WO2015031725A1 - Transition metal-catalyzed imidation of arenes - Google Patents
Transition metal-catalyzed imidation of arenes Download PDFInfo
- Publication number
- WO2015031725A1 WO2015031725A1 PCT/US2014/053357 US2014053357W WO2015031725A1 WO 2015031725 A1 WO2015031725 A1 WO 2015031725A1 US 2014053357 W US2014053357 W US 2014053357W WO 2015031725 A1 WO2015031725 A1 WO 2015031725A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- substituted
- unsubstituted
- instance
- certain embodiments
- heteroaryl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CCC*(Cc1*2cccc1)(Cc1cccc*11)OS21=C Chemical compound CCC*(Cc1*2cccc1)(Cc1cccc*11)OS21=C 0.000 description 20
- CPOAKRDGWLCBRK-PLNGDYQASA-N C/C(/C=C)=C/N=N\N Chemical compound C/C(/C=C)=C/N=N\N CPOAKRDGWLCBRK-PLNGDYQASA-N 0.000 description 1
- KAIXVZLWZZDPOB-YVMONPNESA-N C/C(/C=C)=N/N=C\N Chemical compound C/C(/C=C)=N/N=C\N KAIXVZLWZZDPOB-YVMONPNESA-N 0.000 description 1
- QFXSZIQEXNKRDU-UTOQUPLUSA-N C/C(/N)=C/N=N\C=C Chemical compound C/C(/N)=C/N=N\C=C QFXSZIQEXNKRDU-UTOQUPLUSA-N 0.000 description 1
- LQCRLOOGZKIXQC-UHFFFAOYSA-N C/C(/N)=N/C=N\C=C Chemical compound C/C(/N)=N/C=N\C=C LQCRLOOGZKIXQC-UHFFFAOYSA-N 0.000 description 1
- JONNOLYHXZFDIU-DAXSKMNVSA-N C/C(/N)=N/N=C\C=C Chemical compound C/C(/N)=N/N=C\C=C JONNOLYHXZFDIU-DAXSKMNVSA-N 0.000 description 1
- OGHDWKZGJMDUKU-UHFFFAOYSA-N CC1=C[I]=NC=C1 Chemical compound CC1=C[I]=NC=C1 OGHDWKZGJMDUKU-UHFFFAOYSA-N 0.000 description 1
- BSKHPKMHTQYZBB-UHFFFAOYSA-N Cc1ccccn1 Chemical compound Cc1ccccn1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 1
- LVILGAOSPDLNRM-UHFFFAOYSA-N Cc1ccncn1 Chemical compound Cc1ccncn1 LVILGAOSPDLNRM-UHFFFAOYSA-N 0.000 description 1
- ITQTTZVARXURQS-UHFFFAOYSA-N Cc1cnccc1 Chemical compound Cc1cnccc1 ITQTTZVARXURQS-UHFFFAOYSA-N 0.000 description 1
- TWGNOYAGHYUFFR-UHFFFAOYSA-N Cc1cncnc1 Chemical compound Cc1cncnc1 TWGNOYAGHYUFFR-UHFFFAOYSA-N 0.000 description 1
- AIKUBOPKWKZULG-UHFFFAOYSA-N Cc1cnncc1 Chemical compound Cc1cnncc1 AIKUBOPKWKZULG-UHFFFAOYSA-N 0.000 description 1
- CAWHJQAVHZEVTJ-UHFFFAOYSA-N Cc1nccnc1 Chemical compound Cc1nccnc1 CAWHJQAVHZEVTJ-UHFFFAOYSA-N 0.000 description 1
- MXDRPNGTQDRKQM-UHFFFAOYSA-N Cc1nnccc1 Chemical compound Cc1nnccc1 MXDRPNGTQDRKQM-UHFFFAOYSA-N 0.000 description 1
- FXKQPQOOZSXQAG-UHFFFAOYSA-N Cc1nnncc1 Chemical compound Cc1nnncc1 FXKQPQOOZSXQAG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/46—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with hetero atoms directly attached to the ring nitrogen atom
- C07D207/48—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C303/00—Preparation of esters or amides of sulfuric acids; Preparation of sulfonic acids or of their esters, halides, anhydrides or amides
- C07C303/36—Preparation of esters or amides of sulfuric acids; Preparation of sulfonic acids or of their esters, halides, anhydrides or amides of amides of sulfonic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/34—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/72—Nitrogen atoms
- C07D213/76—Nitrogen atoms to which a second hetero atom is attached
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/48—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D221/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00
- C07D221/02—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00 condensed with carbocyclic rings or ring systems
- C07D221/04—Ortho- or peri-condensed ring systems
- C07D221/06—Ring systems of three rings
- C07D221/10—Aza-phenanthrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/26—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D333/30—Hetero atoms other than halogen
- C07D333/36—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/52—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes
- C07D333/62—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the hetero ring
- C07D333/68—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
- C07D333/70—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/006—Palladium compounds
- C07F15/0066—Palladium compounds without a metal-carbon linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids [RP(=O)(OH)2]; Thiophosphonic acids ; [RP(=X1)(X2H)2(X1, X2 are each independently O, S or Se)]
- C07F9/40—Esters thereof
- C07F9/4003—Esters thereof the acid moiety containing a substituent or a structure which is considered as characteristic
- C07F9/4021—Esters of aromatic acids (P-C aromatic linkage)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/05—Isotopically modified compounds, e.g. labelled
Definitions
- the present invention provides novel transition metal complexes (e.g., complexes of any one of Formulae (CI) to (C25)) that include an amine-N-oxide motif.
- the invention also provides methods of using the inventive transition metal complexes in preparing N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I)) and aryl or heteroaryl amines (e.g., compounds of Formula (II)).
- the inventive methods involve imidation of arenes and heteroarenes (e.g., compounds of Formula (A)) using an imidating agent (e.g.
- a compound of Formula (B) such as N-fluorobenzenesulfonimide (NFBS or NFSI)) in the presence of a single-electron reductant (e.g., an Ag(I) or Ru(II) salt).
- NFBS N-fluorobenzenesulfonimide
- a single-electron reductant e.g., an Ag(I) or Ru(II) salt
- the present invention provides methods for preparing an N-aryl or N-heteroaryl sulfonimide (e.g. , a compound of Formula (I), or a salt thereof).
- the method typically involves contacting a compound of Formula (A), or a salt thereof, with a compound of Formula (B), or a salt thereof, in the presence of a transition metal complex of any one of Formula (CI) to (C25) and a single-electron reductant to provide the compound of Formula (I), or a salt thereof.
- the compound of Formula (B) is N- fluorobenzenesulfonimide (NFBS or NFSI).
- the transition metal complex is of Formula (CI).
- the transition metal complex is of Formula (C2).
- the transition metal complex is of Formula (C3).
- the transition metal complex is of Formula (C4).
- the transition metal complex is of any one of Formulae (C5) to (C25).
- the single-electron reductant is an Ag(I) or Ru(II) salt.
- a transition metal complex of Formula (CI) for use in the imidation reactions described herein is prepared by contacting a ligand of Formula (Dl), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of Formula (CI):
- a transition metal complex of Formula (C2) is prepared by contacting a ligand of Formula (D2), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of Formula (C2):
- the inventive methods of further include the step of deprotecting a compound of Formula (I), or a salt thereof, with a reductant (e.g. , a mixture of an alkaline earth metal and an alcohol; or a mixture of zinc and an acid), a strong acid, or a nucleophile (e.g. , an alkyl lithium, phenyl lithium, or a Grignard reagent), to provide a compound of Formula (II), or a salt thereof.
- a reductant e.g. , a mixture of an alkaline earth metal and an alcohol; or a mixture of zinc and an acid
- a strong acid e.g. , an alkyl lithium, phenyl lithium, or a Grignard reagent
- the present invention provides complexes of any one of Formulae (CI) to (C25).
- the complex of the invention is of Formula (CI).
- the complex of the invention is of Formula (C2).
- the complex of the invention is of Formula (C3).
- the complex of the invention is of Formula (C4).
- the complex of the invention is of any one of Formulae (C5) to (C25).
- kits are useful.
- the kits are useful for preparing the compounds (e.g. , N-aryl or N-heteroaryl sulfonimides and aryl or heteroaryl amines) described herein.
- the kits are useful for preparing compounds of Formula (I), and salts thereof.
- the kits are useful for preparing compounds of Formula (II), and salts thereof.
- a kit of the invention includes a compound of Formula (B) (e.g., N-fluorobenzenesulfonimide (NFBS or NFSI)), or a salt thereof; a transition metal complex of any one of Formulae (Cl)- (C25); and a single-electron reductant (e.g., an Au(I) or Ru(II) salt).
- a kit further includes an arene or heteroarene described herein (e.g., a compound of Formula (A), or a salt thereof).
- a kit further includes a deprotecting agent (e.g., a reductant, a strong acid, or a nucleophile).
- a kit of the invention may also include a container, solvent, organic or inorganic agent, and/or solid support, as described herein.
- a kit of the invention may further include instructions for using the kit.
- Compounds described herein can comprise one or more asymmetric centers, and thus can exist in various isomeric forms, e.g., enantiomers and/or diastereomers.
- the compounds described herein can be in the form of an individual enantiomer, diastereomer or geometric isomer, or can be in the form of a mixture of stereoisomers, including racemic mixtures and mixtures enriched in one or more stereoisomer.
- Isomers can be isolated from mixtures by methods known to those skilled in the art, including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts; or preferred isomers can be prepared by asymmetric syntheses.
- HPLC high pressure liquid chromatography
- C ⁇ is intended to encompass, C 1 ; C 2 , C 3 , C 4 , C5, C 6 , Ci_6, Ci-5, Ci ⁇ , Ci-3, Ci-2, C 2 -6, C 2 _5, C 2 _4, C 2 _ 3 , C 3 _6, C 3 _ 5 , C 3 ⁇ , C 4 _ 6 , C 4 _ 5 , and C 5 _6.
- aliphatic includes both saturated and unsaturated, straight chain (i.e. , unbranched), branched, acyclic, cyclic, or polycyclic aliphatic hydrocarbons, which are optionally substituted with one or more functional groups.
- aliphatic is intended herein to include, but is not limited to, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, and cycloalkynyl moieties.
- alkyl includes straight, branched and cyclic alkyl groups.
- alkyl alkenyl
- alkynyl alkynyl
- lower alkyl is used to indicate those alkyl groups (cyclic, acyclic, substituted, unsubstituted, branched or unbranched) having 1-6 carbon atoms.
- the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-20 aliphatic carbon atoms. In certain other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1- 10 aliphatic carbon atoms. In yet other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-8 aliphatic carbon atoms. In still other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-6 aliphatic carbon atoms. In yet other
- the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-4 carbon atoms.
- Illustrative aliphatic groups thus include, but are not limited to, for example, methyl, ethyl, n-propyl, isopropyl, cyclopropyl, -CH 2 -cyclopropyl, vinyl, allyl, n-butyl, sec- butyl, isobutyl, tert-butyl, cyclobutyl, -CH 2 -cyclobutyl, n-pentyl, sec-pentyl, isopentyl, tert- pentyl, cyclopentyl, -CH 2 -cyclopentyl, n-hexyl, sec-hexyl, cyclohexyl, -CH 2 -cyclohexyl moieties and the like, which again, may bear one or more substituents.
- Alkenyl groups include, but are not limited to, for example, ethenyl, propenyl, butenyl, l-methyl-2-buten- l- yl, and the like.
- Representative alkynyl groups include, but are not limited to, ethynyl, 2- propynyl (propargyl), 1-propynyl, and the like.
- alkyl refers to a radical of a straight-chain or branched saturated hydrocarbon group having from 1 to 10 carbon atoms (“Ci-w alkyl”). In some embodiments, an alkyl group has 1 to 9 carbon atoms ("Q-9 alkyl”). In some embodiments, an alkyl group has 1 to 8 carbon atoms ("Q-e alkyl”). In some embodiments, an alkyl group has 1 to 7 carbon atoms (“C ⁇ alkyl”). In some embodiments, an alkyl group has 1 to 6 carbon atoms (“Ci_6 alkyl”). In some embodiments, an alkyl group has 1 to 5 carbon atoms (“Q-s alkyl”).
- an alkyl group has 1 to 4 carbon atoms ("Ci ⁇ alkyl”). In some embodiments, an alkyl group has 1 to 3 carbon atoms ("C ⁇ alkyl”). In some embodiments, an alkyl group has 1 to 2 carbon atoms ("C ⁇ alkyl”). In some embodiments, an alkyl group has 1 carbon atom (“Q alkyl”). In some embodiments, an alkyl group has 2 to 6 carbon atoms (“C 2 -6 alkyl”).
- Q-6 alkyl groups include methyl (CO, ethyl (C 2 ), n-propyl (C 3 ), isopropyl (C 3 ), n-butyl (C 4 ), tert-butyl (C 4 ), sec-butyl (C 4 ), iso-butyl (C 4 ), n-pentyl (C 5 ), 3-pentanyl (C 5 ), amyl (C 5 ), neopentyl (C 5 ), 3-methyl-2-butanyl (C 5 ), tertiary amyl (C 5 ), and n-hexyl (C 6 ).
- Additional examples of alkyl groups include n-heptyl (C 7 ), n-octyl (C 8 ), and the like. Unless otherwise specified, each instance of an alkyl group is
- the alkyl group is an unsubstituted Q_ 10 alkyl (e.g., -CH 3 ). In certain embodiments, the alkyl group is a substituted C ⁇ o alkyl.
- haloalkyl is a substituted alkyl group, wherein one or more of the hydrogen atoms are independently replaced by a halogen, e.g., fluoro, bromo, chloro, or iodo.
- a halogen e.g., fluoro, bromo, chloro, or iodo.
- Perhaloalkyl is a subset of haloalkyl, and refers to an alkyl group wherein all of the hydrogen atoms are independently replaced by a halogen, e.g., fluoro, bromo, chloro, or iodo.
- the haloalkyl moiety has 1 to 8 carbon atoms ("Q-s haloalkyl").
- the haloalkyl moiety has 1 to 6 carbon atoms ("C ⁇ haloalkyl”). In some embodiments, the haloalkyl moiety has 1 to 4 carbon atoms ("C ⁇ haloalkyl”). In some embodiments, the haloalkyl moiety has 1 to 3 carbon atoms (“Ci_ 3 haloalkyl”). In some embodiments, the haloalkyl moiety has 1 to 2 carbon atoms (“C ⁇ haloalkyl”). In some embodiments, all of the haloalkyl hydrogen atoms are replaced with fluoro to provide a perfluoroalkyl group.
- haloalkyl hydrogen atoms are replaced with chloro to provide a "perchloroalkyl" group.
- haloalkyl groups include -CF , -CF 2 CF 3 , -CF 2 CF 2 CF 3 , -CCI 3 , -CFC1 2 , -CF 2 C1, and the like.
- alkenyl refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon double bonds, and no triple bonds (“C2-20 alkenyl”).
- an alkenyl group has 2 to 10 carbon atoms ("C 2 -io alkenyl”).
- an alkenyl group has 2 to 9 carbon atoms ("C2-9 alkenyl”).
- an alkenyl group has 2 to 8 carbon atoms (“C 2 - 8 alkenyl”).
- an alkenyl group has 2 to 7 carbon atoms (“C 2 _ 7 alkenyl”).
- an alkenyl group has 2 to 6 carbon atoms ("C 2 -6 alkenyl”). In some embodiments, an alkenyl group has 2 to 5 carbon atoms (“C 2 _5 alkenyl”). In some embodiments, an alkenyl group has 2 to 4 carbon atoms (“C 2 ⁇ alkenyl”). In some embodiments,
- an alkenyl group has 2 to 3 carbon atoms ("C 2 _3 alkenyl").
- an alkenyl group has 2 carbon atoms ("C 2 alkenyl").
- the one or more carbon- carbon double bonds can be internal (such as in 2-butenyl) or terminal (such as in 1-butenyl).
- Examples of C 2 ⁇ alkenyl groups include ethenyl (C 2 ), 1-propenyl (C 3 ), 2-propenyl (C 3 ), 1- butenyl (C 4 ), 2-butenyl (C 4 ), butadienyl (C 4 ), and the like.
- C 2 -6 alkenyl groups include the aforementioned C 2 ⁇ alkenyl groups as well as pentenyl (C 5 ), pentadienyl (C 5 ), hexenyl (C 6 ), and the like. Additional examples of alkenyl include heptenyl (C 7 ), octenyl (Cg), octatrienyl (Cg), and the like. Unless otherwise specified, each instance of an alkenyl group is independently optionally substituted, i.e. , unsubstituted (an "unsubstituted alkenyl") or substituted (a "substituted alkenyl”) with one or more substituents. In certain
- Alkynyl refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon triple bonds, and optionally one or more double bonds ("C 2 - 2 o alkynyl").
- an alkynyl group has 2 to 10 carbon atoms ("C 2 -io alkynyl”).
- an alkynyl group has 2 to 9 carbon atoms (“C 2 _9 alkynyl”).
- an alkynyl group has 2 to 8 carbon atoms (“C 2 _8 alkynyl”).
- an alkynyl group has 2 to 7 carbon atoms ("C 2 _ 7 alkynyl”). In some embodiments, an alkynyl group has 2 to 6 carbon atoms ("C 2 -6 alkynyl”). In some embodiments, an alkynyl group has 2 to 5 carbon atoms (“C 2 _5 alkynyl”). In some embodiments, an alkynyl group has 2 to 4 carbon atoms ("C 2 ⁇ alkynyl”). In some
- an alkynyl group has 2 to 3 carbon atoms ("C 2 _ 3 alkynyl").
- an alkynyl group has 2 carbon atoms ("C 2 alkynyl").
- the one or more carbon- carbon triple bonds can be internal (such as in 2-butynyl) or terminal (such as in 1-butynyl).
- Examples of C 2 - alkynyl groups include, without limitation, ethynyl (C 2 ), 1-propynyl (C 3 ), 2-propynyl (C 3 ), 1-butynyl (C 4 ), 2-butynyl (C 4 ), and the like.
- C 2 -6 alkenyl groups include the aforementioned C 2 - alkynyl groups as well as pentynyl (C 5 ), hexynyl (C 6 ), and the like. Additional examples of alkynyl include heptynyl (C 7 ), octynyl (Cg), and the like. Unless otherwise specified, each instance of an alkynyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted alkynyl") or substituted (a "substituted alkynyl") with one or more substituents. In certain embodiments, the alkynyl group is unsubstituted C 2 _ 10 alkynyl. In certain embodiments, the alkynyl group is substituted C2-10 alkynyl.
- Carbocyclyl or “carbocyclic” refers to a radical of a non-aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (" -io carbocyclyl") and zero heteroatoms in the non-aromatic ring system.
- a carbocyclyl group has 3 to 8 ring carbon atoms ("C 3 _ 8 carbocyclyl”).
- a carbocyclyl group has 3 to 6 ring carbon atoms ("C 3 _6 carbocyclyl”).
- a carbocyclyl group has 3 to 6 ring carbon atoms (“C 3 _6 carbocyclyl”).
- a carbocyclyl group has 5 to 10 ring carbon atoms ("Cs-io carbocyclyl").
- Exemplary C 3 _ 6 carbocyclyl groups include, without limitation, cyclopropyl (C 3 ), cyclopropenyl (C 3 ), cyclobutyl (C 4 ), cyclobutenyl (C 4 ), cyclopentyl (C 5 ), cyclopentenyl (C 5 ), cyclohexyl (C 6 ), cyclohexenyl (C 6 ), cyclohexadienyl (C 6 ), and the like.
- Exemplary C 3 _ 8 carbocyclyl groups include, without limitation, the aforementioned C 3 _ 6 carbocyclyl groups as well as cycloheptyl (C 7 ), cycloheptenyl (C 7 ), cycloheptadienyl (C 7 ), cycloheptatrienyl (C 7 ), cyclooctyl (C 8 ), cyclooctenyl (C 8 ),
- C 3 _io carbocyclyl groups include, without limitation, the aforementioned C 3 _ 8 carbocyclyl groups as well as cyclononyl (C 9 ), cyclononenyl (C 9 ), cyclodecyl (C 10 ), cyclodecenyl (C 10 ), octahydro-lH-indenyl (C 9 ), decahydronaphthalenyl (C 10 ), spiro[4.5]decanyl (C 10 ), and the like.
- the carbocyclyl group is either monocyclic (“monocyclic carbocyclyl”) or contain a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic carbocyclyl”) and can be saturated or can be partially unsaturated.
- “Carbocyclyl” also includes ring systems wherein the carbocyclic ring, as defined above, is fused with one or more aryl or heteroaryl groups wherein the point of attachment is on the carbocyclic ring, and in such instances, the number of carbons continue to designate the number of carbons in the carbocyclic ring system.
- each instance of a carbocyclyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted carbocyclyl") or substituted (a "substituted carbocyclyl") with one or more substituents.
- the carbocyclyl group is unsubstituted C 3 _io carbocyclyl. In certain embodiments, the carbocyclyl group is substituted C 3 _ 10 carbocyclyl.
- “carbocyclyl” is a monocyclic, saturated carbocyclyl group having from 3 to 10 ring carbon atoms ("C 3 _io cycloalkyl”). In some embodiments, a cycloalkyl group has 3 to 8 ring carbon atoms ("C 3 _ 8 cycloalkyl”). In some embodiments, a cycloalkyl group has 3 to 6 ring carbon atoms ("C 3 _6 cycloalkyl”). In some embodiments, a cycloalkyl group has 5 to 6 ring carbon atoms ("Cs_6 cycloalkyl").
- a cycloalkyl group has 5 to 10 ring carbon atoms ("Cs-io cycloalkyl").
- C 5 _6 cycloalkyl groups include cyclopentyl (C 5 ) and cyclohexyl (C 5 ).
- C 3 _ 6 cycloalkyl groups include the aforementioned C 5 _6 cycloalkyl groups as well as cyclopropyl (C 3 ) and cyclobutyl (C 4 ).
- C 3 _ 8 cycloalkyl groups include the aforementioned C 3 _6 cycloalkyl groups as well as cycloheptyl (C 7 ) and cyclooctyl (C 8 ).
- each instance of a cycloalkyl group is independently unsubstituted (an "unsubstituted cycloalkyl") or substituted (a "substituted cycloalkyl”) with one or more substituents.
- the cycloalkyl group is unsubstituted C 3 _ 10 cycloalkyl.
- the cycloalkyl group is substituted C 3 _ 10 cycloalkyl.
- Heterocyclyl or “heterocyclic” refers to a radical of a 3- to 10-membered non- aromatic ring system having ring carbon atoms and 1 to 4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon ("3-10 membered heterocyclyl").
- the point of attachment can be a carbon or nitrogen atom, as valency permits.
- a heterocyclyl group can either be monocyclic ("monocyclic heterocyclyl”) or a fused, bridged, or spiro ring system, such as a bicyclic system ("bicyclic heterocyclyl”), and can be saturated or can be partially unsaturated.
- Heterocyclyl bicyclic ring systems can include one or more heteroatoms in one or both rings.
- Heterocyclyl also includes ring systems wherein the heterocyclic ring, as defined above, is fused with one or more carbocyclyl groups wherein the point of attachment is either on the carbocyclyl or heterocyclic ring, or ring systems wherein the heterocyclic ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclic ring, and in such instances, the number of ring members continue to designate the number of ring members in the
- heterocyclic ring system Unless otherwise specified, each instance of heterocyclyl is independently optionally substituted, i.e., unsubstituted (an “unsubstituted heterocyclyl”) or substituted (a "substituted heterocyclyl”) with one or more substituents.
- each instance of heterocyclyl is independently optionally substituted, i.e., unsubstituted (an “unsubstituted heterocyclyl”) or substituted (a "substituted heterocyclyl”) with one or more substituents.
- the heterocyclyl group is unsubstituted 3-10 membered heterocyclyl. In certain embodiments, the heterocyclyl group is substituted 3-10 membered heterocyclyl.
- a heterocyclyl group is a 5-10 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon ("5-10 membered heterocyclyl").
- a heterocyclyl group is a 5-8 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-8 membered heterocyclyl").
- a heterocyclyl group is a 5-6 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-6 membered heterocyclyl").
- the 5-6 membered heterocyclyl has 1-3 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5-6 membered heterocyclyl has 1-2 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5-6 membered heterocyclyl has one ring heteroatom selected from nitrogen, oxygen, and sulfur.
- Exemplary 3-membered heterocyclyl groups containing one heteroatom include, without limitation, azirdinyl, oxiranyl, and thiiranyl.
- Exemplary 4-membered heterocyclyl groups containing one heteroatom include, without limitation, azetidinyl, oxetanyl, and thietanyl.
- Exemplary 5-membered heterocyclyl groups containing one heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl,
- Exemplary 5- membered heterocyclyl groups containing two heteroatoms include, without limitation, dioxolanyl, oxasulfuranyl, disulfuranyl, and oxazolidin-2-one.
- Exemplary 5-membered heterocyclyl groups containing three heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl.
- Exemplary 6-membered heterocyclyl groups containing one heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl.
- Exemplary 6-membered heterocyclyl groups containing two heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, and dioxanyl.
- Exemplary 6- membered heterocyclyl groups containing three heteroatoms include, without limitation, triazinanyl.
- Exemplary 7-membered heterocyclyl groups containing one heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl.
- Exemplary 8-membered heterocyclyl groups containing one heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl.
- Exemplary 5-membered heterocyclyl groups fused to a C 6 aryl ring include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, benzoxazolinonyl, and the like.
- Exemplary 6-membered heterocyclyl groups fused to an aryl ring include, without limitation, tetrahydroquinolinyl,
- Aryl refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g. , having 6, 10, or 14 pi electrons shared in a cyclic array) having 6-14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system ("C6-14 aryl").
- an aryl group has six ring carbon atoms ("C 6 aryl”; e.g., phenyl).
- an aryl group has ten ring carbon atoms ("Cio aryl”; e.g., naphthyl such as 1-naphthyl and 2-naphthyl). In some embodiments, an aryl group has fourteen ring carbon atoms ("Cw aryl”; e.g. , anthracyl).
- Aryl also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system. Unless otherwise specified, each instance of an aryl group is independently optionally substituted, i.e. , unsubstituted (an "unsubstituted aryl”) or substituted (a
- substituted aryl with one or more substituents.
- the aryl group is unsubstituted C 6 -i4 aryl.
- the aryl group is substituted C 6 -i4 aryl.
- Alkyl is a subset of alkyl and aryl and refers to an optionally substituted alkyl group substituted by an optionally substituted aryl group. In certain embodiments, the aralkyl is optionally substituted benzyl. In certain embodiments, the aralkyl is benzyl. In certain embodiments, the aralkyl is optionally substituted phenethyl. In certain embodiments, the aralkyl is phenethyl.
- Heteroaryl refers to a radical of a 5-10 membered monocyclic or bicyclic 4n+2 aromatic ring system (e.g., having 6 or 10 pi electrons shared in a cyclic array) having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-10 membered heteroaryl").
- the point of attachment can be a carbon or nitrogen atom, as valency permits.
- Heteroaryl bicyclic ring systems can include one or more heteroatoms in one or both rings.
- Heteroaryl includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the point of attachment is on the heteroaryl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heteroaryl ring system.
- Heteroaryl also includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more aryl groups wherein the point of attachment is either on the aryl or heteroaryl ring, and in such instances, the number of ring members designates the number of ring members in the fused (aryl/heteroaryl) ring system.
- Bicyclic heteroaryl groups wherein one ring does not contain a heteroatom e.g. , indolyl, quinolinyl, carbazolyl, and the like
- the point of attachment can be on either ring, i.e., either the ring bearing a heteroatom (e.g., 2-indolyl) or the ring that does not contain a heteroatom (e.g., 5- indolyl).
- a heteroaryl group is a 5-10 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-10 membered heteroaryl").
- a heteroaryl group is a 5-8 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-8 membered heteroaryl").
- a heteroaryl group is a 5-6 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-6 membered heteroaryl").
- the 5-6 membered heteroaryl has 1-3 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5-6 membered heteroaryl has 1-2 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5-6 membered heteroaryl has 1 ring heteroatom selected from nitrogen, oxygen, and sulfur.
- each instance of a heteroaryl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted heteroaryl") or substituted (a "substituted heteroaryl") with one or more substituents.
- the heteroaryl group is unsubstituted 5-14 membered heteroaryl. In certain embodiments, the heteroaryl group is substituted 5-14 membered heteroaryl.
- Exemplary 5-membered heteroaryl groups containing one heteroatom include, without limitation, pyrrolyl, furanyl, and thiophenyl.
- Exemplary 5-membered heteroaryl groups containing two heteroatoms include, without limitation, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, and isothiazolyl.
- Exemplary 5-membered heteroaryl groups containing three heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl.
- Exemplary 5-membered heteroaryl groups containing four heteroatoms include, without limitation, tetrazolyl.
- Exemplary 6-membered heteroaryl groups containing one heteroatom include, without limitation, pyridinyl.
- Exemplary 6-membered heteroaryl groups containing two heteroatoms include, without limitation, pyridazinyl, pyrimidinyl, and pyrazinyl.
- Exemplary 6-membered heteroaryl groups containing three or four heteroatoms include, without limitation, triazinyl and tetrazinyl, respectively.
- Exemplary 7-membered heteroaryl groups containing one heteroatom include, without limitation, azepinyl, oxepinyl, and thiepinyl.
- Exemplary 5,6-bicyclic heteroaryl groups include, without limitation, indolyl, isoindolyl, indazolyl, benzotriazolyl, benzothiophenyl, isobenzothiophenyl, benzofuranyl, benzoisofuranyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzoxadiazolyl, benzthiazolyl, benzisothiazolyl, benzthiadiazolyl, indolizinyl, and purinyl.
- Exemplary 6,6- bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl.
- Heteroaralkyl is a subset of alkyl and heteroaryl and refers to an optionally substituted alkyl group substituted by an optionally substituted heteroaryl group.
- Unsaturated or “partially unsaturated” refers to a group that includes at least one double or triple bond.
- a “partially unsaturated” ring system is further intended to encompass rings having multiple sites of unsaturation, but is not intended to include aromatic groups
- saturated refers to a group that does not contain a double or triple bond, i.e., contains all single bonds.
- Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, which are divalent bridging groups, are further referred to using the suffix -ene, e.g., alkylene, alkenylene, alkynylene, carbocyclylene, heterocyclylene, arylene, and
- Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups are optionally substituted (e.g., "substituted” or “unsubstituted” alkyl, "substituted” or
- substituted carbocyclyl, "substituted” or “unsubstituted” heterocyclyl, "substituted” or “unsubstituted” aryl or “substituted” or “unsubstituted” heteroaryl group).
- substituted whether preceded by the term “optionally” or not, means that at least one hydrogen present on a group (e.g. , a carbon or nitrogen atom) is replaced with a permissible substituent, e.g. , a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction.
- a "substituted" group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position.
- substituted is contemplated to include substitution with all permissible substituents of organic compounds, any of the substituents described herein that results in the formation of a stable compound.
- the present invention contemplates any and all such combinations in order to arrive at a stable compound.
- heteroatoms such as nitrogen may have hydrogen substituents and/or any suitable substituent as described herein which satisfy the valencies of the heteroatoms and results in the formation of a stable moiety.
- the substituent is a carbon atom substituent. In certain embodiments, the substituent is a nitrogen atom substituent. In certain embodiments, the substituent is an oxygen atom substituent. In certain embodiments, the substituent is a sulfur atom substituent.
- each instance of R aa is, independently, selected from C ⁇ o alkyl, C ⁇ o perhaloalkyl, C 2 _ 10 alkenyl, C 2 _ 10 alkynyl, C 3 _ 10 carbocyclyl, 3-14 membered heterocyclyl, C 6 -i4 aryl, and 5-14 membered heteroaryl, or two R aa groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R dd groups;
- each instance of R cc is, independently, selected from hydrogen, Ci-io alkyl, Q_ io perhaloalkyl, C 2 _ 10 alkenyl, C 2 _ 10 alkynyl, C 3 _ 10 carbocyclyl, 3-14 membered heterocyclyl, C 6 -i4 aryl, and 5-14 membered heteroaryl, or two R cc groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R dd groups;
- each instance of R ee is, independently, selected from Q_6 alkyl, C ⁇
- each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R gg groups;
- each instance of R is, independently, selected from hydrogen, Q_6 alkyl, Q_6 perhaloalkyl, C 2 _ 6 alkenyl, C 2 _ 6 alkynyl, C 3 _ 10 carbocyclyl, 3-10 membered heterocyclyl, C 6 - ff
- io aryl and 5-10 membered heteroaryl, or two R groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R gg groups; and
- Halo or "halogen” refers to fluorine (fluoro, -F), chlorine (chloro, -CI), bromine (bromo, -Br), or iodine (iodo, -I).
- Nitrogen atoms can be substituted or unsubstituted as valency permits, and include primary, secondary, tertiary, and quatemarynitrogen atoms.
- the substituent present on a nitrogen atom is a nitrogen protecting group (also referred to as an amino protecting group).
- Nitrogen protecting groups such as carbamate groups include, but are not limited to, methyl carbamate, ethyl carbamante, 9-fluorenylmethyl carbamate (Fmoc), 9-(2-sulfo)fluorenylmethyl carbamate, 9-(2,7-dibromo)fluoroenylmethyl carbamate, 2,7-di-i-butyl-[9-( 10,10-dioxo-l 0, 10,10,10-tetrahydrothioxanthyl)]methyl carbamate (DBD-Tmoc), 4-methoxyphenacyl carbamate (Phenoc), 2,2,2-trichloroethyl carbamate (Troc), 2-trimethylsilylethyl carbamate (Teoc), 2-phenylethyl carbamate (hZ), 1- (l-adamantyl)-l-methylethyl
- Nitrogen protecting groups such as sulfonamide groups include, but are not limited to, p-toluenesulfonamide (Ts), benzenesulfonamide, 2,3,6,-trimethyl-4- methoxybenzenesulfonamide (Mtr), 2,4,6-trimethoxybenzenesulfonamide (Mtb), 2,6- dimethyl-4-methoxybenzenesulfonamide (Pme), 2,3,5, 6-tetramethyl-4- methoxybenzenesulfonamide (Mte), 4-methoxybenzenesulfonamide (Mbs), 2,4,6- trimethylbenzenesulfonamide (Mts), 2,6-dimethoxy-4-methylbenzenesulfonamide (iMds), 2,2,5,7, 8-pentamethylchroman-6-sulfonamide (Pmc), methanes
- Ts p-toluenesulfonamide
- Mtr 2,3,
- nitrogen protecting groups include, but are not limited to, phenothiazinyl- (lO)-acyl derivative, N'-p-toluenesulfonylaminoacyl derivative, N'-phenylaminothioacyl derivative, N-benzoylphenylalanyl derivative, N-acetylmethionine derivative, 4,5-diphenyl- 3-oxazolin-2-one, N-phthalimide, N-dithiasuccinimide (Dts), N-2,3-diphenylmaleimide, N-2,5-dimethylpyrrole, N-l,l,4,4-tetramethyldisilylazacyclopentane adduct (STABASE), 5-substituted l,3-dimethyl-l,3,5-triazacyclohexan-2-one, 5-substituted 1,3-dibenzyl- l,3,5-triazacyclohexan-2-one
- benzenesulfenamide o-nitrobenzenesulfenamide (Nps), 2,4-dinitrobenzenesulfenamide, pentachlorobenzenesulfenamide, 2-nitro-4-methoxybenzenesulfenamide,
- oxygen protecting groups include, but are not limited to, methyl, i-butyloxycarbonyl (BOC or Boc), methoxylmethyl (MOM), methylthiomethyl (MTM), i-butylthiomethyl, (phenyldimethylsilyl)methoxymethyl (SMOM), benzyloxymethyl (BOM), p-methoxybenzyloxymethyl (PMBM), (4- methoxyphenoxy)methyl (p-AOM), guaiacolmethyl (GUM), i-butoxymethyl, 4- pentenyloxymethyl (POM), siloxymethyl, 2-methoxyethoxymethyl (MEM), 2,2,2- trichloroethoxymethyl, bis(2-chloroethoxy)methyl, 2-(trimethylsilyl)ethoxymethyl
- DPMS diphenylmethylsilyl
- TMPS i-butylmethoxyphenylsilyl
- the sulfur atom substituent present on a sulfur atom is a sulfur protecting group (also referred to as a thiol protecting group).
- Sulfur protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3 r edition, John Wiley & Sons, 1999, incorporated herein by reference.
- salt refers to ionic compounds that result from the neutralization reaction of an acid and a base.
- a salt is composed of one or more cations (positively charged ions) and one or more anions (negative ions) so that the salt is electrically neutral (without a net charge).
- Salts of the compounds of this invention include those derived from inorganic and organic acids and bases.
- acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, and perchloric acid, or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid, or malonic acid or by using other methods known in the art such as ion exchange.
- Other salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate,
- ethanesulfonate formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2-hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2- naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, propionate, stearate, succinate, sulfate, tartrate, thiocyanate, p-toluenesulfonate, undecanoate, valerate salts, and the like.
- Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N + (Ci ⁇ alkyl) 4 salts.
- Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like.
- Further salts include ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, lower alkyl sulfonate, and aryl sulfonate.
- tautomers refers to two or more interconvertable compounds resulting from at least one formal migration of a hydrogen atom and at least one change in valency (e.g., a single bond to a double bond, a triple bond to a single bond, or vice versa).
- the exact ratio of the tautomers depends on several factors, including temperature, solvent, and pH. Tautomerizations (i.e., the reaction providing a tautomeric pair) may catalyzed by acid or base.
- Exemplary tautomerizations include keto-to-enol, amide-to-imide, lactam- to-lactim, enamine-to-imine, and enamine-to-(a different enamine) tautomerizations.
- isomers compounds that have the same molecular formula but differ in the nature or sequence of bonding of their atoms or the arrangement of their atoms in space are termed "isomers”. Isomers that differ in the arrangement of their atoms in space are termed "stereoisomers”.
- enantiomers and those that are non-superimposable mirror images of each other are termed "enantiomers".
- An enantiomer can be characterized by the absolute configuration of its asymmetric center and is described by the R- and S-sequencing rules of Cahn and Prelog, or by the manner in which the molecule rotates the plane of polarized light and designated as dextrorotatory or levorotatory (i.e., as (+) or (-)-isomers respectively).
- a chiral compound can exist as either individual enantiomer or as a mixture thereof. A mixture containing equal proportions of the enantiomers is called a "racemic mixture".
- solvate refers to forms of the compound that are associated with a solvent, usually by a solvolysis reaction. This physical association may include hydrogen bonding.
- solvents include water, methanol, ethanol, acetic acid, DMSO, THF, diethyl ether, and the like.
- the compounds described herein may be prepared, e.g., in crystalline form, and may be solvated.
- Suitable solvates include pharmaceutically acceptable solvates and further include both stoichiometric solvates and non- stoichiometric solvates. In certain instances, the solvate will be capable of isolation, for example, when one or more solvent molecules are incorporated in the crystal lattice of a crystalline solid.
- “Solvate” encompasses both solution-phase and isolatable solvates. Representative solvates include hydrates, ethanolates, and methanolates.
- hydrate refers to a compound that is associated with water.
- the number of the water molecules contained in a hydrate of a compound is in a definite ratio to the number of the compound molecules in the hydrate. Therefore, a hydrate of a compound may be represented, for example, by the general formula R x H 2 0, wherein R is the compound, and x is a number greater than 0.
- a given compound may form more than one type of hydrate, including, e.g., monohydrates (x is 1), lower hydrates (x is a number greater than 0 and smaller than 1, e.g., hemihydrates (R-0.5 H 2 0)), and polyhydrates (x is a number greater than 1, e.g., dihydrates (R-2 H 2 0) and hexahydrates (R-6 H 2 0)).
- polymorph refers to a crystalline form of a compound (or a salt, hydrate, or solvate thereof) in a particular crystal packing arrangement. All polymorphs have the same elemental composition. Different crystalline forms usually have different X-ray diffraction patterns, infrared spectra, melting points, density, hardness, crystal shape, optical and electrical properties, stability, and solubility. Recrystallization solvent, rate of
- crystallization, storage temperature, and other factors may cause one crystal form to dominate.
- Various polymorphs of a compound can be prepared by crystallization under different conditions.
- complex refers to an association of at least one atom or ion (which is referred to as a "central atom,” “central ion,” or “acceptor,” and is usually a metallic cation) and a surrounding array of bound ligands or donors).
- Ligands are generally bound to a central atom or central ion by a coordinate covalent bond (e.g. , ligands may donate electrons from a lone electron pair into an empty orbital of the central atom or central ion) and are referred to as being “coordinated" to the central atom or central ion.
- a complex may include one or more donors, which can be the same or different.
- a complex may also include one or more acceptors, which can be the same or different.
- ligand refers to an ion or molecule that binds to a central atom or ion (e.g., a central metal atom or ion) to form a coordination complex.
- Ligands are usually electron donors, and the central atom or ion is electron acceptors.
- the bonding between the central atom or ion and the ligand typically involves formal donation of one or more of the ligand' s electron pairs. The nature of such bonding can range from covalent to ionic, and the bond order can range from one to three.
- One central atom or ion may bind to one or more ligands of the same or different type.
- a ligand may be capable of binding a central atom or ion through multiple sites, usually because the ligand includes lone pairs on more than one atom of the ligand.
- Ligands in a complex may affect the reactivity (e.g. , ligand substitution rates and redox) of the central atom or ion.
- Exemplary ligands include charge-neutral ligands ("ligand molecules," e.g. , CH 3 CN, amides (e.g.
- N,N-dimethylformamide (DMF), N,N- dimethylacetamide (DMA), or N-methyl-2-pyrrolidone (NMP)
- dimethyl sulfoxide (DMSO)
- amines e.g., ammonia; ethylenediamine (en); pyridine (py); 2,2'-bipyridine (bipy); and 1,10- phenanthroline (phen)
- phosphines e.g., PPh 3
- ethers e.g. , tetrahydrofuran (THF), 2- methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl t-butyl ether
- ketones e.g., acetone and butanone
- chlorohydrocarbons e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, and 1,2-dichloroethane (DCE)
- esters e.g., propylene carbonate and ethyl acetate
- CO N 2
- water water
- alkenes anionic ligands
- transition metal refers to elements that are in the d-block and f-block of the Periodic Chart of the Elements, which may exhibit a variety of oxidation states, and which may form numerous complex ions.
- d-block refers to those elements that have electrons filling the 3d, 4d, 5d, and 6d orbitals
- f-block refers to those elements (including lanthanides and the actinides) that have electrons filling the 4f and 5f orbitals.
- Exemplary transition metals include palladium, nickel, cobalt, copper, platinum, silver, manganese, zinc, iridium, rhodium, iron, and ruthenium.
- Transition metal also includes alloys, metal/metal composites, metal ceramic composites, and metal polymer composites, as well as other metal composites.
- catalysis refers to the increase in rate of a chemical reaction due to the participation of a substance called a "catalyst.”
- the amount and nature of a catalyst remains essentially unchanged during a reaction.
- a catalyst is regenerated, or the nature of a catalyst is essentially restored after a reaction.
- a catalyst may participate in multiple chemical transformations. The effect of a catalyst may vary due to the presence of other substances known as inhibitors or poisons (which reduce the catalytic activity) or promoters (which increase the activity).
- Catalyzed reactions have lower activation energy (rate-limiting free energy of activation) than the corresponding uncatalyzed reaction, resulting in a higher reaction rate at the same temperature.
- Catalysts may affect the reaction environment favorably, bind to the reagents to polarize bonds, form specific intermediates that are not typically produced by a uncatalyzed reaction, or cause dissociation of reagents to reactive forms.
- arene refers to a compound including an aryl moiety, wherein at least one carbon atom of the aryl ring system is directly attached to a hydrogen atom.
- heteroaryl refers to a compound including a heteroaryl moiety, wherein at least one carbon atom of the heteroaryl ring system is directly attached to a hydrogen atom.
- a "counterion” or “anionic counterion” is a negatively charged group associated with a positively charged group in order to maintain electronic neutrality.
- An anionic counterion may be monovalent (i.e., including one formal negative charge).
- An anionic counterion may also be multivalent (i.e., including more than one formal negative charge), such as divalent or trivalent.
- Exemplary counterions include halide ions (e.g., F , CT, Br , ⁇ ), NO 3 , CIO 4 , OH , H 2 PO 4 , HSO 4 , sulfonate ions (e.g., methansulfonate,
- non-coordinating anionic counterion refers to an anion that interacts weakly with cations.
- exemplary non-coordinating anions include, but are not limited to, C10 4 , ⁇ , BF 4 , PF 4 , PF 6 , and SbF 6 ⁇ .
- Other examples of non-coordinating anions include, but are not limited to, B[3,5-(CF ) 2 C 6 H ] 4 ] ⁇ , BPh 4 , Al(OC(CF ) ) 4 ⁇ , or a carborane anion (e.g., CB U H 12 or (HCBnMe 5 Br 6 ) ).
- Figure 1 depicts an X-ray structure of complex 1 (elipsoids drawn at 50% probability).
- Figure 2 shows a proposed mechanism of a catalytic imidation reaction of the invention.
- Figure 3 shows calculated valence orbitals of complex 1.
- Figure 4 illustrates the rate law for the reduction of N-fluorobenzenesulfonimide (NFBS or NFSI) catalyzed by complex 1.
- NFBS N-fluorobenzenesulfonimide
- Figure 5 illustrates the rate law for an imidation reaction of the invention catalyzed by 1 and in the presence of Ag(bipy) 2 C10 4 .
- Figure 6 illustrates the rate law showing the order in Ag(bipy) 2 C10 4 .
- Figure 7 shows the results of experiments for the determination of the order in an arene substrate.
- Figure 8 shows the results of experiments for the determination of the order in NFBS.
- Figure 9 shows the results of experiments for the determination of the order in complex 1.
- Figure 10 shows the results of experiments for the determination of the resting state.
- Figure 11 shows the 1H NMR spectra of Solution A (left panels) and Solution B (right panels) after 10 minutes (top panels) and 4 hours (bottom panels) of oxidation of Ru(bipy) (PF 6 )2 mediated by complex 1.
- Figure 12 shows 1 H NMR peak broadness at 23 °C, -20 °C, and -40 °C during oxidation of Ru(bipy) 3 (PF 6 )2 mediated by complex 1.
- Figure 13 shows EPR (electron paramagnetic resonance) spectra of Solution B and pure Ru(bipy) 3 (PF 6 ) 3 .
- Figure 14 shows the rates of NFBS consumption by Ru(bipy) 3 (PF 6 )2 or by
- Figure 15 shows the EPR spectra of Solutions E and F.
- Figure 16 shows the optimized structure of complex 1 with the B3PW91 functional with the BS I basis set.
- the present invention provides novel transition metal complexes (e.g., complexes of any one of Formulae (CI) to (C25)) that include an amine-N-oxide motif. These transition metal complexes have been found useful in the imidation of arenes and heteroarenes.
- the invention also provides methods of using the transition metal complexes as catalysts in the preparation of N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof) and aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof) through imidation of arenes or heteroarenes (e.g., compounds of Formula (A), and salts thereof).
- the methods of the invention involves an imidating agent (e.g., a compound of Formula (B), such as N-fluorobenzenesulfonimide (NFBS or NFSI)) in the presence of a transition metal complex of the invention and a single-electron reductant (e.g., an Ag(I) or Ru(II) salt).
- an imidating agent e.g., a compound of Formula (B), such as N-fluorobenzenesulfonimide (NFBS or NFSI)
- NFBS or NFSI N-fluorobenzenesulfonimide
- a single-electron reductant e.g., an Ag(I) or Ru(II) salt
- inventive methods may involve a catalytic, intermolecular C-H imidation that proceeds via a mechanism different from the conventional metallation-functionalization sequence and may enable the transfer of the imido group without the formation of conventionally targeted organometallic
- oxidation of the catalyst e.g., complexes of any one of Formulae (CI) to (C25)
- the methods of the invention may be practiced at or below room temperature.
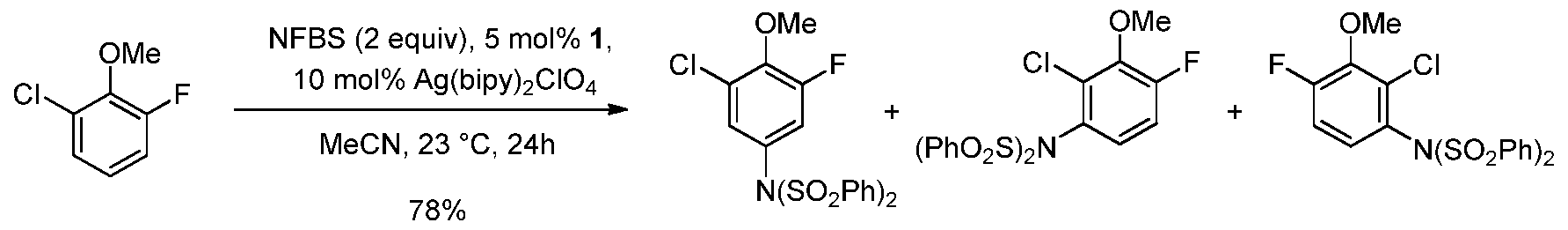
- the imidation methods of the invention do not require coordinating directing groups on the starting material and give synthetically useful yields with the arene or heteroarene starting material as the limiting reagent (e.g., one equivalent of arene or heteroarene and more than one equivalents of imidating agent is typically used).
- the present invention provides methods of preparing N-aryl sulfonimides or N-heteroaryl sulfonimides through a catalytic imidation of an arene or heteroarene (e.g., a compound of Formula (A), or a salt thereof) using an imidating agent in the presence of a catalyst and a single-electron reductant.
- the imidating agent employed in the methods of the invention may be an N-fluorosulfonimide, such as a compound of Formula (B) (e.g., N-fluorobenzenesulfonimide (NFBS or NFSI)), or a salt thereof.
- the catalyst useful in the inventive methods may be a transition metal complex, such as a complex of any one of Formulae (CI) to (C25).
- the invention provides methods of preparing compounds of Formula (I):
- inventive methods typically include contacting a compound of Formula (A), or a salt thereof, with a compound of Formula (B), or a salt thereof, in the presence of a transition metal complex of any one of Formulae (CI) to (C25) and a single-electron reductant to provide the compound of Formula (I), or the salt thereof:
- each instance of R A1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R A1 are joined to form a substituted or unsubstituted heterocyclic ring;
- k 0, 1, 2, 3, 4, or 5;
- each instance of m is independently 0, 1, 2, 3, 4, or 5;
- each instance of M is a transition metal ion
- each instance of Ring B is independently substituted or unsubstituted heteroaryl; each instance of is independently a single or double bond;
- each instance of r is independently 0, 1, 2, 3, or 4;
- each instance of R D is independently substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of R D are joined to form a substituted or unsubstituted heterocyclic ring;
- each instance of R is independently absent, hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or p
- n 0 or 1 ;
- p 1 or 2;
- each instance of X is an anionic counterion
- q is 1, 2, or 3.
- the arene or heteroarene involved in the inventive methods may be a compound of Formula (A), or a salt thereof.
- Compounds of Formula (A) include substituted or unsubstituted aryl or substituted or unsubstituted heteroaryl as Ring A.
- Ring A is substituted aryl. In certain embodiments, Ring A is unsubstituted aryl. In certain embodiments, Ring A is 6- to 14-membered aryl. In certain embodiments, Ring A is 6- to 10-membered aryl. In certain embodiments, Ring A is monocyclic aryl. In certain embodiments, Ring A is unsubstituted phenyl. In certain embo Ring A is
- Ring A is of the formula: . In certain . In certain embodiments, Ring A is of
- Ring A is of the formula: .
- Ring A is of the formula: . In certain embodiments, Ring A is
- Ring A is of the formula:
- n certa n em o ments ng s o t e ormu a: . n certa n em o ments,
- Rin A is of the formula: .
- Ring A is of the formula:
- Ring A is bicyclic aryl. In certain embodiments, Ring A is substituted naphthyl. In certain embodiments, Ring A is unsubstituted naphthyl. In certain embodiments, Ring A is substituted or unsubstituted aryl fused with one or more substituted or
- Ring A of Formula (I) may also be substituted or unsubstituted heteroaryl.
- Ring A is substituted heteroaryl.
- Ring A is unsubstituted heteroaryl.
- Ring A is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- Ring A is 6-membered, monocyclic heteroaryl.
- Ring A is of the formula: .
