WO2024178304A1 - Kras modulators - Google Patents
Kras modulators Download PDFInfo
- Publication number
- WO2024178304A1 WO2024178304A1 PCT/US2024/017034 US2024017034W WO2024178304A1 WO 2024178304 A1 WO2024178304 A1 WO 2024178304A1 US 2024017034 W US2024017034 W US 2024017034W WO 2024178304 A1 WO2024178304 A1 WO 2024178304A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- pharmaceutically acceptable
- solvate
- acceptable salt
- optionally substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/05—Isotopically modified compounds, e.g. labelled
Definitions
- KRAS Zirsten rat sarcoma viral oncogene homologue
- KRAS protein is a GTPase and involved in cellular signaling such as regulation of cell proliferation.
- KRAS can activate cellular signaling pathways including, but not limited to, the mitogen-activated protein kinase (MAPK) pathway.
- MAPK mitogen-activated protein kinase
- X 1 is N or C-CN
- X 2 is N, C-H, C-F, or C-Cl
- X 3 is N, C-H, C-F, C-Cl, or C-CF3
- X 4 is O, S, or NR 5
- X is O, S, or NR 5
- Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system
- One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof: WSGR Docket No. 62619-720.601 wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, C-Cl, or C-CF 3 ; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R 5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH
- X 3 is N, C-H, C-F, C-Cl, or C-CF 3 ;
- X 4 is O, S, or NR 5 ;
- X is O, S, or NR 5 ;
- Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system;
- R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl;
- each R 5 is hydrogen or optionally substituted C1-C4 alkyl;
- Y is a divalent moiety selected from: (a) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
- One embodiment provides a pharmaceutical composition comprising a compound of Formula (I), (II), (III), or pharmaceutically acceptable salt or solvate thereof, and at least one pharmaceutically acceptable excipient.
- One embodiment provides a method of treating cancer in a patient in need thereof comprising administering to the patient a compound of Formula (I), (II), (III), or pharmaceutically acceptable salt or solvate thereof.
- One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (II), (III), wherein the KRAS protein is contacted in an in vitro setting.
- One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (II), (III), wherein the KRAS protein is contacted in an in vivo setting.
- “Amino” refers to the –NH 2 radical.
- “Cyano” refers to the -CN radical.
- “Nitro” refers to the -NO2 radical.
- “Oxa” refers to the -O- radical.
- Alkyl refers to a straight or branched hydrocarbon chain radical consisting solely of carbon and hydrogen atoms, containing no unsaturation, having from one to fifteen carbon atoms (e.g., C 1 -C 15 alkyl).
- an alkyl comprises one to thirteen carbon atoms (e.g., C1-C13 alkyl).
- an alkyl comprises one to eight carbon atoms (e.g., C1-C8 alkyl).
- an alkyl comprises one to five carbon atoms (e.g., C1-C5 alkyl).
- an alkyl comprises one to four carbon atoms (e.g., C 1 -C 4 alkyl). In other embodiments, an alkyl comprises one to three carbon atoms (e.g., C1-C3 alkyl). In other embodiments, an alkyl comprises one to two carbon atoms (e.g., C 1 -C 2 alkyl). In other embodiments, an alkyl comprises one carbon atom (e.g., C 1 alkyl). In other embodiments, an alkyl comprises five to fifteen carbon atoms (e.g., C5-C15 alkyl). In other embodiments, an alkyl comprises five to eight carbon atoms (e.g., C5-C8 alkyl).
- an alkyl comprises two to five carbon atoms (e.g., C 2 -C 5 alkyl). In other embodiments, an alkyl comprises three to five carbon atoms (e.g., C3-C5 alkyl).
- the alkyl group is selected from methyl, ethyl, 1-propyl (n-propyl), 1-methylethyl (iso-propyl), 1-butyl (n-butyl), 1-methylpropyl (sec- butyl), 2-methylpropyl (iso-butyl), 1,1-dimethylethyl (tert-butyl), 1-pentyl (n-pentyl).
- alkyl is attached to the rest of the molecule by a single bond.
- an alkyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -OR a , -SR a , -OC(O)-R a , -N(R a ) 2 , - C(O)R a , -C(O)OR a , -C(O)N(R a )2, -N(R a )C(O)OR a , -OC(O)-N(R a )2, -N(R a )C(O)R a , -N(R a )S(O)tR a (where t is 1 or 2), -S(O) t OR a (where t is 1 or 2), -S(
- an optionally substituted alkyl is a haloalkyl. In other embodiments, an optionally substituted alkyl is a fluoroalkyl. In other embodiments, an optionally substituted alkyl is a -CF3 group.
- Alkoxy refers to a radical bonded through an oxygen atom of the formula –O-alkyl, where alkyl is an alkyl chain as defined above.
- Alkenyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon double bond, and having from two to twelve carbon atoms.
- an alkenyl comprises two to eight carbon atoms. In other embodiments, an alkenyl comprises two to four carbon atoms.
- the alkenyl is attached to the rest of the molecule by a single bond, for example, ethenyl (i.e., vinyl), prop-1-enyl (i.e., allyl), but-1-enyl, pent-1-enyl, penta-1,4-dienyl, and the like.
- an alkenyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -OR a , -SR a , - OC(O)-R a , -N(R a )2, -C(O)R a , -C(O)OR a , -C(O)N(R a )2, -N(R a )C(O)OR a , -OC(O)-N(R a )2, - N(R a )C(O)R a , -N(R a )S(O)tR a (where t is 1 or 2), -S(O)tOR a (where t is 1 or 2), -S(O)tOR a (where t is 1 or 2), -S(O)
- Alkynyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon triple bond, having from two to twelve carbon atoms.
- an alkynyl comprises two to eight carbon atoms.
- an alkynyl comprises two to six carbon atoms.
- an alkynyl comprises two to four carbon atoms.
- the alkynyl is attached to the rest of the molecule by a single bond, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, and the like.
- an alkynyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, WSGR Docket No.
- Alkylene or “alkylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing no unsaturation, and having from one to twelve carbon atoms, for example, methylene, ethylene, propylene, n-butylene, and the like.
- the alkylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- the points of attachment of the alkylene chain to the rest of the molecule and to the radical group are through one carbon in the alkylene chain or through any two carbons within the chain.
- an alkylene comprises one to eight carbon atoms (e.g., C1-C8 alkylene). In other embodiments, an alkylene comprises one to five carbon atoms (e.g., C1-C5 alkylene). In other embodiments, an alkylene comprises one to four carbon atoms (e.g., C 1 -C 4 alkylene). In other embodiments, an alkylene comprises one to three carbon atoms (e.g., C1-C3 alkylene). In other embodiments, an alkylene comprises one to two carbon atoms (e.g., C 1 -C 2 alkylene). In other embodiments, an alkylene comprises one carbon atom (e.g., C 1 alkylene).
- an alkylene comprises five to eight carbon atoms (e.g., C5-C8 alkylene). In other embodiments, an alkylene comprises two to five carbon atoms (e.g., C2-C5 alkylene). In other embodiments, an alkylene comprises three to five carbon atoms (e.g., C 3 -C 5 alkylene).
- an alkylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -OR a , -SR a , -OC(O)- R a , -N(R a ) 2 , -C(O)R a , -C(O)OR a , -C(O)N(R a ) 2 , -N(R a )C(O)OR a , -OC(O)-N(R a ) 2 , -N(R a )C(O)R a , - N(R a )S(O)tR a (where t is 1 or 2), -S(O)tOR a (where t is 1 or 2), -S(O)tOR a (where t is 1 or 2), -
- alkenylene or “alkenylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon double bond, and having from two to twelve carbon atoms.
- the alkenylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- an alkenylene comprises two to eight carbon atoms (e.g., C2-C8 alkenylene).
- an alkenylene comprises two to five carbon atoms (e.g., C 2 -C 5 alkenylene).
- an alkenylene comprises two to four carbon atoms (e.g., C2-C4 alkenylene). In other embodiments, an alkenylene comprises two to three carbon atoms (e.g., C2-C3 alkenylene). In other embodiments, an alkenylene comprises two carbon atoms (e.g., C 2 alkenylene). In other embodiments, an alkenylene comprises five to eight carbon atoms (e.g., C5-C8 alkenylene). In other embodiments, an alkenylene comprises three to five carbon atoms (e.g., C3-C5 alkenylene).
- an alkenylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -OR a , -SR a , -OC(O)-R a , -N(R a )2, -C(O)R a , - C(O)OR a , -C(O)N(R a ) 2 , -N(R a )C(O)OR a , -OC(O)-N(R a ) 2 , -N(R a )C(O)R a , -N(R a )S(O) t R a (where t is 1 or 2), -S(O) t OR a (where t is 1 or 2), -S(O) t R a (where t is 1 or 2),
- Alkynylene or “alkynylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon triple bond, and having from two to twelve carbon atoms.
- the alkynylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- an alkynylene comprises two to eight carbon atoms (e.g., C2-C8 alkynylene).
- an alkynylene comprises two to five carbon atoms (e.g., C2-C5 alkynylene). In other embodiments, an alkynylene comprises two to four carbon atoms (e.g., C 2 -C 4 alkynylene). In other embodiments, an alkynylene comprises two to three carbon atoms (e.g., C2-C3 alkynylene). In other embodiments, an alkynylene comprises two carbon atoms (e.g., C 2 alkynylene). In other embodiments, an alkynylene comprises five to eight carbon atoms (e.g., C 5 -C 8 alkynylene).
- an alkynylene comprises three to five carbon atoms (e.g., C3-C5 alkynylene).
- an alkynylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -OR a , -SR a , -OC(O)-R a , -N(R a ) 2 , -C(O)R a , - C(O)OR a , -C(O)N(R a )2, -N(R a )C(O)OR a , -OC(O)-N(R a )2, -N(R a )C(O)R a , -N(R a )S(O)tR a (where t is 1 or
- Aryl refers to a radical derived from an aromatic monocyclic or multicyclic hydrocarbon ring system by removing a hydrogen atom from a ring carbon atom.
- the aromatic monocyclic or multicyclic hydrocarbon ring system contains only hydrogen and carbon from five to eighteen carbon atoms, where at least one of the rings in the ring system is fully unsaturated, i.e., it contains a cyclic, delocalized (4n+2) –electron system in accordance with the Hückel theory.
- aryl groups include, but are not limited to, groups such as benzene, fluorene, indane, indene, tetralin and naphthalene.
- aryl or the prefix “ar-“ (such as in “aralkyl”) is meant to include aryl radicals optionally substituted by one or more substituents independently selected from optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, halo, cyano, nitro, - R b -OR a , -R b -OC(O)-R a , -R b -OC(O)-OR a , -R b -OC(O)-N(R a )2, -R b -N(R a )2, -R b -C(O)R a , -R b
- “Aralkyl” refers to a radical of the formula -R c -aryl where R c is an alkylene chain as defined above, for example, methylene, ethylene, and the like.
- the alkylene chain part of the aralkyl radical is optionally substituted as described above for an alkylene chain.
- the aryl part of the aralkyl radical is optionally substituted as described above for an aryl group.
- “Aralkenyl” refers to a radical of the formula –R d -aryl where R d is an alkenylene chain as defined above.
- the aryl part of the aralkenyl radical is optionally substituted as described above for an aryl group.
- alkenylene chain part of the aralkenyl radical is optionally substituted as defined above for an alkenylene group.
- “Aralkynyl” refers to a radical of the formula -R e -aryl, where R e is an alkynylene chain as defined above.
- the aryl part of the aralkynyl radical is optionally substituted as described above for an aryl group.
- the alkynylene chain part of the aralkynyl radical is optionally substituted as defined above for an alkynylene chain.
- Alkoxy refers to a radical bonded through an oxygen atom of the formula -O-R c -aryl where R c is an alkylene chain as defined above, for example, methylene, ethylene, and the like.
- the alkylene chain part of the aralkyl radical is optionally substituted as described above for an alkylene chain.
- the aryl part of the aralkyl radical is optionally substituted as described above for an aryl group.
- Carbocyclyl refers to a stable non-aromatic monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, having from three to fifteen carbon atoms.
- a carbocyclyl comprises three to ten carbon atoms.
- a carbocyclyl comprises five to seven carbon atoms.
- the carbocyclyl is attached to the rest of the molecule by a single bond. Carbocyclyl is saturated (i.e., containing single C-C bonds only) or unsaturated (i.e., containing one or more double bonds or triple bonds).
- a fully saturated carbocyclyl radical is also referred to as “cycloalkyl.”
- monocyclic cycloalkyls include, e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl.
- An unsaturated carbocyclyl is also referred to as “cycloalkenyl.”
- Examples of monocyclic cycloalkenyls include, e.g., cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl.
- Polycyclic carbocyclyl radicals include, for example, adamantyl, norbornyl (i.e., bicyclo[2.2.1]heptanyl), norbornenyl, decalinyl, 7,7-dimethyl-bicyclo[2.2.1]heptanyl, and the like.
- carbocyclyl is meant to include carbocyclyl radicals that are optionally substituted by one or more substituents independently selected from optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted exocyclic alkylidene, halo, oxo, thioxo, cyano, nitro, -R b -OR a , -R b - OC(O)-R a , -R b -OC(O)-OR a , -R b -OC(O)-N(R a ) 2 , -R b -N(R a ) 2 , -R b -C(O)R a , -R b -C(O)OR a , -R b - C(O)N(R a )2, -R b -O-
- each R b is independently a direct bond or a straight or branched alkylene or alkenylene chain
- R c is a straight or branched alkylene or alkenylene chain
- each of the R a , R b , or R c substituents is unsubstituted unless otherwise indicated.
- “Carbocyclylalkyl” refers to a radical of the formula –R c -carbocyclyl where R c is an alkylene chain as defined above. The alkylene chain and the carbocyclyl radical is optionally substituted as defined above.
- Carbocyclylalkynyl refers to a radical of the formula –R c -carbocyclyl where R c is an alkynylene chain as defined above. The alkynylene chain and the carbocyclyl radical is optionally substituted as defined above.
- Carbocyclylalkoxy refers to a radical bonded through an oxygen atom of the formula –O- R c -carbocyclyl where R c is an alkylene chain as defined above. The alkylene chain and the carbocyclyl radical is optionally substituted as defined above.
- Halo or “halogen” refers to bromo, chloro, fluoro or iodo substituents.
- Fluoroalkyl refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, as defined above, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, 1-fluoromethyl-2-fluoroethyl, and the like.
- the alkyl part of the fluoroalkyl radical is optionally substituted as defined above for an alkyl group.
- Heterocyclyl refers to a stable 3- to 18-membered non-aromatic ring radical that comprises two to twelve carbon atoms and from one to six heteroatoms selected from nitrogen, oxygen and sulfur. Unless stated otherwise specifically in the specification, the heterocyclyl radical is a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which optionally includes fused or bridged ring systems. The heteroatoms in the heterocyclyl radical are optionally oxidized. One or more nitrogen atoms, if present, are optionally quaternized. The heterocyclyl radical is partially or fully saturated. The heterocyclyl is attached to the rest of the molecule through any atom of the ring(s).
- heterocyclyl radicals include, but are not limited to, dioxolanyl, thienyl[1,3]dithianyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isothiazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydropyranyl, thiomorpholinyl, thiamorpholinyl, 1-oxo-thio
- heterocyclyl is meant to include heterocyclyl radicals as defined above that are optionally substituted by one or more substituents selected from optionally substituted alkyl, optionally WSGR Docket No.
- N-heterocyclyl or “N-attached heterocyclyl” refers to a heterocyclyl radical as defined above containing at least one nitrogen and where the point of attachment of the heterocyclyl radical to the rest of the molecule is through a nitrogen atom in the heterocyclyl radical.
- An N-heterocyclyl radical is optionally substituted as described above for heterocyclyl radicals. Examples of such N-heterocyclyl radicals include, but are not limited to, 1-morpholinyl, 1- piperidinyl, 1-piperazinyl, 1-pyrrolidinyl, pyrazolidinyl, and imidazolidinyl.
- C-heterocyclyl or “C-attached heterocyclyl” refers to a heterocyclyl radical as defined above containing at least one heteroatom and where the point of attachment of the heterocyclyl radical to the rest of the molecule is through a carbon atom in the heterocyclyl radical.
- a C-heterocyclyl radical is optionally substituted as described above for heterocyclyl radicals. Examples of such C-heterocyclyl radicals include, but are not limited to, 2-morpholinyl, 2- or 3- or 4-piperidinyl, 2-piperazinyl, 2- or 3-pyrrolidinyl, and the like.
- Heterocyclylalkyl refers to a radical of the formula –R c -heterocyclyl where R c is an alkylene chain as defined above. If the heterocyclyl is a nitrogen-containing heterocyclyl, the heterocyclyl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heterocyclylalkyl radical is optionally substituted as defined above for an alkylene chain.
- the WSGR Docket No. 62619-720.601 heterocyclyl part of the heterocyclylalkyl radical is optionally substituted as defined above for a heterocyclyl group.
- Heterocyclylalkoxy refers to a radical bonded through an oxygen atom of the formula –O- R c -heterocyclyl where R c is an alkylene chain as defined above. If the heterocyclyl is a nitrogen-containing heterocyclyl, the heterocyclyl is optionally attached to the alkyl radical at the nitrogen atom.
- the alkylene chain of the heterocyclylalkoxy radical is optionally substituted as defined above for an alkylene chain.
- the heterocyclyl part of the heterocyclylalkoxy radical is optionally substituted as defined above for a heterocyclyl group.
- Heteroaryl refers to a radical derived from a 3- to 18-membered aromatic ring radical that comprises two to seventeen carbon atoms and from one to six heteroatoms selected from nitrogen, oxygen, and sulfur.
- the heteroaryl radical is a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, wherein at least one of the rings in the ring system is fully unsaturated, i.e., it contains a cyclic, delocalized (4n+2) –electron system in accordance with the Hückel theory.
- Heteroaryl includes fused or bridged ring systems.
- the heteroatom(s) in the heteroaryl radical is optionally oxidized.
- heteroaryl is attached to the rest of the molecule through any atom of the ring(s).
- heteroaryls include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzindolyl, 1,3-benzodioxolyl, benzofuranyl, benzooxazolyl, benzo[d]thiazolyl, benzothiadiazolyl, benzo[b][1,4]dioxepinyl, benzo[b][1,4]oxazinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzoxazolyl, benzodioxolyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothienyl (benzothion
- quinolinyl isoquinolinyl, tetrahydroquinolinyl, 5,6,7,8-tetrahydroquinazolinyl, 5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidinyl, 6,7,8,9-tetrahydro-5H-cyclohepta[4,5]thieno[2,3-d]pyrimidinyl, 5,6,7,8-tetrahydropyrido[4,5-c]pyridazinyl, thiazolyl, thiadiazolyl, triazolyl, tetrazolyl, triazinyl, thieno[2,3-d]pyrimidinyl, thieno[3,2-d]pyrimidinyl, thieno[2,3-c]pridinyl, and thiophenyl (i.e.
- heteroaryl is meant to include heteroaryl radicals as defined above which are optionally substituted by one or more substituents selected from optionally substituted alkyl, optionally substituted cycloalkylalkyl, optionally substituted heterocyclylalkyl, optionally substituted alkenyl, optionally substituted alkynyl, halo, optionally substituted fluoroalkyl, optionally substituted haloalkenyl, optionally substituted haloalkynyl, oxo, thioxo, cyano, nitro, -R b -OR a , -R b -OC(O)-R a , -R b -OC(O)-OR a , -R b - OC(O)-N(R a ) 2 , -R b -N(R a ) 2 , -R b -N(R a ) 2 , -R b -N(R
- N-heteroaryl refers to a heteroaryl radical as defined above containing at least one nitrogen and where the point of attachment of the heteroaryl radical to the rest of the molecule is through a nitrogen atom in the heteroaryl radical.
- An N-heteroaryl radical is optionally substituted as described above for heteroaryl radicals.
- C-heteroaryl refers to a heteroaryl radical as defined above and where the point of attachment of the heteroaryl radical to the rest of the molecule is through a carbon atom in the WSGR Docket No. 62619-720.601 heteroaryl radical.
- a C-heteroaryl radical is optionally substituted as described above for heteroaryl radicals.
- Heteroarylalkyl refers to a radical of the formula –R c -heteroaryl, where R c is an alkylene chain as defined above. If the heteroaryl is a nitrogen-containing heteroaryl, the heteroaryl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heteroarylalkyl radical is optionally substituted as defined above for an alkylene chain. The heteroaryl part of the heteroarylalkyl radical is optionally substituted as defined above for a heteroaryl group.
- Heteroarylalkoxy refers to a radical bonded through an oxygen atom of the formula –O- R c -heteroaryl, where R c is an alkylene chain as defined above. If the heteroaryl is a nitrogen-containing heteroaryl, the heteroaryl is optionally attached to the alkyl radical at the nitrogen atom.
- the alkylene chain of the heteroarylalkoxy radical is optionally substituted as defined above for an alkylene chain.
- the heteroaryl part of the heteroarylalkoxy radical is optionally substituted as defined above for a heteroaryl group.
- the compounds disclosed herein in some embodiments, contain one or more asymmetric centers and thus give rise to enantiomers, diastereomers, and other stereoisomeric forms that are defined, in terms of absolute stereochemistry, as (R)- or (S)-. Unless stated otherwise, it is intended that all stereoisomeric forms of the compounds disclosed herein are contemplated by this disclosure. When the compounds described herein contain alkene double bonds, and unless specified otherwise, it is intended that this disclosure includes both E and Z geometric isomers (e.g., cis or trans.) Likewise, all possible isomers, as well as their racemic and optically pure forms, and all tautomeric forms are also intended to be included.
- geometric isomer refers to E or Z geometric isomers (e.g., cis or trans) of an alkene double bond.
- positional isomer refers to structural isomers around a central ring, such as ortho-, meta-, and para- isomers around a benzene ring.
- a “tautomer” refers to a molecule wherein a proton shift from one atom of a molecule to another atom of the same molecule is possible.
- tautomeric equilibrium includes: WSGR Docket No. 62619-720.601
- the compounds disclosed herein are used in different enriched isotopic forms, e.g., enriched in the content of 2 H, 3 H, 11 C, 13 C and/or 14 C.
- the compound is deuterated in at least one position.
- deuterated forms can be made by the procedure described in, for example, U.S. Patent Nos.5,846,514 and 6,334,997. As described in U.S.
- Patent Nos.5,846,514 and 6,334,997 deuteration can, in some instances, improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs.
- structures depicted herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13 C- or 14 C-enriched carbon are within the scope of the present disclosure.
- the compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds.
- the compounds may be labeled with isotopes, such as for example, deuterium ( 2 H), tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C).
- isotopes such as for example, deuterium ( 2 H), tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C).
- Isotopic substitution with 2 H, 11 C, 13 C, 14 C, 15 C, 12 N, 13 N, 15 N, 16 N, 16 O, 17 O, 14 F, 15 F, 16 F, 17 F, 18 F, 33 S, 34 S, 35 S, 36 S, 35 Cl, 37 Cl, 79 Br, 81 Br, 125 I are all contemplated.
- isotopic substitution with 18 F is contemplated. All isotopic variations of the compounds of the present invention, whether radioactive or not, are encompassed within the scope of the present invention.
- the compounds disclosed herein have some or all of the 1 H atoms replaced with 2 H atoms.
- the methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
- WSGR Docket No. 62619-720.601 [0068] Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S.
- Deuterium-transfer reagents suitable for use in nucleophilic substitution reactions are readily available and may be employed to transfer a deuterium- substituted carbon atom under nucleophilic substitution reaction conditions to the reaction substrate.
- CD3I is illustrated, by way of example only, in the reaction schemes below.
- Deuterium-transfer reagents, such as lithium aluminum deuteride (LiAlD4) are employed to transfer deuterium under reducing conditions to the reaction substrate.
- LiAlD4 is illustrated, by way of example only, in the reaction schemes below.
- the compounds disclosed herein contain one deuterium atom. In another embodiment, the compounds disclosed herein contain two deuterium atoms. In another embodiment, the compounds disclosed herein contain three deuterium atoms. In another embodiment, the compounds disclosed herein contain four deuterium atoms. In another embodiment, the compounds disclosed herein contain five deuterium atoms. In another embodiment, the compounds disclosed herein contain six deuterium atoms.
- the compounds disclosed herein contain more than six deuterium atoms. In another embodiment, the compound disclosed herein is fully substituted with deuterium atoms and contains no non-exchangeable 1 H hydrogen atoms. In one embodiment, the level of deuterium incorporation is determined by synthetic methods in which a deuterated synthetic building block is used as a starting material.
- “Pharmaceutically acceptable salt” includes both acid and base addition salts. A pharmaceutically acceptable salt of any one of the KRAS inhibitory compounds described herein is intended to encompass any and all pharmaceutically suitable salt forms. Preferred pharmaceutically acceptable salts of the compounds described herein are pharmaceutically acceptable acid addition salts and pharmaceutically acceptable base addition salts.
- “Pharmaceutically acceptable acid addition salt” refers to those salts which retain the biological effectiveness and properties of the free bases, which are not biologically or otherwise undesirable, and which are formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, hydroiodic acid, hydrofluoric acid, phosphorous acid, and the like. Also included are salts that are formed with organic acids such as aliphatic mono- and dicarboxylic acids, phenyl-substituted alkanoic acids, hydroxy alkanoic acids, alkanedioic acids, aromatic acids, aliphatic and. Aromatic sulfonic acids, etc.
- acetic acid trifluoroacetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid, salicylic acid, and the like.
- Exemplary salts thus include sulfates, pyrosulfates, bisulfates, sulfites, bisulfites, nitrates, phosphates, monohydrogenphosphates, dihydrogenphosphates, metaphosphates, pyrophosphates, chlorides, bromides, iodides, acetates, trifluoroacetates, propionates, caprylates, isobutyrates, oxalates, malonates, succinate suberates, sebacates, fumarates, maleates, mandelates, benzoates, chlorobenzoates, methylbenzoates, dinitrobenzoates, phthalates, benzenesulfonates, toluenesulfonates, phenylacetates, citrates, lactates, malates, tartrates, methanesulfonates, and the like.
- salts of amino acids such as arginates, gluconates, and galacturonates
- Acid addition salts of basic compounds are, in some embodiments, prepared by contacting the free base forms with a sufficient amount of the desired acid to produce the salt according to methods and techniques with which a skilled artisan is familiar.
- “Pharmaceutically acceptable base addition salt” refers to those salts that retain the biological effectiveness and properties of the free acids, which are not biologically or otherwise undesirable.
- salts are prepared from addition of an inorganic base or an organic base to the free acid.
- Pharmaceutically acceptable base addition salts are, in some embodiments, formed with metals or amines, such as alkali and alkaline earth metals or organic amines.
- Salts derived from inorganic bases include, but are not limited to, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum salts and the like.
- Salts derived from organic bases include, but are not limited to, salts of primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines and basic ion exchange resins, for example, isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, ethanolamine, diethanolamine, 2-dimethylaminoethanol, 2-diethylaminoethanol, dicyclohexylamine, lysine, arginine, histidine, caffeine, procaine, N,N-dibenzylethylenediamine, chloroprocaine, hydrabamine, choline, betaine, ethylenediamine, ethylenedianiline, N- methylglucamine, glucosamine, methylglucamine, theobromine, purines, piperazine, piperidine, N-ethylpiperidine, polyamine resins and the like.
- solvates refers to a composition of matter that is the solvent addition form.
- solvates contain either stoichiometric or non-stoichiometric amounts of a solvent, and are formed during the process of making with pharmaceutically acceptable solvents such as water, ethanol, and the like. Hydrates are formed when the solvent is water, or alcoholates are formed when the solvent is alcohol. Solvates of compounds described herein are conveniently prepared or formed during the processes described herein. The compounds provided herein exist in either unsolvated or solvated forms.
- the term “subject” or “patient” encompasses mammals.
- mammals include, but are not limited to, any member of the Mammalian class: humans, non-human primates such as chimpanzees, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like.
- the mammal is a human.
- “treatment” or “treating,” or “palliating” or “ameliorating” are used interchangeably. These terms refer to an approach for obtaining beneficial or desired results WSGR Docket No.
- compositions are, in some embodiments, administered to a patient at risk of developing a particular disease, or to a patient reporting one or more of the physiological symptoms of a disease, even though a diagnosis of this disease has not been made.
- Ras proteins are important for activating signaling networks for controlling cell differentiation, proliferation, and survival, encoded by three genes HRAS, KRAS, and NRAS. The three genes share significant sequence homology and largely overlapping functions. Activation of RAS is facilitated by guanine nucleotide exchange factors (GEF), and activation causes conformational changes.
- GEF guanine nucleotide exchange factors
- the KRAS gene encodes two highly related protein isoforms, KRAS-4A and KRAS-4B, which comprise of 189 and 188 amino acids.
- KRAS generally refers to KRAS-4B, because of the high level of mRNA encoding KRAS-4B in cells.
- KRAS has two major domains, the catalytic G domain and a hypervariable region (HVR).
- HVR hypervariable region
- KRAS G domain is the basis of biological function of GTPase proteins.
- the G domain comprises 6 beta-strands of the protein core, surrounded by five alpha-helices, and comprises residues 1-166.
- the G domain also consists of other regions: switch I, switch II, and the P loop.
- KRAS-GTP binding alters the conformation of the switches I and II in the G domain. When activated, KRAS binds to its downstream molecules as monomers or dimers to mediate series of signaling cascades.
- KRAS also has a flexible C-terminal, the hypervariable region (HVR), which is important for localizing KRAS to the membrane.
- HVR hypervariable region
- the RAS family comprises three isoforms, but about 85% of RAS-related cancers are caused by mutations in the KRAS isoform.
- the mutations in KRAS isoform occurs most frequently in solid tumors such as colorectal carcinoma, lung adenocarcinoma, and pancreatic ductal carcinoma.
- nearly 80% of KRAS mutant tumors are located within codon 12, with the most common mutations being p.G12D, p.G12V, and p.G12C.
- KRAS protein functions as a molecular switch in growth factor signaling pathways by regulating proliferation by alternating between a GDP-bound inactive form and a GTP-bound WSGR Docket No. 62619-720.601 active form.
- the GTP-bound active form is capable of engaging downstream effector proteins to trigger a pro-proliferative response. This regulation cycle is impaired by mutations in codon 12 which disrupts association of GTPase activating proteins, which impairs the inactivation of KRAS, which leads to accumulation of the pro-proliferative form.
- EGF epidermal growth factor
- PDGF platelet-derived growth factor
- FGF fibroblast growth factors
- Upstream regulation can promote binding of GTP and KRAS, converting KRAS from an inactive to an active state.
- Molecules upstream of KRAS mainly mediate the activation or inactivation of KRAS by regulating guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs) (L. Huang et al., Signal Transduction and Targeted Therapy, 2021, 6, 386).
- GEFs guanine nucleotide exchange factors
- GAPs GTPase-activating proteins
- SHP2 Src homology phosphatase 2
- SHP2 is a common signaling regulatory that mediates receptor tyrosine kinases signals to KRAS-ERK signaling, and dephosphorylation substrates of SHP2 have been shown to promote KRAS activation.
- the RAF-MEK-ERK pathway is a downstream target of KRAS signaling.
- Another pathway KRAS involved in is the PI3K-AKT-mTOR pathway (L. Huang et al., Signal Transduction and Targeted Therapy, 2021, 6, 386).
- KRAS was previously considered to be an undruggable protein, but recently there have been advances in targeting codon 12, and specifically in G12C inhibitors. Many efforts have been focused on indirectly targeting KRAS, so there remains an unmet need of targeting KRAS, which the compounds provided herein fulfill. With the discovery of a new allosteric site of KRAS, G12C, several covalently binding inhibitors of KRAS have emerged and are under clinical investigation. However, KRAS inhibition is a complex issue with a lack of understanding of the underlying principles, and there still remains an unmet need for new inhibitors which target other KRAS mutations such as, but not limited to G12D and G12V.
- KRAS mutations are frequently found in colorectal cancer, pancreatic cancer, and non- small cell lung cancer (M.H. Hofmann et al., Cancer Discov 2022; 12:924-37).
- the KRAS allelic distribution varies between the tumor types, with G12C mutations in 13.6% of lung adenocarcinomas, whereas the G12D and G12V mutations are most common in colorectal and pancreatic cancer.
- the G12D, G12V, and G12C mutations are the three most frequent allele mutations.
- KRAS mutations, especially at codon 12 is strongly associated with cellular KRAS dependency, indicating that KRAS acts as an oncogenic driver.
- KRAS G12C inhibitors such as sotorasib (AMG510) and adagrasib (MRTX849). Sotorasib is the first to be approved for clinical use. Both inhibitors rely on the interaction with the nucleophilic cysteine 12 in the GDP state and occupy the switch II pocket.
- KRAS inhibitory compounds are KRAS inhibitory compounds.
- One embodiment provides a compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, C-Cl, or C-CF 3 ; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R 5 is hydrogen or optionally
- One embodiment provides a compound having the structure of Formula (Ia), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; WSGR Docket No.
- X 3 is N, C-H, C-F, or C-Cl;
- X 4 is O, S, or NR 5 ;
- X is O, S, or NR 5 ;
- Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system;
- R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl;
- each R 5 is hydrogen or optionally substituted C1-C4 alkyl;
- Y is a divalent moiety selected from: (a) –(CH2)m-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) -(CH 2 )p-O-(CH 2 )q-, wherein up to 4
- One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH 2 )m-. [0095] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)-. [0096] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2CH2)-.
- One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH 2 )p-O-(CH 2 )q-. [0098] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH 2 )-O-(CH 2 )-. [0099] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -( CH2CH2)-O-(CH2)-.
- One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH 2 )-O-(CH 2 CH 2 )-.
- One embodiment provides a compound having the structure of Formula (Ib), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, C-Cl, or C-CF 3 ; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; WSGR Docket
- each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R 5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from –(CH2)m-, or –(CH2)p-O-(CH2)q-; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; p is 1-4; and q is 1-4.
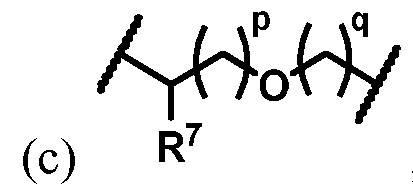
- One embodiment provides a compound having the structure of Formula (Ic), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, or C-Cl; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R 5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from –(CH2)m-, or –(CH2)p-O
- One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH 2 )m-. [0104] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH 2 )-. WSGR Docket No. 62619-720.601 [0105] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2CH2)-.
- One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH 2 )p-O-(CH 2 )q-. [0107] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2)-. [0108] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2CH2)-O-(CH2)-.
- One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH 2 )-O-(CH 2 CH 2 )-.
- One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, C-Cl, or C-CF 3 ; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected
- R 3 is hydrogen, R 2 is alkyl, R 4 is alkyl, and R 2 and R 4 join to form a ring; or R 2 is hydrogen, R 3 is alkyl, R 4 is alkyl, and R 3 and R 4 join to form a ring; or R 2 is hydrogen, and R 3 and R 4 join to form a -CH2OCH2- group; or R 2 is hydrogen, and R 3 and R 4 join to form an -O- group; and n is 0, 1, 2, 3, 4, 5, or 6.
- One embodiment provides a compound having the structure of Formula (IIa), or a pharmaceutically acceptable salt or solvate thereof: wherein: X 1 is N or C-CN; X 2 is N, C-H, C-F, or C-Cl; X 3 is N, C-H, C-F, or C-Cl; X 4 is O, S, or NR 5 ; X is O, S, or NR 5 ; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R 1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene, ; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R 5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH 2 -, or -CD 2 -; R 3 is hydrogen, R
- One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein W is O. [0113] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein W is -CH2-, or -CD2-. WSGR Docket No. 62619-720.601 [0114] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R 2 and R 4 join together to form a - CH2CH2-.
- One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R 3 and R 4 join together to form a - CH2CH2-.
- One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R 3 and R 4 join together to form a - CH2CH2CH2-.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0119] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
- One embodiment provides the compound wherein W is oxo.
- One embodiment provides the compound wherein W is oximo.
- One embodiment provides the compound wherein W is optionally substituted alkyl oximo.
- One embodiment provides the compound wherein W is O-methyl oximo.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 7 is an optionally substituted C1 alkyl.
- One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein p is 1 or 2; and q is 1 or 2. [0128] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 1 is N. [0129] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 1 is C-CN.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 2 is N.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 2 is C-H, C-F, or C-Cl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 3 is N.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 3 is C-H, C-F, or C-Cl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 3 is C-CF 3 .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X 4 is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R is hydrogen.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R is methyl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 0.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 1.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 2.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted aryl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is an optionally substituted naphthyl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted naphthyl is an optionally substituted 1-naphthyl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted 1-naphthyl is further substituted at the 8-position.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is described by Formula (a): wherein: R 12 is hydrogen; R 13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R 14 is hydrogen; R 15 is hydrogen or F; R 16 is hydrogen or F; R 17 is fluorine or -CN; and R 18 3, -OCD3, -OCH2F, or -OCD2F.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: WSGR Docket No. 62619-720.601 [0144] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: [0145] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: [0147]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is: .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: [0150]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted heteroaryl.
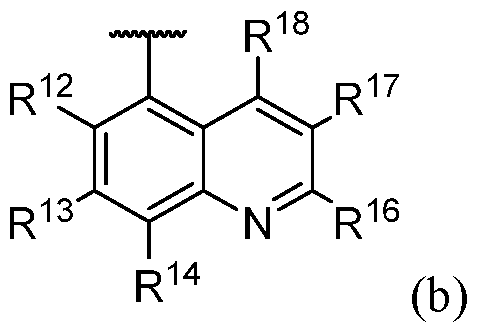
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is described by Formula (b): wherein: R 12 is hydrogen; R 13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R 14 is hydrogen; R 16 is hydrogen or F; R 17 is fluorine or -CN; and R 18 3 , -OCD 3 , -OCH 2 F, or -OCD 2 F.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from: WSGR Docket No. 62619-720.601 .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from: [0155]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is an optionally substituted phenyl.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted phenyl is substituted with an -OH group at the 3-position.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
- WSGR Docket No. 62619-720.601 One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: .
- WSGR Docket No. 62619-720.601 One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: [0162]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is a bond.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1-C4 alkylene.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1 alkylene.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III),or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is an optionally substituted 5- to 10-membered heterocyclyl.
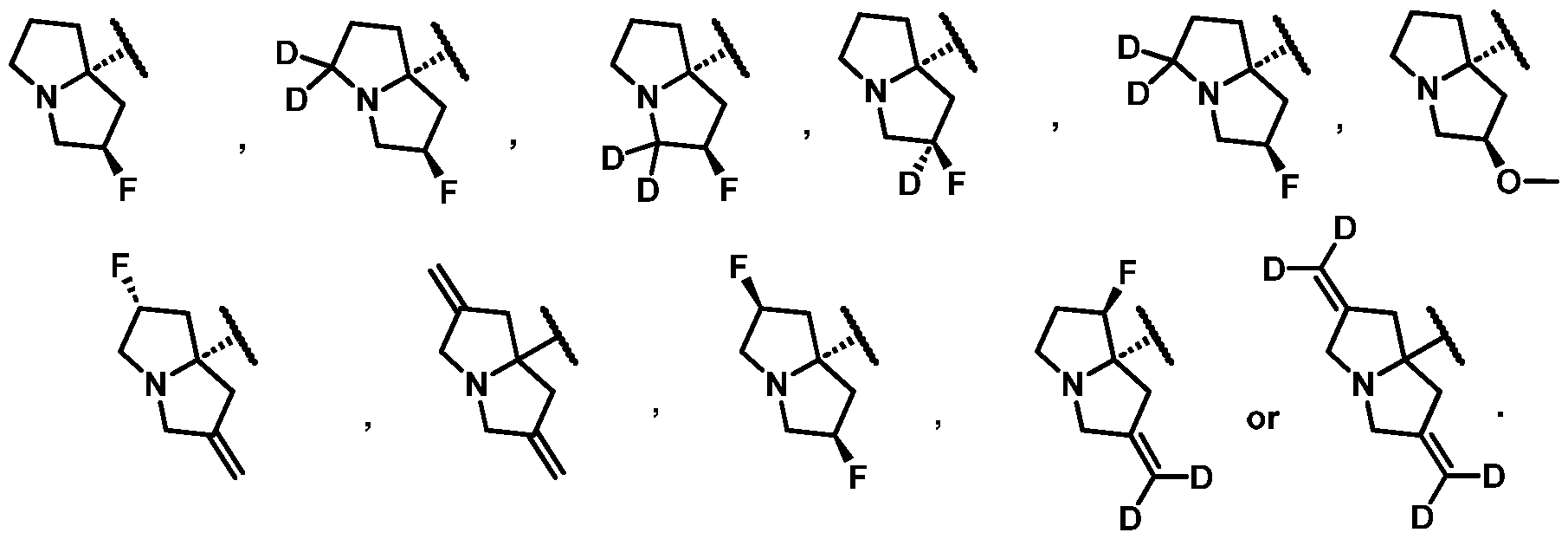
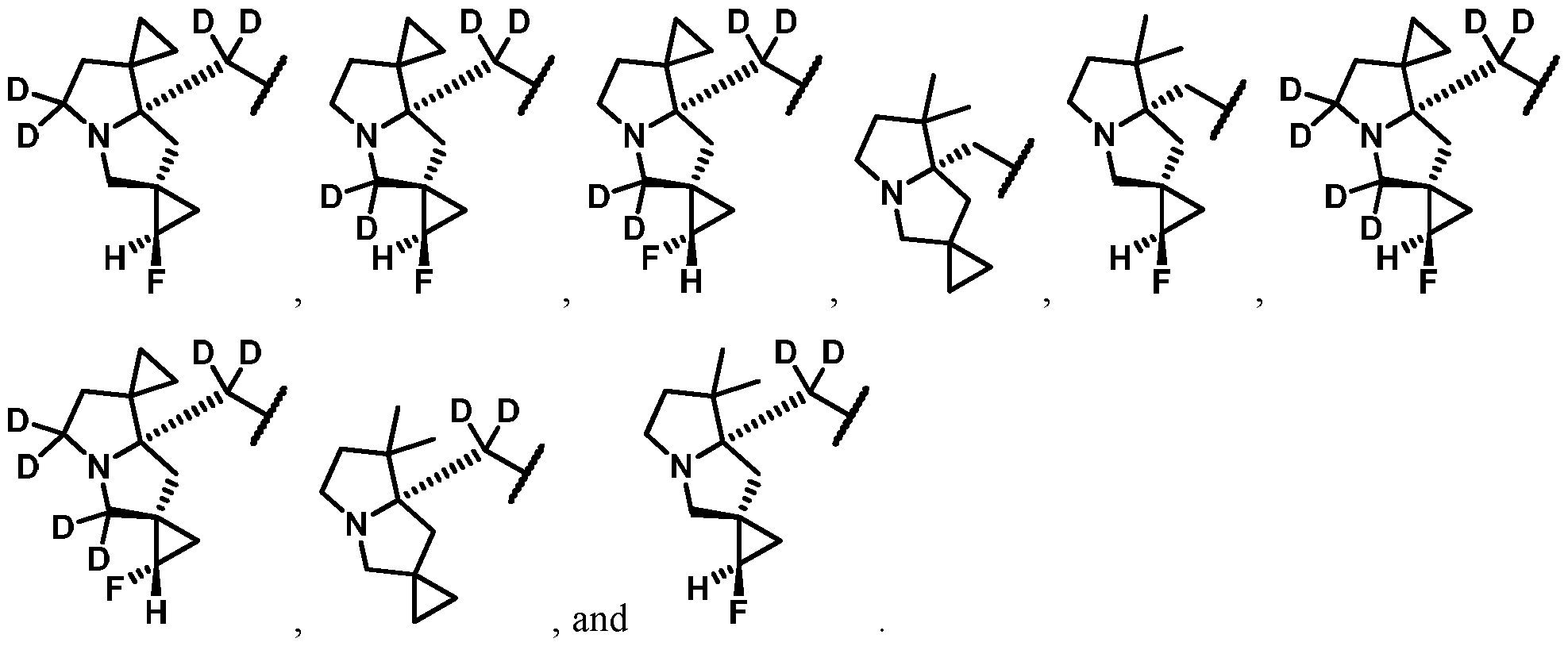
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: WSGR Docket No. 62619-720.601 [0171] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: .
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (c): wherein, each R 20 -R 30 is independently selected from hydrogen or deuterium.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (d): wherein, R 31 is selected from hydrogen, F, Cl, -CN, -OH, or optionally substituted C1-C4 alkyl; R 32 is hydrogen, deuterium or optionally substituted C1-C4 alkyl; and R 33 is hydrogen, deuterium, or F.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: [0176]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
- [0177] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: [0179]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: [0180]
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0182] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: . Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. WSGR Docket No.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0184] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: , WSGR Docket No. 62619-720.601 .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: , .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III),or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- WSGR Docket No. 62619-720.601 One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: WSGR Docket No. 62619-720.601 embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- R 1 is selected from: , .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: WSGR Docket No. 62619-720.601 .
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: , , , , , , . provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- R 1 is selected from: WSGR Docket No.
- Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
- One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R 1 is selected from: [0197]
- One embodiment provides a compound of the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof, having a structure represented in Fig.1.
- One embodiment provides a compound of the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof, having a structure represented in Fig.2.
- One embodiment provides a compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof, WSGR Docket No. 62619-720.601 Formula (I), wherein the portion of the structure of Formula (I) enclosed within the box is provided in Fig.3, Fig.4, Fig.5, or Fig.6.
- One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof, Formula (II), wherein the portion of the structure of Formula (II) enclosed within the box is provided in Fig.7, Fig.8, Fig.9, or Fig.10.
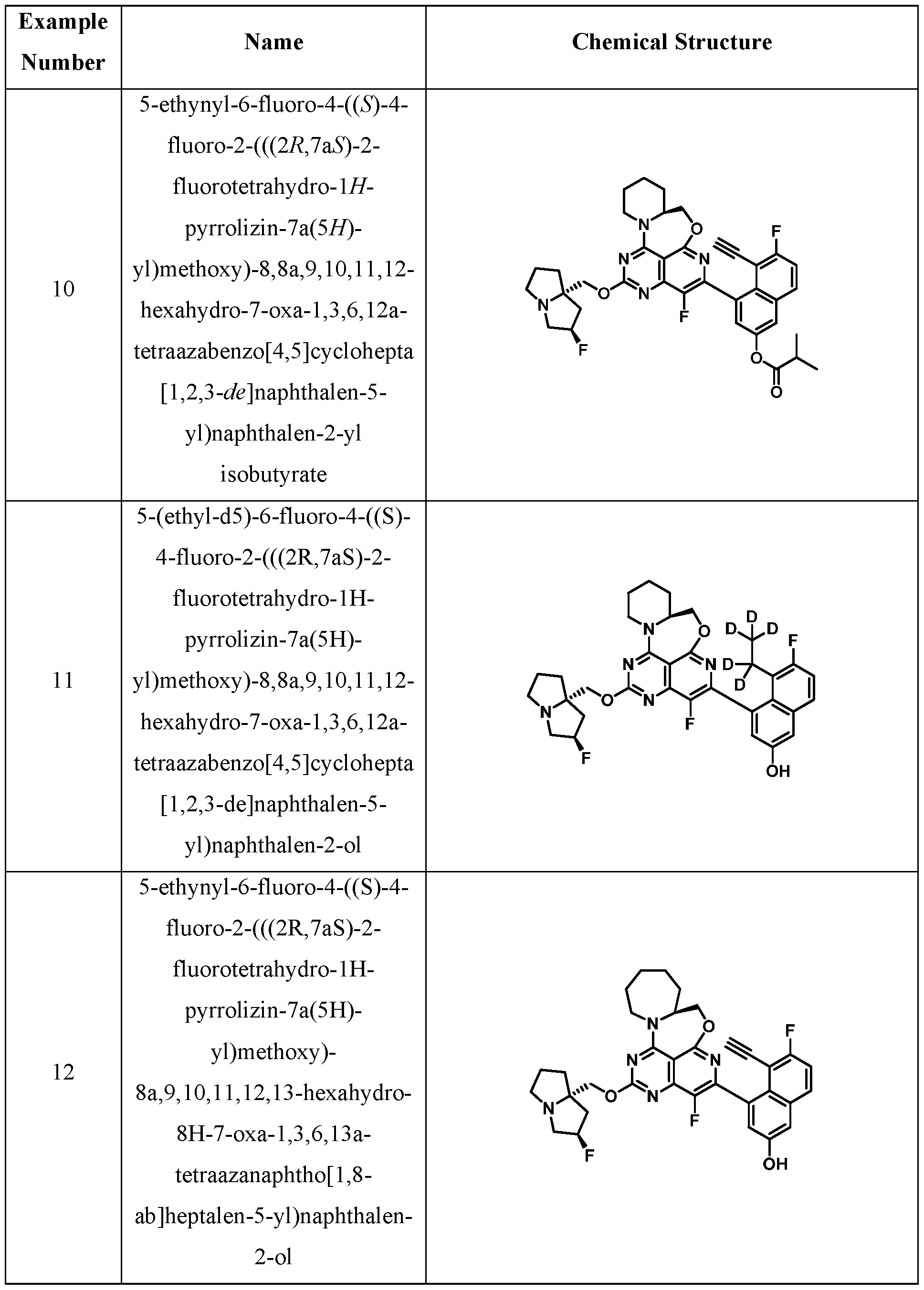
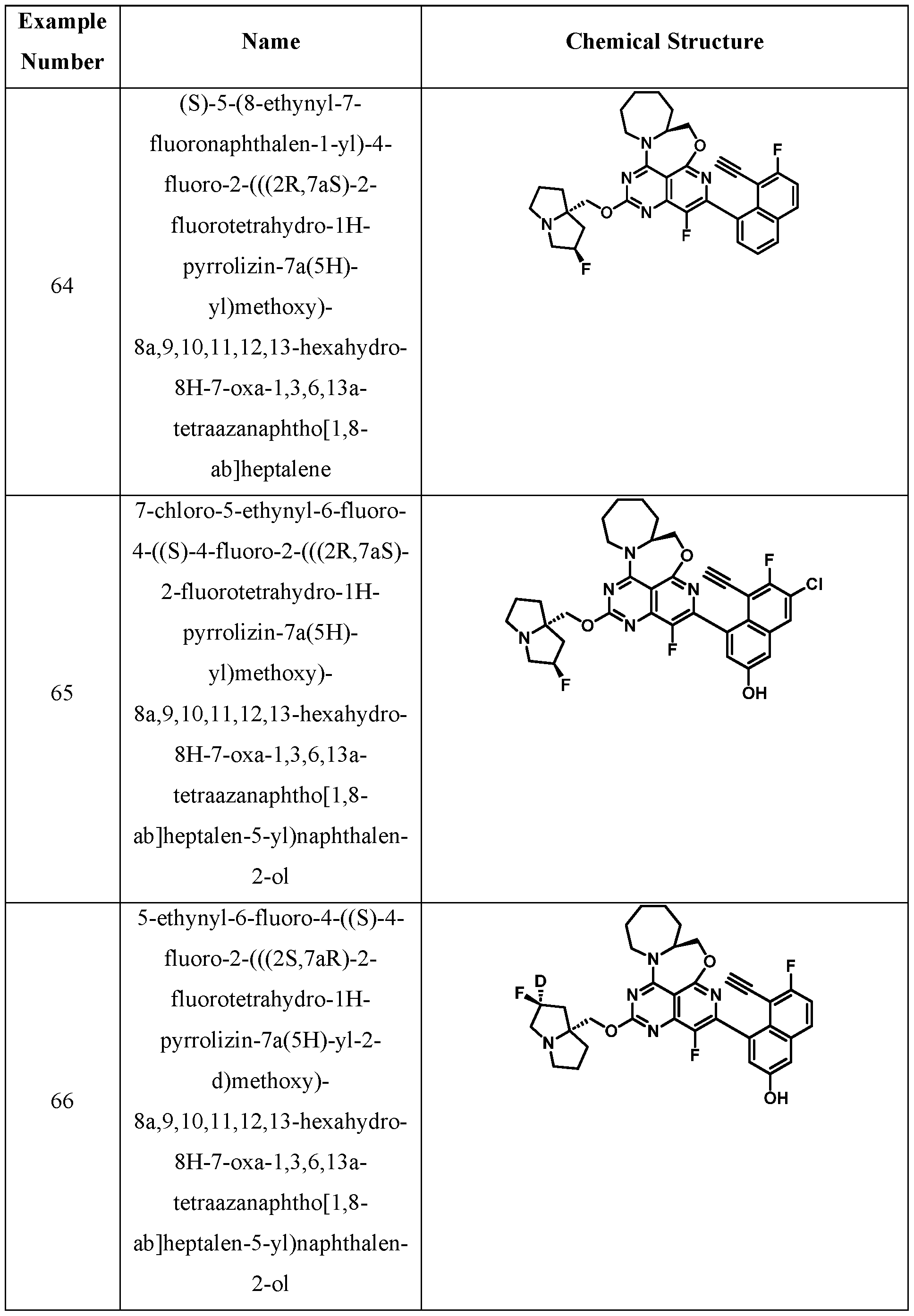
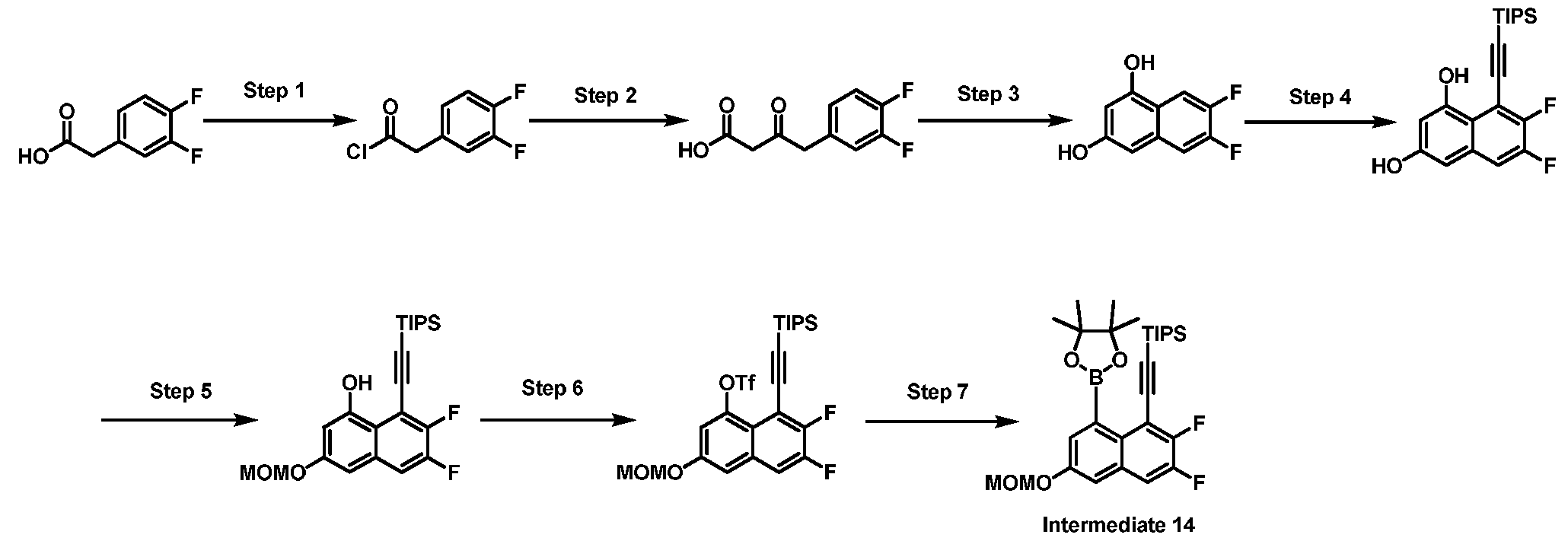
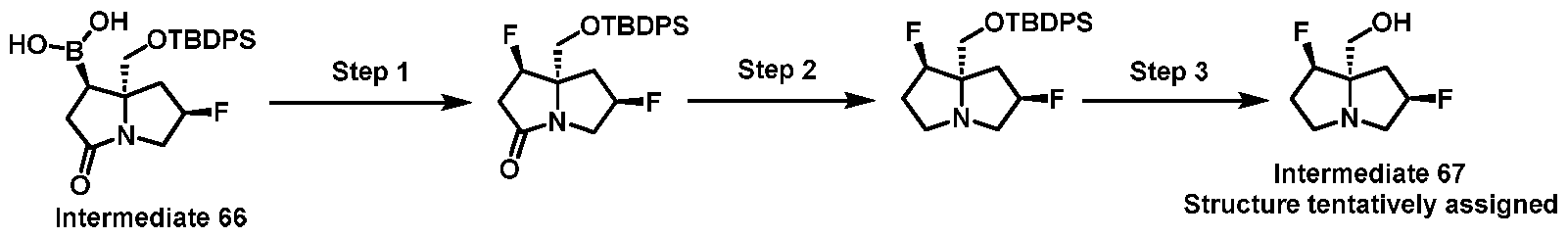
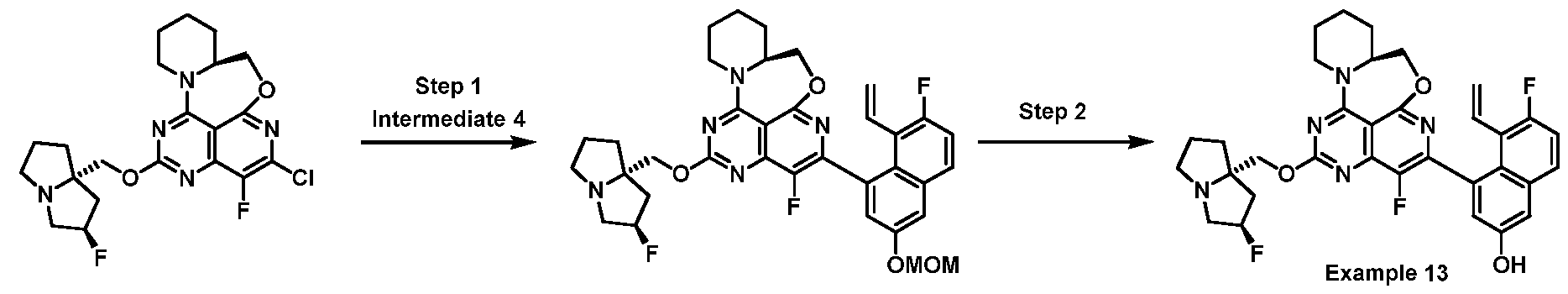
- One embodiment provides a KRAS inhibitory compound, or a pharmaceutically acceptable salt or solvate thereof, having a structure presented in Table 1.
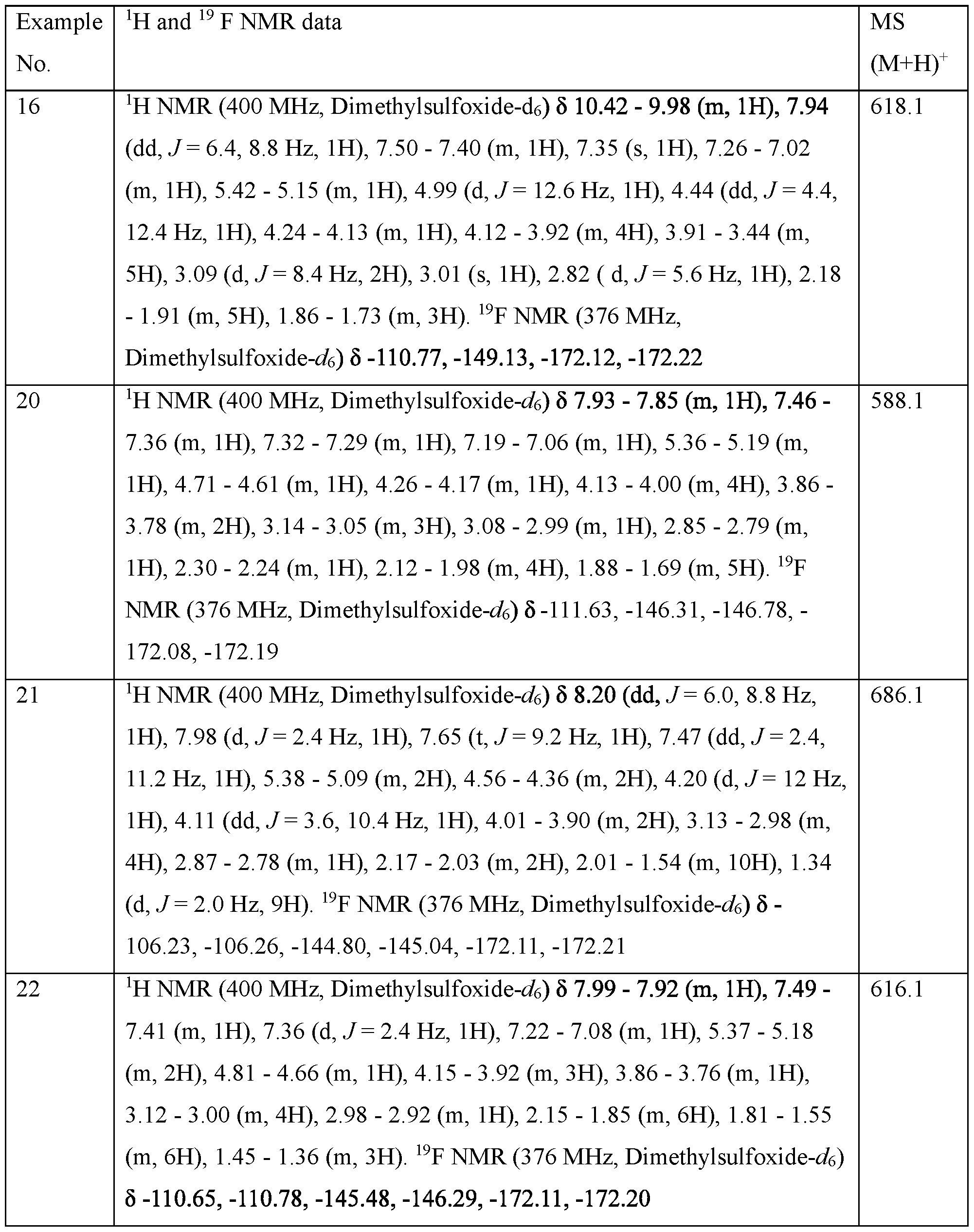
- WSGR Docket No. 62619-720.601 Table 1 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-
- WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601
- WSGR Docket No. 62619-720.601 WSGR Docket No. 62619-720.601
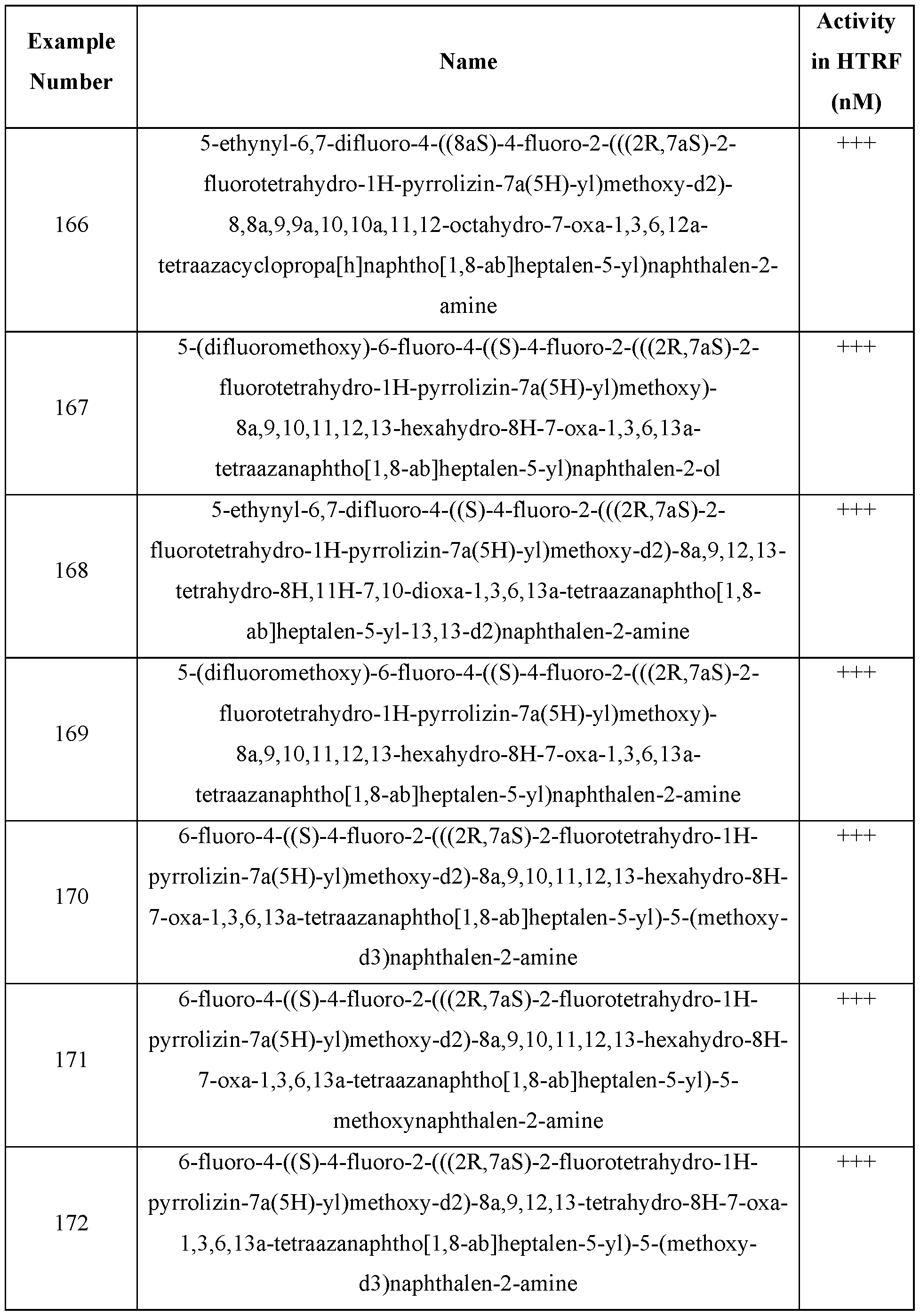
- One embodiment provides a KRAS inhibitory compound, or a pharmaceutically acceptable salt or solvate thereof, having a structure presented in Table 2.
- Table 2 WSGR Docket No. 62619-720.601
- WSGR Docket No. 62619-720.601 Preparation of Compounds [0203]
- the compounds used in the synthetic chemistry reactions described herein are made according to organic synthesis techniques known to those skilled in this art, starting from commercially available chemicals and/or from compounds described in the chemical literature.
- “Commercially available chemicals” are obtained from standard commercial sources including Acros Organics (Pittsburgh, PA), Aldrich Chemical (Milwaukee, WI, including Sigma Chemical and Fluka), Apin Chemicals Ltd. (Milton Park, UK), Avocado Research (Lancashire, U.K.), BDH Inc. (Toronto, Canada), Bionet (Cornwall, U.K.), Chemservice Inc. (West Chester, PA), Crescent Chemical Co. (Hauppauge, NY), Eastman Organic Chemicals, Eastman Kodak Company (Rochester, NY), Fisher Scientific Co. (Pittsburgh, PA), Fisons Chemicals (Leicestershire, UK), Frontier Scientific (Logan, UT), ICN Biomedicals, Inc.
- Suitable reference books and treatise that detail the synthesis of reactants useful in the preparation of compounds described herein, or provide references to articles that describe the preparation include for example, “Synthetic Organic Chemistry”, John Wiley & Sons, Inc., New York; S. R. Sandler et al., “Organic Functional Group Preparations,” 2 nd Ed., Academic Press, New York, 1983; H. O. House, “Modern Synthetic Reactions”, 2 nd Ed., W. A. Benjamin, Inc. Menlo WSGR Docket No. 62619-720.601 Park, Calif.1972; T. L.
- the KRAS inhibitory compound described herein is administered as a pure chemical.
- the KRAS inhibitory compound described herein is combined with a pharmaceutically suitable or acceptable carrier (also referred to herein as a pharmaceutically suitable (or acceptable) excipient, physiologically suitable (or acceptable) WSGR Docket No. 62619-720.601 excipient, or physiologically suitable (or acceptable) carrier) selected on the basis of a chosen route of administration and standard pharmaceutical practice as described, for example, in Remington: The Science and Practice of Pharmacy (Gennaro, 21 st Ed. Mack Pub. Co., Easton, PA (2005)).
- a pharmaceutical composition comprising at least one KRAS inhibitory compound as described herein, or a stereoisomer, pharmaceutically acceptable salt, hydrate, or solvate thereof, together with one or more pharmaceutically acceptable carriers.
- the carrier(s) or excipient(s)
- the carrier(s) is acceptable or suitable if the carrier is compatible with the other ingredients of the composition and not deleterious to the recipient (i.e., the subject or the patient) of the composition.
- One embodiment provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof.
- One embodiment provides a method of preparing a pharmaceutical composition comprising mixing a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable carrier.
- the KRAS inhibitory compound as described by Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof is substantially pure, in that it contains less than about 5%, or less than about 2%, or less than about 1%, or less than about 0.5%, or less than about 0.1%, of other organic small molecules, such as unreacted intermediates or synthesis by-products that are created, for example, in one or more of the steps of a synthesis method.
- a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof.
- One embodiment provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of any of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof.
- One embodiment provides a method of preparing a pharmaceutical composition comprising mixing a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable carrier.
- the KRAS inhibitory compound as described by Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof is substantially pure, in that it contains less than about 5%, or less than about 2%, or less than about 1%, or less than about 0.5%, or less than about 0.1%, of other organic small molecules, such as unreacted intermediates or synthesis by- products that are created, for example, in one or more of the steps of a synthesis method.
- Suitable oral dosage forms include, for example, tablets, pills, sachets, or capsules of hard or soft gelatin, methylcellulose or of another suitable material easily dissolved in the digestive tract.
- suitable nontoxic solid carriers include, for example, pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, talcum, cellulose, glucose, sucrose, magnesium carbonate, and the like.
- pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, talcum, cellulose, glucose, sucrose, magnesium carbonate, and the like See, e.g., Remington: The Science and Practice of Pharmacy (Gennaro, 21 st Ed. Mack Pub. Co., Easton, PA (2005)).
- the KRAS inhibitory compound as described by Formula (I), or Table 1 or Table 2, or pharmaceutically acceptable salt or solvate thereof is formulated for administration by injection.
- the injection formulation is an aqueous formulation.
- the injection formulation is a non-aqueous formulation.
- the injection formulation is an oil-based formulation, such as sesame oil, or the like.
- the dose of the composition comprising at least one KRAS inhibitory compound as described herein differs depending upon the subject or patient's (e.g., human) condition. In some embodiments, such factors include general health status, age, and other factors.
- Pharmaceutical compositions are administered in a manner appropriate to the disease to be treated (or prevented). An appropriate dose and a suitable duration and frequency of administration will be determined by such factors as the condition of the patient, the type and severity of the patient's disease, the particular form of the active ingredient, and the method of administration.
- an appropriate dose and treatment regimen provides the composition(s) in an amount sufficient to provide therapeutic and/or prophylactic benefit (e.g., an improved clinical outcome, such as more frequent complete or partial remissions, or longer disease-free and/or overall survival, or a lessening of symptom severity.
- Optimal doses are generally determined using experimental models and/or clinical trials. The optimal dose depends upon the body mass, weight, or blood volume of the patient. [0218] Oral doses typically range from about 1.0 mg to about 1000 mg, one to four times, or more, per day.
- One embodiment provides a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of the human or animal body.
- One embodiment provides a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treating cancer.
- WSGR Docket No. 62619-720.601 Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer.
- One embodiment provides a pharmaceutical composition comprising a compound of Formula (I) or (II), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient.
- One embodiment provides a use of a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, in the manufacture of a medicament for the treatment of cancer.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a pharmaceutical composition comprising a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient.
- Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer.
- One embodiment provides a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of the human or animal body.
- One embodiment provides a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treating cancer.
- Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer.
- One embodiment provides a pharmaceutical composition comprising a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient.
- One embodiment provides a use of a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, in the manufacture of a medicament for the treatment of cancer.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a pharmaceutical composition comprising a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate WSGR Docket No. 62619-720.601 thereof, and a pharmaceutically acceptable excipient.
- Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a compound of any one of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof.
- a method of treating cancer in a patient in need thereof comprising administering to the patient a pharmaceutical composition comprising a compound of any one of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient.
- Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer.
- One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or Table 1 or Table 2. Another embodiment provides the method of inhibiting KRAS protein activity, wherein the KRAS protein is contacted in an in vivo setting. Another embodiment provides the method of inhibiting KRAS protein activity, wherein the KRAS protein is contacted in an in vitro setting.
- Step 2 2,6-Dichloro-3-fluoro-5-iodopyridin-4-amine
- 2-iodopyrrolidine-2,5-dione 112 g, 497 mmol
- 4-methylbenzenesulfonic acid monohydrate 3.94 g, 20.7 mmol
- the reaction mixture was stirred at 70 °C for 4 h.
- the reaction mixture was quenched with Na2SO3 (200 mL) at 20 °C and extracted with EtOAc (500 mL x 3).
- Step 3 Ethyl 4-amino-2,6-dichloro-5-fluoronicotinate WSGR Docket No. 62619-720.601 [0240] To a mixture of 2,6-dichloro-3-fluoro-5-iodo-pyridin-4-amine (42 g, 137 mmol), TEA (66. 7 mL, 479 mmol) and dichloropalladiumtriphenylphosphane (4.80 g, 6.84 mmol) in EtOH (1050 mL) was purged with CO three times, and the mixture was stirred under CO (1.5 Mpa) at 100 °C for 40 h in a 2000 mL of autoclave.
- Step 5 5,7-Dichloro-8-fluoropyrido[4,3-d]pyrimidine-2,4(1H,3H)-dione
- ethyl 2,6-dichloro-5-fluoro-4-(3-(2,2,2-trichloroacetyl)ureido)nicotinate 50.6 g, 115 mmol
- MeOH 500 mL
- NH3 in MeOH 7 M, 45.81 mL
- Step 6 2,5,7-Trichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol
- 5 To a solution of 5,7-dichloro-8-fluoropyrido[4,3-d]pyrimidine-2,4(1H,3H)-dione (10 g, 40.0 mmol) in POCl3 (100 mL) was added DIPEA (20.9 mL, 120 mmol). The mixture was stirred at 105 °C for 16 h. The reaction mixture was concentrated under reduced pressure. The residue was diluted with 1,4-dioxane (40 mL) and the resulting solution was added dropwise to aq. K2CO3 (20%, 200 mL).
- Step 2 2-(8-Ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl-1,3,2- dioxaborolane [0251] To a solution of 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- 10% purity). The suspension was degassed, purged with H2 three times, and stirred at 25 °C for 16 h under H2 (15 psi). The reaction mixture was filtered and concentrated under reduced pressure.
- Step 2 2-(8-(Ethynyl-d)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
- 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (1.9 g, 5.334 mmol) in MeCN (30 mL) under nitrogen atmosphere were added K2CO3 (2.21 g, 16.002 mmol) and D2O (3.0 mL, 164.778 mmol) at room temperature.
- Step 3 2-(8-(Ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0258] To a solution of 2-(8-(ethynyl-d)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (1.5 g, 4.199 mmol) in EtOAc (38 mL) under nitrogen atmosphere was added PtO 2 (0.14 g, 0.630 mmol) at room temperature.
- Step 2 2-(7-Fluoro-3-(methoxymethoxy)-8-vinylnaphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane
- 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane 13.0 g, 36.5 mmol
- quinoline (6.00 g, 46.5 mmol, 5.48 mL) in methanol (50.0 mL) was added Lindlar catalyst (7.80 g, 37.8 mmol).
- Step 1 tert-Butyl (S)-2-(hydroxymethyl)azepane-1-carboxylate
- BH 3 61.72 mL, 61.72 mmol, 1M in THF
- Step 2 (S)-Azepan-2-ylmethanol [0268] To an ice-cooled solution of tert-butyl (S)-2-(hydroxymethyl)azepane-1-carboxylate (30 g, 130.82 mmol) in DCM (300 mL) was added 4N HCl (gas) in 1,4-dioxane (300 mL, 9873.83 mmol) dropwise. The reaction mixture was stirred in an ice bath for 1.5 hours. The ice bath was removed, and the resulting mixture was concentrated under reduced pressure. The residue was dissolved in MeOH (50 mL), and Amberlyst-A-21 resin (8 g) was added at room temperature.
- Step 3 (S)-5-(Azepan-2-ylmethoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol
- NaH 0.89 g, 37.25 mmol, 60% in mineral oil
- Step 3 (S)-5-(Azepan-2-ylmethoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol
- Step 4 (S)-2,5-Dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0272] To an ice-cooled solution of (S)-5-(azepan-2-ylmethoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4-ol (1.9 g, 5.26 mmol) and DIEA (4.08 g, 31.56 mmol) in DCM (380 mL) under nitrogen atmosphere was added POCl3 (4.03 g, 26.30 mmol) dropwise.
- Step 5 (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0274] To a mixture of (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- pyrrolizin-7a(5H)-yl)methanol (556.69 mg
- Step 1 1-(tert-Butyl) 2-methyl (2R,4R)-4-((tert-butyldimethylsilyl)oxy)pyrrolidine-1,2- dicarboxylate
- TBSCl adazole-6.25 g, 91.734 mmol
- the ice bath was removed, and the reaction mixture was stirred at room temperature for 16 hours.
- Step 2 1-(tert-Butyl) 2-methyl (4R)-4-((tert-butyldimethylsilyl)oxy)-2-(2- (chloromethyl)allyl)pyrrolidine-1,2-dicarboxylate
- 1-(tert-butyl) 2-methyl (2R,4R)-4-((tert- butyldimethylsilyl)oxy)pyrrolidine-1,2-dicarboxylate 15 g, 41.720 mmol
- THF 300 mL
- LiHMDS 54.24 mL, 54.236 mmol, 1M in THF
- Step 3 1-(tert-Butyl) 2-methyl (4R)-2-(2-(chloromethyl)allyl)-4-hydroxypyrrolidine-1,2- dicarboxylate
- 1-tert-butyl 2-methyl (4R)-4-[(tert-butyldimethylsilyl)oxy]-2-[2- (chloromethyl)prop-2-en-1-yl]pyrrolidine-1,2-dicarboxylate 36 g, 80.345 mmol
- TBAF 96.41 mL, 96.414 mmol, 1M in THF
- Step 4 1-(tert-Butyl) 2-methyl (4S)-2-(2-(chloromethyl)allyl)-4-fluoropyrrolidine-1,2- dicarboxylate
- 1-(tert-butyl) 2-methyl (4R)-2-(2-(chloromethyl)allyl)-4- hydroxypyrrolidine-1,2-dicarboxylate 7.8 g, 23.367 mmol
- DCM DCM
- BAST 7.75 g, 35.050 mmol
- Step 7 ((2S,7aR)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol & ((2S,7aS)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0289] To an ice-cooled solution of methyl (2S)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizine- 7a(5H)-carboxylate (7.5 g, 37.646 mmol) in THF (75 mL) under N 2 atmosphere was added LiAlH 4 (37.65 mL, 75.292 mmol, 2M in THF) dropwise.
- Step 1 (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine
- TBDPSCl TBDPSCl
- Step 2 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one (a mixture) [0294] To an ice-cooled solution of (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2- fluorohexahydro-1H-pyrrolizine (10 g, 25.15 mmol) and RuCl 3 .H 2 O (2.83 g, 12.57 mmol) in CCl 4 (50 mL) and H 2 O (50 mL) was added NaIO 4 (26.90 g, 125.75 mmol).
- Step 3 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one & [0296] A mixture of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one (6 g) was separated by Prep-Achiral-SFC with the following conditions: Column: 2, Mobile Phase B
- Step 2 ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-3,3-d2)methanol
- (2R,7aS)-2-fluoro-7a-(hydroxymethyl)hexahydro-3H- pyrrolizin-3-one 400 mg, 2.310 mmol
- LiAlD4 4.85 mL, 4.85 mmol, 1M in THF
- Step 2 ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methanol
- 6R,7aS -6-fluoro-7a-(hydroxymethyl)-tetrahydro-1H- pyrrolizin-3-one
- LiAlD4 (1.82 mL, 1.819 mmol, 1M in THF
- Step 2 ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methan-d2-ol
- (2R,7aS)-2-fluoro-hexahydropyrrolizine-7a-carboxylic acid 200 mg, 1.155 mmol
- Me-THF Me-THF
- LiAlD4 2.43 mL, 2.425 mmol, 1M in THF
- Step 2 ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methan-d2-ol
- (2R,7aS)-2-fluorotetrahydro-1H-pyrrolizine-7a(5H)- carboxylic-5,5-d 2 acid 80 mg, 0.457 mmol
- LiAlD4 0.96 mL, 0.960 mmol, 1M in THF
- Step 2 4-(3,4-Difluorophenyl)-3-oxobutanoic acid [0321] To a stirred solution of 1,3-ditrimethylsilyl propanedioate (3.91 g, 15.74 mmol) in THF (50 mL) under nitrogen atmosphere was added n-BuLi (5.2 mL, 13.08 mmol, 2.5 M in n-Hexane) dropwise at -78 °C.
- Step 3 6,7-Difluoronaphthalene-1,3-diol
- a solution of 4-(3,4-difluorophenyl)-3-oxobutanoic acid (800 mg, 3.73 mmol) in trifluoromethanesulfonic acid (8 mL) under nitrogen atmosphere was stirred at 25°C for 16 hours.
- the resulting mixture was diluted with EA (150 mL), washed with water (4 x 60 mL) and brine (60 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure.
- Step 4 6,7-Difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol
- 6 mL 1,4-dioxane
- [Ru(p-cymene)Cl2]2 162.34 mg, 0.26 mmol
- KOAc 520.35 mg, 5.30 mmol
- Step 5 6,7-Difluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-ol
- 627 To an ice-cooled solution of 6,7-difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol (600 mg, 1.59 mmol) and DIEA (267.75 mg, 2.07 mmol) in DCM (6 mL) under nitrogen atmosphere was added bromo(methoxy)methane (189.18 mg, 1.51 mmol) dropwise.
- Step 6 6,7-Difluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate
- DCM dimethyl methoxycarbonate
- Step 7 ((2,3-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-1-yl)ethynyl)triisopropylsilane [0331] To a stirred solution of 6,7-difluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (500 mg, 0.90 mmol) and WSGR Docket No.
- Step 2 4-(3-Bromo-4-fluorophenyl)-3-oxobutanoic acid
- 1,3-ditrimethylsilyl propanedioate 23.71 g, 95.43 mmol
- THF 250 mL
- n-BuLi 2.5 M in n-hexane, 32 mL, 80 mmol
- Step 3 6-Bromo-7-fluoronaphthalene-1,3-diol
- Step 4 ((6-Bromo-7-fluoronaphthalene-1,3-diyl)bis(oxy))bis(tert-butyldimethylsilane) [0340] To an ice-cooled solution of 6-bromo-7-fluoronaphthalene-1,3-diol (5.0 g, 19.44 mmol) in DCM (50 mL) under nitrogen atmosphere were added Et3N (13.52 mL, 97.24 mmol) and TBSOTf (17.88 mL, 77.80 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours.
- Step 5 ((7-Fluoronaphthalene-1,3-diyl-6-d)bis(oxy))bis(tert-butyldimethylsilane)
- Step 5 To a stirred solution of ((6-bromo-7-fluoronaphthalene-1,3-diyl)bis(oxy))bis(tert- butyldimethylsilane) (4.4 g, 9.06 mmol) in CH3OD (50 mL) under D2 atmosphere was added Pd/C (4.4 g, 10% wt) at room temperature. The resulting mixture was stirred at room temperature for 1 hour under D 2 atmosphere. The resulting mixture was filtered and concentrated under reduced pressure.
- Step 7 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-6-d-1,3-diol
- Step 7 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-6-d-1,3-diol
- Step 8 7-Fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-6-d-1-ol
- Step 8 To an ice-cooled solution of 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-6-d-1,3-diol (630 mg, 1.75 mmol) and DIEA (0.23 g, 1.75 mmol) in DCM (10 mL) under nitrogen atmosphere was added bromo(methoxy)methane (0.20 g, 1.58 mmol) dropwise.
- Step 9 7-Fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl-6-d trifluoromethanesulfonate
- DCM dimethyl methoxycarbonate
- Step 10 ((2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-1-yl-3-d)ethynyl)triisopropylsilane [0352] To a stirred solution of 7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl-6-d trifluoromethanesulfonate (610 mg, crude) and B2Pin2 (347.03 mg, 1.37 mmol) in 1,4-dioxane (7 mL) under nitrogen atmosphere were added KOAc (335.30 mg, 3.42 mmol) and Pd(dppf)Cl2.CH2Cl2 (83.33.
- Step 1 6-Bromo-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol
- 6-bromo-7-fluoronaphthalene-1,3-diol (refer to Intermediate 15 for detail procedures, 2 g, 7.780 mmol) and [Ru(p-Cymene)Cl2]2 (0.48 g, 0.778 mmol) in 1,4-dioxane (20 mL) under nitrogen atmosphere were added (2-bromoethynyl)tris(propan-2-yl)silane (2.24 g, 8.558 mmol) and KOAc (1.53 g, 15.560 mmol) at room temperature.
- Step 2 6-Bromo-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- ol
- 6-bromo-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene- 1,3-diol (2 g, 4.572 mmol) and DIEA (768.23 mg, 5.944 mmol) in DCM (20 mL) under nitrogen atmosphere was added bromo(methoxy)methane (542.80 mg, 4.343 mmol) dropwise.
- Step 3 3-Fluoro-5-hydroxy-7-(methoxymethoxy)-4-((triisopropylsilyl)ethynyl)-2- naphthonitrile
- 6-bromo-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-ol (1 g, 2.077 mmol) and Pd(PPh3)4 (1.20 g, 1.038 mmol) in DMF (10 mL) under nitrogen atmosphere was added Zn(CN) 2 (0.27 g, 2.285 mmol) at room temperature.
- Step 4 6-Cyano-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate
- 3-fluoro-5-hydroxy-7-(methoxymethoxy)-4- ((triisopropylsilyl)ethynyl)-2-naphthonitrile 500 mg, 1.169 mmol
- DIEA 453.40 mg, 3.507 mmol
- Tf2O 494.88 mg, 1.754 mmol
- Step 5 3-Fluoro-7-(methoxymethoxy)-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4- ((triisopropylsilyl)ethynyl)-2-naphthonitrile
- 6-cyano-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate 700 mg, crude
- B 2 Pin 2 594.05 mg, 2.340 mmol
- Pd(dppf)Cl2.CH2Cl2 85.59 mg, 0.117 mmol
- KOAc 344.38 mg, 3.510 mmol
- Step 2 (S)-Azepan-2-ylmethanol [0372] To a solution of (S)-azepane-2-carboxylic acid (2.2 g, 12.2 mmol) in THF (40 mL) was added dropwise LiAlH 4 (2.5 M in n-hexane, 24.5 mL) at 0 °C under N 2 atmosphere. The reaction was warmed up to 20 °C slowly and stirred for 1 h. Na 2 SO 4 ⁇ 10H 2 O (29.5 g, 90 mmol) was added into the mixture slowly at 0 °C, and the mixture was stirred for 30 min.
- LiAlH 4 2.5 M in n-hexane, 24.5 mL
- Step 1 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol
- Step 1 To a solution of 7-fluoronaphthalene-1,3-diol (50 g, 281 mmol) and 2- bromoethynyl(triisopropyl)silane (77 g, 295 mmol) in 1,4-dioxane (334 mL) were added KOAc (55.1 g, 561 mmol) and dichlororuthenium:1-isopropyl-4-methyl-benzene (17.2 g, 28.1 mmol).
