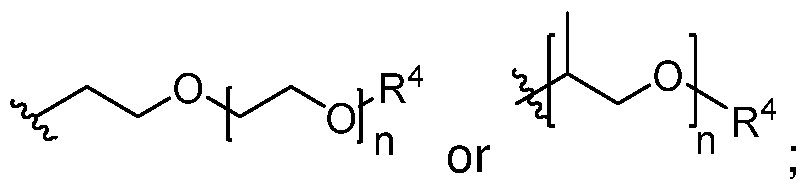WO2022076365A1 - Aromatic enol ethers - Google Patents
Aromatic enol ethers Download PDFInfo
- Publication number
- WO2022076365A1 WO2022076365A1 PCT/US2021/053505 US2021053505W WO2022076365A1 WO 2022076365 A1 WO2022076365 A1 WO 2022076365A1 US 2021053505 W US2021053505 W US 2021053505W WO 2022076365 A1 WO2022076365 A1 WO 2022076365A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- benzene
- found
- prop
- exact mass
- bis
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/03—Ethers having all ether-oxygen atoms bound to acyclic carbon atoms
- C07C43/14—Unsaturated ethers
- C07C43/164—Unsaturated ethers containing six-membered aromatic rings
- C07C43/166—Unsaturated ethers containing six-membered aromatic rings having unsaturation outside the aromatic rings
Definitions
- This application relates to chemistry generally.
- this application relates to enol ethers and more particularly to aromatic enol ethers.
- Enol ethers are useful in a variety of chemical applications such as plasticizers, diluents, wetting agents and paint additives and as intermediates in chemical processes.
- Plasticizers, diluents, wetting agents and paint additives often are volatile and evaporate into the atmosphere during use.
- coalescing aids that are added to water-based paints, aid in film formation in latex emulsions. The coalescing aids lower the glass transition temperature (Tg) of the latex polymer and as the paint dries, the polymers that have been softened by the coalescing aid are allowed to flow together and form a film after the water has left the system. Coalescing aids that are volatile evaporate out of the film. This allows the polymer to return to the original Tg thereby giving harder films for better block and print resistant coatings.
- Tg glass transition temperature
- A is (C8-20) alkylaryl
- R 1a and R 1b are independently (Ci-i2)alkyl, (C2-i2)alkenyl, (C3- s)cycloalkyl, or 5- to 9-membered aryl or each R 4 is independently (Ci-i2)alkyl, or -C(O)R 5 ; each R 5 is (Ci-i2)alkyl unsubstituted or substituted by R 6 , (C2-i2)alkenyl unsubstituted or substituted by R 6 , (C3-8)cycloalkyl, or 5- to 9- membered aryl; each R 6 is (Ci-4)alkoxy, or oxo; and each n is independently an integer from 1 to 15 or each R 7 is independently (Ci-i2)alkyl.
- Alkyl means an aliphatic hydrocarbon.
- the alkyl can specify the number of carbon atoms, for example (C1 -5)alkyl.
- the alkyl group can be unbranched or branched. In some embodiments, the alkyl group is branched. In some embodiments, the alkyl group is unbranched.
- alkanes include methane, ethane, propane, isopropyl (i.e., branched propyl), butyl, and the like.
- alkenyl means an aliphatic hydrocarbon with one or more unsaturated carbon-carbon bonds.
- the alkenyl can specify the number of carbon atoms, for example (C2-12)alkenyl.
- the alkyl group can be unbranched or branched. In some embodiments, the alkyl group is branched. In some embodiments, the alkyl group is unbranched.
- Non-limiting examples of alkanes include ethenyl, propenyl, butenyl, hexa-3,5- dienyl, and the like.
- Alcohol means a chemical containing one or more hydroxyl groups.
- Aldehyde means a chemical containing one or more -C(O)H groups.
- Cycloalkyl means a cyclic hydrocarbon compound.
- the cycloalkyl can specify the number of carbon atoms in ring system, for example (C3- 8)cycloalkyl.
- Non-limiting examples of cycloalkyl include cyclopropyl, cyclobutyl, cyclohexyl, and cyclooctyl.
- Aryl means a ring system made up carbon atoms that has at least one ring that is aromatic.
- the carbon units making up the aryl ring may be specified, for example 5- to 9-membered aryl.
- Non-limiting examples of aryl include phenyl, naphthyl, 2,3-dihydro-1 H-indene, and 1 , 2,3,4- tetrahydronaphthalene.
- Values may be expressed as “about” or “approximately” a given number.
- ranges may be expressed herein as from “about” one particular value and/or to “about” or another particular value. When such a range is expressed, another aspect includes from the one particular value and/or to the other particular value.
- values are expressed as approximations, by use of the antecedent “about,” it will be understood that the particular value forms another aspect.
- the term “and/or,” when used in a list of two or more items, means that any one of the listed items can be employed by itself or any combination of two or more of the listed items can be employed. For example, if a compound is described as containing components A, B, and/or C, the compound can contain A alone; B alone; C alone; A and B in combination; A and C in combination, B and C in combination; or A, B, and C in combination.
- the terms “comprising,” “comprises,” and “comprise” are open-ended transition terms used to transition from a subject recited before the term to one or more elements recited after the term, where the element or elements listed after the transition term are not necessarily the only elements that make up the subject.
- Y is chosen from A, B, and C means Y can be individually A, B, or C.
- Y is chosen from A, B, or C means Y can be individually A, B, or C; or a combination of A and B, A and C, B and C, or A, B, and C.
- novel enol ethers which can be used in applications such as (but not limited to) plasticizers, diluents, wetting agents, coalescing aids and paint additives.
- the invention is a compound according to Formula I:
- A is (C8-20) alkylaryl
- R 1a and R 1b are independently (Ci-i2)alkyl, (C2-i2)alkenyl, (C3- s)cycloalkyl, or 5- to 9-membered aryl or each R 4 is independently (Ci-i2)alkyl, or -C(O)R 5 ; each R 5 is (Ci-i2)alkyl unsubstituted or substituted by R 6 , (C2-i2)alkenyl unsubstituted or substituted by R 6 , (C3-8)cycloalkyl, or 5- to 9- membered aryl; each R 6 is (Ci-4)alkoxy, or oxo; and each n is independently an integer from 1 to 15 or each R 7 is independently (Ci-i2)alkyl.
- a in Formulas I is 1 ,2-, 1 ,3-, or 1 ,4- disubstituted phenyl.
- each n is an integer from 1 to 3.
- each R 4 is hydrogen.
- each R 4 is (Ci-i2)alkyl.
- each R 4 is independently ethyl.
- each R 4 is (C2-12)alkenyl.
- each R 4 is -C(O)R 5 .
- each R 5 is (Ci-i2)alkyl unsubstituted or substituted by R 6 . In some embodiments, each R 5 is (Ci- i2)alkenyl unsubstituted or substituted by R 6 . In some embodiments, each R 5 is (C3-8)cycloalkyl. In some embodiments, each R 5 is 5- to 9-membered aryl. [0025] In some embodiments of Formulas I each n is an integer from 1 to 2. In some embodiments, each n is an integer from 1 to 3. In some embodiments, each n is an integer from 1 to 4. In some embodiments, each n is an integer from 1 to 5.
- n is an integer from 1 to 6. In some embodiments, n is an integer from 1 to 7. In some embodiments, n is an integer from 1 to 8. In some embodiments, n is an integer from 1 to 9. In some embodiments, n is an integer from 1 to 10. In some embodiments, n is an integer from 1 to 11 . In some embodiments, n is an integer from 1 to 12. In some embodiments, n is an integer from 1 to 13. In some embodiments, n is an integer from 1 to 14. In some embodiments, n is an integer from 1 to 15. [0026] In some embodiments, the compounds of Formulas I have a volatile organic content of less than 50 wt % according to ASTM D6886.
- the volatile organic content is less than 30 wt %. In some embodiments, the volatile organic content is less than 10 wt %. In some embodiments, the volatile organic content is less than 5 wt %. In some embodiments, the volatile organic content is less than 3 wt %. In some embodiments, the volatile organic content is less than 2 wt %. In some embodiments, the volatile organic content is less than 1 wt %. In some embodiments, the volatile organic content is less than 0.8 wt %.
- the enol ether compounds disclosed in the present application exhibit a low volatile organic content (less than 50 wt %, but as low as 0.7 wt % according to ASTM D6886).
- the enol ethers can be used as reactive filmhardening compounds. Reactive film-hardening compounds react with components in coating compositions to form crosslinks in the films providing improved film properties.
- the enol ether compounds of this invention can be used as reactive film-hardening additives, we mean when added to a coating composition, that a harder film is obtained upon curing the composition than is obtained in the absence of the invention enol ether additives, or that the coating composition exhibits a higher gel fraction than in the absence of the enol ether additive, or that both coating composition hardness and increased gel fraction properties are improved by the addition of the enol ether reactive film-hardening additives.
- the increase in hardness observed in a coating that contains the enol ether additives described herein may be the result of a chemical reaction, so that the additives described herein may be described as “reactive” enol ether film-hardening additives.
- the enol ether materials described herein can also facilitate the individual latex particles coming together to form a continuous film at a given temperature by reducing the minimum film-forming temperature (MFFT) of the latex polymer.
- the compound comprises the compounds represented by Formulas I, II and II.
- the compounds of Formula I are enol ethers represented by Formulas 20-69:
- the enol ethers depicted by Formulas 5-69 are representative of the enol ethers claimed herein. Isomers of the enol ethers depicted by Formulas 5-69 are expected to be produced during synthesis of the enol ethers depicted by Formulas 5-69. All isomers of the enol ethers depicted by Formulas 5-69 and are within the scope of the claims set forth herein.
- the compounds depicted by Formulas I include those having a weight percent volatile content of less than 50%, as measured according to ASTM Method D6886. This test may be conducted generally by heating the sample in a forced air oven at 110°C. for 60 minutes.
- the weight loss after the test is deemed to result from a loss of volatiles originally present in the sample; the percent volatile present in the original sample may then be calculated.
- the values cited herein may be obtained from a sample of the additive itself.
