KR20150024444A - 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 - Google Patents
변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 Download PDFInfo
- Publication number
- KR20150024444A KR20150024444A KR1020157003616A KR20157003616A KR20150024444A KR 20150024444 A KR20150024444 A KR 20150024444A KR 1020157003616 A KR1020157003616 A KR 1020157003616A KR 20157003616 A KR20157003616 A KR 20157003616A KR 20150024444 A KR20150024444 A KR 20150024444A
- Authority
- KR
- South Korea
- Prior art keywords
- glu
- gly
- leu
- pro
- ala
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/71—Receptors; Cell surface antigens; Cell surface determinants for growth factors; for growth regulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/06—Free radical scavengers or antioxidants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K19/00—Hybrid peptides, i.e. peptides covalently bound to nucleic acids, or non-covalently bound protein-protein complexes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Cell Biology (AREA)
- Physical Education & Sports Medicine (AREA)
- Diabetes (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- Urology & Nephrology (AREA)
- Obesity (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Hematology (AREA)
- Microbiology (AREA)
- Neurology (AREA)
- Virology (AREA)
- Rheumatology (AREA)
- Epidemiology (AREA)
Abstract
Description
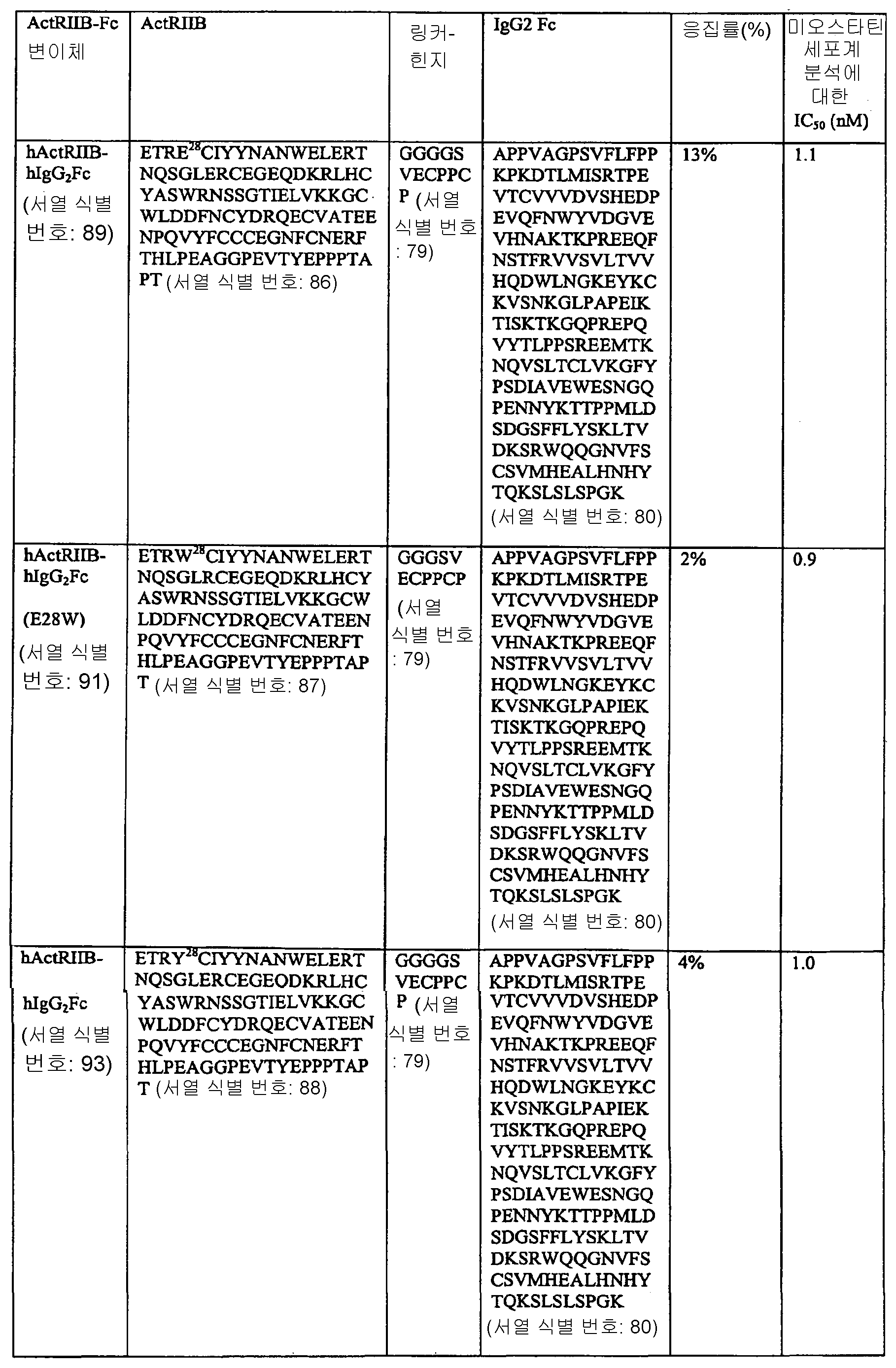
도 2는 가용성 ActRIIB5-인간 IgG1 Fc의 아미노산 서열(서열 식별 번호: 99)을 나타낸다. 신호 펩타이드 서열은 굵은체이며, 성숙 ActRIIB5 가용성 도메인이 뒤따르고, 부분 힌지 영역을 포함한 인간 IgG1 Fc는 이탤릭체이다. E28 및 R40에는 밑줄이 쳐 있다. 링커 서열(GGGGS)(서열 식별 번호: 75)은 이탤릭체이며 밑줄이 쳐 있다.
도 3은 인히빈-α 넉아웃 마우스에서 고환(도 3a) 및 난소(도 3b) 질량에 대한 가용성 vActRIIB-Fc E28W 치료의 효과를 도시한다.
도 4는 수컷(도 4a) 및 암컷(도 4b) 인히빈-α 넉아웃 마우스에서 생존률에 대한 가용성 vActRIIB-Fc E28W 치료의 효과를 도시한다.
도 5는 결장 26 종양-함유 마우스에서 체중에 대한 가용성 vActRIIB-Fc E28W 치료의 효과를 도시한다.
도 6은 결장 26 종양-함유 마우스의 생존에 대한 가용성 vActRIIB-Fc E28W 치료의 효과를 도시한다.