- Step 2 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3- diylbis(trifluoromethanesulfonate) [0377] To a solution of 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol (140 g, 390 mmol) and DIPEA (408 mL, 2.34 mol) in CH2Cl2 (3.5 L) was added Tf2O (258 ml, 1.56 mol) dropwise under N 2 atmosphere at 0 °C.
- Step 3 3-((Diphenylmethylene)amino)-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate WSGR Docket No.
- Step 1 (2R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-2,3,5,7a-tetrahydro- 1H-pyrrolizine
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro- 5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one (refer to Intermediate 36 for detail procedures, 1 g, 2.44 mmol) in DCM (100 mL) under nitrogen atmosphere was added tetrafluoroboranuide; trimethyloxidanium (1.81 g, 12.21 mmol) at room temperature.
- Step 2 ((2R,7aR)-2-Fluoro-2,3-dihydro-1H-pyrrolizin-7a(5H)-yl)methanol
- (2R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluoro- 2,3,5,7a-tetrahydro-1H-pyrrolizine 200 mg, 0.50 mmol
- TBAF 0.75 mL, 0.75 mmol, 1 M solution in THF
- Step 1 (S)-5-Chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0389] A solution of (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 5 for detail procedures, 1.2 g, 3.49 mmol) and KF (304.72 mg, 5.24 mmol) in DMSO (60 mL) under nitrogen atmosphere was heated at 80 °C for 16 hours.
- Step 2 (S)-2,4-difluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0391] To a stirred mixture of (S)-5-chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (200 mg, 0.61 mmol) and ((2-fluoro-6- (methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-y
- Step 1 A mixture of ethyl (2S,7aS)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aR)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate- 2-d [0394] To an ice-cooled solution of ethyl 2,5-dioxo-tetrahydropyrrolizine-7a-carboxylate (20 g, 94.68 mmol) in ethyl alcohol (150 mL) was added NaBD4 (796.53 mg, 18.93 mmol).
- Step 2 A mixture of ethyl (2S,7aR)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aS)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate-2-d [0396] To a stirred solution of ethyl (2S,7aS)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aR)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate- WSGR Docket No.
- Step 3 A mixture of (6S,7aR)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one- 6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one-6-d.
- Step 4 A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro- 3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro- 3H-pyrrolizin-3-one-6-d [0400] To an ice-cooled solution of a mixture of (6S,7aR)-6-fluoro-7a-(hydroxymethyl)hexahydro- 3H-pyrrolizin-3-one-6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one- 6-d (500 mg, 2.87 mmol) and imidazole (390.81 mg, 5.74 mmol) in DMF (5 mL) under nitrogen atmosphere was added
- Step 5 (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d [0402] A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d (2.5 g, 6.06 mmol
- the first eluting peak (RT1: 6.63 min) was concentrated under reduced pressure to give the title compound (Intermediate 24, 1 g, 40% yield) as a white solid.
- 1 H NMR (400 MHz, DMSO-d6) 1H), 2.81 - 2.56 (m, 1H), 2.28 - 1.78 (m, 5H), 0.99 (s, 9H).
- the second eluting peak (RT1: 8.07 min) was concentrated under reduced pressure to give the title compound (Intermediate 25, 1 g, 40% yield) as a white solid.
- Step 2 ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2-d)methanol
- 6R,7aS -6-fluoro-7a-(hydroxymethyl)hexahydro-3H- pyrrolizin-3-one-6-d
- LiAlH4 4.8 mL, 4.8 mmol, 1 M in THF
- Step 2 4-(3-Chloro-4-fluorophenyl)-3-oxobutanoic acid
- 2-(3-chloro-4-fluorophenyl)acetyl chloride (10.9 g, crude) in THF (110 mL) under nitrogen atmosphere was added n-butyllithium (2.5 M in n-hexane, 25.2 mL, 63.17 mmol) dropwise at -78 °C.
- the resulting mixture was stirred in an ice bath for 0.5 hours.
- the reaction was quenched with sat. aq. NH 4 Cl (500 mL) in an ice bath and concentrated under reduced pressure.
- the combined organic layers were washed with brine (500 mL), dried over WSGR Docket No. 62619-720.601 anhydrous Na 2 SO 4 , filtered, and concentrated under reduced pressure.
- the residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (8 g, 65% yield for two steps) as a black oil.
- Step 3 6-Chloro-7-fluoronaphthalene-1,3-diol
- 4-(3-Chloro-4-fluorophenyl)-3-oxobutanoic acid (8 g, 34.68 mmol) was dissolved in CF3SO3H (150 mL) at room temperature, and the mixture was stirred at this temperature for 16 hours under nitrogen atmosphere. The resulting mixture was diluted with EtOAc (800 mL), washed with water (4 x 300 mL) and brine (500 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure.
- Step 4 6-Chloro-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol
- 6-chloro-7-fluoronaphthalene-1,3-diol 6.5 g, 30.57 mmol
- 1,4- dioxane 65 mL
- (2-bromoethynyl)triisopropylsilane 8787.10 mg, 33.63 mmol
- [Ru(p-cymene)Cl2]2 (1872.31 mg, 3.05 mmol
- KOAc (6001.16 mg, 61.14 mmol) at room temperature.
- Step 5 6-Chloro-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- ol
- 6-chloro-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene- 1,3-diol (6 g, 15.26 mmol) and DIEA (2565.40 mg, 19.84 mmol) in DCM (60 mL) under nitrogen atmosphere was added bromo(methoxy)methane (1717.19 mg, 13.74 mmol) dropwise.
- Step 6 6-Chloro-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate WSGR Docket No.
- Step 7 ((3-Chloro-2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane [0425] To a stirred solution of 6-chloro-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (5 g, crude) and bis(pinacolato)diboron (4462.29 mg, 17.57 mmol) in 1,4-dioxane (50 mL) under nitrogen atmosphere were added AcOK (2586.86 mg, 26.358 mmol) and Pd(dppf)Cl2•CH2Cl2 (715.73 mg, 0.87 mmol) at room temperature.
- Step 2 A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl-d 2 )-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl-d2)- 6-fluorohexahydro-3H-pyrrolizin-3-one-6-d [0430] To an ice-cooled solution of (6S,7aR)-6-fluoro-7a-(hydroxymethyl-d 2 )hexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H-pyrrolizin-3-one-6- d (2.1 g mixture, 11.91 mmol) and 1H-imidazole (1.62 g, 23.83 mmol)
- Step 2 ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2,5,5-d3)methan-d2-ol
- ((6R,7aS)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H- pyrrolizin-3-one-6-d (380 mg, 2.15 mmol) in 2-methyl-THF (4 mL) under nitrogen atmosphere was added LiAlD4 (4.53 mL, 4.53 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour.
- Step 2 (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-5,6,7,7a-tetrahydro- 3H-pyrrolizin-3-one
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- (phenylselanyl)hexahydro-3H-pyrrolizin-3-one 5.2 g, 9.177 mmol
- DCM 80 mL
- H 2 O 2 (20 mL, 30% w/w
- Step 3 ((6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-3-oxohexahydro-1H- pyrrolizin-1-yl)boronic acid
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro- 5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one 800 mg, 1.953 mmol) in EtOH (20 mL) were added 2- (Dicyclohexylphosphino)biphenyl (68.46 mg, 0.195 mmol), CuCl (19.34 mg, 0.195 mmol), (dihydroxyboranyl)boronic acid (210.13 mg, 2.344 mmol) and t-BuONa (56.31 mg, 0.586 mmol) at room temperature
- Step 4 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-3H- pyrrolizin-3-one
- ((6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-3- oxohexahydro-1H-pyrrolizin-1-yl)boronic acid 600 mg, 1.317 mmol
- AgNO 3 (223.81 mg, 1.317 mmol
- Selectfluor (2.80 g, 7.902 mmol) in DCM (9 mL) were added H2O (9 mL), H3PO4 (1.2 mL, 20.634 mmol) and TFA (4.8 mL, 64.623 mmol) under nitrogen atmosphere.
- the resulting mixture was quenched with MeOH (40 mL) and 2 M HCl (40 mL) in an ice bath, and then heated at 60 °C for another 1 hour. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The mixture was diluted with EtOAc (80 mL) and saturated aqueous NaHCO 3 (100 mL). After separation, the aqueous phase was extracted with EtOAc (3 x 80 mL). The combined organic layers were washed brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure.
- Step 6 ((6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-1,6- difluorohexahydro-1H-pyrrolizine (680 mg, 1.636 mmol) in THF (7 mL) was added pyridine hydrofluoride (347.48 mg, 2.454 mmol, 70% w/w) at room temperature.
- the reaction mixture was stirred at room temperature for 2 hours.
- the resulting mixture was concentrated under reduced pressure.
- Step 2 (2R)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,6-difluorohexahydro-1H- pyrrolizine
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6- difluorohexahydro-3H-pyrrolizin-3-one (2 g, 4.65 mmol) in THF (50 mL) under nitrogen atmosphere was added BH 3 •Me 2 S (2.33 mL, 23.3 mmol) dropwise.
- the reaction mixture was heated at 60 °C for 16 hours.
- Step 3 ((2R)-2,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- (2R)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6- difluorohexahydro-1H-pyrrolizine (1.5 g, 3.60 mmol) in THF (30 mL) under nitrogen atmosphere was added TBAF (5.41 mL, 5.41 mmol) dropwise at room temperature. The resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was concentrated under reduced pressure.
- Step 1 (8aS)-5-Chloro-2-(((2R,7aS)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0466] To an ice-cooled solution of Intermediate 37 (182.2 mg, 1.02 mmol) in THF (5 mL) under nitrogen atmosphere was added NaH (51.41 mg, 1.28 mmol, 60% dispersion in mineral oil).
- Step 2 (S)-5-Chloro-2-(((2R,6S,7as)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene & (S)-5-chloro-2-(((2R,6R)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0468] (8aS)-5-Chloro-2-(((2R,7as)-2
- the first eluting peak (RT1: 10.136 min) was concentrated and lyophilized to give the title compound (Intermediate 38, 48 mg, 20% yield) as an off-white solid.
- the second eluting peak (RT2: 13.282 min) was concentrated and lyophilized to give the title compound (Intermediate 39, 130 mg, 54% yield) as an off-white solid.
- Step 1 5-Chloro-6-fluoro-1,4-dihydro-1,4-epoxynaphthalene
- 1-bromo-3-chloro-2,4-difluorobenzene 10 g, 43.97 mmol
- furan 6.02 g, 88.38 mmol
- n-BuLi 2.5 M in n-hexane, 21.00 mL, 52.5 mmol
- the resulting mixture was stirred at -15 °C for 30 min. Then the resulting mixture was warmed to room temperature and stirred for another 16 hours.
- Step 4 8-Chloro-7-fluoro-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-1-yl pivalate
- 8-chloro-7-fluoronaphthalen-1-yl pivalate 2.5 g, 8.90 mmol
- bis(pinacolato)diboron 2.26 g, 8.90 mmol
- n-hexane 75 mL
- Step 5 8-Chloro-7-fluoro-3-hydroxynaphthalen-1-yl pivalate
- H2O2 4.87 g, 42.96 mmol, 30% aq.
- AcOH 21.08 g, 350.99 mmol
- Step 8 8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl trifluoromethanesulfonate
- DCM 7.5 mL
- DIEA 546.24 mg, 4.22 mmol
- Tf2O 604.18 mg, 2.14 mmol
- the resulting mixture was stirred at -40 °C for 1.5 hours.
- the resulting mixture was diluted with H2O (15 mL) and extracted with EtOAc (3 x 80 mL).
- Step 9 2-(8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0492] To a solution of 8-chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl trifluoromethanesulfonate (1.05 g, crude) and AcOK (795.32 mg, 8.10 mmol) in 1,4-dioxane (12 mL) under nitrogen atmosphere were added bis(pinacolato)diboron (1.37g, 5.40 mmol) and Pd(dppf)Cl2•CH2Cl2 (220.05 mg, 0.27 mmol) at room temperature.
- Step 2 3-((Diphenylmethylene)amino)-6,7-difluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate [0497] To a stirred solution of 6,7-difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diyl bis(trifluoromethanesulfonate) (3 g, crude), Pd2(dba)3 (428.83 mg, 0.468 mmol), XantPhos (812.90 mg, 1.405 mmol) and Cs 2 CO 3 (3051.55 mg, 9.366 mmol) in toluene (30 mL) was added diphenylmethanimine (933.59 mg, 5.151 mmol) at room temperature under nitrogen atmosphere.
- Step 3 N-(6,7-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine [0499] To a stirred solution of 3-((diphenylmethylene)amino)-6,7-difluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (2 g, 2.977 mmol) and 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (1.512 g, 5.954 mmol) in 1,4-dioxane (20 mL) under nitrogen atmosphere were added KOAc (876.53 mg
- Step 2 tert-Butyl 3-oxo-4-(2,4,5-trifluorophenyl)butanoate
- a solution of 2,2-dimethyl-5-(2-(2,4,5-trifluorophenyl)acetyl)-1,3-dioxane-4,6-dione (40 g, 126.48 mmol) in 2-methyl-2-propanol (680 mL) was heated at 88 °C for 5 hours. The reaction mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% EA in PE to afford the title compound (30 g, 82% yield) as a white oil.
- Step 5 5,7,8-Trifluoro-3-((triisopropylsilyl)oxy)naphthalen-1-ol
- DIEA 1,3-diol
- chlorotriisopropylsilane 734.85 mg, 3.81 mmol
- Step 7 Triisopropyl((5,6,8-trifluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)oxy)silane
- 514 To a stirred mixture of 5,7,8-trifluoro-3-((triisopropylsilyl)oxy)naphthalen-1-yl trifluoromethanesulfonate (750 mg, 1.49 mmol), bis(pinacolato)diboron (757.97 mg, 2.98 mmol) and Pd(dppf)Cl 2 .DCM (121.58 mg, 0.14 mmol) in 1,4-dioxane (8 mL) was added KOAc (439.41 mg, 4.47 mmol) at room temperature under argon atmosphere.
- Step 2 (5R,6aR)-6a-(((tert-Butyldiphenylsilyl)oxy)methyl)-5- fluorooctahydrocyclopropa[a]pyrrolizine
- 5R,6aR -6a-(((tert-butyldiphenylsilyl)oxy)methyl)-5- fluorohexahydrocyclopropa[a]pyrrolizin-2(1H)-one
- BH3•Me2S 0.413 mL, 4.13 mmol, 10M in DMS
- Step 3 ((5R,6aR)-5-Fluorohexahydrocyclopropa[a]pyrrolizin-6a(4H)-yl)methanol
- 5R,6aR -6a-(((tert-butyldiphenylsilyl)oxy)methyl)-5- fluorooctahydrocyclopropa[a]pyrrolizine (230 mg, 0.56 mmol) in THF (3.5 mL) was added TBAF (1 M in THF, 0.84 mL, 0.84 mmol) at room temperature. The resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure.
- Step 1 (2R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine-3,3-d2
- TBDPSCl 736.52 mg, 2.68 mmol
- Step 3 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-5,5-d 2
- the mixture was separated by prep-Achiral-HPLC with the following conditions: Column: 2; Mobile Phase B: MeOH(0.1% 2 M NH 3 WSGR Docket No. 62619-720.601 Back Pressure(bar): 100; Detector: UV 220 nm; RT1 (min): 5.17; RT2 (min): 7.97.
- Step 4 (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one-5,5-d 2
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-5,5-d 2 550 mg, 1.33 mmol
- THF 5.5 mL
- TBAF (1 M in THF, 1.46 mL, 1.463 mmol
- Step 1 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-hydroxyhexahydro- 3H-pyrrolizin-3-one
- LDA 9.11 mL, 18.222 mmol
- Step 2 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H- pyrrolizin-2-ol
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-hydroxyhexahydro-3H-pyrrolizin-3-one 100 mg, 0.234 mmol
- THF 10 mL
- BH 3 -Me 2 S (0.12 mL, 1.170 mmol
- Step 3 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorotetrahydro-1H- pyrrolizin-2(3H)-one
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-ol 500 mg, 1.209 mmol
- TEA 366.99 mg, 3.627 mmol
- DCM 5 mL
- DMSO DMSO
- Step 4 (6R,7aS)-6-fluoro-7a-(hydroxymethyl)tetrahydro-1H-pyrrolizin-2(3H)-one
- 6R,7aS -6-fluoro-7a-(hydroxymethyl)tetrahydro-1H-pyrrolizin-2(3H)-one
- TBAF 0.0573 mL, 0.729 mmol
- Step 2 ((2R,7aS)-2-Fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- (2R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methylenehexahydro-1H-pyrrolizine (298 mg, 0.728 mmol) in THF (3 mL) was added TBAF (1.09 mL, 1.092 mmol, 1M in THF) at room temperature. The mixture was stirred at room temperature for 1 hour.
- Step 2 (2-Bromoethyl)diphenylsulfonium trifluoromethanesulfonate
- the crude was purified by RP-Flash with the following conditions: Column: C18, 40 g, 4HCO3; Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 10 min; Flow rate: 35 mL/min; Detector: UV 210 nm.
- the product-containing fractions were collected and evaporated in vacuo and then lyophilized overnight to give the title compound (170 mg, 19% yield) as a yellow oil.
- Step 2 (6'R,7a'S)-7a'-(((tert-Butyldiphenylsilyl)oxy)methyl)-6'-fluorotetrahydro-1'H,3'H- spiro[cyclopropane-1,2'-pyrrolizine]
- 6'R,7a'S)-7a'-(((tert-butyldiphenylsilyl)oxy)methyl)-6'- fluorotetrahydro-1'H,3'H-spiro[cyclopropane-1,2'-pyrrolizin]-3'-one (170 mg, 0.38 mmol) in THF (5 mL) was added BH3•Me2S (0.19 mL, 1.94 mmol) dropwise under nitrogen atmosphere.
- the ice bath was removed, and the resulting mixture was heated at 60 °C for 16 hours.
- the reaction mixture was cooled to room temperature and quenched with MeOH (5 mL) and 2 M HCl aq. (5 mL) at room temperature.
- the resulting mixture was heated at 60 °C for 1 hour.
- the mixture was WSGR Docket No. 62619-720.601 cooled to room temperature and concentrated under reduced pressure.
- the mixture was diluted with EtOAc (30 mL) and saturated aqueous NaHCO3 (20 mL). After separation, the aqueous phase was extracted with EtOAc (3 x 20 mL).
- Step 3 ((6'R,7a'S)-6'-Fluorodihydro-1'H,3'H-spiro[cyclopropane-1,2'-pyrrolizin]-7a'(5'H)- yl)methanol
- 6'R,7'aS)-7'a- ⁇ [(tert-butyldiphenylsilyl)oxy]methyl ⁇ -6'-fluoro- tetrahydro-1'H-spiro[cyclopropane-1,2'-pyrrolizine] (116 mg, 0.27 mmol) in THF (1.5 mL) was added TBAF (1 M solution in THF, 0.41 mL, 0.41 mmol) at room temperature.
- Step 2 (6R,7aS)-7a-((((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol [0565] To a stirred solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-hexahydropyrrolizin-2-ol (135 mg, 0.77 mmol) and Cs2CO3 (131.53 mg, 0.40 mmol) in 1,4-dioxane (5 mL) under argon atmosphere was added (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,
- Step 3 (2S,6R,7aS)-7a-(((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol & (2R,6R,7aS)-7a-(((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol [0567] (6R,7aS)-7a-((((S)-5-chloro-4-
- the first eluting peak (RT1: 7.089) was collected and concentrated to give the title compound (Intermediate 51, 30 mg, 13% yield) as light-yellow lyophilized powder.
- the second eluting peak (RT2: 8.309 min) was collected and concentrated to give the title compound (Intermediate 52, 150 mg, 65% yield) as light-yellow lyophilized powder.
- MS: m/z 482.20 [M + H] + .
- Step 2 (2R,6S,7R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine-6,7-d2
- Step 3 ((2R,6S,7R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,7-d 2 )methanol
- Step 1 tert-Butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate
- (2S)-1-(tert-butoxycarbonyl)azepane-2-carboxylic acid (2 g, 8.220 mmol)
- N,O-dimethylhydroxylaminehydrochloride (1.20 g, 12.330 mmol) in DCM (20 mL) under nitrogen atmosphere were added DIEA (4.25 g, 32.880 mmo) and HATU (4.69 g, 12.330 mmol).
- Step 2 tert-Butyl (2S)-2-acetylazepane-1-carboxylate
- tert-butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate 2.2 g, 7.682 mmol
- bromo(methyl) magnesium 11.5 mL, 1.362 mmol
- the reaction was quenched with NH4Cl (sat) (30 mL) in an ice bath and extracted with EA (3 x 50 mL).
- Step 3 tert-Butyl (2S)-2-(1-hydroxyethyl)azepane-1-carboxylate
- BH 3 .THF 12 mL, 12.000 mmol, 1 M in DMS.
- the ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours.
- the reaction was quenched with water (10 mL) in an ice bath and extracted with EtOAc (3 x 15 mL).
- Step 4 (S)-1-(Azepan-2-yl)ethan-1-ol WSGR Docket No. 62619-720.601 [0583] To an ice-cooled stirred solution of tert-butyl (2S)-2-(1-hydroxyethyl)azepane-1- carboxylate (1.9 g, 7.808 mmol) in DCM (20 mL) was added HCl (20 mL, 4 M in 1,4-dioxane) dropwise. The ice bath was removed, and the reaction mixture was stirred at room temperature for 2 hours. The mixture was concentrated under reduced pressure.
- Step 5 (S)-5-(1-(Azepan-2-yl)ethoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol
- (S)-1-(azepan-2-yl)ethan-1-ol (1.17 g, 8.195 mmol) in THF (40 mL) under nitrogen atmosphere was added NaH (1.19 g, 29.800 mmol, 60% dispersion in mineral oil). The mixture was stirred in an ice bath for 0.5 hours. Intermediate 1 (2 g, 7.450 mmol) was added to the above mixture, and the reaction mixture was stirred at room temperature for 2.5 hours.
- Step 6 (S)-2,5-Dichloro-4-fluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0587] To an ice-cooled solution of (S)-5-(1-(azepan-2-yl)ethoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4-ol (1.7 g, 4.531 mmol) and DIEA (3.51 g, 27.186 mmol) in DCM (340 mL) under nitrogen atmosphere was added POCl3 (3.47 g, 22.655 mmol) dropwise.
- Step 7 (S)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0589] To a stirred solution of (S)-2,5-dichloro-4-fluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (1.1 g, 3.079 mmol) in DMSO (20 mL) was added KF (0.30 g, 5.234 mmol) at room temperature under nitrogen atmosphere.
- Step 8 (8S,8aS)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene & (8R,8aS)-5-chloro-2,4-difluoro-8-methyl- 8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0591] (S)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (850 mg, 2.494 mmol) was separated
- the first eluting peak (RT1: 1.731 min) was concentrated and lyophilized to give the title compound (Intermediate 54, 280 mg, 32% yield) as a yellow lyophilized powder.
- MS: m/z 341.05 [M + H] + .
- the second eluting peak (RT2: 1.995 min) was concentrated and lyophilized to give the title compound (Intermediate 55, 380 mg, 44% yield) as a yellow lyophilized powder.
- MS: m/z 341.05 [M + H] + .
- Step 2 (2R,6S,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6-methylhexahydro- 1H-pyrrolizine
- (2S,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-methylhexahydro-3H-pyrrolizin-3-one 550 mg, 1.29 mmol
- THF 6 mL
- BH 3 •Me 2 S (0.65 mL, 6.46 mmol
- the reaction mixture was heated at 60 °C for 16 hours.
- the resulting mixture was cooled in an ice bath, quenched with MeOH (7 mL) and 2 M HCl (7 mL).
- the resulting mixture was heated at 60 °C for another 1 hour.
- the resulting mixture was cooled and concentrated under reduced pressure.
- the resulting mixture was diluted with EtOAc (100 mL), washed with saturated aqueous NaHCO3 (3 x 50 mL) and brine (50 mL).
- the organic layer was dried over anhydrous Na 2 SO 4 , filtered and concentrated under reduced pressure.
- Step 3 ((2R,6S,7aS)-2-Fluoro-6-methyltetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- (2R,6S,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methylhexahydro-1H-pyrrolizine 350 mg, 0.85 mmol
- MeOH 4 mL
- NH4F (1259.65 mg, 34.00 mmol
- the ice bath was removed, and the reaction mixture was heated at 60 °C for 16 hours.
- the reaction mixture was cooled in an ice bath, then MeOH (10 mL) and HCl aq. (2 M, 10 mL) were added.
- the resulting mixture was heated at 60 °C for 1 hour.
- the resulting mixture was cooled, quenched with saturated aq. NaHCO3 (50 mL) in an ice bath, and extracted with EA (3 x 50 mL).
- the combined organic layers were washed with brine (50 mL), dried over anhydrous Na 2 SO 4 , filtered and concentrated under reduced pressure.
- Step 3 ((6R,7aR)-2,6-difluoro-2-methyltetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6-difluoro-2- methylhexahydro-1H-pyrrolizine 300 mg, 0.69 mmol
- NH4F (1034.49 mg, 27.92 mmol)
- Step 2 (R)-(1,4-Oxazepan-3-yl)methanol [0610] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-5-one (200 mg, 1.38 mmol) in THF (5 mL) was added LiAlH4 (2.5 M in THF, 1.10 mL) at 0 °C.
- Step 3 (S)-5-((1,4-Oxazepan-3-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one
- (R)-(1,4-oxazepan-3-yl)methanol 130 mg, 991 ⁇ mol
- THF 10 mL
- NaH 176 mg, 4.40 mmol, 60% purity
- Step 4 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0614] To a solution of (S)-5-((1,4-oxazepan-3-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (400 mg, 1.10 mmol) in CH 2 Cl 2 (150 mL) were added DIEA (6.61 mmol, 1.15 mL) and POCl3 hr.
- Example 59 (S)-5-Chloro-2,4-difluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene WSGR Docket No. 62619-720.601 [0616] To a stirred solution of Intermediate 58 (900 mg, 2.60 mmol) in DMSO (9 mL) under nitrogen atmosphere was added KF (227.23 mg, 3.91 mmol) at room temperature, and the mixture was heated at 80 °C for 16 hours.
- Step 2 (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H- pyrrolizin-2-yl-2-d 4-methylbenzenesulfonate
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-d-2-ol 400 mg, crude
- DMAP 17.68 mg, 0.14 mmol
- TEA 292.88 mg, 2.89 mmol
- 4-methylbenzene- 1-sulfonyl chloride 275.89 mg, 1.44 mmol.
- Step 3 ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,6-d2)methanol
- Step 3 4-(4-Bromophenyl)-3-oxobutanoic acid [0630] To a stirred solution of tert-butyl 4-(4-bromophenyl)-3-oxobutanoate (12.5 g, 39.91 mmol) in DCM (125 mL) was added TFA (125 mL) dropwise at room temperature. The resulting mixture was stirred at room temperature for 2 hours.
- Step 5 6,8-Dihydroxynaphthalene-2-carbonitrile
- Step 6 6,8-Dihydroxy-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile WSGR Docket No. 62619-720.601
- To a stirred solution of 6,8-dihydroxynaphthalene-2-carbonitrile (4.3 g, 23.22 mmol) and (2-bromoethynyl)triisopropylsilane (6.67 g, 25.54 mmol) in 1,4-dioxane (43 mL) under nitrogen atmosphere were added bis(1-methyl-4-(propan-2-yl)benzene); bis(dichlororuthenium) (1421.99 mg, 2.32 mmol) and KOAc (4.56 g, 46.44 mmol) at room temperature.
- Step 7 8-Hydroxy-6-(methoxymethoxy)-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile
- 6-38 To an ice-cooled solution of 6,8-dihydroxy-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile (4 g, 10.94 mmol) and DIEA (3.54 g, 27.35 mmol) in DCM (80 mL) under nitrogen atmosphere was added bromo(methoxymethoxy)methane (1.37 g, 10.94 mmol) dropwise. The reaction mixture was stirred in an ice bath for 1 hour.
- Step 8 7-Cyano-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate
- DCM 8-Hydroxy-6-(methoxymethoxy)-1-((triisopropylsilyl)ethynyl)-2- naphthonitrile
- Step 9 6-(Methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1- ((triisopropylsilyl)ethynyl)-2-naphthonitrile
- 7-cyano-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate 1.0 g, crude
- bis(pinacolato)diboron 937.64 mg, 3.69 mmol
- 1,4-dioxane 10 mL
- Pd(dppf)Cl 2 •CH 2 Cl 2 150.39 mg, 0.18 mmol
- KOAc 543.56 mg, 5.53 mmol
- Step 2 (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methyl-2- (phenylselanyl)hexahydro-3H-pyrrolizin-3-one
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-3H-pyrrolizin-3-one 550 mg, 1.29 mmol
- LiHMDS 1.5 mL, 1 M in THF
- Step 3 (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methyl-5,6,7,7a- tetrahydro-3H-pyrrolizin-3-one
- (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-methyl-2-(phenylselanyl)hexahydro-3H-pyrrolizin-3-one 500 mg, 0.86 mmol
- CH2Cl2 6 mL
- hydrogen peroxide 1.5 mL, 30wt%) dropwise.
- Step 4 (2R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6-methyl-2,3,5,7a- tetrahydro-1H-pyrrolizine
- 6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methyl-5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one 300 mg, 0.70 mmol
- 9-BBN 6 mL, 3 mmol, 0.5 M in THF
- Step 2 (1R,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-1H- pyrrolizine
- (1R,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)- 1,6-difluorohexahydro-3H-pyrrolizin-3-one 330 mg, 0.768 mmol
- THF 8 mL
- BH 3 - Me2S (0.38 mL, 3.840 mmol
- Step 2 (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-2-methylhexahydro-1H-pyrrolizin-2-ol
- 6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-1H-pyrrolizin-2-ol (414.8 mg, 0.97 mmol) in CH 3 OH (4.2 mL) under nitrogen atmosphere was added NH4F (1437.37 mg, 38.81 mmol) at room temperature. The reaction mixture was heated at 65 °C for 8 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure.
- Step 2 ((6R,7aR)-6-Fluoro-1-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- 6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-1- methylenehexahydro-1H-pyrrolizine (170 mg, 0.41 mmol) and NH4F (614.84 mg, 16.60 mmol) in MeOH (2 mL) under nitrogen atmosphere was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure.
- Step 3 Benzyl (S)-3-(1-hydroxypropyl)-1,4-oxazepane-4-carboxylate [0686] To a solution of (S)-benzyl 3-formyl-1,4-oxazepane-4-carboxylate (810 mg, 3.08 mmol) in THF (10 mL) was stirred at -78 °C for 10 min under N2 atmosphere, and then EtMgBr (3 M, 2 mL) was added dropwise at -78 °C. The resulting mixture was stirred at 20 °C for 1 hr under N 2 atmosphere.
- Step 4 (S)-1-(1,4-Oxazepan-3-yl)propan-1-ol [0688] To a solution of (S)-benzyl 3-(1-hydroxypropyl)-1,4-oxazepane-4-carboxylate (450 mg, 1.53 mmol) in MeOH (10 mL) was added Pd/C (200 mg, 10% purity). The mixture was degassed, purged with H 2 three times, and stirred at 25 °C for 4 hr under H 2 (15 Psi) atmosphere. The reaction mixture was filtered and concentrated to give the title compound (230 mg, yield: 94%) as a colorless oil.
- Step 5 (S)-5-(1-(1,4-Oxazepan-3-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one
- (S)-1-(1,4-oxazepan-3-yl)propan-1-ol (220 mg, 1.38 mmol) in THF (20 mL) was degassed, purged with N2 three times. NaH (221 mg, 5.53 mmol, 60% purity) was added at 0 °C. The mixture was stirred at 0 °C for 0.5 hr.
- Step 6 (8R,8aS)-2,5-Dichloro-8-ethyl-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0692] To a solution of (S)-5-(1-(1,4-oxazepan-3-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (450 mg, 1.15 mmol) in CH2Cl2 (100 mL) was added dropwise DIPEA (1.2 mL, 6.90 mmol) at 0 °C.
- Step 2 (S)-4-Benzyl-3-(chloromethyl)-1,4-oxazepane [0699] To a solution of (R)-(4-benzyl-1,4-oxazepan-3-yl)methanol (4.5 g, 20.3 mmol), TEA (6.17 g, 61.0 mmol) in THF (45 mL) was added MsCl (2.4 g, 21.0 mmol) at -10 °C. The mixture was stirred at 25 °C for 0.5 hr. The reaction mixture was diluted with H 2 O (30 mL) and extracted with EtOAc (20 mL x 3).
- Step 3 (R)-3-(Azidomethyl)-4-benzyl-1,4-oxazepane [0701] To a solution of (S)-4-benzyl-3-(chloromethyl)-1,4-oxazepane (2.9 g, 12.1 mmol) in DMF (30 mL) was added NaN3 (2.3 g, 35.2 mmol). The mixture was stirred at 70 °C for 3 hr. The reaction mixture was diluted with H 2 O (200 mL) and extracted with EtOAc (100 mL x 2).
- Step 4 tert-Butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)carbamate [0703] To a solution of (R)-3-(azidomethyl)-4-benzyl-1,4-oxazepane (2.7 g, 11.0 mmol) in MeOH (30 mL) was added Pd/C (500 mg, 10% purity). The mixture was stirred at 25 °C for 2 hr under H2 (15 Psi). Then the mixture was filtered and concentrated under reduced pressure. Then THF (20 mL), DIPEA (1.46 g, 11.3 mmol) and Boc 2 O (1.98 g, 9.06 mmol) were added.
- Step 5 tert-Butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)(methyl)carbamate
- a solution of tert-butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)carbamate (1.6 g, 4.99 mmol) in THF (5 mL) was degassed and purged with N2 three times. NaH (599 mg, 15.0 mmol, 60% purity) was added at 0 °C, and the mixture was stirred at 25 °C for 0.5 hr under N 2 atmosphere.
- Step 6 tert-Butyl (R)-((1,4-oxazepan-3-yl)methyl)(methyl)carbamate
- tert-butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)(methyl)carbamate 745 mg, 2.23 mmol
- Pd(OH)2 745 mg, 20% purity
- the mixture was stirred at 25 °C for 3 hr under H2 (15 Psi).
- the reaction mixture was filtered and concentrated under reduced pressure to give the title compound (600 mg, yield: 77%) as a colourless oil.
- Step 7 tert-Butyl (R)-((4-(5,7-dichloro-8-fluoro-2-(methylthio)pyrido[4,3-d]pyrimidin-4- yl)-1,4-oxazepan-3-yl)methyl)(methyl)carbamate
- DIPEA 2.5 g, 19.5 mmol
- POCl3 2.8 g, 18.2 mmol
- Step 8 (R)-5-Chloro-4-fluoro-7-methyl-2-(methylthio)-7,8,8a,9,12,13-hexahydro-11H-10- oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalene [0711] To a solution of tert-butyl (R)-((4-(5,7-dichloro-8-fluoro-2-(methylthio)pyrido[4,3- d]pyrimidin-4-yl)-1,4-oxazepan-3-yl)methyl)(methyl)carbamate (800 mg, 1.58 mmol) in MeCN (55 mL) was added HCl/1,4-dioxane (4 M, 9 mL).
- Step 1 Methyl N-(tert-butoxycarbonyl)-O-(tert-butyldiphenylsilyl)-L-serinate
- Step 1 Methyl N-(tert-butoxycarbonyl)-O-(tert-butyldiphenylsilyl)-L-serinate
- Step 2 tert-Butyl (R)-(1-((tert-butyldiphenylsilyl)oxy)-3-hydroxypropan-2-yl-3,3- d 2 )carbamate
- methyl N-(tert-butoxycarbonyl)-O-(tert-butyldiphenylsilyl)-L-serinate 17.1 g, 24.5 mmol
- LiAlD4 3.26 g, 85.8 mmol
- Step 3 tert-Butyl (R)-(1-(benzyloxy)-3-((tert-butyldiphenylsilyl)oxy)propan-2-yl-1,1- d 2 )carbamate WSGR Docket No. 62619-720.601 [0718] To a solution of tert-butyl (R)-(1-((tert-butyldiphenylsilyl)oxy)-3-hydroxypropan-2-yl-3,3- d2)carbamate (7.7 g, 17.8 mmol) in DMF (90 mL) was added NaH (460 mg, 11.5 mmol, 60% purity in oil) at 0 °C under N2 atmosphere.
- Step 4 tert-Butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-1,1-d 2 )carbamate
- tert-butyl (R)-(1-(benzyloxy)-3-((tert-butyldiphenylsilyl)oxy)propan-2-yl- 1,1-d 2 )carbamate 3.5 g, 6.71 mmol
- THF 26 mL
- TBAF 1,3.4 mL, 1 M in THF
- Step 5 tert-Butyl (S)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy-3,3- d2)propanoate
- tert-butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-1,1-d 2 )carbamate 1.7 g, 6.00 mmol
- t-BuOH 18 mL
- Cs2CO3 2.35 g, 7.20 mmol
- tert-butyl acrylate 18.8 g, 146 mmol
- Step 7 (S)-3-((Benzyloxy)methyl-d2)-1,4-oxazepan-5-one [0726] To a solution of (S)-3-(2-amino-3-(benzyloxy)propoxy-3,3-d2)propanoic acid (1.5 g, 5.88 mmol) in CH 2 Cl 2 (150 mL) were added HATU (2.68 g, 7.05 mmol) and TEA (2.97 g, 29.4 mmol). The mixture was stirred at 25 °C for 3 hr. The mixture was diluted with H2O (500 mL) and extracted with CH2Cl2 (500 mL ⁇ 2).
- Step 8 (R)-3-(Hydroxymethyl-d 2 )-1,4-oxazepan-5-one
- (S)-3-((benzyloxy)methyl-d2)-1,4-oxazepan-5-one 900 mg, 3.79 mmol
- CH2Cl2 20 mL
- BCl3 1 M in CH2Cl2, 7.59 mL
- the reaction mixture was stirred at 25 °C for 1 hr.
- the reaction mixture quenched with MeOH (100 mL) and concentrated under reduced pressure.
- the resulting residue neutralized with NH3 H2O (pH ⁇ 7-8).
- Step 9 (R)-(1,4-Oxazepan-3-yl)methan-d2-ol [0730] To a solution of (R)-3-(hydroxymethyl-d 2 )-1,4-oxazepan-5-one (470 mg, 3.19 mmol) in THF (5 mL) was added LiAlH4 (242 mg, 6.39 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 hr. The reaction mixture was quenched with H 2 O (0.24 mL), 15% NaOH (0.24 mL), and H 2 O (0.72 mL).
- Step 10 (S)-5-((1,4-Oxazepan-3-yl)methoxy-d2)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0732] To a solution of NaH (336 mg, 8.41 mmol, 60% purity in oil) in THF (8 mL) was added (R)-(1,4-oxazepan-3-yl)methan-d 2 -ol (280 mg, 2.10 mmol) in THF (3 mL) at 0 °C under N 2 atmosphere.
- Step 11 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-8,8-d 2 .
- Step 2 (S)-5-((1,4-Oxazepan-3-yl-5,5-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one
- (R)-(1,4-oxazepan-3-yl-5,5-d2)methanol 130 mg, 976 ⁇ mol
- NaH 156 mg, 3.90 mmol, 60% purity
- Step 3 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-13,13-d 2 [0743] To a solution of (S)-5-((1,4-oxazepan-3-yl-5,5-d2)methoxy)-2,7-dichloro-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (400 mg, 1.1 mmol) in CH 2 Cl 2 (150 mL) were added DIEA (849 mg, 6.57 mmol) and POCl 3 (839 mg, 5.48 mmol) at 0 °C.
- Step 1 tert-Butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-3,3-d2)carbamate
- (R)-3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propanoic acid 5 g, 16.9 mmol
- LiAlD 4 2.73 g, 59.3 mmol
- the mixture was stirred at 25 °C for 2 hr.
- Step 2 tert- Butyl (R)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy-1,1- d 2 )propanoate
- tert-butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-3,3-d2)carbamate 3. g, 10.9 mmol
- Cs2CO3 4.28 g, 13.1 mmol
- tert-butyl acrylate (26.5 g, 207 mmol). The mixture was stirred at 25 °C for 16 hr.
- Step 3 (R)-3-(2-Amino-3-(benzyloxy)propoxy-1,1-d2)propanoic acid
- tert-butyl (R)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy- 1,1-d 2 )propanoate 3.0 g, 7.53 mmol
- CH 2 Cl 2 35 mL
- TFA 35 mL
- the mixture WSGR Docket No. 62619-720.601 was stirred at 25 °C for 2 hr.
- the reaction mixture was concentrated under reduced pressure.
- the title compound (2.78 g, TFA salt) was obtained as a yellow oil.
- Step 4 (R)-3-((Benzyloxy)methyl)-1,4-oxazepan-5-one-2,2-d2 [0754] To a solution of (R)-3-(2-amino-3-(benzyloxy)propoxy-1,1-d 2 )propanoic acid (2.78 g, 10.9 mmol) in CH2Cl2 (300 mL) were added HATU (4.97 g, 13.1 mmol) and TEA (5.51 g, 54.4 mmol). The mixture was stirred at 25 °C for 3 hr.
- Step 5 (R)-3-(Hydroxymethyl)-1,4-oxazepan-5-one-2,2-d2
- (R)-3-((benzyloxy)methyl)-1,4-oxazepan-5-one-2,2-d2 was added dropwise a solution of BCl 3 (1 M in CH 2 Cl 2 , 10.5 mL) at 0 - 5 °C under N2 over 10 min.
- the mixture was stirred at 25 °C for 1 hr.
- the reaction mixture quenched with MeOH (100 mL) and concentrated under reduced pressure.
- Step 6 (R)-(1,4-Oxazepan-3-yl-2,2-d2)methanol [0758] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-5-one-2,2-d2 (630 mg, 4.28 mmol) in THF (10 mL) was added LiAlH 4 (325 mg, 8.56 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 hr. The reaction mixture was quenched with H2O (0.33 mL), 15% NaOH (0.33 mL), and H 2 O (1 mL).
- Step 7 (S)-5-((1,4-Oxazepan-3-yl-2,2-d 2 )methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0760] To a solution of (R)-(1,4-oxazepan-3-yl-2,2-d2)methanol (300 mg, 2.25 mmol) in THF (20 mL) was added NaH (360 mg, 9.01 mmol, 60% purity) at 0 °C.
- Step 8 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-9,9-d2 [0762] To a solution of (S)-5-((1,4-oxazepan-3-yl-2,2-d 2 )methoxy)-2,7-dichloro-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (780 mg, 2.14 mmol) in CH 2 Cl 2 (130 mL) was added DIEA (2.23 mL, 12.8 mmol) and POCl3 (1 mL, 10.7 mmol).
- Step 2 tert -Butyl (R)-6,6-difluoro-3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate
- a mixture of tert-butyl (R)-3-((benzyloxy)methyl)-6,6-difluoro-1,4-oxazepane-4- 2 MeOH (30 mL) was degassed and purged with H2 for three times, and the mixture was stirred at 25 °C for 3 hr under H 2 atmosphere. The reaction mixture was filtered and concentrated under reduced pressure.
- Step 3 (R)-(6,6-Difluoro-1,4-oxazepan-3-yl)methanol
- a mixture of tert-butyl (R)-6,6-difluoro-3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate (1.34 g, 5.01 mmol) in HCl/EtOAc (2 M, 10.3 mL) was stirred at 25 °C for 0.5 hr.
- the reaction WSGR Docket No. 62619-720.601 mixture was concentrated under reduced pressure.
- the crude product was dissolved in CH 3 CN : MeOH (5 mL : 1 mL).
- Step 5 (S)-2,5-Dichloro-4,12,12-trifluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0775] To a solution of (S)-2,7-dichloro-5-((6,6-difluoro-1,4-oxazepan-3-yl)methoxy)-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (1.1 g, 2.76 mmol) in CH2Cl2 (300 mL) were added DIEA (2.40 mL, 13.7 mmol) and POCl 3 (1.54 mL, 16.5 mmol) at 0 °C.
- Step 2 tert-Butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-iodoazepane-1- carboxylate
- Step 3 tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,6,7-tetrahydro-1H- azepine-1-carboxylate & tert-butyl (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro- 1H-azepine-1-carboxylate [0784] To a solution of tert-butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-iodoazepane-1- carboxylate (5.1 g, 8.59 mmol) in xylene (100 mL) was added DBU (25.5 mL, 169 mmol).
- Step 4 (S)-2-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepine [0787] To a solution of Intermediate 82 (700 mg, 1.50 mmol) in CH 2 Cl 2 (6 mL) was added TFA (2.33 mL, 31.4 mmol). The mixture was stirred at 25 °C for 2 h. The reaction mixture concentrated under reduced pressure.
- Step 2 (S)-4-(7-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepin-1-yl)- 2,5,7-trichloro-8-fluoropyrido[4,3-d]pyrimidine [0789] To a solution of 2,4,5,7-tetrachloro-8-fluoropyrido[4,3-d]pyrimidine (455 mg, 1.59 mmol) in MeCN (8 mL) was added DIPEA (0.83 mL, 4.70 mmol) and (S)-2-(((tert- butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H N 2 , and the mixture was stirred at 25 °C for 16 h.
- Step 3 4-((S)-2-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepin-1-yl)- 5,7-dichloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)pyrido[4,3-d]pyrimidine WSGR Docket No.
- Step 4 (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0793] To a solution of 4-((S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H- azepin-1-yl)-5,7-dichloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- in THF, 2.98 mL) and stirred at 25 °C for 16 h.
- Step 2 (S)-(2,3,6,7-Tetrahydro-1H-azepin-2-yl)methanol
- a mixture of tert-butyl (S)-2-(hydroxymethyl)-2,3,6,7-tetrahydro-1H-azepine-1-carboxylate (369 mg, 1.62 mmol) in HCl (2M in EtOAc, 3 mL) was stirred at 25 °C for 0.5 h. The reaction mixture was concentrated to dryness. The residue was dissolved in MeCN (5mL) and MeOH (1mL), and then K2CO3 was added to adjust the pH to around 8.
- Step 3 (S)-2,7-Dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H)-one [0800] To a solution of (S)-(2,3,6,7-tetrahydro-1H-azepin-2-yl)methanol (200 mg, 1.57 mmol) in THF (5 mL) was added NaH (252 mg, 6.29 mmol, 60% purity) at 0 °C, and the mixture was stirred at 0 °C for 0.5 h under N 2 .
- Step 4 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0802] To a solution of (S)-2,7-dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H 2 Cl 2 (70 mL) were added 3 2. The mixture was stirred at 0 °C for 1 h. The reaction mixture was quenched with sat.
- Step 2 tert-Butyl (S)-2-propionylazepane-1-carboxylate
- a mixture of of tert-butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate (2.10 g, 7.33 mmol) in THF (20 mL) was degassed and purged with N2 three times.
- CH3CH2MgBr (3 M, 7.30 mL) was added dropwise at -78 °C under N 2 atmosphere. The reaction was slowly warmed up to 20 °C and stirred for 1 h. The reaction mixture was quenched with sat.
- Step 3 (S)-1-(Azepan-2-yl)propan-1-one [0811] To a solution of tert-butyl (S)-2-propionylazepane-1-carboxylate (1.54 g, 6.03 mmol) in CH2Cl2 (20 mL) was added HCl/EtOAc (4 M, 10 mL), and the mixture was stirred at 20 °C for 1 h. The reaction mixture was concentrated under reduced pressure to give the title compound (1.15 g, HCl salt) as a white solid, which was used in the next step without further purification.
- Step 5 5-(1-((S)-Azepan-2-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one
- N2 atmosphere a solution of 1-((S)-azepan-2-yl)propan-1-ol (300 mg, 1.91 mmol) in THF (18 mL) was added NaH (305 mg, 7.63 mmol, 60% purity) at 0 °C, and the mixture was stirred at 30 °C for 1 h under N2 atmosphere.
- Step 7 (8S,8aS)-5-Chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene & (8R,8aS)-5-chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0819] A mixture of (8aS)-2-fluorote
- Step 2 tert-Butyl (S)-4,4-difluoro-2-(hydroxymethyl)azepane-1-carboxylate
- tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-4,4-difluoroazepane- 1-carboxylate 753 mg, 1.49 mmol
- THF 10 mL
- TBAF (1 M in THF, 2.24 mL
- Step 5 (S)-2,5-Dichloro-4,10,10-trifluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene WSGR Docket No.
- Step 6 (S)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0832] A solution of (S)-2,5-dichloro-4,10,10-trifluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H (5 mL) was stirred at 110 °C under N2 for 16 h.
- Step 2 8-Bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one O-methyl oxime
- NBS 10.6 g, 59.6 mmol
- Pd(OAc) 2 1.49 g, 6.6 mmol
- the reaction mixture was concentrated under reduced pressure, diluted with H2O (100 mL), and extracted with EtOAc (100 mL x 3).
- Step 3 8-Bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one
- O-methyl oxime 16 g, 56.31 mmol
- HCl 6 M, 160 mL
- EtOAc 200 mL x 3
- Step 4 8-Bromo-2-fluoro-6-methoxy-3,4-dihydronaphthalen-1(2H)-one
- H 2 SO 4 3.36 mL, 63.03 mmol
- SelectFluor 80 g, 225.8 mmol
- Step 3 N-(6-Fluoro-5-(fluoromethoxy)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)-1,1-diphenylmethanimine
- Step 2 tert-Butyl (3S)-8,8-difluoro-3-(hydroxymethyl)-4-azabicyclo[5.1.0]octane-4- carboxylate
- the residue was purified by silica gel flash chromatography (eluent: 0 ⁇ 30% of EtOAc in petroleum ether) to give the title compound (160 mg, 74% yield) as a colorless WSGR Docket No. 62619-720.601 oil.
- Step 3 ((3S)-8,8-Difluoro-4-azabicyclo[5.1.0]octan-3-yl)methanol [0865] To a solution of tert-butyl (3S)-8,8-difluoro-3-(hydroxymethyl)-4-azabicyclo[5.1.0]octane- reaction mixture was concentrated to dryness. The residue was dissolved in MeCN (10 mL) and MeOH (2 mL), and K2CO3 was added to adjust the pH to around 8. The mixture was filtered and concentrated under reduced pressure to give the title compound (90 mg, crude) as a colorless oil, which was used in the next step without further purification.
- Step 4 2,7-Dichloro-5-(((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methoxy)-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one
- ((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methanol 90 mg, 508 2
- the mixture was stirred at 0 °C under N 2 for 0.5 h.
- a solution of Intermediate 1 167 mg, 559 h under N2 atmosphere.
- the reaction mixture was quenched with sat. NaHCO3 aq.
- Step 5 (8aS)-2,5-Dichloro-4,10,10-trifluoro-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa- 1,3,6,12a-tetraazacyclopropa[h]naphtho[1,8-ab]heptalene [0869] To a solution of 2,7-dichloro-5-(((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methoxy)- 8-fluoropyrido[4,3-d]pyrimidin-4(3H 2 Cl 2 (80 mL) was added 3 0 °C under N2 for 2 h.
- Step 6 (8aS)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalene [0871] A mixture of (8aS)-2,5-dichloro-4,10,10-trifluoro-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa- 1,3,6,12a-tetraazacyclopropa[h]naphtho[1,8-ab R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5
- Step 2 (S)-7-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,5,6,7-tetrahydro-2H-azepin-2-one
- (S)-N-(1-((tert-butyldiphenylsilyl)oxy)hex-5-en-2-yl)acrylamide (13.4 g, 32.9 mmol) in CH 2 Cl 2 (1400 mL) was added Grubb's II (2.79 g, 3.29 mmol) at 25 °C under N 2 .
- the mixture was stirred at 40 °C for 1 h under N2.
- the reaction mixture was concentrated under reduced pressure.
- Step 3 (S)-7-(((tert-Butyldiphenylsilyl)oxy)methyl)azepan-2-one [0889] To a solution of (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)-1,5,6,7-tetrahydro-2H-azepin- 2-one (9 g, 23.7 mmol) in MeOH (180 mL) was added 10% Pd/C (4 g, 60% purity) under Ar2. The suspension was degassed and purged with H2 three times. The mixture was stirred at 25 °C for 2 h under H 2 (15 Psi).
- Step 4 (S)-7-(hydroxymethyl)azepan-2-one [0891] To a solution of (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)azepan-2-one (3 g, 7.86 mmol) in THF (30 mL) was added TBAF (7.86 mL, 1 M in THF) at 0 °C. The mixture was stirred at 25 °C for 1 h. The reaction mixture was partitioned between H 2 O (100 mL) and EtOAc (120 mL).
- Step 5 (S)-(Azepan-2-yl-7,7-d 2 )methanol
- (S)-7-(hydroxymethyl)azepan-2-one 1.1 g, 7.68 mmol
- LiAlD4 530 mg, 11.5 mmol
- the mixture was stirred at 0 °C for 2 h under N 2 .
- D 2 O (0.53 mL) was added dropwise to the reaction mixture at 0 °C. 15 wt% NaOH (0.53 mL) was added dropwise and then water (1.5 mL) was added.
- Step 6 (S)-5-((Azepan-2-yl-7,7-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0895] To a solution of (S)-(azepan-2-yl-7,7-d2)methanol (900 mg, 6.86 mmol) in THF (18 mL) was added NaH (1.10 g, 27.4 mmol, 60% purity) at 0 °C under N 2 , and the mixture was stirred for 30 min at 0 °C under N 2 .
- Step 7 (S)-2,5-Dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-13,13-d2 [0897] To a solution of (S)-5-((azepan-2-yl-7,7-d 2 )methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (380 mg, 1.05 mmol) in CH2Cl2 (120 mL) was added DIPEA (811 mg, 6.28 mmol, 1.09 mL) and POCl 3 2 .
- Step 2 2,5-Dichloro-4-fluoro-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
- DIEA 640 mg, 4.96 mmol
- POCl 3 633 mg, 4.13 mmol
- Step 2 tert-Butyl (S)-(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl-1,1-d2)carbamate
- tert-butyl (S)-(1-hydroxypent-4-en-2-yl-1,1-d 2 )carbamate 7.16 g, 35.2 mmol
- CH2Cl2 30 mL
- imidazole 2.40 g, 35.2 mmol
- TBDPSCl 11.62 g, 42.27 mmol
- Step 3 (S)-1-((tert-Butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2-amine [0913] To a solution of tert-butyl (S)-(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl-1,1- d 2 )carbamate (8.6 g, 19.47 mmol) in CH 2 Cl 2 (20 mL) was added TFA (5 mL, 67.3 mmol). The mixture was stirred at 25 °C for 2 h. The reation mixture was concentrated under reduced pressure.
- Step 4 (S)-N-(but-3-en-1-yl-1,1-d2)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2- amine
- (S)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2-amine (2 g, 5.86 mmol) in DMF (40 mL) were added K2CO3 (4.05 g, 29.3 mmol) and NaI (4.39 g, 29.3 mmol).
- Step 5 tert-Butyl (S)-(but-3-en-1-yl-1,1-d 2 )(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl- 1,1-d2)carbamate [0917] To a solution of (S)-N-(but-3-en-1-yl-1,1-d2)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1- d 2 -2-amine (3.48 g, 8.75 mmol) and K 2 CO 3 (4.05 g, 29.3 mmol) in DMF (50 mL) was added Boc2O (2.86 g, 13.1 mmol).
- Step 6 tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl-d2)-2,3,6,7-tetrahydro-1H- azepine-1-carboxylate-7,7-d 2
- tert-butyl (S)-(but-3-en-1-yl-1,1-d 2 )(1-((tert-butyldiphenylsilyl)oxy)pent-4- en-2-yl-1,1-d2)carbamate (2.8 g, 5.63 mmol) in CH2Cl2 (470 mL) under N2 was added Grubb’s II 2 three times and stirred at 40 °C for 1 h under N 2 .
- Step 7 tert-Butyl (S)-2-(hydroxymethyl-d 2 )-2,3,6,7-tetrahydro-1H-azepine-1-carboxylate- 7,7-d2
- tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl-d 2 )-2,3,6,7- tetrahydro-1H-azepine-1-carboxylate-7,7-d 2 (1.6 g, 3.41 mmol) in THF (10 mL) was added TBAF (4.09 mL, 1 M in THF). The mixture was stirred at 25 °C for 16 h.
- Step 8 (S)-(2,3,6,7-Tetrahydro-1H-azepin-2-yl-7,7-d 2 )methan-d 2 -ol WSGR Docket No. 62619-720.601 [0923] To a solution of tert-butyl (S)-2-(hydroxymethyl-d 2 )-2,3,6,7-tetrahydro-1H-azepine-1- carboxylate-7,7-d2 (490 mg, 2.12 mmol) in CH2Cl2 (5 mL) at 25 °C was added HCl (2.40 mL, 2 M in EtOAc), and the mixture was stirred at 25 °C for 1 h. The reaction mixture was concentrated under reduced pressure.
- Step 9 (S)-2,7-Dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7-d2)methoxy- d 2 )pyrido[4,3-d]pyrimidin-4(3H)-one [0925] To a solution of (S)-(2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7-d2)methan-d2-ol (260 mg, 1.98 mmol) in THF (15 mL) and DMF (2 mL) was added NaH (317 mg, 7.93 mmol, 60% purity) at 0 °C under N 2 .
- Step 10 (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-8,8,13,13-d 4 [0927] To a solution of (S)-2,7-dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7- d2)methoxy-d2)pyrido[4,3-d]pyrimidin-4(3H)-one (719 mg, 1.98 mmol) in CH2Cl2 (216 mL) were added DIPEA (2.07 mL,11.9 mmol) and POCl 3 2 , and then the mixture was stirred at 0 °C for 1 h under N2.
- Step 2 5-Allyl-1,2-difluoro-3-(methoxy-d 3 )benzene
- Step 7 4-(2-Bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoic acid [0944] To a solution of tert-butyl 4-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoate (18 g, 41.4 mmol) in CH 2 Cl 2 (50 mL) was added TFA (66.0 mL, 888 mmol).
- Step 8 5-Bromo-6,7-difluoro-8-(methoxy-d3)naphthalene-1,3-diol
- a solution of 4-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoic acid (18 g, 55.2 mmol) in trifluoromethanesulfonic acid (150 mL) was stirred at 20 °C for 16 h.
- the reaction mixture was poured into ice water (200 mL) slowly, and the precipitate was filtered, and the filter cake was washed with water (50 ml x 3), sat. NaHCO3 aq.
- Step 10 6,7-Difluoro-8-(methoxy-d 3 )naphthalene-1,3-diyl bis(trifluoromethanesulfonate) [0950] To a solution of 6,7-difluoro-8-(trideuteriomethoxy)naphthalene-1,3-diol (7.6 g, 33.16 mmol) and DIPEA (35 mL, 199 mmol) in CH2Cl2 (140 mL) was added dropwise Tf2O (22 mL, 133 mmol) at 0 °C under N 2 .
- Step 12 N-(6,7-Difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)-1,1-diphenylmethanimine
- a solution of 3-((diphenylmethylene)amino)-6,7-difluoro-8-(methoxy-d3)naphthalen-1-yl trifluoromethanesulfonate (4 g, 7.63 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2- dioxaborolane) (2.32 g, 9.15 mmol), AcOK (2.25 g, 22.9 mmol)
- Step 13 6,7-Difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-amine
- N-(6,7-difluoro-5-(methoxy-d 3 )-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-2-yl)-1,1-diphenylmethanimine (18 g, 16.1 mmol) in EtOH (135 mL) was added hydroxylamine;hydrochloride (2.24 g, 32.3 mmol) and AcOK (4.75 g, 48.4 mmol).
- Step 1 Methylene-d 2 bis(4-methylbenzenesulfonate) [0959] To a solution of (tosyloxy)silver (15.7 g, 56.3 mmol) in ACN (50 mL) was added dibromomethane-d2 (4.5 g, 25.6 mmol). The mixture was stirred at 81 °C for 16 h under N2. The reaction mixture was filtered to remove all the salts, and the mixture was concentrated. The residue was dissolved in CH2Cl2 (500 mL) and precipitate formed. The precipitate was filtered off to remove all the salts.
- Step 2 Fluoromethyl-d24-methylbenzenesulfonate
- methylene-d 2 bis(4-methylbenzenesulfonate) (2 g, 5.58 mmol)
- ACN 20 mL
- CsF 1.27 g, 8.37 mmol
- hexaetheylene glycol (2.22 mL , 8.37 mmol, 95% purity).
- the mixture was stirred at 81 °C for 16 h under N 2 .
- the reaction mixture was quenched with H 2 O (50 mL) at 25 °C and extracted with EtOAc (50 mL x 3).
- Step 2 Methyl 2-(2-(chloromethyl)allyl)-4-methylenepyrrolidine-2-carboxylate [0970] To an ice-cooled solution of 1-(tert-butyl) 2-methyl 2-(2-(chloromethyl)allyl)-4- methylenepyrrolidine-1,2-dicarboxylate (18 g, 54.57 mmol) in MeCN (180 mL) was added HCl (4 M in 1,4-dioxane, 179.96 mL, 719.84 mmol) under nitrogen atmosphere. The resulting mixture stirred for 1 hour. The resulting mixture was concentrated under reduced pressure to give the title compound (12 g, crude) as a yellow solid.
- Step 3 Methyl 2,6-dimethylenetetrahydro-1H-pyrrolizine-7a(5H)-carboxylate
- Step 4 (2,6-dimethylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
- 2,6-dimethylenetetrahydro-1H-pyrrolizine-7a(5H)-carboxylate 2.3 g, 11.90 mmol
- LiAlH4 11.85 mL, 2 mmol
- the resulting mixture was heated at room temperature for 1 hour.
- the resulting mixture was quenched with water (0.076 mL), NaOH (15% in water, 0.76 mL) and water (0.228 mL) at 0 °C.
- Example 1 5-Ethynyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol
- Step 1 (R)-2,7-Dichloro-8-fluoro-5-(2-(piperidin-2-yl)ethoxy)pyrido[4,3-d]pyrimidin- 4(3H)-one [0981] To a solution of (R)-2-(piperidin-2-yl)ethan-1-ol (288 mg, 2.23 mmol) in THF (30 mL) was added NaH (297 mg, 7.45 mmol, 60% purity) at 0 °C under N 2 atmosphere, and the mixture was stirred for 0.5 h.
- Step 3 (R)-2-Chloro-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de]naphthalene [0985] A mixture of (R)-2,12-dichloro-1-fluoro-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H 2 three times, and then the mixture was stirred at 100 °C for 16 h under N
- Step 4 6-Fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de]naphthalen-2-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [0987] A mixture of (R)-2-chloro-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-t
- Step 5 5-Ethynyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol WSGR Docket No.
- Step 3 (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalene [0996] A mixture of (S)-2,5-dichloro-4-fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (700 mg, 2.13 mmol), ((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol (677.13 mg, 4.25 mmol
- Step 4 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [0998] A mixture of (S)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydr
- Step 5 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1000] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3
- Step 2 5-Ethyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol [1005] To a solution of (R)-2-(8-ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-1-fluoro-12- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-
- Example 6 5-Ethynyl-6-fluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1011] Example 6 was prepared in a similar manner to Example 1.
- Example 7 5-Ethynyl-6-fluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,11,12-tetrahydro-8H-7,10-dioxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1013] Example 7 was prepared in a similar manner to Example 1.
- Step 2 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl dimethylcarbamate [1020] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cycloh
- Step 2 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl isobutyrate [1025] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- WSGR Docket No.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Provided herein are inhibitors of KRAS, pharmaceutical compositions comprising the inhibitory compounds, and methods for using the KRAS inhibitory compounds for the treatment of diseases or disorders.
Description
WSGR Docket No. 62619-720.601 KRAS MODULATORS CROSS REFERENCE [0001] This application claims the benefit of US Provisional Application No.63/486,821, filed February 24, 2023; US Provisional Application No.63/497,819, filed April 24, 2023; US Provisional Application No.63/500,469, filed May 5, 2023; and US Provisional Application No. 63/587,912, filed October 4, 2023; all of which are incorporated herein by reference in their entirety. BACKGROUND [0002] KRAS (Kirsten rat sarcoma viral oncogene homologue) is an oncoprotein that is a part of the RAS/MAPK pathway, and relays signals from outside of the cell to the cell’s nucleus. KRAS protein is a GTPase and involved in cellular signaling such as regulation of cell proliferation. KRAS can activate cellular signaling pathways including, but not limited to, the mitogen-activated protein kinase (MAPK) pathway. KRAS was previously considered un-targetable, but recent studies have shown that targeting codon 12 can lead to therapeutic effects. There remains an unmet need to identify and develop novel compounds for KRAS inhibition. BRIEF SUMMARY OF THE INVENTION [0003] Provided herein are inhibitors of KRAS, pharmaceutical compositions comprising said inhibitory compounds, and methods for using said inhibitory compounds for the treatment of cancer and neoplastic disease. [0004] One embodiment provides a compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system;
WSGR Docket No. 62619-720.601 R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from: (a) –(CH2)m-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) -(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (c) -CH=CH-(CH2)p-O-(CH2)q-; (d) –(CH2)p-CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e) –(CH2)p-CH=CH-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f) -CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (g) –CH=CH-CH=CH-, wherein up to 2 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (h) -C(=CH2)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (i) –(c-C3H4)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (j) –(CH2)p-(c-C3H4)-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; and p is 1-4 and q is 1-4. [0005] One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof:
WSGR Docket No. 62619-720.601
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH2-, or -CD2-; R3 is hydrogen, R2 is alkyl, R4 is alkyl, and R2 and R4 join to form a ring; or R2 is hydrogen, R3 is alkyl, R4 is alkyl, and R3 and R4 join to form a ring; or R2 is hydrogen, and R3 and R4 join to form a -CH2OCH2- group; or R2 is hydrogen, and R3 and R4 join to form an -O- group; and n is 0, 1, 2, 3, 4, 5, or 6. [0006] A compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl;
WSGR Docket No. 62619-720.601 X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from: (a)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b)
wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (d)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
WSGR Docket No. 62619-720.601
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (h) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; R7 is an optionally substituted C1-C4 alkyl; W is selected from oxo, oximo, or optionally substituted alkyl oximo; n is 1 or 2; p is 1-4; and q is 1-4. [0007] One embodiment provides a pharmaceutical composition comprising a compound of Formula (I), (II), (III), or pharmaceutically acceptable salt or solvate thereof, and at least one pharmaceutically acceptable excipient. [0008] One embodiment provides a method of treating cancer in a patient in need thereof comprising administering to the patient a compound of Formula (I), (II), (III), or pharmaceutically acceptable salt or solvate thereof. [0009] One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (II), (III), wherein the KRAS protein is contacted in an in vitro setting. [0010] One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (II), (III), wherein the KRAS protein is contacted in an in vivo setting. BRIEF DESCRIPTION OF THE DRAWINGS [0011] The features of the invention are set forth with particularity in the appended claims. A better understanding of the features of the present invention will be obtained by reference to the following detailed description that sets forth illustrative embodiments, in which the principles of the invention are utilized, and the accompanying drawings of which: [0012] Figure 1 provides additional structures of compounds of Formula (I); [0013] Figure 2 provides additional structures of compounds of Formula (II); [0014] Figure 3 provides additional structures of compounds of Formula (I); [0015] Figure 4 provides additional structures of compounds of Formula (I);
WSGR Docket No. 62619-720.601 [0016] Figure 5 provides additional structures of compounds of Formula (I); [0017] Figure 6 provides additional structures of compounds of Formula (I); [0018] Figure 7 provides additional structures of compounds of Formula (II); [0019] Figure 8 provides additional structures of compounds of Formula (II); [0020] Figure 9 provides additional structures of compounds of Formula (II); and [0021] Figure 10 provides additional structures of compounds of Formula (II). INCORPORATION BY REFERENCE [0022] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference for the specific purposes identified herein. DETAILED DESCRIPTION OF THE INVENTION [0023] As used herein and in the appended claims, the singular forms “a,” “and,” and “the” include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to “an agent” includes a plurality of such agents, and reference to “the cell” includes reference to one or more cells (or to a plurality of cells) and equivalents thereof known to those skilled in the art, and so forth. When ranges are used herein for physical properties, such as molecular weight, or chemical properties, such as chemical formulae, all combinations and sub-combinations of ranges and specific embodiments therein are intended to be included. The term “about” when referring to a number or a numerical range means that the number or numerical range referred to is an approximation within experimental variability (or within statistical experimental error), and thus the number or numerical range, in some instances, will vary between 1% and 15% of the stated number or numerical range. The term “comprising” (and related terms such as “comprise” or “comprises” or “having” or “including”) is not intended to exclude that in other certain embodiments, for example, an embodiment of any composition of matter, composition, method, or process, or the like, described herein, “consist of” or “consist essentially of” the described features. Definitions [0024] As used in the specification and appended claims, unless specified to the contrary, the following terms have the meaning indicated below. [0025] “Amino” refers to the –NH2 radical. [0026] “Cyano” refers to the -CN radical. [0027] “Nitro” refers to the -NO2 radical. [0028] “Oxa” refers to the -O- radical. [0029] “Oxo” refers to the =O radical.
WSGR Docket No. 62619-720.601 [0030] “Thioxo” refers to the =S radical. [0031] “Imino” refers to the =N-H radical. [0032] “Oximo” refers to the =N-OH radical. [0033] “Hydrazino” refers to the =N-NH2 radical. [0034] “Alkyl” refers to a straight or branched hydrocarbon chain radical consisting solely of carbon and hydrogen atoms, containing no unsaturation, having from one to fifteen carbon atoms (e.g., C1-C15 alkyl). In certain embodiments, an alkyl comprises one to thirteen carbon atoms (e.g., C1-C13 alkyl). In certain embodiments, an alkyl comprises one to eight carbon atoms (e.g., C1-C8 alkyl). In other embodiments, an alkyl comprises one to five carbon atoms (e.g., C1-C5 alkyl). In other embodiments, an alkyl comprises one to four carbon atoms (e.g., C1-C4 alkyl). In other embodiments, an alkyl comprises one to three carbon atoms (e.g., C1-C3 alkyl). In other embodiments, an alkyl comprises one to two carbon atoms (e.g., C1-C2 alkyl). In other embodiments, an alkyl comprises one carbon atom (e.g., C1 alkyl). In other embodiments, an alkyl comprises five to fifteen carbon atoms (e.g., C5-C15 alkyl). In other embodiments, an alkyl comprises five to eight carbon atoms (e.g., C5-C8 alkyl). In other embodiments, an alkyl comprises two to five carbon atoms (e.g., C2-C5 alkyl). In other embodiments, an alkyl comprises three to five carbon atoms (e.g., C3-C5 alkyl). In other embodiments, the alkyl group is selected from methyl, ethyl, 1-propyl (n-propyl), 1-methylethyl (iso-propyl), 1-butyl (n-butyl), 1-methylpropyl (sec- butyl), 2-methylpropyl (iso-butyl), 1,1-dimethylethyl (tert-butyl), 1-pentyl (n-pentyl). The alkyl is attached to the rest of the molecule by a single bond. Unless stated otherwise specifically in the specification, an alkyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -ORa, -SRa, -OC(O)-Ra, -N(Ra)2, - C(O)Ra, -C(O)ORa, -C(O)N(Ra)2, -N(Ra)C(O)ORa, -OC(O)-N(Ra)2, -N(Ra)C(O)Ra, -N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, oxo or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, oxo or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or
WSGR Docket No. 62619-720.601 trifluoromethyl). In certain embodiments, an optionally substituted alkyl is a haloalkyl. In other embodiments, an optionally substituted alkyl is a fluoroalkyl. In other embodiments, an optionally substituted alkyl is a -CF3 group. [0035] “Alkoxy” refers to a radical bonded through an oxygen atom of the formula –O-alkyl, where alkyl is an alkyl chain as defined above. [0036] “Alkenyl” refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon double bond, and having from two to twelve carbon atoms. In certain embodiments, an alkenyl comprises two to eight carbon atoms. In other embodiments, an alkenyl comprises two to four carbon atoms. The alkenyl is attached to the rest of the molecule by a single bond, for example, ethenyl (i.e., vinyl), prop-1-enyl (i.e., allyl), but-1-enyl, pent-1-enyl, penta-1,4-dienyl, and the like. Unless stated otherwise specifically in the specification, an alkenyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -ORa, -SRa, - OC(O)-Ra, -N(Ra)2, -C(O)Ra, -C(O)ORa, -C(O)N(Ra)2, -N(Ra)C(O)ORa, -OC(O)-N(Ra)2, - N(Ra)C(O)Ra, -N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl). [0037] “Alkynyl” refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon triple bond, having from two to twelve carbon atoms. In certain embodiments, an alkynyl comprises two to eight carbon atoms. In other embodiments, an alkynyl comprises two to six carbon atoms. In other embodiments, an alkynyl comprises two to four carbon atoms. The alkynyl is attached to the rest of the molecule by a single bond, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, and the like. Unless stated otherwise specifically in the specification, an alkynyl group is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo,
WSGR Docket No. 62619-720.601 trimethylsilanyl, -ORa, -SRa, -OC(O)-Ra, -N(Ra)2, -C(O)Ra, -C(O)ORa, -C(O)N(Ra)2, - N(Ra)C(O)ORa, -OC(O)-N(Ra)2, -N(Ra)C(O)Ra, -N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl). [0038] “Alkylene” or “alkylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing no unsaturation, and having from one to twelve carbon atoms, for example, methylene, ethylene, propylene, n-butylene, and the like. The alkylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. The points of attachment of the alkylene chain to the rest of the molecule and to the radical group are through one carbon in the alkylene chain or through any two carbons within the chain. In certain embodiments, an alkylene comprises one to eight carbon atoms (e.g., C1-C8 alkylene). In other embodiments, an alkylene comprises one to five carbon atoms (e.g., C1-C5 alkylene). In other embodiments, an alkylene comprises one to four carbon atoms (e.g., C1-C4 alkylene). In other embodiments, an alkylene comprises one to three carbon atoms (e.g., C1-C3 alkylene). In other embodiments, an alkylene comprises one to two carbon atoms (e.g., C1-C2 alkylene). In other embodiments, an alkylene comprises one carbon atom (e.g., C1 alkylene). In other embodiments, an alkylene comprises five to eight carbon atoms (e.g., C5-C8 alkylene). In other embodiments, an alkylene comprises two to five carbon atoms (e.g., C2-C5 alkylene). In other embodiments, an alkylene comprises three to five carbon atoms (e.g., C3-C5 alkylene). Unless stated otherwise specifically in the specification, an alkylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -ORa, -SRa, -OC(O)- Ra, -N(Ra)2, -C(O)Ra, -C(O)ORa, -C(O)N(Ra)2, -N(Ra)C(O)ORa, -OC(O)-N(Ra)2, -N(Ra)C(O)Ra, - N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally
WSGR Docket No. 62619-720.601 substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl). [0039] “Alkenylene” or “alkenylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon double bond, and having from two to twelve carbon atoms. The alkenylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. In certain embodiments, an alkenylene comprises two to eight carbon atoms (e.g., C2-C8 alkenylene). In other embodiments, an alkenylene comprises two to five carbon atoms (e.g., C2-C5 alkenylene). In other embodiments, an alkenylene comprises two to four carbon atoms (e.g., C2-C4 alkenylene). In other embodiments, an alkenylene comprises two to three carbon atoms (e.g., C2-C3 alkenylene). In other embodiments, an alkenylene comprises two carbon atoms (e.g., C2 alkenylene). In other embodiments, an alkenylene comprises five to eight carbon atoms (e.g., C5-C8 alkenylene). In other embodiments, an alkenylene comprises three to five carbon atoms (e.g., C3-C5 alkenylene). Unless stated otherwise specifically in the specification, an alkenylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -ORa, -SRa, -OC(O)-Ra, -N(Ra)2, -C(O)Ra, - C(O)ORa, -C(O)N(Ra)2, -N(Ra)C(O)ORa, -OC(O)-N(Ra)2, -N(Ra)C(O)Ra, -N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or
WSGR Docket No. 62619-720.601 trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl). [0040] “Alkynylene” or “alkynylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon triple bond, and having from two to twelve carbon atoms. The alkynylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. In certain embodiments, an alkynylene comprises two to eight carbon atoms (e.g., C2-C8 alkynylene). In other embodiments, an alkynylene comprises two to five carbon atoms (e.g., C2-C5 alkynylene). In other embodiments, an alkynylene comprises two to four carbon atoms (e.g., C2-C4 alkynylene). In other embodiments, an alkynylene comprises two to three carbon atoms (e.g., C2-C3 alkynylene). In other embodiments, an alkynylene comprises two carbon atoms (e.g., C2 alkynylene). In other embodiments, an alkynylene comprises five to eight carbon atoms (e.g., C5-C8 alkynylene). In other embodiments, an alkynylene comprises three to five carbon atoms (e.g., C3-C5 alkynylene). Unless stated otherwise specifically in the specification, an alkynylene chain is optionally substituted by one or more of the following substituents: halo, cyano, nitro, oxo, thioxo, imino, oximo, trimethylsilanyl, -ORa, -SRa, -OC(O)-Ra, -N(Ra)2, -C(O)Ra, - C(O)ORa, -C(O)N(Ra)2, -N(Ra)C(O)ORa, -OC(O)-N(Ra)2, -N(Ra)C(O)Ra, -N(Ra)S(O)tRa (where t is 1 or 2), -S(O)tORa (where t is 1 or 2), -S(O)tRa (where t is 1 or 2) and -S(O)tN(Ra)2 (where t is 1 or 2) where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, carbocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), carbocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl). [0041] “Aryl” refers to a radical derived from an aromatic monocyclic or multicyclic hydrocarbon ring system by removing a hydrogen atom from a ring carbon atom. The aromatic monocyclic or multicyclic hydrocarbon ring system contains only hydrogen and carbon from five to eighteen carbon atoms, where at least one of the rings in the ring system is fully unsaturated, i.e., it contains a cyclic, delocalized (4n+2) –electron system in accordance with the Hückel theory. The ring
WSGR Docket No. 62619-720.601 system from which aryl groups are derived include, but are not limited to, groups such as benzene, fluorene, indane, indene, tetralin and naphthalene. Unless stated otherwise specifically in the specification, the term “aryl” or the prefix “ar-“ (such as in “aralkyl”) is meant to include aryl radicals optionally substituted by one or more substituents independently selected from optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, halo, cyano, nitro, - Rb-ORa, -Rb-OC(O)-Ra, -Rb-OC(O)-ORa, -Rb-OC(O)-N(Ra)2, -Rb-N(Ra)2, -Rb-C(O)Ra, -Rb- C(O)ORa, -Rb-C(O)N(Ra)2, -Rb-O-Rc-C(O)N(Ra)2, -Rb-N(Ra)C(O)ORa, -Rb-N(Ra)C(O)Ra, -Rb- N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb-S(O)tN(Ra)2 (where t is 1 or 2), where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, cycloalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), cycloalkylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), each Rb is independently a direct bond or a straight or branched alkylene or alkenylene chain, and Rc is a straight or branched alkylene or alkenylene chain, and where each of the Ra, Rb, or Rc substituents is unsubstituted unless otherwise indicated. [0042] “Aralkyl” refers to a radical of the formula -Rc-aryl where Rc is an alkylene chain as defined above, for example, methylene, ethylene, and the like. The alkylene chain part of the aralkyl radical is optionally substituted as described above for an alkylene chain. The aryl part of the aralkyl radical is optionally substituted as described above for an aryl group. [0043] “Aralkenyl” refers to a radical of the formula –Rd-aryl where Rd is an alkenylene chain as defined above. The aryl part of the aralkenyl radical is optionally substituted as described above for an aryl group. The alkenylene chain part of the aralkenyl radical is optionally substituted as defined above for an alkenylene group. [0044] “Aralkynyl” refers to a radical of the formula -Re-aryl, where Re is an alkynylene chain as defined above. The aryl part of the aralkynyl radical is optionally substituted as described above for an aryl group. The alkynylene chain part of the aralkynyl radical is optionally substituted as defined above for an alkynylene chain.
