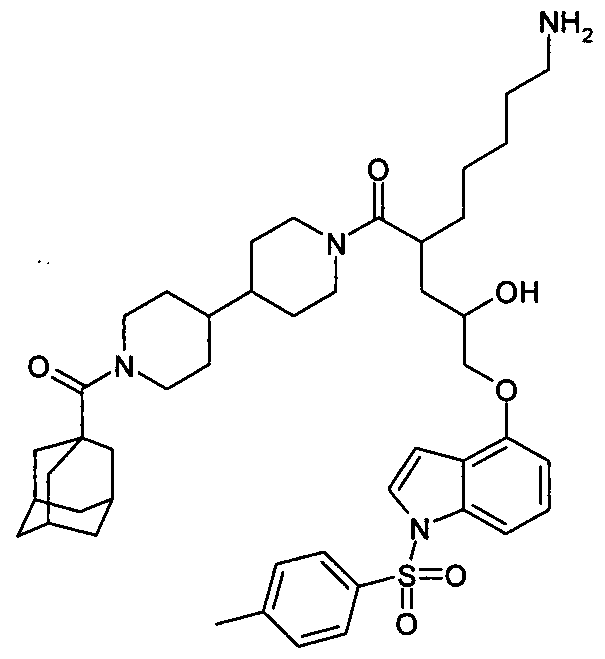
WO2012040754A2 - Novel thiazolamine derivates as differentiation accelerators - Google Patents
Novel thiazolamine derivates as differentiation accelerators Download PDFInfo
- Publication number
- WO2012040754A2 WO2012040754A2 PCT/AT2011/000393 AT2011000393W WO2012040754A2 WO 2012040754 A2 WO2012040754 A2 WO 2012040754A2 AT 2011000393 W AT2011000393 W AT 2011000393W WO 2012040754 A2 WO2012040754 A2 WO 2012040754A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- benzyl
- arc
- thiazolamine
- aniline
- ylidene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- SYTYSHBXSMDTPX-GHVJWSGMSA-N C(c1ccccc1)N(C=C(c1ccccc1)S1)/C1=N\c1ccccc1 Chemical compound C(c1ccccc1)N(C=C(c1ccccc1)S1)/C1=N\c1ccccc1 SYTYSHBXSMDTPX-GHVJWSGMSA-N 0.000 description 1
- ISOYGZVWGGRWJG-WUKNDPDISA-N C(c1ccccc1)N(C=CS1)/C1=N\c1ccccc1 Chemical compound C(c1ccccc1)N(C=CS1)/C1=N\c1ccccc1 ISOYGZVWGGRWJG-WUKNDPDISA-N 0.000 description 1
- VJQDGAUHVGYSNY-UHFFFAOYSA-N C(c1ccccc1)N(c1ncc[s]1)c1ccccc1 Chemical compound C(c1ccccc1)N(c1ncc[s]1)c1ccccc1 VJQDGAUHVGYSNY-UHFFFAOYSA-N 0.000 description 1
- MGKYEWWFHSESJN-UHFFFAOYSA-N CCOC(c(cc1)ccc1N(C)c1c[s]c(-c2ccccc2)n1)=O Chemical compound CCOC(c(cc1)ccc1N(C)c1c[s]c(-c2ccccc2)n1)=O MGKYEWWFHSESJN-UHFFFAOYSA-N 0.000 description 1
- XGQHKYJRIXHCGJ-UHFFFAOYSA-N Fc(cncc1)c1-c1cnc(N(Cc2ccccc2)c2ccccc2)[s]1 Chemical compound Fc(cncc1)c1-c1cnc(N(Cc2ccccc2)c2ccccc2)[s]1 XGQHKYJRIXHCGJ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/38—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the present invention relates to novel thiazolamine derivatives for use as accelerators of cell differentiation.
- Molecules containing a thiazolamine moiety exhibit a number of interesting biological activities, depending on their substituents and the substitution pattern on the thiazole ring.
- compounds of the formula below have been shown to be potent inhibitors of histone deacetylase and tyrosine kinase:
- Ri and R 2 are selected from aryl, heteroaryl, hydrogen and acyl and R 3 is selected from aryl and heteroaryl
- R 3 is selected from aryl and heteroaryl
- N-phenylthiazolamine derivatives are also known as selective aurora kinase inhibitors (CB Andersen et al., ACS Chemical Biology 3 (3), 180-192 (2008)).
- molecules of the formula below have been reported to induce autophagic cell death in renal cancer cells:
- Ri and R 2 are selected from aryl and heteroaryl (MP Hay et al., J.Med.Chem. 53 (2), 787-797 (2010)). Due to the increasing importance of methods for influencing cell differentiation processes, attempts have been made to identify or synthesize molecules that have activity in promoting cell differentiation or undifferentiation. With regard to the use of thiazolamines for such purposes, N. Bouquier et al., Chem. Biol.
- thiazolamine derivatives have the ability to Inhibition of the Trio / RhoG / Rac1 signaling pathway, inter alia, inhibits the differentiation of stimulated PC12 and C2C12 cells into neurites or myotubes.
- the thiazolamine derivatives used for this purpose are in all cases two- or three-membered fused ring systems in which a nitrogen of the thiazolamine represents a bridgehead atom, the following structure being predominant:
- R stands for variously substituted aryls or heteroaryls.
- WO 2005/047524 A2 discloses molecules of similar size based on purines which induce the dedifferentiation of lineage-determined mammalian cells ("lineage-committed mammalian cells").
- Molecules in such size ranges ie, molecular weights below about 1,000 g / mol, are often referred to as "small molecules", and the majority of all synthetic drugs are in this molecular weight range.
- chemical compounds which promote the differentiation of cells for example in the form of such "small molecules” are desirable for numerous applications, for example for stem cell therapy. Examples here- for are hardly known.
- a compound called the adhesin has been reported to be able to accelerate the differentiation of hippocampal neurons (Hoshino, M. et al., Biochemical Journal 427, 297 (2010)).
- Wrenchnolol and Adhesamine are certainly complex compounds whose synthesis is associated with relatively high costs.
- Another interesting compound is neuropathiazole, a 4-thiazolamine that has been reported to induce the differentiation of multipotential neural progenitor cells of the hippocampus into neurons in adults.
- the object of the present invention was to produce further small molecules, in particular those based on thiazolamine, which are capable of accelerating the differentiation of mammalian cells and which are relatively easy to synthesize.
- R 1 to R 3 are independently selected or absent from hydrogen, benzyl, aryl and heteroaryl, with at most one of the radicals R 1 to R 3 being hydrogen and R 4 being selected from benzyl, aryl and heteroaryl,
- R 1 to R 4 are each independently optionally substituted with one or more substituents selected from halogen (F, Cl, Br, I), hydroxy, Ci -3 - alkyl, Ci -3 alkoxy, carboxy, C 1-3 alkylcarbonyl , d-3-alkoxycarbonyl, nitro, amino, mono- and di-Ci-3-alkylamino and cyano, in which the alkyl groups are optionally halogenated substituted;
- n 0 to 2;
- the above thiazolamine derivatives can be obtained in only three synthetic steps, whereby the cost and the cost of preparation compared to the compounds of the prior art can be significantly reduced.