- Ring A is of
- Ring A is of the formula:
- Ring A is of the
- Ring A is of the formula:
- Ring A is of the formula:
- Ring A is 5-membered, monocyclic heteroaryl. In certain embodiments, Ring A is 5-membered, monocyclic heteroaryl, wherein one atom in the heteroaryl ring system is nitrogen, ox en, or sulfur. In certain embodiments, Ring A is of
- Ring A is of the
- Ring A is 5-membered, monocyclic heteroaryl, wherein two atoms in the heteroaryl ring system are independently nitrogen, oxygen, or sulfur. In certain embodiments, Ring A is of the formula: , or . In certain embodiments, Ring A is of the
- Ring A is of th e f . In certain embodiments, Ring A is of
- Ring A is of the formula In certain embodiments, Ring A
- Ring A is 5-membered, monocyclic heteroaryl, wherein three atoms in the heteroaryl ring system are independently nitrogen, oxygen, or sulfur.
- three atoms in the heteroaryl ring system are independently nitrogen, oxygen, or sulfur.
- Ring A is of the or ⁇ * . n certa n em o ments, Ring A is of the formula:
- Ring A is 5-membered, monocyclic heteroaryl, wherein four atoms in the heteroaryl ring system are nitrogen, oxygen, or sulfur.
- Ring A is of the formula:
- Ring A is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- the bicyclic heteroaryl described herein may be substituted or unsubstituted.
- Ring A is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- Ring A is 5- or 6- membered, monocyclic heteroaryl fused with phenyl, wherein one, two, or three atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- Ring A is 5- or 6-membered, monocyclic heteroaryl fused with another 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- Ring A is substituted or unsubstituted indole.
- Ring A is substituted or unsubstituted isoindole.
- Ring A is substituted or unsubstituted indazole. In certain embodiments, Ring A is substituted or unsubstituted benzothiophene. In certain embodiments, Ring A is substituted or unsubstituted isobenzothiophene. In certain embodiments, Ring A is substituted or unsubstituted benzofuran. In certain embodiments, Ring A is substituted or unsubstituted benzoisofuran. In certain embodiments, Ring A is substituted or unsubstituted benzimidazole. In certain embodiments, Ring A is substituted or unsubstituted benzoxazole. In certain embodiments, Ring A is substituted or unsubstituted benzisoxazole.
- Ring A is substituted or unsubstituted benzothiazole. In certain embodiments, Ring A is substituted or unsubstituted benzisothiazole. In certain embodiments, Ring A is substituted or unsubstituted benzotriazole. In certain embodiments, Ring A is substituted or unsubstituted benzoxadiazole. In certain embodiments, Ring A is substituted or unsubstituted quinoline. In certain embodiments, Ring A is substituted or unsubstituted isoquinoline. In certain embodiments, Ring A is substituted or unsubstituted cinnoline. In certain embodiments, Ring A is substituted or unsubstituted quinoxaline. In certain embodiments, Ring A is substituted or unsubstituted phthalazine. In certain embodiments, Ring A is substituted or unsubstituted quinazoline.
- Compounds of Formula (I) or (A) may include one or more substituent R A .
- at least one instance of R A is H.
- at least one instance of R A is halogen.
- at least one instance of R A is F.
- at least one instance of R A is CI.
- at least one instance of R A is Br.
- at least one instance of R A is I (iodine).
- at least one instance of R A is substituted acyl.
- at least one instance of R A is unsubstituted acyl.
- at least one instance of R A is substituted alkyl.
- At least one instance of R A is unsubstituted alkyl. In certain embodiments, at least one instance of R A is C 1-12 alkyl. In certain embodiments, at least one instance of R A is substituted Ci_6 alkyl. In certain embodiments, at least one instance of R A is unsubstituted C 1-6 alkyl. In certain embodiments, at least one instance of R A is substituted methyl. In certain embodiments, at least one instance of R A is -CH 2 F. In certain embodiments, at least one instance of R A is -CHF 2 . In certain embodiments, at least one instance of R A is -CF 3 . In certain embodiments, at least one instance of R A is Bn.
- At least one instance of R A is unsubstituted methyl. In certain embodiments, at least one instance of R A is ethyl. In certain embodiments, at least one instance of R A is propyl. In certain embodiments, at least one instance of R A is butyl. In certain embodiments, at least one instance of R A is pentyl. In certain embodiments, at least one instance of R A is hexyl. In certain embodiments, at least one instance of R A is substituted alkenyl. In certain embodiments, at least one instance of R A is unsubstituted alkenyl. In certain embodiments, at least one instance of R A is substituted or unsubstituted C 1-6 alkenyl.
- At least one instance of R A is vinyl. In certain embodiments, at least one instance of R A is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R A is ethynyl. In certain
- At least one instance of R A is substituted carbocyclyl. In certain embodiments, at least one instance of R A is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R A is saturated carbocyclyl. In certain embodiments, at least one instance of R A is unsaturated carbocyclyl. In certain embodiments, at least one instance of R A is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R A is monocyclic carbocyclyl. In certain embodiments, at least one instance of R A is 3- to 7-membered, monocyclic carbocyclyl.
- At least one instance of R A is cylcopropyl. In certain embodiments, at least one instance of R A is cyclobutyl. In certain embodiments, at least one instance of R A is cyclopentyl. In certain embodiments, at least one instance of R A is cyclohexyl. In certain embodiments, at least one instance of R A is cycloheptyl. In certain embodiments, at least one instance of R A is bicyclic carbocyclyl. In certain embodiments, at least one instance of R A is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R A is substituted heterocyclyl. In certain embodiments, at least one instance of R A is
- At least one instance of R A is saturated heterocyclyl. In certain embodiments, at least one instance of R A is unsaturated heterocyclyl. In certain embodiments, at least one instance of R A is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R A is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R A is monocyclic heterocyclyl. In certain embodiments, at least one instance of R A is 3- to 7-membered, monocyclic heterocyclyl.
- At least one instance of R A is bicyclic heterocyclyl. In certain embodiments, at least one instance of R A is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R A is substituted aryl. In certain embodiments, at least one instance of R A is unsubstituted aryl. In certain embodiments, at least one instance of R A is 6- to 14-membered aryl. In certain embodiments, at least one instance of R A is 6- to 10-membered aryl. In certain embodiments, at least one instance of R A is substituted phenyl. In certain embodiments, at least one instance of R A is unsubstituted phenyl.
- At least one instance of R A is substituted naphthyl. In certain embodiments, at least one instance of R A is unsubstituted naphthyl. In certain embodiments, at least one instance of R A is substituted heteroaryl. In certain embodiments, at least one instance of R A is unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R A is monocyclic heteroaryl. In certain embodiments, at least one instance of R A is 5-membered, monocyclic heteroaryl.
- At least one instance of R A is 6-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R A is substituted pyridyl. In certain embodiments, at least one instance of R A is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R A is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R A is 9-membered, bicyclic heteroaryl.
- At least one instance of R A is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R A is -OR A1 . In certain embodiments, at least one instance of R A is -OR A1 , wherein R A1 is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R A is -OMe. In certain embodiments, at least one instance of R A is -OEt. In certain embodiments, at least one instance of R A is -OPr. In certain embodiments, at least one instance of R A is -OBu. In certain embodiments, at least one instance of R A is -O(pentyl).
- At least one instance of R A is - O(hexyl). In certain embodiments, at least one instance of R A is -OBn. certain embodiments, at least one instance of R A is -OPh. In certain embodiments, at least one instance of R A is - OH. In certain embodiments, at least one instance of R A is -SR A1 . In certain embodiments, at least one instance of R A is -SMe. In certain embodiments, at least one instance of R A is -SH. In certain embodiments, at least one instance of R A is -N(R A1 ) 2 . In certain embodiments, at least one instance of R A is -NMe 2 .
- at least one instance of R A is -N0 2 .
- two R A groups may be joined to form a substituted or unsubstituted carbocyclic ring.
- two instances of R A are joined to form a saturated or unsaturated carbocyclic ring.
- two instances of R A are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system.
- two instances of R A are joined to form a 3- to 7-membered, monocyclic carbocyclic ring.
- two instances of R A are joined to form a 3-membered carbocyclic ring.
- two instances of R A are joined to form a 4-membered carbocyclic ring. In certain embodiments, two instances of R A are joined to form a 5-membered carbocyclic ring. In certain embodiments, two instances of R A are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of R A are joined to form a 7-membered carbocyclic ring. In certain embodiments,
- two instances of R A are joined to form a 5- to 13-membered, bicyclic carbocyclic ring. [0089] In certain embodiments, two instances of R are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R A are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R A are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R A are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R A are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R A are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- two instances of R A are joined to form a substituted or unsubstituted aryl ring. In certain embodiments, two instances of R A are joined to form a 6- to 14-membered aryl ring. In certain embodiments, two instances of R A are joined to form a 6- to 10-membered aryl ring. In certain embodiments, two instances of R A are joined to form a monocyclic aryl ring. In certain embodiments, two instances of R A are joined to form an unsubstituted phenyl ring. In certain embodiments, two instances of R A are joined to form a substituted phenyl ring. In certain embodiments, two instances of R A are joined to form a bicyclic aryl ring. In certain embodiments, two instances of R A are joined to form a naphthyl ring.
- two instances of R A are joined to form a substituted or unsubstituted heteroaryl ring.
- two instances of R A are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R A are joined to form a substituted or unsubstituted pyridyl ring.
- two instances of R A are joined to form a 9- or 10-membered, bicyclic heteroaryl ring, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- At least one instance of R A is halogen, substituted or unsubstituted alkyl, or -OR A1 . In certain embodiments, at least one instance of R A is halogen, substituted or unsubstituted C 1-6 alkyl, or -OR A1 , wherein R A1 is substituted or unsubstituted Ci-6 alkyl.
- At least one instance of R A1 is H. In certain embodiments, at least one instance of R A1 is substituted acyl. In certain embodiments, at least one instance of R A1 is unsubstituted acyl. In certain embodiments, at least one instance of R A1 is acetyl. In certain embodiments, at least one instance of R A1 is substituted alkyl. In certain embodiments,
- At least one instance of R A1 is unsubstituted alkyl. In certain embodiments, at least one instance of R A1 is C 1-12 alkyl. In certain embodiments, at least one instance of R A1 is Ci-6 alkyl. In certain embodiments, at least one instance of R A1 is methyl. In certain embodiments, at least one instance of R A1 is ethyl. In certain embodiments, at least one instance of R A1 is propyl. In certain embodiments, at least one instance of R A1 is butyl. In certain embodiments, at least one instance of R A1 is pentyl. In certain embodiments, at least one instance of R A1 is hexyl.
- At least one instance of R A1 is substituted alkenyl. In certain embodiments, at least one instance of R A1 is unsubstituted alkenyl. In certain embodiments, at least one instance of R A1 is vinyl. In certain embodiments,
- At least one instance of R A1 is substituted alkynyl. In certain embodiments, at least one instance of R A1 is unsubstituted alkynyl. In certain embodiments, at least one instance of R A1 is ethynyl. In certain embodiments, at least one instance of R A1 is substituted carbocyclyl. In certain embodiments, at least one instance of R A1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R A1 is saturated carbocyclyl. In certain embodiments, at least one instance of R A1 is unsaturated carbocyclyl.
- At least one instance of R A1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R A1 is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R A1 is cylcopropyl. In certain embodiments, at least one instance of R A1 is cyclobutyl. In certain embodiments, at least one instance of R A1 is cyclopentyl. In certain embodiments, at least one instance of R A1 is cyclohexyl. In certain embodiments, at least one instance of R A1 is cycloheptyl.
- At least one instance of R A1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R A1 is substituted heterocyclyl. In certain embodiments, at least one instance of R A1 is unsubstituted
- heterocyclyl In certain embodiments, at least one instance of R A1 is saturated heterocyclyl. In certain embodiments, at least one instance of R A1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R A1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R A1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R A1 is 5- to 13-membered, bicyclic
- R A1 is substituted or
- At least one instance of R A1 is 6- to 14-membered aryl. In certain embodiments, at least one instance of R A1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R A1 is monocyclic aryl. In certain embodiments, at least one instance of R A1 is unsubstituted phenyl. In certain embodiments, at least one instance of R A1 is substituted phenyl. In certain embodiments, at least one instance of R A1 is bicyclic aryl. In certain embodiments, at least one instance of R A1 is naphthyl.
- At least one instance of R A1 is substituted or unsubstituted heteroaryl.
- at least one instance of R A1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R A1 is pyridyl.
- at least one instance of R A1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- At least one instance of R A1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R A1 is a nitrogen protecting group when attached to a nitrogen atom.
- at least one instance of R A1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom.
- R A1 is an oxygen protecting group when attached to an oxygen atom.
- R A1 is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ⁇ -Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom.
- R A1 is a sulfur protecting group when attached to a sulfur atom.
- R A1 is acetamidomethyl, ⁇ -Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
- two instances of R A1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R A1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R A1 are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R A1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R A1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- k is 0. In certain embodiments, k is 1. In certain embodiments, k is 2. In certain embodiments, k is 3. In certain embodiments, k is 4. In certain embodiments, k is 5.
- k is 1; and R A is halogen, substituted or unsubstituted alkyl, or -OR A1 . In certain embodiments, k is 1; and R A is halogen, substituted or
- R A1 is substituted or unsubstituted C 1-6 alkyl, or -OR A1 , wherein R A1 is substituted or unsubstituted C 1-6 alkyl.
- k is 2; and each instance of R A is independently halogen, substituted or unsubstituted alkyl, or -OR A1 .
- k is 2; and each instance of R A is independently halogen, substituted or unsubstituted C 1-6 alkyl, or -OR A1 , wherein each instance of R A1 is independently substituted or unsubstituted C 1-6 alkyl.
- Exemplary compounds of Formula (A) include, but are not limited to, compounds of any one of the formulae:
- the methods of the invention also involves an imidating agent, such as an N- fluorosulfonimide, e.g., a compound of Formula (B), or a salt thereof.
- an imidating agent such as an N- fluorosulfonimide, e.g., a compound of Formula (B), or a salt thereof.
- Formula (B) may include one or more substituents R .
- at least one instance of R B is H.
- at least one instance of R B is halogen.
- at least one instance of R is F.
- at least one instance of R B is CI.
- at least one instance of R B is Br.
- at least one instance of R B is I (iodine).
- at least one instance of R B is substituted acyl.
- at least one instance of R is unsubstituted acyl.
- at least one instance of R is substituted alkyl.
- At least one instance of R is unsubstituted alkyl.
- at least one instance of R B is C 1-12 alkyl.
- at least one instance of R B is substituted Ci-6 alkyl.
- at least one instance of R is unsubstituted C 1-6 alkyl.
- at least one instance of R is substituted methyl.
- At least one instance of R is -CH 2 F. In certain embodiments, at least one instance of R B is -CHF 2 . In certain embodiments, at least one instance of R B is -CF 3 . In certain embodiments, at least one instance of R is Bn. In certain embodiments, at least one instance of R B is unsubstituted methyl. In certain embodiments, at least one instance of R B is ethyl. In certain embodiments, at least one instance of R is propyl. In certain embodiments, at least one instance of R B is butyl. In certain embodiments, at least one instance of R B is pentyl. In certain embodiments, at least one instance of R is hexyl.
- At least one instance of R is substituted alkenyl. In certain embodiments, at least one instance of R B is unsubstituted alkenyl. In certain embodiments, at least one instance of R B is substituted or unsubstituted C 1-6 alkenyl. In certain embodiments, at least one instance of R is vinyl. In certain embodiments, at least one instance of R is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R B is ethynyl. In certain embodiments, at least one instance of R B is substituted carbocyclyl.
- At least one instance of R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R is saturated carbocyclyl. In certain embodiments, at least one instance of R is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of
- R B is monocyclic carbocyclyl. In certain embodiments, at least one instance of R B is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl. In certain embodiments, at least one instance of R is cyclopentyl. In certain embodiments, at least one instance of R B is cyclohexyl. In certain embodiments, at least one instance of R B is cycloheptyl. In certain embodiments, at least one instance of R is bicyclic carbocyclyl.
- At least one instance of R is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is unsubstituted heterocyclyl. In certain
- At least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl. In certain embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heterocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic heterocyclyl.
- At least one instance of R B is bicyclic heterocyclyl. In certain embodiments, at least one instance of R B is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R is substituted aryl. In certain embodiments, at least one instance of R is unsubstituted aryl. In certain embodiments, at least one instance of R is 6- to 14-membered aryl. In certain embodiments, at least one instance of R is 6- to 10-membered aryl. In certain embodiments, at least one instance of R is substituted phenyl. In certain embodiments, at least one instance of R B is unsubstituted phenyl.
- At least one instance of R B is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R is substituted heteroaryl. In certain embodiments, at least one instance of R is unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl. In certain embodiments, at least one instance of R is 5-membered, monocyclic heteroaryl.
- At least one instance of R is 6-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is substituted pyridyl. In certain embodiments, at least one instance of R is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R is 9-membered, bicyclic heteroaryl.
- At least one instance of R is 10-membered, bicyclic heteroaryl.
- at least one instance of R B is -OR B 1.
- at least one instance of R B is -OR B 1 , wherein R B 1 is substituted or unsubstituted alkyl.
- at least one instance of R B is -OMe.
- at least one instance of R B is -OEt.
- at least one instance of R is -OPr.
- at least one instance of R B is -OBu.
- at least one instance of R B is -O(pentyl).
- At least one instance of R is -O(hexyl). In certain embodiments, at least one instance of R is -OBn. In certain embodiments, at least one instance of R is - OPh. In certain embodiments, at least one instance of R is -OH. In certain embodiments, at least one instance of R B is -SR B 1. In certain embodiments, at least one instance of R B is - SMe. In certain embodiments, at least one instance of R is -SH. In certain embodiments, at least one instance of R B is -N(R B 1 ) 2 . In certain embodiments, at least one instance of R B is - NMe 2 . In certain embodiments, at least one instance of R is -NH 2 .
- at least one instance of R B is -N0 2 .
- at least one instance of R B is
- two R B groups may be joined to form a substituted or unsubstituted carbocyclic ring.
- two instances of R are joined to form a saturated or unsaturated carbocyclic ring.
- two instances of R are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system.
- two instances of R are joined to form a 3- to 7-membered, monocyclic carbocyclic ring.
- two instances of R are joined to form a 3-membered carbocyclic ring.
- two instances of R are joined to form a 4-membered carbocyclic ring.
- two instances of R are joined to form a 5-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 7-membered carbocyclic ring. In certain embodiments,
- two instances of R are joined to form a 5- to 13-membered, bicyclic carbocyclic ring.
- two instances of R B are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- two instances of R B are joined to form a substituted or unsubstituted aryl ring. In certain embodiments, two instances of R are joined to form a 6- to 14-membered aryl ring. In certain embodiments, two instances of R are joined to form a 6- to 10-membered aryl ring. In certain embodiments, two instances of R are joined to form a monocyclic aryl ring. In certain embodiments, two instances of R are joined to form an unsubstituted phenyl ring. In certain embodiments, two instances of R are joined to form a substituted phenyl ring. In certain embodiments, two instances of R are joined to form a bicyclic aryl ring. In certain embodiments, two instances of R are joined to form a naphthyl ring.
- two instances of R B are joined to form a substituted or unsubstituted heteroaryl ring.
- two instances of R are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R are joined to form a substituted or unsubstituted pyridyl ring.
- each instance of R is H.
- at least one instance of R is halogen or substituted or unsubstituted alkyl.
- at least one instance of R is halogen or substituted or unsubstituted C 1-6 alkyl.
- At least one instance of R B1 is H. In certain embodiments, at least one instance of R B 1 is substituted acyl. In certain embodiments, at least one instance of R B 1 is unsubstituted acyl. In certain embodiments, at least one instance of R B 1 is acetyl. In certain embodiments, at least one instance of R B 1 is substituted alkyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkyl. In certain embodiments, at least one instance of R B 1 is C 1-12 alkyl. In certain embodiments, at least one instance of R B 1 is C 1-6 alkyl. In certain embodiments, at least one instance of R B 1 is methyl.
- At least one instance of R B 1 is ethyl. In certain embodiments, at least one instance of R B 1 is propyl. In certain embodiments, at least one instance of R B 1 is butyl. In certain embodiments, at least one instance of R B 1 is pentyl. In certain embodiments, at least one instance of R B 1 is hexyl. In certain embodiments, at least one instance of R B 1 is substituted alkenyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkenyl. In certain embodiments, at least one instance of R B 1 is vinyl. In certain embodiments, at least one instance of R B 1 is substituted alkynyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkynyl. In certain embodiments, at least one instance of R B 1 is ethynyl. In certain
- At least one instance of R B 1 is substituted carbocyclyl. In certain embodiments, at least one instance of R B 1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R B 1 is saturated carbocyclyl. In certain embodiments, at least one instance of R B 1 is unsaturated carbocyclyl. In certain embodiments, at least one instance of R B 1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R B 1 is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R B 1 is cylcopropyl.
- At least one instance of R B 1 is cyclobutyl. In certain embodiments, at least one instance of R B 1 is cyclopentyl. In certain embodiments, at least one instance of R B 1 is cyclohexyl. In certain embodiments, at least one instance of R B 1 is cycloheptyl. In certain embodiments, at least one instance of R B 1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R B 1 is substituted heterocyclyl. In certain embodiments, at least one instance of R B 1 is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R B 1 is saturated heterocyclyl.
- At least one instance of R B 1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R B 1 is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R B 1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R B 1 is 5- to 13- membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R B 1 is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R B 1 is 6- to
- At least one instance of R B 1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R B 1 is monocyclic aryl. In certain embodiments, at least one instance of R B 1 is unsubstituted phenyl. In certain embodiments, at least one instance of R B 1 is substituted phenyl. In certain embodiments, at least one instance of R B 1 is bicyclic aryl. In certain embodiments, at least one instance of R B 1 is naphthyl. In certain embodiments, at least one instance of R B 1 is substituted or unsubstituted heteroaryl.
- At least one instance of R B 1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R B 1 is pyridyl.
- at least one instance of R B 1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- At least one instance of R B 1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R B 1 is a nitrogen protecting group when attached to a nitrogen atom.
- at least one instance of R B 1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom.
- R B 1 is an oxygen protecting group when attached to an oxygen atom.
- R B 1 is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ⁇ -Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom.
- R B 1 is a sulfur protecting group when attached to a sulfur atom.
- R B 1 is acetamidomethyl, ⁇ -Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
- two instances of R B1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R B 1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
- R B 1 are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system.
- two instances of R B 1 are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R B 1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring.
- two instances of R B 1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- At least one instance of m is 0. In certain embodiments, at least one instance of m is 1. In certain embodiments, at least one instance of m is 2. In certain embodiments, at least one instance of m is 3. In certain embodiments, at least one instance of m is 4. In certain embodiments, at least one instance of m is 5. In certain embodiments, both instances of m are 0. In certain embodiments, both instances of m are 1. In certain embodiments,
- both instances of m are 2. In certain embodiments, both instances of m are 3. In certain embodiments, both instances of m are 4. In certain embodiments, both instances of m are 5.
- a compound of Formula (B) is commercially available.
- An exemplary compound of Formula (B) is N-fluorobenzenesulfonimide (NFBS or NFSI).
- the methods of the invention involve a catalyst.
- the catalyst employed in the inventive methods includes a transition metal complex, such as a complex of any one of Formulae (CI) to (C25).
- Complexes of any one of Formulae (CI) to (C25) include a transition metal ion M.
- M is a transition metal cation.
- M is a monovalent transition metal ion.
- M is Ag + , Cu + , or Co + .
- M is a divalent transition metal ion.
- M is Pd 2+ , Ni 2+ , Cu 2+ , Ir 2+ , Zn 2+ , Mn 2+ , or Fe 2+ .
- M is a trivalent transition metal ion.
- M is Rh 3+ .
- M is a tetravalent, pentavalent, or hexavalent transition metal ion.
- Complexes of Formula (CI) include at least two instances of Ring B.
- all instances of Ring B are independently substituted or unsubstituted heteroaryl.
- at least one instance of Ring B is monocyclic, 5- membered heteroaryl, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- At least one instance of Ring B is of the formula: . In certain In certain
- At least one instance of Ring B is of the formula: e instance of Ring B is of the formula: , or embodiments, at least one instance of Ring B is of the formula: , 5 or In certain embodiments at least one instance of
- Ring B is of the formula: embodiments, at least one instance of Ring B is of the formula:
- Ring B is of the formula:
- At least one instance of Ring B is of the
- At least one instance of Ring B is of the formula: ,
- At least one instance of Ring B is of the
- Ring B is of the formula: . In certain embodiments,
- At least one instance of Ring B is of the formula: . In certain embodiments, at least
- Ring B is of the formula: .
- at least one instance of Ring B is monocyclic, 6-membered heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of Ring B is of the
- At least one instance of Ring B is of the
- At least one instance of Ring B is of the formula: , (RC, ' , , or .
- At least one instance of Ring B is of the formula:
- each instance of Ring B is substituted or unsubstituted, monocyclic, 5- or 6-membered heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of Ring B is bicyclic, 9- or 10-membered heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- Complexes of Formula (CI) may include one or more substituents R c .
- at least one instance of R is H.
- at least one instance of R c is halogen.
- at least one instance of R c is F.
- at least one instance of R is CI.
- at least one instance of R is Br.
- at least one instance of R c is I (iodine).
- at least one instance of R is substituted acyl.
- at least one instance of R c is unsubstituted acyl.
- at least one instance of R c is substituted alkyl.
- at least one instance of R is unsubstituted alkyl.
- At least one instance of R is C 1-12 alkyl. In certain embodiments, at least one instance of R is substituted C 1-6 alkyl. In certain embodiments, at least one instance of R c is unsubstituted C 1-6 alkyl. In certain embodiments, at least one instance of R c is substituted methyl. In certain embodiments, at least one instance of R is -CH 2 F. In certain embodiments, at least one instance of R is -CHF 2 . In certain embodiments, at least one instance of R c is -CF 3 . In certain embodiments, at least one instance of R c is Bn. In certain embodiments, at least one instance of R is unsubstituted methyl. In certain embodiments, at least one instance of R c is ethyl. In certain embodiments, at least one instance of R c is propyl.
- At least one instance of R is butyl. In certain embodiments, at least one instance of R c is pentyl. In certain embodiments, at least one instance of R c is hexyl. In certain embodiments, at least one instance of R is substituted alkenyl. In certain embodiments,
- At least one instance of R is unsubstituted alkenyl. In certain embodiments, at least one instance of R is substituted or unsubstituted C 1-6 alkenyl. In certain embodiments, at least one instance of R c is vinyl. In certain embodiments, at least one instance of R c is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R is ethynyl. In certain
- At least one instance of R is substituted carbocyclyl. In certain embodiments, at least one instance of R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R c is saturated carbocyclyl. In certain embodiments, at least one instance of R c is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R is monocyclic carbocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R c is cyclobutyl. In certain embodiments, at least one instance of R c is
- At least one instance of R is cyclohexyl. In certain embodiments, at least one instance of R is cycloheptyl. In certain embodiments, at least one instance of R c is bicyclic carbocyclyl. In certain embodiments, at least one instance of R c is bicyclic, 5- to 13-membered carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is
- At least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl.
- At least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- At least one instance of R is monocyclic heterocyclyl. In certain embodiments, at least one instance of R is monocylic, 3- to 7-membered heterocyclyl. In certain embodiments, at least one instance of R is bicyclic heterocyclyl. In certain embodiments, at least one instance of R is bicyclic, 5- to 13-membered heterocyclyl. In certain embodiments, at least one instance of R is substituted aryl. In certain embodiments, at least one instance of R is unsubstituted aryl. In certain embodiments, at least one instance of R c is 6- to 14-membered aryl. In certain embodiments, at least one instance of R c is 6- to
- At least one instance of R is substituted phenyl.
- At least one instance of R is unsubstituted phenyl. In certain embodiments, at least one instance of R is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R c is substituted heteroaryl. In certain embodiments, at least one instance of R c is unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl.
- At least one instance of R is monocyclic, 5-membered heteroaryl. In certain embodiments, at least one instance of R is monocyclic, 6-membered heteroaryl. In certain embodiments, at least one instance of R c is substituted pyridyl. In certain embodiments, at least one instance of R c is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- At least one instance of R is bicyclic, 9-membered heteroaryl. In certain embodiments, at least one instance of R is bicyclic, 10-membered heteroaryl. In certain embodiments, at least one instance of R C is -OR CI . In certain embodiments, at least one instance of R C is -OR CI , wherein R CI is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is -OMe. In certain embodiments, at least one instance of R c is -OEt. In certain embodiments, at least one instance of R c is -OPr. In certain embodiments, at least one instance of R is -OBu. In certain embodiments, at least one instance of R c is -O(pentyl). In certain embodiments, at least one instance of R c is -
- At least one instance of R is -OBn.
- At least one instance of R is -OPh. In certain embodiments, at least one instance of R C is -OH. In certain embodiments, at least one instance of R C is -SR CI . In certain embodiments, at least one instance of R is -SMe. In certain embodiments, at least one instance of R C is -SH. In certain embodiments, at least one instance of R C is -N(R CI ) 2 . In certain embodiments, at least one instance of R is -NMe 2 . In certain embodiments, at least one instance of R c is -NH 2 . In certain embodiments, at least one instance of R c is -CN. In certain embodiments, at least one instance of R is -SCN.
- at least one instance of R c is -
- at least one instance of R c is -N0 2 .
- two R c groups may be joined to form a substituted or unsubstituted carbocyclic ring.
- two instances of R are joined to form a saturated or unsaturated carbocyclic ring.
- two instances of R are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system.
- two instances of R are joined to form a 3- to 7-membered, monocyclic carbocyclic ring.
- two instances of R are joined to form a 3-membered carbocyclic ring.
- two instances of R are joined to form a 4-membered carbocyclic ring.
- two instances of R are joined to form a 5-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 7-membered carbocyclic ring. In certain embodiments,
- two instances of R are joined to form a 5- to 13-membered, bicyclic carbocyclic ring.
- two instances of R c are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- two instances of R c are joined to form a substituted or unsubstituted aryl ring.
- two instances of R are joined to form a 6- to 14-membered aryl ring.
- two instances of R are joined to form a 6- to 10-membered aryl ring.
- two instances of R are joined to form a monocyclic aryl ring.
- two instances of R are joined to form an unsubstituted phenyl ring.
- two instances of R are joined to form a substituted phenyl ring.
- two instances of R are joined to form a bicyclic aryl ring.
- two instances of R are joined to form a naphthyl ring.
- two instances of R c are joined to form a substituted or unsubstituted heteroaryl ring.
- two instances of R are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R are joined to form a substituted or unsubstituted pyridyl ring.
- two instances of R are joined to form a 9- or 10-membered, bicyclic heteroaryl ring, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- each instance of R c is H.
- at least one instance of R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR CI .
- at least one instance of R c is halogen, substituted or unsubstituted C 1-6 alkyl, substituted or unsubstituted phenyl, or -OR CI , wherein R CI is substituted or unsubstituted C 1-6 alkyl.
- At least one instance of R C 1 is H. In certain embodiments, at least one instance of R C 1 is substituted acyl. In certain embodiments, at least one instance of R CI is unsubstituted acyl. In certain embodiments, at least one instance of R CI is acetyl. In certain embodiments, at least one instance of R CI is substituted alkyl. In certain embodiments, at least one instance of R C 1 is unsubstituted alkyl. In certain embodiments, at least one instance of R CI is C 1-12 alkyl. In certain embodiments, at least one instance of R CI is C 1-6 alkyl. In certain embodiments, at least one instance of R CI is methyl.
- At least one instance of R CI is ethyl. In certain embodiments, at least one instance of R CI is propyl. In certain embodiments, at least one instance of R C 1 is butyl. In certain embodiments, at least one instance of R CI is pentyl. In certain embodiments, at least one instance of R CI is hexyl. In certain embodiments, at least one instance of R C 1 is substituted alkenyl. In certain embodiments, at least one instance of R CI is unsubstituted alkenyl. In certain embodiments, at least one instance of R CI is vinyl. In certain embodiments, at least one instance of R CI is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R CI is ethynyl. In certain
- At least one instance of R CI is substituted carbocyclyl. In certain embodiments, at least one instance of R C 1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R CI is saturated carbocyclyl. In certain embodiments, at least one instance of R CI is unsaturated carbocyclyl. In certain embodiments, at least one instance of R C 1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R CI is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R CI is cylcopropyl.
- At least one instance of R CI is cyclobutyl. In certain embodiments, at least one instance of R CI is cyclopentyl. In certain embodiments, at least one instance of R CI is cyclohexyl. In certain embodiments, at least one instance of R CI is cycloheptyl. In certain embodiments, at least one instance of R C 1 is bicyclic, 5- to 13-membered carbocyclyl. In certain embodiments, at least one instance of R CI is substituted heterocyclyl. In certain embodiments, at least one instance of R CI is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R CI is saturated heterocyclyl.
- At least one instance of R C 1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R CI is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R C 1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R C 1 is monocyclic, 3- to
- At least one instance of R CI is bicyclic, 5- to 13-membered heterocyclyl. In certain embodiments, at least one instance of R C 1 is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R CI is 6- to
- At least one instance of R C 1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R C 1 is monocyclic aryl. In certain embodiments, at least one instance of R CI is unsubstituted phenyl. In certain embodiments, at least one instance of R CI is substituted phenyl. In certain embodiments, at least one instance of R CI is bicyclic aryl. In certain embodiments, at least one instance of R CI is naphthyl. In certain embodiments, at least one instance of R CI is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of R CI is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are
- At least one instance of R CI is pyridyl.
- at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- R is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of
- At least one instance of R is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least
- R is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts
- R is an oxygen protecting group
- R is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ⁇ -Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to
- R is a sulfur protecting group when attached to a
- R is acetamidomethyl, ⁇ -Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
- two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
- R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in
- heterocyclic ring system two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R are joined to form a 3- to 7-membered, monocyclic
- heterocyclic ring In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- At least one instance of r is 0. In certain embodiments, at least one instance of r is 1. In certain embodiments, at least one instance of r is 2. In certain embodiments, at least one instance of r is 3. In certain embodiments, at least one instance of r is 4. In certain embodiments, at least one instance of r is 5. In certain embodiments, each instance of r is 0. In certain embodiments, each instance of r is 1. In certain embodiments, each instance of r is 2. In certain embodiments, each instance of r is 3. In certain
- each instance of r is 4. In certain embodiments, each instance of r is 5.
- At least one instance of r is 1 ; and at least one instance of
- R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR .
- at least one instance of r is 1 ; and at least one instance of R is halogen, substituted or unsubstituted C . alkyl, substituted or unsubstituted phenyl, or - OR CI , wherein R CI is substituted or unsubstituted C 1-6 alkyl.
- each instance of r is 1; and each instance of R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR C 1.
- each instance of r is 1; and each instance of R is halogen, substituted or unsubstituted C 1-6 alkyl, substituted or unsubstituted phenyl, or -OR CI , wherein R CI is substituted or unsubstituted C 1-6 alkyl.
- Complexes of Formula (CI) or (C2) include more than one substituent R D .
- R D is substituted acyl.
- at least one instance of R D is unsubstituted acyl.
- at least one instance of R D is substituted alkyl.
- at least one instance of R D is unsubstituted alkyl.
- at least one instance of R D is C 1-12 alkyl.
- At least one instance of R D is substituted C 1-6 alkyl. In certain embodiments, at least one instance of R D is unsubstituted C 1-6 alkyl. In certain embodiments, at least one instance of R D is substituted methyl. In certain embodiments, at least one instance of R D is - CH 2 F. In certain embodiments, at least one instance of R D is -CHF 2 . In certain embodiments, at least one instance of R D is -CF 3 . In certain embodiments, at least one instance of R D is Bn. In certain embodiments, at least one instance of R D is unsubstituted methyl. In certain embodiments, at least one instance of R D is ethyl.
- At least one instance of R D is propyl. In certain embodiments, at least one instance of R D is butyl. In certain embodiments, at least one instance of R D is pentyl. In certain embodiments, at least one instance of R D is hexyl. In certain embodiments, at least one instance of R D is substituted alkenyl. In certain embodiments, at least one instance of R D is unsubstituted alkenyl. In certain embodiments, at least one instance of R D is substituted or unsubstituted C 1-6 alkenyl. In certain embodiments, at least one instance of R D is vinyl. In certain embodiments, at least one instance of R D is substituted alkynyl.
- At least one instance of R D is unsubstituted alkynyl. In certain embodiments, at least one instance of R D is ethynyl. In certain embodiments, at least one instance of R D is substituted carbocyclyl. In certain embodiments, at least one instance of R D is unsubstituted carbocyclyl. In certain embodiments,
- At least one instance of R D is saturated carbocyclyl. In certain embodiments, at least one instance of R D is unsaturated carbocyclyl. In certain embodiments, at least one instance of R D is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R D is monocyclic carbocyclyl. In certain embodiments, at least one instance of R D is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R D is cylcopropyl. In certain embodiments, at least one instance of R D is cyclobutyl. In certain embodiments, at least one instance of R is cyclopentyl.
- At least one instance of R is cyclohexyl. In certain embodiments, at least one instance of R D is cycloheptyl. In certain embodiments, at least one instance of R D is bicyclic carbocyclyl. In certain embodiments, at least one instance of R D is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R D is substituted heterocyclyl. In certain embodiments, at least one instance of R D is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R D is saturated heterocyclyl. In certain embodiments, at least one instance of R D is unsaturated heterocyclyl.
- At least one instance of R D is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R D is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R D is monocyclic heterocyclyl. In certain embodiments, at least one instance of R D is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R D is bicyclic heterocyclyl. In certain embodiments, at least one instance of R D is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R D is substituted aryl. In certain embodiments, at least one instance of R D is unsubstituted aryl. In certain embodiments,
- At least one instance of R D is 6- to 14-membered aryl. In certain embodiments, at least one instance of R D is 6- to 10-membered aryl. In certain embodiments, at least one instance of R D is substituted phenyl. In certain embodiments, at least one instance of R D is unsubstituted phenyl. In certain embodiments, at least one instance of R D is substituted naphthyl. In certain embodiments, at least one instance of R D is unsubstituted naphthyl. In certain embodiments, at least one instance of R D is substituted heteroaryl. In certain embodiments, at least one instance of R D is unsubstituted heteroaryl.
- At least one instance of R D is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R D is monocyclic heteroaryl.
- at least one instance of R D is 5-membered, monocyclic heteroaryl.
- at least one instance of R D is 6-membered, monocyclic heteroaryl.
- at least one instance of R D is substituted pyridyl.
- At least one instance of R D is unsubstituted 2-pyridyl, unsubstituted 3- pyridyl, or unsubstituted 4-pyridyl.
- at least one instance of R D is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- at least one instance of R is 9-membered, bicyclic heteroaryl.
- at least one instance of R is 10-membered, bicyclic heteroaryl.
- at least one instance of R D is a nitrogen protecting group.
- at least one instance of R D is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts.
- two instances of R D are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R D are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R D are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R D are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R D are joined to form a 3- to 7-membered, monocyclic heterocyclic ring.
- two instances of R D are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R D are joined to form a
- heterocyclic ring of 5 wherein the heterocyclic ring is substituted or unsubstituted.
- two instances of R 1 are joined to form a 5- to 13-membered, bicyclic heterocyclic ring.
- two instances of R D are joined to form a substituted or unsubstituted, 7- to 11-membered, bicyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- heterocyclic ring of the formula: , wherein the heterocyclic ring is substituted or unsubstituted.
- At least one instance of R D is H, substituted or
- R D is H, substituted or unsubstituted C 1-6 alkyl, or two instances of R are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- each instance of R D is H, substituted or unsubstituted alkyl, or two instances of R D are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, each instance of R D is H, substituted or
- At least one instance of R D1 is H. In certain embodiments, at least one instance of R D1 is substituted acyl. In certain embodiments, at least one instance of R D1 is unsubstituted acyl. In certain embodiments, at least one instance of R D1 is acetyl. In certain embodiments, at least one instance of R D1 is substituted alkyl. In certain
- At least one instance of R D1 is unsubstituted alkyl. In certain embodiments, at least one instance of R D1 is C 1-12 alkyl. In certain embodiments, at least one instance of R D1 is Ci-6 alkyl. In certain embodiments, at least one instance of R D1 is methyl. In certain embodiments, at least one instance of R D1 is ethyl. In certain embodiments, at least one instance of R D1 is propyl. In certain embodiments, at least one instance of R D1 is butyl. In certain embodiments, at least one instance of R D1 is pentyl. In certain embodiments, at least one instance of R D1 is hexyl.
- At least one instance of R D1 is substituted alkenyl. In certain embodiments, at least one instance of R D1 is unsubstituted alkenyl. In certain embodiments, at least one instance of R D1 is vinyl. In certain embodiments,
- At least one instance of R D1 is substituted alkynyl. In certain embodiments, at least one instance of R D1 is unsubstituted alkynyl. In certain embodiments, at least one instance of R D1 is ethynyl. In certain embodiments, at least one instance of R D1 is substituted carbocyclyl. In certain embodiments, at least one instance of R D1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R D1 is saturated carbocyclyl. In certain embodiments, at least one instance of R D1 is unsaturated carbocyclyl.
- At least one instance of R D1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R D1 is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R D1 is cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl. In certain embodiments, at least one instance of R D1 is cyclopentyl. In certain embodiments, at least one instance of R D1 is cyclohexyl. In certain embodiments, at least one instance of R D1 is cycloheptyl.
- At least one instance of R D1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R D1 is substituted heterocyclyl. In certain embodiments, at least one instance of R D1 is unsubstituted
- heterocyclyl In certain embodiments, at least one instance of R D1 is saturated heterocyclyl. In certain embodiments, at least one instance of R D1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R D1 is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R D1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R D1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R D1 is 5- to 13-membered, bicyclic
- R D1 is substituted or
- At least one instance of R D1 is 6- to 14-membered aryl. In certain embodiments, at least one instance of R D1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R D1 is monocyclic aryl. In certain embodiments, at least one instance of R D1 is unsubstituted phenyl. In certain embodiments, at least one instance of R D1 is substituted phenyl. In certain embodiments, at least one instance of R D1 is bicyclic aryl. In certain embodiments, at least one instance of R D1 is naphthyl.
- At least one instance of R D1 is substituted or unsubstituted heteroaryl.
- at least one instance of R D1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R D1 is pyridyl.
- at least one instance of R D1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- At least one instance of R D1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R D1 is a nitrogen protecting group when attached to a nitrogen atom.
- at least one instance of R D1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom.
- R D1 is an oxygen protecting group when attached to an oxygen atom.
- R is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, i-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom.
- R D1 is a sulfur protecting group when attached to a sulfur atom.
- R D1 is acetamidomethyl, ⁇ -Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
- two instances of R D1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R D1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R D1 are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R D1 are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R D1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R D1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- Complexes of Formula (CI) or (C2) may include one or more substituent R E .
- At least one instance of R is absent. In certain embodiments, at least one instance of R E is H. In certain embodiments, at least one instance of R E is substituted p
- acyl In certain embodiments, at least one instance of R is unsubstituted acyl. In certain embodiments,
- At least one instance of R is substituted alkyl. In certain embodiments, at least one instance of R E is unsubstituted alkyl. In certain embodiments, at least one instance of R E p
- R is C 1-12 alkyl. In certain embodiments, at least one instance of R is substituted C 1-6 alkyl. In p
- At least one instance of R is unsubstituted C 1-6 alkyl.
- At least one instance of R is substituted methyl. In certain embodiments, at least one instance of R E is -CH 2 F. In certain embodiments, at least one instance of R E is - p
- At least one instance of R is -CF 3 . In certain embodiments, at least one instance of R E is Bn. In certain embodiments, at least one instance of R E is
- At least one instance of R is ethyl.
- At least one instance of R is propyl. In certain embodiments, at least one instance of R E is butyl. In certain embodiments, at least one instance of R E is pentyl. In
- At least one instance of R is hexyl. In certain embodiments, at least one instance of R E is substituted alkenyl. In certain embodiments, at least one instance of R E p
- R is unsubstituted alkenyl. In certain embodiments, at least one instance of R is substituted or p
- At least one instance of R is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R is ethynyl. In certain embodiments, at least one instance of R is substituted carbocyclyl. In certain embodiments, at least one instance of
- R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R is saturated carbocyclyl. In certain embodiments, at least one instance of R is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R is monocyclic carbocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl.
- At least one instance of R is cyclopentyl. In certain embodiments, at least one instance of R is cyclohexyl. In certain embodiments, at least one instance of R is cycloheptyl. In certain embodiments, at least one instance of R is bicyclic carbocyclyl. In certain embodiments, at least one instance of R is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is unsubstituted
- heterocyclyl In certain embodiments, at least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl. In certain embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of
- R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R is monocyclic heterocyclyl.
- at least one instance of R is 3- to 7-membered, monocyclic heterocyclyl.
- at least one instance of R is bicyclic heterocyclyl.
- at least one instance of R is 5- to 13-membered, bicyclic heterocyclyl.
- at least one instance of R is substituted aryl.
- R is unsubstituted aryl. In certain embodiments, at least one instance of R is 6- to 14- membered aryl. In certain embodiments, at least one instance of R is 6- to 10-membered aryl. In certain embodiments, at least one instance of R is substituted phenyl. In certain embodiments, at least one instance of R is unsubstituted phenyl. In certain embodiments, at least one instance of R is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R is substituted heteroaryl. In certain embodiments, at least one instance of R is unsubstituted heteroaryl.
- At least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl. In certain embodiments, at least one instance of R is
- At least one instance of R is 6- membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is substituted pyridyl. In certain embodiments, at least one instance of R is unsubstituted 2- pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R is 9-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is a nitrogen protecting group. In certain embodiments, at least one instance of
- R is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts.
- two instances of R E are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
- R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system.
- two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring.
- two instances of R are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- two instances of R are joined to form a substituted or unsubstituted, 7- to 11 -membered, bicyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- At least one instance of R E is H or substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is H or substituted or unsubstituted C 1-6 alkyl. In certain embodiments, each instance of R is H or substituted or unsubstituted alkyl. In certain embodiments, each instance of R is H or substituted or unsubstituted C 1-6 alkyl.
- At least one instance of R E1 is H. In certain embodiments, at least one instance of R El is substituted acyl. In certain embodiments, at least one instance of R El is unsubstituted acyl. In certain embodiments, at least one instance of R El is acetyl. In certain embodiments, at least one instance of R El is substituted alkyl. In certain embodiments, at least one instance of R El is unsubstituted alkyl. In certain embodiments, at least one instance of R El is C 1-12 alkyl. In certain embodiments, at least one instance of R El is C 1-6 alkyl. In certain embodiments, at least one instance of R El is methyl.
- At least one instance of R El is ethyl. In certain embodiments, at least one instance of R El is propyl. In certain embodiments, at least one instance of R El is butyl. In certain embodiments, at least one instance of R El is pentyl. In certain embodiments, at least one instance of R El is hexyl. In certain embodiments, at least one instance of R El is substituted alkenyl. In certain embodiments, at least one instance of R El is unsubstituted alkenyl. In certain embodiments, at least one instance of R El is vinyl. In certain embodiments, at least one instance of R El is substituted alkynyl. In certain embodiments, at least one instance of R El is unsubstituted alkynyl. In certain embodiments, at least one instance of R El is ethynyl. In certain
- At least one instance of R El is substituted carbocyclyl. In certain embodiments, at least one instance of R El is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R El is saturated carbocyclyl. In certain embodiments, at least one instance of R El is unsaturated carbocyclyl. In certain embodiments, at least one instance of R El is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R El is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R El is cylcopropyl.
- At least one instance of R El is cyclobutyl. In certain embodiments, at least one instance of R El is cyclopentyl. In certain embodiments, at least one instance of R El is cyclohexyl. In certain embodiments, at least one instance of R El is cycloheptyl. In certain embodiments, at least one instance of R El is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R El is substituted heterocyclyl. In certain embodiments, at least one instance of R El is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R El is saturated heterocyclyl.
- At least one instance of R El is unsaturated heterocyclyl. In certain embodiments, at least one instance of R El is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R El is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R El is 5- to 13- membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R El is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R El is 6- to
- At least one instance of R El is 6- to 10-membered aryl. In certain embodiments, at least one instance of R El is monocyclic aryl. In certain embodiments, at least one instance of R El is unsubstituted phenyl. In certain embodiments, at least one instance of R El is substituted phenyl. In certain embodiments, at least one instance of R El is bicyclic aryl. In certain embodiments, at least one instance of R El is naphthyl. In certain embodiments, at least one instance of R El is substituted or unsubstituted heteroaryl.
- At least one instance of R El is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R El is pyridyl.
- at least one instance of R El is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits.
- At least one instance of R El is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- at least one instance of R El is a nitrogen protecting group when attached to a nitrogen atom.
- at least one instance of R El is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom.
- R El is an oxygen protecting group when attached to an oxygen atom.
- R El is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ⁇ -Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom.
- R El is a sulfur protecting group when attached to a sulfur atom.
- R El is acetamidomethyl, ⁇ -Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
- two instances of R E1 are joined to form a substituted or unsubstituted heterocyclic ring.
- two instances of R El are joined to form a saturated or unsaturated heterocyclic ring.
- R El are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system.
- two instances of R El are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
- two instances of R El are joined to form a 3- to 7-membered, monocyclic heterocyclic ring.
- two instances of R El are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
- At least one instance of n is 0. In certain embodiments, at least one instance of n is 1. In certain embodiments, each instance of n is 0. In certain embodiments, each instance of n is 1.
- p is 1. In certain embodiments, p is 2.
- Complexes of any one of Formulae (CI) to (C25) are typically electrically neutral compounds and include one or more anionic counterions X.
- each instance of X is a non-coordinating anionic counterion.
- each instance of X is a monovalent anionic counterion.
- each instance of X is C10 4
- each instance of X is B[3,5-
- each instance of X is a divalent anionic counterion. In certain embodiments, each instance of X is S0 4 " .
- q is 1. In certain embodiments, q is 2. In certain embodiments, q is 3.
- M is a monovalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 1.
- M is a divalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 2.
- M is a trivalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 3.
- the transition metal complex is of Formula (CI). In certain embodiments, the transition metal complex is of the formula:
- lex is of the formula:
- lex is of the formula:
- each instance of X is a monovalent anionic counterion (e.g. , C10 4 , ⁇ , BF 4 , PF 4 PF 6 , or SbF 6 ).
- the transition metal complex is of any one of the formulae:
- each instance of X is a monovalent anionic counterion (e.g. , C10 4 , ⁇ , BF 4 , PF 4 , PF 6 , or SbF 6 ).
- the transition metal complex is of Formula (C2).
- the transition metal complex is of the formula: wherein each instance of X is a monovalent anionic counterion (e.g. , C10 4 , ⁇ , BF 4 , PF 4 , PF 6 , or SbF 6 -).
- the transition metal complex is of Formula (C3).
- the transition metal complex is of the formula: wherein each instance of X is a monovalent anionic counterion (e.g. , C10 4 , ⁇ , BF 4 , PF 4 , PF 6 , or SbF 6 ).
- the transition metal complex is of the formula:
- the transition metal complex is of Formula (C4). In certain embodiments the transition metal complex is of any one of the formulae:
- each instance of X is a monovalent anionic counterion (e.g. , C10 4 , ⁇ , BF 4 , PF 4 , PF 6 , or SbF 6 ).
- Exemplary complexes of Formula (C4) include, but are not limited to:
- the transition metal complex is of any one of Formulae (C5) to (C25).
- the methods of the invention may involve a single-electron reductant.
- the single-electron reductant is an inorganic single-electron reductant.
- the single-electron reductant is a single-electron reductive, transition metal salt.
- the single-electron reductant is an Ag(I) salt.
- the single-electron reductant is Ag(2,2'-bipyridine) 2 C10 4 (i.e. , Ag(bipy) 2 C10 4 ).
- the single-electron reductant is a Ru(II) salt.
- the single-electron reductant is Ru(2,2'-bipyridine)3(PF6) 2 (i.e. , Ru(bipy)3(PF6) 2 ).
- the single-electron reductant is an lr(0) complex, Cu(I) salt, Sm(II) salt, Co(II) salt, Cr(II) salt, Fe(II) salt, Ni(III) salt, Nd(III) salt, Yb(III) salt, or Ti(III) salt.
- the single-electron reductant is Cu(bipy) 2 PF 6 , Cu(bipy) 2 (OTf) 2 ,
- the single- electron reductant is an organic single-electron reductant. In certain embodiments, the single- electron reductant is commercially available.