- the weight percent volatile of a film-hardening aid may be used herein as a yardstick to measure the amount of VOC the additive would contribute to the VOC in a particular end use such as a component of a coating composition.
- mL is milliliter; wt % is weight percent; eq is equivalent(s); hrs or h is hour(s); mm is millimeter; m is meter; GC is gas chromatography; °C is degree Celsius; min is minute; tR is retention time; VOC is volatile organic compound; MeP is methyl palmitate; w/v is weight/volume; pL is microliter.
- RFHA reactive film-hardening additive.
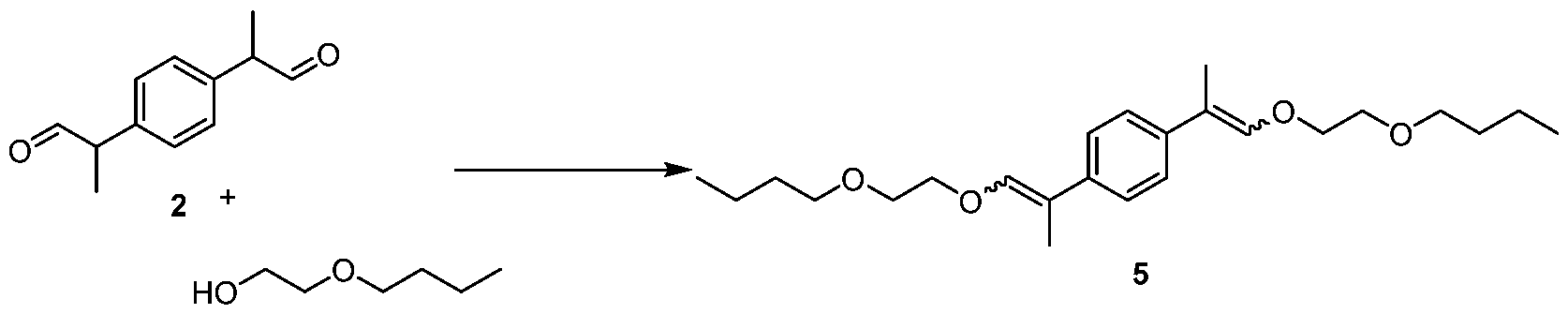
- the dicarbinol 1 was then dissolved in formic acid (88%, 98.0 g) contained within a 500 mL, 4-necked round-bottom flask fitted with thermocouple, overhead stirrer, and nitrogen inlet atop a reflux condenser. The mixture was heated to 100 °C. After 6 hrs, additional formic acid was added (98.0 g). After an additional 2 hrs, GC indicated >99% conversion to dialdehyde 2. The volatiles were then removed under reduced pressure using a rotary evaporator. The residue was taken up in 250 mL of toluene and then washed with a saturated solution of NaHCOa. After layer separation, the organics were dried with MgSC , filtered, and then concentrated.
- Dicarbinol 3 was prepared in a similar manner to di-carbinol 2 using procedure described in Method A. [LC-MS (Column B) tR : 4.55 min, 4.68 min (Exact mass: 254.15 m/z, found 254.2 m/z)]. Di-aldehyde 4 was prepared in a similar manner to di-aldehyde 2 using Method A.
- Example 3 A mixture of (E,E/Z,Z) - 1,4-bis(1 (1-(( 1 -m ethoxypropan-2- yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [6a], (E,Z) - methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en-2-yl)-4-(3-((1 -((1 - methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [6b], and 1 ,4-bis(3-((1 -((1 -methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en- 2-yl)benzene [6c] GC-MS tR : 25.80 min, 26.28 min, 26.80 min, 27.30 min, 28.38 min, 28.94
- Example 4 A mixture of (E,E/Z,Z) - 1,4-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-12-en-13-yl)benzene [7a], (E,Z) - 4,7,10-trimethyl-13-(4- (4,7,10-trimethyl-2,5,8,11 -tetraoxatetradec-13-en-13-yl)phenyl)-2,5,8,11 - tetraoxatetradec-12-ene [7b], and 1,4-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-13-en-13-yl)benzene [7c]
- Example 5 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [8a], (E,Z) - 1-(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [8b], and 1 ,4-bis(3-(2-(2- methoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [8c] GC-MS tR : 23.9 min, 24.29 min, 24.48 min, 25.64 min, 25.96 min, 27.63 min (Exact mass: 394.24 m/z, found: 394.3 m/z).
- Example 6 a mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [9a], (EZZ) - 1-(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [9b], and 1 ,4-bis(3-(2-(2- ethoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [9c].
- Example 7 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10a], (E,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [1 Ob], 1 ,4-bis(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [10c].
- GC-MS tR 29.70 min, 30.67 min, 31 .30 min, 33.27 min, 34.18 min, 37.58 min (Exact mass: 450.30 m/z, found: 450.3 m/z).
- Example 8 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11a], (E,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11 b], 1 ,4-bis(3-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [11c].
- Example 9 A mixture of (E,E/Z,Z) - 1,4-di(2,5,8,11-tetraoxatetradec-12- en-13-yl)benzene [12a], (E,Z) - 13-(4-(2,5,8,11-tetraoxatetradec-13-en-13- yl)phenyl)-2,5,8,11-tetraoxatetradec-12-ene [12b], and 1 ,4-di(2,5,8,11 - tetraoxatetradec-13-en-13-yl)benzene [12c].
- GC-MS tR 39.51 min, 41 .51 min, 42.78 min, 46.43 min, 48.26 min, 55.04 min (Exact mass: 482.29 m/z, found: 482.4 m/z).
- Example 10 A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-((1-methoxypropan-2- yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [13a], (E/Z) - 1 -(1 -((1 -(Ci- met hoxy propan-2-y l)oxy)pro pan-2-y l)oxy)prop-1 -en-2-yl)-3-(3-((1 -((1 - methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en-2-yl)benzene [13b], and 1 ,3-bis(3-((1 -((1 -methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en- 2-yl)benzene [13c].
- Example 11 A mixture of (E,E/Z,Z) - 1,3-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-12-en-13-yl)benzene [14a], (E/Z) - 4,7,10-trimethyl-13-(3- (4,7,10-trimethyl-2,5,8,11 -tetraoxatetradec-13-en-13-yl)phenyl)-2,5,8,11 - tetraoxatetradec-12-ene [14b], and 1,3-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-13-en-13-yl)benzene [14c]
- Example 12 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [15a], (E/Z) - 1-(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- methoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [15b], 1 ,3-bis(3-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [15c].
- Example 13-1 and Example 13-2 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [16a], (E/Z) - 1-(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [16b], and 1 ,3-bis(3-(2-(2- ethoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [16c].
- Example 14 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [17a], (E/Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17b], and 1 ,3-bis(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [17c]
- Example 15 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18a], (E/Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18b], and 1 ,3-bis(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18c].
- Example 16 A mixture of (E,E/Z,Z) - 1,3-di(2,5,8,11-tetraoxatetradec-12- en-13-yl)benzene [19a], (E/Z) - 13-(3-(2,5,8,11-tetraoxatetradec-13-en-13- yl)phenyl)-2,5,8,11-tetraoxatetradec-12-ene [19b], and 1 ,3-di(2,5,8,11 - tetraoxatetradec-13-en-13-yl)benzene [19c].
- reaction mixture was cooled to ambient temperature and then diluted with a minimal amount of toluene. This mixture was then poured into water containing 1 .25 equiv. of NaOH. The layers were separated and the organics were washed with water three times. After washing, the organics were dried with MgSO4, filtered, and the volatiles were removed under reduced pressure. The mixture was then transferred to a 4-necked flask containing glass boiling chips and fitted with a thermocouple and short-path condenser.
