WO2025094002A1 - Lurbinectedin formulation - Google Patents
Lurbinectedin formulation Download PDFInfo
- Publication number
- WO2025094002A1 WO2025094002A1 PCT/IB2024/060468 IB2024060468W WO2025094002A1 WO 2025094002 A1 WO2025094002 A1 WO 2025094002A1 IB 2024060468 W IB2024060468 W IB 2024060468W WO 2025094002 A1 WO2025094002 A1 WO 2025094002A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pharmaceutical formulation
- stable liquid
- injectable pharmaceutical
- liquid ready
- lurbinectedin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4995—Pyrazines or piperazines forming part of bridged ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
Definitions
- the present invention provides a stable, liquid ready -to-use injectable pharmaceutical formulation comprising lurbinectedin.
- the invention further relates to a process for preparing such formulation.
- the invention relates the use of non-aqueous solvents to prepare stable therapeutic formulation by dissolving lurbinectedin in an aprotic solvent(s) without the need for lyophilisation.
- Lurbinectedin is an alkylating agent with the chemical name of (l’R,6R,6aR,7R,13S,14S,16R)-8,14-dihydroxy-6’,9-dimethoxy-4,10,23- trimethyl-19-oxo2’,3’,4’,6,7,9’,12,13,14,16-decahydro-6aH-spiro[7,13-azano- 6,16-(epithiopropanooxymethano)[l,3]dioxolo[7,8] isoquinolino[3,2-b][3] benzazocine -20,l ’-pyrido[3,4-b]indol]-5-yl acetate.
- Lurbinectedin is a member of the ecteinascidin family and binds to guanine residues in the minor groove of DNA, forming adducts and resulting in a bent of the DNA helix towards the major groove.
- the adduct formation triggers a cascade of events that can affect the subsequent activity of DNA binding proteins, including some transcription factors, and DNA repair pathways, resulting in perturbation of the cell cycle and eventual cell death.
- ZEPZELC A® discloses solubility properties of lurbinectedin. It is insoluble or practically insoluble in water. However, its solubility increases at acidic pH.
- ZEPZELC A® has a solubility of 7.2 mg/ml in 0.05M lactic acid and 90.4 mg/ml in 0.5M lactic acid.
- the recommended dosage of ZEPZELC A® is 3.2 mg/m 2 by intravenous infusion over 60 minutes every 21 days until disease progression or unacceptable toxicity.
- Such formulation provides challenges to delivering them in final injectable form such as through further dilution.
- a well- designed formulation must, at a minimum step and less complex method which ensures error free administration.
- these compositions tend to be unstable, with sedimentation and/or precipitation occurring in under 24 hours following rehydration or reconstitution.
- Lurbinectedin is unstable at room temperature and undergoes degradation. Thus, it needs to be stored at 2-8°C and stability improves below -20°C. Thermolabile nature of lurbinectedin make it difficult to handle and process at room temperature.
- US Patent No. 7763615 discloses lurbinectedin as a composition of matter.
- PCT Publication WO2021098992 discloses a pharmaceutical formulation prepared by lyophilizing an aqueous stock solution comprising lurbinectedin, an organic carboxylic acid, sodium hydroxide, and sucrose to produce a lyophilized powder, wherein the concentration of lurbinectedin in the aqueous stock solution is 0.5 mg/ml. It also discloses method of reducing lurbinectedin degradation in a lyophilized formulation. It further discloses the degradation pathway of the lurbinectedin. Lurbinectedin undergoes alkaline hydrolysis in presence of water i.e. deacetylation to produce impurities B, D and G.
- PCT Publication WO2021043949 discloses lurbinectedin or a pharmaceutically acceptable salt or ester thereof for use in the treatment of malignant mesothelioma. It further discloses injection composition that can be prepared by combining lurbinectedin with water, or other physiologically suitable diluent, so as to form a solution. A surfactant can be added to facilitate the formation of a homogeneous solution or suspension, but fails to disclose any example and/or stability.
- PCT Publication WO2021228414 discloses methods for the treatment of SCLC patients by administering therapeutic amounts of lurbinectedin by intravenous infusion. It further discloses Stable lyophilized formulations of lurbinectedin, which stable even after prolonged storage. It further teaches that, lurbinectedin aqueous solution have very limited stability of 14 days with 25 °C / 60% RH conditions.
- PCT Publication WO2024121864 discloses ready-to-use injectable composition of lurbinectedin. It exemplifies aqueous composition of lurbinectedin in polymer based dual or multi-chamber bag. It further discloses few examples with nonaqueous solvents such as ethanol and propylene glycol, but without manufacturing condition and stability data.
- vasodilation may lead to facial flushing and hypotension. Both vasodilation and hypotension can lead to reflex tachycardia. It is further reported in literature that, propylene glycol as a nonaqueous solvent may pose toxicity issues.
- Lurbinectedin is insoluble or practically insoluble in water but its solubility increases at acidic pH. This insolubility and corresponding instability of lurbinectedin in aqueous solution has necessitated lyophilization of bulk solutions, in order to increase the storage stability of the pharmaceutical product.
- the lurbinectedin injectable product required for parenteral administration is currently available only in the form of lyophilized powder (marketed as ZEPZELCA®), which must be reconstituted before administration to the patient.
- the lurbinectedin drug product is presented as a lyophilized powder for solution for infusion with a strength of 4 mg/vial.
- the drug product is reconstituted with water for injection (8 ml) to give a solution of 0.5 mg/ml lurbinectedin prior to use.
- the reconstituted solution is further diluted with 100 mL or 250mL glucose (5%) solution or sodium chloride (0.9%) solutions for infusion.
- the ZEPZELCA® solution can be stored prior to administration for up to 24 hours following reconstitution, including infusion time, at either room temperature/ ambient light or under refrigeration conditions (2°C-8°C).
- a stable liquid ready-to-use injectable pharmaceutical formulation comprising lurbinectedin.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable non-aqueous solvent.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable aprotic polar solvent.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin wherein the formulation is free of water.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvent(s), wherein the formulation may further comprise co-solvent(s), solubilizing agent(s), excipient(s), adjuant(s).
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutical acceptable non-aqueous solvent selected from dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N- dimethylacetamide (DMA), propylene carbonate, aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a Cl 6- 17 fatty alcohol group, and benzyl benzoate, triacetin, polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxy propylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylo
- DMSO dimethylsulfox
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable non-aqueous solvent(s) selected from N,N- dimethylacetamide, glycerol monocaprylocaprate and diethylene glycol monoethyl ether, polyethylene glycol 12-hydroxy stearate.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in N,N- dimethylacetamide.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in glycerol monocaprylocaprate.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in diethylene glycol monoethyl ether.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in polyethylene glycol 12-hydroxy stearate.