- Complexes of Formula (CI) may be synthesized by contacting a ligand of Formula (Dl), or a salt thereof, with a transition metal salt of Formula (E) to provide the complexes of Formul
- Ring B, R , R , R , r, n, M, X, p, and q are as described herein;
- each instance of L is independently a ligand molecule
- s 0, 1, 2, 3, or 4.
- the transition metal salt of Formula (E) is N-(2-aminoethyl)-2-aminoethyl
- Transition metal salts of Formula (E) may include one or more ligand molecules L.
- at least one instance of L is a ligand molecule described herein.
- each instance of L is a ligand molecule described herein.
- at least one instance of L is a molecule of a suitable solvent described herein.
- each instance of L is a molecule of a suitable solvent described herein.
- at least one instance of L is acetonitrile (ACN).
- each instance of L is acetonitrile.
- at least one instance of L is an amide (e.g.
- At least one instance of L is dimethyl sulfoxide (DMSO).
- at least one instance of L is an ether (e.g. , tetrahydrofuran (THF), 2-methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl i-butyl ether (MTBE), dimethoxyethane (DME), or diglyme).
- at least one instance of L is a ketone (e.g.
- At least one instance of L is a chlorohydrocarbon (e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, or 1,2-dichloroethane (DCE)).
- at least one instance of L is an ester (e.g. , propylene carbonate or ethyl acetate).
- at least one instance of L is commercially available. In certain embodiments, each instance of L is commercially available.
- s is 0. In certain embodiments, s is 1. In certain embodiments, s is 2. In certain embodiments, s is 3. In certain embodiments, s is 4.
- the ligand of Formula (Dl) is commercially available.
- the transition metal salt of Formula (E) is commercially available.
- Complexes of Formula (C2) may be prepared by contacting a ligand of Formula (D2), or a salt thereof, with a transition metal salt of Formula (E) to provide the complexes of Formula (C2):
- Complexes of any one of Formulae (C3) to (C25) may be synthesized by contacting a ligand of any one of Formulae (D3) to (D25), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of any one of Formulae (C3) to (C25), respectively:
- the ligand of any one of Formulae (D3) to (D25), or the salt thereof is commercially available.
- Complexes of any one of Formulae (CI) to (C25) may be isolated or may be generated in situ and reacted with a reagent or intermediate involved in a method of the invention.
- N-aryl sulfonimides or N-heteroaryl sulfonimides obtained by methods of the invention may be partially deprotected to provide N-aryl sulfonamides or N-heteroaryl sulfonamides or fully deprotected to provide aryl amines or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof).
- the N-aryl sulfonimides or N-heteroaryl sulfonimides are partially or fully deprotected by reacting them with a reductant, a strong acid, or a nucleophile.
- the reductant employed in the deprotection step is a mixture of an alkaline earth metal and an alcohol.
- the reductant is a mixture of magnesium and an alcohol.
- the reductant is a mixture of magnesium and an unsubstituted Ci_6 alkyl alcohol (e.g. , MeOH, EtOH, i-PrOH, or i-BuOH).
- the reductant is a mixture of zinc and an acid.
- the reductant is a mixture of zinc and an acid selected from the group consisting of HC1, HBr, HI, HC10 4 , HN0 3 , H 2 S0 4 , CH 3 S0 3 H, CF S0 3 H, CH C0 2 H, and CF C0 2 H.
- the reductant is a mixture of an alkali metal and ammonia.
- the reductant is a mixture of zinc and HC1.
- the strong acid employed in the deprotection step may be a strong inorganic acid or strong organic acid.
- the strong acid is an acid having a ⁇ ⁇ value of less than 3, less than 2, less than 1, or less than 0 at ambient temperature.
- the strong acid is HC1, HBr, HI, HC10 4 , HN0 3 , H 2 S0 4 , CH 3 S0 3 H, CF 3 S0 3 H, or CF 3 C0 2 H.
- the nucleophile useful in the deprotection step may be an alkyl lithium or phenyl lithium.
- the nucleophile is an unsubstituted Ci_6 alkyl lithium (e.g. , MeLi, w-BuLi, sec-BuLi, or ⁇ -BuLi).
- the nucleophile may also be a Grignard reagent.
- the nucleophile is of Formula: YMgZ, wherein Y is unsubstituted Ci_6 alkyl, unsubstituted phenyl, or -Si(unsubstituted Ci_6 alkyl) ; and Z is halogen.
- the nucleophile is MeMgCl, MeMgBr, or MeMgl. In certain embodiments, the nucleophile is EtMgCl, EtMgBr, or EtMgl. In certain embodiments, the nucleophile is PhMgCl, PhMgBr, or PhMgl. In certain embodiments, the nucleophile is TMSMgCl or TMSMgBr.
- the various step(s) of the inventive methods for preparing the N-aryl or N- heteroaryl sulfonimides e.g., compounds of Formula (I), and salts thereof
- aryl or heteroaryl amines e.g., compounds of Formula (II), and salts thereof
- catalysts e.g. , complexes of any one of Formulae (CI) to (C25)
- a suitable condition is a combination of physical and chemical parameters under which an intended product (e.g. , a compound of any one of Formulae (I), (II), and (CI) to (C25), or a salt thereof) may be formed using the inventive methods.
- a suitable condition may include a suitable solvent.
- the suitable solvent is a solvent described herein.
- the suitable solvent is an aprotic solvent.
- the suitable solvent is acetonitrile (ACN).
- the suitable solvent is an amide solvent (e.g., N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMA), or N-methyl-2-pyrrolidone (NMP).
- the suitable solvent is dimethyl sulfoxide (DMSO).
- the suitable solvent is an ether solvent (e.g., tetrahydrofuran (THF), 2-methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl i-butyl ether (MTBE), dimethoxyethane (DME), or diglyme).
- the suitable solvent is a ketone solvent (e.g. , acetone or butanone).
- the suitable solvent is a chlorohydrocarbon solvent (e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, or 1,2-dichloroethane (DCE)).
- the suitable solvent is an ester solvent (e.g. , propylene carbonate or ethyl acetate).
- the suitable solvent is a pro tic solvent.
- the suitable solvent is a mixture of two or more solvents described herein. In certain embodiments, the suitable solvent is commercially available.
- a suitable condition may also include a suitable temperature under which a step of a method of preparing the compounds of the invention is performed.
- the suitable temperature is at least about 0 °C, at least about 4 °C, at least about 10 °C, at least about 20 °C, at least about 23 °C, at least about 40 °C, at least about 50 °C, at least about 60 °C, at least about 80 °C, or at least about 100 °C.
- the suitable temperature is at most about 100 °C, at most about 80 °C, at most about 60 °C, at most about 50 °C, at most about 40 °C, at most about 23 °C, at most about 20 °C, at most about 10 °C, at most about 4 °C, or at most about 0 °C. Combinations of the above-referenced ranges (e.g., at least about 0 °C and at most about 60 °C) are also within the scope of the invention.
- a suitable temperature may be a variable temperature during a step of a method of preparing the compounds of the invention.
- a suitable condition may also include a suitable pressure under which a step of a method of preparing the compounds of the invention is performed.
- the suitable pressure is about 1 atmosphere.
- a suitable condition may also include a suitable atmosphere under which a step of a method of preparing the compounds of the invention is performed.
- the suitable atmosphere is air.
- the suitable atmosphere is an inert atmosphere.
- the suitable atmosphere is a nitrogen or argon atmosphere.
- a suitable condition may also include a suitable time duration that a step of a method of preparing the compounds of the invention lasts.
- the suitable time duration is in the order of minutes (e.g., about 10 minutes or about 30 minutes), hours (e.g. , about 1 hour, about 2 hours, about 4 hours, about 8 hours, or about 16 hours), or days (e.g., about 1 day).
- a suitable condition may also include agitating (e.g., stirring, sonicating, and/or shaking) one or more reactants, reagents, solvents, and/or solid supports involved in a method of the invention, or a mixture thereof.
- agitating e.g., stirring, sonicating, and/or shaking
- One or more intermediates resulting from a step of a method of preparing the compounds of the invention may be isolated and/or purified, and the isolated and/or purified intermediate may be reacted in a next step of the method.
- the isolated and/or purified intermediates may be substantially free of impurities or may contain one or more other components, such as reagents and solvents employed in the step yielding the intermediates, and byproducts.
- the one or more intermediates may also be reacted in a next step without being isolated and/or purified.
- the intermediates and/or intended products of the methods of preparing the N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof) and aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof) may be isolated and/or purified using methods known in the art, such as chromatography normal phase chromatography (e.g. , silica gel flash chromatography), reverse phase chromatography (e.g., high performance liquid chromatography (HPLC)), precipitation, decanting, filtration, centrifuge, trituration, crystallization, recrystallization, liquid-liquid phase separation, evaporation, and drying.
- chromatography normal phase chromatography e.g. , silica gel flash chromatography
- reverse phase chromatography e.g., high performance liquid chromatography (HPLC)
- precipitation decanting, filtration, centrifuge, trituration, crystallization
- the intended products described herein are substantially pure (e.g. , substantially free of impurities) (e.g., at least about 90%, at least about 95%, at least about 98%, at least about 99%, at least about 99.5%, at least about 99.9%, or more pure).
- la (I) is of the formula:
- Compounds of Formula (I) that may be prepared by the inventive methods include, but are not limited to, compounds of any one of the formulae:
- the transition metal complexes of the invention include amine-N-oxide ligands, such as ligands of Formula (Dl) to (D25).
- the inventive transition metal complexes are the transition metal complexes described herein
- the inventive transition metal complexes are complexes of Formula (CI).
- the inventive transition metal complexes are complexes of Formula (C2).
- the inventive transition metal complexes are complexes of Formula (C3).
- the inventive transition metal complexes are complexes of Formula (C4).
- the inventive transition metal complexes are complexes of any one of Formulae (C5) to (C25).
- the inventive transition metal complexes are electrically neutral.
- the complexes of the invention may be prepared according to the methods described herein or by processes known in the art. For example, one can use synthetic chemistry transformations (including protecting group methodologies), e.g., those described in R. Larock,
- kits e.g., packs.
- the kits are useful for preparing the compounds described herein (e.g., N-aryl or N-heteroaryl sulfonimides and aryl or heteroaryl amines) from an arene or heteroarene (e.g., a compound of Formula (A), or a salt thereof).
- the kits are useful for preparing the compounds of Formula (I), and salts thereof.
- the kits are useful for preparing the compounds of Formula (II), and salts thereof.
- a kit of the invention may include an N-fluorosulfonimide as an imidating agent; a catalyst; and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (CI); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (C2); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (C3); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (C4); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C5) to (C25); and a single-electron reductant.
- a kit of the invention includes N-fluorobenzenesulfonimide (NFBS or NFSI); a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant.
- a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and an Ag(I) or Ru(II) salt.
- kits may further include an arene or heteroarene.
- a kit of the invention further includes a compound of Formula (A), or a salt thereof.
- a kit of the invention may also include a deprotecting agent that is useful in deprotecting the N-aryl or N-heteroaryl sulfonimides prepared by the inventive methods to provide partially deprotected N-aryl or N-heteroaryl sulfonamides or fully deprotected aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof).
- a deprotecting agent that is useful in deprotecting the N-aryl or N-heteroaryl sulfonimides prepared by the inventive methods to provide partially deprotected N-aryl or N-heteroaryl sulfonamides or fully deprotected aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof).
- a kit of the invention further includes a reductant.
- a kit of the invention further includes an alkaline earth metal (e.g. , magnesium) and an alcohol (e.g., an unsubstituted Ci_6 alkyl alcohol).
- a kit of the invention further includes zinc and an acid (e.g. , HC1, HBr, HI, HC10 4 , HN0 3 , H 2 S0 4 , CH 3 S0 3 H, CF S0 3 H, CH C0 2 H, or CF C0 2 H).
- a kit of the invention further includes a strong acid (e.g.
- kits of the invention further includes a nucleophile.
- a kit of the invention further includes alkyl lithium (e.g. , an
- a kit of the invention further includes a Grignard reagent (e.g. , a compound of Formula: YMgZ, wherein Y and Z are as described herein).
- a Grignard reagent e.g. , a compound of Formula: YMgZ, wherein Y and Z are as described herein.
- kits provided may further include a container (e.g. , a vial, ampule, bottle, syringe, flask, tube, beaker, dish, microtiter plate, and/or dispenser package, or other suitable container), a solvent (e.g. , a suitable solvent described herein), or an organic or inorganic agent (e.g. , a phase-transfer agent, a solubilizing agent, a stabilizing agent, an anti- oxidative agent, protecting agent, deprotecting agent, and/or a preservative agent).
- a container e.g. , a vial, ampule, bottle, syringe, flask, tube, beaker, dish, microtiter plate, and/or dispenser package, or other suitable container
- a solvent e.g. , a suitable solvent described herein
- an organic or inorganic agent e.g. , a phase-transfer agent, a solubilizing agent, a stabilizing
- kits further include instructions for using the kits of the invention.
- the kits and instructions provide for preparing the compounds described herein (e.g., N-aryl or N-heteroaryl sulfonimides, aryl or heteroaryl amines, and transition metal complexes).
- the kits and instructions provide for preparing the compounds of Formula (I), and salts thereof.
- the kits and instructions provide for preparing the compounds described herein (e.g., N-aryl or N-heteroaryl sulfonimides, aryl or heteroaryl amines, and transition metal complexes).
- the kits and instructions provide for preparing the compounds of Formula (I), and salts thereof.
- kits and instructions provide for preparing the compounds of Formula (II), and salts thereof.
- the kits and instructions provide for preparing the complexes of any one of Formulae (C1)-(C25).
- Example 1 Preparation of the Complexes of any one of Formulae (CI) to (C25), the N-Aryl or ⁇ -Heteroaryl Sulfonimides of Formula (I), and the Aryl or Heteroaryl Amines of Formula (I).
- Complexes of any one of Formulae (CI) to (C25) may be prepared from ligands of Formula (Dl) to (D25), or salts thereof, with transition metal salts of Formula (E).
- complex 1 a complex of Formula (CI), is synthesized by reacting N-(2- pyridylmethyl)pyrrolidine-N-oxide with a palladium(II) salt (e.g.,
- the ligand N-(2-pyridylmethyl)pyrrolidine-N- oxide can be prepared in two steps from commercially available starting materials.
- the complexes of the invention may also be s nthesized by methods known in the art.
- N-fluorobenzenesulfonimide (NFBS or NFS I) is used as an imidating agent, complex 1 may act as a catalyst, and Ag(bipy) 2 C10 4 may work as a single- electron reductant. Both complex 1 and the Ag(bipy) 2 C10 4 are required in a reaction of Scheme 1 ; control experiments in which either complex 1 or the Ag(bipy) 2 C10 4 is omitted gave only trace amounts of imidated products. In another set of experiments, Ag(bipy) 2 C10 4 can be replaced with Ru(bipy) 3 (PF 6 ) 2 with similar results (Table 1, entry 2a). The reactions proceed similarly well in the presence or absence of light. Complex 1 involves an unusual pyridine-N- oxide ligand motif.
- complex 1 is easily prepared and conveniently handled, it can be isolated, and the isolated complex 1 can be employed in the methods of the invention.
- Complex 1 can also be generated in situ by reacting N-(2-pyridylmethyl)pyrrolidine-N-oxide with a palladium(II) salt (e.g., Pd(NCMe)4(OTf) 2 ), and the in situ generated complex 1 can be used in the inventive methods.
- a palladium(II) salt e.g., Pd(NCMe)4(OTf) 2
- a variety of substrates i.e., arenes and heteroarenes, can be efficiently imidated (e.g., Table 1).
- Regio selectivity is substrate-intrinsic.
- Resonance donors such as alkoxy and halogen groups, may direct imidation ortho/para, similar to the regio selectivity in an electrophilic aromatic substitution reaction.
- inductive donors e.g., 2c and 2j
- Arenes and heteroarenes more electron-poor than those shown in Table 1 may show diminished reactivity and give lower yields.
- the sulfonyl protecting groups of the N-aryl or N-heteroaryl sulfonimides can be removed using methods of the invention to provide aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof).
- aryl or heteroaryl amines e.g., compounds of Formula (II), and salts thereof.
- treatment of compound 2b with magnesium in methanol under sonication produced aryl amine 3 in 85% yield (Scheme 2).
- Triethylamine (5mL) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with (hexanes/EtOAc 5: 1 (v/v) with 1% triethylamine) to afford 11.5 g of the title compound (83% yield), which is spectroscopically identical to the compound prepared according to the standard procedure (vide supra).
- the intermediate of Formula (G) formally transfers a sulfonimidyl radical to the bound arene substrate, which expels a delocalized radical intermediate and regenerates complex 1.
- the delocalized radical intermediate is subsequently oxidized by the Ag(II) intermediate, then deprotonated to furnish the sulfonimidated product.
- the rate of the non-productive catalytic reduction of NFBS is substantially identical to the rate of the productive imidation reaction (Scheme 3).
- the rate law and the substantially identical rate for both reactions shown in Scheme 3 are consistent with a turnover-limiting oxidation of complex 1 by NFBS to yield a common, short-lived, high-valent palladium intermediate of Formula (F).
- Oxidation of a transition metal complex of the invention typically requires strongly ⁇ -donating anionic ligands such as hydrocarbyl ligands, which are absent from complex l.
- anionic ligands such as hydrocarbyl ligands, which are absent from complex l.
- 8 DFT (density functional theory) calculations reveal that the highest occupied molecular orbital (HOMO) of complex 1 is an extended M-L ⁇ -antibonding orbital of d xz parentage from palladium, instead of the J z 2 -based orbital more typical of square-planar d complexes.
- a single-electron reductant e.g. , an Ag(I) salt
- G intermediate of Formula (G)
- EPR electron paramagnetic resonance
- KIE indicates a rehybridization of the C-H bond from sp 2 to sp 3 in the product-determining transition state, consistent with a C-N bond formation via inner-sphere addition of a dibenzenesulfonimidyl radical to the bound arene in an intermediate of Formula (G).
- Electrophilic addition would also be consistent with the observed intramolecular isotope effect.
- inverse secondary KIE values for electrophilic substitution are rare; 11 commonly, KIEs close to unity are observed for electrophilic processes due to the opposing effects of rehybridization and hyperconjugation on the zero-point vibrational energy of the affected C-H bond.
- 12 Inner-sphere attack from an intermediate, such as an intermediate of Formula (G), to form the putative aryl radical is supported by an intermolecular competition KIE 1.03 + 0.02. The absence of a KIE in this case is consistent with irreversible substrate bindin prior to C-N bond formation. 9 ' 1013
- Fluorobenzene was chosen as the substrate for the determination of the rate law of the catalytic imidation reaction because fluorobenzene is a competent substrate for the imidation reaction (81 yield), and the consumption of fluorobenzene and appearance of the products could be followed concurrently with the consumption of NFBS by 19 F NMR.
- the data was acquired at 50 °C because this temperature was found to facilitate a more convenient rate of reaction for kinetic analysis. To justify the acquisition of kinetic data with fluorobenzene as a substrate at a reaction temperature of 50 °C, the catalytic imidation on fluorobenzene under these conditions has been performed , and no substantial diminution in yield has been found.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
The present invention provides novel transition metal complexes (e.g., complexes of any one of Formulae (C1) to (C25)) that include an amine-N-oxide motif. The invention also provides methods of using these transition metal complexes in preparing N-aryl or N- heteroaryl sulfonimides (e.g., compounds of Formula (I)) and aryl or heteroaryl amines (e.g., compounds of Formula (II)). The inventive methods involve imidation of arenes and heteroarenes (e.g., compounds of Formula (A)) using an imidating agent (e.g., a compound of Formula (B), such as N-fluorobenzenesulfonimide (NFBS or NFSI)) in the presence of a single-electron reductant (e.g., an Ag(I) or Ru(II) salt).
Description
TRANSITION METAL-CATALYZED IMIDATION OF ARENES
RELATED APPLICATIONS
[0001] The present application claims priority under 35 U.S.C. § 119(e) to U.S.
provisional patent application, U.S. S.N. 61/872,466, filed August 30, 2013, which is incorporated herein by reference.
GOVERNMENT SUPPORT
[0002] This invention was made with U.S. Government support under grant number GM088237 awarded by the National Institutes of Health (NIH) and the National Institute of General Medical Sciences (NIGMS) and grant numbers CHE-0952753 and DGE0644491 awarded by the National Science Foundation (NSF). The U.S. Government has certain rights in the invention.
BACKGROUND OF THE INVENTION
[0003] Transition metal catalysis holds promise as a strategy for the direct
functionalization of otherwise unreactive C-H bonds. Many reported transition metal catalyzed aryl C-H functionalization approaches rely on metallation of the arene followed by functionalization of the aryl-metal σ-bond.1 To accelerate an otherwise sluggish metallation step, coordinating directing groups are commonly employed to accelerate carbon-metal bond formation; without such directing groups, often multiple equivalents or even solvent quantities of arene are required to drive the metallation. 2 With few exceptions, 3 catalytic C-H functionalization without the aid of directing groups remains an unmet challenge.
[0004] Historically, an important method of introducing an amino group into arenes has been electrophilic aromatic nitration followed by reduction of the nitro group.4 However, nitration of arenes typically requires strongly acidic or oxidizing reaction conditions, which limits its use on substrates with sensitive functional groups. Alternatively, several aniline derivatives can be reliably prepared by transition-metal catalyzed directed ortho C-H amidation,5 but the requirement for a coordinating directing group limits the potential substrate scope. Non-chelation-assisted transition metal catalyzed C-H amination reactions,6 as well as metal-free approaches involving the use of an oxidant and amine to introduce the C-N bond, have also been reported; however, these methods require a minimum of 1.5 equivalents, and not uncommonly, solvent quantities of arene substrate, and yields are often
based on the amidating reagent, not the arene. Therefore, there remains a need for a facile method of introducing amino or amino functionalized groups into arenes.
SUMMARY OF THE INVENTION
[0005] The present invention provides novel transition metal complexes (e.g., complexes of any one of Formulae (CI) to (C25)) that include an amine-N-oxide motif. The invention also provides methods of using the inventive transition metal complexes in preparing N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I)) and aryl or heteroaryl amines (e.g., compounds of Formula (II)). The inventive methods involve imidation of arenes and heteroarenes (e.g., compounds of Formula (A)) using an imidating agent (e.g. , a compound of Formula (B), such as N-fluorobenzenesulfonimide (NFBS or NFSI)) in the presence of a single-electron reductant (e.g., an Ag(I) or Ru(II) salt).
(C3) (C4)
(C17) (C18)
(C19)
(C24)
(C25)
[0006] In one aspect, the present invention provides methods for preparing an N-aryl or N-heteroaryl sulfonimide (e.g. , a compound of Formula (I), or a salt thereof). The method typically involves contacting a compound of Formula (A), or a salt thereof, with a compound of Formula (B), or a salt thereof, in the presence of a transition metal complex of any one of Formula (CI) to (C25) and a single-electron reductant to provide the compound of Formula (I), or a salt thereof. In certain embodiments, the compound of Formula (B) is N- fluorobenzenesulfonimide (NFBS or NFSI). In certain embodiments, the transition metal
complex is of Formula (CI). In certain embodiments, the transition metal complex is of Formula (C2). In certain embodiments, the transition metal complex is of Formula (C3). In certain embodiments, the transition metal complex is of Formula (C4). In certain
embodiments, the transition metal complex is of any one of Formulae (C5) to (C25). In certain embodiments, the single-electron reductant is an Ag(I) or Ru(II) salt.
[0007] In certain embodiments, a transition metal complex of Formula (CI) for use in the imidation reactions described herein is prepared by contacting a ligand of Formula (Dl), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of Formula (CI):
(Dl) (E).
[0008] In certain embodiments, a transition metal complex of Formula (C2) is prepared by contacting a ligand of Formula (D2), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of Formula (C2):
[0009] In certain embodiments, the inventive methods of further include the step of deprotecting a compound of Formula (I), or a salt thereof, with a reductant (e.g. , a mixture of an alkaline earth metal and an alcohol; or a mixture of zinc and an acid), a strong acid, or a nucleophile (e.g. , an alkyl lithium, phenyl lithium, or a Grignard reagent), to provide a compound of Formula (II), or a salt thereof.
[0010] In another aspect, the present invention provides complexes of any one of Formulae (CI) to (C25). In certain embodiments, the complex of the invention is of Formula (CI). In certain embodiments, the complex of the invention is of Formula (C2). In certain embodiments, the complex of the invention is of Formula (C3). In certain embodiments, the complex of the invention is of Formula (C4). In certain embodiments, the complex of the invention is of any one of Formulae (C5) to (C25).
[0011] In another aspect, the present invention provides kits. In certain embodiments, the kits are useful for preparing the compounds (e.g. , N-aryl or N-heteroaryl sulfonimides and aryl or heteroaryl amines) described herein. In certain embodiments, the kits are useful for preparing compounds of Formula (I), and salts thereof. In certain embodiments, the kits are
useful for preparing compounds of Formula (II), and salts thereof. In certain embodiments, a kit of the invention includes a compound of Formula (B) (e.g., N-fluorobenzenesulfonimide (NFBS or NFSI)), or a salt thereof; a transition metal complex of any one of Formulae (Cl)- (C25); and a single-electron reductant (e.g., an Au(I) or Ru(II) salt). In certain embodiments, a kit further includes an arene or heteroarene described herein (e.g., a compound of Formula (A), or a salt thereof). In certain embodiments, a kit further includes a deprotecting agent (e.g., a reductant, a strong acid, or a nucleophile). A kit of the invention may also include a container, solvent, organic or inorganic agent, and/or solid support, as described herein. A kit of the invention may further include instructions for using the kit.
DEFINITIONS
[0012] Definitions of specific functional groups and chemical terms are described in more detail below. The chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 75th Ed., inside cover, and specific functional groups are generally defined as described therein. Additionally, general principles of organic chemistry, as well as specific functional moieties and reactivity, are described in Thomas Sorrell, Organic Chemistry, University Science Books, Sausalito, 1999; Smith and March, March's Advanced Organic Chemistry, 5th Edition, John Wiley & Sons, Inc., New York, 2001; Larock, Comprehensive Organic Transformations, VCH
Publishers, Inc., New York, 1989; and Carruthers, Some Modern Methods of Organic
Synthesis, 3 rd Edition, Cambridge University Press, Cambridge, 1987.
[0013] Compounds described herein can comprise one or more asymmetric centers, and thus can exist in various isomeric forms, e.g., enantiomers and/or diastereomers. For example, the compounds described herein can be in the form of an individual enantiomer, diastereomer or geometric isomer, or can be in the form of a mixture of stereoisomers, including racemic mixtures and mixtures enriched in one or more stereoisomer. Isomers can be isolated from mixtures by methods known to those skilled in the art, including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts; or preferred isomers can be prepared by asymmetric syntheses. See, for example, Jacques et ah, Enantiomers, Racemates and Resolutions (Wiley Interscience, New York, 1981); Wilen et ah, Tetrahedron 33:2725 (1977); Eliel, Stereochemistry of Carbon Compounds (McGraw- Hill, NY, 1962); and Wilen, Tables of Resolving Agents and Optical Resolutions p. 268 (E.L. Eliel, Ed., Univ. of Notre Dame Press, Notre Dame, IN 1972). The invention additionally
encompasses compounds described herein as individual isomers substantially free of other isomers, and alternatively, as mixtures of various isomers.
[0014] When a range of values is listed, it is intended to encompass each value and subrange within the range. For example "C^" is intended to encompass, C1 ; C2, C3, C4, C5, C6, Ci_6, Ci-5, Ci^, Ci-3, Ci-2, C2-6, C2_5, C2_4, C2_3, C3_6, C3_5, C3^, C4_6, C4_5, and C5_6.
[0015] The term "aliphatic" includes both saturated and unsaturated, straight chain (i.e. , unbranched), branched, acyclic, cyclic, or polycyclic aliphatic hydrocarbons, which are optionally substituted with one or more functional groups. As will be appreciated by one of ordinary skill in the art, "aliphatic" is intended herein to include, but is not limited to, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, and cycloalkynyl moieties. Thus, the term "alkyl" includes straight, branched and cyclic alkyl groups. An analogous convention applies to other generic terms such as "alkenyl", "alkynyl", and the like. Furthermore, the terms "alkyl", "alkenyl", "alkynyl", and the like encompass both substituted and unsubstituted groups. In certain embodiments, "lower alkyl" is used to indicate those alkyl groups (cyclic, acyclic, substituted, unsubstituted, branched or unbranched) having 1-6 carbon atoms.
[0016] In certain embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-20 aliphatic carbon atoms. In certain other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1- 10 aliphatic carbon atoms. In yet other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-8 aliphatic carbon atoms. In still other embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-6 aliphatic carbon atoms. In yet other
embodiments, the alkyl, alkenyl, and alkynyl groups employed in the invention contain 1-4 carbon atoms. Illustrative aliphatic groups thus include, but are not limited to, for example, methyl, ethyl, n-propyl, isopropyl, cyclopropyl, -CH2-cyclopropyl, vinyl, allyl, n-butyl, sec- butyl, isobutyl, tert-butyl, cyclobutyl, -CH2-cyclobutyl, n-pentyl, sec-pentyl, isopentyl, tert- pentyl, cyclopentyl, -CH2-cyclopentyl, n-hexyl, sec-hexyl, cyclohexyl, -CH2-cyclohexyl moieties and the like, which again, may bear one or more substituents. Alkenyl groups include, but are not limited to, for example, ethenyl, propenyl, butenyl, l-methyl-2-buten- l- yl, and the like. Representative alkynyl groups include, but are not limited to, ethynyl, 2- propynyl (propargyl), 1-propynyl, and the like.
[0017] The term "alkyl" refers to a radical of a straight-chain or branched saturated hydrocarbon group having from 1 to 10 carbon atoms ("Ci-w alkyl"). In some embodiments, an alkyl group has 1 to 9 carbon atoms ("Q-9 alkyl"). In some embodiments, an alkyl group has 1 to 8 carbon atoms ("Q-e alkyl"). In some embodiments, an alkyl group has 1 to 7
carbon atoms ("C^ alkyl"). In some embodiments, an alkyl group has 1 to 6 carbon atoms ("Ci_6 alkyl"). In some embodiments, an alkyl group has 1 to 5 carbon atoms ("Q-s alkyl"). In some embodiments, an alkyl group has 1 to 4 carbon atoms ("Ci^ alkyl"). In some embodiments, an alkyl group has 1 to 3 carbon atoms ("C^ alkyl"). In some embodiments, an alkyl group has 1 to 2 carbon atoms ("C^ alkyl"). In some embodiments, an alkyl group has 1 carbon atom ("Q alkyl"). In some embodiments, an alkyl group has 2 to 6 carbon atoms ("C2-6 alkyl"). Examples of Q-6 alkyl groups include methyl (CO, ethyl (C2), n-propyl (C3), isopropyl (C3), n-butyl (C4), tert-butyl (C4), sec-butyl (C4), iso-butyl (C4), n-pentyl (C5), 3-pentanyl (C5), amyl (C5), neopentyl (C5), 3-methyl-2-butanyl (C5), tertiary amyl (C5), and n-hexyl (C6). Additional examples of alkyl groups include n-heptyl (C7), n-octyl (C8), and the like. Unless otherwise specified, each instance of an alkyl group is
independently unsubstituted (an "unsubstituted alkyl") or substituted (a "substituted alkyl") with one or more substituents. In certain embodiments, the alkyl group is an unsubstituted Q_ 10 alkyl (e.g., -CH3). In certain embodiments, the alkyl group is a substituted C^o alkyl.
[0018] The term "haloalkyl" is a substituted alkyl group, wherein one or more of the hydrogen atoms are independently replaced by a halogen, e.g., fluoro, bromo, chloro, or iodo. "Perhaloalkyl" is a subset of haloalkyl, and refers to an alkyl group wherein all of the hydrogen atoms are independently replaced by a halogen, e.g., fluoro, bromo, chloro, or iodo. In some embodiments, the haloalkyl moiety has 1 to 8 carbon atoms ("Q-s haloalkyl"). In some embodiments, the haloalkyl moiety has 1 to 6 carbon atoms ("C^ haloalkyl"). In some embodiments, the haloalkyl moiety has 1 to 4 carbon atoms ("C^ haloalkyl"). In some embodiments, the haloalkyl moiety has 1 to 3 carbon atoms ("Ci_3 haloalkyl"). In some embodiments, the haloalkyl moiety has 1 to 2 carbon atoms ("C^ haloalkyl"). In some embodiments, all of the haloalkyl hydrogen atoms are replaced with fluoro to provide a perfluoroalkyl group. In some embodiments, all of the haloalkyl hydrogen atoms are replaced with chloro to provide a "perchloroalkyl" group. Examples of haloalkyl groups include -CF , -CF2CF3, -CF2CF2CF3, -CCI3, -CFC12, -CF2C1, and the like.
[0019] "Alkenyl" refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon double bonds, and no triple bonds ("C2-20 alkenyl"). In some embodiments, an alkenyl group has 2 to 10 carbon atoms ("C2-io alkenyl"). In some embodiments, an alkenyl group has 2 to 9 carbon atoms ("C2-9 alkenyl"). In some embodiments, an alkenyl group has 2 to 8 carbon atoms ("C2-8 alkenyl"). In some embodiments, an alkenyl group has 2 to 7 carbon atoms ("C2_7 alkenyl"). In some embodiments, an alkenyl group has 2 to 6 carbon atoms ("C2-6 alkenyl"). In some
embodiments, an alkenyl group has 2 to 5 carbon atoms ("C2_5 alkenyl"). In some embodiments, an alkenyl group has 2 to 4 carbon atoms ("C2^ alkenyl"). In some
embodiments, an alkenyl group has 2 to 3 carbon atoms ("C2_3 alkenyl"). In some
embodiments, an alkenyl group has 2 carbon atoms ("C2 alkenyl"). The one or more carbon- carbon double bonds can be internal (such as in 2-butenyl) or terminal (such as in 1-butenyl). Examples of C2^ alkenyl groups include ethenyl (C2), 1-propenyl (C3), 2-propenyl (C3), 1- butenyl (C4), 2-butenyl (C4), butadienyl (C4), and the like. Examples of C2-6 alkenyl groups include the aforementioned C2^ alkenyl groups as well as pentenyl (C5), pentadienyl (C5), hexenyl (C6), and the like. Additional examples of alkenyl include heptenyl (C7), octenyl (Cg), octatrienyl (Cg), and the like. Unless otherwise specified, each instance of an alkenyl group is independently optionally substituted, i.e. , unsubstituted (an "unsubstituted alkenyl") or substituted (a "substituted alkenyl") with one or more substituents. In certain
embodiments, the alkenyl group is unsubstituted C2_10 alkenyl. In certain embodiments, the alkenyl group is substituted C2_10 alkenyl. In an alkenyl group, a C=C double bond for which the stereochemistry is not specified (e.g. , -CH=CHCH3 or ^^^ ) may be an or (z)- double bond.
[0020] "Alkynyl" refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon-carbon triple bonds, and optionally one or more double bonds ("C2-2o alkynyl"). In some embodiments, an alkynyl group has 2 to 10 carbon atoms ("C2-io alkynyl"). In some embodiments, an alkynyl group has 2 to 9 carbon atoms ("C2_9 alkynyl"). In some embodiments, an alkynyl group has 2 to 8 carbon atoms ("C2_8 alkynyl"). In some embodiments, an alkynyl group has 2 to 7 carbon atoms ("C2_7 alkynyl"). In some embodiments, an alkynyl group has 2 to 6 carbon atoms ("C2-6 alkynyl"). In some embodiments, an alkynyl group has 2 to 5 carbon atoms ("C2_5 alkynyl"). In some embodiments, an alkynyl group has 2 to 4 carbon atoms ("C2^ alkynyl"). In some
embodiments, an alkynyl group has 2 to 3 carbon atoms ("C2_3 alkynyl"). In some
embodiments, an alkynyl group has 2 carbon atoms ("C2 alkynyl"). The one or more carbon- carbon triple bonds can be internal (such as in 2-butynyl) or terminal (such as in 1-butynyl). Examples of C2- alkynyl groups include, without limitation, ethynyl (C2), 1-propynyl (C3), 2-propynyl (C3), 1-butynyl (C4), 2-butynyl (C4), and the like. Examples of C2-6 alkenyl groups include the aforementioned C2- alkynyl groups as well as pentynyl (C5), hexynyl (C6), and the like. Additional examples of alkynyl include heptynyl (C7), octynyl (Cg), and the like. Unless otherwise specified, each instance of an alkynyl group is independently
optionally substituted, i.e., unsubstituted (an "unsubstituted alkynyl") or substituted (a "substituted alkynyl") with one or more substituents. In certain embodiments, the alkynyl group is unsubstituted C2_10 alkynyl. In certain embodiments, the alkynyl group is substituted C2-10 alkynyl.
[0021] "Carbocyclyl" or "carbocyclic" refers to a radical of a non-aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (" -io carbocyclyl") and zero heteroatoms in the non-aromatic ring system. In some embodiments, a carbocyclyl group has 3 to 8 ring carbon atoms ("C3_8 carbocyclyl"). In some embodiments, a carbocyclyl group has 3 to 6 ring carbon atoms ("C3_6 carbocyclyl"). In some embodiments, a carbocyclyl group has 3 to 6 ring carbon atoms ("C3_6 carbocyclyl"). In some embodiments, a carbocyclyl group has 5 to 10 ring carbon atoms ("Cs-io carbocyclyl"). Exemplary C3_6 carbocyclyl groups include, without limitation, cyclopropyl (C3), cyclopropenyl (C3), cyclobutyl (C4), cyclobutenyl (C4), cyclopentyl (C5), cyclopentenyl (C5), cyclohexyl (C6), cyclohexenyl (C6), cyclohexadienyl (C6), and the like. Exemplary C3_8 carbocyclyl groups include, without limitation, the aforementioned C3_6 carbocyclyl groups as well as cycloheptyl (C7), cycloheptenyl (C7), cycloheptadienyl (C7), cycloheptatrienyl (C7), cyclooctyl (C8), cyclooctenyl (C8),
bicyclo[2.2.1]heptanyl (C7), bicyclo[2.2.2]octanyl (C8), and the like. Exemplary C3_io carbocyclyl groups include, without limitation, the aforementioned C3_8 carbocyclyl groups as well as cyclononyl (C9), cyclononenyl (C9), cyclodecyl (C10), cyclodecenyl (C10), octahydro-lH-indenyl (C9), decahydronaphthalenyl (C10), spiro[4.5]decanyl (C10), and the like. As the foregoing examples illustrate, in certain embodiments, the carbocyclyl group is either monocyclic ("monocyclic carbocyclyl") or contain a fused, bridged or spiro ring system such as a bicyclic system ("bicyclic carbocyclyl") and can be saturated or can be partially unsaturated. "Carbocyclyl" also includes ring systems wherein the carbocyclic ring, as defined above, is fused with one or more aryl or heteroaryl groups wherein the point of attachment is on the carbocyclic ring, and in such instances, the number of carbons continue to designate the number of carbons in the carbocyclic ring system. Unless otherwise specified, each instance of a carbocyclyl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted carbocyclyl") or substituted (a "substituted carbocyclyl") with one or more substituents. In certain embodiments, the carbocyclyl group is unsubstituted C3_io carbocyclyl. In certain embodiments, the carbocyclyl group is substituted C3_10 carbocyclyl.
[0022] In some embodiments, "carbocyclyl" is a monocyclic, saturated carbocyclyl group having from 3 to 10 ring carbon atoms ("C3_io cycloalkyl"). In some embodiments, a
cycloalkyl group has 3 to 8 ring carbon atoms ("C3_8 cycloalkyl"). In some embodiments, a cycloalkyl group has 3 to 6 ring carbon atoms ("C3_6 cycloalkyl"). In some embodiments, a cycloalkyl group has 5 to 6 ring carbon atoms ("Cs_6 cycloalkyl"). In some embodiments, a cycloalkyl group has 5 to 10 ring carbon atoms ("Cs-io cycloalkyl"). Examples of C5_6 cycloalkyl groups include cyclopentyl (C5) and cyclohexyl (C5). Examples of C3_6 cycloalkyl groups include the aforementioned C5_6 cycloalkyl groups as well as cyclopropyl (C3) and cyclobutyl (C4). Examples of C3_8 cycloalkyl groups include the aforementioned C3_6 cycloalkyl groups as well as cycloheptyl (C7) and cyclooctyl (C8). Unless otherwise specified, each instance of a cycloalkyl group is independently unsubstituted (an "unsubstituted cycloalkyl") or substituted (a "substituted cycloalkyl") with one or more substituents. In certain embodiments, the cycloalkyl group is unsubstituted C3_10 cycloalkyl. In certain embodiments, the cycloalkyl group is substituted C3_10 cycloalkyl.
[0023] "Heterocyclyl" or "heterocyclic" refers to a radical of a 3- to 10-membered non- aromatic ring system having ring carbon atoms and 1 to 4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon ("3-10 membered heterocyclyl"). In heterocyclyl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. A heterocyclyl group can either be monocyclic ("monocyclic heterocyclyl") or a fused, bridged, or spiro ring system, such as a bicyclic system ("bicyclic heterocyclyl"), and can be saturated or can be partially unsaturated. Heterocyclyl bicyclic ring systems can include one or more heteroatoms in one or both rings. "Heterocyclyl" also includes ring systems wherein the heterocyclic ring, as defined above, is fused with one or more carbocyclyl groups wherein the point of attachment is either on the carbocyclyl or heterocyclic ring, or ring systems wherein the heterocyclic ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclic ring, and in such instances, the number of ring members continue to designate the number of ring members in the
heterocyclic ring system. Unless otherwise specified, each instance of heterocyclyl is independently optionally substituted, i.e., unsubstituted (an "unsubstituted heterocyclyl") or substituted (a "substituted heterocyclyl") with one or more substituents. In certain
embodiments, the heterocyclyl group is unsubstituted 3-10 membered heterocyclyl. In certain embodiments, the heterocyclyl group is substituted 3-10 membered heterocyclyl.
[0024] In some embodiments, a heterocyclyl group is a 5-10 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon ("5-10
membered heterocyclyl"). In some embodiments, a heterocyclyl group is a 5-8 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-8 membered heterocyclyl"). In some embodiments, a heterocyclyl group is a 5-6 membered non-aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-6 membered heterocyclyl"). In some embodiments, the 5-6 membered heterocyclyl has 1-3 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5-6 membered heterocyclyl has 1-2 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5-6 membered heterocyclyl has one ring heteroatom selected from nitrogen, oxygen, and sulfur.
[0025] Exemplary 3-membered heterocyclyl groups containing one heteroatom include, without limitation, azirdinyl, oxiranyl, and thiiranyl. Exemplary 4-membered heterocyclyl groups containing one heteroatom include, without limitation, azetidinyl, oxetanyl, and thietanyl. Exemplary 5-membered heterocyclyl groups containing one heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl,
dihydrothiophenyl, pyrrolidinyl, dihydropyrrolyl, and pyrrolyl-2,5-dione. Exemplary 5- membered heterocyclyl groups containing two heteroatoms include, without limitation, dioxolanyl, oxasulfuranyl, disulfuranyl, and oxazolidin-2-one. Exemplary 5-membered heterocyclyl groups containing three heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl. Exemplary 6-membered heterocyclyl groups containing one heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl. Exemplary 6-membered heterocyclyl groups containing two heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, and dioxanyl. Exemplary 6- membered heterocyclyl groups containing three heteroatoms include, without limitation, triazinanyl. Exemplary 7-membered heterocyclyl groups containing one heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl. Exemplary 8-membered heterocyclyl groups containing one heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl. Exemplary 5-membered heterocyclyl groups fused to a C6 aryl ring (also referred to herein as a 5,6-bicyclic heterocyclic ring) include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, benzoxazolinonyl, and the like. Exemplary 6-membered heterocyclyl groups fused to an aryl ring (also referred to herein as a 6,6-bicyclic heterocyclic ring) include, without limitation, tetrahydroquinolinyl,
tetrahydroisoquinolinyl, and the like.
[0026] "Aryl" refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g. , having 6, 10, or 14 pi electrons shared in a cyclic array) having 6-14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system ("C6-14 aryl"). In some embodiments, an aryl group has six ring carbon atoms ("C6 aryl"; e.g., phenyl). In some embodiments, an aryl group has ten ring carbon atoms ("Cio aryl"; e.g., naphthyl such as 1-naphthyl and 2-naphthyl). In some embodiments, an aryl group has fourteen ring carbon atoms ("Cw aryl"; e.g. , anthracyl). "Aryl" also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system. Unless otherwise specified, each instance of an aryl group is independently optionally substituted, i.e. , unsubstituted (an "unsubstituted aryl") or substituted (a
"substituted aryl") with one or more substituents. In certain embodiments, the aryl group is unsubstituted C6-i4 aryl. In certain embodiments, the aryl group is substituted C6-i4 aryl.
[0027] "Aralkyl" is a subset of alkyl and aryl and refers to an optionally substituted alkyl group substituted by an optionally substituted aryl group. In certain embodiments, the aralkyl is optionally substituted benzyl. In certain embodiments, the aralkyl is benzyl. In certain embodiments, the aralkyl is optionally substituted phenethyl. In certain embodiments, the aralkyl is phenethyl.
[0028] "Heteroaryl" refers to a radical of a 5-10 membered monocyclic or bicyclic 4n+2 aromatic ring system (e.g., having 6 or 10 pi electrons shared in a cyclic array) having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-10 membered heteroaryl"). In heteroaryl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. Heteroaryl bicyclic ring systems can include one or more heteroatoms in one or both rings. "Heteroaryl" includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the point of attachment is on the heteroaryl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heteroaryl ring system. "Heteroaryl" also includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more aryl groups wherein the point of attachment is either on the aryl or heteroaryl ring, and in such instances, the number of ring members designates the number of ring members in the fused (aryl/heteroaryl) ring system. Bicyclic heteroaryl groups wherein one ring does not contain a heteroatom (e.g. , indolyl, quinolinyl,
carbazolyl, and the like) the point of attachment can be on either ring, i.e., either the ring bearing a heteroatom (e.g., 2-indolyl) or the ring that does not contain a heteroatom (e.g., 5- indolyl).
[0029] In some embodiments, a heteroaryl group is a 5-10 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-10 membered heteroaryl"). In some embodiments, a heteroaryl group is a 5-8 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-8 membered heteroaryl"). In some embodiments, a heteroaryl group is a 5-6 membered aromatic ring system having ring carbon atoms and 1-4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur ("5-6 membered heteroaryl"). In some embodiments, the 5-6 membered heteroaryl has 1-3 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5-6 membered heteroaryl has 1-2 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5-6 membered heteroaryl has 1 ring heteroatom selected from nitrogen, oxygen, and sulfur. Unless otherwise specified, each instance of a heteroaryl group is independently optionally substituted, i.e., unsubstituted (an "unsubstituted heteroaryl") or substituted (a "substituted heteroaryl") with one or more substituents. In certain embodiments, the heteroaryl group is unsubstituted 5-14 membered heteroaryl. In certain embodiments, the heteroaryl group is substituted 5-14 membered heteroaryl.
[0030] Exemplary 5-membered heteroaryl groups containing one heteroatom include, without limitation, pyrrolyl, furanyl, and thiophenyl. Exemplary 5-membered heteroaryl groups containing two heteroatoms include, without limitation, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, and isothiazolyl. Exemplary 5-membered heteroaryl groups containing three heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl. Exemplary 5-membered heteroaryl groups containing four heteroatoms include, without limitation, tetrazolyl. Exemplary 6-membered heteroaryl groups containing one heteroatom include, without limitation, pyridinyl. Exemplary 6-membered heteroaryl groups containing two heteroatoms include, without limitation, pyridazinyl, pyrimidinyl, and pyrazinyl. Exemplary 6-membered heteroaryl groups containing three or four heteroatoms include, without limitation, triazinyl and tetrazinyl, respectively. Exemplary 7-membered heteroaryl groups containing one heteroatom include, without limitation, azepinyl, oxepinyl,
and thiepinyl. Exemplary 5,6-bicyclic heteroaryl groups include, without limitation, indolyl, isoindolyl, indazolyl, benzotriazolyl, benzothiophenyl, isobenzothiophenyl, benzofuranyl, benzoisofuranyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzoxadiazolyl, benzthiazolyl, benzisothiazolyl, benzthiadiazolyl, indolizinyl, and purinyl. Exemplary 6,6- bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl.
[0031] "Heteroaralkyl" is a subset of alkyl and heteroaryl and refers to an optionally substituted alkyl group substituted by an optionally substituted heteroaryl group.
[0032] "Unsaturated" or "partially unsaturated" refers to a group that includes at least one double or triple bond. A "partially unsaturated" ring system is further intended to encompass rings having multiple sites of unsaturation, but is not intended to include aromatic groups
(e.g., aryl or heteroaryl groups) as herein defined. Likewise, "saturated" refers to a group that does not contain a double or triple bond, i.e., contains all single bonds.
[0033] Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, which are divalent bridging groups, are further referred to using the suffix -ene, e.g., alkylene, alkenylene, alkynylene, carbocyclylene, heterocyclylene, arylene, and
heteroarylene.
[0034] An atom, moiety, or group described herein may be unsubstituted or substituted, as valency permits, unless otherwise provided expressly. The term "optionally substituted" refers to substituted or unsubstituted.
[0035] Alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups are optionally substituted (e.g., "substituted" or "unsubstituted" alkyl, "substituted" or
"unsubstituted" alkenyl, "substituted" or "unsubstituted" alkynyl, "substituted" or
"unsubstituted" carbocyclyl, "substituted" or "unsubstituted" heterocyclyl, "substituted" or "unsubstituted" aryl or "substituted" or "unsubstituted" heteroaryl group). In general, the term "substituted", whether preceded by the term "optionally" or not, means that at least one hydrogen present on a group (e.g. , a carbon or nitrogen atom) is replaced with a permissible substituent, e.g. , a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction. Unless otherwise indicated, a "substituted" group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position. The term "substituted" is contemplated to include substitution with all permissible substituents of organic compounds, any of the substituents described herein that
results in the formation of a stable compound. The present invention contemplates any and all such combinations in order to arrive at a stable compound. For purposes of this invention, heteroatoms such as nitrogen may have hydrogen substituents and/or any suitable substituent as described herein which satisfy the valencies of the heteroatoms and results in the formation of a stable moiety. In certain embodiments, the substituent is a carbon atom substituent. In certain embodiments, the substituent is a nitrogen atom substituent. In certain embodiments, the substituent is an oxygen atom substituent. In certain embodiments, the substituent is a sulfur atom substituent.