- Example 17 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [23], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
- Example 18 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-4-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [24], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
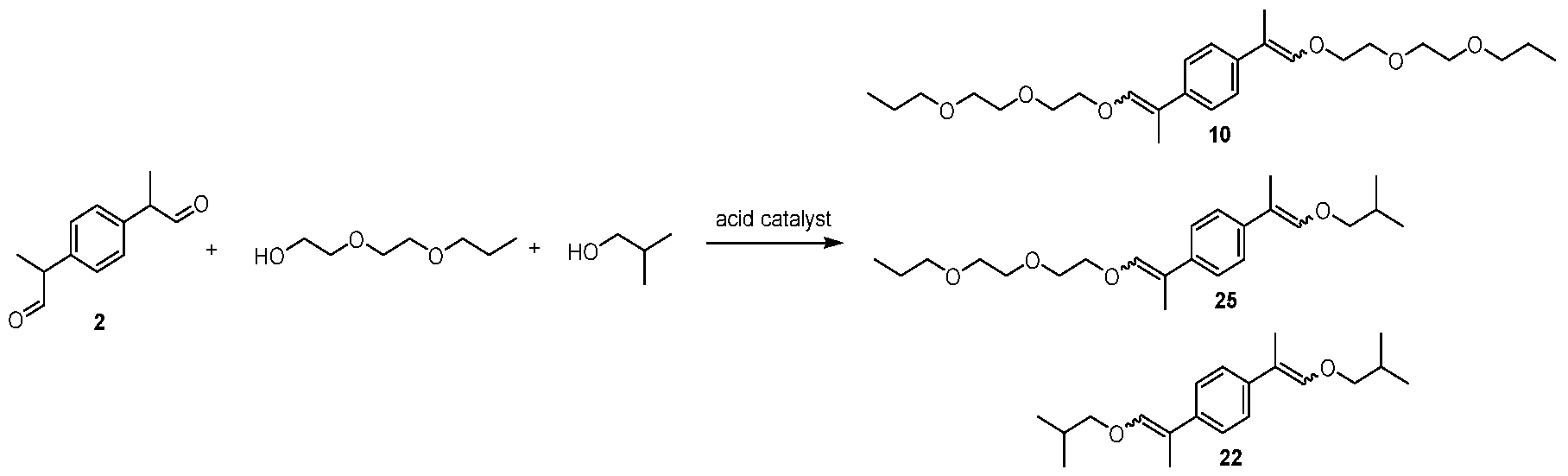
- Example 19 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-4-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [25], and (E,E/Z,Z) - 1,4-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [22]
- Example 20 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1 -(1 -(2- (2-isopropoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [26], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
- Example 21 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-4-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [27], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
- Example 22 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-4-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-
- Example 23 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [29], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
- Example 24 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -butoxyprop-1 -en-2-yl)benzene [30], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21] GC-MS tR : 35.97 min, 41 .73 min, 48.77 min (11 , Exact mass: 478.3 m/z, found: 478.4 m/z), 22.88 min, 24.29 min, 24.45 min, 26.14 min (30, Exact mass: 390.3 m/z, found: 390.3 m/z), 18.15 min, 18.68 min,
- Example 25 A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -isobutoxyprop-1 -en-2- yl)benzene [31], and (E,E/Z,Z) - 1,4-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [22]
- Example 26 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17], (E,E/Z,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [35], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
- Example 27 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17] (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-3-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [36], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
- Example 28 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-3-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [37], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34]
- Example 29 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1 -(1 -(2- (2-isopropoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [38], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32] GC-MS tR : 25.17 min, 26.96 min, 29.48 min (45, Exact mass: 450.3 m/z, found: 450.4 m/z), 19.66 min, 20.12 min, 20.38 min, 20.98 min (38, Exact mass: 362.2 m/z, found: 362.3 m/z), 16.79 min, 17.13
- Example 30 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-3-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [39], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
- Example 31 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-3-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en- 2-yl)benzene [40], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34]
- Example 32 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [41], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
- Example 33 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -butoxyprop-1 -en-2-yl)benzene [42], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
- Example 34 A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -isobutoxyprop-1 -en-2- yl)benzene [43], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34] GC-MS tR : 32.67 min, 36.22 min, 41 .37 min (18, Exact mass: 478.3 m/z, found: 478.4 m/z), 21 .27 min, 22.14 min, 22.36 min, 23.47 min (43, Exact mass: 390.3 m/z, found: 390.3 m/z), 17.18
- Example 35 A mixture of (E,E/Z,Z) - 1 , 4-bis(1-((1 -methoxy propan-2- yl)oxy)prop-1-en-2-yl)benzene [46], (E,E/Z,Z) - 1-(1-((1-methoxypropan-2- yl)oxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2-yl)benzene [47], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
- Example 36 A mixture of (E,E/Z,Z) - 1,4-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [46], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)- 4-(1 -(( 1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [48], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
- Example 37 (E,E/Z,Z) - 1,4-bis(1-((1-methoxypropan-2-yl)oxy)prop-1-en- 2-yl)benzene [46], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1-((1 ⁇ methoxypropan-2-yl)oxy)prop-1-en-2-yl)benzene [49], and (E,E/Z,Z) - 1,4- bis(1 -isobutoxyprop-1 -en-2-yl)benzene [22] GC-MS tR : 19.77 min (46, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.15 min, 18.24 min, 18.69 min, 19.40 min (49, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.58 min, 17.64 min (22, Exact mass: 302.2 m/
- Example 38 A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1-((1-methoxypropan-2- yl)oxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2-yl)benzene [51 ], and (E,E/Z,Z) - 1 ,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
- Example 39 A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)- 3-(1 -(( 1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [52], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33] GC-MS tR : 19.06 min (50, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.86 min, 18.37 min, 18.91 min (52, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.68 min, 18.21 min, 18.73 min (33, Exact mass: 302.2
- Example 40 A mixture of (E,E/Z,Z) - 1, 3-bis(1-((1 -methoxy propan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2- yl)-3-(1 -((1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [53], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2-yl)benzene [34]
- Example 41 A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [54], (E,E/Z,Z) - 1-(1-(3-methoxypropoxy)prop-1-en-2- yl)-4-(1 -propoxyprop-1 -en-2-yl)benzene [55], and (E,E/Z,Z) - 1,4-bis(1- propoxyprop-1 -en-2-yl)benzene [20]
- Example 42 A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [54], (E,E/Z,Z) - 1-(1-butoxyprop-1-en-2-yl)-4-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [56], and (E,E/Z,Z) - 1,4-bis(1- butoxyprop-1 -en-2-yl)benzene [21 ] GC-MS tR : 19.71 min, 20.51 min, 21 .41 min (54, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.87 min, 19.50 min, 20.22 min (56, Exact mass: 318.2 m/z, found: 318.2 m/z), 18.14 min, 18.69 min, 19.24 min (21 , Exact mass: 302.2 m/z, found: 30
- Example 43 (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop-1-en-2- yl)benzene [54], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1 -(3- methoxypropoxy)prop-1-en-2-yl)benzene [57], and (E,E/Z,Z) - 1,4-bis(1- isobutoxyprop-1 -en-2-yl)benzene [22]
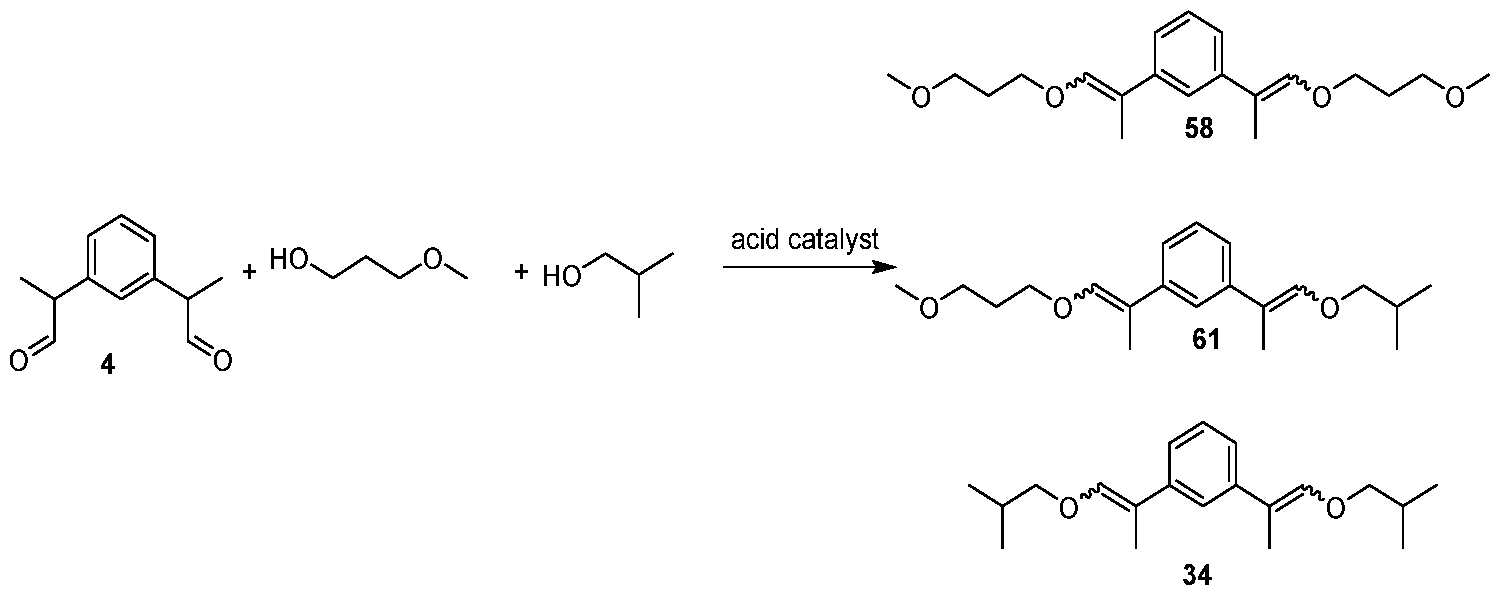
- Example 44 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1 -en-2-yl)benzene [58], 1 -(1 -(3-methoxypropoxy)prop-1 -en-2-yl)-3-(1 - propoxyprop-1 -en-2-yl)benzene [59], and (E,E/Z,Z) - 1,3-bis(1- propoxyprop-1 -en-2-yl)benzene [32] GC-MS tR : 19.05 min, 19.74 min, 20.53 min (58, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.83 min, 18.19 min, 18.36 min, 18.75 min (59, Exact mass: 304.2 m/z, found: 304.1 m/z), 16.73 min, 17.14 min, 17.42 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
- Example 45 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [58], (E,E/Z,Z) - 1-(1-butoxyprop-1-en-2-yl)-3-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [60], and (E,E/Z,Z) - 1,3-bis(1- butoxyprop-1 -en-2-yl)benzene [33]
- Example 46 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [58], (E,E/Z,Z) - 1-(1-isobutoxyprop-1-en-2-yl)-3-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [61], and (E,E/Z,Z) - 1,3-bis(1- isobutoxyprop-1 -en-2-yl)benzene [34]
- Example 47 A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [62], (E,E/Z,Z) - 1-(1-(3-methoxybutoxy)prop-1-en-2-yl)-4- (1 -propoxyprop-1 -en-2-yl)benzene [63], and (E,E/Z,Z) - 1,4-bis(1- propoxyprop-1 -en-2-yl)benzene [20] GC-MS tR : 20.25 min, 21 .33 min, 22.72 min (62, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.52 min, 18.94 min, 19.27 min, 19.75 min (63, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.16 min, 17.54 min, 17.83 min (20, Exact mass: 274.2 m/z,
- Example 48 A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [62], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)-4-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [64], and (E,E/Z,Z) - 1,4-bis(1- butoxyprop-1 -en-2-yl)benzene [21 ]
- Example 49 (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1-en-2- yl)benzene [62], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [65], and (E,E/Z,Z) - 1,4-bis(1- isobutoxyprop-1 -en-2-yl)benzene [22] GC-MS tR : 20.29 min, 21 .33 min, 22.74 min (62, Exact mass: 362.2 m/z, found: 363.2 m/z), 18.75 min, 19.34 min, 19.53 min, 20.22 min (65, Exact mass: 332.2 m/z, found: 332.3 m/z), 17.58 min, 18.06 min, 18.53 min (22, Exact mass: 302.2 m/z
- Example 50 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1-(3-methoxybutoxy)prop-1-en-2-yl)-3- (1 -propoxyprop-1 -en-2-yl)benzene [67], and (E,E/Z,Z) - 1,3-bis(1- propoxyprop-1 -en-2-yl)benzene [32]
- Example 51 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)-3-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [68], and (E,E/Z,Z) - 1,3-bis(1- butoxyprop-1 -en-2-yl)benzene [33]
- Example 52 A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-3-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [69], and (E,E/Z,Z) - 1,3-bis(1- isobutoxyprop-1 -en-2-yl)benzene [34] GC-MS tR : 19.54 min, 20.42 min, 21 .52 min (66, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.21 min, 18.75 min, 18.93 min, 19.47 min (69, Exact mass: 332.2 m/z, found: 332.2 m/z), 17.11 min, 17.57 min, 18.05 min (34, Exact mass: 302.2 m/
- Mass spectra were acquired with a Micromass LCT mass spectrometer, which was coupled to the LC. Mass spectra were collected using electrospray ionization in both the positive-ion and negative ion modes. Ammonium acetate (50 mM in MeOH) was added post column (0.1 mL/min) to enhance ionization efficiency. ES+/ES- scan range was 60- 3300 amu (25 and 75V). GC-MS Instrument Parameters - Agilent 6890N GO with Agilent 5975B VL MSD
- Sample Prep 100 pL sample diluted to 1 mL with toluene; Column: DB-5 30 m x 0.25 mm x 0.25 pm; Oven Ramp: 0-4.5 mins at 40 °C; Ramp 20C/min to 280C, Hold 53.5 mins; Injector: Temperature - 250 °C; Split Flow
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Disclosed are aromatic enol ether compounds that have utility as film-hardening additives for coating formulations. The aromatic enol ether compounds have particular utility as film-hardening additives for water-based coating formulations. The aromatic enol ether compounds provide improvements in hardness and hardness related properties such as block resistance, without contributing to the volatile organic content of the coating formulation.