| 변이체 | SPR 정규화된 RU (RU = 반응 단위) |
IC50(nM) 액티빈 |
| ActRIIB-IgG1Fc(서열 식별 번호: 58) | 35 | 8.20 |
| vActRIIB-IgG1Fc, E28(서열 식별 번호: 60) | 20 | 25.30 |
| vActRIIB-IgG1Fc, E28W(서열 식별 번호: 62) | 128 | 2.07 |
| vActRIIB-IgG1Fc, E28Y(서열 식별 번호: 64) | 115 | 2.10 |
| vActRIIB-IgG1Fc, R40G(서열 식별 번호: 66) | 18 | |
| ActRIIB5-IgG1Fc(서열 식별 번호: 68) | 37 | |
| vActRIIB5-IgG1Fc, E28A(서열 식별 번호: 70) | 8 | |
| vActRIIB5-IgG1Fc, E28W(서열 식별 번호: 72) | 45 | 16.86 |
| 서열 식별 번호 | 설명 |
| 1 | ActRIIB5 세포외 도메인, 폴리뉴클레오타이드 |
| 2 | ActRIIB5 세포외 도메인, 폴리펩타이드 |
| 3 | vActRIIB5 E28A 폴리뉴클레오타이드 |
| 4 | vActRIIB5 E28A 폴리펩타이드 |
| 5 | vActRIIB5 E28A 및 R40A 폴리뉴클레오타이드 |
| 6 | vActRIIB5 E28A 및 R40A 폴리펩타이드 |
| 7 | vActRIIB5 E28W 폴리뉴클레오타이드 |
| 8 | vActRIIB5 E28W 폴리펩타이드 |
| 9 | vActRIIB5 E28Y 폴리뉴클레오타이드 |
| 10 | vActRIIB5 E28Y 폴리펩타이드 |
| 11 | vActRIIB5 E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, W 또는 Y 폴리뉴클레오타이드 |
| 12 | vActRIIB5 E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, W 또는 Y 폴리펩타이드 |
| 13 | vActRIIB5 E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, W 또는 Y 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 14 | vActRIIB5 E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, W 또는 Y 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리펩타이드 |
| 15 | vActRIIB5 R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 16 | vActRIIB5 R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리펩타이드 |
| 17 | ActRIIB 세포외 도메인, 폴리뉴클레오타이드 |
| 18 | ActRIIB 세포외 도메인, 폴리펩타이드 |
| 19 | vActRIIB E28A 폴리뉴클레오타이드 |
| 20 | vActRIIB E28A 폴리펩타이드 |
| 21 | vActRIIB E28A 및 R40A 폴리뉴클레오타이드 |
| 22 | vActRIIB E28A 및 R40A 폴리펩타이드 |
| 23 | vActRIIB E28W 폴리뉴클레오타이드 |
| 24 | vActRIIB E28W 폴리펩타이드 |
| 25 | vActRIIB E28Y 폴리뉴클레오타이드 |
| 26 | vActRIIB E28Y 폴리펩타이드 |
| 27 | vActRIIB E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, W 또는 Y 폴리뉴클레오타이드 |
| 28 | vActRIIB E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, W 또는 Y 폴리펩타이드 |
| 29 | vActRIIB E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, Y 또는 W 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 30 | vActRIIB E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, Y 또는 W 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리펩타이드 |
| 31 | vActRIIB R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 32 | vActRIIB R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리펩타이드 |
| 33 | vActRIIB R64A, E28A 폴리뉴클레오타이드 |
| 34 | vActRIIB R64A, E28A 폴리펩타이드 |
| 35 | vActRIIB R64A, E28A 및 R40A 폴리뉴클레오타이드 |
| 36 | vActRIIB R64A, E28A 및 R40A 폴리펩타이드 |
| 37 | vActRIIB R64A, E28W 폴리뉴클레오타이드 |
| 38 | vActRIIB R64A, E28W 폴리펩타이드 |
| 39 | vActRIIB R64A, E28Y 폴리뉴클레오타이드 |
| 40 | vActRIIB R64A, E28Y 폴리펩타이드 |
| 41 | vActRIIB R64A, E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, Y 또는 W 폴리뉴클레오타이드 |
| 42 | vActRIIB R64A, E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, Y 또는 W 폴리펩타이드 |
| 43 | vActRIIB R64A, E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, W 또는 Y 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 44 | vActRIIB R64A, E28X 및 R40X 이때 X(28)는 A, F, Q, V, I, L, M, K, H, W 또는 Y 이때 X(40)는 A, G, Q, M, H, K 또는 N 폴리펩타이드 |
| 45 | vActRIIB R64A, R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리뉴클레오타이드 |
| 46 | vActRIIB R64A, R40X, 이때 X는 G, Q, M, H, K 또는 N 폴리펩타이드 |
| 47 | 서열 수납 번호 NP_001097(야생형 ActRIIB) 폴리펩타이드 |
| 48 | 서열 수납 번호 NM_002192(액티빈 A) 폴리펩타이드 |
| 49 | 서열 수납 번호 AAB86694(미오스타틴) 폴리펩타이드 |
| 50 | 서열 수납 번호 O95390 (GDF-11) 폴리펩타이드 |
| 51 | vActRIIB5 E28X 및 R40X, 이때 X는 임의의 아미노산 폴리뉴클레오타이드 |
| 52 | vActRIIB5 E28X 및 R40X, 이때 X는 임의의 아미노산 폴리펩타이드 |
| 53 | vActRIIB5 E28X 및 R40X, 이때 X는 임의의 아미노산 폴리뉴클레오타이드 |
| 54 | vActRIIB5 E28X 및 R40X, 이때 X는 임의의 아미노산 폴리펩타이드 |