WSGR Docket No. 62619-720.601 [0045] “Aralkoxy” refers to a radical bonded through an oxygen atom of the formula -O-Rc-aryl where Rc is an alkylene chain as defined above, for example, methylene, ethylene, and the like. The alkylene chain part of the aralkyl radical is optionally substituted as described above for an alkylene chain. The aryl part of the aralkyl radical is optionally substituted as described above for an aryl group. [0046] “Carbocyclyl” refers to a stable non-aromatic monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, having from three to fifteen carbon atoms. In certain embodiments, a carbocyclyl comprises three to ten carbon atoms. In other embodiments, a carbocyclyl comprises five to seven carbon atoms. The carbocyclyl is attached to the rest of the molecule by a single bond. Carbocyclyl is saturated (i.e., containing single C-C bonds only) or unsaturated (i.e., containing one or more double bonds or triple bonds). A fully saturated carbocyclyl radical is also referred to as “cycloalkyl.” Examples of monocyclic cycloalkyls include, e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl. An unsaturated carbocyclyl is also referred to as “cycloalkenyl.” Examples of monocyclic cycloalkenyls include, e.g., cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl. Polycyclic carbocyclyl radicals include, for example, adamantyl, norbornyl (i.e., bicyclo[2.2.1]heptanyl), norbornenyl, decalinyl, 7,7-dimethyl-bicyclo[2.2.1]heptanyl, and the like. Unless otherwise stated specifically in the specification, the term “carbocyclyl” is meant to include carbocyclyl radicals that are optionally substituted by one or more substituents independently selected from optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted exocyclic alkylidene, halo, oxo, thioxo, cyano, nitro, -Rb-ORa, -Rb- OC(O)-Ra, -Rb-OC(O)-ORa, -Rb-OC(O)-N(Ra)2, -Rb-N(Ra)2, -Rb-C(O)Ra, -Rb-C(O)ORa, -Rb- C(O)N(Ra)2, -Rb-O-Rc-C(O)N(Ra)2, -Rb-N(Ra)C(O)ORa, -Rb-N(Ra)C(O)Ra, -Rb-N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb- S(O)tN(Ra)2 (where t is 1 or 2), where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, cycloalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), cycloalkylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or
WSGR Docket No. 62619-720.601 trifluoromethyl), each Rb is independently a direct bond or a straight or branched alkylene or alkenylene chain, and Rc is a straight or branched alkylene or alkenylene chain, and where each of the Ra, Rb, or Rc substituents is unsubstituted unless otherwise indicated. [0047] “Carbocyclylalkyl” refers to a radical of the formula –Rc-carbocyclyl where Rc is an alkylene chain as defined above. The alkylene chain and the carbocyclyl radical is optionally substituted as defined above. [0048] “Carbocyclylalkynyl” refers to a radical of the formula –Rc-carbocyclyl where Rc is an alkynylene chain as defined above. The alkynylene chain and the carbocyclyl radical is optionally substituted as defined above. [0049] “Carbocyclylalkoxy” refers to a radical bonded through an oxygen atom of the formula –O- Rc-carbocyclyl where Rc is an alkylene chain as defined above. The alkylene chain and the carbocyclyl radical is optionally substituted as defined above. [0050] “Halo” or “halogen” refers to bromo, chloro, fluoro or iodo substituents. [0051] “Fluoroalkyl” refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, as defined above, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, 1-fluoromethyl-2-fluoroethyl, and the like. In some embodiments, the alkyl part of the fluoroalkyl radical is optionally substituted as defined above for an alkyl group. [0052] “Heterocyclyl” refers to a stable 3- to 18-membered non-aromatic ring radical that comprises two to twelve carbon atoms and from one to six heteroatoms selected from nitrogen, oxygen and sulfur. Unless stated otherwise specifically in the specification, the heterocyclyl radical is a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which optionally includes fused or bridged ring systems. The heteroatoms in the heterocyclyl radical are optionally oxidized. One or more nitrogen atoms, if present, are optionally quaternized. The heterocyclyl radical is partially or fully saturated. The heterocyclyl is attached to the rest of the molecule through any atom of the ring(s). Examples of such heterocyclyl radicals include, but are not limited to, dioxolanyl, thienyl[1,3]dithianyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isothiazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydropyranyl, thiomorpholinyl, thiamorpholinyl, 1-oxo-thiomorpholinyl, and 1,1-dioxo-thiomorpholinyl. Unless stated otherwise specifically in the specification, the term “heterocyclyl” is meant to include heterocyclyl radicals as defined above that are optionally substituted by one or more substituents selected from optionally substituted alkyl, optionally
WSGR Docket No. 62619-720.601 substituted alkenyl, optionally substituted alkynyl, optionally substituted exocyclic alkylidene, halo, fluoroalkyl, oxo, thioxo, cyano, nitro, -Rb-ORa, -Rb-OC(O)-Ra, -Rb-OC(O)-ORa, -Rb-OC(O)- N(Ra)2, -Rb-N(Ra)2, -Rb-C(O)Ra, -Rb-C(O)ORa, -Rb-C(O)N(Ra)2, -Rb-O-Rc-C(O)N(Ra)2, -Rb- N(Ra)C(O)ORa, -Rb-N(Ra)C(O)Ra, -Rb-N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb-S(O)tN(Ra)2 (where t is 1 or 2), where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, cycloalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), cycloalkylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), each Rb is independently a direct bond or a straight or branched alkylene or alkenylene chain, and Rc is a straight or branched alkylene or alkenylene chain, and where each of the Ra, Rb, or Rc substituents is unsubstituted unless otherwise indicated. [0053] “N-heterocyclyl” or “N-attached heterocyclyl” refers to a heterocyclyl radical as defined above containing at least one nitrogen and where the point of attachment of the heterocyclyl radical to the rest of the molecule is through a nitrogen atom in the heterocyclyl radical. An N-heterocyclyl radical is optionally substituted as described above for heterocyclyl radicals. Examples of such N-heterocyclyl radicals include, but are not limited to, 1-morpholinyl, 1- piperidinyl, 1-piperazinyl, 1-pyrrolidinyl, pyrazolidinyl, and imidazolidinyl. [0054] “C-heterocyclyl” or “C-attached heterocyclyl” refers to a heterocyclyl radical as defined above containing at least one heteroatom and where the point of attachment of the heterocyclyl radical to the rest of the molecule is through a carbon atom in the heterocyclyl radical. A C-heterocyclyl radical is optionally substituted as described above for heterocyclyl radicals. Examples of such C-heterocyclyl radicals include, but are not limited to, 2-morpholinyl, 2- or 3- or 4-piperidinyl, 2-piperazinyl, 2- or 3-pyrrolidinyl, and the like. [0055] “Heterocyclylalkyl” refers to a radical of the formula –Rc-heterocyclyl where Rc is an alkylene chain as defined above. If the heterocyclyl is a nitrogen-containing heterocyclyl, the heterocyclyl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heterocyclylalkyl radical is optionally substituted as defined above for an alkylene chain. The
WSGR Docket No. 62619-720.601 heterocyclyl part of the heterocyclylalkyl radical is optionally substituted as defined above for a heterocyclyl group. [0056] “Heterocyclylalkoxy” refers to a radical bonded through an oxygen atom of the formula –O- Rc-heterocyclyl where Rc is an alkylene chain as defined above. If the heterocyclyl is a nitrogen-containing heterocyclyl, the heterocyclyl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heterocyclylalkoxy radical is optionally substituted as defined above for an alkylene chain. The heterocyclyl part of the heterocyclylalkoxy radical is optionally substituted as defined above for a heterocyclyl group. [0057] “Heteroaryl” refers to a radical derived from a 3- to 18-membered aromatic ring radical that comprises two to seventeen carbon atoms and from one to six heteroatoms selected from nitrogen, oxygen, and sulfur. As used herein, the heteroaryl radical is a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, wherein at least one of the rings in the ring system is fully unsaturated, i.e., it contains a cyclic, delocalized (4n+2) –electron system in accordance with the Hückel theory. Heteroaryl includes fused or bridged ring systems. The heteroatom(s) in the heteroaryl radical is optionally oxidized. One or more nitrogen atoms, if present, are optionally quaternized. The heteroaryl is attached to the rest of the molecule through any atom of the ring(s). Examples of heteroaryls include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzindolyl, 1,3-benzodioxolyl, benzofuranyl, benzooxazolyl, benzo[d]thiazolyl, benzothiadiazolyl, benzo[b][1,4]dioxepinyl, benzo[b][1,4]oxazinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzoxazolyl, benzodioxolyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothiophenyl), benzothieno[3,2-d]pyrimidinyl, benzotriazolyl, benzo[4,6]imidazo[1,2-a]pyridinyl, carbazolyl, cinnolinyl, cyclopenta[d]pyrimidinyl, 6,7-dihydro-5H-cyclopenta[4,5]thieno[2,3-d]pyrimidinyl, 5,6-dihydrobenzo[h]quinazolinyl, 5,6-dihydrobenzo[h]cinnolinyl, 6,7-dihydro-5H-benzo[6,7]cyclohepta[1,2-c]pyridazinyl, dibenzofuranyl, dibenzothiophenyl, furanyl, furanonyl, furo[3,2-c]pyridinyl, 5,6,7,8,9,10-hexahydrocycloocta[d]pyrimidinyl, 5,6,7,8,9,10-hexahydrocycloocta[d]pyridazinyl, 5,6,7,8,9,10-hexahydrocycloocta[d]pyridinyl, isothiazolyl, imidazolyl, indazolyl, indolyl, indazolyl, isoindolyl, indolinyl, isoindolinyl, isoquinolyl, indolizinyl, isoxazolyl, 5,8-methano-5,6,7,8-tetrahydroquinazolinyl, naphthyridinyl, 1,6-naphthyridinonyl, oxadiazolyl, 2-oxoazepinyl, oxazolyl, oxiranyl, 5,6,6a,7,8,9,10,10a-octahydrobenzo[h]quinazolinyl, 1-phenyl-1H-pyrrolyl, phenazinyl, phenothiazinyl, phenoxazinyl, phthalazinyl, pteridinyl, purinyl, pyrrolyl, pyrazolyl, pyrazolo[3,4-d]pyrimidinyl, pyridinyl, pyrido[3,2-d]pyrimidinyl, pyrido[3,4-d]pyrimidinyl, pyrazinyl, pyrimidinyl, pyridazinyl, pyrrolyl, quinazolinyl, quinoxalinyl,
WSGR Docket No. 62619-720.601 quinolinyl, isoquinolinyl, tetrahydroquinolinyl, 5,6,7,8-tetrahydroquinazolinyl, 5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidinyl, 6,7,8,9-tetrahydro-5H-cyclohepta[4,5]thieno[2,3-d]pyrimidinyl, 5,6,7,8-tetrahydropyrido[4,5-c]pyridazinyl, thiazolyl, thiadiazolyl, triazolyl, tetrazolyl, triazinyl, thieno[2,3-d]pyrimidinyl, thieno[3,2-d]pyrimidinyl, thieno[2,3-c]pridinyl, and thiophenyl (i.e. thienyl). Unless stated otherwise specifically in the specification, the term “heteroaryl” is meant to include heteroaryl radicals as defined above which are optionally substituted by one or more substituents selected from optionally substituted alkyl, optionally substituted cycloalkylalkyl, optionally substituted heterocyclylalkyl, optionally substituted alkenyl, optionally substituted alkynyl, halo, optionally substituted fluoroalkyl, optionally substituted haloalkenyl, optionally substituted haloalkynyl, oxo, thioxo, cyano, nitro, -Rb-ORa, -Rb-OC(O)-Ra, -Rb-OC(O)-ORa, -Rb- OC(O)-N(Ra)2, -Rb-N(Ra)2, -Rb-C(O)Ra, -Rb-C(O)ORa, -Rb-C(O)N(Ra)2, -Rb-O-Rc-C(O)N(Ra)2, - Rb-N(Ra)C(O)ORa, -Rb-N(Ra)C(O)Ra, -Rb-N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb-S(O)tN(Ra)2 (where t is 1 or 2), where each Ra is independently hydrogen, alkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), fluoroalkyl, cycloalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), cycloalkylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), aralkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heterocyclylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), heteroaryl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), or heteroarylalkyl (optionally substituted with halogen, hydroxy, methoxy, or trifluoromethyl), each Rb is independently a direct bond or a straight or branched alkylene or alkenylene chain, and Rc is a straight or branched alkylene or alkenylene chain, and where each of the Ra, Rb, or Rc substituents is unsubstituted unless otherwise indicated. [0058] “N-heteroaryl” refers to a heteroaryl radical as defined above containing at least one nitrogen and where the point of attachment of the heteroaryl radical to the rest of the molecule is through a nitrogen atom in the heteroaryl radical. An N-heteroaryl radical is optionally substituted as described above for heteroaryl radicals. [0059] “C-heteroaryl” refers to a heteroaryl radical as defined above and where the point of attachment of the heteroaryl radical to the rest of the molecule is through a carbon atom in the
WSGR Docket No. 62619-720.601 heteroaryl radical. A C-heteroaryl radical is optionally substituted as described above for heteroaryl radicals. [0060] “Heteroarylalkyl” refers to a radical of the formula –Rc-heteroaryl, where Rc is an alkylene chain as defined above. If the heteroaryl is a nitrogen-containing heteroaryl, the heteroaryl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heteroarylalkyl radical is optionally substituted as defined above for an alkylene chain. The heteroaryl part of the heteroarylalkyl radical is optionally substituted as defined above for a heteroaryl group. [0061] “Heteroarylalkoxy” refers to a radical bonded through an oxygen atom of the formula –O- Rc-heteroaryl, where Rc is an alkylene chain as defined above. If the heteroaryl is a nitrogen-containing heteroaryl, the heteroaryl is optionally attached to the alkyl radical at the nitrogen atom. The alkylene chain of the heteroarylalkoxy radical is optionally substituted as defined above for an alkylene chain. The heteroaryl part of the heteroarylalkoxy radical is optionally substituted as defined above for a heteroaryl group. [0062] The compounds disclosed herein, in some embodiments, contain one or more asymmetric centers and thus give rise to enantiomers, diastereomers, and other stereoisomeric forms that are defined, in terms of absolute stereochemistry, as (R)- or (S)-. Unless stated otherwise, it is intended that all stereoisomeric forms of the compounds disclosed herein are contemplated by this disclosure. When the compounds described herein contain alkene double bonds, and unless specified otherwise, it is intended that this disclosure includes both E and Z geometric isomers (e.g., cis or trans.) Likewise, all possible isomers, as well as their racemic and optically pure forms, and all tautomeric forms are also intended to be included. The term “geometric isomer” refers to E or Z geometric isomers (e.g., cis or trans) of an alkene double bond. The term “positional isomer” refers to structural isomers around a central ring, such as ortho-, meta-, and para- isomers around a benzene ring. [0063] A “tautomer” refers to a molecule wherein a proton shift from one atom of a molecule to another atom of the same molecule is possible. The compounds presented herein, in certain embodiments, exist as tautomers. In circumstances where tautomerization is possible, a chemical equilibrium of the tautomers will exist. The exact ratio of the tautomers depends on several factors, including physical state, temperature, solvent, and pH. Some examples of tautomeric equilibrium include:
WSGR Docket No. 62619-720.601
[0064] The compounds disclosed herein, in some embodiments, are used in different enriched isotopic forms, e.g., enriched in the content of 2H, 3H, 11C, 13C and/or 14C. In one particular embodiment, the compound is deuterated in at least one position. Such deuterated forms can be made by the procedure described in, for example, U.S. Patent Nos.5,846,514 and 6,334,997. As described in U.S. Patent Nos.5,846,514 and 6,334,997, deuteration can, in some instances, improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs. [0065] Unless otherwise stated, structures depicted herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13C- or 14C-enriched carbon are within the scope of the present disclosure. [0066] The compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds. For example, the compounds may be labeled with isotopes, such as for example, deuterium (2H), tritium (3H), iodine-125 (125I) or carbon-14 (14C). Isotopic substitution with 2H, 11C, 13C, 14C, 15C, 12N, 13N, 15N, 16N, 16O, 17O, 14F, 15F, 16F, 17F, 18F, 33S, 34S, 35S, 36S, 35Cl, 37Cl, 79Br, 81Br, 125I are all contemplated. In some embodiments, isotopic substitution with 18F is contemplated. All isotopic variations of the compounds of the present invention, whether radioactive or not, are encompassed within the scope of the present invention. [0067] In certain embodiments, the compounds disclosed herein have some or all of the 1H atoms replaced with 2H atoms. The methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
WSGR Docket No. 62619-720.601 [0068] Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S. The Synthesis of Radiolabeled Compounds via Organometallic Intermediates, Tetrahedron, 1989, 45(21), 6601-21; and Evans, E. Anthony. Synthesis of radiolabeled compounds, J. Radioanal. Chem., 1981, 64(1-2), 9-32. [0069] Deuterated starting materials are readily available and are subjected to the synthetic methods described herein to provide for the synthesis of deuterium-containing compounds. Large numbers of deuterium-containing reagents and building blocks are available commercially from chemical vendors, such as Aldrich Chemical Co. [0070] Deuterium-transfer reagents suitable for use in nucleophilic substitution reactions, such as iodomethane-d3 (CD3I), are readily available and may be employed to transfer a deuterium- substituted carbon atom under nucleophilic substitution reaction conditions to the reaction substrate. The use of CD3I is illustrated, by way of example only, in the reaction schemes below.
[0071] Deuterium-transfer reagents, such as lithium aluminum deuteride (LiAlD4), are employed to transfer deuterium under reducing conditions to the reaction substrate. The use of LiAlD4 is illustrated, by way of example only, in the reaction schemes below.
[0072] Deuterium gas and palladium catalyst are employed to reduce unsaturated carbon-carbon linkages and to perform a reductive substitution of aryl carbon-halogen bonds as illustrated, by way of example only, in the reaction schemes below.
WSGR Docket No. 62619-720.601 [0073] In one embodiment, the compounds disclosed herein contain one deuterium atom. In another embodiment, the compounds disclosed herein contain two deuterium atoms. In another embodiment, the compounds disclosed herein contain three deuterium atoms. In another embodiment, the compounds disclosed herein contain four deuterium atoms. In another embodiment, the compounds disclosed herein contain five deuterium atoms. In another embodiment, the compounds disclosed herein contain six deuterium atoms. In another embodiment, the compounds disclosed herein contain more than six deuterium atoms. In another embodiment, the compound disclosed herein is fully substituted with deuterium atoms and contains no non-exchangeable 1H hydrogen atoms. In one embodiment, the level of deuterium incorporation is determined by synthetic methods in which a deuterated synthetic building block is used as a starting material. [0074] “Pharmaceutically acceptable salt” includes both acid and base addition salts. A pharmaceutically acceptable salt of any one of the KRAS inhibitory compounds described herein is intended to encompass any and all pharmaceutically suitable salt forms. Preferred pharmaceutically acceptable salts of the compounds described herein are pharmaceutically acceptable acid addition salts and pharmaceutically acceptable base addition salts. [0075] “Pharmaceutically acceptable acid addition salt” refers to those salts which retain the biological effectiveness and properties of the free bases, which are not biologically or otherwise undesirable, and which are formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, hydroiodic acid, hydrofluoric acid, phosphorous acid, and the like. Also included are salts that are formed with organic acids such as aliphatic mono- and dicarboxylic acids, phenyl-substituted alkanoic acids, hydroxy alkanoic acids, alkanedioic acids, aromatic acids, aliphatic and. Aromatic sulfonic acids, etc. and include, for example, acetic acid, trifluoroacetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid, salicylic acid, and the like. Exemplary salts thus include sulfates, pyrosulfates, bisulfates, sulfites, bisulfites, nitrates, phosphates, monohydrogenphosphates, dihydrogenphosphates, metaphosphates, pyrophosphates, chlorides, bromides, iodides, acetates, trifluoroacetates, propionates, caprylates, isobutyrates, oxalates, malonates, succinate suberates, sebacates, fumarates, maleates, mandelates, benzoates, chlorobenzoates, methylbenzoates, dinitrobenzoates, phthalates, benzenesulfonates, toluenesulfonates, phenylacetates, citrates, lactates, malates, tartrates, methanesulfonates, and the like. Also contemplated are salts of amino acids, such as arginates, gluconates, and galacturonates (see, for example, Berge S.M. et al.,
WSGR Docket No. 62619-720.601 “Pharmaceutical Salts,” Journal of Pharmaceutical Science, 66:1-19 (1997)). Acid addition salts of basic compounds are, in some embodiments, prepared by contacting the free base forms with a sufficient amount of the desired acid to produce the salt according to methods and techniques with which a skilled artisan is familiar. [0076] “Pharmaceutically acceptable base addition salt” refers to those salts that retain the biological effectiveness and properties of the free acids, which are not biologically or otherwise undesirable. These salts are prepared from addition of an inorganic base or an organic base to the free acid. Pharmaceutically acceptable base addition salts are, in some embodiments, formed with metals or amines, such as alkali and alkaline earth metals or organic amines. Salts derived from inorganic bases include, but are not limited to, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum salts and the like. Salts derived from organic bases include, but are not limited to, salts of primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines and basic ion exchange resins, for example, isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, ethanolamine, diethanolamine, 2-dimethylaminoethanol, 2-diethylaminoethanol, dicyclohexylamine, lysine, arginine, histidine, caffeine, procaine, N,N-dibenzylethylenediamine, chloroprocaine, hydrabamine, choline, betaine, ethylenediamine, ethylenedianiline, N- methylglucamine, glucosamine, methylglucamine, theobromine, purines, piperazine, piperidine, N-ethylpiperidine, polyamine resins and the like. See Berge et al., supra. [0077] “Pharmaceutically acceptable solvate” refers to a composition of matter that is the solvent addition form. In some embodiments, solvates contain either stoichiometric or non-stoichiometric amounts of a solvent, and are formed during the process of making with pharmaceutically acceptable solvents such as water, ethanol, and the like. Hydrates are formed when the solvent is water, or alcoholates are formed when the solvent is alcohol. Solvates of compounds described herein are conveniently prepared or formed during the processes described herein. The compounds provided herein exist in either unsolvated or solvated forms. [0078] The term “subject” or “patient” encompasses mammals. Examples of mammals include, but are not limited to, any member of the Mammalian class: humans, non-human primates such as chimpanzees, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like. In one aspect, the mammal is a human. [0079] As used herein, “treatment” or “treating,” or “palliating” or “ameliorating” are used interchangeably. These terms refer to an approach for obtaining beneficial or desired results
WSGR Docket No. 62619-720.601 including but not limited to therapeutic benefit and/or a prophylactic benefit. By “therapeutic benefit” is meant eradication or amelioration of the underlying disorder being treated. Also, a therapeutic benefit is achieved with the eradication or amelioration of one or more of the physiological symptoms associated with the underlying disorder such that an improvement is observed in the patient, notwithstanding that the patient is still afflicted with the underlying disorder. For prophylactic benefit, the compositions are, in some embodiments, administered to a patient at risk of developing a particular disease, or to a patient reporting one or more of the physiological symptoms of a disease, even though a diagnosis of this disease has not been made. KRAS Protein and Function [0080] RAS mutation is frequent in cancer, with approximately 19% of patients with cancer harboring RAS mutations (I.A. Prior et al., Cancer Res 2020; 80:2969-74). Ras proteins are important for activating signaling networks for controlling cell differentiation, proliferation, and survival, encoded by three genes HRAS, KRAS, and NRAS. The three genes share significant sequence homology and largely overlapping functions. Activation of RAS is facilitated by guanine nucleotide exchange factors (GEF), and activation causes conformational changes. [0081] The KRAS gene encodes two highly related protein isoforms, KRAS-4A and KRAS-4B, which comprise of 189 and 188 amino acids. KRAS generally refers to KRAS-4B, because of the high level of mRNA encoding KRAS-4B in cells. KRAS has two major domains, the catalytic G domain and a hypervariable region (HVR). [0082] KRAS G domain is the basis of biological function of GTPase proteins. The G domain comprises 6 beta-strands of the protein core, surrounded by five alpha-helices, and comprises residues 1-166. The G domain also consists of other regions: switch I, switch II, and the P loop. KRAS-GTP binding alters the conformation of the switches I and II in the G domain. When activated, KRAS binds to its downstream molecules as monomers or dimers to mediate series of signaling cascades. KRAS also has a flexible C-terminal, the hypervariable region (HVR), which is important for localizing KRAS to the membrane. [0083] The RAS family comprises three isoforms, but about 85% of RAS-related cancers are caused by mutations in the KRAS isoform. The mutations in KRAS isoform occurs most frequently in solid tumors such as colorectal carcinoma, lung adenocarcinoma, and pancreatic ductal carcinoma. Further, nearly 80% of KRAS mutant tumors are located within codon 12, with the most common mutations being p.G12D, p.G12V, and p.G12C. [0084] KRAS protein functions as a molecular switch in growth factor signaling pathways by regulating proliferation by alternating between a GDP-bound inactive form and a GTP-bound
WSGR Docket No. 62619-720.601 active form. The GTP-bound active form is capable of engaging downstream effector proteins to trigger a pro-proliferative response. This regulation cycle is impaired by mutations in codon 12 which disrupts association of GTPase activating proteins, which impairs the inactivation of KRAS, which leads to accumulation of the pro-proliferative form. Many growth factors such as but not limited to epidermal growth factor (EGF), platelet-derived growth factor (PDGF), and fibroblast growth factors (FGF) can activate KRAS proteins through intermediary molecules after activating receptor tyrosine kinases. Upstream regulation can promote binding of GTP and KRAS, converting KRAS from an inactive to an active state. Molecules upstream of KRAS mainly mediate the activation or inactivation of KRAS by regulating guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs) (L. Huang et al., Signal Transduction and Targeted Therapy, 2021, 6, 386). Another molecule in KRAS activation is Src homology phosphatase 2 (SHP2) which plays a role in KRAS activation. SHP2 is a common signaling regulatory that mediates receptor tyrosine kinases signals to KRAS-ERK signaling, and dephosphorylation substrates of SHP2 have been shown to promote KRAS activation. [0085] The RAF-MEK-ERK pathway is a downstream target of KRAS signaling. Another pathway KRAS involved in is the PI3K-AKT-mTOR pathway (L. Huang et al., Signal Transduction and Targeted Therapy, 2021, 6, 386). [0086] KRAS was previously considered to be an undruggable protein, but recently there have been advances in targeting codon 12, and specifically in G12C inhibitors. Many efforts have been focused on indirectly targeting KRAS, so there remains an unmet need of targeting KRAS, which the compounds provided herein fulfill. With the discovery of a new allosteric site of KRAS, G12C, several covalently binding inhibitors of KRAS have emerged and are under clinical investigation. However, KRAS inhibition is a complex issue with a lack of understanding of the underlying principles, and there still remains an unmet need for new inhibitors which target other KRAS mutations such as, but not limited to G12D and G12V. [0087] KRAS mutations are frequently found in colorectal cancer, pancreatic cancer, and non- small cell lung cancer (M.H. Hofmann et al., Cancer Discov 2022; 12:924-37). The KRAS allelic distribution varies between the tumor types, with G12C mutations in 13.6% of lung adenocarcinomas, whereas the G12D and G12V mutations are most common in colorectal and pancreatic cancer. The G12D, G12V, and G12C mutations are the three most frequent allele mutations. KRAS mutations, especially at codon 12, is strongly associated with cellular KRAS dependency, indicating that KRAS acts as an oncogenic driver. Prior Art Small Molecules Inhibitors
WSGR Docket No. 62619-720.601 [0088] There have been advances for KRAS G12C inhibitors such as sotorasib (AMG510) and adagrasib (MRTX849). Sotorasib is the first to be approved for clinical use. Both inhibitors rely on the interaction with the nucleophilic cysteine 12 in the GDP state and occupy the switch II pocket. [0089] NMR studies have shown that MRTX849 can engage mutant KRAS proteins lacking the nucleophilic mutant cysteine 12, but that the engagement is selected for inactive GDP-loaded state of KRAS protein, but that AMG510 had weak binding and relies on irreversible reaction of the mutant cysteine12 for KRAS (G12C) inhibitory activity (J.D. Vasta et al., Nature Chemical Biology, 2022, 18, 596-604). [0090] AMG510 and additional KRAS inhibitors are described in Discovery of a Covalent Inhibitor of KRASG12C (AMG 510) for the Treatment of Solid Tumors (B.A Lanman et al., J. Med. Chem.2020, 63, 52-65). Novel Compounds Inhibiting KRAS [0091] In one aspect, provided herein are KRAS inhibitory compounds. [0092] One embodiment provides a compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl;
WSGR Docket No. 62619-720.601 Y is a divalent moiety selected from: (a) –(CH2)m-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) -(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (c) -CH=CH-(CH2)p-O-(CH2)q-; (d) –(CH2)p-CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e) –(CH2)p-CH=CH-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f) -CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (g) –CH=CH-CH=CH-, wherein up to 2 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (h) -C(=CH2)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (i) –(c-C3H4)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (j) –(CH2)p-(c-C3H4)-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; and p is 1-4 and q is 1-4. [0093] One embodiment provides a compound having the structure of Formula (Ia), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl;
WSGR Docket No. 62619-720.601 X3 is N, C-H, C-F, or C-Cl; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from: (a) –(CH2)m-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) -(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (c) -CH=CH-(CH2)p-O-(CH2)q-; (d) –(CH2)p-CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e) –(CH2)p-CH=CH-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f) -CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (g) –CH=CH-CH=CH-, wherein up to 2 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (h) -C(=CH2)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (i) –(c-C3H4)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (j) –(CH2)p-(c-C3H4)-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; n is 0, 1, 2, 3, 4, 5, or 6;
WSGR Docket No. 62619-720.601 m is 1-9; and p is 1-4 and q is 1-4. [0094] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)m-. [0095] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)-. [0096] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2CH2)-. [0097] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)p-O-(CH2)q-. [0098] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2)-. [0099] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -( CH2CH2)-O-(CH2)-. [0100] One embodiment provides the compound of Formula (I) or (Ia), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2CH2)-. [0101] One embodiment provides a compound having the structure of Formula (Ib), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl;
WSGR Docket No. 62619-720.601 each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from –(CH2)m-, or –(CH2)p-O-(CH2)q-; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; p is 1-4; and q is 1-4. [0102] One embodiment provides a compound having the structure of Formula (Ic), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, or C-Cl; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from –(CH2)m-, or –(CH2)p-O-(CH2)q-; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; p is 1-4; and q is 1-4. [0103] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)m-. [0104] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)-.
WSGR Docket No. 62619-720.601 [0105] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2CH2)-. [0106] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)p-O-(CH2)q-. [0107] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2)-. [0108] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2CH2)-O-(CH2)-. [0109] One embodiment provides the compound of Formula (Ib) or (Ic), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2CH2)-. [0110] One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH2-, or -CD2-;
WSGR Docket No. 62619-720.601 R3 is hydrogen, R2 is alkyl, R4 is alkyl, and R2 and R4 join to form a ring; or R2 is hydrogen, R3 is alkyl, R4 is alkyl, and R3 and R4 join to form a ring; or R2 is hydrogen, and R3 and R4 join to form a -CH2OCH2- group; or R2 is hydrogen, and R3 and R4 join to form an -O- group; and n is 0, 1, 2, 3, 4, 5, or 6. [0111] One embodiment provides a compound having the structure of Formula (IIa), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, or C-Cl; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH2-, or -CD2-; R3 is hydrogen, R2 is alkyl, R4 is alkyl, and R2 and R4 join to form a ring; or R2 is hydrogen, R3 is alkyl, R4 is alkyl, and R3 and R4 join to form a ring; or R2 is hydrogen, and R3 and R4 join to form a -CH2OCH2- group; or R2 is hydrogen, and R3 and R4 join to form an -O- group; and n is 0, 1, 2, 3, 4, 5, or 6. [0112] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein W is O. [0113] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein W is -CH2-, or -CD2-.
WSGR Docket No. 62619-720.601 [0114] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R2 and R4 join together to form a - CH2CH2-. [0115] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R3 and R4 join together to form a - CH2CH2-. [0116] One embodiment provides a compound having the structure of Formula (II) or (IIa), or a pharmaceutically acceptable salt or solvate thereof, wherein R3 and R4 join together to form a - CH2CH2CH2-. [0117] A compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from:
WSGR Docket No. 62619-720.601 (a)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (d)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
(h) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; R7 is an optionally substituted C1-C4 alkyl; W is selected from oxo, oximo, or optionally substituted alkyl oximo; n is 1 or 2; p is 1-4; and q is 1-4.
WSGR Docket No. 62619-720.601 [0118] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0119] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0120] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0121] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0122] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0123] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. [0124] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
WSGR Docket No. 62619-720.601 [0125] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Y is , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. One embodiment provides the compound wherein W is oxo. One embodiment provides the compound wherein W is oximo. One embodiment provides the compound wherein W is optionally substituted alkyl oximo. One embodiment provides the compound wherein W is O-methyl oximo. [0126] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R7 is an optionally substituted C1 alkyl. [0127] One embodiment provides a compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof, wherein p is 1 or 2; and q is 1 or 2. [0128] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X1 is N. [0129] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X1 is C-CN. [0130] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X2 is N. [0131] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X2 is C-H, C-F, or C-Cl. [0132] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X3 is N. [0133] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X3 is C-H, C-F, or C-Cl. [0134] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X3 is C-CF3. [0135] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein X4 is O. [0136] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R is hydrogen. [0137] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R is methyl.
WSGR Docket No. 62619-720.601 [0138] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 0. [0139] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 1. [0140] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein n is 2. [0141] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted aryl. One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is an optionally substituted naphthyl. One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted naphthyl is an optionally substituted 1-naphthyl. One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted 1-naphthyl is further substituted at the 8-position. [0142] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is described by Formula (a):
wherein: R12 is hydrogen; R13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R14 is hydrogen; R15 is hydrogen or F; R16 is hydrogen or F; R17 is fluorine or -CN; and R18 3, -OCD3, -OCH2F, or -OCD2F. [0143] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0144] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
[0145] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0146] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
[0147] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is:
. [0148] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0149] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
[0150] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0151] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0152] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted heteroaryl. One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is described by Formula (b):
wherein: R12 is hydrogen; R13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R14 is hydrogen; R16 is hydrogen or F; R17 is fluorine or -CN; and R18 3, -OCD3, -OCH2F, or -OCD2F. [0153] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from:
WSGR Docket No. 62619-720.601
. [0154] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from:
[0155] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is an optionally substituted phenyl. One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted phenyl is substituted with an -OH group at the 3-position. [0156] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0157] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
. [0158] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
. [0159] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
.
WSGR Docket No. 62619-720.601 [0160] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
. [0161] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
[0162] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
[0163] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is a bond. [0164] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1-C4 alkylene. [0165] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is
. [0166] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is
. [0167] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1 alkylene. [0168] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III),or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0169] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is an optionally substituted 5- to 10-membered heterocyclyl. [0170] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601
[0171] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
. [0172] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (c):
wherein, each R20-R30 is independently selected from hydrogen or deuterium. [0173] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (d):
wherein, R31 is selected from hydrogen, F, Cl, -CN, -OH, or optionally substituted C1-C4 alkyl; R32 is hydrogen, deuterium or optionally substituted C1-C4 alkyl; and R33 is hydrogen, deuterium, or F. [0174] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601
[0175] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
[0176] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: ,
[0177] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601 [0178] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
[0179] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
[0180] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601
[0181] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0182] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
WSGR Docket No. 62619-720.601 [0183] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0184] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
WSGR Docket No. 62619-720.601
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0185] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0186] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III),or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
WSGR Docket No. 62619-720.601 [0187] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0188] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0189] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601
embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0190] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0191] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601
. Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0192] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
, , , , , ,
. provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0193] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601 Another embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. [0194] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
salt or solvate thereof, wherein X is O. [0195] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
embodiment provides the compound, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O.
WSGR Docket No. 62619-720.601 [0196] One embodiment provides the compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
[0197] One embodiment provides a compound of the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof, having a structure represented in Fig.1. [0198] One embodiment provides a compound of the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof, having a structure represented in Fig.2. [0199] One embodiment provides a compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof,
WSGR Docket No. 62619-720.601
Formula (I), wherein the portion of the structure of Formula (I) enclosed within the box is provided in Fig.3, Fig.4, Fig.5, or Fig.6. [0200] One embodiment provides a compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof,
Formula (II), wherein the portion of the structure of Formula (II) enclosed within the box is provided in Fig.7, Fig.8, Fig.9, or Fig.10. [0201] One embodiment provides a KRAS inhibitory compound, or a pharmaceutically acceptable salt or solvate thereof, having a structure presented in Table 1.
WSGR Docket No. 62619-720.601 Table 1
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601 [0202] One embodiment provides a KRAS inhibitory compound, or a pharmaceutically acceptable salt or solvate thereof, having a structure presented in Table 2. Table 2
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
Preparation of Compounds [0203] The compounds used in the synthetic chemistry reactions described herein are made according to organic synthesis techniques known to those skilled in this art, starting from commercially available chemicals and/or from compounds described in the chemical literature. “Commercially available chemicals” are obtained from standard commercial sources including Acros Organics (Pittsburgh, PA), Aldrich Chemical (Milwaukee, WI, including Sigma Chemical and Fluka), Apin Chemicals Ltd. (Milton Park, UK), Avocado Research (Lancashire, U.K.), BDH Inc. (Toronto, Canada), Bionet (Cornwall, U.K.), Chemservice Inc. (West Chester, PA), Crescent Chemical Co. (Hauppauge, NY), Eastman Organic Chemicals, Eastman Kodak Company (Rochester, NY), Fisher Scientific Co. (Pittsburgh, PA), Fisons Chemicals (Leicestershire, UK), Frontier Scientific (Logan, UT), ICN Biomedicals, Inc. (Costa Mesa, CA), Key Organics (Cornwall, U.K.), Lancaster Synthesis (Windham, NH), Maybridge Chemical Co. Ltd. (Cornwall, U.K.), Parish Chemical Co. (Orem, UT), Pfaltz & Bauer, Inc. (Waterbury, CN), Polyorganix (Houston, TX), Pierce Chemical Co. (Rockford, IL), Riedel de Haen AG (Hanover, Germany), Spectrum Quality Product, Inc. (New Brunswick, NJ), TCI America (Portland, OR), Trans World Chemicals, Inc. (Rockville, MD), and Wako Chemicals USA, Inc. (Richmond, VA). [0204] Suitable reference books and treatise that detail the synthesis of reactants useful in the preparation of compounds described herein, or provide references to articles that describe the preparation, include for example, “Synthetic Organic Chemistry”, John Wiley & Sons, Inc., New York; S. R. Sandler et al., “Organic Functional Group Preparations,” 2nd Ed., Academic Press, New York, 1983; H. O. House, “Modern Synthetic Reactions”, 2nd Ed., W. A. Benjamin, Inc. Menlo
WSGR Docket No. 62619-720.601 Park, Calif.1972; T. L. Gilchrist, “Heterocyclic Chemistry”, 2nd Ed., John Wiley & Sons, New York, 1992; J. March, “Advanced Organic Chemistry: Reactions, Mechanisms and Structure”, 4th Ed., Wiley-Interscience, New York, 1992. Additional suitable reference books and treatise that detail the synthesis of re“ctants useful in the prepa”ation of compounds described herein, or provide references to articles that describe the preparation, include for example, Fuhrhop, J. and Penzlin G. “Organic Synthesis: Concepts, Methods, Starting Materials”, Second, Revised and Enlarged Edition (1994) John Wiley & Sons ISBN: 3-527-29074-5; Hoffman, R.V. “Organic Chemistry, An Intermediate Text" (1996) Oxford University Press, ISBN 0-19-509618-5; Larock, R. C. “Comprehensive Organic Transformations: A Guide to Functional Group Preparations” 2nd Edition (1999) Wiley-VCH, ISBN: 0-471-19031-4; March, J. “Advanced Organic Chemistry: Reactions, Mechanisms, and Structure” 4th Edition (1992) John Wiley & Sons, ISBN: 0-471- 60180-2; Otera, J. (editor) “Modern Carbonyl Chemistry” (2000) Wiley-VCH, ISBN: 3-527- 29871-1; Patai, S. “Patai’s 1992 Guide to the Chemistry of Functional Groups” (1992) Interscience ISBN: 0-471-93022-9; Solomons, T. W. G. “Organic Chemistry” 7th Edition (2000) John Wiley & Sons, ISBN: 0-471-19095-0; Stowell, J.C., “Intermediate Organic Chemistry” 2nd Edition (1993) Wiley-Interscience, ISBN: 0-471-57456-2; “Industrial Organic Chemicals: Starting Materials and Intermediates: An Ullmann’s Encyclopedia” (1999) John Wiley & Sons, ISBN: 3-527-29645-X, in 8 volumes; “Organic Reactions” (1942-2000) John Wiley & Sons, in over 55 volumes; and “Chemistry of Functional Groups” John Wiley & Sons, in 73 volumes. [0205] Specific and analogous reactants are optionally identified through the indices of known chemicals prepared by the Chemical Abstract Service of the American Chemical Society, which are available in most public and university libraries, as well as through on-line databases (contact the American Chemical Society, Washington, D.C. for more details). Chemicals that are known but not commercially available in catalogs are optionally prepared by custom chemical synthesis houses, where many of the standard chemical supply houses (e.g., those listed above) provide custom synthesis services. A reference useful for the preparation and selection of pharmaceutical salts of the compounds described herein is P. H. Stahl & C. G. Wermuth “Handbook of Pharmaceutical Salts”, Verlag Helvetica Chimica Acta, Zurich, 2002. Pharmaceutical Compositions [0206] In certain embodiments, the KRAS inhibitory compound described herein is administered as a pure chemical. In other embodiments, the KRAS inhibitory compound described herein is combined with a pharmaceutically suitable or acceptable carrier (also referred to herein as a pharmaceutically suitable (or acceptable) excipient, physiologically suitable (or acceptable)
WSGR Docket No. 62619-720.601 excipient, or physiologically suitable (or acceptable) carrier) selected on the basis of a chosen route of administration and standard pharmaceutical practice as described, for example, in Remington: The Science and Practice of Pharmacy (Gennaro, 21st Ed. Mack Pub. Co., Easton, PA (2005)). [0207] Provided herein is a pharmaceutical composition comprising at least one KRAS inhibitory compound as described herein, or a stereoisomer, pharmaceutically acceptable salt, hydrate, or solvate thereof, together with one or more pharmaceutically acceptable carriers. The carrier(s) (or excipient(s)) is acceptable or suitable if the carrier is compatible with the other ingredients of the composition and not deleterious to the recipient (i.e., the subject or the patient) of the composition. [0208] One embodiment provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof. [0209] One embodiment provides a method of preparing a pharmaceutical composition comprising mixing a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable carrier. [0210] In certain embodiments, the KRAS inhibitory compound as described by Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, is substantially pure, in that it contains less than about 5%, or less than about 2%, or less than about 1%, or less than about 0.5%, or less than about 0.1%, of other organic small molecules, such as unreacted intermediates or synthesis by-products that are created, for example, in one or more of the steps of a synthesis method. [0211] One embodiment provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof. One embodiment provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a compound of any of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof. [0212] One embodiment provides a method of preparing a pharmaceutical composition comprising mixing a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable carrier. [0213] In certain embodiments, the KRAS inhibitory compound as described by Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, is substantially pure, in that it contains less than about 5%, or less than about 2%, or less than about 1%, or less than about 0.5%, or less than about 0.1%, of other organic small molecules, such as unreacted intermediates or synthesis by- products that are created, for example, in one or more of the steps of a synthesis method.
WSGR Docket No. 62619-720.601 [0214] Suitable oral dosage forms include, for example, tablets, pills, sachets, or capsules of hard or soft gelatin, methylcellulose or of another suitable material easily dissolved in the digestive tract. In some embodiments, suitable nontoxic solid carriers are used which include, for example, pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, talcum, cellulose, glucose, sucrose, magnesium carbonate, and the like. (See, e.g., Remington: The Science and Practice of Pharmacy (Gennaro, 21st Ed. Mack Pub. Co., Easton, PA (2005)). [0215] In some embodiments, the KRAS inhibitory compound as described by Formula (I), or Table 1 or Table 2, or pharmaceutically acceptable salt or solvate thereof, is formulated for administration by injection. In some instances, the injection formulation is an aqueous formulation. In some instances, the injection formulation is a non-aqueous formulation. In some instances, the injection formulation is an oil-based formulation, such as sesame oil, or the like. [0216] The dose of the composition comprising at least one KRAS inhibitory compound as described herein differs depending upon the subject or patient's (e.g., human) condition. In some embodiments, such factors include general health status, age, and other factors. [0217] Pharmaceutical compositions are administered in a manner appropriate to the disease to be treated (or prevented). An appropriate dose and a suitable duration and frequency of administration will be determined by such factors as the condition of the patient, the type and severity of the patient's disease, the particular form of the active ingredient, and the method of administration. In general, an appropriate dose and treatment regimen provides the composition(s) in an amount sufficient to provide therapeutic and/or prophylactic benefit (e.g., an improved clinical outcome, such as more frequent complete or partial remissions, or longer disease-free and/or overall survival, or a lessening of symptom severity. Optimal doses are generally determined using experimental models and/or clinical trials. The optimal dose depends upon the body mass, weight, or blood volume of the patient. [0218] Oral doses typically range from about 1.0 mg to about 1000 mg, one to four times, or more, per day. Methods of Treatment [0219] One embodiment provides a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of the human or animal body. [0220] One embodiment provides a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treating cancer.
WSGR Docket No. 62619-720.601 Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. [0221] One embodiment provides a pharmaceutical composition comprising a compound of Formula (I) or (II), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient. [0222] One embodiment provides a use of a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, in the manufacture of a medicament for the treatment of cancer. [0223] In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof. In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a pharmaceutical composition comprising a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient. Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. [0224] One embodiment provides a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of the human or animal body. [0225] One embodiment provides a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, for use in a method of treating cancer. Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. [0226] One embodiment provides a pharmaceutical composition comprising a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient. [0227] One embodiment provides a use of a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof, in the manufacture of a medicament for the treatment of cancer. [0228] In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate thereof. In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a pharmaceutical composition comprising a compound of Table 1 or Table 2, or a pharmaceutically acceptable salt or solvate
WSGR Docket No. 62619-720.601 thereof, and a pharmaceutically acceptable excipient. Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. [0229] In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a compound of any one of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof. In some embodiments is provided a method of treating cancer in a patient in need thereof, comprising administering to the patient a pharmaceutical composition comprising a compound of any one of Figures 1-10, or a pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient. Another embodiment provides the method wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. [0230] Provided herein is the method wherein the pharmaceutical composition is administered orally. Provided herein is the method wherein the pharmaceutical composition is administered by injection. [0231] One embodiment provides a method of inhibiting KRAS protein activity comprising contacting the KRAS protein with a compound of Formula (I), (Ia), (Ib), (Ic), (II), (IIa), or (III), or Table 1 or Table 2. Another embodiment provides the method of inhibiting KRAS protein activity, wherein the KRAS protein is contacted in an in vivo setting. Another embodiment provides the method of inhibiting KRAS protein activity, wherein the KRAS protein is contacted in an in vitro setting. [0232] Other embodiments and uses will be apparent to one skilled in the art in light of the present disclosures. The following examples are provided merely as illustrative of various embodiments and shall not be construed to limit the invention in any way. EXAMPLES I. Chemical Synthesis [0233] In some embodiments, the KRAS inhibitory compounds disclosed herein are synthesized according to the following examples. As used below, and throughout the description of the invention, the following abbreviations, unless otherwise indicated, shall be understood to have the following meanings: ACN acetonitrile oC degrees Celsius H chemical shift in parts per million downfield from tetramethylsilane DCM dichloromethane (CH2Cl2) DIAD diisopropyl azodicarboxylate
WSGR Docket No. 62619-720.601 DIEA diisopropylethylamine DMF dimethylformamide DMSO dimethylsulfoxide EA ethyl acetate EtOAc ethyl acetate ESI electrospray ionization Et ethyl g gram(s) h hour(s) HPLC high performance liquid chromatography Hz hertz J coupling constant (in NMR spectrometry) LCMS liquid chromatography mass spectrometry micro m multiplet (spectral); meter(s); milli M molar M+ parent molecular ion Me methyl MsCl methanesulfonyl chloride MHz megahertz min minute(s) mol mole(s); molecular (as in mol wt) mL milliliter MS mass spectrometry nm nanometer(s) NMR nuclear magnetic resonance pH potential of hydrogen; a measure of the acidity or basicity of an aqueous solution PE petroleum ether RT room temperature s singlet (spectral) t triplet (spectral) SFC Supercritical fluid chromatography
WSGR Docket No. 62619-720.601 T temperature TFA trifluoroacetic acid THF tetrahydrofuran TPP Triphenylphosphine Experimental Procedures [0234] Intermediate 1: 2,5,7-Trichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol
[0235] Step 1: 2,6-Dichloro-3-fluoropyridin-4-amine [0236] To a solution of 2,6-dichloropyridin-4-amine (26 g, 160 mmol) in MeOH (250 mL) and H2O (50 mL) was added SelectFluor (85 g, 240 mmol). The mixture was degassed and purged with N2 three times, and the mixture was stirred at 45 °C for 16 h under N2 atmosphere. The reaction mixture was diluted with EtOAc (1000 mL), washed with H2O (200 mL x 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (30 g, crude) as a yellow solid, which was used into the next step without further purification. MS: m/z = 181.0 [M+H]+. [0237] Step 2: 2,6-Dichloro-3-fluoro-5-iodopyridin-4-amine [0238] To a solution of 2,6-dichloro-3-fluoro-pyridin-4-amine (75 g, 414 mmol) in MeCN (300 mL) were added 1-iodopyrrolidine-2,5-dione (112 g, 497 mmol) and 4-methylbenzenesulfonic acid monohydrate (3.94 g, 20.7 mmol). The mixture was stirred at 70 °C for 4 h. The reaction mixture was quenched with Na2SO3 (200 mL) at 20 °C and extracted with EtOAc (500 mL x 3). The combined organic phases were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by re-crystallization from toluene (500 mL) at 100 °C for 30 min, cooled to room temperature, filtered and the filter cake was dried to give the title compound (76.7 g, 50% yield for two steps) as a yellow solid. MS: m/z = 306.8 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 19F NMR (376 MHz, Dimethylsulfoxide- d6 -140.69. [0239] Step 3: Ethyl 4-amino-2,6-dichloro-5-fluoronicotinate
WSGR Docket No. 62619-720.601 [0240] To a mixture of 2,6-dichloro-3-fluoro-5-iodo-pyridin-4-amine (42 g, 137 mmol), TEA (66. 7 mL, 479 mmol) and dichloropalladiumtriphenylphosphane (4.80 g, 6.84 mmol) in EtOH (1050 mL) was purged with CO three times, and the mixture was stirred under CO (1.5 Mpa) at 100 °C for 40 h in a 2000 mL of autoclave. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 10% of EtOAc in Petroleum ether) to give the title compound (31 g, 33% yield) as a yellow solid. MS: m/z = [0241] Step 4: Ethyl 2,6-dichloro-5-fluoro-4-(3-(2,2,2-trichloroacetyl)ureido)nicotinate [0242] To a solution of ethyl 4-amino-2,6-dichloro-5-fluoronicotinate (16.7 g, 66.0 mmol) in THF (250 mL) was added 2,2,2-trichloroacetyl isocyanate (9.39 mL, 79.1 mmol). The mixture was degassed and purged with N2 three times, and then the mixture was stirred at 25 °C for 1 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The crude was triturated with MTBE (100 mL) at 25 °C for 2 h, filltered and the filter cake was dried to give the title compound (20.6 g, 70% yield) as a gray solid. MS: m/z = 441.8 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 7.2 Hz, 14.4 Hz, 3H), 1.28 (t, J = 7.2 Hz, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 -127.67. [0243] Step 5: 5,7-Dichloro-8-fluoropyrido[4,3-d]pyrimidine-2,4(1H,3H)-dione [0244] To a solution of ethyl 2,6-dichloro-5-fluoro-4-(3-(2,2,2-trichloroacetyl)ureido)nicotinate (50.6 g, 115 mmol) in MeOH (500 mL) was added NH3 in MeOH (7 M, 45.81 mL). The mixture was stirred at 25 °C for 1 h. The reaction mixture was concentrated under reduced pressure. The crude was triturated with MTBE (200 mL) at 25 °C for 3 h, filtered, and the filter cake was dried to give the title compound (29.6 g, 99.9% yield) as a white solid. MS: m/z = 249.8 [M + H]+.1H NMR (400 MHz, Methanol-d4 19F NMR (376 MHz, Methanol-d4 -139.59. [0245] Step 6: 2,5,7-Trichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol [0246] To a solution of 5,7-dichloro-8-fluoropyrido[4,3-d]pyrimidine-2,4(1H,3H)-dione (10 g, 40.0 mmol) in POCl3 (100 mL) was added DIPEA (20.9 mL, 120 mmol). The mixture was stirred at 105 °C for 16 h. The reaction mixture was concentrated under reduced pressure. The residue was diluted with 1,4-dioxane (40 mL) and the resulting solution was added dropwise to aq. K2CO3 (20%, 200 mL). The mixture was stirred for 2 h at 25 °C. The pH of the mixture wsa adjusted to 2 - 3 with con. HCl. The mixture was filtered, and the filter cake was collected, and dried to give the title compound (Intermediate 1, 10.54 g, 90% yield,) as a brown solid. MS: m/z = 267.8 [M + H]+. 19F NMR (376 MHz, Methanol-d4 -134.27.
WSGR Docket No. 62619-720.601 [0247] Intermediate 2: 2-(8-Ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
[0248] Step 1: 2-(8-Ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0249] To a solution of ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- CsF (445 mg, 2.93 mmol). The mixture was stirred at 25 °C for 0.5 h. The residue was diluted with ice-water (10 mL) and extracted with CH2Cl2 (10 mL x 2). The combined organic layers were washed with water (20 mL x 3), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (300 mg, 85% yield) as an off-white solid, which was used in the next step without further purification. MS: m/z = 357.2 [M + H]+. [0250] Step 2: 2-(8-Ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl-1,3,2- dioxaborolane [0251] To a solution of 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- 10% purity). The suspension was degassed, purged with H2 three times, and stirred at 25 °C for 16 h under H2 (15 psi). The reaction mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 5% of EtOAc in Petrol Ether) to give the title compound (Intermediate 2, 160 mg, 53% yield) as an off-white solid. 1H NMR (400 MHz, Dimethylsulfoxide-d6 7.56 (dd, J = 6.0, 8.8 Hz, 1H), 7.41 - 7.35 (m, 2H), 7.20 (t, J = 9.2 Hz, 1H), 5.27 (s, 2H), 3.50 (s, 3H), 3.13 (m, J = 2.4, 7.6 Hz, 2H), 1.44 (s, 12H), 1.27 (t, J = 7.6 Hz, 3H). 19F NMR (400 MHz, Dimethylsulfoxide-d6 -119.45. [0252] Intermediate 3: 2-(8-(Ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
[0253] Step 1: 2-(8-Ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane
WSGR Docket No. 62619-720.601 [0254] To a solution of ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (5 g, 9.755 mmol) in DMF (25 mL) under nitrogen atmosphere was added CsF (14.82 g, 97.550 mmol) at room temperature. The mixture was stirred at this temperature for 16 hours. The resulting mixture was diluted with EtOAc (500 mL), washed with water (3 x 100 mL) and brine (100 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 80% EA in PE to afford the title compound (3.0 g, 86% yield) as a yellow powder. MS: m/z = 357.55 [M + H]+. 1H NMR (300 MHz, Chloroform-d J = 2.4 Hz, 1H), 7.40 (d, J = 2.4 Hz, 1H), 7.23 (t, J = 9.0 Hz, 1H), 5.28 (s, 2H), 3.68 (s, 1H), 3.50 (s, 3H), 1.44 (s, 12H). [0255] Step 2: 2-(8-(Ethynyl-d)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane [0256] To a solution of 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (1.9 g, 5.334 mmol) in MeCN (30 mL) under nitrogen atmosphere were added K2CO3 (2.21 g, 16.002 mmol) and D2O (3.0 mL, 164.778 mmol) at room temperature. The mixture was stirred at room temperature for 4 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 20% EA in PE to afford the title compound (1.5 g, 78% yield) as an off-white powder. MS: m/z = 358.15 [M + H]+. 1H NMR (300 MHz, Chloroform-d J = 2.4 Hz, 1H), 7.40 (d, J = 2.7 Hz, 1H), 7.23 (t, J = 9.0 Hz, 1H), 5.28 (s, 2H), 3.50 (s, 3H), 1.44 (s, 12H). [0257] Step 3: 2-(8-(Ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0258] To a solution of 2-(8-(ethynyl-d)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (1.5 g, 4.199 mmol) in EtOAc (38 mL) under nitrogen atmosphere was added PtO2 (0.14 g, 0.630 mmol) at room temperature. The reaction mixture was degassed, purged with nitrogen three times, and followed by flushing with deuterium gas. The resulting mixture was stirred at room temperature for 2 hours under an atmosphere of deuterium gas (1.5-2 atm). The solid was filtered off and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 1-50% EtOAc in petroleum ether to give the title compound (Intermediate 3, 1.2 g, 78.24% yield) as a light yellow solid. MS: m/z = 366.30 [M + H]+. 1H NMR (400 MHz, Chloroform-d 2H), 7.23 - 7.15 (m, 1H), 5.27 (s, 2H), 3.50 (s, 3H), 1.44 (s, 12H).
WSGR Docket No. 62619-720.601 [0259] Intermediate 4: 2-(7-Fluoro-3-(methoxymethoxy)-8-vinylnaphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
[0260] Step 1: 2-(8-Ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0261] To a solution of ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (10.0 g, 19.5 mmol) in DMSO (100 mL) was added CsF (29.6 g, 195 mmol). The mixture was stirred at 20 °C for 2 h. The reaction mixture was poured into ice-water (300 mL), filtered and washed with water (500 mL). The filter cake was dried to give a residue. The residue was washed with petroleum ether (80 mL) to give the title compound (6.90 g, 99% yield) as an off-white solid. MS: m/z = 356.8 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 9.2 Hz, 1H), 7.58 (d, J = 2.4 Hz, 1H), 7.49 (t, J = 8.8 Hz, 1H), 7.34 (d, J = 2.4 Hz, 1H), 5.32 (s, 2H), 5.03 (s, 1H), 3.44 - 3.39 (m, 3H), 1.37 (s, 12H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 -110.39. [0262] Step 2: 2-(7-Fluoro-3-(methoxymethoxy)-8-vinylnaphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0263] To a solution of 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (13.0 g, 36.5 mmol), quinoline (6.00 g, 46.5 mmol, 5.48 mL) in methanol (50.0 mL) was added Lindlar catalyst (7.80 g, 37.8 mmol). The mixture was stirred at 40 °C for 1 h under H2 (30 psi). The reaction mixture was filtered and washed with methanol (100 mL x 3). The filtrate was dried to afford the residue. The residue was purified by prep-HPLC (column: 3H2O) - ACN]; gradient: 55% - 85% B over 15 min ) to give the title compound (Intermediate 4, 6.67 g, 50% yield) as a yellow solid. MS: m/z = 359.0 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 5.6, 8.8 Hz, 1H), 7.53 (d, J = 2.4 Hz, 1H), 7.40 (t, J = 9.6 Hz, 1H), 7.28 (d, J = 2.8 Hz, 1H), 7.00 (dd, J = 11.2, 17.6 Hz, 1H), 5.72 (dd, J = 1.6, 11.6 Hz, 1H), 5.56 (d, J = 17.6 Hz, 1H), 5.31 (s, 2H), 3.42 (s, 3H), 1.34 (s, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0264] Intermediate 5: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene
WSGR Docket No. 62619-720.601
[0265] Step 1: tert-Butyl (S)-2-(hydroxymethyl)azepane-1-carboxylate [0266] To an ice-cooled solution of (S)-1-(tert-butoxycarbonyl)azepane-2-carboxylic acid (10 g, 41.10 mmol) in THF (100 mL) was added BH3 (61.72 mL, 61.72 mmol, 1M in THF) dropwise. The mixture was stirred in an ice bath for 1.5 hours. MeOH (20 mL) was added dropwise to the above mixture. The ice bath was removed, and the resulting mixture was heated at 60 °C for 20 mins. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (9 g, 95% yield) as a white oil. MS: m/z = 230.15 [M + H]+. [0267] Step 2: (S)-Azepan-2-ylmethanol [0268] To an ice-cooled solution of tert-butyl (S)-2-(hydroxymethyl)azepane-1-carboxylate (30 g, 130.82 mmol) in DCM (300 mL) was added 4N HCl (gas) in 1,4-dioxane (300 mL, 9873.83 mmol) dropwise. The reaction mixture was stirred in an ice bath for 1.5 hours. The ice bath was removed, and the resulting mixture was concentrated under reduced pressure. The residue was dissolved in MeOH (50 mL), and Amberlyst-A-21 resin (8 g) was added at room temperature. The resulting mixture was stirred at room temperature for 1 hour. The mixture was filtered and concentrated under reduced pressure to give the title compound (15 g, 88% yield) as a light-yellow solid. MS: m/z = 130.15 [M + H]+. 1H NMR (400 MHz, DMSO-d6 3.35 - 3.23 (m, 2H), 3.20 - 3.11 (m, 1H), 1.98 - 1.76 (m, 5H), 1.70 - 1.59 (m, 3H). [0269] Step 3: (S)-5-(Azepan-2-ylmethoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol [0270] To an ice-cooled solution of (S)-azepan-2-ylmethanol (1.80 g, 13.97 mmol) in THF (60 mL) under nitrogen atmosphere was added NaH (0.89 g, 37.25 mmol, 60% in mineral oil). After the mixture was stirred in an ice bath for 0.5 hour, Intermediate 1 (2.5 g, 9.31 mmol) was added. The ice bath was removed, and the reaction mixture was stirred at room temperature for 2.5 hours. The resulting mixture was quenched with saturated aq. NH4Cl (200 mL) and extracted with EtOAc (3 x
WSGR Docket No. 62619-720.601 100 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 15% MeOH in DCM to afford the title compound (2 g, 59% yield) as a yellow solid. MS: m/z = 360.95, 362.95 [M + H]+. 1H NMR (300 MHz, DMSO- d6 1H), 3.18 - 3.10 (m, 1H), 2.02 - 1.68 (m, 6H), 1.62 - 1.45 (m, 2H). [0271] Step 4: (S)-2,5-Dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0272] To an ice-cooled solution of (S)-5-(azepan-2-ylmethoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4-ol (1.9 g, 5.26 mmol) and DIEA (4.08 g, 31.56 mmol) in DCM (380 mL) under nitrogen atmosphere was added POCl3 (4.03 g, 26.30 mmol) dropwise. The resulting mixture was stirred in an ice bath for 2 hours. The resulting mixture was quenched with saturated aq. NaHCO3 (200 mL) and extracted with CH2Cl2 (3 x 100 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous sodium, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 6% EA in DCM to give the title compound (600 mg, 33% yield) as a yellow solid. MS: m/z = 343.05, 345.05 [M + H]+. 1H NMR (400 MHz, Chloroform-d J = 12.8 Hz, 1H), 3.96 - 3.93 (m, 1H), 3.21 - 3.14 (m, 1H), 2.16 - 1.23 (m, 8H). [0273] Step 5: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0274] To a mixture of (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- pyrrolizin-7a(5H)-yl)methanol (556.69 mg, 3.49 mmol) in 1,4-dioxane (12 mL) under nitrogen atmosphere were added 4A molecular sieves and Cs2CO3 (1424.12 mg, 4.37 mmol) at room temperature. The reaction mixture was heated at 90 °C for 16 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 5% MeOH in DCM to afford the title compound (Intermediate 5, 580 mg, 71% yield) as a light yellow solid. MS: m/z = 466.20 [M + H]+. 1H NMR (300 MHz, Chloroform-d 1H), 4.44 - 4.33 (m, 1H), 4.27 - 4.17 (m, 2H), 3.92 - 3.85 (m, 1H), 3.46 - 2.86 (m, 5H), 2.38 - 1.23 (m, 14H). [0275] Intermediate 6 & 7: ((2S,7aR)-2-Fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol & ((2S,7aS)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
WSGR Docket No. 62619-720.601
[0276] Step 1: 1-(tert-Butyl) 2-methyl (2R,4R)-4-((tert-butyldimethylsilyl)oxy)pyrrolidine-1,2- dicarboxylate [0277] To an ice-cooled solution of 1-tert-butyl 2-methyl (2R,4R)-4-hydroxypyrrolidine-1,2- dicarboxylate (15 g, 61.156 mmol) and imidazole (6.25 g, 91.734 mmol) in DMF (150 mL) under nitrogen atmosphere was added TBSCl (11.98 g, 79.503 mmol). The ice bath was removed, and the reaction mixture was stirred at room temperature for 16 hours. The resulting mixture was diluted with water (1500 mL) and extracted with EtOAc (500 mL x 4). The combined organic layers were washed with brine (400 mL x 3), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EtOAc in PE to afford the title compound (20.25 g, 92% yield) as a yellow oil. MS: m/z = 360.10 [M + H]+. 1H NMR (400 MHz, Chloroform-d 3H), 3.67 - 3.53 (m, 1H), 3.35 - 3.24 (m, 1H), 2.36 - 2.20 (m, 1H), 2.15 - 2.02 (m, 1H), 1.48 - 1.40 (m, 9H), 0.90 - 0.84 (m, 9H), 0.07 - 0.02 (m, 6H). [0278] Step 2: 1-(tert-Butyl) 2-methyl (4R)-4-((tert-butyldimethylsilyl)oxy)-2-(2- (chloromethyl)allyl)pyrrolidine-1,2-dicarboxylate [0279] To a stirred solution of 1-(tert-butyl) 2-methyl (2R,4R)-4-((tert- butyldimethylsilyl)oxy)pyrrolidine-1,2-dicarboxylate (15 g, 41.720 mmol) in THF (300 mL) under nitrogen atmosphere was added LiHMDS (54.24 mL, 54.236 mmol, 1M in THF) dropwise at - 78 °C. The reaction mixture was stirred at -78 °C for 20 min. To the above mixture was added 3- chloro-2-(chloromethyl)prop-1-ene (13.56 g, 108.472 mmol) dropwise at -78 °C. The resulting mixture was stirred at -40 °C for 2 hours. The resulting mixture was quenched with sat. aq. NH4Cl (400 mL) in an ice bath and extracted with EtOAc (400 mL x 3). The combined organic layers were washed with brine (500 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (13.5 g, 72% yield) as a yellow oil. MS: m/z = 448.15 [M + H]+. 1H NMR (400 MHz, Chloroform-d 4.31 - 4.18 (m, 1H), 4.06 - 3.95 (m, 2H), 3.74 - 3.71 (m, 3H), 3.68 - 3.53 (m, 1H), 3.36 - 3.12 (m,
WSGR Docket No. 62619-720.601 2H), 2.68 - 2.61 (m, 1H), 2.34 - 2.20 (m, 1H), 2.16 - 2.00 (m, 1H), 1.46 - 1.44 (m, 9H), 0.87 - 0.83 (m, 9H), 0.05 - 0.00 (m, 6H). [0280] Step 3: 1-(tert-Butyl) 2-methyl (4R)-2-(2-(chloromethyl)allyl)-4-hydroxypyrrolidine-1,2- dicarboxylate [0281] To a solution of 1-tert-butyl 2-methyl (4R)-4-[(tert-butyldimethylsilyl)oxy]-2-[2- (chloromethyl)prop-2-en-1-yl]pyrrolidine-1,2-dicarboxylate (36 g, 80.345 mmol) in THF (360 mL) was added TBAF (96.41 mL, 96.414 mmol, 1M in THF) dropwise at room temperature. The resulting mixture was stirred at room temperature for 16 hours and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-70% EA in PE to give the title compound (23.5 g, 87% yield) as a yellow oil. MS: m/z = 334.05 [M + H]+. [0282] Step 4: 1-(tert-Butyl) 2-methyl (4S)-2-(2-(chloromethyl)allyl)-4-fluoropyrrolidine-1,2- dicarboxylate [0283] To a stirred solution of 1-(tert-butyl) 2-methyl (4R)-2-(2-(chloromethyl)allyl)-4- hydroxypyrrolidine-1,2-dicarboxylate (7.8 g, 23.367 mmol) in DCM (78 mL) under nitrogen atmosphere was added BAST (7.75 g, 35.050 mmol) dropwise at -78 °C. The reaction mixture was stirred at room temperature for 16 hours. The resulting mixture was quenched with water (200 mL) in an ice bath and extracted with DCM (200 mL x 3). The combined organic layers were washed with brine (400 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-70% EA in PE to give 1-tert-butyl 2-methyl (4S)-2-[2-(chloromethyl)prop-2-en-1-yl]-4-fluoropyrrolidine-1,2- dicarboxylate (5 g, 63% yield) as a yellow oil. MS: m/z = 336.05 [M + H]+. 1H NMR (400 MHz, Chloroform-d 2.86 - 2.80 (m, 1H), 2.64 - 2.25 (m, 2H), 1.50 - 1.40 (m, 9H). 19F NMR (376 MHz, Chloroform-d) -173.56 - -174.33 (m, 1F). [0284] Step 5: Methyl (4S)-2-(2-(chloromethyl)allyl)-4-fluoropyrrolidine-2-carboxylate 2,2,2- trifluoroacetate [0285] To an ice-cooled solution of 1-(tert-butyl) 2-methyl (4S)-2-(2-(chloromethyl)allyl)-4- fluoropyrrolidine-1,2-dicarboxylate (15 g, 44.669 mmol) in DCM (150 mL) was added TFA (30 mL). The ice bath was removed, and the reaction mixture was stirred at room temperature for 4 hours. The resulting mixture was concentrated under reduced pressure to give the title compound (15 g, crude) as a brown oil. MS: m/z = 236.05 [M + H]+. [0286] Step 6: Methyl (2S)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizine-7a(5H)-carboxylate
WSGR Docket No. 62619-720.601 [0287] An ice-cooled solution of methyl (4S)-2-(2-(chloromethyl)allyl)-4-fluoropyrrolidine-2- carboxylate 2,2,2-trifluoroacetate (15 g, crude) in MeOH (20 mL) was basified to pH = 8 with NH3(g) (7M in MeOH). The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-100% EA in PE to give the title compound (8 g, 89% yield) as a light-yellow oil. MS: m/z = 199.95 [M + H]+. 1H NMR (400 MHz, Chloroform-d 3.59 - 3.51 (m, 1H), 3.45 - 3.27 (m, 1H), 3.22 - 2.99 (m, 2H), 2.81 - 2.70 (m, 1H), 2.63 - 2.45 (m, 1H), 2.34 - 2.21 (m, 1H). 19F NMR (376 MHz, Chloroform-d -172.17 (s, 1F). [0288] Step 7: ((2S,7aR)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol & ((2S,7aS)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0289] To an ice-cooled solution of methyl (2S)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizine- 7a(5H)-carboxylate (7.5 g, 37.646 mmol) in THF (75 mL) under N2 atmosphere was added LiAlH4 (37.65 mL, 75.292 mmol, 2M in THF) dropwise. The ice bath was removed, and the reaction mixture was stirred at room temperature for 2 hours. The resulting mixture was quenched with water (2.86 mL), 10% aq. NaOH (2.86 mL) and water (8.58 mL) in an ice bath. The resulting mixture was stirred in an ice bath for 15 min, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM to give two isomers. The first eluting isomer was concentrated to give the title compound (Intermediate 6, 5.3 g, 82% yield) as a yellow oil. MS: m/z = 172.00 [M + H]+. 1H NMR (400 MHz, Chloroform-d (m, 1H), 3.32 - 3.15 (m, 3H), 3.15 - 3.02 (m, 1H), 2.87 (br., 1H), 2.72 - 2.62 (m, 1H), 2.38 - 2.30 (m, 1H), 2.26 - 2.01 (m, 2H). 19F NMR (376 MHz, Chloroform-d) -171.45 (s, 1F). The second eluting isomer was concentrated to give the title compound (Intermediate 7, 950 mg, 14% yield) as a yellow oil. MS: m/z = 172.00 [M + H]+. 1H NMR (400 MHz, Chloroform-d 5.35 - 5.15 (m, 1H), 4.98 - 4.92 (m, 2H), 3.88 - 3.81 (m, 1H), 3.58 - 3.53 (m, 1H), 3.43 - 3.26 (m, 3H), 3.21 - 3.07 (m, 1H), 2.74 - 2.65 (m, 1H), 2.46 - 2.38 (m, 1H), 2.37 - 2.21 (m, 1H), 2.17 - 2.04 (m, 1H). 19F NMR (376 MHz, Chloroform-d (s, 1F). [0290] Intermediate 8 & 9: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro- 3H-pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one &
WSGR Docket No. 62619-720.601
[0291] Step 1: (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine [0292] To an ice-cooled solution of ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol (5 g, 31.40 mmol) and imidazole (4.28 g, 62.81 mmol) in DMF (50 mL) was added TBDPSCl (10.36 g, 37.68 mmol). The ice bath was removed, and the reaction mixture was stirred at room temperature for 2 hours. The resulting mixture was diluted with EtOAc (400 mL), washed with water (3 x 100 mL) and brine (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20 % EA in PE to afford the title compound (11 g, 88% yield) as a light-yellow oil. MS: m/z = 398.20 [M + H]+. [0293] Step 2: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one (a mixture) [0294] To an ice-cooled solution of (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2- fluorohexahydro-1H-pyrrolizine (10 g, 25.15 mmol) and RuCl3.H2O (2.83 g, 12.57 mmol) in CCl4 (50 mL) and H2O (50 mL) was added NaIO4 (26.90 g, 125.75 mmol). The reaction mixture was stirred at 10 °C for 2 hours. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-100% EA in PE to afford a mixture of the title compounds (6.5 g, 62% yield) as a white solid. MS: m/z = 412.15 [M + H]+. [0295] Step 3: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one & [0296] A mixture of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one & (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H- pyrrolizin-3-one (6 g) was separated by Prep-Achiral-SFC with the following conditions: Column: 2, Mobile Phase B: MeOH (0.1% 2M NH3 Back Pressure(bar): 100; Wave Length: 220 nm; RT1: 3.78 min; RT2: 5.1 min. The first eluting isomer (RT1: 3.78 min) was concentrated and lyophilized overnight to give the title compound
WSGR Docket No. 62619-720.601 (Intermediate 8, 1.3 g, 21% yield) as a white solid. MS: m/z = 412.20 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 53.2, 4.0 Hz, 1H), 3.98 - 3.84 (m, 1H), 3.63 - 3.44 (m, 2H), 3.10 - 2.90 (m, 1H), 2.74 - 2.60 (m, 1H), 2.26 - 1.83 (m, 5H), 0.99 (s, 9H). The second eluting isomer (RT2: 5.1 min) was concentrated and lyophilized overnight to give the title compound (Intermediate 9, 4.5 g, 75% yield) as a white solid. MS: m/z = 412.20 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 54.4, 8.4 Hz, 1H), 3.66 - 3.51 (m, 2H), 3.46 - 3.37 (m, 1H), 2.91 - 2.74 (m, 2H), 2.11 - 1.58 (m, 5H), 0.99 (s, 9H). [0297] Intermediate 10: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-3,3-d2)methanol
[0298] Step 1: (2R,7aS)-2-Fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one [0299] To an ice-cooled solution of Intermediate 9 (750 mg, 1.822 mmol) in THF (8 mL) was added TBFA (1.82 mL, 1.822 mmol, 1M in THF) dropwise. The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-15% MeOH in CH2Cl2 to afford the title compound (300 mg, 95% yield) as a yellow solid. 1H NMR (400 MHz, DMSO-d6 J = 54.8, 8.4 Hz, 1H), 5.17 (t, J = 5.6 Hz, 1H), 3.63 - 3.51 (m, 1H), 3.26 (d, J = 5.2 Hz, 2H), 2.30 - 2.89 (m, 1H), 2.78 - 2.69 (m, 1H), 2.05 - 1.76 (m, 4H), 1.66 - 1.54 (m, 1H). [0300] Step 2: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-3,3-d2)methanol [0301] To an ice-cooled solution of (2R,7aS)-2-fluoro-7a-(hydroxymethyl)hexahydro-3H- pyrrolizin-3-one (400 mg, 2.310 mmol) in Me-THF (5 mL) under nitrogen atmosphere was added LiAlD4 (4.85 mL, 4.85 mmol, 1M in THF) dropwise. The ice bath was removed, and the reaction mixture was heated at 90 °C for 2 hours. The resulting mixture was cooled, quenched with MeOH (5 mL) in an ice bath, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-15% MeOH in CH2Cl2 (contained 5% NH3.H2O) to afford the title compound (Intermediate 10, 93 mg, 24% yield) as a light yellow solid. 1H NMR (400 MHz, DMSO-d6 - 1.92 (m, 1H), 1.92 - 1.72 (m, 3H), 1.71 - 1.57 (m, 2H). [0302] Intermediate 11: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methanol
WSGR Docket No. 62619-720.601 [0303] Step 1: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)-tetrahydro-1H-pyrrolizin-3-one [0304] To an ice-cooled solution of Intermediate 8 (4.5 g, 10.933 mmol) in THF (50 mL) under nitrogen atmosphere was added TBAF (16.40 mL, 16.399 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1.5 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 10% methanol in DCM to afford the title compound (1.74 g, 91% yield) as an off-white solid. MS: m/z = 174.10 [M + H]+. [0305] Step 2: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methanol [0306] To an ice-cooled solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-tetrahydro-1H- pyrrolizin-3-one (150 mg, 0.866 mmol) in Me-THF (2 mL) under nitrogen atmosphere was added LiAlD4 (1.82 mL, 1.819 mmol, 1M in THF) dropwise. The ice bath was removed, and the resulting mixture was heated at 90 °C for 2 hours. The resulting mixture was cooled and quenched with MeOH (2 mL) in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 1-20% MeOH in DCM (contained 2.5% NH3.H2O) to afford the title compound (Intermediate 11, 120 mg, 85% yield) as a light-yellow oil. MS: m/z = 162.10 [M + H]+. [0307] Intermediate 12: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methan-d2-ol
[0308] Step 1: (2R,7aS)-2-Fluoro-hexahydropyrrolizine-7a-carboxylic acid [0309] To a stirred solution of ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol (500 mg, 3.141 mmol) and RuCl3.H2O (35.40 mg, 0.157 mmol) in CCl4 (4 mL), MeCN (4 mL) and H2O (6 mL) under nitrogen atmosphere was added NaIO4 (2686.99 mg, 12.564 mmol) at room temperature. The reaction mixture was stirred at room temperature for 5 hours. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 (25% AcOH) / MeOH (5:1) to afford the title compound (200 mg, 36% yield) as a brown, yellow semi-solid. MS: m/z = 174.10 [M + H]+. [0310] Step 2: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methan-d2-ol [0311] To an ice-cooled solution of (2R,7aS)-2-fluoro-hexahydropyrrolizine-7a-carboxylic acid (200 mg, 1.155 mmol) in Me-THF (2 mL) under nitrogen atmosphere was added LiAlD4 (2.43 mL, 2.425 mmol, 1M in THF) dropwise. The ice bath was removed, and the reaction mixture was heated at 90 °C for 2 hours. The reaction mixture was cooled and quenched with MeOH (5 mL) in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was
WSGR Docket No. 62619-720.601 purified by silica gel flash chromatography, eluted with 1-20% methanol in dichloromethane to afford the title compound (Intermediate 12, 100 mg, 53% yield) as a yellow oil. MS: m/z = 162.15 [M + H]+. [0312] Intermediate 13: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methan-d2-ol
[0313] Step 1: (2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizine-7a(5H)-carboxylic-5,5-d2 acid [0314] To a stirred solution of Intermediate 11 (200 mg, 1.241 mmol) and RuCl3.H2O (8.39 mg, 0.037 mmol) in CCl4 (1 mL), MeCN (1 mL) and H2O (1.5 mL) under nitrogen atmosphere was added NaIO4 (1.06 g, 4.964 mmol) at room temperature. The reaction mixture was stirred at room temperature for 5 hours. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 5% MeOH in DCM (containing 2% TFA) to afford the title compound (80 mg, 37% yield) as a colorless oil. 1H NMR (300 MHz, Chloroform-d 1.95 (m, 6H). [0315] Step 2: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-5,5-d2)methan-d2-ol [0316] To an ice-cooled solution of (2R,7aS)-2-fluorotetrahydro-1H-pyrrolizine-7a(5H)- carboxylic-5,5-d2 acid (80 mg, 0.457 mmol) in Me-THF (2 mL) under nitrogen atmosphere was added LiAlD4 (0.96 mL, 0.960 mmol, 1M in THF) dropwise. The reaction mixture was stirred at 90 °C for 2 hours. The resulting mixture was cooled and quenched with MeOH in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 1-10% MeOH in CH2Cl2 (containing 2.5% NH3 H2O) to afford the title compound (Intermediate 13, 35 mg, 47% yield) as a colorless oil. MS: m/z = 164.15 [M + H]+.
WSGR Docket No. 62619-720.601 [0317] Intermediate 14: ((2,3-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane
[0318] Step 1: 2-(3,4-Difluorophenyl)acetyl chloride [0319] To an ice-cooled solution of 2-(3,4-difluorophenyl)acetic acid (5 g, 29.04 mmol) (5 g, 29.04 mmol) in 1,4-dioxane (50 mL) under nitrogen atmosphere was added SOCl2 (10 mL, 137.86 mmol) dropwise. The ice bath was removed, and the resulting mixture was heated at 80 °C for 1 hour. The resulting mixture was cooled to room temperature and concentrated under reduced pressure to give the title compound (5.5 g, crude used through) as a colorless oil. [0320] Step 2: 4-(3,4-Difluorophenyl)-3-oxobutanoic acid [0321] To a stirred solution of 1,3-ditrimethylsilyl propanedioate (3.91 g, 15.74 mmol) in THF (50 mL) under nitrogen atmosphere was added n-BuLi (5.2 mL, 13.08 mmol, 2.5 M in n-Hexane) dropwise at -78 °C. The resulting mixture was stirred in an ice bath for 0.5 hours. To the above mixture was added a solution of 2-(3,4-difluorophenyl)acetic acid (2 g, crude) in THF (5 mL) dropwise at -78 °C. The resulting mixture was stirred in an ice bath for another 0.5 hours. The reaction mixture was quenched with sat. NH4Cl (aq.) (100 mL) in an ice bath and concentrated under reduced pressure. The resulting mixture was acidified to pH = 2 with 1 M HCl (aq.) and extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (80 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-80% EA in PE to afford the title compound (850 mg, 34% yield for two steps) as a black oil. MS: m/z =213.00 [M - H]+. [0322] Step 3: 6,7-Difluoronaphthalene-1,3-diol [0323] A solution of 4-(3,4-difluorophenyl)-3-oxobutanoic acid (800 mg, 3.73 mmol) in trifluoromethanesulfonic acid (8 mL) under nitrogen atmosphere was stirred at 25°C for 16 hours. The resulting mixture was diluted with EA (150 mL), washed with water (4 x 60 mL) and brine (60 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The
WSGR Docket No. 62619-720.601 residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (550 mg, 75% yield) as a black oil. MS: m/z = 195.05 [M - H]+. [0324] Step 4: 6,7-Difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol [0325] To a stirred solution of 6,7-difluoronaphthalene-1,3-diol (520 mg, 2.65 mmol) and (2- bromoethynyl)triisopropylsilane (761.91 mg, 2.91 mmol) in 1,4-dioxane (6 mL) under nitrogen atmosphere were added [Ru(p-cymene)Cl2]2 (162.34 mg, 0.26 mmol) and KOAc (520.35 mg, 5.30 mmol) at room temperature. The resulting mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (600 mg, 60% yield) as a black solid. MS: m/z = 377.00 [M + H]+. [0326] Step 5: 6,7-Difluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-ol [0327] To an ice-cooled solution of 6,7-difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol (600 mg, 1.59 mmol) and DIEA (267.75 mg, 2.07 mmol) in DCM (6 mL) under nitrogen atmosphere was added bromo(methoxy)methane (189.18 mg, 1.51 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was diluted with CH2Cl2 (100 mL), washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (450 mg, 67% yield) as a yellow oil. MS: m/z = 418.95 [M - H]+. [0328] Step 6: 6,7-Difluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate [0329] To a stirred solution of 6,7-difluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-ol (450 mg, 1.07 mmol) in DCM (5 mL) under nitrogen atmosphere were added DIEA (414.87 mg, 3.21 mmol) and Tf2O (452.82 mg, 1.60 mmol) dropwise at -40 °C. The resulting mixture was stirred at -40 °C for 1 hour. The resulting mixture was quenched with sat. aq. NaHCO3 (80 mL) in an ice bath and extracted with DCM (3 x 80 mL). The combined organic layers were washed with brine (80 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (550 mg, 93% yield) as a yellow oil. MS: m/z = 550.90 [M - H]+. [0330] Step 7: ((2,3-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-1-yl)ethynyl)triisopropylsilane [0331] To a stirred solution of 6,7-difluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (500 mg, 0.90 mmol) and
WSGR Docket No. 62619-720.601 B2Pin2 (459.51 mg, 1.81 mmol) in toluene (5 mL) under nitrogen atmosphere were added Pd(dppf)Cl2.CH2Cl2 (73.70 mg, 0.09 mmol) and AcOK (266.39 mg, 2.71 mmol) at room temperature. The reaction mixture was stirred at 130 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (Intermediate 14, 210 mg, 43% yield) as a yellow solid. MS: m/z = 529.15 [M - H]+. 1H NMR (400 MHz, DMSO-d6 J = 2.4 Hz, 1H), 7.37 (d, J = 2.8 Hz, 1H), 5.32 (s, 2H), 3.42 (s, 3H), 1.34 (s, 12H), 1.29 - 1.01 (m, 21H). 19F NMR (376 MHz, DMSO- d6 [0332] Intermediate 15: ((2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl-3-d)ethynyl)triisopropylsilane
[0333] Step 1: 2-(3-Bromo-4-fluorophenyl)acetyl chloride [0334] To a stirred solution of 2-(3-bromo-4-fluorophenyl)acetic acid (15 g, 64.37 mmol) in 1,4- dioxane (150 mL) under nitrogen atmosphere was added SOCl2 (100 mL) dropwise at room temperature. The resulting mixture was heated at 80 °C for 2 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure to give the title compound (16 g, crude used directly) as a colorless oil. [0335] Step 2: 4-(3-Bromo-4-fluorophenyl)-3-oxobutanoic acid [0336] To a stirred solution of 1,3-ditrimethylsilyl propanedioate (23.71 g, 95.43 mmol) in THF (250 mL) under nitrogen atmosphere were added n-BuLi (2.5 M in n-hexane, 32 mL, 80 mmol) dropwise at -78 °C. The reaction mixture was stirred in an ice bath for 0.5 hour. To the above mixture was added a solution of 2-(3-Bromo-4-fluorophenyl)acetyl chloride (16 g, crude) in THF (35 mL) dropwise at -78°C. The reaction mixture was stirred in an ice bath for 2 hours. The
WSGR Docket No. 62619-720.601 resulting mixture was quenched with sat. NH4Cl (aq.) (500 mL) in an ice bath and concentrated under reduced pressure. The resulting mixture was acidified to pH = 2 with 1 M HCl (aq.) and extracted with EtOAc (3 x 500 mL). The combined organic layers were washed with brine (500 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to give the title compound (10.8 g, 60% yield for two steps) as a yellow oil. MS: m/z = 272.85, 274.85 [M - H]-. [0337] Step 3: 6-Bromo-7-fluoronaphthalene-1,3-diol [0338] A solution of 4-(3-bromo-4-fluorophenyl)-3-oxobutanoic acid (10.8 g, 29.26 mmol) in CF3SO3H (150 mL) under nitrogen atmosphere was stirred at room temperature for 16 hours. The resulting mixture was diluted with EtOAc (800 mL), washed with water (4 x 300 mL) and brine (400 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (7 g, 69% yield) as a brown solid. MS: m/z =254.95, 256.95 [M - H]-. [0339] Step 4: ((6-Bromo-7-fluoronaphthalene-1,3-diyl)bis(oxy))bis(tert-butyldimethylsilane) [0340] To an ice-cooled solution of 6-bromo-7-fluoronaphthalene-1,3-diol (5.0 g, 19.44 mmol) in DCM (50 mL) under nitrogen atmosphere were added Et3N (13.52 mL, 97.24 mmol) and TBSOTf (17.88 mL, 77.80 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was diluted with CH2Cl2 (400 mL), washed with brine (5 x 100 mL), dried over anhydrous Na2SO4. filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to the title compound (4.4 g, 46% yield) as a light yellow oil. MS: m/z =485.10, 487.10 [M + H]+. [0341] Step 5: ((7-Fluoronaphthalene-1,3-diyl-6-d)bis(oxy))bis(tert-butyldimethylsilane) [0342] To a stirred solution of ((6-bromo-7-fluoronaphthalene-1,3-diyl)bis(oxy))bis(tert- butyldimethylsilane) (4.4 g, 9.06 mmol) in CH3OD (50 mL) under D2 atmosphere was added Pd/C (4.4 g, 10% wt) at room temperature. The resulting mixture was stirred at room temperature for 1 hour under D2 atmosphere. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-60% EA in PE to afford the title compound (2.0 g, 54% yield) as a light yellow oil. MS: m/z = 408.25 [M + H]+. [0343] Step 6: 7-Fluoronaphthalene-6-d-1,3-diol [0344] To a stirred solution of ((7-fluoronaphthalene-1,3-diyl-6-d)bis(oxy))bis(tert- butyldimethylsilane) (2 g, 4.90 mmol) in THF (30 mL) under nitrogen atmosphere were added
WSGR Docket No. 62619-720.601 TBAF (12.26 mL, 12.26 mmol, 1 M in THF) dropwise at room temperature. The reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was diluted with EtOAc (300 mL), washed with brine (5 x 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (550 mg, 63% yield) as a light-yellow oil. MS: m/z = 180.10 [M + H]+. 1H NMR (300 MHz, Methanol-d4 J = 2.1 Hz, 1H), 6.50 (d, J = 2.1, 0.9 Hz, 1H). [0345] Step 7: 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-6-d-1,3-diol [0346] To a stirred solution of 7-fluoronaphthalene-6-d-1,3-diol (550 mg, 3.07 mmol), [Ru(p- cymene)Cl2]2 (962.45 g, 3.68 mmol) and KOAc (662.79 mg, 6.75 mmol) in 1,4-dioxane (10 mL) under nitrogen atmosphere was added (2-bromoethynyl)triisopropylsilane (1.60 g, 6.13 mmol) at room temperature. The reaction mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (640 mg, 58% yield) as a brown solid. MS: m/z = 360.20 [M + H]+. [0347] Step 8: 7-Fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-6-d-1-ol [0348] To an ice-cooled solution of 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-6-d-1,3-diol (630 mg, 1.75 mmol) and DIEA (0.23 g, 1.75 mmol) in DCM (10 mL) under nitrogen atmosphere was added bromo(methoxy)methane (0.20 g, 1.58 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with water (100 mL) and extracted with DCM (3 x 200 mL). The combined organic layers were washed with brine (300 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (490 mg, 69% yield) as a brown solid. MS: m/z = 404.25 [M + H]+. 1H NMR (300 MHz, Chloroform-d 1H), 6.97 - 6.96 (m, 1H), 6.80 - 6.79 (m, 1H), 5.25 (s, 2H), 3.50 (s, 3H), 1.22 - 1.15 (m, 21H). [0349] Step 9: 7-Fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl-6-d trifluoromethanesulfonate [0350] To a stirred solution of 7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-6-d-1-ol (470 mg, 1.16 mmol) in DCM (8 mL) under nitrogen atmosphere were added DIEA (451.55 mg, 3.49 mmol) and Tf2O (0.30 mL, 1.74 mmol) dropwise at -40 °C. The reaction mixture was stirred at -40 °C for 1 hour. The resulting mixture was quenched with saturated aq. NaHCO3 (30 mL) and extracted with DCM (3 x 50 mL). The
WSGR Docket No. 62619-720.601 combined organic layers were washed with brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the title compound (610 mg) as a light- yellow oil, which was used in the next step without further purification. MS: m/z = 534.15 [M - H]-. [0351] Step 10: ((2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-1-yl-3-d)ethynyl)triisopropylsilane [0352] To a stirred solution of 7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl-6-d trifluoromethanesulfonate (610 mg, crude) and B2Pin2 (347.03 mg, 1.37 mmol) in 1,4-dioxane (7 mL) under nitrogen atmosphere were added KOAc (335.30 mg, 3.42 mmol) and Pd(dppf)Cl2.CH2Cl2 (83.33. mg, 0.11 mmol) at room temperature. The reaction mixture was heated at 110 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (Intermediate 15, 360 mg, 60% yield for two steps) as a yellow solid. MS: m/z = 514.30 [M + H]+. 1H NMR (300 MHz, DMSO-d6 J = 6.0 Hz, 1H), 7.58 (d, J = 3.0 Hz, 1H), 7.39 (d, J = 3.0 Hz, 1H), 5.32 (s, 2H), 3.42 (s, 3H), 1.35 (s, 12H), 1.23 - 1.10 (m, 21H). [0353] Intermediate 16: ((2,4-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane
[0354] Intermediate 16 was prepared in a similar manner to Intermediate 14. 1H NMR (300 MHz, DMSO-d6 J = 2.4 Hz, 1H), 5.37 (s, 2H), 3.42 (s, 3H), 1.35 (s, 12H), 1.16 - 1.08 (m, 21H). [0355] Intermediate 17: ((2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl-4-d)ethynyl)triisopropylsilane
WSGR Docket No. 62619-720.601
[0358] Step 1: 6-Bromo-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol [0359] To a stirred solution 6-bromo-7-fluoronaphthalene-1,3-diol (refer to Intermediate 15 for detail procedures, 2 g, 7.780 mmol) and [Ru(p-Cymene)Cl2]2 (0.48 g, 0.778 mmol) in 1,4-dioxane (20 mL) under nitrogen atmosphere were added (2-bromoethynyl)tris(propan-2-yl)silane (2.24 g, 8.558 mmol) and KOAc (1.53 g, 15.560 mmol) at room temperature. The reaction mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (2 g, 58% yield) as a brown solid. MS: m/z = 436.90, 438.90 [M + H]+.