- such derivatives have been shown to be effective in promoting cell differentiation in a C2C12 mouse cell model.
- the invention relates to the use of compounds of the above formula as cell differentiation accelerators.
- Aryl and heteroaryl are to be understood here as mononuclear and polynuclear aromatic ring systems, preferably those having 5 to 10 ring atoms, more preferably aromatic rings having 5 or 6 ring atoms.
- the heteroatoms, of which one or more Others may be present, are preferably selected from nitrogen, sulfur and oxygen, with nitrogen and sulfur being particularly preferred and a single nitrogen heteroatom in the heteroaryl radical is more preferred.
- nitrogen and sulfur being particularly preferred and a single nitrogen heteroatom in the heteroaryl radical is more preferred.
- phenyl is preferred among the aryl and heteroaryl radicals.
- an optional double bond shown in broken line in the formula is present, either endo or exocyclic, ie extending from C2 to either the thiazole or amine nitrogen.
- R 1 to R 3 are preferably selected independently of one another from benzyl and phenyl, and on the two nitrogen atoms preferably only a total of two substituents are present. These two substituents are in particularly preferred embodiments each a phenyl and a benzyl group.
- This radical is particularly preferably bonded to C 5 of the thiazole ring and is preferably selected from aryl and heteroaryl, more preferably from phenyl, thiophenyl and pyridinyl wherein phenyl is particularly preferred.
- the optional substituents from R 1 to R 4 are preferably selected from fluoro, methyl, trifluoromethyl, nitro, carboxy and C 1-3 -alkoxycarbonyl, more preferably from fluoro, methyl and nitro.
- the inventors of the present application believe that, in the presence of larger or even bulky substituents, the interaction of the thiazolamine derivatives of the invention with translation factors or other components of the cells essential to induce cell differentiation is hindered the effect of the invention would be impaired. Furthermore, it is believed that more electron attractive substituents are to be preferred because they appear to exert an activating effect.
- Preferred thiazolamine derivatives of the invention are as follows
- GC-MS measurements were taken on a Thermo Finnigan Focus GC / DSQ II using a standard capillary acid BGB 5 (30 mx 0.32 mm ID) and using one of the following standardized temperature profiles: STD 100-280 ° C Short method: 2 Minutes at 100 ° C, at 18 ° C / min to 280 ° C, 5 minutes at 280 ° C; or STD 100-280 ° C Long Procedure: 2 minutes at 100 ° C, at 6 ° C / min to 150 ° C, 3 minutes at 150 ° C, at 10 ° C / min to 280 ° C, 10 minutes at 280 ° C. Thin-layer chromatography was carried out with silica gel plates 60 F245 from Merck.
- N-phenyl-2-thiazolamine 3 (30 mg, 0.17 mmol, 1.0 equiv.), Benzylbromide (38 mg, 0.22 mmol, 1.3 equiv.) And NaH (4.1 mg, 0). 17 mmol, 1.0 equiv) were dissolved in dry DMF (2 ml) and stirred at room temperature for 5 h. The reaction mixture was then diluted with EtOAc, concentrated on silica gel on a rotary evaporator and purified by MPLC (LP: EtOAc 85:15 + 5% TEA). The title compound 21 was obtained as a beige solid (30 mg, 66% of theory).
- N-phenyl-2-thiazolamine 3 (264 mg, 1.50 mmol, 1.0 equiv.), Benzyl bromide (334 mg, 1.96 mmol, 1.3 equiv.) And triethylamine (152 mg, 1.50 mmol , 1.0 equiv) were dissolved at 0 ° C in dry dioxane (4 ml), allowed to warm slowly and stirred at 120 ° C on a heating block for 3 days. The reaction mixture was then diluted with EtOAc, concentrated on silica gel on a rotary evaporator and purified by MPLC (LP: EtOAc 85:15 + 4% TEA). The title compound 22 was obtained as a pale yellow solid (287 mg, 72% of theory).
- the cell material was produced at the Institute of Pharmacology of the Medical University of Vienna.
- C2C12 mouse cells were used as a model to determine the differentiation-promoting properties of the compounds prepared as described above.
- the C2C12 cells were incubated for two days in culture dishes with growth medium (Dulbecco's modified Eagle's medium, DMEM, with 4.5 g / l glucose, 4 mM L-glutamine, 50 U / ml penicillin, 50 pg / ml streptomycin and 20% fetal calf serum) were incubated before the test compounds were added in differentiation medium (substantially identical to the growth medium, except that it contained 2% horse serum instead of fetal calf serum).
- growth medium Dulbecco's modified Eagle's medium, DMEM, with 4.5 g / l glucose, 4 mM L-glutamine, 50 U / ml penicillin, 50 pg / ml streptomycin and 20% fetal cal
- the compounds were first dissolved in DMSO, and these solutions were added to aliquots of the differentiation medium. As a comparison was pure DMSO dissolved in differentiation medium. First, 5 ⁇ solutions of the compounds were used, each experiment being carried out in duplicate.
- the latter was removed, after which 3 ml of the above solutions of the respective compound and DMSO in differentiation medium were added to each cell culture. Subsequently, the cells were incubated at 37 ° C for 7 days. Meanwhile, the medium was exchanged twice, after 3 and 5 days. After 7 days, the cells were harvested, the medium was removed, and the cells were washed with PBS (phosphate buffered saline) to remove any residual differentiation medium. This is of essential importance as the medium contains proteins that could influence subsequent proteome analyzes.
- PBS phosphate buffered saline
- test compounds also developed more or less toxicity to the cells, as evidenced by dead cells. showed material in the samples. Both effects, ie acceleration of differentiation and toxicity, were enhanced during the following days.
- the present invention provides a group of compounds capable of accelerating the differentiation of cells which are accessible in a relatively simple and inexpensive manner and which show little to no toxicity.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Thiazole And Isothizaole Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Neue Thiazolamin-Derivate als Differenzierungsbeschleuniger New thiazolamine derivatives as differentiation accelerators
Die vorliegende Erfindung betrifft neue Thiazolamin-Derivate zur Verwendung als Beschleuniger der Differenzierung von Zellen. The present invention relates to novel thiazolamine derivatives for use as accelerators of cell differentiation.