| 55 | vActRIIB5 R64A, E28X 및 R40X, 이때 X는 임의의 아미노산 폴리뉴클레오타이드 |
| 56 | vActRIIB5 R64A, E28X 및 R40X, 이때 X는 임의의 아미노산 폴리펩타이드 |
| 57 | ActRIIB-IgG1Fc 성숙 폴리뉴클레오타이드 |
| 58 | ActRIIB-IgG1Fc 성숙 폴리펩타이드 |
| 59 | vActRIIB-IgG1Fc E28A (E10A) 성숙 폴리뉴클레오타이드 |
| 60 | vActRIIB-IgG1Fc E28A (E10A) 성숙 폴리펩타이드 |
| 61 | vActRIIB-IgG1Fc E28W (E10W) 성숙 폴리뉴클레오타이드 |
| 62 | vActRIIB-IgG1Fc E28W (E10W) 성숙 폴리펩타이드 |
| 63 | vActRIIB-IgG1Fc E28Y (E10Y) 성숙 폴리뉴클레오타이드 |
| 64 | vActRIIB-IgG1Fc E28Y (E10Y) 성숙 폴리펩타이드 |
| 65 | vActRIIB-IgG1Fc R40G (R22G) 성숙 폴리뉴클레오타이드 |
| 66 | vActRIIB-IgG1Fc 성숙 R40G (R22G) 성숙 폴리펩타이드 |
| 67 | vActRIIB-IgG1Fc 성숙 폴리뉴클레오타이드 |
| 68 | vActRIIB-IgG1Fc 성숙 폴리펩타이드 |
| 69 | vActRIIB-IgG1Fc E28A (E10A) 성숙 폴리뉴클레오타이드 |
| 70 | vActRIIB-IgG1Fc E28A (E10A) 성숙 폴리펩타이드 |
| 71 | vActRIIB-IgG1Fc E28W (E10W) 성숙 폴리뉴클레오타이드 E10W |
| 72 | vActRIIB-IgG1Fc E28W (E10W) 성숙 폴리펩타이드 E10W |
| 73 | 도 1 및 2에 도시된 신호 서열 |
| 74 | 대체 신호 서열 |
| 75 | 링커 |
| 76 | IgG1에 대한 완전 힌지 영역 |
| 77 | IgG2에 대한 완전 힌지 영역 |
| 78 | IgG4에 대한 완전 힌지 영역 |
| 79 | 힌지 링커 |
| 80 | IgG2 Fc 폴리펩타이드 |
| 81 | IgG2 Fc 뉴클레오타이드 축퇴 |
| 82 | IgG1 Fc 폴리펩타이드 |
| 83 | IgG1 Fc 폴리뉴클레오타이드 |
| 84 | IgG4 Fc 폴리펩타이드 |
| 85 | IgG4 Fc 폴리뉴클레오타이드-축퇴 |
| 86 | ActRIIB 성숙 절단형 야생형 폴리펩타이드 |
| 87 | vActRIIB (E4W) (E28W) 성숙 절단형 폴리펩타이드 |
| 88 | vActRIIB (E4Y) (E28Y) 성숙 절단형 폴리펩타이드 |
| 89 | ActRIIB-IgG2Fc 성숙 절단형 폴리펩타이드 |
| 90 | ActRIIB-IgG2Fc 성숙 절단형 폴리뉴클레오타이드 축퇴 |
| 91 | vActRIIB-IgG2Fc (E4W) E28W 성숙 절단형 폴리펩타이드 |
| 92 | vActRIIB-IgG2Fc (E4W) E28W 성숙 절단형 폴리뉴클레오타이드 |
| 93 | vActRIIB-IgG2Fc (E4Y) E28Y 성숙 절단형 폴리펩타이드 |
| 94 | vActRIIB-IgG2Fc (E4Y) E28Y 성숙 절단형 폴리뉴클레오타이드 |
| 95 | vActRIIB-IgG2Fc (E4A) E28A 성숙 절단형 폴리펩타이드 |
| 96 | vActRIIB-IgG2Fc (E4A) E28A 성숙 절단형 폴리뉴클레오타이드 축퇴 |
| 97 | vActRIIB-IgG2Fc (E4X) E28X, 이때 X는 A, F, Q, V, I, L, M, K, H, W 또는 Y 성숙 절단형 폴리펩타이드 |
| 98 | 도 1 - ActRIIB-IgG1 Fc |
| 99 | 도 2 - ActRIIB5-IgG1 Fc |
| 야생형 + 비히클 | 넉아웃 + 비히클 | 넉아웃 + E28W | |
| 체중 | 28.64 ± 1.11 | 21.36 ± 0.99* | 33.10 ± 1.56*# |
| 제지방 사체(g) | 12.07 ± 0.36 | 8.00 ± 0.29* | 14.90 ± 0.77*# |
| 장딴지 근육(g) | 0.324 ± 0.014 | 0.209 ± 0.012* | 0.426 ± 0.024*# |
| *: P < 0.05 대 야생형 + 비히클; #: P < 0.05 대 넉아웃 +비히클 | |||
| 그룹/일(체중 변화(%)) | 3일(%) | 7일(%) | 14일(%) |
| 비부유 + PBS | 2.4% | 2.9% | 4.8% |
| 부유 + 비히클 | -10.0% | -3.0% | -0.2% |
| 부유 + E28W, 30 mg/ml | -9.7% | 2.1% | 12.6% |
Claims (16)
- 단백질을 생산하는 방법으로서,
상기 방법은,
단백질을 암호화하는 핵산 분자를 포함하는 숙주 세포를 얻는 단계;
상기 숙주 세포를 배양하여 단백질을 발현시키는 단계: 및
단백질을 생산하는 단계를 포함하며,
상기 단백질은,
(a) 위치 28에서의 아미노산 치환을 제외하고, 서열 식별 번호: 18에 기재된 폴리펩타이드 서열을 갖는 폴리펩타이드;
(b) 위치 28에서의 아미노산 치환을 제외하고, 서열 식별 번호: 18의 아미노산 19 내지 134에 기재된 폴리펩타이드 서열을 갖는 폴리펩타이드;
(c) 위치 28에서의 아미노산 치환을 제외하고, 서열 식별 번호: 18의 아미노산 23 내지 134에 기재된 폴리펩타이드 서열을 갖는 폴리펩타이드;
(d) 위치 28에서의 아미노산 치환을 제외하고, 서열 식별 번호: 18의 아미노산 25 내지 134에 기재된 폴리펩타이드 서열을 갖는 폴리펩타이드; 및
(e) 위치 28에서의 아미노산 치환을 제외하고, 상기 (a) 내지 (d) 중 어느 하나와 99% 이상의 동일성을 갖는 폴리펩타이드로 이루어진 군으로부터 선택된 폴리펩타이드를 포함하며,
상기 폴리펩타이드는 미오스타틴 또는 액티빈 A에 결합할 수 있는 것인, 방법. - 제 1 항에 있어서,
단백질은 이종 폴리펩타이드에 링커로 연결된 폴리펩타이드를 포함하는 방법. - 제 2 항에 있어서,
링커가 펩타이드 링커를 포함하고, 이종 폴리펩타이드가 인간 Fc 도메인을 포함하는 방법. - 제 3 항에 있어서,
링커가 서열 식별 번호: 79의 서열을 포함하고, 이종 폴리펩타이드가 서열 식별 번호: 80의 서열을 포함하는 방법. - 제 4 항에 있어서,
링커가 서열 식별 번호: 79의 서열로 이루어지고, 이종 폴리펩타이드가 서열 식별 번호: 80의 서열로 이루어진 방법. - 제 1 항에 있어서,
폴리펩타이드가 하나 이상의 이종 폴리펩타이드에 융합되는 방법. - 제 6 항에 있어서,
이종 폴리펩타이드가 인간 Fc 도메인인 방법. - 제 7 항에 있어서,
단백질은 링커를 추가로 포함하는 방법. - 제 8 항에 있어서,
단백질은 서열 식별 번호: 91에 기재된 서열을 갖는 방법. - 제 1 항에 있어서,
핵산 분자는 재조합 발현 벡터에 존재하는 방법. - 제 1 항에 있어서,
숙주 세포는 핵산 분자를 포함하는 벡터로 형질전환된 방법. - 제 1 항에 있어서,
숙주 세포는 포유류 세포인 방법. - 제 1 항에 있어서,
숙주 세포는 박테리아 세포인 방법. - 제 1 항에 있어서,
숙주 세포는 숙주 세포 및 세포 배양배지를 포함하는 세포 배양물에서 얻어지는 방법. - 제 1 항에 있어서,
단백질을 분리하는 단계를 더 포함하는 방법. - 제 15 항에 있어서,
단백질을 단백질 및 담체를 포함하는 약학적 조성물로 생산하는 가공 단계를 더 포함하는 방법.