- the formulation has a pH from about 1 to about 10. In yet another aspect of the invention, the formulation has a pH from about 1 to about 8.
- the formulation has a pH from about 2 to about 7.
- the formulation has a pH from about 3 to about 5.
- the formulation has a pH from about 3 to about 11.
- the formulation has a pH from about 6 to about 11.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin in concentration from about 0.001% w/v to about 1% w/v.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprises 4 mg lurbinectedin.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprises 0.5 mg/ml lurbinectedin.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin in suitable container such as vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
- suitable container such as vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin for single-dose administration.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin for multi-dose administration.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin wherein formulation manufacturing process temperature is about 2°C to about 8°C.
- a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin, with improved solubility and stability.
- At least 90% purity of the lurbinectedin is retained after storage for 12 months at 2°C to 8°C.
- At least 90% purity of the lurbinectedin is retained after storage for 24 months at 2°C to 8°C.
- At least 90% purity of the lurbinectedin is retained after storage for 36 months at 2°C to 8°C.
- the present inventors while working on liquid, ready-to-use formulations of lurbinectedin, surprisingly found that stable and clear solution of lurbinectedin can be formulated with a suitable non-aqueous solvent.
- the present invention also provides a simple, cost-effective, reproducible and quick method of preparing a stable liquid ready-to-use, injectable pharmaceutical formulation of lurbinectedin.
- a stable liquid ready - to-use injectable pharmaceutical formulation comprising lurbinectedin.
- a "ready-to-use” or “RTU” formulation is a sterile, nonaqueous injectable formulation that is stable and has not been reconstituted from a lyophilizate.
- the RTU formulation prepared after dilution is also a sterile, aqueous or non-aqueous or a combination thereof, injectable formulation that is stable and has been diluted from a concentrated, liquid solution.
- Lurbinectedin refers to Lurbinectedin and the pharmaceutically acceptable salts, solvates, hydrates, co-crystals and anhydrous forms thereof. Lurbinectedin can be crystalline or amorphous.
- a “stable" formulation is defined as no aggregation observed when the said pharmaceutical preparation is kept for stability studies carried out at 2° C to 8° C (Real time study) and 25° C/60% relative humidity (Accelerated study) for at least 12 months and wherein the loss of purity of lurbinectedin would not be more than 5%. It further refers to, lurbinectedin containing composition or formulation having sufficient physical and chemical stability to allow storage at a convenient temperature, such as between about 2° C. and about 8° C., for a commercially reasonable period of time.
- the phrase “physical stability” refers to maintenance of colour or colourless state, dissolved oxygen level, head space oxygen level and particulate matter
- chemical stability relates to formation of drug- related impurities in terms of total impurities, single maximum individual impurity, or maximum individual unknown impurity.
- stability is required for commercially relevant times after manufacturing, such as for about 6, 12, 18, 24, or 36 months, during which time a product is kept in its original packaging under specified storage conditions.
- Ecteinascidins such as lurbinectedin, are complex chemical entities whose stability behaviour in formulations is unpredictable.
- the formulations of the present invention are stable when stored at 2-8°C over a period of at least 12 months.
- shelf life refers to the amount of time the pharmaceutical formulation may be stored without loss of potency and/or performance profile, i.e. formulations that stay within the specification defined herein, upon storage at about 2°C to about 8°C for 12 months, and/or at about -20°C for 6 months.
- the liquid injectable formulation after storage at 2-8° C for 2/3/6/12 months, contains at least 90% by weight of the initial amount of lurbinectedin.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin of the present disclosure minimize the amount of a lurbinectedin degradation product resulting from deacetylation of lurbinectedin (“Impurity D”) when the composition is stored for prolonged times.
- the amount of impurity D present is less than 0.3%, 0.4%, 0.5%, 0.6%, 0.7% or 0.8% w/w of the total lurbinectedin weight in the formulation after prolonged storage at 2° C to 8° C.
- “Pharmaceutically acceptable” ingredient, excipient or component is one that is suitable for use with humans and/or animals without undue adverse side effects (such as toxicity, irritation and allergic response) commensurate with a reasonable benefit/risk ratio.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and composition is free of water.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in aprotic polar solvent.
- aprotic polar solvent refers to a polar solvent which does not contain acidic hydrogen and thus does not act as a hydrogen bond donor.
- aprotic polar solvent system refers to a solution wherein the solvent is a single aprotic polar solvent (for example, neat DMSO, DMA), or a mixture of two or more aprotic polar solvents (for example, a mixture of DMSO and NMP or DMA and glycerol monocaprylocaprate and diethylene glycol monoethyl ether).
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvents, co-solvents, and/or solubilizing agents.
- the formulation may optionally include a co-solvent, while in other embodiment it can include a co-solvent.
- the formulation can include a single/only one biocompatible non-aqueous solvent (i.e., in neat or pure form).
- the formulation includes a mixture of two, three, four, or more biocompatible non-aqueous solvents.
- the formulation can exclude co-solvents, salts, and other ingredients that can help with or increase the solubility of the small molecule drug in the non-aqueous solvent.
- the formulation can consist of or consist essentially of a small molecule drug and a non-aqueous solvent (or mixture of non-aqueous solvents) and still be directly injected through parenteral administration to a subject.
- the formulation of the present invention can be non-aqueous or substantially nonaqueous (e.g., less than 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.5% or less of water by weight or volume).
- the small molecule drug has previously been dried in the presence of a buffer prior to being solubilized in the non-aqueous solvent.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvents, co-solvents, and/or solubilizing agents and at least one pharmaceutically acceptable excipient or adjuvant.
- excipient refers to a natural or synthetic substance formulated alongside the active or therapeutic ingredient (an ingredient that is not the active ingredient) of a medication, included for the purpose of stabilization, or to confer a therapeutic enhancement on the active ingredient in the final dosage form, such as facilitating drug absorption, reducing viscosity, enhancing solubility, adjusting tonicity, mitigating injection site discomfort, depressing the freezing point, or enhancing stability.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin is single-dose.
- a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin is multidose.
- a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin, having improved solubility and stability.
- At least 90% purity of the lurbinectedin is retained after storage for 12 months at 2°C to 8°C. In yet another embodiment of the invention, at least 90% purity of the lurbinectedin is retained after storage for 24 months at 2°C to 8°C.
- At least 90% purity of the lurbinectedin is retained after storage for 36 months at 2°C to 8°C.
- a “single-dose” or “multi-dose” refers to a sterile formulation packed in a container for parenteral administration.
- a single-dose or multi-dose formulation is designed for use with a single/multiple patient/s as a single/multiple injection/s or infusion/s.
- Single-dose or multi-dose formulation may be dispensing in vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin in concentration from about 0.001% w/v to about 1% w/v.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprises 4 mg lurbinectedin.
- a stable liquid ready -to-use, injectable pharmaceutical formulation comprises 0.5 mg/ml lurbinectedin.