Moleküle, die eine Thiazolamin-Gruppierung enthalten, zeigen in Abhängigkeit von deren Substituenten und dem Substitutionsmuster am Thiazolring eine Reihe von interessanten biologischen Aktivitäten. So haben sich etwa Verbindungen der nachstehenden Formel als wirksame Inhibitoren von Histon-Deacetylase und Tyrosin-Kinase erwiesen: Molecules containing a thiazolamine moiety exhibit a number of interesting biological activities, depending on their substituents and the substitution pattern on the thiazole ring. For example, compounds of the formula below have been shown to be potent inhibitors of histone deacetylase and tyrosine kinase:
worin Ri und R2 aus Aryl, Heteroaryl, Wasserstoff und Acyl ausgewählt sind und R3 aus Aryl und Heteroaryl ausgewählt ist (S. Mahboobi et al., J. Med. Chem. 52(8), 2265-2279 (2009)). N-Phenylthiazolamin-Derivate sind zudem als selektive Aurora- Kinase-Inhibitoren bekannt (C.B. Andersen et al., ACS Chemical Biology 3(3), 180- 192 (2008)). Über Moleküle der nachstehenden Formel wurde hingegen berichtet, dass sie bei Nierenkrebszellen autophagen Zelltod auslösen können: wherein Ri and R 2 are selected from aryl, heteroaryl, hydrogen and acyl and R 3 is selected from aryl and heteroaryl (Mahboobi, S., et al., J. Med. Chem. 52 (8), 2265-2279 (2009)) , N-phenylthiazolamine derivatives are also known as selective aurora kinase inhibitors (CB Andersen et al., ACS Chemical Biology 3 (3), 180-192 (2008)). However, molecules of the formula below have been reported to induce autophagic cell death in renal cancer cells:
worin Ri und R2 aus Aryl und Heteroaryl ausgewählt sind (M.P. Hay et al., J. Med. Chem. 53(2), 787-797 (2010)). Aufgrund der zunehmenden Bedeutung von Verfahren zur Beeinflussung der Zell- differenzierungsprozesse wurden Versuche unternommen, Moleküle zu identifizieren oder zu synthetisieren, die Aktivität bei der Förderung von Zelldifferenzierung oder -entdifferenzierung aufweisen. Bezüglich der Verwendung von Thiazolaminen für sol- che Zwecke haben N. Bouquier et al., Chem. Biol. 16, 657-666 (2009), nachgewiesen, dass manche, im weitesten Sinne als Thiazolamin-Derivate zu bezeichnende Substanzen die Fähigkeit aufweisen, durch Inhibierung des Trio/RhoG/ Rac1-Signali- sierungswegs u.a. die Differenzierung von stimulierten PC12- und C2C12-Zellen zu Neuriten bzw. Myotuben zu hemmen. Bei den dazu eingesetzten Thiazolamin-Deri- vaten handelt es sich durchwegs um zwei- oder dreikernige kondensierte Ringsysteme, bei denen ein Stickstoff des Thiazolamins ein Brückenkopfatom darstellt, wobei die folgende Struktur überwiegt: wherein Ri and R 2 are selected from aryl and heteroaryl (MP Hay et al., J.Med.Chem. 53 (2), 787-797 (2010)). Due to the increasing importance of methods for influencing cell differentiation processes, attempts have been made to identify or synthesize molecules that have activity in promoting cell differentiation or undifferentiation. With regard to the use of thiazolamines for such purposes, N. Bouquier et al., Chem. Biol. 16, 657-666 (2009), have demonstrated that some substances, broadly referred to as thiazolamine derivatives, have the ability to Inhibition of the Trio / RhoG / Rac1 signaling pathway, inter alia, inhibits the differentiation of stimulated PC12 and C2C12 cells into neurites or myotubes. The thiazolamine derivatives used for this purpose are in all cases two- or three-membered fused ring systems in which a nitrogen of the thiazolamine represents a bridgehead atom, the following structure being predominant:
und bei den übrigen Thiazolamin-Derivaten der kondensierte Benzolring fehlt. R steht in der obigen Formel für verschiedenartig substituierte Aryle bzw. Heteroaryle. and in the other thiazolamine derivatives of the condensed benzene ring is missing. In the above formula, R stands for variously substituted aryls or heteroaryls.
Aus der WO 2005/047524 A2 sind Moleküle ähnlicher Größe auf der Basis von Purinen bekannt, die die Entdifferenzierung von Stammbaum-determinierten Säugerzel- len ("lineage-committed mammalian cells") induzieren. Moleküle in solchen Größenbereichen, d.h. mit Molekulargewichten unterhalb von etwa 1.000 g/mol, werden häufig als "Kleinmoleküle" bezeichnet, und die Mehrzahl aller synthetischen Arzneimittel liegt in diesem Molekulargewichtsbereich. Im Gegensatz zur obigen Literatur, in der Differenzierungsinhibitoren und Entdifferen- zierungsmittel beschrieben werden, sind jedoch für zahlreiche Anwendungen, wie z.B. für die Stammzelltherapie, chemische Verbindungen wünschenswert, die die Differenzierung von Zellen fördern, z.B. in Form solcher "Kleinmoleküle". Beispiele hier- für sind jedoch kaum bekannt. So wurde etwa für eine als Adhesamin bezeichnete Verbindung berichtet, dass sie in der Lage ist, die Differenzierung von Hippocampus- Neuronen zu beschleunigen (M. Hoshino et al., Biochemical Journal 427, 297 (2010)). WO 2005/047524 A2 discloses molecules of similar size based on purines which induce the dedifferentiation of lineage-determined mammalian cells ("lineage-committed mammalian cells"). Molecules in such size ranges, ie, molecular weights below about 1,000 g / mol, are often referred to as "small molecules", and the majority of all synthetic drugs are in this molecular weight range. In contrast to the above literature, in which differentiation inhibitors and de-differentiating agents are described, however, chemical compounds which promote the differentiation of cells, for example in the form of such "small molecules", are desirable for numerous applications, for example for stem cell therapy. Examples here- for are hardly known. For example, a compound called the adhesin has been reported to be able to accelerate the differentiation of hippocampal neurons (Hoshino, M. et al., Biochemical Journal 427, 297 (2010)).
"Adhesamin" "Adhesamin"
Und für Derivate einer aufgrund ihrer - an einen Schraubenschlüssel erinnernden - Form Wrenchnolol genannten Verbindung wurden von einigen Forschergruppen die Transkription und damit die Expression von Genen aktivierende Wirkungen gezeigt, wodurch unter anderem Myogenese von C2C12-Zellen induziert wurde (D. Jung et al., J. Am. Chem. Soc. 131 , 4774-4782 (2009)). And for derivatives of a compound known as wrenchnolol, which is reminiscent of a wrench, some groups of researchers have shown transcription and thus the expression of genes activating effects, among other things, inducing myogenesis of C2C12 cells (Jung, D. et al. J. Am. Chem. Soc. 131, 4774-4782 (2009)).
"Wrenchnolol" "Wrenchnolol"
Sowohl Wrenchnolol als auch Adhesamin sind freilich komplexe Verbindunge deren Synthese mit relativ hohem Aufwand verbunden ist. Eine weitere interessante Verbindung ist Neuropathiazol, ein 4-Thiazolamin, von dem berichtet wurde, dass es die Differenzierung multipotenter neuraler Vorläuferzellen des Hippocampus bei Erwachsenen zu Neuronen induziert. Both Wrenchnolol and Adhesamine are certainly complex compounds whose synthesis is associated with relatively high costs. Another interesting compound is neuropathiazole, a 4-thiazolamine that has been reported to induce the differentiation of multipotential neural progenitor cells of the hippocampus into neurons in adults.
