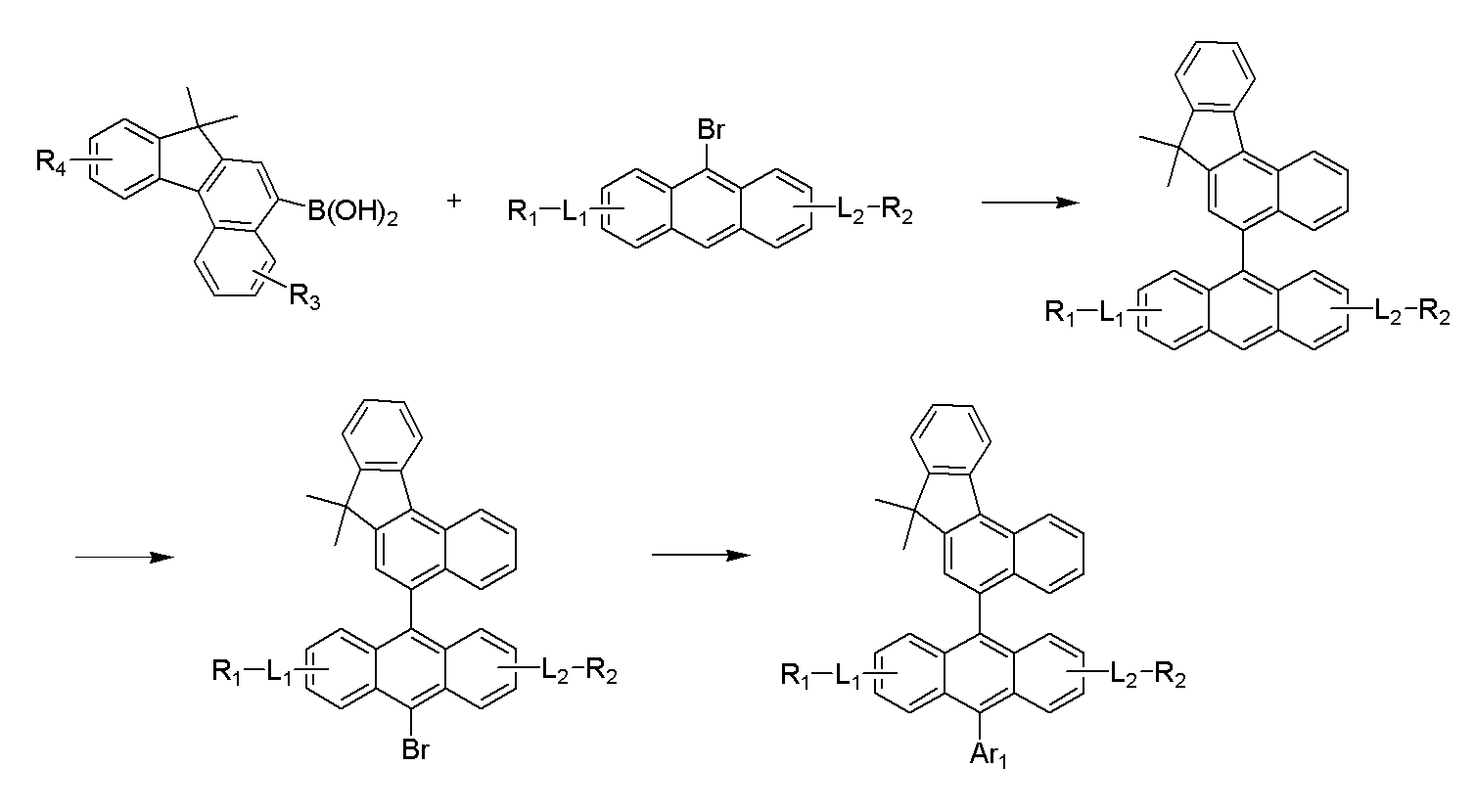
본 발명에 따른 유기 발광 화합물은 하기 반응식 1과 2에 나타난 바와 같이, 제조될 수 있으나, 이에 한정되는 것은 아니다.The organic light emitting compound according to the present invention may be prepared as shown in Schemes 1 and 2, but is not limited thereto.
[반응식 1]Scheme 1
[반응식 2]Scheme 2
[상기 반응식 1및 2에서 R1 내지 R4, L1, L2 및 Ar1은 상기 화학식 1에서의 정의와 동일하다.][R 1 to R 4 , L 1 , L 2 and Ar 1 in Schemes 1 and 2 are the same as defined in Formula 1 above.]
본 발명은 유기 전계 발광 소자를 제공하며, 본 발명에 따른 유기 전계 발광 소자는 제1전극; 제2전극; 및 상기 제1전극 및 제2전극 사이에 개재되는 1층 이상의 유기물층으로 이루어진 유기 전계 발광 소자에 있어서, 상기 유기물층은 상기 화학식 1의 유기 발광 화합물을 하나 이상 포함하는 것을 특징으로 한다. 상기 유기 발광 화합물은 발광층의 호스트 물질로 사용되어진다. The present invention provides an organic electroluminescent device, the organic electroluminescent device according to the present invention comprises a first electrode; A second electrode; And at least one organic material layer interposed between the first electrode and the second electrode, wherein the organic material layer includes at least one organic light emitting compound of Formula 1. The organic light emitting compound is used as a host material of the light emitting layer.
또한, 상기 유기물층은 발광층을 포함하며, 상기 발광층은 상기 화학식 1의 하나 이상의 유기 발광 화합물 이외에 하나 이상의 도판트를 더 포함하는 것을 특징으로 하며, 본 발명의 유기 발광 소자에 적용되는 도판트는 특별히 제한되지는 않는다.The organic material layer may include a light emitting layer, and the light emitting layer may further include one or more dopants in addition to the one or more organic light emitting compounds of Formula 1, and the dopant applied to the organic light emitting device of the present invention is not particularly limited. Does not.
본 발명의 유기 전계 발광 소자에 적용되는 도판트는 하기 화학식 2 또는 화학식 3의 화합물로부터 선택되는 것이 바람직하다.The dopant applied to the organic electroluminescent device of the present invention is preferably selected from the compounds of the following formula (2) or (3).