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US90545907P | 2007-03-06 | 2007-03-06 | |
| US60/905,459 | 2007-03-06 | ||
| US6547408P | 2008-02-11 | 2008-02-11 | |
| US61/065,474 | 2008-02-11 | ||
| PCT/US2008/003119 WO2008109167A2 (en) | 2007-03-06 | 2008-03-06 | Variant activin receptor polypeptides and uses thereof |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020147010346A Division KR101633728B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167016449A Division KR20160075861A (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20150024444A true KR20150024444A (ko) | 2015-03-06 |
| KR101633727B1 KR101633727B1 (ko) | 2016-06-28 |
Family
ID=39739001
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167016449A Abandoned KR20160075861A (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020147010346A Expired - Fee Related KR101633728B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020157003616A Expired - Fee Related KR101633727B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020097020320A Ceased KR20090127146A (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020127008467A Expired - Fee Related KR101428344B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167016449A Abandoned KR20160075861A (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020147010346A Expired - Fee Related KR101633728B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020097020320A Ceased KR20090127146A (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
| KR1020127008467A Expired - Fee Related KR101428344B1 (ko) | 2007-03-06 | 2008-03-06 | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 |
Country Status (29)
| Country | Link |
|---|---|
| US (5) | US7947646B2 (ko) |
| EP (2) | EP3141605A1 (ko) |
| JP (1) | JP5496682B2 (ko) |
| KR (5) | KR20160075861A (ko) |
| CN (2) | CN105924516A (ko) |
| AR (1) | AR065611A1 (ko) |
| AU (1) | AU2008223338C9 (ko) |
| BR (1) | BRPI0808332A2 (ko) |
| CA (1) | CA2679841C (ko) |
| CL (2) | CL2008000664A1 (ko) |
| CR (2) | CR11054A (ko) |
| CY (1) | CY1118857T1 (ko) |
| DK (1) | DK2132314T3 (ko) |
| EA (2) | EA201490822A1 (ko) |
| ES (1) | ES2613043T3 (ko) |
| HR (1) | HRP20170323T1 (ko) |
| HU (1) | HUE032260T2 (ko) |
| IL (2) | IL200605A (ko) |
| LT (1) | LT2132314T (ko) |
| MX (3) | MX389715B (ko) |
| NZ (1) | NZ579369A (ko) |
| PE (3) | PE20171325A1 (ko) |
| PH (1) | PH12013502308A1 (ko) |
| PL (1) | PL2132314T3 (ko) |
| PT (1) | PT2132314T (ko) |
| RS (1) | RS55726B1 (ko) |
| SI (1) | SI2132314T1 (ko) |
| TW (3) | TWI573802B (ko) |
| WO (1) | WO2008109167A2 (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102015118132A1 (de) | 2015-02-17 | 2016-08-18 | Hyundai Motor Company | Zahnstangeneinheit einer Fahrzeuglenkvorrichtung |
Families Citing this family (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1771470B1 (en) | 2004-07-23 | 2013-06-26 | Acceleron Pharma Inc. | Actrii receptor polypeptides, methods and compositions |
| BRPI0616923A2 (pt) | 2005-10-06 | 2011-07-05 | Lilly Co Eli | anticorpos monoclonais, seus usos e composição farmacêutica |
| UA92504C2 (en) | 2005-10-12 | 2010-11-10 | Эли Лилли Энд Компани | Anti-myostatin monoclonal antibody |
| US8067562B2 (en) | 2005-11-01 | 2011-11-29 | Amgen Inc. | Isolated nucleic acid molecule comprising the amino acid sequence of SEQ ID NO:1 |
| KR20190006086A (ko) | 2005-11-23 | 2019-01-16 | 악셀레론 파마 인코포레이티드 | 액티빈-actrⅱa 길항제 및 골 성장을 촉진하기 위한 이들의 용도 |
| US8128933B2 (en) | 2005-11-23 | 2012-03-06 | Acceleron Pharma, Inc. | Method of promoting bone growth by an anti-activin B antibody |
| CA2661836A1 (en) | 2006-09-05 | 2008-03-13 | Eli Lilly And Company | Anti-myostatin antibodies |
| US8895016B2 (en) | 2006-12-18 | 2014-11-25 | Acceleron Pharma, Inc. | Antagonists of activin-actriia and uses for increasing red blood cell levels |
| DK2124999T3 (da) | 2006-12-18 | 2013-01-14 | Acceleron Pharma Inc | Activin-actrii antagonister og anvendelser til behandling af anæmi |
| EP2111229B1 (en) | 2007-02-01 | 2013-04-10 | Acceleron Pharma, Inc. | Pharmaceutical compositions comprising Activin-ActRIIA antagonists for use in preventing or treating breast cancer metastasis or breast cancer related bone loss |
| TWI782836B (zh) | 2007-02-02 | 2022-11-01 | 美商艾瑟勒朗法瑪公司 | 衍生自ActRIIB的變體與其用途 |
| ES2756725T3 (es) | 2007-02-09 | 2020-04-27 | Acceleron Pharma Inc | Composiciones farmacéuticas que comprenden antagonistas de Activina-ActRIIA |
| TWI573802B (zh) | 2007-03-06 | 2017-03-11 | 安美基公司 | 變異之活動素受體多肽及其用途 |
| US8501678B2 (en) | 2007-03-06 | 2013-08-06 | Atara Biotherapeutics, Inc. | Variant activin receptor polypeptides and uses thereof |
| EP3243524A1 (en) | 2007-09-18 | 2017-11-15 | Acceleron Pharma Inc. | Activin-actriia antagonists and uses for decreasing or inhibiting fsh secretion |
| US8216997B2 (en) | 2008-08-14 | 2012-07-10 | Acceleron Pharma, Inc. | Methods for increasing red blood cell levels and treating anemia using a combination of GDF traps and erythropoietin receptor activators |
| SMT202300222T1 (it) | 2008-08-14 | 2023-09-06 | Acceleron Pharma Inc | Trappole di gdf |
| AU2016210719B2 (en) * | 2008-11-26 | 2018-03-29 | Amgen Inc. | Variants of activin IIB receptor polypeptides and uses thereof |
| SG10201703067QA (en) * | 2008-11-26 | 2017-05-30 | Amgen Inc | Variants Of Activin IIB Receptor Polypeptides And Uses Thereof |
| AU2013216639B2 (en) * | 2008-11-26 | 2016-05-05 | Amgen Inc. | Variants of activin IIB receptor polypeptides and uses thereof |
| US8138142B2 (en) | 2009-01-13 | 2012-03-20 | Acceleron Pharma Inc. | Methods for increasing adiponectin in a patient in need thereof |
| JP5766179B2 (ja) | 2009-04-27 | 2015-08-19 | ノバルティス アーゲー | 筋肉増殖を増加させるための組成物および方法 |
| BRPI1010587A2 (pt) | 2009-06-08 | 2019-04-09 | Acceleron Pharma Inc. | métodos para aumentar adipócitos termogênicos |
| EP2440577A4 (en) * | 2009-06-12 | 2013-01-23 | Acceleron Pharma Inc | SHORTEN ACTRIIB FC FUSION PROTEINS |
| EP3818988A1 (en) | 2009-11-03 | 2021-05-12 | Acceleron Pharma Inc. | Methods for treating fatty liver disease |