WSGR Docket No. 62619-720.601 [0360] Step 2: 6-Bromo-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- ol [0361] To an ice-cooled solution of 6-bromo-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene- 1,3-diol (2 g, 4.572 mmol) and DIEA (768.23 mg, 5.944 mmol) in DCM (20 mL) under nitrogen atmosphere was added bromo(methoxy)methane (542.80 mg, 4.343 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with water (50 mL), then extracted with DCM (3 x 100 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (2 g, 90% yield) as a brown solid. MS: m/z = 480.85, 482.85 [M + H]+. [0362] Step 3: 3-Fluoro-5-hydroxy-7-(methoxymethoxy)-4-((triisopropylsilyl)ethynyl)-2- naphthonitrile [0363] To a stirred solution 6-bromo-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-ol (1 g, 2.077 mmol) and Pd(PPh3)4 (1.20 g, 1.038 mmol) in DMF (10 mL) under nitrogen atmosphere was added Zn(CN)2 (0.27 g, 2.285 mmol) at room temperature. The reaction mixture was heated at 100 °C for 16 hours. The resulting mixture was cooled to room temperature and filtered. The filtrate was diluted with EtOAc (200mL), washed with H2O (3 x 80 mL) and brine (100 mL). The organic layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-30% EA in PE to afford the title compound (500 mg, 56% yield) as a yellow solid. MS: m/z = 426.20 [M - H]-. [0364] Step 4: 6-Cyano-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate [0365] To a stirred solution of 3-fluoro-5-hydroxy-7-(methoxymethoxy)-4- ((triisopropylsilyl)ethynyl)-2-naphthonitrile (500 mg, 1.169 mmol) and DIEA (453.40 mg, 3.507 mmol) in DCM (5 mL) under nitrogen atmosphere was added Tf2O (494.88 mg, 1.754 mmol) dropwise at -40 °C. The reaction mixture was stirred at -40 °C for 1 hour. The resulting mixture was quenched with sat. aq. NaHCO3 (50 mL) and extracted with DCM (3 x 50 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the title compound (700 mg, crude used through) as a brown solid. MS: m/z = 558.15 [M - H]-.
WSGR Docket No. 62619-720.601 [0366] Step 5: 3-Fluoro-7-(methoxymethoxy)-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4- ((triisopropylsilyl)ethynyl)-2-naphthonitrile [0367] To a stirred solution of 6-cyano-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (700 mg, crude) and B2Pin2 (594.05 mg, 2.340 mmol) in toluene (8 mL) under nitrogen atmosphere were added Pd(dppf)Cl2.CH2Cl2 (85.59 mg, 0.117 mmol) and KOAc (344.38 mg, 3.510 mmol) at room temperature. The reaction mixture was heated at 110 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (Intermediate 18, 270 mg, 42% yield) as a white solid. 1H NMR (400 MHz, DMSO-d6) J = 2.4 Hz, 1H), 7.60 (d, J = 2.8 Hz, 1H), 5.41 (s, 2H), 3.48 (s, 3H), 1.40 (s, 12H), 1.24 - 1.12 (m, 21H). [0368] Intermediate 19: (S)-Azepan-2-ylmethanol
[0369] Step 1: (S)-Azepane-2-carboxylic acid [0370] To a solution of (2S)-1-tert-butoxycarbonylazepane-2-carboxylic acid (3.00 g, 12.3 mmol) in CH2Cl2 (15 mL) was added HCl/1,4-dioxane (4 M, 15 mL) and stirred at 0°C for 1 h. The reaction mixture was concentrated under reduced pressure to give the title compound (2.1 g, crude) as a white solid, which was used in the next step without further purification. MS: m/z = 143.8 [M + H]+. [0371] Step 2: (S)-Azepan-2-ylmethanol [0372] To a solution of (S)-azepane-2-carboxylic acid (2.2 g, 12.2 mmol) in THF (40 mL) was added dropwise LiAlH4 (2.5 M in n-hexane, 24.5 mL) at 0 °C under N2 atmosphere. The reaction was warmed up to 20 °C slowly and stirred for 1 h. Na2SO4·10H2O (29.5 g, 90 mmol) was added into the mixture slowly at 0 °C, and the mixture was stirred for 30 min. Then the mixture was filtered and concentrated under reduced pressure to give the title compound (Intermediate 19, 1 g, crude) as a brown solid, which was used in the next step without further purification. MS: m/z = 130.4 [M+ H]+.1H NMR (400 MHz, Chloroform-d J = 4.4, 10.4 Hz, 1H), 3.11 (t, J = 10.0 Hz, 1H), 2.97 - 2.88 (m, 1H), 2.79 - 2.71 (m, 2H), 1.80 - 1.70 (m, 4H), 1.61 - 1.44 (m, 4H). [0373] Intermediate 20 & 21: N-(6-Fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine & 6-Fluoro-4-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[0374] Step 1: 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol [0375] To a solution of 7-fluoronaphthalene-1,3-diol (50 g, 281 mmol) and 2- bromoethynyl(triisopropyl)silane (77 g, 295 mmol) in 1,4-dioxane (334 mL) were added KOAc (55.1 g, 561 mmol) and dichlororuthenium:1-isopropyl-4-methyl-benzene (17.2 g, 28.1 mmol). The mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 5% of EtOAc in petroleum ether) to give the title compound (70 g, 194 mmol, 69% yield) as a brown solid. MS: m/z = 359.1 [M+ H]+.1H NMR (400 MHz, Chloroform-d J = 5.6, 9.2 Hz, 1H), 7.17 (t, J = 8.8 Hz, 1H), 6.77 - 6.62 (m, 2H), 1.21 - 1.15 (m, 21H). 19F NMR (376 MHz, Chloroform-d [0376] Step 2: 7-Fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3- diylbis(trifluoromethanesulfonate) [0377] To a solution of 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol (140 g, 390 mmol) and DIPEA (408 mL, 2.34 mol) in CH2Cl2 (3.5 L) was added Tf2O (258 ml, 1.56 mol) dropwise under N2 atmosphere at 0 °C. The mixture was stirred at 0 °C under N2 atmosphere for 2 h. The reaction mixture was partitioned between CH2Cl2 (500 mL) and H2O (2 L). The organic phase was separated, washed with brine (500 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 3% of EtOAc in petroleum ether) to give 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3- diyl bis(trifluoromethanesulfonate) (218 g, 340 mmol, 87% yield) as a brown solid. 1H NMR (400 MHz, Chloroform-d J = 5.2, 9.2 Hz, 1H), 7.80 (d, J = 2.4 Hz, 1H), 7.54 - 7.47 (m, 2H), 1.26 - 1.21 (m, 3H), 1.19 - 1.15 (m, 18H). [0378] Step 3: 3-((Diphenylmethylene)amino)-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate
WSGR Docket No. 62619-720.601 [0379] A mixture of 7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3- diylbis(trifluoromethanesulfonate) (94 g, 151 mmol), diphenylmethanimine (54.7 g, 302 mmol, 50.7 mL), (5-diphenylphosphanyl-9,9-dimethyl-xanthen-4-yl)-diphenyl-phosphane (17.5 g, 30.2 mmol), Pd2(dba)3 (6.91 g, 7.55 mmol) and Cs2CO3 (148 g, 453 mmol) in toluene (1800 mL) was degassed, purged with N2 three times, and stirred at 100 °C under N2 atmosphere for 2 h. The reaction mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 3% of EtOAc in petroleum ether) to give the crude product (80 g) as a brown solid. The crude was triturated with MeOH (150 mL) at 25°C for 20 min and filtered. The filter cake was dried under reduced pressure to give the title compound (70 g, 94.2 mmol, 62% yield) as a yellow solid. MS: m/z = 654.3 [M+ H]+. 1H NMR (400 MHz, Chloroform-d J = 8.0 Hz, 2H), 7.57 - 7.51 (m, 2H), 7.48 - 7.41 (m, 2H), 7.27 (s, 2H), 7.26 - 7.19 (m, 2H), 7.17 - 7.01 (m, 4H), 1.22 - 1.13 (m, 21H). 19F NMR (376 MHz, [0380] Step 4: N-(6-Fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine & 6-Fluoro-4-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [0381] A mixture of 3-((diphenylmethylene)amino)-7-fluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (50 g, 76.5 mmol), 4,4,5,5- tetramethyl-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1,3,2-dioxaborolane (39 g, 153 mmol), KOAc (30 g, 306 mmol), and Pd(dppf)Cl2 (11.2 g, 15.3 mmol) in 1,4-dioxane (600 mL) was degassed, purged with N2 three times, and stirred at 100 °C under N2 atmosphere for 16 h. The reaction mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0~100% of EtOAc in petroleum ether) to give the title compound (Intermediate 20, 9 g, 14.3 mmol, 19% yield) as a yellow solid and the title compound (Intermediate 21, 10 g, 21.4 mmol, 28% yield) as a brown oil. Spectra for Intermediate 20: MS: m/z = 632.4 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 7.60 - 7.53 (m, 1H), 7.53 - 7.47 (m, 2H), 7.42 (t, J = 9.2 Hz, 1H), 7.34 - 7.27 (m, 4H), 7.22 - 7.16 (m, 2H), 7.14 - 7.10 (m, 1H), 1.27 (s, 12H), 1.14 - 1.08 (m, 21H), 19F NMR (376 MHz, m/z = 468.3 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 9.0 Hz, 1H), 7.23 - 7.17 (m, 1H), 6.89 (d, J = 2.4 Hz, 1H), 5.48 (s, 2H), 1.33 (s, 12H), 1.17 - 1.11 (m, 21H). 19F NMR (376 [0382] Intermediate 22: ((2R,7aR)-2-Fluoro-2,3-dihydro-1H-pyrrolizin-7a(5H)-yl)methanol
WSGR Docket No. 62619-720.601
[0383] Step 1: (2R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-2,3,5,7a-tetrahydro- 1H-pyrrolizine [0384] To a stirred solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro- 5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one (refer to Intermediate 36 for detail procedures, 1 g, 2.44 mmol) in DCM (100 mL) under nitrogen atmosphere was added tetrafluoroboranuide; trimethyloxidanium (1.81 g, 12.21 mmol) at room temperature. The resulting mixture was heated at 35 °C for 3.5 hours. To the above mixture were added MeOH (200 mL) and NaBH4 (1.85 g, 48.84 mmol) in an ice bath. The resulting mixture was stirred in an ice bath for 40 min. The resulting mixture was quenched with NaHCO3 (40 mL) in an ice bath, then extracted with EA (3 x 500 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by prep-TLC (EA : PE = 1 : 3) to afford the title compound (220 mg, 22% yield) as a yellow oil. MS: m/z = 396.25 [M + H]+. [0385] Step 2: ((2R,7aR)-2-Fluoro-2,3-dihydro-1H-pyrrolizin-7a(5H)-yl)methanol [0386] To a stirred solution of (2R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluoro- 2,3,5,7a-tetrahydro-1H-pyrrolizine (200 mg, 0.50 mmol) in THF (2 mL) under nitrogen atmosphere was added TBAF (0.75 mL, 0.75 mmol, 1 M solution in THF) dropwise at room temperature. The resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 10% MeOH in CH2Cl2 to afford the title compound (Intermediate 22, 45 mg, 56% yield) as a light-yellow oil. MS: m/z = 158.20 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 5.82 - 5.66 (m, 2H), 5.32 - 5.17 (m, 1H), 4.14 - 4.09 (m, 1H), 3.76 - 3.71 (m, 1H), 3.53 - 3.51 (m, 1H), 3.40 - 3.20 (m, 3H), 2.60 (bs, 1H), 2.16 - 2.00 (m, 2H). [0387] Intermediate 23: (S)-2,4-Difluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601 [0388] Step 1: (S)-5-Chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0389] A solution of (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 5 for detail procedures, 1.2 g, 3.49 mmol) and KF (304.72 mg, 5.24 mmol) in DMSO (60 mL) under nitrogen atmosphere was heated at 80 °C for 16 hours. The resulting mixture was cooled to room temperature. The resulting mixture was diluted with EA (1000 mL), washed with water (3 x 200 mL) and brine (200 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (15 : 1) to afford the title compound (870 mg, 76% yield) as a light yellow solid. MS: m/z = 327.05 [M + H]+. [0390] Step 2: (S)-2,4-difluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0391] To a stirred mixture of (S)-5-chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (200 mg, 0.61 mmol) and ((2-fluoro-6- (methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-1- yl)ethynyl)triisopropylsilane (470.61 mg, 0.91 mmol) in H2O (2 mL) and THF (10 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (89.16 mg, 0.12 mmol) and K3PO4 (779.60 mg, 3.67 mmol) at room temperature. The resulting mixture was heated at 80 °C for 3 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by prep-TLC (PE : EA = 1 : 2) to afford the title compound (Intermediate 23, 280 mg, 67% yield) as a light yellow solid. MS: m/z = 677.45 [M + H]+. [0392] Intermediate 24 & 25: (6S,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d
WSGR Docket No. 62619-720.601
[0393] Step 1: A mixture of ethyl (2S,7aS)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aR)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate- 2-d [0394] To an ice-cooled solution of ethyl 2,5-dioxo-tetrahydropyrrolizine-7a-carboxylate (20 g, 94.68 mmol) in ethyl alcohol (150 mL) was added NaBD4 (796.53 mg, 18.93 mmol). The resulting mixture was stirred in an ice bath for 10 min. The resulting mixture was quenched with sat. aq. NH4Cl (15 mL), diluted with brine (15 mL), and extracted with EtOAc (3 x 150 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (10 / 1) to give two peaks. The first eluting peak (less polar isomer mixture) was concentrated under reduced pressure to give a mixture of the title compounds (3 g, 17% yield) as an off-white solid. The second eluting peak (polar isomer mixture) was concentrated under reduced pressure to give mixture of ethyl (2S,7aS)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate-2-d & ethyl (2R,7aR)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate-2-d (10 g, 56% (brs, 1H), 4.15 - 4.01 (m, 2H), 3.64 (d, J = 12.0 Hz, 1H), 2.79 - 2.74 (m, 1H), 2.62 - 2.53 (m, 2H), 2.32 - 2.07 (m, 3H), 1.84 (d, J = 12.9 Hz, 1H), 1.17 (t, J = 6.9 Hz, 3H). [0395] Step 2: A mixture of ethyl (2S,7aR)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aS)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate-2-d [0396] To a stirred solution of ethyl (2S,7aS)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d & ethyl (2R,7aR)-2-hydroxy-5-oxotetrahydro-1H-pyrrolizine-7a(5H)-carboxylate-
WSGR Docket No. 62619-720.601 2-d (1 g, 4.66 mmol) in DCM (10 mL) under nitrogen atmosphere was added DAST (1.13 g, 7.00 mmol) dropwise at - 70 °C. The resulting mixture was warmed to room temperature and stirred at room temperature for 16 hours. The resulting mixture was quenched with MeOH (5 mL) in an ice bath, diluted with water (10 mL), and extracted with DCM (3 x 20 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (2 / 1) to afford a mixture of the title compounds (420 mg, 41% yield) as a colorless oil. MS: m/z = 217.05 [M + (m, 3H), 2.49 - 2.41 (m, 1H), 2.30 - 2.07 (m, 2H), 1.38 - 1.29 (m, 3H). [0397] Step 3: A mixture of (6S,7aR)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one- 6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one-6-d. [0398] To an ice-cooled solution of a mixture of ethyl ((2S,7aR)-2-fluoro-5-oxotetrahydro-1H- pyrrolizine-7a(5H)-carboxylate-2-d & ethyl (2R,7aS)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine- 7a(5H)-carboxylate-2-d (100 mg, 0.46 mmol) in EtOH (3 mL) was added NaBH4 (19.24 mg, 0.50 mmol). The resulting mixture was stirred in an ice bath for 30 min. The resulting mixture was quenched with saturated aq. NH4Cl (10 mL) and extracted with EA (3 x 10 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (5 / 1) to afford a mixture of the title compounds (60 mg, 74% yield) as a white solid. MS: m/z = 175.10 [M + H]+. [0399] Step 4: A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro- 3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro- 3H-pyrrolizin-3-one-6-d [0400] To an ice-cooled solution of a mixture of (6S,7aR)-6-fluoro-7a-(hydroxymethyl)hexahydro- 3H-pyrrolizin-3-one-6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one- 6-d (500 mg, 2.87 mmol) and imidazole (390.81 mg, 5.74 mmol) in DMF (5 mL) under nitrogen atmosphere was added tert-butylchlorodiphenylsilane (946.74 mg, 3.44 mmol). The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was diluted with water (300 mL), extracted with CH2Cl2 (3 x 500 mL). The organic layer was dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 20% EA in PE to give a mixture of the title compounds (999 mg, 84% yield) as a white solid. MS: m/z = 413.20 [M + H]+
WSGR Docket No. 62619-720.601 [0401] Step 5: (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d [0402] A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-6-d (2.5 g, 6.06 mmol) was separated by Chiral-SFC with following conditions: Back Pressure(bar): 100; Detector: UV 220 nm; RT1 (min): 6.63; RT2 (min): 8.07. [0403] The first eluting peak (RT1: 6.63 min) was concentrated under reduced pressure to give the title compound (Intermediate 24, 1 g, 40% yield) as a white solid. 1H NMR (400 MHz, DMSO-d6) 1H), 2.81 - 2.56 (m, 1H), 2.28 - 1.78 (m, 5H), 0.99 (s, 9H). The second eluting peak (RT1: 8.07 min) was concentrated under reduced pressure to give the title compound (Intermediate 25, 1 g, 40% yield) as a white solid. 1 5H), 3.95 - 3.86 (m, 1H), 3.71 - 3.37 (m, 2H), 3.05 - 2.93 (m, 1H), 2.71 - 2.62 (m, 1H), 2.37 - 1.79 (m, 5H), 0.99 (s, 9H). [0404] Intermediate 26: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2-d)methanol
[0405] Step 1: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one-6-d [0406] To a stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d (940 mg, 2.27 mmol) in THF (10 mL) under nitrogen atmosphere was added TBAF (3.6 mL, 13.76 mmol, 1 M in THF) dropwise at room temperature. The resulting mixture was stirred at room temperature for 1 hour and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (10 : 1) to afford the title compound (330 mg, 83% yield) as a yellow oil. MS: m/z = 175.10 [M + H]+. [0407] Step 2: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2-d)methanol [0408] To an ice-cooled solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H- pyrrolizin-3-one-6-d (400 mg, 2.29 mmol) in Me-THF (4 mL) under nitrogen atmosphere was added LiAlH4 (4.8 mL, 4.8 mmol, 1 M in THF) dropwise. The ice bath was removed, and the resulting mixture was heated at 90 °C for 2 hours. The resulting mixture was cooled and quenched
WSGR Docket No. 62619-720.601 with MeOH (50 mL) in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (5 : 1) to afford the title compound (Intermediate 26, 200 mg, 54% yield) as a yellow oil. 1H NMR 1H), 2.79 - 2.75 (m, 1H), 2.07 - 1.94 (m, 1H), 1.90 - 1.77 (m, 3H), 1.72 - 1.60 (m, 2H). [0409] Intermediate 27: ((2S,7aR)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2-d)methanol
[0410] Intermediate 27 was prepared in a similar manner to Intermediate 26. MS: m/z = 161.15 [M + H]+. [0411] Intermediate 28: ((3-Chloro-2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane
[0412] Step 1: 2-(3-Chloro-4-fluorophenyl)acetyl chloride [0413] To a stirred solution of 2-(3-chloro-4-fluorophenyl)acetic acid (10 g, 53.02 mmol) in 1,4- dioxane (100 mL) under nitrogen atmosphere was added SOCl2 (20.00 mL) dropwise at room temperature. The resulting mixture was heated at 80 °C for 2 hours. The resulting mixture was concentrated under reduced pressure to give crude product (10.9 g crude), which was used in the next step without further purification. [0414] Step 2: 4-(3-Chloro-4-fluorophenyl)-3-oxobutanoic acid [0415] To a stirred solution of 2-(3-chloro-4-fluorophenyl)acetyl chloride (10.9 g, crude) in THF (110 mL) under nitrogen atmosphere was added n-butyllithium (2.5 M in n-hexane, 25.2 mL, 63.17 mmol) dropwise at -78 °C. The resulting mixture was stirred in an ice bath for 0.5 hours. The reaction was quenched with sat. aq. NH4Cl (500 mL) in an ice bath and concentrated under reduced pressure. The resulting mixture was acidified to pH = 2 with aq.1 M. HCl and extracted with EA (3 x 500 mL). The combined organic layers were washed with brine (500 mL), dried over
WSGR Docket No. 62619-720.601 anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (8 g, 65% yield for two steps) as a black oil. MS: m/z = 227.00 [M - H]-. [0416] Step 3: 6-Chloro-7-fluoronaphthalene-1,3-diol [0417] 4-(3-Chloro-4-fluorophenyl)-3-oxobutanoic acid (8 g, 34.68 mmol) was dissolved in CF3SO3H (150 mL) at room temperature, and the mixture was stirred at this temperature for 16 hours under nitrogen atmosphere. The resulting mixture was diluted with EtOAc (800 mL), washed with water (4 x 300 mL) and brine (500 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (6.5 g, 88% yield) as a black oil. MS: m/z = 210.85 [M - H]-. [0418] Step 4: 6-Chloro-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol [0419] To a stirred solution of 6-chloro-7-fluoronaphthalene-1,3-diol (6.5 g, 30.57 mmol) in 1,4- dioxane (65 mL) under nitrogen atmosphere were added (2-bromoethynyl)triisopropylsilane (8787.10 mg, 33.63 mmol), [Ru(p-cymene)Cl2]2 (1872.31 mg, 3.05 mmol) and KOAc (6001.16 mg, 61.14 mmol) at room temperature. The resulting mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (6 g, 49% yield) as a black oil. MS: m/z = 391.10 [M - H]-. [0420] Step 5: 6-Chloro-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- ol [0421] To an ice-cooled solution of 6-chloro-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalene- 1,3-diol (6 g, 15.26 mmol) and DIEA (2565.40 mg, 19.84 mmol) in DCM (60 mL) under nitrogen atmosphere was added bromo(methoxy)methane (1717.19 mg, 13.74 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with water (100 mL) and extracted with DCM (3 x 300 mL). The combined organic layers were washed with brine (300 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (5 g, 74% yield) as a brown oil. MS: m/z = 434.85 [M - H]-. [0422] Step 6: 6-Chloro-7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1- yl trifluoromethanesulfonate
WSGR Docket No. 62619-720.601 [0423] To a stirred solution of 6-chloro-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-ol (4.8 g, 10.98 mmol) in DCM (50 mL) under nitrogen atmosphere were added DIEA (4.26 g, 32.94 mmol) and Tf2O (4.65 g, 16.47 mmol) dropwise at - 40 °C. The resulting mixture was stirred at -40 °C for 1 hour. The resulting mixture was quenched with sat. aq. NaHCO3 (50 mL) and extracted with DCM (3 x 300 mL). The combined organic layers were washed with brine (300 mL), dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure to give a crude product (5 g, crude), which was used in the next step without further purification. MS: m/z = 567.10 [M - H]-. [0424] Step 7: ((3-Chloro-2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane [0425] To a stirred solution of 6-chloro-7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (5 g, crude) and bis(pinacolato)diboron (4462.29 mg, 17.57 mmol) in 1,4-dioxane (50 mL) under nitrogen atmosphere were added AcOK (2586.86 mg, 26.358 mmol) and Pd(dppf)Cl2•CH2Cl2 (715.73 mg, 0.87 mmol) at room temperature. The resulting mixture was heated at 90 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (Intermediate 28, 2 g, 33% yield for two steps) as an orange solid. 1H Hz, 1H), 5.32 (s, 2H), 3.34 (s, 3H), 1.34 (s, 12H), 1.22 - 1.10 (m, 21H). [0426] Intermediate 29 & 30: (6S,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl-d2)-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl-d2)- 6-fluorohexahydro-3H-pyrrolizin-3-one-6-d
[0427] Step 1: A mixture of (6S,7aR)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H-pyrrolizin-3- one-6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H-pyrrolizin-3-one-6-d [0428] To an ice-cooled solution of ethyl (2S,7aR)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine- 7a(5H)-carboxylate-2-d & ethyl (2R,7aS)-2-fluoro-5-oxotetrahydro-1H-pyrrolizine-7a(5H)- carboxylate-2-d (refer to Intermediate 24 & 25 for detail procedures, 5 g mixture, 23.12 mmol) in EtOH (50 mL) under nitrogen atmosphere was added sodium tetrahydroborate-d4 (1.06 g, 25.43 mmol) in portions. The resulting mixture was stirred in an ice bath for 1 hour under nitrogen
WSGR Docket No. 62619-720.601 atmosphere. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (10 : 1) to afford a mixture of the title compounds (3 g, 73% yield) as a white solid. MS: m/z = 177.05 [M + H]+. [0429] Step 2: A mixture of (6S,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl-d2)-6- fluorohexahydro-3H-pyrrolizin-3-one-6-d & (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl-d2)- 6-fluorohexahydro-3H-pyrrolizin-3-one-6-d [0430] To an ice-cooled solution of (6S,7aR)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H- pyrrolizin-3-one-6-d & (6R,7aS)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H-pyrrolizin-3-one-6- d (2.1 g mixture, 11.91 mmol) and 1H-imidazole (1.62 g, 23.83 mmol) in DMF (21 mL) under nitrogen atmosphere was added tert-butylchlorodiphenylsilane (3.93 g, 14.30 mmol). The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was diluted with CH2Cl2 (300 mL), washed with water (3 x 100 mL) and brine (100 mL). The organic layer was dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 20% EA in PE to give a mixture of the title compounds (4.7 g, 95% yield) as a white solid. MS: m/z = 415.25 [M + H]+. The mixture (2.5 g mixture, 6.03 mmol) was separated by Prep-SFC with 2, Mobile Phase B: IPA: MeCN =1 : 1; Flow rate: 140 mL/min; Gradient: Isocratic 25% B; Column (min): 8.07. The first eluting peak (RT1: 6.63 min) was concentrated under reduced pressure to give the title compound (Intermediate 29, 1 g, 40% yield) as white solid. 1H NMR (400 MHz, DMSO-d6 2.74 - 2.59 (m, 1H), 2.27 - 2.05 (m, 3H), 2.00 -1.81 (m, 2H), 0.99 (s, 9H). The second eluting peak (RT1: 8.07 min) was concentrated under reduced pressure to give the title compound (Intermediate 30, 1 g, 40% yield) as a white solid. 1H NMR (400 MHz, DMSO-d6 7.42 (m, 6H), 3.94 - 3.86 (m, 1H), 3.05 - 2.92 (m, 1H), 2.78 - 2.58 (m, 1H), 2.27 - 2.06 (m, 3H), 2.00 - 1.80 (m, 2H), 0.99 (s, 9H). [0431] Intermediate 31: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2,5,5-d3)methan- d2-ol
[0432] Step 1: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl-d2)hexahydro-3H-pyrrolizin-3-one-6-d
WSGR Docket No. 62619-720.601 [0433] To an ice-cooled solution of Intermediate 30 (1 g, 2.41 mmol) in THF (10 mL) under nitrogen atmosphere was added TBAF (3.61 mL, 3.61 mmol, 1 M in THF) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 9% methanol in dichloromethane to give the title compound (400 mg, 3.05 (m, 1H), 2.81 - 2.71 (m, 1H), 2.45 - 2.38 (m, 1H), 2.32 - 2.18 (m, 2H), 2.08 - 1.93 (m, 2H). [0434] Step 2: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2,5,5-d3)methan-d2-ol [0435] To an ice-cooled solution of ((6R,7aS)-6-fluoro-7a-(hydroxymethyl-d2)hexahydro-3H- pyrrolizin-3-one-6-d (380 mg, 2.15 mmol) in 2-methyl-THF (4 mL) under nitrogen atmosphere was added LiAlD4 (4.53 mL, 4.53 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with MeOH (5 mL) in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 13% methanol in dichloromethane (contained 2.5% NH3•H2O) to afford the title compound (Intermediate 31, 200 mg, 56% yield) as a colorless oil. MS: m/z = 165.20 [M + H]+. [0436] Intermediate 32: ((2S,7aR)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-2,5,5-d3)methan-d2- ol
[0437] Intermediate 32 was prepared in a similar manner to Intermediate 31. MS: m/z = 165.20 M + H]+. [0438] Intermediate 33: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene
[0439] To an ice-cooled solution of Intermediate 12 (370.07 mg, 2.30 mmol) in THF (5 mL) was added NaH (55.09 mg, 2.30 mmol, 60% in mineral oil). The mixture was stirred in an ice bath for 0.5 hour before ((S)-5-chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-
WSGR Docket No. 62619-720.601 tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 23 for detail procedures, 500 mg, 1.53 mmol) was added. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (20 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 5% MeOH in DCM to afford the title compound (Intermediate 33, 550 mg, 76% yield) as yellow solid. MS: m/z = 468.20 [M + H]+. [0440] Intermediate 34: 2-(7,8-Difluoro-3-(methoxymethoxy)naphthalen-1-yl-5-d)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
[0441] Intermediate 34 was prepared in a similar manner to Intermediate 17. 1H NMR (400 MHz, DMSO-d6 [0442] Intermediate 35: N-(1,6-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine
[0443] To a stirred mixture of Intermediate 20 (500 mg, 0.79 mmol) in MeCN (62.5 mL) was added Selectfluor (280.40 mg, 0.79 mmol) at room temperature. The resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (10 : 1) to afford
WSGR Docket No. 62619-720.601 the title compound (Intermediate 35, 100 mg, 19% yield) as a light yellow solid. MS: m/z = 650.30 [M + H]+. [0444] Intermediate 36: ((6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
[0445] Step 1: (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- (phenylselanyl)hexahydro-3H-pyrrolizin-3-one [0446] To a solution of Intermediate 8 (5 g, 12.148 mmol) in THF (90 mL) was added LiHMDS (1 M in THF, 15.79 mL, 15.792 mmol) dropwise at -60 °C under nitrogen atmosphere, and the mixture was stirred at -60 °C for 1 h. A solution of diphenyl di-selenide (4.17 g, 13.363 mmol) in THF (10 mL) was added dropwise. The resulting mixture was stirred at -60 °C for 2 hours. Then the mixture was quenched with saturated aq. NH4Cl (300 mL) and extracted with EtOAc (3 × 400 mL). The combined organic phase was washed with brine (500 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-40% EA in PE to give the title compound (5.2 g, 75% yield) as a yellow oil. MS: m/z = 568.25 [M + H]+. 1H NMR (400 MHz, Chloroform-d 15H), 5.53 - 5.04 (m, 1H), 4.47 - 3.79 (m, 3H), 3.54 - 3.26 (m, 2H), 3.16 - 2.44 (m, 2H), 2.19 - 2.06 (m, 1H), 2.00 - 1.69 (m, 1H), 1.06 - 0.95 (m, 9H). 19F NMR (376 MHz, CDCl3 172.70 (d, 1F). [0447] Step 2: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-5,6,7,7a-tetrahydro- 3H-pyrrolizin-3-one [0448] To an ice-cooled solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- (phenylselanyl)hexahydro-3H-pyrrolizin-3-one (5.2 g, 9.177 mmol) in DCM (80 mL) was added H2O2 (20 mL, 30% w/w) dropwise under N2 atmosphere. The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. Then the mixture was diluted with water (200 mL) and extracted with DCM (3 x 200 mL). The combined organic phase was washed brine (300 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-35% EA in PE to give the title compound (3.3 g, 87% yield) as a white solid. MS: m/z = 410.20 [M + H]+. 1H NMR (400
WSGR Docket No. 62619-720.601 MHz, Chloroform-d J = 5.6 Hz, 1H), 6.05 (d, J = 5.6 Hz, 1H), 5.46 - 5.25 (m, 1H), 4.17 - 4.00 (m, 1H), 3.68 - 3.46 (m, 2H), 3.43 - 3.24 (m, 1H), 2.50 - 2.30 (m, 1H), 2.13 - 1.94 (m, 1H), 1.05 (s, 9H). 19F NMR (376 MHz, CDCl3 1F). [0449] Step 3: ((6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-3-oxohexahydro-1H- pyrrolizin-1-yl)boronic acid [0450] To a stirred solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro- 5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one (800 mg, 1.953 mmol) in EtOH (20 mL) were added 2- (Dicyclohexylphosphino)biphenyl (68.46 mg, 0.195 mmol), CuCl (19.34 mg, 0.195 mmol), (dihydroxyboranyl)boronic acid (210.13 mg, 2.344 mmol) and t-BuONa (56.31 mg, 0.586 mmol) at room temperature. The reaction mixture was stirred at room temperature for 2 hours. The resulting mixture was filtered and concentrated under reduced pressure. The residue (combined from 4 batches, 3.56 g crude, total 7.817 mmol, theoretical 100% yield assumed) was purified by RP-Flash aq. FA, Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 10 min; Flow rate: 85 mL/min; Detector: UV 254 & 210 nm. The product-containing fractions were collected and concentrated to give the title compound (3.2 g, 89% yield) as a white solid. MS: m/z = 456.20 [M + H]+. [0451] Step 4: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-3H- pyrrolizin-3-one [0452] To an ice-cooled mixture of ((6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-3- oxohexahydro-1H-pyrrolizin-1-yl)boronic acid (600 mg, 1.317 mmol), AgNO3 (223.81 mg, 1.317 mmol) and Selectfluor (2.80 g, 7.902 mmol) in DCM (9 mL) were added H2O (9 mL), H3PO4 (1.2 mL, 20.634 mmol) and TFA (4.8 mL, 64.623 mmol) under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was heated at 50 °C for 16 hours. The resulting mixture was cooled to room temperature, diluted with water (50 mL), and extracted with EtOAc (3 x 40 mL). The combined organic phase was washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue (combined from 6 batches, 2.945 g crude, total 6.855 mmol, theoretical 100% yield assumed) was purified by silica gel flash chromatography, eluted with 0-40% EA in PE to give the title compound (1.1 g, crude, containing ~30% product in step 2) as a light yellow solid. MS: m/z = 430.20 [M + H]+. [0453] Step 5: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-1H- pyrrolizine
WSGR Docket No. 62619-720.601 [0454] To an ice-cooled stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-1,6- difluorohexahydro-3H-pyrrolizin-3-one (1.1 g, 2.561 mmol) in THF (40 mL) was added BH3-Me2S (1.28 mL, 12.800 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was heated at 60 °C for 16 hours. The resulting mixture was quenched with MeOH (40 mL) and 2 M HCl (40 mL) in an ice bath, and then heated at 60 °C for another 1 hour. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The mixture was diluted with EtOAc (80 mL) and saturated aqueous NaHCO3 (100 mL). After separation, the aqueous phase was extracted with EtOAc (3 x 80 mL). The combined organic layers were washed brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to give the title compound (710 mg, 66% yield) as a yellow oil. MS: m/z = 416.15 [M + H]+. 1H NMR (300 MHz, Chloroform-d J = 54.3 Hz, 1H), 4.93 (dd, J = 53.7, 3.9 Hz, 1H), 3.30 (s, 2H), 3.22 - 2.91 (m, 4H), 2.63 - 2.43 (m, 1H), 2.27 - 1.53 (m, 3H), 1.06 (s, 9H). 19F NMR (282 MHz, CDCl3 180.35 (d, 1F). [0455] Step 6: ((6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0456] To a stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-1,6- difluorohexahydro-1H-pyrrolizine (680 mg, 1.636 mmol) in THF (7 mL) was added pyridine hydrofluoride (347.48 mg, 2.454 mmol, 70% w/w) at room temperature. The reaction mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM to give the title compound (Intermediate 36, 220 mg, 75% yield) as a light-yellow solid. MS: m/z = 178.15 [M + H]+. [0457] Intermediate 37: ((2R)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
[0458] Step 1: (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6-difluorohexahydro-3H- pyrrolizin-3-one [0459] To a stirred solution of Intermediate 8 (2.7 g, 6.56 mmol) in THF (12 mL) under nitrogen atmosphere was added LDA (2 M in THF, 4.92 mL, 9.84 mmol) dropwise at -78 °C. The reaction mixture was stirred at -78 °C for 30 min. To the above mixture was added NFSI (3.10 g, 9.84 mmol) in THF (12 mL) dropwise at -78 °C. The resulting mixture was stirred in an ice bath for 2
WSGR Docket No. 62619-720.601 hours. The resulting mixture was quenched with aq. NH4Cl (50 mL) in an ice bath and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with saturated aq. NaHCO3 (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 1-40% EA in PE to afford the title compound (2.0 g, 70% yield) as a light-yellow oil. MS: m/z = 430.30 [M + H]+. [0460] Step 2: (2R)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,6-difluorohexahydro-1H- pyrrolizine [0461] To an ice-cooled solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6- difluorohexahydro-3H-pyrrolizin-3-one (2 g, 4.65 mmol) in THF (50 mL) under nitrogen atmosphere was added BH3•Me2S (2.33 mL, 23.3 mmol) dropwise. The reaction mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled to room temperature and quenched with MeOH (30 mL) and 4 M HCl aq. (30 mL). The resulting mixture was heated at 60 °C for 1 hour. The resulting mixture was cooled to room temperature and extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (10 : 1) to afford the title compound (1.5 g, 77% yield) as a colorless oil. MS: m/z = 416.25 [M + H]+. [0462] Step 3: ((2R)-2,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0463] To a stirred solution of (2R)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6- difluorohexahydro-1H-pyrrolizine (1.5 g, 3.60 mmol) in THF (30 mL) under nitrogen atmosphere was added TBAF (5.41 mL, 5.41 mmol) dropwise at room temperature. The resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (10 : 1) to afford the title compound (Intermediate 37, 330 mg, 51% yield) as a light yellow oil. MS: m/z = 178.00 [M + H]+. [0464] Intermediate 38 & 39: (S)-5-Chloro-2-(((2R,6S,7as)-2,6-difluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene & (S)-5-chloro-2-(((2R,6R)-2,6-difluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601
[0465] Step 1: (8aS)-5-Chloro-2-(((2R,7aS)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0466] To an ice-cooled solution of Intermediate 37 (182.2 mg, 1.02 mmol) in THF (5 mL) under nitrogen atmosphere was added NaH (51.41 mg, 1.28 mmol, 60% dispersion in mineral oil). The resulting mixture was stirred in an ice bath for 30 min. To the above mixture was added (S)-5- chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene (refer to Intermediate 23 for detail procedures, 280 mg, 0.85 mmol) at 0 °C. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (20 mL) in an ice bath and extracted with CH2Cl2 (3 x 30 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by prep-TLC (CH2Cl2 / MeOH, 15 : 1) to afford the title compound (240 mg, 57% yield) as a light yellow solid. MS: m/z = 484.25 [M + H]+. [0467] Step 2: (S)-5-Chloro-2-(((2R,6S,7as)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene & (S)-5-chloro-2-(((2R,6R)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0468] (8aS)-5-Chloro-2-(((2R,7aS)-2,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (240 mg, 0.49 mmol) was separated by Chiral-HPLC with the following conditions: Column: CHIRALPAK 3-MeOH; Mobile Phase B: EtOH; Flow rate: 20 mL/min; Detector: UV 220 & 254 nm; RT1 (min): 10.136; RT2 (min): 13.282. The first eluting peak (RT1: 10.136 min) was concentrated and lyophilized to give the title compound (Intermediate 38, 48 mg, 20% yield) as an off-white solid. The second eluting peak (RT2: 13.282 min) was concentrated and lyophilized to give the title compound (Intermediate 39, 130 mg, 54% yield) as an off-white solid.
WSGR Docket No. 62619-720.601 [0469] Intermediate 40: 2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)-1-naphthonitrile
[0470] Step 1: 2-(8-Ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0471] To a stirred solution of ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (5 g, 9.75 mmol) in DMF (50 mL) was added CsF (22.23 g, 146.32 mmol) at room temperature. The resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was diluted with EtOAc (300 mL), washed with water (3 x 50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 35% EA in PE to afford the title compound (3 g, 86% yield) as a white solid. 1H NMR (400 MHz, Chloroform-d 1H), 3.50 (s, 3H), 1.44 (s, 12H). 19F NMR (376 MHz, Chloroform-d [0472] Step 2: 2-Fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1- naphthonitrile [0473] To a stirred solution of 2-(8-ethynyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (12 g, 33.68 mmol) in THF (120 mL) under nitrogen atmosphere were added picoline N-oxide (7.35 g, 67.37 mmol) and tert-butyl nitrite (6.95 g, 67.37 mmol) dropwise at room temperature. The resulting mixture was heated at 70 °C for 3 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 13% EA in PE to afford the title compound (Intermediate 40, 7.4 g, 61% yield) as a yellow solid. 1H NMR (400 MHz, Chloroform-d 5.29 (s, 2H), 3.50 (s, 3H), 1.48 (s, 12H). 19F NMR (376 MHz, Chloroform-d [0474] Intermediate 41: 2-(8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane
WSGR Docket No. 62619-720.601
[0475] Step 1: 5-Chloro-6-fluoro-1,4-dihydro-1,4-epoxynaphthalene [0476] To a solution of 1-bromo-3-chloro-2,4-difluorobenzene (10 g, 43.97 mmol) and furan (6.02 g, 88.38 mmol) in toluene (10 mL) was added n-BuLi (2.5 M in n-hexane, 21.00 mL, 52.5 mmol) at -15 °C under nitrogen atmosphere. The resulting mixture was stirred at -15 °C for 30 min. Then the resulting mixture was warmed to room temperature and stirred for another 16 hours. The resulting mixture was quenched with H2O (80 mL). The precipitate was filtered off. The filtrate was extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 6% EA in PE to afford the title compound (3.9 g, 45% yield) as a yellow oil. 1H NMR (400 MHz, Chloroform-d 3H), 6.75 - 6.71 (m, 1H), 5.88 (s, H), 5.74 (d, J = 1.6 Hz, 1H). 19F NMR (376 MHz, Chloroform-d) [0477] Step 2: 8-Chloro-7-fluoronaphthalen-1-ol [0478] To an ice-cooled solution of 5-chloro-6-fluoro-1,4-dihydro-1,4-epoxynaphthalene (3.9 g, 19.83 mmol) in EtOH (41 mL) was added HCl (12 M aq., 20.48 mL, 245.76 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was heated at 80 °C for 5 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was acidified to pH 7 with saturated aqueous NaHCO3 (120 mL) and extracted with EtOAc (3 x 200 mL). The combined organic layers were washed with brine (80 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (3.5 g, 89% yield) as an off-white solid. 1H NMR (400 MHz,
WSGR Docket No. 62619-720.601 Chloroform-d 7.07 (m, 1H). 19F NMR (376 MHz, Chloroform-d [0479] Step 3: 8-Chloro-7-fluoronaphthalen-1-yl pivalate [0480] To a solution of 8-chloro-7-fluoronaphthalen-1-ol (3.5 g, 17.80 mmol) and 4A molecular sieve in DCM (35.53 mL) under nitrogen atmosphere was added DIEA (14.08 g, 108.94 mmol) at room temperature. The resulting mixture was stirred at room temperature for 30 min. Then 2,2- dimethylpropanoyl chloride (3.03 g, 25.10 mmol) was added to the above mixture at -45 °C under nitrogen atmosphere. The resulting mixture was stirred at -45 °C for 1 hour. The resulting mixture was diluted with H2O (80 mL) and extracted with EtOAc (3 x 200 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 46% EA (contained 90% PE) in PE to afford the title compound (3.5 g, 70% yield) as a white solid. 1H NMR (400 MHz, Chloroform-d 7.31 (m, 1H), 7.13 -7.12 (m, 1H), 1.45 (d, J = 1.2 Hz, 9H). 19F NMR (376 MHz, Chloroform-d -110.42 (s, 1F). [0481] Step 4: 8-Chloro-7-fluoro-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-1-yl pivalate [0482] To a solution of 8-chloro-7-fluoronaphthalen-1-yl pivalate (2.5 g, 8.90 mmol) and bis(pinacolato)diboron (2.26 g, 8.90 mmol) in n-hexane (75 mL) were added 4,4'-di-tert-butyl-2,2'- 0.44 mmol) at room temperature under nitrogen atmosphere. The resulting mixture was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure to afford the title compound(5.7 g, crude used through) as a brown solid. [0483] Step 5: 8-Chloro-7-fluoro-3-hydroxynaphthalen-1-yl pivalate [0484] To an ice-cooled solution of 8-chloro-7-fluoro-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-1-yl pivalate (2.1 g, crude) in THF (13 mL) were added H2O2 (4.87 g, 42.96 mmol, 30% aq.) and AcOH (21.08 g, 350.99 mmol) under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was quenched with sat. aq. sodium sulfite (0.5 mL). The resulting mixture was extracted with EtOAc (10 mL x 3). The combined organic layers were washed with brine (2 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 17% EA in PE to afford the title compound (180 mg, 18% yield for two steps) as a yellow solid. 1H NMR (400 MHz, DMSO-d6
WSGR Docket No. 62619-720.601 1H), 7.89 - 7.85 (m, 1H), 7.55 - 7.51 (m, 1H), 7.23 - 7.22 (m, 1H), 6.88 (d, J = 2.0 Hz, 1H), 1.37 (s, 9H). 19F NMR (376 MHz, DMSO-d6 [0485] Step 6: 8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl pivalate [0486] To an ice-cooled solution of 8-chloro-7-fluoro-3-hydroxynaphthalen-1-yl pivalate (1.78 g, 5.99 mmol) in DCM (20 mL) were added DIEA (2.31g, 17.87 mmol) and bromo(methoxy)methane (1.35 g, 10.79 mmol) under nitrogen atmosphere. The resulting mixture was stirred in an ice bath for 1.5 hours. The resulting mixture was diluted with H2O (20 mL) and extracted with EtOAc (3 x 70 mL). The combined organic layers were washed with brine (15 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 37% EA (contained 85% PE) in PE to afford the title compound (1.4 g, 68% yield) as a yellow solid. 1H NMR (400 MHz, DMSO-d6 1H), 7.64 - 7.57 (m, 2H), 7.13 - 7.12 (m, 1H), 5.35 (s, 2H), 3.43 (s, 3H), 1.38 (s, 9H). 19F NMR (376 MHz, DMSO-d6 [0487] Step 7: 8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-ol [0488] To an ice-cooled solution of 8-chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl pivalate (700 mg, 2.05 mmol) in CH3OH (12 mL) was added KOH (460.98 mg, 8.21 mmol) under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was diluted with H2O (30 mL) and extracted with EtOAc (3 x 150 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 37% EA in PE to afford the title compound (370 mg, 70% yield) as a yellow solid. 1H NMR (400 MHz, DMSO-d6 7.71 (m, 1H), 7.47 - 7.41 (m, 1H), 7.00 - 6.99 (m, 1H), 6.74 - 6.73 (m, 1H), 5.25 (s, 2H), 3.41 (s, 3H). [0489] Step 8: 8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl trifluoromethanesulfonate [0490] To a solution of 8-chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-ol (364 mg, 1.41 mmol) in DCM (7.5 mL) was added DIEA (546.24 mg, 4.22 mmol) and Tf2O (604.18 mg, 2.14 mmol) at -40 °C under nitrogen atmosphere. The resulting mixture was stirred at -40 °C for 1.5 hours. The resulting mixture was diluted with H2O (15 mL) and extracted with EtOAc (3 x 80 mL). The combined organic layers were washed with brine (15 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (1.05 g, crude), which was used in the next step without further purification. MS: m/z = 386.95 [M - H]+.
WSGR Docket No. 62619-720.601 [0491] Step 9: 2-(8-Chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane [0492] To a solution of 8-chloro-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl trifluoromethanesulfonate (1.05 g, crude) and AcOK (795.32 mg, 8.10 mmol) in 1,4-dioxane (12 mL) under nitrogen atmosphere were added bis(pinacolato)diboron (1.37g, 5.40 mmol) and Pd(dppf)Cl2•CH2Cl2 (220.05 mg, 0.27 mmol) at room temperature. The resulting mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by prep-TLC (PE : EA = 8 : 1) to afford the title compound (Intermediate 41, 440 mg, 85% yield for two steps) as a yellow oil. MS: m/z = 367.30 [M + H]+. 1H NMR (300 MHz, DMSO-d6 7.34 - 7.33 (m, 1H), 5.34 (s, 2H), 3.42 (s, 3H), 1.37 (s, 12H). [0493] Intermediate 42: N-(6,7-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine
[0494] Step 1: 6,7-Difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diyl bis(trifluoromethanesulfonate) [0495] To a stirred solution of 6,7-difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diol (refer to Intermediate 14 for detail procedures, 1.8 g, 4.781 mmol) in DCM (18 mL) were added DIEA (3.7 g, 28.686 mmol) and Tf2O (4.4 g, 14.343 mmol) at -40 °C under nitrogen atmosphere. The resulting mixture was stirred at -40 °C for 3 hours. The resulting mixture was quenched with saturated aq. NaHCO3 (50 mL), extracted with CH2Cl2 (3 x 100 mL), washed with brine (100 mL), dried over anhydrous Na2SO4, and concentrated under reduced pressure to afford the title compound (3 g, crude used directly) as a brown oil. MS: m/z = 638.95 [M - H]-. [0496] Step 2: 3-((Diphenylmethylene)amino)-6,7-difluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate [0497] To a stirred solution of 6,7-difluoro-8-((triisopropylsilyl)ethynyl)naphthalene-1,3-diyl bis(trifluoromethanesulfonate) (3 g, crude), Pd2(dba)3 (428.83 mg, 0.468 mmol), XantPhos (812.90 mg, 1.405 mmol) and Cs2CO3 (3051.55 mg, 9.366 mmol) in toluene (30 mL) was added diphenylmethanimine (933.59 mg, 5.151 mmol) at room temperature under nitrogen atmosphere. The resulting mixture was heated at 100 °C for 2 hours. The mixture was cooled to room
WSGR Docket No. 62619-720.601 temperature. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (10:1) to afford the title compound (3 g, 93% yield for two steps) as a yellow oil. MS: m/z =672.20 [M + H]+. [0498] Step 3: N-(6,7-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine [0499] To a stirred solution of 3-((diphenylmethylene)amino)-6,7-difluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (2 g, 2.977 mmol) and 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (1.512 g, 5.954 mmol) in 1,4-dioxane (20 mL) under nitrogen atmosphere were added KOAc (876.53 mg, 8.931 mmol) and Pd(dppf)Cl2.CH2Cl2 (12.13 mg, 0.015 mmol) at room temperature. The reaction mixture was heated at 110 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-20% EA in PE to afford the title compound (Intermediate 42, 1.2 g, 62% yield) as a yellow solid. MS: m/z = 650.65 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.70 - 7.68 (m, 2H), 7.59 - 7.55 (m, 1H), 7.51 - 7.48 (m, 2H), 7.31 - 7.29 (m, 4H), 7.19 - 7.17 (m, 2H), 7.09 - 7.08 (m, 1H), 1.27 (s, 12H), 1.13 - 1.07 (m, 21H). [0500] Intermediate 43: Triisopropyl((5,6,8-trifluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)oxy)silane
[0501] Step 1: 2,2-Dimethyl-5-(2-(2,4,5-trifluorophenyl)acetyl)-1,3-dioxane-4,6-dione [0502] To an ice-cooled solution of (2,4,5-trifluorophenyl)acetic acid (10 g, 52.598 mmol), meldrum's acid (7.58 g, 52.59 mmol) and DMAP (28.27 g, 231.43 mmol) in DCM (100 mL) was added DCC (11.94 g, 57.85 mmol) in DCM (10 mL) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was filtered. The filter cake was washed with DCM (3 x 50 mL). The filtrate was washed with 1 M NaHSO4 (3 x 50 mL) and extracted with CH2Cl2 (3 x 50 mL). The combined organic layers were washed with
WSGR Docket No. 62619-720.601 brine (3 x 50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to afford the title compound (65 g, 97% yield) as a light-yellow oil. MS: m/z = 314.90 [M - H]-. [0503] Step 2: tert-Butyl 3-oxo-4-(2,4,5-trifluorophenyl)butanoate [0504] A solution of 2,2-dimethyl-5-(2-(2,4,5-trifluorophenyl)acetyl)-1,3-dioxane-4,6-dione (40 g, 126.48 mmol) in 2-methyl-2-propanol (680 mL) was heated at 88 °C for 5 hours. The reaction mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% EA in PE to afford the title compound (30 g, 82% yield) as a white oil. MS: m/z = 287.10 [M - H]-. [0505] Step 3: 3-Oxo-4-(2,4,5-trifluorophenyl)butanoic acid [0506] To a solution of tert-butyl 3-oxo-4-(2,4,5-trifluorophenyl)butanoate (30 g, 104.07 mmol) in DCM (480 mL) was added TFA (300.0 mL) at room temperature under argon atmosphere. The reaction mixture was stirred at room temperature for 4 hours. The resulting mixture was co- evaporated with toluene (500 mL) three times to give the title compound (20 g, 82% yield) as a yellow solid. MS: m/z = 231.00 [M - H]-. [0507] Step 4: 5,7,8-Trifluoronaphthalene-1,3-diol [0508] 3-Oxo-4-(2,4,5-trifluorophenyl)butanoic acid (10 g, 43.3 mmol) was dissolved in CF3SO3H (300 mL, 10.57 mmol) at room temperature, and the mixture was stirred at this temperature for 16 hours under nitrogen atmosphere. The resulting mixture was diluted with EtOAc (800 mL), washed with water (4 x 300 mL) and brine (400 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0 - 50% EA in PE to afford the title compound (1.4 g, 15%) as a white solid. MS: m/z = 212.95 [M - H]-. 1H NMR (400 MHz, DMSO-d6 1H), 6.65 (s, 2H). [0509] Step 5: 5,7,8-Trifluoro-3-((triisopropylsilyl)oxy)naphthalen-1-ol [0510] To an ice-cooled solution of 5,7,8-trifluoronaphthalene-1,3-diol (742 mg, 3.46 mmol) in CH2Cl2 (8 mL) were added DIEA (1.34 g, 10.39 mmol) and chlorotriisopropylsilane (734.85 mg, 3.81 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with water/ice (7 mL). The resulting mixture was diluted with CH2Cl2 (40 mL), washed with water/ice (3 x 20 mL), washed with brine (5 x 20 mL), dried over anhydrous Na2SO4. filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (642 mg, 50% yield) as a yellow oil. MS: m/z = 369.15 [M - H]-. [0511] Step 6: 5,7,8-Trifluoro-3-((triisopropylsilyl)oxy)naphthalen-1-yl trifluoromethanesulfonate
WSGR Docket No. 62619-720.601 [0512] To an ice-cooled solution of 5,7,8-trifluoro-3-[(triisopropylsilyl)oxy]naphthalen-1-ol (640 mg, 1.72 mmol) in DCM (7 mL) were added DIEA (334.90 mg, 2.59 mmol) and Tf2O (1949.47 mg, 6.91 mmol) dropwise. The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was diluted with CH2Cl2 (40 mL), washed with brine (5 x 10 mL), dried over anhydrous Na2SO4. filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% EA in PE to afford the title compound (750 mg, 86% yield) as a yellow oil. MS: m/z = 500.95 [M - H]-. [0513] Step 7: Triisopropyl((5,6,8-trifluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)oxy)silane [0514] To a stirred mixture of 5,7,8-trifluoro-3-((triisopropylsilyl)oxy)naphthalen-1-yl trifluoromethanesulfonate (750 mg, 1.49 mmol), bis(pinacolato)diboron (757.97 mg, 2.98 mmol) and Pd(dppf)Cl2.DCM (121.58 mg, 0.14 mmol) in 1,4-dioxane (8 mL) was added KOAc (439.41 mg, 4.47 mmol) at room temperature under argon atmosphere. The resulting mixture was heated at 110 °C and stirred for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-5% EA in PE to afford the title compound (Intermediate 43, 340 mg, 47% yield) as a yellow solid. MS: m/z = 481.50 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.34 (s, 2H), 1.36 (s, 12H), 1.35 - 1.34 (m, 3 H), 1.09 (d, J = 8.0 Hz, 18H). [0515] Intermediate 44: ((5R,6aR)-5-Fluorohexahydrocyclopropa[a]pyrrolizin-6a(4H)-yl)methanol
[0516] Step 1: (5R,6aR)-6a-(((tert-Butyldiphenylsilyl)oxy)methyl)-5- fluorohexahydrocyclopropa[a]pyrrolizin-2(1H)-one [0517] To an ice-cooled stirred solution of trimethyl(oxo)-lambda6-sulfanylium iodide (322.39 mg, 1.46 mmol) in DMSO (0.4 mL) was added NaH (48.83 mg, 1.22 mmol, 60% dispersion in mineral oil) under nitrogen atmosphere. The mixture was stirred at room temperature for 30 min and then heated at 50 °C for 1 hour. Then a solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)- 6-fluoro-5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one (refer to Intermediate 36 for detail procedures, 100 mg, 0.24 mmol) in DMSO (0.4 mL) was added. The reaction mixture was heated at 50 °C for 1.5 hours. The resulting mixture was cooled, quenched with H2O (50 mL) in an ice bath, and extracted with EtOAc (3 x 100 mL). The combined organic layers were dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 30% PE in EA to afford the title compound (17.5 mg,
WSGR Docket No. 62619-720.601 16% yield) as a yellow solid. MS: m/z = 424.25 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 7.66 - 7.62 (m, 4H), 7.48 - 7.37 (m, 6H), 5.30 - 5.08 (m, 1H), 3.88 - 3.76 (m, 1H), 3.69 - 3.53 (m, 2H), 3.09 - 2.91 (m, 1H), 2.36 - 2.18 (m, 1H), 2.20 - 1.90 (m, 2H), 1.67 - 1.58 (m, 1H), 1.06 (s, 9H), 1.05 - 0.94 (m, 1H), 0.84 - 0.79 (m, 1H). [0518] Step 2: (5R,6aR)-6a-(((tert-Butyldiphenylsilyl)oxy)methyl)-5- fluorooctahydrocyclopropa[a]pyrrolizine [0519] To an ice-cooled stirred solution of (5R,6aR)-6a-(((tert-butyldiphenylsilyl)oxy)methyl)-5- fluorohexahydrocyclopropa[a]pyrrolizin-2(1H)-one (350 mg, 0.82 mmol) in THF (7 mL) was added BH3•Me2S (0.413 mL, 4.13 mmol, 10M in DMS) dropwise under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled and quenched with MeOH (7 mL) and 2 M HCl aq. (7 mL) in an ice bath. The resulting mixture was heated at 60 °C for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was diluted with EtOAc (80 mL) and washed with saturated aqueous NaHCO3 (100 mL). The aqueous phase was extracted with EtOAc (3 x 80 mL). The combined organic layers were washed brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to give the title compound (230 mg, 67% yield) as a yellow oil. 1H NMR (400 MHz, Methanol-d4) 7.72 - 7.68 (m, 4H), 7.44 - 7.37 (m, 6H), 5.26 - 5.17 (m, 1H), 3.50 - 3.38 (m, 2H), 3.29 - 3.23 (m, 2H), 2.84 - 2.72 (m, 2H), 2.35 - 2.25 (m, 1H), 2.05 - 1.85 (m, 1H), 1.70 - 1.64 (m, 1H), 1.51 - 1.49 (m, 1H), 1.07 (s, 9H), 0.68 - 0.66 (m, 1H), 0.41 - 0.38 (m, 1H). [0520] Step 3: ((5R,6aR)-5-Fluorohexahydrocyclopropa[a]pyrrolizin-6a(4H)-yl)methanol [0521] To a stirred solution of (5R,6aR)-6a-(((tert-butyldiphenylsilyl)oxy)methyl)-5- fluorooctahydrocyclopropa[a]pyrrolizine (230 mg, 0.56 mmol) in THF (3.5 mL) was added TBAF (1 M in THF, 0.84 mL, 0.84 mmol) at room temperature. The resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM to give the title compound (Intermediate 44, 70 mg, 72% yield) as a light-yellow solid. MS: m/z = 172.10 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 5.24 - 5.07 (m, 1H), 4.40 - 4.30 (m, 1H), 3.24 - 3.08 (m, 4H), 2.77 - 2.68 (m, 2H), 2.30 - 2.21 (m, 1H), 1.85 - 1.74 (m, 1H), 1.62 - 1.45 (m, 2H), 0.64 - 0.58 (m, 1H), 0.25 - 0.22 (m, 1H). [0522] Intermediate 45: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-3,3,5,5-d4)methanol
WSGR Docket No. 62619-720.601
[0523] Step 1: (2R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine-3,3-d2 [0524] To a stirred solution of Intermediate 10 (360 mg, 2.23 mmol) and imidazole (304.04 mg, 4.46 mmol) in DMF (3.6 mL) was added TBDPSCl (736.52 mg, 2.68 mmol) at room temperature. The resulting mixture was stirred at room temperature for 4 hours. The resulting mixture was diluted with EtOAc (50 mL) and washed with brine (5 x 30 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-55% EA in PE to afford the title compound (770 mg, 86% yield) as a light-yellow solid. MS: m/z = 400.30 [M + H]+. 1H NMR (400 MHz, Chloroform-d 3.10 (m, 3H), 2.95 - 2.83 (m, 1H), 2.15 - 1.94 (m, 2H), 1.91 - 1.56 (m, 4H), 1.06 (s, 9H). 19F NMR (376 MHz, Chloroform-d [0525] Step 2: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-5,5-d2 [0526] To an ice-cooled mixture of (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2- fluorohexahydro-1H-pyrrolizine-3,3-d2 (750 mg, 1.87 mmol) and RuCl3•H2O (211.55 mg, 0.94 mmol) in CCl4 (7.5 mL) and H2O (7.5 mL) were added NaIO4 (2007.12 mg, 9.38 mmol) in portions under nitrogen atmosphere. The resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was diluted with EtOAc (50 mL), filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-55% EA in PE to afford the title compound (600 mg, a mixture, 77% yield) as a light-yellow solid. MS: m/z = 414.15 [M + H]+. [0527] Step 3: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-5,5-d2 [0528] The mixture was separated by prep-Achiral-HPLC with the following conditions: Column: 2; Mobile Phase B: MeOH(0.1% 2 M NH3
WSGR Docket No. 62619-720.601 Back Pressure(bar): 100; Detector: UV 220 nm; RT1 (min): 5.17; RT2 (min): 7.97. The first eluting peak (RT1: 5.17 min) was concentrated and lyophilized to give (6R,7aS)-7a-(((tert- butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H-pyrrolizin-3-one-5,5-d2 (550 mg, 96% yield) as an off-white solid. MS: m/z = 414.15 [M + H]+. 1H NMR (400 MHz, Chloroform-d 7.68 - 7.58 (m, 4H), 7.48 - 7.33 (m, 6H), 5.24 (dd, J = 52.8, 4.8 Hz, 1H), 3.68 - 3.39 (m, 2H), 2.90 - 2.76 (m, 1H), 2.41 - 1.90 (m, 5H), 1.05 (s, 9H). 19F NMR (376 MHz, Chloroform-d 171.35 (m, 1F). The second eluting peak (RT1: 7.97 min) was concentrated and lyophilized to give (2R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-3H-pyrrolizin-3-one (20 mg, 3% yield) as an off-white solid. MS: m/z = 412.10 [M + H]+. [0529] Step 4: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)hexahydro-3H-pyrrolizin-3-one-5,5-d2 [0530] To a stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-5,5-d2 (550 mg, 1.33 mmol) in THF (5.5 mL) was added TBAF (1 M in THF, 1.46 mL, 1.463 mmol) dropwise at room temperature. The mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-5% MeOH in CH2Cl2 to afford the title compound (220 mg, 94% yield) as a light yellow solid. 1H NMR (400 MHz, DMSO-d6 1H), 2.20 - 2.09 (m, 3H), 1.92 - 1.82 (m, 2H). 19F NMR (376 MHz, DMSO-d6 [0531] Step 5: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-3,3,5,5-d4)methanol [0532] To an iced-cooled stirred solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)hexahydro-3H- pyrrolizin-3-one-5,5-d2 (210 mg, 1.20 mmol) in 2-methyl-THF (4.2 mL) was added LiAlD4 (1 M in THF, 2.52 mL, 2.52 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was heated at 90 °C for 2.5 hours. The resulting mixture was cooled and quenched with water (0.11 mL), 10% aq. NaOH (0.11 mL) and water (0.33 mL) in an ice bath. The resulting mixture was stirred in an ice bath for 15 min, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM (contained 2% NH3.H2O) to give the title compound (Intermediate 45, 120 mg, 61% yield) as a light-yellow solid. 1H NMR (400 MHz, DMSO-d6 3.02 (m, 2H), 2.07 - 1.61 (m, 6H). [0533] Intermediate 46: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)tetrahydro-1H-pyrrolizin-2(3H)-one
WSGR Docket No. 62619-720.601
[0534] Step 1: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-hydroxyhexahydro- 3H-pyrrolizin-3-one [0535] To a stirred solution of Intermediate 8 (5 g, 12.148 mmol) in THF (50 mL) was added LDA (9.11 mL, 18.222 mmol) dropwise at -40 °C under nitrogen atmosphere. The resulting mixture was stirred at -40 °C for 0.5 hours. To the above mixture was added (4aR,7R,8aR)-9,9- dimethyltetrahydro-4H-4a,7-methanobenzo[c][1,2]oxazireno[2,3-b]isothiazole 3,3-dioxide (4.18 g, 18.222 mmol) over 5 min at -40 °C. The resulting mixture was stirred at -40 °C for an additional 1 hour. The reaction was quenched with sat. aq. NH4Cl (8 mL) in an ice bath and extracted with EtOAc (3 x 40 mL). The combined organic layers were washed with brine (3 x 30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (1:1) to afford the title compound (3.8 g, 73% yield) as an off-white semi-solid. MS: m/z = 428.20 [M + H]+. [0536] Step 2: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H- pyrrolizin-2-ol [0537] To an ice-cooled stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-hydroxyhexahydro-3H-pyrrolizin-3-one (100 mg, 0.234 mmol) in THF (10 mL) was added BH3-Me2S (0.12 mL, 1.170 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled and quenched with MeOH in an ice bath. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (1:1) to afford the title compound (66 mg, 68% yield) as a colorless oil. MS: m/z = 414.25 [M + H]+. [0538] Step 3: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorotetrahydro-1H- pyrrolizin-2(3H)-one [0539] To an ice-cooled solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-ol (500 mg, 1.209 mmol) and TEA (366.99 mg, 3.627 mmol) in DCM (5 mL) and DMSO (5 mL) was added sulfur trioxide pyridine complex (577.21 mg, 3.627 mmol). The ice bath was removed, and the resulting mixture was stirred at room temperature for 3
WSGR Docket No. 62619-720.601 hours. The resulting mixture was quenched with saturated aq. NaCl (10 mL) and aq.1 M K2CO3 (10 mL), and extracted with EtOAc (3 x 20 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (1:1) to afford the title compound (250 mg, 50% yield) as a white solid. MS: m/z = 412.20 [M + H]+. [0540] Step 4: (6R,7aS)-6-fluoro-7a-(hydroxymethyl)tetrahydro-1H-pyrrolizin-2(3H)-one [0541] To an ice-cooled stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorotetrahydro-1H-pyrrolizin-2(3H)-one (200 mg, 0.486 mmol) in THF (2 mL) was added TBAF (0.73 mL, 0.729 mmol) dropwise under nitrogen atmosphere. The resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (15:1) to afford the title compound (Intermediate 46, 80 mg, 95% yield) as a light yellow solid. MS: m/z = 174.10 [M + H]+. [0542] Intermediate 47: N-(6,8-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine
[0543] Intermediate 47 was prepared in a similar manner to Intermediate 42. MS: m/z = 650.40 [M + H]+. [0544] Intermediate 48: ((2R,7aS)-2-Fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol
[0545] Step 1: (2R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methylenehexahydro-1H-pyrrolizine [0546] To an ice-cooled solution of methyltriphenylphosphanium bromide (1.51 g, 4.227 mmol) in THF (6 mL) were added t-BuOK (3.95 mL, 3.945 mmol, 1M in THF) dropwise under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. Then a solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorotetrahydro-1H-pyrrolizin-2(3H)-one (refer to Intermediate 46 for detail procedures, 580 mg, 1.409 mmol) in THF (10 mL) was added dropwise to the above mixture in an ice bath. The ice
WSGR Docket No. 62619-720.601 bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The reaction was quenched with sat. NH4Cl (aq.) (50 mL) in an ice bath and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-25% EA in PE to afford the title compound (298 mg, 51% yield) as an off-white oil. MS: m/z = 410.20 [M + H]+. [0547] Step 2: ((2R,7aS)-2-Fluoro-6-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0548] To a solution of (2R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methylenehexahydro-1H-pyrrolizine (298 mg, 0.728 mmol) in THF (3 mL) was added TBAF (1.09 mL, 1.092 mmol, 1M in THF) at room temperature. The mixture was stirred at room temperature for 1 hour. The solvent was removed, and the residue was purified by silica gel flash chromatography, eluted with 5% methanol in dichloromethane to give the title compound (Intermediate 48, 110 mg, 88% yield) as an off-white oil. MS: m/z =172.15 [M + H]+. [0549] Intermediate 49: (2-Bromoethyl)diphenylsulfonium trifluoromethanesulfonate
[0550] Step 1: 2-Bromoethyl trifluoromethanesulfonate [0551] To a stirred solution of trifluoromethanesulfonic anhydride (1.88 mL, 11.17 mmol) in DCM (5 mL) was added pyridine (0.9 mL) in portions at - 20 °C. The resulting mixture was stirred at - 20 °C for 10 min. 2-Bromoethanol (0.71 mL, 10.02 mmol) was added to the mixture. The resulting mixture was stirred at -20 °C for 15 min. The precipitate was filtered and washed with Et2 mL). The precipitate was filtered and washed with Et2 concentrated under reduced pressure to give the title compound (2.36 g, crude), which was used in the next step without further purification. [0552] Step 2: (2-Bromoethyl)diphenylsulfonium trifluoromethanesulfonate [0553] trifluoromethanesulfonate (2.36 g, crude) in toluene (9 mL) at room temperature. The resulting mixture was heated at 100 °C for 7 hours. The resulting mixture was cooled to room temperature, and Et2O (20 mL) was added. The resulting mixture was stirred at room temperature for 16 hours. The precipitate was filtered, washed with Et2O (3 mL), and dried under reduced pressure to afford the title compound (Intermediate 49, 2 g, 45% yield for two steps). 1H NMR (400 MHz, Chloroform-d
WSGR Docket No. 62619-720.601 [0554] Intermediate 50: ((6'R,7a'S)-6'-Fluorodihydro-1'H,3'H-spiro[cyclopropane-1,2'-pyrrolizin]- 7a'(5'H)-yl)methanol
[0555] Step 1: (6'R,7a'S)-7a'-(((tert-Butyldiphenylsilyl)oxy)methyl)-6'-fluorotetrahydro-1'H,3'H- spiro[cyclopropane-1,2'-pyrrolizin]-3'-one [0556] To a stirred solution of Intermediate 8 (300 mg, 0.72 mmol) in THF (3 mL) was added LiHMDS (1.82 mL, 1.82 mmol, 1 M in THF) at -30 °C under nitrogen atmosphere. The resulting mixture was stirred at -30 °C for 1 hour. Then a solution of Intermediate 49 (387.72 mg, 0.87 mmol) in THF (1.5 mL) was added. The resulting mixture was at -30 °C for 2 hours, warmed to room temperature, and stirred for 16 hours at room temperature. The mixture was quenched with sat. aq. NH4Cl (10 mL) and extracted with EtOAc (3 x 15 mL). The combined organic phase was washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue (combined from three batches, total 853 mg, 1.949 mmol, theoretical 100% yield assumed) was purified by prep-TLC (PE / EA = 3 / 1) to give a crude product. The crude was purified by RP-Flash with the following conditions: Column: C18, 40 g, 4HCO3; Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 10 min; Flow rate: 35 mL/min; Detector: UV 210 nm. The product-containing fractions were collected and evaporated in vacuo and then lyophilized overnight to give the title compound (170 mg, 19% yield) as a yellow oil. MS: m/z = 438.25 [M + H]+. 1H NMR (400 MHz, Chloroform-d 1H), 4.20 - 4.04 (m, 1H), 3.66 - 3.47 (m, 2H), 3.34 - 3.19 (m, 1H), 2.56 - 2.40 (m, 1H), 2.29 - 2.26 (m, 1H), 2.09 - 1.94 (m, 1H), 1.87 - 1.84 (m, 1H), 1.28 - 1.25 (m, 1H), 1.05 (s, 9H), 0.98 - 0.89 (m, 1H), 0.65 - 0.55 (m, 2H). 19F NMR (376 MHz, Chloroform-d -171.90 (s, 1F). [0557] Step 2: (6'R,7a'S)-7a'-(((tert-Butyldiphenylsilyl)oxy)methyl)-6'-fluorotetrahydro-1'H,3'H- spiro[cyclopropane-1,2'-pyrrolizine] [0558] To an ice-cooled stirred solution of (6'R,7a'S)-7a'-(((tert-butyldiphenylsilyl)oxy)methyl)-6'- fluorotetrahydro-1'H,3'H-spiro[cyclopropane-1,2'-pyrrolizin]-3'-one (170 mg, 0.38 mmol) in THF (5 mL) was added BH3•Me2S (0.19 mL, 1.94 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was heated at 60 °C for 16 hours. The reaction mixture was cooled to room temperature and quenched with MeOH (5 mL) and 2 M HCl aq. (5 mL) at room temperature. The resulting mixture was heated at 60 °C for 1 hour. The mixture was
WSGR Docket No. 62619-720.601 cooled to room temperature and concentrated under reduced pressure. The mixture was diluted with EtOAc (30 mL) and saturated aqueous NaHCO3 (20 mL). After separation, the aqueous phase was extracted with EtOAc (3 x 20 mL). The combined organic layers were washed brine (30 mL), dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified silica gel flash chromatography, eluted with 0-35% EA in PE to give the title compound (116 mg, 70% yield) as a yellow oil. MS: m/z = 424.20 [M + H]+. [0559] Step 3: ((6'R,7a'S)-6'-Fluorodihydro-1'H,3'H-spiro[cyclopropane-1,2'-pyrrolizin]-7a'(5'H)- yl)methanol [0560] To a stirred solution of (6'R,7'aS)-7'a-{[(tert-butyldiphenylsilyl)oxy]methyl}-6'-fluoro- tetrahydro-1'H-spiro[cyclopropane-1,2'-pyrrolizine] (116 mg, 0.27 mmol) in THF (1.5 mL) was added TBAF (1 M solution in THF, 0.41 mL, 0.41 mmol) at room temperature. The resulting mixture was stirred at room temperature for 5 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by prep-TLC (DCM / MeOH = 10 / 1) to give the title compound (Intermediate 50, 36 mg, 70% yield) as a yellow oil. MS: m/z = 186.10 [M + H]+. [0561] Intermediate 51 & 52: (2S,6R,7aS)-7a-((((S)-5-Chloro-4-fluoro-8a,9,10,11,12,13- hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-ol & (2R,6R,7aS)-7a-((((S)-5-chloro-4-fluoro-8a,9,10,11,12,13- hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-ol
[0562] Step 1: (6R,7aS)-6-Fluoro-7a-(hydroxymethyl)-hexahydropyrrolizin-2-ol [0563] To an ice-cooled stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-ol (refer to Intermediate 46 for detail procedures, 1 g, 2.42 mmol) in THF (10 mL) was added TBAF (3.63 mL, 3.63 mmol) dropwise under nitrogen atmosphere. The iced bath was removed, and the reaction mixture was stirred at room temperature for 1.5 hours. The resulting mixture was concentrated and purified by silica gel flash chromatography, eluted with
WSGR Docket No. 62619-720.601 0-20% MeOH in CH2Cl2 (contained 2% NH3•H2O) to afford the title compound (135 mg, 31% yield) as a colorless oil. MS: m/z = 176.15 [M + H]+. [0564] Step 2: (6R,7aS)-7a-((((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol [0565] To a stirred solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-hexahydropyrrolizin-2-ol (135 mg, 0.77 mmol) and Cs2CO3 (131.53 mg, 0.40 mmol) in 1,4-dioxane (5 mL) under argon atmosphere was added (S)-2,5-dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 5 for detail procedures, 211.55 mg, 0.62 mmol) at room temperature. The resulting mixture was heated at 80 °C for 16 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in CH2Cl2 to afford the title compound (230 mg, 61% yield) as a light-yellow solid. MS: m/z = 482.20 [M + H]+. [0566] Step 3: (2S,6R,7aS)-7a-((((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol & (2R,6R,7aS)-7a-((((S)-5-chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol [0567] (6R,7aS)-7a-((((S)-5-Chloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol (230 mg) was separated by Prep-Chiral-HPLC with following conditions: Column: CHIRALPAK IA, 2 * 25 3-MeOH), Mobile Phase B: EtOH; Flow rate: 20 mL/min; Gradient: isocratic 10; RT1: 7.089 min; RT2: 8.309 min. The first eluting peak (RT1: 7.089) was collected and concentrated to give the title compound (Intermediate 51, 30 mg, 13% yield) as light-yellow lyophilized powder. The second eluting peak (RT2: 8.309 min) was collected and concentrated to give the title compound (Intermediate 52, 150 mg, 65% yield) as light-yellow lyophilized powder. MS: m/z = 482.20 [M + H]+. [0568] Intermediate 53: ((2R,6S,7R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,7- d2)methanol
[0569] Step 1: (1R,2S,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-3H- pyrrolizin-3-one-1,2-d2 [0570] To a mixture of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-5,6,7,7a- tetrahydro-3H-pyrrolizin-3-one (refer to Intermediate 36 for detail procedures, 500 mg, 1.22 mmol)
WSGR Docket No. 62619-720.601 in D2O (5 mL) and DCM (20 mL) was added Pd/C (493.68 mg, 0.46 mmol, 10% wt) under nitrogen atmosphere. The reaction mixture was flushing with deuterium gas. The resulting mixture was stirred at room temperature for 3 hours under an atmosphere of deuterium gas. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 10 % EA in PE to afford the title compound (cis isomer) (430 mg, 85% yield) as a white solid. MS: m/z = 414.20 [M + H]+. [0571] Step 2: (2R,6S,7R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluorohexahydro-1H- pyrrolizine-6,7-d2 [0572] To an ice-cooled mixture of (1R,2S,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-3H-pyrrolizin-3-one-1,2-d2 (cis isomer) (430 mg, 1.04 mmol) in THF (3 mL) was added BH3•Me2S (0.52 mL, 5.20 mmol) under nitrogen atmosphere. The ice bath was removed, and the resulting mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled to room temperature, quenched with MeOH (10 mL) and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 16% EA in PE to afford the title compound (cis isomer) (390 mg, 93% yield) as a yellow solid. MS: m/z = 400.10 [M + H]+. [0573] Step 3: ((2R,6S,7R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,7-d2)methanol [0574] To a solution of (2R,6S,7R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2- fluorohexahydro-1H-pyrrolizine-6,7-d2 (cis isomer) (390 mg, 0.97 mmol) in MeOH (4 mL) under nitrogen atmosphere was added NH4F (1445.81 mg, 39.04 mmol) at room temperature. The resulting mixture was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 15% MeOH in DCM to afford the title compound (cis isomer) (Intermediate 53, 100 mg, 63% yield) as a yellow oil. MS: m/z = 162.15 [M + H]+. [0575] Intermediate 54 & 55: (8S,8aS)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13- hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene & (8R,8aS)-5-chloro-2,4- difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene
WSGR Docket No. 62619-720.601 [0576] Step 1: tert-Butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate [0577] To an ice-cooled stirred solution of (2S)-1-(tert-butoxycarbonyl)azepane-2-carboxylic acid (2 g, 8.220 mmol) and N,O-dimethylhydroxylaminehydrochloride (1.20 g, 12.330 mmol) in DCM (20 mL) under nitrogen atmosphere were added DIEA (4.25 g, 32.880 mmo) and HATU (4.69 g, 12.330 mmol). The ice bath was removed, and the resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was diluted with water (30 mL) and extracted with DCM (3 x 50 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA/PE (1/2) to afford the title compound (2.2 g, 93% yield) as a yellow oil. MS: m/z = 287.15 [M + H]+. [0578] Step 2: tert-Butyl (2S)-2-acetylazepane-1-carboxylate [0579] To a stirred solution of tert-butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate (2.2 g, 7.682 mmol) in THF (20 mL) was added bromo(methyl) magnesium (11.5 mL, 1.362 mmol) dropwise at -78 °C under nitrogen atmosphere. The resulting mixture was warmed to room temperature slowly and stirred for 3 hours. The reaction was quenched with NH4Cl (sat) (30 mL) in an ice bath and extracted with EA (3 x 50 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA/PE (1/2) to afford the title compound (1.7 g, 92% yield) as a yellow oil. 1H NMR (400 MHz, Chloroform-d 1H), 3.10 - 2.76 (m, 1H), 2.25 - 2.21 (m, 1H), 2.20 - 2.08 (m, 3H), 1.99 - 1.66 (m, 3H), 1.65 - 1.50 (m, 2H), 1.43 (s, 9H), 1.39 - 1.21 (m, 2H). [0580] Step 3: tert-Butyl (2S)-2-(1-hydroxyethyl)azepane-1-carboxylate [0581] To an ice-cooled stirred solution of tert-butyl (2S)-2-acetylazepane-1-carboxylate (2 g, 8.287 mmol) in THF (10 mL) was added BH3.THF (12 mL, 12.000 mmol, 1 M in DMS). The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The reaction was quenched with water (10 mL) in an ice bath and extracted with EtOAc (3 x 15 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA/PE (1/1) to afford the title compound (1.9 g, 94% yield) as a yellow oil. MS: m/z = 244.15 [M + H]+. 1H NMR (400 MHz, DMSO-d6 2.93 - 2.82 (m, 1H), 2.10 - 1.82 (m, 3H), 1.80 - 1.68 (m, 2H), 1.45 - 1.25 (m, 11H), 1.10 - 0.93 (m, 4H). [0582] Step 4: (S)-1-(Azepan-2-yl)ethan-1-ol
WSGR Docket No. 62619-720.601 [0583] To an ice-cooled stirred solution of tert-butyl (2S)-2-(1-hydroxyethyl)azepane-1- carboxylate (1.9 g, 7.808 mmol) in DCM (20 mL) was added HCl (20 mL, 4 M in 1,4-dioxane) dropwise. The ice bath was removed, and the reaction mixture was stirred at room temperature for 2 hours. The mixture was concentrated under reduced pressure. The residue was dissolved in methanol (10 mL), and the mixture was then basified to pH 7 with Amberlyst-A-21 resin. The precipitate was filtered off and washed with methanol (3 x 20 mL). The combined filtrate was concentrated under reduced pressure to give the title compound (1.1 g, 98% yield) as a light yellow solid. MS: m/z = 144.15 [M+ H]+. [0584] Step 5: (S)-5-(1-(Azepan-2-yl)ethoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin-4-ol [0585] To an ice-cooled solution of (S)-1-(azepan-2-yl)ethan-1-ol (1.17 g, 8.195 mmol) in THF (40 mL) under nitrogen atmosphere was added NaH (1.19 g, 29.800 mmol, 60% dispersion in mineral oil). The mixture was stirred in an ice bath for 0.5 hours. Intermediate 1 (2 g, 7.450 mmol) was added to the above mixture, and the reaction mixture was stirred at room temperature for 2.5 hours. The resulting mixture was quenched with saturated aq. NH4Cl (150 mL) and extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (80 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 8% MeOH in DCM to afford the title compound (1.7 g, 60% yield) as a yellow solid. MS: m/z = 375.05, 377.05 [M + H]+. [0586] Step 6: (S)-2,5-Dichloro-4-fluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0587] To an ice-cooled solution of (S)-5-(1-(azepan-2-yl)ethoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4-ol (1.7 g, 4.531 mmol) and DIEA (3.51 g, 27.186 mmol) in DCM (340 mL) under nitrogen atmosphere was added POCl3 (3.47 g, 22.655 mmol) dropwise. The reaction mixture was stirred in an ice bath for 2 hours. The resulting mixture was quenched with sat. aq. NaHCO3 (100 mL) and extracted with DCM (3 x 80 mL). The combined organic layers were washed with brine (60 mL), dried over anhydrous sodium, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 3% EA in DCM to afford the title compound (700 mg, 43% yield) as a yellow solid. MS: m/z = 357.05 [M + H]+. [0588] Step 7: (S)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0589] To a stirred solution of (S)-2,5-dichloro-4-fluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (1.1 g, 3.079 mmol) in DMSO (20 mL) was added KF (0.30 g, 5.234 mmol) at room temperature under nitrogen atmosphere. The mixture was
WSGR Docket No. 62619-720.601 heated at 80 °C for 16 hours and cooled to room temperature. The resulting mixture was diluted with EtOAc (200 mL) and washed with brine (3 x 150 mL). The organic layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 4% EA in DCM to afford the title compound (850 mg, 81% yield) as a yellow solid. MS: m/z = 341.05 [M + H]+. 1H NMR (400 MHz, Chloroform-d 5.21 - 5.03 (m, 1H), 4.79 - 4.58 (m, 1H), 3.80 - 3.75 (m, 1H), 3.14 - 2.96 (m, 1H), 2.18 - 1.90 (m, 2H), 1.85 - 1.31 (m, 9H). [0590] Step 8: (8S,8aS)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene & (8R,8aS)-5-chloro-2,4-difluoro-8-methyl- 8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0591] (S)-5-Chloro-2,4-difluoro-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (850 mg, 2.494 mmol) was separated by Prep-SFC-HPLC with the following conditions: Mobile Phase A: CO2, Mobile Phase B: MEOH (0.1% 2M NH3-MEOH); Pressure(bar): 100; Wave Length: 220 nm; Sample Solvent: MeOH. The first eluting peak (RT1: 1.731 min) was concentrated and lyophilized to give the title compound (Intermediate 54, 280 mg, 32% yield) as a yellow lyophilized powder. MS: m/z = 341.05 [M + H]+. The second eluting peak (RT2: 1.995 min) was concentrated and lyophilized to give the title compound (Intermediate 55, 380 mg, 44% yield) as a yellow lyophilized powder. MS: m/z = 341.05 [M + H]+. [0592] Intermediate 56: ((2R,6S,7aS)-2-Fluoro-6-methyltetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol
[0593] Step 1: (2S,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methylhexahydro- 3H-pyrrolizin-3-one & (2R,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-3H-pyrrolizin-3-one [0594] To an ice-cooled stirred solution of Intermediate 8 (1 g, 2.43 mmol) in THF (20 mL) under nitrogen atmosphere was added LiHMDS (2.91 mL, 2.91 mmol, 1M in THF) dropwise. The mixture was stirred in an ice bath for 1 hour. Methyl iodide (0.38 g, 2.67 mmol) in THF (2 mL)
WSGR Docket No. 62619-720.601 was added to the above solution in an ice bath. The resulting mixture was stirred in an ice bath for 2 hours. The mixture was quenched with saturated aq. NH4Cl (50 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (1 / 3) to afford two peaks. The first eluting peak was concentrated under reduced pressure to afford the title compound (550 mg, 53% yield) as a yellow solid. MS: m/z = 426.20 [M + H]+. 1H NMR (300 MHz, Chloroform-d) 7.65 -7.60 (m, 4H), 7.48 - 7.37 (m, 6H), 5.35 - 5.17 (m, 1H), 4.23 - 4.11 (m, 1H), 3.60 - 3.41 (m, 2H), 3.20 - 3.04 (m, 1H), 2.83 - 2.76 (m, 1H), 2.38 - 2.19 (m, 2H), 2.04 - 1.89 (m, 1H), 1.70 - 1.63 (m, 1H), 1.18 - 1.12 (m, 3H), 1.04 (s, 9H). The second eluting peak was concentrated under reduced pressure to afford the title compound (330 mg, 31% yield) as a yellow solid. MS: m/z = 426.25 [M + H]+. 1H NMR (300 MHz, Chloroform-d) 7.66 - 7.60 (m, 4H), 7.48 - 7.37 (m, 6H), 5.46 - 5.25 (m, 1H), 4.18 - 4.06 (m, 1H), 3.61 - 3.57 (m, 1H), 3.43 - 3.37 (m, 1H), 3.34 - 3.22 (m, 1H), 2.70 - 2.42 (m, 2H), 2.26 - 2.18 (m, 1H), 1.97 - 1.81 (m, 1H), 1.67 - 1.61 (m, 1H), 1.20 - 1.17 (m, 3H), 1.04 (s, 9H). [0595] Step 2: (2R,6S,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6-methylhexahydro- 1H-pyrrolizine [0596] To an ice-cooled solution of (2S,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-methylhexahydro-3H-pyrrolizin-3-one (550 mg, 1.29 mmol) in THF (6 mL) under nitrogen atmosphere was added BH3•Me2S (0.65 mL, 6.46 mmol) dropwise. The reaction mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled in an ice bath, quenched with MeOH (7 mL) and 2 M HCl (7 mL). The resulting mixture was heated at 60 °C for another 1 hour. The resulting mixture was cooled and concentrated under reduced pressure. The resulting mixture was diluted with EtOAc (100 mL), washed with saturated aqueous NaHCO3 (3 x 50 mL) and brine (50 mL). The organic layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to give the title compound (350 mg, 65% yield) as a yellow oil. MS: m/z = 412.20 [M + H]+. [0597] Step 3: ((2R,6S,7aS)-2-Fluoro-6-methyltetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0598] To a mixture of (2R,6S,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methylhexahydro-1H-pyrrolizine (350 mg, 0.85 mmol) in MeOH (4 mL) was added NH4F (1259.65 mg, 34.00 mmol) under nitrogen atmosphere. The reaction mixture was heated at 65 °C for 4 hours. The resulting mixture was cooled and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 15% MeOH in DCM to afford
WSGR Docket No. 62619-720.601 the title compound (Intermediate 56, 60 mg, 40% yield) as a yellow oil. MS: m/z = 174.10 [M + H]+. [0599] Intermediate 57: ((6R,7aR)-2,6-Difluoro-2-methyltetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol
[0600] Step 1: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,6-difluoro-2- methylhexahydro-3H-pyrrolizin-3-one [0601] To a stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-3H-pyrrolizin-3-one (refer to Intermediate 62 for detail procedures, 1 g, 2.35 mmol) in THF (10 mL) was added LiHMDS (4.70 mL, 4.70 mmol) at -40 °C and then stirred at this temperature for 2 hours. Then NFSI (1.11 g, 3.52 mmol) was added to the above mixture, and the reaction mixture was stirred in an ice bath for 2 hours. The resulting mixture was quenched with saturated aq. NH4Cl (50 mL), extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 30% EA in PE to afford the title compound (700 mg, 67% yield) as a yellow oil. MS: m/z = 444.20 [M + H]+. 19F NMR (282 MHz, Chloroform-d) -141.07 (s, 1F), -172.34 (s, 1F). [0602] Step 2: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,6-difluoro-2- methylhexahydro-1H-pyrrolizine [0603] To an ice-cooled solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6- difluoro-2-methylhexahydro-3H-pyrrolizin-3-one (500 mg, 1.12 mmol) in THF (5 mL) was added BH3-Me2S (428.08 mg, 5.63 mmol) under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was heated at 60 °C for 16 hours. The reaction mixture was cooled in an ice bath, then MeOH (10 mL) and HCl aq. (2 M, 10 mL) were added. The resulting mixture was heated at 60 °C for 1 hour. The resulting mixture was cooled, quenched with saturated aq. NaHCO3 (50 mL) in an ice bath, and extracted with EA (3 x 50 mL). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 10% MeOH in DCM to afford the title compound (300 mg, 61% yield) as a white solid. MS: m/z = 430.15 [M + H]+. 1H NMR (400 MHz, Chloroform-d 3.74 - 3.71 (m, 1H), 3.49 - 3.40 (m, 2H), 3.14 - 2.97 (m, 3H), 2.44 - 1.91 (m, 4H), 1.47 - 1.39 (m, 3H), 1.06 (s, 9H). 19F NMR (376 MHz, Chloroform-d) -140.45 (s, 1F), -173.92 (s, 1F).