[0036] Exemplary carbon atom substituents include, but are not limited to, halogen, -CN, -NO2, -N3, -SO2H, -S03H, -OH, -ORaa, -ON(Rbb)2, -N(Rbb)2, -N(Rbb)3 +X , -N(ORcc)Rbb, - SH, -SRaa, -SSRCC, -C(=0)Raa, -C02H, -CHO, -C(ORcc)2, -C02Raa, -OC(=0)Raa, - OCOaR^, -C(=0)N(Rbb)2, -OC(=0)N(Rbb)2, -NRbbC(=0)Raa, -NRbbC02Raa, - NRbbC(=0)N(Rbb)2, -C(=NRbb)Raa, -C(=NRbb)ORaa, -OC(=NRbb)Raa, -OC(=NRbb)ORaa, - C(=NRbb)N(Rbb)2, -OC(=NRbb)N(Rbb)2, -NRbbC(=NRbb)N(Rbb)2, -C(=0)NRbbS02Raa, - NRbbS02Raa, -S02N(Rbb)2, -S02Raa, -S02ORaa, -OS02Raa, -S(=0)Raa, -OS(=0)Raa, - Si(Raa)3, -OSi(Raa)3 -C(=S)N(Rbb)2, -C(=0)SRaa, -C(=S)SRaa, -SC(=S)SRaa, -SC(=0)SRaa, -OC(=0)SRaa, -SC(=0)ORaa, -SC(=0)Raa, -P(=0)2Raa, -OP(=0)2Raa, -P(=0)(Raa)2, - OP(=0)(Raa)2, -OP(=0)(ORcc)2, -P(=0)2N(Rbb)2, -OP(=0)2N(Rbb)2, -P(=0)(NRbb)2, - OP(=0)(NRbb)2, -NRbbP(=0)(ORcc)2, -NRbbP(=0)(NRbb)2, -P(RCC)2, -P(RCC)3, -OP(Rcc)2, - OP(Rcc)3, -B(Raa)2, -B(ORcc)2, -BRaa(ORcc), Ci_i0 alkyl, Ci_i0 perhaloalkyl, C2_10 alkenyl, C2_io alkynyl, C3_io carbocyclyl, 3-14 membered heterocyclyl, C6-i4 aryl, and 5-14 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups; or two geminal hydrogens on a carbon atom are replaced with the group =0, =S, =NN(Rbb)2,
=NNRbbC(=0)Raa, =NNRbbC(=0)ORaa, =NNRbbS(=0)2Raa, =NRbb, or =NORcc;
each instance of Raa is, independently, selected from C^o alkyl, C^o perhaloalkyl, C2_10 alkenyl, C2_10 alkynyl, C3_10 carbocyclyl, 3-14 membered heterocyclyl, C6-i4 aryl, and 5-14 membered heteroaryl, or two Raa groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups;
each instance of Rbb is, independently, selected from hydrogen, -OH, -ORaa, - N(RCC)2, -CN, -C(=0)Raa, -C(=0)N(Rcc)2, -C02Raa, -S02Raa, -C(=NRcc)ORaa, - C(=NRCC)N(RCC)2, -S02N(Rcc)2, -S02Rcc, -S02ORcc, -SORaa, -C(=S)N(RCC)2, -C(=0)SRcc, -
C(=S)SRCC, -P(=0)2Raa, -P(=0)(Raa)2, -P(=0)2N(Rcc)2, -P(=0)(NRcc)2, C 10 alkyl, C 10 perhaloalkyl, C2_io alkenyl, C2_io alkynyl, C3_io carbocyclyl, 3-14 membered heterocyclyl, C6-i4 aryl, and 5-14 membered heteroaryl, or two Rbb groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups;
each instance of Rcc is, independently, selected from hydrogen, Ci-io alkyl, Q_ io perhaloalkyl, C2_10 alkenyl, C2_10 alkynyl, C3_10 carbocyclyl, 3-14 membered heterocyclyl, C6-i4 aryl, and 5-14 membered heteroaryl, or two Rcc groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups;
each instance of Rdd is, independently, selected from halogen, -CN, -N02, - N3, -S02H, -S03H, -OH, -ORee, -ON(Rff)2, -N(Rff)2, -N(Rff)3 +X , -N(ORee)Rff, -SH, - SRee, -SSRee, -C(=0)Ree, -C02H, -C02Ree, -OC(=0)Ree, -OC02Ree, -C(=0)N(Rff)2, - OC(=0)N(Rff)2, -NRffC(=0)Ree, -NRffC02Ree, -NRffC(=0)N(Rff)2, -C(=NRff)ORee, - OC(=NRff)Ree, -OC(=NRff)ORee, -C(=NRff)N(Rff)2, -OC(=NRff)N(Rff)2, - NRffC(=NRff)N(Rff)2,-NRffS02Ree, -S02N(Rff)2, -S02Ree, -S02ORee, -OS02Ree, -S(=0)Ree, -Si(Ree)3, -OSi(Ree)3, -C(=S)N(Rff)2, -C(=0)SRee, -C(=S)SRee, -SC(=S)SRee, -P(=0)2Ree, - P(=0)(Ree)2, -OP(=0)(Ree)2, -OP(=0)(ORee)2, Ci_6 alkyl, Ci_6 perhaloalkyl, C2_6 alkenyl, C2 6 alkynyl, C3_io carbocyclyl, 3-10 membered heterocyclyl, C6-io aryl, 5-10 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups, or two geminal Rdd substituents can be joined to form =0 or =S;
each instance of Ree is, independently, selected from Q_6 alkyl, C^
perhaloalkyl, C2_6 alkenyl, C2_6 alkynyl, C3_io carbocyclyl, C6-io aryl, 3-10 membered heterocyclyl, and 3-10 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups;
ff
each instance of R is, independently, selected from hydrogen, Q_6 alkyl, Q_6 perhaloalkyl, C2_6 alkenyl, C2_6 alkynyl, C3_10 carbocyclyl, 3-10 membered heterocyclyl, C6- ff
io aryl and 5-10 membered heteroaryl, or two R groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl,
carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups; and
each instance of Rgg is, independently, halogen, -CN, -N02, -N3, -S02H, - S03H, -OH, -OCi-6 alkyl, -ON(Ci_6 alkyl)2, -N(Ci_6 alkyl)2, -N(Ci_6 alkyl)3 +X- -NH(Ci_6 alkyl)2 +X~, -NH2(Ci_6 alkyl) +X~, -NH3 +X , -N(OCi_6 alkyl)(Ci_6 alkyl), -N(OH)(Ci_6 alkyl), -NH(OH), -SH, -SCi_6 alkyl, -SS(Ci_e alkyl), -C(=0)(Ci_6 alkyl), -C02H, -C02(Ci_6 alkyl),
alkyl), -OC02(Ci 6 alkyl), -C(=0)NH2, -C(=0)N(Ci_6 alkyl)2, - OC(=0)NH(Ci_6 alkyl), -NHC(=0)( Ci_6 alkyl), -N(Ci_6 alkyl)C(=0)( Ci_6 alkyl), - NHC02(Ci_6 alkyl), -NHC(=0)N(Ci_6 alkyl)2, -NHC(=0)NH(Ci 6 alkyl), -NHC(=0)NH2, - C(=NH)0(Ci_6 alkyl),-OC(=NH)(Ci_6 alkyl), -OC(=NH)OCi 6 alkyl, -C(=NH)N(Ci_6 alkyl)2, -C(=NH)NH(Ci_6 alkyl), -C(=NH)NH2, -OC(=NH)N(Ci_6 alkyl)2, -OC(NH)NH(Ci 6 alkyl), -OC(NH)NH2, -NHC(NH)N(Ci_6 alkyl)2, -NHC(=NH)NH2, -NHS02(Ci 6 alkyl), - S02N(d_6 alkyl)2, -S02NH(C^ alkyl), -S02NH2,-S02Ci_6 alkyl, -S02Od 6 alkyl, - OS02Ci^, alkyl, -SOQ 6 alkyl, -Si(Ci_e alkyl)3, -OSi(Ci_6 alkyl)3 -C(=S)N(C^ alkyl)2, C(=S)NH(Ci_6 alkyl), C(=S)NH2, -C(=0)S(Ci_6 alkyl), -C(=S)SCi_6 alkyl, -SC(=S)SCi_6 alkyl, -P(=0)2(Ci_i alkyl), -P(=0)(Ci_6 alkyl)2, -OP(=0)(Ci_6 alkyl)2, -OP(=0)(OCi_6 alkyl)2, Ci_6 alkyl, C^ perhaloalkyl, C2_6 alkenyl, C2_6 alkynyl, C3_10 carbocyclyl, C6-io aryl, 3-10 membered heterocyclyl, 5-10 membered heteroaryl; or two geminal Rgg substituents can be joined to form =0 or =S; wherein X~ is a counterion.
[0037] "Halo" or "halogen" refers to fluorine (fluoro, -F), chlorine (chloro, -CI), bromine (bromo, -Br), or iodine (iodo, -I).
[0038] "Acyl" refers to a moiety selected from the group consisting of -C(=0)Raa,-CHO, -COaR^, -C(=0)N(Rbb)2, -C(=NRbb)Raa, -C(=NRbb)ORaa, -C(=NRbb)N(Rbb)2, - C(=0)NRbbS02Raa, -C(=S)N(Rbb)2, -C(=0)SRaa, or -C(=S)SRaa, wherein Raa and Rbb are as defined herein.
[0039] Nitrogen atoms can be substituted or unsubstituted as valency permits, and include primary, secondary, tertiary, and quatemarynitrogen atoms. Exemplary nitrogen atom substituents include, but are not limited to, hydrogen, -OH, -ORaa, -N(RCC)2, -CN, - C(=0)Raa, -C(=0)N(Rcc)2, -C02Raa, -S02Raa, -C(=NRbb)Raa, -C(=NRcc)ORaa, - C(=NRCC)N(RCC)2, -S02N(Rcc)2, -S02Rcc, -S02ORcc, -SORaa, -C(=S)N(RCC)2, -C(=0)SRcc, - C(=S)SRCC, -P(=0)2Raa, -P(=0)(Raa)2, -P(=0)2N(Rcc)2, -P(=0)(NRcc)2, Ci_i0 alkyl, Ci_i0 perhaloalkyl, C2_io alkenyl, C2_io alkynyl, C3_io carbocyclyl, 3-14 membered heterocyclyl, C6-i4 aryl, and 5-14 membered heteroaryl, or two Rcc groups attached to a nitrogen atom are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein
each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups, and wherein Raa, Rbb, Rcc, and Rdd are as defined above.
[0040] In certain embodiments, the substituent present on a nitrogen atom is a nitrogen protecting group (also referred to as an amino protecting group). Nitrogen protecting groups include, but are not limited to, -OH, -ORaa, -N(RCC)2, -C(=0)Raa, -C(=0)N(Rcc)2, -CO^, -S02Raa, -C(=NRcc)Raa, -C(=NRcc)ORaa, -C(=NRCC)N(RCC)2, -S02N(Rcc)2, -S02Rcc, - S02ORcc, -SORaa, -C(=S)N(RCC)2, -C(=0)SRcc, -C(=S)SRCC, Ci_i0 alkyl (e.g., aralkyl, heteroaralkyl), C2_io alkenyl, C2_io alkynyl, Ο3_10 carbocyclyl, 3-14 membered heterocyclyl, Ce-M aryl, and 5-14 membered heteroaryl groups, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aralkyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R groups, and wherein Raa, Rbb, Rcc and Rdd are as defined herein. Nitrogen protecting groups are well known in the art and include those described in detail in Protecting
Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3 rd edition, John Wiley & Sons, 1999, incorporated herein by reference.
[0041] For example, nitrogen protecting groups such as amide groups (e.g., -C(=0)Raa) include, but are not limited to, formamide, acetamide, chloroacetamide, trichloroacetamide, trifluoroacetamide, phenylacetamide, 3-phenylpropanamide, picolinamide, 3- pyridylcarboxamide, N-benzoylphenylalanyl derivative, benzamide, p-phenylbenzamide, o- nitophenylacetamide, o-nitrophenoxyacetamide, acetoacetamide, (N - dithiobenzyloxyacylamino)acetamide, 3-(p-hydroxyphenyl)propanamide, 3-(o- nitrophenyl)propanamide, 2-methyl-2-(o-nitrophenoxy)propanamide, 2-methyl-2-(o- phenylazophenoxy)propanamide, 4-chlorobutanamide, 3-methyl-3-nitrobutanamide, o- nitrocinnamide, N-acetylmethionine derivative, o-nitrobenzamide, and o—
(benzoyloxymethyl)benzamide.
[0042] Nitrogen protecting groups such as carbamate groups (e.g., -C(=0)ORaa) include, but are not limited to, methyl carbamate, ethyl carbamante, 9-fluorenylmethyl carbamate (Fmoc), 9-(2-sulfo)fluorenylmethyl carbamate, 9-(2,7-dibromo)fluoroenylmethyl carbamate, 2,7-di-i-butyl-[9-( 10,10-dioxo-l 0, 10,10,10-tetrahydrothioxanthyl)]methyl carbamate (DBD-Tmoc), 4-methoxyphenacyl carbamate (Phenoc), 2,2,2-trichloroethyl carbamate (Troc), 2-trimethylsilylethyl carbamate (Teoc), 2-phenylethyl carbamate (hZ), 1- (l-adamantyl)-l-methylethyl carbamate (Adpoc), l,l-dimethyl-2-haloethyl carbamate, l,l-dimethyl-2,2-dibromoethyl carbamate (DB-i-BOC), l,l-dimethyl-2,2,2-trichloroethyl
carbamate (TCBOC), l-methyl-l-(4-biphenylyl)ethyl carbamate (Bpoc), 1— (3,5— di— i— butylphenyl)-l-methylethyl carbamate (i-Bumeoc), 2-(2'- and 4'-pyridyl)ethyl carbamate (Pyoc), 2-(N,N-dicyclohexylcarboxamido)ethyl carbamate, i-butyl carbamate (BOC or Boc), 1-adamantyl carbamate (Adoc), vinyl carbamate (Voc), allyl carbamate (Alloc), 1- isopropylallyl carbamate (Ipaoc), cinnamyl carbamate (Coc), 4-nitrocinnamyl carbamate (Noc), 8-quinolyl carbamate, N-hydroxypiperidinyl carbamate, alkyldithio carbamate, benzyl carbamate (Cbz), p-methoxybenzyl carbamate (Moz), /?-nitobenzyl carbamate, p- bromobenzyl carbamate, p-chlorobenzyl carbamate, 2,4-dichlorobenzyl carbamate, 4- methylsulfinylbenzyl carbamate (Msz), 9-anthrylmethyl carbamate, diphenylmethyl carbamate, 2-methylthioethyl carbamate, 2-methylsulfonylethyl carbamate, 2-(p- toluenesulfonyl)ethyl carbamate, [2-(l,3-dithianyl)]methyl carbamate (Dmoc), 4- methylthiophenyl carbamate (Mtpc), 2,4-dimethylthiophenyl carbamate (Bmpc), 2- phosphonioethyl carbamate (Peoc), 2-triphenylphosphonioisopropyl carbamate (Ppoc), 1, 1- dimethyl-2-cyanoethyl carbamate, m-chloro-p-acyloxybenzyl carbamate, p- (dihydroxyboryl)benzyl carbamate, 5-benzisoxazolylmethyl carbamate, 2-(trifluoromethyl)- 6-chromonylmethyl carbamate (Tcroc), m-nitrophenyl carbamate, 3,5-dimethoxybenzyl carbamate, o-nitrobenzyl carbamate, 3,4-dimethoxy-6-nitrobenzyl carbamate, phenyl (o- nitrophenyl)methyl carbamate, i-amyl carbamate, S-benzyl thiocarbamate, p-cyanobenzyl carbamate, cyclobutyl carbamate, cyclohexyl carbamate, cyclopentyl carbamate,
cyclopropylmethyl carbamate, p-decyloxybenzyl carbamate, 2,2-dimethoxyacylvinyl carbamate, o-(N,N-dimethylcarboxamido)benzyl carbamate, l,l-dimethyl-3-(N,N- dimethylcarboxamido)propyl carbamate, 1, 1-dimethylpropynyl carbamate, di(2- pyridyl)methyl carbamate, 2-furanylmethyl carbamate, 2-iodoethyl carbamate, isoborynl carbamate, isobutyl carbamate, isonicotinyl carbamate, /?-(/ -methoxyphenylazo)benzyl carbamate, 1-methylcyclobutyl carbamate, 1-methylcyclohexyl carbamate, 1-methyl-l- cyclopropylmethyl carbamate, l-methyl-l-(3,5-dimethoxyphenyl)ethyl carbamate, 1- methyl-l-(p-phenylazophenyl)ethyl carbamate, 1 -methyl- 1-phenylethyl carbamate, 1- methyl-l-(4-pyridyl)ethyl carbamate, phenyl carbamate, /?-(phenylazo)benzyl carbamate, 2,4,6-tri-i-butylphenyl carbamate, 4-(trimethylammonium)benzyl carbamate, and 2,4,6- trimethylbenzyl carbamate.
[0043] Nitrogen protecting groups such as sulfonamide groups (e.g. , -S(=0)2Raa) include, but are not limited to, p-toluenesulfonamide (Ts), benzenesulfonamide, 2,3,6,-trimethyl-4- methoxybenzenesulfonamide (Mtr), 2,4,6-trimethoxybenzenesulfonamide (Mtb), 2,6-
dimethyl-4-methoxybenzenesulfonamide (Pme), 2,3,5, 6-tetramethyl-4- methoxybenzenesulfonamide (Mte), 4-methoxybenzenesulfonamide (Mbs), 2,4,6- trimethylbenzenesulfonamide (Mts), 2,6-dimethoxy-4-methylbenzenesulfonamide (iMds), 2,2,5,7, 8-pentamethylchroman-6-sulfonamide (Pmc), methanesulfonamide (Ms), β- trimethylsilylethanesulfonamide (SES), 9-anthracenesulfonamide, 4-(4',8'- dimethoxynaphthylmethyl)benzenesulfonamide (DNMBS), benzylsulfonamide,
trifluoromethylsulfonamide, and phenacylsulfonamide.
[0044] Other nitrogen protecting groups include, but are not limited to, phenothiazinyl- (lO)-acyl derivative, N'-p-toluenesulfonylaminoacyl derivative, N'-phenylaminothioacyl derivative, N-benzoylphenylalanyl derivative, N-acetylmethionine derivative, 4,5-diphenyl- 3-oxazolin-2-one, N-phthalimide, N-dithiasuccinimide (Dts), N-2,3-diphenylmaleimide, N-2,5-dimethylpyrrole, N-l,l,4,4-tetramethyldisilylazacyclopentane adduct (STABASE), 5-substituted l,3-dimethyl-l,3,5-triazacyclohexan-2-one, 5-substituted 1,3-dibenzyl- l,3,5-triazacyclohexan-2-one, 1-substituted 3,5-dinitro-4-pyridone, N-methylamine, N- allylamine, N-[2-(trimethylsilyl)ethoxy]methylamine (SEM), N-3-acetoxypropylamine, N- (l-isopropyl-4-nitro-2-oxo-3-pyroolin-3-yl)amine, quaternary ammonium salts, N- benzylamine, N-di(4-methoxyphenyl)methylamine, N-5-dibenzosuberylamine, N- triphenylmethylamine (Tr), N-[(4-methoxyphenyl)diphenylmethyl] amine (MMTr), N-9- phenylfluorenylamine (PhF), N-2,7-dichloro-9-fluorenylmethyleneamine, N- ferrocenylmethylamino (Fcm), N-2-picolylamino N'-oxide, N-1,1- dimethylthiomethyleneamine, N-benzylideneamine, N-p-methoxybenzylideneamine, N- diphenylmethyleneamine, N-[(2-pyridyl)mesityl]methyleneamine, N-(N',N'- dimethylaminomethylene)amine, N,N'-isopropylidenediamine, N-p-nitrobenzylideneamine, N-salicylideneamine, N-5-chlorosalicylideneamine, N-(5-chloro-2- hydroxyphenyl)phenylmethyleneamine, N-cyclohexylideneamine, N-(5,5-dimethyl-3-oxo- l-cyclohexenyl)amine, N-borane derivative, N-diphenylborinic acid derivative, N- [phenyl(pentaacylchromium- or tungsten)acyl] amine, N-copper chelate, N-zinc chelate, N- nitroamine, N-nitrosoamine, amine N-oxide, diphenylphosphinamide (Dpp),
dimethylthiophosphinamide (Mpt), diphenylthiophosphinamide (Ppt), dialkyl
phosphoramidates, dibenzyl phosphoramidate, diphenyl phosphoramidate,
benzenesulfenamide, o-nitrobenzenesulfenamide (Nps), 2,4-dinitrobenzenesulfenamide, pentachlorobenzenesulfenamide, 2-nitro-4-methoxybenzenesulfenamide,
triphenylmethylsulfenamide, and 3-nitropyridinesulfenamide (Npys).
[0045] Exemplary oxygen atom substituents include, but are not limited to, -Raa, - C(=0)SRaa, -C(=0)Raa, -C02Raa, -C(=0)N(Rbb)2, -C(=NRbb)Raa, -C(=NRbb)ORaa, - C(=NRbb)N(Rbb)2, -S(=0)Raa, -SOaR^, -Si(Raa)3 -P(RCC)2, -P(RCC) , -P(=0)2Raa, - P(=0)(Raa)2, -P(=0)(ORcc)2, -P(=0)2N(Rbb)2, and -P(=0)(NRbb)2, wherein Raa, Rbb, and Rcc are as defined herein. In certain embodiments, the oxygen atom substituent present on an oxygen atom is an oxygen protecting group (also referred to as a hydroxyl protecting group). Oxygen protecting groups are well known in the art and include those described in detail in
Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3 rd edition, John Wiley & Sons, 1999, incorporated herein by reference. Exemplary oxygen protecting groups include, but are not limited to, methyl, i-butyloxycarbonyl (BOC or Boc), methoxylmethyl (MOM), methylthiomethyl (MTM), i-butylthiomethyl, (phenyldimethylsilyl)methoxymethyl (SMOM), benzyloxymethyl (BOM), p-methoxybenzyloxymethyl (PMBM), (4- methoxyphenoxy)methyl (p-AOM), guaiacolmethyl (GUM), i-butoxymethyl, 4- pentenyloxymethyl (POM), siloxymethyl, 2-methoxyethoxymethyl (MEM), 2,2,2- trichloroethoxymethyl, bis(2-chloroethoxy)methyl, 2-(trimethylsilyl)ethoxymethyl
(SEMOR), tetrahydropyranyl (THP), 3-bromotetrahydropyranyl, tetrahydrothiopyranyl, 1- methoxycyclohexyl, 4-methoxytetrahydropyranyl (MTHP), 4- methoxytetrahydrothiopyranyl, 4-methoxytetrahydrothiopyranyl S,S-dioxide, l-[(2-chloro- 4-methyl)phenyl]-4-methoxypiperidin-4-yl (CTMP), l,4-dioxan-2-yl, tetrahydrofuranyl, tetrahydrothiofuranyl, 2,3,3a,4,5,6,7,7a-octahydro-7,8,8-trimethyl-4,7-methanobenzofuran- 2-yl, 1-ethoxyethyl, l-(2-chloroethoxy)ethyl, 1-methyl-l-methoxyethyl, 1-methyl-l- benzyloxyethyl, l-methyl-l-benzyloxy-2-fluoroethyl, 2,2,2-trichloroethyl, 2- trimethylsilylethyl, 2-(phenylselenyl)ethyl, i-butyl, allyl, p-chlorophenyl, p-methoxyphenyl, 2,4-dinitrophenyl, benzyl (Bn), /?-methoxybenzyl, 3,4-dimethoxybenzyl, o-nitrobenzyl, p- nitrobenzyl, p-halobenzyl, 2,6-dichlorobenzyl, p-cyanobenzyl, p-phenylbenzyl, 2-picolyl, 4-picolyl, 3-methyl-2-picolyl N-oxido, diphenylmethyl, p^'-dinitrobenzhydryl, 5- dibenzosuberyl, triphenylmethyl, a-naphthyldiphenylmethyl, p- methoxyphenyldiphenylmethyl, di(p-methoxyphenyl)phenylmethyl, tri(p- methoxyphenyl)methyl, 4-(4'-bromophenacyloxyphenyl)diphenylmethyl, 4,4',4"-tris(4,5- dichlorophthalimidophenyl)methyl, 4,4',4"-tris(levulinoyloxyphenyl)methyl, 4,4',4"- tris(benzoyloxyphenyl)methyl, 3-(imidazol-l-yl)bis(4',4"-dimethoxyphenyl)methyl, 1 , 1- bis(4-methoxyphenyl)- -pyrenylmethyl, 9-anthryl, 9-(9-phenyl)xanthenyl, 9-(9-phenyl- 10-oxo)anthryl, l,3-benzodisulfuran-2-yl, benzisothiazolyl S,S-dioxido, trimethylsilyl
(TMS), triethylsilyl (TES), triisopropylsilyl (TIPS), dimethylisopropylsilyl (IPDMS), diethylisopropylsilyl (DEIPS), dimethylthexylsilyl, i-butyldimethylsilyl (TBDMS), t- butyldiphenylsilyl (TBDPS), tribenzylsilyl, tri-p-xylylsilyl, triphenylsilyl,
diphenylmethylsilyl (DPMS), i-butylmethoxyphenylsilyl (TBMPS), formate,
benzoylformate, acetate, chloroacetate, dichloroacetate, trichloroacetate, trifhioroacetate, methoxyacetate, triphenylmethoxyacetate, phenoxyacetate, p-chlorophenoxyacetate, 3- phenylpropionate, 4-oxopentanoate (levulinate), 4,4-(ethylenedithio)pentanoate
(levulinoyldithioacetal), pivaloate, adamantoate, crotonate, 4-methoxycrotonate, benzoate, p- phenylbenzoate, 2,4,6-trimethylbenzoate (mesitoate), alkyl methyl carbonate, 9- fluorenylmethyl carbonate (Fmoc), alkyl ethyl carbonate, alkyl 2,2,2-trichloroethyl carbonate (Troc), 2-(trimethylsilyl)ethyl carbonate (TMSEC), 2-(phenylsulfonyl) ethyl carbonate (Psec), 2-(triphenylphosphonio) ethyl carbonate (Peoc), alkyl isobutyl carbonate, alkyl vinyl carbonate alkyl allyl carbonate, alkyl p-nitrophenyl carbonate, alkyl benzyl carbonate, alkyl /7-methoxybenzyl carbonate, alkyl 3,4-dimethoxybenzyl carbonate, alkyl o-nitrobenzyl carbonate, alkyl p-nitrobenzyl carbonate, alkyl S-benzyl thiocarbonate, 4-ethoxy-l- napththyl carbonate, methyl dithiocarbonate, 2-iodobenzoate, 4-azidobutyrate, 4-nitro-4- methylpentanoate, o-(dibromomethyl)benzoate, 2-formylbenzenesulfonate, 2- (methylthiomethoxy)ethyl, 4-(methylthiomethoxy)butyrate, 2-
(methylthiomethoxymethyl)benzoate, 2,6-dichloro-4-methylphenoxyacetate, 2,6-dichloro- 4-( 1 , 1 ,3,3-tetramethylbutyl)phenoxyacetate, 2,4-bis( 1 , l-dimethylpropyl)phenoxyacetate, chlorodiphenylacetate, isobutyrate, monosuccinoate, (E)-2-methyl-2-butenoate, o- (methoxyacyl)benzoate, a-naphthoate, nitrate, alkyl Ν,Ν,Ν',Ν'- tetramethylphosphorodiamidate, alkyl N-phenylcarbamate, borate, dimethylphosphinothioyl, alkyl 2,4-dinitrophenylsulfenate, sulfate, methanesulfonate (mesylate), benzylsulfonate, and tosylate (Ts).
[0046] Exemplary sulfur atom substituents include, but are not limited to, -Raa, - C(=0)SRaa, -C(=0)Raa, -C02Raa, -C(=0)N(Rbb)2, -C(=NRbb)Raa, -C(=NRbb)ORaa, - C(=NRbb)N(Rbb)2, -S(=0)Raa, -SOaR^, -Si(Raa)3 -P(RCC)2, -P(RCC)3, -P(=0)2Raa, - P(=0)(Raa)2, -P(=0)(ORcc)2, -P(=0)2N(Rbb)2, and -P(=0)(NRbb)2, wherein Raa, Rbb, and Rcc are as defined herein. In certain embodiments, the sulfur atom substituent present on a sulfur atom is a sulfur protecting group (also referred to as a thiol protecting group). Sulfur protecting groups are well known in the art and include those described in detail in Protecting
Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3r edition, John Wiley & Sons, 1999, incorporated herein by reference.
[0047] The invention is not intended to be limited in any manner by the above exemplary listing of substituents.
[0048] The term "salt" refers to ionic compounds that result from the neutralization reaction of an acid and a base. A salt is composed of one or more cations (positively charged ions) and one or more anions (negative ions) so that the salt is electrically neutral (without a net charge). Salts of the compounds of this invention include those derived from inorganic and organic acids and bases. Examples of acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, and perchloric acid, or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid, or malonic acid or by using other methods known in the art such as ion exchange. Other salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate,
camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate,
ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2-hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2- naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, propionate, stearate, succinate, sulfate, tartrate, thiocyanate, p-toluenesulfonate, undecanoate, valerate salts, and the like. Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N+(Ci^ alkyl)4 salts. Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like. Further salts include ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, lower alkyl sulfonate, and aryl sulfonate.
[0049] The term "tautomers" or "tautomeric" refers to two or more interconvertable compounds resulting from at least one formal migration of a hydrogen atom and at least one change in valency (e.g., a single bond to a double bond, a triple bond to a single bond, or vice versa). The exact ratio of the tautomers depends on several factors, including temperature, solvent, and pH. Tautomerizations (i.e., the reaction providing a tautomeric pair) may catalyzed by acid or base. Exemplary tautomerizations include keto-to-enol, amide-to-imide, lactam- to-lactim, enamine-to-imine, and enamine-to-(a different enamine) tautomerizations.
[0050] It is also to be understood that compounds that have the same molecular formula but differ in the nature or sequence of bonding of their atoms or the arrangement of their atoms in space are termed "isomers". Isomers that differ in the arrangement of their atoms in space are termed "stereoisomers".
[0051] Stereoisomers that are not mirror images of one another are termed
"diastereomers" and those that are non-superimposable mirror images of each other are termed "enantiomers". When a compound has an asymmetric center, for example, it is bonded to four different groups, a pair of enantiomers is possible. An enantiomer can be characterized by the absolute configuration of its asymmetric center and is described by the R- and S-sequencing rules of Cahn and Prelog, or by the manner in which the molecule rotates the plane of polarized light and designated as dextrorotatory or levorotatory (i.e., as (+) or (-)-isomers respectively). A chiral compound can exist as either individual enantiomer or as a mixture thereof. A mixture containing equal proportions of the enantiomers is called a "racemic mixture".
[0052] The term "solvate" refers to forms of the compound that are associated with a solvent, usually by a solvolysis reaction. This physical association may include hydrogen bonding. Conventional solvents include water, methanol, ethanol, acetic acid, DMSO, THF, diethyl ether, and the like. The compounds described herein may be prepared, e.g., in crystalline form, and may be solvated. Suitable solvates include pharmaceutically acceptable solvates and further include both stoichiometric solvates and non- stoichiometric solvates. In certain instances, the solvate will be capable of isolation, for example, when one or more solvent molecules are incorporated in the crystal lattice of a crystalline solid. "Solvate" encompasses both solution-phase and isolatable solvates. Representative solvates include hydrates, ethanolates, and methanolates.
[0053] The term "hydrate" refers to a compound that is associated with water. Typically, the number of the water molecules contained in a hydrate of a compound is in a definite ratio to the number of the compound molecules in the hydrate. Therefore, a hydrate of a compound may be represented, for example, by the general formula R x H20, wherein R is the compound, and x is a number greater than 0. A given compound may form more than one type of hydrate, including, e.g., monohydrates (x is 1), lower hydrates (x is a number greater than 0 and smaller than 1, e.g., hemihydrates (R-0.5 H20)), and polyhydrates (x is a number greater than 1, e.g., dihydrates (R-2 H20) and hexahydrates (R-6 H20)).
[0054] The term "polymorph" refers to a crystalline form of a compound (or a salt, hydrate, or solvate thereof) in a particular crystal packing arrangement. All polymorphs have the same elemental composition. Different crystalline forms usually have different X-ray diffraction patterns, infrared spectra, melting points, density, hardness, crystal shape, optical and electrical properties, stability, and solubility. Recrystallization solvent, rate of
crystallization, storage temperature, and other factors may cause one crystal form to dominate. Various polymorphs of a compound can be prepared by crystallization under different conditions.
[0055] The term "complex" or "coordination complex" refers to an association of at least one atom or ion (which is referred to as a "central atom," "central ion," or "acceptor," and is usually a metallic cation) and a surrounding array of bound ligands or donors). Ligands are generally bound to a central atom or central ion by a coordinate covalent bond (e.g. , ligands may donate electrons from a lone electron pair into an empty orbital of the central atom or central ion) and are referred to as being "coordinated" to the central atom or central ion. There are also organic ligands such as alkenes whose -bonds can coordinate to empty orbitals of an acceptor. A complex may include one or more donors, which can be the same or different. A complex may also include one or more acceptors, which can be the same or different.
[0056] The term "ligand" refers to an ion or molecule that binds to a central atom or ion (e.g., a central metal atom or ion) to form a coordination complex. Ligands are usually electron donors, and the central atom or ion is electron acceptors. The bonding between the central atom or ion and the ligand typically involves formal donation of one or more of the ligand' s electron pairs. The nature of such bonding can range from covalent to ionic, and the bond order can range from one to three. One central atom or ion may bind to one or more ligands of the same or different type. A ligand may be capable of binding a central atom or ion through multiple sites, usually because the ligand includes lone pairs on more than one atom of the ligand. Ligands in a complex may affect the reactivity (e.g. , ligand substitution rates and redox) of the central atom or ion. Exemplary ligands include charge-neutral ligands ("ligand molecules," e.g. , CH3CN, amides (e.g. , N,N-dimethylformamide (DMF), N,N- dimethylacetamide (DMA), or N-methyl-2-pyrrolidone (NMP)), dimethyl sulfoxide (DMSO), amines (e.g., ammonia; ethylenediamine (en); pyridine (py); 2,2'-bipyridine (bipy); and 1,10- phenanthroline (phen)), phosphines (e.g., PPh3), ethers (e.g. , tetrahydrofuran (THF), 2- methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl t-butyl ether
(MTBE), dimethoxyethane (DME), and diglyme), ketones (e.g., acetone and butanone),
chlorohydrocarbons (e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, and 1,2-dichloroethane (DCE)), esters (e.g., propylene carbonate and ethyl acetate), CO, N2, water, and alkenes) and anionic ligands ("ligand ions," e.g., halides, hydride, alkyls, S2 ~, S- CN", 0-N02 ", N-N2 ~ O-H", [0-C(=0)-C(=0)-0]2 ", 0-N-O", N=C=S", CN").
[0057] The term "transition metal" refers to elements that are in the d-block and f-block of the Periodic Chart of the Elements, which may exhibit a variety of oxidation states, and which may form numerous complex ions. As used herein, the term "d-block" refers to those elements that have electrons filling the 3d, 4d, 5d, and 6d orbitals, and the term "f-block" refers to those elements (including lanthanides and the actinides) that have electrons filling the 4f and 5f orbitals. Exemplary transition metals include palladium, nickel, cobalt, copper, platinum, silver, manganese, zinc, iridium, rhodium, iron, and ruthenium. The term
"transition metal" also includes alloys, metal/metal composites, metal ceramic composites, and metal polymer composites, as well as other metal composites.
[0058] The term "catalysis," "catalyze," or "catalytic" refers to the increase in rate of a chemical reaction due to the participation of a substance called a "catalyst." In certain embodiments, the amount and nature of a catalyst remains essentially unchanged during a reaction. In certain embodiments, a catalyst is regenerated, or the nature of a catalyst is essentially restored after a reaction. A catalyst may participate in multiple chemical transformations. The effect of a catalyst may vary due to the presence of other substances known as inhibitors or poisons (which reduce the catalytic activity) or promoters (which increase the activity). Catalyzed reactions have lower activation energy (rate-limiting free energy of activation) than the corresponding uncatalyzed reaction, resulting in a higher reaction rate at the same temperature. Catalysts may affect the reaction environment favorably, bind to the reagents to polarize bonds, form specific intermediates that are not typically produced by a uncatalyzed reaction, or cause dissociation of reagents to reactive forms.
[0059] The term "arene" refers to a compound including an aryl moiety, wherein at least one carbon atom of the aryl ring system is directly attached to a hydrogen atom.
[0060] The term "heteroarene" refers to a compound including a heteroaryl moiety, wherein at least one carbon atom of the heteroaryl ring system is directly attached to a hydrogen atom.
[0061] A "counterion" or "anionic counterion" is a negatively charged group associated with a positively charged group in order to maintain electronic neutrality. An anionic counterion may be monovalent (i.e., including one formal negative charge). An anionic
counterion may also be multivalent (i.e., including more than one formal negative charge), such as divalent or trivalent. Exemplary counterions include halide ions (e.g., F , CT, Br , Γ), NO3 , CIO4 , OH , H2PO4 , HSO4 , sulfonate ions (e.g., methansulfonate,
trifluoromethanesulfonate, /7-toluenesulfonate, benzenesulfonate, 10-camphor sulfonate, naphthalene-2-sulfonate, naphthalene-l-sulfonic acid-5-sulfonate, ethan-l-sulfonic acid- 2-sulfonate, and the like), carboxylate ions (e.g., acetate, ethanoate, propanoate, benzoate, glycerate, lactate, tartrate, glycolate, and the like), BF4 , PF4 , PF6 , SbF6 ~, B[3,5- (CF3)2C6H3]4r, BPh4 , Al(OC(CF3)3)4 ~, and a carborane anion (e.g., CBUH12 or
(HCBnMesBre) ).
[0062] The term "non-coordinating anionic counterion" refers to an anion that interacts weakly with cations. Exemplary non-coordinating anions include, but are not limited to, C104 , ΟΤΓ, BF4 , PF4 , PF6 , and SbF6 ~. Other examples of non-coordinating anions include, but are not limited to, B[3,5-(CF )2C6H ]4]~, BPh4 , Al(OC(CF ) )4 ~, or a carborane anion (e.g., CBUH12 or (HCBnMe5Br6) ).
BRIEF DESCRIPTION OF THE DRAWINGS
[0063] Figure 1 depicts an X-ray structure of complex 1 (elipsoids drawn at 50% probability).
[0064] Figure 2 shows a proposed mechanism of a catalytic imidation reaction of the invention.
[0065] Figure 3 shows calculated valence orbitals of complex 1.
[0066] Figure 4 illustrates the rate law for the reduction of N-fluorobenzenesulfonimide (NFBS or NFSI) catalyzed by complex 1.
[0067] Figure 5 illustrates the rate law for an imidation reaction of the invention catalyzed by 1 and in the presence of Ag(bipy)2C104.
[0068] Figure 6 illustrates the rate law showing the order in Ag(bipy)2C104.
[0069] Figure 7 shows the results of experiments for the determination of the order in an arene substrate.
[0070] Figure 8 shows the results of experiments for the determination of the order in NFBS.
[0071] Figure 9 shows the results of experiments for the determination of the order in complex 1.
[0072] Figure 10 shows the results of experiments for the determination of the resting state. In-situ lH NMR (proton nuclear magnetic resonance) spectra of: catalytic imidation (top), pure complex 1 (bottom).
[0073] Figure 11 shows the 1H NMR spectra of Solution A (left panels) and Solution B (right panels) after 10 minutes (top panels) and 4 hours (bottom panels) of oxidation of Ru(bipy) (PF6)2 mediated by complex 1.
[0074] Figure 12 shows 1H NMR peak broadness at 23 °C, -20 °C, and -40 °C during oxidation of Ru(bipy)3(PF6)2 mediated by complex 1.
[0075] Figure 13 shows EPR (electron paramagnetic resonance) spectra of Solution B and pure Ru(bipy)3(PF6)3.
[0076] Figure 14 shows the rates of NFBS consumption by Ru(bipy)3(PF6)2 or by
Ru(bipy)3(PF6)2 in the presence of complex 1.
[0077] Figure 15 shows the EPR spectra of Solutions E and F.
[0078] Figure 16 shows the optimized structure of complex 1 with the B3PW91 functional with the BS I basis set.
DETAILED DESCRIPTION OF CERTAIN EMBODIMENTS OF THE INVENTION
[0079] The present invention provides novel transition metal complexes (e.g., complexes of any one of Formulae (CI) to (C25)) that include an amine-N-oxide motif. These transition metal complexes have been found useful in the imidation of arenes and heteroarenes. The invention also provides methods of using the transition metal complexes as catalysts in the preparation of N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof) and aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof) through imidation of arenes or heteroarenes (e.g., compounds of Formula (A), and salts thereof). The methods of the invention involves an imidating agent (e.g., a compound of Formula (B), such as N-fluorobenzenesulfonimide (NFBS or NFSI)) in the presence of a transition metal complex of the invention and a single-electron reductant (e.g., an Ag(I) or Ru(II) salt).
[0080] Without wishing to be bound by any particular theory, the inventive methods may involve a catalytic, intermolecular C-H imidation that proceeds via a mechanism different from the conventional metallation-functionalization sequence and may enable the transfer of the imido group without the formation of conventionally targeted organometallic
intermediates. It is thought that oxidation of the catalyst (e.g., complexes of any one of Formulae (CI) to (C25)) employed in the inventive methods may occur as the turnover-
limiting step, while C-H bond functionalization occurs subsequently at a high oxidation state of the catalyst. Also in contrast to previously reported metal-catalyzed amidation reactions, the methods of the invention may be practiced at or below room temperature. The imidation methods of the invention do not require coordinating directing groups on the starting material and give synthetically useful yields with the arene or heteroarene starting material as the limiting reagent (e.g., one equivalent of arene or heteroarene and more than one equivalents of imidating agent is typically used).
Methods of Preparing Compounds of Formula (I) or (II)
[0081] In one aspect, the present invention provides methods of preparing N-aryl sulfonimides or N-heteroaryl sulfonimides through a catalytic imidation of an arene or heteroarene (e.g., a compound of Formula (A), or a salt thereof) using an imidating agent in the presence of a catalyst and a single-electron reductant. The imidating agent employed in the methods of the invention may be an N-fluorosulfonimide, such as a compound of Formula (B) (e.g., N-fluorobenzenesulfonimide (NFBS or NFSI)), or a salt thereof. The catalyst useful in the inventive methods may be a transition metal complex, such as a complex of any one of Formulae (CI) to (C25).
[0082] In certain embodiments, the invention provides methods of preparing compounds of Formula (I):
and salts thereof. The inventive methods typically include contacting a compound of Formula (A), or a salt thereof, with a compound of Formula (B), or a salt thereof, in the presence of a transition metal complex of any one of Formulae (CI) to (C25) and a single-electron reductant to provide the compound of Formula (I), or the salt thereof:
C12)
(C15) (C16)
(C19)
(C21) (C22)
(C25)
wherein:
Ring A is substituted or unsubstituted aryl or substituted or unsubstituted heteroaryl; each instance of RA is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -ORA1, -N(RA1)2, -SRA1, -CN, -SCN, -C(=NRA1)RA1, -C(=NRA1)ORA1, -C(=NRA1)N(RA1)2, - C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2, -N02, -NRA1C(=0)RA1, -NRA1C(=0)ORA1, - NRA1C(=0)N(RA1)2, -OC(=0)RA1, -OC(=0)ORA1, -OC(=0)N(RA1)2, -S(=0)2RA1, - S(=0)2ORA1, -S(=0)2N(RA1)2, -OS(=0)2RA1, -N(RA1)S(=0)2RA1, -N(RA1)S(=0)2N(RA1)2, - Si(RA1)4, or -P(=0)(ORA1)2, or two instances of RA are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or
unsubstituted aryl, or substituted or unsubstituted heteroaryl ring;
each instance of RA1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of RA1 are joined to form a substituted or unsubstituted heterocyclic ring;
k is 0, 1, 2, 3, 4, or 5;
each instance of Ring B is substituted or unsubstituted heteroaryl;
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR B 1 , -N(RB1)2, -SRB1, -CN, -SCN, -C(=NRB1)RB1, -C(=NRB1)ORB1, -C(=NRB1)N(RB1)2, - C(=0)RB1, -C(=0)ORB1, -C(=0)N(RB1)2, -N02, -NRB1C(=0)RB1, -NRB1C(=0)ORB1, - NRB1C(=0)N(RB1)2, -OC(=0)RB1, -OC(=0)ORB1, or -OC(=0)N(RB1)2, or two instances of R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R B 1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R B 1 are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of m is independently 0, 1, 2, 3, 4, or 5;
each instance of M is a transition metal ion;
each instance of Ring B is independently substituted or unsubstituted heteroaryl; each instance of is independently a single or double bond;
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR C 1 , -N(RC1)2, -SRC1, -CN, -SCN, -C(=NRC1)RC1, -C(=NRcl)ORcl, -C(=NRC1)N(RC1)2, - C(=0)Rcl, -C(=0)ORcl, -C(=0)N(Rcl)2, -N02, -NRclC(=0)Rcl, -NRclC(=0)ORcl, - NRclC(=0)N(Rcl)2, -OC(=0)Rcl, -OC(=0)ORcl, or -OC(=0)N(Rcl)2, or two instances of R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R CI is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a
nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R CI are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of r is independently 0, 1, 2, 3, or 4;
each instance of RD is independently substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring;
p
each instance of R is independently absent, hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or p
two instances of R are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of n is 0 or 1 ;
p is 1 or 2;
each instance of X is an anionic counterion; and
q is 1, 2, or 3.
[0083] The arene or heteroarene involved in the inventive methods may be a compound of Formula (A), or a salt thereof. Compounds of Formula (A) include substituted or unsubstituted aryl or substituted or unsubstituted heteroaryl as Ring A. In certain
embodiments, Ring A is substituted aryl. In certain embodiments, Ring A is unsubstituted aryl. In certain embodiments, Ring A is 6- to 14-membered aryl. In certain embodiments, Ring A is 6- to 10-membered aryl. In certain embodiments, Ring A is monocyclic aryl. In certain embodiments, Ring A is unsubstituted phenyl. In certain embo Ring A is
substituted phenyl. In certain embodiments, Ring A is of the formula:
. In certain
. In certain embodiments, Ring A is of
the formula: . In certain embodiments, Ring A is of the formula: . In
of the formula: . In certain embodiments, Ring A is of the formula:
n certa n em o ments, ng s o t e ormu a: . n certa n em o ments,
certain embodiments, Ring A is bicyclic aryl. In certain embodiments, Ring A is substituted naphthyl. In certain embodiments, Ring A is unsubstituted naphthyl. In certain embodiments, Ring A is substituted or unsubstituted aryl fused with one or more substituted or
unsubstituted carbocyclyl moieties or substituted or unsubstituted heterocyclyl moieties, wherein the point of attachment is on the aryl ring.
[0084] Ring A of Formula (I) may also be substituted or unsubstituted heteroaryl. In certain embodiments, Ring A is substituted heteroaryl. In certain embodiments, Ring A is unsubstituted heteroaryl. In certain embodiments, Ring A is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, Ring A is 6-membered, monocyclic heteroaryl. In certain embodiments, Ring A is of the
formula: . In certain embodiments, Ring A is of
or . In certain embodiments, Ring A is of the formula:
[0085] In certain embodiments, Ring A is 5-membered, monocyclic heteroaryl. In certain embodiments, Ring A is 5-membered, monocyclic heteroaryl, wherein one atom in the heteroaryl ring system is nitrogen, ox en, or sulfur. In certain embodiments, Ring A is of
formula:
. In certain embodiments, Ring A is 5-membered, monocyclic heteroaryl, wherein two atoms in the heteroaryl ring system are independently nitrogen, oxygen, or sulfur. In certain embodiments, Ring A is of the formula:
, or . In certain embodiments, Ring A is of the
formula: . In certain embodiments, Ring
Ring A is of the formula In certain embodiments, Ring A
embodiments, Ring A is 5-membered, monocyclic heteroaryl, wherein three atoms in the heteroaryl ring system are independently nitrogen, oxygen, or sulfur. In certain embodiments,
embodiments, Ring A is 5-membered, monocyclic heteroaryl, wherein four atoms in the heteroaryl ring system are nitrogen, oxygen, or sulfur. In certain embodiments Ring A is of the formula:
[0086] In certain embodiments, Ring A is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. The bicyclic heteroaryl described herein may be substituted or unsubstituted. In certain embodiments, Ring A is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, Ring A is 5- or 6- membered, monocyclic heteroaryl fused with phenyl, wherein one, two, or three atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, Ring A is 5- or 6-membered, monocyclic heteroaryl fused with another 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the bicyclic heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, Ring A is substituted or unsubstituted indole. In certain embodiments, Ring A is substituted or unsubstituted isoindole. In certain embodiments, Ring A is substituted or unsubstituted indazole. In certain embodiments, Ring A is substituted or unsubstituted benzothiophene. In certain embodiments, Ring A is substituted or unsubstituted isobenzothiophene. In certain embodiments, Ring A is substituted or unsubstituted benzofuran. In certain embodiments,
Ring A is substituted or unsubstituted benzoisofuran. In certain embodiments, Ring A is substituted or unsubstituted benzimidazole. In certain embodiments, Ring A is substituted or unsubstituted benzoxazole. In certain embodiments, Ring A is substituted or unsubstituted benzisoxazole. In certain embodiments, Ring A is substituted or unsubstituted benzothiazole. In certain embodiments, Ring A is substituted or unsubstituted benzisothiazole. In certain embodiments, Ring A is substituted or unsubstituted benzotriazole. In certain embodiments, Ring A is substituted or unsubstituted benzoxadiazole. In certain embodiments, Ring A is substituted or unsubstituted quinoline. In certain embodiments, Ring A is substituted or unsubstituted isoquinoline. In certain embodiments, Ring A is substituted or unsubstituted cinnoline. In certain embodiments, Ring A is substituted or unsubstituted quinoxaline. In certain embodiments, Ring A is substituted or unsubstituted phthalazine. In certain embodiments, Ring A is substituted or unsubstituted quinazoline.
[0087] Compounds of Formula (I) or (A) may include one or more substituent RA. In certain embodiments, at least one instance of RA is H. In certain embodiments, at least one instance of RA is halogen. In certain embodiments, at least one instance of RA is F. In certain embodiments, at least one instance of RA is CI. In certain embodiments, at least one instance of RA is Br. In certain embodiments, at least one instance of RA is I (iodine). In certain embodiments, at least one instance of RA is substituted acyl. In certain embodiments, at least one instance of RA is unsubstituted acyl. In certain embodiments, at least one instance of RA is substituted alkyl. In certain embodiments, at least one instance of RA is unsubstituted alkyl. In certain embodiments, at least one instance of RA is C1-12 alkyl. In certain embodiments, at least one instance of RA is substituted Ci_6 alkyl. In certain embodiments, at least one instance of RA is unsubstituted C1-6 alkyl. In certain embodiments, at least one instance of RA is substituted methyl. In certain embodiments, at least one instance of RA is -CH2F. In certain embodiments, at least one instance of RA is -CHF2. In certain embodiments, at least one instance of RA is -CF3. In certain embodiments, at least one instance of RA is Bn. In certain embodiments, at least one instance of RA is unsubstituted methyl. In certain embodiments, at least one instance of RA is ethyl. In certain embodiments, at least one instance of RA is propyl. In certain embodiments, at least one instance of RA is butyl. In certain embodiments, at least one instance of RA is pentyl. In certain embodiments, at least one instance of RA is hexyl. In certain embodiments, at least one instance of RA is substituted alkenyl. In certain embodiments, at least one instance of RA is unsubstituted alkenyl. In certain embodiments, at least one instance of RA is substituted or unsubstituted C1-6 alkenyl. In certain embodiments, at least one instance of RA is vinyl. In certain embodiments, at least one instance of RA is
substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of RA is ethynyl. In certain
embodiments, at least one instance of RA is substituted carbocyclyl. In certain embodiments, at least one instance of RA is unsubstituted carbocyclyl. In certain embodiments, at least one instance of RA is saturated carbocyclyl. In certain embodiments, at least one instance of RA is unsaturated carbocyclyl. In certain embodiments, at least one instance of RA is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of RA is monocyclic carbocyclyl. In certain embodiments, at least one instance of RA is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of RA is cylcopropyl. In certain embodiments, at least one instance of RA is cyclobutyl. In certain embodiments, at least one instance of RA is cyclopentyl. In certain embodiments, at least one instance of RA is cyclohexyl. In certain embodiments, at least one instance of RA is cycloheptyl. In certain embodiments, at least one instance of RA is bicyclic carbocyclyl. In certain embodiments, at least one instance of RA is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of RA is substituted heterocyclyl. In certain embodiments, at least one instance of RA is
unsubstituted heterocyclyl. In certain embodiments, at least one instance of RA is saturated heterocyclyl. In certain embodiments, at least one instance of RA is unsaturated heterocyclyl. In certain embodiments, at least one instance of RA is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of RA is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RA is monocyclic heterocyclyl. In certain embodiments, at least one instance of RA is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of RA is bicyclic heterocyclyl. In certain embodiments, at least one instance of RA is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of RA is substituted aryl. In certain embodiments, at least one instance of RA is unsubstituted aryl. In certain embodiments, at least one instance of RA is 6- to 14-membered aryl. In certain embodiments, at least one instance of RA is 6- to 10-membered aryl. In certain embodiments, at least one instance of RA is substituted phenyl. In certain embodiments, at least one instance of RA is unsubstituted phenyl. In certain embodiments, at least one instance of RA is substituted naphthyl. In certain embodiments, at least one instance of RA is unsubstituted naphthyl. In certain embodiments, at least one instance of RA is substituted heteroaryl. In certain embodiments, at least one instance of RA is
unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RA is monocyclic heteroaryl. In certain embodiments, at least one instance of RA is 5-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of RA is 6-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of RA is substituted pyridyl. In certain embodiments, at least one instance of RA is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of RA is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of RA is 9-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of RA is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of RA is -ORA1. In certain embodiments, at least one instance of RA is -ORA1, wherein RA1 is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of RA is -OMe. In certain embodiments, at least one instance of RA is -OEt. In certain embodiments, at least one instance of RA is -OPr. In certain embodiments, at least one instance of RA is -OBu. In certain embodiments, at least one instance of RA is -O(pentyl). In certain embodiments, at least one instance of RA is - O(hexyl). In certain embodiments, at least one instance of RA is -OBn. certain embodiments, at least one instance of RA is -OPh. In certain embodiments, at least one instance of RA is - OH. In certain embodiments, at least one instance of RA is -SRA1. In certain embodiments, at least one instance of RA is -SMe. In certain embodiments, at least one instance of RA is -SH. In certain embodiments, at least one instance of RA is -N(RA1)2. In certain embodiments, at least one instance of RA is -NMe2. In certain embodiments, at least one instance of RA is - NH2. In certain embodiments, at least one instance of RA is -CN. In certain embodiments, at least one instance of RA is -SCN. In certain embodiments, at least one instance of RA is - C(=NRA1)RA1, -C(=NRA1)ORA1, or -C(=NRA1)N(RA1)2. In certain embodiments, at least one instance of RA is -C(=0)RA1. In certain embodiments, at least one instance of RA is - C(=0)RA1, wherein RA1 is substituted or unsubstituted alkyl or substituted or unsubstituted aryl. In certain embodiments, at least one instance of RA is -C(=0)RA1, wherein RA1 is substituted or unsubstituted C1-6 alkyl or substituted or unsubstituted phenyl. In certain embodiments, at least one instance of RA is -C(=0)ORA1. In certain embodiments, at least one instance of RA is -C(=0)ORA1, wherein RA1 is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of RA is -C(=0)N(RA1)2. In certain embodiments,
at least one instance of RA is -C(=0)N(RA1)2, wherein each instance of RA1 is independently selected from the group consisting of hydrogen, substituted or unsubstituted alkyl, or a nitrogen protecting group. In certain embodiments, at least one instance of RA is - C(=0)N(RA1)2, wherein each instance of RA1 is independently selected from the group consisting of hydrogen, substituted or unsubstituted C1-6 alkyl, Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts. In certain embodiments, at least one instance of RA is -C(=0)NH2. In certain embodiments, at least one instance of RA is -N02. In certain embodiments, at least one instance of RA is -NRA1C(=0)RA1, -NRA1C(=0)ORA1, or - NRA1C(=0)N(RA1)2. In certain embodiments, at least one instance of RA is -OC(=0)RA1, - OC(=0)ORA1, or -OC(=0)N(RA1)2. In certain embodiments, at least one instance of RA is - S(=0)2RA1. In certain embodiments, at least one instance of RA is -S(=0)2RA1, wherein RA1 is substituted or unsubstituted aryl. In certain embodiments, at least one instance of RA is - S(=0)2RA1, wherein RA1 is substituted or unsubstituted phenyl. In certain embodiments, at least one instance of RA is -S(=0)2ORA1 or -S(=0)2N(RA1)2. In certain embodiments, at least one instance of RA is -OS(=0)2RA1 or -N(RA1)S(=0)2RA1. In certain embodiments, at least one instance of RA is -N(RA1)S(=0)2N(RA1)2. In certain embodiments, at least one instance of RA is -Si(RA1)4. In certain embodiments, at least one instance of RA is -Si(RA1)4, wherein each instance of RA1 is independently unsubstituted alkyl. In certain embodiments, at least one instance of RA is -P(=0)(ORA1)2. In certain embodiments, at least one instance of RA is - P(=0)(ORA1)2, wherein each instance of RA1 is independently unsubstituted alkyl.