[화학식 2] [Formula 2]
[화학식 3](3)
[상기 화학식 3에서,[In Formula 3,
L은 치환 또는 비치환된 (C6-C30)아릴렌, 치환 또는 비치환된 (C3-C30)헤테로아릴렌이고, L is substituted or unsubstituted (C6-C30) arylene, substituted or unsubstituted (C3-C30) heteroarylene,
R151 내지 R154는 서로 독립적으로 수소, 중수소, 할로겐, 치환 또는 비치환된(C1-C30)알킬, 치환 또는 비치환된(C6-C30)아릴, 치환 또는 비치환된(C3-C30)헤테로아릴, 치환 또는 비치환된 5원 내지 7원의 헤테로시클로알킬, 치환 또는 비치 환된 방향족고리가 하나이상 융합된 5원 내지 7원의 헤테로시클로알킬, 치환 또는 비치환된(C3-C30)시클로알킬, 치환 또는 비치환된 방향족고리가 하나이상 융합된 (C3-C30)시클로알킬, 치환 또는 비치환된 아다만틸, 치환 또는 비치환된(C7-C30)바이시클로알킬, 시아노, NR11R12, BR13R14, PR15R16, P(=O)R17R18[R11 내지 R18은 서로 독립적으로 치환 또는 비치환된(C1-C30)알킬, 치환 또는 비치환된(C6-C30)아릴 또는 치환 또는 비치환된(C3-C30)헤테로아릴이다.], 치환 또는 비치환된 트리(C1-C30)알킬실릴, 치환 또는 비치환된 디(C1-C30)알킬(C6-C30)아릴실릴, 치환 또는 비치환된 트리(C6-C30)아릴실릴, 치환 또는 비치환된 (C6-C30)아르(C1-C30)알킬, 치환 또는 비치환된(C1-C30)알킬옥시, 치환 또는 비치환된(C1-C30)알킬티오, 치환 또는 비치환된(C6-C30)아릴옥시, 치환 또는 비치환된(C6-C30)아릴티오, 치환 또는 비치환된(C1-C30)알콕시카보닐, 치환 또는 비치환된(C1-C30)알킬카보닐, 치환 또는 비치환된(C6-C30)아릴카보닐, 치환 또는 비치환된(C2-C30)알케닐, 치환 또는 비치환된(C2-C30)알키닐, 치환 또는 비치환된(C6-C30)아릴옥시카보닐, 치환 또는 비치환된(C1-C30)알콕시카보닐옥시, 치환 또는 비치환된(C1-C30)알킬카보닐옥시, 치환 또는 비치환된(C6-C30)아릴카보닐옥시, 치환 또는 비치환된(C6-C30)아릴옥시카보닐옥시, 카르복실, 나이트로, 하이드록시이거나 인접 탄소와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 융합고리를 형성할 수 있고;R 151 to R 154 are each independently Hydrogen, deuterium, halogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C3-C30) heteroaryl, substituted or unsubstituted 5 member 5-7 membered heterocycloalkyl, substituted or unsubstituted aromatic ring 5-7 membered fused heterocycloalkyl, substituted or unsubstituted (C3-C30) cycloalkyl, substituted or unsubstituted aromatic ring One or more fused (C3-C30) cycloalkyl, substituted or unsubstituted adamantyl, substituted or unsubstituted (C7-C30) bicycloalkyl, cyano, NR 11 R 12 , BR 13 R 14 , PR 15 R 16 , P (═O) R 17 R 18 [R 11 to R 18 are independently of each other substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl or substituted or unsubstituted. Substituted (C3-C30) heteroaryl], substituted or unsubstituted tri (C1-C30) alkylsilyl, substituted or unsubstituted di (C1-C30) alkyl (C6-C30) arylsilyl, substituted or unsubstituted Tri (C6-C30) arylsilyl, substituted or unsubstituted (C6-C30) ar (C1-C30) alkyl, substituted or unsubstituted (C1-C30) alkyloxy, substituted or unsubstituted (C1-C30 Alkylthio, substituted or unsubstituted (C6-C30) aryloxy, substituted or unsubstituted (C6-C30) arylthio, substituted or unsubstituted (C1-C30) alkoxycarbonyl, substituted or unsubstituted ( C1-C30) alkylcarbonyl, substituted or unsubstituted (C6-C30) arylcarbonyl, substituted or unsubstituted (C2-C30) alkenyl, substituted or unsubstituted (C2-C30) alkynyl, substituted or Unsubstituted (C6-C30) aryloxycarbonyl, substituted or unsubstituted (C1-C30) alkoxycarbonyloxy, substituted or unsubstituted (C1-C30) alkylcarbonyloxy, substituted or unsubstituted (C6 -C30) arylcarbonyloxy, substituted or unsubstituted (C6-C30) aryloxycarbonyloxy, carboxyl, nitro, (C3-C30) alkyl with or without hydroxy or fused ring with adjacent carbon Opened with lene or (C3-C30) alkenylene It is may form a fused ring;
상기 헤테로시클로알킬 및 헤테로아릴은 B, N, O, S, P(=O), Si 및 P로부터 선택된 하나 이상의 헤테로원자를 포함한다.] Wherein said heterocycloalkyl and heteroaryl include one or more heteroatoms selected from B, N, O, S, P (= 0), Si and P.]
상기 발광층의 의미는 발광이 이루어지는 층으로서 단일 층일 수 있으며, 또한 2개 이상의 층이 적층된 복수의 층일 수 있다. 본 발명의 구성에서의 호스트-도판트를 혼합하여 사용하는 경우, 본 발명의 발광 호스트에 의한 발광 효율의 현저한 개선을 확인할 수 있었다. 이는 0.5 내지 10중량%의 도핑 농도로 구성할 수 있는데, 기존의 다른 호스트 재료에 비하여 정공, 전자에 대한 전도성이 매우 뛰어나며, 물질 안정성을 매우 우수하여 발광효율 뿐만 아니라, 수명도 현저히 개선시키는 특성을 보여 주고 있다.The light emitting layer may be a single layer as a light emitting layer, or may be a plurality of layers in which two or more layers are stacked. In the case of using a mixture of the host and dopants in the configuration of the present invention, a significant improvement in the luminous efficiency by the light emitting host of the present invention was confirmed. It can be composed of a doping concentration of 0.5 to 10% by weight, and has excellent conductivity for holes and electrons compared to other host materials, and has excellent material stability, which significantly improves luminous efficiency and lifetime. Is showing.
상기 화학식 3의 도판트 화합물은 출원번호 제10-2009-0023442호에 기재된 화합물로 예시될 수 있으며, 보다 바람직하게는 하기 구조에서 선택될 수 있으나, 이에 한정되는 것은 아니다. The dopant compound of Chemical Formula 3 may be exemplified by the compound described in Application No. 10-2009-0023442, and more preferably may be selected from the following structures, but is not limited thereto.