| JP6267425B2 (ja) | 2009-11-17 | 2018-01-24 | アクセルロン ファーマ, インコーポレイテッド | 筋ジストロフィー治療のためのユートロフィン誘導に関するactriibタンパク質およびその改変体およびその使用 |
| JO3340B1 (ar) | 2010-05-26 | 2019-03-13 | Regeneron Pharma | مضادات حيوية لـعامل تمايز النمو 8 البشري |
| US8999343B2 (en) | 2010-08-16 | 2015-04-07 | Amgen Inc. | Antibodies that bind myostatin, compositions and methods |
| CN103298832A (zh) | 2010-11-08 | 2013-09-11 | 阿塞勒隆制药公司 | Actriia结合剂及其用途 |
| PH12013500720A1 (en) | 2010-11-12 | 2013-06-03 | Colgate Palmolive Co | Oral care product and methods of use and manufacture thereof |
| HUE040276T2 (hu) * | 2011-07-01 | 2019-02-28 | Novartis Ag | Eljárás metabolikus rendellenességek kezelésére |
| US8765385B2 (en) * | 2011-10-27 | 2014-07-01 | Ravindra Kumar | Method of detection of neutralizing anti-actriib antibodies |
| AU2012339722B2 (en) | 2011-11-14 | 2017-09-14 | Regeneron Pharmaceuticals, Inc. | Compositions and methods for increasing muscle mass and muscle strength by specifically antagonizing GDF8 and/or Activin A |
| HK1203384A1 (en) * | 2011-12-19 | 2015-12-11 | Amgen Inc. | Variant activin receptor polypeptides, alone or in combination with chemotherapy, and uses thereof |
| WO2013142114A1 (en) * | 2012-03-19 | 2013-09-26 | The Brigham And Women's Hosptial, Inc. | Growth differentiation factor (gdf) for treatment of diastolic heart failure |
| JP2015525230A (ja) | 2012-06-11 | 2015-09-03 | アムジエン・インコーポレーテツド | デュアル受容体アンタゴニスト性抗原結合タンパク質およびその使用 |
| AU2013334660B2 (en) | 2012-10-24 | 2018-08-09 | Celgene Corporation | Methods for treating anemia |
| NZ707477A (en) | 2012-11-02 | 2019-09-27 | Celgene Corp | Activin-actrii antagonists and uses for treating bone and other disorders |
| SG10201706148WA (en) | 2013-02-01 | 2017-08-30 | Santa Maria Biotherapeutics Inc | Administration of an anti-activin-a compound to a subject |
| US20160038588A1 (en) * | 2013-03-15 | 2016-02-11 | Amgen Inc. | Myostatin Antagonism in Human Subjects |
| EP2983694B1 (en) | 2013-04-08 | 2022-06-22 | President and Fellows of Harvard College | Compositions for rejuvenating skeletal muscle stem cells |
| TW201920262A (zh) | 2013-07-30 | 2019-06-01 | 美商再生元醫藥公司 | 抗活化素a之抗體及其用途 |
| US10017566B2 (en) | 2013-11-12 | 2018-07-10 | The Brigham And Women's Hospital, Inc. | Growth differentiation factor (GDF) for treatment of diastolic heart failure |
| KR20170035891A (ko) * | 2014-06-13 | 2017-03-31 | 산타 마리아 바이오테라퓨틱스, 인코포레이티드 | 제형화된 수용체 폴리펩타이드 및 관련 방법 |
| WO2015192111A1 (en) | 2014-06-13 | 2015-12-17 | Acceleron Pharma, Inc. | Methods and compositions for treating ulcers |
| MA41052A (fr) * | 2014-10-09 | 2017-08-15 | Celgene Corp | Traitement d'une maladie cardiovasculaire à l'aide de pièges de ligands d'actrii |
| JP2018501307A (ja) | 2014-12-03 | 2018-01-18 | セルジーン コーポレイション | アクチビン−ActRIIアンタゴニスト及び貧血を治療するための使用 |
| MX2017007519A (es) * | 2014-12-08 | 2017-08-22 | Novartis Ag | Antagonistas de miostatina o activina para el tratamiento de sarcopenia. |
| EP3256148A1 (en) | 2015-02-12 | 2017-12-20 | INSERM - Institut National de la Santé et de la Recherche Médicale | Methods for predicting the responsiveness of a patient affected with malignant hematological disease to chemotherapy treatment and methods of treatment of such disease |
| US10227392B2 (en) | 2015-04-06 | 2019-03-12 | Acceleron Pharma Inc. | ALK7:ActRIIB heteromultimers and uses thereof |
| MA41919A (fr) | 2015-04-06 | 2018-02-13 | Acceleron Pharma Inc | Hétéromultimères alk4:actriib et leurs utilisations |
| CN107849114B (zh) | 2015-04-06 | 2021-08-20 | 阿塞勒隆制药公司 | 单臂i型和ii型受体融合蛋白和其用途 |
| WO2016168613A1 (en) | 2015-04-15 | 2016-10-20 | Regeneron Pharmaceuticals, Inc. | Methods of increasing strength and functionality with gdf8 inhibitors |
| PT3286206T (pt) | 2015-04-22 | 2021-04-01 | Biogen Ma Inc | Novas proteínas híbridas bloqueadoras de ligando actriib para tratar doenças de desgaste muscular |
| RU2651776C2 (ru) * | 2015-12-01 | 2018-04-23 | Общество с ограниченной ответственностью "Международный биотехнологический центр "Генериум" ("МБЦ "Генериум") | Биспецифические антитела против cd3*cd19 |
| CA3010799A1 (en) | 2016-01-06 | 2017-07-13 | President And Fellows Of Harvard College | Treatment with gdf11 prevents weight gain, improves glucose tolerance and reduces hepatosteatosis |
| JP6987072B2 (ja) | 2016-03-10 | 2021-12-22 | アクセレロン ファーマ インコーポレーテッド | アクチビン2型受容体結合タンパク質及びその使用 |
| BR112019006993A2 (pt) | 2016-10-05 | 2019-09-03 | Acceleron Pharma Inc | heteromultímeros de alk4:actriib e usos dos mesmos |
| JOP20190085A1 (ar) * | 2016-10-20 | 2019-04-17 | Biogen Ma Inc | طرق علاج الضمور العضلي ومرض العظام باستخدام بروتينات احتجاز مركب ترابطي actriib هجين حديثة |
| CN110430890B (zh) | 2016-11-10 | 2024-09-10 | 科乐斯疗法公司 | 激活素受体iia型变体及其使用方法 |
| EP3706777B1 (en) | 2017-11-09 | 2024-05-22 | Keros Therapeutics, Inc. | Activin receptor type iia variants and methods of use thereof |
| IL320014A (en) | 2018-01-12 | 2025-06-01 | Keros Therapeutics Inc | Activin receptor type iib variants and methods of use thereof |
| CN111787981B (zh) | 2018-03-01 | 2025-12-30 | 瑞泽恩制药公司 | 增加肌肉量和减少脂肪量的方法 |
| JP7405772B2 (ja) | 2018-05-09 | 2023-12-26 | ケロス セラピューティクス インコーポレイテッド | アクチビンiia型受容体変異体および同変異体を含む医薬組成物 |
| WO2021189010A1 (en) | 2020-03-20 | 2021-09-23 | Keros Therapeutics, Inc. | Methods of using activin receptor type iib variants |
| IL296394A (en) | 2020-03-20 | 2022-11-01 | Keros Therapeutics Inc | Type ii activin receptor chimeras and methods of using them |
| EP4142769A4 (en) | 2020-04-28 | 2024-05-15 | Acceleron Pharma Inc. | ACTRII PROTEINS AND USE IN THE TREATMENT OF POSTCAPILLARY PULMONARY HYPERTENSION |
| US20250041385A1 (en) * | 2021-12-10 | 2025-02-06 | Biogen Ma Inc. | Modified actrii proteins and methods of use thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004039948A2 (en) * | 2002-10-25 | 2004-05-13 | Wyeth | Actriib fusion polypeptides and uses therefor |
| US20060034831A1 (en) * | 2004-08-12 | 2006-02-16 | Wyeth | Combination therapy for diabetes, obesity and cardiovascular diseases using GDF-8 inhibitors |