- the liquid, the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of O. lmg/ml to 20mg/ml of lurbinectedin.
- the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of 0.5mg/ml to lOmg/ml of lurbinectedin.
- the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of 0.5mg/ml to 4mg/ml of lurbinectedin.
- the liquid, ready -to-use formulation according to present invention possess number of advantages as compared to solutions prepared from sterile powders or lyophilizate immediately before use.
- RTU liquids are less likely to be contaminated by particles or microbes, render the dissolution step superfluous and may be used immediately.
- a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent(s) selected from dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N-dimethylacetamide (DMA), propylene carbonate, aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a C16-17 fatty alcohol group, and benzyl benzoate, triacetin, polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxypropylene glycol (MPEG), glycerol, glycofurol, glycerol monocap
- non-aqueous solvents suitable for the formulations of the present invention include but not limited to alkyl alcohols, polysorbates or polyoxy ethylene ethers, polyethylene glycol ethers, N-methyl-2-pyrrolidone (NMP), monothioglycerol and mixtures thereof.
- examples include aprotic polar solvents, alkyl or aryl benzoate solvents, lipid solvents, protic solvents, or a mixture thereof.
- aprotic solvents include dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N-dimethylacetamide (DMA), propylene carbonate, or mixtures thereof.
- aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a Cl 6- 17 fatty alcohol group, and benzyl benzoate.
- a non-limiting example of a lipid is triacetin, which is the triester of glycerol and acetic acid.
- Non-limiting examples of protic solvents include polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxypropylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylocaprate and diethylene glycol monoethyl ether or mixtures thereof.
- PEG polyethylene glycol
- PG propylene glycol
- PVP polyvinylpyrrolidone
- MPEG methoxypropylene glycol
- glycerol glycofurol
- glycerol monocaprylocaprate diethylene glycol monoethyl ether or mixtures thereof.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent(s) selected from N,N-dimethylacetamide, glycerol monocaprylocaprate and diethylene glycol monoethyl ether, polyethylene glycol 12-hydroxy stearate either alone or in combination thereof.
- a suitable non-aqueous solvent(s) selected from N,N-dimethylacetamide, glycerol monocaprylocaprate and diethylene glycol monoethyl ether, polyethylene glycol 12-hydroxy stearate either alone or in combination thereof.
- a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in N,N-dimethylacetamide. (all four)
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in glycerol monocaprylocaprate.
- a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in diethylene glycol monoethyl ether.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in polyethylene glycol 12-hydroxy stearate.
- the formulation has a pH from about 1 to about 10.
- the formulation has a pH from about
- the formulation has a pH from about
- the formulation has a pH from about
- the formulation has a pH from about 3 to about 11.
- the formulation has a pH from about 6 to about 11.
- the liquid, ready-to-use parenteral pharmaceutical formulation of lurbinectedin may be further admixed with at least lOOmL or 250 mL of diluent (0.9% Sodium Chloride Injection USP or 5% Dextrose Injection USP) prior to actual clinical use.
- Lurbinectedin formulation may be further diluted in 500 ml 0.9% w/v sodium chloride or 5% w/v dextrose.
- the pH of the admixture may be different than the ready-to-use parenteral pharmaceutical formulation of present invention.
- a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin in vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
- Alkyl alcohols include for example, ethanol/anhydrous ethanol/dehydrated alcohol/absolute alcohol, ethylene glycol, propylene glycol, butylene glycol, glycerine or glycerol.
- Polyoxyl ethylene ethers include polysorbate-20 (Tween-20), polysorbate-40 (Tween-40), polysorbate-60 (Tween-60), and polysorbate-80 (Tween-80).
- the polyethylene glycol ethers include polyethoxylated castor oil, such as Cremophor®, polyalkylene glycols, such as polyethylene glycol, polypropylene glycol, and polybutylene glycol, diemthyl acetamide, niacinamide, a diol such as a straight chain, branched or cyclic aliphatic diol, a triol such as straight chain, branched or cyclic aliphatic triol, a polyoxyethylene ether and a polyethylene glycol ether and mixtures thereof.
- polyethoxylated castor oil such as Cremophor®
- polyalkylene glycols such as polyethylene glycol, polypropylene glycol, and polybutylene glycol
- diemthyl acetamide such as a straight chain, branched or cyclic aliphatic diol
- a triol such as straight chain, branched or cyclic aliphatic triol
- formulation of the present invention includes use of acidifying agent such as citric acid.
- the acidifying agents can include citric acid anhydrous, lactic acid, a buffering agent such as citrate, lactate, phosphate, acetate, sulfate and HC1 based buffers.
- the invention includes stable liquid formulations of lurbinectedin wherein excipient is at least one non-aqueous solvent selected from the group comprising ethanol, propylene glycol, polyethylene glycol, N,N- dimethylacetamide, glycerol, polysorbate 20, polysorbate 80, polyethoxylated castor oil or combinations thereof.
- excipient is at least one non-aqueous solvent selected from the group comprising ethanol, propylene glycol, polyethylene glycol, N,N- dimethylacetamide, glycerol, polysorbate 20, polysorbate 80, polyethoxylated castor oil or combinations thereof.
- the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is ethanol. In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is anhydrous ethanol.
- the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is n-methylpyrrolidone.
- the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is N,N-dimethylacetamide.
- the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is propylene glycol.
- the invention includes pharmaceutical stable liquid formulations of lurbinectedin, wherein non aqueous solvent or combination of non-aqueous solvents are present in the formulation in a range from about 10 to 100% by weight.
- the drug is dissolved/solubilized in the stated vehicle/solvent system with or without acidifying/protective agents under nitrogen environment followed by vial filling, stoppering and sealing.
- lurbinectedin soluble in propylene glycol at room temperature and insoluble at 2-8°C.
- it is difficult to manufacture at room temperature.