본 발명의 유기 전계 발광 소자는 상기 화학식 1의 유기 발광 화합물을 포함하고, 동시에 아릴아민계 화합물 또는 스티릴아릴아민계 화합물로 이루어진 군으로부터 선택된 하나 이상의 화합물을 포함할 수 있으며, 상기 아릴아민계 화합물 또 는 스티릴아릴아민계 화합물은 출원번호 제10-2008-0123276호, 제10-2008-0107606호 또는 제10-2008-0118428호에 예시되어 있으나, 이에 한정되지는 않는다.The organic electroluminescent device of the present invention may include one or more compounds selected from the group consisting of an organic light emitting compound of Formula 1 and an arylamine compound or a styrylarylamine compound, and the arylamine compound Or styrylarylamine-based compound is exemplified in, but not limited to, the application number 10-2008-0123276, 10-2008-0107606 or 10-2008-0118428.
또한, 본 발명의 유기 전계 발광 소자에 있어서, 유기물층에 상기 화학식 1의 유기 발광 화합물 이외에 1족, 2족, 4주기, 5주기 전이금속, 란탄계열금속 및 d-전이원소의 유기금속으로 이루어진 군으로부터 선택되는 하나 이상의 금속 또는 착체화합물을 더 포함할 수도 있고, 상기 유기물층은 발광층 및 전하생성층을 동시에 포함할 수 있다.In addition, in the organic electroluminescent device of the present invention, in the organic material layer, in addition to the organic light emitting compound of Chemical Formula 1, a group consisting of Group 1, Group 2, 4 cycle, 5 cycle transition metal, lanthanide series metal and organic metal of d-transition element It may further comprise one or more metals or complex compounds selected from, the organic material layer may include a light emitting layer and a charge generating layer at the same time.
또한, 상기 유기물층에 화학식 1의 유기발광 화합물 이외에 청색, 녹색 또는 적색 발광을 하는 유기 화합물층 하나 이상을 동시에 포함하여 백색 유기 전계 발광 소자를 형성할 수 있으며, 상기 청색, 녹색 또는 적색 발광을 하는 화합물은 출원번호 제10-2008-0123276호, 제10-2008-0107606호 또는 제10-2008-0118428호에 예시되어 있으나, 이에 한정되지는 않는다.In addition, the organic material layer may simultaneously include one or more organic compound layers emitting blue, green, or red light in addition to the organic light emitting compound of Formula 1 to form a white organic electroluminescent device, and the blue, green, or red light emitting compound It is illustrated in the application No. 10-2008-0123276, 10-2008-0107606 or 10-2008-0118428, but is not limited thereto.
본 발명의 유기 전계 발광 소자에 있어서, 한 쌍의 전극의 적어도 한쪽의 내측표면에, 칼코제나이드(chalcogenide)층, 할로겐화 금속층 및 금속 산화물층으로부터 선택되는 일층(이하, 이들을 "표면층"이라고 지칭함) 이상을 배치하는 것이 바람직하다. 구체적으로는, 발광 매체층 측의 양극 표면에 규소 및 알루미늄의 금속의 칼코제나이드(산화물을 포함한다)층을, 또한 발광매체층 측의 음극 표면에 할로겐화 금속층 또는 금속 산화물층을 배치하는 것이 바람직하다. 이것에 의해, 구동의 안정화를 얻을 수 있다.In the organic electroluminescent device of the present invention, at least one inner surface of a pair of electrodes is selected from a chalcogenide layer, a halogenated metal layer, and a metal oxide layer (hereinafter, these are referred to as "surface layers"). It is preferable to arrange | position the above. Specifically, it is preferable to dispose a chalcogenide (containing oxide) layer of a metal of silicon and aluminum on the anode surface of the light emitting medium layer side and a metal halide or metal oxide layer on the cathode surface of the light emitting medium layer side. Do. As a result, drive stabilization can be obtained.
상기 칼코제나이드로서는 예컨대 SiOx(1≤X≤2), AlOX(1≤X≤1.5), SiON, SiAlON 등을 바람직하게 들 수 있으며, 할로겐화 금속으로서는 예컨대 LiF, MgF2, CaF2, 불화 희토류 금속 등을 바람직하게 들 수 있으며, 금속 산화물로서는 예컨대 Cs2O, Li2O, MgO, SrO, BaO, CaO 등을 바람직하게 들 수 있다.Examples of the chalcogenide include SiO x (1 ≦ X ≦ 2 ), AlO X (1 ≦ X ≦ 1.5), SiON, SiAlON, and the like, and examples of the metal halide include LiF, MgF 2 , CaF 2 , and fluoride. Rare earth metals and the like are preferable. Examples of the metal oxides include Cs 2 O, Li 2 O, MgO, SrO, BaO, CaO and the like.
또한, 본 발명의 유기 전계 발광 소자에 있어서, 이렇게 제작된 한 쌍의 전극의 적어도 한쪽의 표면에 전자 전달 화합물과 환원성 도판트의 혼합 영역 또는 정공 전달 화합물과 산화성 도판트의 혼합 영역을 배치하는 것도 바람직하다. 이러한 방식으로, 전자 전달 화합물이 음이온으로 환원되므로 혼합 영역으로부터 발광 매체에 전자를 주입 및 전달하기 용이해진다. 또한, 정공 전달 화합물은 산화되어 양이온으로 되므로 혼합 영역으로부터 발광 매체에 정공을 주입 및 전달하기 용이해진다. 바람직한 산화성 도판트로서는 각종 루이스산 및 억셉터(acceptor) 화합물을 들 수 있다. 바람직한 환원성 도판트로서는 알칼리 금속, 알칼리 금속 화합물, 알칼리 토류 금속, 희토류 금속 및 이들의 혼합물을 들 수 있다.Further, in the organic electroluminescent device of the present invention, disposing a mixed region of an electron transfer compound and a reducing dopant or a mixed region of a hole transfer compound and an oxidative dopant on at least one surface of the pair of electrodes thus produced desirable. In this way, the electron transfer compound is reduced to an anion, thereby facilitating injection and transfer of electrons from the mixed region into the light emitting medium. In addition, since the hole transport compound is oxidized to become a cation, it is easy to inject and transfer holes from the mixed region to the light emitting medium. Preferred oxidative dopants include various Lewis acids and acceptor compounds. Preferred reducing dopants include alkali metals, alkali metal compounds, alkaline earth metals, rare earth metals and mixtures thereof.