[0088] In certain embodiments, two RA groups may be joined to form a substituted or unsubstituted carbocyclic ring. In certain embodiments, two instances of RA are joined to form a saturated or unsaturated carbocyclic ring. In certain embodiments, two instances of RA are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, two instances of RA are joined to form a 3- to 7-membered, monocyclic carbocyclic ring. In certain embodiments, two instances of RA are joined to form a 3-membered carbocyclic ring. In certain embodiments, two instances of RA are joined to form a 4-membered carbocyclic ring. In certain embodiments, two instances of RA are joined to form a 5-membered carbocyclic ring. In certain embodiments, two instances of RA are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of RA are joined to form a 7-membered carbocyclic ring. In certain
embodiments, two instances of RA are joined to form a 5- to 13-membered, bicyclic carbocyclic ring.
[0089] In certain embodiments, two instances of R are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of RA are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of RA are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of RA are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RA are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of RA are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[0090] In certain embodiments, two instances of RA are joined to form a substituted or unsubstituted aryl ring. In certain embodiments, two instances of RA are joined to form a 6- to 14-membered aryl ring. In certain embodiments, two instances of RA are joined to form a 6- to 10-membered aryl ring. In certain embodiments, two instances of RA are joined to form a monocyclic aryl ring. In certain embodiments, two instances of RA are joined to form an unsubstituted phenyl ring. In certain embodiments, two instances of RA are joined to form a substituted phenyl ring. In certain embodiments, two instances of RA are joined to form a bicyclic aryl ring. In certain embodiments, two instances of RA are joined to form a naphthyl ring.
[0091] In certain embodiments, two instances of RA are joined to form a substituted or unsubstituted heteroaryl ring. In certain embodiments, two instances of RA are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RA are joined to form a substituted or unsubstituted pyridyl ring. In certain embodiments, two instances of RA are joined to form a 9- or 10-membered, bicyclic heteroaryl ring, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[0092] In certain embodiments, at least one instance of RA is halogen, substituted or unsubstituted alkyl, or -ORA1. In certain embodiments, at least one instance of RA is halogen, substituted or unsubstituted C1-6 alkyl, or -ORA1, wherein RA1 is substituted or unsubstituted Ci-6 alkyl. In certain embodiments, at least one instance of RA is halogen, substituted or unsubstituted alkyl, -ORA1, substituted or unsubstituted acyl, -CN, -N02, -S(=0)2RA1, - Si(RA1)4, or -P(=0)(ORA1)2. In certain embodiments, at least one instance of RA is halogen,
substituted or unsubstituted Ci_6 alkyl, -ORA1, -C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2, - CN, -NO2, -S(=0)2RA1, -Si(RA1)4, or -P(=0)(ORA1)2, wherein each instance of RA1 is independently hydrogen, substituted or unsubstituted C1-6 alkyl, or substituted or
unsubstituted phenyl.
[0093] In certain embodiments, at least one instance of RA1 is H. In certain embodiments, at least one instance of RA1 is substituted acyl. In certain embodiments, at least one instance of RA1 is unsubstituted acyl. In certain embodiments, at least one instance of RA1 is acetyl. In certain embodiments, at least one instance of RA1 is substituted alkyl. In certain
embodiments, at least one instance of RA1 is unsubstituted alkyl. In certain embodiments, at least one instance of RA1 is C1-12 alkyl. In certain embodiments, at least one instance of RA1 is Ci-6 alkyl. In certain embodiments, at least one instance of RA1 is methyl. In certain embodiments, at least one instance of RA1 is ethyl. In certain embodiments, at least one instance of RA1 is propyl. In certain embodiments, at least one instance of RA1 is butyl. In certain embodiments, at least one instance of RA1 is pentyl. In certain embodiments, at least one instance of RA1 is hexyl. In certain embodiments, at least one instance of RA1 is substituted alkenyl. In certain embodiments, at least one instance of RA1 is unsubstituted alkenyl. In certain embodiments, at least one instance of RA1 is vinyl. In certain
embodiments, at least one instance of RA1 is substituted alkynyl. In certain embodiments, at least one instance of RA1 is unsubstituted alkynyl. In certain embodiments, at least one instance of RA1 is ethynyl. In certain embodiments, at least one instance of RA1 is substituted carbocyclyl. In certain embodiments, at least one instance of RA1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of RA1 is saturated carbocyclyl. In certain embodiments, at least one instance of RA1 is unsaturated carbocyclyl. In certain embodiments, at least one instance of RA1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of RA1 is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of RA1 is cylcopropyl. In certain embodiments, at least one instance of RA1 is cyclobutyl. In certain embodiments, at least one instance of RA1 is cyclopentyl. In certain embodiments, at least one instance of RA1 is cyclohexyl. In certain embodiments, at least one instance of RA1 is cycloheptyl. In certain embodiments, at least one instance of RA1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of RA1 is substituted heterocyclyl. In certain embodiments, at least one instance of RA1 is unsubstituted
heterocyclyl. In certain embodiments, at least one instance of RA1 is saturated heterocyclyl. In certain embodiments, at least one instance of RA1 is unsaturated heterocyclyl. In certain
embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of RA1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RA1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of RA1 is 5- to 13-membered, bicyclic
heterocyclyl. In certain embodiments, at least one instance of RA1 is substituted or
unsubstituted aryl. In certain embodiments, at least one instance of RA1 is 6- to 14-membered aryl. In certain embodiments, at least one instance of RA1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of RA1 is monocyclic aryl. In certain embodiments, at least one instance of RA1 is unsubstituted phenyl. In certain embodiments, at least one instance of RA1 is substituted phenyl. In certain embodiments, at least one instance of RA1 is bicyclic aryl. In certain embodiments, at least one instance of RA1 is naphthyl. In certain embodiments, at least one instance of RA1 is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of RA1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RA1 is pyridyl. In certain embodiments, at least one instance of RA1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of RA1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RA1 is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least one instance of RA1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom. In certain embodiments, RA1 is an oxygen protecting group when attached to an oxygen atom. In certain embodiments, RA1 is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ί-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom. In certain embodiments, RA1 is a sulfur protecting group when attached to a sulfur atom. In certain embodiments, RA1 is acetamidomethyl, ί-Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
[0094] In certain embodiments, two instances of RA1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of RA1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of RA1 are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RA1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of RA1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[0095] In certain embodiments, k is 0. In certain embodiments, k is 1. In certain embodiments, k is 2. In certain embodiments, k is 3. In certain embodiments, k is 4. In certain embodiments, k is 5.
[0096] In certain embodiments, k is 1; and RA is halogen, substituted or unsubstituted alkyl, or -ORA1. In certain embodiments, k is 1; and RA is halogen, substituted or
unsubstituted C1-6 alkyl, or -ORA1, wherein RA1 is substituted or unsubstituted C1-6 alkyl. In certain embodiments, k is 2; and each instance of RA is independently halogen, substituted or unsubstituted alkyl, or -ORA1. In certain embodiments, k is 2; and each instance of RA is independently halogen, substituted or unsubstituted C1-6 alkyl, or -ORA1, wherein each instance of RA1 is independently substituted or unsubstituted C1-6 alkyl. In certain
embodiments, k is 1; and RA is halogen, substituted or unsubstituted alkyl, -ORA1, substituted or unsubstituted acyl, -CN, -N02, -S(=0)2RA1, -Si(RA1)4, or -P(=0)(ORA1)2. In certain embodiments, k is 1; and RA is halogen, substituted or unsubstituted C1-6 alkyl, -ORA1, - C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2, -CN, -N02, -S(=0)2RA1, -Si(RA1)4, or - P(=0)(ORA1)2, wherein each instance of RA1 is independently hydrogen, substituted or unsubstituted C1-6 alkyl, or substituted or unsubstituted phenyl. In certain embodiments, k is 2; and each instance of RA is independently halogen, substituted or unsubstituted alkyl, - ORA1, substituted or unsubstituted acyl, -CN, -N02, -S(=0)2RA1, -Si(RA1)4, or - P(=0)(ORA1)2. In certain embodiments, k is 2; and each instance of RA is independently halogen, substituted or unsubstituted C1-6 alkyl, -ORA1, -C(=0)RA1, -C(=0)ORA1, - C(=0)N(RA1)2, -CN, -N02, -S(=0)2RA1, -Si(RA1)4, or -P(=0)(ORA1)2, wherein each instance of RA1 is independently hydrogen, substituted or unsubstituted C1-6 alkyl, or substituted or unsubstituted phenyl.
[0097] Exemplary compounds of Formula (A) include, but are not limited to, compounds of any one of the formulae:
lr Is It lu
lv lw lx,
and salts thereof.
[0098] The methods of the invention also involves an imidating agent, such as an N- fluorosulfonimide, e.g., a compound of Formula (B), or a salt thereof. Compounds of
Formula (B) may include one or more substituents R . In certain embodiments, at least one instance of R B is H. In certain embodiments, at least one instance of R B is halogen. In certain embodiments, at least one instance of R is F. In certain embodiments, at least one instance of R B is CI. In certain embodiments, at least one instance of R B is Br. In certain embodiments, at least one instance of R B is I (iodine). In certain embodiments, at least one instance of R B is substituted acyl. In certain embodiments, at least one instance of R is unsubstituted acyl. In
certain embodiments, at least one instance of R is substituted alkyl. In certain embodiments, at least one instance of R is unsubstituted alkyl. In certain embodiments, at least one instance of R B is C1-12 alkyl. In certain embodiments, at least one instance of R B is substituted Ci-6 alkyl. In certain embodiments, at least one instance of R is unsubstituted C1-6 alkyl. In certain embodiments, at least one instance of R is substituted methyl. In certain
embodiments, at least one instance of R is -CH2F. In certain embodiments, at least one instance of R B is -CHF2. In certain embodiments, at least one instance of R B is -CF3. In certain embodiments, at least one instance of R is Bn. In certain embodiments, at least one instance of R B is unsubstituted methyl. In certain embodiments, at least one instance of R B is ethyl. In certain embodiments, at least one instance of R is propyl. In certain embodiments, at least one instance of R B is butyl. In certain embodiments, at least one instance of R B is pentyl. In certain embodiments, at least one instance of R is hexyl. In certain embodiments, at least one instance of R is substituted alkenyl. In certain embodiments, at least one instance of R B is unsubstituted alkenyl. In certain embodiments, at least one instance of R B is substituted or unsubstituted C1-6 alkenyl. In certain embodiments, at least one instance of R is vinyl. In certain embodiments, at least one instance of R is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R B is ethynyl. In certain embodiments, at least one instance of R B is substituted carbocyclyl. In certain embodiments, at least one instance of R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R is saturated carbocyclyl. In certain embodiments, at least one instance of R is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of
R B is monocyclic carbocyclyl. In certain embodiments, at least one instance of R B is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl. In certain embodiments, at least one instance of R is cyclopentyl. In certain embodiments, at least one instance of R B is cyclohexyl. In certain embodiments, at least one instance of R B is cycloheptyl. In certain embodiments, at least one instance of R is bicyclic carbocyclyl. In certain embodiments, at least one instance of R is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is unsubstituted heterocyclyl. In certain
embodiments, at least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl. In certain embodiments, at least one
instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heterocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R B is bicyclic heterocyclyl. In certain embodiments, at least one instance of R B is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R is substituted aryl. In certain embodiments, at least one instance of R is unsubstituted aryl. In certain embodiments, at least one instance of R is 6- to 14-membered aryl. In certain embodiments, at least one instance of R is 6- to 10-membered aryl. In certain embodiments, at least one instance of R is substituted phenyl. In certain embodiments, at least one instance of R B is unsubstituted phenyl. In certain embodiments, at least one instance of R B is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R is substituted heteroaryl. In certain embodiments, at least one instance of R is unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl. In certain embodiments, at least one instance of R is 5-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is 6-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is substituted pyridyl. In certain embodiments, at least one instance of R is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R is 9-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R B is -OR B 1. In certain embodiments, at least one instance of R B is -OR B 1 , wherein R B 1 is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R B is -OMe. In certain embodiments, at least one instance of R B is -OEt. In certain embodiments, at least one instance of R is -OPr. In certain embodiments, at least one instance of R B is -OBu. In certain embodiments, at least one instance of R B is -O(pentyl). In certain embodiments, at least one instance of R is -O(hexyl). In certain embodiments, at
least one instance of R is -OBn. In certain embodiments, at least one instance of R is - OPh. In certain embodiments, at least one instance of R is -OH. In certain embodiments, at least one instance of R B is -SR B 1. In certain embodiments, at least one instance of R B is - SMe. In certain embodiments, at least one instance of R is -SH. In certain embodiments, at least one instance of R B is -N(R B 1 )2. In certain embodiments, at least one instance of R B is - NMe2. In certain embodiments, at least one instance of R is -NH2. In certain embodiments, at least one instance of R B is -CN. In certain embodiments, at least one instance of R B is - SCN. In certain embodiments, at least one instance of RB is -C(=NRB1)RB1, -C(=NRB1)ORB1, or -C(=NRB1)N(RB1)2. In certain embodiments, at least one instance of RB is -C(=0)RB1. In certain embodiments, at least one instance of R B is -C(=0)R B 1 , wherein R B 1 is substituted or unsubstituted alkyl or substituted or unsubstituted aryl. In certain embodiments, at least one instance of R B is -C(=0)R B 1 , wherein R B 1 is substituted or unsubstituted C1-6 alkyl or substituted or unsubstituted phenyl. In certain embodiments, at least one instance of R is -
C(=0)OR B 1. In certain embodiments, at least one instance of R B is -C(=0)OR B 1 , wherein R B 1 is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is - C(=0)N(RB1)2. In certain embodiments, at least one instance of RB is -C(=0)N(RB1)2, wherein each instance of R B 1 is independently selected from the group consisting of hydrogen, substituted or unsubstituted alkyl, or a nitrogen protecting group. In certain embodiments, at least one instance of R B is -C(=0)N(R B 1 )2, wherein each instance of R B 1 is independently selected from the group consisting of hydrogen, substituted or unsubstituted C1-6 alkyl, Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts. In certain embodiments, at least one instance of R is -C(=0)NH2. In certain embodiments, at least one instance of R B is -N02. In certain embodiments, at least one instance of R B is
-NRB1C(=0)RB1, -NRB1C(=0)ORB1, or -NRB1C(=0)N(RB1)2. In certain embodiments, at least one instance of RB is -OC(=0)RB1, -OC(=0)ORB1, or -OC(=0)N(RB1)2.
[0099] In certain embodiments, two RB groups may be joined to form a substituted or unsubstituted carbocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated carbocyclic ring. In certain embodiments, two instances of R are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic carbocyclic ring. In certain embodiments, two instances of R are joined to form a 3-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 4-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 5-membered carbocyclic ring. In certain embodiments, two
instances of R are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 7-membered carbocyclic ring. In certain
embodiments, two instances of R are joined to form a 5- to 13-membered, bicyclic carbocyclic ring.
[00100] In certain embodiments, two instances of RB are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00101] In certain embodiments, two instances of RB are joined to form a substituted or unsubstituted aryl ring. In certain embodiments, two instances of R are joined to form a 6- to 14-membered aryl ring. In certain embodiments, two instances of R are joined to form a 6- to 10-membered aryl ring. In certain embodiments, two instances of R are joined to form a monocyclic aryl ring. In certain embodiments, two instances of R are joined to form an unsubstituted phenyl ring. In certain embodiments, two instances of R are joined to form a substituted phenyl ring. In certain embodiments, two instances of R are joined to form a bicyclic aryl ring. In certain embodiments, two instances of R are joined to form a naphthyl ring.
[00102] In certain embodiments, two instances of RB are joined to form a substituted or unsubstituted heteroaryl ring. In certain embodiments, two instances of R are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a substituted or unsubstituted pyridyl ring. In certain embodiments, two instances of R are joined to form a 9- or 10-membered, bicyclic heteroaryl ring, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[00103] In certain embodiments, each instance of R is H. In certain embodiments, at least one instance of R is halogen or substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is halogen or substituted or unsubstituted C1-6 alkyl.
[00104] In certain embodiments, at least one instance of RB1 is H. In certain embodiments, at least one instance of R B 1 is substituted acyl. In certain embodiments, at least one instance of R B 1 is unsubstituted acyl. In certain embodiments, at least one instance of R B 1 is acetyl. In certain embodiments, at least one instance of R B 1 is substituted alkyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkyl. In certain embodiments, at least one instance of R B 1 is C1-12 alkyl. In certain embodiments, at least one instance of R B 1 is C1-6 alkyl. In certain embodiments, at least one instance of R B 1 is methyl. In certain embodiments, at least one instance of R B 1 is ethyl. In certain embodiments, at least one instance of R B 1 is propyl. In certain embodiments, at least one instance of R B 1 is butyl. In certain embodiments, at least one instance of R B 1 is pentyl. In certain embodiments, at least one instance of R B 1 is hexyl. In certain embodiments, at least one instance of R B 1 is substituted alkenyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkenyl. In certain embodiments, at least one instance of R B 1 is vinyl. In certain embodiments, at least one instance of R B 1 is substituted alkynyl. In certain embodiments, at least one instance of R B 1 is unsubstituted alkynyl. In certain embodiments, at least one instance of R B 1 is ethynyl. In certain
embodiments, at least one instance of R B 1 is substituted carbocyclyl. In certain embodiments, at least one instance of R B 1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R B 1 is saturated carbocyclyl. In certain embodiments, at least one instance of R B 1 is unsaturated carbocyclyl. In certain embodiments, at least one instance of R B 1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R B 1 is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R B 1 is cylcopropyl. In certain embodiments, at least one instance of R B 1 is cyclobutyl. In certain embodiments, at least one instance of R B 1 is cyclopentyl. In certain embodiments, at least one instance of R B 1 is cyclohexyl. In certain embodiments, at least one instance of R B 1 is cycloheptyl. In certain embodiments, at least one instance of R B 1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R B 1 is substituted heterocyclyl. In certain embodiments, at least one instance of R B 1 is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R B 1 is saturated heterocyclyl. In certain embodiments, at least one instance of R B 1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R B 1 is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments,
at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R B 1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R B 1 is 5- to 13- membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R B 1 is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R B 1 is 6- to
14-membered aryl. In certain embodiments, at least one instance of R B 1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R B 1 is monocyclic aryl. In certain embodiments, at least one instance of R B 1 is unsubstituted phenyl. In certain embodiments, at least one instance of R B 1 is substituted phenyl. In certain embodiments, at least one instance of R B 1 is bicyclic aryl. In certain embodiments, at least one instance of R B 1 is naphthyl. In certain embodiments, at least one instance of R B 1 is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of R B 1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R B 1 is pyridyl. In certain embodiments, at least one instance of R B 1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R B 1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R B 1 is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least one instance of R B 1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom. In certain embodiments, R B 1 is an oxygen protecting group when attached to an oxygen atom. In certain embodiments, R B 1 is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ί-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom. In certain embodiments, R B 1 is a sulfur protecting group when attached to a sulfur atom. In certain embodiments, R B 1 is acetamidomethyl, ί-Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
[00105] In certain embodiments, two instances of RB1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R B 1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
R B 1 are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R B 1 are joined to form
a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R B 1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R B 1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00106] In certain embodiments, at least one instance of m is 0. In certain embodiments, at least one instance of m is 1. In certain embodiments, at least one instance of m is 2. In certain embodiments, at least one instance of m is 3. In certain embodiments, at least one instance of m is 4. In certain embodiments, at least one instance of m is 5. In certain embodiments, both instances of m are 0. In certain embodiments, both instances of m are 1. In certain
embodiments, both instances of m are 2. In certain embodiments, both instances of m are 3. In certain embodiments, both instances of m are 4. In certain embodiments, both instances of m are 5.
[00107] In certain embodiments, a compound of Formula (B) is commercially available. An exemplary compound of Formula (B) is N-fluorobenzenesulfonimide (NFBS or NFSI).
[00108] The methods of the invention involve a catalyst. In certain embodiments, the catalyst employed in the inventive methods includes a transition metal complex, such as a complex of any one of Formulae (CI) to (C25). Complexes of any one of Formulae (CI) to (C25) include a transition metal ion M. In certain embodiments, M is a transition metal cation. In certain embodiments, M is a monovalent transition metal ion. In certain
embodiments, M is Ag+, Cu+, or Co+. In certain embodiments, M is a divalent transition metal ion. In certain embodiments, M is Pd2+, Ni2+, Cu2+, Ir2+, Zn2+, Mn2+, or Fe2+. In certain embodiments, M is a trivalent transition metal ion. In certain embodiments, M is Rh3+. In certain embodiments, M is a tetravalent, pentavalent, or hexavalent transition metal ion.
[00109] Complexes of Formula (CI) include at least two instances of Ring B. In certain embodiments, all instances of Ring B are independently substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of Ring B is monocyclic, 5- membered heteroaryl, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain
n certain , or
e formula:
. In certain embodiments at least one instance of Ring B is of the formula:
e instance of Ring B is of the formula: , or embodiments, at least one instance of Ring B is of the formula:
, 5 or In certain embodiments at least one instance of
or R . In certain embodiments, at least one instance of Ring B is of the
formula:
one instance of Ring B is of the formula:
. In certain embodiments, at least one instance of Ring B is monocyclic, 6-membered heteroaryl, wherein one, two, three, or four
atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of Ring B is of the
formula: . In certain embodiments, at least one instance of Ring B is of the
formula:
. In certain embodiments, at least one instance of Ring B is of the formula:
, (RC,' , , or . In certain
certain embodiments, each instance of Ring B is substituted or unsubstituted, monocyclic, 5- or 6-membered heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of Ring B is bicyclic, 9- or 10-membered heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[00110] Complexes of Formula (CI) may include one or more substituents Rc. In certain embodiments, at least one instance of R is H. In certain embodiments, at least one instance of R c is halogen. In certain embodiments, at least one instance of R c is F. In certain embodiments, at least one instance of R is CI. In certain embodiments, at least one instance of R c is Br. In certain embodiments, at least one instance of R c is I (iodine). In certain embodiments, at least one instance of R is substituted acyl. In certain embodiments, at least one instance of R c is unsubstituted acyl. In certain embodiments, at least one instance of R c is substituted alkyl. In certain embodiments, at least one instance of R is unsubstituted alkyl.
In certain embodiments, at least one instance of R is C1-12 alkyl. In certain embodiments, at least one instance of R is substituted C1-6 alkyl. In certain embodiments, at least one instance of R c is unsubstituted C1-6 alkyl. In certain embodiments, at least one instance of R c is substituted methyl. In certain embodiments, at least one instance of R is -CH2F. In certain
embodiments, at least one instance of R is -CHF2. In certain embodiments, at least one instance of R c is -CF3. In certain embodiments, at least one instance of R c is Bn. In certain embodiments, at least one instance of R is unsubstituted methyl. In certain embodiments, at least one instance of R c is ethyl. In certain embodiments, at least one instance of R c is propyl.
In certain embodiments, at least one instance of R is butyl. In certain embodiments, at least one instance of R c is pentyl. In certain embodiments, at least one instance of R c is hexyl. In certain embodiments, at least one instance of R is substituted alkenyl. In certain
embodiments, at least one instance of R is unsubstituted alkenyl. In certain embodiments, at least one instance of R is substituted or unsubstituted C1-6 alkenyl. In certain embodiments, at least one instance of R c is vinyl. In certain embodiments, at least one instance of R c is substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R is ethynyl. In certain
embodiments, at least one instance of R is substituted carbocyclyl. In certain embodiments, at least one instance of R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R c is saturated carbocyclyl. In certain embodiments, at least one instance of R c is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R is monocyclic carbocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R c is cyclobutyl. In certain embodiments, at least one instance of R c is
cyclopentyl. In certain embodiments, at least one instance of R is cyclohexyl. In certain embodiments, at least one instance of R is cycloheptyl. In certain embodiments, at least one instance of R c is bicyclic carbocyclyl. In certain embodiments, at least one instance of R c is bicyclic, 5- to 13-membered carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is
unsubstituted heterocyclyl. In certain embodiments, at least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl.
In certain embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
In certain embodiments, at least one instance of R is monocyclic heterocyclyl. In certain embodiments, at least one instance of R is monocylic, 3- to 7-membered heterocyclyl. In
certain embodiments, at least one instance of R is bicyclic heterocyclyl. In certain embodiments, at least one instance of R is bicyclic, 5- to 13-membered heterocyclyl. In certain embodiments, at least one instance of R is substituted aryl. In certain embodiments, at least one instance of R is unsubstituted aryl. In certain embodiments, at least one instance of R c is 6- to 14-membered aryl. In certain embodiments, at least one instance of R c is 6- to
10-membered aryl. In certain embodiments, at least one instance of R is substituted phenyl.
In certain embodiments, at least one instance of R is unsubstituted phenyl. In certain embodiments, at least one instance of R is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R c is substituted heteroaryl. In certain embodiments, at least one instance of R c is unsubstituted heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl. In certain embodiments, at least one instance of R is monocyclic, 5-membered heteroaryl. In certain embodiments, at least one instance of R is monocyclic, 6-membered heteroaryl. In certain embodiments, at least one instance of R c is substituted pyridyl. In certain embodiments, at least one instance of R c is unsubstituted 2-pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R is bicyclic, 9-membered heteroaryl. In certain embodiments, at least one instance of R is bicyclic, 10-membered heteroaryl. In certain embodiments, at least one instance of R C is -OR CI . In certain embodiments, at least one instance of R C is -OR CI , wherein R CI is substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is -OMe. In certain embodiments, at least one instance of R c is -OEt. In certain embodiments, at least one instance of R c is -OPr. In certain embodiments, at least one instance of R is -OBu. In certain embodiments, at least one instance of R c is -O(pentyl). In certain embodiments, at least one instance of R c is -
O(hexyl). In certain embodiments, at least one instance of R is -OBn. In certain
embodiments, at least one instance of R is -OPh. In certain embodiments, at least one instance of R C is -OH. In certain embodiments, at least one instance of R C is -SR CI . In certain embodiments, at least one instance of R is -SMe. In certain embodiments, at least one instance of R C is -SH. In certain embodiments, at least one instance of R C is -N(R CI )2. In certain embodiments, at least one instance of R is -NMe2. In certain embodiments, at least
one instance of R c is -NH2. In certain embodiments, at least one instance of R c is -CN. In certain embodiments, at least one instance of R is -SCN. In certain embodiments, at least one instance of Rc is -C(=NRC1)RC1, -C(=NRcl)ORcl, or -C(=NRC1)N(RC1)2. In certain embodiments, at least one instance of R C is -C(=0)R CI . In certain embodiments, at least one instance of R C is -C(=0)OR CI . In certain embodiments, at least one instance of R c is -
C(=0)N(R CI )2. In certain embodiments, at least one instance of R c is -N02. In certain embodiments, at least one instance of Rc is -NRclC(=0)Rcl, -NRclC(=0)ORcl, or - NRclC(=0)N(Rcl)2. In certain embodiments, at least one instance of Rc is -OC(=0)Rcl, - OC(=0)ORcl, or -OC(=0)N(Rcl)2.
[00111] In certain embodiments, two Rc groups may be joined to form a substituted or unsubstituted carbocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated carbocyclic ring. In certain embodiments, two instances of R are joined to form a carbocyclic ring including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic carbocyclic ring. In certain embodiments, two instances of R are joined to form a 3-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 4-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 5-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 6-membered carbocyclic ring. In certain embodiments, two instances of R are joined to form a 7-membered carbocyclic ring. In certain
embodiments, two instances of R are joined to form a 5- to 13-membered, bicyclic carbocyclic ring.
[00112] In certain embodiments, two instances of Rc are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00113] In certain embodiments, two instances of Rc are joined to form a substituted or unsubstituted aryl ring. In certain embodiments, two instances of R are joined to form a 6- to
14-membered aryl ring. In certain embodiments, two instances of R are joined to form a 6- to 10-membered aryl ring. In certain embodiments, two instances of R are joined to form a monocyclic aryl ring. In certain embodiments, two instances of R are joined to form an unsubstituted phenyl ring. In certain embodiments, two instances of R are joined to form a substituted phenyl ring. In certain embodiments, two instances of R are joined to form a bicyclic aryl ring. In certain embodiments, two instances of R are joined to form a naphthyl ring.
[00114] In certain embodiments, two instances of Rc are joined to form a substituted or unsubstituted heteroaryl ring. In certain embodiments, two instances of R are joined to form a 5- or 6-membered, monocyclic heteroaryl ring, wherein one, two, or three atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a substituted or unsubstituted pyridyl ring. In certain embodiments, two instances of R are joined to form a 9- or 10-membered, bicyclic heteroaryl ring, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[00115] In certain embodiments, each instance of Rc is H. In certain embodiments, at least one instance of R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR CI . In certain embodiments, at least one instance of R c is halogen, substituted or unsubstituted C1-6 alkyl, substituted or unsubstituted phenyl, or -OR CI , wherein R CI is substituted or unsubstituted C1-6 alkyl.
[00116] In certain embodiments, at least one instance of R C 1 is H. In certain embodiments, at least one instance of R C 1 is substituted acyl. In certain embodiments, at least one instance of R CI is unsubstituted acyl. In certain embodiments, at least one instance of R CI is acetyl. In certain embodiments, at least one instance of R CI is substituted alkyl. In certain embodiments, at least one instance of R C 1 is unsubstituted alkyl. In certain embodiments, at least one instance of R CI is C1-12 alkyl. In certain embodiments, at least one instance of R CI is C1-6 alkyl. In certain embodiments, at least one instance of R CI is methyl. In certain embodiments, at least one instance of R CI is ethyl. In certain embodiments, at least one instance of R CI is propyl. In certain embodiments, at least one instance of R C 1 is butyl. In certain embodiments, at least one instance of R CI is pentyl. In certain embodiments, at least one instance of R CI is hexyl. In certain embodiments, at least one instance of R C 1 is substituted alkenyl. In certain embodiments, at least one instance of R CI is unsubstituted alkenyl. In certain embodiments, at least one instance of R CI is vinyl. In certain embodiments, at least one instance of R CI is
substituted alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R CI is ethynyl. In certain
embodiments, at least one instance of R CI is substituted carbocyclyl. In certain embodiments, at least one instance of R C 1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R CI is saturated carbocyclyl. In certain embodiments, at least one instance of R CI is unsaturated carbocyclyl. In certain embodiments, at least one instance of R C 1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R CI is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R CI is cylcopropyl. In certain embodiments, at least one instance of R CI is cyclobutyl. In certain embodiments, at least one instance of R CI is cyclopentyl. In certain embodiments, at least one instance of R CI is cyclohexyl. In certain embodiments, at least one instance of R CI is cycloheptyl. In certain embodiments, at least one instance of R C 1 is bicyclic, 5- to 13-membered carbocyclyl. In certain embodiments, at least one instance of R CI is substituted heterocyclyl. In certain embodiments, at least one instance of R CI is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R CI is saturated heterocyclyl. In certain embodiments, at least one instance of R C 1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of R CI is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of R C 1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R C 1 is monocyclic, 3- to
7-membered heterocyclyl. In certain embodiments, at least one instance of R CI is bicyclic, 5- to 13-membered heterocyclyl. In certain embodiments, at least one instance of R C 1 is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R CI is 6- to
14-membered aryl. In certain embodiments, at least one instance of R C 1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of R C 1 is monocyclic aryl. In certain embodiments, at least one instance of R CI is unsubstituted phenyl. In certain embodiments, at least one instance of R CI is substituted phenyl. In certain embodiments, at least one instance of R CI is bicyclic aryl. In certain embodiments, at least one instance of R CI is naphthyl. In certain embodiments, at least one instance of R CI is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of R CI is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are
independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R CI is pyridyl. In certain embodiments, at least one
instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one
C 1
instance of R is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of
C 1 nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least
CI
one instance of R is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts
CI
when attached to a nitrogen atom. In certain embodiments, R is an oxygen protecting group
CI
when attached to an oxygen atom. In certain embodiments, R is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ί-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to
CI
an oxygen atom. In certain embodiments, R is a sulfur protecting group when attached to a
CI
sulfur atom. In certain embodiments, R is acetamidomethyl, ί-Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
C 1
[00117] In certain embodiments, two instances of R are joined to form a substituted or
CI
unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
C 1
R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in
C 1
the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain
CI
embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic
CI
heterocyclic ring. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00118] In certain embodiments, at least one instance of r is 0. In certain embodiments, at least one instance of r is 1. In certain embodiments, at least one instance of r is 2. In certain embodiments, at least one instance of r is 3. In certain embodiments, at least one instance of r is 4. In certain embodiments, at least one instance of r is 5. In certain embodiments, each instance of r is 0. In certain embodiments, each instance of r is 1. In certain embodiments, each instance of r is 2. In certain embodiments, each instance of r is 3. In certain
embodiments, each instance of r is 4. In certain embodiments, each instance of r is 5.
[00119] In certain embodiments, at least one instance of r is 1 ; and at least one instance of
C CI
R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR . In certain embodiments, at least one instance of r is 1 ; and at least one instance of R is halogen, substituted or unsubstituted C . alkyl, substituted or unsubstituted phenyl, or -
OR CI , wherein R CI is substituted or unsubstituted C1-6 alkyl. In certain embodiments, each instance of r is 1; and each instance of R is halogen, substituted or unsubstituted alkyl, substituted or unsubstituted aryl, or -OR C 1. In certain embodiments, each instance of r is 1; and each instance of R is halogen, substituted or unsubstituted C1-6 alkyl, substituted or unsubstituted phenyl, or -OR CI , wherein R CI is substituted or unsubstituted C1-6 alkyl.
[00120] Complexes of Formula (CI) or (C2) include more than one substituent RD. In certain embodiments, at least one instance of RD is substituted acyl. In certain embodiments, at least one instance of RD is unsubstituted acyl. In certain embodiments, at least one instance of RD is substituted alkyl. In certain embodiments, at least one instance of RD is unsubstituted alkyl. In certain embodiments, at least one instance of RD is C1-12 alkyl. In certain
embodiments, at least one instance of RD is substituted C1-6 alkyl. In certain embodiments, at least one instance of RD is unsubstituted C1-6 alkyl. In certain embodiments, at least one instance of RD is substituted methyl. In certain embodiments, at least one instance of RD is - CH2F. In certain embodiments, at least one instance of RD is -CHF2. In certain embodiments, at least one instance of RD is -CF3. In certain embodiments, at least one instance of RD is Bn. In certain embodiments, at least one instance of RD is unsubstituted methyl. In certain embodiments, at least one instance of RD is ethyl. In certain embodiments, at least one instance of RD is propyl. In certain embodiments, at least one instance of RD is butyl. In certain embodiments, at least one instance of RD is pentyl. In certain embodiments, at least one instance of RD is hexyl. In certain embodiments, at least one instance of RD is substituted alkenyl. In certain embodiments, at least one instance of RD is unsubstituted alkenyl. In certain embodiments, at least one instance of RD is substituted or unsubstituted C1-6 alkenyl. In certain embodiments, at least one instance of RD is vinyl. In certain embodiments, at least one instance of RD is substituted alkynyl. In certain embodiments, at least one instance of RD is unsubstituted alkynyl. In certain embodiments, at least one instance of RD is ethynyl. In certain embodiments, at least one instance of RD is substituted carbocyclyl. In certain embodiments, at least one instance of RD is unsubstituted carbocyclyl. In certain
embodiments, at least one instance of RD is saturated carbocyclyl. In certain embodiments, at least one instance of RD is unsaturated carbocyclyl. In certain embodiments, at least one instance of RD is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of RD is monocyclic carbocyclyl. In certain embodiments, at least one instance of RD is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of RD is cylcopropyl. In certain embodiments, at least one instance of RD is cyclobutyl. In certain embodiments, at
least one instance of R is cyclopentyl. In certain embodiments, at least one instance of R is cyclohexyl. In certain embodiments, at least one instance of RD is cycloheptyl. In certain embodiments, at least one instance of RD is bicyclic carbocyclyl. In certain embodiments, at least one instance of RD is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of RD is substituted heterocyclyl. In certain embodiments, at least one instance of RD is unsubstituted heterocyclyl. In certain embodiments, at least one instance of RD is saturated heterocyclyl. In certain embodiments, at least one instance of RD is unsaturated heterocyclyl. In certain embodiments, at least one instance of RD is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of RD is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RD is monocyclic heterocyclyl. In certain embodiments, at least one instance of RD is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of RD is bicyclic heterocyclyl. In certain embodiments, at least one instance of RD is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of RD is substituted aryl. In certain embodiments, at least one instance of RD is unsubstituted aryl. In certain
embodiments, at least one instance of RD is 6- to 14-membered aryl. In certain embodiments, at least one instance of RD is 6- to 10-membered aryl. In certain embodiments, at least one instance of RD is substituted phenyl. In certain embodiments, at least one instance of RD is unsubstituted phenyl. In certain embodiments, at least one instance of RD is substituted naphthyl. In certain embodiments, at least one instance of RD is unsubstituted naphthyl. In certain embodiments, at least one instance of RD is substituted heteroaryl. In certain embodiments, at least one instance of RD is unsubstituted heteroaryl. In certain embodiments, at least one instance of RD is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RD is monocyclic heteroaryl. In certain embodiments, at least one instance of RD is 5-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of RD is 6-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of RD is substituted pyridyl. In certain embodiments, at least one instance of RD is unsubstituted 2-pyridyl, unsubstituted 3- pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of RD is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of
R is 9-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of RD is a nitrogen protecting group. In certain embodiments, at least one instance of RD is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts.
[00121] In certain embodiments, two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of RD are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of RD are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of RD are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RD are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of RD are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RD are joined to form a
heterocyclic ring of
5 wherein the heterocyclic ring is substituted or unsubstituted. In certain embodiments, two instances of R1 are joined to form a 5- to 13-membered, bicyclic heterocyclic ring. In certain embodiments, two instances of RD are joined to form a substituted or unsubstituted, 7- to 11-membered, bicyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments,
two instances of R are joined to form a heterocyclic ring of the formula:
, wherein the heterocyclic ring is substituted or unsubstituted.
[00122] In certain embodiments, at least one instance of RD is H, substituted or
unsubstituted alkyl, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, at least one instance of RD is H, substituted or
unsubstituted C1-6 alkyl, or two instances of R are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, each instance of RD is H, substituted or unsubstituted alkyl, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, each instance of RD is H, substituted or
unsubstituted C1-6 alkyl, or two instances of RD are joined to form a substituted or
unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[00123] In certain embodiments, at least one instance of RD1 is H. In certain embodiments, at least one instance of RD1 is substituted acyl. In certain embodiments, at least one instance of RD1 is unsubstituted acyl. In certain embodiments, at least one instance of RD1 is acetyl. In certain embodiments, at least one instance of RD1 is substituted alkyl. In certain
embodiments, at least one instance of RD1 is unsubstituted alkyl. In certain embodiments, at least one instance of RD1 is C1-12 alkyl. In certain embodiments, at least one instance of RD1 is Ci-6 alkyl. In certain embodiments, at least one instance of RD1 is methyl. In certain embodiments, at least one instance of RD1 is ethyl. In certain embodiments, at least one instance of RD1 is propyl. In certain embodiments, at least one instance of RD1 is butyl. In certain embodiments, at least one instance of RD1 is pentyl. In certain embodiments, at least one instance of RD1 is hexyl. In certain embodiments, at least one instance of RD1 is substituted alkenyl. In certain embodiments, at least one instance of RD1 is unsubstituted alkenyl. In certain embodiments, at least one instance of RD1 is vinyl. In certain
embodiments, at least one instance of RD1 is substituted alkynyl. In certain embodiments, at least one instance of RD1 is unsubstituted alkynyl. In certain embodiments, at least one instance of RD1 is ethynyl. In certain embodiments, at least one instance of RD1 is substituted carbocyclyl. In certain embodiments, at least one instance of RD1 is unsubstituted carbocyclyl. In certain embodiments, at least one instance of RD1 is saturated carbocyclyl. In certain embodiments, at least one instance of RD1 is unsaturated carbocyclyl. In certain embodiments, at least one instance of RD1 is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of RD1 is 3- to 7- membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of RD1 is
cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl. In certain embodiments, at least one instance of RD1 is cyclopentyl. In certain embodiments, at least one instance of RD1 is cyclohexyl. In certain embodiments, at least one instance of RD1 is cycloheptyl. In certain embodiments, at least one instance of RD1 is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of RD1 is substituted heterocyclyl. In certain embodiments, at least one instance of RD1 is unsubstituted
heterocyclyl. In certain embodiments, at least one instance of RD1 is saturated heterocyclyl. In certain embodiments, at least one instance of RD1 is unsaturated heterocyclyl. In certain embodiments, at least one instance of RD1 is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of RD1 is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RD1 is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of RD1 is 5- to 13-membered, bicyclic
heterocyclyl. In certain embodiments, at least one instance of RD1 is substituted or
unsubstituted aryl. In certain embodiments, at least one instance of RD1 is 6- to 14-membered aryl. In certain embodiments, at least one instance of RD1 is 6- to 10-membered aryl. In certain embodiments, at least one instance of RD1 is monocyclic aryl. In certain embodiments, at least one instance of RD1 is unsubstituted phenyl. In certain embodiments, at least one instance of RD1 is substituted phenyl. In certain embodiments, at least one instance of RD1 is bicyclic aryl. In certain embodiments, at least one instance of RD1 is naphthyl. In certain embodiments, at least one instance of RD1 is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of RD1 is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RD1 is pyridyl. In certain embodiments, at least one instance of RD1 is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of RD1 is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of RD1 is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least one instance of RD1 is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom. In certain embodiments, RD1 is an oxygen protecting group when
attached to an oxygen atom. In certain embodiments, R is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, i-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom. In certain embodiments, RD1 is a sulfur protecting group when attached to a sulfur atom. In certain embodiments, RD1 is acetamidomethyl, ί-Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
[00124] In certain embodiments, two instances of RD1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of RD1 are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of RD1 are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of RD1 are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of RD1 are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of RD1 are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00125] Complexes of Formula (CI) or (C2) may include one or more substituent RE. In p
certain embodiments, at least one instance of R is absent. In certain embodiments, at least one instance of R E is H. In certain embodiments, at least one instance of R E is substituted p
acyl. In certain embodiments, at least one instance of R is unsubstituted acyl. In certain
p
embodiments, at least one instance of R is substituted alkyl. In certain embodiments, at least one instance of R E is unsubstituted alkyl. In certain embodiments, at least one instance of R E p
is C1-12 alkyl. In certain embodiments, at least one instance of R is substituted C1-6 alkyl. In p
certain embodiments, at least one instance of R is unsubstituted C1-6 alkyl. In certain
p
embodiments, at least one instance of R is substituted methyl. In certain embodiments, at least one instance of R E is -CH2F. In certain embodiments, at least one instance of R E is - p
CHF2. In certain embodiments, at least one instance of R is -CF3. In certain embodiments, at least one instance of R E is Bn. In certain embodiments, at least one instance of R E is
p
unsubstituted methyl. In certain embodiments, at least one instance of R is ethyl. In certain p
embodiments, at least one instance of R is propyl. In certain embodiments, at least one instance of R E is butyl. In certain embodiments, at least one instance of R E is pentyl. In
p
certain embodiments, at least one instance of R is hexyl. In certain embodiments, at least one instance of R E is substituted alkenyl. In certain embodiments, at least one instance of R E p
is unsubstituted alkenyl. In certain embodiments, at least one instance of R is substituted or p
unsubstituted Ci_6 alkenyl. In certain embodiments, at least one instance of R is substituted
alkynyl. In certain embodiments, at least one instance of R is unsubstituted alkynyl. In certain embodiments, at least one instance of R is ethynyl. In certain embodiments, at least one instance of R is substituted carbocyclyl. In certain embodiments, at least one instance of
R is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R is saturated carbocyclyl. In certain embodiments, at least one instance of R is unsaturated carbocyclyl. In certain embodiments, at least one instance of R is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R is monocyclic carbocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R is cylcopropyl. In certain embodiments, at least one instance of R is cyclobutyl. In certain embodiments, at least one instance of R is cyclopentyl. In certain embodiments, at least one instance of R is cyclohexyl. In certain embodiments, at least one instance of R is cycloheptyl. In certain embodiments, at least one instance of R is bicyclic carbocyclyl. In certain embodiments, at least one instance of R is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R is substituted heterocyclyl. In certain embodiments, at least one instance of R is unsubstituted
heterocyclyl. In certain embodiments, at least one instance of R is saturated heterocyclyl. In certain embodiments, at least one instance of R is unsaturated heterocyclyl. In certain embodiments, at least one instance of R is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, at least one instance of
R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heterocyclyl. In certain embodiments, at least one instance of R is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R is bicyclic heterocyclyl. In certain embodiments, at least one instance of R is 5- to 13-membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R is substituted aryl. In certain embodiments, at least one instance of
R is unsubstituted aryl. In certain embodiments, at least one instance of R is 6- to 14- membered aryl. In certain embodiments, at least one instance of R is 6- to 10-membered aryl. In certain embodiments, at least one instance of R is substituted phenyl. In certain embodiments, at least one instance of R is unsubstituted phenyl. In certain embodiments, at least one instance of R is substituted naphthyl. In certain embodiments, at least one instance of R is unsubstituted naphthyl. In certain embodiments, at least one instance of R is substituted heteroaryl. In certain embodiments, at least one instance of R is unsubstituted
heteroaryl. In certain embodiments, at least one instance of R is heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R is monocyclic heteroaryl. In certain embodiments, at least one instance of R is
5-membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is 6- membered, monocyclic heteroaryl. In certain embodiments, at least one instance of R is substituted pyridyl. In certain embodiments, at least one instance of R is unsubstituted 2- pyridyl, unsubstituted 3-pyridyl, or unsubstituted 4-pyridyl. In certain embodiments, at least one instance of R is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R is 9-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is 10-membered, bicyclic heteroaryl. In certain embodiments, at least one instance of R is a nitrogen protecting group. In certain embodiments, at least one instance of
R is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts.
[00126] In certain embodiments, two instances of RE are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
R are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R are joined to form a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R are joined to form a 5- to 13- membered, bicyclic heterocyclic ring. In certain embodiments, two instances of R are joined to form a substituted or unsubstituted, 7- to 11 -membered, bicyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
[00127] In certain embodiments, at least one instance of RE is H or substituted or unsubstituted alkyl. In certain embodiments, at least one instance of R is H or substituted or
unsubstituted C1-6 alkyl. In certain embodiments, each instance of R is H or substituted or unsubstituted alkyl. In certain embodiments, each instance of R is H or substituted or unsubstituted C1-6 alkyl.
[00128] In certain embodiments, at least one instance of RE1 is H. In certain embodiments, at least one instance of R El is substituted acyl. In certain embodiments, at least one instance of R El is unsubstituted acyl. In certain embodiments, at least one instance of R El is acetyl. In certain embodiments, at least one instance of R El is substituted alkyl. In certain embodiments, at least one instance of R El is unsubstituted alkyl. In certain embodiments, at least one instance of R El is C1-12 alkyl. In certain embodiments, at least one instance of R El is C1-6 alkyl. In certain embodiments, at least one instance of R El is methyl. In certain embodiments, at least one instance of R El is ethyl. In certain embodiments, at least one instance of R El is propyl. In certain embodiments, at least one instance of R El is butyl. In certain embodiments, at least one instance of R El is pentyl. In certain embodiments, at least one instance of R El is hexyl. In certain embodiments, at least one instance of R El is substituted alkenyl. In certain embodiments, at least one instance of R El is unsubstituted alkenyl. In certain embodiments, at least one instance of R El is vinyl. In certain embodiments, at least one instance of R El is substituted alkynyl. In certain embodiments, at least one instance of R El is unsubstituted alkynyl. In certain embodiments, at least one instance of R El is ethynyl. In certain
embodiments, at least one instance of R El is substituted carbocyclyl. In certain embodiments, at least one instance of R El is unsubstituted carbocyclyl. In certain embodiments, at least one instance of R El is saturated carbocyclyl. In certain embodiments, at least one instance of R El is unsaturated carbocyclyl. In certain embodiments, at least one instance of R El is carbocyclyl including zero, one, two, or three double bonds in the carbocyclic ring system. In certain embodiments, at least one instance of R El is 3- to 7-membered, monocyclic carbocyclyl. In certain embodiments, at least one instance of R El is cylcopropyl. In certain embodiments, at least one instance of R El is cyclobutyl. In certain embodiments, at least one instance of R El is cyclopentyl. In certain embodiments, at least one instance of R El is cyclohexyl. In certain embodiments, at least one instance of R El is cycloheptyl. In certain embodiments, at least one instance of R El is 5- to 13-membered, bicyclic carbocyclyl. In certain embodiments, at least one instance of R El is substituted heterocyclyl. In certain embodiments, at least one instance of R El is unsubstituted heterocyclyl. In certain embodiments, at least one instance of R El is saturated heterocyclyl. In certain embodiments, at least one instance of R El is unsaturated heterocyclyl. In certain embodiments, at least one instance of R El is heterocyclyl including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments,
at least one instance of R is heterocyclyl, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R El is 3- to 7-membered, monocyclic heterocyclyl. In certain embodiments, at least one instance of R El is 5- to 13- membered, bicyclic heterocyclyl. In certain embodiments, at least one instance of R El is substituted or unsubstituted aryl. In certain embodiments, at least one instance of R El is 6- to
14-membered aryl. In certain embodiments, at least one instance of R El is 6- to 10-membered aryl. In certain embodiments, at least one instance of R El is monocyclic aryl. In certain embodiments, at least one instance of R El is unsubstituted phenyl. In certain embodiments, at least one instance of R El is substituted phenyl. In certain embodiments, at least one instance of R El is bicyclic aryl. In certain embodiments, at least one instance of R El is naphthyl. In certain embodiments, at least one instance of R El is substituted or unsubstituted heteroaryl. In certain embodiments, at least one instance of R El is 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R El is pyridyl. In certain embodiments, at least one instance of R El is bicyclic heteroaryl, wherein the point of attachment may be on any atom of the bicyclic heteroaryl ring system, as valency permits. In certain embodiments, at least one instance of R El is 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, at least one instance of R El is a nitrogen protecting group when attached to a nitrogen atom. In certain embodiments, at least one instance of R El is Bn, Boc, Cbz, Fmoc, trifluoroacetyl, triphenylmethyl, acetyl, or Ts when attached to a nitrogen atom. In certain embodiments, R El is an oxygen protecting group when attached to an oxygen atom. In certain embodiments, R El is silyl, TBDPS, TBDMS, TIPS, TES, TMS, MOM, THP, ί-Bu, Bn, allyl, acetyl, pivaloyl, or benzoyl when attached to an oxygen atom. In certain embodiments, R El is a sulfur protecting group when attached to a sulfur atom. In certain embodiments, R El is acetamidomethyl, ί-Bu, 3-nitro-2-pyridine sulfenyl, 2-pyridine-sulfenyl, or triphenylmethyl when attached to a sulfur atom.