Family Cites Families (95)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2899886A (en) * | 1959-08-18 | rodth | ||
| US3773919A (en) | 1969-10-23 | 1973-11-20 | Du Pont | Polylactide-drug mixtures |
| JPS5349966A (en) | 1976-10-18 | 1978-05-06 | Hitachi Ltd | Manufacture of semiconductor integrated circuit |
| US4263428A (en) | 1978-03-24 | 1981-04-21 | The Regents Of The University Of California | Bis-anthracycline nucleic acid function inhibitors and improved method for administering the same |
| IE52535B1 (en) | 1981-02-16 | 1987-12-09 | Ici Plc | Continuous release pharmaceutical compositions |
| US4411993A (en) | 1981-04-29 | 1983-10-25 | Steven Gillis | Hybridoma antibody which inhibits interleukin 2 activity |
| USRE32011E (en) | 1981-12-14 | 1985-10-22 | Scripps Clinic And Research Foundation | Ultrapurification of factor VIII using monoclonal antibodies |
| DE3374837D1 (en) | 1982-02-17 | 1988-01-21 | Ciba Geigy Ag | Lipids in the aqueous phase |
| US4543439A (en) | 1982-12-13 | 1985-09-24 | Massachusetts Institute Of Technology | Production and use of monoclonal antibodies to phosphotyrosine-containing proteins |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| HUT35524A (en) | 1983-08-02 | 1985-07-29 | Hoechst Ag | Process for preparing pharmaceutical compositions containing regulatory /regulative/ peptides providing for the retarded release of the active substance |
| EP0143949B1 (en) | 1983-11-01 | 1988-10-12 | TERUMO KABUSHIKI KAISHA trading as TERUMO CORPORATION | Pharmaceutical composition containing urokinase |
| US4902614A (en) | 1984-12-03 | 1990-02-20 | Teijin Limited | Monoclonal antibody to human protein C |
| US5618920A (en) | 1985-11-01 | 1997-04-08 | Xoma Corporation | Modular assembly of antibody genes, antibodies prepared thereby and use |
| US5750375A (en) | 1988-01-22 | 1998-05-12 | Zymogenetics, Inc. | Methods of producing secreted receptor analogs and biologically active dimerized polypeptide fusions |
| US5567584A (en) | 1988-01-22 | 1996-10-22 | Zymogenetics, Inc. | Methods of using biologically active dimerized polypeptide fusions to detect PDGF |
| JP2877509B2 (ja) | 1989-05-19 | 1999-03-31 | アムジエン・インコーポレーテツド | メタロプロテイナーゼ阻害剤 |
| US6300129B1 (en) | 1990-08-29 | 2001-10-09 | Genpharm International | Transgenic non-human animals for producing heterologous antibodies |
| US6565841B1 (en) | 1991-03-15 | 2003-05-20 | Amgen, Inc. | Pulmonary administration of granulocyte colony stimulating factor |
| US20050186593A1 (en) * | 1991-05-10 | 2005-08-25 | The Salk Institute For Biological Studies | Cloning and recombinant production of CRF receptor(s) |
| US6162896A (en) * | 1991-05-10 | 2000-12-19 | The Salk Institute For Biological Studies | Recombinant vertebrate activin receptors |
| US5885794A (en) * | 1991-05-10 | 1999-03-23 | The Salk Institute For Biological Studies | Recombinant production of vertebrate activin receptor polypeptides and identification of receptor DNAs in the activin/TGF-β superfamily |
| EP0521187B1 (fr) * | 1991-07-05 | 1995-11-02 | Societe Des Produits Nestle S.A. | Dispositif d'extraction de cartouches adaptable à toute machine espresso |
| DE69216159T2 (de) * | 1991-07-05 | 1997-04-10 | Nestle Sa | Stabile Kaffeekassette und Verfahren zu ihrer Herstellung |
| WO1993015722A1 (en) | 1992-02-07 | 1993-08-19 | Syntex (Usa) Inc. | Controlled delivery of pharmaceuticals from preformed porous microparticles |
| US6153407A (en) | 1992-07-28 | 2000-11-28 | Beth Israel Deaconess Medical Center | Erythropoietin DNA having modified 5' and 3' sequences and its use to prepare EPO therapeutics |
| US5837821A (en) | 1992-11-04 | 1998-11-17 | City Of Hope | Antibody construct |
| GB9223377D0 (en) | 1992-11-04 | 1992-12-23 | Medarex Inc | Humanized antibodies to fc receptors for immunoglobulin on human mononuclear phagocytes |
| US6607884B1 (en) | 1993-03-19 | 2003-08-19 | The Johns Hopkins University School Of Medicine | Methods of detecting growth differentiation factor-8 |
| US6465239B1 (en) | 1993-03-19 | 2002-10-15 | The John Hopkins University School Of Medicine | Growth differentiation factor-8 nucleic acid and polypeptides from aquatic species and non-human transgenic aquatic species |
| US5994618A (en) | 1997-02-05 | 1999-11-30 | Johns Hopkins University School Of Medicine | Growth differentiation factor-8 transgenic mice |
| DK0690873T3 (da) | 1993-03-19 | 2003-09-29 | Univ Johns Hopkins Med | Vækstdifferentieringsfaktor-8 |
| AU702163B2 (en) | 1994-04-29 | 1999-02-18 | Creative Biomolecules, Inc. | Morphogenic protein-specific cell surface receptors and uses therefor |
| US6891082B2 (en) * | 1997-08-01 | 2005-05-10 | The Johns Hopkins University School Of Medicine | Transgenic non-human animals expressing a truncated activintype II receptor |
| US6656475B1 (en) | 1997-08-01 | 2003-12-02 | The Johns Hopkins University School Of Medicine | Growth differentiation factor receptors, agonists and antagonists thereof, and methods of using same |
| US6472179B2 (en) | 1998-09-25 | 2002-10-29 | Regeneron Pharmaceuticals, Inc. | Receptor based antagonists and methods of making and using |
| US6660843B1 (en) | 1998-10-23 | 2003-12-09 | Amgen Inc. | Modified peptides as therapeutic agents |
| IL143034A0 (en) | 1998-11-13 | 2002-04-21 | Immunex Corp | Human tslp dna and polypeptides |
| AU772694B2 (en) | 1999-01-21 | 2004-05-06 | Metamorphix International, Inc. | Growth differentiation factor inhibitors and uses therefor |
| JP2000262405A (ja) * | 1999-03-18 | 2000-09-26 | Soc Prod Nestle Sa | 飲料抽出用密封カートリッジ |
| JP4487376B2 (ja) | 2000-03-31 | 2010-06-23 | 味の素株式会社 | 腎疾患治療剤 |
| TWI236360B (en) * | 2000-06-30 | 2005-07-21 | Nestle Sa | Capsule cage |
| US6812339B1 (en) * | 2000-09-08 | 2004-11-02 | Applera Corporation | Polymorphisms in known genes associated with human disease, methods of detection and uses thereof |
| EP1190959B1 (fr) * | 2000-09-26 | 2004-03-31 | Societe Des Produits Nestle S.A. | Cartouche fermée pour la confection d'une boisson, prévue pour être extraite sous pression |