또한 환원성 도판트층을 전하생성층으로 사용하여 두 개 이상의 발광층을 가진 백색 유기 전계 발광 소자를 제작할 수 있다.In addition, the reducing dopant layer By using the charge generating layer, a white organic electroluminescent device having two or more light emitting layers may be manufactured.
본 발명에 따른 유기 발광 화합물은 발광 효율이 좋고 재료의 수명특성이 뛰어나 소자의 구동수명이 매우 우수한 OLED 소자를 제조할 수 있는 장점이 있다.The organic light emitting compound according to the present invention has the advantage of being able to manufacture an OLED device having a good luminous efficiency and excellent life characteristics of the material and excellent driving life of the device.
이하에서, 본 발명의 상세한 이해를 위하여 본 발명의 대표 화합물을 들어 본 발명에 따른 유기 발광 화합물, 이의 제조방법 및 소자의 발광특성을 설명하나, 이는 단지 그 실시 양태를 예시하기 위한 것일 뿐, 본 발명의 범위를 한정하는 것은 아니다.Hereinafter, the organic light emitting compound according to the present invention, a method for preparing the same and a light emitting property of the device are described for the detailed understanding of the present invention, but the present invention is only intended to illustrate the embodiments of the present invention. It does not limit the scope of the invention.
[제조예 1] 화합물 1의 제조Preparation Example 1 Preparation of Compound 1
화합물 compound AA 의 제조Manufacture
메틸 2-브로모벤조에이트(methyl 2-bromobenzoate) 40 g(152.6 mmol)과 나프탈렌-1-일보로닉 산(naphthalen-1-ylboronic acid) 31.5 g(183.2 mmol)과 테트라키스(트리페닐포스핀)팔라듐[Pd(PPh3)4] 8.8 g(7.62 mmol)를 2구 플라스크에 첨가한다. 톨루엔 1 L를 첨가하면서 교반을 시키고 2 M의 포테슘 카보네이트(Potassuim carbonate) 228 mL(458 mmol)와 에탄올 228 mL를 첨가한다. 100 ℃에서 5시간동안 환류시킨다. 반응이 종결되면 실온으로 냉각시킨 후 증류수와 에틸아세테이트로 추 출한다. 유기층을 MgSO4로 건조 시킨 후 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 A 35 g(87 %)를 분리한다.40 g (152.6 mmol) of methyl 2-bromobenzoate, 31.5 g (183.2 mmol) of naphthalen-1-ylboronic acid and tetrakis (triphenylphosphine) 8.8 g (7.62 mmol) of palladium [Pd (PPh 3 ) 4 ] are added to a two neck flask. Stir with the addition of 1 L of toluene and add 228 mL (458 mmol) of 2 M Potassium carbonate and 228 mL of ethanol. Reflux at 100 ° C. for 5 hours. After the reaction is completed, the reaction mixture is cooled to room temperature and extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and then the solvent was removed using a rotary evaporator. Compound A 35 g (87%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound BB 의 제조Manufacture
화합물 A 24 g(91.49 mmol)를 1구 플라스크에 넣고 진공 분위기로 만들고 아르곤으로 채운다. 테트라하이드로퓨란 1 L를 넣고 -75 ℃에서 10분간 교반한다. MeLi(1.6 M in hexane) 257 mL(0.41 mol)를 첨가하고 -75 ℃에서 10분간 교반하고 상온에서 3시간 교반한다. 반응이 종결되면 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 후 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 B 20 g(83 %)를 분리한다.24 g (91.49 mmol) of Compound A are placed in a 1-neck flask, vacuumed and filled with argon. Add 1 L of tetrahydrofuran and stir at −75 ° C. for 10 minutes. Add 257 mL (0.41 mol) of MeLi (1.6 M in hexane), stir at −75 ° C. for 10 minutes, and stir at room temperature for 3 hours. After the reaction is completed, the mixture is extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and then the solvent was removed using a rotary evaporator. Compound B 20 g (83%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound CC 의 제조Manufacture
화합물 B 20 g(76.23 mmol)을 1구 플라스크에 넣고 AcOH 300 mL를 넣어 0 ℃에서 10분간 교반한다. H3PO4 400 mL를 첨가하고 상온에서 1시간 동안 교반한다. 반응이 종결되면 NaOH로 중화시키고 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 C 13.5 g(72 %)를 분리한다.20 g (76.23 mmol) of Compound B were added to a 1-neck flask, and 300 mL of AcOH was added thereto and stirred at 0 ° C. for 10 minutes. Add 400 mL of H 3 PO 4 and stir at room temperature for 1 hour. Upon completion of the reaction, neutralize with NaOH and extract with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 and the solvent was removed using a rotary evaporator. Compound C 13.5 g (72%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound DD 의 제조Manufacture
화합물 C 13.5 g(55.25 mmol)를 1구 플라스크에 넣고 진공 분위기로 만들고 아르곤으로 채운다. 테트라하이드로퓨란 500 mL를 넣고 0 ℃에서 10분간 교반한다. NBS 19.6 g(0.11 mol)를 첨가하고 상온에서 하루 동안 교반한다. 반응이 종결되면 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 D 13 g(73 %)를 분리한다.13.5 g (55.25 mmol) of compound C are placed in a 1-neck flask, vacuumed and filled with argon. Add 500 mL of tetrahydrofuran and stir at 0 ° C. for 10 minutes. Add 19.6 g (0.11 mol) of NBS and stir at room temperature for one day. After the reaction is completed, the mixture is extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 and the solvent was removed using a rotary evaporator. Compound D 13 g (73%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound EE 의 제조Manufacture
화합물 D 13 g(42.21 mmol)를 1구 플라스크에 넣고 진공 분위기로 만들고 아르곤으로 채운다. 테트라하이드로퓨란 500 mL를 넣고 -78 ℃에서 10분간 교반한다. n-BuLi(2.5 M in hexane) 24.1 mL(60.32 mmol)를 적가하고 1시간 30분간 -78 ℃에서 교반한다. 트라이메틸보레이트 6.85 mL(60.32 mol)를 -78 ℃에서 첨가한다. 30분간 -78 ℃에서 교반한 후 실온에서 4시간 동안 교반한다. 반응이 종결되면 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 E 8 g(69 %)를 분리한다.13 g (42.21 mmol) of compound D are placed in a 1-neck flask, vacuumed and filled with argon. Add 500 mL of tetrahydrofuran and stir at -78 ° C for 10 minutes. 24.1 mL (60.32 mmol) of n-BuLi (2.5 M in hexane) is added dropwise and stirred at −78 ° C. for 1 hour 30 minutes. 6.85 mL (60.32 mol) of trimethylborate is added at -78 ° C. Stir for 30 minutes at -78 ° C and then for 4 hours at room temperature. After the reaction is completed, the mixture is extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 and the solvent was removed using a rotary evaporator. Compound E 8 g (69%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound FF 의 제조Manufacture