WSGR Docket No. 62619-720.601 [0604] Step 3: ((6R,7aR)-2,6-difluoro-2-methyltetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0605] A solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2,6-difluoro-2- methylhexahydro-1H-pyrrolizine (300 mg, 0.69 mmol) and NH4F (1034.49 mg, 27.92 mmol) in MeOH (5 mL) under nitrogen atmosphere was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 10% MeOH in DCM to afford the title compound (Intermediate 57, 110 mg, 82% yield) as a yellow solid. MS: m/z = 192.10 [M + H]+. 1H NMR (300 MHz, Chloroform-d 1.52 - 1.44 (m, 3H). 19 178.22 (m, 1F). [0606] Intermediate 58: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0607] Step 1: (R)-3-(Hydroxymethyl)-1,4-oxazepan-5-one [0608] To a solution of (S)-3-((benzyloxy)methyl)-1,4-oxazepan-5-one (400 mg, 1.70 mmol) in CH2Cl2 (8 mL) was added dropwise a solution of BCl3 (1 M, 3.40 mL) in CH2Cl2 (5 mL) at 0 – 5 °C under N2 over 10 min. The mixture was stirred at 25 °C for 0.5 hr. The reaction mixture was quenched with MeOH (8 mL) and concentrated. The resulting residue was diluted with CH2Cl2 (10 mL) at 0 °C and basified with TEA (1 mL). The mixture was concentrated. After purification by silica gel flash chromatography (Eluent of MeOH in CH2Cl2 = 0 to 2%), the title compound (200 mg, yield: 81%) was obtained as a white solid. 1H NMR (400 MHz, Chloroform-d 4.02 - 3.86 (m, 2H), 3.79 - 3.56 (m, 4H), 3.55 - 3.48 (m, 1H), 2.93 - 2.84 (m, 1H), 2.77 (br s, 1H), 2.58 - 2.49 (m, 1H). [0609] Step 2: (R)-(1,4-Oxazepan-3-yl)methanol [0610] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-5-one (200 mg, 1.38 mmol) in THF (5 mL) was added LiAlH4 (2.5 M in THF, 1.10 mL) at 0 °C. Then the mixture was stirred at 25 °C
WSGR Docket No. 62619-720.601 for 20 hr. The reaction mixture was quenched with H2O (0.1 mL), 15% NaOH (0.1 mL ) and H2O (0.3 mL). The solution was filtered and the filtrate was concentrated to give the title compound (140 mg, yield: 77%) as a yellow oil. 1H NMR (400 MHz, Chloroform-d 3.74 - 3.68 (m, 1H), 3.54 - 3.47 (m, 1H), 3.41 - 3.27 (m, 2H), 3.16 - 2.98 (m, 2H), 2.93 - 2.82 (m, 1H), 2.04 (br s, 2H), 1.90 - 1.80 (m, 2H). [0611] Step 3: (S)-5-((1,4-Oxazepan-3-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0612] A mixture of (R)-(1,4-oxazepan-3-yl)methanol (130 mg, 991 µmol) in THF (10 mL) was degassed and purged with N2 three times, and NaH (176 mg, 4.40 mmol, 60% purity) was added at 0°C. The mixture was stirred at 0 °C for 0.5 hr. The mixture was added to a solution of Intermediate 1 (295 mg, 1.10 mmol) in THF (10 mL) quickly at 0 °C under N2. The mixture was stirred at 25 °C for 1.5 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (1 mL) and dried over MgSO4. The residue was washed with CH2Cl2 (30 mL), filtered and concentrated to give the title compound (400 mg, crude) as a yellow solid. MS: m/z = 362.9 [M + H]+. [0613] Step 4: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0614] To a solution of (S)-5-((1,4-oxazepan-3-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (400 mg, 1.10 mmol) in CH2Cl2 (150 mL) were added DIEA (6.61 mmol, 1.15 mL) and POCl3 hr. The reaction mixture was diluted with saturated NaHCO3 (100 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (MeOH in CH2Cl2 = 0 to 6%), the title compound (Intermediate 58, 65 mg, yield: 17% over two steps) was obtained as a yellow solid. MS: m/z = 344.8 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 13.6, 4.8 Hz, 1H), 4.44 (d, J = 13.8 Hz, 1H), 4.20 (dd, J = 12.8, 4.0 Hz, 1H), 4.14 - 3.96 (m, 2H), 3.75 - 3.65 (m, 1H), 3.44 - 3.33 (m, 1H), 2.10 - 1.93 (m, 2H). [0615] Example 59: (S)-5-Chloro-2,4-difluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601
[0616] To a stirred solution of Intermediate 58 (900 mg, 2.60 mmol) in DMSO (9 mL) under nitrogen atmosphere was added KF (227.23 mg, 3.91 mmol) at room temperature, and the mixture was heated at 80 °C for 16 hours. The resulting mixture was cooled to room temperature, diluted with EtOAc (100 mL), and washed with brine (5 x 30 mL). The organic layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / EA (5 : 1) to afford the title compound (Intermediate 59, 750 mg, 87% yield) as a light yellow solid. MS: m/z = 329.00 [M + H]+. [0617] Intermediate 60: ((2R,7aS)-2-Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,6-d2)methanol
[0618] Step 1: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H- pyrrolizin-2-d-2-ol [0619] To an ice-cooled solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorotetrahydro-1H-pyrrolizin-2(3H)-one (refer to Intermediate 46 for detail procedures, 500 mg, 1.21 mmol) in Et2O (5 mL) under nitrogen atmosphere was added LiAlD4 (1 M in THF, 1.21 mL) dropwise. The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The reaction was quenched with H2O (0.05 mL), 15% aq. NaOH (0.05 mL) and H2O (0.15 mL) at room temperature for 10 min. The resulting mixture was filtered and concentrated under reduced pressure to afford the title compound (400 mg, crude used through) as a colorless oil. MS: m/z = 415.20 [M + H]+. [0620] Step 2: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H- pyrrolizin-2-yl-2-d 4-methylbenzenesulfonate [0621] To an ice-cooled solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-d-2-ol (400 mg, crude), DMAP (17.68 mg, 0.14 mmol) and TEA (292.88 mg, 2.89 mmol) in DCM (4 mL) under nitrogen atmosphere was added 4-methylbenzene- 1-sulfonyl chloride (275.89 mg, 1.44 mmol). The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with H2O (20 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue
WSGR Docket No. 62619-720.601 was purified by silica gel flash chromatography, eluted with 0-50% EA in PE to afford the title compound (400 mg, 57% yield for two steps) as a colorless oil. MS: m/z = 569.30 [M + H]+. [0622] Step 3: ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl-6,6-d2)methanol [0623] To an ice-cooled solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorohexahydro-1H-pyrrolizin-2-yl-2-d 4-methylbenzenesulfonate (400 mg, 0.70 mmol) in 2- methyloxolane (4 mL) under nitrogen atmosphere was added LiAlD4 (1 M in THF, 0.70 mL) dropwise. The ice bath was removed, and the reaction mixture was heated at 90 °C for 2 hours. The resulting mixture was cooled to room temperature, quenched with H2O (0.03 mL), 15% aq. NaOH (0.03 mL) and H2O (0.09 mL). The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM (2% NH3.H2O) to afford the title compound (Intermediate 60, 60 mg, 52% yield) as a colorless oil. MS: m/z = 162.10 [M + H]+. [0624] Intermediate 61: 6-(Methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1- ((triisopropylsilyl)ethynyl)-2-naphthonitrile
[0625] Step 1: 5-(2-(4-Bromophenyl)acetyl)-2,2-dimethyl-1,3-dioxane-4,6-dione [0626] To an ice-cooled solution of 2-(4-bromophenyl)acetic acid (25 g, 116.25 mmol) and 2,2- dimethyl-1,3-dioxane-4,6-dione (16.76 g, 116.25 mmol) in DCM (250 mL) were added DCC (26.39 g, 127.87 mmol) and DMAP (15.62 g, 127.87 mmol) under nitrogen atmosphere. The reaction mixture was stirred at room temperature for 16 hours. The resulting mixture was filtered, and the filter cake was washed with DCM (3 x 500 mL). The filtrate was washed with 1 M NaHSO4 aq. (2 x 500 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to afford the title compound (39 g, crude used through) as a light-yellow solid. MS: m/z = 340.95, 342.99 [M + H]+. [0627] Step 2: tert-Butyl 4-(4-bromophenyl)-3-oxobutanoate
WSGR Docket No. 62619-720.601 [0628] A solution of 5-(2-(4-bromophenyl)acetyl)-2,2-dimethyl-1,3-dioxane-4,6-dione (39 g, crude) in t-BuOH (480 mL) was heated at 88 °C for 5 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (10 : 1) to afford the title compound (31 g, 85% yield for two steps) as a light-yellow solid. MS: m/z = 312.85, 314.85 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 7.48 - 7.43 (m, 2H), 7.12 - 7.06 (m, 2H), 3.79 (s, 2H), 3.38 (s, 2H), 1.46 (s, 9H). [0629] Step 3: 4-(4-Bromophenyl)-3-oxobutanoic acid [0630] To a stirred solution of tert-butyl 4-(4-bromophenyl)-3-oxobutanoate (12.5 g, 39.91 mmol) in DCM (125 mL) was added TFA (125 mL) dropwise at room temperature. The resulting mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure to afford the title compound (10 g, crude used through) as a light-yellow solid. MS: m/z = 254.80, 256.80 [M - H]-. [0631] Step 4: 7-Bromonaphthalene-1,3-diol [0632] A solution of 4-(4-bromophenyl)-3-oxobutanoic acid (10 g, crude) in CF3SO3H (300 mL) was heated at 45 °C for 16 hours. The resulting mixture was quenched with saturated aq. NaHCO3 (2000 mL) in an ice bath and extracted with EtOAc (3 x 1000 mL). The combined organic layers were washed with brine (1000 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (5 : 1) to afford the title compound (7.0 g, 73% yield for two steps) as a brown solid. MS: m/z = 238.90, 240.90 [M + H]+. [0633] Step 5: 6,8-Dihydroxynaphthalene-2-carbonitrile [0634] To a stirred solution of 7-bromonaphthalene-1,3-diol (7 g, 29.28 mmol) in DMF (42 mL) under nitrogen atmosphere was added CuCN (3.15 g, 35.13 mmol) at room temperature. The reaction mixture was heated at 160 °C for 3 hours. The resulting mixture was cooled to room temperature and quenched with water (50 mL) at room temperature. The resulting mixture was extracted with EtOAc (3 x 300 mL) and washed with brine (3 x 200 mL). The organic layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (1 : 1) to afford the title compound (4.3 g, 79% yield) as a pink solid. MS: m/z = 183.90 [M - H]-. 1H NMR (400 MHz, DMSO-d6) 10.69 (s, 1H), 10.14 (s, 1H), 8.35 (d, J = 2.0 Hz, 1H), 7.72 (d, J = 8.4 Hz, 1H), 7.56 (dd, J = 8.4, 1.8 Hz, 1H), 6.69 (d, J = 2.0 Hz, 1H), 6.61 (d, J = 2.0 Hz, 1H). [0635] Step 6: 6,8-Dihydroxy-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile
WSGR Docket No. 62619-720.601 [0636] To a stirred solution of 6,8-dihydroxynaphthalene-2-carbonitrile (4.3 g, 23.22 mmol) and (2-bromoethynyl)triisopropylsilane (6.67 g, 25.54 mmol) in 1,4-dioxane (43 mL) under nitrogen atmosphere were added bis(1-methyl-4-(propan-2-yl)benzene); bis(dichlororuthenium) (1421.99 mg, 2.32 mmol) and KOAc (4.56 g, 46.44 mmol) at room temperature. The reaction mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (3 : 1) to afford the title compound (6.9 g, 81% yield) as a light-yellow solid. MS: m/z = 364.00 [M - H]-. 1H NMR (400 MHz, DMSO-d6) 10.60 (s, 1H), 10.19 (s, 1H), 7.69 (d, J = 8.8 Hz, 1H), 7.54 (d, J = 8.8 Hz, 1H), 6.69 - 6.65 (m, 2H), 1.16 (s, 21H). [0637] Step 7: 8-Hydroxy-6-(methoxymethoxy)-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile [0638] To an ice-cooled solution of 6,8-dihydroxy-1-((triisopropylsilyl)ethynyl)-2-naphthonitrile (4 g, 10.94 mmol) and DIEA (3.54 g, 27.35 mmol) in DCM (80 mL) under nitrogen atmosphere was added bromo(methoxymethoxy)methane (1.37 g, 10.94 mmol) dropwise. The reaction mixture was stirred in an ice bath for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with PE / EA (10 : 1) to afford the title compound (1.8 g, 40% yield) as a light yellow solid. MS: m/z = 410.45 [M + H]+. [0639] Step 8: 7-Cyano-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate [0640] To a stirred solution of 8-Hydroxy-6-(methoxymethoxy)-1-((triisopropylsilyl)ethynyl)-2- naphthonitrile (800 mg, 1.953 mmol) in DCM (8 mL) were added DIEA (757.30 mg, 5.85 mmol) and Tf2O (826.55 mg, 2.92 mmol) dropwise at -40 °C under nitrogen atmosphere. The reaction mixture was stirred at -40 °C for 1 hour. The resulting mixture was quenched with saturated aq. NaHCO3 (20 mL) in an ice bath and extracted with EA (3 x 20 mL). The combined organic layer was washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to afford the title compound (1.0 g, crude used through) as a yellow solid. [0641] Step 9: 6-(Methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1- ((triisopropylsilyl)ethynyl)-2-naphthonitrile [0642] To a stirred solution of 7-cyano-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl trifluoromethanesulfonate (1.0 g, crude) and bis(pinacolato)diboron (937.64 mg, 3.69 mmol) in 1,4-dioxane (10 mL) under nitrogen atmosphere were added Pd(dppf)Cl2•CH2Cl2 (150.39 mg, 0.18 mmol) and KOAc (543.56 mg, 5.53 mmol) at room temperature. The reaction mixture was heated at 110 °C for 2 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue
WSGR Docket No. 62619-720.601 was purified by silica gel flash chromatography, eluted with PE / EA (10 : 1) to afford the title compound (Intermediate 61, 550 mg, 54% yield for two steps) as an off-white solid. MS: m/z = 520.75 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 7.69 (d, J = 8.8 Hz, 1H), 7.57 - 7.54 (m, 2H), 7.37 (d, J = 2.4 Hz, 1H), 5.30 (s, 2H), 3.50 (s, 3H), 1.43 (s, 12H), 1.26 (s, 21H). [0643] Intermediate 62: ((2R,7aR)-2-Fluoro-6-methyl-2,3-dihydro-1H-pyrrolizin-7a(5H)- yl)methanol
[0644] Step 1: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methylhexahydro- 3H-pyrrolizin-3-one [0645] To an ice-cooled stirred solution of Intermediate 8 (250 mg, 0.607 mmol) in THF (5 mL) under nitrogen atmosphere was added LiHMDS (0.72 mL, 0.72 mmol, 1 M in THF) dropwise. The mixture was stirred in an ice bath for 1 hour. MeI (102.20 mg, 0.72 mmol) in THF (0.5 mL) was added dropwise to the above mixture in an ice bath. The resulting mixture was stirred in an ice bath for 2 hours. The resulting mixture was quenched with saturated aq. NH4Cl (20 mL) and extracted with EtOAc (3 x 20 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (1 / 3) to afford the title compound (175 mg, 67% yield). MS: m/z = 426.15 [M + H]+. [0646] Step 2: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methyl-2- (phenylselanyl)hexahydro-3H-pyrrolizin-3-one [0647] To a stirred solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-3H-pyrrolizin-3-one (550 mg, 1.29 mmol) in THF (10 mL) under nitrogen atmosphere was added LiHMDS (1.5 mL, 1 M in THF) dropwise at -78 °C. The mixture was stirred at -78 °C for 1 hour. Diphenyl diselenide (484.05 mg, 1.55 mmol) in THF (1 mL) was added dropwise. The dry ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (30 mL) in an ice bath and extracted with EA (3 x 50 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by
WSGR Docket No. 62619-720.601 silica gel flash chromatography, eluted with EA / PE (1 / 3) to afford the title compound (330 mg, 44% yield) as a yellow oil. MS: m/z = 582.15 [M + H]+. [0648] Step 3: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methyl-5,6,7,7a- tetrahydro-3H-pyrrolizin-3-one [0649] To an ice-cooled stirred solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluoro-2-methyl-2-(phenylselanyl)hexahydro-3H-pyrrolizin-3-one (500 mg, 0.86 mmol) in CH2Cl2 (6 mL) was added hydrogen peroxide (1.5 mL, 30wt%) dropwise. The reaction mixture was stirred in an ice bath for 2 hours. The resulting mixture was diluted with water (15 mL) and extracted with CH2Cl2 (3 x 15 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with EA / PE (1 / 3) to afford the title compound (330 mg, 91% yield) as a yellow solid. MS: m/z = 424.15 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 7.65 - 7.60 (m, 4H), 7.47 - 7.38 (m, 6H), 6.71 (s, 1H), 5.45 - 5.31 (m, 1H), 4.13 - 4.02 (m, 1H), 3.54 (s, 2H), 3.44 - 3.32 (m, 1H), 2.46 - 2.34 (m, 1H), 2.01- 1.89 (m, 1H), 1.86 (s, 3H), 1.04 (s, 9H). [0650] Step 4: (2R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-2-fluoro-6-methyl-2,3,5,7a- tetrahydro-1H-pyrrolizine [0651] To a stirred solution of (6R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methyl-5,6,7,7a-tetrahydro-3H-pyrrolizin-3-one (300 mg, 0.70 mmol) in THF (2 mL) under nitrogen atmosphere was added 9-BBN (6 mL, 3 mmol, 0.5 M in THF) at room temperature. The reaction mixture was heated at 50 °C for 1 hour. Additional 9-BBN (1 mL, 0.5 mmol, 0.5 M in THF) was added to the mixture and the reaction mixture was stirred at 50 °C for another 16 hours. The resulting mixture was cooled to room temperature, quenched with methanol (3 mL) and concentrated under reduced pressure. The residue was purified by RP- Flash with the following conditions: C18 Column, 20 - 30 um, 100 A, 40 g; Mobile Phase A: 5 mM aq. NH4HCO3; Mobile Phase B: MeCN; Flow rate: 40 mL/min; Gradient: 5% B hold 5 min, 5% B to 85% B in 20 min, 85% B hold 5 min, 85% B to 95% B in 5 min; Detector: UV 254 & 210 nm. The collected fractions were combined, concentrated and then lyophilized overnight to give the title compound (217 mg, 75% yield) as a light yellow solid. MS: m/z = 410.20 [M + H]+. 1H NMR (400 MHz, Chloroform-d) 7.73 - 7.65 (m, 4H), 7.44 - 7.34 (m, 6H), 5.33 - 5.32 (m, 1H), 5.27 - 5.11 (m, 1H), 3.98 - 3.87 (m, 1H), 3.54 - 3.47 (m, 3H), 3.26 - 3.08 (m, 2H), 2.16 - 1.98 (m, 2H), 1.69 (s, 3H), 1.05 (s, 9H). [0652] Step 5: ((2R,7aR)-2-Fluoro-6-methyl-2,3-dihydro-1H-pyrrolizin-7a(5H)-yl)methanol
WSGR Docket No. 62619-720.601 [0653] To a stirred solution of (2R,7aR)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-2-fluoro-6- methyl-2,3,5,7a-tetrahydro-1H-pyrrolizine (500 mg, 1.22 mmol) in MeOH (5 mL) was added NH4F (1.81 g, 48.84 mmol) at room temperature. The reaction mixture was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (15 / 1) to afford the title compound (Intermediate 62, 150 mg, 72% yield) as a yellow oil. MS: m/z = 172.10 [M + H]+. 1H NMR (300 MHz, Chloroform-d) 5.35 - 5.14 (m, 2H), 4.01 - 3.96 (m, 1H), 3.68 - 3.46 (m, 2H), 3.37 - 3.22 (m, 2H), 3.23 - 3.08 (m, 1H), 2.94 (bs, 1H), 2.17 - 1.82 (m, 2H), 1.74 (s, 3H). [0654] Intermediate 63: 22,3-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)-1-naphthonitrile
[0655] Step 1: 2-(8-Ethynyl-6,7-difluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane [0656] To a stirred solution of Intermediate 14 (2.5 g, 4.71 mmol) in DMF (25 mL) was added CsF (7.16 g, 47.12 mmol) at room temperature. The reaction mixture was stirred at room temperature for 3 hours. The resulting mixture was diluted with EtOAc (300 mL), washed with water (3 x 80 mL) and brine (80 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 9% EA in PE to afford the title compound (1.45 g, 82% yield) as a yellow solid. 1H NMR (300 MHz, Chloroform-d) 7.50 - 7.30 (m, 3H), 5.27 (s, 2H), 3.73 (s, 1H), 3.50 (s, 3H), 1.44 (s, 12H). 19F NMR (376 MHz, Chloroform-d) -132.16 - -132.22 (d, 1F), -137.25 - -137.31 (d, 1F). [0657] Step 2: 22,3-Difluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)-1-naphthonitrile [0658] To a solution of 2-(8-ethynyl-6,7-difluoro-3-(methoxymethoxy)naphthalen-1-yl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (1.15 g, 3.07 mmol) in THF (11 mL) under nitrogen atmosphere were added tert-butyl nitrite (1.27 g, 12.29 mmol) and 2-methylpyridin-1-ium-1-olate (1.34 g, 12.29 mmol) at room temperature. The reaction mixture was heated at 70 °C for 5 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 11% EA in PE to afford the
WSGR Docket No. 62619-720.601 title compound (Intermediate 63,479 mg, 41% yield) as a yellow solid. 1H NMR (400 MHz, Chloroform-d) 7.72 - 7.68 (m, 1H), 7.60 - 7.59 (m, 1H), 7.40 - 7.39 (m, 1H), 5.29 (s, 2H), 3.50 (s, 3H), 1.47 (s, 12H). 19F NMR (376 MHz, Chloroform-d) -127.27 - -127.33 (d, 1F), -136.19 - - 136.24 (d, 1F). [0659] Intermediate 64: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene
[0660] To an ice-cooled solution of Intermediate 12 (551.78 mg, 3.42 mmol) in THF (8 mL) under nitrogen atmosphere was added NaH (136.89 mg, 3.42 mmol, 60% dispersion in mineral oil). The mixture was stirred in an ice bath for 0.5 hour, and Intermediate 59 (750 mg, 2.28 mmol) was added. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (20 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 6% MeOH in DCM to afford the title compound (Intermediate 64, 870 mg, 81% yield) as an off-white solid. MS: m/z = 470.15 [M + H]+. [0661] Intermediate 65 & 66: ((1S,6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-3- oxohexahydro-1H-pyrrolizin-1-yl)boronic acid & ((1R,6R,7aR)-7a-(((tert- butyldiphenylsilyl)oxy)methyl)-6-fluoro-3-oxohexahydro-1H-pyrrolizin-1-yl)boronic acid
[0662] ((6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-3-oxohexahydro-1H- pyrrolizin-1-yl)boronic acid (refer to Intermediate 36 for detail procedures, 7.3 g, 16.030 mmol) was purified by HP-FLASH to give two isomers with the following conditions: Column: C18 gel Gradient: 0% B hold 10 min, up to 40% B within 5 min, 40% B to 80% B within 30 min, up to 95%
WSGR Docket No. 62619-720.601 B within 3 min, 95% B hold 10 min; Flow rate: 85 mL/min; Detector: UV 254 & 210 nm. The first eluting peak was collected and concentrated under reduced pressure to give 1.9 g crude product. The crude product was re-purified by RP-Flash with the following conditions: Column: C18 gel Gradient: 0% B hold 10 min, up to 40% B within 5 min, 40% B to 80% B within 30 min, up to 95% B within 3 min, 95% B hold 10 min; Flow rate: 85 mL/min; Detector: UV 254 & 210 nm. The product-containing fractions were collected and concentrated under reduced pressure to give the title compound (Intermediate 65, 1.6 g, 21% yield) as a white solid. MS: m/z = 456.20 [M + H]+. 1H NMR (400 MHz, Chloroform-d J = 52.4 Hz, 1H), 4.10 - 3.91 (m, 1H), 3.81 - 3.30 (m, 3H), 3.07 - 2.87 (m, 1H), 2.65 - 2.34 (m, 2H), 2.07 - 1.65 (m, 2H), 1.07 (s, 9H). 19F NMR (376 MHz, Chloroform-d second eluting peak was collected and concentrated under reduced pressure to give the title compound (Intermediate 66, 4.33 g, 59% yield) as a white solid. MS: m/z = 456.20 [M + H]+. 1H NMR (400 MHz, Chloroform-d J = 54.8 Hz, 1H), 4.12 - 3.90 (m, 1H), 3.59 (s, 2H), 3.53 - 3.47 (m, 1H), 3.13 - 2.90 (m, 1H), 2.83 - 2.33 (m, 2H), 1.96 - 1.73 (m, 2H), 1.08 (s, 9H). 19F NMR (376 MHz, Chloroform-d 1F). [0663] Intermediate 67: ((1R,6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
[0664] Step 1: (1R,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-3H- pyrrolizin-3-one [0665] To an ice-cooled mixture of Intermediate 66 (720 mg, 1.581 mmol), AgNO3 (268.57 mg, 1.581 mmol) and Selectfluor (3.36 g, 9.486 mmol) in DCM (8 mL) were added water (12 mL), H3PO4 (0.8 mL, 13.756 mmol) and TFA (3.2 mL, 43.082 mmol) under nitrogen atmosphere. The reaction mixture was heated at 50 °C for 16 hours. The resulting mixture was cooled to room temperature, diluted with water (80 mL), and extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-35% EtOAc in PE to give the title compound (278 mg, 40% yield) as a yellow solid. MS: m/z = 430.10 [M + H]+.
WSGR Docket No. 62619-720.601 [0666] Step 2: (1R,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,6-difluorohexahydro-1H- pyrrolizine [0667] To an ice-cooled stirred solution of (1R,6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)- 1,6-difluorohexahydro-3H-pyrrolizin-3-one (330 mg, 0.768 mmol) in THF (8 mL) was added BH3- Me2S (0.38 mL, 3.840 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was heated at 60 °C for 16 hours. The resulting mixture was cooled and quenched with MeOH (8 mL) and 2 M HCl (8 mL) in an ice bath. The ice bath was removed, and the resulting mixture was heated at 60 °C for another 1 hour. The resulting mixture was cooled and concentrated under reduced pressure. The residue was diluted with EtOAc (30 mL) and washed with saturated aqueous NaHCO3 (30 mL). The aqueous phase was extracted with EtOAc (3 x 30 mL). The combined organic layers were washed brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-35% EtOAc in PE to give the title compound (144 mg, 45% yield) as a yellow solid. MS: m/z = 416.15 [M + H]+. 1H NMR (400 MHz, Chloroform-d (m, 4H), 7.49 - 7.30 (m, 6H), 5.17 (d, J = 54.4 Hz, 1H), 5.05 - 4.83 (m, 1H), 3.43 - 3.23 (m, 2H), 3.21 - 2.93 (m, 4H), 2.65 - 2.43 (m, 1H), 2.30 - 1.79 (m, 3H), 1.07 (s, 9H). 19F NMR (376 MHz, Chloroform-d [0668] Step 3: ((1R,6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0669] To a stirred solution of (1R,6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-1,6- difluorohexahydro-1H-pyrrolizine (400 mg, 0.962 mmol) in THF (7 mL) was added TBAF (1.44 mL, 1.443 mmol, 1 M in THF) at room temperature. The reaction mixture was stirred at room temperature for 16 hours. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-10% MeOH in DCM to give the title compound (Intermediate 67, 100 mg, 58% yield) as a yellow solid. MS: m/z = 178.05 [M + H]+. [0670] Intermediate 68: (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-2-methylhexahydro-1H-pyrrolizin- 2-ol
[0671] Step 1: (6R,7aS)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-2-methylhexahydro- 1H-pyrrolizin-2-ol [0672] To an ice-cooled solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6- fluorotetrahydro-1H-pyrrolizin-2(3H)-one (refer to Intermediate 46 for detail procedures, 500 mg,
WSGR Docket No. 62619-720.601 1.21 mmol) in THF (5 mL) under nitrogen atmosphere was added CH3MgBr (3.67 mL, 31.83 mmol). The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aqueous NH4Cl (15 mL), extracted with EtOAc (3 x 20 mL). The combined organic layers were washed with brine (8 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 61% EA in PE to afford the title compound (390 mg, 75% yield) as a colorless oil. MS: m/z = 428.20 [M + H]+. 1H NMR (400 MHz, DMSO- d6) 7.69 - 7.63 (m, 4H), 7.47 - 7.41 (m, 6H), 5.35 - 5.18 (m, 1H), 3.29 (s, 2H), 3.19 - 3.09 (m, 2H), 3.00 - 2.71 (m, 2H), 2.46 - 2.21 (m, 2H), 1.99 - 1.65 (m, 2H), 1.15 (s, 3H), 0.99 (s, 9H). 19F NMR (376 MHz, DMSO-d6) -172.22 (s, 1F). [0673] Step 2: (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-2-methylhexahydro-1H-pyrrolizin-2-ol [0674] To a solution of (6R,7aS)-7a-(((tert-butyldiphenylsilyl)oxy)methyl)-6-fluoro-2- methylhexahydro-1H-pyrrolizin-2-ol (414.8 mg, 0.97 mmol) in CH3OH (4.2 mL) under nitrogen atmosphere was added NH4F (1437.37 mg, 38.81 mmol) at room temperature. The reaction mixture was heated at 65 °C for 8 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 25% CH3OH in CH2Cl2 (2% NH4•H2O) to afford the title compound (Intermediate 68, 200 mg, 54% yield) as an off-white solid. MS: m/z = 190.15 [M + H]+. [0675] Intermediate 69: ((6R,7aR)-6-Fluoro-1-methylenetetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol
[0676] Step 1: (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-1- methylenehexahydro-1H-pyrrolizine [0677] To an ice-cooled stirred solution of potassium 2-methylpropan-2-olate (408.94 mg, 3.64 mmol) in THF (10 mL) under nitrogen atmosphere was added methyltriphenylphosphanium bromide (1215.07 mg, 3.40 mmol). The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. To the above mixture was added (6R,7aS)-7a-(((tert- butyldiphenylsilyl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-1-one (500 mg, 1.21 mmol) over 3 min in an ice bath. The ice bath was removed, and the resulting mixture was stirred at room temperature for 16 hours. The resulting mixture was quenched with saturated aq. NH4Cl (100 mL), extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue
WSGR Docket No. 62619-720.601 was purified by Prep-TLC (PE / EA = 4 : 1) to afford the title compound (350 mg, 70% yield) as a colorless oil. MS: m/z = 410.10 [M + H]+. 1H NMR (300 MHz, Chloroform-d) 7.77 - 7.66 (m, 4H), 7.45 - 7.34 (m, 6H), 5.32 - 5.14 (m, 1H), 5.02 - 4.83 (m, 2H), 3.50 (s, 2H), 3.38 - 3.09 (m, 3H), 2.94 - 2.80 (m, 1H), 2.58 (s, 2H), 2.43 - 2.03 (m, 2H), 1.06 (s, 9H). 19F NMR (282 MHz, Chloroform-d) -169.04 (s, 1F). [0678] Step 2: ((6R,7aR)-6-Fluoro-1-methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0679] A solution of (6R,7aR)-7a-(((tert-Butyldiphenylsilyl)oxy)methyl)-6-fluoro-1- methylenehexahydro-1H-pyrrolizine (170 mg, 0.41 mmol) and NH4F (614.84 mg, 16.60 mmol) in MeOH (2 mL) under nitrogen atmosphere was heated at 65 °C for 4 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by Prep-TLC (DCM / MeOH = 10 : 1) to afford the title compound (Intermediate 69, 55 mg, 77% yield) as a colorless oil. MS: m/z = 172.05 [M + H]+. [0680] Intermediate 70: (8R,8aS)-2,5-Dichloro-8-ethyl-4-fluoro-8a,9,12,13-tetrahydro-8H,11H- 7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0681] Step 1: Benzyl (R)-3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate [0682] To a solution of (R)-(1,4-oxazepan-3-yl)methanol (3 g, 22.8 mmol) in CH2Cl2 (35 mL) were added TEA (4.63 g, 45.7 mmol), DMAP (279 mg, 2.29 mmol) and CbzCl (4.29 g, 25.1 mmol). The mixture was stirred at 25 °C for 2 hr. The reaction mixture was diluted with water (30 mL) and extracted with CH2Cl2 (30 mL x 2). The combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 30% EtOAc in petroleum ether), the title compound (1 g, yield: 15%) was obtained as a colorless oil. 1H NMR (400 MHz, Chloroform-d 5.22 - 5.08 (m, 2H), 4.44 - 4.24 (m, 1H), 4.08 - 4.02 (m, 1H), 4.01 - 3.87 (m, 2H), 3.78 - 3.58 (m, 3H), 3.55 - 3.47 (m, 1H), 3.35 - 3.23 (m, 1H), 1.98 - 1.81 (m, 1H), 1.76 - 1.66 (m, 1H). [0683] Step 2: (S)-Benzyl 3-formyl-1,4-oxazepane-4-carboxylate
WSGR Docket No. 62619-720.601 [0684] To a solution of (R)-benzyl 3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate (1 g, 3.77 mmol) in CH2Cl2 (10 mL) was added DMP (2.4 g, 5.65 mmol). The mixture was stirred at 0 °C for 1 hr. The reaction mixture was filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 50% EtOAc in petroleum ether), (S)- benzyl 3-formyl-1,4-oxazepane-4-carboxylate (810 mg, yield: 81%) was obtained as a colorless oil. 1H NMR (400 MHz, Chloroform-d 4.70 - 4.48 (m, 1H), 4.27 - 4.08 (m, 1H), 3.99 - 3.92 (m, 1H), 3.87 - 3.80 (m, 1H), 3.79 - 3.63 (m, 3H), 1.93 - 1.81 (m, 2H). [0685] Step 3: Benzyl (S)-3-(1-hydroxypropyl)-1,4-oxazepane-4-carboxylate [0686] To a solution of (S)-benzyl 3-formyl-1,4-oxazepane-4-carboxylate (810 mg, 3.08 mmol) in THF (10 mL) was stirred at -78 °C for 10 min under N2 atmosphere, and then EtMgBr (3 M, 2 mL) was added dropwise at -78 °C. The resulting mixture was stirred at 20 °C for 1 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (10 mL) at 0 °C and extracted with EtOAc (15 mL x 2). The combined organic layers were washed with brine (20 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 50% EtOAc in petroleum ether), the title compound (450 mg, yield: 50%) was obtained as a yellow solid. 1H NMR (400 MHz, Chloroform-d (m, 5H), 5.21 - 5.06 (m, 2H), 4.28 - 4.16 (m, 1H), 4.16 - 4.05 (m, 2H), 4.01 - 3.88 (m, 2H), 3.78 - 3.70 (m, 2H), 3.55 - 3.43 (m, 1H), 3.36 - 3.25 (m, 1H), 1.76 - 1.62 (m, 2H), 1.52 - 1.32 (m, 2H), 1.02 - 0.88 (m, 3H). [0687] Step 4: (S)-1-(1,4-Oxazepan-3-yl)propan-1-ol [0688] To a solution of (S)-benzyl 3-(1-hydroxypropyl)-1,4-oxazepane-4-carboxylate (450 mg, 1.53 mmol) in MeOH (10 mL) was added Pd/C (200 mg, 10% purity). The mixture was degassed, purged with H2 three times, and stirred at 25 °C for 4 hr under H2 (15 Psi) atmosphere. The reaction mixture was filtered and concentrated to give the title compound (230 mg, yield: 94%) as a colorless oil. 1H NMR (400 MHz, Chloroform -d - 3.55 (m, 1H), 3.54 - 3.36 (m, 1H), 3.20 - 3.04 (m, 1H), 3.03 - 2.71 (m, 2H), 1.90 - 1.74 (m, 2H), 1.50 - 1.36 (m, 2H), 1.04 - 0.95 (m, 3H). [0689] Step 5: (S)-5-(1-(1,4-Oxazepan-3-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0690] A mixture of (S)-1-(1,4-oxazepan-3-yl)propan-1-ol (220 mg, 1.38 mmol) in THF (20 mL) was degassed, purged with N2 three times. NaH (221 mg, 5.53 mmol, 60% purity) was added at 0 °C. The mixture was stirred at 0 °C for 0.5 hr. Then a solution of Intermediate 1 (371 mg, 1.38
WSGR Docket No. 62619-720.601 mmol) in THF (5 mL) was added to the mixture at 0 °C. The mixture was stirred at 25 °C for 0.5 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (0.2 mL) and dried over MgSO4. The residue was washed with CH2Cl2 (100 mL), filtered and concentrated under reduced pressure. The title compound (480 mg, yield: 88%) was obtained as a yellow solid. MS: m/z = 391.0, 393.0 [M + H]+. [0691] Step 6: (8R,8aS)-2,5-Dichloro-8-ethyl-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0692] To a solution of (S)-5-(1-(1,4-oxazepan-3-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (450 mg, 1.15 mmol) in CH2Cl2 (100 mL) was added dropwise DIPEA (1.2 mL, 6.90 mmol) at 0 °C. The mixture was stirred 0 °C for 10 min, and then POCl3 mmol) was added dropwise at 0 °C. The resulting mixture was stirred at 0 °C for 50 min. The reaction mixture was diluted with saturated NaHCO3 (30 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 10% EtOAc in CH2Cl2), the title compound (Intermediate 70, 100 mg, yield: 16%) was obtained as a yellow solid. MS: m/z = 372.8, 374.8 [M + H]+. [0693] Intermediate 71: (8R,8aS)-5-Chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
[0694] mixture was stirred at 110 °C for 16 hr under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 7% MeOH in CH2Cl2), the title compound (Intermediate 71, 100 mg, yield: 61%) was obtained as a yellow solid. MS: m/z = 498.0, 499.9 [M + H]+.19F NMR (400 MHz, Chloroform-d) [0695] Intermediate 72: (R)-5-Chloro-4-fluoro-7-methyl-2-(methylthio)-7,8,8a,9,12,13-hexahydro- 11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601
[0696] Step: (R)-(4-Benzyl-1,4-oxazepan-3-yl)methanol [0697] To a solution of (R)-(1,4-oxazepan-3-yl)methanol (3 g, 22.9 mmol), DIPEA (7.4 g, 57.4 mmol) in CH2Cl2 (30 mL) was added dropwise bromomethylbenzene (3.9 g, 22.9 mmol) at -10 °C. The mixture was stirred at 25 °C for 0.5 hr. The reaction mixture was diluted with H2O (100 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (150 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography ( eluent of 0 ~ 50% EtOAc in Petroleum ether), the title compound (4.6 g, yield: 82%) was obtained as a yellow oil. MS: m/z = 222.1 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 12.8, 7.6 Hz, 1H), 3.55 - 3.38 (m, 2H), 3.08 - 2.99 (m, 2H), 2.82 - 2.75 (m, 1H), 1.96 - 1.85 (m, 1H), 1.80 - 1.69 (m, 1H). [0698] Step 2: (S)-4-Benzyl-3-(chloromethyl)-1,4-oxazepane [0699] To a solution of (R)-(4-benzyl-1,4-oxazepan-3-yl)methanol (4.5 g, 20.3 mmol), TEA (6.17 g, 61.0 mmol) in THF (45 mL) was added MsCl (2.4 g, 21.0 mmol) at -10 °C. The mixture was stirred at 25 °C for 0.5 hr. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (30 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. The title compound (3 g, yield: 55%) was obtained as a light yellow oil. 1H NMR (400 MHz, Chloroform-d - 3.87 (m, 1H), 3.87 - 3.69 (m, 4H), 3.69 - 3.55 (m, 2H), 3.50 - 3.40 (m, 1H), 3.04 - 2.87 (m, 2H), 2.86 - 2.76 (m, 1H), 1.90 - 1.76 (m, 1H), 1.68 - 1.58 (m, 1H). [0700] Step 3: (R)-3-(Azidomethyl)-4-benzyl-1,4-oxazepane [0701] To a solution of (S)-4-benzyl-3-(chloromethyl)-1,4-oxazepane (2.9 g, 12.1 mmol) in DMF (30 mL) was added NaN3 (2.3 g, 35.2 mmol). The mixture was stirred at 70 °C for 3 hr. The reaction mixture was diluted with H2O (200 mL) and extracted with EtOAc (100 mL x 2). The combined organic layers were washed with brine (200 mL), dried over Na2SO4, filtered and concentrated under reduced pressure, the title compound (2.7 g, yield: 77%) was obtained as a yellow oil. MS: m/z = 246.9 [M + H]+.1H NMR (400 MHz, Chloroform-d
WSGR Docket No. 62619-720.601 3.87 - 3.78 (m, 3H), 3.76 - 3.68 (m, 3H), 3.35 - 3.23 (m, 2H), 3.04 - 2.93 (m, 2H), 2.90 - 2.83 (m, 1H), 1.97 - 1.84 (m, 1H), 1.68 - 1.61 (m, 1H). [0702] Step 4: tert-Butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)carbamate [0703] To a solution of (R)-3-(azidomethyl)-4-benzyl-1,4-oxazepane (2.7 g, 11.0 mmol) in MeOH (30 mL) was added Pd/C (500 mg, 10% purity). The mixture was stirred at 25 °C for 2 hr under H2 (15 Psi). Then the mixture was filtered and concentrated under reduced pressure. Then THF (20 mL), DIPEA (1.46 g, 11.3 mmol) and Boc2O (1.98 g, 9.06 mmol) were added. The mixture was stirred at 25 °C for 3 hr. The reaction mixture was diluted with H2O (50 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (30 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 25% EtOAc in petroleum ether), the title compound (1.6 g, yield: 42%) was obtained as a yellow oil. MS: m/z = 321.2 [M + H]+. [0704] Step 5: tert-Butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)(methyl)carbamate [0705] A solution of tert-butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)carbamate (1.6 g, 4.99 mmol) in THF (5 mL) was degassed and purged with N2 three times. NaH (599 mg, 15.0 mmol, 60% purity) was added at 0 °C, and the mixture was stirred at 25 °C for 0.5 hr under N2 atmosphere. Then CH3I (2.13 g, 15.0 mmol) was added to the mixture at 0 °C, and the mixture was stirred at 25 °C for 17.5 hr. The mixture was quenched with saturated NH4Cl (20 mL) at 0 °C. The mixture was diluted with water (50 mL) and extracted with EtOAc (100 mL x 2). The combined organic layers were washed with brine (200 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 10% EtOAc in petroleum ether), the title compound (745 mg, yield: 31%) was obtained as a colorless oil. MS: m/z = 335.0 [M + H]+.1H NMR (400 MHz, Chloroform-d 3.91 (m, 1H), 3.84 - 3.77 (m, 2H), 3.75 - 3.66 (m, 2H), 3.57 - 3.30 (m, 2H), 3.24 - 3.12 (m, 1H), 3.08 - 2.92 (m, 2H), 2.90 - 2.77 (m, 4H), 1.93 - 1.84 (m, 1H), 1.70 - 1.62 (m, 1H), 1.46 - 1.41 (m, 9H). [0706] Step 6: tert-Butyl (R)-((1,4-oxazepan-3-yl)methyl)(methyl)carbamate [0707] To a solution of tert-butyl (R)-((4-benzyl-1,4-oxazepan-3-yl)methyl)(methyl)carbamate (745 mg, 2.23 mmol) in MeOH (10 mL) was added Pd(OH)2 (745 mg, 20% purity). The mixture was stirred at 25 °C for 3 hr under H2 (15 Psi). The reaction mixture was filtered and concentrated under reduced pressure to give the title compound (600 mg, yield: 77%) as a colourless oil. 1H NMR (400 MHz, Chloroform-d
WSGR Docket No. 62619-720.601 3.15 - 3.00 (m, 3H), 2.88 (s, 3H), 2.85 - 2.79 (m, 1H), 1.86 - 1.83 (m, 1H), 1.65 - 1.61 (m, 1H), 1.45 (s, 9H). [0708] Step 7: tert-Butyl (R)-((4-(5,7-dichloro-8-fluoro-2-(methylthio)pyrido[4,3-d]pyrimidin-4- yl)-1,4-oxazepan-3-yl)methyl)(methyl)carbamate [0709] To a solution of 5,7-dichloro-8-fluoro-2-(methylthio)pyrido[4,3-d]pyrimidin-4-ol (611 mg, 2.18 mmol) in toluene (50 mL) were added DIPEA (2.5 g, 19.5 mmol) and POCl3 (2.8 g, 18.2 mmol). The mixture was stirred at 80 °C for 1.5 hr and then concentarted under reduce pressure. The residue was dissolved in CH2Cl2 (25 mL). A solution of tert-butyl (R)-((1,4-oxazepan-3- yl)methyl)(methyl)carbamate (523 mg, 2.14 mmol), CH2Cl2 (50 mL) and DIEA (5.0 g, 39.0 mmol) was added to the resulting mixture. The mixture was strried at 25 °C for 0.5 hr. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 30 % EtOAc in petroleum ether), the title compound (900 mg, yield: 71%) was obtained as a yellow oil. MS: m/z = 506.1, 507.9 [M + H]+. [0710] Step 8: (R)-5-Chloro-4-fluoro-7-methyl-2-(methylthio)-7,8,8a,9,12,13-hexahydro-11H-10- oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalene [0711] To a solution of tert-butyl (R)-((4-(5,7-dichloro-8-fluoro-2-(methylthio)pyrido[4,3- d]pyrimidin-4-yl)-1,4-oxazepan-3-yl)methyl)(methyl)carbamate (800 mg, 1.58 mmol) in MeCN (55 mL) was added HCl/1,4-dioxane (4 M, 9 mL). The mixture was stirred at 25 °C for 0.5 hr. The mixture was concentarted under reduce pressure. Then MeCN (50 mL) and DIPEA (6.9 g, 51.7 mmol) were added to the residue. The mixture was stirred at 80 °C for 1.5 hr. The reaction mixture was diluted with H2O (100 mL) and extracted with EtOAc (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 30% EtOAc in petroleum ether), the title compound (Intermediate 72, 530 mg, yield: 63%) was obtained as a yellow solid. MS: m/z = 369.8, 371.7 [M + H]+.1H NMR (400 MHz, Chloroform-d 5.00 (m, 0.5H), 4.68 - 4.57 (m, 0.5H), 4.11 - 3.87 (m, 3H), 3.82 - 3.63 (m, 3H), 3.60 - 3.43 (m, 2H), 3.34 - 3.21 (m, 3H), 2.58 (s, 3H), 2.08 - 1.95 (m, 2H). [0712] Intermediate 73: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene-8,8-d2
WSGR Docket No. 62619-720.601
[0713] Step 1: Methyl N-(tert-butoxycarbonyl)-O-(tert-butyldiphenylsilyl)-L-serinate [0714] To a solution of methyl (tert-butoxycarbonyl)-L-serinate (20 g, 91.2 mmol) in CH2Cl2 (200 mL) were added imidazole (12.4 g, 182 mmol) and tert-butylchlorodiphenylsilane (35 mL, 137 mmol). The mixture was stirred at 25 °C for 16 hr. The mixture was diluted with H2O (400 mL) and extracted with CH2Cl2 (400 mL × 2). The combined organic layers were washed with brine (400 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 2% EtOAc in petroleum ether), the title compound (27 g, yield: 49%) was obtained as a colourless oil. MS: m/z = 480.2 [M + Na]+.1H NMR (400 MHz, Chloroform-d J = 8.4 Hz, 1H), 4.44 - 4.36 (m, 1H), 4.07 (dd, J = 10.0, 2.8 Hz, 1H), 3.89 (dd, J = 10.0, 2.8 Hz, 1H), 3.74 (s, 3H), 1.46 (s, 9H), 1.03 (s, 9H). [0715] Step 2: tert-Butyl (R)-(1-((tert-butyldiphenylsilyl)oxy)-3-hydroxypropan-2-yl-3,3- d2)carbamate [0716] To a solution of methyl N-(tert-butoxycarbonyl)-O-(tert-butyldiphenylsilyl)-L-serinate (17 g, 24.5 mmol) in THF (170 mL) was added LiAlD4 (3.26 g, 85.8 mmol) at 0 °C. The mixture was stirred at 25 °C for 2 hr under N2 atmosphere. The reaction mixture was quenched with Na2SO4 10H2O (30 g) at 0 °C, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 15% EtOAc in petroleum ether), the title compound (7.7 g, yield: 48%) was obtained as a colorless oil. MS: m/z = 454.3 [M + Na]+.1H NMR (400 MHz, Chloroform-d 3.82 - 3.75 (m, 2H), 3.74 - 3.68 (s, 1H), 2.37 (s, 1H), 1.44 (s, 9H), 1.07 (s, 9H). [0717] Step 3: tert-Butyl (R)-(1-(benzyloxy)-3-((tert-butyldiphenylsilyl)oxy)propan-2-yl-1,1- d2)carbamate
WSGR Docket No. 62619-720.601 [0718] To a solution of tert-butyl (R)-(1-((tert-butyldiphenylsilyl)oxy)-3-hydroxypropan-2-yl-3,3- d2)carbamate (7.7 g, 17.8 mmol) in DMF (90 mL) was added NaH (460 mg, 11.5 mmol, 60% purity in oil) at 0 °C under N2 atmosphere. The mixture was stirred at 25 °C for 0.5 hr, then BnBr(1.47 g, 8.62 mmol) in THF (10 mL) was added into the mixture at 0 °C under N2 atmosphere. The mixture was stirred at 0 °C for 16 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (5 mL) and dried over MgSO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 7% EtOAc in petroleum ether), the title compound (3.5 g, yield: 83%) was obtained as a colorless oil. MS: m/z = 544.3 [M + Na]+. [0719] Step 4: tert-Butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-1,1-d2)carbamate [0720] To a solution of tert-butyl (R)-(1-(benzyloxy)-3-((tert-butyldiphenylsilyl)oxy)propan-2-yl- 1,1-d2)carbamate (3.5 g, 6.71 mmol) in THF (26 mL) was added TBAF (13.4 mL, 1 M in THF). The mixture was stirred at 25 °C for 2 hr. The mixture was diluted with H2O (150 mL) and extracted with EtOAc (150 mL × 2). The combined organic layers were washed with brine (150 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 6% EtOAc in petroleum ether), the title compound (1.7 g, yield: 84%) was obtained as a colorless oil. MS: m/z = 306.1 [M + Na]+.1H NMR (400 MHz, Chloroform-d 3.73 - 3.61 (m, 2H), 1.44 (s, 9H). [0721] Step 5: tert-Butyl (S)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy-3,3- d2)propanoate [0722] To a solution of tert-butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-1,1-d2)carbamate (1.7 g, 6.00 mmol) in t-BuOH (18 mL) were added Cs2CO3 (2.35 g, 7.20 mmol) and tert-butyl acrylate (18.8 g, 146 mmol). The mixture was stirred at 25 °C for 16 hr. The mixture was diluted with H2O (50 mL) and extracted with EtOAc (50 mL × 2). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 6% EtOAc in petroleum ether), the title compound (1.6 g, yield: 61%) was obtained as a yellow oil. MS: m/z = 434.2 [M + Na]+.1H NMR (400 MHz, Chloroform-d 3.83 (m, 1H), 3.69 - 3.65 (m, 2H), 3.61 - 3.55 (m, 1H), 3.52 - 3.46 (m, 1H), 2.47 - 2.42 (m, 2H), 1.45 (s, 9H), 1.43 (s, 9H). [0723] Step 6: (S)-3-(2-Amino-3-(benzyloxy)propoxy-3,3-d2)propanoic acid
WSGR Docket No. 62619-720.601 [0724] To a solution of tert-butyl (S)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy- 3,3-d2)propanoate (1.6 g, 3.89 mmol) in CH2Cl2 (20 mL) was added TFA (19.2 mL). The mixture was stirred at 25 °C for 2 hr. The reaction mixture was concentrated under reduced pressure. The title compound (1.5 g, TFA salt) was obtained as a yellow oil. MS: m/z = 256.1[M + H]+. [0725] Step 7: (S)-3-((Benzyloxy)methyl-d2)-1,4-oxazepan-5-one [0726] To a solution of (S)-3-(2-amino-3-(benzyloxy)propoxy-3,3-d2)propanoic acid (1.5 g, 5.88 mmol) in CH2Cl2 (150 mL) were added HATU (2.68 g, 7.05 mmol) and TEA (2.97 g, 29.4 mmol). The mixture was stirred at 25 °C for 3 hr. The mixture was diluted with H2O (500 mL) and extracted with CH2Cl2 (500 mL × 2). The combined organic layers were washed with brine (500 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 48% EtOAc in petroleum ether), the title compound (1.39 g, yield: 61%) was obtained as a colorless oil. MS: m/z = 238.1 [M + H]+.1H NMR (400 MHz, Chloroform-d (m, 1H), 3.85 - 3.79 (m, 1H), 3.70 - 3.63 (m, 1H), 3.60 - 3.30 (m, 2H), 2.90 - 2.80 (m, 1H), 2.59 - 2.51 (m, 1H). [0727] Step 8: (R)-3-(Hydroxymethyl-d2)-1,4-oxazepan-5-one [0728] To a solution of (S)-3-((benzyloxy)methyl-d2)-1,4-oxazepan-5-one (900 mg, 3.79 mmol) in CH2Cl2 (20 mL) was added dropwise a solution of BCl3 (1 M in CH2Cl2, 7.59 mL) at 0 - 5 °C under N2 atmosphere over 10 min. The mixture was stirred at 25 °C for 1 hr. The reaction mixture quenched with MeOH (100 mL) and concentrated under reduced pressure. The resulting residue neutralized with NH3 H2O (pH~7-8). The mixture was concentrated. After purification by silica gel flash chromatography (Eluent of 0 ~ 10% MeOH in CH2Cl2), the title compound (470 mg, yield: 84%) was obtained as an off white solid. 1H NMR (400 MHz, Chloroform-d 1H), 3.97 - 3.89 (m, 1H), 3.82 - 3.47 (m, 5H), 2.93 - 2.83 (m, 1H), 2.54 (dd, J = 16.0, 6.0 Hz, 1H). [0729] Step 9: (R)-(1,4-Oxazepan-3-yl)methan-d2-ol [0730] To a solution of (R)-3-(hydroxymethyl-d2)-1,4-oxazepan-5-one (470 mg, 3.19 mmol) in THF (5 mL) was added LiAlH4 (242 mg, 6.39 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 hr. The reaction mixture was quenched with H2O (0.24 mL), 15% NaOH (0.24 mL), and H2O (0.72 mL). Then the mixture was filtered and concentrated under reduced pressure to give the title compound (280 mg, yield: 66%) as a white solid. 1H NMR (400 MHz, Chloroform-d 3.85 - 3.79 (m, 1H), 3.74 - 3.67 (m, 1H), 3.56 - 3.20 (m, 2H), 3.06 - 3.16 (m, 1H), 3.02 (dd, J = 8.0, 4.0 Hz, 1H), 2.89 - 2.80 (m, 1H), 2.14 - 2.03 (m, 2H), 1.88 – 1.80 (m, 2H).
WSGR Docket No. 62619-720.601 [0731] Step 10: (S)-5-((1,4-Oxazepan-3-yl)methoxy-d2)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0732] To a solution of NaH (336 mg, 8.41 mmol, 60% purity in oil) in THF (8 mL) was added (R)-(1,4-oxazepan-3-yl)methan-d2-ol (280 mg, 2.10 mmol) in THF (3 mL) at 0 °C under N2 atmosphere. The mixture was stirred at 25 °C for 0.5 hr, then 2,5,7-trichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (564 mg, 2.10 mmol) in THF (3 mL) was added at 0 °C under N2 atmosphere. The mixture was stirred at 25 °C for 1 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (2 mL), dried over MgSO4, filtered. The filter cake was washed with CH2Cl2 (20 mL), and the filtrate was concentrated under reduced pressure to give the title compound (760 mg, yield: 78%) as a light yellow solid. MS: m/z = 364.9 [M + H]+. [0733] Step 11: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-8,8-d2. [0734] To a solution of (S)-5-((1,4-oxazepan-3-yl)methoxy-d2)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (760 mg, 2.08 mmol) in CH2Cl2 (300 mL) were added DIPEA (2.17 mL, 12.5 mmol) and then POCl3 2 atmosphere. The mixture was stirred at 0 °C for 2 hr under N2 atmosphere. The reaction mixture was added into saturated NaHCO3 (100 mL) slowly and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated. After purification by silica gel flash chromatography (Eluent of 0 ~ 28% EtOAc in petroleum ether), the title compound (Intermediate 73, 100 mg, yield: 10%) was obtained as a light yellow solid. MS: m/z = 346.7 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 4.60 - 4.49 (m, 1H), 4.36 - 4.33 (m, 1H), 4.12 - 4.01 (m, 1H), 3.90 - 3.81 (m, 2H), 1.94 - 1.86 (m, 2H). [0735] Intermediate 74: 4-Fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene-8,8-d2
[0736] mixture was stirred at 100 °C for 16 hr under N2 atmosphere. The mixture was concentrated. After
WSGR Docket No. 62619-720.601 purification by silica gel flash chromatography (Eluent of 0 ~ 8% MeOH in CH2Cl2), the title compound (Intermediate 74, 48 mg, yield: 30%) was obtained as a light yellow solid. MS: m/z = 472.0 [M + H]+. [0737] Intermediate 75: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene-13,13-d2
[0738] Step 1: (R)-(1,4-Oxazepan-3-yl-5,5-d2)methanol [0739] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-5-one (refer to Intermediate 58 for detail procedures, 200 mg, 1.38 mmol) in THF (5 mL) was added LiAlD4 (126 mg, 2.76 mmol) at 0 °C. The mixture was stirred at 25 °C for 16 hr. The reaction mixture was quenched with H2O (0.1 mL), 15% NaOH (0.1 mL), and H2O (0.3 mL). The mixture was filtered and concentrated under reduce pressure to give the title compound (150 mg, crude) as a yellow oil. 1H NMR (400 MHz, Chloroform-d (m, 2H), 3.08 - 3.00 (m, 1H), 1.84 - 1.79 (m, 2H). [0740] Step 2: (S)-5-((1,4-Oxazepan-3-yl-5,5-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0741] A mixture of (R)-(1,4-oxazepan-3-yl-5,5-d2)methanol (130 mg, 976 µmol) in THF (10 mL) was degassed and purged with N2 three times. NaH (156 mg, 3.90 mmol, 60% purity) was added at 0°C. The mixture was stirred at 0 °C for 0.5 hr. Then a solution of Intermediate 1 (288 mg, 1.07 mmol) in THF (10 mL) was added to the mixture quickly at 0 °C under N2, and the mixture was stirred at 25 °C for 1.5 hr under N2 atmosphere. The reaction mixture was quenched with saturated NH4Cl (1 mL) and dried over MgSO4. The residue was washed with CH2Cl2 (30 mL), filtered and concentrated to give the title compound (400 mg, crude) as a yellow solid. MS: m/z = 364.9 [M + H]+. [0742] Step 3: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-13,13-d2 [0743] To a solution of (S)-5-((1,4-oxazepan-3-yl-5,5-d2)methoxy)-2,7-dichloro-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (400 mg, 1.1 mmol) in CH2Cl2 (150 mL) were added DIEA (849 mg, 6.57 mmol) and POCl3 (839 mg, 5.48 mmol) at 0 °C. The mixture was stirred at 0 °C for 1 hr. The reaction mixture was quenched with saturated NaHCO3 (100 mL) and extracted with CH2Cl2 (200 mL x 2). The combined organic layers were washed with brine (100 mL), dried
WSGR Docket No. 62619-720.601 over Na2SO4, filtered and concentrated. After purification by silica gel flash chromatography (Eluent of 0 ~ 50% EtOAc in petroleum ether), the title compound (Intermediate 74, 60 mg, yield: 18% for three steps) was obtained as a yellow solid. MS: m/z = 346.8 [M + H]+.1H NMR (400 MHz, Chloroform-d (m, 1H), 4.05 - 3.98 (m, 1H), 3.96 - 3.90 (m, 1H), 3.44 - 3.38 (m, 1H), 2.02 - 1.99 (m, 2H). [0744] Intermediate 76: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene-13,13-d2
[0745] purged with N2 three times, and the mixture was stirred at 105 °C for 16 hr under N2 atmosphere. The reaction mixture was diluted with H2O (10 mL) and extracted with CH2Cl2 (20 mL x 3). The combined organic layers dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 4% MeOH in CH2Cl2), the title compound (Intermediate 76, 125 mg, yield: 48%) was obtained as a yellow solid. MS: m/z = 472.0 [M + H]+.1H NMR (400 MHz, Chloroform-d (dd, J = 13.6, 4.8 Hz, 1H), 4.39 (d, J = 13.2 Hz, 1H), 4.18 (dd, J = 12.4, 3.6 Hz, 1H), 4.07 - 3.94 (m, 2H), 3.73 - 3.65 (m, 1H), 3.39 - 3.26 (m, 2H), 3.25 - 3.12 (m, 2H), 3.04 - 2.93 (m, 1H), 2.25 - 2.10 (m, 4H), 2.02 - 1.89 (m, 4H). 19F NMR (376 MHz, Chloroform-d [0746] Intermediate 77: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene-9,9-d2
WSGR Docket No. 62619-720.601
[0747] Step 1: tert-Butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-3,3-d2)carbamate [0748] To a solution of (R)-3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propanoic acid (5 g, 16.9 mmol) in THF (50 mL) was added LiAlD4 (2.73 g, 59.3 mmol) at 0 °C. The mixture was stirred at 25 °C for 2 hr. The reaction mixture was quenched with Na2SO4 10H2O (15 g) at 0 °C, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 15% EtOAc in petroleum ether), the title compound (3.1 g, yield: 53%) was obtained as a colorless oil. MS: m/z = 184.1 [M + H - 100]+.1H NMR (400 MHz, Chloroform-d (m, 5H), 5.17 (br s, 1H), 4.52 (s, 2H), 3.79 (br s, 1H), 3.67 - 3.60 (m, 2H), 1.44 (s, 9H). [0749] Step 2: tert- Butyl (R)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy-1,1- d2)propanoate [0750] To a solution of tert-butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl-3,3-d2)carbamate (3.1 g, 10.9 mmol) in t-BuOH (30 mL) were added Cs2CO3 (4.28 g, 13.1 mmol) and tert-butyl acrylate (26.5 g, 207 mmol). The mixture was stirred at 25 °C for 16 hr. The mixture was diluted with H2O (500 mL) and extracted with EtOAc (500 mL × 2). The combined organic layers were washed with brine (500 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 4% EtOAc in petroleum ether), the title compound (3.4 g, yield: 69%) was obtained as a colorless oil. MS: m/z = 434.3 [M + Na]+.1H NMR (400 MHz, Chloroform-d 1H), 3.69 - 3.64 (m, 2H), 3.59 - 3.48 (m, 2H), 2.45 (t, J = 5.6 Hz, 6.4H), 1.45 (s, 9H), 1.43 (s, 9H). [0751] Step 3: (R)-3-(2-Amino-3-(benzyloxy)propoxy-1,1-d2)propanoic acid [0752] To a solution of tert-butyl (R)-3-(3-(benzyloxy)-2-((tert-butoxycarbonyl)amino)propoxy- 1,1-d2)propanoate (3.1 g, 7.53 mmol) in CH2Cl2 (35 mL) was added TFA (35 mL). The mixture
WSGR Docket No. 62619-720.601 was stirred at 25 °C for 2 hr. The reaction mixture was concentrated under reduced pressure. The title compound (2.78 g, TFA salt) was obtained as a yellow oil. MS: m/z = 256.2 [M + H]+. [0753] Step 4: (R)-3-((Benzyloxy)methyl)-1,4-oxazepan-5-one-2,2-d2 [0754] To a solution of (R)-3-(2-amino-3-(benzyloxy)propoxy-1,1-d2)propanoic acid (2.78 g, 10.9 mmol) in CH2Cl2 (300 mL) were added HATU (4.97 g, 13.1 mmol) and TEA (5.51 g, 54.4 mmol). The mixture was stirred at 25 °C for 3 hr. The mixture was diluted with H2O (500 mL) and extracted with CH2Cl2 (500 mL × 2). The combined organic layers were washed with brine (500 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 46% EtOAc in petroleum ether), the title compound (2.58 g, yield: 97%) was obtained as a colorless oil. MS: m/z = 238.1 [M + H]+.1H NMR (400 MHz, Chloroform-d 3.80 - 3.76 (m, 1H), 3.69 - 3.61 (m, 1H), 3.53 (dd, J = 9.6, 4.8 Hz, 1H), 3.39 (dd, J = 9.6, 7.6 Hz, 1H), 2.87 - 2.79 (m, 1H), 2.53 (dd, J = 15.6, 5.6 Hz, 1H). [0755] Step 5: (R)-3-(Hydroxymethyl)-1,4-oxazepan-5-one-2,2-d2 [0756] To a solution of (R)-3-((benzyloxy)methyl)-1,4-oxazepan-5-one-2,2-d2 (2.5 g, 10.5 mmol) in CH2Cl2 (60 mL) was added dropwise a solution of BCl3 (1 M in CH2Cl2, 10.5 mL) at 0 - 5 °C under N2 over 10 min. The mixture was stirred at 25 °C for 1 hr. The reaction mixture quenched with MeOH (100 mL) and concentrated under reduced pressure. The resulting residue neutralized with NH3 H2O (pH~7-8). The mixture was concentrated. After purification by silica gel flash chromatography (Eluent of MeOH in CH2Cl2 = 0 to 14%), the title compund (630 mg, yield: 41%) was obtained as an off white solid. 1H NMR (400 MHz, Chloroform-d 3.91 (m, 1H), 3.77 - 3.64 (m, 3H), 3.63 - 3.52 (m, 2H), 2.91 - 2.84 (m, 1H), 2.52 (dd, J = 15.6, 5.2 Hz, 1H). [0757] Step 6: (R)-(1,4-Oxazepan-3-yl-2,2-d2)methanol [0758] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-5-one-2,2-d2 (630 mg, 4.28 mmol) in THF (10 mL) was added LiAlH4 (325 mg, 8.56 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 hr. The reaction mixture was quenched with H2O (0.33 mL), 15% NaOH (0.33 mL), and H2O (1 mL). Then the mixture was filtered and concentrated under reduced pressure, the title compound (370 mg, yield: 65%) was obtained as a white solid. 1H NMR (400 MHz, Chloroform- d J = 10.4, 4.4 Hz, 1H), 3.32 (dd, J = 10.4, 8.0 Hz, 1H), 3.15 – 3.05 (m, 1H), 3.02 (dd, J = 8.0, 4.4 Hz, 1H), 2.89 - 2.81 (m, 1H), 2.13 (br s, 2H), 1.89 - 1.80 (m, 2H).
WSGR Docket No. 62619-720.601 [0759] Step 7: (S)-5-((1,4-Oxazepan-3-yl-2,2-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0760] To a solution of (R)-(1,4-oxazepan-3-yl-2,2-d2)methanol (300 mg, 2.25 mmol) in THF (20 mL) was added NaH (360 mg, 9.01 mmol, 60% purity) at 0 °C. The mixture was stirred at 0 °C for 30 min, and a solution of Intermediate 1 (605 mg, 2.25 mmol) in THF (20 mL) was added dropwise at 0 °C. The resulting mixture was stirred at 25 °C for 2 hr. The reaction mixture was quenched with saturated NH4Cl (2 mL) and dried over MgSO4. The residue was washed with CH2Cl2 (100 mL), filtered and concentrated under reduced pressure to give the title compound (822 mg, crude) as a yellow solid. MS: m/z = 365.1 [M + H]+. [0761] Step 8: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-9,9-d2 [0762] To a solution of (S)-5-((1,4-oxazepan-3-yl-2,2-d2)methoxy)-2,7-dichloro-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (780 mg, 2.14 mmol) in CH2Cl2 (130 mL) was added DIEA (2.23 mL, 12.8 mmol) and POCl3 (1 mL, 10.7 mmol). The mixture was stirred at 0 °C for 1 hr. The reaction mixture was diluted with saturated NaHCO3 (150 mL) and extracted with CH2Cl2 (150 mL x 2). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of 0 ~ 7% MeOH in CH2Cl2), the title compound (Intermediate 77, 120 mg, yield: 12%) was obtained as a yellow solid. MS: m/z = 347.0 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 13.6, 4.8 Hz, 1H), 4.44 (dd, J = 13.6, 0.8 Hz, 1H), 4.09 (d, J = 4.4 Hz, 1H), 4.05 - 3.97 (m, 1H), 3.46 - 3.29 (m, 2H), 2.26 - 2.15 (m, 1H), 2.06 - 1.96 (m, 1H). [0763] Intermediate 78: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene-9,9-d2
[0764] Intermediate 78 was prepared in a similar manner to Intermediate 76. MS: m/z = 472.1 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 13.2, 4.4 Hz, 1H), 4.40 (d, J = 13.2 Hz, 1H), 4.06 - 3.97 (m, 2H), 3.38 - 3.20 (m, 4H), 3.18 – 3.11 (m, 1H), 3.02 - 2.92 (m, 1H), 2.30 - 2.10 (m, 4H), 2.02 - 1.80 (m, 4H).
WSGR Docket No. 62619-720.601 [0765] Intermediate 79: (S)-2,5-Dichloro-4,12,12-trifluoro-8a,9,12,13-tetrahydro-8H,11H-7,10- dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0766] Step 1: tert-Butyl (R)-3-((benzyloxy)methyl)-6,6-difluoro-1,4-oxazepane-4-carboxylate [0767] To a solution of tert-butyl (R)-3-((benzyloxy)methyl)-6-oxo-1,4-oxazepane-4-carboxylate (2.5 g, 7.45 mmol) in CH2Cl2 (30 mL) was added BAST (9.80 mL, 44.7 mmol). The mixture was stirred at 40 °C for 3 hr. The reaction mixture was diluted with H2O (10 mL) and extracted with CH2Cl2 (20 mL x 2). The combined organic layers dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 7% EtOAc in petroleum ether), the title compound (2.26 g, yield: 80%) was obtained as a colorless oil. MS: m/z = 258.0 [M – 100 + H]+.1H NMR (400 MHz, Chloroform-d 4.44 (m, 3H), 4.34 - 4.22 (m, 1H), 4.02 - 3.93 (m, 1H), 3.91 - 3.68 (m, 3H), 3.65 - 3.45 (m, 3H), 1.50 - 1.44 (m, 9H).19F NMR (376 MHz, Chloroform-d 107.85, -108.04. [0768] Step 2: tert -Butyl (R)-6,6-difluoro-3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate [0769] A mixture of tert-butyl (R)-3-((benzyloxy)methyl)-6,6-difluoro-1,4-oxazepane-4- 2 MeOH (30 mL) was degassed and purged with H2 for three times, and the mixture was stirred at 25 °C for 3 hr under H2 atmosphere. The reaction mixture was filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (Eluent of petroleum ether in EtOAc = 5/1), the title compound (1.34 g, yield: 79%) was obtained as a colorless oil. 1H NMR (400 MHz, Chloroform-d 3.68 (m, 4H), 3.67 - 3.47 (m, 1H), 1.48 (s, 9H).19F NMR (376 MHz, Chloroform-d 105.31, -106.46, -107.14. [0770] Step 3: (R)-(6,6-Difluoro-1,4-oxazepan-3-yl)methanol [0771] A mixture of tert-butyl (R)-6,6-difluoro-3-(hydroxymethyl)-1,4-oxazepane-4-carboxylate (1.34 g, 5.01 mmol) in HCl/EtOAc (2 M, 10.3 mL) was stirred at 25 °C for 0.5 hr. The reaction
WSGR Docket No. 62619-720.601 mixture was concentrated under reduced pressure. The crude product was dissolved in CH3CN : MeOH (5 mL : 1 mL). The pH of the mixture was adjusted to about 6 with K2CO3. The mixture was filtered and concentrated under reduced pressure. After trituration with CH2Cl2 : MeOH = 20 : 1 at 25 °C for 5 min, the title compound (640 mg, yield: 76%) was obtained as an off-white solid. 1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 3.32 - 3.25 (m, 2H), 3.24 - 3.16 (m, 2H), 3.05 - 2.90 (m, 1H), 2.88 - 2.77 (m, 1H), 2.60 - 2.52 (m, 1H).19F NMR (376 MHz, DMSO-d6 [0772] Step 4: (S)-2,7-Dichloro-5-((6,6-difluoro-1,4-oxazepan-3-yl)methoxy)-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0773] A mixture of NaH (612 mg, 15.3 mmol, 60% purity) in THF (10 mL) was degassed and purged with N2 three times, and a solution of (R)-(6,6-difluoro-1,4-oxazepan-3-yl)methanol (640 mg, 3.83 mmol) in THF (10 mL) was added dropwise at 25 °C under N2 atmosphere. The mixture was stirred at 25 °C for 1 hr. Then a solution of Intermediate 1 (1.13 g, 4.21 mmol) in THF (10 mL) was added quickly at 25 °C under N2 atmosphere. The mixture was stirred at 25 °C for 1 hr under N2 atmosphere. The mixture was quenched with saturated NH4Cl (2 mL), dried over MgSO4, filtered, washed with CH2Cl2 (30 mL), and concentrated under reduced pressure. The title compound (1.1 g crude) was obtained as an off-white solid. MS: m/z = 398.7 [M + H]+. [0774] Step 5: (S)-2,5-Dichloro-4,12,12-trifluoro-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0775] To a solution of (S)-2,7-dichloro-5-((6,6-difluoro-1,4-oxazepan-3-yl)methoxy)-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one (1.1 g, 2.76 mmol) in CH2Cl2 (300 mL) were added DIEA (2.40 mL, 13.7 mmol) and POCl3 (1.54 mL, 16.5 mmol) at 0 °C. The mixture was stirred at 0 °C for 4 hr. The reaction mixture diluted with saturated NaHCO3 (60 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 30% EtOAc in petroleum ether), the title compound (Intermediate 79, 260 mg, yield: 18% for two steps) was obtained as an off-white solid. MS: m/z = 380.9 [M + H]+.1H NMR (400 MHz, Chloroform-d 6.02 (t, J = 13.6 Hz, 1H), 4.73 (dd, J = 14.0, 3.6 Hz, 1H), 4.48 (d, J = 14.0 Hz, 1H), 4.37 (d, J = 12.8 Hz, 1H), 4.23 - 4.07 (m, 2H), 3.81 (dd, J = 12.4, 10.4 Hz, 1H), 3.63 - 3.46 (m, 2H).19F NMR (376 MHz, Chloroform-d [0776] Intermediate 80: (S)-5-Chloro-4,12,12-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601
[0777] Intermediate 80 was prepared in a similar manner to Intermediate 76. MS: m/z = 506.1 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 14.4 Hz, 1H), 5.37 - 5.15 (m, 1H), 4.68 (dd, J = 13.6, 4.0 Hz, 1H), 4.42 (d, J = 13.6 Hz, 1H), 4.38 - 4.29 (m, 1H), 4.19 - 4.05 (m, 2H), 3.85 - 3.74 (m, 1H), 3.56 - 3.43 (m, 2H), 3.31 - 3.09 (m, 3H), 3.04 - 2.90 (m, 1H), 2.35 - 2.04 (m, 3H), 2.00 - 1.83 (m, 3H).19F NMR (376 MHz, Chloroform-d 139.84, -173.17. [0778] Intermediate 81 & 82: tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,6,7- tetrahydro-1H-azepine-1-carboxylate & tert-butyl (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)- 2,3,4,7-tetrahydro-1H-azepine-1-carboxylate
[0779] Step 1: tert-Butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-hydroxyazepane-1- carboxylate [0780] To a solution of tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-oxoazepane-1- carboxylate (6.7 g, 13.9 mmol) in MeOH (140 mL) was added NaBH4 (810 mg, 21.4 mmol) at 0 °C under N2 and the mixture was stirred at 0 °C for 1 h under N2. The reaction mixture was quenched with HCl (1M in H2O, 30 mL) at 0 °C and extracted with EtOAc (100 mL x 3). The combined organic layers were washed with brine (60 ml), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 50% of EtOAc in petroleum ether) to give the title compound (5.7 g, 85% yield) as a colorless oil. MS: m/z = 506.3 [M + Na]+.1H NMR (400 MHz, Chloroform-d 4H), 7.48 - 7.28 (m, 6H), 4.28 - 3.95 (m, 2H), 3.87 - 3.48 (m, 4H), 3.28 - 2.78 (m, 1H), 2.03 - 1.93 (m, 2H), 1.69 - 1.52 (m, 4H), 1.48 - 1.37 (m, 9H), 1.04 (s, 9H).
WSGR Docket No. 62619-720.601 [0781] Step 2: tert-Butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-iodoazepane-1- carboxylate [0782] A mixture of tert-butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-hydroxyazepane-1- carboxylate (5.7 g, 11.8 mmol), imidazole (1.6 g, 23.6 mmol), PPh3 (4.64 g, 17.7 mmol) and I2 (4.49 g, 17.7 mmol) in toluene (80 mL) was stirred at 110 °C for 16 h under N2. The reaction mixture was quenched with sat. Na2SO3 aq. (50 mL) and extracted with EtOAc (40 mL x 3). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of EtOAc in petroleum ether) to give the title compound (5.1 g, 73% yield) as a colorless oil. MS: m/z = 616.2 [M + Na]+. 1H NMR (400 MHz, Chloroform-d 4.27 - 3.96 (m, 2H), 3.86 - 3.51 (m, 3H), 3.37 - 2.77 (m, 1H), 2.74 - 2.32 (m, 2H), 2.31 - 2.06 (m, 2H), 2.05 - 1.78 (m, 2H), 1.52 - 1.35 (m, 9H), 1.13 - 1.01 (m, 9H). [0783] Step 3: tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,6,7-tetrahydro-1H- azepine-1-carboxylate & tert-butyl (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro- 1H-azepine-1-carboxylate [0784] To a solution of tert-butyl (2S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-5-iodoazepane-1- carboxylate (5.1 g, 8.59 mmol) in xylene (100 mL) was added DBU (25.5 mL, 169 mmol). The mixture was stirred at 140 °C for 16 h. The mixture was diluted with EtOAc (100 mL) and washed with brine (100 mL × 3). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of EtOAc in petroleum ether) to give a mixture of the title compounds (3.5 g) as an off-white oil. The mixture was purified by SFC (column: (s, s) WHELK- 2-i-PrOH (0.1%NH3·H2O)]; B%: 15%, isocratic elution mode) to give the title compound (Intermediate 81, 2.5 g, 61% yield, peak 1) as a colorless oil and the title compound (Intermediate 82, 700 mg, 17% yield, peak 2) as a colorless oil. Spectra for Intermediate 81: MS: m/z = 488.2 [M + Na]+.1H NMR (400 MHz, Chloroform-d (m, 4H), 7.49 - 7.31 (m, 6H), 5.75 - 5.40 (m, 2H), 4.36 - 4.11 (m, 1H), 3.92 - 3.48 (m, 4H), 2.97 - 2.75 (m, 1H), 2.63 - 2.41 (m, 1H), 2.32 - 2.10 (m, 2H), 1.51 - 1.35 (m, 9H), 1.06 (s, 9H). Spectra for Intermediate 82: MS: m/z = 488.2 [M + Na]+.1H NMR (400 MHz, Chloroform-d (m, 4H), 7.53 - 7.28 (m, 6H), 5.76 - 5.55 (m, 2H), 4.48 - 4.20 (m, 1H), 4.20 - 3.95 (m, 1H), 3.76 - 3.55 (m, 3H), 2.38 - 2.11 (m, 2H), 2.05 - 1.85 (m, 2H), 1.43 ( d, J = 12.0 Hz, 9H), 1.05 (s, 9H). [0785] Intermediate 83: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601
[0786] Step 4: (S)-2-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepine [0787] To a solution of Intermediate 82 (700 mg, 1.50 mmol) in CH2Cl2 (6 mL) was added TFA (2.33 mL, 31.4 mmol). The mixture was stirred at 25 °C for 2 h. The reaction mixture concentrated under reduced pressure. Then the mixture was diluted with ACN : MeOH=10 : 1 (5 mL), stirred with K2CO3 for about 5 min, filtered and concentrated under reduced pressure to give the title compound (580 mg, crude) as a yellow oil, which was used in next step without purification. MS: m/z = 366.2 [M + H]+.1H NMR (400 MHz, Chloroform-d 7.53 - 7.40 (m, 6H), 5.71 (s, 2H), 3.64 - 3.44 (m, 3H), 3.38 - 3.33 (m, 2H), 3.10 (s, 1H), 2.36 - 2.07 (m, 2H), 1.92 - 1.43 (m, 2H), 1.02 (s, 9H). [0788] Step 2: (S)-4-(7-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepin-1-yl)- 2,5,7-trichloro-8-fluoropyrido[4,3-d]pyrimidine [0789] To a solution of 2,4,5,7-tetrachloro-8-fluoropyrido[4,3-d]pyrimidine (455 mg, 1.59 mmol) in MeCN (8 mL) was added DIPEA (0.83 mL, 4.70 mmol) and (S)-2-(((tert- butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H N2, and the mixture was stirred at 25 °C for 16 h. The reaction mixture was partitioned between EtOAc (80 mL) and H2O (50 mL). The organic phase was separated, washed with brine (30 mL × 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of EtOAc in petroleum ether) to give the title compound (600 mg, 61% yield) as a yellow oil. MS: m/z = 616.9 [M + H]+.1H NMR (400 MHz, Chloroform-d 1H), 4.59 - 4.16 (m, 2H), 4.03 - 3.67 (m, 2H), 2.25 - 1.89 (m, 3H), 2.01 - 1.89 (m, 1H), 1.52 - 1.50 (m, 1H), 0.93 (s, 9H).19F NMR (376 MHz, Chloroform-d [0790] Step 3: 4-((S)-2-(((tert-Butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepin-1-yl)- 5,7-dichloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)pyrido[4,3-d]pyrimidine
WSGR Docket No. 62619-720.601 [0791] A solution of (S)-4-(2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H-azepin- 1-yl)-2,5,7-trichloro-8-fluoropyrido[4,3-d mmol) and ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol (310 mg, 1.95 mmol) in 1,4-dioxane (12 mL) was stirred at 115 °C for 16 h. The mixture was concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (550 mg, 76% yield) as a yellow solid. MS: m/z = 738.3 [M + 1]+. [0792] Step 4: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0793] To a solution of 4-((S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,4,7-tetrahydro-1H- azepin-1-yl)-5,7-dichloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- in THF, 2.98 mL) and stirred at 25 °C for 16 h. The reaction mixture was partitioned between EtOAc (150 mL) and H2O (90 mL). The organic phase was separated, washed with brine (60 mL × 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (Intermediate 83, 197 mg, 57% yield) as a yellow solid. MS: m/z = 464.1 [M + 1]+. [0794] Intermediate 84: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
[0795] Step 1: tert-Butyl (S)-2-(hydroxymethyl)-2,3,6,7-tetrahydro-1H-azepine-1-carboxylate [0796] A solution of Intermediate 81 (800 mg, 1.72 mmol) and TBAF (1 M in THF, 1.89 mL) in THF (16 mL) was stirred at 25 °C for 16 h. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (30 mL x 3). The combined organic phases were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 17% of EtOAc
WSGR Docket No. 62619-720.601 in petroleum ether) to give the title compound (369 mg, 1.62 mmol) as a colorless oil. 1H NMR (400 MHz, Dimethylsulfoxide-d6 3.64 - 3.47 (m, 1H), 3.45 - 3.34 (m, 3H), 2.57 - 2.54 (m, 1H), 2.41 - 2.30 (m, 1H), 2.24 - 2.08 (m, 2H), 1.39 (s, 9H). [0797] Step 2: (S)-(2,3,6,7-Tetrahydro-1H-azepin-2-yl)methanol [0798] A mixture of tert-butyl (S)-2-(hydroxymethyl)-2,3,6,7-tetrahydro-1H-azepine-1-carboxylate (369 mg, 1.62 mmol) in HCl (2M in EtOAc, 3 mL) was stirred at 25 °C for 0.5 h. The reaction mixture was concentrated to dryness. The residue was dissolved in MeCN (5mL) and MeOH (1mL), and then K2CO3 was added to adjust the pH to around 8. The mixture was filtered and concentrated under reduced pressure to give the title compound (220 mg, crude) as an off-white solid, which was used in the next step without further purification. MS: m/z = 127.9 [M+ H]+. [0799] Step 3: (S)-2,7-Dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H)-one [0800] To a solution of (S)-(2,3,6,7-tetrahydro-1H-azepin-2-yl)methanol (200 mg, 1.57 mmol) in THF (5 mL) was added NaH (252 mg, 6.29 mmol, 60% purity) at 0 °C, and the mixture was stirred at 0 °C for 0.5 h under N2. Then a solution of Intermediate 1 (422 mg, 1.57 mmol) in THF (2 ml) was added at 0 °C, and the resulting mixture was stirred at 20 °C for 0.5 h. The reaction mixture was quenched with sat. NaHCO3 aq. (0.2 mL), diluted with CH2Cl2 (20 ml), dried over MgSO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 45% of EtOAc in petroleum ether) to give the title compound (230 mg, 41% yield over two steps) as an off-white solid. MS: m/z = 358.9 [M + H]+. [0801] Step 4: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0802] To a solution of (S)-2,7-dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H 2Cl2 (70 mL) were added 3 2. The mixture was stirred at 0 °C for 1 h. The reaction mixture was quenched with sat. NaHCO3 aq. (100 mL) and extracted with CH2Cl2 (40 mL x 2). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 50% of EtOAc in petroleum ether) to give the title compound (Intermediate 84, 97 mg, 44% yield) as an off-white solid. MS: m/z = 340.9 [M + H]+.