Description
AROMATIC ENOL ETHERS
FIELD OF THE INVENTION
[0001] This application relates to chemistry generally. In particular, this application relates to enol ethers and more particularly to aromatic enol ethers.
BACKGROUND OF THE INVENTION
[0002] Enol ethers are useful in a variety of chemical applications such as plasticizers, diluents, wetting agents and paint additives and as intermediates in chemical processes. Plasticizers, diluents, wetting agents and paint additives often are volatile and evaporate into the atmosphere during use. For example, coalescing aids that are added to water-based paints, aid in film formation in latex emulsions. The coalescing aids lower the glass transition temperature (Tg) of the latex polymer and as the paint dries, the polymers that have been softened by the coalescing aid are allowed to flow together and form a film after the water has left the system. Coalescing aids that are volatile evaporate out of the film. This allows the polymer to return to the original Tg thereby giving harder films for better block and print resistant coatings.
[0003] Due to environmental concerns, the use of volatile materials such as paint additives, plasticizers, diluents, wetting agents and coalescing aids are increasing undesirable. There is a need for materials that can be used as plasticizers, diluents, wetting agents and paint additives that exhibit low volatility.
SUMMARY OF THE INVENTION
[0004] The invention is set forth in the appended claims.
I wherein:
A is (C8-20) alkylaryl;
R1a and R1b are independently (Ci-i2)alkyl, (C2-i2)alkenyl, (C3- s)cycloalkyl, or 5- to 9-membered aryl or
each R4 is independently (Ci-i2)alkyl, or -C(O)R5; each R5 is (Ci-i2)alkyl unsubstituted or substituted by R6, (C2-i2)alkenyl unsubstituted or substituted by R6, (C3-8)cycloalkyl, or 5- to 9- membered aryl; each R6 is (Ci-4)alkoxy, or oxo; and each n is independently an integer from 1 to 15 or
each R7 is independently (Ci-i2)alkyl.
DETAILED DESCRIPTION
Definitions:
[0006] In this specification and in the claims that follow, reference will be made to a number of terms, which shall be defined to have the following meanings.
[0007] “Alkyl” means an aliphatic hydrocarbon. The alkyl can specify the number of carbon atoms, for example (C1 -5)alkyl. Unless otherwise specified, the alkyl group can be unbranched or branched. In some embodiments, the alkyl group is branched. In some embodiments, the alkyl group is unbranched. Non-limiting examples of alkanes include methane, ethane, propane, isopropyl (i.e., branched propyl), butyl, and the like.
[0008] “Alkenyl” means an aliphatic hydrocarbon with one or more unsaturated carbon-carbon bonds. The alkenyl can specify the number of carbon atoms, for example (C2-12)alkenyl. Unless otherwise specified, the alkyl group can be unbranched or branched. In some embodiments, the alkyl group is branched. In some embodiments, the alkyl group is unbranched.
Non-limiting examples of alkanes include ethenyl, propenyl, butenyl, hexa-3,5- dienyl, and the like.
[0009] “Alcohol” means a chemical containing one or more hydroxyl groups.
[0010] “Aldehyde” means a chemical containing one or more -C(O)H groups.
[0011] “Cycloalkyl” means a cyclic hydrocarbon compound. The cycloalkyl can specify the number of carbon atoms in ring system, for example (C3- 8)cycloalkyl. Non-limiting examples of cycloalkyl include cyclopropyl, cyclobutyl, cyclohexyl, and cyclooctyl.
[0012] “Aryl” means a ring system made up carbon atoms that has at least one ring that is aromatic. The carbon units making up the aryl ring may be specified, for example 5- to 9-membered aryl. Non-limiting examples of aryl include phenyl, naphthyl, 2,3-dihydro-1 H-indene, and 1 , 2,3,4- tetrahydronaphthalene.
[0013] Values may be expressed as “about” or “approximately” a given number. Similarly, ranges may be expressed herein as from “about” one particular value and/or to “about” or another particular value. When such a range is expressed, another aspect includes from the one particular value and/or to the other particular value. Similarly, when values are expressed as
approximations, by use of the antecedent “about,” it will be understood that the particular value forms another aspect.
[0014] As used herein, the terms “a,” “an,” and “the” mean one or more.
[0015] As used herein, the term “and/or,” when used in a list of two or more items, means that any one of the listed items can be employed by itself or any combination of two or more of the listed items can be employed. For example, if a compound is described as containing components A, B, and/or C, the compound can contain A alone; B alone; C alone; A and B in combination; A and C in combination, B and C in combination; or A, B, and C in combination.
[0016] As used herein, the terms “comprising,” “comprises,” and “comprise” are open-ended transition terms used to transition from a subject recited before the term to one or more elements recited after the term, where the element or elements listed after the transition term are not necessarily the only elements that make up the subject.
[0017] As used herein, the terms “having,” “has,” and “have” have the same open-ended meaning as “comprising,” “comprises,” and “comprise” provided above.
[0018] As used herein, the terms “including,” “includes,” and “include” have the same open-ended meaning as “comprising,” “comprises,” and “comprise” provided above.
[0019] “Chosen from” as used herein can be used with “or” or “and.” For example, Y is chosen from A, B, and C means Y can be individually A, B, or C. Alternatively, Y is chosen from A, B, or C means Y can be individually A, B, or C; or a combination of A and B, A and C, B and C, or A, B, and C.
[0020] Presented herein are novel enol ethers which can be used in applications such as (but not limited to) plasticizers, diluents, wetting agents, coalescing aids and paint additives.
I wherein:
A is (C8-20) alkylaryl;
R1a and R1b are independently (Ci-i2)alkyl, (C2-i2)alkenyl, (C3- s)cycloalkyl, or 5- to 9-membered aryl or
each R4 is independently (Ci-i2)alkyl, or -C(O)R5; each R5 is (Ci-i2)alkyl unsubstituted or substituted by R6, (C2-i2)alkenyl unsubstituted or substituted by R6, (C3-8)cycloalkyl, or 5- to 9- membered aryl; each R6 is (Ci-4)alkoxy, or oxo; and each n is independently an integer from 1 to 15 or
each R7 is independently (Ci-i2)alkyl.
[0022] In some embodiments, A in Formulas I, is 1 ,2-, 1 ,3-, or 1 ,4- disubstituted phenyl. In some embodiments, each n is an integer from 1 to 3. [0023] In some embodiments of Formulas I, each R4 is hydrogen. In some embodiments, each R4 is (Ci-i2)alkyl. In some embodiments, each R4 is independently ethyl. In some embodiments, each R4 is (C2-12)alkenyl. In some embodiments, each R4 is -C(O)R5.
[0024] In some embodiments of Formulas I, each R5 is (Ci-i2)alkyl
unsubstituted or substituted by R6. In some embodiments, each R5 is (Ci- i2)alkenyl unsubstituted or substituted by R6. In some embodiments, each R5 is (C3-8)cycloalkyl. In some embodiments, each R5 is 5- to 9-membered aryl. [0025] In some embodiments of Formulas I each n is an integer from 1 to 2. In some embodiments, each n is an integer from 1 to 3. In some embodiments, each n is an integer from 1 to 4. In some embodiments, each n is an integer from 1 to 5. In some embodiments, n is an integer from 1 to 6. In some embodiments, n is an integer from 1 to 7. In some embodiments, n is an integer from 1 to 8. In some embodiments, n is an integer from 1 to 9. In some embodiments, n is an integer from 1 to 10. In some embodiments, n is an integer from 1 to 11 . In some embodiments, n is an integer from 1 to 12. In some embodiments, n is an integer from 1 to 13. In some embodiments, n is an integer from 1 to 14. In some embodiments, n is an integer from 1 to 15. [0026] In some embodiments, the compounds of Formulas I have a volatile organic content of less than 50 wt % according to ASTM D6886. In some embodiments, the volatile organic content is less than 30 wt %. In some embodiments, the volatile organic content is less than 10 wt %. In some embodiments, the volatile organic content is less than 5 wt %. In some embodiments, the volatile organic content is less than 3 wt %. In some embodiments, the volatile organic content is less than 2 wt %. In some embodiments, the volatile organic content is less than 1 wt %. In some embodiments, the volatile organic content is less than 0.8 wt %.