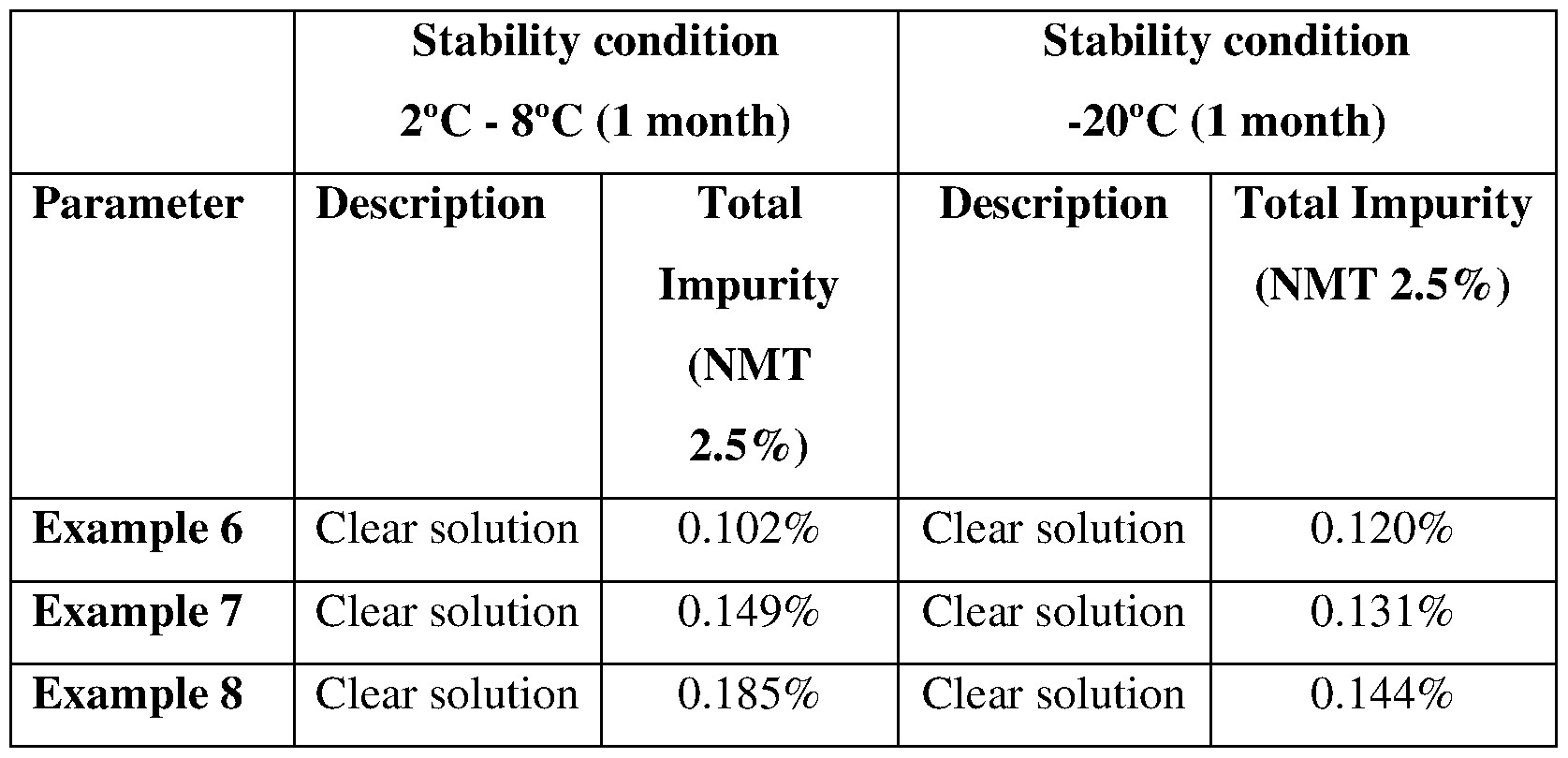
- Stability data of Lurbinectedin in Pharmaceutical formulations of Example 6 to 8, are summarised in Table 3. As can be seen, all Lurbinectedin formulations are stable for 1 Month at 2-8 °C & for 1 Month at 20 °C. Moreover, formulation with N,N- dimethylacetamide is more stable.
- Stability data of lurbinectedin in Pharmaceutical formulations of Example 10 to 18, is summarised in Table 4. As can be seen, PEG and Tween 80 containing formulations i.e. Example 10 and 11 are not stable. In addition to this, formulation of Example 12 and 14 to 15 are not possible.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The present invention provides a stable, liquid ready-to-use injectable pharmaceutical formulation comprising lurbinectedin. The invention further relates to a process for preparing such formulation.
Description
LURBINECTEDIN FORMULATION
FIELD OF THE INVENTION:
The present invention provides a stable, liquid ready -to-use injectable pharmaceutical formulation comprising lurbinectedin. The invention further relates to a process for preparing such formulation. In particular, the invention relates the use of non-aqueous solvents to prepare stable therapeutic formulation by dissolving lurbinectedin in an aprotic solvent(s) without the need for lyophilisation.
BACKGROUND OF THE INVENTION:
Lurbinectedin is an alkylating agent with the chemical name of (l’R,6R,6aR,7R,13S,14S,16R)-8,14-dihydroxy-6’,9-dimethoxy-4,10,23- trimethyl-19-oxo2’,3’,4’,6,7,9’,12,13,14,16-decahydro-6aH-spiro[7,13-azano- 6,16-(epithiopropanooxymethano)[l,3]dioxolo[7,8] isoquinolino[3,2-b][3] benzazocine -20,l ’-pyrido[3,4-b]indol]-5-yl acetate.
Lurbinectedin
Lurbinectedin is a member of the ecteinascidin family and binds to guanine residues in the minor groove of DNA, forming adducts and resulting in a bent of the DNA
helix towards the major groove. The adduct formation triggers a cascade of events that can affect the subsequent activity of DNA binding proteins, including some transcription factors, and DNA repair pathways, resulting in perturbation of the cell cycle and eventual cell death. The US FDA approved lurbinectedin in 2020 as an orphan drug for the treatment of metastatic small cell lung cancer. In addition to this Canadian label of ZEPZELC A® discloses solubility properties of lurbinectedin. It is insoluble or practically insoluble in water. However, its solubility increases at acidic pH. For example, it has a solubility of 7.2 mg/ml in 0.05M lactic acid and 90.4 mg/ml in 0.5M lactic acid. The recommended dosage of ZEPZELC A® is 3.2 mg/m2 by intravenous infusion over 60 minutes every 21 days until disease progression or unacceptable toxicity. Such formulation provides challenges to delivering them in final injectable form such as through further dilution. A well- designed formulation must, at a minimum step and less complex method which ensures error free administration. In addition, these compositions tend to be unstable, with sedimentation and/or precipitation occurring in under 24 hours following rehydration or reconstitution.
Lurbinectedin is unstable at room temperature and undergoes degradation. Thus, it needs to be stored at 2-8°C and stability improves below -20°C. Thermolabile nature of lurbinectedin make it difficult to handle and process at room temperature.
US Patent No. 7763615 discloses lurbinectedin as a composition of matter.
PCT Publication WO2021098992 discloses a pharmaceutical formulation prepared by lyophilizing an aqueous stock solution comprising lurbinectedin, an organic carboxylic acid, sodium hydroxide, and sucrose to produce a lyophilized powder, wherein the concentration of lurbinectedin in the aqueous stock solution is 0.5 mg/ml. It also discloses method of reducing lurbinectedin degradation in a lyophilized formulation. It further discloses the degradation pathway of the lurbinectedin. Lurbinectedin undergoes alkaline hydrolysis in presence of water i.e. deacetylation to produce impurities B, D and G.