2-클로로안트라센-9,10(4aH,9aH)-다이온 50 g(0.2 mol)과 페닐보로닉 산 37.6 g(0.3 mol)와 테트라키스(트리페닐포스핀)팔라듐 9.5 g(8.24 mmol)를 2구 플라스크에 첨가한다. 톨루엔을 첨가하면서 교반을 시키고 2 M의 포테슘 카보네이트 500 mL(1.0 mol)과 에탄올 500 mL를 첨가한다. 120 ℃에서 5시간동안 환류시킨다. 반응이 종결되면 실온으로 냉각시킨 후 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 F 56 g(95 %)를 분리한다.50 g (0.2 mol) of 2-chloroanthracene-9,10 (4aH, 9aH) -dione, 37.6 g (0.3 mol) of phenylboronic acid, and 9.5 g (8.24 mmol) of tetrakis (triphenylphosphine) palladium Is added to a two-necked flask. Stir with the addition of toluene and add 500 mL (1.0 mol) of 2 M potassium carbonate and 500 mL of ethanol. Reflux at 120 ° C. for 5 hours. After the reaction is completed, the reaction mixture is cooled to room temperature and extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and the solvent was removed using a rotary evaporator. Compound F 56 g (95%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound GG 의 제조Manufacture
화합물 F 50 g(0.17 mol)를 1구 플라스크에 넣고 AcOH 1 L를 넣어 10분 간 교반한다. H3PO2 380 g(5.76 mol)와 HI 781 g(6.11 mol)를 첨가하고 150 ℃에서 하루 동안 교반한다. 반응이 종결되면 NaOH 용액과 HCl로 중화시키고 생성된 고체를 필터 한다. 필터 한 고체를 에틸아세테이트에 넣고 100 ℃ 환류 재결정하고 화합물 G 40 g(90 %)를 분리한다.50 g (0.17 mol) of Compound F was added to a 1-neck flask, and 1 L of AcOH was added thereto and stirred for 10 minutes. 380 g (5.76 mol) of H 3 PO 2 and 781 g (6.11 mol) of HI are added and stirred at 150 ° C. for one day. Upon completion of the reaction, neutralize with NaOH solution and HCl and filter the solid produced. The filtered solid was put in ethyl acetate, recrystallized at reflux at 100 ° C., and 40 g (90%) of Compound G was separated.
화합물 compound HH 의 제조Manufacture
화합물 G 40 g(0.15 mol)를 1구 플라스크에 넣고 진공 분위기로 만들고 아르곤으로 채운다. 다이클로로메탄 500 mL를 넣고 0 ℃에서 10분간 교반한다. NBS 28 g(0.15 mol)를 첨가하고 상온에서 하루 동안 교반한다. 반응이 종결되면 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 H 48 g(92 %)를 분리한다.40 g (0.15 mol) of compound G are placed in a 1-neck flask, vacuumed and filled with argon. Add 500 mL of dichloromethane and stir at 0 ° C. for 10 minutes. Add 28 g (0.15 mol) of NBS and stir at room temperature for one day. After the reaction is completed, the mixture is extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and the solvent was removed using a rotary evaporator. Compound H 48 g (92%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound II 의 제조Manufacture
화합물 H 48 g(0.14 mol)과 화합물 E 43 g(0.15 mol)와 테트라키스(트리페닐포스핀)팔라듐 8.3 g(7.2 mmol)를 2구 플라스크에 첨가한다. 톨루엔을 첨가하면서 교반을 시키고 2 M의 포테슘 카보네이트 720 mL(1.4 mol)과 에탄올 720 mL를 첨가한다. 120 ℃에서 5시간동안 환류시킨다. 반응이 종결되면 실온으로 냉각시킨 후 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 I 60 g(84 %)를 분리한다.Compound H 48 g (0.14 mol) and compound E 43 g (0.15 mol) and tetrakis (triphenylphosphine) palladium, 8.3 g (7.2 mmol) 2 is added to the flask. Stir with the addition of toluene and add 720 mL (1.4 mol) of 2 M potassium carbonate and 720 mL of ethanol. Reflux at 120 ° C. for 5 hours. After the reaction is completed, the reaction mixture is cooled to room temperature and extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 and the solvent was removed using a rotary evaporator, and 60 g (84%) of Compound I was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound JJ 의 제조Manufacture
화합물 I 60 g(0.12 mol)를 1구 플라스크에 넣고 진공 분위기로 만들고 아르곤으로 채운다. 다이클로로메탄 600 mL를 넣고 0 ℃에서 10분간 교반한다. NBS 38.7 g(0.21 mol)를 첨가하고 상온에서 하루 동안 교반한다. 반응이 종결되면 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 J 60 g(86 %)를 분리한다.60 g (0.12 mol) of compound I are placed in a 1-neck flask, vacuumed and filled with argon. Add 600 mL of dichloromethane and stir at 0 ° C. for 10 minutes. Add 38.7 g (0.21 mol) of NBS and stir at room temperature for one day. After the reaction is completed, the mixture is extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and the solvent was removed using a rotary evaporator. Compound J 60 g (86%) was isolated by column chromatography using hexane and ethyl acetate as developing solvents.
화합물 compound 1One 의 제조Manufacture
화합물 J 10 g(17.37 mmol)과 페닐보로닉 산 3.1 g(26.06 mmol)과 테트라키스(트리페닐포스핀)팔라듐 1.0 g(0.86 mmol)를 2구 플라스크에 첨가한다. 톨루엔을 첨가하면서 교반을 시키고 2 M의 포테슘 카보네이트 86 mL(0.17 mol)과 에탄올 86 mL를 첨가다. 120 ℃에서 5시간동안 환류시킨다. 반응이 종결되면 실온으로 냉각시 킨 후 증류수와 에틸아세테이트로 추출한다. 유기층을 MgSO4로 건조 시킨 다음 회전 증발기로 용매를 제거한 후 헥산과 에틸아세테이트를 전개 용매로 하여 컬럼 크로마토그래피 작업으로 화합물 1 8.5 g(85 %)를 분리한다.10 g (17.37 mmol) of compound J, 3.1 g (26.06 mmol) of phenylboronic acid and 1.0 g (0.86 mmol) of tetrakis (triphenylphosphine) palladium are added to a two neck flask. Stir with the addition of toluene and add 86 mL (0.17 mol) of 2 M potassium carbonate and 86 mL of ethanol. Reflux at 120 ° C. for 5 hours. After the reaction is completed, the mixture is cooled to room temperature and extracted with distilled water and ethyl acetate. The organic layer was dried over MgSO 4 , and the solvent was removed using a rotary evaporator, and then 8.5 g (85%) of Compound 1 was separated by column chromatography using hexane and ethyl acetate as developing solvents.