Compounds
[0027] The enol ether compounds disclosed in the present application exhibit a low volatile organic content (less than 50 wt %, but as low as 0.7 wt % according to ASTM D6886). The enol ethers can be used as reactive filmhardening compounds. Reactive film-hardening compounds react with components in coating compositions to form crosslinks in the films providing improved film properties. When we say that the enol ether compounds of this invention can be used as reactive film-hardening additives, we mean when
added to a coating composition, that a harder film is obtained upon curing the composition than is obtained in the absence of the invention enol ether additives, or that the coating composition exhibits a higher gel fraction than in the absence of the enol ether additive, or that both coating composition hardness and increased gel fraction properties are improved by the addition of the enol ether reactive film-hardening additives.
[0028] Not wishing to be bound by any theory, the increase in hardness observed in a coating that contains the enol ether additives described herein may be the result of a chemical reaction, so that the additives described herein may be described as “reactive” enol ether film-hardening additives. [0029] The enol ether materials described herein can also facilitate the individual latex particles coming together to form a continuous film at a given temperature by reducing the minimum film-forming temperature (MFFT) of the latex polymer.
[0030] In some embodiments, the compound comprises the compounds represented by Formulas I, II and II.
[0031] In some embodiments, the compounds of Formula I are enol ethers represented by Formulas 20-69:
10
and isomers thereof. [0032] The enol ethers depicted by Formulas 5-69 are representative of the enol ethers claimed herein. Isomers of the enol ethers depicted by Formulas 5-69 are expected to be produced during synthesis of the enol ethers depicted by Formulas 5-69. All isomers of the enol ethers depicted by Formulas 5-69 and are within the scope of the claims set forth herein. [0033] The compounds depicted by Formulas I include those having a
weight percent volatile content of less than 50%, as measured according to ASTM Method D6886. This test may be conducted generally by heating the sample in a forced air oven at 110°C. for 60 minutes. The weight loss after the test is deemed to result from a loss of volatiles originally present in the sample; the percent volatile present in the original sample may then be calculated. Although the cited test can be conducted on coating compositions containing other components such as latex polymers, the values cited herein may be obtained from a sample of the additive itself. The weight percent volatile of a film-hardening aid may be used herein as a yardstick to measure the amount of VOC the additive would contribute to the VOC in a particular end use such as a component of a coating composition.
EXAMPLES
[0034] This invention can be further illustrated by the following examples thereof, although it will be understood that these examples are included merely for purposes of illustration and are not intended to limit the scope of the invention unless otherwise specifically indicated. In particular more than one method to make enol ethers is available to the skilled artisan. Methods A, B, and C are described herein.
Abbreviations:
[0035] mL is milliliter; wt % is weight percent; eq is equivalent(s); hrs or h is hour(s); mm is millimeter; m is meter; GC is gas chromatography; °C is degree Celsius; min is minute; tR is retention time; VOC is volatile organic compound; MeP is methyl palmitate; w/v is weight/volume; pL is microliter. RFHA is reactive film-hardening additive.
Preparation of dicarbinol [11 and 2,2'-(1 ,4-phenylene)dipropanal [21
Preparation of dicarbinol 1 :
[0036] KOH (49.2 g, 788 mmol) was dissolved in MeOH (400 mL) contained within a 1 L, 4-necked round-bottom flask fitted with thermocouple, overhead stirrer, and nitrogen inlet atop a reflux condenser. During the addition of KOH, internal temperature reached 60 °C and was maintained there by heating mantle. The solid di-epoxide was added over the course of 1 .5 hrs. The reaction was monitored by 1H NMR (aliquot was taken and dissolved in DMSO-de. Once di-epoxide was completely consumed, the reaction was cooled to ambient temperature, and acetic acid (47.3g, 788 mmol) was added dropwise. Once addition was complete, the volatiles were removed under reduced pressure using a rotary evaporator. The residue was taken up in 250 mL of toluene and then washed with 250 mL of water. The aqueous layer was back-extracted with 250 mL of EtOAc (ethyl acetate). The organics were combined, dried with MgSO4 and simultaneously treated with 5g of activated carbon. The mixture was filtered and volatiles were removed under reduced pressure using a rotary evaporator. Dicarbinol 1 was isolated as a white solid. LC-MS (Column A) tR : 3.80 min (Exact mass : 254.15 m/z, found 254.2 m/z ).
Preparation of 2,2'-(1 ,4-phenylene)dipropanal [21:
[0037] The dicarbinol 1 was then dissolved in formic acid (88%, 98.0 g) contained within a 500 mL, 4-necked round-bottom flask fitted with thermocouple, overhead stirrer, and nitrogen inlet atop a reflux condenser. The mixture was heated to 100 °C. After 6 hrs, additional formic acid was added (98.0 g). After an additional 2 hrs, GC indicated >99% conversion to
dialdehyde 2. The volatiles were then removed under reduced pressure using a rotary evaporator. The residue was taken up in 250 mL of toluene and then washed with a saturated solution of NaHCOa. After layer separation, the organics were dried with MgSC , filtered, and then concentrated. The crude material was then Kugelrohr - distilled at 150 °C/1 mm Hg to isolate the dialdehyde 2 as a colorless oil. GC-MS tR : 14.47 min (Exact mass: 190.10 m/z, found: 190.1 m/z).
[0038] Dicarbinol 3 was prepared in a similar manner to di-carbinol 2 using procedure described in Method A. [LC-MS (Column B) tR : 4.55 min, 4.68 min (Exact mass: 254.15 m/z, found 254.2 m/z)]. Di-aldehyde 4 was prepared in a similar manner to di-aldehyde 2 using Method A.
GC-MS tR : 14.22 min (Exact mass: 190.10 m/z, found: 190.1 m/z).
Method A:
[0039] Di-aldehyde was added to a nitrogen-swept, round-bottom flask fitted with overhead-stirrer, thermocouple, and Dean-Stark. Glycol alcohol solvent (5 equiv.) was added all at once, followed by the addition of toluene (2x mass of aldehyde used). Sodium methanesulfonate (0.025 equiv.) was added to the flask, followed by the addition of methanesulfonic acid (0.025 equiv.). The reaction was heated to reflux and held at that temperature for 15 hrs. Toluene was removed under reduced pressure using a rotary evaporator. Then 50% caustic (0.024 equiv.) was added all at once. The mixture was fractionally distilled under reduced pressure.
Example 1 : Preparation of (E,E/Z,Z) - 1,4-bis(1-(2-butoxyethoxy)prop-1- en-2-yl)benzene [5]
GC-MS tR : 22.83 min, 24.34 min, 26.08 min (Exact mass: 390.28 m/z, found 390.3 m/z).
Example 2: Preparation of (E,E/Z,Z) - 1 ,4-bis(1 -((1-((1 -methoxy propan-2- yl)oxy)propan-2-yl)oxy)prop-1 -en-2-yl)benzene [6]
mixture of isomers
GC-MS tR : 25.8 min, 26.2 min, 28.3 min, 28.9 min (Exact mass: 450.30 m/z, found 450 m/z).
Method B:
[0040] To a 4-necked round-bottom flask fitted with an overhead stirrer, thermocouple, and nitrogen inlet was added acetic anhydride (5 equiv.) and sodium bisulfate monohydrate (0.025 equiv.). The mixture was then heated to 65 °C and held for 30 minutes. The dicarbinol was then added dropwise over the course of 4-5 hrs via a pressure-equalizing addition funnel. Once the addition was complete, the reaction was checked by GC. Once complete, the mix was transferred to a 1 -neck round-bottom flask and the excess acetic anhydride/acetic acid was removed under reduced pressure using a rotary evaporatory. The crude was taken up in toluene. The organics were then washed with 10% caustic (x2) solution and then 5% ammonium hydroxide solution. The mixture was dried with MgSO4 and simultaneously treated with activated carbon. After filtration, the volatiles were removed under reduced pressure using a rotary evaporator. Light-boiling impurities were removed by distillation. The enol ether / 1 ,1 -disubstituted olefin was Kugelrohr-distilled to afford product blends.
Example 3: A mixture of (E,E/Z,Z) - 1,4-bis(1 (1-((1 -methoxypropan-2- yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [6a], (E,Z) -
methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en-2-yl)-4-(3-((1 -((1 - methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [6b], and 1 ,4-bis(3-((1 -((1 -methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en- 2-yl)benzene [6c]
GC-MS tR : 25.80 min, 26.28 min, 26.80 min, 27.30 min, 28.38 min, 28.94 min (Exact mass: 450.30 m/z, found: 450.4 m/z).
Example 4: A mixture of (E,E/Z,Z) - 1,4-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-12-en-13-yl)benzene [7a], (E,Z) - 4,7,10-trimethyl-13-(4- (4,7,10-trimethyl-2,5,8,11 -tetraoxatetradec-13-en-13-yl)phenyl)-2,5,8,11 - tetraoxatetradec-12-ene [7b], and 1,4-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-13-en-13-yl)benzene [7c]
GC-MS tR : 43.08 (broad peak), 69.53 min (broad peak) (Exact mass: 566.38 m/z, found: 566.5 m/z).
Example 5: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [8a], (E,Z) - 1-(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [8b], and 1 ,4-bis(3-(2-(2- methoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [8c]
GC-MS tR : 23.9 min, 24.29 min, 24.48 min, 25.64 min, 25.96 min, 27.63 min (Exact mass: 394.24 m/z, found: 394.3 m/z).
Example 6: a mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [9a], (EZZ) - 1-(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [9b], and 1 ,4-bis(3-(2-(2- ethoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [9c].
GC-MS tR : 25.64 min, 26.23 min, 26.55 min, 28.00 min, 28.47 min, 30.67 min (Exact mass: 422.27 m/z, found: 422.3 m/z).
Example 7: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10a], (E,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [1 Ob], 1 ,4-bis(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [10c].
GC-MS tR : 29.70 min, 30.67 min, 31 .30 min, 33.27 min, 34.18 min, 37.58 min (Exact mass: 450.30 m/z, found: 450.3 m/z).
Example 8: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11a], (E,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)-4-(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11 b], 1 ,4-bis(3-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [11c].
GC-MS tR : 36.07 min, 37.71 min, 38.86 min, 41 .45 min (Exact mass: 478.33 m/z, found: 478.4 m/z).