PCT Publication WO2021043949 discloses lurbinectedin or a pharmaceutically acceptable salt or ester thereof for use in the treatment of malignant mesothelioma. It further discloses injection composition that can be prepared by combining lurbinectedin with water, or other physiologically suitable diluent, so as to form a solution. A surfactant can be added to facilitate the formation of a homogeneous solution or suspension, but fails to disclose any example and/or stability.
PCT Publication WO2021228414 discloses methods for the treatment of SCLC patients by administering therapeutic amounts of lurbinectedin by intravenous infusion. It further discloses Stable lyophilized formulations of lurbinectedin, which stable even after prolonged storage. It further teaches that, lurbinectedin aqueous solution have very limited stability of 14 days with 25 °C / 60% RH conditions.
PCT Publication WO2024121864 discloses ready-to-use injectable composition of lurbinectedin. It exemplifies aqueous composition of lurbinectedin in polymer based dual or multi-chamber bag. It further discloses few examples with nonaqueous solvents such as ethanol and propylene glycol, but without manufacturing condition and stability data.
It is reported in literature that, ethanol induced vasodilation may lead to facial flushing and hypotension. Both vasodilation and hypotension can lead to reflex tachycardia. It is further reported in literature that, propylene glycol as a nonaqueous solvent may pose toxicity issues.
Lurbinectedin is insoluble or practically insoluble in water but its solubility increases at acidic pH. This insolubility and corresponding instability of lurbinectedin in aqueous solution has necessitated lyophilization of bulk solutions, in order to increase the storage stability of the pharmaceutical product. The lurbinectedin injectable product required for parenteral administration is currently available only in the form of lyophilized powder (marketed as ZEPZELCA®), which must be reconstituted before administration to the patient.
The lurbinectedin drug product is presented as a lyophilized powder for solution for infusion with a strength of 4 mg/vial. The drug product is reconstituted with water for injection (8 ml) to give a solution of 0.5 mg/ml lurbinectedin prior to use. For administration purpose, the reconstituted solution is further diluted with 100 mL or 250mL glucose (5%) solution or sodium chloride (0.9%) solutions for infusion. If not used immediately after reconstitution or dilution, the ZEPZELCA® solution can be stored prior to administration for up to 24 hours following reconstitution, including infusion time, at either room temperature/ ambient light or under refrigeration conditions (2°C-8°C).
The use of such freeze-dried or lyophilized preparations has considerable disadvantages. First of all, the process of preparing these lyophilizates is complicated, time consuming and is cost intensive over the entire lifecycle of the product. Further, the reconstitution requires sterile diluent and results in additional requirement for handling by the healthcare personnel administering the drug and usually every additional step increases the chances of error. Lurbinectedin being a cytotoxic drug, requires special handling and disposal procedures. Thus, the step of reconstitution requires dedicated space for conducting the operation under sterile and laminar conditions, and it increases the risk of the personnel coming in contact with the cytotoxic drug. Furthermore, other errors in the handling of these lyophilizates can lead to serious problems, such as deviation in the concentration of the active substance leading to dosing errors, or microbial contamination of the solution during the reconstitution step. It is also important to note that provision of a lyophilized product means increased oncologic waste to be handled, which increases the burden on the facility handling the drug administration.
In view of the potential of lurbinectedin formulations as antitumoral agents, there is a need to provide a formulation that can solve problems that conventional formulations and manufacturing methodologies possess, or do not completely solve. Thus, there is a need for a stable liquid ready -to-use injectable pharmaceutical formulation of lurbinectedin which avoids reconstitution of the
product by healthcare professionals and obviates the aforementioned shortcomings, namely complexity, time and cost that of currently available lyophilized formulations.
SUMMARY OF THE INVENTION
In one of the aspect of the invention, there is provided a stable liquid ready-to-use injectable pharmaceutical formulation comprising lurbinectedin.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable non-aqueous solvent.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable aprotic polar solvent.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin wherein the formulation is free of water.
In yet another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvent(s), wherein the formulation may further comprise co-solvent(s), solubilizing agent(s), excipient(s), adjuant(s).
In yet another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutical acceptable non-aqueous solvent selected from dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N- dimethylacetamide (DMA), propylene carbonate, aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl
benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a Cl 6- 17 fatty alcohol group, and benzyl benzoate, triacetin, polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxy propylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylocaprate and diethylene glycol monoethyl ether.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in one or more pharmaceutically acceptable non-aqueous solvent(s) selected from N,N- dimethylacetamide, glycerol monocaprylocaprate and diethylene glycol monoethyl ether, polyethylene glycol 12-hydroxy stearate.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in N,N- dimethylacetamide.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in glycerol monocaprylocaprate.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in diethylene glycol monoethyl ether.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in polyethylene glycol 12-hydroxy stearate.
In another aspect of the invention, the formulation has a pH from about 1 to about 10.
In yet another aspect of the invention, the formulation has a pH from about 1 to about 8.
In yet another aspect of the invention, the formulation has a pH from about 2 to about 7.
In yet another aspect of the invention, the formulation has a pH from about 3 to about 5.
In another aspect of the invention, the formulation has a pH from about 3 to about 11.
In another aspect of the invention, the formulation has a pH from about 6 to about 11.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin in concentration from about 0.001% w/v to about 1% w/v.
In yet another aspect of the invention, a stable liquid ready-to-use, injectable pharmaceutical formulation comprises 4 mg lurbinectedin.
In yet another aspect of the invention, a stable liquid ready-to-use, injectable pharmaceutical formulation comprises 0.5 mg/ml lurbinectedin.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin in suitable container such as vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin for single-dose administration.
In yet another aspect of the invention, there is provided a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin for multi-dose administration.
In another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin wherein formulation manufacturing process temperature is about 2°C to about 8°C.
In yet another aspect of the invention, there is provided a stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin, with improved solubility and stability.
In another aspect of the invention, at least 90% purity of the lurbinectedin is retained after storage for 12 months at 2°C to 8°C.
In yet another aspect of the invention, at least 90% purity of the lurbinectedin is retained after storage for 24 months at 2°C to 8°C.
In another aspect of the invention, at least 90% purity of the lurbinectedin is retained after storage for 36 months at 2°C to 8°C.
DETAILED DESCRIPTION OF THE INVENTION
The present inventors while working on liquid, ready-to-use formulations of lurbinectedin, surprisingly found that stable and clear solution of lurbinectedin can be formulated with a suitable non-aqueous solvent. The present invention also provides a simple, cost-effective, reproducible and quick method of preparing a stable liquid ready-to-use, injectable pharmaceutical formulation of lurbinectedin.
In one of the embodiment of the invention, there is provided a stable liquid ready - to-use injectable pharmaceutical formulation comprising lurbinectedin.