상기 제조예 1의 방법을 이용하여 유기 발광 화합물 1 내지 화합물 120을 제조하였으며, 표 1에 제조된 유기 발광 화합물들의 1H NMR 및 MS/FAB를 나타내었다.The organic light emitting compounds 1 to 120 were prepared using the method of Preparation Example 1, and the 1 H NMR and the MS / FAB of the organic light emitting compounds prepared in Table 1 are shown.
[표 1]TABLE 1
[실시예 1] 본 발명에 따른 유기 발광 화합물을 이용한 OLED 소자 제작Example 1 Fabrication of an OLED Device Using an Organic Light Emitting Compound According to the Present Invention
본 발명의 발광 재료를 이용한 구조의 OLED 소자를 제작하였다.An OLED device having a structure using the light emitting material of the present invention was produced.
우선, OLED용 글래스(삼성-코닝사 제조)로부터 얻어진 투명전극 ITO 박막(15 Ω/□)을, 트리클로로에틸렌, 아세톤, 에탄올, 증류수를 순차적으로 사용하여 초음파 세척을 실시한 후, 이소프로판올에 넣어 보관한 후 사용하였다.First, a transparent electrode ITO thin film (15 Ω / □) obtained from an OLED glass (manufactured by Samsung Corning Corporation) was subjected to ultrasonic cleaning using trichloroethylene, acetone, ethanol and distilled water sequentially, and then stored in isopropanol. It was used after.
다음으로, 진공 증착 장비의 기판 폴더에 ITO 기판을 설치하고, 진공 증착 장비 내의 셀에 하기 구조의 4,4',4"-tris(N,N-(2-naphthyl)-phenylamino)triphenylamine (2-TNATA)을 넣고, 챔버 내의 진공도가 10-6 torr에 도달할 때까지 배기시킨 후, 셀에 전류를 인가하여 2-TNATA를 증발시켜 ITO 기판 상에 60 nm 두께의 정공주입층을 증착하였다.Next, an ITO substrate is installed in the substrate folder of the vacuum deposition apparatus, and 4,4 ', 4 "-tris (N, N- (2-naphthyl) -phenylamino) triphenylamine (2) having the structure -TNATA), evacuated until the vacuum in the chamber reached 10 -6 torr, and then applied a current to the cell to evaporate 2-TNATA to deposit a 60 nm thick hole injection layer on the ITO substrate.
이어서, 진공 증착 장비 내의 다른 셀에 하기구조 N,N'-bis(α-naphthyl)-N,N'-diphenyl-4,4'-diamine (NPB)을 넣고, 셀에 전류를 인가하여 NPB를 증발시켜 정공주입층 위에 20 nm 두께의 정공전달층을 증착하였다.The NPB -diphenyl-4,4'-diamine into the (NPB), by applying a current to the cell - Then, to another cell of the vacuum vapor-deposit device structure, N, N 'N, N -bis (α-naphthyl)' A 20 nm thick hole transport layer was deposited on the hole injection layer by evaporation.
정공주입층, 정공전달층을 형성시킨 후, 그 위에 발광층을 다음과 같이 증착시켰다. 진공 증착 장비 내의 한쪽 셀에 호스트로서 본 발명에 따른 화합물 1 를 넣고, 또 다른 셀에는 도판트로서 화합물 A 을 각각 넣은 후, 두 물질을 다른 속도로 증발시켜 호스트를 기준으로 2 내지 5 중량%로 도핑함으로써 상기 정공 전달층 위에 30 nm 두께의 발광층을 증착하였다. After the hole injection layer and the hole transport layer were formed, the light emitting layer was deposited thereon as follows. Compound 1 according to the present invention is added as a host to one cell in a vacuum deposition apparatus, and Compound A is added as a dopant to another cell, and then the two materials are evaporated at different rates to 2 to 5% by weight based on the host. A doped 30 nm thick light emitting layer was deposited on the hole transport layer by doping.
이어서 전자전달층으로써 하기 구조의 tris(8-hydroxyquinoline)-aluminum(III) (Alq)를 20 nm 두께로 증착한 다음, 전자주입층으로 하기 구조의 화합물 lithium quinolate (Liq)를 1 내지 2 nm 두께로 증착한 후, 다른 진공 증착 장비를 이용하여 Al 음극을 150 nm의 두께로 증착하여 OLED를 제작하였다. Subsequently, tris (8-hydroxyquinoline) -aluminum (III) (Alq) having the following structure was deposited as an electron transport layer to a thickness of 20 nm, and then the compound lithium quinolate (Liq) having the following structure as the electron injection layer was 1 to 2 nm thick. After deposition, the Al cathode was deposited to a thickness of 150 nm using another vacuum deposition equipment to produce an OLED.
재료 별로 각 화합물은 10-6 torr 하에서 진공 승화 정제하여 OLED 발광재료로 사용하였다.Each compound was vacuum sublimated and purified under 10 -6 torr to be used as an OLED light emitting material.
[비교예 1] 종래의 발광 재료를 이용한 OLED 소자 제작Comparative Example 1 Fabrication of OLED Device Using Conventional Light-Emitting Material
상기 실시예 1과 동일한 방법으로 정공주입층, 정공전달층을 형성시킨 후, 상기 진공 증착 장비 내의 다른 셀에 발광 호스트 재료인 tris(8-hydroxyquinoline)-aluminum(III) (Alq)를 넣고, 또 다른 셀에는 하기 구조의 Coumarin 545T(C545T)를 각각 넣은 후, 두 물질을 다른 속도로 증발시켜 도핑함으로써 상기 정공 전달층 위에 30 nm 두께의 발광층을 증착하였다. 이 때의 도핑 농도는 Alq 기준으로 1 내지 3 중량%가 바람직하다.After the hole injection layer and the hole transport layer were formed in the same manner as in Example 1, tris (8-hydroxyquinoline) -aluminum (III) (Alq), which is a light emitting host material, was placed in another cell in the vacuum deposition apparatus. In another cell, Coumarin 545T (C545T) having the following structure was put in each, and the light emitting layer having a thickness of 30 nm was deposited on the hole transport layer by evaporating and doping the two materials at different rates. The doping concentration at this time is preferably 1 to 3% by weight based on Alq.