Example 9: A mixture of (E,E/Z,Z) - 1,4-di(2,5,8,11-tetraoxatetradec-12- en-13-yl)benzene [12a], (E,Z) - 13-(4-(2,5,8,11-tetraoxatetradec-13-en-13- yl)phenyl)-2,5,8,11-tetraoxatetradec-12-ene [12b], and 1 ,4-di(2,5,8,11 - tetraoxatetradec-13-en-13-yl)benzene [12c].
GC-MS tR : 39.51 min, 41 .51 min, 42.78 min, 46.43 min, 48.26 min, 55.04 min (Exact mass: 482.29 m/z, found: 482.4 m/z).
Example 10: A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-((1-methoxypropan-2- yl)oxy)propan-2-yl)oxy)prop-1-en-2-yl)benzene [13a], (E/Z) - 1 -(1 -((1 -(Ci- met hoxy propan-2-y l)oxy)pro pan-2-y l)oxy)prop-1 -en-2-yl)-3-(3-((1 -((1 - methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en-2-yl)benzene [13b], and 1 ,3-bis(3-((1 -((1 -methoxypropan-2-yl)oxy)propan-2-yl)oxy)prop-1 -en- 2-yl)benzene [13c].
GC-MS tR : 22.78 min, 23.14 min, 23.45 min, 23.91 min, 24.27 min, 24.59 min, 25.17 min, 25.58 min, 26.05 min (Exact mass: 450.30 m/z, found: 450.4 m/z).
Example 11 : A mixture of (E,E/Z,Z) - 1,3-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-12-en-13-yl)benzene [14a], (E/Z) - 4,7,10-trimethyl-13-(3- (4,7,10-trimethyl-2,5,8,11 -tetraoxatetradec-13-en-13-yl)phenyl)-2,5,8,11 - tetraoxatetradec-12-ene [14b], and 1,3-bis(4,7,10-trimethyl-2,5,8,11- tetraoxatetradec-13-en-13-yl)benzene [14c]
GC-MS tR : 41 .84 - 43.72 min (broad peak) (Exact mass: 566.38 m/z, found: 566.5 m/z).
Example 12: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [15a], (E/Z) - 1-(1-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- methoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [15b], 1 ,3-bis(3-(2-(2- methoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [15c].
GC-MS tR : 22.57 min, 22.91 min, 23.08 min, 23.79 min, 24.08 min, 25.43 min (Exact mass: 394.24 m/z, found: 394.3 m/z).
Example 13-1 and Example 13-2: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [16a], (E/Z) - 1-(1-(2-(2- ethoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2-
ethoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [16b], and 1 ,3-bis(3-(2-(2- ethoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [16c].
GC-MS tR : 24.07 min, 24.73 min, 24.80 min, 25.72 min, 25.87 min, 27.78 min (Exact mass: 422.27 m/z, found: 422.3 m/z). 0.05 equiv. of sodium bisulfate used.
Example 13-1 : 0.025 equiv. of sodium bisulfate used. Ratio of 16a:16b: 16c = 1.0:1 .7:3.6.
Example 13-2: 0.50 equiv. of sodium bisulfate used. Ratio of 16a:16b:16c = 1.0:2.7:10.
Example 14: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [17a], (E/Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17b], and 1 ,3-bis(3-(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)benzene [17c]
GC-MS tR : 27.42 min, 28.16 min, 28.63 min, 29.80 min, 30.61 min, 33.20 min (Exact mass: 450.30 m/z, found: 450.4 m/z).
Example 15: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18a], (E/Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)-3-(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18b], and 1 ,3-bis(3-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18c].
GC-MS tR : 32.75 min, 33.89 min, 34.68 min, 36.20 min, 37.42 min, 41 .29 min (Exact mass: 478.33 m/z, found: 478.4 m/z).
Example 16: A mixture of (E,E/Z,Z) - 1,3-di(2,5,8,11-tetraoxatetradec-12- en-13-yl)benzene [19a], (E/Z) - 13-(3-(2,5,8,11-tetraoxatetradec-13-en-13-
yl)phenyl)-2,5,8,11-tetraoxatetradec-12-ene [19b], and 1 ,3-di(2,5,8,11 - tetraoxatetradec-13-en-13-yl)benzene [19c].
GC-MS tR : 35.57 min, 36.92 min, 37.81 min, 39.66 min, 41 .11 min, 45.59 min (Exact mass: 482.29 m/z, found: 482.3 m/z).
Method C:
[0041] To a 4-necked round-bottom flask fitted with an overhead stirrer, thermocouple, and nitrogen inlet was added dialdehyde 2 or 4 (1 equiv.), glycol ether solvent (5 equiv.) and lower-boiling, aliphatic alcohol (5 equiv.). Phosphoric acid (0.5 equiv., 85% in water) was added all at once. A liquid level was marked on the reaction flask. The mixture was sparged (subsurface) with nitrogen at a flow rate of 250 mL/min. The system was then pulled under vacuum. The reaction mixture was heated to 75°C. The lower- boiling alcohol component was continuously removed from the reaction to facilitate water-removal. Over the course of the reaction, fresh lower-boiling alcohol component was added back to the reaction flask such that the liquid volume was maintained up to the marked liquid level on the flask. Once GC and NMR confirmed >99.5% conversion of aldehyde, the reaction mixture was cooled to ambient temperature and then diluted with a minimal amount of toluene. This mixture was then poured into water containing 1 .25 equiv. of NaOH. The layers were separated and the organics were washed with water three times. After washing, the organics were dried with MgSO4, filtered, and
the volatiles were removed under reduced pressure. The mixture was then transferred to a 4-necked flask containing glass boiling chips and fitted with a thermocouple and short-path condenser. The excess, unreacted alcohols were first removed under reduced pressure with heat. Once complete, 0.05 equiv. of phosphoric acid was added to the reactor. The mixture was further heated to facilitate conversion to enol ether. The reaction was heated until the rate of alcohol production dropped substantially and NMR or GC analysis indicated >95% conversion to the desired product.
Example 17: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [23], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 29.61 min, 33.23 min, 37.51 min (10, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.85 min, 21 .57 min, 21 .83 min, 22.75 min (23, Exact mass: 362.2 m/z, found: 362.3 m/z), 17.20 min, 17.52 min, 17.83 min (20, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 18: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-4-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2-
yl)benzene [24], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
GC-MS tR : 29.65 min, 33.18 min, 37.42 min (10, Exact mass: 450.3 m/z, found: 450.4 m/z), 21 .71 min, 22.84 min, 22.97 min, 24.34 min (24, Exact mass: 376.3 m/z, found: 376.3 m/z), 18.15 min, 18.68 min, 19.22 min (21 , Exact mass: 302.2 m/z, found: 302.3 m/z).
Example 19: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [10], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-4-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [25], and (E,E/Z,Z) - 1,4-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [22]
GC-MS tR : 29.60 min, 33.18 min, 37.48 min (10, Exact mass: 450.3 m/z, found: 450.4 m/z), 21.17 min, 22.28 min, 23.51 min (25, Exact mass: 376.3
m/z, found: 376.3 m/z), 17.58 min, 18.06 min, 18.55 min (22, Exact mass: 302.2 m/z, found: 302.3 m/z).
Example 20: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1 -(1 -(2- (2-isopropoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [26], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 27.02 min, 29.68 min, 32.85 min (44, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.32 min, 21 .01 min, 21 .23 min, 21 .98 min (26, Exact mass: 362.2 m/z, found: 362.3 m/z), 17.17 min, 17.55 min, 17.84 min (20, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 21 : A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-4-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [27], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
GC-MS tR : 26.94 min, 29.67 min, 32.88 min (44, Exact mass: 450.3 m/z, found: 450.4 m/z), 21 .08 min, 22.19 min, 23.43 min (27, Exact mass: 376.3 m/z, found: 376.3 m/z), 18.14 min, 18.68 min, 19.22 min (21 , Exact mass:
302.2 m/z, found: 302.3 m/z).