WSGR Docket No. 62619-720.601 [0803] Intermediate 85: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0804] 2R,7aS)-2-fluorotetrahydro-1H- (4 mL) was degassed and purged with N2 three times, and the mixture was stirred at 115 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of MeOH in CH2Cl2) to give the title compound (Intermediate 85, 65 mg, 49% yield) as a yellow solid. MS: m/z = 464.2 [M + H]+. [0805] Intermediate 86 & 87: (8S,8aS)-5-Chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene & (8R,8aS)-5-chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0806] Step 1: tert-Butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate [0807] To a solution of (S)-1-(tert-butoxycarbonyl)azepane-2-carboxylic acid (2 g, 8.22 mmol) in CH2Cl2 (14 mL) were added N,O-dimethylhydroxylamine hydrochloride (882 mg, 9.04 mmol),
WSGR Docket No. 62619-720.601 DIPEA (2.12 g, 16.4 mmol) and HATU (3.44 g, 9.04 mmol), and the mixture was stirred at 0 °C for 2 h. The reaction mixture was quenched with H2O (40 mL) and extracted with EtOAc (40 mL x 2). The combined organic layers were washed with brine (40 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 25% of EtOAc in Petroleum ether) to give the title compound (2.10 g, 89% yield) as a yellow oil. 1H NMR (400 MHz, Chloroform-d (m, 1H), 3.86 - 3.71 (m, 3H), 3.23 - 3.11 (m, 4H), 2.34 - 2.19 (m, 1H), 1.96 - 1.85 (m, 1H), 1.84 - 1.49 (m, 4H), 1.46 - 1.42 (m, 9H), 1.35 - 1.23 (m, 2H). [0808] Step 2: tert-Butyl (S)-2-propionylazepane-1-carboxylate [0809] A mixture of of tert-butyl (S)-2-(methoxy(methyl)carbamoyl)azepane-1-carboxylate (2.10 g, 7.33 mmol) in THF (20 mL) was degassed and purged with N2 three times. CH3CH2MgBr (3 M, 7.30 mL) was added dropwise at -78 °C under N2 atmosphere. The reaction was slowly warmed up to 20 °C and stirred for 1 h. The reaction mixture was quenched with sat. aqueous NH4Cl (40 mL) at -78 °C and extracted with EtOAc (40 mL x 2). The combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 25% of EtOAc in Petroleum ether) to give the title compound (1.54 g, 76% yield) as a yellow oil. 1H NMR (400 MHz, Chloroform-d (m, 2H), 2.26 - 2.06 (m, 1H), 1.91 - 1.72 (m, 3H), 1.65 - 1.51 (m, 2H), 1.48 - 1.40 (m, 9H), 1.37 - 1.19 (m, 2H), 1.08 - 0.99 (m, 3H). [0810] Step 3: (S)-1-(Azepan-2-yl)propan-1-one [0811] To a solution of tert-butyl (S)-2-propionylazepane-1-carboxylate (1.54 g, 6.03 mmol) in CH2Cl2 (20 mL) was added HCl/EtOAc (4 M, 10 mL), and the mixture was stirred at 20 °C for 1 h. The reaction mixture was concentrated under reduced pressure to give the title compound (1.15 g, HCl salt) as a white solid, which was used in the next step without further purification. 1H NMR (400 MHz, Chloroform-d J = 7.2 Hz, 2H), 2.35 - 2.21 (m, 1H), 2.08 - 1.71 (m, 7H), 1.68 - 1.57 (m, 1H), 1.17 - 1.06 (m, 3H). [0812] Step 4: 1-((S)-Azepan-2-yl)propan-1-ol [0813] To a solution of (S)-1-(azepan-2-yl)propan-1-one (1.15 g, 6.00 mmol, HCl salt) in THF (10 mL) was added dropwise LiAlH4 (2.5 M in n-hexane, 9.60 mL) at 0 °C under N2 atmosphere. The reaction was stirred at 20 °C for 1 h under N2 atmosphere. Na2SO4·10H2O (10 g) was slowly added to the mixture at 20 °C, then the mixture was stirred for 30 min. The mixture was filtered and concentrated under reduced pressure to give the title compound (880 mg, crude) as a yellow oil,
WSGR Docket No. 62619-720.601 which was used in the next step without further purification. 1H NMR (400 MHz, Chloroform-d 3.25 - 3.36 (m, 1H), 3.08 - 2.92 (m, 1H), 2.79 - 2.63 (m, 2H), 2.39 - 2.12 (m, 2H), 1.76 - 1.61 (m, 4H), 1.52 - 1.38 (m, 4H), 1.34 - 1.22 (m, 2H), 0.99 - 0.93 (m, 3H). [0814] Step 5: 5-(1-((S)-Azepan-2-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0815] To a solution of 1-((S)-azepan-2-yl)propan-1-ol (300 mg, 1.91 mmol) in THF (18 mL) was added NaH (305 mg, 7.63 mmol, 60% purity) at 0 °C, and the mixture was stirred at 30 °C for 1 h under N2 atmosphere. Then a solution of Intermediate 1 (563 mg, 2.10 mmol) in THF (2 mL) was added to the mixture, and the mixture was stirred at 30 °C for 2 h. The reaction mixture was quenched with sat. NH4Cl aq. (5.0 mL) at 20 °C, dried over anhydrous MgSO4, filtered and concentrated under reduced pressure to give the title compound (720 mg, crude) as a yellow solid. MS: m/z = 388.9 [M + H]+ [0816] Step 6: (8aS)-2,5-Dichloro-8-ethyl-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [0817] A mixture of 5-(1-((S)-azepan-2-yl)propoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one (716 mg, 1.84 mmol) and DIPEA (1.43 g, 11.0 mmol) in CH2Cl2 (265 mL) was degassed and purged with N2 three times. POCl3 (1.41 g, 9.20 mmol) was added dropwise. The reaction was stirred at 0 °C for 1 h under N2 atmosphere. The reaction mixture was quenched with sat. aq. NaHCO3 (300 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 22% of EtOAc in Petroleum ether) to give the title compound (230 mg, 31% yield over 4 steps) as a yellow solid. MS: m/z = 371.0 [M + H]+ .1H NMR (400 MHz, Chloroform-d (dd, J = 5.2, 8.8 Hz, 1H), 3.77 (dd, J = 4.4, 10.8 Hz, 1H), 3.20 - 3.02 (m, 1H), 2.02 - 1.91 (m, 2H), 1.76 - 1.68 (m, 4H), 1.58 (br s, 4H), 1.14 (t, J = 7.2 Hz, 3H).19F NMR (376 MHz, Chloroform-d -140.36. [0818] Step 7: (8S,8aS)-5-Chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene & (8R,8aS)-5-chloro-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0819] A mixture of (8aS)-2,5-dichloro-8-ethyl-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab R,7aS)-2-fluorotetrahydro-1H-
WSGR Docket No. 62619-720.601 pyrrolizin-7a(5H dioxane (5 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 115 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was separated by SFC (column: DAICEL CHIRALCEL AD (250 mm x 30 2 - EtOH (0.1% NH3H2O)]; B%: 30%, isocratic elution mode) to give the title compound (Intermediate 86, 21 mg, 7% yield, SFC Peak 1: 1.494min) as a yellow solid and the title compound (Intermediate 87, 90 mg, 29% yield, SFC Peak 2: 1.623min) as a yellow solid. MS: m/z = 494.2 [M + H]+. [0820] Intermediate 88: (S)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
[0821] Step 1: tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-4,4-difluoroazepane-1- carboxylate [0822] To a solution of tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-4-oxoazepane-1- carboxylate (2 g, 4.15 mmol) in CH2Cl2 (30 mL) was added BAST (5.5 mL, 24.9 mmol). The mixture was stirred at 40 °C for 16 h. The reaction mixture was quenched with sat. NaHCO3 aq. (30 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (40 mL), dried over anhydrous Na2SO4, filtered and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of EtOAc in petroleum ether) to give the title compound (753 mg, 36% yield) as a colourless oil. MS: m/z = 526.3 [M + H]+. 1H NMR (400 MHz, Chloroform-d 6H), 4.36 - 3.88 (m, 2H), 3.87 - 3.77 (m, 1H), 3.70 - 3.57 (m, 2H), 3.18 - 2.99 (m, 1H), 2.52 - 2.34 (m, 2H), 2.23 - 2.11 (m, 1H), 1.94 - 1.72 (m, 2H), 1.68 - 1.63 (m, 1H), 1.47 - 1.36 (m, 9H), 1.05 (s, 9H).19F NMR (376 MHz, Chloroform-d 104.15, -104.70.
WSGR Docket No. 62619-720.601 [0823] Step 2: tert-Butyl (S)-4,4-difluoro-2-(hydroxymethyl)azepane-1-carboxylate [0824] To a solution of tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-4,4-difluoroazepane- 1-carboxylate (753 mg, 1.49 mmol) in THF (10 mL) was added TBAF (1 M in THF, 2.24 mL). The mixture was stirred at 25 °C for 12 h. The reaction mixture was quenched with water (20 mL) at 20 °C and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 33% of EtOAc in petroleum ether) to give the title compound (330 mg, 83% yield) as a colourless oil. MS: m/z = 280.2 [M + H]+.1H NMR (400 MHz, Chloroform-d 3.05 - 2.92 (m, 1H), 2.44 - 2.10 (m, 3H), 1.79 - 1.57 (m, 4H), 1.47 (s, 9H).19F NMR (376 MHz, Chloroform-d [0825] Step 3: (S)-(4,4-Difluoroazepan-2-yl)methanol [0826] A solution of tert-butyl (2S)-4,4-difluoro-2-(hydroxymethyl)azepane-1-carboxylate (330 mg, 1.24 mmol) in HCl (2M in 1,4-dioxane, 4 mL) was stirred at 25 °C for 10 min. The reaction mixture was concentrated. The residue was dissolved in MeCN (5mL) and MeOH (1mL), and then K2CO3 was added to adjust the pH value to around 8. The mixture was filtered and concentrated under reduced pressure to give the title compound (205 mg, crude) as a yellow solid, which was used in the next step without further purification. 1H NMR (400 MHz, Chloroform-d (m, 1H), 3.23 - 3.06 (m, 2H), 2.95 - 2.80 (m, 2H), 2.74 (s, 1H), 2.28 - 2.08 (m, 3H), 1.94 - 1.67 (m, 3H).19F NMR (376 MHz, Chloroform-d [0827] Step 4: (S)-2,7-Dichloro-5-((4,4-difluoroazepan-2-yl)methoxy)-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one [0828] To a mixture of (S)-(4,4-difluoroazepan-2-yl)methanol (200 mg, 1.21 mmol) in THF (6 mL) was added NaH (194 mg, 4.84 mmol, 60% purity) at 0 °C under N2, and the mixture was stirred at 0 °C under N2 for 0.5 h. Then, a solution of Intermediate 1 (325 mg, 1.21 mmol) in THF (2 mL) was added to the mixture, and the mixture was stirred at 25 °C for 2 h under N2 atmosphere. The reaction mixture was quenched with sat. NaHCO3 aq. (2 mL) at 25 °C, dried over anhydrous MgSO4, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 15% of MeOH in CH2Cl2) to give the title compound (360 mg, 75% yield) as a yellow oil. MS: m/z = 396.9 [M + H]+. [0829] Step 5: (S)-2,5-Dichloro-4,10,10-trifluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
WSGR Docket No. 62619-720.601 [0830] To a solution of (S)-2,7-dichloro-5-((4,4-difluoroazepan-2-yl)methoxy)-8-fluoropyrido[4,3- d]pyrimidin-4(3H 2Cl2 5.44 mmol) and POCl3 2, and the mixture was stirred at 25 °C under N2 for 2 h. The reaction mixture was quenched with sat. aq.NaHCO3 (20 mL) and extracted with CH2Cl2 (60 mL x 3). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 2% of MeOH in CH2Cl2) to give the title compound (140 mg, 34% yield) as a yellow oil. MS: m/z = 378.9 [M + H]+. [0831] Step 6: (S)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [0832] A solution of (S)-2,5-dichloro-4,10,10-trifluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H (5 mL) was stirred at 110 °C under N2 for 16 h. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of MeOH in CH2Cl2) to give the title compound (Intermediate 88, 127 mg, 68% yield) as a yellow solid. MS: m/z = 502.2 [M + H]+. [0833] Intermediate 89: 6-Fluoro-5-(fluoromethoxy)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-ol
[0834] Step 1 (S)-7-(Hydroxymethyl)azepan-4-one O-methyl oxime
WSGR Docket No. 62619-720.601 [0835] Pyridine (55 mL, 681 mmol) was added dropwise to a solution of 6-methoxy-3,4- dihydronaphthalen-1(2H)-one (80 g, 454 mmol) and O-methylhydroxylamine hydrochloride (45.5 g, 544.8 mmol) in EtOH (1200 mL) at 20 °C. The mixture was stirred at 20 °C for 16 h. The mixture was concentrated under reduced pressure. The residue was diluted with H2O (500 mL) and extracted with EtOAc (500 mL x 3). The combined organic layers were washed with brine (500 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (95.7 g, 98% purity) as a brown oil, which was used in the next step without further purification. MS: m/z = 206.0 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 8.8 Hz, 1H), 6.75 (dd, J = 2.8, 8.8 Hz, 1H), 6.64 (d, J = 2.4 Hz, 1H), 3.96 (s, 3H), 3.81 (s, 3H), 2.71 (t, J = 6.4 Hz, 4H), 1.90 - 1.75 (m, 2H). [0836] Step 2: 8-Bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one O-methyl oxime [0837] To a solution of 6-methoxy-3,4-dihydronaphthalen-1(2H)-one O-methyl oxime (13.6 g, 66.3 mmol) in AcOH (500 mL) was added NBS (10.6 g, 59.6 mmol) and Pd(OAc)2 (1.49 g, 6.6 mmol). The mixture was stirred at 90 °C for 1 h under N2. The reaction mixture was concentrated under reduced pressure, diluted with H2O (100 mL), and extracted with EtOAc (100 mL x 3). The combined organic layers were washed with brine (200 mL x 2), dried over Na2SO4, filtered and concentrated under reduced pressure to give the title compound (20 g, crude) as a black solid, which was used in the next step without further purification. [0838] Step 3: 8-Bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one [0839] To a solution of 8-bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one O-methyl oxime (16 g, 56.31 mmol) in 1,4-dioxane (160 mL) was added HCl (6 M, 160 mL), and the mixture was stirred at 90 °C for 2 h. The reaction mixture was quenched with sat. Na2CO3 aq. (300 mL) at 0 °C and extracted with EtOAc (200 mL x 3). The combined organic layers were washed with brine (300 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 7% of EtOAc in Petroleum ether) to give the title compound (10 g, 36% yield for 3 step) as a white solid. MS: m/z = 255.0 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.4 Hz, 1H), 6.94 (d, J = 2.4 Hz, 1H), 3.83 (s, 3H), 2.95 - 2.92 (m, 2H), 2.57 - 2.54 (m, 2H), 1.99 - 1.93(m, 2H). [0840] Step 4: 8-Bromo-2-fluoro-6-methoxy-3,4-dihydronaphthalen-1(2H)-one [0841] To a solution of 8-bromo-6-methoxy-3,4-dihydronaphthalen-1(2H)-one (48 g, 188 mmol) in MeOH (480 mL) were added H2SO4 (3.36 mL, 63.03 mmol) and SelectFluor (80 g, 225.8 mmol). The mixture was stirred at 50 °C for 5 h. The mixture was concentrated, diluted with H2O (400 mL), and extracted with EtOAc (400 mL x 3). The combined organic layers were washed with
WSGR Docket No. 62619-720.601 brine (500 mL), dried over anhydrous Na2SO4, filtered and the filtrate concentrated under reduced pressure to give the title compound (48 g, 86% yield) as a brown solid, which was used in the next step without further purification. MS: m/z = 272.9 [M + H]+.1H NMR (400 MHz, Chloroform-d 7.14 - 7.15 (m, 1H), 7.70 -7.69 (m, 1H), 5.13 - 4.97 (m, 1H),3.86 (s, 3H), 3.14 - 3.09 (m, 2H), 2.54 - 2.46 (m, 1H), 2.36 - 2.30 (m, 1H).19F NMR (376 MHz, Chloroform-d [0842] Step 5: 2,8-Dibromo-2-fluoro-6-methoxy-3,4-dihydronaphthalen-1(2H)-one [0843] To a solution of 8-bromo-2-fluoro-6-methoxy-3,4-dihydronaphthalen-1(2H)-one (47 g, 173.6 mmol) in ACN (470 mL) was added PyBr3 (60.54 g, 189.3 mmol). The mixture was stirred at 70 °C for 2 h under N2. The reaction mixture was quenched with H2O (500 mL) at 25 °C and extracted with EtOAc (500 mL x 3). The combined organic layers were washed with brine (500 mL x 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The crude was triturated with mixture solvent (Petroleum ether/EtOAc =10/1, 50 mL) at 25 °C for 30 min to give the title compound (55 g, 90% yield) as a brown solid. MS: m/z = 350.8 [M + H]+. 1H NMR (400 MHz, Chloroform-d J = 2.4 Hz, 1H), 6.72 (d, J = 1.6 Hz, 1H) ,3.89 (s, 3H), 3.38 - 3.30 (m, 1H), 3.13 - 3.07 (m, 1H), 2.85 - 2.79 (m, 1H), 2.67 - 2.57 (m, 1H).19F NMR (376 MHz, Chloroform-d [0844] Step 6: 8-Bromo-2-fluoro-6-methoxynaphthalen-1-ol [0845] To a solution of 2,8-dibromo-2-fluoro-6-methoxy-3,4-dihydronaphthalen-1(2H)-one (54 g, 153.4 mmol) in DMF (540 mL) was added LiBr (29.3 g, 337.5 mmol). The mixture was stirred at 100 °C for 1 h. The reaction mixture was quenched with H2O (500 mL) at 25 °C and extracted with EtOAc (300 mL x 3). The combined organic layers were washed with brine (400 mL x 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was triturated with mixture solvent (Petroleum ether/EtOAc = 10/1, 50 mL) at 25 °C for 30 min to give the title compound (40.17 g, 96% yield) as a brown solid. MS: m/z = 270.9 [M + H]+.1H NMR (400 MHz, Chloroform-d 7.10 (m, 1H), 7.06 - 7.05 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0846] Step 7: 8-Bromo-2-fluoronaphthalene-1,6-diol [0847] To a solution of 8-bromo-2-fluoro-6-methoxynaphthalen-1-ol (6 g, 22.13 mmol) in CH2Cl2 (6.5 mL) was added BBr3 (6.40 mL, 66.40 mmol) at 0 °C. The mixture was stirred at 25 °C for 0.5 h. The reaction mixture was quenched with H2O (100 mL) at 25°C. The pH of the mixture was adjusted to 7 with. NaHCO3 aq. The mixture was extracted with EtOAc (100 mL x 3). The combined organic layers were washed with brine (200 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was triturated with CH2Cl2 (6 mL) at 25 °C
WSGR Docket No. 62619-720.601 for 30 min to give the title compound (4.86 g, 86% yield) as a white solid. 1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0848] Step 8: 8-Bromo-2-fluoronaphthalene-1,6-diol [0849] To a solution of 8-bromo-2-fluoronaphthalene-1,6-diol (1 g, 3.89 mmol) in DMF (15 mL) were added K2CO3 (1.08 g, 7.78 mmol) and CH2FBr (439.32 mg, 3.89 mmol) at 0 °C. The mixture was stirred at 25 °C for 3 h. The reaction mixture was quenched with H2O (30 mL) at 25°C and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 7% of EtOAc in Petroleum ether) to give the title compound (737 mg, 66% yield) as a white solid. MS: m/z = 286.8 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 1H), 5.83 (s, 1H), 5.70 (s, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0850] Step 9: 6-Fluoro-5-(fluoromethoxy)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-ol [0851] A mixture of 8-bromo-2-fluoronaphthalene-1,6-diol (737 mg, 2.55 mmol), 4,4,4',4',5,5,5',5'- octamethyl-2,2'-bi(1,3,2-dioxaborolane) (1.29 g, 5.10 mmol), Ad2nBuP-Pd- G3(cataCXium®APdG3 3PO4 (1.62 g, 7.65 mmol) in 1,4-dioxane (20 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 7% of EtOAc in Petroleum ether) to give the title compound (Intermediate 89, 766 mg, 74% yield) as a yellow oil. MS: m/z = 334.8 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 2H), 7.15 - 7.14 (m, 1H), 5.91 - 5.77 (m, 2H), 1.50 (s, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0852] Intermediate 90: N-(6-Fluoro-5-(fluoromethoxy)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-2-yl)-1,1-diphenylmethanimine
[0853] Step 1: 4-Bromo-6-fluoro-5-(fluoromethoxy)naphthalen-2-yl trifluoromethanesulfonate [0854] To a solution of 4-bromo-6-fluoro-5-(fluoromethoxy)naphthalen-2-ol (1 g, 3.46 mmol) in CH2Cl2 (20 mL) were added DIPEA (1.81 mL, 10.38 mmol) and Tf2
WSGR Docket No. 62619-720.601 dropwise at 0 °C and stirred at 0 °C for 0.5 h under N2. The reaction mixture was diluted with water (20 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of EtOAc in Petroleum ether) to give the title compound (1.39 g, 92% yield) as a colorless oil. 1H NMR (400 MHz, Chloroform-d) 7.91 - 7.63 (m, 3H), 7.57 - 7.45 (m, 1H), 5.89 - 5.69 (m, 2H).19F NMR (376 MHz, Chloroform-d [0855] Step 2: N-(4-Bromo-6-fluoro-5-(fluoromethoxy)naphthalen-2-yl)-1,1-diphenylmethanimine [0856] A mixture of 4-bromo-6-fluoro-5-(fluoromethoxy)naphthalen-2-yl Cs2CO3 (2.07 g, 6.34 mmol) and XantPhosPdG3 degassed and purged with N2 three times, and the mixture was stirred at 80 °C for 2 h under N2 atmosphere. The reaction mixture was filtered, and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 1% of EtOAc in Petroleum ether) to give the title compound (487 mg, 44% yield) as a yellow solid. MS: m/z = 452.0 [M + H]+.1H NMR (400 MHz, Chloroform-d 7.43 - 7.33 (m, 4H), 7.26 - 7.00 (m, 6H), 5.83 - 5.57 (m, 2H).19F NMR (376 MHz, Chloroform-d -129.39, -151.84. [0857] Step 3: N-(6-Fluoro-5-(fluoromethoxy)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)-1,1-diphenylmethanimine [0858] A mixture of N-(4-bromo-6-fluoro-5-(fluoromethoxy)naphthalen-2-yl)-1,1- diphenylmethanimine (467 mg, 1.03 mmol), 4,4,5,5-tetramethyl-2-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)-1,3,2-dioxaborolane (524mg, 2.07 mmol), KOAc (304 mg, 3.10 mmol), 1,4-dioxane (10 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 1 h under N2 atmosphere. The reaction mixture was filtered, and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 5% of EtOAc in Petroleum ether) to give the title compound (Intermediate 90, 390 mg, 66% yield) as a yellow solid. MS: m/z = 500.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide- d6 J = 2.0 Hz, 1H), 5.86 - 5.68 (m, 2H), 1.39 (s, 12H).19F NMR (376 MHz, Chloroform-d 148.53, -148.57.
WSGR Docket No. 62619-720.601 [0859] Intermediate 91: (8aS)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalene
[0860] Step 1: tert-Butyl (3S)-3-(((tert-butyldiphenylsilyl)oxy)methyl)-8,8-difluoro-4- azabicyclo[5.1.0]octane-4-carboxylate [0861] To a solution of tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl)-2,3,6,7-tetrahydro- 1H mmol,) and TMSCF3(535 mg, 3.76 mmol). The mixture was refluxed for 72 h. The reaction mixture was quenched with sat. aq. NaHCO3 (30 mL). The reaction mixture was extracted with EtOAc (50mL x 3). The combined organic layers were washed with brine (60 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of EtOAc in petroleum ether) to give the title compound (380 mg, 91% yield) as a yellow oil. MS: m/z = 416.2 [M + H - Boc]+.1H NMR (400 MHz, Chloroform-d (m, 1H), 3.96 - 3.77 (m, 1H), 3.71 - 3.49 (m, 2H), 3.02 - 2.71 (m, 1H), 2.37 - 2.16 (m, 1H), 2.13 - 1.67 (m, 4H), 1.48 - 1.40 (m, 9H), 1.06 - 1.01 (m, 9H).19F NMR (376 MHz, Chloroform-d 124.05, -124.46, -126.68, -126.82,-127.08, -127.23, -150.22, -150.34, -150.62, -150.75, -151.23, - 151.30. [0862] Step 2: tert-Butyl (3S)-8,8-difluoro-3-(hydroxymethyl)-4-azabicyclo[5.1.0]octane-4- carboxylate [0863] To a solution of tert-butyl (3S)-3-(((tert-butyldiphenylsilyl)oxy)methyl)-8,8-difluoro-4- under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 30% of EtOAc in petroleum ether) to give the title compound (160 mg, 74% yield) as a colorless
WSGR Docket No. 62619-720.601 oil. 1H NMR (400 MHz, Chloroform-d 2H), 3.09 - 2.54 (m, 1H), 2.40 - 2.12 (m, 1H), 1.88 - 1.67 (m, 2H), 1.65 - 1.52 (m, 4H), 1.51 - 1.42 (m, 8H).19F NMR (376 MHz, Chloroform-d 127.32, -127.38, -127.50, -149.99, -150.01, -150.13, -150.48, -150.97, -151.37. [0864] Step 3: ((3S)-8,8-Difluoro-4-azabicyclo[5.1.0]octan-3-yl)methanol [0865] To a solution of tert-butyl (3S)-8,8-difluoro-3-(hydroxymethyl)-4-azabicyclo[5.1.0]octane- reaction mixture was concentrated to dryness. The residue was dissolved in MeCN (10 mL) and MeOH (2 mL), and K2CO3 was added to adjust the pH to around 8. The mixture was filtered and concentrated under reduced pressure to give the title compound (90 mg, crude) as a colorless oil, which was used in the next step without further purification. [0866] Step 4: 2,7-Dichloro-5-(((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methoxy)-8- fluoropyrido[4,3-d]pyrimidin-4(3H)-one [0867] To a mixture of ((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methanol (90 mg, 508 2, and the mixture was stirred at 0 °C under N2 for 0.5 h. Then a solution of Intermediate 1 (167 mg, 559 h under N2 atmosphere. The reaction mixture was quenched with sat. NaHCO3 aq. (3 mL) at 0 °C, dried over anhydrous MgSO4 (6 g), filtered and concentrated under reduced pressure to give the title compound (200 mg, crude) as a yellow oil, which was used in the next step without further purification. MS: m/z = 408.9 [M + H]+. [0868] Step 5: (8aS)-2,5-Dichloro-4,10,10-trifluoro-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa- 1,3,6,12a-tetraazacyclopropa[h]naphtho[1,8-ab]heptalene [0869] To a solution of 2,7-dichloro-5-(((3S)-8,8-difluoro-4-azabicyclo[5.1.0]octan-3-yl)methoxy)- 8-fluoropyrido[4,3-d]pyrimidin-4(3H 2Cl2 (80 mL) was added 3 0 °C under N2 for 2 h. The reaction mixture was quenched with sat. aq. NaHCO3 (100 mL) and extracted with CH2Cl2 (100 mL x 3). The combined organic layers were washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 3% of MeOH in CH2Cl2) to give the title compound (80 mg, 34% yield over 3 steps ) as a yellow oil. MS: m/z = 390.9 [M + H]+.
WSGR Docket No. 62619-720.601 [0870] Step 6: (8aS)-5-Chloro-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalene [0871] A mixture of (8aS)-2,5-dichloro-4,10,10-trifluoro-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa- 1,3,6,12a-tetraazacyclopropa[h]naphtho[1,8-ab R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H 2 three times, and the mixture was stirred at 110 °C under N2 for 16 h. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (Intermediate 91, 60 mg, 48% yield) as a yellow solid. MS: m/z = 514.1 [M + H]+. [0872] Intermediate 92: 6-Fluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-ol
[0873] Step 1: 4-Bromo-6-fluoro-5-(methoxy-d3)naphthalen-2-ol [0874] To a solution of 8-bromo-2-fluoronaphthalene-1,6-diol (refer to Intermediate 89 for detail procedures, 500 mg, 1.95 mmol) and K2CO3 (807 mg, 5.84 mmol) in DMF (3 mL) was added dropwise a solution of CD3I (277 mg, 1.95 mmol) in DMF (1 mL) at 0 °C under N2. The mixture was stirred at 0 °C for 2 h. The reaction mixture was quenched with H2O (50 mL) at 25°C and extracted with EtOAc (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of EtOAc in Petroleum ether) to give the title compound (380 mg, 69% yield, confirmed by 2D-NMR) as a white solid. MS: m/z = 273.7 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 5.6, 9.2 Hz, 1H), 7.51 - 7.39 (m, 2H), 7.21 (d, J = 2.4 Hz, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0875] Step 2: 6-Fluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-ol A mixture of 4-bromo-6-fluoro-5-(methoxy-d3)naphthalen-2-ol (60 mg, 219 3P-Pd-G3 3PO4 purged with N2 three times, and the mixture was stirred at 110 °C for 16 h under N2 atmosphere.
WSGR Docket No. 62619-720.601 The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of EtOAc in Petroleum ether) to give the title compound (Intermediate 92, 40 mg, 56% yield) as a yellow oil. MS: m/z = 319.8 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.4 Hz, 1H), 7.04 (d, J = 2.0 Hz, 1H), 1.36 (s, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 137.48. [0876] Intermediate 93: 6-Fluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-amine
[0877] Step 1: 4-Bromo-6-fluoro-5-(methoxy-d3)naphthalen-2-amine [0878] To a solution of 4-bromo-6-fluoro-5-(methoxy-d3)naphthalen-2-ol (refer to Intermediate 92 and stirred at 20 °C for 1h under N2 was added to the mixture and stirred at 20 °C for 5 h. NaOH (88 mg, 2.19 mmol) was added, and the mixture was stirred at 50 °C for 3h under N2. NaOH (5 M in H2O, 1 mL) and EtOH (1 mL) were added to the reaction, and the mixture was refluxed for 3h under N2. The mixture was quenched with H2O (50 mL) at 25°C and extracted with EtOAc (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The mixture was purified by prep-HPLC (column: 62% B over 10 min) to give the title compound (40 mg, 20% yield) as a brown oil. MS: m/z = 272.9 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.0 Hz, 1H), 5.57 (s, 2H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0879] Step 2: 6-Fluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2- amine [0880] A mixture of 4-bromo-6-fluoro-5-(methoxy-d3 3 (20 mg, 3PO4 2 three times, and the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure to give a residue. The residue was purified by
WSGR Docket No. 62619-720.601 silica gel flash chromatography (eluent: 0 ~ 17% of EtOAc in Petroleum ether) to give the title compound (Intermediate 93, 40 mg, 80% yield) as a yellow oil. MS: m/z = 320.9 [M - H]-. [0881] Intermediate 94: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene
[0882] and purged with N2 three times, and the mixture was stirred at 100 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 3% of CH2Cl2 in MeOH) to give the title compound (Intermediate 94, 97 mg, 43% yield) as a yellow solid. MS: m/z = 466.0 [M + H]+. [0883] Intermediate 95: (S)-2,5-Dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene-13,13-d2
[0884] Step 1: (S)-N-(1-((tert-butyldiphenylsilyl)oxy)hex-5-en-2-yl)acrylamide [0885] To a solution of (S)-1-((tert-butyldiphenylsilyl)oxy)hex-5-en-2-amine (13 g, 36.8 mmol) in CH2Cl2 (230 mL) was added TEA (68 mL, 55.1 mmol) at 0 °C under N2. A solution of acryloyl chloride (3.14 mL, 38.6 mmol) in CH2Cl2 (30 mL) was added dropwise to the mixture at 0 °C under N2, and the mixture was stirred at 0 °C for 1 h. The reaction mixture was partitioned between H2O (150 mL) and CH2Cl2 (300 mL). The organic phase was separated, washed with brine (150 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 25% of EtOAc in petroleum ether) to give the title compound (13.4 g, 89% yield) as a colorless oil. MS: m/z = 408.1
WSGR Docket No. 62619-720.601 [M + H]+.1HNMR (400 MHz, Dimethylsulfoxide-d6 J = 8.4 Hz, 1H), 7.69 - 7.53 (m, 4H), 7.50 - 7.30 (m, 6H), 6.36 - 6.19 (m, 1H), 6.13 - 6.01 (m, 1H), 5.87 - 5.70 (m, 1H), 5.48 - 5.65 (m, 1H), 5.08 - 4.87 (m, 2H), 4.06 - 3.83 (m, 1H), 3.66 - 3.47 (m, 2H), 2.12 - 1.92 (m, 2H), 1.83 - 1.64 (m, 1H), 1.56 - 1.41 (m, 1H), 0.98 (s, 9H). [0886] Step 2: (S)-7-(((tert-Butyldiphenylsilyl)oxy)methyl)-1,5,6,7-tetrahydro-2H-azepin-2-one [0887] To a solution of (S)-N-(1-((tert-butyldiphenylsilyl)oxy)hex-5-en-2-yl)acrylamide (13.4 g, 32.9 mmol) in CH2Cl2 (1400 mL) was added Grubb's II (2.79 g, 3.29 mmol) at 25 °C under N2. The mixture was stirred at 40 °C for 1 h under N2. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 30 % of EtOAc in petroleum ether) to give the title compound (9 g, 72% yield) as a brown gum. MS: m/z = 380.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 6H), 7.42 - 7.37 (m, 1H), 6.30 - 6.13 (m, 1H), 5.72 - 5.58 (m, 1H), 3.71 - 3.64 (m, 1H), 3.62 - 3.55 (m, 1H), 3.41 - 3.39 (m, 1H), 2.41 - 2.27 (m, 2H), 1.99 - 1.94 (m, 1H), 1.83 - 1.71 (m, 1H), 1.00 (s, 9H). [0888] Step 3: (S)-7-(((tert-Butyldiphenylsilyl)oxy)methyl)azepan-2-one [0889] To a solution of (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)-1,5,6,7-tetrahydro-2H-azepin- 2-one (9 g, 23.7 mmol) in MeOH (180 mL) was added 10% Pd/C (4 g, 60% purity) under Ar2. The suspension was degassed and purged with H2 three times. The mixture was stirred at 25 °C for 2 h under H2 (15 Psi). The reaction mixture was filtered and concentrated under reduced pressure to give the title compound (9 g, crude) as a colorless gum, which was used in the next step without further purification. MS: m/z = 382.0 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 - 7.55 (m, 4H), 7.54 - 7.34 (m, 6H), 6.95 - 6.75 (m, 1H), 3.69 - 3.61 (m, 1H), 3.57 - 3.50 (m, 1H), 3.47 - 3.38 (m, 1H), 2.48 - 2.34 (m, 1H), 2.20 - 2.05 (m, 1H), 1.96 - 1.84 (m, 2H), 1.81 - 1.67 (m, 1H), 1.54 - 1.41 (m, 1H), 1.41 - 1.28 (m, 1H), 1.21 - 1.09 (m, 1H), 1.01 (s, 9H). [0890] Step 4: (S)-7-(hydroxymethyl)azepan-2-one [0891] To a solution of (S)-7-(((tert-butyldiphenylsilyl)oxy)methyl)azepan-2-one (3 g, 7.86 mmol) in THF (30 mL) was added TBAF (7.86 mL, 1 M in THF) at 0 °C. The mixture was stirred at 25 °C for 1 h. The reaction mixture was partitioned between H2O (100 mL) and EtOAc (120 mL). The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (1.1 g, 98% yield) as a colorless gum. MS: m/z = 143.8 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 2.48 - 2.38 (m, 1H), 2.20 - 2.10 (m, 1H), 1.92 - 1.82 (m, 1H), 1.80 - 1.68 (m, 2H), 1.53 - 1.42 (m, 1H), 1.36 - 1.28 (m, 1H), 1.17 - 1.03 (m, 1H).
WSGR Docket No. 62619-720.601 [0892] Step 5: (S)-(Azepan-2-yl-7,7-d2)methanol [0893] To a solution of (S)-7-(hydroxymethyl)azepan-2-one (1.1 g, 7.68 mmol) in THF (20 mL) was added LiAlD4 (530 mg, 11.5 mmol) in portion at 0 °C under N2. The mixture was stirred at 0 °C for 2 h under N2. D2O (0.53 mL) was added dropwise to the reaction mixture at 0 °C. 15 wt% NaOH (0.53 mL) was added dropwise and then water (1.5 mL) was added. The mixture was stirred at 25 °C for 0.5 h, filtered and concentrated under reduced pressure to give the title compound (900 mg, crude) as a colorless gum, which was used in the next step without purification. MS: m/z = 131.9 [M + H]+. [0894] Step 6: (S)-5-((Azepan-2-yl-7,7-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0895] To a solution of (S)-(azepan-2-yl-7,7-d2)methanol (900 mg, 6.86 mmol) in THF (18 mL) was added NaH (1.10 g, 27.4 mmol, 60% purity) at 0 °C under N2, and the mixture was stirred for 30 min at 0 °C under N2. A solution of Intermediate 1 (1.84 g, 6.86 mmol) in THF (2 mL) was added to the mixture at 0 °C, and the mixture was stirred at 0 °C for 0.5 h under N2. The mixture was stirred at 25 °C for 2.5 h under N2. The reaction mixture was quenched with sat. NaHCO3 aq. (0.6 mL), diluted with CH2Cl2 (50 ml), dried over MgSO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 50% of MeOH in CH2Cl2) to give the title compound (380 mg, 1.05 mmol, 15% yield) as a yellow solid. MS: m/z = 362.9 [M+ H]+. [0896] Step 7: (S)-2,5-Dichloro-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-13,13-d2 [0897] To a solution of (S)-5-((azepan-2-yl-7,7-d2)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H)-one (380 mg, 1.05 mmol) in CH2Cl2 (120 mL) was added DIPEA (811 mg, 6.28 mmol, 1.09 mL) and POCl3 2. The mixture was stirred for 1 h at 0 °C under N2. The reaction mixture was quenched with sat. NaHCO3 aq. (100 mL) and extracted with CH2Cl2 (40 mL × 2). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0~3% of MeOH in CH2Cl2) to give the title compound (Intermediate 95, 180 mg, 45% yield) as a yellow solid. MS: m/z = 345.0 [M + H]+. [0898] Intermediate 96: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene-13,13-d2
WSGR Docket No. 62619-720.601
[0899] stirred at 105 °C for 16 h under N2. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0~10 % of MeOH in CH2Cl2) to give the title compound (Intermeidate 96, 100 mg, 41% yield) as a yellow solid. MS: m/z = 470.1 [M + H]+. [0900] Intermediate 97: 2,5-Dichloro-4-fluoro-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene
[0901] Step 1: 5-((1,4-Oxazepan-5-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3-d]pyrimidin- 4(3H)-one [0902] A mixture of NaH (135 mg, 3.39 mmol, 60% purity in oil) in THF (5 mL) was degassed and purged with N2 THF (5 mL) was added dropwise at 0 °C under N2 atmosphere. The mixture was stirred at 0 °C for in THF (5 mL) at 0 °C, and then the mixture was stirred at 25 °C for 1 hr under N2 atmosphere. The reaction mixture was diluted with sat. NH4Cl aq. (1 mL), dried over MgSO4, filtered and concentrated under reduced pressure to give the title compound (300 mg, crude) as a yellow solid. MS: m/z = 362.9 [M + H]+. [0903] Step 2: 2,5-Dichloro-4-fluoro-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [0904] To a solution of 5-((1,4-oxazepan-5-yl)methoxy)-2,7-dichloro-8-fluoropyrido[4,3- d]pyrimidin-4(3H 2Cl2 (120 mL) were added DIEA (640 mg, 4.96 mmol) and POCl3 (633 mg, 4.13 mmol) at 0 °C. The mixture was stirred at 0 °C for 1 hr. The reaction mixture was poured into sat. NaHCO3 aq. (100 mL) and extracted with CH2Cl2 (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered
WSGR Docket No. 62619-720.601 and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 50% EtOAc in petroleum ether), the title compound (Intermediate 97, 100 mg, yield: 34% for two steps) was obtained as a yellow solid. MS: m/z = 344.9 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 14.8 Hz, 1H), 4.65 (dd, J = 13.6, 4.8 Hz, 1H), 4.46 (d, J = 13.2 Hz, 1H), 4.25 - 4.14 (m, 1H), 4.11 - 3.96 (m, 2H), 3.81 - 3.66 (m, 1H), 3.54 - 3.38 (m, 1H), 3.36 - 3.22 (m, 1H), 2.20 - 2.09 (m, 2H).19F NMR (376 MHz, Chloroform-d [0905] Intermediate 98: 5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[0906] Intermediate 98 was prepared in a similar manner to Example 76. MS: m/z = 469.9 [M + H]+.1H NMR (400 MHz, Chloroform-d 1H), 4.15 - 3.96 (m, 3H), 3.89 - 3.58 (m, 2H), 3.43 - 3.35 (m, 1H), 3.27 - 3.21 (m, 2H), 3.17 - 3.06 (m, 1H), 3.02 - 2.92 (m, 1H), 2.26 - 2.05 (m, 5H), 1.97 - 1.82 (m, 3H).19F NMR (376 MHz, Chloroform-d [0907] Intermediate 99: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-8,8,13,13-d4
[0908] Step 1: tert-Butyl (S)-(1-hydroxypent-4-en-2-yl-1,1-d2)carbamate [0909] To a solution of (S)-2-((tert-butoxycarbonyl)amino)pent-4-enoic acid (10 g, 39.7 mmol) in THF (150 mL) was added LiAlD4 (3.77 g, 99.3 mmol) in portion at 0 °C under N2. The mixture
WSGR Docket No. 62619-720.601 was stirred at 0 °C for 2 h. D2O (3.77 mL) was added dropwise at 0 °C. 15 wt% NaOH aq. (3.77 mL) was added. And then water (11.3 mL) was added. The mixture was stirred at 25 °C for 0.5 h, filtered and concentrated under redued pressure to give the title compound (7.16 g, 89% yield) as a yellow oil. 1H NMR (400 MHz, Chloroform-d 3.54 (m, 1H), 2.69 - 2.17 (m, 3H), 1.56 - 1.35 (m, 9H). [0910] Step 2: tert-Butyl (S)-(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl-1,1-d2)carbamate [0911] To a solution of tert-butyl (S)-(1-hydroxypent-4-en-2-yl-1,1-d2)carbamate (7.16 g, 35.2 mmol) in CH2Cl2 (30 mL) was added imidazole (2.40 g, 35.2 mmol) and TBDPSCl (11.62 g, 42.27 mmol). The mixture was stirred at 25 °C for 2 h. The reaction mixture was dilluted with H2O (50 mL) and extracted with EtOAc (100 mL × 3). The combined organic layers were washed with brine (100 mL), dried over anhysrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 7% of EtOAc in petroleum ether) to give the title compound (8.6 g, 45% yield) as a yellow gum. MS: m/z =342.0 [M – Boc + H]+.1H NMR (400 MHz, Chloroform-d 5.84 - 5.63 (m, 1H), 5.14 - 4.95 (m, 2H), 4.78 - 4.59 (m, 1H), 3.85 - 3.52 (m, 1H), 2.49 - 2.26 (m, 2H), 1.43 (s, 9H), 1.07 (s, 9H). [0912] Step 3: (S)-1-((tert-Butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2-amine [0913] To a solution of tert-butyl (S)-(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl-1,1- d2)carbamate (8.6 g, 19.47 mmol) in CH2Cl2 (20 mL) was added TFA (5 mL, 67.3 mmol). The mixture was stirred at 25 °C for 2 h. The reation mixture was concentrated under reduced pressure. The residue was disolved in ACN:MeOH=5:1 (10 mL), and K2CO3 (3 g) was added. The mixture was stirred for 5 min, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 7% of MeOH in CH2Cl2) to give the title compound (6.2 g, 93% yield) as a yellow oil. MS: m/z =342.3 [M + H]+.1H NMR (400 MHz, Chloroform-d) 2.50 - 2.25 (m, 2H), 1.07 (s, 9H). [0914] Step 4: (S)-N-(but-3-en-1-yl-1,1-d2)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2- amine [0915] To a solution of (S)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1-d2-2-amine (2 g, 5.86 mmol) in DMF (40 mL) were added K2CO3 (4.05 g, 29.3 mmol) and NaI (4.39 g, 29.3 mmol). A solution of but-3-en-1-yl-1,1-d24-methylbenzenesulfonate (1.20 g, 5.27 mmol) in DMF (10 mL) was added dropwise at 25 °C. The mixture was stirred at 25 °C for 16 h. The mixture was used in the next step directly.
WSGR Docket No. 62619-720.601 [0916] Step 5: tert-Butyl (S)-(but-3-en-1-yl-1,1-d2)(1-((tert-butyldiphenylsilyl)oxy)pent-4-en-2-yl- 1,1-d2)carbamate [0917] To a solution of (S)-N-(but-3-en-1-yl-1,1-d2)-1-((tert-butyldiphenylsilyl)oxy)pent-4-en-1,1- d2-2-amine (3.48 g, 8.75 mmol) and K2CO3 (4.05 g, 29.3 mmol) in DMF (50 mL) was added Boc2O (2.86 g, 13.1 mmol). The mixture was stirred at 25 °C for 1 h. The reaction mixture was partitioned between EtOAc (100 mL) and H2O (100 mL). The organic phase was separated, washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 4% of EtOAc in petroleum ether) to give the title compound (3 g, 69% yield over 2 steps) as a yellow oil. MS: m/z =498.3 [M + H]+. [0918] Step 6: tert-Butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl-d2)-2,3,6,7-tetrahydro-1H- azepine-1-carboxylate-7,7-d2 [0919] To a solution of tert-butyl (S)-(but-3-en-1-yl-1,1-d2)(1-((tert-butyldiphenylsilyl)oxy)pent-4- en-2-yl-1,1-d2)carbamate (2.8 g, 5.63 mmol) in CH2Cl2 (470 mL) under N2 was added Grubb’s II 2 three times and stirred at 40 °C for 1 h under N2. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 10% of EtOAc in petroleum ether) to give the title compound (1.6 g, 61% yield) as a brown oil. MS: m/z =492.1 [M + Na]+.1H NMR (400 MHz, Chloroform-d (m, 1H), 2.92 - 2.77 (m, 1H), 2.62 - 2.40 (m, 1H), 2.31 - 2.08 (m, 2H), 1.48 - 1.34 (m, 9H), 1.04 (s, 9H). [0920] Step 7: tert-Butyl (S)-2-(hydroxymethyl-d2)-2,3,6,7-tetrahydro-1H-azepine-1-carboxylate- 7,7-d2 [0921] To a solution of tert-butyl (S)-2-(((tert-butyldiphenylsilyl)oxy)methyl-d2)-2,3,6,7- tetrahydro-1H-azepine-1-carboxylate-7,7-d2 (1.6 g, 3.41 mmol) in THF (10 mL) was added TBAF (4.09 mL, 1 M in THF). The mixture was stirred at 25 °C for 16 h. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (30 mL x 2). The combined organic layers were washed with brine (40 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 30% of EtOAc in petroleum ether) to give the title compound (490 mg, 62% yield) as a colorless oil. 1H NMR (400 MHz, Chloroform-d 2.34 (m, 2H), 2.30 - 2.10 (m, 2H), 1.53 - 1.41 (m, 9H). [0922] Step 8: (S)-(2,3,6,7-Tetrahydro-1H-azepin-2-yl-7,7-d2)methan-d2-ol
WSGR Docket No. 62619-720.601 [0923] To a solution of tert-butyl (S)-2-(hydroxymethyl-d2)-2,3,6,7-tetrahydro-1H-azepine-1- carboxylate-7,7-d2 (490 mg, 2.12 mmol) in CH2Cl2 (5 mL) at 25 °C was added HCl (2.40 mL, 2 M in EtOAc), and the mixture was stirred at 25 °C for 1 h. The reaction mixture was concentrated under reduced pressure. The residue was diluted with ACN:MeOH=5:1 (5 mL), and K2CO3 (300 mg) was added. The mixture was stirred for about 5 min, filtered and concentrated under reduced pressure to give the title compound (260 mg, 93% yield) as a colorless oil. MS: m/z =131.9 [M + H]+.1H NMR (400 MHz, Chloroform-d (m, 1H), 2.89 - 2.43 (m, 3H), 2.29 - 2.21 (m, 1H). [0924] Step 9: (S)-2,7-Dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7-d2)methoxy- d2)pyrido[4,3-d]pyrimidin-4(3H)-one [0925] To a solution of (S)-(2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7-d2)methan-d2-ol (260 mg, 1.98 mmol) in THF (15 mL) and DMF (2 mL) was added NaH (317 mg, 7.93 mmol, 60% purity) at 0 °C under N2. The mixture was stirred for 0.5 h at 0 °C under N2. Intermediate 1 (479 mg, 1.78 mmol) was added to the mixture at 0 °C under N2. The mixture was stirred at 0 °C for 0.5 h under N2. The reaction mixture was diluted with sat. NH4Cl aq. (1.5 mL) and dried over MgSO4, filtered and concentrated under reduced pressure to give the title compound (719 mg) as a yellow solid, which was used in the next step directly. MS: m/z =362.8 [M + H]+. [0926] Step 10: (S)-2,5-Dichloro-4-fluoro-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene-8,8,13,13-d4 [0927] To a solution of (S)-2,7-dichloro-8-fluoro-5-((2,3,6,7-tetrahydro-1H-azepin-2-yl-7,7- d2)methoxy-d2)pyrido[4,3-d]pyrimidin-4(3H)-one (719 mg, 1.98 mmol) in CH2Cl2 (216 mL) were added DIPEA (2.07 mL,11.9 mmol) and POCl3 2, and then the mixture was stirred at 0 °C for 1 h under N2. The reaction mixture was diluted with warm sat. NaHCO3 aq. (60 mL) and extracted with CH2Cl2 (300 mL x 2). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 3% of MeOH in CH2Cl2) to give the title compound (Intermediate 99, 100 mg, 10% yield over 3 steps) as a yellow solid. MS: m/z =344.8 [M + H]+. [0928] Intermediate 100: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene-8,8,13,13-d4
WSGR Docket No. 62619-720.601
[0929] Intermediate 100 was prepared in a similar manner to Intermediate 96. MS: m/z =470.1 [M + H]+.1H NMR (400 MHz, Chloroform-d (s, 0.5H), 4.04 (dd, J = 4.8, 9.2 Hz, 1H), 3.30 - 3.14 (m, 2H), 3.05 - 2.89 (m, 1H), 2.44 - 2.11 (m, 5H), 2.00 - 1.85 (m, 3H), 1.40 - 1.35 (m, 1H), 1.34 - 1.24 (m, 2H).19F NMR (376 MHz, Chloroform-d [0930] Intermediate 101: 6,7-Difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-2-amine
[0931] Step 1: 5-Bromo-1,2-difluoro-3-(methoxy-d3)benzene [0932] To a solution of 5-bromo-2,3-difluorophenol (20 g, 95.7 mmol) in DMF (200 mL) was added K2CO3 (40 g, 287 mmol) at 0 °C under N2, and then trideuterio(iodo)methane (6.42 mL, 105 mmol) in DMF (200 mL) was added dropwise to the mixture at 0 °C under N2. The mixture was stirred at 0 °C for 2 h under N2. The mixture was diluted with H2O (200 mL) and extracted with EtOAc (100 mL × 2). The combined organic layers were washed with brine (100mL × 2), dried over anhydrous Na2SO4 and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 2% of EtOAc in petroleum ether) to give the title
WSGR Docket No. 62619-720.601 compound (33.2 g, 77% yield) as a colorless oil. 1H NMR (400 MHz, Dimethylsulfoxide-d6 7.37 - 7.30 (m, 1H), 7.28 - 7.23 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 136.97, -161.52, -161.58. [0933] Step 2: 5-Allyl-1,2-difluoro-3-(methoxy-d3)benzene [0934] A mixture of 5-bromo-1,2-difluoro-3-(methoxy-d3)benzene (33.2 g, 147 mmol), 2-allyl- 4,4,5,5-tetramethyl-1,3,2-dioxaborolane (49 g, 294 mmol), CsF (67 g, 441 mmol) and Pd(PPh3)4 (7.49 g, 7.35 mmol) in THF (2500 mL) was degassed and purged with N2 three times, and the mixture was refluxed for 16 h under N2 atmosphere. The mixture was diluted with H2O (500 mL) and extracted with EtOAc (300 mL × 2). The combined organic layers were washed with brine (300mL), dried over anhydrous Na2SO4 and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 2% of EtOAc in petroleum ether) to give the title compound (24.6 g, 89% yield) as a yellow oil. 1H NMR (400 MHz, Dimethylsulfoxide-d6) 19F NMR (376 MHz, Dimethylsulfoxide-d6 [0935] Step 3: 2-(3,4-Difluoro-5-(methoxy-d3)phenyl)acetic acid [0936] To a solution of 5-allyl-1,2-difluoro-3-(methoxy-d3)benzene (24.58 g, 131 mmol) in ACN (320 mL), EtOAc (320 mL) and H2O (520 mL) was added RuCl3 (5.45 g, 26.3 mmol) at 0 °C. Then NaIO4 (98 g, 458 mmol) was added slowly over 1 h while the reaction temperature was maintained bewteen 0 and 10 °C. The resulting mixture was stirred at 0 °C for 1 h. The reaction mixture was filtered, and the filter cake was washed with EtOAc (200 mL). The filtrate was quenched with sat. Na2S2O3 aq. (200 mL) and extracted with EtOAc (200 mL x 2). The combined organic phases were washed with brine (800 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was triturated with petrol ether (500 mL) at 25 °C for 10 min, filtered, and the filter cake was washed with petrol ether (100 mL) and dried under reduced pressure to give the title compound (19.8 g, 74% yield) as a black-brown solid. 1H NMR (400 MHz, Dimethylsulfoxide-d6 19F NMR (376 MHz, Dimethylsulfoxide-d6 [0937] Step 4: 2-(2-Bromo-3,4-difluoro-5-(methoxy-d3)phenyl)acetic acid [0938] To a solution of 2-(3,4-difluoro-5-(methoxy-d3)phenyl)acetic acid (18 g, 87.7 mmol) in acetic acid (80 mL) was added Br2 (6 mL, 116 mmol) at 0 °C. The mixture was stirred at 20 °C for 3 h. The reaction mixture was quenched with water (160 mL) and sat. Na2S2O3 aq. (15 mL) at 25 °C. The precipitate was filtered, and the filter cake was washed with water (100 mL) and dried under reduced pressure to give the title compound (18 g, 66% yield, confirmed by 2D-NMR) as an
WSGR Docket No. 62619-720.601 off-white solid. 1H NMR (400 MHz, Dimethylsulfoxide-d6 3.74 (s, 2H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0939] Step 5: 5-(2-(2-Bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-1-hydroxyethylidene)-2,2- dimethyl-1,3-dioxane-4,6-dione [0940] To a solution of 2-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)acetic acid (18 g, 63.4 mmol) in ACN (180 mL) was added 2,2-dimethyl-1,3-dioxane-4,6-dione (10 g, 69.7 mmol), DMAP (774 mg, 6.34 mmol) and DIPEA (24 mL, 139 mmol). t-BuCOCl (7.80 mL, 63.4 mmol) was added dropwise at 0 °C under N2. The mixture was stirred at 20 °C for 3 h under N2. The reaction mixture was quenched with HCl (200 mL, 1N in water) at 0 °C and the precipitate was filtered. The filter cake was washed with water (50 mL) and dried under reduced pressure to give the title compound (21.3 g, 80% yield) as a white solid. MS: m/z = 409.9 [M - H]-.1H NMR (400 MHz, Chloroform-d 19F NMR (376 MHz, Chloroform-d [0941] Step 6: tert-Butyl 4-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoate [0942] A mixture of 5-(2-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-1-hydroxyethylidene)-2,2- dimethyl-1,3-dioxane-4,6-dione (21.3 g, 43.1 mmol) in t-BuOH (500 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 90 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated to give the title compound (18 g, crude) as a brown solid, which was used in the next step without further purification. MS: m/z = 380.0 [M - H]-. [0943] Step 7: 4-(2-Bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoic acid [0944] To a solution of tert-butyl 4-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoate (18 g, 41.4 mmol) in CH2Cl2 (50 mL) was added TFA (66.0 mL, 888 mmol). The mixture was stirred at 25 °C for 0.5 h. The reaction mixture was concentrated under reduced pressure to the title compound (18 g, crude) as a black-brown solid, which was used in the next step without further purification. MS: m/z = 279.7 [M - COOH]-. [0945] Step 8: 5-Bromo-6,7-difluoro-8-(methoxy-d3)naphthalene-1,3-diol [0946] A solution of 4-(2-bromo-3,4-difluoro-5-(methoxy-d3)phenyl)-3-oxobutanoic acid (18 g, 55.2 mmol) in trifluoromethanesulfonic acid (150 mL) was stirred at 20 °C for 16 h. The reaction mixture was poured into ice water (200 mL) slowly, and the precipitate was filtered, and the filter cake was washed with water (50 ml x 3), sat. NaHCO3 aq. (50 mL), and water (50 ml) in sequence, dried under reduced pressure to give the title compound (12 g, 65% yield over 3 steps) as a black- brown solid. MS: m/z = 307.8 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6
WSGR Docket No. 62619-720.601 1H), 10.03 (s, 1H), 6.91 (d, J = 2 Hz, 1H), 6.55 (d, J = 2 Hz, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0947] Step 9: 6,7-Difluoro-8-(methoxy-d3)naphthalene-1,3-diol [0948] To a solution of 5-bromo-6,7-difluoro-8-(methoxy-d3)naphthalene-1,3-diol (12 g, 38.9 mmol) in MeOH (120 mL) was added Pd/C (4.15 g, 3.89 mmol, 10% purity) under N2. The mixture was degassed and purged with H2 three times, and the mixture was stirred at 25 °C for 2 h under H2 atmosphere (15 psi). The mixture was filtered and washed with MeOH (100 mL), the filtrate was concentrated. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 20% of EtOAc in petroleum ether) to give the title compound (7.6 g, 77% yield) as a white solid. MS: m/z = 227.9 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 1H), 6.59 (d, J = 2 Hz, 1H), 6.46 (d, J = 1.6 Hz, 1H).19F NMR (376 MHz, Dimethylsulfoxide- d6 [0949] Step 10: 6,7-Difluoro-8-(methoxy-d3)naphthalene-1,3-diyl bis(trifluoromethanesulfonate) [0950] To a solution of 6,7-difluoro-8-(trideuteriomethoxy)naphthalene-1,3-diol (7.6 g, 33.16 mmol) and DIPEA (35 mL, 199 mmol) in CH2Cl2 (140 mL) was added dropwise Tf2O (22 mL, 133 mmol) at 0 °C under N2. The mixture was stirred at 0 °C for 1 h under N2 atmosphere. The reaction mixture was quenched with H2O (140 mL) and extracted with CH2Cl2 (140 mL × 3). The combined organic layers were washed with brine (280 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 4% of CH2Cl2 in petroleum ether) to give the title compound (14.9 g, 91% yield) as a yellow solid. MS: m/z = 491.7 [M - H]-.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.4 Hz, 1H), 8.13 - 8.06 (m, 1H), 8.01 (d, J = 1.6 Hz, 1H).19F NMR (376 MHz, Dimethylsulfoxide- d6 [0951] Step 11: 3-((Diphenylmethylene)amino)-6,7-difluoro-8-(methoxy-d3)naphthalen-1-yl trifluoromethanesulfonate [0952] A solution of 6,7-difluoro-8-(methoxy-d3)naphthalene-1,3-diyl bis(trifluoromethanesulfonate) (9.3 g, 18.9 mmol), diphenylmethanimine (3.16 mL, 18.9 mmol), Cs2CO3 (18.4 g, 56.56 mmol) and XantPhosPdG3 (900 mg, 0.95 mmol) in 1,4-dioxane (180 mL) was degassed and purged with N2 three times and the mixture was stirred at 80 °C under N2 for 2 h. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 4% of EtOAc in petroleum ether) to give the title compound (4 g, 70% yield) as a yellow oil. MS: m/z = 525.1 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.0 Hz, 1H), 7.33 -
WSGR Docket No. 62619-720.601 7.31 (m, 2H), 7.22 -7.19 (m, 1H), 7.09 (s, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 73.20, -133.44, -133.49, -154.47, -154.52. [0953] Step 12: N-(6,7-Difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-yl)-1,1-diphenylmethanimine [0954] A solution of 3-((diphenylmethylene)amino)-6,7-difluoro-8-(methoxy-d3)naphthalen-1-yl trifluoromethanesulfonate (4 g, 7.63 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2- dioxaborolane) (2.32 g, 9.15 mmol), AcOK (2.25 g, 22.9 mmol) and Cy3PPdG3 (496 mg, 763 2 three times, and the mixture was stirred at 100 °C under N2 for 1 h.. The reaction mixture was diluted with H2O (150 mL) and extracted with EtOAc (50 mL × 3). The combined organic layers were washed with brine (75 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (8 g, crude) as a black, brown oil, which was used in the next step without further purification. MS: m/z = 502.9 [M + H]+. [0955] Step 13: 6,7-Difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-amine [0956] To a solution of N-(6,7-difluoro-5-(methoxy-d3)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)naphthalen-2-yl)-1,1-diphenylmethanimine (18 g, 16.1 mmol) in EtOH (135 mL) was added hydroxylamine;hydrochloride (2.24 g, 32.3 mmol) and AcOK (4.75 g, 48.4 mmol). The mixture was stirred at 25 °C for 0.5 h. The reaction mixture was concentrated. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 20% of EtOAc in petroleum ether) to give the title compound (Intermediate 101, 4.4 g, 76% yield over 2 steps) as a yellow solid. MS: m/z = 338.8 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 1.6 Hz, 1H), 6.76 (d, J = 2 Hz, 1H), 5.51 (s, 2H), 1.34 (s, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 138.12, -138.18, -163.21, -163.26. [0957] Intermediate 102: 6-Fluoro-5-(fluoromethoxy-d2)-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [0958] Step 1: Methylene-d2 bis(4-methylbenzenesulfonate) [0959] To a solution of (tosyloxy)silver (15.7 g, 56.3 mmol) in ACN (50 mL) was added dibromomethane-d2 (4.5 g, 25.6 mmol). The mixture was stirred at 81 °C for 16 h under N2. The reaction mixture was filtered to remove all the salts, and the mixture was concentrated. The residue was dissolved in CH2Cl2 (500 mL) and precipitate formed. The precipitate was filtered off to remove all the salts. The filtrate was concentrated to give the title compound (2.73 g, 28% yield) as a white solid. MS: m/z = 381.1 [M + Na]+.1H NMR (400 MHz, Chloroform-d 4H), 7.31 - 7.26 (m, 4H), 2.47 (s, 6H). [0960] Step 2: Fluoromethyl-d24-methylbenzenesulfonate [0961] To a solution of methylene-d2 bis(4-methylbenzenesulfonate) (2 g, 5.58 mmol) in ACN (20 mL) were added CsF (1.27 g, 8.37 mmol) and hexaetheylene glycol (2.22 mL , 8.37 mmol, 95% purity). The mixture was stirred at 81 °C for 16 h under N2. The reaction mixture was quenched with H2O (50 mL) at 25 °C and extracted with EtOAc (50 mL x 3). The combined organic layers were washed with brine 50 mL , dried over Na2SO4, filtered and concentrated under reduced pressure to give the title compound (700 mg, crude) as a colourless oil, which was used in the next step without further purification. 1H NMR (400 MHz, Chloroform-d 7.31 (m, 2H), 2.50 - 2.42 (m, 3H).19F NMR (376 MHz, Chloroform-d [0962] Step 3: 4-Bromo-6-fluoro-5-(fluoromethoxy-d2)naphthalen-2-ol [0963] To a solution of fluoromethyl-d24-methylbenzenesulfonate (700 mg, 3.39 mmol,) in DMF (20 mL) was added K2CO3 (938 mg, 6.79 mmol) and 8-bromo-2-fluoronaphthalene-1,6-diol (872 mg, 3.39 mmol). The mixture was stirred at 50 °C for 16 h under N2. The reaction mixture was partitioned between H2O (500 mL) and CH2Cl2 (300 mL). The organic phase was separated, washed with CH2Cl2 (500 mL x 3), dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by f silica gel lash chromatography (eluent: 0 ~ 9% of EtOAc in petroleum ether) to give the title compound (400 mg, 38% yield over 2 steps, confirmed by 2D- NMR) as a white solid. MS: m/z = 290.7 [M - H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 10.24 (s, 1H), 7.75 - 7.68 (m, 1H), 7.56 - 7.46 (m, 2H), 7.29 - 7.23 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0964] Step 4: 6-Fluoro-5-(fluoromethoxy-d2)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)naphthalen-2-ol [0965] A mixture of 4-bromo-6-fluoro-5-(fluoromethoxy-d2)naphthalen-2-ol (70 mg, 240.48 3PO4 (153 3
WSGR Docket No. 62619-720.601 with N2 three times, and the mixture was stirred at 100 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 11% of EtOAc in petroleum ether) to give the title compound (Intermediate 102, 67 mg, 81% yield) as a white solid. MS: m/z = 336.8 [M - H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 1.35 (s, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0966] Intermediate 103: (2,6-Dimethylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol
[0967] Step 1: 1-(tert-Butyl) 2-methyl 2-(2-(chloromethyl)allyl)-4-methylenepyrrolidine-1,2- dicarboxylate [0968] To a solution of 1-tert-butyl 2-methyl 4-methylidenepyrrolidine-1,2-dicarboxylate (18 g, 74.60 mmol) in THF (180 mL) was added lithiobis(trimethylsilyl)amine (1M in THF, 149.20 mL, 149.20 mmol) dropwise at -78°C under nitrogen atmosphere. The resulting mixture was stirred for 1 hour. Then 3-chloro-2-(chloromethyl)prop-1-ene (46.62 g, 373.00 mmol) was added dropwise at -78°C, and the mixture was stirred for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl at 0 °C. The resulting mixture was extracted with EtOAc (3 x 300 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 17% EA in PE to afford the title compound (18 g, 73% yield) as a yellow solid. MS: m/z = 330.15 [M + H]+. 1H NMR (300 MHz, Chloroform-d 5.30 (m, 1H), 5.05 - 4.92 (m, 3H), 4.27 - 4.14 (m, 1H), 4.05 - 3.93 (m, 3H), 3.73 (s, 3H), 3.37 - 3.19 (m, 1H), 2.84 - 2.67 (m, 1.45 (s, 9H). [0969] Step 2: Methyl 2-(2-(chloromethyl)allyl)-4-methylenepyrrolidine-2-carboxylate [0970] To an ice-cooled solution of 1-(tert-butyl) 2-methyl 2-(2-(chloromethyl)allyl)-4- methylenepyrrolidine-1,2-dicarboxylate (18 g, 54.57 mmol) in MeCN (180 mL) was added HCl (4 M in 1,4-dioxane, 179.96 mL, 719.84 mmol) under nitrogen atmosphere. The resulting mixture stirred for 1 hour. The resulting mixture was concentrated under reduced pressure to give the title compound (12 g, crude) as a yellow solid. The crude product was used in the next step without further purification. MS: m/z = 230.00 [M + H]+. [0971] Step 3: Methyl 2,6-dimethylenetetrahydro-1H-pyrrolizine-7a(5H)-carboxylate [0972] A mixture of methyl 2-(2-(chloromethyl)allyl)-4-methylenepyrrolidine-2-carboxylate (12 g, 36.38 mmol), NaHCO3 (14.98 g, 178.27 mmol), and KI (603.98 mg, 3.63 mmol) in ACN (270 mL)
WSGR Docket No. 62619-720.601 was stirred for 2 hours at room temperature under nitrogen atmosphere. The resulting mixture was filtered, and the filter cake was washed with MeCN (3 x 100 ml). The filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 70% EA in PE to afford the title compound (6 g, 85% yield) as a yellow solid. MS: m/z = 194.10 [M + H]+. 1H NMR (300 MHz, Chloroform-d 3H), 3.30 - 3.23 (m, 2H), 3.04 - 2.96 (m, 2H), 2.62 - 2.55 (m, 2H). [0973] Step 4: (2,6-dimethylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol [0974] To an ice-cooled solution of 2,6-dimethylenetetrahydro-1H-pyrrolizine-7a(5H)-carboxylate (2.3 g, 11.90 mmol) in THF (70 mL) under nitrogen atmosphere was added LiAlH4 (11.85 mL, 2 mmol) at 0 °C under nitrogen atmosphere. The resulting mixture was heated at room temperature for 1 hour. The resulting mixture was quenched with water (0.076 mL), NaOH (15% in water, 0.76 mL) and water (0.228 mL) at 0 °C. The resulting mixture was filtered. The filtrate was extracted with EtOAc (3 x 100 mL). The combined organic layers were washed with water (2 x 100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0 - 10% methanol in dichloromethane to afford the title compound (Intermediate 103, 1.8 g, 91% yield) as an off-white solid. MS: m/z =166.00 [M + H]+. 1H NMR (300 MHz, Chloroform-d 3.29 - 3.22 (m, 4H), 2.58 - 2.50 (m, 2H), 2.44 - 2.36 (m, 2H). [0975] Intermediate 104: 7-Fluoro-6-methyl-1-(tetrahydro-2H-pyran-2-yl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)-1H-indazole
[0976] A mixture of 7-fluoro-6-methyl-1-(tetrahydro-2H-pyran-2-yl)-5-(trifluoromethyl)-1H- indazol-4-yl trifluoromethanesulfonate (700 mg, 1.55 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'- bi(1,3,2-dioxaborolane) (474 mg, 1.87 mmol), potassium acetate (305 mg, 3.11 mmol), and Ad2nBuP-Pd-G3 (cataCXiumAPdG3 and purged with N2 three times, and the mixture was stirred at 100 °C for 12h under N2 atmosphere. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (30 mL x 2). The combined organic layers were washed with brine (30mL), dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 7% of ethyl acetate in petroleum ether) to give the title compound (Intermediate 104 275 mg, 41% yield) as a brown solid. MS: m/z = 428.9 [M + H]+. 1H NMR (400 MHz,
WSGR Docket No. 62619-720.601 Chloroform-d J = 1.6 Hz, 1H), 5.91 (dd, J = 2.4, 9.6 Hz, 1H), 4.09 - 3.97 (m, 1H), 3.83 - 3.67 (m, 1H), 2.63 - 2.53 (m, 1H), 2.49 (br s, 3H), 2.22 - 2.14 (m, 1H), 2.14 – 2.05 (m, 1H), 1.81 - 1.72 (m, 2H), 1.68 - 1.61 (m, 1H), 1.44 (s, 12H).19F NMR (376 MHz, Chloroform-d 132.27. [0977] Intermediate 105: 6,7-Difluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-amine
[0978] To a solution of Intermediate 42 (4,300 g, crude) in EtOH (15 L) at room temperature were added NaOAc (1,085.87 g, 13,236.878 mmol) and NH2OH.HCl (919.83 g, 13,236.878 mmol) at room temperature. The resulting mixture was stirred for 8 h at room temperature. Additional NaOAc (1,085.87 g, 13,236.878 mmol) and NH2OH.HCl (919.83 g, 13,236.878 mmol) were added at room temperature. The resulting mixture was stirred for additional 16 h at room temperature. The resulting mixture was diluted with MTBE (15 L). The resulting mixture was washed with of brine (30 L). The aqueous layer was extracted with MTBE (1x2 L). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography, eluted with PE / EA (5:1) to afford the title compound (Intermediate 105, 560 g, 52.8%) as a yellow solid. MS: m/z = 527.3 [M + H]+. 1H- NMR: (400 MHz, DMSO-d6 J = 11.8, 8.3 Hz, 1H), 7.19 (d, J = 2.3 Hz, 1H), 6.85 (d, J = 2.3 Hz, 1H), 5.66 (s, 2H), 1.33 (s, 12H), 1.10 (d, J = 4.4 Hz, 21H). 19F-NMR: (400 MHz, DMSO-d6 J = 23.5 Hz), -139.06 (d, J = 23.5 Hz). [0979] Example 1: 5-Ethynyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[0980] Step 1: (R)-2,7-Dichloro-8-fluoro-5-(2-(piperidin-2-yl)ethoxy)pyrido[4,3-d]pyrimidin- 4(3H)-one [0981] To a solution of (R)-2-(piperidin-2-yl)ethan-1-ol (288 mg, 2.23 mmol) in THF (30 mL) was added NaH (297 mg, 7.45 mmol, 60% purity) at 0 °C under N2 atmosphere, and the mixture was stirred for 0.5 h. Then a solution of Intermediate 1 (500 mg, 1.86 mmol) in THF (9 mL) was added, and the mixture was stirred at 0 °C for 2.5 h under N2 atmosphere. The reaction mixture was quenched with water (6 mL) and concentrated under reduced pressure. The residue was purified by reversed-phase HPLC (column: C18; mobile phase: [Water (NH4OH) - ACN)]; B%: 5% - 60%, 30 min) to give the title compound (230 mg, 33% yield) as a white solid. MS: m/z = 361.1 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 4.56 - 4.45 (m, 1H), 4.32 (dt, J = 4.0, 9.8 Hz, 1H), 3.36 (s, 1H), 2.94 (t, J = 11.6 Hz, 1H), 2.16 - 1.95 (m, 2H), 1.88 - 1.70 (m, 3H), 1.61 - 1.42 (m, 3H). 19F NMR (376 MHz, Dimethylsulfoxide- d6 [0982] Step 2: (R)-2,12-Dichloro-1-fluoro-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalene [0983] To a solution of (R)-2,7-dichloro-8-fluoro-5-(2-(piperidin-2-yl)ethoxy)pyrido[4,3- d]pyrimidin-4(3H 2Cl2 mmol) and POCl3 reaction mixture was quenched with sat. aq. NaHCO3. (50 mL) and extracted with CH2Cl2 (50 mL x 2). The combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 10% of MeOH in CH2Cl2) to give the title compound (105 mg, 45% yield) as a yellow solid. MS: m/z = 344.0 [M + H]+.
WSGR Docket No. 62619-720.601 [0984] Step 3: (R)-2-Chloro-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de]naphthalene [0985] A mixture of (R)-2,12-dichloro-1-fluoro-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H 2 three times, and then the mixture was stirred at 100 °C for 16 h under N2 atmosphere. The reaction mixture was diluted with EtOAc (50 mL) and washed with water (50 mL x 2). The combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 10% of MeOH in CH2Cl2) to give the title compound (45 mg, 59% yield) as a yellow solid. MS: m/z = 466.1 [M + H]+. [0986] Step 4: 6-Fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de]naphthalen-2-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [0987] A mixture of (R)-2-chloro-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de 2CO3 Ad2nBuP-Pd-G3 (cataCXiumA® Pd G3 2O (1 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 100 °C for 3 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 4% of MeOH in CH2Cl2) to give the title compound (40 mg, 52% yield) as a yellow solid. MS: m/z = 772.5 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 - 7.02 (m, 1H), 5.41 - 5.19 (m, 1H), 5.17 - 5.05 (m, 1H), 4.50 - 4.35 (m, 1H), 4.16 - 3.92 (m, 4H), 3.22 - 2.96 (m, 4H), 2.91 - 2.79 (m, 1H), 2.18 - 1.95 (m, 4H), 1.92 - 1.60 (m, 10H), 0.84 (dd, J = 3.6, 6.8 Hz, 18H), 0.71 - 0.63 (m, 3H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 109.17, -143.43, -147.87, -172.20, -172.30. [0988] Step 5: 5-Ethynyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [0989] To a solution of 6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- stirred at 25 °C for 1 h. The reaction mixture was diluted with H2O (30 mL), extracted with EtOAc (30 mL x 2), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The crude product was dissolved with CH2Cl2 solid were precipitated. The precipitate was filtered and washed with petroleum ether (5 mL) and dried to give the title compound (Example 1, 14 mg, 43 % yield) as a yellow solid. MS: m/z = 616.3 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 9.2 Hz, 1H), 7.49 - 7.42 (m, 1H), 7.36 (d, J = 2.4 Hz, 1H), 7.25 - 7.05 (m, 1H), 5.40 - 5.20 (m, 1H), 5.18 - 5.05 (m, 1H), 4.39 (m, 1H), 4.19 - 4.07 (m, 2H), 4.06 (s, 1H), 4.01 - 3.91 (m, 2H), 3.18 - 3.00 (m, 4H), 2.88 - 2.80 (m, 1H), 2.42 - 2.36 (m, 1H), 2.14 - 2.04 (m, 2H), 1.87 - 1.63 (m, 8H), 1.59 - 1.42 (m, 2H), 1.40 (s, 1H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 147.66, -149.34, -172.11, -172.17. [0990] Example 2: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[0991] Step 1: (S)-2,7-Dichloro-8-fluoro-5-(piperidin-2-ylmethoxy)pyrido[4,3-d]pyrimidin-4(3H)- one [0992] To a solution of (S)-piperidin-2-ylmethanol (944 mg, 8.19 mmol) in THF (40 mL) was added NaH (1.19 g, 29.8 mmol, 60% purity) at 0 °C, the mixture was stirred for 0.5 h. Then a solution of Intermediate 1 (2 g, 7.45 mmol) was added, the mixture was stirred at 0 °C for 2.5 h. The reaction mixture was quenched with sat. NH4Cl aq. (3 mL) and concentrated under reduced
WSGR Docket No. 62619-720.601 pressure to give a residue. The residue was purified by reversed-phase HPLC (column: C18; mobile phase: [Water (NH4OH) - ACN)]; B%: 5% - 40%, 30 min) to give the title compound (1.4 g, 44% yield) as a white solid. MS: m/z = 347.2 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 4.0, 11.2 Hz, 1H), 4.21 (dd, J = 7.6, 11.2 Hz, 1H), 3.41 - 3.33 (m, 2H), 3.27 - 3.16 (m, 1H), 2.95 - 2.83 (m, 1H), 1.86 - 1.68 (m, 3H), 1.58 - 1.43 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [0993] Step 2: (S)-2,5-Dichloro-4-fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [0994] To a solution of (S)-2,7-dichloro-8-fluoro-5-(piperidin-2-ylmethoxy)pyrido[4,3- d]pyrimidin-4(3H)-one (1 g, 2.88 mmol) in CH2Cl2 (300 mL) was added DIPEA (3.01 mL, 17.28 mmol) and POCl3 (1.34 mL, 14.40 mmol) at 0 °C and the mixture was stirred at 0 °C for 2 h. The reaction mixture was quenched with sat. NaHCO3 aq. (200 mL) and extracted with CH2Cl2 (200 mL x 2). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (Eluent: 0 - 4% of MeOH in CH2Cl2) to give the title compound (550 mg, 46% yield) as a yellow solid. MS: m/z = 328.9[M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 12.8 Hz, 1H), 4.57 - 4.48 (m, 2H), 3.99 (br d, J = 10 Hz, 1H), 3.14 - 3.03 (m, 1H), 1.89 - 1.64 (m, 6H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 - 141.94. [0995] Step 3: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalene [0996] A mixture of (S)-2,5-dichloro-4-fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (700 mg, 2.13 mmol), ((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol (677.13 mg, 4.25 mmol), and DIPEA (1.85 mL, 10.6 mmol) in 1,4-dioxane (10 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 100 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 4% of MeOH in CH2Cl2) to give the title compound (950 mg, 81 % yield) as a yellow solid. MS: m/z = 452.1 [M + H]+. 1H NMR (400 MHz, Chloroform-d6 - 4.01 (m, 1H), 3.80 - 3.74 (m, 1H), 3.69 - 3.57 (m, 3H), 3.52 - 3.44 (m, 1H), 3.37 - 3.28 (m, 1H), 3.26 - 3.17 (m, 1H), 2.98 - 2.92 (m, 1H), 2.75 - 2.56 (m, 1H), 2.45 - 2.25 (m, 4H), 2.24 - 2.09 (m,
WSGR Docket No. 62619-720.601 4H), 1.89 - 1.82 (m, 2H), 1.78 - 1.71 (m, 2H).19F NMR (376 MHz, Chloroform-d6 172.63. [0997] Step 4: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [0998] A mixture of (S)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalene (850 mg, 1.88 mmol), 6-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-ol (969 mg, 2.07 mmol), K3PO4 (2.00 g, 9.40 mmol), and Ad2nBuP-Pd-G3 (cataCXiumA® Pd G3 2O (5 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 100 °C for 3 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 4% of MeOH in CH2Cl2) to give the title compound (650 mg, 42% yield) as a yellow solid. MS: m/z = 758.2 [M + H]+. 1H NMR (400 MHz, Chloroform-d6 5.12 (m, 2H), 4.53 - 4.10 (m, 4H), 3.88 - 3.56 (m, 1H), 3.47 - 3.12 (m, 3H), 3.08 - 2.97 (m, 1H), 2.87 - 2.76 (m, 1H), 2.07 - 1.83 (m, 6H), 1.82 - 1.47 (m, 6H), 0.96 - 0.83 (m, 18H), 0.69 - 0.55 (m, 3H). 19F NMR (376 MHz, Chloroform-d6 172.90. [0999] Step 5: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1000] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- stirred at 25 °C for 1 h. The reaction mixture was diluted with EtOAc (100 mL) and washed with water (100 mL x 3). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was dissolved with CH2Cl2 (1 mL) and n-hexane (10 mL) was added till the solid was precipitated. The precipitate was filtered and washed with n-hexane (10 mL x 3) and dried to give the title compound (Example 2, 500 mg, 92% yield) as a yellow solid. MS: m/z = 602.2 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 9.2 Hz, 1H), 7.45 (t, J = 8.8 Hz,
WSGR Docket No. 62619-720.601 1H), 7.36 (d, J = 2.4 Hz, 1H), 7.14 (dd, J = 2.4, 14.4 Hz, 1H), 5.40 - 5.07 (m, 2H), 4.57 - 4.35 (m, 2H), 4.16 - 3.89 (m, 4H), 3.14 - 2.99 (m, 4H), 2.87 - 2.79 (m, 1H), 2.02 - 1.65 (m, 10H), 1.64 - 1.43 (m, 2H).19F NMR (376 MHz, Dimethylsulfoxide-d6 -110.72, -145.20, -172.09. [1001] Example 3: 5-Ethyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol
[1002] Step 1: (R)-2-(8-ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-1-fluoro-12- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4- oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalene [1003] A mixture of (R)-2-chloro-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3- de 2nBuP-Pd-G3 (cataCXiumA® Pd G3 3PO4 (150 mg, 708 2 three times, and then the mixture was stirred at 100 °C for 16 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 10% of MeOH in CH2Cl2) to give the title compound (80 mg, 48% yield) as a yellow solid. MS: m/z = 664.2 [M + H]+.1H NMR (400 MHz, Chloroform-d6 J = 6.4, 8.0 Hz, 1H), 7.47 (s, 1H), 7.33 (s, 1H), 7.24 - 7.14 (m, 1H), 5.49 - 5.14 (m, 4H), 4.63 - 4.03 (m, 5H), 3.72 - 3.18 (m, 6H), 3.16 - 2.95 (m, 2H), 2.76 - 2.45 (m, 2H), 2.42 - 1.94 (m, 10H), 1.80 - 1.68 (m, 4H), 1.00 - 0.85 (m, 3H). 19F NMR (376 MHz, Chloroform-d6 173.19. [1004] Step 2: 5-Ethyl-6-fluoro-4-((R)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol [1005] To a solution of (R)-2-(8-ethyl-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-1-fluoro-12- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4- oxa-3,10a,11,13-tetraazabenzo[4,5]cycloocta[1,2,3-de mL) was added TFA (0.2 mL, 2.69 mmol). The mixture was stirred at 25 °C for 1h. The reaction
WSGR Docket No. 62619-720.601 mixture was concentrated under reduced pressure. The crude was purified by reversed-phase HPLC (C18; mobile phase: [water (0.1% TFA)-ACN]; gradient: 10% - 40% B over 30 min) to give the title compound as a TFA salt. The residue was purified again by Agilent StratoSpheres SPE(PL-HCO3 MP SPE) to give the title compound (Example 3, 19.7 mg, 27% yield) as a white solid. MS: m/z = 620.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 7.74 (dd, J = 6.0, 8.4 Hz, 1H), 7.39 - 7.27 (m, 2H), 7.14 - 6.89 (m, 1H), 5.36 - 5.09 (m, 2H), 4.47 - 4.36(m, 1H), 4.23 - 4.09 (m, 2H), 4.04 - 3.90 (m, 2H), 3.13 - 3.07 (m, 3H), 3.01 (s, 1H), 2.86 - 2.78 (m, 1H), 2.25 - 1.89 (m, 6H), 1.87 - 1.43 (m, 10H), 0.87 - 0.75 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1006] Example 4: 5-Ethyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1007] 2 atmosphere. The suspension was degassed and purged with H2 three times, and then the mixture was stirred at 40 °C for 2 h under H2 (30 Psi) atmosphere. The reaction mixture was filtered, washed with MeOH (10 mL x 2), and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 - 8% of MeOH in CH2Cl2) to give the title compound (Example 4, 34 mg, 60% yield) as a white solid. MS: m/z = 606.2 [M+ H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 4.8 Hz, 1H), 7.74 (dd, J = 6.0, 8.8 Hz, 1H), 7.39 - 7.27 (m, 2H), 7.01 (dd, J = 2.4, 18.4 Hz, 1H), 5.39 - 5.20 (m, 1H), 5.14 (t, J = 12.4 Hz, 1H), 4.54 - 4.41 (m, 2H), 4.16 (d, J = 10.0 Hz, 1H), 4.07 - 3.91 (m, 2H), 3.17 - 2.99 (m, 4H), 2.90 - 2.79 (m, 1H), 2.44 - 2.25 (m, 2H), 2.17 - 2.00 (m, 3H), 1.89 - 1.47 (m, 9H), 0.84 - 0.78 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1008] Example 5: 5-Ethynyl-6-fluoro-4-((S)-1-fluoro-12-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-6,6a,7,8,9,10-hexahydro-5H-4-oxa-3,10a,11,13- tetraazabenzo[4,5]cycloocta[1,2,3-de]naphthalen-2-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1009] Example 5 was prepared in a similar manner to Example 1. MS: m/z = 616.2 [M + H]+.1H J = 6.0, 9.2 Hz, 1H), 7.49 - 7.40 (m, 1H), 7.37 - 7.32 (m, 1H), 7.27 - 6.99 (m, 1H), 5.42 - 5.19 (m, 1H), 5.18 - 5.05 (m, 1H), 4.48 - 4.33 (m, 1H), 4.17 - 3.94 (m, 5H), 3.09 (d, J = 8.4 Hz, 3H), 3.01 (s, 1H), 2.88 - 2.78 (m, 1H), 2.41 (t, J = 13.2 Hz, 1H), 2.12 (d, J = 2.8 Hz, 1H), 2.07 - 1.99 (m, 2H), 1.94 - 1.58 (m, 10H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 -172.30. [1010] Example 6: 5-Ethynyl-6-fluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1011] Example 6 was prepared in a similar manner to Example 1. MS: m/z = 602.1 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 8.8 Hz, 1H), 7.36 (s, 1H), 7.15 (d, J = 10.4 Hz, 1H), 5.34 - 5.10 (m, 2H), 4.49 - 4.38 (m,2H), 4.10 - 3.93 (m, 4H), 3.10 -
WSGR Docket No. 62619-720.601 3.01 (m, 4H), 2.85 - 2.80 (m, 1H), 2.12 - 2.01 (m, 2H), 1.88 - 1.56 (m, 9H).19F NMR (376 MHz, Dimethylsulfoxide-d -110.795, -110.825, -145.241, -145.511, -172.092, -172.204. [1012] Example 7: 5-Ethynyl-6-fluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,11,12-tetrahydro-8H-7,10-dioxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1013] Example 7 was prepared in a similar manner to Example 1. MS: m/z = 604.4 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 9.2 Hz, 1H), 7.46 (t, J = 9.2 Hz, 1H), 7.37 (d, J = 2.4 Hz, 1H), 7.14 (d, J = 2.4 Hz, 1H), 5.37 - 5.19 (m, 1H), 5.03 - 4.89 (m, 1H), 4.57 - 4.39 (m, 2H), 4.16 - 3.98 (m, 6H), 3.59 - 3.54 (m, 1H), 3.49 - 3.41 (m, 1H), 3.22 - 3.04 (m, 3H), 3.01 (s, 1H), 2.86 - 2.79 (m, 1H), 2.15 - 1.99 (m, 3H), 1.87 - 1.74 (m, 3H) 19F NMR (376 MHz, Dimethylsulfoxide-d6 [1014] Example 8: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,11,12-tetrahydro-8H-7,10-dioxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [1015] Example 8 was prepared in a similar manner to Example 1. MS: m/z = 604.1 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 9.2 Hz, 1H), 7.44 (t, J = 9.2 Hz, 1H), 7.35 (d, J = 2.4 Hz, 1H), 7.16 - 7.12 (m, 1H), 5.35 - 5.20 (m, 1H), 5.01 - 4.89 (m, 1H), 4.55 - 4.41 (m, 2H), 4.13 - 3.96 (m, 6H), 3.63 - 3.52 (m, 2H), 3.45 (s, 1H), 3.13 - 3.06 (m, 2H), 3.01 (s, 1H), 2.87 - 2.80 (m, 1H), 2.14 - 1.99 (m, 3H), 1.87 - 1.76 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 -110.99, -144.91, -145.05, -172.09, -172.21. [1016] Example 9: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl dimethylcarbamate
[1017] Step 1: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl dimethylcarbamate [1018] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- 2Cl2 (4 mL) were added TEA (119 mg, 1.11 mmol). The mixture was stirred at 25 °C for 1 h. The reaction mixture was diluted with CH2Cl2 (30 mL) and washed with sat. aq. NaHCO3 (20 mL x 2). The combined organic layers were washed with brine (10 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (250 mg, crude) as a yellow solid, which was used into the next step without further purification. MS: m/z = 829.3 [M + H]+. [1019] Step 2: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl dimethylcarbamate [1020] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-
WSGR Docket No. 62619-720.601 The mixture was stirred at 25 °C for 2 h. The reaction mixture was purified by reversed-phase HPLC (column: C18; mobile phase: [Water (NH4OH) - ACN)]; B%: 30% - 70%, 30 min) to give the title compound (Example 9, 71.2 mg, 28% yield over two steps) as a yellow solid. MS: m/z = 673.4 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 5.6, 8.8 Hz, 1H), 7.96 (d, J = 2.4 Hz, 1H), 7.62 (t, J = 9.2 Hz, 1H), 7.46 (dd, J = 2.4, 11.2 Hz, 1H), 5.34 - 5.20 (m, 2H), 4.50 - 4.47 (m, 2H), 4.17 (d, J = 12.4 Hz, 1H), 4.10 (dd, J = 1.6, 10.4 Hz, 1H), 4.01 - 3.90 (m, 2H), 3.10 - 3.01 (m, 7H), 2.94 (s, 3H), 2.86 - 2.79 (m, 1H), 2.12 – 2.05 (m, 3H), 1.86 - 1.71 (m, 9H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 [1021] Example 10: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl isobutyrate
[1022] Step 1: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl isobutyrate [1023] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- 2Cl2 (3 mL) were added TEA sat. aq. NaHCO3 (20 mL) and extracted with CH2Cl2 (20 mL x 2). The combined organic layers were washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to give the title compound (150 mg, crude) as a yellow solid, which was used into the next step without further purification. MS: m/z = 828.2 [M + H]+. [1024] Step 2: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-yl isobutyrate [1025] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-
WSGR Docket No. 62619-720.601 tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- mixture was stirred at 25 °C for 1 h. The reaction mixture was purified by prep-HPLC (neutral 4HCO3) - ACN]; gradient: 50% ~ 80% B over 10 min) to give the title compound (Example 10, 17.6 mg, 16% yield over two steps) as an off-white solid. MS: m/z = 672.1 [M+ H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.4, 8.4 Hz, 1H), 7.98 (d, J = 2.0 Hz, 1H), 7.64 (t, J = 8.8 Hz, 1H), 7.48 (d, J = 12.0 Hz, 1H), 5.36 - 5.10 (m, 2H), 4.55 - 4.38 (m, 2H), 4.21 (d, J = 12.0 Hz, 1H), 4.11 (dd, J = 2.8, 10.0 Hz, 1H), 4.00 - 3.91 (m, 2H), 3.11 - 3.00 (m, 4H), 2.92 - 2.80 (m, 2H), 2.13 - 1.98 (m, 3H), 1.92 - 1.60 (m, 9H), 1.27 (d, J = 6.8 Hz, 6H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 [1026] Example 11: 5-(Ethyl-d5)-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1027] Step 1: (S)-5-(8-(Ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7- oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1028] To a mixture of (S)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexa hydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (refer to Example 2 for detail procedures, 60 mg, 0.13 mmol) and Intermediate 3 (72.74 mg, 0.20 mmol) in THF (2.2 mL) and H2O (0.44 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (19.34 mg, 0.02 mmol) and K3PO4 (169.10 mg, 0.79 mmol) at room temperature. The reaction mixture was stirred at 80 °C for 3 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 7% MeOH in DCM to afford the title compound (55 mg, 63% yield) as a yellow solid. MS: m/z = 655.35 [M + H]+.