[00129] In certain embodiments, two instances of RE1 are joined to form a substituted or unsubstituted heterocyclic ring. In certain embodiments, two instances of R El are joined to form a saturated or unsaturated heterocyclic ring. In certain embodiments, two instances of
R El are joined to form a heterocyclic ring including zero, one, two, or three double bonds in the heterocyclic ring system. In certain embodiments, two instances of R El are joined to form
a heterocyclic ring, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur. In certain embodiments, two instances of R El are joined to form a 3- to 7-membered, monocyclic heterocyclic ring. In certain embodiments, two instances of R El are joined to form a 5- to 13- membered, bicyclic heterocyclic ring.
[00130] In certain embodiments, at least one instance of n is 0. In certain embodiments, at least one instance of n is 1. In certain embodiments, each instance of n is 0. In certain embodiments, each instance of n is 1.
[00131] In certain embodiments, p is 1. In certain embodiments, p is 2.
[00132] Complexes of any one of Formulae (CI) to (C25) are typically electrically neutral compounds and include one or more anionic counterions X. In certain embodiments, each instance of X is a non-coordinating anionic counterion. In certain embodiments, each instance of X is a monovalent anionic counterion. In certain embodiments, each instance of X is C104
, ΟΤΓ, BF4 , PF4 ~, PF6 ~, or SbF6 ~. In certain embodiments, each instance of X is B[3,5-
(CF3)2C6H3]4r, BPh4 , Al(OC(CF3)3)4 ~, or a carborane anion (e.g. , CBUH12 or
(HCBnMe5Br6D. In certain embodiments, each instance of X is a divalent anionic counterion. In certain embodiments, each instance of X is S04 ".
[00133] In certain embodiments, q is 1. In certain embodiments, q is 2. In certain embodiments, q is 3.
[00134] In certain embodiments, M is a monovalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 1. In certain embodiments, M is a divalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 2. In certain embodiments, M is a trivalent transition metal ion; p is 1; X is a monovalent anionic counterion; and q is 3.
[00135] In certain embodiments, the transition metal complex is of Formula (CI). In certain embodiments, the transition metal complex is of the formula:
[00137] In certain embodiment lex is of the formula:
wherein each instance of X is a monovalent anionic counterion (e.g. , C104 , ΟΤΓ, BF4 , PF4 PF6 , or SbF6 ).
[00138] In certain embodimen lex is of the formula:
[00139] In certain embodimen lex is of the formula:
[00140] In certain embodiments, the transition metal complex is of any one of the formulae:
wherein each instance of X is a monovalent anionic counterion (e.g. , C104 , ΟΤΓ, BF4 , PF4 , PF6 , or SbF6 ).
[00141] Other exemplary complexes of Formula (CI) include, but are not limited to:
[00142] In certain embodiments, the transition metal complex is of Formula (C2). In certain embodiments, the transition metal complex is of the formula:
wherein each instance of X is a monovalent anionic counterion (e.g. , C104 , ΟΤΓ, BF4 , PF4 , PF6 , or SbF6-).
[00143] In certain embodiments, the transition metal complex is of Formula (C3). In certain embodiments, the transition metal complex is of the formula:
wherein each instance of X is a monovalent anionic counterion (e.g. , C104 , ΟΤΓ, BF4 , PF4 , PF6 , or SbF6 ).
[00144] In certain embodiments, the transition metal complex is of the formula:
[00145] In certain embodiments, the transition metal complex is of Formula (C4). In certain embodiments the transition metal complex is of any one of the formulae:
wherein each instance of X is a monovalent anionic counterion (e.g. , C104 , ΟΤΓ, BF4 , PF4 , PF6 , or SbF6 ).
[00146] Exemplary complexes of Formula (C4) include, but are not limited to:
[00147] In certain embodiments, the transition metal complex is of any one of Formulae (C5) to (C25).
[00148] The methods of the invention may involve a single-electron reductant. In certain embodiments, the single-electron reductant is an inorganic single-electron reductant. In certain embodiments, the single-electron reductant is a single-electron reductive, transition metal salt. In certain embodiments, the single-electron reductant is an Ag(I) salt. In certain embodiments, the single-electron reductant is Ag(2,2'-bipyridine)2C104 (i.e. , Ag(bipy)2C104). In certain embodiments, the single-electron reductant is a Ru(II) salt. In certain embodiments, the single-electron reductant is Ru(2,2'-bipyridine)3(PF6)2 (i.e. , Ru(bipy)3(PF6)2). In certain embodiments, the single-electron reductant is an lr(0) complex, Cu(I) salt, Sm(II) salt, Co(II)
salt, Cr(II) salt, Fe(II) salt, Ni(III) salt, Nd(III) salt, Yb(III) salt, or Ti(III) salt. In certain embodiments, the single-electron reductant is Cu(bipy)2PF6, Cu(bipy)2(OTf)2,
Ni(bipy)3(C104)3, Co(bipy)3(C104)3, Fe(bipy)3(C104)2, Ir(2-phenylpyridine)3 (i.e. , Ir(Phpy)3), or Ru(l,10-phenanthroline)3Cl2 (i.e. , Ru(phen)3Cl2) In certain embodiments, the single- electron reductant is an organic single-electron reductant. In certain embodiments, the single- electron reductant is commercially available.
[00149] Complexes of Formula (CI) may be synthesized by contacting a ligand of Formula (Dl), or a salt thereof, with a transition metal salt of Formula (E) to provide the complexes of Formul
(Dl) (E);
wherein:
C D E
Ring B, R , R , R , r, n, M, X, p, and q are as described herein;
each instance of L is independently a ligand molecule; and
s is 0, 1, 2, 3, or 4.
[00150] In certain embodiments, the transition metal salt of Formula (E) is
Pd(MeCN)4(OTf)2.
[00151] Transition metal salts of Formula (E) may include one or more ligand molecules L. In certain embodiments, at least one instance of L is a ligand molecule described herein. In certain embodiments, each instance of L is a ligand molecule described herein. In certain embodiments, at least one instance of L is a molecule of a suitable solvent described herein. In certain embodiments, each instance of L is a molecule of a suitable solvent described herein. In certain embodiments, at least one instance of L is acetonitrile (ACN). In certain embodiments, each instance of L is acetonitrile. In certain embodiments, at least one instance of L is an amide (e.g. , N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMA), or N-methyl-2-pyrrolidone (NMP)). In certain embodiments, at least one instance of L is dimethyl sulfoxide (DMSO). In certain embodiments, at least one instance of L is an ether (e.g. , tetrahydrofuran (THF), 2-methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl i-butyl ether (MTBE), dimethoxyethane (DME), or diglyme). In certain embodiments, at least one instance of L is a ketone (e.g. , acetone or butanone). In certain embodiments, at least one instance of L is a chlorohydrocarbon (e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, or 1,2-dichloroethane (DCE)). In certain
embodiments, at least one instance of L is an ester (e.g. , propylene carbonate or ethyl acetate). . In certain embodiments, at least one instance of L is commercially available. In certain embodiments, each instance of L is commercially available.
[00152] In certain embodiments, s is 0. In certain embodiments, s is 1. In certain embodiments, s is 2. In certain embodiments, s is 3. In certain embodiments, s is 4.
[00153] In certain embodiments, the ligand of Formula (Dl) is commercially available. In certain embodiments, the transition metal salt of Formula (E) is commercially available.
[00154] Complexes of Formula (C2) may be prepared by contacting a ligand of Formula (D2), or a salt thereof, with a transition metal salt of Formula (E) to provide the complexes of Formula (C2):
D E
wherein R , R , and n are as described herein. In certain embodiments, the ligand of Formula
(D2) is commercially available.
[00155] Complexes of any one of Formulae (C3) to (C25) may be synthesized by contacting a ligand of any one of Formulae (D3) to (D25), or a salt thereof, with a transition metal salt of Formula (E) to provide the complex of any one of Formulae (C3) to (C25), respectively:
D9) (D10) (Dll)
(D12) D13) (D14)
D15) D16) (D17)
(D18) (D19) (D20)
(D24) (D25).
[00156] In certain embodiments, the ligand of any one of Formulae (D3) to (D25), or the salt thereof, is commercially available.
[00157] Complexes of any one of Formulae (CI) to (C25) may be isolated or may be generated in situ and reacted with a reagent or intermediate involved in a method of the invention.
[00158] The N-aryl sulfonimides or N-heteroaryl sulfonimides (e.g. , compounds of Formula (I), and salts thereof) obtained by methods of the invention may be partially deprotected to provide N-aryl sulfonamides or N-heteroaryl sulfonamides or fully deprotected to provide aryl amines or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof).
[00159] In certain embodiments, the N-aryl sulfonimides or N-heteroaryl sulfonimides (e.g. , compounds of Formula (I), and salts thereof) are partially or fully deprotected by reacting them with a reductant, a strong acid, or a nucleophile. In certain embodiments, the reductant employed in the deprotection step is a mixture of an alkaline earth metal and an alcohol. In certain embodiments, the reductant is a mixture of magnesium and an alcohol. In certain embodiments, the reductant is a mixture of magnesium and an unsubstituted Ci_6 alkyl alcohol (e.g. , MeOH, EtOH, i-PrOH, or i-BuOH). In certain embodiments, the reductant is a mixture of zinc and an acid. In certain embodiments, the reductant is a mixture of zinc and an acid selected from the group consisting of HC1, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF S03H, CH C02H, and CF C02H. In certain embodiments, the reductant is a mixture of an alkali metal and ammonia. In certain embodiments, the reductant is a mixture of zinc and HC1.
[00160] The strong acid employed in the deprotection step may be a strong inorganic acid or strong organic acid. In certain embodiments, the strong acid is an acid having a ρΚΆ value of less than 3, less than 2, less than 1, or less than 0 at ambient temperature. In certain embodiments, the strong acid is HC1, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF3S03H, or CF3C02H.
[00161] The nucleophile useful in the deprotection step may be an alkyl lithium or phenyl lithium. In certain embodiments, the nucleophile is an unsubstituted Ci_6 alkyl lithium (e.g. , MeLi, w-BuLi, sec-BuLi, or ί-BuLi). The nucleophile may also be a Grignard reagent. In certain embodiments, the nucleophile is of Formula: YMgZ, wherein Y is unsubstituted Ci_6 alkyl, unsubstituted phenyl, or -Si(unsubstituted Ci_6 alkyl) ; and Z is halogen. In certain embodiments, the nucleophile is MeMgCl, MeMgBr, or MeMgl. In certain embodiments, the
nucleophile is EtMgCl, EtMgBr, or EtMgl. In certain embodiments, the nucleophile is PhMgCl, PhMgBr, or PhMgl. In certain embodiments, the nucleophile is TMSMgCl or TMSMgBr.
[00162] The various step(s) of the inventive methods for preparing the N-aryl or N- heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof), aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof), and/or catalysts (e.g. , complexes of any one of Formulae (CI) to (C25)) may be performed under any suitable conditions as would be appreciated by one of skill in the art. A suitable condition is a combination of physical and chemical parameters under which an intended product (e.g. , a compound of any one of Formulae (I), (II), and (CI) to (C25), or a salt thereof) may be formed using the inventive methods. A suitable condition may include a suitable solvent. In certain
embodiments, the suitable solvent is a solvent described herein.
[00163] In certain embodiments, the suitable solvent is an aprotic solvent. In certain embodiments, the suitable solvent is acetonitrile (ACN). In certain embodiments, the suitable solvent is an amide solvent (e.g., N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMA), or N-methyl-2-pyrrolidone (NMP). In certain embodiments, the suitable solvent is dimethyl sulfoxide (DMSO). In certain embodiments, the suitable solvent is an ether solvent (e.g., tetrahydrofuran (THF), 2-methly-tetrahydrofuran, tetrahydropyran, dioxane, diethyl ether, methyl i-butyl ether (MTBE), dimethoxyethane (DME), or diglyme). In certain embodiments, the suitable solvent is a ketone solvent (e.g. , acetone or butanone). In certain embodiments, the suitable solvent is a chlorohydrocarbon solvent (e.g. , dichloromethane (DCM), chloroform, carbon tetrachloride, or 1,2-dichloroethane (DCE)). In certain embodiments, the suitable solvent is an ester solvent (e.g. , propylene carbonate or ethyl acetate). In certain embodiments, the suitable solvent is a pro tic solvent. In certain
embodiments, the suitable solvent is a mixture of two or more solvents described herein. In certain embodiments, the suitable solvent is commercially available.
[00164] A suitable condition may also include a suitable temperature under which a step of a method of preparing the compounds of the invention is performed. In certain embodiments, the suitable temperature is at least about 0 °C, at least about 4 °C, at least about 10 °C, at least about 20 °C, at least about 23 °C, at least about 40 °C, at least about 50 °C, at least about 60 °C, at least about 80 °C, or at least about 100 °C. In certain embodiments, the suitable temperature is at most about 100 °C, at most about 80 °C, at most about 60 °C, at most about 50 °C, at most about 40 °C, at most about 23 °C, at most about 20 °C, at most about 10 °C, at
most about 4 °C, or at most about 0 °C. Combinations of the above-referenced ranges (e.g., at least about 0 °C and at most about 60 °C) are also within the scope of the invention. A suitable temperature may be a variable temperature during a step of a method of preparing the compounds of the invention.
[00165] A suitable condition may also include a suitable pressure under which a step of a method of preparing the compounds of the invention is performed. In certain embodiments, the suitable pressure is about 1 atmosphere.
[00166] A suitable condition may also include a suitable atmosphere under which a step of a method of preparing the compounds of the invention is performed. In certain embodiments, the suitable atmosphere is air. In certain embodiments, the suitable atmosphere is an inert atmosphere. In certain embodiments, the suitable atmosphere is a nitrogen or argon atmosphere.
[00167] A suitable condition may also include a suitable time duration that a step of a method of preparing the compounds of the invention lasts. In certain embodiments, the suitable time duration is in the order of minutes (e.g., about 10 minutes or about 30 minutes), hours (e.g. , about 1 hour, about 2 hours, about 4 hours, about 8 hours, or about 16 hours), or days (e.g., about 1 day).
[00168] A suitable condition may also include agitating (e.g., stirring, sonicating, and/or shaking) one or more reactants, reagents, solvents, and/or solid supports involved in a method of the invention, or a mixture thereof.
[00169] One or more intermediates resulting from a step of a method of preparing the compounds of the invention may be isolated and/or purified, and the isolated and/or purified intermediate may be reacted in a next step of the method. The isolated and/or purified intermediates may be substantially free of impurities or may contain one or more other components, such as reagents and solvents employed in the step yielding the intermediates, and byproducts. The one or more intermediates may also be reacted in a next step without being isolated and/or purified. The intermediates and/or intended products of the methods of preparing the N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof) and aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof) may be isolated and/or purified using methods known in the art, such as chromatography normal phase chromatography (e.g. , silica gel flash chromatography), reverse phase chromatography (e.g., high performance liquid chromatography (HPLC)), precipitation, decanting, filtration, centrifuge, trituration, crystallization, recrystallization, liquid-liquid
phase separation, evaporation, and drying. In certain embodiments, the intended products described herein are substantially pure (e.g. , substantially free of impurities) (e.g., at least about 90%, at least about 95%, at least about 98%, at least about 99%, at least about 99.5%, at least about 99.9%, or more pure).
[00170] In certain embodiment la (I) is of the formula:
or a salt thereof.
[00171] Compounds of Formula (I) that may be prepared by the inventive methods include, but are not limited to, compounds of any one of the formulae:
and salts thereof.
Complexes
[00172] Another aspect of the present invention relates to transition metal complexes of any one of Formulae (CI) to (C25). The transition metal complexes of the invention include amine-N-oxide ligands, such as ligands of Formula (Dl) to (D25). In certain embodiments, the inventive transition metal complexes are the transition metal complexes described herein
In certain embodiments, the inventive transition metal complexes are complexes of Formula (CI). In certain embodiments, the inventive transition metal complexes are complexes of Formula (C2). In certain embodiments, the inventive transition metal complexes are complexes of Formula (C3). In certain embodiments, the inventive transition metal complexes are complexes of Formula (C4). In certain embodiments, the inventive transition metal complexes are complexes of any one of Formulae (C5) to (C25). In certain
embodiments, the inventive transition metal complexes are electrically neutral. The complexes of the invention may be prepared according to the methods described herein or by processes known in the art. For example, one can use synthetic chemistry transformations (including protecting group methodologies), e.g., those described in R. Larock,
Comprehensive Organic Transformations, VCH Publishers (1989); T.W. Greene and P.G.M. Wuts, Protective Groups in Organic Synthesis, 3rd Ed., John Wiley and Sons (1999); L.
Fieser and M. Fieser, Fieser and Fieser's Reagents for Organic Synthesis, John Wiley and Sons (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995) and subsequent editions thereof.
Kits
[00173] Also encompassed by the invention are kits (e.g., packs). In certain embodiments, the kits are useful for preparing the compounds described herein (e.g., N-aryl or N-heteroaryl sulfonimides and aryl or heteroaryl amines) from an arene or heteroarene (e.g., a compound of Formula (A), or a salt thereof). In certain embodiments, the kits are useful for preparing the compounds of Formula (I), and salts thereof. In certain embodiments, the kits are useful for preparing the compounds of Formula (II), and salts thereof.
[00174] A kit of the invention may include an N-fluorosulfonimide as an imidating agent; a catalyst; and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (CI); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (C2); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal
complex of Formula (C3); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of Formula (C4); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C5) to (C25); and a single-electron reductant. In certain embodiments, a kit of the invention includes N-fluorobenzenesulfonimide (NFBS or NFSI); a transition metal complex of any one of Formulae (C1)-(C25); and a single-electron reductant. In certain embodiments, a kit of the invention includes a compound of Formula (B), or a salt thereof; a transition metal complex of any one of Formulae (C1)-(C25); and an Ag(I) or Ru(II) salt.
[00175] An inventive kit may further include an arene or heteroarene. In certain embodiments, a kit of the invention further includes a compound of Formula (A), or a salt thereof.
[00176] A kit of the invention may also include a deprotecting agent that is useful in deprotecting the N-aryl or N-heteroaryl sulfonimides prepared by the inventive methods to provide partially deprotected N-aryl or N-heteroaryl sulfonamides or fully deprotected aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof). In certain
embodiments, a kit of the invention further includes a reductant. In certain embodiments, a kit of the invention further includes an alkaline earth metal (e.g. , magnesium) and an alcohol (e.g., an unsubstituted Ci_6 alkyl alcohol). In certain embodiments, a kit of the invention further includes zinc and an acid (e.g. , HC1, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF S03H, CH C02H, or CF C02H). In certain embodiments, a kit of the invention further includes a strong acid (e.g. , HC1, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF3S03H, or CF3C02H). In certain embodiments, a kit of the invention further includes a nucleophile. In certain embodiments, a kit of the invention further includes alkyl lithium (e.g. , an
unsubstituted C . alkyl lithium) or phenyl lithium. In certain embodiments, a kit of the invention further includes a Grignard reagent (e.g. , a compound of Formula: YMgZ, wherein Y and Z are as described herein).
[00177] The kits provided may further include a container (e.g. , a vial, ampule, bottle, syringe, flask, tube, beaker, dish, microtiter plate, and/or dispenser package, or other suitable container), a solvent (e.g. , a suitable solvent described herein), or an organic or inorganic agent (e.g. , a phase-transfer agent, a solubilizing agent, a stabilizing agent, an anti- oxidative agent, protecting agent, deprotecting agent, and/or a preservative agent). In some
embodiments, the kits further include instructions for using the kits of the invention. In
certain embodiments, the kits and instructions provide for preparing the compounds described herein (e.g., N-aryl or N-heteroaryl sulfonimides, aryl or heteroaryl amines, and transition metal complexes). In certain embodiments, the kits and instructions provide for preparing the compounds of Formula (I), and salts thereof. In certain embodiments, the kits and
instructions provide for preparing the compounds of Formula (II), and salts thereof. In certain embodiments, the kits and instructions provide for preparing the complexes of any one of Formulae (C1)-(C25).
EXAMPLES
[00178] In order that the invention described herein may be more fully understood, the following examples are set forth. The synthetic and biological examples described in this application are offered to illustrate the compounds, compositions, and methods provided herein and are not to be construed in any way as limiting their scope.
Example 1. Preparation of the Complexes of any one of Formulae (CI) to (C25), the N-Aryl or Ή-Heteroaryl Sulfonimides of Formula (I), and the Aryl or Heteroaryl Amines of Formula
(II)
[00179] Complexes of any one of Formulae (CI) to (C25) may be prepared from ligands of Formula (Dl) to (D25), or salts thereof, with transition metal salts of Formula (E). In one typical example, complex 1, a complex of Formula (CI), is synthesized by reacting N-(2- pyridylmethyl)pyrrolidine-N-oxide with a palladium(II) salt (e.g.,
tetrakis(acetonitrile)palladium(II) triflate). The ligand N-(2-pyridylmethyl)pyrrolidine-N- oxide can be prepared in two steps from commercially available starting materials. The complexes of the invention may also be s nthesized by methods known in the art.
[00180] An X-ray structure of complex 1 is shown in Figure 1.
[00181] An exemplary imidation of arenes (e.g., compounds of Formula Ar-H, wherein Ar is substituted or unsubstituted aryl) or heteroarenes by the inventive methods is illustrated in Scheme 1.
5 mol% 1
Ph02S„ ..S02Ph 10 mol% Ag{bipy)2CI04
Ar-H N Ar-N{S02Ph)2
F MeCH, 23 "C
1 equiv. 2 equtv. 24 h 2
Scheme 1. Exemplary synthesis of compounds of Formula (I)
[00182] In Scheme 1, N-fluorobenzenesulfonimide (NFBS or NFS I) is used as an imidating agent, complex 1 may act as a catalyst, and Ag(bipy)2C104 may work as a single- electron reductant. Both complex 1 and the Ag(bipy)2C104 are required in a reaction of Scheme 1 ; control experiments in which either complex 1 or the Ag(bipy)2C104 is omitted gave only trace amounts of imidated products. In another set of experiments, Ag(bipy)2C104 can be replaced with Ru(bipy)3(PF6)2 with similar results (Table 1, entry 2a). The reactions proceed similarly well in the presence or absence of light. Complex 1 involves an unusual pyridine-N- oxide ligand motif.
[00183] It was found that the pyridine-N-oxide ligand motif is exceptionally effective for the catalytic imidation. Because complex 1 is easily prepared and conveniently handled, it can be isolated, and the isolated complex 1 can be employed in the methods of the invention. Complex 1 can also be generated in situ by reacting N-(2-pyridylmethyl)pyrrolidine-N-oxide with a palladium(II) salt (e.g., Pd(NCMe)4(OTf)2), and the in situ generated complex 1 can be used in the inventive methods.
[00184] A variety of substrates, i.e., arenes and heteroarenes, can be efficiently imidated (e.g., Table 1). Regio selectivity is substrate-intrinsic. Resonance donors, such as alkoxy and halogen groups, may direct imidation ortho/para, similar to the regio selectivity in an electrophilic aromatic substitution reaction. In some cases, inductive donors (e.g., 2c and 2j) do not direct as effectively as resonance donors. Arenes and heteroarenes more electron-poor than those shown in Table 1 may show diminished reactivity and give lower yields.
Competitive C-H fluorination and/or double imidation are major side reactions for more electron-rich arenes, though performing the reactions at lower temperature (about 4 °C or lower) can substantially reduce both side reactions for some substrates (e.g., 2n-p, 2r, and 2s). The imidation methods of the invention work well at a small or large scale. For example, compound 2b can be synthesized on a 11-gram scale with the inventive methodology.
Table 1. Exemplary synthesis of compounds of Formula (I) under conditions of
Is
2r
8β%, ¾:ΐ 82%*
aRu(bipy)3(PF6)2 (2.5 mol ) used in place of Ag(bipy)2C104. Reaction performed at about 50 °C.
^Reaction performed at about 4 °C.
[00186] The sulfonyl protecting groups of the N-aryl or N-heteroaryl sulfonimides (e.g., compounds of Formula (I), and salts thereof) can be removed using methods of the invention
to provide aryl or heteroaryl amines (e.g., compounds of Formula (II), and salts thereof). In one set of experiments, treatment of compound 2b with magnesium in methanol under sonication produced aryl amine 3 in 85% yield (Scheme 2).
2b 85% 3
Scheme 2. Exemplary synthesis of compounds of Formula (II)
Materials and Methods
[00187] All air- and moisture-insensitive reactions were carried out under an ambient atmosphere, magnetically stirred, and monitored by thin layer chromatography (TLC) using EMD TLC plates pre-coated with 250 μιη thickness silica gel 60 F254 plates and visualized by fluorescence quenching under UV light. Flash chromatography was performed on
Dynamic Adsorbents Silica Gel 40-63 μιη particle size using a forced flow of eluent at 0.3-
13
0.5 bar pressure.
[00188] All air- and moisture- sensitive manipulations were performed using oven-dried glassware, including standard Schlenk and glovebox techniques under an atmosphere of nitrogen. Acetonitrile and acetonitrile-<i were dried over P205 and vacuum-distilled. MeOH
-4 ° was degassed at -30 °C under dynamic vacuum (10"" Torr) for one hour and stored over 3A sieves. All chemicals were used as received. All deuterated solvents were purchased from Cambridge Isotope Laboratories. NMR spectra were recorded on either a Varian Unity/Inova 600 spectrometer operating at 600 MHz for 1H acquisitions, a Varian Unity/Inova 500
1 13
spectrometer operating at 500 MHz and 125 MHz for H and C acquisitions, respectively, or Varian Mercury 400 spectrometer operating at 375 MHz and 101 MHz for 19F and 13C acquisitions, respectively. Chemical shifts are reported in ppm with the solvent resonance as the internal standard (1H: CDC13, δ 7.26; (CD3)2SO, δ 2.50; (CD3)2CO, δ 2.05; CD3CN, δ 1.94), (13C: CDC13, δ 77.16; (CD3)2SO, δ 39.52; (CD3)2CO, δ 29.84; CD3CN, δ 1.32),14 or added 3-nitrofluorobenzene (-112.0 ppm) for 19F spectra. Signals are listed in ppm, and multiplicity identified as s = singlet, br = broad, d = doublet, t = triplet, q = quartet, quin = quintet, sep = septet, m = multiplet; coupling constants in Hz; integration. High-resolution mass spectra were obtained using an Agilent ESI-TOF (6210) mass spectrometer or a Bruker
q-TOF Maxis Impact mass spectrometer. Concentration under reduced pressure was performed by rotary evaporation at 25-30 °C at appropriate pressure. Purified compounds were further dried under high vacuum (0.01-0.05 Torr). Yields refer to purified and spectroscopically pure compounds.
Standard procedure for C-H imidation reactions
[00189] Under N2 atmosphere, an oven-dried 4 mL vial was charged with (hetero)arene (0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%),
Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol,
2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added to react with the remaining NFBS and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel. It was observed that addition of triethylamine to the appropriate solvent system (1% of the final volume) aided the removal of dibenzenesulfonimide, which otherwise co-eluted with the desired product.
2-(Pyrrolidin-l-ylmethyl)pyridine (SI)
^ ^ K2C03 (2 equiv) fX
M DMF, 8 on0 °C, 18 h
2 equiv.
56%
[00190] To a solution of 2-(chloromethyl)pyridine (66.2 g, 0.519 mol, 1.00 equiv) in DMF (650 ml, c = 0.800 M) was added potassium carbonate (144 g, 1.04 mol, 2.00 equiv) and the mixture was stirred at 80 °C for 5 min. Pyrrolidine (73.8 g, 1.04 mol, 2.00 equiv) was then added, and the reaction mixture was stirred at 80 °C for 18 h. H20 (3.50 L) was added and the mixture was extracted with Et20 (4 x 250 ml). The combined organic layers were washed with H20 (2 x 200 mL), brine (200 mL), dried (MgS04), and concentrated in vacuo to afford 47.3 g of the title compound as a brown oil (56% yield), which was analytically pure and was used in the next step without further purification.
[00191] NMR Spectroscopy: 1H NMR (600 MHz, CD3CN, 23 °C, δ): 8.51-8.56 (m, 1H), 7.63 (td, J = 1.1, 1.8 Hz, 1H), 7.38 (d, J = 8.1 Hz, 1H), 7.14 (ddd, J = 7.5, 4.9, 1.1 Hz, 1H), 3.76 (s, 2H), 2.53-2.60 (m, 4H), 1.76-1.82 (m, 4H). These spectroscopic data correspond to previously reported data.15
l-(Pyridin-2-ylmethyl)pyrrolidine 1-oxide (S2)
92% S2
[00192] To a suspension of K2C03 (91.0 g, 0.656 mol, 3.00 equiv) and 2-(pyrrolidin-l- ylmethyl)pyridine (SI) (35.5 g, 0.219 mol, 1.00 equiv) in dichloromethane (1.20 L, c = 0.183 M) at -78 °C was added m-CPBA (56.6 g, 0.328 mol, 1.50 equiv). The resulting mixture was slowly warmed from -78 °C to 23 °C over 5 h and was stirred at 23 °C for 15 h. The solids were removed by filtration and washed with dichloromethane (3 x 20 mL). The combined filtrates were concentrated in vacuo to afford 39.0 g of the title compound (92% yield) as a brown solid, which was used directly for the preparation of 1. The characterization data was recorded on pure product, which was obtained by triturating the title compound (500 mg) with THF (3 x 2 mL, 15 min each time) at 23 °C.
[00193] Rf = 0.39 (CH2Cl2/MeOH 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.56-8.60 (m, 1H), 7.92 (d, J = 7.6 Hz, 1H), 7.77 (td, J = 1.6, 1.8 Hz, 1H), 7.31-7.36 (m, 1H), 4.96 (s, 2H), 3.73-3.81 (m, 2H), 3.66-3.73 (m, 2H), 2.29-2.40 (m, 2H), 2.03-2.12 (m, 2H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 150.6, 149.4, 137.2, 128.3, 124.5, 70.8, 67.1, 21.5. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C10H15N2O ([M+ H]+), 179.1179, found, 179.1181.
Palladium complex 1
[00194] l-(Pyridin-2-ylmethyl)pyrrolidine l-0xide (S2) (3.13 g, 17.6 mmol, 2.00 equiv) and Pd(MeCN)4(OTf)2 (5.00 g, 8.80 mmol, 1.00 equiv) were dissolved in acetonitrile (70.0 mL, c = 0.125 M). After stirring at 23 °C for 2 h, the reaction mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo. The product was triturated with THF
(3 x 10 mL, 15 min each time) at 23 °C and dried under vacuum for 8 h to afford 4.79 g of the title compound as a light brown solid (72% yield).
[00195] Melting point: 188 °C (decomp). NMR Spectroscopy: 1H NMR (600 MHz, CD3CN, 23 °C, δ): 8.45 (dd, J = 5.9, 1.2 Hz, 2H), 8.20 (td, J = 7.6, 1.8 Hz, 2H), 7.71-7.78 (m, 4H), 5.15 (s, 4H), 3.39-3.50 (m, 8H), 2.23-2.32 (m, 4H), 2.05-2.14 (m, 4H). 13C NMR (125 MHz, CD3CN, 23 °C, δ): 150.5, 149.0, 143.0, 129.3, 127.5, 71.7, 68.9, 22.5. Anal: calcd for C22H28F6N408S2Pd: C, 34.72; H, 3.71; N, 7.36; found: C, 34.66; H, 3.42; N, 7.24. UV- VIS Spectroscopy (MeCN, 23 °C): 265 nm (ε = 1.53 x 103 M"1 cm"1); 226 nm (ε = 4.77 x 103 M"1 cm"1). X-ray data included in X-Ray Crystallographic Analysis Section.
[00196] A solution of silver perchlorate (3.27 g, 15.8 mmol, 1.00 equiv) and 2,2'- bipyridine (5.05 g, 32.4 mmol, 2.05 equiv) in acetonitrile (100 mL, c = 0.158 M) was stirred at 23 °C for 2.5 h. The reaction mixture was then concentrated in vacuo. The resulting solid was triturated with Et20 and dried under vacuum to afford 7.93 g of the title compound as a yellow powder (97% yield).
[00197] NMR Spectroscopy: 1H NMR (600 MHz, CD3CN, 23 °C, δ): 8.66 (dd, J = 4.1, 1.8 Hz, 2H), 8.34 (dt, J = 8.2, 1.2 Hz, 2H), 8.04 (td, J = 7.6, 1.8 Hz, 2H), 7.53-7.56 (m, 2H). 13C NMR (125 MHz, CD3CN, 23 °C, δ): 153.5, 151.7, 139.9, 126.4, 123.5. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C2oHi6AgN4 + (Ag(bipy)2 +), 419.0420, found, 419.0427.
N-Phenyl-N-(phenylsulfonyl)benzenesulfonamide (2a)
76% 2a
[00198] Ag(bipy)2Cl04-catalyzed synthesis of 2a, 23 °C: Under N2 atmosphere, an oven- dried 4 mL vial was charged with benzene (23.4 mg, 26.8 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μιηοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40
M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h.
Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 84.7 mg of the title compound as a colorless solid (76% yield).
NFBS (2 equiv), 5 mol% 1 ,
88% 2a
[00199] Ru(bipy)3(PF6)2-catalyzed synthesis of 2a, 50 °C: Under N2 atmosphere, an oven- dried 4 mL vial was charged with benzene (39.1 mg, 44.7 μί, 0.500 mmol, 1.00 equiv), palladium complex 1 (19.0 mg, 25.0 μιηοΐ, 5.00 mol%), Ru(bipy)3(PF6)2 (10.7 mg,
13.0 μιηοΐ, 2.5 mol%), and NFBS (0.315 g, 1.00 mmol, 2.00 equiv). Acetonitrile (2.5 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 50 °C for 24 h. Subsequently, triethylamine (50.8 mg, 70.0 μί, 0.500 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 165 mg of the title compound as a colorless solid (88% yield). N(S02ph)2
81 % 2a
[00200] Ag(bipy)2Cl04-catalyzed synthesis of 2a, 50 °C: Under N2 atmosphere, an oven- dried 4 mL vial was charged with benzene (23.4 mg, 26.8 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μιηοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 50 °C for 24 h.
Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 90.7 mg of the title compound as a colorless solid (81% yield).
[00201] Rf = 0.51 (hexanes/EtOAc 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.95 (dd, J = 8.8, 1.2 Hz, 4H), Ί .66-1.69 (m, 2H), 7.53-7.57 (m, 4H),
7.44-7.47 (m, 1H), 7.34-7.38 (m, 2H), 7.02-7.05 (m, 2H). 1JC NMR (125 MHz, CDC13, 23 °C, δ): 139.6, 134.3, 134.1, 131.7, 130.4, 129.4, 129.1, 128.7. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C18Hi6N04S2 ([M + H]+), 374.0515, found, 374.0524.
Methyl 4-methoxy-3-(N-(phenylsulfonyl)phenylsulfonamido)benzoate (2b)
NFBS (2 equiv), 5 mol% 1 ,
99% 2b
[00202] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 4- methoxybenzoate (49.9 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 7:3 (v/v) with 1% triethylamine), to afford 137 mg of the title compound as a colorless solid (99% yield).
[00203] Alternative procedure for the preparation of 2b: Under N2 atmosphere, an oven- dried 4 mL vial was charged with 4-methoxybenzoate (49.9 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μιηοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (1.5 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 50 °C for 24 h.
Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 97 mg of the title compound as a colorless solid (70% yield).
Preparation of 2b under ambient atmosphere
[00204] A 4 mL vial was charged with 4-methoxybenzoate (166.2 mg, 1.000 mmol, 1.00 equiv), palladium complex 1 (38.1 mg, 50.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104
(51.7 mg, 10.0 μπιοΐ, 10.0 mol%), and NFBS (636.0 mg, 2.017 mmol, 2.02 equiv).
Acetonitrile (2.5 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed
vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 333.7 mg of the title compound as a colorless solid (72% yield).
Preparation of 2b under nitrogen, with components weighed out under air
[00205] Rf = 0.56 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.11 (dd, J = 8.5, 2.1 Hz, 1H), 7.96 (d, J = 8.2 Hz, 4H), 7.81 (d, J = 2.3 Hz, 1H), 7.62-7.71 (m, 2H), 7.54 (t, J = 7.6 Hz, 4H), 6.87 (d, J = 8.8 Hz, 1H), 3.88 (s, 3H), 3.44 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 165.8, 161.3, 140.0, 134.8, 134.0, 133.9, 129.0, 128.9, 123.2, 123.0, 111.7, 55.7, 52.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C21H20NO7S2 ([M+ H]+), 462.0676, found, 462.0690.
81 % 2c
[00206] Under N2 atmosphere, an oven-dried 4 mL vial was charged with (4- bromophenyl)trimethylsilane (68.8 mg, 58.6 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μιηοΐ,
10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 128 mg of the title compound as a colorless solid (81% yield).
[00207] Rf = 0.57 (hexanes/EtOAc 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDCI3, 23 °C, δ): 8.00-8.04 (m, 4H), 7.67-7.71 (m, 2H), 7.62 (d, J = 8.2 Hz, 1H), 7.53-7.58 (m, 4H), 7.39 (dd, J = 7.9, 1.5 Hz, 1H), 7.02 (d, J = 1.8 Hz, 1H), 0.18 (s, 9 H). 13C NMR (125 MHz, CDCI3, 23 °C, δ): 141.3, 139.5, 138.6, 136.4, 134.3, 134.0, 133.5, 129.5, 129.0, 127.7, -1.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C21H22BrNNa04S2Si [M + Na]+, 547.9815, found, 547.9821.
N-(5-Cyano-2-methoxyphenyl)-N-
(phenylsulfonyl)benzenesulfonamidebenzenesulfonamide (2d)
NFBS (2 equiv), 5 mol% 1 ,
NC\^^. 10 mol% Ag(bipy)2CI04 NC. ^ .NiSOzP Jz
OMe MeCN, 23 °C, 24h ^ OMe
99% 2d
[00208] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 4- methoxybenzonitrile (39.9 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 3:2 (v/v) with 1% triethylamine), to afford 128 mg of the title compound as an off-white solid (99% yield).
[00209] Rf = 0.42 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.94 (d, J = 8.2 Hz, 4H), 7.66-7.72 (m, 3H), 7.56 (t, J = 7.6 Hz, 4H), 7.40 (d, J = 2.3 Hz, 1H), 6.91 (d, J = 8.2 Hz, 1H), 3.44 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 161.3, 139.7, 137.0, 136.4, 134.2, 129.0, 128.9, 124.1, 117.9, 112.9, 104.7, 55.9. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C20H17N2O5S2 ([M + H]+), 429.0573, found, 429.0566.
2e, 71 % 2e-ll, 5%
[00210] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 2- methoxybenzoate (49.9 mg, 43.1 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine
(30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 7:3 (v/v) with 1% triethylamine), to afford 110 mg of the mixture of the title compound and methyl 5-(N-(henylsulfonyl)phenylsulfonamido)-2-((/V- (phenylsulfonyl)phenylsulfonamido)methoxy)benzoate (2e-II) (76% yield). Purification for characterization was accomplished by preparative TLC.
[00211] Data for 2e: colorless solid; R/ = 0.31 (hexanes/EtOAc 1 : 1 (v/v)). NMR
Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.90-7.94 (m, 4H), 7.66-7.70 (m, 2H), 7.53-7.58 (m, 4H), 7.44 (d, J = 2.3 Hz, 1H), 7.11 (dd, J = 9.1, 2.6 Hz, 1H), 6.92 (d, J = 8.8 Hz, 1H), 3.92 (s, 3H), 3.82 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 165.1, 160.5, 139.3, 136.5, 135.0, 134.2, 129.2, 128.6, 126.1, 120.7, 112.6, 56.4, 52.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C21H20NO7S2 [M + H]+, 462.0676, found, 462.0686.
Data for 2e-II: colorless solid; NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.04-8.07 (m, 4H), 7.91-7.93 (m, 4H), 7.64-7.73 (m, 4H), 7.51- 7.59 (m, 9H), 7.01-7.06 (m, 1H), 6.79-6.83 (m, 1H), 5.83 (s, 2H), 3.72 (s, 3H).
99% 2f
[00212] Under N2 atmosphere, an oven-dried 4 mL vial was charged with l-methoxy-4- nitrobenzene (45.9 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 1: 1 (v/v) with 1% triethylamine), to afford 134 mg of the title compound as an off-white solid (99% yield).
[00213] Rf = 0.27 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.34 (dd, J = 9.1, 2.6 Hz, 1H), 7.99 (d, J = 2.9 Hz, 1H), 7.96 (dt, J = 1.0, 1.8 Hz, 3H), 7.70 (tt, J = 1.6, 1.2 Hz, 2H), 7.57 (t, J = 7.6 Hz, 4H), 6.94 (d, J = 9.4 Hz, 1H),
3.52 (s, 3H). UC NMR (125 MHz, CDC13, 23 °C, δ): 162.9, 141.2, 139.7, 134.3, 129.3, 129.1, 128.9, 128.0, 123.6, 111.7, 56.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for Q9H17N2O7S2 ([M+ H]+), 449.0472, found, 449.0484.
N-(2-Methoxy-5-(trifluoromethoxy)phenyl)-N-(phenylsulfonyl)benzenesulfonamide (2g)
NFBS (2 equiv), 5 mol% 1 ,
61 %
[00214] Under N2 atmosphere, an oven-dried 4 mL vial was charged with l-methoxy-4- (trifluoromethoxy)benzene (57.6 mg, 45.5 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 89.2 mg of the title compound as a colorless solid (61 yield).
[00215] Rf = 0.30 (hexanes/EtOAc 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.97 (d, J = 7.6 Hz, 4H), Ί .65-1.69 (m, 2H), 7.54 (t, J = 7.6 Hz, 4H), 7.29 (dd, J = 9.5, 2.9 Hz, 1H), 6.95 (d, J = 2.9 Hz, 1H), 6.85 (d, J = 8.6 Hz, 1H), 3.43 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 156.8, 141.8, 139.8, 134.0, 128.9, 128.9, 126.5, 125.1, 123.4, 120.6 (q, J = 255 Hz), 112.4, 55.8. 19F NMR (375 MHz, CDC13, 23 °C, δ): -59.9. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C20H2oF3N206S2 [M + NH4]+, 505.0709, found, 505.0721.
N-(2-Methoxy-5-propionylphenyl)-N-(phenylsulfonyl)benzenesulfonamide (2h)
99%
2h
[00216] Under N2 atmosphere, an oven-dried 4 mL vial was charged with l-(4- methoxyphenyl)propan-l-one (49.2 mg, 45.7 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ,
10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 7:3 (v/v) with 1% triethylamine), to afford 136 mg of the title compound as a colorless solid (99% yield).
[00217] Rf = 0.33 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.05 (dd, J = 6.5, 2.3 Hz, 1H), Ί .93-1.91 (m, 4H), 7.64-7.68 (m, 3H), 7.54 (t, J = 8.2 Hz, 4H), 6.89 (d, J = 8.8 Hz, 1H), 3.45 (s, 3H), 2.84 (q, J = 7.0 Hz, 2H), 1.18 (t, J = 7.0 Hz, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 198.2, 161.2, 139.9, 134.0, 133.4, 132.4, 130.2, 128.9, 128.8, 122.9, 111.8, 55.7, 31.6, 8.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C22H21NNa06S2 ([M + Na]+), 482.0702, found, 482.0716.
N-(4-(tert-Butyl)phenyl)-N-(phenylsulfonyl)benzenesulfonamide (2j) and N-(3-(tert- Butyl)phenyl) -N- (phenylsulf onyl)benzenesulf onamide (2j -II)
NFBS (2 equiv), 5 mol% 1
^^ .N(S02Ph)2
10 mol% Ag(bipy)2CI04
(Bu"^^ MeCN, 23 °C, 24h 'Bu^ ,Bu/^ii^^N(S02Ph)2
87% 2j, 52% 2j-ll, 35%
[00218] Under N2 atmosphere, an oven-dried 4 mL vial was charged with tert- butylbenzene (40.3 mg, 46.4 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (9: 1 (v/v) with 1% triethylamine), to afford 112.0 mg of the mixture of the title compounds as a colorless solid (87% yield).
[00219] The products could not readily be separated by silica gel chromatography or preparative TLC, so they were characterized as a mixture. Data for 2j and 2j-II: R/ = 0.63
(hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.94- 7.97 (m, 10H), 7.65-7.69 (m, 5H), 7.52-7.58 (m, 10H), 7.44-7.48 (m, 1H), 7.34-7.38 (m, 3H), 7.29-7.33 (m, 1H), 6.92-6.97 (m, 4H), 6.84 (t, J = 2.1 Hz, 1H), 1.33 (s, 13.5H), 1.20 (s, 9H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 153.7, 152.5, 139.7, 139.7, 134.1, 134.0, 134.0, 131.5, 131.0, 129.1, 129.0, 128.7, 128.7, 128.5, 127.3, 126.4, 35.0, 34.7, 31.4, 31.1. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C22H27N2O4S2 ([M + NH4]+), 447.1407, found, 447.1399.
N-(3-Chloro-5-fluoro-4-methoxyphenyl)-N-(phenylsulfonyl)benzenesulfonamide (2k)
2k, 54% 2k-ll, 12% 2 -III, 12%
[00220] Under N2 atmosphere, an oven-dried 4 mL vial was charged with l-chloro-3- fluoro-2-methoxybenzene (48.2 mg, 38.9 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 106.7 mg of a mixture of the title compound and its two constitutional isomers (78% yield). Purification for characterization was accomplished by preparative TLC.
[00221] Data for 2k: colorless solid; R/ = 0.59 (hexanes/EtOAc 7:3 (v/v)). NMR
Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.93-7.97 (m, 4H), 7.70-7.74 (m, 2H), 7.57-7.61 (m, 4H), 6.83 (t, J = 2.1 Hz, 1H), 6.72 (dd, J = 10.9, 2.6 Hz, 1H), 4.04 (d, J = 2.3 Hz, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 155.1 (d, J = 251.3 Hz), 146.4 (d, J = 12.5 Hz), 139.1, 134.5, 129.4, 129.1 (d, J = 3.8 Hz), 128.8 (d, J = 1.1 Hz), 128.7, 128.4 (d, J = 5.5 Hz), 119.4 (d, J =20 Hz), 61.7 (d, J = 6.3 Hz). 19F NMR (375 MHz, CDC13, 23 °C, δ): -129.0 (d, J = 9.8 Hz). Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for
[M + NH4]+, 473.0402, found, 473.0415.
4-Methoxy-3-(N-(phenylsulfonyl)phenylsulfonamido)benzamide (21)
60%
2I
[00222] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 4- methoxybenzamide (45.3 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (1:4 (v/v) with 1% triethylamine), to afford 80.0 mg of the title compound as an off-white solid (60% yield).
[00223] R/= 0.39 (EtO Ac). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.94-7.98 (m, 5H), 7.65-7.69 (m, 2H), 7.52-7.56 (m, 4H), 7.50 (d, J = 2.3 Hz, 1H), 6.90 (d, J = 8.8 Hz, 1H), 3.45 (s, 3H). 13C NMR (125 MHz, (CD3)2CO, 23 °C, δ): 167.2, 160.9, 140.9, 134.9, 133.7, 132.6, 129.7, 129.6, 127.8, 123.4, 112.6, 55.9. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C2oHi9N206S2 ([M + H]+), 447.0679, found, 447.0684.
Diethyl (4-methoxy-3-(N-(phenylsulfonyl)phenylsulfonamido)phenyl)phosphonate (2m)
[00224] Under N2 atmosphere, an oven-dried 4 mL vial was charged with diethyl (4- methoxyphenyl)phosphonate (73.3 mg, 65.4 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ,
10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on
silica gel, eluting with hexanes/EtOAc (3:2 to 1:9 (v/v) with 1% triethylamine), to afford 138.4 mg of the title compound as a colorless solid (86% yield).
[00225] R/= 0.52 (EtOAc). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.92-7.94 (m, 4H), 7.88 (ddd, J = 12.8, 8.4, 1.8 Hz, 1H), Ί .63-1.61 (m, 2H), 7.52 (t, J = 1.9 Hz, 4H), 7.46 (dd, J = 13.2, 2.1 Hz, 1H), 6.91 (dd, J = 8.5, 3.8 Hz, 1H), 4.00-4.14 (m, 4H), 3.44 (s, 3H), 1.30 (t, J = 7.3 Hz, 6H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 160.8 (d, J = 3.4 Hz), 139.9, 136.8 (d, J = 10.0 Hz), 136.3 (d, J = 10.0 Hz), 133.9, 128.9, 128.8, 123.2 (d, J = 20.0 Hz), 120.6 (d, J = 193.4 Hz), 112.1 (d, J = 17.5 Hz), 62.3 (d, J = 5.0 Hz), 55.6, 16.4 (d, J = 6.3 Hz). Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C23H27N08PS2 [M + H]+, 540.0910, found, 540.0922.
N-(4-Fluorophenyl)-N-(phenylsulfonyl)benzenesulfonamide (2n)
2n, 44% 2n-ll, 1 1 % 2n-lll, 26%
[00226] Under N2 atmosphere, an oven-dried 4 mL vial was charged with fluorobenzene (28.8 mg, 28.2 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford two fractions: fraction A (36.9 mg) contained a 7.1: 1 ratio of 2n:2n-II, and fraction B (58.6 mg) contained a 1.35: 1:2.35 ratio of 2n:2n-II:2n- III. The total yield is therefore 95.5 mg (81% yield) with a product ratio of 2n:2n-II:2n-III = 4: 1:2.3.
[00227] The major component (2n) was further separated for characterization from fraction A by column chromatography eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v).
Compound 2n-II was assigned by COSY of the mixture in fraction A (see S87 for details). Compound 2n-III was assigned by process of elimination. Data for 2n:
[00228] R/= 0.47 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.95-7.92 (m, 4H), 7.71-7.67 (m, 2H), 7.58-7.54 (m, 4H), 7.07-7.02 (m, 2H), 7.02-6.98 (m, 2H),. 13C NMR (100 MHz, CDC13, 23 °C, δ): 166.1 (d, J = 252.6 Hz), 139.2 (s), 134.3 (s), 134.1 (s), 133.5 (d, J = 9.3 Hz), 130.1 (s), 128.8 (d, J = 54.4 Hz), 116.4 (d, J = 23.2). 19F NMR (125 MHz, CDC13, 23 °C, δ): -108.7. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C18Hi5FN04S2 ([M + H]+), 392.0421, found, 392.0416.
N-(4- soPropylphenyl)-N-(phenylsulfonyl)benzenesulfonamide (2o)
63% 2o, 39% 2o-ll, 12% 2o-lll, 12%
[00229] Under N2 atmosphere, an oven-dried 4 mL vial was charged with cumene (36.1 mg, 40.3 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ,
5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 with 1% triethylamine), to afford 78.4 mg of the mixture of the title compounds as a yellow solid (63% yield).