이어서 실시예 1과 동일한 방법으로 전자전달층과 전자주입층을 증착한 후, 다른 진공 증착 장비를 이용하여 Al 음극을 150 nm의 두께로 증착하여 OLED를 제작하였다.Subsequently, an electron transport layer and an electron injection layer were deposited in the same manner as in Example 1, and then another OLED was manufactured by depositing an Al cathode to a thickness of 150 nm using another vacuum deposition equipment.
[비교예 2] 종래의 발광 재료를 이용한 OLED 소자 제작Comparative Example 2 Fabrication of OLED Device Using Conventional Light-Emitting Material
상기 실시예 1과 동일한 방법으로 정공주입층, 정공전달층을 형성시킨 후, 상기 진공 증착 장비 내의 다른 셀에 발광 호스트 재료인 dinaphthylanthracene(DNA)를 넣고, 또 다른 셀에는 화합물 A를 각각 넣은 후, 두 물질을 다른 속도로 증발시켜 호스트를 기준으로 2 내지 5 중량%로 도핑함으로써 상기 정공 전달층 위에 30 nm 두께의 발광층을 증착하였다.After the hole injection layer and the hole transport layer were formed in the same manner as in Example 1, dinaphthylanthracene (DNA), which is a light emitting host material, was placed in another cell in the vacuum deposition apparatus, and Compound A was put in another cell, respectively. A 30 nm thick light emitting layer was deposited on the hole transport layer by evaporating the two materials at different rates and doping at 2-5 wt% based on the host.
이어서 실시예 1과 동일한 방법으로 전자전달층과 전자주입층을 증착한 후, 다른 진공 증착 장비를 이용하여 Al 음극을 150 nm의 두께로 증착하여 OLED를 제작하였다.Subsequently, an electron transport layer and an electron injection layer were deposited in the same manner as in Example 1, and then another OLED was manufactured by depositing an Al cathode to a thickness of 150 nm using another vacuum deposition equipment.
상기 실시예 1 및 비교예 1 또는 2 에서 제조된 본 발명에 따른 유기 발광 화합물과 종래의 발광 화합물을 각각 함유하는 OLED 소자의 발광 효율을 각각 5,000 cd/m2에서 측정하여 하기 표 2에 나타내었다. To the above Example 1 and Comparative Example 1, or the luminous efficiency of the OLED device containing each of the organic light-emitting compound and a conventional electroluminescent compound according to the present invention produced in the second are measured at 5,000 cd / m 2 are shown in Table 2 .
[표 2]TABLE 2
상기 표 2에 나타난 바와 같이, 본 발명의 재료를 녹색 발광 소자에 적용한 결과, 본 발명의 유기화합물이 비교예 1 및 2의 대비 동등이상의 색순도를 유지하면서 구동전압도 낮고 발광효율이 개선되었음을 확인할 수 있었다. As shown in Table 2, as a result of applying the material of the present invention to the green light emitting device, it can be seen that the organic compound of the present invention has a low driving voltage and improved luminous efficiency while maintaining color purity equal to or higher than that of Comparative Examples 1 and 2. there was.
Claims (8)
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020090027846A KR20100109293A (en) | 2009-03-31 | 2009-03-31 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| JP2012503312A JP5781499B2 (en) | 2009-03-31 | 2010-03-24 | Novel organic electroluminescent compound and organic electroluminescent device using the same |
| PCT/KR2010/001813 WO2010114253A2 (en) | 2009-03-31 | 2010-03-24 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| CN2010800250539A CN102449110A (en) | 2009-03-31 | 2010-03-24 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| TW099109823A TW201105776A (en) | 2009-03-31 | 2010-03-31 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020090027846A KR20100109293A (en) | 2009-03-31 | 2009-03-31 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20100109293A true KR20100109293A (en) | 2010-10-08 |
Family
ID=42828814
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020090027846A Abandoned KR20100109293A (en) | 2009-03-31 | 2009-03-31 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Country Status (5)
| Country | Link |
|---|---|
| JP (1) | JP5781499B2 (en) |
| KR (1) | KR20100109293A (en) |
| CN (1) | CN102449110A (en) |
| TW (1) | TW201105776A (en) |
| WO (1) | WO2010114253A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20130098227A (en) * | 2012-02-27 | 2013-09-04 | 주식회사 엘지화학 | Organic light emitting diode |
| KR20210073587A (en) * | 2019-02-20 | 2021-06-18 | 주식회사 엘지화학 | Anthracene derivatives and organic electronic devices using same |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5352304B2 (en) * | 2008-04-02 | 2013-11-27 | 株式会社半導体エネルギー研究所 | Anthracene derivative, light emitting material, light emitting element material, coating composition, light emitting element, and light emitting device |
| EP2112212B1 (en) | 2008-04-24 | 2013-05-29 | Semiconductor Energy Laboratory Co., Ltd. | Anthracene derivative, light-emitting material, material for light-emitting element, composition for coating light-emitting element, light-emitting device, and electronic device |
| KR101427605B1 (en) * | 2009-03-31 | 2014-08-07 | 롬엔드하스전자재료코리아유한회사 | Novel organic light emitting compounds and organic electroluminescent devices employing the same |
| KR101754445B1 (en) | 2009-05-29 | 2017-07-05 | 이데미쓰 고산 가부시키가이샤 | Anthracene derivative and organic electroluminescent element using the same |
| JP5699524B2 (en) * | 2010-10-21 | 2015-04-15 | コニカミノルタ株式会社 | Organic photoelectric conversion element and solar cell |
| US9312500B2 (en) | 2012-08-31 | 2016-04-12 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device |
| JP2015233024A (en) * | 2012-09-03 | 2015-12-24 | 出光興産株式会社 | Organic electroluminescent device |
| US9748492B2 (en) | 2012-11-02 | 2017-08-29 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| KR20150010016A (en) * | 2013-07-17 | 2015-01-28 | 롬엔드하스전자재료코리아유한회사 | Organic electroluminescent device |
| JP6266632B2 (en) | 2013-09-06 | 2018-01-24 | 出光興産株式会社 | Anthracene derivative and organic electroluminescence device using the same |
| CN106033793B (en) * | 2015-03-09 | 2018-11-13 | 广东阿格蕾雅光电材料有限公司 | Organic electroluminescence device |
| CN106033798B (en) * | 2015-03-09 | 2017-11-14 | 广东阿格蕾雅光电材料有限公司 | Organic electroluminescence device |
| CN106032350B (en) * | 2015-03-09 | 2019-03-01 | 广东阿格蕾雅光电材料有限公司 | Organic electronic material |
| CN106033801B (en) * | 2015-03-09 | 2018-04-06 | 广东阿格蕾雅光电材料有限公司 | Organic electroluminescence device |
| CN106831448B (en) * | 2016-12-16 | 2019-04-02 | 烟台九目化学制品有限公司 | A kind of novel aromatic amine luminescent material and its application |
| CN107954942B (en) * | 2017-12-08 | 2019-04-09 | 北京鼎材科技有限公司 | Benzopyrazine Substituted Anthracene Derivatives and Organic Electroluminescent Devices |