WSGR Docket No. 62619-720.601 [1029] Step 2: 5-(Ethyl-d5)-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1030] To an ice-cooled mixture of (S)-5-(8-(ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1- yl)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12- hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (55 mg, 0.08 mmol) in MeCN (4 mL) under nitrogen atmosphere was added 4 N HCl in 1,4-dioxane (0.74 mL) dropwise. The reaction mixture was stirred in an ice bath for 1 hour. The resulting mixture was quenched with saturated aq. NaHCO3 (10 mL), extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by RP-Flash with the following conditions: C18 spherical, 20 - 30 um, 100 A, 40 g; Mobile Phase A: 5 mM aq. NH4HCO3, Mobile Phase B: MeCN; Flow rate: 40 mL/min; Gradient: 5% B hold 3 min, 5% B to 67% B in 15 min, 67% B hold 3 min, 67% B to 95% B in 4 min; Detector: UV 254 & 210 nm; RT: 20 min. The collected fractions were combined, concentrated, and lyophilized overnight to give the title compound (Example 11, 19.8 mg, 36% yield) as a light-yellow lyophilized powder. MS: m/z = 611.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.28 (m, 2H), 7.02 - 7.01 (m, 1H), 5.35 - 5.22 (m, 1H), 5.14 (t, J = 12.4 Hz, 1H), 4.55 - 4.41 (m, 2H), 4.14 (d, J = 10.4 Hz, 1H), 4.04 - 3.88 (m, 2H), 3.15 - 2.98 (m, 4H), 2.89 - 2.78 (m, 1H), 2.17 - 1.41 (m, 12H). 19F NMR (376 MHz, DMSO-d6 [1031] Example 12: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1032] Example 12 was prepared in a similar manner to Example 1. MS: m/z = 616.2 [M + H]+ . 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 2.8, 9.2 Hz, 1H),
WSGR Docket No. 62619-720.601 7.26 (s, 1H), 7.18 - 7.07 (m, 1H), 5.38 - 5.20 (m, 1H), 4.95 - 4.84 (m, 1H), 4.59 - 4.49 (m, 1H), 4.44 - 4.37 (m, 1H), 4.10 (dd, J = 3.2, 10.4 Hz, 2H), 4.05 - 3.96 (m, 2H), 3.76 (s, 1H), 3.10 - 3.00 (m, 3H), 2.85 - 2.79 (m, 1H), 2.16 - 1.95 (m, 4H), 1.88 - 1.54 (m, 9H), 1.37 - 1.20 (m, 1H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 [1033] Example 13: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-vinylnaphthalen-2-ol
[1034] Step 1: (S)-4-Fluoro-5-(7-fluoro-3-(methoxymethoxy)-8-vinylnaphthalen-1-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7- oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1035] A mixture of (S)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- 3PO4 (254 mg, 1.19 mmol), Ad2nBuP-Pd-G3 (cataCXium®APdG3) (29 2O (0.4 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (250 mg, 97% yield) as yellow solid. MS: m/z = 648.4 [M + H]+. [1036] Step 2: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-vinylnaphthalen-2-ol [1037] To a solution of (S)-4-fluoro-5-(7-fluoro-3-(methoxymethoxy)-8-vinylnaphthalen-1-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7- oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de mL) was added HCl/EtOAc (4 M, 0.3 mL). The mixture was stirred at 20 °C for 20 min. The pH of the reaction mixture was adjusted to 8 with sat. aq. NaHCO3. The mixture was partitioned between EtOAc (3×10 mL) and H2O (20mL). The combined organic layers were separated, washed with sat. NaCl (20mL), dried over Na2SO4, filtered and concentrated under reduced
WSGR Docket No. 62619-720.601 pressure. The crude product was triturated with DCM:PE=1:50 at 20 °C for 20 min to give the title compound (Example 13, 76.9 mg, 38% yield) as a yellow solid. MS: m/z = 604.2 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 4.0 Hz, 1H), 7.83 (dd, J = 6.0, 8.8 Hz, 1H), 7.38 (t, J = 9.6 Hz, 1H), 7.32 (d, J = 2.4 Hz, 1H), 7.12 (dd, J = 2.0, 20.0 Hz, 1H), 6.20 - 5.86 (m, 1H), 5.38 - 5.19 (m, 1H), 5.18 - 5.09 (m, 1H), 5.08 - 5.00 (m, 1H), 4.91 (dd, J = 12.0, 19.2 Hz, 1H), 4.54 - 4.38 (m, 2H), 4.14 - 4.11 (m, 1H), 4.03 - 3.88 (m, 2H), 3.15 - 3.03 (m, 3H), 3.02 (s, 1H), 2.87 - 2.78 (m, 1H), 2.17 - 2.09 (m, 1H), 2.07 - 1.98 (m, 2H), 1.94 - 1.64 (m, 9H). [1038] Example 14: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-7-d-2-ol
[1039] Example 14 was prepared in a similar manner to Example 1. MS: m/z = 603.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 7.6 Hz, 1H), 7.37 - 7.36 (m, 1H), 7.17 -7.13 (m, 1H), 5.42 - 5.05 (m, 2H), 4.60 - 4.32 (m, 2H), 4.14 - 3.88 (m, 4H), 3.20 - 2.95 (m, 4H), 2.90 - 2.78 (m, 1H), 2.20 - 1.40 (m, 12H). 19F NMR (376 MHz, DMSO-d6 1F), -145.20 - -145.54 (d, 1F), -172.10 - -172.21 (d, 1F). [1040] Example 15: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-8-d-2-ol
WSGR Docket No. 62619-720.601
[1041] Example 15 was prepared in a similar manner to Example 1. MS: m/z = 603.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 8.8 Hz, 1H), 7.37 (d, J = 1.6 Hz, 1H), 7.13 (d, J = 1.6 Hz, 1H), 5.28 (d, J = 54.8 Hz, 1H), 5.20 - 5.06 (m, 1H), 4.51 - 4.38 (m, 2H), 4.12 - 3.90 (m, 4H), 3.18 - 2.99 (m, 4H), 2.90 - 2.78 (m, 1H), 2.15 - 1.40 (m, 12H). 19F NMR (376 MHz, DMSO-d6 [1042] Example 17: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-5,5-d2)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1043] Example 17 was prepared in a similar manner to Example 1. MS: m/z = 604.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 8.8 Hz, 1H), 7.37 (d, J=2.4 Hz, 1H), 7.17 - 7.12 (m, 1H), 5.28 (d, J = 54.4 Hz, 1H), 5.20 - 5.07 (m, 1H), 4.56 - 4.38 (m, 2H), 4.18 - 4.86 (m, 4H), 3.18 - 2.90 (m, 3H), 2.20 - 1.40 (m, 12H). 19F NMR (376 MHz, DMSO-d6
WSGR Docket No. 62619-720.601 [1044] Example 18: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1045] Example 18 was prepared in a similar manner to Example 1. MS: m/z = 620.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 2.4 Hz, 1H), 7.15 - 7.10 (m, 1H), 5.35 - 5.10 (m, 2H), 4.51 -4.42 (m, 2H), 4.30 - 4.27 (m, 1H), 4.14 - 4.10 (m, 1H), 4.00 - 3.94 (m, 2H), 3.11 - 3.02 (m, 4H), 2.86 - 2.80 (m, 1H), 2.13 - 1.55 (m, 12H). 19F NMR (376 MHz, DMSO-d6 172.11 - -172.22 (d, 1F). [1046] Example 19: 5-Ethynyl-6,8-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1047] Example 19 was prepared in a similar manner to Example 1. MS: m/z = 620.20 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 10.0 Hz, 1H), 7.41 (d, J = 2.4 Hz, 1H), 7.26 - 7.22 (m, 1H), 5.35 - 5.10 (m, 2H), 4.56 - 4.38 (m, 2H), 4.13 - 4.07 (m, 2H), 4.00 - 3.94 (m,
WSGR Docket No. 62619-720.601 2H), 3.18 - 2.94 (m, 4H), 2.90 - 2.80 (m, 1H), 2.18 - 1.42 (m, 12H). 19F NMR (376 MHz, DMSO- d6 [1048] Example 24: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1049] Example 24 was prepared in a similar manner to Example 1. MS: m/z = 604.3 [M + H]+.1H NMR (400 MHz, DMSO-d6 J = 8.8 Hz, 1H), 7.37 (d, J = 2.4 Hz, 1H), 7.18 - 7.13 (m, 1H), 5.31 (d, J = 54.4 Hz, 1H), 5.22 - 5.09 (m, 1H), 4.55 - 4.36 (m, 2H), 4.15 - 4.02 (m, 1H), 4.00 - 3.88 (m, 1H), 3.26 - 2.80 (m, 5H), 2.26 - 1.40 (m, 12H). 19F NMR (376 MHz, DMSO-d6 [1050] Example 25: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-3,3-d2)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1051] Example 25 was prepared in a similar manner to Example 1. MS: m/z = 604.10 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 8.8 Hz, 1H), 7.37
WSGR Docket No. 62619-720.601 (d, J = 2.4 Hz, 1H), 7.17 - 7.12 (m, 1H), 5.28 (d, J = 55.2 Hz, 1H), 5.21 - 5.08 (m, 1H), 4.53 - 4.38 (m, 2H), 4.14 - 3.89 (m, 4H), 3.18 - 2.98 (m, 2H), 2.91 - 2.78 (m, 1H), 2.19 - 1.40 (m, 12H). 19F NMR (376 MHz, DMSO-d6 [1052] Example 26: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-5,5-d2)methoxy-d2)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1053] Example 26 was prepared in a similar manner to Example 1. MS: m/z = 606.30 [M + H]+. 1H NMR (400 MHz, Methanol-d4 5.44 - 5.16 (m, 2H), 4.60 - 4.42 (m, 2H), 4.02 - 3.90 (m, 1H), 3.52 - 3.48 (m, 1H), 3.35 - 3.02 (m, 3H), 2.39 - 2.07 (m, 3H), 2.06 - 1.75 (m, 8H), 1.72 - 1.58 (m, 1H). 19F NMR (376 MHz, Methanol- d4 [1054] Example 33: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-methoxytetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1055] Step 1: (S)-5-Chloro-2,4-difluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1056] To a solution of (S)-2,5-dichloro-4-fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (refer to Example 2 for detail procedures, 500 mg, 1.51 mmol) in DMSO (10 mL) was added KF (132.37 mg, 2.27 mmol) at room temperature. The reaction mixture was heated at 80 °C for 16 hours. The resulting mixture was cooled to room temperature, diluted with EtOAc (100 mL), and washed with water (2 x 30 mL) and brine (30 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 50% EA in PE to afford the title compound (220 mg, 46% yield) as an white solid. MS: m/z = 313.05 [M + H]+. [1057] Step 2: (S)-2,4-Difluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1058] To a mixture of (S)-5-Chloro-2,4-difluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (120 mg, 0.38 mmol) and ((2-fluoro-6- (methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-1- yl)ethynyl)triisopropylsilane (295.03 mg, 0.57 mmol) in THF (7.6 mL) and H2O (1.5 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (55.90 mg, 0.07 mmol) and K3PO4 (488.75 mg, 2.30 mmol) at room temperature. The reaction mixture was heated at 80 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 56% EA in PE
WSGR Docket No. 62619-720.601 to afford the title compound (130 mg, 51% yield) as an off-white solid. MS: m/z = 663.45 [M + H]+. [1059] Step 3: (S)-4-Fluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-2-(((2R,7aS)-2-methoxytetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1060] To an ice-cooled solution of ((2R,7aS)-2-methoxytetrahydro-1H-pyrrolizin-7a(5H)- yl)methanol (31.00 mg, 0.18 mmol) in THF (1.5 mL) under nitrogen atmosphere was added NaH (12.01 mg, 0.181 mmol, 60% dispersion in mineral oil). After stirring in an ice bath for 0.5 hours, (S)-2,4-difluoro-5-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)- 8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (80 mg, 0.121 mmol) was added to the above mixture. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (20 mL) in an ice bath, and extracted with EtOAc (3 x 30 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by prep-TLC (CH2Cl2 / MeOH = 10 : 1) to afford the title compound (95 mg, 96% yield) as a light yellow solid. MS: m/z = 814.35 [M + H]+. [1061] Step 4: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-methoxytetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [1062] To an ice-cooled solution of (S)-4-fluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-2-(((2R,7aS)-2-methoxytetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (95 mg, 0.12 mmol) in MeCN (2 mL) under nitrogen atmosphere was added HCl (4 M in 1,4-dioxane, 1.02 mL, 4.09 mmol) dropwise. The reaction mixture was stirred in an ice bath for 1 hour. The resulting mixture was quenched with saturated aq. NaHCO3 (30 mL) in an ice bath and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated under reduced pressure to afford the title compound (80 mg, crude used directly) as a light-yellow solid. MS: m/z = 770.35 [M + H]+.
WSGR Docket No. 62619-720.601 [1063] Step 5: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-methoxytetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1064] To a stirred solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-methoxytetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- ol (80 mg, crude) in DMF (1 mL) under nitrogen atmosphere was added CsF (157.82 mg, 1.04 mmol) at room temperature. The reaction mixture was stirred at room temperature for 3 hours. The resulting mixture was purified by RP-Flash directly with the following conditions: column, C18; Mobile phase A: 5 mM aq. NH4HCO3; Mobile phase B: MeCN; Gradient: 2% B to 54% B in 20 min; Detector, UV 254 nm. The collected fractions were combined, concentrated and lyophilized overnight to afford the title compound (Example 33, 37.3 mg, 58% yield for two steps) as an off-white lyophilized powder. MS: m/z = 614.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) J = 2.4 Hz, 1H), 7.16 - 7.12 (m, 1H), 5.20 - 5.15 (m, 1H), 4.62 - 4.29 (m, 2H), 4.10 - 3.94 (m, 5H), 3.20 (s, 3H), 3.08 - 2.92 (m, 3H), 2.80 - 2.64 (m, 2H), 2.11 - 2.08 (m, 1H), 1.95 - 1.44 (m, 11H). 19F NMR (376 MHz, DMSO- d6 [1065] Example 34: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2S,7aS)-2-fluoro-6- methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa- 1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
, , , , (m, 2H), 4.59 - 4.37 (m, 2H), 4.17 - 3.86 (m, 4H), 3.67 (d, J = 13.6 Hz, 1H), 3.47 (d, J = 13.6 Hz,
WSGR Docket No. 62619-720.601 1H), 3.30 - 3.11 (m, 1H), 3.09 - 2.97 (m, 2H), 2.62 (s, 2H), 2.25 - 2.10 (m, 2H), 2.00 - 1.43 (m, 6H). 19F NMR (376 MHz, DMSO-d6) -110.70 - -110.73 (d, 1F), -145.23 - -145.49 (d, 1F), - 172.17 - -172.20 (d, 1F). [1067] Example 35: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluoro-6- methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa- 1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol
[1068] Step 1: (S)-5-Chloro-4-fluoro-2-(((2S,7aR)-2-fluoro-6-methylenetetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene [1069] To a mixture of (S)-2,5-dichloro-4-fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (refer to Example 2 for detail procedures, 150 mg, 0.456 mmol) and Intermediate 6 (156.05 mg, 0.912 mmol) in 1,4-dioxane (2 mL) were added DIEA (294.49 mg, 2.280 mmol) and 4A molecular sieves at room temperature. The reaction mixture was heated at 100 °C for 16 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by prep-TLC (DCM / MeOH = 30 / 1) to give the title compound (155 mg, 73% yield) as a yellow solid. MS: m/z = 464.10 [M + H]+. [1070] Step 2: 6-Fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluoro-6-methylenetetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- ol [1071] To a stirred mixture of (S)-5-Chloro-4-fluoro-2-(((2S,7aR)-2-fluoro-6-methylenetetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalene (145 mg, 0.313 mmol) and 6-fluoro-4-(4,4,5,5-
WSGR Docket No. 62619-720.601 tetramethyl-1,3,2-dioxaborolan-2-yl)-5-[2-(triisopropylsilyl)ethynyl]naphthalen-2-ol (292.86 mg, 0.626 mmol) in THF (6 mL) and H2O (1.2 mL) under nitrogen atmosphere were added K3PO4 (398.07 mg, 1.878 mmol) and CataCXium A Pd G3 (45.53 mg, 0.063 mmol) at room temperature. The reaction mixture was heated at 60 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by prep- TLC (DCM / MeOH = 15 / 1) to give the title compound (110 mg, 45% yield) as a yellow solid. MS: m/z = 770.40 [M + H]+. [1072] Step 3: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluoro-6-methylenetetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)naphthalen-2-ol [1073] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluoro-6-methylenetetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a- tetraazabenzo[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2- ol (105 mg, 0.136 mmol) in DMF (2 mL) was added CsF (207.15 mg, 1.360 mmol) at room temperature. The reaction mixture was stirred at room temperature for 3 hours. The resulting mixture was purified by RP-Flash with the following conditions: Column: C18 gel column,40g, 20- 4HCO3, Mobile Phase B: MeCN; Gradient: 0% B hold 10 min, up to 95% B within 30 min, 95% B hold 5 min; Flow rate: 40 mL/min; Detector: UV 254 & 210 nm. The product-containing fractions were collected, concentrated and lyophilized overnight to give the title compound (Example 35, 40 mg, 46% yield) as a light-yellow lyophilized powder. MS: m/z = 614.35 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.45 (t, J = 8.8 Hz, 1H), 7.37 (d, J = 2.4 Hz, 1H), 7.18 - 7.11 (m, 1H), 5.32 (d, J = 52.8 Hz, 1H), 5.20 - 5.06 (m, 1H), 4.98 - 4.88 (m, 2H), 4.55 - 4.35 (m, 2H), 4.14 - 3.88 (m, 4H), 3.67 (d, J = 13.6 Hz, 1H), 3.47 (d, J = 13.6 Hz, 1H), 3.29 - 3.11 (m, 1H), 3.09 - 2.96 (m, 2H), 2.62 (s, 2H), 2.25 - 2.08 (m, 2H), 2.00 - 1.36 (m, 6H). 19F NMR (376 MHz, DMSO-d6 145.23 - -145.49 (d, 1F), -172.17 - -172.20 (d, 1F). [1074] Example 38: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-yl dimethylcarbamate
WSGR Docket No. 62619-720.601 [1075] 2Cl2 (10 mL) was added TEA The reaction mixture was quenched with H2O (20 mL) and extracted with CH2Cl2 (20 mL x 3). The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (Eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (Example 38, 40 mg, 59% yield) as a yellow solid. MS: m/z = 687.2 [M+ H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 7.95 (t, J = 2.4 Hz, 1H), 7.62 (t, J = 9.2 Hz, 1H), 7.48 (dd, J = 22.4, 2.4 Hz, 1H), 5.37 - 5.23 (m, 1H), 4.90 - 4.88 (m, 1H), 4.57 - 4.50 (m, 1H), 4.42 (dd, J = 12.8, 8.8 Hz, 1H), 4.17 – 3.94 (m, 4H), 3.27 - 3.09 (m, 7H), 2.94 - 2.86 (m, 4H), 2.08 - 1.97 (m, 4H), 1.89 - 1.56 (m, 9H), 1.35 - 1.23 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 172.14, - 172.26. [1076] Example 43: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1077] Step 1: N-(6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine [1078] To a stirred mixture of Intermediate 5 (200 mg, 0.42 mmol) and Intermediate 20 (406.74 mg, 0.64 mmol) in H2O (1.5 mL) and THF (7.5 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (62.52 mg, 0.08 mmol) and K3PO4 (546.69 mg, 2.57 mmol) at room temperature. The reaction mixture was stirred at 80 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was
WSGR Docket No. 62619-720.601 purified by prep-TLC (CH2Cl2 / MeOH 15 : 1) to afford the title compound (380 mg, 94% yield) as a light yellow solid. MS: m/z = 935.65 [M + H]+. [1079] Step 2: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1080] To a stirred solution of N-(6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine (360 mg, 0.38 mmol) in EtOH (5 mL) under nitrogen atmosphere were added NaOAc (63.16 mg, 0.77 mmol) and NH2OH•HCl (53.50 mg, 0.77 mmol) at room temperature. The reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (12 : 1) to afford the title compound (260 mg, 87% yield) as a light yellow solid. MS: m/z = 771.15 [M + H]+. [1081] Step 3: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-amine [1082] A mixture of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine (250 mg, 0.32 mmol) and CsF (738.82 mg, 4.86 mmol) in DMF (3 mL) was stirred at room temperature for 1 hour. The resulting mixture was filtered and purified by RP-Flash with the following conditions: Column: C18, 40 g, 4HCO3; Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 10 min; Flow rate: 40 mL/min; Detector: UV 254 & 210 nm. The product-containing fractions were collected, concentrated and lyophilized overnight to give the title compound (Example 43, 140 mg, 70% yield) as a light-yellow lyophilized powder. MS: m/z = 615.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6 1H), 7.07 - 6.99 (m, 2H), 5.70 - 5.56 (m, 2H), 5.28 (d, J = 54.0 Hz, 1H), 4.95 - 4.84 (m, 1H), 4.60 - 4.49 (m, 1H), 4.49 - 4.38 (m, 1H), 4.12 - 4.10 (m, 2H), 4.01 - 3.74 (m, 2H), 3.32 - 3.18 (m, 1H), 3.10 - 3.01 (m, 3H), 2.87 - 2.80 (m, 1H), 2.18 - 1.47 (m, 13H), 1.41 - 1.15 (m, 1H). 19F NMR (376 MHz, DMSO-d6
WSGR Docket No. 62619-720.601 [1083] Example 44: 5-Ethynyl-6,8-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1084] Example 44 was prepared in a similar manner to Example 1. MS: m/z = 634.05 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 9.6 Hz, 1H), 7.41 (s, 1H), 7.29 - 7.23 (m, 1H), 5.8 (d, J = 56.4 Hz, 1H), 4.92 - 4.89 (m, 1H), 4.59 - 4.41 (m, 2H), 4.13 - 3.84 (m, 4H), 3.28 - 3.25 (m, 1H), 3.10 - 3.02 (m, 3H), 2.86 - 2.82 (m, 1H), 2.18 - 1.57 (m, 13H), 1.35 - 1.23 (m, 1H). 19F NMR (376 MHz, DMSO-d6 145.99 - -146.85 (d, 1F), -172.11- -172.22 (d, 1F). [1085] Example 45: 4-Ethynyl-3-fluoro-5-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-7-hydroxy-2-naphthonitrile
[1086] Example 45 was prepared in a similar manner to Example 1. MS: m/z = 641.05 [M + H]+. 1H NMR (400 MHz, DMSO-d6 5.28 (d, J = 54.0 Hz, 1H), 4.98 - 4.82 (m, 1H), 4.68 - 4.30 (m, 3H), 4.24 - 3.90 (m, 4H), 3.20 - 2.95
WSGR Docket No. 62619-720.601 (m, 3H), 2.90 - 2.78 (m, 1H), 2.22 - 1.50 (m, 14H). 19F NMR (376 MHz, DMSO-d6 111.72 (d, 1F), 146.03 - 146.88 (d, 1F), 172.11 - 172.22 (d, 1F). [1087] Example 46: 5-(Ethyl-d5)-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1088] Step 1: (S)-5-(8-(Ethyl-d5)-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-4-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1089] To a mixture of Intermediate 5 (100 mg, 0.21 mmol) and Intermediate 3 (156.79 mg, 0.43 mmol) in THF (4.29 mL) and H2O (0.85 mL) under nitrogen atmosphere were added CataCXiumAPdG3 (31.21 mg, 0.043 mmol) and K3PO4 (273.34 mg, 1.29 mmol) at room temperature. The reaction mixture was heated at 80 °C for 3 hours. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 7% MeOH in DCM to afford the title compound (75 mg, 52% yield) as a yellow solid. MS: m/z = 669.30 [M + H]+. [1090] Step 2: 5-(Ethyl-d5)-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol [1091] To an ice-cooled mixture of Intermediate 5 (46 mg, 0.07 mmol) in MeCN (3.04 mL) under nitrogen atmosphere was added HCl (4 M in 1,4-dioxane, 0.6 mL) dropwise. The reaction mixture was stirred in an ice bath for 1 hour. The resulting mixture was quenched with saturated aqueous NaHCO3 (2 mL) in an ice bath and concentrated under reduced pressure. The residue was purified by RP-Flash with the following conditions: C18 spherical, 20 - 30 um, 100 A, 20 g; Mobile Phase A: 5 mM aq. NH4HCO3; Mobile Phase B: MeCN; Flow rate: 40 mL/min; Gradient: 5% B to 5% B in 3 min, 5% B to 67% B in 10 min, 67% B to 67% B in 3 min, 67% B to 95% B in 5 min; Detector: UV 254 & 210 nm; RT: 20 min. The collected fractions were combined, concentrated, and lyophilized overnight to give the title compound (Example 46, 29.2 mg, 67% yield) as a white lyophilized powder. MS: m/z = 625.15 [M + H]+. 1H NMR (300 MHz, DMSO-d6 (m, 1H), 7.75 - 7.74 (m, 1H), 7.34 - 7.31 (m, 2H), 7.08 - 6.98 (m, 1H), 5.37 - 5.19 (m, 1H), 5.00 -
WSGR Docket No. 62619-720.601 4.80 (m, 1H), 4.55 - 4.45 (m, 2H), 4.16 - 4.01 (m, 3H), 3.30 - 3.20 (m, 1H), 3.17 - 3.01 (m, 3H), 2.86 - 2.80 (m, 1H), 2.20 - 1.20 (m, 14H). 19F NMR (282 MHz, DMSO-d6 1F), -145.35 - -145.95 (m, 1F), -172.13 - -172.15 (d, 1F). [1092] Example 49: (8aS)-4-Fluoro-5-(7-fluoro-6-methyl-5-(trifluoromethyl)-1H-indazol-4-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[1093] Step 1: (8aS)-4-Fluoro-5-(7-fluoro-6-methyl-1-(tetrahydro-2H-pyran-2-yl)-5- (trifluoromethyl)-1H-indazol-4-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1094] K3PO4 2nBuP-Pd-G3 (cataCXiumAPdG3 dioxane (2.5 mL) and H2O (0.5 mL) was degassed and purged with N2 three times, and the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (68 mg, 86% yield) as a yellow solid. MS: m/z = 732.4 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 5.39 - 5.18 (m, 1H), 4.93 - 4.83 (m, 1H), 4.63 - 4.53 (m, 1H), 4.49 - 4.42 (m, 1H), 4.18 - 4.01 (m, 3H), 3.97 - 3.87 (m, 1H), 3.75 - 3.66 (m, 1H), 3.30 (s, 5H), 3.13 - 2.98 (m, 3H), 2.11 - 1.97 (m, 6H), 1.93 - 1.81 (m, 3H), 1.79 - 1.66 (m, 6H), 1.63 - 1.52 (m, 5H).19F NMR (376 MHz, Dimethylsulfoxide-d6 147.07, -172.16, -172.18. [1095] Step 2: (8aS)-4-Fluoro-5-(7-fluoro-6-methyl-5-(trifluoromethyl)-1H-indazol-4-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1096] To a solution of (8aS)-4-fluoro-5-(7-fluoro-6-methyl-1-(tetrahydro-2H-pyran-2-yl)-5- (trifluoromethyl)-1H-indazol-4-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene 2Cl2 (2 mL) was added HCl (4M in EtOAc 0.5 mL). The mixture was stirred at 20 °C for 1 h. The reaction mixture was concentrated under reduced pressure. The residue was dissolved with CH2Cl2 (1 mL), and n-hexane (5 mL) was added to the solution to give
WSGR Docket No. 62619-720.601 precipitate. The mixture was filtered, and the filter cake was washed with n-hexane (5 mL), dried under reduced pressure to give the title compuond (Example 49, 21.1 mg, 36% yield) as a yellow solid. MS: m/z = 648.1 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 7.90 - 7.79 (m, 1H), 5.31 - 5.12 (m, 1H), 4.91 - 4.73 (m, 1H), 4.63 - 4.31 (m, 2H), 4.21 - 3.84 (m, 3H), 3.23 - 3.16 (m, 1H), 3.09 - 2.93 (m, 3H), 2.83 - 2.70 (m, 1H), 2.54 - 2.47 (m, 3H), 2.10 - 2.04 (m, 1H), 2.02 - 1.98 (m, 1H), 1.97 - 1.86 (m, 3H), 1.83 - 1.78 (m, 1H), 1.77 - 1.61 (m, 5H), 1.60 - 1.45 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 147.02, -147.08, -172.17. [1097] Example 51: 5-Ethyl-6,8-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1098] A solution of Example 44 (50 mg, 0.07 mmol) and Pd/C (50 mg, 10wt%) in MeOH (3 mL) under hydrogen atmosphere was stirred at room temperature for 1 hour. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by RP-Flash with the following conditions: C18 spherical, 20 - 30 um, 100 A, 25 g; Mobile Phase A: 5 mM aq. NH4HCO3; Mobile Phase B: MeCN; Flow rate: 25 mL/min; Gradient: 3% B to 10% B in 3 min, 10% B to 80% B in 15 min, 80% B to 80% B in 3 min, 80% B to 95% B in 4 min; Detector: UV 254 & 210 nm; RT: 25 min. The product collected fractions were combined, concentrated and then lyophilized overnight to give the title compound (Example 51, 26.4 mg, 51% yield) as a white powder. MS: m/z = 638.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.43 - 7.40 (m, 2H), 7.16 - 7.07 (m, 1H), 5.35 - 5.22 (m, 1H), 4.94 - 4.84 (m, 1H), 4.56 - 4.46 (m, 2H), 4.15 - 4.01 (m, 3H), 3.29 - 3.20 (m, 1H), 3.13 - 3.01 (m, 3H), 2.86 - 2.80 (m, 1H), 2.41 - 2.30 (m, 1H), 2.21 - 1.49 (m, 15H), 0.85 - 0.74 (m, 3H). 19F NMR (376 MHz, DMSO-d6) -117.16 - - 117.38 (m, 1F), -121.40 - -121.56 (m, 1F), -145.33 - -145.91 (d, 1F), -172.14 - -172.17 (d, 1F). [1099] Example 52: N-(5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-yl)formamide
WSGR Docket No. 62619-720.601
[1100] To a stirred solution of Example 43 (75 mg, 0.12 mmol) in THF (3 mL) were added TEA (12.35 mg, 0.12 mmol) and 0.035 mL mixed anhydride (see Note as below) at room temperature. The resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was purified by RP A: 5 mM aq. NH4HCO3; Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 10 min; Flow rate: 40 mL/min; Detector: UV 254 & 210 nm. The product- containing fractions were collected, concentrated and lyophilized overnight to give the title compound (47.3 mg, 60% yield) as a light-yellow lyophilized powder. MS: m/z = 643.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 1H), 7.80 - 7.71 (m, 1H), 7.58 - 7.53 (m, 1H), 5.35 - 5.22 (m, 1H), 4.96 - 4.84 (m, 1H), 4.56 - 4.42 (m, 2H), 4.16 - 3.92 (m, 4H), 3.32 - 3.18 (m, 1H), 3.10 - 3.01 (m, 3H), 2.86 - 2.80 (m, 1H), 2.14 - 1.56 (m, 13H), 1.42 - 1.17 (m, 1H). 19F NMR (376 MHz, DMSO-d6 145.88 - -146.73 (m, 1F), -172.10 - -172.22 (d, 1F). Note: Mixed anhydride was prepared by stirring formic acid (575 mg, 12.49 mmol) and acetyl acetate (1020 mg, 9.99 mmol) at 60 °C for 2 hours. [1101] Example 58: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-5,5-d2)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1102] Example 58 was prepared in a similar manner to Example 33. MS: m/z = 618.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6
WSGR Docket No. 62619-720.601 1H), 7.21 - 7.13 (m, 1H), 5.35 - 5.21 (m, 1H), 4.94 - 4.86 (m, 1H), 4.59 -4.52 (m, 1H), 4.44 - 4.41 (m, 1H), 4.13 - 3.82 (m, 4H), 3.33 - 3.21 (m, 1H), 3.14 - 3.01 (m, 2H), 2.15 - 1.53 (m, 13H), 1.35 - 1.33 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.67 - -110.81 (d, 1F), -145.96 - -146.856 (d, 1F), -172.16 - -172.27 (d, 1F). [1103] Example 59: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1104] Example 59 was prepared in a similar manner to Example 33. MS: m/z = 618.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.14 - 10.12 (m, 1H), 7.98 - 7.94 (m, 1H), 7.48 - 7.44 (m, 1H), 7.37 (s, 1H), 7.20 - 7.13 (m, 1H), 5.35 - 5.22 (m, 1H), 4.98 - 4.82 (m, 1H), 4.56 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.12 - 3.83 (m, 2H), 3.33 - 3.19 (m, 1H), 3.17 - 3.01 (m, 3H), 2.88 - 2.80 (m, 1H), 2.14 - 1.47 (m, 13H), 1.43 - 1.26 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.66 - - 110.79 (m, 1F), -145.97 - -146.87 (m, 1F), -172.10 - -172.21 (d, 1F). [1105] Example 60: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-3,3-d2)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [1106] Example 60 was prepared in a similar manner to Example 33. MS: m/z = 618.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.15 - 10.13 (m, 1H), 7.97 - 7.94 (m, 1H), 7.48 - 7.44 (m, 1H), 7.37 (s, 1H), 7.20 - 7.13 (m, 1H), 5.35 - 5.21 (m, 1H), 4.95 - 4.85 (m, 1H), 4.56 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.18 - 3.82 (m, 4H), 3.33 - 3.20 (m, 1H), 3.12 - 3.09 (m, 1H), 2.85 - 2.82 (m, 1H), 2.13 - 1.50 (m, 13H), 1.40 - 1.29 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.66 - - 110.79 (m, 1F), -145.96 - -146.86 (m, 1F), -172.63 - -172.74 (d, 1F). [1107] Example 61: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-5,5-d2)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1108] Example 61 was prepared in a similar manner to Example 33. MS: m/z = 620.20 [M + H]+. 7.37 (s, 1H), 7.20 - 7.12 (m, 1H), 5.35 - 5.21 (m, 1H), 4.91 - 4.89 (m, 1H), 4.56 - 4.51 (m, 1H), 4.41 - 4.41 (m, 1H), 4.13 - 3.82 (m, 2H), 3.30 - 3.19 (m, 1H), 3.14 - 2.93 (m, 2H), 2.22 - 1.46 (m, -146.86 (d, 1F), -172.16 - -172.27 (d, 1F). [1109] Example 62: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-2-d)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1110] Example 62 was prepared in a similar manner to Example 33. MS: m/z = 617.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.37 (s, 1H), 7.20 - 7.12 (m, 1H), 4.97 - 4.82 (m, 1H), 4.61 - 4.41 (m, 2H), 4.13 - 3.83 (m, 4H), 3.29 - 3.18 (m, 1H), 3.16 - 2.98 (m, 3H), 2.90 - 2.78 (m, 1H), 2.13 - 1.56 (m, 13H), 1.39 - 1.25 (m, 1H). 19F NMR (376 MHz, DMSO-d6 172.83 - -172.94 (d, 1F). [1111] Example 63: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-2,5,5-d3)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1112] Example 63 was prepared in a similar manner to Example 33. MS: m/z = 621.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 8.8 Hz, 1H), 7.37 (s, 1H), 7.13 (d, J = 2.4 Hz, 1H), 5.05 - 4.85 (m, 1H), 4.65 - 4.30 (m, 2H), 4.20 - 3.75 (m, 2H), 3.30 - 2.90 (m, 3H), 2.20 - 1.20 (m, 14H). 19F NMR (376 MHz, DMSO-d6 110.80 (d, 1F), -145.97 - - 146.88 (d, 1F), -172.90 (s, 1F).
WSGR Docket No. 62619-720.601 [1113] Example 64: (S)-5-(8-Ethynyl-7-fluoronaphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene
[1114] Step 1: (S)-4-Fluoro-5-(7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1115] To a mixture of Intermediate 5 (60 mg, 0.13 mmol) and ((2-fluoro-8-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (87.40 mg, 0.19 mmol) in THF (1.70 mL) and H2O (0.34 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (18.76 mg, 0.026 mmol) and K3PO4 (164.01 mg, 0.77 mmol) at room temperature. The resulting mixture was heated at 80 °C for 3 hours and cooled to room temperature. The resulting mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 5% methanol in dichloromethane to afford the title compound (70 mg, 71% yield) as a yellow solid. MS: m/z = 756.40 [M + H]+. [1116] Step 2: (S)-5-(8-Ethynyl-7-fluoronaphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene [1117] To a solution of (S)-4-fluoro-5-(7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (70 mg, 0.09 mmol) in DMF (1 mL) was added CsF (140.65 mg, 0.93 mmol) at room temperature. The resulting mixture was stirred at room temperature for 3 hours. The solid was filtered out. The filtrate was purified by RP-Flash directly with the following conditions: Column, C18; Mobile phase A: 5 mM aq. NH4HCO3; Mobile phase B: MeCN; Gradient: 5% B - 50% B in 30 min, 50% B - 60% B in 10 min, 60% B - 95% B in 5 min, 56% B hold 3 min; Flow rate: 40 mL/min; Detector UV: 254 & 210 nm. The collected fractions were combined, concentrated, and then lyophilized overnight to give the title compound (Example 64, 34.7 mg, 62% yield) as a yellow lyophilized powder. MS: m/z = 600.30 [M + H]+. 1H NMR (300 MHz, DMSO-d6 (m, 1H), 4.59 - 4.41 (m, 2H), 4.16 - 3.91 (m, 4H), 3.32 - 3.18 (m, 1H), 3.11 - 3.00 (m, 3H), 2.88 -
WSGR Docket No. 62619-720.601 2.82 (m, 1H), 2.22 - 1.47 (m, 13H), 1.42 - 1.16 (m, 1H). 19F NMR (282 MHz, DMSO-d6 - -105.82 (d, 1F), -145.89 - -146.75 (d, 1F), -172.09 - -172.20 (d, 1F). [1118] Example 65: 7-Chloro-5-ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1119] Example 65 was prepared in a similar manner to Example 1. MS: m/z = 650.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.37 - 10.34 (m, 1H), 8.27 - 8.25 (m, 1H), 7.36 (s, 1H), 7.22 - 7.15 (m, 1H), 5.35 - 5.22 (m, 1H), 4.90 - 4.80 (m, 1H), 4.55 - 4.52 (m, 1H), 4.44 - 4.41 (m, 1H), 4.27 - 3.98 (m, 4H), 3.28 - 3.18 (m, 1H), 3.10 - 3.01 (m, 3H), 2.85 - 2.82 (m, 1H), 2.13 - 1.54 (m, 13H), 1.40 - 1.30 (m, 1H). 19F NMR (376 MHz, DMSO-d6 -146.88 (m, 1F), -172.10 - -172.21 (d, 1F). [1120] Example 66: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-2-d)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [1121] Example 66 was prepared in a similar manner to Example 33. MS: m/z = 617.35 [M + H]+. 1 7.20 - 7.13 (m, 1H), 4.95 - 4.85 (m, 1H), 4.56 - 4.41 (m, 2H), 4.12 - 3.80 (m, 4H), 3.11 - 2.97 (m, 3H), 2.86 - 2.80 (m, 1H), 2.17 - 1.22 (m, 15H). 19 110.80 (d, 1F), -145.97 - -146.79 (d, 1F), -172.78 - -172.88 (d, 1F). [1122] Example 67: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2S,7aR)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-2,5,5-d3)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1123] Example 67 was prepared in a similar manner to Example 33. MS: m/z = 621.35 [M + H]+. 1H NMR (300 MHz, DMSO-d6 7.12 (m, 1H), 4.96 - 4.82 (m, 1H), 4.60 - 4.40 (m, 2H), 4.18 - 3.79 (m, 2H), 3.29 - 3.18 (m, 1H), 3.13 - 3.00 (m, 2H), 2.16 - 1.48 (m, 13H), 1.42 - 1.28 (m, 1H). 19F NMR (282 MHz, DMSO-d6 110.64 - -110.80 (d, 1F), -145.97 - 146.80 (d, 1F), -172.84 - -172.94 (d, 1F). [1124] Example 68: 5-(Ethyl-d5)-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1125] Example 68 was prepared in a similar manner to Example 46. MS: m/z = 627.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.07 - 6.97 (m, 1H), 5.35 - 5.21 (m, 1H), 4.92 - 4.85 (m, 1H), 4.58 - 4.44 (m, 2H), 4.20 - 4.10 (m,
WSGR Docket No. 62619-720.601 1H), 3.30 - 3.20 (m, 1H), 3.09 - 3.01 (m, 3H), 2.86 - 4.80 (m, 1H), 2.20 - 1.50 (m, 13H), 1.30 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6 172.13 - -172.16 (d, 1F). [1126] Example 69: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-vinylnaphthalen-2-ol
[1127] Example 69 was prepared in a similar manner to Example 13. MS: m/z = 618.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.04 - 9.95 (m, 1H), 7.85 - 7.81 (m, 1H), 7.39 -7.32 (m, 2H), 7.19 - 7.09 (m, 1H), 6.16 - 5.92 (m, 1H), 5.35 - 5.21 (m, 1H), 5.10 - 4.72 (m, 3H), 4.60 - 4.44 (m, 2H), 4.14 - 4.10 (m, 2H), 4.02 - 3.99 (m, 1H), 3.28 - 3.21 (m, 1H), 3.14 - 3.01 (m, 3H), 2.86 - 2.80 (m, 1H), 2.19 - 1.50 (m, 13H), 1.30 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -117.90 - - 118.06 (d, 1F), -145.77 - -146.81 (d, 1F), -172.13- -172.17 (d, 1F). [1128] Example 70: 5,6-Difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-8-d-2-ol
[1129] Example 70 was prepared in a similar manner to Example 13. MS: m/z = 611.35 [M + H]+. 1H NMR (400 MHz, DMSO-d6 1H), 5.35 - 5.22 (m, 1H), 4.91 - 4.88 (m, 1H), 4.59 - 4.44 (m, 2H), 4.14 - 3.96 (m, 3H), 3.32 - 3.20 (m, 1H), 3.14 - 2.99 (m, 3H), 2.85 - 2.77 (m, 1H), 2.18 - 1.50 (m, 13H), 1.30 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6 -172.11 - -172.15 (d, 1F). [1130] Example 71: 5-Ethynyl-1,6-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[1131] Example 71 was prepared in a similar manner to Example 43. MS: m/z = 633.35 [M + H]+. 1H NMR (300 MHz, DMSO-d6) 7.99 - 7.94 (m, 1H), 7.52 - 7.46 (m, 1H), 7.28 - 7.16 (m, 1H), 5.68 - 5.65 (m, 2H), 5.34 - 5.20 (m, 1H), 5.00 - 4.81 (m, 1H), 4.56 - 4.40 (m, 2H), 4.14 - 3.85 (m, 4H), 3.30 - 3.01 (m, 4H), 2.90 - 2.80 (m, 1H), 2.21 - 2.13 (m, 1H), 2.17 - 1.44 (m, 12H), 1.40 - 1.20 (m, 1H). 19F NMR (282 MHz, DMSO-d6) -112.01 - -112.15 (m, 1F), -145.68 - -146.51 (m, 2F), - 172.11, -172.22 (d, 1F). [1132] Example 72: 5-Ethynyl-6,8-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1133] Example 72 was prepared in a similar manner to Example 1. MS: m/z = 636.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.29 - 7.22 (m, 1H), 5.36 - 5.22 (m, 1H), 4.95 - 4.85 (m, 1H), 4.58 - 4.41 (m, 2H), 4.13 - 3.83 (m, 2H), 3.30 - 3.20 (m, 1H), 3.15 - 3.03 (m, 3H), 2.90 - 2.80 (m, 1H), 2.20 - 1.47 (m, 13H), 1.41 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6 145.98 - -146.84 (d, 1F), -172.12 - -172.23 (d, 1F).
WSGR Docket No. 62619-720.601 [1134] Example 76: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1135] Step 1: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1136] ((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H with H2O (20 mL) and extracted with EtOAc (30 mL x 3). The combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (MeOH in CH2Cl2 = 0 to 6%), the title compound (50 mg, yield: 51 %) was obtained as a yellow solid. MS: m/z = 468.0 [M + H]+.1H NMR (400 MHz, Chloroform-d 5.36 (m, 1H), 5.29 - 5.17 (m, 1H), 4.64 - 4.92 (m, 1H), 4.44 - 4.15 (m, 2H), 4.15 - 3.82 (m, 3H), 3.73 - 2.99 (m, 7H), 2.78 - 2.48 (m, 1H), 2.37 - 1.92 (m, 8H). [1137] Step 2: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen- 5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [1138] A mixture of (S)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene 3PO4 cataCXiumAPdG3 2O (1 mL) was degassed and purged with N2 three times, then the mixture was stirred at 110 °C for 2 hr under N2 atmosphere. The reaction mixture was diluted with H2O (20 mL) and extracted with EtOAc (30 mL x 3). The
WSGR Docket No. 62619-720.601 combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (MeOH in CH2Cl2 = 0 to 8%), the title compound (50 mg, yield: 59%) was obtained as a yellow solid. MS: m/z = 774.2 [M + H]+.1H NMR (400 MHz, Chloroform-d 5.43 - 5.33 (m, 1H), 5.29 - 5.21 (m, 1H), 5.08 - 4.98 (m, 1H), 4.86 - 4.75 (m, 1H), 4.60 (dd, J = 13.6, 5.2 Hz, 1H), 4.41 (d, J = 12.8 Hz, 1H), 4.29 - 4.24 (m, 2H), 4.16 - 4.08 (m, 2H), 3.78 - 3.74 (m, 2H), 3.51 - 3.46 (m, 1H), 3.30 - 3.25 (m, 2H), 3.03 - 3.00 (m, 1H), 2.01 - 1.90 (m, 8H), 0.89 - 0.87 (m, 18H), 0.68 - 0.64 (m, 3H).19FNMR (400 MHz, Chloroform-d 172.842. [1139] Step 3: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-ol [1140] A mixture of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab 2 atmosphere. The reaction mixture was diluted with H2O (10 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with brine (30 mL), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (MeOH in CH2Cl2 = 0 to 8%), the title compound (Example 76, 21.4 mg, yield: 52%) was obtained as a yellow solid. MS: m/z = 618.4 [M + H]+.1H NMR (400 MHz, Methanol- d4 (m, 1H), 4.77 - 4.71 (m, 1H), 4.61 - 4.56 (m, 3H), 4.45 - 4.28 (m, 3H), 4.25 - 4.15 (m, 1H), 4.06 - 3.95 (m, 1H), 3.90 - 3.80 (m, 1H), 3.75 - 3.40 (m, 4H), 3.14 - 3.04 (m, 1H), 2.44 - 2.16 (m, 4H), 2.08 - 1.91 (m, 4H).19FNMR (400 MHz, Chloroform-d [1141] Example 78: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[1142] Example 78 was prepared in a similar manner to Example 43. MS: m/z = 617.35 [M + H]+. 1H NMR (400 MHz, DMSO-d6 5.64 - 5.61 (m, 2H), 5.36 - 5.22 (m, 1H), 4.96 - 4.83 (m, 1H), 4.56 - 4.50 (m, 1H), 4.43 - 4.40 (m, 1H), 4.13 - 4.11 (m, 1H), 4.01 - 3.75 (m, 1H), 3.32 - 3.18 (m, 1H), 3.15 - 3.02 (m, 3H), 2.88 - 2.82 (m, 1H), 2.14 - 1.53 (m, 13H), 1.38 - 1.17 (m, 1H). 19F NMR (376 MHz, DMSO-d6 113.44 (d, 1F), -145.88 - -146.75 (d, 1F), -172.10 - -172.22 (d, 1F). [1143] Example 79: 4-((S)-2-(((2R,6R)-2,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)- 4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-5- ethynyl-6-fluoronaphthalen-2-ol
[1144] Example 79 was prepared in a similar manner to Example 1. MS: m/z = 634.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.20 - 7.13 (m, 1H), 5.41 - 5.28 (m, 2H), 5.00 - 4.85 (m, 1H), 4.58 - 4.41 (m, 2H), 4.23 - 3.86 (m, 4H), 3.48 - 2.94 (m, 5H), 2.44 - 1.87 (m, 6H), 1.80 - 1.27 (m, 6H). 19F NMR (376 MHz, DMSO- d6
WSGR Docket No. 62619-720.601 [1145] Example 80: 4-((S)-2-(((2R,6S,7aS)-2,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
[1146] Example 80 was prepared in a similar manner to Example 1. MS: m/z = 634.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.37 (s, 1H), 7.20 - 7.13 (m, 1H), 5.40 (s, 1H), 5.27 (s, 1H), 4.92 - 4.89 (m, 1H), 4.58 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.14 - 3.82 (m, 4H), 3.34 - 3.08 (m, 5H), 2.30 - 2.16 (m, 4H), 2.08 - 1.81 (m, 2H), 1.78 - 1.42 (m, 6H). 19F NMR (376 MHz, DMSO-d6 - -146.74 (m, 1F), -176.19 - -176.72 (m, 2F). [1147] Example 82: 4-((8R,8aS)-8-ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
[1148] Example 82 was prepared in a similar manner to Example 76. MS: m/z = 644.4 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 5.6, 8.8 Hz, 1H), 7.46 (t, J = 9.2 Hz, 1H), 7.37 (d, J = 2.4 Hz, 1H), 7.24 - 7.12 (m, 1H), 5.39 - 5.19 (m, 1H), 5.01 - 4.86 (m, 1H), 4.42 - 4.34 (m, 1H), 4.14 - 4.07 (m, 1H), 4.05 - 3.75 (m, 3H), 3.28 - 3.18 (m, 1H), 3.14 - 3.00 (m, 3H), 2.88 - 2.79 (m, 1H), 2.16 - 2.04 (m, 2H), 2.02 - 1.84 (m, 4H), 1.81 - 1.65 (m, 6H), 1.61 - 1.49 (m, 2H), 1.47 - 1.20 (m, 2H), 1.07 - 0.96 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6
WSGR Docket No. 62619-720.601 [1149] Example 83: 5-Ethynyl-6-fluoro-4-((S)-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1150] Example 82 was prepared in a similar manner to Example 76. MS: m/z = 652.2 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 7.42 (m, 1H), 7.37 (s, 1H), 7.25 - 7.11 (m, 1H), 5.37 - 5.19 (m, 1H), 4.90 - 4.71 (m, 1H), 4.68 - 4.56 (m, 1H), 4.51 - 4.36 (m, 2H), 4.19 - 3.96 (m, 3H), 3.82 (s, 1H), 3.13 - 3.00 (m, 3H), 2.87 - 2.79 (m, 1H), 2.32 - 1.71 (m, 12H).19F NMR (376 MHz, Dimethylsulfoxide-d6 84.06, -84.71, -89.83, -89.98, -90.47, -90.62, -110.58, -110.76, -145.68, -146.68, -172.12, -172.21. [1151] Example 84: 4-((8aS)-2-(((6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
[1152] Step 1: (8aS)-5-Chloro-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [1153] To an ice-cooled stirred solution of Intermediate 36 (211.51 mg, 1.193 mmol) in THF (10 mL) was added NaH (47.74 mg, 1.193 mmol, 60% dispersion in mineral oil) in portions under nitrogen atmosphere. The reaction mixture was stirred in an ice bath for 30 min. To the above mixture was added (S)-5-chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 23 for detail procedures, 300 mg, 0.918
WSGR Docket No. 62619-720.601 mmol). The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with sat. NH4Cl aq. (40 mL) in an ice bath and extracted with CH2Cl2 (3 x 50 mL). The combined organic layers were washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by Prep-TLC (CH2Cl2 / MeOH = 17 / 1) to afford the title compound (350 mg, 78% yield) as a light-yellow solid. MS: m/z = 484.05 [M + H]+. 1H NMR (400 MHz, Chloroform-d 5.44 - 5.21 (m, 1H), 5.20 - 5.00 (m, 2H), 4.63 - 4.52 (m, 1H), 4.36 (d, J = 12.8 Hz, 1H), 4.24 - 4.09 (m, 2H), 3.93 - 3.84 (m, 1H), 3.41 - 3.05 (m, 5H), 2.84 - 2.63 (m, 1H), 2.40 - 1.88 (m, 5H), 1.88 - 1.67 (m, 3H), 1.67 - 1.55 (m, 2H), 1.41 - 1.22 (m, 1H). 19F NMR (376 MHz, Chloroform-d 141.87 (s, 1F), -175.76 (s, 1F), -180.10 - -180.13 (d, 1F). [1154] Step 2: (8aS)-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-5-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)- 8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene [1155] To a mixture of (8aS)-5-chloro-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene (150 mg, 0.31 mmol) and ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (238.31 mg, 0.46 mmol) in THF (6 mL) and H2O (1.2 mL) were added CataCXium A Pd G3 (45.15 mg, 0.06 mmol) and K3PO4 (394.77 mg, 1.86 mmol) at room temperature under nitrogen atmosphere. The resulting mixture was irradiated with microwave radiation at 80 °C for 2 hours and cooled to room temperature. After filtration, the filtrate was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 6% methanol in dichloromethane to afford the title compound (200 mg, 77% yield) as a yellow solid. MS: m/z = 834.35 [M + H]+. [1156] Step 3: 4-((8aS)-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-6- fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [1157] To an ice-cooled mixture of (8aS)-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-4-fluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (200 mg, 0.24 mmol) in MeCN (10 mL) was added HCl (4 M in 1,4-dioxane, 2.10 mL, 8.40 mmol) dropwise. The resulting mixture was stirred in an ice bath for 1 hour. The resulting mixture was acidified to pH 7 - 8 with sat. aq. NaHCO3 (30 mL) and extracted with EtOAc (3 x 20 mL). The combined organic layers were washed with brine (30 mL), dried
WSGR Docket No. 62619-720.601 over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (180 mg, crude) as a yellow solid. MS: m/z = 790.35 [M + H]+. [1158] Step 3: 4-((8aS)-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-5- ethynyl-6-fluoronaphthalen-2-ol [1159] To a solution of 4-((8aS)-2-(((6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol (180 mg, crude) in DMF (2.5 mL) was added CsF (346.11 mg, 2.28 mmol) at room temperature. The resulting mixture was stirred at room temperature for 3 hours. The solid was filtered out. The filtrate was purified by RP-Flash directly with the following conditions: Column, C18; Mobile phase A: 5 mM aq. NH4HCO3; Mobile phase B: MeCN; Gradient: 5% B - 95% B in 40 min, 42% hold 5 min; Flow rate: 40 mL/min; Detector UV: 254 & 210 nm. The collected fractions were combined, concentrated and then lyophilized overnight to give the title compound (Example 84, 108.6 mg, 71% yield for two steps) as a yellow lyophilized powder.MS: m/z = 634.10 [M + H]+. 1H NMR (400 MHz, DMSO-d6 1H), 7.20 - 7.12 (m, 1H), 5.39 - 5.25 (m, 1H), 5.09 (s, 1H), 4.95 - 4.89 (m, 1H), 4.56 - 4.41 (m, 2H), 4.14 - 3.81 (m, 4H), 3.31 - 3.21 (m, 1H), 3.17 - 2.95 (m, 4H), 2.42 - 2.36 (m, 1H), 2.15 - 1.80 (m, 5H), 1.72 - 1.47 (m, 5H), 1.45 - 1.27 (m, 1H). 19F NMR (376 MHz, DMSO-d6 110.78 (d, 1F), -145.92 - 146.81 (d, 1F), -173.54 - -173.75 (m, 1F), -172.32 - -178.35 (d, 1F). [1160] Example 86: 5-Ethynyl-6,8-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601 [1161] Example 86 was 1H NMR (400 MHz, 2H), 5.34 - 5.21 (m, 1H) (m, 1H), 4.01 - 3.75 (m, 1H), 2.13 (d, J = 4.4 Hz, DMSO-d6) -111.47 - - - -172.22 (m, 1F). [1162] Example 88: 5- pyrrolizin-7a(5H)-yl) tetraazanaphtho[1,8-ab] [1163] Example 88 was 1H NMR (400 MHz, 1H), 7.09 - 6.96 (m, 2H) 4.60 (m, 1H), 4.49 -
3.69 - 3.52 (m, 1H), 3.48 - 3.38 (m, 2H), 3.19 - 2.96 (m, 3H), 2.93 - 2.78 (m, 1H), 2.17 - 2.00 (m, 4H), 1.95 - 1.74 (m, 4H).19F NMR (400 MHz, Dimethylsulfoxide-d6 145.386, -145.993, -172.118, -172.245. [1164] Example 89: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[1165] Example 89 was prepared in a similar manner to Example 43. MS: m/z = 635.35 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 12 Hz, 2H), 5.34 - 5.21 (m, 1H), 4.89 (s, 1H), 4.54 - 4.43 (m, 1H), 4.42 (d, J = 12.8 Hz, 1H), 4.20 - 3.96 (m, 2H), 3.33 - 3.09 (m, 1H), 3.07 - 3.01 (m, 3H), 2.83 - 2.81 (m, 1H), 2.13 - 1.55 (m, 13H), 1.31 - 1.28 (m, 1H). 19F NMR (376 MHz, DMSO-d6 145.97 - -146.81 (m, 1F), -172.10 - -172.21 (m, 1F). [1166] Example 91: 2-Fluoro-8-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6-hydroxy-1-naphthonitrile
[1167] Example 91 was prepared in a similar manner to Example 13. MS: m/z = 619.15 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.34 (m, 2H), 5.35 - 5.21 (m, 1H), 4.92 - 4.80 (m, 1H), 4.70 - 4.30 (m, 2H), 4.14 - 4.11 (m, 1H), 3.33 - 3.24 (m, 1H), 3.10 - 2.90 (m, 3H), 2.83 - 2.82 (m, 1H), 2.16 - 1.45 (m, 13H), 1.20 - 1.10 (m, 1H). 19F NMR (376 MHz, DMSO-d6 172.10 - -172.16 (d, 1F). [1168] Example 92: 5-Ethynyl-6-fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[1169] Step 1: (8R,8aS)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalene [1170] To an ice-cooled solution of Intermediate 12 (198.71 mg, 1.233 mmol) in THF (5 mL) under nitrogen atmosphere was added NaH (49.30 mg, 1.233 mmol, 60% dispersion in mineral oil). The mixture was stirred in an ice bath for 0.5 hours. Intermediate 55 (280 mg, 0.822 mmol) was added to the above mixture. The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (50 mL) and extracted with EtOAc (3 x 30 mL). The combined organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 8% methanol in dichloromethane to afford the title compound (280 mg, 70% yield) as a yellow solid. MS: m/z = 482.20 [M + H]+. [1171] Step 2: N-(6-Fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine [1172] To a mixture of (8R,8aS)-5-chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (280 mg, 0.581 mmol) and Intermediate 20 (733.98 mg, 1.162 mmol) in THF (10 mL) and H2O (2 mL) were added CataCXium A Pd G3 (84.62 mg, 0.116 mmol) and K3PO4 (739.89 mg, 3.486 mmol) at room temperature under nitrogen atmosphere. The reaction mixture was heated at 80 °C for 3 hours and cooled to room temperature. The mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash
WSGR Docket No. 62619-720.601 chromatography, eluted with 6% methanol in dichloromethane to afford the title compound (430 mg, 77% yield) as a yellow solid. MS: m/z = 951.45 [M + H]+. [1173] Step 3: 6-Fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-
8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1174] To a solution of N-(6-fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine (430 mg, 0.452 mmol) in EtOH (10 mL) were added NaOAc (74.16 mg, 0.904 mmol) and NH2OH.HCl (62.82 mg, 0.904 mmol) at room temperature. The reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with CH2Cl2 / MeOH (9:1) to afford the title compound (250 mg, 70% yield) as a yellow solid. MS: m/z = 787.55 [M + H]+. [1175] Step 4: 5-ethynyl-6-fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine [1176] To a solution of 6-fluoro-4-((8R,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine (230 mg, 0.292 mmol) in DMF (2.5 mL) was added CsF (887.81 mg, 5.840 mmol) at room temperature. The reaction mixture was stirred at room temperature for 1 hour. The solid was filtered out. The filtrate was purified by RP-Flash directly with the following conditions: Column, C18; Mobile phase A: 5 mM aq. NH4HCO3; Mobile phase B: MeCN; Gradient: 5 ~ 95% in 40 min, 60% hold 5 mins; Flow rate: 40 mL/min; Detector UV: 254 & 210 nm. The collected fractions were combined, concentrated and then lyophilized overnight to give the title compound (129.6 mg, 70% yield) as a yellow lyophilized powder. MS: m/z = 631.00 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.72 (m, 1H), 7.37 - 7.30 (m, 1H), 7.16 - 6.96 (m, 2H), 7.65 - 5.57 (m, 2H), 5.28 (d, J = 54.4 Hz, 1H), 5.04 - 4.88 (m, 1H), 4.78 - 4.66 (m, 1H), 4.12 - 3.70 (m, 2H), 3.35 - 3.00 (m, 4H), 2.91 - 2.82 (m, 1H), 2.21 - 1.20 (m, 17H). 19F NMR (376 MHz, DMSO-d6 146.03 - -147.29 (m, 1F), -172.07 - -172.22 (m, 1F).
WSGR Docket No. 62619-720.601 [1177] Example 93: 5-Ethynyl-6-fluoro-4-((8S,8aS)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8-methyl-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1178] Example 93 was prepared in a similar manner to Example 92. MS: m/z = 631.35 [M + H]+. 1H NMR (400 MHz, DMSO-d6 5.63 - 5.49 (m, 2H), 5.35 - 5.22 (m, 1H), 5.02 - 4.90 (m, 1H), 4.68 - 4.66 (m, 1H),4.11 - 3.73 (m, 2H), 3.30 - 3.02 (m, 4H), 2.85 - 2.80 (m, 1H), 2.15 - 1.58 (m, 12H), 1.50 - 1.20 (m, 5H). 19F NMR (376 MHz, DMSO-d6 1F). [1179] Example 94: 5-Chloro-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1180] Example 94 was prepared in a similar manner to Example 13. MS: m/z = 628.10 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.41 - 7.40 (m, 1H), 7.25 - 7.15 (m, 1H), 5.38 - 5.20 (m, 1H), 4.83 - 4.75 (m, 1H), 4.57 - 4.44 (m, 2H), 4.12 (s, 1H), 3.38 - 3.21 (m, 1H), 3.20 - 3.04 (m, 3H).2.87 - 2.81 (m, 1H), 2.13 - 1.61 (m, 14H). 19F NMR (376 MHz, DMSO-d6 172.14 - -172.19 (d, 1F).
WSGR Docket No. 62619-720.601 [1181] Example 95: 5,6,8-Trifluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-ol
[1182] To a solution of Intermediate 43 (246.42 mg, 0.51 mmol), Intermediate 33 (100 mg, 0.21 mmol), CataCXium A Pd G3 (32.1 mg, 0.04 mmol) and K3PO4 (272.17 mg, 1.28 mmol) in THF (0.3 mL) was added H2O (0.91 mL, 50.53 mmol) at room temperature under argon atmosphere. The resulting mixture was heated at 80 °C for 3 hours. The resulting mixture was cooled to room temperature, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-15% MeOH in CH2Cl2 to afford the title compound (Example 95, 30.5 mg, 22% yield) as a white lyophilized powder. MS: m/z = 630.10 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 56.0 Hz, 1H), 4.91 - 4.86 (m, 1H), 4.59 - 4.55 (m, 1H), 4.49 - 4.44 (m, 1H), 4.13 (s, 1H), 3.31 - 3.25 (m, 1H), 3.13 - 3.01 (m, 3H), 2.86 - 2.80 (m, 1H), 2.18 - 1.53 (m, 13H), 1.25 - 1.15 (m, 1H). 19F NMR (376 MHz, DMSO-d6 147.43 (m, 2F), -172.12 - -172.16 (m, 1F). [1183] Example 96: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-3,3,5,5-d4)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1184] Example 96 was prepared in a similar manner to Example 33. MS: m/z = 620.60 [M + H]+. 1
WSGR Docket No. 62619-720.601 1H), 7.20 - 7.13 (m, 1H), 5.35 - 5.20 (m, 1H), 4.95 - 4.85 (m, 1H), 4.55 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.13 - 3.82 (m, 4H), 3.33 - 3.18 (m, 1H), 2.14 - 1.55 (m, 13H), 1.41 - 1.18 (m, 1H). 19F 172.79 (d, 1F). [1185] Example 97: (6R,7aS)-7a-((((S)-5-(8-Ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4-fluoro- 8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2- yl)oxy)methyl)-6-fluorotetrahydro-1H-pyrrolizin-2(3H)-one
[1186] Example 97 was prepared in a similar manner to Example 33. MS: m/z = 630.30 [M + H]+. 1 7.03 (m, 1H), 5.5 - 5.2 (m, 1H), 4.88 (s, 1H), 4.60 - 4.51 (m, 1H), 4.45-4.41 (m, 1H), 4.37 - 4.20 (m, 2H), 4.12 (s, 1H), 3.98-3.63 (m, 1H), 3.57 (d, J = 18.8 Hz, 3H), 3.15 (s, 1H), 2.75 - 2.58 (m, 2H), 2.40 - 2.16 (m, 2H), 2.07 - 1.83 (m, 2H), 1.72-1.67 (m, 5H), 1.33 (s, 1H), 1.24 (s, 1H). 19F 172.39 (m, 1F). [1187] Example 98: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluoro-6- methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1188] Example 98 was prepared in a similar manner to Example 33. MS: m/z = 628.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 1.6 Hz, 1H), 7.20 - 7.12 (m, 1H), 5.39 - 5.23 (m, 1H), 4.92 - 4.83 (m, 3H), 4.59 - 4.50 (m, 1H), 4.44 - 4.40 (m, 1H), 4.15 - 4.00 (m, 3H), 3.83 (s, 1H), 3.66 (d, J = 12.0 Hz, 1H), 3.47 (d, J = 14.0 Hz, 1H), 3.27 - 2.98 (m, 3H), 2.61 (d, J = 8.0 Hz, 2H), 2.25 - 2.13 (m, 2H), 1.98 - 1.85 (m, 2H), 1.73 - 1.56 (m, 5H), 1.36 - 1.32 (m, 1H).19F NMR (376 MHz, DMSO-d6) -110.67- -110.80 (m, 1F), -145.92 - -146.85 (m, 1F), -172.16- -172.21 (m, 1F). [1189] Example 100: 5-Ethynyl-6-fluoro-4-((S)-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1190] Example 100 was prepared in a similar manner to Example 43. MS: m/z = 651.2 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 5.68 - 5.56 (m, 2H), 5.44 - 5.20 (m, 1H), 4.91 - 4.74 (m, 1H), 4.68 - 4.55 (m, 1H), 4.48 - 4.35 (m, 2H), 4.29 - 4.00 (m, 2H), 3.98-3.72 (m, 1H), 3.24 - 3.05 (m, 4H), 2.95 - 2.83 (m, 1H), 2.36
WSGR Docket No. 62619-720.601 - 2.09 (m, 5H), 2.06 - 1.77 (m, 7H).19F NMR (376 MHz, Dimethylsulfoxide-d6 84.01, -84.65, -89.71, -90.31, -90.36, -111.48, -113.23, -113.41 -145.54, -146.53, -172.18. [1191] Example 103: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(fluoromethoxy)naphthalen-2-ol
[1192] A mixture of Intermediate 89 Ad2nBuP-Pd-G3(cataCXium®APdG3 3PO4 dioxane (10 mL) and H2O (2 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 2 h under N2 atmosphere. The mixture was concentrated under reduced pressure. The mixture was purified by reversed-phase HPLC (C18, mobile phase: [water (NH4HCO3) - MeCN]; gradient: 6% ~ 80% B over 30 min) to give the title compound (Example 103, 132 mg, 73% yield) as a white solid. MS: m/z = 640.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 7.16 - 7.07 (m, 1H), 5.34 - 5.85 (m, 4H), 4.56 - 4.43 (m, 2H), 4.13 - 3.99 (m, 3H), 3.20 - 3.19 (m, 1H), 3.09 - 3.01 (m, 3H), 2.83 - 2.80 (m, 1H), 2.13 - 1.52 (m, 14H).19F NMR (376 MHz, 149.66, -149.69, -172.10, -172.18. [1193] Example 106: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aR)-2-fluoro-6-methyl-2,3- dihydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [1194] Example 106 was prepared in a similar manner to Example 33. MS: m/z = 628.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.15 - 10.13 (m, 1H), 7.98 - 7.94 (m, 1H), 7.48 - 7.44 (m, 1H), 7.37 (s, 1H), 7.21 - 7.13 (m, 1H), 5.51 - 5.49 (m, 1H), 5.35 - 5.22 (m, 1H), 4.94 - 4.81 (m, 1H), 4.56 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.16 - 3.80 (m, 5H), 3.52 - 3.49 (m, 1H), 3.30 - 3.02 (m, 3H), 2.17 - 1.82 (m, 4H), 1.75 - 1.50 (m, 8H), 1.40 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.64 - -110.79 (d, 1F), -145.92 - -146.81 (d, 1F), -176.29 - -176.35 (d, 1F). [1195] Example 107: 5-Ethynyl-6-fluoro-4-((8aS)-4-fluoro-2-(((5R,6aR)-5- fluorohexahydrocyclopropa[a]pyrrolizin-6a(4H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7- oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1196] Example 107 was prepared in a similar manner to Example 33. MS: m/z = 628.30 [M + H]+. 1 1H), 7.37 - 7.36 (m, 1H), 7.21 - 7.13 (m, 1H), 5.55 - 5.41 (m, 1H), 4.95 - 4.81 (m, 1H), 4.66 - 4.52 (m, 1H), 4.47 - 4.41 (m, 1H), 4.17 - 3.78 (m, 4H), 3.30 - 3.24 (m, 3H), 2.82 - 2.75 (m, 2H), 2.44 - 2.30 (m, 1H), 2.08 - 1.82 (m, 3H), 1.80 - 1.46 (m, 7H), 1.43 - 1.16 (m, 1H), 0.69 - 0.67 (m, 1H), 0.32 - 0.31 (m, 1H). 19 146.76 (d, 1F), -172.32 - -172.53 (d, 1F). [1197] Example 108: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,6S,7R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl-6,7-d2)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1198] Example 108 was prepared in a similar manner to Example 33. MS: m/z = 618.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 1H), 7.37 (s, 1H), 7.21 - 7.13 (m, 1H), 5.35 - 5.22 (m, 1H), 4.95 - 4.85 (m, 1H), 4.56 - 4.41 (m, 2H), 4.13 - 3.83 (m, 4H), 3.32 - 3.20 (m, 1H), 3.10 - 3.00 (m, 3H), 2.85 - 2.80 (m, 1H), 2.21 - 1.25 (m, 12H). 19F NMR (376 MHz, DMSO-d6 172.10 - -172.20 (d, 1F). [1199] Example 109: (2R,6R,7aS)-7a-((((S)-5-(8-Ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2- yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol
[1200] Example 109 was prepared in a similar manner to Example 1. MS: m/z = 632.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.12 (m, 1H), 5.35 - 5.21 (m, 1H), 4.95 - 4.85 (m, 1H), 4.70 (s, 1H), 4.59 - 4.50 (m, 1H), 4.43 - 4.40(m, 1H), 4.35 (s, 1H), 4.31 - 4.27 (m, 1H), 4.18 - 4.11 (m, 2H), 4.09 - 3.83 (m, 1H), 3.27 - 3.20 (m, 1H), 3.14 - 2.98 (m, 4H), 2.17 - 1.85 (m, 6H), 1.74 - 1.57 (m, 5H), 1.34 - 1.21 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.78 - -110.89 (m, 1F), -145.97 - -146.82 (m, 1F), -173.38 - - 173.44 (m, 1F).