The products could not readily be separated by silica gel chromatography or preparative TLC, so they were characterized as a mixture. Data for 2o and 2o-II and 2o-III:
[00230] Rf = 0.54 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (500 MHz, CDCI3, 23 °C, δ): 8.00-7.90 (m, 6.7H), 7.71-7.64 (m, 3.5H), 7.59-7.50 (m, 6.8H), 7.44-7.36 (m, 0.9H), 7.32-7.28 (m, 0.6H), 7.24-7.17 (m, 2H), 7.12-7.16 (m, 0.4H), 6.97-6.88 (m, 2.3H), 6.81-6.72 (m, 0.6H), 3.02 (sept, 0.3H), 2.94 (sept, 1H), 2.83 (sept, 0.3H), 1.26 (d, 6H), 1.14 (d, 1.9H), 1.04 (d, 1.9H). 13C NMR (100 MHz, CDC13, 23 °C, δ): 151.3, 151.2, 150.0, 139.6, 139.5, 139.2, 134.1, 134.0, 133.8, 131.6, 131.4, 131.2, 130.7, 129.6, 129.0, 128.9, 128.8, 128.6, 128.5, 127.7, 127.3, 125.9, 33.8, 33.6, 28.4, 24.0, 23.7, 23.6. Mass
Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C21H2iN04S2 ([M + H]+), 416.0985, found, 416.0986.
Propylphenyl)-N-(phenylsulfonyl)benzenesulfonamide (2p)
2p-IV, 3%
[00231] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 2-fluoro-5- bromotoluene (56.7 mg, 38.3 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 4: 1 with 1% triethylamine), to afford 71.0 mg of the mixture of the title compounds as a yellow solid (63% yield).
[00232] The products could not readily be separated by silica gel chromatography or preparative TLC, so they were characterized as a mixture. Compounds 2p-2p-IV were assigned through a combination of 1-D TOCSY and NOESY experiments (see page S92- S93). Data for 2p and 2p-II and 2p-III:
[00233] R/= 0.57 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.07 (d, J = 8.2 Hz, 1.3H), 8.00 (d, J = 8.2 Hz, 4H), 7.96 (d, J = 8.2 Hz, 0.4H), 7.91 (d, J = 8.2 Hz, 0.6H), 7.72-7.68 (m, 2.9H), 7.66-7.62 (m, 0.3H), 7.59-7.54 (m, 5.7H), 7.53-7.49 (m, 0.7H), 7.46-7.42 (m, 1.5H), 7.31-7.27 (m, 0.2H), 7.25-7.22 (m, 0.2H), 7.01 (dd, J = 8.7, 8.7 Hz, 0.45H), 6.87 (dd, J = 9.6, 9.6 Hz, 0.2H), 6.81 (d, J = 9.6 Hz, 1H), 5.01 (s, 0.3H), 2.30 (s, 3H), 2.21 (s, 0.2H), 1.84 (s, 1.0H). 13C NMR (125 MHz, CDC13, 23 °C, δ): Peaks are not listed because the mixture of four compounds, as well as splitting of aryl carbons by 19F, precluded assignment. See S91 for spectrum. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for Ci9H15BrNNa04S2 ([M + Na]+), 505.9504, found, 505.9502.
N-(Benzo[h]quinolin-5-yl)-N-(phenylsulfonyl)benzenesulfonamide (2q)
88% 2q, 55% 2q-ll, 28% 2q-lll, 6%
[00234] Under N2 atmosphere, an oven-dried 4 mL vial was charged with
benzo[/z]quinoline (56.7 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 27.5 mg of the title compound as a colorless solid, along with 97.6 mg of a 6.7:4.3: 1 mixture of 2q, 2q-II, and 2q-III (88% yield overall). Compounds 2q-II and 2q-III were separated from 2q by preparative thin layer chromatography (eluting twice with 1:4 EtOAc:hexanes), and were assigned as a mixture through a combination of one-dimensional TOCSY and NOESY NMR experiments (see page S97). Data for 2q:
[00235] R/= 0.47 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 9.32 (d, J = 8.7 Hz, 1H), 8.96 (dd, / = 4.3 Hz, 1.7 Hz, 1H), 7.96 (dd, J = 7.96, 1.0 Hz, 4H), 7.88 (dd, J = 8.5, 1.8 Hz, 1H), 7.83 (ddd, J = 8.1, 6.7, 1.4 Hz, 1H), 7.77 (d, J = 7.8 Hz, 1H), 7.72 (m, 3H), 7.54 (m, 4 H), 7.46 (s, 1H), 7.31 (dd, / = 8.1, 4.4 Hz. 1H); 13C NMR (125 MHz, CDC13, 23 °C, δ): 149.3, 147.0, 138.8, 134.3, 132.8, 132.6, 132.3, 131.9, 129.1, 129.0, 128.8, 128.6, 128.5, 125.8, 124.7, 121.8. Mass Spectrometry: HRMS (ESI- TOF) (m/z): calcd for C25H18N2Na04S2 ([M + Na]+), 497.0600, found, 497.0595.
Methyl 3-(N-(phenylsulfonyl)phenylsulfonamido)benzo[/>]thiophene-2-carboxylate (2r)
79% 2r
[00236] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl benzo[b]thiophene-2-carboxylate (27.7 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 115 mg of the title compound as a colorlesssolid (79% yield).
[00237] R/= 0.38 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.98-8.00 (m, 4H), 7.83 (d, J = 8.2 Hz, 1H), 7.66-7.70 (m, 2H), 7.53 (t, J = 7.9 Hz, 4H), 7.46 (td, J = 1.6, 1.2 Hz, 1H), 7.30-7.33 (m, 1H), 7.25-7.28 (m, 1H), 3.39 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 160.9, 139.8, 138.2, 137.4, 134.6, 134.2, 129.4, 128.9, 128.8, 127.8, 125.4, 124.8, 122.8, 52.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C22H2iN206S3 [M + NH4]+, 505.0556, found, 505.0569.
N-(Phenylsulfonyl)-N-(thiophen-2-yl)benzenesulfonamide (2s)
NFBS (2 equiv), 5 mol% 1 ,
[00238] Under N2 atmosphere, an oven-dried 4 mL vial was charged with thiophene (25.2 mg, 24.4 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ,
5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The
residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (99: 1 to 5.6: 1 (v/v) with 1% triethylamine), to afford 59.7 mg of the title compound as a colorless solid (52% yield).
[00239] R/= 0.53 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.99 (dd, J = 8.3, 1.1 Hz, 4H), 7.68-7.71 (m, 2H), 7.55-7.59 (m, 4H), 7.38 (dd, J = 5.5, 1.7 Hz, 1H), 6.94 (dd, J = 5.5, 3.9 Hz, 1H), 6.74 (dd, J = 3.9, 1.1 Hz, 1H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 138.8, 134.4, 134.0, 131.4, 129.2, 128.9, 128.8, 125.8. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C16Hi3NNa04S3 ([M + Na]+), 401.9899, found, 401.9913.
N-(5-Bromothiophen-2-yl)-N-(phenylsulfonyl)benzenesulfonamide (2t)
NFBS (2 equiv), 5 mol% 1 ,
58% 2t
[00240] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 2- bromothiophene (48.9 mg, 29.0 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 80.3 mg of the title compound as a colorless solid (58% yield).
[00241] Rf = 0.52 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDCI3, 23 °C, δ): 7.97-8.02 (m, 4H), 7.71 (t, J = 7.3 Hz, 2H), 7.58 (t, J = 7.9 Hz, 4H), 6.94 (d, J = 4.1 Hz, 1H), 6.50 (d, J = 4.1 Hz, 1H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 138.6, 134.6, 134.3, 132.3, 129.3, 128.9, 128.8, 115.8. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C16H12BrNNa04S3 [M + Na]+, 481.8983, found, 481.8987.
N-(5-Cyanothiophen-2-yl)-N-(phenylsulfonyl)benzenesulfonamide (2u)
84% 2u
[00242] Under N2 atmosphere, an oven-dried 4 mL vial was charged with thiophene-2- carbonitrile (27.9 μί, 32.7 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol ), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol ), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with
hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 102 mg of the title compound as a colorlesssolid (84% yield).
[00243] R/= 0.41 (hexanes/EtOAc 3:2 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.97 (d, / = 7.6 Hz, 4H), 7.74 (t, / = 7.6 Hz, 2H), 7.60 (t, / = 7.9 Hz, 4H), 7.46 (d, / = 4.1 Hz, 1H), 6.77 (d, / = 4.1 Hz, 1H). 13C NMR (100 MHz, CDC13, 23 °C, δ): 140.3, 138.1, 135.8, 135.0, 131.6, 129.5, 128.9, 113.2, 112.6. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C17H12N2Na04S3 ([M + Na]+), 426.9851, found, 426.9861.
N-(Phenylsulfonyl)-N-(l-(phenylsulfonyl)-lH-pyrrol-2-yl)benzenesulfonamide (2v)
C. NFBS (2 equiv), 5 mol% 1 ,
10 mol% Ag(bipy)2CIO4 - N(S02P ) 2
N *■ N
S02Ph M^CN, 4 °C, 24h ¾o2ph
60%
2v
[00244] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 1- (phenylsulfonyl)-lH-pyrrole (62.2 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting
with hexanes/EtOAc (19: 1 to 4: 1 (v/v) with 1% triethylamine), to afford 90.5 mg of the title compound as a colorless solid (60% yield).
[00245] R/= 0.46 (hexanes/EtOAc 3:2 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.95-7.99 (m, 6H), 7.67-7.70 (m, 2H), 7.58-7.61 (m, 1H), 7.52-7.56 (m, 4H), 7.47-7.51 (m, 2H), 7.22 (dd, J = 3.5, 1.8 Hz, 1H), 6.26 (t, J = 3.5 Hz, 1H), 6.04 (dd, J = 3.5, 1.8 Hz, 1H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 139.0, 138.4, 134.4, 134.3, 129.8, 129.1, 128.9, 128.5, 124.8, 122.6, 117.7, 111.0. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C22H19N206S3 ([M + H]+), 503.0400, found, 503.0397.
52% 2w
[00246] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 1 -(1 -methyl- 1H- pyrrol-2-yl)ethanone (36.9 mg, 35.5 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 3: 1 (v/v) with 1% triethylamine), to afford 65.8 mg of the title compound as a colorless solid (52% yield).
[00247] Rf = 0.46 (hexanes/EtOAc 3:2 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDCI3, 23 °C, δ): 7.90-7.94 (m, 4H), 7.68-7.72 (m, 2H), 7.56 (t, J = 8.2 Hz, 4H), 6.89 (d, J = 4.7 Hz, 1H), 5.92 (d, J = 4.1 Hz, 1H), 3.42 (s, 3H), 2.44 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 189.1, 138.7, 134.7, 131.4, 129.3, 128.9, 127.5, 117.5, 111.4, 33.1, 27.3. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C19Hi9N205S2 ([M + H]+), 419.0730, found, 419.0734.
N-(6-Chloro-4-methoxypyridin-3-yl)-N-(phenylsulfonyl)benzenesulfonamide (2x) and N- (2-Chloro-4-methoxypyridin-3-yl)-N-(phenylsulfonyl)benzenesulfonamide (2x-II)
86%
2x, 55% 2x-ll, 31 %
[00248] Under N2 atmosphere, an oven-dried 4 mL vial was charged 2-chloro-4- methoxypyridine (43.1 mg, 34.2 μί, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (7:3 to 2:3 (v/v) with 1% triethylamine), to afford 72.4 mg (55% yield) of 2x and 40.2 mg (31% yield) of 2x-II.
[00249] Data for 2x: yellow solid; R/= 0.61 (hexanes/EtOAc 1: 1 (v/v)). NMR
Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.94-7.98 (m, 4H), 7.91 (s, 1H), 7.66- 7.71 (m, 2H), 7.54-7.58 (m, 4H), 6.82 (s, 1H), 3.50 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 164.8, 154.7, 152.3, 139.6, 134.3, 129.1, 128.9, 120.6, 108.2, 56.1. Mass
Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C18H16C1N205S2 ([M + H]+), 439.0184, found, 439.0200.
Data for 2x-II: yellow oil; R/ = 0.22 (hexanes/EtOAc 1: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.29 (d, J = 5.9 Hz, 1H), 8.03 - 8.06 (m, 4H), Ί .65-1.69 (m, 2H), 7.55 (t, J = 7.9 Hz, 4H), 6.77 (d, J = 5.9 Hz, 1H), 3.46 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 165.9, 155.0, 151.5, 139.9, 134.2, 129.5, 128.8, 119.1, 107.2, 56.2. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C18H16C1N205S2 ([M + H]+), 439.0184, found, 439.0184.
N-(6-Methoxy-2-methylquinolin-7-yl)-N-(phenylsulfonyl)benzenesulfonamide (2y) and N^V'-(6-Methoxy-2-methylquinoline-5,7-diyl)bis(N- (phenylsulfonyl)benzenesulfonamide) (2y-II)
82%
2y-ll
[00250] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 6- methoxyquinoline-2-carbonitrile (55.3 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 4 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 1: 1 (v/v) with 1% triethylamine), to afford 115 mg of the title compound (82% yield). When the reaction was performed at 23 °C, a 6:5 mixture of 2y and 2y-II was formed in 77% yield.
[00251] Data for 2y: colorless solid; Rf = 0.33 (hexanes/EtOAc 1 : 1 (v/v)). NMR
Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.28 (d, J = 9.4 Hz, 1H), 8.01 (d, J = 8.8 Hz, 1H), 7.91 (dd, J = 8.8, 1.2 Hz, 4H), 7.68-7.72 (m, 2H), 7.52-7.56 (m, 5H), 7.51 (d, J = 2.9 Hz, 1H), 3.49 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 158.4, 143.9, 139.6, 134.9, 134.3, 133.4, 131.9, 130.5, 129.3, 128.9, 124.4, 117.8, 117.5, 115.8, 56.1. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C23H18N305S2 ([M + H]+), 480.0682, found, 480.0695.
[00252] Data for 2y-II: colorless solid; R/ = 0.40 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.03 (d, J = 8.2 Hz, 1H), 7.88 (dd, J = 8.8, 1.2 Hz, 4H), 7.85 (dd, J = 8.8, 1.2 Hz, 4H), 7.70-7.74 (m, 2H), 7.64-7.68 (m, 2H), 7.53- 7.57 (m, 5H), 7.48-7.52 (m, 4H), 7.40 (d, J = 8.8 Hz, 1H), 3.42 (s, 3 H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 157.8, 141.2, 139.2, 138.3, 135.6, 134.5, 134.0, 131.7, 130.8, 129.5, 129.3, 129.0, 128.9, 124.7, 123.6, 118.1, 116.5, 56.5. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C35H27N409S4 ([M + H]+), 775.0655, found, 775.0668.
(E)-4-methoxy-4-oxobut-2-en-l-yl-4-methoxy-3-(N-(phenylsulfonyl)phenylsulfonamido) benzoate (2z)
[00253] Under N2 atmosphere, an oven-dried 4 mL vial was charged with (E)-4-methoxy- 4-oxobut-2-en-l-yl 4-methoxybenzoate (75.1 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μπιοΐ,
10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (9: 1 to 7:3 (v/v) with 1% triethylamine), to afford 132.7 mg of the title compound as an off-white solid (81% yield).
[00254] Rf = 0.25 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.13 (dd, J = 8.8, 2.3 Hz, 1H), 7.96 (d, J = 8.2 Hz, 4H), 7.76 (d, J = 1.8 Hz, 1H), 7.66 (t, J = 7.8 Hz, 2H), 7.54 (t, J = 7.9 Hz, 4H), 7.02 (dt, J = 15.4, 4.6 Hz, 1H), 6.90 (d, J = 8.8 Hz, 1H), 6.05 (dt, J = 15.8, 1.8 Hz, 1H), 4.94 (dd, J = 4.7, 1.8 Hz, 2H), 3.78 (s, 3H), 3.47 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 166.3, 164.4, 161.7, 141.5, 139.9, 134.8, 134.1, 134.0, 128.9, 128.9, 123.1, 122.4, 122.0, 111.8, 63.1, 55.7, 51.9. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C25H27N209S2 ([M+ NH4]+), 563.1152, found, 563.1162.
N-(2-Methoxy-5-(4-nitrobenzoyl)phenyl)-N-(phenylsulfonyl)benzenesulfonamide (2aa)
89%
2aa
[00255] Under N2 atmosphere, an oven-dried 4 mL vial was charged with (4- methoxyphenyl)(4-nitrophenyl)methanone (45.9 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (16.0 mg, 30.0 μηιοΐ,
10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c =
0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 3:2 (v/v) with 1% triethylamine), to afford 148 mg of the title compound as an off-white solid (89% yield).
[00256] Rf = 0.47 (hexanes/EtOAc 1 : 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.33 (dt, J = 8.8, 1.8 Hz, 2H), 7.98 (dd, J = 8.5, 2.1 Hz, 1H), 7.95 (dd, J = 7.6, 1.2 Hz, 4H), 7.87 (dt, J = 8.8, 2.3 Hz, 2H), 7.67 (t, J = 7.6 Hz, 2H), 7.58 (d, J = 1.8 Hz, 1H), 7.54 (t, J = 8.2 Hz, 4H), 6.97 (d, J = 8.2 Hz, 1H), 3.49 (s, 3H). 13C NMR (125 MHz, CDC13, 23 °C, δ): 192.2, 161.9, 149.9, 142.9, 139.9, 135.8, 134.6, 134.1, 130.6, 129.1, 128.9, 128.9, 123.8, 123.2, 112.2, 55.9. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C26H21N208S2 ([M + H]+), 553.0734, found, 553.0723.
Isopropyl 2-(4-(4-chlorobenzoyl)-2-(N-(phenylsulfonyl)phenylsulfonamido)phenoxy)-2- methylpropanoate (2ab)
[00257] Under N2 atmosphere, an oven-dried 4 mL vial was charged Fenofibrate
(isopropyl 2-(4-(4-chlorobenzoyl)phenoxy)-2-methylpropanoate) (108.2 mg, 0.300 mmol, 1.00 equiv), palladium complex 1 (11.4 mg, 15.0 μιηοΐ, 5.00 mol%), Ag(bipy)2C104
(16.0 mg, 30.0 μηιοΐ, 10.0 mol%), and NFBS (0.189 g, 0.600 mmol, 2.00 equiv). Acetonitrile (0.75 mL, c = 0.40 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, triethylamine (30.5 mg, 42.0 μί, 0.300 mmol, 1.00 equiv) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (19: 1 to 5.7: 1 (v/v) with 1% triethylamine), to afford 148.4 mg of the title compound as a colorless solid (75% yield).
[00258] R/= 0.57 (hexanes/EtOAc 7:3 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 8.01-8.04 (m, 4H), 7.86 (dd, J = 8.8, 2.3 Hz, 1H), 7.68-7.70 (m, 2H), 7.64-7.68 (m, 2H), 7.56 (d, J = 2.3 Hz, 1H), 7.53-7.56 (m, 4H), 7.43-7.46 (m, 2H), 6.70 (d, J = 8.8 Hz, 1H), 5.07 (sep, J = 6.3 Hz, 1H), 1.34 (s, 6H), 1.25 (d, J = 6.5 Hz, 6H). 13C NMR
(125 MHz, CDCI3, 23 °C, δ): 192.8, 172.6, 158.0, 140.4, 138.9, 136.0, 135.7, 133.8, 133.1, 131.3, 129.8, 129.0, 129.0, 128.8, 124.4, 115.9, 80.5, 69.5, 24.3, 21.7. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C32H34C1N208S2 ([M + NH4]+), 673.144, found,
673.1455.
Methyl 4-methoxy-3-(N-(phenylsulfonyl)phenylsulfonamido)benzoate (2b)
83% 2b
[00259] To a flame dried 250 mL round bottom flask charged with stir bar, N-fluoro-N- (phenylsulfonyl)benzenesulfonamide (14.2 g, 45.0 mmol, 1.50 equiv), palladium complex 1 (0.457 g, 0.600 mmol, 2.00 mol%), and Ag(bipy)2C104 (0.624 g, 1.20 mmol, 4.00 mol%) in dry MeCN (30.0 mL, c = 1.00 M) was added methyl 4-methoxybenzoate (4.99 g, 30.0 mmol, 1.00 equiv). The reaction vessel was sealed and stirred at 23 °C for 40 h. Triethylamine (5mL) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with (hexanes/EtOAc 5: 1 (v/v) with 1% triethylamine) to afford 11.5 g of the title compound (83% yield), which is spectroscopically identical to the compound prepared according to the standard procedure (vide supra).
Methyl 3-amino-4-methoxybenzoate (3b)
Me02C _^¾^ N(S02Ph)2 Mg Μβ02Ο. ^^ΝΗ2
MeOH, 23 °C, 2h ^— ^,,
OMe ' ' OMe
85%
2b 3b
0.108 mmol scale
[00260] To a solution of methyl 4-methoxy-3-(N-
(phenylsulfonyl)phenylsulfonamido)benzoate (2b) (49.8 mg, 0.108 mmol, 1 equiv) in dry MeOH (3.6 mL, c = 0.030 M) was added Mg powder (31.6 mg, 1.30 mmol, 12 equiv) and the suspension was sonicated under nitrogen atmosphere at 23 °C for 2 h. Saturated solution of NH4C1 (2.0 mL) was then added and the resulting mixture was extracted with diethyl ether (3 x 15 mL). The combined organic layers were dried (MgS04), filtered, and concentrated in vacuo. The residue was purified by flash chromatography on silica gel, eluting with
hexanes/EtOAc (5: 1 (v/v) with 1% triethylamine) to afford 16.7 mg of the title compound as a yellow solid (85% yield).
[00261] Rf = 0.70 (CH2Cl2/MeOH 9: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, (CD3)2SO, 23 °C, δ): 7.26 (d, J = 2.3 Hz, 1H), 7.21 (dd, J = 8.2, 2.3 Hz, 1H), 6.87 (d, J = 8.8 Hz, 1H), 4.97 (br s, 2H), 3.82 (s, 3H), 3.76 (s, 3 H). 13C NMR (125 MHz, (CD3)2SO, 23 °C, δ): 166.5, 150.1, 137.7, 122.0, 118.3, 113.8, 109.8, 55.5, 51.6. Mass Spectrometry: HRMS (ESI-TOF) (m/z): calcd for C9H12N03 ([M + H]+), 182.0812, found, 182.0815.
0.217 mmol scale:
[00262] To a solution of methyl 4-methoxy-3-(/V-
(phenylsulfonyl)phenylsulfonamido)benzoate (2b) (100 mg, 0.217 mmol, 1 equiv) in dry MeOH (7.2 mL, c = 0.030 M) was added Mg powder (63.2 mg, 2.60 mmol, 12 equiv) and the suspension was sonicated under nitrogen atmosphere at 23 °C for 2 h. Saturated solution of NH4CI (4.0 mL) was then added and the resulting mixture was extracted with diethyl ether (3 x 30 mL). The combined organic layers were dried (MgS04), filtered, and concentrated in vacuo. The residue was purified by flash chromatography on silica gel, eluting with hexanes/EtOAc (5: 1 (v/v) with 1% triethylamine) to afford 29.0 mg of the title compound as a yellow solid (74% yield). Spectroscopic data are the same as described above.
Compound Cla
[00263] Palladium acetate (2.87 g, 12.8 mmol, 1.00 equivalent) was suspended in dry acetonitrile (20.0 mL), followed by the addition of 50% tetrafluoroboric acid diethyl ether complex (4.72 g, 3.97 mL, 26.9 mmol, 2.10 equivalents). After stirring the mixture at 23 °C for 0.5 h, a solution of l-(pyridin-2-ylmethyl)pyrrolidine 1-oxide (4.79 g, 26.9 mmol, 2.10 equivalents) in dry acetonitrile (15.0 mL) was added to the mixture. The resulting mixture was stirred at 23 °C for 1 hour and concentrated under reduced pressure. The resulting residue was triturated with dichloromethane (3 x 10 mL) and tetrahydrofuran (2 x 8 mL) at
23 °C and dried under vacuum for 8 h to afford 6.24 g of compound Cla as a light brown solid (76% yield). 1H NMR (600 MHz, CD3CN, 23 °C): δ 8.45 (dd, J = 5.8, 1.3 Hz, 2H), 8.21 (td, J = 7.7, 1.3 Hz, 2H), 7.76 (td, J = 7.7, 1.3 Hz, 2H), 7.71 (d, J = 7.7 Hz, 2H), 5.11 (s, 4H), 3.42-3.44 (m, 8H), 2.15-2.31 (m, 4H), 2.09-2.12 (m, 4H) ppm. 13C NMR (125 MHz, CD3CN, 23 °C): δ 150.5, 148.9, 143.0, 129.3, 127.5, 71.8, 68.9, 22.5 ppm. Anal: calcd for C2oH28B2F8N402Pd: C, 37.74; H, 4.43, N, 8.80; found: C, 37.03; H, 4.26; N, 8.55.
Exemple 2. Control Experiments: Catalytic Imidation in the Absence ofl,
Ag(bipy)2ClC>4,and/ or light
[00264] Table 2. Imidation in the Absence of 1, Ag(bipy)2C104,and/ or light
Standard
[00265] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 4- methoxybenzoate (8.3 mg, 0.05 mmol, 1.0 equiv), palladium complex 1 (1.9 mg, 2.5 μιηοΐ, 5.0 mol%), Ag(bipy)2C104 (2.6 mg, 5.0 μηιοΐ, 10 mol%), and NFBS (32 mg, 0.10 mmol, 2.0 equiv). Acetonitrile (0.250 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, dimethylurea (2.2 mg, 0.025 mmol, 0.50 equiv) was added as an internal standard for the NMR measurement and the reaction mixture was concentrated in vacuo. The NMR of the crude reaction mixture indicated full conversation of starting material to the desired product 2b.
In the absence of 1
[00266] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 4- methoxybenzoate (8.3 mg, 0.050 mmol, 1.0 equiv), Ag(bipy)2C104 (2.6 mg, 5.0 μιηοΐ, 10 mol%), and NFBS (32 mg, 0.10 mmol, 2.0 equiv). Acetonitrile (0.250 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h.
Subsequently, dimethylurea (2.2 mg, 0.025 mmol, 0.50 equiv) was added as an internal standard for the NMR measurement and the reaction mixture was concentrated in vacuo. The NMR of the crude reaction mixture indicated less than 10% conversation of starting material to the desired product 2b.
In the absence of Ag(bipy)2004
[00267] Under N2 atmosphere, an oven-dried 4 mL vial was charged with methyl 4- methoxybenzoate (8.3 mg, 0.050 mmol, 1.0 equiv), palladium complex 1 (1.9 mg, 2.5 μιηοΐ, 5.0 mol%), and NFBS (32 mg, 0.10 mmol, 2.0 equiv). Acetonitrile (0.250 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 24 h.
Subsequently, dimethylurea (2.2 mg, 0.025 mmol, 0.50 equiv) was added as an internal standard for the NMR measurement and the reaction mixture was concentrated in vacuo. The NMR of the crude reaction mixture indicated full conversation of starting material to the desired product 2b.
In the absence of light
[00268] Under N2 atmosphere, an oven-dried amber 4 mL vial was charged with methyl 4- methoxybenzoate (8.3 mg, 0.050 mmol, 1.0 equiv), palladium complex 1 (1.9 mg, 2.5 μιηοΐ, 5.0 mol%), Ag(bipy)2C104 (2.6 mg, 5.0 μπιοΐ, 10 mol%), and NFBS (32 mg, 0.10 mmol, 2.0 equiv). The vial was wrapped with electrical tape and acetonitrile (0.250 mL, c = 0.20 M) was added. The reaction mixture was stirred in a sealed vial at 23 °C for 24 h. Subsequently, dimethylurea (2.2 mg, 0.025 mmol, 0.50 equiv) was added as an internal standard for the NMR measurement and the reaction mixture was concentrated in vacuo. The NMR of the crude reaction mixture indicated full conversation of starting material to the desired product 2b.
Example 3. Control Experiments: Evaluation of Palladium Catalysts Other than 1
[00269] Under N2 atmosphere, in a 4 mL vial was prepared a solution of NFBS (387.6 mg, 1.200 mmol, 2.0 equiv) and Ag(bipy)2C104 (31.2 mg, 0.0604 mmol, 10 mol%) in acetonitrile (3.00 mL). In 5 separate vials were weighed palladium source (0.0050 mmol, 5.0 mol%), and
ligand (0.010 mmol, 10.0 mol ) if applicable (see Table 3 for details). To each of the five Pd/ligand-containing vials was added 0.500 mL of the NFBS/Ag solution. Finally, fluorobenzene (9.3 μί, 0.100 mmol, 1.0 equiv) was added to each vial. The five reactions were stirred magnetically at room temperature for 40 h, after which 2.0 \L 4- fluoronitrobenzene was added to each of the reaction mixtures as an internal standard and the product yields were analyzed by 19F NMR {Table 3).
[00270] Table 3. Evaluation of Palladium Catalysts Other than 1
Exampel 4. X-ray Crystallographic Analysis
Palladium complex 1 (CCDC 943631)
[00271] X-Ray quality crystals of 1 were grown by layering Et20 (ca. 0.2 mL) on top of a solution of ca. 5 mg of 1 in 0.2 mL MeCN A crystal was mounted on a nylon loop using Paratone-N oil, and transferred to a Bruker APEX II CCD diffractometer (MoKcc radiation, λ=0.71073 A) equipped with an Oxford Cryosystems nitrogen flow apparatus. The sample was held at 100 K during the experiment. The collection method involved 0.5 ° scans in ω at
28 ° in 2Θ. Data integration down to 0.82 A resolution was carried out using SAINT V7.46 A (Bruker diffractometer, 2009) with reflection spot size optimisation. Absorption corrections were made with the program SADABS (Bruker diffractometer, 2009). The structure was solved by the direct methods procedure and refined by least- squares methods again F2 using SHELXS-97 and SHELXL-97 (Sheldrick, 2008). Non-hydrogen atoms were refined anisotropically, and hydrogen atoms were allowed to ride on the respective atoms. Restraints on bond lengths and constraints of the atomic displacement parameters on each pair of disorder fragments (SADI and EADP instructions of SHELXL97), as well as the restraints of the atomic displacement parameters (SIMU/DELU instructions of SHELXL97) if necessary,
have been applied for the disorder refinement. Crystal data as well as details
collection and refinement are summarized in Table 4.
[00272] Table 4. X-ray crystallographic data
Computer programs: APEX2 v2009.3.0 (Bruker-AXS, 2009), SAINT 7.46A (Bruker-AXS, 2009), SHELXS97 (Sheldrick, 2008), SHELXL97 (Sheldrick, 2008), Bruker SHELXTL.
Example 5. Mechanistic Studies
[00273] Mechanistic investigations indicate a mechanism with the following unusual features: oxidation of a transition metal complex, such as a Pd(II) complex (e.g., complex 1),
prior to the activation of a substrate (e.g. , an arene) enabled by an amine-N- oxide ligand and C-N bond formation distinct from conventional palladium-catalyzed C-H functionalization (Figure 2). Turnover-limiting oxidation of complex 1 yields a Pd(IV) complex of Formula
(F) . Single-electron reduction of the complex of Formula (F) by a single-electron reductant (e.g., an Ag(I) salt) and substrate association follow to form Pd(III) intermediate of Formula
(G) and an Ag(II) intermediate. The intermediate of Formula (G) formally transfers a sulfonimidyl radical to the bound arene substrate, which expels a delocalized radical intermediate and regenerates complex 1. The delocalized radical intermediate is subsequently oxidized by the Ag(II) intermediate, then deprotonated to furnish the sulfonimidated product.
[00274] Turnover-limiting oxidation of complex 1 by NFBS is supported by the measured rate law of the reaction, which is first order with respect to both the transition metal complex of the invention (e.g., complex 1) and imidating agent (e.g., NFBS), and zero-order with respect to the substrate (e.g. , a compound of Formula (I)) and the single-electron reductant (e.g., Ag(bipy)2C104). When only NFBS and complex 1 are combined in acetonitrile, catalytic reduction of NFBS to HN(S02Ph)2 and HF is observed, with the reducing equivalents evidently derived from the solvent. The rate of the non-productive catalytic reduction of NFBS is substantially identical to the rate of the productive imidation reaction (Scheme 3). The rate law and the substantially identical rate for both reactions shown in Scheme 3 are consistent with a turnover-limiting oxidation of complex 1 by NFBS to yield a common, short-lived, high-valent palladium intermediate of Formula (F).
PhC½S. ,„SG2Ph cat. 1 P C S. ,S02Ph
N + H- F
F MeCN, 50 °C H
raie = k [1] [NFBSl, /f = 1.26x10"2 s^ "
Scheme 3. Kinetic studies of the methods of the invention
[00275] Oxidation of a transition metal complex of the invention (e.g., a Pd(II) complex) typically requires strongly σ-donating anionic ligands such as hydrocarbyl ligands, which are absent from complex l.8 DFT (density functional theory) calculations reveal that the highest occupied molecular orbital (HOMO) of complex 1 is an extended M-L π-antibonding orbital of dxz parentage from palladium, instead of the Jz 2-based orbital more typical of square-planar d complexes. The oxygen lone pairs of the amine -N-oxide ligand interact strongly with the Pd-based dxz orbital, driving it higher in energy than the Jz 2-based orbital (Figure 3). This interaction may explain how complex 1 can be oxidized by NFBS, despite its two formal positive charges.
[00276] It is proposed that a single-electron reductant (e.g. , an Ag(I) salt) may serve as a co-catalyst to provide access to intermediate of Formula (G), which is the putative intermediate responsible for C-N bond formation. Ag(bipy)2 2+ was observed by electron paramagnetic resonance (EPR) spectroscopy during catalysis, which implicates the redox reactivity of the single-electron reductant. Inner- sphere reactivity of the single-electron reductant can be ruled out because coordinatively saturated Ru(bipy)3(PF6)2 is also an effective single-electron reductant. The action of the single-electron reductant on an intermediate of Formula (F) to yield the C-N bond forming species explains why for most substrates, substrate consumption is not observed in the absence of the single-electron reductant. Because the oxidation of complex 1 by NFBS is turnover-limiting, the C-N bond forming step cannot be studied kinetically. Therefore, competition kinetic isotope effect (KIE) experiments were employed to probe this step (Scheme 4). An inverse secondary
intramolecular of 0.80 + 0.01 for imidation of 1,3,5-trideuterobenzene was measured. Inverse secondary KIEs are unusual for palladium-catalyzed C-H functionalization reactions; C-H palladations at Pd(II)9 and Pd(IV)10 typically display primary KIE values. The measured
KIE indicates a rehybridization of the C-H bond from sp 2 to sp 3 in the product-determining transition state, consistent with a C-N bond formation via inner-sphere addition of a dibenzenesulfonimidyl radical to the bound arene in an intermediate of Formula (G).
Electrophilic addition would also be consistent with the observed intramolecular isotope effect. However, inverse secondary KIE values for electrophilic substitution are rare;11 commonly, KIEs close to unity are observed for electrophilic processes due to the opposing effects of rehybridization and hyperconjugation on the zero-point vibrational energy of the affected C-H bond. 12 Inner-sphere attack from an intermediate, such as an intermediate of Formula (G), to form the putative aryl radical is supported by an intermolecular competition KIE 1.03 + 0.02. The absence of a KIE in this case is consistent with irreversible substrate bindin prior to C-N bond formation.9'1013
Intermolecular KIE: 2a 2a-ds s 1.03 ± 0.02
Scheme 4. Intramolecular and intermolecular kinetic isotope effect studies
Rate Law for NFBS Reduction Catalyzed by 1
Ph02S. ,S02P 1.25-10.0 μιηοΙ 1 ^ PhOzS. ,S02P
[. MeCN, 50 °C H + H_F
0.0501 mmol
[00277] This experiment was carried out under an N2 atmosphere. A 0.167 M solution of NFBS in CD3CN was prepared in a vial and sealed with a septum cap. In five separate septum-sealed screw cap NMR tubes, solutions of 1 (1.25, 2.5, 3.75, 5.0, and 10 μιηοΐ) and 3- nitro-fluorobenzene (0.019-0.038 μιηοΐ) in 0.200 mL CD3CN were prepared in the following way:
1.25 and 2.50 μτηοΐ reactions: A stock solution of 3.8 mg 1 (5.0 μιηοΐ) and 8.0
of 3-nitrofluorobenzene (0.076 μτηοΐ) in 0.400 mL CD3CN was prepared. To one NMR tube (1.25 μιηοΐ 1) was added 0.100 mL of the stock solution and 0.100 mL pure CD CN. To the other tube (2.5 μιηοΐ 1) was added 0.200 mL of the stock solution.
[00278] 3.75 and 5.0 μητοΐ reactions: A stock solution of 7.6 mg 1 (10 μιηοΐ) in 0.400 mL CD CN was prepared. To one NMR tube (3.75 μιηοΐ 1) was added 0.150 mL of the stock solution, 4.0 μΐ^ (0.038 μιηοΐ) 3-nitrofluorobenzene, and 0.050 mL pure CD CN. To the other tube (5.0 μιηοΐ 1) was added 0.200 mL of the stock solution and 4.0 μΐ^ (0.038 μιηοΐ) 3- nitrofluorobenzene .
[00279] 10 μηιοΐ reaction: A solution of 7.6 mg (10 μτηοΐ) 1 and and 4.0 μί (0.038 μτηοΐ) 3-nitrofluorobenzene in 0.200 mL CD CN was prepared in an NMR tube.
[00280] For each reaction, 0.300 mL of the NFBS solution was added to the NMR tube via syringe, the sample was immediately inserted into the NMR probe pre-heated at 50 °C, and the consumption of NFBS was followed by integration of the 19F NMR spectra with 3-nitrofluorobenzene. The reactions were followed over 15-30% conversion, except the last one (10.0 μιηοΐ 1), which was followed through 90% conversion (>3 half-lives). The plot of ln[NFBS] vs. time shows excellent linearity throughout, indicating an overall reaction order of unity. Pseudo-first-order analysis was therefore applied to determine the dependence of k0bs on [1]. A plot of
vs ln[l] reveals a first order dependence on [1]. The overall rate law is therefore rate = fc[l][NFBS]. A plot of k0bs vs. [1] revealed a slope of 1.26xl0"2 M_1s_1, which is the value of k. The results are shown in Figure 4.
Rate Law of Imidation Catalyzed by 1 and Ag(bipy)2C104
[00281] Fluorobenzene was chosen as the substrate for the determination of the rate law of the catalytic imidation reaction because fluorobenzene is a competent substrate for the
imidation reaction (81 yield), and the consumption of fluorobenzene and appearance of the products could be followed concurrently with the consumption of NFBS by 19F NMR. The data was acquired at 50 °C because this temperature was found to facilitate a more convenient rate of reaction for kinetic analysis. To justify the acquisition of kinetic data with fluorobenzene as a substrate at a reaction temperature of 50 °C, the catalytic imidation on fluorobenzene under these conditions has been performed , and no substantial diminution in yield has been found.
1 .0 equiv 2.0 equiv 76%
[00282] Under N2 atmosphere, an oven-dried 4 mL vial was charged with fluorobenzene (9.3 μί, 0.10 mmol, 1.0 equiv), palladium complex 1 (7.6 mg, 10 μιηοΐ, 10 mol%),
Ag(bipy)2C104 (5.2 mg, 10 μπιοΐ, 10 mol%), and NFBS (63.0 mg, 0.20 mmol, 2.0 equiv). Acetonitrile (0.500 mL, c = 0.20 M) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 12 h. Subsequently, 4-nitrofluorobenzene (2.0 μί, 0.0188 mmol) was added as an internal standard and the yield of the imidated products was measured by 19F NMR {Table 5).
0.050 mmol 0.100 mmol
[00284] This experiment was carried out under an N2 atmosphere. A 0.333 M solution of NFBS in CD3CN was prepared in a vial and sealed with a septum cap. In a septum-sealed screw cap NMR tube was prepared a solution containing 1 (7.6 mg, 10 μιηοΐ), Ag(bipy)2C104 (2.6 mg, 5.0 μιηοΐ), fluorobenzene (4.7 μί, 0.050 mmol), and 3-nitro-fluorobenzene (2.0 μί, 0.019 μπιοΐ) in 0.200 mL CD3CN. With a syringe, 0.300 mL of the NFBS solution was added
to the NMR tube, the sample was immediately inserted into the NMR probe pre-heated to 50 °C, and the consumption of NFBS and fluorobenzene were followed over 4.5 h by measuring against 3-nitro-fluorobenzene by 19F NMR. The consumption of NFBS follows clean first- order kinetics through ca. 95% conversion (>4 half-lives). The results are shown in Figure 5.
[00285] This observation can be explained considering the presence of a competitive, nonproductive NFBS reduction pathway. This nonproductive pathway causes NFBS to be expended before the arene substrate, which prematurely slows rate of fluorobenzene consumption. Since the consumption of fluorobenzene was followed only for the first ca. 20% for the purpose of rate law determination, pseudo first-order kinetic analysis is still appropriate.
Determination of order in Ag(bipy)2Cl04
0.10 mmol 0.100 mmol
[00286] This experiment was carried out under an N2 atmosphere. A 0.333 M solution of NFBS in CD3CN was prepared in a vial and sealed with a septum cap. In five separate septum-sealed screw cap NMR tubes, solutions containing 1 (10.0 μιηοΐ), Ag(bipy)2C104 (1.3, 2.6, 5.0, 7.5 μιηοΐ), fluorobenzene (0.10 mmol), and 3-nitro-fluorobenzene (0.019 mmol) in 0.200 mL CD3CN were prepared in the following way. A stock solution containing 22.8 mg 1 (0.030 mmol), 27.9 fluorobenzene (0.300 mmol), and 6.0 μΐ, 3- nitrofluorobenzene (0.056 mmol) in CD3CN was prepared. Into four vials were weighed 0.8 mg, 1.6 mg, 3.1 mg, and 4.7 mg Ag(bipy)2C104, respectively. The content of each vial was dissolved in 0.240 mL of the stock solution, and 0.200 mL of this mixture was transferred to an NMR tube. For each reaction, 0.300 mL of the NFBS solution was added to the NMR tube via syringe, the sample was immediately inserted into the NMR probe pre-heated to 50 °C, and the consumption of NFBS and fluorobenzene were followed by 19F NMR, with 3-nitro- fluorobenzene as internal standard. The reactions were followed over ca. 20% conversion, and the results were subjected to pseudo first-order kinetic analysis. The results are shown in Figure 6.
Determination of order in arene substrate
0.050 - 0.25 0.100 mmol
mmol
[00287] This experiment was carried out under an N2 atmosphere. A 0.333 M solution of NFBS in CD3CN was prepared in a vial and sealed with a septum cap. In five separate septum-sealed screw cap NMR tubes were prepared solutions containing 1 (7.6 mg, 10 μιηοΐ), Ag(bipy)2C104 (1.3 mg, 2.5 μιηοΐ), and 3-nitro-fluorobenzene (2.0
0.019 μιηοΐ) in 0.200 mL CD3CN. To each tube was then added 4.7, 9.4, 14.1, 18.8, and 27.9 μΐ.
fluorobenzene (0.050, 0.10, 0.150, 0.200, and 0.300 mmol, respectively). For each reaction, 0.300 mL of the NFBS solution was added to the NMR tube via syringe, the sample was immediately inserted into the NMR probe pre-heated to 50 °C, and the consumption of NFBS was followed by 19F NMR, with 3-nitro-fluorobenzene as internal standard. The reactions were followed over ca. 20% conversion, and the results were subjected to pseudo first-order kinetic analysis. The results are shown in Figure 7.
0.10 mmol 0.0500 - 0.150
mmol
[00288] This experiment was carried out under an N2 atmosphere. A solution of NFBS (0.500 M) in CD CN was prepared in a vial and sealed with a septum cap. In five separate septum-sealed screw cap NMR tubes were prepared solutions containing 1 (7.6 mg, 10.0 μιηοΐ), Ag(bipy)2C104 (1.3 mg, 2.5 μιηοΐ), fluorobenzene (9.3
0.10 mmol), and 3-nitro- fluorobenzene (2.0
0.019 μιηοΐ) in the volume of CD3CN which would make 0.500 mL after addition of the NFBS solution (see below). The NFBS solution was added to the NMR tube via syringe (0.10 mL, 0.15 mL, 0.20 mL, 0.25 mL, and 0.30 mL). The sample was immediately inserted into the NMR probe pre-heated to 50 °C, and the consumption of fluorobenzene was followed by 19F NMR, with 3-nitro-fluorobenzene as internal standard. The reactions were followed over ca. 20% conversion, and the results were subjected to pseudo first-order kinetic analysis. The results are shown in Figure 8.
Determination of order in 1
0.10 mmol 0.100 mmol
[00289] This experiment was carried out under an N2 atmosphere. A 0.333 M solution of NFBS in CD3CN was prepared in a vial and sealed with a septum cap. In five separate septum-sealed screw cap NMR tubes, solutions containing 1 (2.50, 3.75, 5.00, 7.50, and 10.0 μιηοΐ), Ag(bipy)2C104 (1.3 mg, 2.5 μιηοΐ), fluorobenzene (9.3
0.10 mmol), and 3- nitro-fluorobenzene (2.0
0.019 μιηοΐ) in 0.200 mL CD3CN were prepared in the following way:
[00290] 3.75 and 7.5 μητοΐ 1: Two stock solutions were prepared. Solution A contained 11.4 mg 1 (0.0150 mmol) in CD3CN, and Solution B contained 5.2 mg Ag(bipy)2C104 (0.010 mmol), 37.2 μϊ^ fluorobenzene (0.400 mmol), and 8.0 μΐ^ 3-nitrofluorobenzene (0.075 mmol) in 0.200 mL CD3CN. To one NMR tube (3.75 μηιοΐ 1) were added 0.075 mL Solution A, 0.050 mL Solution B, and 0.075 mL pure CD3CN. To another NMR tube (7.5 μιηοΐ 1) were added 0.150 mL Solution A and 0.050 mL Solution B.
[00291] 2.5 and 5.0 μητοΐ 1: Two stock solutions were prepared. Solution A contained 7.6 mg 1 (0.015 mmol) in CD3CN, and Solution B contained 5.2 mg Ag(bipy)2C104 (0.010 mmol), 37.2 μϊ^ fluorobenzene (0.400 mmol), and 8.0 μΐ^ 3-nitrofluorobenzene (0.075 mmol) in 0.200 mL CD3CN. To one NMR tube (2.5 μιηοΐ 1) were added 0.075 mL Solution A, 0.050 mL Solution B, and 0.075 mL pure CD3CN. To another NMR tube (5.0 μιηοΐ 1) were added 0.150 mL Solution A and 0.050 mL Solution B.
[00292] 10 μηιοΐ 1: Two stock solutions were prepared. Solution A contained 15.2 mg 1 (0.020 mmol) in CD3CN, and Solution B contained 5.2 mg Ag(bipy)2C104 (0.010 mmol), 37.2
3-nitrofluorobenzene (0.076 mmol) in 0.200 mL CD3CN. To an NMR tube was added 0.150 mL Solution A and 0.050 mL Solution B.
For each reaction, 0.300 mL of the NFBS solution was added to the NMR tube via syringe, the sample was immediately inserted into the NMR probe pre-heated to 50 °C, and the consumption of NFBS and fluorobenzene were followed by by 19F NMR, with 3-nitrofluorobenzene as internal standard. The reactions were followed over ca. 20% conversion, and the results were subjected to pseudo first-order kinetic analysis. The results are shown in Figure 9.
Determination of the Resting State
0.10 mmol 0.200 mmol
[00293] Under an N2 atmosphere, into a vial (A) a solution of 1 (15.6 mg, 0.204 mmol) and 1,2-dichloroethane (8.0 μί, 0.10 mmol) in CD3CN (1.000 mL) was prepared. In a separate vial (B) were weighed NFBS (63.0 mg, 0.200 mmol, 2.00 equiv) and Ag(bipy)2C104 (5.2 mg, 0.010 mmol, 10 mol%). The contents of vial B were dissolved in half of the solution in vial A (0.500 mL), and fluorobenzene (9.3 μί, 0.10 mmol) was added to vial B. The contents of each vial was transferred to an NMR tube. The solution from vial A (containing only 1 and 1,2-dichloroethane) was analyzed by 1H NMR, and the actual ratio of complex 1: 1,2-dichloroethane was measured to be 0.22: 1. The solution from vial B (catalytic imidation reaction) was inserted into an NMR probe pre-heated to 50 °C. After 12 minutes, the conversion was measured to be ca. 31% by 19F NMR. A 1H NMR of the reaction mixture was also recorded, and the actual ratio of complex 1: 1,2-dichloroethane was measured to be 0.15: 1. Complex 1 was therefore found to account for 67% of the palladium-containing species. The diminution in the amount of 1 may be attributed to decomposition during catalysis. The results are shown in Figure 10.
Role of the co-catalyst
[00294] The co-catalyst is proposed to be responsible for the reduction of intermediate F in order to generate intermediate G, the actual C-N bond forming species. Evidence supporting this proposal is outlined below.
1 equiv NFBS
Ru(bipy)3(PF6)2 - Ru(bipy)3 3+
MeCN, 23 °C
1 equiv 1
1 equiv NFBS
Ru(bipy)3(PF6)2 Ru(bipy)3 3+
MeCN, 23 °C
[00295] Under an N2 atmosphere, two solutions were prepared in NMR tubes: Solution A contained Ru(bipy)3(PF6)2 (5.0 mg, 5.8 μπιοΐ) and NFBS (1.8 mg, 5.7 μπιοΐ) in CD3CN (0.80 mL), and Solution B contained 1 (4.4 mg, 5.8 μιηοΐ), Ru(bipy) (PF6)2 (5.0 mg, 5.8 μιηοΐ) and
NFBS (1.8 mg, 5.7 μηιοΐ) in CD3CN (0.80 niL). After 10 minutes, each solution was analyzed by 1H NMR (Figure 11, top panels). The spectrum of Solution A showed sharp signals for Ru(bipy)3(PF6)2 and NFBS, while Solution B showed sharp signals for 1 and NFBS, but dramatically broadened signals for Ru(bipy)3(PF6)2. After standing for 4 hours at room temperature, the solutions were analyzed again by 1H and 19F NMR (Figure 11, bottom panels). The 1H NMR spectrum of Solution A showed sharp signals for NFBS but broadened signals for Ru(bipy)2(PF6)2, and the 19F NMR of Solution A showed only 9 % NFBS consumption. The 1H NMR spectrum of Solution B showed no sign of Ru(bipy)3(PF6)2 (Figure 12), and the 19F NMR of solution B showed 61% NFBS conversion.
[00296] The broadening of the signals due to Ru(bipy) 2+ is attributed to partial oxidation to Ru(bipy)3 3+, with rapid redox exchange between the Ru(II) and Ru(III) species causing the broadening. This interpretation is supported by low temperature NMR of Solution A after 4 hours of standing at room temperature, which shows the peaks due to Ru(bipy) 2+ sharpening as temperature decreases, consistent with slower exchange at lower temperature (Figure 12).
[00297] EPR spectroscopy provides evidence for the formation of Ru(bipy)3 3+ in Solution B. After 5 hours, Solution B was transferred to an EPR tube and frozen in liquid nitrogen. The resulting glass was analyzed by EPR spectroscopy along with a sample containing pure Ru(bipy) (PF6)3 prepared according to a literature procedure.16 The EPR spectrum of Solution B (Figure 13) shows a paramagnetic resonance assignable to Ru(bipy)3 3+, along with a partially overlapping second resonance (possibly a Pd(III) species generated upon oxidation of Ru(bipy) 2+).
[00298] The data shown in Figures 11 to 13 combined demonstrate an acceleration of the oxidation of Ru(bipy)2 2+ to Ru(bipy)2 3+ in the presence of 1.
Comparison of rates of NFBS reduction by 1, and 1 + Ru(bipy)3(PF )2
[00299] Under an N2 atmosphere, two solutions were prepared in NMR tubes: Solution C contained 1 (4.4 mg, 5.8 μιηοΐ) and NFBS (1.8 mg, 5.7 μιηοΐ) in CD3CN (0.60 mL), and Solution D contained 1 (4.4 mg, 5.8 μιηοΐ), Ru(bipy)3(PF6)2 (5.0 mg, 5.8 μιηοΐ) and NFBS (1.8 mg, 5.7 μιηοΐ) in CD CN (0.60 mL). The consumption of NFBS in both solutions was followed by 19F NMR over 26 hours, and the rates were found to be identical (Figure 14).