As used herein, a "ready-to-use" or "RTU" formulation is a sterile, nonaqueous injectable formulation that is stable and has not been reconstituted from a lyophilizate. The RTU formulation prepared after dilution is also a sterile, aqueous or non-aqueous or a combination thereof, injectable formulation that is stable and has been diluted from a concentrated, liquid solution.
As used herein, “Lurbinectedin” refers to Lurbinectedin and the pharmaceutically acceptable salts, solvates, hydrates, co-crystals and anhydrous forms thereof. Lurbinectedin can be crystalline or amorphous.
As used herein, a "stable" formulation is defined as no aggregation observed when the said pharmaceutical preparation is kept for stability studies carried out at 2° C to 8° C (Real time study) and 25° C/60% relative humidity (Accelerated study) for at least 12 months and wherein the loss of purity of lurbinectedin would not be more than 5%. It further refers to, lurbinectedin containing composition or formulation having sufficient physical and chemical stability to allow storage at a convenient temperature, such as between about 2° C. and about 8° C., for a commercially reasonable period of time. The phrase “physical stability” refers to maintenance of colour or colourless state, dissolved oxygen level, head space oxygen level and particulate matter, and the phrase “chemical stability” relates to formation of drug- related impurities in terms of total impurities, single maximum individual impurity, or maximum individual unknown impurity. For pharmaceutical products, stability is required for commercially relevant times after manufacturing, such as for about 6, 12, 18, 24, or 36 months, during which time a product is kept in its original packaging under specified storage conditions. Ecteinascidins, such as lurbinectedin, are complex chemical entities whose stability behaviour in formulations is unpredictable. The formulations of the present invention are stable when stored at
2-8°C over a period of at least 12 months. The stable formulations provided herein are designed to have shelf life of at least 12 months. The term “shelf life”, as used herein, refers to the amount of time the pharmaceutical formulation may be stored without loss of potency and/or performance profile, i.e. formulations that stay within the specification defined herein, upon storage at about 2°C to about 8°C for 12 months, and/or at about -20°C for 6 months. The liquid injectable formulation, after storage at 2-8° C for 2/3/6/12 months, contains at least 90% by weight of the initial amount of lurbinectedin.
Another embodiment of the invention, a stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin of the present disclosure minimize the amount of a lurbinectedin degradation product resulting from deacetylation of lurbinectedin (“Impurity D”) when the composition is stored for prolonged times.
In yet another embodiment, the amount of impurity D present is less than 0.3%, 0.4%, 0.5%, 0.6%, 0.7% or 0.8% w/w of the total lurbinectedin weight in the formulation after prolonged storage at 2° C to 8° C.
“Pharmaceutically acceptable” ingredient, excipient or component is one that is suitable for use with humans and/or animals without undue adverse side effects (such as toxicity, irritation and allergic response) commensurate with a reasonable benefit/risk ratio.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and composition is free of water.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in aprotic polar solvent.
As used herein, the term “aprotic polar solvent” refers to a polar solvent which does not contain acidic hydrogen and thus does not act as a hydrogen bond donor.
As used herein, the term “aprotic polar solvent system” refers to a solution wherein the solvent is a single aprotic polar solvent (for example, neat DMSO, DMA), or a mixture of two or more aprotic polar solvents (for example, a mixture of DMSO and NMP or DMA and glycerol monocaprylocaprate and diethylene glycol monoethyl ether).
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvents, co-solvents, and/or solubilizing agents.
In certain embodiment, the formulation may optionally include a co-solvent, while in other embodiment it can include a co-solvent. In one instance, the formulation can include a single/only one biocompatible non-aqueous solvent (i.e., in neat or pure form). In other embodiment, the formulation includes a mixture of two, three, four, or more biocompatible non-aqueous solvents. In still additional embodiment, the formulation can exclude co-solvents, salts, and other ingredients that can help with or increase the solubility of the small molecule drug in the non-aqueous solvent. For instance, the formulation can consist of or consist essentially of a small molecule drug and a non-aqueous solvent (or mixture of non-aqueous solvents) and still be directly injected through parenteral administration to a subject. Further, the formulation of the present invention can be non-aqueous or substantially nonaqueous (e.g., less than 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.5% or less of water by weight or volume). In some instances, the small molecule drug has
previously been dried in the presence of a buffer prior to being solubilized in the non-aqueous solvent.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin and one or more pharmaceutically acceptable non-aqueous solvents, co-solvents, and/or solubilizing agents and at least one pharmaceutically acceptable excipient or adjuvant.
The term “excipient” as used herein refers to a natural or synthetic substance formulated alongside the active or therapeutic ingredient (an ingredient that is not the active ingredient) of a medication, included for the purpose of stabilization, or to confer a therapeutic enhancement on the active ingredient in the final dosage form, such as facilitating drug absorption, reducing viscosity, enhancing solubility, adjusting tonicity, mitigating injection site discomfort, depressing the freezing point, or enhancing stability.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin is single-dose.
In yet another embodiment of the invention, there is provided a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin is multidose.
In yet another embodiment of the invention, there is provided a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin, having improved solubility and stability.
In another embodiment of the invention, at least 90% purity of the lurbinectedin is retained after storage for 12 months at 2°C to 8°C.
In yet another embodiment of the invention, at least 90% purity of the lurbinectedin is retained after storage for 24 months at 2°C to 8°C.
In another embodiment of the invention, at least 90% purity of the lurbinectedin is retained after storage for 36 months at 2°C to 8°C.
As used herein, a “single-dose” or “multi-dose” refers to a sterile formulation packed in a container for parenteral administration. A single-dose or multi-dose formulation is designed for use with a single/multiple patient/s as a single/multiple injection/s or infusion/s. Single-dose or multi-dose formulation may be dispensing in vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
Other objects, features and advantages of the present invention will become apparent from the following detailed description. It should be understood, however, that the detailed description and the examples, while indicating specific embodiments of the invention, are given by way of illustration only. Additionally, it is contemplated that changes and modifications within the spirit and scope of the invention will become apparent to those skilled in the art from this detailed description.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin in concentration from about 0.001% w/v to about 1% w/v.
In yet another embodiment of the invention, a stable liquid ready -to-use, injectable pharmaceutical formulation comprises 4 mg lurbinectedin.
In yet another embodiment of the invention, a stable liquid ready -to-use, injectable pharmaceutical formulation comprises 0.5 mg/ml lurbinectedin.
The ready -to-use liquid parenteral formulation as claimed in claim 1, having a concentration of lurbinectedin of from about 0.001% w/v to about 1% w/v.
The liquid, the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of O. lmg/ml to 20mg/ml of lurbinectedin.
In one of the embodiment, the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of 0.5mg/ml to lOmg/ml of lurbinectedin.