| EP1203554A1 (fr) * | 2000-11-03 | 2002-05-08 | Societe Des Produits Nestle S.A. | Dispositif pour l'extraction d'une substance alimentaire contenue dans un élément de recharge |
| DK1208782T3 (da) * | 2000-11-28 | 2004-12-13 | Nestle Sa | Perkolationsindretning |
| US20050188854A1 (en) * | 2002-02-07 | 2005-09-01 | The Coca-Cola Co. | Coffee and tea dispenser |
| US6786134B2 (en) * | 2002-02-07 | 2004-09-07 | The Coca-Cola Company | Coffee and tea dispenser |
| GB0209896D0 (en) | 2002-04-30 | 2002-06-05 | Molmed Spa | Conjugate |
| FR2842092B1 (fr) * | 2002-07-12 | 2004-12-24 | Seb Sa | Machine a cafe fonctionnant avec des doses |
| CN101287484B (zh) | 2002-12-20 | 2012-10-10 | 安姆根有限公司 | 抑制肌肉生长抑制素的结合剂 |
| BRPI0410927A (pt) | 2003-06-02 | 2006-06-27 | Wyeth Corp | métodos terapêuticos e profiláticos para distúrbios neuromusculares |
| US8327754B2 (en) * | 2003-07-22 | 2012-12-11 | The Coca-Cola Company | Coffee and tea pod |
| US20060196364A1 (en) * | 2003-07-22 | 2006-09-07 | The Coca-Cola Company | Coffee & Tea Pod |
| ES2655435T3 (es) | 2003-09-12 | 2018-02-20 | Amgen Inc. | Formulación de disolución rápida de cinacalcet |
| US7074445B2 (en) * | 2003-10-30 | 2006-07-11 | Frito-Lay North America, Inc. | Method for adhering large seasoning bits to a food substrate |
| US8110665B2 (en) | 2003-11-13 | 2012-02-07 | Hanmi Holdings Co., Ltd. | Pharmaceutical composition comprising an immunoglobulin FC region as a carrier |
| ATE555133T1 (de) | 2003-11-13 | 2012-05-15 | Hanmi Holdings Co Ltd | Igg fc fragment für einen arzneimittelträger und verfahren zu dessen herstellung |
| GB2411106B (en) * | 2004-02-17 | 2006-11-22 | Kraft Foods R & D Inc | Cartridge for the preparation of beverages |
| US7323169B2 (en) | 2004-04-23 | 2008-01-29 | Amgen Inc. | Sustained release formulations |
| EP1771470B1 (en) | 2004-07-23 | 2013-06-26 | Acceleron Pharma Inc. | Actrii receptor polypeptides, methods and compositions |
| GB2416480B (en) * | 2004-07-27 | 2007-12-27 | Kraft Foods R & D Inc | A system for the preparation of beverages |
| EA011879B1 (ru) | 2004-09-24 | 2009-06-30 | Эмджин Инк. | МОЛЕКУЛЫ С МОДИФИЦИРОВАННЫМ Fc ФРАГМЕНТОМ |
| EP1640756A1 (en) * | 2004-09-27 | 2006-03-29 | Barco N.V. | Methods and systems for illuminating |
| KR100754667B1 (ko) | 2005-04-08 | 2007-09-03 | 한미약품 주식회사 | 비펩타이드성 중합체로 개질된 면역글로불린 Fc 단편 및이를 포함하는 약제학적 조성물 |
| CA2621371C (en) | 2005-09-07 | 2018-05-15 | Amgen Fremont Inc. | Human monoclonal antibodies to activin receptor-like kinase-1 |
| PL1775234T3 (pl) * | 2005-10-14 | 2009-01-30 | Nestec Sa | Kapsułka do przygotowywania napoju |
| US8067562B2 (en) | 2005-11-01 | 2011-11-29 | Amgen Inc. | Isolated nucleic acid molecule comprising the amino acid sequence of SEQ ID NO:1 |
| US20080014860A1 (en) * | 2006-07-14 | 2008-01-17 | Lynn Byron Heitman | Method and apparatus for eliminating register boxes, improving penetration sealing, improving airflow and reducing the labor costs to install ceiling registers |
| CL2007002567A1 (es) | 2006-09-08 | 2008-02-01 | Amgen Inc | Proteinas aisladas de enlace a activina a humana. |
| DK2124999T3 (da) | 2006-12-18 | 2013-01-14 | Acceleron Pharma Inc | Activin-actrii antagonister og anvendelser til behandling af anæmi |
| TWI782836B (zh) * | 2007-02-02 | 2022-11-01 | 美商艾瑟勒朗法瑪公司 | 衍生自ActRIIB的變體與其用途 |
| TWI573802B (zh) | 2007-03-06 | 2017-03-11 | 安美基公司 | 變異之活動素受體多肽及其用途 |
| US8501678B2 (en) | 2007-03-06 | 2013-08-06 | Atara Biotherapeutics, Inc. | Variant activin receptor polypeptides and uses thereof |
| ES2449753T3 (es) | 2007-03-19 | 2014-03-21 | National Research Council Of Canada | Proteínas de fusión que comprenden dos dominios de unión tgf-beta |
| US8431175B2 (en) * | 2007-06-05 | 2013-04-30 | Nestec S.A. | Method for preparing a beverage or food liquid and system using brewing centrifugal force |
| JP5082676B2 (ja) * | 2007-08-23 | 2012-11-28 | 株式会社サタケ | 光学式穀物選別機 |
| EP3243524A1 (en) | 2007-09-18 | 2017-11-15 | Acceleron Pharma Inc. | Activin-actriia antagonists and uses for decreasing or inhibiting fsh secretion |
| AU2009262970A1 (en) | 2008-06-26 | 2009-12-30 | Acceleron Pharma Inc. | Methods for dosing an activin-ActRIIa antagonist and monitoring of treated patients |
| PL2299851T3 (pl) * | 2008-07-08 | 2019-01-31 | Nestec S.A. | System żywieniowy o kontrolowanych porcjach oraz sposób stosowania kapsułek |
| US8151694B2 (en) * | 2008-08-01 | 2012-04-10 | Keurig, Incorporated | Beverage forming apparatus with centrifugal pump |
| US8216997B2 (en) | 2008-08-14 | 2012-07-10 | Acceleron Pharma, Inc. | Methods for increasing red blood cell levels and treating anemia using a combination of GDF traps and erythropoietin receptor activators |
| SMT202300222T1 (it) | 2008-08-14 | 2023-09-06 | Acceleron Pharma Inc | Trappole di gdf |
| US8512784B2 (en) * | 2008-09-02 | 2013-08-20 | Nestec S.A. | Method and device for controlled beverage production using centrifugal forces |
| SG10201703067QA (en) | 2008-11-26 | 2017-05-30 | Amgen Inc | Variants Of Activin IIB Receptor Polypeptides And Uses Thereof |
| US8138142B2 (en) | 2009-01-13 | 2012-03-20 | Acceleron Pharma Inc. | Methods for increasing adiponectin in a patient in need thereof |
| JP5766179B2 (ja) | 2009-04-27 | 2015-08-19 | ノバルティス アーゲー | 筋肉増殖を増加させるための組成物および方法 |
| BRPI1010587A2 (pt) | 2009-06-08 | 2019-04-09 | Acceleron Pharma Inc. | métodos para aumentar adipócitos termogênicos |
| US20120295814A1 (en) | 2010-01-08 | 2012-11-22 | The Brigham And Women's Hospital, Inc. | CA-125 Immune Complexes as Biomarkers of Ovarian Cancer |
| WO2011123691A1 (en) | 2010-03-31 | 2011-10-06 | Keryx Biopharmaceuticals, Inc. | Perifosine and capecitabine as a combined treatment for cancer |
| CN103298832A (zh) | 2010-11-08 | 2013-09-11 | 阿塞勒隆制药公司 | Actriia结合剂及其用途 |
| HK1203384A1 (en) | 2011-12-19 | 2015-12-11 | Amgen Inc. | Variant activin receptor polypeptides, alone or in combination with chemotherapy, and uses thereof |
| WO2013106715A1 (en) | 2012-01-13 | 2013-07-18 | Allergan, Inc. | Crosslinked hyaluronic acid-collagen gels for improving tissue graft viability and soft tissue augmentation |