Discussion
[00300] The above data indicate the following: (1) NFBS reacts with palladium catalyst 1 more rapidly than with Ru(bipy)3 2+, (2) Ru(bipy)3 3+ forms much more rapidly in the presence
of 1 and NFBS than with NFBS alone, (3) the rate of NFBS oxidation of Ru(bipy)3 mediated by 1 is limited by the rate of oxidation of 1 by NFBS. These observations combined are consistent with a scenario in which palladium catalyst 1 is oxidized by NFBS to give the putative high-valent intermediate F, followed by single electron oxidation of Ru(bipy)3 2+ by F to yield Ru(bipy) 3+ and a Pd(III) intermediate (possibly a progenitor to G in Figure 2).
Observation of Agu(bipy)2 in the catalytic imidation reaction
[00301] Under an N2 atmosphere, two solutions were prepared. Solution E contained 1 (3.8 mg, 5.0 μπιοΐ), Ag(bipy)2C104 (5.2 mg, 10 μπιοΐ), NFBS (63.1 mg, 0.200 mmol), and fluorobenzene (9.4
0.10 mmol) in 0.50 mL MeCN. Solution F contained Ag(bipy)2C104 (5.2 mg, 10 μιηοΐ) and NFBS (63.1 mg, 0.200 mmol) in 0.50 mL MeCN. Both solutions were transferred to EPR tubes and were frozen in liquid nitrogen after 1 hour, and the resulting glasses were analyzed by EPR spectroscopy (Figure 15). Both spectra show the same signal, assigned to an Ag(bipy)2 2+ species.
[00302] The above data demonstrates that an Ag(bipy)2 2+ species is present in the catalytic imidation reaction mixture.
Competition Kinetic Isotope Effect Experiments
Intramolecular Competition KIE Experiment
Intramolecular KIE: 2a-d32a-d2 = 0.80 ± 0.01
[00303] Under N2 atmosphere, an oven-dried 4 mL vial was charged with 1,3,5- trideuterobenzene (89.2 μί, 1.00 mmol, 2.00 equiv), palladium complex 1 (19.0 mg, 25.0 μπιοΐ, 5.00 mol%), Ag(bipy)2C104 (25.8 mg, 50.0 μπιοΐ, 10.0 mol%), and NFBS (0.158 g, 0.500 mmol, 1.00 equiv). Acetonitrile (1.25 mL) was added and the reaction mixture was stirred in a sealed vial at 23 °C for 29 h. Subsequently, triethylamine (200 μί) was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (4: 1 (v/v)), to afford 118.0 mg of the title compound as a colorless solid (0.314 mmol, 63% yield based on NFBS).
[00304] Rf = 0.51 (hexanes/EtOAc 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDCI3, 23 °C, δ): 7.95 (dd, J = 8.8, 1.2 Hz, 4H), Ί .66-1.69 (m, 2H), 7.53-7.57 (m, 4H), 7.44-7.47 (m, 0.57H), 7.34-7.38 (m, 1.01H), 7.02-7.05 (m, 1.11H).
[00305] Ratio of 2a-di/2a-d2, measured by !H NMR: the 1H NMR spectrum was recorded nine times with seven minutes between spectra to assure full relaxation. The peak at 7.03 ppm (the 2- and 6-positions in 2a-<¾) were integrated against the peak at 7.95 ppm (the 2- and 6- positions in the sulphonyl phenyl groups, set to 4. OH) in each spectrum. The average of these measurements yielded 2a-<¾/2a-<¾ = 0.80, with a standard deviation of 0.0056 (95% confidence interval: + 0.011).
[00306] Ratio of 2a-d3/2a-d2, measured by mass spectrometry: the mixture was analyzed three times by GC/MS (EI detector) in single ion mode, counting M+* at m/z = 375 and 376. Because the peak at m/z=376 has a contribution from both 2a-<¾ and (M+1) for 2a-<¾ (natural abundance: 19.5%), the following formula was necessary to extract the ratio of 2a-</3/2a-<¾:
[00307] Where A376 and A375 are the areas for the peaks at m/z = 376 and 375, respectively.
Through this treatment, the three measurements yielded 2a-<¾/2a-<¾ = 0.80 (average), with a standard deviation of 0.017 (95% confidence interval: + 0.034).
Intermolecular Competition KIE Experiment
Intermolecular KIE: 2a/2a-d5
[00308] Under N2 atmosphere, an oven-dried 4 mL vial (A) was charged with palladium complex 1 (19.0 mg, 25.0 μηιοΐ, 5.00 mol%), Ag(bipy)2C104 (25.8 mg, 50.0 μηιοΐ,
10.0 mol%), and NFBS (0.158 g, 0.500 mmol, 1.00 equiv). In a separate vial (B), a solution of C6¾ (90.2 μί, 1.00 mmol) and C6D6 (88.6 μί, 1.00 mmol) was prepared in 2.50 mL acetonitrile. The contents of vial A were dissolved in 1.25 mL of the solution in vial B, and the reaction mixture was stirred in the sealed vial at 23 °C for 8 h. Subsequently,
triethylamine (200 was added and the reaction mixture was concentrated in vacuo. The residue was purified by chromatography on silica gel, eluting with hexanes/EtOAc (4: 1 (v/v)), to afford 73.3 mg of the title compound as a colorless solid (0.195 mmol, 39% yield
based on NFBS). The remainder of the solution in vial B was subjected to GC/MS analysis, and the actual ratio of C6¾ to C6D6 was measured.
[00309] Rf = 0.51 (hexanes/EtOAc 4: 1 (v/v)). NMR Spectroscopy: 1H NMR (600 MHz, CDC13, 23 °C, δ): 7.95 (dd, J = 8.8, 1.2 Hz, 4H), Ί .66-1.69 (m, 2H), 7.53-7.57 (m, 4H), 7.44-7.47 (m, 0.53H), 7.34-7.38 (m, 1.04H), 7.02-7.05 (m, 1.04H).
[00310] Ratio of 2a/2a-ds, measured by !H NMR: the 1H NMR spectrum was recorded nine times with seven minutes between spectra to assure full relaxation. The peak at 7.03 ppm (the 2- and 6-positions in 2a) were integrated against the peak at 7.95 ppm (the 2- and 6- positions in the sulphonyl phenyl groups, set to 4. OH) in each spectrum. The average of these measurements, correcting for the measured actual starting ratio of C6¾ to C6D6, yielded
2a/2a-rf5 = 1.03, with a standard deviation of 0.010 (95% confidence interval: + 0.020).
[00311] Ratio of 2a/2a-ds, measured by mass spectrometry: the mixture was analyzed three times by GC/MS (EI detector) in single ion mode, counting M+* at m/z = 373 and 378. Division of the areas of the resulting peaks yielded 2a/2a-ds = 1.03, with a standard deviation of 0.0038 (95% confidence interval: + 0.076). Correcting for the measured actual starting ratio of C6¾ to C6D6, this corresponds to kulko = 0.99 + 0.076.
DFT Calculations
[00312] Density functional theory (DFT) calculations were performed using Gaussian09 17 on the Odyssey cluster at Harvard University. Geometry optimization was carried out using the atomic coordinates from the crystal structure of 1 as a starting point. BS I includes SDD quasirelativistic pseudopotentials on Pd (MWB28) with basis sets (Pd: (8s7p6d)/[6s5p3d] 18 ) extended by polarization functions (Pd: f, 1.472 19) and 6-31G(d,p) 20 on H, C, N. All geometry optimizations were performed using the B3PW91 functional with the BS I basis set. Molecular orbitals were generated using an isosurface value of 0.03 with B3PW91/BS I.
Images were generated using Chem3D. 21 An optimized structure of complex 1 with the B3PW91 functional with the BS I basis set is shown in Figure 16 and the corresponding Cartesian coordinates are shown in Table 6.
[00313] Table 6. Cartesian coordinates of an optimized structure of complex 1 with the
B3PW91 functional with the BS I basis set.
0,000129 c 5,259697 4,363332 -1.960754
0 0,526134 0.062967 c 2.9S 2 0,244798 -2,030643
H 6342291 -L852818 0.827158 € 4.223733 3,705119 -1.30134:1
'0358592 0.527153 C 2.248429 -1.091817 -2.252473
€ 5.344906 -2.479767 1.476335 c 1.516529 -1.406731 -0.922865
H 4,399548 -1,95177? 1,504105 H 7.599111 1JS2212 -1.S03755
C 8.6S95 3 -1.744523 O.01S094 H 2,413855 2.314095 -0.119451
H 9,584661 •2,31427? 0.119394 H 3,581863 1.627823 1.049809
H & 36S98 -1.62791 -1.0497.54 H 7.350353 4,195998 -2,5659?
C 5,504448 ' .733 3 2,053905 H 1,013898 0,592376 -0.204295
H 4.668135 -4.195557 2.566382 H 1,85393 -0,3¾718? 1,030546
C 10.23979 0,190486 0,037144 H $.098374 S.344066 -2.397061
H 10,984836 -0.592736 0.203955 H 2.361554 1.098422 -2.364916
H 10,144756 0,38688$ -1,030801 H 3,959494 0.310608 -2,458258
C 7,552038 -2.45029 0.744641 H 3.245019 4.165691 -1,213924 c 6.733743 -4,363 IB 1.961056 H 1,558621 -1.010462 -3.094782
H 6.899983 •$,343865 2.397388 H 2.967636 -1,876068 -2.491919
C 9,03959 -0,244825 2.030555 H 1.923926 -2.308985 -0,461364
H 9.637431 ♦1.098483 2,364786 H 0,445004 -1.56236 -1,0626
H 8.03956? -0.310526 2,458308
€ 7,774736 -3.70504 1,301551
H 8.753386 -4.165735 1,21.4057
C 9.7S0721 1.09174 2.252214
H 10,440573 1.010404 3.0S4488
H 9.031599 1,876081 2,491626
C 10. 8256 1.40848
H 10.075292 2,306760 0,461018
H 11,5S4128 1,561904 106216
0 4,055564 -0.526119 -0,062922
H 5.656352 1,853038 -0.826886
n 3-085538 0.35851 -0.527216
c 6.6537 2.480106 -1.47601
€ 3,33902$ U 7 -0.015062
C 6,49406 3,?33385 -2.0535S
C -0.190761 -0,037405
€ 4.446532 2.450378 -0.744452
[00314] The valence orbitals and LUMO (lowest unoccupied molecular orbital) of complex 1 with energies in Hartrees are shown in Figure 3.
[00315] Imidation reactions of the invention involving N-heteroarenes may proceed through a mechanism similar to the one described herein.
[00316] In summary, the present invention provides novel transition metal complexes including a amine-N- oxide motif and uses of the complexes in imidating arenes or heteroarenes. The inventive methods typically give synthetically useful yields using one
equivalent of arene or heteroarenes. The methods of the invention also do not require coordinating directing groups at or below room temperature. It is thought that the reactions involved in the inventive methods may proceed through a mechanism different from conventional strategies. The inventive transition metal complexes may be oxidized prior to the activation of the arenes or heteroarenes. C-H functionalization proceeds from a high oxidation state complex without the formation of conventional organometallic intermediates.
REFERENCES
[00317] (1) (a) Chen, X.; Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Angew. Chem. Int. Ed. 2009, 48, 5094-5115; (b) Lyons, T. W.; Sanford, M. S. Chem. Rev. 2010, 110, 1147-1169; (c) Engle, K. M.; Mei, T.-S.; Wasa, M.; Yu, J.-Q. Acc. Chem. Res. 2011, 45, 788-802; (d) Neufeldt, S. R.; Sanford, M. S. Acc. Chem. Res. 2012, 45, 936-946.
[00318] (2) Kuhl, N.; Hopkinson, M. N.; Wencel-Delord, J.; Glorius, F. Angew. Chem. Int. Ed. 2012, 51, 10236-10254.
[00319] (3) (a) Mkhalid, I. A. I.; Barnard, J. H.; Marder, T. B.; Murphy, J. M.; Hartwig, J. F. Chem. Rev. 2009, 110, 890-931; (b) Nagib, D. A.; MacMillan, D. W. C. Nature 2011, 480, 224-228.
[00320] (4) (a) Olah, G. A.; Ripudaman, M.; Narang, S. C. Nitration: Methods and Mechanisms; VCH Publishers, Inc.: New York, 1989.
[00321] (5) (a) Grohmann, C; Wang, H.; Glorius, F. Org. Lett. 2011, 14, 656-659; (b) Ng, K.-H.; Chan, A. S. C; Yu, W.-Y. /. Am. Chem. Soc. 2010, 132, 12862-12864; (c) Ng, K.-H.; Zhou, Z.; Yu, W.-Y. Org. Lett. 2011, 14, 272-275; (d) Sun, K.; Li, Y.; Xiong, T.; Zhang, J.; Zhang, Q. /. Am. Chem. Soc. 2011, 133, 1694-1697; (e) Thu, H.-Y.; Yu, W.-Y.; Che, C.-M. /. Am. Chem. Soc. 2006, 128, 9048-9049; (f) Xiao, B.; Gong, T.-J.; Xu, J.; Liu, Z.-J.; Liu, L. /. Am. Chem. Soc. 2011, 133, 1466-1474; (g) Yoo, E. J.; Ma, S.; Mei, T.-S.; Chan, K. S. L.; Yu, J.-Q. /. Am. Chem. Soc. 2011, 133, 7652-7655.
[00322] (6) (a) Froehr, T.; Sindlinger, C. P.; Kloeckner, U.; Finkbeiner, P.; Nachtsheim, B. J. Org. Lett. 2011, 13, 3754-3757; (b) Guo, S.; Qian, B.; Xie, Y.; Xia, C; Huang, H. Org. Lett. 2010, 13, 522-525; (c) Diaz-Requejo, M. M.; Belderrafn, T. R.; Nicasio, M. C;
Trofimenko, S.; Perez, P. J. /. Am. Chem. Soc. 2003, 125, 12078-12079; (d) Li, Z.; Capretto, D. A.; Rahaman, R. O.; He, C. /. Am. Chem. Soc. 2007, 129, 12058-12059; (e) Shrestha, R.; Mukherjee, P.; Tan, Y.; Litman, Z. C; Hartwig, J. F. /. Am. Chem. Soc 2013 ASAP.
[00323] (7) (a) Kim, H. J.; Kim, J.; Cho, S. H.; Chang, S. /. Am. Chem. Soc. 2011, 133, 16382-16385; (b) Antonchick, A. P.; Samanta, R.; Kulikov, K.; Lategahn, J. Angew. Chem.
Int. Ed. 2011, 50, 8605-8608; (c) Samanta, R.; Bauer, J. O.; Strohmann, C; Antonchick, A. P. Org. Lett. 2012, 14, 5518-5521.
[00324] (8) (a) Oloo, W.; Zavalij, P. Y.; Zhang, J.; Khaskin, E.; Vedernikov, A. N. /. Am. Chem. Soc. 2010, 132, 14400-14402; (b) McCall, A. S.; Kraft, S. Organometallics 2012, 31, 3527-3538; (c) McCall, A. S.; Wang, H.; Desper, J. M.; Kraft, S. /. Am. Chem. Soc. 2011, 133, 1832-1848.
[00325] (9) Simmons, E. M.; Hartwig, J. F. Angew. Chem. Int. Ed. 2012, 51, 3066-3072.
[00326] (10) (a) Rosewall, C. F.; Sibbald, P. A.; Liskin, D. V.; Michael, F. E. /. Am.
Chem. Soc. 2009, 131, 9488-9489; (b) Sibbald, P. A.; Rosewall, C. F.; Swartz, R. D.;
Michael, F. E. /. Am. Chem. Soc. 2009, 131, 15945-15951; (c) Racowski, J. M.; Ball, N. D.; Sanford, M. S. /. Am. Chem. Soc. 2011, 133, 18022-18025.
[00327] (11) (a) Olah, G. A. Acc. Chem. Res. 1971, 4, 240-248; (b) Olah, G. A.; Kuhn, S. J.; Flood, S. H. /. Am. Chem. Soc. 1961, 83, 4571-4580; (c) Nakane, R.; Kurihara, O.;
Takematsu, A. /. Org. Chem. 1971, 36, 2753-2756.
[00328] (12) (a) Streitwieser, A.; Jagow, R. H.; Fahey, R. C; Suzuki, S. /. Am. Chem. Soc. 1958, 80, 2326-2332; (b) Taylor, R. Electrophilic Aromatic Substitution; John Wiley & Sons: West Sussex, England, 1990, pg. 33; (c) Melander, L. Isotope Effects on Reaction Rates; The Ronald Press Company: New York, 1960, pg. 112.
[00329] (13) Still, W. C; Kahn, M.; Mitra, A. /. Org. Chem. 1978, 43, 2925-2927.
[00330] (14) Fulmer, G. R.; Miller, A. J. M.; Sherden, N. H.; Gottlieb, H. E.; Nudelman, A.; Stoltz, B. M.; Bercaw, J. E.; Goldberg, K. I. Organometallics. 2010, 29, 2176.
[00331] (15) Sanders, E. B.; Secor, H. V.; Seeman, J. I. J. Org. Chem. 1978, 43, 324.
[00332] (16) Biner, M.; Buergi, H. B.; Ludi, A.; Roehr, C. /. Am. Chem. Soc. 1992, 114, 5197-5203.
[00333] (17) Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; , J. A. Montgomery, J.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd; J. J. Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Normand; J. Raghavachari, K.; Rendell, A.; Burant, J. C; Iyengar, S. S.; Cossi, J. M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adam, C; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C; Ochterski, J. W.;
Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J.
J.; Dapprich, S.; Daniels; A. D. Farkas, O.; Foresman, J. B.; Ortiz, J. V.; Cioslowski; J. Fox, D. J. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford CT, 2009.
[00334] (18) (a) Andrae, D.; Haussermann, U.; Dolg, M.; Stoll, H.; Preuss, H. Theor. Chim. Acta 1990, 77, 123-141. (b) Andrae, D.; Haussermann, U.; Dolg, M.; Stoll, H.; Preuss, H. Theor. Chim. Acta 1991, 78, 247-266.
[00335] (19) Ehlers, A. W.; Bohme, M.; Dapprich, S.; Gobbi, A.; Hollwarth, A.; Jonas, V.; Kohler, K. F.; Stegmann, R.; Veldkamp, A.; Frenking, G. Chem. Phys. Lett. 1993, 208, 111- 114.
[00336] (20) Hariharan, P. C; Pople, J. A. Theor. Chim. Acta 1973, 28, 213-222.
[00337] (21) Dennington, R., II; Keith, T. A.; Millam, J. M. GaussView, Version 5.0.8; Semichem, Inc.
EQUIVALENTS AND SCOPE
[00338] In the claims articles such as "a," "an," and "the" may mean one or more than one unless indicated to the contrary or otherwise evident from the context. Claims or descriptions that include "or" between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. The invention includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. The invention includes embodiments in which more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process.
[00339] Furthermore, the invention encompasses all variations, combinations, and permutations in which one or more limitations, elements, clauses, and descriptive terms from one or more of the listed claims is introduced into another claim. For example, any claim that is dependent on another claim can be modified to include one or more limitations found in any other claim that is dependent on the same base claim. Where elements are presented as lists, e.g., in Markush group format, each subgroup of the elements is also disclosed, and any element(s) can be removed from the group. It should it be understood that, in general, where the invention, or aspects of the invention, is/are referred to as comprising particular elements and/or features, certain embodiments of the invention or aspects of the invention consist, or consist essentially of, such elements and/or features. For purposes of simplicity, those embodiments have not been specifically set forth in haec verba herein. It is also noted that the terms "comprising" and "containing" are intended to be open and permits the inclusion of
additional elements or steps. Where ranges are given, endpoints are included. Furthermore, unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or sub-range within the stated ranges in different embodiments of the invention, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise.
[00340] This application refers to various issued patents, published patent applications, journal articles, and other publications, all of which are incorporated herein by reference. If there is a conflict between any of the incorporated references and the instant specification, the specification shall control. In addition, any particular embodiment of the present invention that falls within the prior art may be explicitly excluded from any one or more of the claims. Because such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the invention can be excluded from any claim, for any reason, whether or not related to the existence of prior art.
[00341] Those skilled in the art will recognize or be able to ascertain using no more than routine experimentation many equivalents to the specific embodiments described herein. The scope of the present embodiments described herein is not intended to be limited to the above Description, but rather is as set forth in the appended claims. Those of ordinary skill in the art will appreciate that various changes and modifications to this description may be made without departing from the spirit or scope of the present invention, as defined in the following claims.
Claims
What is claimed is:
1. A method of preparin
or a salt thereof, comprising contacting a compound of Formula (A), or a salt thereof, with a compound of Formula (B), or a salt thereof, in the presence of a transition metal complex and a single-electron reductant to provide the compound of Formula (I), or the salt thereof:
(C3) (C4)
(C19)
(C24)
(C25)
Ring A is substituted or unsubstituted aryl or substituted or unsubstituted heteroaryl; each instance of RA is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR Al -N(RA1)2, -SRA1, -CN, -SCN, -C(=NRA1)RA1, -C(=NRA1)ORA1, -C(=NRA1)N(RA1)2, - C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2, -N02, -NRA1C(=0)RA1, -NRA1C(=0)ORA1,
NRA1C(=0)N(RA1)2, -OC(=0)R , -OC(=0)OR , -OC(=0)N(RA1)2, -S(=0)2R , - S(=0)2ORA1, -S(=0)2N(RA1)2, -OS(=0)2RA1, -N(RA1)S(=0)2RA1, -N(RA1)S(=0)2N(RA1)2, - Si(RA1)4, or -P(=0)(ORA1)2, or two instances of RA are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or
unsubstituted aryl, or substituted or unsubstituted heteroaryl ring;
each instance of RA1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of RA1 are joined to form a substituted or unsubstituted heterocyclic ring;
k is 0, 1, 2, 3, 4, or 5;
each instance of Ring B is substituted or unsubstituted heteroaryl;
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR B 1 , -N(RB1)2, -SRB1, -CN, -SCN, -C(=NRB1)RB1, -C(=NRB1)ORB1, -C(=NRB1)N(RB1)2, - C(=0)RB1, -C(=0)ORB1, -C(=0)N(RB1)2, -N02, -NRB1C(=0)RB1, -NRB1C(=0)ORB1, - NRB1C(=0)N(RB1)2, -OC(=0)RB1, -OC(=0)ORB1, or -OC(=0)N(RB1)2, or two instances of R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R B 1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R B 1 are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of m is independently 0, 1, 2, 3, 4, or 5;
each instance of M is a transition metal ion;
each instance of Ring B is independently substituted or unsubstituted heteroaryl; each instance of is independently a single or double bond;
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR C 1 , -N(RC1)2, -SRC1, -CN, -SCN, -C(=NRC1)RC1, -C(=NRcl)ORcl, -C(=NRC1)N(RC1)2, - C(=0)Rcl, -C(=0)ORcl, -C(=0)N(Rcl)2, -N02, -NRclC(=0)Rcl, -NRclC(=0)ORcl, - NRclC(=0)N(Rcl)2, -OC(=0)Rcl, -OC(=0)ORcl, or -OC(=0)N(Rcl)2, or two instances of R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R CI is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R C 1 are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of r is independently 0, 1, 2, 3, or 4;
each instance of RD is independently substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring;
p
each instance of R is independently absent, hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or p
two instances of R are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of n is 0 or 1 ;
p is 1 or 2;
each instance of X is an anionic counterion; and
q is 1, 2, or 3.
2. The method of claim 1 further comprising the step of deprotecting the compound of Formula (I), or the salt thereof, with a reductant, a strong acid, or a nucleophile, to provide a compound of Formula (II), or a salt thereof:
3. The method of claim 1 or 2, wherein the transition metal complex is of Formula (CI).
4. The method of claim 1 or 2, wherein the transition metal complex is of the formula:
5. The method of claim 1 or 2 metal complex is of the formula:
6. The method of claim 1 or 2, wherein the transition metal complex is of Formula (C2).
8. The method of claim 1 or 2, wherein the transition metal complex is of any one of Formulae (C3-C25).
9. The method of any one of claims 1-8, wherein Ring A is substituted or unsubstituted phenyl.
The method of an one of claims 1-8, wherein Ring A is of the formula:
11. The method of an one of claims 1-8, wherein Ring A is of the formula:
The method of an one of claims 1-8, wherein Ring A is of the formula:
13. The method of any one of claims 1-8, wherein Ring A is substituted or unsubstituted, 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
14. The method of any one of claims 1-8, wherein Ring A is of the formula:
148
16. The method of any one of claims 1-8, wherein Ring A is substituted or unsubstituted, 9- or 10-membered, bicyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
17. The method of any one of claims 1-16, wherein at least one instance of RA is halogen.
18. The method of any one of claims 1-16, wherein at least one instance of RA is substituted or unsubstituted acyl.
19. The method of any one of claims 1-16, wherein at least one instance of RA is of the formula: -C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2.
20. The method of any one of claims 1-16, wherein at least one instance of RA is -ORA1.
21. The method of any one of claims 1-16, wherein at least one instance of RA is -CN or -N02.
22. The method of any one of claims 1-16, wherein at least one instance of RA is - S(=0)2RA1.
23. The method of any one of claims 1-16, wherein at least one instance of RA is - Si(RA1)4.
24. The method of any one of claims 1-16, wherein at least one instance of RA is - P(=0)(ORA1)2.
25. The method of any one of claims 1-24, wherein k is 0.
26. The method of any one of claims 1-24, wherein k is 1.
27. The method of any one of claims 1-24, wherein k is 2.
28. The method of any one of claims 1-24, wherein k is 3.
29. The method of any one of claims 1-28, wherein all instances of Ring B are substituted or unsubstituted, 5- or 6-membered, monocyclic heteroaryl, wherein one, two, three, or four atoms in the heteroaryl ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
31. of any one of claims 1-28, wherein all instances of Ring B are of the
32. The method of any one of claims 1-31, wherein all instances of R are hydrogen.
33. The method of any one of claims 1-31, wherein both instances of m are 0.
34. The method of any one of claims 1-33, wherein each instance of M is a monovalent transition metal ion.
35. The method of any one of claims 1-33, wherein each instance of M is Ag+, Cu+, or
Co+.
36. The method of any one of claims 1-33, wherein each instance of M is a divalent transition metal ion.
37. The method of any one of claims 1-33, wherein each instance of M is Pd2+, Ni2+,
Cu , Ir , Zn , Mn , or Fe 2+
38. The method of any one of claims 1-33, wherein each instance of M is a trivalent transition metal ion.
39. The method of any one of claims 1-33, wherein each instance of M is Rh3+.
40. The method of any one of claims 1-39, wherein at least one instance of R is hydrogen.
41. The method of any one of claims 1-39, wherein at least one instance of R is substituted or unsubstituted alkyl.
42. The method of any one of claims 1-39, wherein at least one instance of R is substituted or unsubstituted aryl.
43. The method of any one of claims 1-39, wherein at least one instance of R C is -OR CI .
44. The method of any one of claims 1-43, wherein all instances of r are 0.
45. The method of any one of claims 1-43, wherein all instances of r are 1.
46. The method of any one of claims 1-45, wherein at least one instance of RD is substituted or unsubstituted alkyl.
47. The method of any one of claims 1-45, wherein two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring.
48. The method of any one of claims 1-45, wherein two instances of RD are joined to form a substituted or unsubstituted, 4- to 7-membered, monocyclic heterocyclic ring including zero, one, or two double bonds in the heterocyclic ring system, wherein one, two, or three atoms in the heterocyclic ring system are independently selected from the group consisting of nitrogen, oxygen, and sulfur.
49. The method of any one of claims 1-48, wherein at least one instance of R is absent.
50. The method of any one of claims 1-48, wherein at least one instance of R is hydrogen.
51. The method of any one of claims 1-48, wherein at least one instance of R is substituted or unsubstituted alkyl.
52. The method of any one of claims 1-51, wherein each instance of n is 0.
53. The method of any one of claims 1-51, wherein each instance of n is 1.
54. The method of any one of claims 1-53, wherein p is 1.
55. The method of any one of claims 1-54, wherein each instance of X is a monovalent anionic counterion.
56. The method of any one of claims 1-54, wherein each instance of X is C104 , ΟΤΓ, BF , PF4 , PF6 , or SbF6 .
57. The method of any one of claims 1-56, wherein q is 1.
58. The method of any one of claims 1-56, wherein q is 2.
59. The method of any one of claims 1-56, wherein q is 3.
60. The method of any one of claims 1-59, wherein the single-electron reductant is a single-electron reductive, transition metal salt.
61. The method of any one of claims 1-59, wherein the single-electron reductant is a Ag(I) salt or Ru(II) salt.
62. The method of any one of claims 1-59, wherein the single-electron reductant is Ag(2,2'-bipyridine)2C104 or Ru(2,2'-bipyridine)3(PF6)2.
63. The method of any one of claims 2-62, wherein the reductant is a mixture of an alkaline earth metal and an alcohol; or a mixture of zinc and an acid.
64. The method of claim 63, wherein the alkaline earth metal is magnesium; and the alcohol is an unsubstituted C1-6 alkyl alcohol.
65. The method of claim 63, wherein the acid is HCl, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF3S03H, CH3C02H, or CF3C02H.
66. The method of any one of claims 2-62, wherein the strong acid is HCl, HBr, HI, HC104, HN03, H2S04, CH3S03H, CF3S03H, or CF3C02H.
67. The method of any one of claims 2-62, wherein the nucleophile is an alkyl lithium, phenyl lithium, or a Grignard reagent.
68. The method of claim 67, wherein the alkyl lithium is an unsubstituted C . alkyl lithium.
69. The method of claim 67, wherein the Grignard reagent is of the formula: YMgZ; wherein:
Y is unsubstituted C1-6 alkyl, unsubstituted phenyl, or -Si(unsubstituted C1-6 alkyl)3; and
Z is halogen.
70. A transition metal complex of Formula (CI):
each instance of M is a transition metal ion;
each instance of Ring B is independently substituted or unsubstituted heteroaryl; each instance of is independently a single or double bond;
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR C 1 , -N(RC1)2, -SRC1, -CN, -SCN, -C(=NRC1)RC1, -C(=NRcl)ORcl, -C(=NRC1)N(RC1)2, - C(=0)Rcl, -C(=0)ORcl, -C(=0)N(Rcl)2, -N02, -NRclC(=0)Rcl, -NRclC(=0)ORcl, - NRclC(=0)N(Rcl)2, -OC(=0)Rcl, -OC(=0)ORcl, or -OC(=0)N(Rcl)2, or two instances of R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R CI is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R C 1 are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of r is independently 0, 1, 2, 3, or 4;
each instance of RD is independently substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of n is 0 or 1 ;
p is 1 or 2;
each instance of X is an anionic counterion; and
q is 1, 2, or 3.
71. The transition metal complex of claim 70, wherein the transition metal complex is of the formula:
72. The transition metal complex of claim 70, wherein the transition metal complex is of the formula:
73. The transition metal complex of claim 70, wherein the transition metal complex is of the formula:
wherein each instance of X is a monovalent anionic counterion.
74. The transition metal complex of claim 70, wherein the transition metal complex is of the formula:
75. The transition metal complex of claim 70, wherein the transition metal complex is of the formula:
wherein each instance of X is a monovalent anionic counterion. 76. A transition metal complex of Formula (C2):
each instance of M is a transition metal ion;
each instance of RD is independently substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of RD are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of R is independently hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, or a nitrogen protecting group, or two instances of R are joined to form a substituted or unsubstituted heterocyclic ring;
each instance of n is 0 or 1 ;
p is 1 or 2;
each instance of X is an anionic counterion; and
q is 1, 2, or 3.
77. The transition metal complex of claim 76, wherein the transition metal complex is of the formula:
wherein each instance of X is a monovalent anionic counterion.
A transition metal complex of any one of Formulae (C3)-(C25):
(CIO)
C15)
(C19) (C20)
(C25) wherein:
each instance of M is a transition metal ion;
p is 1 or 2;
each instance of X is an anionic counterion; and
q is 1, 2, or 3.
79. The transition metal complex of claim 78, wherein the transition metal compli the formula:
wherein each instance of X is a monovalent anionic counterion.
80. The transition metal complex of claim 78, wherein the transition metal complex is of the formula:
81. The transition metal complex of claim 78, wherein the transition metal complex is of the formula
A kit comprising:
a compound of Formula (B), or a salt thereof:
a transition metal complex of any one of claims 70-81; and
a single-electron reductant;
wherein:
each instance of R is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -OR B 1 , -N(RB1)2, -SRB1, -CN, -SCN, -C(=NRB1)RB1, -C(=NRB1)ORB1, -C(=NRB1)N(RB1)2, - C(=0)RB1, -C(=0)ORB1, -C(=0)N(RB1)2, -N02, -NRB1C(=0)RB1, -NRB1C(=0)ORB1, -
NRB1C(=0)N(RB1)2, -OC(=0)RB1, -OC(=0)ORB1, or -OC(=0)N(RB1)2, or two instances of
R are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl ring; each instance of R B 1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group when attached to an oxygen atom, or a sulfur protecting group when attached to a sulfur atom, or two instances of R B 1 are joined to form a substituted or unsubstituted heterocyclic ring; and
each instance of m is independently 0, 1, 2, 3, 4, or 5. 83. The kit of claim 82 further com rising a compound of Formula (A), or a salt thereof:
wherein:
Ring A is substituted or unsubstituted aryl or substituted or unsubstituted heteroaryl; each instance of RA is independently hydrogen, halogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, -ORA1, -N(RA1)2, -SRA1, -CN, -SCN, -C(=NRA1)RA1, -C(=NRA1)ORA1, -C(=NRA1)N(RA1)2, - C(=0)RA1, -C(=0)ORA1, -C(=0)N(RA1)2, -N02, -NRA1C(=0)RA1, -NRA1C(=0)ORA1, - NRA1C(=0)N(RA1)2, -OC(=0)RA1, -OC(=0)ORA1, -OC(=0)N(RA1)2, -S(=0)2RA1, - Si(RA1)4, or -P(=0)(ORA1)2, or two instances of RA are joined to form a substituted or unsubstituted carbocyclic, substituted or unsubstituted heterocyclic, substituted or
unsubstituted aryl, or substituted or unsubstituted heteroaryl ring;
each instance of RA1 is independently hydrogen, substituted or unsubstituted acyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted carbocyclyl, substituted or unsubstituted heterocyclyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, a nitrogen protecting group when attached to a nitrogen atom, an oxygen protecting group
when attached to an oxygen atom, a sulfur protecting group when attached to a sulfur atom, or a silicon protecting group when attached to a silicon atom, or two instances of RA1 are joined to form a substituted or unsubstituted heterocyclic ring; and
k is 0, 1, 2, 3, 4, or 5.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361872466P | 2013-08-30 | 2013-08-30 | |
| US61/872,466 | 2013-08-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015031725A1 true WO2015031725A1 (en) | 2015-03-05 |
Family
ID=51535577
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2014/053357 Ceased WO2015031725A1 (en) | 2013-08-30 | 2014-08-29 | Transition metal-catalyzed imidation of arenes |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015031725A1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9951069B1 (en) | 2017-01-11 | 2018-04-24 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US9975886B1 (en) | 2017-01-23 | 2018-05-22 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| DE102017111121A1 (en) * | 2017-05-22 | 2018-11-22 | Osram Oled Gmbh | Organic electronic device, use of a sulfonylimide and sulfonylimide |
| CN109942506A (en) * | 2019-03-27 | 2019-06-28 | 上海交通大学 | Asymmetric nickel-catalyzed hydrogenation of N-sulfonimide to prepare chiral amines |
| US10421756B2 (en) | 2015-07-06 | 2019-09-24 | Rodin Therapeutics, Inc. | Heterobicyclic N-aminophenyl-amides as inhibitors of histone deacetylase |
| US10774064B2 (en) | 2016-06-02 | 2020-09-15 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| US10919902B2 (en) | 2015-07-06 | 2021-02-16 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| US11225475B2 (en) | 2017-08-07 | 2022-01-18 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US11993586B2 (en) | 2018-10-22 | 2024-05-28 | Novartis Ag | Crystalline forms of potassium channel modulators |
-
2014
- 2014-08-29 WO PCT/US2014/053357 patent/WO2015031725A1/en not_active Ceased
Non-Patent Citations (62)
| Title |
|---|
| "Encyclopedia of Reagents for Organic Synthesis", 1995, JOHN WILEY AND SONS |
| "Handbook of Chemistry and Physics" |
| ANDRAE, D.; HÄUSSERMANN, U.; DOLG, M.; STOLL, H.; PREUSS, H., THEOR. CHIM. ACTA, vol. 77, 1990, pages 123 - 141 |
| ANDRAE, D.; HÄUSSERMANN, U.; DOLG, M.; STOLL, H.; PREUSS, H., THEOR. CHIM. ACTA, vol. 78, 1991, pages 247 - 266 |
| ANTONCHICK, A. P.; SAMANTA, R; KULIKOV, K.; LATEGAHN, J., ANGEW. CHEM. INT. ED., vol. 50, 2011, pages 8605 - 8608 |
| BINER, M.; BUERGI, H. B.; LUDI, A.; ROEHR, C., J. AM. CHEM. SOC., vol. 114, 1992, pages 5197 - 5203 |
| CARRUTHERS: "Some Modern Methods of Organic Synthesis", 1987, CAMBRIDGE UNIVERSITY PRESS |
| CHEN, X.; ENGLE, K. M.; WANG, D.-H.; YU, J.-Q., ANGEW. CHEM. INT. ED., vol. 48, 2009, pages 5094 - 5115 |
| DENNINGTON, R., II; KEITH, T. A; MILLAM, J. M.: "GaussView", SEMICHEM, INC. |
| DIAZ-REQUEJO, M. M.; BELDERRAIN, T. R.; NICASIO, M. C.; TROFIMENKO, S.; PEREZ, P. J., J. AM. CHEM. SOC., vol. 125, 2003, pages 12078 - 12079 |
| EHLERS, A. W.; BOHME, M.; DAPPRICH, S.; GOBBI, A; HOLLWARTH, A; JONAS, V.; KOHLER, K. F.; STEGMANN, R.; VELDKAMP, A; FRENKING, G., CHEM. PHYS. LETT., vol. 208, 1993, pages 111 - 114 |
| ELIEL: "Stereochemistry of Carbon Compounds", 1962, MCGRAW-HILL |
| ENGLE, K. M.; MEI, T.-S.; WASA, M.; YU, J.-Q., ACC. CHEM. RES., vol. 45, 2011, pages 788 - 802 |
| FRISCH, M. J.; TRUCKS, G. W.; SCHLEGEL, H. B.; SCUSERIA, G. E.; ROBB, M. A.; CHEESEMAN, J. R; SCALMANI, G.; BARONE, V.; MENNUCCI,: "Gaussian 09, Revision A.02", 2009, GAUSSIAN, INC. |
| FROEHR, T.; SINDLINGER, C. P.; KLOECKNER, U.; FINKBEINER, P.; NACHTSHEIM, B., J. ORG. LETT., vol. 13, 2011, pages 3754 - 3757 |
| FULMER, G. R; MILLER, A. J. M.; SHERDEN, N. H.; GOTTLIEB, H. E.; NUDELMAN, A.; STOLTZ, B. M.; BERCAW, J. E.; GOLDBERG, K. I., ORGANOMETALLICS, vol. 29, 2010, pages 2176 |
| GREGORY B. BOURSALIAN ET AL: "Pd-Catalyzed Aryl C-H Imidation with Arene as the Limiting Reagent", JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 135, no. 36, 11 September 2013 (2013-09-11), pages 13278 - 13281, XP055152685, ISSN: 0002-7863, DOI: 10.1021/ja4064926 * |
| GROHMANN, C.; WANG, H.; GLORIUS, F., ORG. LETT., vol. 14, 2011, pages 656 - 659 |
| GUO, S.; QIAN, B.; XIE, Y.; XIA, C.; HUANG, H., ORG. LETT., vol. 13, 2010, pages 522 - 525 |
| HARIHARAN, P. C.; POPLE, J. A., THEOR. CHIM. ACTA, vol. 28, 1973, pages 213 - 222 |
| JACQUES ET AL.: "Enantiomers, Racemates and Resolutions", 1981, WILEY INTERSCIENCE |
| KAI SUN ET AL: "Palladium-Catalyzed C-H Aminations of Anilides with N -Fluorobenzenesulfonimide", JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 133, no. 6, 16 February 2011 (2011-02-16), pages 1694 - 1697, XP055152609, ISSN: 0002-7863, DOI: 10.1021/ja1101695 * |
| KIM, H. J.; KIM, J.; CHO, S. H.; CHANG, S., J. AM. CHEM. SOC., vol. 133, 2011, pages 16382 - 16385 |
| KUHL, N.; HOPKINSON, M. N.; WENCEL-DELORD, J.; GLORIUS, F., ANGEW. CHEM. INT. ED., vol. 51, 2012, pages 10236 - 10254 |
| L. FIESER; M. FIESER: "Fieser and Fieser's Reagents for Organic Synthesis", 1994, JOHN WILEY AND SONS |
| LAROCK: "Comprehensive Organic Transformations", 1989, VCH PUBLISHERS, INC. |
| LI, Z.; CAPRETTO, D. A.; RAHAMAN, R. 0.; HE, C., J. AM. CHEM. SOC., vol. 129, 2007, pages 12058 - 12059 |
| LYONS, T. W.; SANFORD, M. S., CHEM. REV., vol. 110, 2010, pages 1147 - 1169 |
| MCCALL, A. S.; KRAFT, S., ORGANOMETALLICS, vol. 31, 2012, pages 3527 - 3538 |
| MCCALL, A. S.; WANG, H.; DESPER, J. M.; KRAFT, S., J. AM. CHEM. SOC., vol. 133, 2011, pages 1832 - 1848 |
| MELANDER, L.: "Isotope Effects on Reaction Rates", 1960, THE RONALD PRESS COMPANY, pages: 112 |
| MKHALID, I. A. I.; BARNARD, J. H.; MARDER, T. B.; MURPHY, J. M.; HARTWIG, J. F., CHEM. REV., vol. 110, 2009, pages 890 - 931 |
| NAGIB, D. A; MACMILLAN, D. W. C., NATURE, vol. 480, 2011, pages 224 - 228 |
| NAKANE, R.; KURIHARA, 0.; TAKEMATSU, A., J. ORG. CHEM., vol. 36, 1971, pages 2753 - 2756 |
| NEUFELDT, S. R.; SANFORD, M. S., ACC. CHEM. RES., vol. 45, 2012, pages 936 - 946 |
| NG, K.-H.; CHAN, A. S. C.; YU, W.-Y., J. AM. CHEM. SOC., vol. 132, 2010, pages 128 - 1286 |
| NG, K.-H.; ZHOU, Z.; YU, W.-Y., ORG. LETT., vol. 14, 2011, pages 272 - 275 |
| OLAH, G. A., ACC. CHEM. RES., vol. 4, 1971, pages 240 - 248 |
| OLAH, G. A.; KUHN, S. J.; FLOOD, S. H., J. AM. CHEM. SOC., vol. 83, 1961, pages 4571 - 4580 |
| OLAH, G. A.; RIPUDAMAN, M.; NARANG, S. C.: "Nitration: Methods and Mechanisms", 1989, VCH PUBLISHERS, INC. |
| OLOO, W.; ZAVALIJ, P. Y.; ZHANG, J.; KHASKIN, E.; VEDERNIKOV, A. N., J. AM. CHEM. SOC., vol. 132, 2010, pages 14400 - 14402 |
| R. LAROCK: "Comprehensive Organic Transformations", 1989, VCH PUBLISHERS |
| RACOWSKI, J. M.; BALL, N. D.; SANFORD, M. S., J. AM. CHEM. SOC., vol. 133, 2011, pages 18022 - 18025 |
| ROSEWALL, C. F.; SIBBALD, P. A.; LISKIN, D. V.; MICHAEL, F. E., J. AM.CHEM. SOC., vol. 131, 2009, pages 9488 - 9489 |
| SAMANTA, R; BAUER, J. 0.; STROHMANN, C.; ANTONCHICK, A., P. ORG. LETT., vol. 14, 2012, pages 5518 - 5521 |
| SANDERS, E. B.; SECOR, H. V.; SEEMAN, J. 1, J. ORG. CHEM., vol. 43, 1978, pages 324 |
| SHRESTHA, R.; MUKHERJEE, P.; TAN, Y.; LITMAN, Z. C.; HARTWIG, J. F., J. AM. CHEM. SOC, 2013 |
| SIBBALD, P. A.; ROSEWALL, C. F.; SWARTZ, R. D.; MICHAEL, F. E., J. AM. CHEM. SOC., vol. 131, 2009, pages 15945 - 15951 |
| SIMMONS, E. M.; HARTWIG, J. F., ANGEW. CHEM. INT. ED., vol. 51, 2012, pages 3066 - 3072 |
| SMITH; MARCH: "March's Advanced Organic Chemistry", 2001, JOHN WILEY & SONS, INC. |
| STILL, W. C.; KAHN, M.; MITRA, A., J. ORG. CHEM., vol. 43, 1978, pages 2925 - 2927 |
| STREITWIESER, A.; JAGOW, R. H.; FAHEY, R. C.; SUZUKI, S., J. AM. CHEM. SOC., vol. 80, 1958, pages 2326 - 2332 |
| SUN, K.; LI, Y.; XIONG, T.; ZHANG, J.; ZHANG, Q., J. AM. CHEM. SOC., vol. 133, 2011, pages 1694 - 1697 |
| T. W. GREENE; P. G. M. WUTS: "Protecting Groups in Organic Synthesis", 1999, JOHN WILEY & SONS |
| T.W. GREENE; P.G.M. WUTS: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY AND SONS |
| TAYLOR, R.: "Electrophilic Aromatic Substitution", 1990, JOHN WILEY & SONS, pages: 33 |
| THOMAS SORRELL: "Organic Chemistry, University Science Books", 1999 |
| THU, H.-Y.; YU, W.-Y.; CHE, C.-M., J. AM. CHEM. SOC., vol. 128, 2006, pages 9048 - 9049 |
| WILEN ET AL., TETRAHEDRON, vol. 33, 1977, pages 2725 |
| WILEN: "Tables of Resolving Agents and Optical Resolutions", 1972, UNIV. OF NOTRE DAME PRESS, pages: 268 |
| XIAO, B.; GONG, T.-J.; XU, J.; LIU, Z.-J.; LIU, L., J. AM. CHEM. SOC., vol. 133, 2011, pages 1466 - 1474 |
| YOO, E. J.; MA, S.; MEI, T.-S.; CHAN, K. S. L.; YU, J.-Q., J. AM. CHEM. SOC., vol. 133, 2011, pages 7652 - 7655 |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10421756B2 (en) | 2015-07-06 | 2019-09-24 | Rodin Therapeutics, Inc. | Heterobicyclic N-aminophenyl-amides as inhibitors of histone deacetylase |
| US11858939B2 (en) | 2015-07-06 | 2024-01-02 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| US10919902B2 (en) | 2015-07-06 | 2021-02-16 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| US10774064B2 (en) | 2016-06-02 | 2020-09-15 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| US10793567B2 (en) | 2017-01-11 | 2020-10-06 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11225479B2 (en) | 2017-01-11 | 2022-01-18 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10519149B2 (en) | 2017-01-11 | 2019-12-31 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10696673B2 (en) | 2017-01-11 | 2020-06-30 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11987580B2 (en) | 2017-01-11 | 2024-05-21 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11286256B2 (en) | 2017-01-11 | 2022-03-29 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US9951069B1 (en) | 2017-01-11 | 2018-04-24 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10717728B2 (en) | 2017-01-23 | 2020-07-21 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| US9975886B1 (en) | 2017-01-23 | 2018-05-22 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| US10351553B2 (en) | 2017-01-23 | 2019-07-16 | Cadent Therapeutics, Inc. | Potassium channel modulators |
| DE102017111121A1 (en) * | 2017-05-22 | 2018-11-22 | Osram Oled Gmbh | Organic electronic device, use of a sulfonylimide and sulfonylimide |
| US11225475B2 (en) | 2017-08-07 | 2022-01-18 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US11912702B2 (en) | 2017-08-07 | 2024-02-27 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US11993586B2 (en) | 2018-10-22 | 2024-05-28 | Novartis Ag | Crystalline forms of potassium channel modulators |
| CN109942506B (en) * | 2019-03-27 | 2020-12-29 | 上海交通大学 | Asymmetric nickel-catalyzed hydrogenation of N-sulfonimide to prepare chiral amines |
| CN109942506A (en) * | 2019-03-27 | 2019-06-28 | 上海交通大学 | Asymmetric nickel-catalyzed hydrogenation of N-sulfonimide to prepare chiral amines |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2015031725A1 (en) | Transition metal-catalyzed imidation of arenes | |
| Vogt et al. | Activation of nitriles by metal ligand cooperation. reversible formation of ketimido-and enamido-rhenium PNP pincer complexes and relevance to catalytic design | |
| Manz et al. | Pairwise H2/D2 exchange and H2 substitution at a bimetallic dinickel (II) complex featuring two terminal hydrides | |
| Jindabot et al. | Palladium (II) complexes featuring bidentate pyridine–triazole ligands: Synthesis, structures, and catalytic activities for Suzuki–Miyaura coupling reactions | |
| CA3012078A1 (en) | Selective estrogen receptor degraders and uses thereof | |
| EP3426654B1 (en) | Direct palladium-catalyzed aromatic fluorination | |
| WO2014134141A1 (en) | Synthesis of acyclic and cyclic amines using iron-catalyzed nitrene group transfer | |
| WO2019009956A1 (en) | Fe/cu-mediated ketone synthesis | |
| Donnelly et al. | Controlling the Selectivity of C–H Activation in Pyridinium Triazolylidene Iridium Complexes: Mechanistic Details and Influence of Remote Substituents | |
| Tanabe et al. | Reaction Mechanism of the C⋮ N Triple Bond Cleavage of β-Ketonitriles on a Molybdenum (0) Center1 | |
| WO2013003315A2 (en) | Methods for preparing isoquinolines | |
| Herasymchuk et al. | Simple Modular Synthetic Approaches to Asymmetric NN′ N′′, NN′ C, or NN′ P-Type Amido Pincer Ligands: Synthesis, Characterisation, and Preliminary Ligation Studies | |
| Farrer et al. | Platinum (IV) azido complexes undergo copper-free click reactions with alkynes | |
| Komuro et al. | Directed ortho-C–H silylation coupled with trans-selective hydrogenation of arylalkynes catalyzed by ruthenium complexes of a xanthene-based Si, O, Si-chelate ligand,“Xantsil” | |
| Murugan et al. | Versatile formation of Ru (II) hydrazone complexes: Structure, theoretical studies and catalytic activity in α-alkylation | |
| Legzdins et al. | Ligand elaboration mediated by a Cp* W (NO) template: stepwise incorporation of small molecules into a tungsten vinyl fragment | |
| Bertogg et al. | A novel chiral ferrocene-based amidine/amidinato ligand and its rhodium complexes | |
| Salazar et al. | (Alkynyl) dicobalt Hexacarbonyl-Mediated Radical Cyclizations | |
| Maurya et al. | Trinuclear cis-dioxidomolybdenum (VI) complexes of compartmental C3 symmetric ligands: Synthesis, characterization, DFT study and catalytic application for hydropyridines (Hps) via the Hantzsch reaction | |
| Du et al. | Studies with the Ruthenacarborane Complex [Ru (CO)(PPh3)(THF)(η5-7, 8-C2B9H11)]: Reactions with Terminal Alkynes | |
| Mansour et al. | Triazolato Pd (II) and Pt (II) complexes of 2, 6-bis (1-ethylbenzimidazol-2′-yl) pyridine formed via catalyst-free [3+ 2] click reactions | |
| Akhmadiev et al. | Synthesis, structure and catalytic activity of novel five-membered Pd (II) and Pt (II) metallaheterocycles based on 1, 2-bis (3, 5-dimethylisoxazol-4-yl-methylsulfanyl) ethane | |
| WO2020061300A1 (en) | Novel annulation catalysts via direct c-h bond amination | |
| Zwettler et al. | Templated C–C and C–N Bond Formation Facilitated by a Molybdenum (VI) Metal Center | |
| Liu et al. | Reactions of hydrogen with ruthenium and osmium complexes containing tridentate ligands Cy2PCH2CH (CH2) 2PCy2 and 2, 6-(Ph2PCH2) 2C6H3 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14762200 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14762200 Country of ref document: EP Kind code of ref document: A1 |