In one of the embodiment, the stable liquid ready -to-use, injectable pharmaceutical formulation comprising lurbinectedin may have a concentration of 0.5mg/ml to 4mg/ml of lurbinectedin.
The liquid, ready -to-use formulation according to present invention possess number of advantages as compared to solutions prepared from sterile powders or lyophilizate immediately before use. RTU liquids are less likely to be contaminated by particles or microbes, render the dissolution step superfluous and may be used immediately.
In yet another embodiment of the invention, there is provided a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent(s) selected from dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N-dimethylacetamide (DMA), propylene carbonate, aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a C16-17 fatty alcohol group, and benzyl benzoate, triacetin, polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxypropylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylocaprate and diethylene glycol monoethyl ether or mixtures thereof.
The various non-aqueous solvents suitable for the formulations of the present invention include but not limited to alkyl alcohols, polysorbates or polyoxy ethylene ethers, polyethylene glycol ethers, N-methyl-2-pyrrolidone (NMP), monothioglycerol and mixtures thereof. With respect to the biocompatible nonaqueous solvent, examples include aprotic polar solvents, alkyl or aryl benzoate solvents, lipid solvents, protic solvents, or a mixture thereof. Non-limiting examples of aprotic solvents include dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N-dimethylacetamide (DMA), propylene carbonate, or mixtures thereof. In some instances, however, the formulations of the present invention do not have to include the aforementioned solvents (i.e., others can be used). Non-limiting examples of aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a Cl 6- 17 fatty alcohol group, and benzyl benzoate. A non-limiting example of a lipid is triacetin, which is the triester of glycerol and acetic acid. Non-limiting examples of protic solvents include polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxypropylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylocaprate and diethylene glycol monoethyl ether or mixtures thereof.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in a suitable non-aqueous solvent(s) selected from N,N-dimethylacetamide, glycerol monocaprylocaprate and diethylene glycol monoethyl ether, polyethylene glycol 12-hydroxy stearate either alone or in combination thereof.
In yet another embodiment of the invention, there is provided a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in N,N-dimethylacetamide. (all four)
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in glycerol monocaprylocaprate.
In yet another embodiment of the invention, there is provided a stable liquid ready- to-use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in diethylene glycol monoethyl ether.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin dissolved in polyethylene glycol 12-hydroxy stearate.
In another embodiment of the invention, the formulation has a pH from about 1 to about 10.
In yet another embodiment of the invention, the formulation has a pH from about
1 to about 8.
In yet another embodiment of the invention, the formulation has a pH from about
2 to about 7.
In yet another embodiment of the invention, the formulation has a pH from about
3 to about 5.
In another embodiment of the invention, the formulation has a pH from about 3 to about 11.
In another embodiment of the invention, the formulation has a pH from about 6 to about 11.
The liquid, ready-to-use parenteral pharmaceutical formulation of lurbinectedin may be further admixed with at least lOOmL or 250 mL of diluent (0.9% Sodium Chloride Injection USP or 5% Dextrose Injection USP) prior to actual clinical use. Lurbinectedin formulation may be further diluted in 500 ml 0.9% w/v sodium chloride or 5% w/v dextrose. Thus, it is to be understood that, the pH of the admixture may be different than the ready-to-use parenteral pharmaceutical formulation of present invention.
In another embodiment of the invention, there is provided a stable liquid ready-to- use, injectable pharmaceutical formulation comprising lurbinectedin in vial, ampules, bottles, syringes, intravenous bags or multi chamber bag.
Alkyl alcohols include for example, ethanol/anhydrous ethanol/dehydrated alcohol/absolute alcohol, ethylene glycol, propylene glycol, butylene glycol, glycerine or glycerol. Polyoxyl ethylene ethers include polysorbate-20 (Tween-20), polysorbate-40 (Tween-40), polysorbate-60 (Tween-60), and polysorbate-80 (Tween-80). The polyethylene glycol ethers include polyethoxylated castor oil, such as Cremophor®, polyalkylene glycols, such as polyethylene glycol, polypropylene glycol, and polybutylene glycol, diemthyl acetamide, niacinamide, a diol such as a straight chain, branched or cyclic aliphatic diol, a triol such as straight chain, branched or cyclic aliphatic triol, a polyoxyethylene ether and a polyethylene glycol ether and mixtures thereof.
In addition to non-aqueous solvents, formulation of the present invention includes use of acidifying agent such as citric acid. In alternative embodiments, the acidifying agents can include citric acid anhydrous, lactic acid, a buffering agent such as citrate, lactate, phosphate, acetate, sulfate and HC1 based buffers.
In an embodiment the invention includes stable liquid formulations of lurbinectedin wherein excipient is at least one non-aqueous solvent selected from the group comprising ethanol, propylene glycol, polyethylene glycol, N,N-
dimethylacetamide, glycerol, polysorbate 20, polysorbate 80, polyethoxylated castor oil or combinations thereof.
In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is ethanol. In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is anhydrous ethanol.
In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is n-methylpyrrolidone.
In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is N,N-dimethylacetamide.
In an embodiment the invention relates to stable liquid formulations of lurbinectedin wherein non-aqueous solvent is propylene glycol.
In an embodiment the invention includes pharmaceutical stable liquid formulations of lurbinectedin, wherein non aqueous solvent or combination of non-aqueous solvents are present in the formulation in a range from about 10 to 100% by weight.
The present invention is explained in detail with reference to the following examples described below, which are given for the purpose of illustration only and are not intended to limit the scope of the invention.
Examples 1-9:
Process: The drug is dissolved/solubilized in the stated vehicle/solvent system with or without acidifying/protective agents under nitrogen environment followed by vial filling, stoppering and sealing. As can be seen, lurbinectedin soluble in propylene glycol at room temperature and insoluble at 2-8°C. Moreover, considering lurbinetedin degradation associated with temperature condition, it is difficult to manufacture at room temperature.
Stability studies: Table 2:
and Example 9, are summarised in Table 2. As can be seen, n-methyl-pyrrolidone and polyethylene glycol containing formulations i.e. Example 4, 5 and 9 are not stable.
Stability studies:
Stability data of Lurbinectedin in Pharmaceutical formulations of Example 6 to 8, are summarised in Table 3. As can be seen, all Lurbinectedin formulations are stable for 1 Month at 2-8 °C & for 1 Month at 20 °C. Moreover, formulation with N,N- dimethylacetamide is more stable.
Stability data of lurbinectedin in Pharmaceutical formulations of Example 10 to 18, is summarised in Table 4. As can be seen, PEG and Tween 80 containing formulations i.e. Example 10 and 11 are not stable. In addition to this, formulation of Example 12 and 14 to 15 are not possible.
Examples 19-23:
Stability data of lurbinectedin in Pharmaceutical formulations of Example 19 to 23, are summarised in Table 5. As can be seen, preparation of Example 19 is not possible.