WSGR Docket No. 62619-720.601 [1201] Example 110: (2S,6R,7aS)-7a-((((S)-5-(8-Ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2- yl)oxy)methyl)-6-fluorohexahydro-1H-pyrrolizin-2-ol
[1202] Example 110 was prepared in a similar manner to Example 1. MS: m/z = 632.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6 J = 2.4 Hz, 1H), 7.20 - 7.11 (m, 1H), 5.61 - 5.57 (m, 1H), 5.33 - 5.17 (m, 1H), 4.93 - 4.84 (m, 1H), 4.68 (s, 1H), 4.59 - 4.50 (m, 1H), 4.45 - 4.41 (m, 1H), 4.14 - 4.10 (m, 1H), 4.09 - 3.81 (m, 1H), 3.30 - 3.20 (m, 5H), 3.12 - 2.95 (m, 2H), 2.24 - 1.85 (m, 6H), 1.73 - 1.51 (m, 5H), 1.37 - 1.26 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.87 - -110.98 (m, 1F), -146.00 - -146.82 (m, 1F), - 171.61 - -171.65 (m, 1F). [1203] Example 113: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-ol
[1204] Example 113 was prepared in a similar manner to Example 76. MS: m/z = 614.2 [M+ H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 1.2 Hz, 1H), 7.96 (dd, J = 6.0, 9.2 Hz, 1H), 7.46 (dt, J = 1.2, 9.2 Hz, 1H), 7.37 (s, 1H), 7.22 - 7.07 (m, 1H), 5.82 - 5.64 (m, 2H), 5.39 - 5.16 (m, 1H), 5.06 - 4.88 (m, 1H), 4.63 - 4.55 (m, 1H), 4.50 - 4.40 (m, 1H), 4.37 - 4.24 (m, 1H), 4.18 - 3.88 (m, 3H), 3.30 - 3.25 (m, 1H), 3.20 - 3.10 (m, 2H), 3.05 - 2.99 (m, 1H), 2.88 - 2.78 (m, 1H), 2.56 - 2.52 (m, 1H), 2.49 - 2.35 (m, 3H), 2.16 - 1.98 (m, 3H), 1.87 - 1.71 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6
WSGR Docket No. 62619-720.601 [1205] Example 116: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((6'R,7a'S)-6'-fluorodihydro-1'H,3'H- spiro[cyclopropane-1,2'-pyrrolizin]-7a'(5'H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H- naphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1206] Example 116 was prepared in a similar manner to Example 33. MS: m/z = 642.20 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.37 (s, 1H), 7.21 - 7.13 (m, 1H), 5.39 - 5.26 (m, 1H), 4.98 - 4.82 (m, 1H), 4.56 - 4.51 (m, 1H), 4.45 - 4.41 (m, 1H), 4.37 - 4.32 (m, 1H), 4.13 - 3.81 (m, 3H), 3.30 - 3.18 (m, 1H), 3.18 - 2.98 (m, 3H), 2.71 - 2.68 (m, 1H), 2.18 - 1.82 (m, 5H), 1.79 - 1.43 (m, 6H), 1.43 - 1.27 (m, 1H), 0.60 - 0.40 (m, 4H). 19F NMR (376 MHz, DMSO-d6 173.25 - -173.35 (d, 1F). [1207] Example 119: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1208] Example 119 was prepared in a similar manner to Example 92. MS: m/z = 619.45 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 7.76 - 7.74 (m, 1H), 7.35 - 7.30 (m, 1H), 7.06 - 6.99 (m, 2H), 5.63 - 5.62 (m, 2H), 5.35 - 5.22 (m, 1H), 5.05 - 4.89 (m, 1H), 4.67 - 4.64 (m, 1H), 4.52 - 4.47
WSGR Docket No. 62619-720.601 (m, 1H), 4.32 - 4.26 (m, 1H), 4.20 - 4.11 (m, 1H), 4.02 - 3.84 (m, 2H), 3.70 - 3.30 (m, 3H), 3.11 - 3.02 (m, 3H), 2.90 - 2.80 (m, 1H), 2.16 - 1.75 (m, 8H). 19F NMR (376 MHz, DMSO-d6) -113.29 - -113.41 (d, 1F), -145.40 - -146.02 (d, 1F), -172.10 - -172.22 (d, 1F). [1209] Example 120: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1210] Example 120 was prepared in a similar manner to Example 92. MS: m/z = 637.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 2H), 5.35 - 5.22 (m, 1H), 5.04 - 4.89 (m, 1H), 4.73 - 4.60 (m, 1H), 4.50 - 4.46 (m, 1H), 4.30 - 4.28 (m, 1H), 4.21 - 4.05 (m, 2H), 3.93 - 3.88 (m, 1H), 3.70 - 3.60 (m, 1H), 3.59 - 3.35 (m, 2H), 3.10 - 3.02 (m, 3H), 2.85 - 2.80 (m, 1H), 2.15 - 1.77 (m, 8H). 19F NMR (376 MHz, DMSO-d6 - -138.68 (m, 2F), -145.48 - -146.07 (d, 1F), -172.09 - -172.21 (d, 1F). [1211] Example 121: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601 [1212] Example 121 was prepared in a similar manner to Example 1. MS: m/z = 638.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.31 (s, 1H), 8.01 - 7.98 (m, 1H), 7.35 - 7.34 (m, 1H), 7.17 - 7.10 (m, 1H), 5.35 - 5.22 (m, 1H), 5.05 - 4.91 (m, 1H), 4.70 - 4.60 (m, 1H), 4.53 - 4.44 (m, 1H), 4.30 - 4.11 (m, 3H), 3.95 - 3.84 (m, 1H), 3.70 - 3.36 (m, 3H), 3.13 - 2.97 (m, 3H), 2.88 - 2.78 (m, 1H), 2.15 - 1.78 (m, 8H). 19F NMR (376 MHz, DMSO-d6) -134.45 - -134.66 (d, 1F), 137.61 - - 137.68 (d, 1F), -145.56 - -146.16 (d, 1F), -172.09 - -172.21 (d, 1F). [1213] Example 122: 2-Fluoro-8-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6-hydroxy-1-naphthonitrile
[1214] Example 122 was prepared in a similar manner to Example 13. MS: m/z = 621.60 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.55 (s, 1H), 8.34 - 8.30 (m, 1H), 7.67 - 7.63 (m, 1H), 7.51 - 7.34 (m, 2H), 5.35 - 5.22 (m, 1H), 5.01 - 4.95 (m, 1H), 4.70 - 4.66 (m, 1H), 4.55 - 4.32 (m, 2H), 4.13 - 4.10 (m, 1H), 3.94 - 3.89 (m, 1H), 3.75 - 3.33 (m, 2H), 3.22 - 3.02 (m, 4H), 2.86 - 2.83 (m, 1H), 2.17 - 1.76 (m, 8H). 19F NMR (376 MHz, DMSO-d6) -106.74 - -106.89 (d, 1F), -145.78 - -146.73 (d, 1F), -172.10 - -172.16 (d, 1F). [1215] Example 123: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1216] Example 123 was prepared in a similar manner to Example 1. MS: m/z = 636.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6
WSGR Docket No. 62619-720.601 1H), 5.28 (d, J = 54.4 Hz, 1H), 4.89 (s, 1H), 4.64 - 4.48 (m, 1H), 4.47 - 4.38 (m, 1H), 4.33 - 4.01 (m, 2H), 3.33 - 3.18 (m, 1H), 3.17 - 2.95 (m, 3H), 2.91 - 2.75 (m, 1H), 2.25 - 1.43 (m, 13H), 1.43 - 1.29 (m, 1H). 19 F), -146.02 - -146.88 (d, 1F), -172.11 - -172.22 (d, 1F). [1217] Example 125: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-ol
[1218] Example 125 was prepared in a similar manner to Example 76. MS: m/z = 614.2[M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 9.2Hz, 1H), 7.38 - 7.34 (m, 1H), 7.20 - 7.10 (m, 1H), 6.06 - 5.79 (m, 2H), 5.44 - 5.18 (m, 2H), 4.66 - 4.54 (m, 1H), 4.52 - 4.44 (m, 1H), 4.36 - 4.23 (m, 1H), 4.16 - 3.82 (m, 4H), 3.15 - 3.06 (m, 2H), 3.05 - 2.98 (m, 1H), 2.87 - 2.78 (m, 1H), 2.36 - 2.21 (m, 2H), 2.16 - 2.11 (m, 1H), 2.09 - 1.97 (m, 3H), 1.90 - 1.73 (m, 4H).19F NMR (376 MHz, Dimethylsulfoxide-d6 146.42, -172.07, -172.17. [1219] Example 126: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(fluoromethoxy)naphthalen-2-amine
[1220] Step 1: N-(6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- yl)-5-(fluoromethoxy)naphthalen-2-yl)-1,1-diphenylmethanimine [1221] 2nBuP-Pd-G3 (cataCXiumAPdG3 a solution of K3PO4 2O (1 mL). The mixture was stirred at 100 °C under N2 for 2 h. The reaction mixture was concentrated under reduced pressure. The residue was
WSGR Docket No. 62619-720.601 purified by silica gel flash chromatography (eluent: 0 ~ 5% of MeOH in CH2Cl2) to give the title compound (100 mg, 55% yield) as a yellow solid. MS: m/z = 803.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 7.12 - 7.02 (m, 1H), 5.39 - 4.83 (m, 4H), 4.59 - 4.38 (m, 2H), 4.18 - 3.96 (m, 3H), 3.27 - 3.25 (m, 1H), 3.15 - 2.98 (m, 3H), 2.90 - 2.77 (m, 1H), 2.14 - 1.60 (m, 13H), 1.55 - 1.45 (m, 1H). 19F NMR (376 MHz, Dimethylsulfoxide-d6 -172.11, -172.19. [1222] Step 2: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- yl)-5-(fluoromethoxy)naphthalen-2-amine [1223] To a solution of N-(6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(fluoromethoxy)naphthalen-2-yl)-1,1-diphenylmethanimine (100 mg, 124 concentrated under reduced pressure. The residue was purified by reversed-phase column (C18, mobile phase: [water (NH4HCO3) - MeOH]; gradient: 0% ~ 50% B over 30 min) to give the title compound (Example 126, 60 mg, 74% yield) as a yellow solid. MS: m/z = 639.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 2H), 5.60 (d, J = 13.6 Hz, 2H), 5.41 - 5.06 (m, 2H), 5.05 - 4.87 (m, 2H), 4.59 - 4.40 (m, 2H), 4.13 - 4.10 (m, 2H), 4.01 (t, J = 1.2 Hz, 1H), 3.26 - 3.19 (m, 1H), 3.14 - 3.06 (m, 2H), 3.01 (s, 1H), 2.87 - 2.79 (m, 1H), 2.18 - 2.09 (m, 1H), 2.02 - 1.50 (m, 12H), 1.29 - 1.20 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 172.10, -172.18. [1224] Example 128: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-ol
[1225] A mixture of Ad2nBuP-Pd-G3 (cataCXiumAPdG3 3PO4
WSGR Docket No. 62619-720.601 dioxane (5 mL) and H2O (1 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure to give a residue. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (Example 128, 33.1mg, 41% yield) as a yellow solid. MS: m/z = 625.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (d, J = 18.8 Hz, 1H), 7.63 - 7.55 (m, 1H), 7.46 - 7.37 (m, 1H), 7.28 (t, J = 2.8 Hz, 1H), 7.10 - 6.95 (m, 1H), 5.43 - 5.20 (m, 1H), 4.96 - 4.84 (m, 1H), 4.60 - 4.42 (m, 2H), 4.24 - 4.02 (m, 3H), 3.30 - 3.08 (m, 4H), 2.95 - 2.83 (m, 1H), 2.22 - 1.97 (m, 4H), 1.93 - 1.61 (m, 9H), 1.55 - 1.48 (m, 1H). 172.28. [1226] Example 130 & 131: 4-((S,10E,12Z)-4,11-Difluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,14-tetrahydro-7-oxa-1,3,6,14a- tetraazacycloocta[4,5]cyclohepta[1,2,3-de]naphthalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol & 5-Ethynyl-6-fluoro-4-((8aS)-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1227] Step 1: 6-Fluoro-4-((8aS)-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol [1228] 3PO4 (75 2nBuP-Pd-G3 (cataCXiumAPdG3 and H2O (1 mL) was degassed and purged with N2 three times, and the mixture was stirred at 110 °C under N2 for 2 h. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of MeOH in CH2Cl2) to give the title compound (50 mg, 43% yield) as a brown solid. MS: m/z = 820.3 [M + H]+. [1229] Step 2: 4-((S,10E,12Z)-4,11-Difluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8,8a,9,14-tetrahydro-7-oxa-1,3,6,14a-tetraazacycloocta[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol & 5-ethynyl-6-fluoro-4-((8aS)-4,10,10-
WSGR Docket No. 62619-720.601 trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)- 8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a-tetraazacyclopropa[h]naphtho[1,8-ab]heptalen- 5-yl)naphthalen-2-ol [1230] To a solution of 6-fluoro-4-((8aS)-4,10,10-trifluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8,8a,9,9a,10,10a,11,12-octahydro-7-oxa-1,3,6,12a- tetraazacyclopropa[h]naphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol stirred at 20 °C for 2 h. The reaction mixture was purified by prep-HPLC (column: Waters Xbridge 3·H2O) - ACN]; gradient: 35% ~ 55% B over 10 min] to give the title compound (Example 131, 8.0 mg, 21% yield) as a white solid and the title compound (Example 130, 3.9 mg, 10% yield) as a white solid. Spectra for Example 131: MS: m/z = 664.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 7.01 - 6.85 (m, 2H), 5.40 - 5.13 (m, 2H), 4.72 - 4.17 (m, 4H), 4.15 - 4.09 (m, 1H), 4.03 - 3.95 (m, 1H), 3.91 - 3.83 (m, 1H), 3.10 - 3.06 (m, 2H), 3.01 (s, 1H), 2.87 - 2.75 (m, 2H), 2.20 - 2.10 (m, 3H), 2.03 - 1.94 (m, 2H), 1.92 - 1.72 (m, 5H), 1.53 - 1.42 (m, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 145.80, -146.20, -146.46,-147.47, -147.86, -172.04, -172.12. Spectra for Example 130: MS: m/z = 644.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 1H), 7.37 - 7.31 (m, 1H), 7.21 - 7.11 (m, 1H), 6.06 - 5.98 (m, 1H), 5.70 - 5.60 (m, 1H), 5.49 - 5.06 (m, 3H), 4.70 - 4.44 (m, 2H), 4.16 - 3.95 (m, 4H), 4.43 - 3.86 (m, 1H), 3.11 - 3.05 (m, 3H), 3.00 ( s, 1H), 2.86 - 2.78 (m, 2H), 2.20 - 1.70 (m, 10H).19F NMR (376 MHz, Dimethylsulfoxide-d6 97.35, -97.91 , -110.67, -145.17, -145.38, -146.30, -172.10. [1231] Example 135: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl-6,6-d2)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
WSGR Docket No. 62619-720.601
[1232] Example 135 was prepared in a similar manner to Example 33. MS: m/z = 618.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.36 (m, 1H), 7.20 - 7.13 (m, 1H), 5.28 (d,
= 54.4 Hz, 1H), 4.99 - 4.84 (m, 1H), 4.59 - 4.51 (m, 1H), 4.42 (d, J = 12.8 Hz, 1H), 4.18 - 3.77 (m, 4H), 3.29 - 3.18 (m, 1H), 3.16 - 2.95 (m, 3H), 2.82 (d, J = 9.2 Hz, 1H), 2.16 - 1.83 (m, 5H), 1.80 - 1.48 (m, 6H), 1.34 - 1.23 (m, 1H). 19F NMR (376 MHz, DMSO-d6 [1233] Example 136: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-yl methylcarbamate
[1234] To a solution of Example 123 (100 mg, 0.157 mmol) and TEA (47.76 mg, 0.471 mmol) in DCM (2 mL) were added N-methylcarbamoyl chloride (22.07 mg, 0.235 mmol) at room temperature. The reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was diluted with water (10 mL) and extracted with DCM (3 x 10 mL). The combined organic layers were washed with brine (10 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by Prep-TLC (CH2Cl2 / MeOH 13:1) to afford the title compound (Example 136, 73 mg, 67%) as a white lyophilized powder. MS: m/z = 693.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6 7.56 - 7.33 (m, 1H), 5.28 (d, J = 54.0 Hz, 1H), 4.96 - 4.83 (m, 1H), 4.67 - 4.48 (m, 1H), 4.48 - 4.08 (m, 3H), 3.33 - 3.18 (m, 1H), 3.17 - 3.04 (m, 2H), 3.02 (s, 1H), 2.90 - 2.78 (m, 1H), 2.71 (t, J = 4.4 Hz, 3H), 2.21 - 2.11 (m, 1H), 2.10 - 2.03 (m, 1H), 2.03 - 1.94 (m, 2H), 1.94 - 1.48 (m, 9H), 1.44 -
WSGR Docket No. 62619-720.601 1.24 (m, 1H). 19 -145.74 - -146.50 (d, 1F), -172.11 - -172.22 (d, 1F). [1235] Example 137: Methyl (5-ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-yl)carbamate
[1236] Step 1: Methyl (6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)carbamate [1237] To an ice-cooled solution of 6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine (refer to Example 89 for detail procedures, 106.4 mg, 0.13 mmol) in DCM (1 mL) were added DIEA (52.68 mg, 0.41 mmol) and dimethyl dicarbonate (36.25 mg, 0.27 mmol) dropwise under nitrogen atmosphere. The ice bath was removed, and the reaction mixture was stirred at room temperature for 16 hours. The resulting mixture was diluted with H2O (5 mL) and extracted with EtOAc (3 x 15 mL). The combined organic layers were washed wish brine (5 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (110 mg, crude used through) as a light yellow solid. MS: m/z = 849.45 [M + H]+. [1238] Step 2: Methyl (5-ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-yl)carbamate [1239] A solution of methyl (6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)carbamate (110 mg, crude) and CsF (327.47 mg, 2.15 mmol) in DMF (1.2 mL) under nitrogen atmosphere was stirred at room temperature for 16 hours. The resulting mixture was filtered and purified with the following conditions: Column: X-Bridge Prep OBD C18 Column 19 x 150 mm, 5 um; Mobile Phase A: 10 mM aq. NH4HCO3; Mobile Phase B: MeCN; Flow rate: 20 mL/min; Gradient:
WSGR Docket No. 62619-720.601 isocratic 55% - 65%; Detector: UV 254 nm & 220 nm. The product-containing fractions were collected, concentrated and then lyophilized overnight to give the title compound (Example 137, 23.1 mg, 23% yield) as an off-white lyophilized powder. MS: m/z = 693.45 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.17 - 10.11 (m, 1H), 8.26 (s, 1H), 8.18 - 8.13 (m, 1H), 7.66 - 7.57 (m, 1H), 5.35 - 5.21 (m, 1H), 4.95 - 4.82 (m, 1H), 4.64 - 4.39 (m, 2H), 4.33 - 4.09 (m, 2H), 3.73 (s, 3H), 3.32 - 3.21 (m, 1H), 3.13 - 3.01 (m, 3H), 2.89 - 2.82 (m, 1H), 2.17 - 1.48 (m, 13H), 1.46 - 1.23 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -132.32 - -132.52 (m, 1F), -136.96 - -137.04 (m, 1F), - 145.94 - -146.77 (d, 1F), -172.11 - -173.21 (d, 1F). [1240] Example 139: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,6S,7aS)-2-fluoro-6- methyltetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-
2-ol
[1241] Example 139 was prepared in a similar manner to Example 33. MS: m/z = 630.20 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.14 - 10.11 (m, 1H), 7.98 - 7.94 (m, 1H), 7.48 - 7.43 (m, 1H), 7.37 (s, 1H), 7.20 - 7.12 (m, 1H), 5.38 - 5.24 (m, 1H), 4.95 - 4.85 (m, 1H), 4.60 - 4.50 (m, 1H), 4.44 - 4.41 (m, 1H), 4.18 - 3.82 (m, 4H), 3.29 - 2.87 (m, 4H), 2.36 - 1.76 (m, 6H), 1.76 - 1.15 (m, 7H), 0.98 - 0.96 (m, 3H). 19F NMR (376 MHz, DMSO-d6) -110.66 - -110.80 (d, 1F), -145.97 - -146.88 (d, 1F), -171.32 - -171.38 (d, 1F). [1242] Example 140: 2,3-Difluoro-8-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6-hydroxy-1-naphthonitrile
WSGR Docket No. 62619-720.601 [1243] Example 140 was prepared in a similar manner to Example 13. MS: m/z = 637.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.63 (s, 1H), 8.40 - 8.36 (m, 1H), 7.46 - 7.31 (m, 2H), 5.35 - 5.22 (m, 1H), 4.94 - 4.91 (m, 1H), 4.62 - 4.32 (m, 2H), 4.14 - 4.12 (m, 1H), 3.33 - 3.02 (m, 3H), 2.90 - 2.84 (m, 1H), 2.20 - 1.43 (m, 14H), 1.19 - 1.09 (m, 1H). 19F NMR (376 MHz, DMSO- d6) -129.29 - -129.51 (m, 1F), -136.79 - -136.85 (m, 1F), -146.24 - -147.34 (m, 1F), -172.11 - - 172.17 (m, 1F). [1244] Example 141: 1-Ethynyl-8-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6-hydroxy-2-naphthonitrile
[1245] Example 141 was prepared in a similar manner to Example 1. MS: m/z = 625.25 [M + H]+. 1 7.42 (m, 1H), 7.30 - 7.23 (m, 1H), 5.36 - 5.22 (m, 1H), 4.95 - 4.83 (m, 1H), 4.63 - 4.49 (m, 1H), 4.48 - 4.28 (m, 2H), 4.18 - 4.08 (m, 1H), 3.31 - 3.19 (m, 1H), 3.17 - 2.97 (m, 3H), 2.89 - 2.78 (m, 1H), 2.24 - 1.44 (m, 13H), 1.42 - 1.13 (m, 1H). 19 146.93 (d, 1F), -172.11 -172.23 (d, 1F). [1246] Example 142 & 143: (2S,6R,7aS)-7a-((((S)-5-(8-ethynyl-7-fluoro-3-hydroxynaphthalen-1- yl)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2- yl)oxy)methyl)-6-fluoro-2-methylhexahydro-1H-pyrrolizin-2-ol & (2R,6R,7aS)-7a-((((S)-5-(8- Ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluoro-2-methylhexahydro-1H- pyrrolizin-2-ol
WSGR Docket No. 62619-720.601
[1247] Step 1: (6R,7aS)-6-Fluoro-7a-((((S)-4-fluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-2-methylhexahydro-1H-pyrrolizin-2-ol [1248] To an ice-cooled solution of (6R,7aS)-6-fluoro-7a-(hydroxymethyl)-2-methyl-tetrahydro- 1H-pyrrolizin-2-ol (81 mg, 0.42 mmol) in THF (1.5 mL) under nitrogen atmosphere was added NaH (16 mg, 0.40 mmol, 60% dispersion in mineral oil). The mixture was stirred in an ice bath for 30 min. (S)-2,4-Difluoro-5-(7-fluoro-3-(methoxymethoxy)-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalene (150 mg, 0.222 mmol) in THF (0.5 mL) was added in an ice bath. The ice bath was removed, and the reaction mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aqueous NH4Cl (15 mL) and extracted with EtOAc (3 x 20 mL). The combined organic layers were washed with brine (10 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by Prep-TLC (DCM : MeOH = 20 : 1) to afford the title compound (63 mg, 33% yield) as a brown yellow solid. MS: m/z = 846.45 [M + H]+. [1249] Step 2: (6R,7aS)-6-Fluoro-7a-((((S)-4-fluoro-5-(7-fluoro-3-hydroxy-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-2-methylhexahydro-1H-pyrrolizin-2-ol [1250] To an ice-cooled solution of (6R,7aS)-6-fluoro-7a-((((S)-4-fluoro-5-(7-fluoro-3- (methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H- 7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-2-methylhexahydro-1H- pyrrolizin-2-ol (60 mg, 0.07 mmol) in CH3CN (4.75 mL) was added HCl (4 M in 1,4-dioxane, 0.70 mL, 2.81 mmol). The reaction mixture was stirred in an ice bath for 1 hour. The resulting mixture
WSGR Docket No. 62619-720.601 was quenched with saturated aqueous NaHCO3 (20 mL), extracted with EtOAc (3 x 20 mL). The combined organic layers were washed with brine (2 mL), dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the title compound (78 mg, crude used through) as a light-yellow solid. MS: m/z = 802.40 [M + H]+. [1251] Step 3: (2S,6R,7aS)-7a-((((S)-5-(8-ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4-fluoro- 8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2- yl)oxy)methyl)-6-fluoro-2-methylhexahydro-1H-pyrrolizin-2-ol & (2R,6R,7aS)-7a-((((S)-5-(8- ethynyl-7-fluoro-3-hydroxynaphthalen-1-yl)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-6-fluoro-2-methylhexahydro-1H- pyrrolizin-2-ol [1252] To a solution of (6R,7aS)-6-fluoro-7a-((((S)-4-fluoro-5-(7-fluoro-3-hydroxy-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-2-yl)oxy)methyl)-2-methylhexahydro-1H-pyrrolizin-2-ol (78 mg, crude) in DMF (0.8 mL) was added CsF (232 mg, 1.52 mmol) at room temperature. The reaction mixture was stirred at room temperature for 1.5 hours. The resulting mixture was filtered and purified by RP-Flash with following conditions: column, C18; Mobile phase A: 5 mM aq. NH4HCO3; Mobile phase B: MeCN; Flow rate: 40 mL/min; Gradient: 0% B to 64% B in 30 min, 64% B hold 3 min; Detector, UV 254 & 210 nm. The collected fractions were combined, concentrated and then lyophilized overnight to give a crude product. The crude product was purified by Prep-HPLC with following conditions: Column: X-Bridge Prep OBD C18 Column 19 x150 mm, 5 um; Mobile Phase A: 10 mM aq. NH4HCO3; Mobile Phase B: MeCN; Flow rate: 20 mL/min; Gradient: isocratic 40% - 50%; Detector: UV 254 & 210 nm; RT1: 5.0 min; RT2: 6.2 min. The first eluting peak (RT1: 5.0 min) was concentrated and lyophilized to give the title compound (Example 143, 9.7 mg, 14% yield) as an off-white lyophilized powder. MS: m/z = 646.25 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.14 - 10.12 (m, 1H), 7.98 - 7.94 (m, 1H), 7.48 - 7.36 (m, 2H), 7.20 - 7.13 (m, 1H), 5.44 - 5.25 (m, 1H), 4.97 - 4.82 (m, 1H), 4.73 - 4.69 (m, 1H), 4.61 - 4.52 (m, 1H), 4.46 - 4.39 (m, 1H), 4.13 - 3.81 (m, 4H), 3.30 - 3.09 (m, 3H), 3.03 - 2.97 (m, 1H), 2.87 - 2.78 (m, 1H), 2.41 - 2.16 (m, 2H), 2.11 - 1.84 (m, 4H), 1.80 - 1.44 (m, 5H), 1.41 - 1.21 (m, 4H). 19F NMR (376 MHz, DMSO-d6) -110.65 - -110.79 (d, 1F), -145.90 - -146.77 (d, 1F), -173.52 - - 174.19 (d, 1F). The second eluting peak (RT2: 6.2 min) was concentrated and lyophilized to give the title compound (Example 142, 14.4 mg, 21% yield) as an off-white lyophilized powder. MS: m/z = 646.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.06 (bs, 1H), 7.96 - 7.94 (m, 1H), 7.48 - 7.43 (m, 1H), 7.36 (s, 1H), 7.20 - 7.13 (m, 1H), 5.39 - 5.26 (m, 1H), 4.98 - 4.86 (m, 1H),
WSGR Docket No. 62619-720.601 4.63 - 4.40 (m, 4H), 4.20 - 3.82 (m, 3H), 3.30 - 3.20 (m, 1H), 3.10 - 2.88 (m, 4H), 2.19 - 1.84 (m, 6H), 1.79 - 1.51 (m, 5H), 1.28 - 1.21 (m, 4H). 19F NMR (376 MHz, DMSO-d6) -110.66 - -110.83 (m, 1F), -145.97 - -146.82 (m, 1F), -173.11 - -173.14 (m, 1F). [1253] Example 144: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine
[1254] A mixture of Intermediate 93 Ad2nBuP-Pd-G3 (cataCXiumAPdG3 3PO4 dioxane (5 mL) and H2O (1 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The reaction mixture was purified by reversed-phase column (C18, mobile phase: [water (NH4HCO3) - MeOH]; gradient: 5% ~ 45% B over 30 min) to give the title compound (Example 144, 12.2 mg, 21% yield) as a white solid. MS: m/z = 624.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 2H), 5.53 (d, J = 14.8 Hz, 2H), 5.40 - 5.19 (m, 1H), 4.99 - 4.85 (m, 1H), 4.62 - 4.41 (m, 2H), 4.20 - 4.00 (m, 3H), 3.28 - 3.22 (m, 1H), 3.20 - 2.99 (m, 3H), 2.92 - 2.81 (m, 1H), 2.20 - 1.56 (m, 14H). [1255] Example 145: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-amine
[1256] Step 1: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-amine [1257] K3PO4 2nBuP-Pd-G3 (cataCXiumAPdG3
WSGR Docket No. 62619-720.601 (2 mL) and H2O (0.2 mL) was degassed and purged with N2 three times, and then the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 5% of MeOH in CH2Cl2) to give the title compound (84 mg, 84% yield) as a yellow solid. MS: m/z = 769.4 [M + H]+. [1258] Step 2: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- yl)naphthalen-2-amine [1259] To a solution of 6-fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5- added CsF (158 mg, 1.04 mmol). The mixture was stirred at 25 °C for 2 h. The reaction mixture was purified by reversed-phase column (column: C18; mobile phase: [Water (NH4HCO3) - MeOH); B%: 0% - 65%, 20 min) to give the title compound (Example 145, 19.4 mg, 30% yield) as an off- white solid. MS: m/z = 613.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 1H), 7.32 (t, J = 9.2 Hz, 1H), 7.07 - 6.97 (m, 2H), 6.05 - 5.80 (m, 2H), 5.62 (d, J = 9.2 Hz, 2H), 5.45 - 5.16 (m, 2H), 4.64 - 4.43 (m, 2H), 4.36 - 4.22 (m, 1H), 4.17 - 4.09 (m, 1H), 4.03 - 3.79 (m, 3H), 3.16 - 3.06 (m, 2H), 3.05 - 2.99 (m, 1H), 2.87 - 2.79 (m, 1H), 2.32 - 2.19 (m, 2H), 2.16 - 2.10 (m, 1H), 2.08 - 1.97 (m, 3H), 1.88 - 1.73 (m, 4H).19F NMR (376 MHz, Dimethylsulfoxide-d6 113.35, -113.44, -145.60, -146.32, -172.07, -172.17. [1260] Example 147: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl-9,9-d2)naphthalen-2-amine
WSGR Docket No. 62619-720.601 [1261] Example 147 was prepared in a similar manner to Example 92. MS: m/z = 639.3 [M + H]+. 1H NMR (400 MHz, Methanol-d4 4.77 - 4.67 (m, 1H), 4.57 (d, J = 13.2 Hz, 1H), 4.43 - 4.27 (m, 1H), 4.03 - 3.91 (m, 1H), 3.72 - 3.34 (m, 6H), 3.18 - 3.03 (m, 1H), 2.39 - 2.15 (m, 4H), 2.09 - 1.92 (m, 4H).19F NMR (400 MHz, Methanol-d4 146.34, -173.74, -173.79. [1262] Example 148: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-amine
[1263] Example 148 was prepared in a similar manner to Example 92. MS: m/z = 633.2 [M + H]+. 1HNMR (400 MHz, Dimethylsulfoxide-d6 (m, 4H), 5.38 - 5.19 (m, 1H), 4.96 (t, J = 13.4 Hz, 1H), 4.64 - 4.51 (m, 1H), 4.49 - 4.36 (m, 1H), 4.30 (d, J = 10.2 Hz, 1H), 4.23 - 4.00 (m, 1H), 3.14 - 3.00 (m, 4H), 2.86 - 2.80 (m, 1H), 2.45 - 2.26 (m, 2H), 2.26 - 1.65 (m, 8H).19F NMR (376 MHz, Dimethylsulfoxide-d6 138.62, -138.70, -145.82, -146.23, -172.08, -172.20. [1264] Example 149: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(fluoromethoxy-d2)naphthalen-2-ol
[1265] Ad2nBuP-Pd-G3 (cataCXium®APdG3 3PO4
WSGR Docket No. 62619-720.601 dioxane (5 mL) and H2O (1 mL) was degassed and purged with N2 three times, and the mixture was stirred at 110 °C for 1 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 5% of MeOH in CH2Cl2) to give the title compound (Example 149, 42.8 mg, 32% yield) as a yellow solid. MS: m/z = 642.2 [M + H]+.1 10.18 - 10.07 (m, 1H), 7.84 - 7.63 (m, 1H), 7.53 - 7.44 (m, 1H), 7.34 (s, 1H), 7.24 - 7.03 (m, 1H), 5.39 - 5.14 (m, 1H), 4.96 - 4.79 (m, 1H), 4.61 - 4.41 (m, 2H), 4.17 - 4.00 (m, 3H), 3.23 - 2.98 (m, 4H), 2.91 - 2.80 (m, 1H), 2.20 - 2.12 (m, 1H), 2.04 - 1.51 (m, 13H).19 134.5, -134.39, -134.73, -134.77, -146.00, -146.38, -150.39, -150.98, -151.10, -172.14, -172.22. [1266] Example 151: 4-((S)-2-(((1R,6R,7aS)-1,6-Difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-ethynyl-6,7-difluoronaphthalen-2-amine
[1267] Step 1: (S)-N-(4-(2,4-Difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-6,7-difluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)- 1,1-diphenylmethanimine [1268] To a stirred mixture of (S)-5-chloro-2,4-difluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene (refer to Intermediate 23 for detail procedures, 200 mg, 0.612 mmol) and Intermediate 42 (715.86 mg, 1.102 mmol) in THF (12 mL) and water (2.4 mL) under nitrogen atmosphere were added CataCXium A Pd G3 (89.16 mg, 0.122 mmol) and K3PO4 (779.60 mg, 3.672 mmol) at room temperature. The reaction mixture was heated at 80 °C for 3 hours. The resulting mixture was cooled to room temperature, diluted with H2O (25 mL), extracted with DCM (3 x 25 mL). The combined organic layers were washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by
WSGR Docket No. 62619-720.601 silica gel flash chromatography, eluted with 0-60% EtOAc in PE to afford the title compound (185 mg, 37% yield) as a light yellow solid. MS: m/z = 814.10 [M + H]+. [1269] Step 2: N-(4-((S)-2-(((1R,6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6,7-difluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine [1270] To an ice-cooled solution of Intermediate 67 (52.24 mg, 0.295 mmol) in THF (2 mL) was added NaH (15.72 mg, 0.394 mmol, 60% dispersion in mineral oil) under nitrogen atmosphere. The mixture was stirred in an ice bath for 30 min. (S)-N-(4-(2,4-difluoro-8a,9,10,11,12,13- hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-6,7-difluoro-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine (160 mg, 0.197 mmol) was added to the above mixture. The ice bath was removed, and the resulting mixture was stirred at room temperature for 1 hour. The resulting mixture was quenched with saturated aq. NH4Cl (10 mL) in an ice bath, then extracted with DCM (3 x 15 mL). The combined organic layers were washed with brine (15 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography, eluted with 0-15% EA in DCM to give the title compound (80 mg, 41% yield) as a yellow solid. MS: m/z = 971.55 [M + H]+. [1271] Step 3: 4-((S)-2-(((1R,6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-6,7- difluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1272] To a stirred solution of N-(4-((S)-2-(((1R,6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-6,7-difluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)- 1,1-diphenylmethanimine (87 mg, 0.090 mmol) in EtOH (1.5 mL) under nitrogen atmosphere were added NaOAc (14.70 mg, 0.180 mmol) and NH2OH.HCl (12.45 mg, 0.180 mmol) at room temperature. The reaction mixture was stirred at room temperature for 2 hours. The resulting mixture was concentrated under reduced pressure to give the title compound (110 mg, crude used through) as a yellow solid. MS: m/z = 807.40 [M + H]+. [1273] Step 4: 4-((S)-2-(((1R,6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-5- ethynyl-6,7-difluoronaphthalen-2-amine
WSGR Docket No. 62619-720.601 [1274] A mixture of 4-((S)-2-(((1R,6R,7aS)-1,6-difluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-6,7-difluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine (110 mg, crude) and CsF (206.12 mg, 1.350 mmol) in DMF (1 mL) was stirred at room temperature for 1 hour. The resulting mixture was filtered and purified by RP-Flash with the following conditions: Column: 4HCO3, Mobile Phase B: MeCN; Gradient: 0% B hold 5 min, up to 95% B within 30 min, 95% B hold 5 min; Flow rate: 25 mL/min; Detector: UV 254 & 210 nm. The product-containing fractions were collected, concentrated and then lyophilized overnight to give the title compound (Example 151, 28 mg, 47% yield for two steps) as a yellow lyophilized powder. MS: m/z = 651.20 [M + H]+. 1H NMR (400 MHz, DMSO- d6 (m, 2H), 4.63 - 4.39 (m, 2H), 4.23 - 3.91 (m, 4H), 3.33 - 3.18 (m, 1H), 3.16 - 2.90 (m, 4H), 2.49 - 2.31 (m, 1H), 2.30 - 1.80 (m, 5H), 1.79 - 1.44 (m, 5H), 1.43 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6 178.32 - -178.37 (m, 1F). [1275] Example 153: 5-Ethynyl-6,7-difluoro-4-((S)-4,12,12-trifluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
[1276] Example 153 was prepared in a similar manner to Example 92. MS: m/z = 673.3 [M + H]+. 1H NMR (400 MHz, Methanol-d4 J = 14.8 Hz, 1H), 5.45 - 5.18 (m, 1H), 4.80 - 4.74 (m, 1H), 4.73 - 4.72 (m, 1H), 4.62 - 4.56 (m, 1H), 4.45 - 4.32 (m, 2H), 4.14 - 4.05 (m, 1H), 3.94 - 3.69 (m, 3H), 3.61 (d, J = 14.0 Hz, 1H), 3.39 - 3.32 (m, 1H), 3.29 - 3.23 (m, 2H), 3.09 - 3.01 (m, 1H), 2.39 - 2.21 (m, 2H), 2.16 - 2.06 (m, 1H), 2.05 - 1.98 (m, 2H), 1.96 - 1.86 (m, 1H).19F NMR (376 MHz, Methanol-d4
WSGR Docket No. 62619-720.601 102.75, -103.34, -103.42, -103.02, -137.55, -137.60, -137.69, -140.27, -140.33, -145.00, -145.45, - 173.66. [1277] Example 157: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((6R,7aR)-6-fluoro-1- methylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1278] Example 157 was prepared in a similar manner to Example 33. MS: m/z = 628.40 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.62 - 10.10 (m, 1H),7.95 - 7.93 (m, 1H), 7.47 - 7.42 (m, 1H), 7.35 - 7.34 (m, 1H), 7.19 - 7.12 (m, 1H), 5.37 - 5.23 (m, 1H), 5.04 - 5.00 (m, 2H), 4.94 - 4.85 (m, 1H), 4.55 - 4.51 (m, 1H), 4.44 - 4.41 (m, 1H), 4.19 - 3.80 (m, 4H), 3.33 - 3.05 (m, 4H), 2.83 - 2.81 (m, 1H),2.51 - 2.45 (m, 2H), 2.40 - 2.13 (m, 2H), 2.05 - 1.79 (m, 2H)1, 1.69 - 1.50 (m, 5H), 1.30 - 1.20 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.86 - -110.99 (d, 1F), -145.88 - -146.70 (d, 1F), -168.67 - -168.76 (d, 1F). [1279] Example 159: 4-((8R,8aS)-8-Ethyl-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-ethynyl-6,7-difluoronaphthalen-2-amine
WSGR Docket No. 62619-720.601 [1280] Example 159 was prepared in a similar manner to Example 92. MS: m/z = 665.0 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 5.39 - 5.17 (m, 1H), 5.06 - 4.92 (m, 1H), 4.50 - 4.39 (m, 1H), 4.32 - 3.86 (m, 4H), 3.62 - 3.44 (m, 2H), 3.13 - 2.93 (m, 4H), 2.87 - 2.79 (m, 1H), 2.13 - 1.95 (m, 4H), 1.88 - 1.66 (m, 6H), 1.02 - 0.95 (m, 3H). 19F NMR (400 MHz, Dimethylsulfoxide-d6 138.58, -138.64, -138.71, -145.63, -146.62, -172.09, -172.21. [1281] Example 160: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl-8,8-d2)naphthalen-2-amine
[1282] Example 160 was prepared in a similar manner to Example 92. MS: m/z = 639.1 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 7.77 (dd, J = 11.6, 8.4 Hz, 1H), 7.05 - 6.92 (m, 2H), 5.78 (d, J = 6.0 Hz, 2H), 5.41 - 5.15 (m, 1H), 5.05 - 4.87 (m, 1H), 4.70 - 4.42 (m, 1H), 4.31 - 4.25 (m, 1H), 4.21 - 4.03 (m, 1H), 3.94 - 3.83 (m, 1H), 3.69 - 3.38 (m, 3H), 3.12 - 3.04 (m, 2H), 3.01 (s, 1H), 2.87 - 2.78 (m, 1H), 2.12 - 1.76 (m, 8H).19F NMR (400 MHz, Dimethylsulfoxide-d6 137.59, -137.66, -137.74, -137.82, -138.62, -138.68, 145.53, -146.05, -146.10, -172.06, -172.09, - 172.21. [1283] Example 161: 4-((8aS)-2-(((6R,7aR)-2,6-Difluoro-2-methyltetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)-4-fluoro-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
WSGR Docket No. 62619-720.601
[1284] Example 161 was prepared in a similar manner to Example 33. MS: m/z = 648.25 [M + H]+. 1 7.37 - 7.36 (m, 1H), 7.21 - 7.12 (m, 1H), 5.45 - 5.27 (m, 1H), 5.00 - 4.83 (m, 1H), 4.56 - 4.51 (m, 1H), 4.44 - 4.40 (m, 1H), 4.29 - 4.25 (m, 1H), 4.12 - 3.84 (m, 3H), 3.46 - 3.34 (m, 1H), 3.29 - 2.95 (m, 4H), 2.46 - 1.80 (m, 6H), 1.78 - 1.20 (m, 9H). 19 110.80 (m, 1F), -137.97 - -138.10 (m, 1F), -145.85 - -146.85 (m, 1F), -174.06 (s, 1F). [1285] Example 168: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl-13,13-d2)naphthalen-2-amine
[1286] Example 168 was prepared in a similar manner to Example 92. MS: m/z = 639.0 [M + H]+. 1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 5.40 - 5.18 (m, 1H), 4.70 - 4.59 (m, 1H), 4.40 (d, J = 13.6 Hz, 1H), 4.33 - 4.24 (m, 1H), 4.21 - 4.02 (m, 2H), 3.95 - 3.86 (m, 1H), 3.69 - 3.51 (m, 1H), 3.48 - 3.42 (m, 1H), 3.19 - 2.99 (m, 3H), 2.89 - 2.77 (m, 1H), 2.11 - 1.97 (m, 4H), 1.92 - 1.76 (m, 4H), 19F NMR (376 MHz, Dimethylsulfoxide-d6 172.118, -172.230.
WSGR Docket No. 62619-720.601 [1287] Example 172: 6-Fluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine
[1288] K3PO4 2nBuP-Pd-G3 (cataCXiumAPdG3 dioxane (2.5 mL) and H2O (0.5 mL) was degassed and purged with N2 three times, and the mixture was stirred at 100 °C for 0.5 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The reaction mixture was purified by reversed-phase column: (column C18; mobile phase: [Water (NH3·H2O) - ACN)]; B%: 0% ~ 54%, 18 min) and silica gel flash chromatography (eluent: 0 ~ 6% of MeOH in CH2Cl2) to give the title compound (Example 172, 40.9 mg, 25% yield) as an off-white solid. MS: m/z = 624.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 (m, 2H), 5.03 - 4.93 (m, 2H), 5.41 - 5.21 (m, 1H), 5.03 - 4.91 (m, 1H), 4.65 - 4.42 (m, 2H), 4.36 - 4.24 (m, 1H), 3.39 - 3.34 (m, 1H), 3.23 - 3.06 (m, 3H), 2.93 - 2.83 (m, 1H), 2.47 - 2.30 (m, 2H), 2.25 - 1.98 (m, 3H), 1.93 - 1.74 (m, 3H).19F NMR (376 MHz, Dimethylsulfoxide-d6 138.65, -146.30, -146.51, -172.21. [1289] Example 182: 6,7-Difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine
[1290] Example 182 was prepared in a similar manner to Example 172. MS: m/z = 644.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 5.60 (m, 2H), 5.40 - 5.20 (m, 1H), 4.99 - 4.83 (m, 1H), 4.61 - 4.50 (m, 1H), 4.49 - 4.41 (m, 1H), 4.18 - 4.05 (m, 1H), 3.22 - 2.99 (m, 4H), 2.87 - 2.77 (m, 1H), 2.20 - 2.08 (m, 1H), 2.07 - 2.02 (m,
WSGR Docket No. 62619-720.601 1H), 2.01 - 1.83 (m, 4H), 1.79 - 1.59 (m, 6H), 1.56 - 1.44 (m, 1H), 1.28 - 1.21 ( s, 1H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1291] Example 183: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl-13,13-d2)naphthalen-2-amine
[1292] Step 1: 6,7-Difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen- 5-yl-13,13-d2)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1293] K3PO4 2nBuP-Pd-G3 (cataCXiumAPdG3 dioxane (2 mL) and H2O (0.2 mL) was degassed and purged with N2 three times, and the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (95 mg, 59% yield) as a yellow solid. MS: m/z = 793.4 [M + H]+. [1294] Step 2: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl-13,13-d2)naphthalen-2-amine [1295] To a solution of 6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl-13,13-d2 reaction mixture was purified by reversed-phase column (column: C18; mobile phase: [Water (NH4HCO3) - MeOH); B%: 0% - 71%, 23 min) to give 5-ethynyl-6,7-difluoro-4-((S)-4-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,10,11,12,13-hexahydro- 8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl-13,13-d2)naphthalen-2-amine (Example 183, 36.6 mg, 51% yield) as a yellow solid. MS: m/z = 637.4 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 11.6 Hz, 2H), 5.43 - 5.10 (m, 1H), 4.61 - 4.47 (m, 1H), 4.46 - 4.35 (d, 1H), 4.23 - 3.88 (m, 2H), 3.14 - 3.00 (m, 3H),
WSGR Docket No. 62619-720.601 2.90 - 2.74 (m, 1H), 2.18 - 1.86 (m, 6H), 1.82 - 1.43 (m, 8H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1296] Example 184: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl-8,8,13,13-d4)naphthalen-2-amine
[1297] Example 184 was prepared in a similar manner to Example 183. MS: m/z = 637.2 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 8.4, 11.6 Hz, 1H), 7.08 - 6.89 (m, 2H), 5.97 - 5.59 (m, 4H), 5.38 - 5.18 (m, 1H), 4.34 - 4.24 (m, 1H), 4.22 - 4.05 (m, 1H), 3.15 - 2.97 (m, 3H), 2.89 - 2.77 (m, 1H), 2.48 - 2.30 (m, 3H), 2.20 - 1.70 (m, 7H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1298] Example 186 &187: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa- 1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine & 5-Ethynyl-6,7-difluoro-4- ((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)- 8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen- 2-amine
[1299] Step 1: N-(6,7-Difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen- 5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine
WSGR Docket No. 62619-720.601 [1300] CataCXium A Pd G3 3PO4 (623 mg, 2.94 mmol) in THF (10 mL) and H2O (2 mL) was degassed and purged with N2 three times, and the mixture was stirred at 80 °C for 3 hr under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 6% MeOH in CH2Cl2), the title compound (280 mg, yield: 48%) was obtained as a yellow solid. MS: m/z = 957.4 [M + H]+.1H NMR (400 MHz, Chloroform-d J = 7.2 Hz, 2H), 7.50 - 7.26 (m, 5H), 7.25 - 6.90 (m, 6H), 5.40 - 5.15 (m, 1H), 4.59 - 4.31 (m, 2H), 4.30 - 3.41 (m, 6H), 3.40 - 3.16 (m, 4H), 3.03 - 2.93 (m, 1H), 2.30 - 2.20 (m, 2H), 2.00 - 1.84 (m, 2H), 1.73 - 1.60 (m, 4H), 0.94 - 0.84 (m, 18H), 0.77 - 0.62 (m, 3H).19F NMR (376 MHz, Chloroform-d 141.27, -142.73, -142.82, -173.23, -173.30. [1301] Step 2: 6,7-Difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen- 5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1302] To a solution of N-(6,7-difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine (280 2OH HCl (40.6 under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 6% MeOH in CH2Cl2), the title compound (210 mg, yield: 89%) was obtained as a yellow solid. MS: m/z = 793.3 [M + H]+.1H NMR (400 MHz, Chloroform-d 2H), 5.37 - 5.18 (m, 2H), 4.61 - 4.53 (m, 1H), 4.49 - 4.37 (m, 1H), 4.16 - 3.99 (m, 3H), 3.91 - 3.83 (m, 1H), 3.79 - 3.73 (m, 1H), 3.71 - 3.59 (m, 1H), 3.45 - 3.15 (m, 5H), 3.04 - 2.94 (m, 1H), 2.32 - 2.21 (m, 2H), 2.19 - 2.14 (m, 2H), 2.06 - 1.94 (m, 4H), 0.93 - 0.86 (m, 18H), 0.77 - 0.62 (m, 3H). 19F NMR (376 MHz, Chloroform-d 142.18, -173.03, -173.12, -173.18. [1303] Step 3: 5-Ethynyl-6,7-difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)naphthalen-2-amine [1304] To a solution of 6,7-difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab
WSGR Docket No. 62619-720.601 reaction mixture was diluted with H2O (20 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with H2O (20 mL x 3), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 10% MeOH in CH2Cl2), the title compound (150 mg, yield: 85%) was obtained as a yellow solid. MS: m/z = 637.3 [M + H]+. [1305] Step 4: 5-Ethynyl-6,7-difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine & 5-Ethynyl-6,7-difluoro-4-((R)-4- fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8,8a,9,10,12,13- hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine [1306] 5-Ethynyl-6,7-difluoro-4-(4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-8,8a,9,10,12,13-hexahydro-7,11-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen- 5-yl)naphthalen-2-amine(150 mg) was purified by SFC (column: DAICEL CHIRALCEL OD (250 mm * 50 mm, 10 m); mobile phase: [Hexane – EtOH (0.1%IPAm)]; B%:30%, isocratic elution mode) to give the title compound (Example 186, 58.1 mg, yield: 75%) as a yellow powder and the title compound (Example 187, 52.5 mg, yield: 70%) as a yellow powder. Spectra for Example 186: MS: m/z = 637.4 [M + H]+.1 J = 11.6, 8.4 Hz, 1H), 7.04 - 6.95 (m, 2H), 5.77 (d, J = 10.8 Hz, 2H), 5.39 - 5.18 (m, 1H), 5.10 - 4.94 (m, 1H), 4.62 - 4.45 (m, 2H), 4.37 - 4.24 (m, 1H), 4.23 - 3.97 (m, 1H), 3.97 - 3.83 (m, 2H), 3.79 - 3.65 (m, 1H), 3.61 - 3.41 (m, 2H), 3.12 - 3.00 (m, 3H), 2.87 - 2.79 (m, 1H), 2.11 - 1.99 (m, 4H), 1.92 - 1.76 (m, 4H).19F NMR (376 MHz, Dimethylsulfoxide-d6 -145.69, -146.51, -172.08, -172.20. Spectra for Example 187: MS: m/z = 637.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 11.6, 8.8 Hz, 1H), 7.05 - 6.94 (m, 2H), 5.77 (d, J = 10.0 Hz, 2H), 5.39 - 5.18 (m, 1H), 5.08 - 4.95 (m, 1H), 4.62 - 4.46 (m, 2H), 4.35 - 4.25 (m, 1H), 3.97 (s, 1H), 3.95 - 3.83 (m, 2H), 3.75 - 3.65 (m, 1H), 3.59 - 3.48 (m, 2H), 3.10 - 2.94 (m, 3H), 2.87 - 2.78 (m, 1H), 2.13 - 2.00 (m, 4H), 1.91 - 1.75 (m, 4H).19F NMR (376 MHz, Dimethylsulfoxide-d6 172.06, -172.18. [1307] Example 198: 6,7-Difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine
WSGR Docket No. 62619-720.601
[1308] 3PO4 (333 mg, 1.02 mmol) in 1,4-dioxane (1 mL) and H2O (0.2 mL) was degassed and purged with N2 three times, and the mixture was stirred at 80 °C for 3 hr under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 7% MeOH in CH2Cl2), the title compound (Example 198, 67.4 mg, yield: 45%) was obtained as a yellow solid. MS: m/z = 646.4 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 12.0, 7.6 Hz, 1H), 6.93 - 6.73 (m, 2H), 5.79 - 5.58 (m, 2H), 5.38 - 5.20 (m, 1H), 5.07 - 4.93 (m, 1H), 4.71 - 4.58 (m, 1H), 4.57 - 4.47 (m, 1H), 4.37 - 4.24 (m, 1H), 4.18 - 4.06 (m, 1H), 3.95 - 3.85 (m, 1H), 3.69 - 3.51 (m, 1H), 3.49 - 3.38 (m, 2H), 3.17 - 2.98 (m, 3H), 2.90 - 2.78 (m, 1H), 2.17 - 1.97 (m, 4H), 1.93 - 1.74 (m, 4H).19 161.836, -172.148, -172.193. [1309] Example 199: 6,7-Difluoro-4-((S)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7-oxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine
[1310] K3PO4 (273 mg, 1.29 mmol) and Ad2nBuP-Pd-G3 (cataCXiumAPdG3 dioxane (10 mL) and H2O (2 mL) was degassed and purged with N2 for 3 times, and then the mixture was stirred at 110 °C for 0.5 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give 6,7-difluoro-4-((S)-4-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy-d2)-8a,9,12,13-tetrahydro-8H-7- oxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-5-yl)-5-(methoxy-d3)naphthalen-2-amine (Example 199, 116.7 mg, 41% yield) as a yellow solid. MS: m/z = 642.1 [M + H]+.1HNMR (400 MHz, Dimethylsulfoxide-d6
WSGR Docket No. 62619-720.601 (m, 1H), 5.05 - 4.91 (m, 1H), 4.63 - 4.40 (m, 2H), 4.37 - 4.20 (m, 1H), 3.39 - 3.35 (m, 1H), 3.18 - 2.94 (m, 3H), 2.92 - 2.76 (m, 1H), 2.47 - 2.30 (m, 2H), 2.23 - 1.58 (m, 8H).19F NMR (376 MHz, Dimethylsulfoxide-d6 [1311] Example 203: 5-Ethynyl-6-fluoro-4-((S)-4-fluoro-2-(((2R,7aR)-2-fluoro-2,3-dihydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9,10,11,12,13-hexahydro-8H-7-oxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-ol
[1312] Example 203 was prepared in a similar manner to Example 33. MS: m/z = 614.15 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.13 - 10.11 (m, 1H), 7.98 - 7.94 (m, 1H), 7.48 - 7.43 (m, 1H), 7.37 (s, 1H), 7.20 - 7.13 (m, 1H), 5.89 - 5.81 (m, 2H), 5.34 - 5.21 (m, 1H), 4.92 - 4.82 (m, 1H), 4.55 - 4.50 (m, 1H), 4.44 - 4.41 (m, 1H), 4.19 - 3.81 (m, 5H), 3.63 - 3.60 (m, 1H), 3.29 - 3.03 (m, 3H), 2.24 - 1.81 (m, 4H), 1.81 - 1.44 (m, 5H), 1.40 - 1.30 (m, 1H). 19F NMR (376 MHz, DMSO-d6) -110.64 - -110.79 (d, 1F), -145.90 - -146.77 (d, 1F), -176.45 - -176.50 (d, 1F). [1313] Example 204: 5-Ethynyl-6,7-difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a- pentaazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine
WSGR Docket No. 62619-720.601 [1314] Step 1: (R)-N-(6,7-Difluoro-4-(4-fluoro-7-methyl-2-(methylthio)-7,8,8a,9,12,13-hexahydro- 11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalen-5-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine [1315] CataCXium A Pd G3 and K3PO4 and H2O (0.4 mL) was degassed and purged with N2 three times, then the mixture was stirred at 100 °C for 2 hr under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 20% petroleum ether in EtOAc), the title compound (169 mg, yield: 73%) was obtained as a brown solid. MS: m/z = 857.3, 858.3 [M + H]+. [1316] Step 2: N-(6,7-Difluoro-4-((8aR)-4-fluoro-7-methyl-2-(methylsulfinyl)-7,8,8a,9,12,13- hexahydro-11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalen-5-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine [1317] To a solution of (R)-N-(6,7-difluoro-4-(4-fluoro-7-methyl-2-(methylthio)-7,8,8a,9,12,13- hexahydro-11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalen-5-yl)-5- CH2Cl2 25 °C for 2 hr. The reaction mixture was quenched with aqueous Na2SO3 (10 mL) at 0 °C, diluted with H2O (20 mL) and extracted with CH2Cl2 (50 mL x 3). The combined organic layers were washed with brine (20 mL x 3), dried over Na2SO4, filtered and concentrated under reduced pressure. The title compound (250 mg, yield: 85%) was obtained as an brown solid. MS: m/z = 873.5 [M + H]+. [1318] Step 3: N-(6,7-Difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a- pentaazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine [1319] To a solution of N-(6,7-difluoro-4-((8aR)-4-fluoro-7-methyl-2-(methylsulfinyl)- 7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8-ab]heptalen-5-yl)-5- ((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine The resulting mixture was stirred at 0 °C for 1 hr. The reaction mixture was quenched with saturated NaHCO3 (10 mL) at 0 °C, diluted with H2O (20 mL) and extracted with EtOAc (50 mL x
WSGR Docket No. 62619-720.601 3). The combined organic layers were washed with brine (20 mL x 3), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 10% MeOH in CH2Cl2), the title compound (190 mg, yield: 59%) was obtained as a yellow solid. MS: m/z = 827.4, 828.3 [M – 143 + H]+. [1320] Step 4: 6,7-Difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a-pentaazanaphtho[1,8- ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine [1321] To a solution of N-(6,7-difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a- pentaazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1- diphenylmethanimine and NH2OH The mixture was stirred at 25 °C for 2 hr. The reaction mixture was concentrated under reduced pressure. After purification by silica gel flash chromatography (eluent of 0 ~ 10% MeOH in CH2Cl2), the title compound (150 mg, yield: 72%) was obtained as a yellow solid. MS: m/z = 663.3, 664.2 [M – 143 + H]+. [1322] Step 5: 5-Ethynyl-6,7-difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a- pentaazanaphtho[1,8-ab]heptalen-5-yl)naphthalen-2-amine [1323] To a solution of 6,7-difluoro-4-((R)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy-d2)-7-methyl-7,8,8a,9,12,13-hexahydro-11H-10-oxa-1,3,6,7,13a- pentaazanaphtho[1,8-ab]heptalen-5-yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-amine (130 mg, in DMSO (1 mL) The mixture was stirred at 25 °C for 2 hr. The reaction mixture was diluted with H2O (20 mL) and extracted with EtOAc (20 mL x 3). The combined organic layers were washed with H2O (20 mL x 3), dried over Na2SO4, filtered and concentrated under reduced pressure. After purification by prep-HPLC (column: CD06-Waters 4HCO3) - ACN]; gradient: 35% - 65% B over 10 min), The title compound (Example 204, 40.6 mg, yield: 38%) was obtained as a white solid. MS: m/z = 650.5, 651.3 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6) (m, 1H), 7.07 - 6.90 (m, 2H), 5.83 - 5.68 (m, 2H), 5.40 - 5.16 (m, 1H), 4.93 - 4.35 (m, 1H), 4.22 - 4.04 (m, 2H), 3.97 - 3.68 (m, 3H), 3.67 - 3.51 (m, 1H), 3.49 - 3.35 (m, 3H), 3.29 - 3.25 (m, 2H), 3.14 - 3.06 (m, 2H), 3.05 - 2.99 (m, 2H), 2.87 - 2.78 (m, 1H), 2.19 - 1.89 (m, 4H), 1.89 - 1.74 (m, 4H).19F NMR (376 MHz, Dimethylsulfoxide-d6 153.59, -153.97, -154.12, -154.17, -172.08, -172.18.
WSGR Docket No. 62619-720.601 [1324] Example 205: (S)-4-(2-((2,6-Dimethylenetetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-4- fluoro-8,8a,9,10,11,12-hexahydro-7-oxa-1,3,6,12a-tetraazabenzo[4,5]cyclohepta[1,2,3- de]naphthalen-5-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
[1325] Example 205 was prepared in a similar manner to Example 33. MS: m/z = 608.30 [M + H]+. 1H NMR (400 MHz, DMSO-d6) 10.08 (bs, 1H), 7.97 - 7.94 (m, 1H), 7.48 - 7.43 (m, 1H), 7.37 (d, J = 2.4 Hz, 1H), 7.16 - 7.12 (m, 1H), 5.18 - 5.10 (m, 1H), 4.96 - 4.93 (m, 4H), 4.51 - 4.41 (m, 2H), 4.10 - 4.01 (m, 3H), 3.95 - 3.91 (m, 1H), 3.64 - 3.60 (m, 2H), 3.30 - 3.21 (m, 2H), 3.06 - 3.03 (m, 1H), 2.65 - 2.62 (m, 2H), 2.50 - 2.45 (m, 1H), 1.89 - 1.48 (m, 6H). 19F NMR (376 MHz, DMSO-d6) -110.72 (s, 1F), -145.20 - -145.51 (d, 1F). [1326] Example 208: (S)-5-(3-Amino-8-ethynyl-7-fluoronaphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O-methyl oxime
WSGR Docket No. 62619-720.601
[1327] Step 1: tert-Butyl (R)-3-((benzyloxy)methyl)-6-methylene-1,4-oxazepane-4-carboxylate [1328] To a solution of tert-butyl (S)-(1-(benzyloxy)-3-hydroxypropan-2-yl)carbamate (20.0 g, 71.1 mmol) in DMF (120 mL) was added NaH (6.82 g, 60% purity, 170.6 mmol) at 0 °C under N2 atmosphere over 0.5 h, and the mixture was stirred at 0 °C for 0.5 h. 3-Chloro-2- (chloromethyl)prop-1-ene (8.89 g, 71.1 mmol) was added to the mixture and the mixture was stirred at 25 °C for 2 h. The reaction was quenched with NH4Cl (800 mL), diluted with water (300 mL), and extracted with EtOAc (800 mL x 3). The combined organic phases were washed with brine (1000 mL), dried over anhydous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 6% of EtOAc in petroleum ether) to give the title compound (5.9 g, 16% yield) as a yellow oil. MS: m/z = 233.9 [M + H - Boc]+.1H NMR (400 MHz, Chloroform-d (m, 3H), 4.36 - 4.20 (m, 2H), 4.10 - 4.04 (m, 1H), 3.97 - 3.91 (m, 1H), 3.74 - 3.56 (m, 4H), 1.54 - 1.36 (m, 9H). [1329] Step 2: tert-Butyl (R)-3-((benzyloxy)methyl)-6-oxo-1,4-oxazepane-4-carboxylate [1330] To a solution of tert-butyl (R)-3-((benzyloxy)methyl)-6-methylene-1,4-oxazepane-4- carboxylate (470 mg, 1.41 mmol) in THF (10 mL) and H2O (1 mL) were added NaIO4 (905 mg, 4.23 mmol) and K2Os2O42H2 2 atmosphere. The mixture was stirred at 25 °C for 16 h. The reaction mixture was filtered. The filtrate was diluted with EtOAc (200 mL), washed with Na2SO3 solution (50 mL x 2) and brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 15% of EtOAc in petroleum ether) to give the title compound (280 mg, 54% yield) as a colorless oil. MS: m/z = 235.9 [M + H - Boc]+.1H NMR (400 MHz, Chloroform-d 3.81 - 3.66 (m, 4H), 1.46 (s, 9H).
WSGR Docket No. 62619-720.601 [1331] Step 3: tert-Butyl (R)-3-(hydroxymethyl)-6-oxo-1,4-oxazepane-4-carboxylate [1332] To a solution of tert-butyl (R)-3-((benzyloxy)methyl)-6-oxo-1,4-oxazepane-4-carboxylate in MeOH (4 mL) were Pd(OH)2 (240 mg, 167 20% purity) and Pd/C (200 mg, 167 10% purity) under N2. The mixture was degassed, purged with H2 three times, and stirred at 50 °C for 4 h under H2 (15 psi). The reaction mixture was filtered and concentrated under reduced pressure to give the title compound (200 mg, crude) as a colorless oil, which was used in the next step without further purification. 1H NMR (400 MHz, Chloroform-d (m, 8H), 3.51 - 3.23 (m, 1H), 1.53 - 1.39 (m, 9H). [1333] Step 4: (R)-3-(Hydroxymethyl)-1,4-oxazepan-6-one [1334] A solution of tert-butyl (R)-3-(hydroxymethyl)-6-oxo-1,4-oxazepane-4-carboxylate (200 mg, 816 ) in HCl (2 M in EtOAc, 2 mL) was stirred at 25 °C for 0.5 h. The mixture was concentrated under reduced pressure to give the title compound (120 mg, crude) as a colorless oil. [1335] Step 5: (R)-3-(Hydroxymethyl)-1,4-oxazepan-6-one O-methyl oxime [1336] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-6-one (100 mg, 689 ) in EtOH (2 mL) were added NaOAc (227 mg, 2.76 mmol) and O-methylhydroxylamine;hydrochloride (86 mg, 1.03 mmol) at 20 °C under N2. The mixture was stirred at 20 °C for 1 h. The reaction mixture was filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (90 mg, 75% yield over 3 steps) as a colorless oil. MS: m/z = 174.8 [M+ H]+.1H NMR (400 MHz, Chloroform- d (m, 2H), 3.10 - 2.86 (m, 1H). [1337] Step 6: (S)-2,7-Dichloro-8-fluoro-5-((6-(methoxyimino)-1,4-oxazepan-3- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H)-one [1338] To a solution of (R)-3-(hydroxymethyl)-1,4-oxazepan-6-one O-methyl oxime (90 mg, 517 added to the mixture at 0 °C, and the mixture was stirred at 25 °C for 2 h. The reaction mixture was diluted with sat. NaHCO3 aq. (2 mL), dried over MgSO4, filtered, and concentrated under reduced pressure to give the title compound (350 mg, crude) as a yellow solid, which was used in the next step without further purification. MS: m/z = 405.9 [M + H]+. [1339] Step 7: (S)-2,5-Dichloro-4-fluoro-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O-methyl oxime
WSGR Docket No. 62619-720.601 [1340] To a solution of (S)-2,7-dichloro-8-fluoro-5-((6-(methoxyimino)-1,4-oxazepan-3- yl)methoxy)pyrido[4,3-d]pyrimidin-4(3H 2Cl2 (80 mL) were added DIPEA (0.4 mL, 2.22 mmol) and POCl3 (0.2 mL, 1.85 mmol) at 0 °C under N2. The mixture was stirred at 0 °C for 2 h. The mixture was diluted with sat. NaHCO3 aq. (100 mL) and extracted with CH2Cl2 (300 mL x 2). The combined organic layers were washed with brine (300 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 4% of MeOH in CH2Cl2) to give the title compound (100 mg, 50% yield over two steps) as a yellow oil. MS: m/z = 388.0 [M + H]+. [1341] Step 8: (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-12(13H)- one O-methyl oxime [1342] To a solution of (S)-2,5-dichloro-4-fluoro-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H under N2. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 3% of MeOH in CH2Cl2) to give the title compound (Intermediate 17, 150 mg, 78% yield) as a yellow oil. MS: m/z = 511.1 [M + H]+. [1343] Step 9: (S)-5-(3-((Diphenylmethylene)amino)-7-fluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-12(13H)-one O-methyl oxime [1344] A mixture of (S)-5-Chloro-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-12(13H)- one O-methyl oxime N-(6-Fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)-5-((triisopropylsilyl)ethynyl)naphthalen-2-yl)-1,1-diphenylmethanimine K3PO4 2nBuP-Pd-G3 (cataCXiumAPdG3 dioxane (5 mL) and H2O (1 mL) was degassed and purged with N2 three times, and the mixture was stirred at 110 °C for 2 h under N2 atmosphere. The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 8% of MeOH in CH2Cl2) to give the title compound (75 mg, 64% yield) as a yellow solid. MS: m/z = 980.0[M + H]+.
WSGR Docket No. 62619-720.601 [1345] Step 10: (S)-5-(3-Amino-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-4-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H-7,10- dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O-methyl oxime [1346] To a solution of (S)-5-(3-((diphenylmethylene)amino)-7-fluoro-8- ((triisopropylsilyl)ethynyl)naphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8- ab]heptalen-12(13H)-one O 2 The reaction mixture was concentrated under reduced pressure. The residue was purified by silica gel flash chromatography (eluent: 0 ~ 10% of MeOH in CH2Cl2) to give the title compound (60 mg, 70 % yield) as a brown solid. MS: m/z = 816.4 [M + H]+. [1347] Step 11: (S)-5-(3-Amino-8-ethynyl-7-fluoronaphthalen-1-yl)-4-fluoro-2-(((2R,7aS)-2- fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H-7,10-dioxa-1,3,6,13a- tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O-methyl oxime [1348] A solution of (S)-5-(3-amino-7-fluoro-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-4- fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)-8a,9-dihydro-8H,11H- 7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalen-12(13H)-one O-methyl oxime (60 mg, 73.5 mixture was purified by reversed-phase column (column: C18; mobile phase: [water (NH4HCO3) - MeOH]; B%: 5% - 46%, 40 min) to give the title compound (Example 208, 4.0 mg, 10 % yield) as a white solid. MS: m/z = 660.4 [M + H]+.1H NMR (400 MHz, Dimethylsulfoxide-d6 J = 6.0, 8.8 Hz, 1H), 7.32 (t, J = 9.2 Hz, 1H), 7.11 - 6.92 (m, 2H), 6.19 - 5.18 (m, 4H), 4.97 - 4.65 (m, 2H), 4.63 - 4.05 (m, 8H), 4.03 - 3.99 (m, 1H), 3.90 - 3.75 (m, 3H), 3.16 - 2.89 (m, 4H), 2.15 - 1.82 (m, 6H).19F NMR (376 MHz, Dimethylsulfoxide-d6 172.24, -172.33. [1349] Each compound in the Table 3 was prepared in a similar manner (using appropriately substituted reagents) as described in examples.
WSGR Docket No. 62619-720.601 Table 3
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
II. Biological Evaluation [1350] “Ras” refers to a protein in the Ras superfamily of small GTPases, such as in the Ras subfamily. Ras proteins are small guanine nucleotide-binding proteins that act as molecular switches by cycling between active GTP-bound and inactive GDP-bound conformations. The Ras proteins play a critical role in the regulation of cell behaviors, including proliferation, differentiation, and survival. Mutation of any one of the three Ras isoforms (K-Ras, N-Ras, or H- Ras) has been shown to lead to oncogenic transformation, and in fact, K-Ras mutations are by far the most common in human cancers. KRAS G12V, G12D, and G12S variants are common in human cancers and Ras inhibitors binding in or near the Switch II pocket may provide mutant binding selectivity. RAS mutations are known to be often associated with pancreatic, colorectal
WSGR Docket No. 62619-720.601 and non-small-cell lung carcinomas (Lone, A. M. et al. J. Am. Chem. Soc.2011, 133, 11665-74, which is entirely incorporated herein by reference; Dillon, M. B.; Bachovchin, D. A.; Brown, S. J.; Finn, M. G.; Rosen, H.; Cravatt, B. F.; Mowen, K. A. ACS Chem. Biol.2012, 7, 1198-204, which is entirely incorporated herein by reference; Bachovchin, D. A.; Brown, S. J.; Rosen, H.; Cravatt, B. F. Nat. Biotechnol.2009, 27, 387-94, Ostrem, J. M.; Peters, U.; Sos, M. L.; Wells, J. A.; Shokat, K. M. Nature 2013, 503, 548-551. Ryan M.B. and Corcoran R.B, Nat. Rev Clin Onco.2018, 11, (MAPK) signaling pathway. This pathway regulates a wide variety of cellular processes, including proliferation, differentiation, apoptosis, and stress responses. One way to measure the impact of an inhibitor of Ras proteins is to measure downstream phosphorylation of proteins in the MAPK pathway. The MAPK pathway includes e oncogenic conditions. ERK expression and hyperactivation via phosphorylation plays a major role in cancer progression and are downstream of Ras proteins. The assay used for measuring the downstream impact of Ras inhibition is the use of a highly sensitive cell-based readout of ERK phosphorylation (pERK) specifically on Threonine (Thr) 202 and Tyrosine (Tyr) 204 in a KRAS G12V dependent lung adenocarcinoma cell line, NCI-H441 (ATCC Cat# HTB-174). The assay described herein is suitable for assessing inhibition of Ras and can be tailored for any of the predominant oncogenic mutations in any isoform of Ras (e.g., KRAS, HRAS, and NRAS) by changing the cell line. Fluorescence Resonance Energy Transfer (FRET) Assay [1351] pERK was measured using an advanced pERK assay designed for the robust and highly sensitive quantification of ERK phosphorylation on Thr202/Tyr204. This assay is intended for the simple, rapid, and direct detection of endogenous levels of ERK1/2 in cells, but only when ERK is phosphorylated at Thr202 and Tyr204 (Perkin Elmer Catalog # 64AERPEH). pERK was detected in a FRET based sandwich assay format using 2 different specific antibodies, one labelled with Eu3+-Cryptate (donor) and the second with d2 (acceptor). When the dyes are in close proximity, the excitation of the donor with a light source (laser or flash lamp) triggers a Fluorescence Resonance Energy Transfer (FRET) towards the acceptor, which in turn fluoresces at a specific wavelength (665 nm). The specific signal modulates positively in proportion to p-ERK1/2 (Thr202/ Tyr204). The simple mix-and-read protocol eliminates all wash steps for faster analysis and high-quality output. Briefly, cells were diluted to the desired concentration per well based on a growth curve showing ~ 75% confluence within a 24-hour time period at no less than 95%
WSGR Docket No. 62619-720.601 viability. Cell suspension (24 L) were added into each well of 384-well culture plate and to stock plates using TECAN (EVO200) liquid handler using DMSO as the solubilizing and dilution agent. Test compounds were transferred onto adherent cells using an Echo655 liquid handler. DMSO was employed as negative control. Cells were incubated with test compounds for 1 hr in a 37 °C, 5% CO2 incubator following compound addition. Following compound incubation, plates were removed from incubator and lysed with 4X lysis buffer (1X phosphatase inhibitor cocktail included) from the FRET kit. Antibodies and detection reagent were dispensed onto cell lysate and the plate was incubated at room temperature prior to reading. Twenty-five microliters of lysate plus detection reagent was transferred to an optiplate reading plate and measured using a Perkin Elmer Envision Reader using standard HTRF settings. TheEC50 was calculated by fitting the curve using Xlfit (v5.3.1.3). Table 4 provides the assay results for select examples. Activity is defined as “+“ for EC50 less than (<) 10,000 nanomolar, but greater than (>) 500 nanomolar; “++” for EC50 less than (<) 500 nanomolar, but greater than 50 nanomolar; or “+++” for EC50 less than (<) 50 nanomolar. Table 4
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
WSGR Docket No. 62619-720.601
[1352] The examples and embodiments described herein are for illustrative purposes only and various modifications or changes suggested to persons skilled in the art are to be included within the spirit and purview of this application and scope of the appended claims.
Claims
WSGR Docket No. 62619-720.601 CLAIMS We claim: 1. A compound having the structure of Formula (I), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from: (a) –(CH2)m-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b) -(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (c) -CH=CH-(CH2)p-O-(CH2)q-; (d) –(CH2)p-CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e) –(CH2)p-CH=CH-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
WSGR Docket No. 62619-720.601 (f) -CH=CH-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (g) –CH=CH-CH=CH-, wherein up to 2 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (h) -C(=CH2)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (i) –(c-C3H4)-(CH2)p-O-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (j) –(CH2)p-(c-C3H4)-(CH2)q-, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; and p is 1-4 and q is 1-4. 2 A compound having the structure of Formula (Ic), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, or C-Cl; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl;
WSGR Docket No. 62619-720.601 Y is a divalent moiety selected from –(CH2)m-, or -(CH2)p-O-(CH2)q-; n is 0, 1, 2, 3, 4, 5, or 6; m is 1-9; and p is 1-4 and q is 1-4. 3. A compound having the structure of Formula (II), or a pharmaceutically acceptable salt or solvate thereof:
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; W is a O, -CH2-, or -CD2-; R3 is hydrogen, R2 is alkyl, R4 is alkyl, and R2 and R4 join to form a ring; or R2 is hydrogen, R3 is alkyl, R4 is alkyl, and R3 and R4 join to form a ring; or R2 is hydrogen, and R3 and R4 join to form a -CH2OCH2- group; or R2 is hydrogen, and R3 and R4 join to form an -O- group; and n is 0, 1, 2, 3, 4, 5, or 6. 4 A compound having the structure of Formula (III), or a pharmaceutically acceptable salt or solvate thereof:
WSGR Docket No. 62619-720.601
wherein: X1 is N or C-CN; X2 is N, C-H, C-F, or C-Cl; X3 is N, C-H, C-F, C-Cl, or C-CF3; X4 is O, S, or NR5; X is O, S, or NR5; Ar is a monocyclic or bicyclic optionally substituted aryl or heteroaryl ring system; R1 is L-G; wherein L is a bond, optionally substituted C1-C4 alkylene,
; and G is an optionally substituted 5- to 10-membered heterocyclyl; each R is independently selected from hydrogen, deuterium, or optionally substituted C1-C4 alkyl; each R5 is hydrogen or optionally substituted C1-C4 alkyl; Y is a divalent moiety selected from: (a)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (b)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (c) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
WSGR Docket No. 62619-720.601 (d)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (e)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (f)
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; (g) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms;
(h) , wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms; each R6 is independently selected from hydrogen or deuterium; R7 is an optionally substituted C1-C4 alkyl; W is selected from oxo, oximo, or optionally substituted alkyl oximo; n is 1 or 2; p is 1-4; and q is 1-4. 5 The compound of claim 1, 2, 3, or 4, or a pharmaceutically acceptable salt or solvate thereof, wherein X1 is N. 6 The compound of claim 1, 2, 3, or 4, or a pharmaceutically acceptable salt or solvate thereof, wherein X1 is C-CN. 7 The compound of any one of claims 1-6, or a pharmaceutically acceptable salt or solvate thereof, wherein X2 is N. 8 The compound of any one of claims 1-6, or a pharmaceutically acceptable salt or solvate thereof, wherein X2 is C-H, C-F, or C-Cl. 9 The compound of any one of claims 1-8, or a pharmaceutically acceptable salt or solvate thereof, wherein X3 is N.
WSGR Docket No. 62619-720.601 10. The compound of any one of claims 1-8, or a pharmaceutically acceptable salt or solvate thereof, wherein X3 is C-H, C-F, or C-Cl. 11. The compound of any one of claims 1-10, or a pharmaceutically acceptable salt or solvate thereof, wherein X4 is O. 12. The compound of any one of claims 1-11, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted aryl. 13. The compound of claim 12, or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is an optionally substituted naphthyl. 14. The compound of claim 13, or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted naphthyl is an optionally substituted 1-naphthyl. 15. The compound of claim 14, or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted 1-naphthyl is further substituted at the 8-position. 16. The compound of claim 12, or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted aryl is described by Formula (a):
wherein: R12 is hydrogen; R13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R14 is hydrogen; R15 is hydrogen or F; R16 is hydrogen or F; R17 is fluorine or -CN; and R18 3, -OCD3, -OCH2F, or -OCD2F. 17. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
18. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
19. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
20. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
21. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is:
. 22. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
23. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
24. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
25. The compound of any one of claims 1-15, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
26. The compound of any one of claims 1-11, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is a bicyclic optionally substituted heteroaryl. 27. The compound of claim 26, or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is described by Formula (b):
wherein: R12 is hydrogen; R13 is -OH, -NH2, Cl, -OCONHMe, -NHCO2Me; R14 is hydrogen; R16 is hydrogen or F; R17 is fluorine or -CN; and R18 3, -OCD3, -OCH2F, or -OCD2F. 28. The compound of claim 26, or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from:
WSGR Docket No. 62619-720.601
. 29. The compound of claim 26, or a pharmaceutically acceptable salt or solvate thereof, wherein the bicyclic optionally substituted heteroaryl is selected from:
30. The compound of any one of claims 1-11, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is an optionally substituted phenyl. 31. The compound of claim 30, or a pharmaceutically acceptable salt or solvate thereof, wherein the optionally substituted phenyl is substituted with an -OH group at the 3-position. 32. The compound of any one of claims 1-31, or a pharmaceutically acceptable salt or solvate thereof, wherein L is a bond. 33. The compound of any one of claims 1-31, or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1-C4 alkylene. 34. The compound of any one of claims 1-31, or a pharmaceutically acceptable salt or solvate thereof, wherein
. 35. The compound of claim 33, or a pharmaceutically acceptable salt or solvate thereof, wherein L
36. The compound of any one of claims 1-33, or a pharmaceutically acceptable salt or solvate thereof, wherein L is optionally substituted C1 alkylene.
WSGR Docket No. 62619-720.601 37. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is an optionally substituted 5- to 10-membered heterocyclyl. 38. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
39. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
. 40. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (c):
wherein, each R20-R30 is independently selected from hydrogen or deuterium. 41. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is described by Formula (d):
wherein, R31 is selected from hydrogen, F, Cl, -CN, -OH, or optionally substituted C1-C4 alkyl; R32 is hydrogen, deuterium or optionally substituted C1-C4 alkyl; and
WSGR Docket No. 62619-720.601 R33 is hydrogen, deuterium, or F. 42. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
43. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
44. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from: ,
45. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601
46. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
47. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
WSGR Docket No. 62619-720.601 48. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
49. The compound of any one of claims 1-36, or a pharmaceutically acceptable salt or solvate thereof, wherein G is selected from:
50. The compound of any one of claims 1-49, or a pharmaceutically acceptable salt or solvate thereof, wherein X is O. 51. The compound of any one of claims 1-50, or a pharmaceutically acceptable salt or solvate thereof, wherein R is hydrogen. 52. The compound of any one of claims 1-50, or a pharmaceutically acceptable salt or solvate thereof, wherein R is methyl. 53. The compound of any one of claims 1-52, or a pharmaceutically acceptable salt or solvate thereof, wherein n is 0.
WSGR Docket No. 62619-720.601 54. The compound of any one of claims 1-52, or a pharmaceutically acceptable salt or solvate thereof, wherein n is 1. 55. The compound of any one of claims 1-52, or a pharmaceutically acceptable salt or solvate thereof, wherein n is 2. 56. The compound of any one of claims 1-31, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
57. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)m-. 58. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2)-. 59. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is –(CH2CH2)-.
WSGR Docket No. 62619-720.601 60. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)p-O-(CH2)q-. 61. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2)-. 62. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -( CH2CH2)-O-(CH2)-. 63. The compound of any one of claims 1, 2, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is -(CH2)-O-(CH2CH2)-. 64. The compound of any one of claims 3 and 5-63, or a pharmaceutically acceptable salt or solvate thereof, wherein W is O. 65. The compound of any one of claims 3 and 5-63, or a pharmaceutically acceptable salt or solvate thereof, wherein W is -CH2-, or -CD2-. 66. The compound of any one of claims 3 and 5-63, or a pharmaceutically acceptable salt or solvate thereof, wherein R2 and R4 join together to form a -CH2CH2-. 67. The compound of any one of claims 3 and 5-63, or a pharmaceutically acceptable salt or solvate thereof, wherein R3 and R4 join together to form a -CH2CH2-. 68. The compound of any one of claims 3 and 5-63, or a pharmaceutically acceptable salt or solvate thereof, wherein R3 and R4 join together to form a -CH2CH2CH2-. 69. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 70. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 71. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms.
WSGR Docket No. 62619-720.601 72. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 73. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 74. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 75. The compound of any one of claims 4, and 5-56, or a pharmaceutically acceptable salt or solvate thereof, wherein Y is
, wherein up to 4 hydrogen atoms are optionally replaced with deuterium or fluorine atoms. 76. The compound of any one of claims 69-75, or a pharmaceutically acceptable salt or solvate thereof, wherein R7 is an optionally substituted C1 alkyl. 77. The compound of any one of claims 69-76, or a pharmaceutically acceptable salt or solvate thereof, wherein p is 1 or 2; and q is 1 or 2. 78. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
. 79. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
80. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
81. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
82. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
83. The compound of any one of claims 1-11, 32-55, or 57-77, or a pharmaceutically acceptable salt or solvate thereof, wherein Ar is selected from:
WSGR Docket No. 62619-720.601
. 84. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
85. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
WSGR Docket No. 62619-720.601
86. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
. 87. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601
. 88. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
89. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
90. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601 ,
91. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
92 The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
93 The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601
94. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
95. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from:
WSGR Docket No. 62619-720.601 96. The compound of any one of claims 1-31, 51-55 or 57-83, or a pharmaceutically acceptable salt or solvate thereof, wherein R1 is selected from: ,
97. The compound of any one of claims 84-96, or a pharmaceutically acceptable salt or solvate thereof, wherein X is -O-. 98. A compound, or pharmaceutically acceptable salt or solvate thereof, as described in Table 1. 99. A compound, or pharmaceutically acceptable salt or solvate thereof, as described in Table 2. 100. A compound, or pharmaceutically acceptable salt or solvate thereof, as described in any one of Figures 1-10. 101. A pharmaceutical composition comprising a compound, or pharmaceutically acceptable salt or solvate thereof, as described in any one of claims 1-100 and a pharmaceutically acceptable excipient. 102. A method of preparing a pharmaceutical composition comprising mixing a compound, or pharmaceutically acceptable salt or solvate thereof, of any one of claims 1-100, and a pharmaceutically acceptable carrier. 103. A compound of any one of claims 1-100, or pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of the human or animal body. 104. A compound of any one of claims 1-100, or pharmaceutically acceptable salt or solvate thereof, for use in a method of treatment of cancer. 105. Use of a compound of any one of claims 1-100, or pharmaceutically acceptable salt or solvate thereof, in the manufacture of a medicament for the treatment of cancer. 106. A method of treating cancer in a patient in need thereof, comprising administering to the patient a compound as described in any one of claims 1-100, or pharmaceutically acceptable salt or solvate thereof.
WSGR Docket No. 62619-720.601 107. A method of treating cancer in a patient in need thereof, comprising administering to the patient a pharmaceutical composition comprising a compound as described in any one of claims 1-100, or pharmaceutically acceptable salt or solvate thereof, and a pharmaceutically acceptable excipient. 108. The method of any one of claims 104-107, wherein the cancer is lung cancer, non-small cell lung cancer, or colorectal cancer. 109. A method of inhibiting KRAS comprising contacting the enzyme with a compound of any one of claims 1-100, wherein KRAS is contacted in an in vitro setting. 110. A method of inhibiting KRAS comprising contacting the enzyme with a compound of any one of claims 1-100, wherein KRAS is contacted in an in vivo setting.
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363486821P | 2023-02-24 | 2023-02-24 | |
| US63/486,821 | 2023-02-24 | ||
| US202363497819P | 2023-04-24 | 2023-04-24 | |
| US63/497,819 | 2023-04-24 | ||
| US202363500469P | 2023-05-05 | 2023-05-05 | |
| US63/500,469 | 2023-05-05 | ||
| US202363587912P | 2023-10-04 | 2023-10-04 | |
| US63/587,912 | 2023-10-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024178304A1 true WO2024178304A1 (en) | 2024-08-29 |
Family
ID=92501543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/017034 Ceased WO2024178304A1 (en) | 2023-02-24 | 2024-02-23 | Kras modulators |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024178304A1 (en) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025080946A2 (en) | 2023-10-12 | 2025-04-17 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025171296A1 (en) | 2024-02-09 | 2025-08-14 | Revolution Medicines, Inc. | Ras inhibitors |
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
| WO2025240847A1 (en) | 2024-05-17 | 2025-11-20 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
| WO2026035945A1 (en) | 2024-08-07 | 2026-02-12 | Tesseract Medicines Us, Llc | Covalent-induced drug conjugates targeting kras and comprising a topoisomerase payload |
| WO2026035947A1 (en) | 2024-08-07 | 2026-02-12 | Tesseract Medicines Us, Llc | Kras-targeting covalent-induced drug conjugates comprising a topoisomerase payload |
| WO2026050446A1 (en) | 2024-08-29 | 2026-03-05 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026064520A1 (en) | 2024-09-19 | 2026-03-26 | Tesseract Medicines Us, Llc | Covalent-induced drug conjugates targeting kras and comprising a tubulin inhibitor payload |
| WO2026064527A1 (en) | 2024-09-19 | 2026-03-26 | Tesseract Medicines Us, Llc | Kras-targeting covalent-induced drug conjugates comprising a tubulin inhibitor payload |
| WO2026072904A2 (en) | 2024-09-26 | 2026-04-02 | Revolution Medicines, Inc. | Compositions and methods for treating lung cancer |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022173678A1 (en) * | 2021-02-09 | 2022-08-18 | Genentech, Inc. | Tetracyclic oxazepine compounds and uses thereof |
| WO2023001123A1 (en) * | 2021-07-19 | 2023-01-26 | 上海艾力斯医药科技股份有限公司 | New pyridopyrimidine derivative |
| WO2023046135A1 (en) * | 2021-09-27 | 2023-03-30 | Jacobio Pharmaceuticals Co., Ltd. | Polycyclic fused ring derivatives and use thereof |
| CN116332948A (en) * | 2021-12-22 | 2023-06-27 | 翰森生物有限责任公司 | A nitrogen-containing tetracyclic compound and its preparation method and medicinal use |
| WO2024009191A1 (en) * | 2022-07-05 | 2024-01-11 | Pfizer Inc. | Pyrido[4,3-d]pyrimidine compounds |
| WO2024015262A1 (en) * | 2022-07-09 | 2024-01-18 | Blossomhill Therapeutics, Inc. | Fused ring kras inhibitors for treating disease |
| WO2024031088A1 (en) * | 2022-08-05 | 2024-02-08 | Kumquat Biosciences Inc. | Heterocyclic compounds and uses thereof |
| WO2024032704A1 (en) * | 2022-08-11 | 2024-02-15 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024032703A1 (en) * | 2022-08-11 | 2024-02-15 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024041573A1 (en) * | 2022-08-25 | 2024-02-29 | Zai Lab (Shanghai) Co., Ltd. | Fused multi-heterocyclic compounds as kras g12d modulators and uses thereof |
| CN117683051A (en) * | 2022-09-09 | 2024-03-12 | 上海艾力斯医药科技股份有限公司 | Tetraheterocyclic compounds, their preparation methods, their intermediates and their applications |
| WO2024063578A1 (en) * | 2022-09-23 | 2024-03-28 | 일동제약(주) | Novel tetraheterocycle compound |
-
2024
- 2024-02-23 WO PCT/US2024/017034 patent/WO2024178304A1/en not_active Ceased
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022173678A1 (en) * | 2021-02-09 | 2022-08-18 | Genentech, Inc. | Tetracyclic oxazepine compounds and uses thereof |
| WO2023001123A1 (en) * | 2021-07-19 | 2023-01-26 | 上海艾力斯医药科技股份有限公司 | New pyridopyrimidine derivative |
| WO2023046135A1 (en) * | 2021-09-27 | 2023-03-30 | Jacobio Pharmaceuticals Co., Ltd. | Polycyclic fused ring derivatives and use thereof |
| CN116332948A (en) * | 2021-12-22 | 2023-06-27 | 翰森生物有限责任公司 | A nitrogen-containing tetracyclic compound and its preparation method and medicinal use |
| WO2024009191A1 (en) * | 2022-07-05 | 2024-01-11 | Pfizer Inc. | Pyrido[4,3-d]pyrimidine compounds |
| WO2024015262A1 (en) * | 2022-07-09 | 2024-01-18 | Blossomhill Therapeutics, Inc. | Fused ring kras inhibitors for treating disease |
| WO2024031088A1 (en) * | 2022-08-05 | 2024-02-08 | Kumquat Biosciences Inc. | Heterocyclic compounds and uses thereof |
| WO2024032704A1 (en) * | 2022-08-11 | 2024-02-15 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024032703A1 (en) * | 2022-08-11 | 2024-02-15 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024041573A1 (en) * | 2022-08-25 | 2024-02-29 | Zai Lab (Shanghai) Co., Ltd. | Fused multi-heterocyclic compounds as kras g12d modulators and uses thereof |
| CN117683051A (en) * | 2022-09-09 | 2024-03-12 | 上海艾力斯医药科技股份有限公司 | Tetraheterocyclic compounds, their preparation methods, their intermediates and their applications |
| WO2024063578A1 (en) * | 2022-09-23 | 2024-03-28 | 일동제약(주) | Novel tetraheterocycle compound |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
| WO2025080946A2 (en) | 2023-10-12 | 2025-04-17 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025171296A1 (en) | 2024-02-09 | 2025-08-14 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025240847A1 (en) | 2024-05-17 | 2025-11-20 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
| WO2026035945A1 (en) | 2024-08-07 | 2026-02-12 | Tesseract Medicines Us, Llc | Covalent-induced drug conjugates targeting kras and comprising a topoisomerase payload |
| WO2026035947A1 (en) | 2024-08-07 | 2026-02-12 | Tesseract Medicines Us, Llc | Kras-targeting covalent-induced drug conjugates comprising a topoisomerase payload |
| WO2026050446A1 (en) | 2024-08-29 | 2026-03-05 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026064520A1 (en) | 2024-09-19 | 2026-03-26 | Tesseract Medicines Us, Llc | Covalent-induced drug conjugates targeting kras and comprising a tubulin inhibitor payload |
| WO2026064527A1 (en) | 2024-09-19 | 2026-03-26 | Tesseract Medicines Us, Llc | Kras-targeting covalent-induced drug conjugates comprising a tubulin inhibitor payload |
| WO2026072904A2 (en) | 2024-09-26 | 2026-04-02 | Revolution Medicines, Inc. | Compositions and methods for treating lung cancer |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2024178304A1 (en) | Kras modulators | |
| US12595265B2 (en) | Inhibitors of activin receptor-like kinase | |
| US12528828B2 (en) | Substituted pyrido[4,3-d]pyrimidines as KRAS modulators | |
| WO2024178313A1 (en) | Kras modulators | |
| JP2025063176A (en) | KRAS G12D inhibitors | |
| AU2016311426A1 (en) | Compounds and compositions useful for treating disorders related to NTRK | |
| CN113874381A (en) | Inhibitors of RAF kinases | |
| EP4161656A1 (en) | Inhibitors of fibroblast growth factor receptor kinases | |
| WO2024112796A1 (en) | MODULATORS OF TNF-α ACTIVITY | |
| CN115698020B (en) | Macrocyclic RIP2-kinase inhibitors | |
| WO2021247971A1 (en) | Inhibitors of fibroblast growth factor receptor kinases | |
| KR20240152322A (en) | Inhibitor of CDK4/6 kinase | |
| KR20250022815A (en) | HPK1 inhibitors and their applications in medicine | |
| WO2026015385A1 (en) | Substituted pyrido[4,3-d]pyrimidines as kras modulators | |
| WO2025024707A1 (en) | Inhibitors of cdk2/4/6 kinase | |
| HK40063842A (en) | Inhibitors of raf kinases | |
| EA040675B1 (en) | ACTIVIN-LIKE RECEPTOR KINASE INHIBITORS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24761054 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 24761054 Country of ref document: EP Kind code of ref document: A1 |