Example 22: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [44], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-4-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-
2-yl)benzene [28], and (E,E/Z,Z) - 1,4-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [22]
GC-MS tR : 27.00 min, 29.67 min, 32.92 min (44, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.59 min, 21 .61 min, 21 .74 min, 22.71 min (28, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.63 min, 18.06 min, 18.55 min (22, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 23: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2- yl)benzene [29], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 39.95 min, 41 .67 min, 48.54 min (11 , Exact mass: 478.3 m/z, found: 478.4 m/z), 21 .82 min, 22.69 min, 23.13 min, 24.10 min (29, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.20 min, 17.57 min, 17.86 min (20, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 24: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -butoxyprop-1 -en-2-yl)benzene [30], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
GC-MS tR : 35.97 min, 41 .73 min, 48.77 min (11 , Exact mass: 478.3 m/z, found: 478.4 m/z), 22.88 min, 24.29 min, 24.45 min, 26.14 min (30, Exact mass: 390.3 m/z, found: 390.3 m/z), 18.15 min, 18.68 min, 19.22 min (21 , Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 25: A mixture of (E,E/Z,Z) - 1,4-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [11], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-4-(1 -isobutoxyprop-1 -en-2- yl)benzene [31], and (E,E/Z,Z) - 1,4-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [22]
GC-MS tR : 36.01 min, 41 .71 min, 48.67 min (11 , Exact mass: 478.3 m/z, found: 478.4 m/z), 22.21 min, 23.51 min, 23.63 min, 25.13 min (31 , Exact mass: 390.3 m/z, found: 390.3 m/z), 17.60 min, 18.08 min, 18.56 min (22, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 26: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17], (E,E/Z,Z) - 1 -(1 -(2-(2- propoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [35], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 27.34 min, 29.70 min, 32.94 min (17, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.09 min, 20.60 min, 20.88 min, 21 .58 min (35, Exact mass: 362.2 m/z, found: 362.3 m/z), 16.80 min, 17.16 min, 17.46 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 27: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17] (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-3-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [36], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 27.35 min, 29.69 min, 32.96 min (17, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.79 min, 21 .62 min, 21 .77 min, 22.84 min (36, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.71 min, 18.18 min, 18.68 min (33, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 28: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- propoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [17], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-3-(1 -(2-(2-propoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [37], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34]
GC-MS tR : 27.35 min, 29.71 min, 32.95 min (17, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.35 min, 21 .08 min, 21 .24 min, 22.18 min (37, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.16 min, 17.61 min, 18.05 min (34, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 29: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1 -(1 -(2- (2-isopropoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [38], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 25.17 min, 26.96 min, 29.48 min (45, Exact mass: 450.3 m/z, found: 450.4 m/z), 19.66 min, 20.12 min, 20.38 min, 20.98 min (38, Exact mass: 362.2 m/z, found: 362.3 m/z), 16.79 min, 17.13 min, 17.45 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 30: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1-(1- butoxyprop-1 -en-2-yl)-3-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en-2- yl)benzene [39], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 25.16 min, 26.91 min, 29.46 min (45, Exact mass: 450.3 m/z, found: 450.4 m/z), 20.29 min, 21 .07 min, 21 .19 min, 22.14 min (39, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.67 min, 18.17 min, 18.67 min (33, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 31 : A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- isopropoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [45], (E,E/Z,Z) - 1-(1- isobutoxyprop-1 -en-2-yl)-3-(1 -(2-(2-isopropoxyethoxy)ethoxy)prop-1 -en- 2-yl)benzene [40], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34]
GC-MS tR : 25.18 min, 26.96 min, 29.51 min (45, Exact mass: 450.3 m/z, found: 450.4 m/z), 19.88 min, 20.58 min, 20.70 min, 21 .54 min (40, Exact mass: 376.3 m/z, found: 376.3 m/z), 17.16 min, 17.63 min, 18.05 min (34, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 32: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2- yl)benzene [41], and (E,E/Z,Z) - 1,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 32.75 min, 36.17 min, 41 .29 min (18, Exact mass: 478.3 m/z, found: 478.4 m/z), 20.95 min, 21 .58 min, 21 .95 min, 22.72 min (41 , Exact mass: 376.3 m/z, found: 376.3 m/z), 16.82 min, 17.16 min, 17.48 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 33: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1 -(1 -(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -butoxyprop-1 -en-2-yl)benzene [42], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 32.70 min, 36.14 min, 41 .21 min (18, Exact mass: 478.3 m/z, found: 478.4 m/z), 21 .83 min, 22. I min, 23.02 min, 24.29 min (42, Exact mass: 390.3 m/z, found: 390.3 m/z), 17.73 min, 18.18 min, 18.69 min (33, Exact mass: 302.2 m/z, found: 302.1 m/z),
Example 34: A mixture of (E,E/Z,Z) - 1,3-bis(1-(2-(2- butoxyethoxy)ethoxy)prop-1-en-2-yl)benzene [18], (E,E/Z,Z) - 1-(1-(2-(2- butoxyethoxy)ethoxy)prop-1 -en-2-yl)-3-(1 -isobutoxyprop-1 -en-2- yl)benzene [43], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2- yl)benzene [34]
GC-MS tR : 32.67 min, 36.22 min, 41 .37 min (18, Exact mass: 478.3 m/z, found: 478.4 m/z), 21 .27 min, 22.14 min, 22.36 min, 23.47 min (43, Exact mass: 390.3 m/z, found: 390.3 m/z), 17.18 min, 17.64 min, 18.06 min (34, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 35: A mixture of (E,E/Z,Z) - 1 , 4-bis(1-((1 -methoxy propan-2- yl)oxy)prop-1-en-2-yl)benzene [46], (E,E/Z,Z) - 1-(1-((1-methoxypropan-2- yl)oxy)prop-1 -en-2-yl)-4-(1 -propoxyprop-1 -en-2-yl)benzene [47], and (E,E/Z,Z) - 1,4-bis(1 -propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 19.77 min (46, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.28 min, 18.41 min, 18.69 min (47, Exact mass: 304.2 m/z, found: 304.1 m/z), 17.23 min, 17.49 min, 17.64 min (20, Exact mass: 274.2 m/z, found: 274.2 m/z).
Example 36: A mixture of (E,E/Z,Z) - 1,4-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [46], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)- 4-(1 -((1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [48], and (E,E/Z,Z) - 1,4-bis(1 -butoxyprop-1 -en-2-yl)benzene [21]
GC-MS tR : 19.79 min (46, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.88 min, 18.95 min, 19.04 min, 19.50 min (48, Exact mass: 318.2 m/z, found: 318.2 m/z), 18.16 min, 18.70 min, 19.20 min (21 , Exact mass: 302.2 m/z, found: 302.2 m/z).
Example 37: (E,E/Z,Z) - 1,4-bis(1-((1-methoxypropan-2-yl)oxy)prop-1-en- 2-yl)benzene [46], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1-((1 ■ methoxypropan-2-yl)oxy)prop-1-en-2-yl)benzene [49], and (E,E/Z,Z) - 1,4- bis(1 -isobutoxyprop-1 -en-2-yl)benzene [22]
GC-MS tR : 19.77 min (46, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.15 min, 18.24 min, 18.69 min, 19.40 min (49, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.58 min, 17.64 min (22, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 38: A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1-((1-methoxypropan-2- yl)oxy)prop-1 -en-2-yl)-3-(1 -propoxyprop-1 -en-2-yl)benzene [51 ], and (E,E/Z,Z) - 1 ,3-bis(1 -propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 18.54 min, 19.04 min (50, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.78 min, 17.85 min (51 , Exact mass: 304.2 m/z, found: 304.1 m/z),
16.75 min, 17.14 min, 17.46 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 39: A mixture of (E,E/Z,Z) - 1,3-bis(1-((1-methoxypropan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)- 3-(1 -((1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [52], and (E,E/Z,Z) - 1,3-bis(1 -butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 19.06 min (50, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.86 min, 18.37 min, 18.91 min (52, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.68 min, 18.21 min, 18.73 min (33, Exact mass: 302.2 m/z, found: 302.2 m/z).
Example 40: A mixture of (E,E/Z,Z) - 1, 3-bis(1-((1 -methoxy propan-2- yl)oxy)prop-1-en-2-yl)benzene [50], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2- yl)-3-(1 -((1 -methoxypropan-2-yl)oxy)prop-1 -en-2-yl)benzene [53], and (E,E/Z,Z) - 1,3-bis(1 -isobutoxyprop-1 -en-2-yl)benzene [34]
GC-MS tR : 18.46 min, 19.07 min (50, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.96 min (53, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.14 min, 17.69 min (34, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 41 : A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [54], (E,E/Z,Z) - 1-(1-(3-methoxypropoxy)prop-1-en-2- yl)-4-(1 -propoxyprop-1 -en-2-yl)benzene [55], and (E,E/Z,Z) - 1,4-bis(1- propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 19.71 min, 20.54 min, 21 .42 min (54, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.31 min, 18.75 min, 18.90 min, 19.34 min (55, Exact mass: 304.2 m/z, found: 304.1 m/z), 17.19 min, 17.54 min, 17.83 min (20,
Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 42: A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [54], (E,E/Z,Z) - 1-(1-butoxyprop-1-en-2-yl)-4-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [56], and (E,E/Z,Z) - 1,4-bis(1- butoxyprop-1 -en-2-yl)benzene [21 ]
GC-MS tR : 19.71 min, 20.51 min, 21 .41 min (54, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.87 min, 19.50 min, 20.22 min (56, Exact mass: 318.2 m/z, found: 318.2 m/z), 18.14 min, 18.69 min, 19.24 min (21 , Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 43: (E,E/Z,Z) - 1,4-bis(1-(3-methoxypropoxy)prop-1-en-2- yl)benzene [54], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1 -(3- methoxypropoxy)prop-1-en-2-yl)benzene [57], and (E,E/Z,Z) - 1,4-bis(1- isobutoxyprop-1 -en-2-yl)benzene [22]
GC-MS tR : 19.74 min, 20.54 min, 21 .42 min (54, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.61 min, 19.16 min, 19.76 min (57, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.57 min, 18.06 min, 18.53 min (22, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 44: A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1 -en-2-yl)benzene [58], 1 -(1 -(3-methoxypropoxy)prop-1 -en-2-yl)-3-(1 - propoxyprop-1 -en-2-yl)benzene [59], and (E,E/Z,Z) - 1,3-bis(1- propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 19.05 min, 19.74 min, 20.53 min (58, Exact mass: 334.2 m/z, found: 334.2 m/z), 17.83 min, 18.19 min, 18.36 min, 18.75 min (59, Exact mass: 304.2 m/z, found: 304.1 m/z), 16.73 min, 17.14 min, 17.42 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 45: A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [58], (E,E/Z,Z) - 1-(1-butoxyprop-1-en-2-yl)-3-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [60], and (E,E/Z,Z) - 1,3-bis(1- butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 19.06 min, 19.72 min, 20.53 min (58, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.33 min, 18.86 min, 18.89 min, 19.50 min (60, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.65 min, 18.15 min, 18.68 min (33, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 46: A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxypropoxy)prop- 1-en-2-yl)benzene [58], (E,E/Z,Z) - 1-(1-isobutoxyprop-1-en-2-yl)-3-(1-(3- methoxypropoxy)prop-1-en-2-yl)benzene [61], and (E,E/Z,Z) - 1,3-bis(1- isobutoxyprop-1 -en-2-yl)benzene [34]
GC-MS tR : 19.06 min, 19.74 min, 20.51 min (58, Exact mass: 334.2 m/z, found: 334.2 m/z), 18.06 min, 18.55 min, 18.58 min, 19.15 min (61 , Exact mass: 318.2 m/z, found: 318.2 m/z), 17.11 min, 17.61 min, 18.04 min (34,
Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 47: A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [62], (E,E/Z,Z) - 1-(1-(3-methoxybutoxy)prop-1-en-2-yl)-4- (1 -propoxyprop-1 -en-2-yl)benzene [63], and (E,E/Z,Z) - 1,4-bis(1- propoxyprop-1 -en-2-yl)benzene [20]
GC-MS tR : 20.25 min, 21 .33 min, 22.72 min (62, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.52 min, 18.94 min, 19.27 min, 19.75 min (63, Exact mass: 318.2 m/z, found: 318.2 m/z), 17.16 min, 17.54 min, 17.83 min (20, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 48: A mixture of (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [62], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)-4-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [64], and (E,E/Z,Z) - 1,4-bis(1- butoxyprop-1 -en-2-yl)benzene [21 ]
GC-MS tR : 20.25 min, 21 .32 min, 22.71 min (62, Exact mass: 362.2 m/z, found: 362.3 m/z), 19.08 min, 19.74 min, 19.94 min, 20.72 min (64, Exact mass: 332.2 m/z, found: 332.3 m/z),18.12 min, 18.67 min, 19.22 min (21 ,
Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 49: (E,E/Z,Z) - 1,4-bis(1-(3-methoxybutoxy)prop-1-en-2- yl)benzene [62], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-4-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [65], and (E,E/Z,Z) - 1,4-bis(1- isobutoxyprop-1 -en-2-yl)benzene [22]
GC-MS tR : 20.29 min, 21 .33 min, 22.74 min (62, Exact mass: 362.2 m/z, found: 363.2 m/z), 18.75 min, 19.34 min, 19.53 min, 20.22 min (65, Exact mass: 332.2 m/z, found: 332.3 m/z), 17.58 min, 18.06 min, 18.53 min (22, Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 50: A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1-(3-methoxybutoxy)prop-1-en-2-yl)-3- (1 -propoxyprop-1 -en-2-yl)benzene [67], and (E,E/Z,Z) - 1,3-bis(1- propoxyprop-1 -en-2-yl)benzene [32]
GC-MS tR : 19.52 min, 20.39 min, 21 .54 min (66, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.03 min, 18.40 min, 18.68 min, 19.09 min (67, Exact mass: 318.2 m/z, found: 318.2 m/z), 16.76 min, 17.09 min, 17.42 min (32, Exact mass: 274.2 m/z, found: 274.1 m/z).