Neuropathiazol neuropathiazol
In der WO 2007/064891 A1 werden allgemein neue Verbindungen offenbart, die die Differenzierung von Säugetierzellen zu neuronalen Zellen induzieren sollen. Die Verbindungen entsprechen der allgemeinen Formel In WO 2007/064891 A1, new compounds are generally disclosed which are intended to induce the differentiation of mammalian cells into neuronal cells. The compounds correspond to the general formula
die folglich sowohl 2-, 4- als 5-Thiazolamine darstellen kann. Konkrete Synthesewege, basierend auf der altbekannten Thiazolsynthese nach Hantzsch, werden nur für 2- und 4-Thiazolamine angegeben. Tatsächlich hergestellt wurden ausschließlich 4-Thiazolamine, die auch in Differenzierungstests auf ihre Aktivität untersucht wurden. Am besten schnitt dabei das obige Neuropathiazol ab, wobei von insgesamt fünfzehn getesteten 4-Thiazolaminen vier unwirksam waren und zwei sogar antiproliferativ wirkten. Die Frage, ob 2- oder 5-Thiazolamine ähnliche Wirkung zeigen wür- den, bleibt offen. which can thus represent both 2-, 4- as 5-thiazolamines. Concrete synthesis routes, based on the well-known thiazole synthesis according to Hantzsch, are given only for 2- and 4-thiazolamines. In fact, only 4-thiazolamines were prepared, which were also tested in differentiation tests for their activity. The best of these was the above neuropathiazole, of which four out of a total of fifteen tested 4-thiazolamines were inactive and two were even antiproliferative. The question of whether 2- or 5-thiazolamines would show similar effects remains open.
Daher war es das Ziel der vorliegenden Erfindung, weitere Kleinmoleküle, insbesondere solche auf Thiazolamin-Basis, herzustellen, die in der Lage sind, die Differenzierung von Säugerzellen zu beschleunigen, und die relativ einfach zu synthetisieren sind. OFFENBARUNG DER ERFINDUNG Therefore, the object of the present invention was to produce further small molecules, in particular those based on thiazolamine, which are capable of accelerating the differentiation of mammalian cells and which are relatively easy to synthesize. DISCLOSURE OF THE INVENTION
Dieses Ziel wird durch Bereitstellung einer Gruppe neuer 2-Thiazolamin-Derivate erreicht, die der nachstehenden allgemeinen Formel entsprechen: This object is achieved by providing a group of novel 2-thiazolamine derivatives which conform to the following general formula:
Ri bis R3 unabhängig voneinander aus Wasserstoff, Benzyl, Aryl und Heteroaryl ausgewählt sind oder fehlen, wobei maximal einer der Reste R1 bis R3 Wasserstoff ist, und R4 aus Benzyl, Aryl und Heteroaryl ausgewählt ist, R 1 to R 3 are independently selected or absent from hydrogen, benzyl, aryl and heteroaryl, with at most one of the radicals R 1 to R 3 being hydrogen and R 4 being selected from benzyl, aryl and heteroaryl,
wobei R1 bis R4 unabhängig voneinander gegebenenfalls mit einem oder mehreren Substituenten, ausgewählt aus Halogen (F, Cl, Br, I), Hydroxy, Ci-3- Alkyl, Ci-3-Alkoxy, Carboxy, C1-3-Alkylcarbonyl, d-3-Alkoxycarbonyl, Nitro, Amino, Mono- und Di-C-i-3-alkylamino sowie Cyano, in denen die Alkylgruppen gegebenenfalls halogeniert sind, substituiert sind; wherein R 1 to R 4 are each independently optionally substituted with one or more substituents selected from halogen (F, Cl, Br, I), hydroxy, Ci -3 - alkyl, Ci -3 alkoxy, carboxy, C 1-3 alkylcarbonyl , d-3-alkoxycarbonyl, nitro, amino, mono- and di-Ci-3-alkylamino and cyano, in which the alkyl groups are optionally halogenated substituted;
jeweils für eine Einfachbindung oder eine Doppelbindung steht; n = 0 bis 2 ist; each represents a single bond or a double bond; n = 0 to 2;
wobei für den Fall n = 1 der Rest R4 an Position 4 oder 5 des Thiazol- rings gebunden sein kann und für den Fall n = 2 die Reste R4 an den Positionen 4 und 5 des Thiazolrings gebunden sind. where, in the case n = 1, the radical R 4 can be bonded to position 4 or 5 of the thiazole ring and, in the case n = 2, the radicals R 4 are bonded to the positions 4 and 5 of the thiazole ring.
Die obigen Thiazolamin-Derivate können in nur drei Syntheseschritten erhalten werden, wodurch der Aufwand und die Kosten für die Herstellung gegenüber den Verbindungen nach dem Stand der Technik deutlich gesenkt werden können. Zudem haben sich solche Derivate in einem C2C12-Mäusezellen-Modell als wirksam bei der Förde- rung der Zelldifferenzierung erwiesen. Somit betrifft die Erfindung die Verwendung von Verbindungen der obigen Formel als Zelldifferenzierungsbeschleuniger. The above thiazolamine derivatives can be obtained in only three synthetic steps, whereby the cost and the cost of preparation compared to the compounds of the prior art can be significantly reduced. In addition, such derivatives have been shown to be effective in promoting cell differentiation in a C2C12 mouse cell model. Thus, the invention relates to the use of compounds of the above formula as cell differentiation accelerators.
Unter Aryl und Heteroaryl sind hierin ein- und mehrkernige aromatische Ringsysteme zu verstehen, vorzugsweise solche mit 5 bis 10 Ringatomen, noch bevorzugter aro- matische Ringe mit 5 oder 6 Ringatomen. Die Heteroatome, wovon eines oder meh- rere vorhanden sein können, sind vorzugsweise aus Stickstoff, Schwefel und Sauerstoff ausgewählt, wobei Stickstoff und Schwefel besonders bevorzugt werden und ein einziges Stickstoff-Heteroatom im Heteroaryl-Rest noch bevorzugter ist. Unter den Aryl- und Heteroaryl-Resten wird jedoch insbesondere Phenyl bevorzugt. Aryl and heteroaryl are to be understood here as mononuclear and polynuclear aromatic ring systems, preferably those having 5 to 10 ring atoms, more preferably aromatic rings having 5 or 6 ring atoms. The heteroatoms, of which one or more Others may be present, are preferably selected from nitrogen, sulfur and oxygen, with nitrogen and sulfur being particularly preferred and a single nitrogen heteroatom in the heteroaryl radical is more preferred. However, among the aryl and heteroaryl radicals, in particular phenyl is preferred.