Examples 23-27:
Claims
1. A stable liquid ready -to-use, injectable pharmaceutical formulation, comprising lurbinectedin and at least one of a pharmaceutically acceptable non-aqueous solvent.
2. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the pharmaceutically non-aqueous solvent is selected from dimethylsulfoxide (DMSO), dimethylformamide (DMF), ethyl acetate, N,N- dimethylacetamide (DMA), propylene carbonate, aryl or alkyl benzoate solvents include methyl benzoate, ethyl benzoate, propyl benzoate, C12-C15 alkyl benzoates, in which R is a C12-15 alkyl group, C16-17 alkyl benzoate, in which R is a Cl 6- 17 fatty alcohol group, and benzyl benzoate, triacetin, polyethylene glycol (PEG), propylene glycol (PG), polyvinylpyrrolidone (PVP), methoxy propylene glycol (MPEG), glycerol, glycofurol, glycerol monocaprylocaprate and diethylene glycol monoethyl ether or mixtures thereof.
3. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the pharmaceutically acceptable non-aqueous solvent is N,N- dimethylacetamide.
4. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the composition is free of water.
5. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, having a concentration of lurbinectedin of from about 0.001% w/v to about 1% w/v.
6. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the lurbinectedin is present in an amount of about 0.5mg/ml.
7. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, is having a pH value of about 3 to about 11.
8. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, is having a pH value of about 6 to about 11.
9. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, further comprises one or more pharmaceutically acceptable excipient.
10. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 9, wherein one or more pharmaceutically acceptable excipient is selected from co-solvent, solubilizing agent, adjuant, a pH adjuster.
11. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 1, can be provided in vial, ampule, bottle, syringe, intravenous bag or multi chamber bag.
12. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the formulation has a shelf life of 24 months when stored at about 2°C to about 8°C.
13. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 1, wherein the formulation manufacturing process temperature is about 2°C to about 8°C.
14. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 1, can be administered as single-dose or multi-dose.
15. A stable liquid ready-to-use, injectable pharmaceutical formulation comprising lurbinectedin and N,N-dimethylacetamide.
16. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 15, wherein the composition is free of water.
17. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 15, having a concentration of lurbinectedin of from about 0.001% w/v to about 1% w/v.
18. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, wherein the lurbinectedin is present in an amount of about 0.5mg/ml.
19. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, is having a pH value of about 3 to about 11.
20. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, is having a pH value of about 6 to about 11.
21. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, further comprises one or more pharmaceutically acceptable excipient.
22. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, wherein one or more pharmaceutically acceptable excipient is selected from co-solvent, solubilizing agent, adjuant, a pH adjuster.
23. The stable liquid ready-to-use, injectable pharmaceutical formulation as claimed in claim 15, can be provided in vial, ampule, bottle, syringe, intravenous bag or multi chamber bag.
24. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 15, wherein the composition has a shelf life of 24 months when stored at about 2°C to about 8°C.
25. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 15, wherein the formulation manufacturing process temperature is about 2°C to about 8°C.
26. The stable liquid ready -to-use, injectable pharmaceutical formulation as claimed in claim 15, can be administered as single-dose or multi-dose.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN202321074612 | 2023-11-02 | ||
| IN202321074612 | 2023-11-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025094002A1 true WO2025094002A1 (en) | 2025-05-08 |
Family
ID=95582309
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2024/060468 Pending WO2025094002A1 (en) | 2023-11-02 | 2024-10-24 | Lurbinectedin formulation |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025094002A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021098992A1 (en) * | 2019-11-21 | 2021-05-27 | Pharma Mar, S.A. | Methods of treating small cell lung cancer with lurbinectedin formulations |
| WO2021228414A1 (en) * | 2020-05-14 | 2021-11-18 | Pharma Mar, S.A. | Methods of treating small cell lung cancer with lurbinectedin formulations |
| WO2023084329A1 (en) * | 2021-11-15 | 2023-05-19 | Rk Pharma Inc. | Improved process for the preparation of lurbinectedin and its morphs thereof |
-
2024
- 2024-10-24 WO PCT/IB2024/060468 patent/WO2025094002A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021098992A1 (en) * | 2019-11-21 | 2021-05-27 | Pharma Mar, S.A. | Methods of treating small cell lung cancer with lurbinectedin formulations |
| WO2021228414A1 (en) * | 2020-05-14 | 2021-11-18 | Pharma Mar, S.A. | Methods of treating small cell lung cancer with lurbinectedin formulations |
| WO2023084329A1 (en) * | 2021-11-15 | 2023-05-19 | Rk Pharma Inc. | Improved process for the preparation of lurbinectedin and its morphs thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5670335B2 (en) | Bendamustine liquid formulation | |
| EP0650729B1 (en) | Rapamycin formulations for IV injection | |
| US20070249632A1 (en) | Pharmaceutical compositions and use thereof | |
| WO2009097443A2 (en) | Liquid formulations of compounds active at sulfonylurea receptors | |
| US20190008871A1 (en) | Stable carfilzomib formulations | |
| KR20150085136A (en) | Lyophilized cake formulations | |
| US20250009885A1 (en) | Injectable pharmaceutical formulations of lefamulin | |
| WO2013024495A1 (en) | Pharmaceutical formulations of cabazitaxel | |
| WO2011156481A2 (en) | Formulations including amiodarone and salts thereof and methods of their manufacture and use | |
| WO2016001905A2 (en) | Stable liquid ready-to-use injectable formulation of bortezomib | |
| AU702519B2 (en) | Parenteral pharmaceutical compositions containing GF120918A | |
| US20070249640A1 (en) | Pharmaceutical compositions and use thereof | |
| EP3679925B1 (en) | Pharmaceutical composition of docetaxel conjugate and preparation method | |
| WO2025094002A1 (en) | Lurbinectedin formulation | |
| WO2021090183A1 (en) | Liquid melphalan composition | |
| US20190070136A1 (en) | Parenteral compositions of carmustine | |
| WO2022269572A1 (en) | Pharmaceutical compositions | |
| WO2022091122A1 (en) | Stable remdesivir formulations | |
| WO2019130228A1 (en) | Stable liquid compositions of melphalan | |
| WO2024009319A1 (en) | Liquid injectable compositions of trilaciclib | |
| WO2023214433A1 (en) | Stable parenteral compositions of parecoxib | |
| WO2025165852A1 (en) | Liquid formulations of lurbinectedin | |
| HK40033919A (en) | Pharmaceutical composition of docetaxel conjugate and preparation method | |
| HK40033919B (en) | Pharmaceutical composition of docetaxel conjugate and preparation method | |
| WO2011101865A2 (en) | Stable pharmaceutical compositions of clopidogrel for parenteral delivery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24885099 Country of ref document: EP Kind code of ref document: A1 |