| KR102601117B1 (en) * | 2018-08-17 | 2023-11-09 | 주식회사 엘지화학 | Novel compound and organic light emitting device comprising the same |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6929870B2 (en) * | 2000-08-10 | 2005-08-16 | Mitsui Chemicals, Inc. | Hydrocarbon compounds, materials for organic electroluminescent elements and organic electroluminescent elements |
| JP4080213B2 (en) * | 2002-02-01 | 2008-04-23 | 三井化学株式会社 | Organic electroluminescence device |
| TW200304371A (en) * | 2002-02-22 | 2003-10-01 | Akzo Nobel Nv | Substituted 10-ary1-11H-benzo [b] fluorenes and 7-ary1-5, 6-dihydro-benz [a] anthracenes for selective effects on estrogen receptors |
| US6849348B2 (en) * | 2002-12-31 | 2005-02-01 | Eastman Kodak Company | Complex fluorene-containing compounds |
| US7887931B2 (en) * | 2003-10-24 | 2011-02-15 | Global Oled Technology Llc | Electroluminescent device with anthracene derivative host |
| KR100788254B1 (en) * | 2005-08-16 | 2007-12-27 | (주)그라쎌 | Green electroluminescent compounds and organic electroluminescent device using the same |
| DE102006031990A1 (en) * | 2006-07-11 | 2008-01-17 | Merck Patent Gmbh | New materials for organic electroluminescent devices |
| JP5233228B2 (en) * | 2006-10-05 | 2013-07-10 | Jnc株式会社 | Benzofluorene compound, light emitting layer material and organic electroluminescent device using the compound |
| US8465848B2 (en) * | 2006-12-29 | 2013-06-18 | E I Du Pont De Nemours And Company | Benzofluorenes for luminescent applications |
| KR101178219B1 (en) * | 2008-11-21 | 2012-08-29 | 롬엔드하스전자재료코리아유한회사 | Electroluminescent device using the electroluminescent compounds |
| KR101427605B1 (en) * | 2009-03-31 | 2014-08-07 | 롬엔드하스전자재료코리아유한회사 | Novel organic light emitting compounds and organic electroluminescent devices employing the same |
-
2009
- 2009-03-31 KR KR1020090027846A patent/KR20100109293A/en not_active Abandoned
-
2010
- 2010-03-24 WO PCT/KR2010/001813 patent/WO2010114253A2/en not_active Ceased
- 2010-03-24 JP JP2012503312A patent/JP5781499B2/en not_active Expired - Fee Related
- 2010-03-24 CN CN2010800250539A patent/CN102449110A/en active Pending
- 2010-03-31 TW TW099109823A patent/TW201105776A/en unknown
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20130098227A (en) * | 2012-02-27 | 2013-09-04 | 주식회사 엘지화학 | Organic light emitting diode |
| KR20210073587A (en) * | 2019-02-20 | 2021-06-18 | 주식회사 엘지화학 | Anthracene derivatives and organic electronic devices using same |
| US12167681B2 (en) | 2019-02-20 | 2024-12-10 | Lg Chem, Ltd. | Anthracene derivatives and organic electronic device using same |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102449110A (en) | 2012-05-09 |
| JP5781499B2 (en) | 2015-09-24 |
| WO2010114253A3 (en) | 2010-12-23 |
| JP2012522041A (en) | 2012-09-20 |
| TW201105776A (en) | 2011-02-16 |
| WO2010114253A2 (en) | 2010-10-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101427605B1 (en) | Novel organic light emitting compounds and organic electroluminescent devices employing the same | |
| JP5781499B2 (en) | Novel organic electroluminescent compound and organic electroluminescent device using the same | |
| KR101741415B1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20100109050A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20100118700A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20100137198A (en) | Novel organic light emitting compound and organic electroluminescent device comprising same | |
| KR20100108924A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20100108914A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR101784147B1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20110132721A (en) | Novel organic light emitting compound and organic electroluminescent device employing the same | |
| KR20100108903A (en) | Novel compounds for organic electronic material and organic electronic device using the same | |
| KR101645949B1 (en) | Electroluminescent device using the electroluminescent compounds | |
| KR20110008892A (en) | Novel organic light emitting compound and organic electroluminescent device comprising same | |
| KR20100137188A (en) | Novel organic light emitting compound and organic electroluminescent device employing the same | |
| KR20110116635A (en) | Novel Organic Electronic Materials and Organic Electroluminescent Devices Employing the Same | |
| KR20100048447A (en) | Novel compounds for organic electronic material and organic electronic device using the same | |
| KR20100007780A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20100106014A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| JP2013526014A (en) | Novel organic electroluminescent compound and organic electroluminescent device using the same | |
| JP2013546171A (en) | Novel compounds for organic electronic materials and organic electroluminescent devices using the same | |
| KR20110008784A (en) | Novel organic light emitting compound and organic electroluminescent device comprising same | |
| JP2014501699A (en) | Novel compounds for organic electronic materials and organic electroluminescent devices using the same | |
| KR20110113297A (en) | Novel organic electronic material compound and organic electroluminescent device comprising same | |
| KR20120020901A (en) | Novel compounds for organic electronic material and organic electroluminescent device using the same | |
| KR20120020816A (en) | Novel compounds for organic electronic material and organic electroluminescent device using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20090331 |
|
| PG1501 | Laying open of application | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20120402 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20140328 Comment text: Request for Examination of Application Patent event code: PA02011R01I Patent event date: 20090331 Comment text: Patent Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20150821 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20160227 |
|
| NORF | Unpaid initial registration fee | ||
| PC1904 | Unpaid initial registration fee |