Vorzugsweise ist eine in der Formel strichliert dargestellte optionale Doppelbindung vorhanden, und zwar entweder endo- oder exocyclisch, d.h. von C2 entweder zum Thiazol- oder zum Aminstickstoff verlaufend. Weiters sind Ri bis R3 vorzugsweise unabhängig voneinander aus Benzyl und Phenyl ausgewählt, und an den beiden Stickstoffatomen sind vorzugsweise nur insgesamt zwei Substituenten vorhanden. Diese beiden Substituenten sind in besonders bevorzugten Ausführungsformen je eine Phenyl- und eine Benzylgruppe. Vorzugsweise weisen die erfindungsgemäßen Verbindungen nur einen Rest R4 auf, d.h. vorzugsweise gilt: n = 1. Dieser Rest ist besonders bevorzugt an C5 des Thiazol- rings gebunden und ist vorzugsweise aus Aryl und Heteroaryl, noch bevorzugter aus Phenyl, Thiophenyl und Pyridinyl, ausgewählt, wobei Phenyl besonders bevorzugt ist. Preferably, an optional double bond shown in broken line in the formula is present, either endo or exocyclic, ie extending from C2 to either the thiazole or amine nitrogen. Furthermore, R 1 to R 3 are preferably selected independently of one another from benzyl and phenyl, and on the two nitrogen atoms preferably only a total of two substituents are present. These two substituents are in particularly preferred embodiments each a phenyl and a benzyl group. The compounds according to the invention preferably have only one radical R 4 , ie preferably: n = 1. This radical is particularly preferably bonded to C 5 of the thiazole ring and is preferably selected from aryl and heteroaryl, more preferably from phenyl, thiophenyl and pyridinyl wherein phenyl is particularly preferred.
Die optionalen Substituenten von Ri bis R4 sind vorzugsweise aus Fluor, Methyl, Tri- fluormethyl, Nitro, Carboxy und Ci-3-Alkoxycarbonyl, noch bevorzugter aus Fluor, Methyl und Nitro, ausgewählt. Ohne sich auf eine bestimmte Theorie festlegen zu wollen, nehmen die Erfinder des vorliegenden Anmeldungsgegenstands an, dass bei Vorliegen größerer oder gar sperriger Substituenten die Wechselwirkung der erfindungsgemäßen Thiazolamin- Derivate mit Translationsfaktoren oder anderen, zum Induzieren von Zelldifferenzierung wesentlichen Komponenten der Zellen behindert wird, wodurch die Wirkung der Erfindung beeinträchtigt werden würde. Weiters wird angenommen, dass stärker elektronenanziehende Substituenten zu bevorzugen sind, da sie offenbar eine aktivierende Wirkung ausüben. Bevorzugte Thiazolamin-Derivate der Erfindung sind die folgenden, da bei diesenThe optional substituents from R 1 to R 4 are preferably selected from fluoro, methyl, trifluoromethyl, nitro, carboxy and C 1-3 -alkoxycarbonyl, more preferably from fluoro, methyl and nitro. Without wishing to be bound by any particular theory, the inventors of the present application believe that, in the presence of larger or even bulky substituents, the interaction of the thiazolamine derivatives of the invention with translation factors or other components of the cells essential to induce cell differentiation is hindered the effect of the invention would be impaired. Furthermore, it is believed that more electron attractive substituents are to be preferred because they appear to exert an activating effect. Preferred thiazolamine derivatives of the invention are as follows
Verbindungen signifikante Zelldifferenzierungsbeschleunigung ohne übermäßige Toxizität beobachtet wurde: Compounds significant cell differentiation acceleration was observed without excessive toxicity:
N-Benzyl-N,5-diphenylthiazolamin 23, N-benzyl-N, 5-diphenylthiazolamine 23,
N-Benzyl-N-phenyl-5-(2-fluorpyridin-3-yl)thiazolamin 24, N-benzyl-N-phenyl-5- (2-fluoropyridin-3-yl) -thiazolamine 24,
4-(2-(N-Benzyl-N-phenylamino)thiazol-5-yl)benzoesäureethylester 25, Ethyl 4- (2- (N-benzyl-N-phenylamino) thiazol-5-yl) benzoate 25,
N-Benzyl-N-phenyl-5-(m-nitrophenyl)thiazolamin 28, N-benzyl-N-phenyl-5- (m-nitrophenyl) -thiazolamine 28,
N-Benzyl-N-phenyl-5-(p-fluorphenyl)thiazolamin 31a. N-Benzyl-N-phenyl-5- (p -fluorophenyl) -thiazolamine 31a.
N-Benzyl-N-phenyl-4,5-bis(p-fluorphenyl)thiazolamin 31 b, N-benzyl-N-phenyl-4,5-bis (p-fluorophenyl) -thiazolamine 31b,
N-Benzyl-N-phenyl-5-(thiophen-2-yl)thiazolamin 32, N-benzyl-N-phenyl-5- (thiophen-2-yl) -thiazolamine 32,
N-(3-Benzyl-5-phenylthiazol-2(3H)-yliden)anilin 34. N- (3-Benzyl-5-phenylthiazol-2 (3H) -ylidene) aniline 34.
N-(3-Benzyl-5-(2-fluorpyridin-3-yl)thiazol-2(3H)-yliden)anilin 35, N- (3-benzyl-5- (2-fluoropyridin-3-yl) thiazole-2 (3H) -ylidene) aniline 35,
N-(3-Benzyl-5-(2-fluorpyridin-4-yl)thiazol-2(3H)-yliden)anilin 36, N- (3-Benzyl-5- (2-fluoropyridin-4-yl) thiazole-2 (3H) -ylidene) aniline 36,
4-(3-Benzyl-2-(phenylimino)-2,3-dihydrothiazol-5-yl)benzoesäureethylester 37, N-(3-Benzyl-5-o-tolylthiazol-2(3H)-yliden)anilin 38, Ethyl 4- (3-benzyl-2- (phenylimino) -2,3-dihydrothiazol-5-yl) benzoate 37, N- (3-benzyl-5-o-tolylthiazol-2 (3H) -ylidene) aniline 38,
N-(3-Benzyl-5-p-tolylthiazol-2(3H)-yliden)anilin 39, N- (3-benzyl-5-p-tolylthiazol-2 (3H) -ylidene) aniline 39,
N-(3-Benzyl-5-(4-nitrophenyl)thiazol-2(3H)-yliden)anilin 41, N- (3-Benzyl-5- (4-nitrophenyl) thiazole-2 (3H) -ylidene) aniline 41,
N-(3-Benzyl-5-(4-methoxyphenyl)thiazol-2(3H)-yliden)anilin 42, N- (3-Benzyl-5- (4-methoxyphenyl) thiazole-2 (3H) -ylidene) aniline 42,
N-(3-Benzyl-5-(4-fluorphenyl)thiazol-2(3H)-yliden)anilin 43 und N- (3-Benzyl-5- (4-fluorophenyl) thiazole-2 (3H) -ylidene) aniline 43 and
N-(3-Benzyl-5-(2-fluorpyridin-3-yl)thiazol-2(3H)-yliden)anilin-4-carbonsäureethylesterN- (3-benzyl-5- (2-fluoropyridin-3-yl) thiazol-2 (3H) -ylidene) aniline-4-carboxylic acid ethyl ester
46. 46th
Darunter werden aufgrund völliger Nicht-Toxizität folgende Verbindungen noch mehr bevorzugt: Among them, due to total non-toxicity, the following compounds are even more preferred:
N-Benzyl-N,5-diphenylthiazolamin 23, N-benzyl-N, 5-diphenylthiazolamine 23,
N-Benzyl-N-phenyl-5-(m-nitrophenyl)thiazolamin 28, N-benzyl-N-phenyl-5- (m-nitrophenyl) -thiazolamine 28,
N-(3-Benzyl-5-phenylthiazol-2(3H)-yliden)anilin 34. N- (3-Benzyl-5-phenylthiazol-2 (3H) -ylidene) aniline 34.