Example 51 : A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1 -butoxyprop-1 -en-2-yl)-3-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [68], and (E,E/Z,Z) - 1,3-bis(1- butoxyprop-1 -en-2-yl)benzene [33]
GC-MS tR : 19.52 min, 20.39 min, 21 .54 min (66, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.52 min, 19.08 min, 19.25 min, 19.89 min (68, Exact mass: 332.2 m/z, found: 332.3 m/z), 17.67 min, 18.15 min, 18.67 min (33,
Exact mass: 302.2 m/z, found: 302.1 m/z).
Example 52: A mixture of (E,E/Z,Z) - 1,3-bis(1-(3-methoxybutoxy)prop-1- en-2-yl)benzene [66], (E,E/Z,Z) - 1-(1 -isobutoxyprop-1 -en-2-yl)-3-(1 -(3- methoxybutoxy)prop-1-en-2-yl)benzene [69], and (E,E/Z,Z) - 1,3-bis(1- isobutoxyprop-1 -en-2-yl)benzene [34]
GC-MS tR : 19.54 min, 20.42 min, 21 .52 min (66, Exact mass: 362.2 m/z, found: 362.3 m/z), 18.21 min, 18.75 min, 18.93 min, 19.47 min (69, Exact mass: 332.2 m/z, found: 332.2 m/z), 17.11 min, 17.57 min, 18.05 min (34, Exact mass: 302.2 m/z, found: m/z).
[0042] The examples described above were defined as non-VOC by ASTM D6886. This method uses MeP as a standard - if compound tR> MeP tR, compound is defined as non-VOC.
Conditions - Agilent 1100 LC
• Sample Prep: 2-3 mg/mL in DMSO
• Column A: Zorbax XDB-C18 x 4.6 mm, 5 pm
• Column B: Poroshell EC-C18 50 x 4.6 mm, 2.7 pm
• Column Temp: 40 °C
• Injection Volume: 2 pL
• DAD: 190-600 nm collection
• Pump Conditions: Initial - 97% water (2.5 mM NFkOAc) (Solvent A) and 3% acetonitrile (Solvent B)
• Mass spectra were acquired with a Micromass LCT mass spectrometer, which was coupled to the LC. Mass spectra were collected using electrospray ionization in both the positive-ion and negative ion modes. Ammonium acetate (50 mM in MeOH) was added post column (0.1 mL/min) to enhance ionization efficiency. ES+/ES- scan range was 60- 3300 amu (25 and 75V).
GC-MS Instrument Parameters - Agilent 6890N GO with Agilent 5975B VL MSD
[0043] Sample Prep: 100 pL sample diluted to 1 mL with toluene; Column: DB-5 30 m x 0.25 mm x 0.25 pm; Oven Ramp: 0-4.5 mins at 40 °C; Ramp 20C/min to 280C, Hold 53.5 mins; Injector: Temperature - 250 °C; Split Flow
- 65 mL/min; Carrier Flow Rate - 1 .3 mL/min; Volume - 1 .0 pL; MS: Transfer Line - 280 °C; Ion Source Temp - 230 °C; Mass Range - 34 -700 amu. Methyl palmitate tR = 16.6 min using the above method.
[0044] The invention has been described in detail with reference to the embodiments disclosed herein, but it will be understood that variations and modifications can be effected within the spirit and scope of the invention.
Claims
I wherein:
A is (C8-20) alkylaryl;
R1a and R1b are independently (Ci-i2)alkyl, (C2-i2)alkenyl, (C3- s)cycloalkyl, or 5- to 9-membered aryl or
each R4 is independently (Ci-i2)alkyl, or -C(O)R5; each R5 is (Ci-i2)alkyl unsubstituted or substituted by R6, (C2-i2)alkenyl unsubstituted or substituted by R6, (C3-8)cycloalkyl, or 5- to 9- membered aryl; each R6 is (Ci-4)alkoxy, or oxo; and each n is independently an integer from 1 to 15 or
each R7 is independently (Ci-i2)alkyl.
2. The compound of claim 1 wherein A is 1 ,2-, 1 ,3-, or 1 ,4-disubstituted phenyl.
55
3. The compound of claim 1 wherein R4 is hydrogen or ethyl.
4. The compound of claim 1 wherein, n is an integer from 1 to 4.
5. The compound of claim 1 having a volatile organic content of less than
50 wt% according to ASTM D6886.
6. An enol ether compound selected from the group consisting of compounds having Formulas 20-69:
57
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202063087920P | 2020-10-06 | 2020-10-06 | |
| US63/087,920 | 2020-10-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022076365A1 true WO2022076365A1 (en) | 2022-04-14 |
Family
ID=81126200
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2021/053505 Ceased WO2022076365A1 (en) | 2020-10-06 | 2021-10-05 | Aromatic enol ethers |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022076365A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4508653A (en) * | 1983-03-14 | 1985-04-02 | Ashland Oil, Inc. | Method for preparing acyloxy styrenes |
| US20040102474A1 (en) * | 2001-07-26 | 2004-05-27 | Stamford Andrew W. | New neuropeptide Y Y5 receptor antagonists |
| WO2021046007A1 (en) * | 2019-09-04 | 2021-03-11 | Eastman Chemical Company | Aromatic enol ethers |
-
2021
- 2021-10-05 WO PCT/US2021/053505 patent/WO2022076365A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4508653A (en) * | 1983-03-14 | 1985-04-02 | Ashland Oil, Inc. | Method for preparing acyloxy styrenes |
| US20040102474A1 (en) * | 2001-07-26 | 2004-05-27 | Stamford Andrew W. | New neuropeptide Y Y5 receptor antagonists |
| WO2021046007A1 (en) * | 2019-09-04 | 2021-03-11 | Eastman Chemical Company | Aromatic enol ethers |
Non-Patent Citations (1)
| Title |
|---|
| DATABASE PubChem substance ANONYMOUS : "70080-06-7 ", XP055933064, retrieved from NCBI Database accession no. 315010944 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| BE1012526A5 (en) | Stabilizer mixture, containing the composition and method for using an organic matter stabilization. | |
| EP2404924B1 (en) | Aqueous solution of piperazinyl-containing silanol compound and making method | |
| EP4025552A1 (en) | Aromatic enol ethers | |
| US4537923A (en) | Polyalkylpiperidinesulfonic acid esters | |
| WO2022076365A1 (en) | Aromatic enol ethers | |
| CN114072374B (en) | Aromatic enol ethers | |
| WO2022125856A1 (en) | Aromatic enol ethers | |
| FI116844B (en) | Reactive film formers | |
| WO2022076359A1 (en) | A process for making enol ethers | |
| US10815179B1 (en) | Aromatic dicarbinols | |
| US10865171B1 (en) | Process to make aromatic enol ethers and olefin isomers of aromatic enol ethers | |
| US11312873B2 (en) | Aromatic enol ether paint additives | |
| JP2025500462A (en) | 1,3-dipole compounds containing epoxy groups and their synthetic intermediates | |
| US2580494A (en) | N-disubstituted-amino t-hydroxy ketones | |
| CN116445031B (en) | Application of 2,5-tetrahydrofuran dimethanol derivatives in the preparation of water-based coatings | |
| WO2021046046A1 (en) | Method of making a dialdeyhde | |
| WO2021046045A1 (en) | Enol ethers | |
| US5200437A (en) | Coating compositions containing alk-1-enyl ethers | |
| Lovett | Zwitterionic quaternary ammonium alkoxides: organic strong bases | |
| KR102660466B1 (en) | Novel polyacyloxymethyl-4,4'-acyloxybiphenyl compound | |
| US2224865A (en) | Hydrogenated di-naphthyl ether resins and the process of preparing the same | |
| WO2024237153A1 (en) | Bis[1-methyl-1-(4-hydroxyalkoxy-3-alkylphenyl) ethyl ]benzene compound and method for producing same, and crystal and method for producing same | |
| US5189184A (en) | 2-(3'-butenyl)-3,4-dihydro-2h-pyrans, their synthesis and use | |
| US5763683A (en) | 2-methylbutoxy ethoxyethanol | |
| JP5612345B2 (en) | 4- (Cyclopentenyl) cyclohexenone derivative and process for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21878328 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 21878328 Country of ref document: EP Kind code of ref document: A1 |