| WO2013180295A1 (ja) | 2012-06-01 | 2013-12-05 | 日本電信電話株式会社 | パケット転送処理方法およびパケット転送処理装置 |
| US9300829B2 (en) | 2014-04-04 | 2016-03-29 | Canon Kabushiki Kaisha | Image reading apparatus and correction method thereof |
-
2008
- 2008-03-05 TW TW103117408A patent/TWI573802B/zh not_active IP Right Cessation
- 2008-03-05 US US12/074,877 patent/US7947646B2/en active Active
- 2008-03-05 TW TW097107642A patent/TWI454479B/zh not_active IP Right Cessation
- 2008-03-05 TW TW105129851A patent/TW201718635A/zh unknown
- 2008-03-06 WO PCT/US2008/003119 patent/WO2008109167A2/en not_active Ceased
- 2008-03-06 MX MX2017005686A patent/MX389715B/es unknown
- 2008-03-06 ES ES08742032.9T patent/ES2613043T3/es active Active
- 2008-03-06 KR KR1020167016449A patent/KR20160075861A/ko not_active Abandoned
- 2008-03-06 CN CN201610218042.1A patent/CN105924516A/zh active Pending
- 2008-03-06 KR KR1020147010346A patent/KR101633728B1/ko not_active Expired - Fee Related
- 2008-03-06 PT PT87420329T patent/PT2132314T/pt unknown
- 2008-03-06 MX MX2015002054A patent/MX348286B/es unknown
- 2008-03-06 JP JP2009552758A patent/JP5496682B2/ja active Active
- 2008-03-06 EA EA201490822A patent/EA201490822A1/ru unknown
- 2008-03-06 PE PE2017001328A patent/PE20171325A1/es not_active Application Discontinuation
- 2008-03-06 EP EP16194631.4A patent/EP3141605A1/en not_active Withdrawn
- 2008-03-06 MX MX2009009495A patent/MX2009009495A/es active IP Right Grant
- 2008-03-06 HU HUE08742032A patent/HUE032260T2/en unknown
- 2008-03-06 CA CA2679841A patent/CA2679841C/en active Active
- 2008-03-06 EP EP08742032.9A patent/EP2132314B1/en active Active
- 2008-03-06 NZ NZ579369A patent/NZ579369A/en not_active IP Right Cessation
- 2008-03-06 RS RS20170192A patent/RS55726B1/sr unknown
- 2008-03-06 BR BRPI0808332-0A patent/BRPI0808332A2/pt not_active IP Right Cessation
- 2008-03-06 CN CN200880007116.0A patent/CN101679980B8/zh not_active Expired - Fee Related
- 2008-03-06 LT LTEP08742032.9T patent/LT2132314T/lt unknown
- 2008-03-06 SI SI200831764A patent/SI2132314T1/sl unknown
- 2008-03-06 AR ARP080100918A patent/AR065611A1/es not_active Application Discontinuation
- 2008-03-06 KR KR1020157003616A patent/KR101633727B1/ko not_active Expired - Fee Related
- 2008-03-06 KR KR1020097020320A patent/KR20090127146A/ko not_active Ceased
- 2008-03-06 EA EA200970810A patent/EA020510B1/ru not_active IP Right Cessation
- 2008-03-06 PL PL08742032T patent/PL2132314T3/pl unknown
- 2008-03-06 HR HRP20170323TT patent/HRP20170323T1/hr unknown
- 2008-03-06 PE PE2008000436A patent/PE20081804A1/es not_active Application Discontinuation
- 2008-03-06 KR KR1020127008467A patent/KR101428344B1/ko not_active Expired - Fee Related
- 2008-03-06 AU AU2008223338A patent/AU2008223338C9/en active Active
- 2008-03-06 PE PE2012002108A patent/PE20130615A1/es not_active Application Discontinuation
- 2008-03-06 DK DK08742032.9T patent/DK2132314T3/en active
- 2008-03-06 CL CL200800664A patent/CL2008000664A1/es unknown
-
2009
- 2009-08-27 IL IL200605A patent/IL200605A/en not_active IP Right Cessation
- 2009-10-05 CR CR11054A patent/CR11054A/es not_active Application Discontinuation
-
2011
- 2011-04-05 US US13/080,515 patent/US8716459B2/en active Active
-
2013
- 2013-11-11 PH PH12013502308A patent/PH12013502308A1/en unknown
-
2014
- 2014-03-11 US US14/204,460 patent/US9447165B2/en active Active
-
2015
- 2015-02-24 CR CR20150094A patent/CR20150094A/es unknown
- 2015-10-23 CL CL2015003139A patent/CL2015003139A1/es unknown
-
2016
- 2016-06-02 US US15/171,944 patent/US9809638B2/en active Active
- 2016-10-13 IL IL248322A patent/IL248322A0/en unknown
-
2017
- 2017-02-22 CY CY20171100238T patent/CY1118857T1/el unknown
- 2017-09-25 US US15/714,726 patent/US10407487B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004039948A2 (en) * | 2002-10-25 | 2004-05-13 | Wyeth | Actriib fusion polypeptides and uses therefor |
| US20040223966A1 (en) * | 2002-10-25 | 2004-11-11 | Wolfman Neil M. | ActRIIB fusion polypeptides and uses therefor |
| US20060034831A1 (en) * | 2004-08-12 | 2006-02-16 | Wyeth | Combination therapy for diabetes, obesity and cardiovascular diseases using GDF-8 inhibitors |
| WO2006020884A2 (en) * | 2004-08-12 | 2006-02-23 | Wyeth | Combination therapy for diabetes, obesity, and cardiovascular diseases using gdf-8 inhibitors |
Non-Patent Citations (3)
| Title |
|---|
| Cromwell, M.E.M. et al., The AAPS Journal 8:E572-E579, 2006 |
| Harrison et al., J. Biol. Chem. 279, 28036-28044 (2004) |
| Oh et al., Genes Dev 16:2749-54 (2002) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102015118132A1 (de) | 2015-02-17 | 2016-08-18 | Hyundai Motor Company | Zahnstangeneinheit einer Fahrzeuglenkvorrichtung |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101633727B1 (ko) | 변이체 액티빈 수용체 폴리펩타이드 및 이의 용도 | |
| US8501678B2 (en) | Variant activin receptor polypeptides and uses thereof | |
| US9610327B2 (en) | Variant activin receptor polypeptides, alone or in combination with chemotherapy, and uses thereof | |
| HK1235435A1 (en) | Variant activin receptor polypeptides and uses thereof | |
| HK1139983B (en) | Variant activin receptor polypeptides and uses thereof | |
| HK1139983A (en) | Variant activin receptor polypeptides and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| A201 | Request for examination | ||
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A16-div-PA0104 St.27 status event code: A-0-1-A10-A18-div-PA0104 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
St.27 status event code: A-0-1-A10-A16-div-PA0104 St.27 status event code: A-0-1-A10-A18-div-PA0104 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
Fee payment year number: 1 St.27 status event code: A-2-2-U10-U12-oth-PR1002 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| PC1903 | Unpaid annual fee |
Not in force date: 20190622 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE St.27 status event code: A-4-4-U10-U13-oth-PC1903 |
|
| PC1903 | Unpaid annual fee |
Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20190622 St.27 status event code: N-4-6-H10-H13-oth-PC1903 |