N-(3-Benzyl-5-p-tolylthiazol-2(3H)-yliden)anilin 39, N- (3-benzyl-5-p-tolylthiazol-2 (3H) -ylidene) aniline 39,
N-(3-Benzyl-5-(4-methoxyphenyl)thiazol-2(3H)-yliden)anilin 42 und N- (3-Benzyl-5- (4-methoxyphenyl) thiazole-2 (3H) -ylidene) aniline 42 and
N-(3-Benzyl-5-(4-fluorphenyl)thiazol-2(3H)-yliden)anilin 43, N- (3-Benzyl-5- (4-fluorophenyl) thiazole-2 (3H) -ylidene) aniline 43,
wovon die folgenden drei Verbindungen die besten Ergebnisse im Hinblick auf dieof which the following three compounds give the best results in terms of
Beschleunigung der Zelldiffrenzierung geliefert haben: N-Benzyl-N-phenyl-5-(m-nitrophenyl)thiazolamin 28, Acceleration of cell divergence have delivered: N-benzyl-N-phenyl-5- (m-nitrophenyl) -thiazolamine 28,
N-(3-Benzyl-5-p-tolylthiazol-2(3H)-yliden)anilin 39 und N- (3-Benzyl-5-p-tolylthiazol-2 (3H) -ylidene) aniline 39 and
N-(3-Benzyl-5-(4-fluorphenyl)thiazol-2(3H)-yliden)anilin 43. Nachstehend wird die vorliegende Erfindung anhand von Beispielen und Vergleichsbeispielen näher erläutert, die lediglich bevorzugte Ausführungsformen illustrieren und die Erfindung keineswegs einschränken sollen. N- (3-Benzyl-5- (4-fluorophenyl) thiazole-2 (3H) -ylidene) aniline 43. Hereinbelow, the present invention will be further illustrated by Examples and Comparative Examples, which are merely illustrative of preferred embodiments and are not intended to limit the invention in any way ,
BEISPIELE EXAMPLES
A) Synthesen Allgemeines A) Synthesis General
Die Schmelzpunkte wurden auf einem Kofler-Heiztischmikroskop bestimmt und sind unkorrigiert. HR-MS-Analysen wurden am Institut für Chemische Technologien und Analytik an der Technischen Universität Wien durchgeführt. Bei Aufnahme von MS- und MS/MS-Spektren wurden alle Proben mittels LC-IT-TOF-MS, Detektionsmodus: positive Ionen, analysiert. Zur nachfolgenden Auswertung wurden ausschließlich positive lonisierungsspektren herangezogen (wobei das Quasi-Molekülion das [M+H]+- ion ist). Weitere Daten oder Beobachtungen wurden nicht berücksichtigt. GC-MS- Messungen erfolgten auf einem Thermo Finnigan Focus GC / DSQ II unter Verwendung einer Standard-Kapillarsäure BGB 5 (30 m x 0,32 mm ID) und unter Anwendung eines der folgenden standardisierten Temperaturprofile: STD 100-280 °C Kurzverfahren: 2 Minuten bei 100 °C, mit 18 °C/min auf 280°C, 5 Minuten bei 280 °C; oder STD 100-280 °C Langverfahren: 2 Minuten bei 100 °C, mit 6 °C/min auf 150 °C, 3 Minuten bei 150 °C, mit 10 °C/min auf 280 °C, 10 Minuten bei 280 °C. Dünnschichtchromatographie erfolgte mit Kieselgelplatten 60 F245 von Merck. Säulenchromatographie wurde unter Verwendung von Merck-Kieselgel 60 durchgeführt. NMR-Spek- tren wurden auf einem BRUKER AC 200 FT-NMR-Spektrometer mit TMS (Tetra- methylsilan) als internem Standard aufgenommen. Lösungsmittel und Reagenzien wurden aus kommerziellen Quellen bezogen. Die Lösungsmittel wurden vor Gebrauch gegebenenfalls destilliert und getrocknet. Svnthesebeispiele 1-9 - Herstellung der Thiazolamine Melting points were determined on a Kofler hot stage microscope and are uncorrected. HR-MS analyzes were performed at the Institute for Chemical Technologies and Analytics at the Vienna University of Technology. When MS and MS / MS spectra were recorded, all samples were analyzed by LC-IT-TOF-MS, detection mode: positive ions. For the subsequent evaluation, only positive ionization spectra were used (where the quasi-molecular ion is the [M + H] + ion). Further data or observations were not considered. GC-MS measurements were taken on a Thermo Finnigan Focus GC / DSQ II using a standard capillary acid BGB 5 (30 mx 0.32 mm ID) and using one of the following standardized temperature profiles: STD 100-280 ° C Short method: 2 Minutes at 100 ° C, at 18 ° C / min to 280 ° C, 5 minutes at 280 ° C; or STD 100-280 ° C Long Procedure: 2 minutes at 100 ° C, at 6 ° C / min to 150 ° C, 3 minutes at 150 ° C, at 10 ° C / min to 280 ° C, 10 minutes at 280 ° C. Thin-layer chromatography was carried out with silica gel plates 60 F245 from Merck. Column chromatography was performed using Merck Kieselgel 60. NMR spectra were recorded on a BRUKER AC 200 FT NMR spectrometer with TMS (tetramethylsilane) as the internal standard. Solvents and reagents were obtained from commercial sources. The solvents were optionally distilled and dried before use. Synthetic Examples 1-9 - Preparation of Thiazolamines
Allgemeines Verfahren A General Procedure A
1 ,0 Äquivalente des entsprechenden Halogenthiazols, 1 ,5 Äquivalente des entsprechenden Amins und 0,5 Äquivalente p-Toluolsulfonsäure (p-TsOH) wurden in Isopro- panol (i-PrOH) gelöst, und das Reaktionsgemisch wurde bei 80 °C gerührt, bis vollständiger Umsatz zu beobachten war. Das Reaktionsgemisch wurde dann mit Ethyl- acetat (EtOAc) verdünnt und mit gesättigter wässriger NaHC03-Lösung und Kochsalzlösung gewaschen. Die Reinigung erfolgte mittels Kugelrohrdestillation. Synthesebeispiel 1 1.0 equivalents of the corresponding halothiazole, 1.5 equivalents of the corresponding amine and 0.5 equivalents of p-toluenesulfonic acid (p-TsOH) were dissolved in isopropanol (i-PrOH) and the reaction mixture was stirred at 80 ° C., until full turnover was observed. The reaction mixture was then diluted with ethyl acetate (EtOAc) and washed with saturated aqueous NaHCO 3 solution and brine. The purification was carried out by Kugelrohr distillation. Synthetic Example 1
Herstellung von N-Phenyl-2-thiazolamin 3 Preparation of N-phenyl-2-thiazolamine 3
1 2 3 1 2 3
Gemäß dem obigen allgemeinen Verfahren A wurden 41 mg (0,25 mmol) 2-Brom- thiazol 1 in Gegenwart von p-TsOH mit Anilin 2 umgesetzt. Nach 3 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 3 (40 mg, 90 % d.Th.) erhalten. According to general procedure A above, 41 mg (0.25 mmol) of 2-bromothiazole 1 were reacted with aniline 2 in the presence of p-TsOH. After stirring for 3 days and Kugelrohr distillation, the title compound 3 (40 mg, 90% of theory) was obtained.
Fp.: 128-129 °C (Lit.: 128-129°C) Mp: 128-129 ° C (Lit .: 128-129 ° C)
Rf (PE/EtOAc 4:1): 0,51 R f (PE / EtOAc 4: 1): 0.51
1H-NMR (200 MHz, CDCI3) δ: 6,63 (d, J=3,6Hz, 1 H, H5), 7,02-7,11 (m, J=4,3Hz, 1 H, H4'), 7,30 (d, J=3,6Hz, 1 H, H4), 7,35-7,37 (m, 4H, PhH'). 1 H-NMR (200 MHz, CDCl 3 ) δ: 6.63 (d, J = 3.6Hz, 1H, H5), 7.02-7.11 (m, J = 4.3Hz, 1H, H4 '), 7.30 (d, J = 3.6Hz, 1H, H4), 7.35-7.37 (m, 4H, PhH').
13C-NMR (50 MHz, CDCI3) δ: 106,5 (d), 118,0 (d, C2', C6'), 122,5 (d), 129,3 (d, C3\ C5'), 138,0 (d), 141 ,1 (s), 166,6 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 106.5 (d), 118.0 (d, C2 ', C6'), 122.5 (d), 129.3 (d, C3 \ C5 ' ), 138.0 (d), 141, 1 (s), 166.6 (s).
MS m/z (Cl, NH3): 175 (100, M+), 150 (16), 104 (19), 77 (27), 58 (18). MS m / z (CI, NH 3 ): 175 (100, M + ), 150 (16), 104 (19), 77 (27), 58 (18).
Svnthesebeispiel 2 Synthetic Example 2
Herstellung von N-(4-Morpholinophenyl)-2-thiazolamin 9 Preparation of N- (4-morpholinophenyl) -2-thiazolamine 9
Gemäß dem obigen allgemeinen Verfahren A wurden 48 mg (0,40 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 4-Morpholinoanilin 5 umgesetzt. Nach 13 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 9 (90 mg, 87 % d.Th.) als dunkelbrauner Feststoff erhalten. According to general procedure A above, 48 mg (0.40 mmol) of 2-chlorothiazole 4 were reacted with 4-morpholinoaniline 5 in the presence of p-TsOH. After stirring for 13 days and Kugelrohr distillation, the title compound 9 (90 mg, 87% of theory) was obtained as a dark brown solid.
Fp.: 200-201 °C Mp .: 200-201 ° C
Rf (PE/EtOAc 2:1): 0,19 R f (PE / EtOAc 2: 1): 0.19
1H-NMR (200 MHz, CDCI3) δ: 2,99-3,03 (m, 4H, -CH2), 3,70-3,75 (m, 4H, -CH2), 6,97 (d, J=3,5Hz, 1 H, H5), 6,90 (d, J=8,8Hz, 2H, Η2', H6'), 7,17 (d, J=3,6Hz, 1 H, H4), 7,47 (d, J=8,6Hz, 2H, Η3', H5'), 9,89 (s, 1 H, NH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 2.99-3.03 (m, 4H, -CH 2 ), 3.70-3.75 (m, 4H, -CH 2 ), 6.97 (d, J = 3.5Hz, 1H, H5), 6.90 (d, J = 8.8Hz, 2H, Η2 ', H6'), 7.17 (d, J = 3.6Hz, 1H , H4), 7.47 (d, J = 8.6Hz, 2H, Η3 ', H5'), 9.89 (s, 1H, NH).
13C-NMR (50 MHz, CDCI3) δ: 49,2 (t, -CH2), 66,1 (t, -CH2), 107,4 (d), 116,0 (d, C2', C6'), 118,2 (d, C3\ C5'), 134,1 (s), 138,8 (d), 145,9 (s), 164,7 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 49.2 (t, -CH 2 ), 66.1 (t, -CH 2 ), 107.4 (d), 116.0 (d, C2 ') , C6 '), 118.2 (d, C3 \ C5'), 134.1 (s), 138.8 (d), 145.9 (s), 164.7 (s).
MS m/z (Cl, NH3): 261 (100, M+), 203 (58), 175 (10), 101 (16), 77 (8). MS m / z (Cl, NH 3): 261 (100, M +), 203 (58), 175 (10), 101 (16), 77 (8).
HRMS (Cl, NH3): MH+, gef.: 262,1013; ber. für C13H15N3OS: 262,1009. HRMS (Cl, NH 3): MH +, Found .: 262.1013; for C 13 H 15 N 3 OS: 262, 1009.
Svnthesebeispiel 3 Synthetic Example 3
Herstellung von N-(4-Methoxyphenyl)-2-thiazolamin 10 Preparation of N- (4-methoxyphenyl) -2-thiazolamine 10
4 6 10 4 6 10
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 4-Methoxyanilin 6 umgesetzt. Nach 13 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 10 (100 mg, 97 % d.Th.) als dunkelbrauner Feststoff erhalten. Fp.: 126-127 °C (Lit.: 126-127 °C) According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 4-methoxyaniline 6 in the presence of p-TsOH. After stirring for 13 days and Kugelrohr distillation, the title compound 10 (100 mg, 97% of theory) was obtained as a dark brown solid. Mp: 126-127 ° C (Lit .: 126-127 ° C)
Rf (PE/EtOAc 2: 1 ): 0,65 H-NMR (200 MHz, CDCI3) δ: 3,81 (s, 3H, -OCH3), 6,55 (d, J=3,6Hz, 1 H, H5), 6,91 (m, 2H, Η2', Η6'), 7,22 (d, J=3,6Hz, 1 H, H4), 7,23-7,31 (m, 2H, H3*, H5'). 13C-NMR (50 MHz, DMSO-d6) δ: 55,0 (q, -OCH3), 107,4 (d), 1 14, 1 (d, C2', C6'), 1 18,6 (d, C3\ C5'), 134,8 (d), 138,7 (s), 153,9 (s), 164, 1 (s). R f (PE / EtOAc 2: 1): 0.65 H-NMR (200 MHz, CDCl 3 ) δ: 3.81 (s, 3H, -OCH 3 ), 6.55 (d, J = 3.6Hz , 1 H, H5), 6.91 (m, 2H, Η2 ', Η6'), 7.22 (d, J = 3.6Hz, 1H, H4), 7.23-7.31 (m, 2H, H3 *, H5 '). 13 C-NMR (50 MHz, DMSO-d 6 ) δ: 55.0 (q, -OCH 3 ), 107.4 (d), 1 14, 1 (d, C2 ', C6'), 1 18, 6 (d, C3 \ C5 '), 134.8 (d), 138.7 (s), 153.9 (s), 164, 1 (s).
MS m/z (Cl, NH3): 206 (100, M+), 191 (92), 163 (10), 136 (18), 58 (8). MS m / z (Cl, NH 3): 206 (100, M +), 191 (92), 163 (10), 136 (18), 58 (8).
Synthesebeispiel 4 Synthesis Example 4
Herstellung von N-(3-Methoxyphenyl)-2-thiazolamin 1_1 Preparation of N- (3-methoxyphenyl) -2-thiazolamine 1_1
4 7 1 1 4 7 1 1
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 3-Methoxyanilin 7 umgesetzt. Nach 13 d Rüh- ren und Kugelrohrdestillation, Umkristallisation aus LP und MPLC (LP:EtOAc 3: 1 ) wurde die Titelverbindung JM (49 mg, 46 % d.Th.) als brauner Feststoff erhalten. Fp.: 81-83 °C According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 3-methoxyaniline 7 in the presence of p-TsOH. After 13 days of stirring and Kugelrohr distillation, recrystallization from LP and MPLC (LP: EtOAc 3: 1), the title compound JM (49 mg, 46% of theory) was obtained as a brown solid. Mp: 81-83 ° C
Rf (PE/EtOAc 2:1 ): 0,85 R f (PE / EtOAc 2: 1): 0.85
1H-NMR (200 MHz, CDCI3) δ: 3,80 (s, 3H, -OCH3), 6,52 (d, J=3,6Hz, 1 H, H5), 6,90- 6,92 (m, 2H, PhH'), 7,20 (d, J=3,6Hz, 1 H, H4), 7,21 -7,30 (m, 2H, PhH'). 1 H-NMR (200 MHz, CDCl 3 ) δ: 3.80 (s, 3H, -OCH 3 ), 6.52 (d, J = 3.6Hz, 1H, H5), 6.90-6, 92 (m, 2H, PhH '), 7.20 (d, J = 3.6Hz, 1H, H4), 7.21 -7.30 (m, 2H, PhH').
13C-NMR (50 MHz, DMSO-d6) δ: 54,4 (q, -OCH3), 102,0 (d, ArC), 105,9 (d, ArC), 108,4 (d, ArC), 109,3 (d, ArC), 129,5 (d, ArC), 138,3 (d, ArC), 142,9 (s), 159,5 (s), 163,8 (s). 13 C-NMR (50 MHz, DMSO-d 6 ) δ: 54.4 (q, -OCH 3 ), 102.0 (d, ArC), 105.9 (d, ArC), 108.4 (d, ArC), 109.3 (d, ArC), 129.5 (d, ArC), 138.3 (d, ArC), 142.9 (s), 159.5 (s), 163.8 (s) ,
MS m/z (Cl, NH3): 206 (100), 190 (15), 163 (7), 103 (8). Svnthesebeispiel 5 MS m / z (CI, NH 3 ): 206 (100), 190 (15), 163 (7), 103 (8). Synthetic Example 5
Herstellung von N-(2-Methoxyphenyl)-2-thiazolamin Preparation of N- (2-methoxyphenyl) -2-thiazolamine
4 8 12 4 8 12
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 2-Methoxyanilin 8 umgesetzt. Nach 13 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 12 (88 mg, 85 % d.Th.) als rotbrauner Feststoff erhalten. According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 2-methoxyaniline 8 in the presence of p-TsOH. After stirring for 13 days and kugelrohr distillation, the title compound 12 (88 mg, 85% of theory) was obtained as a reddish brown solid.
Fp.: 126-128 °C Mp: 126-128 ° C
Rf (PE/EtOAc 2:1): 0,85 R f (PE / EtOAc 2: 1): 0.85
1H-NMR (200 MHz, CDCI3) δ: 3,89 (s, 3H, -OCH3), 6,65 (d, J=3,6Hz, 1 H, H5), 6,87- 7,02 (m, 3H, PhH'), 7,31 (d, J=3,6Hz, 1 H, H4), 7,83 (bs, 1 H, NH), 7,95-8,00 (m, 1 H, PhH'). 1 H-NMR (200 MHz, CDCl 3 ) δ: 3.89 (s, 3H, -OCH 3 ), 6.65 (d, J = 3.6Hz, 1H, H5), 6.87-7, 02 (m, 3H, PhH '), 7.31 (d, J = 3.6Hz, 1H, H4), 7.83 (bs, 1H, NH), 7.95-8.00 (m, 1H, PhH ').
MS m/z (Cl, NH3): 206 (52, M+), 175 (100), 147 (12), 108 (18), 78 (8). MS m / z (CI, NH 3 ): 206 (52, M + ), 175 (100), 147 (12), 108 (18), 78 (8).
Svnthesebeispiel 6 Synthetic example 6
Herstellun von 4-(Thiazol-2-ylamino)benzoesäureethylester 13 Preparation of 4- (thiazol-2-ylamino) benzoic acid ethyl ester 13
4 17 13 4 17 13
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol).2-Chlor- thiazol 4 in Gegenwart von p-TsOH-mit 4-Aminobenzoesäureethylester ( enzocain) 17 umgesetzt. Nach 1 ,5 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 13 (120 mg, 97 % d.Th.) als hellbrauner Feststoff erhalten. According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted in the presence of p-TsOH with 4-aminobenzoic acid ethyl ester (enzocaine) 17. After stirring for 1.5 h and Kugelrohr distillation, the title compound 13 (120 mg, 97% of theory) was obtained as a tan solid.
Fp.: 155-156 °C Mp: 155-156 ° C
Rf (PE/EtOAc 2:1): 0,75 1H-NMR (200 MHz, CDCI3) δ: 1 ,40 (t, J=7,1 Hz, 3H, -CH3), 4,37 (q, J=7,1 Hz, 2H, -CH2), 6,76 (d, J=3,6Hz, 1 H, H5), 7,36-7,44 (m, 3H, Η2', Η6', H4), 8,04 (d, J=8,8Hz, 2H, Η3', H5'). R f (PE / EtOAc 2: 1): 0.75 1 H-NMR (200 MHz, CDCl 3 ) δ: 1.40 (t, J = 7.1 Hz, 3H, -CH 3 ), 4.37 (q, J = 7.1 Hz, 2H, -CH 2 ), 6.76 (d, J = 3.6Hz, 1H, H5), 7.36-7.44 (m, 3H, Η2 ', Η6', H4), 8.04 (d, J = 8.8Hz, 2H, Η3 ', H5').
13C-NMR (50 MHz, DMSO-d6) δ: 14,4 (q, -CH3), 60,8 (t, -OCH2), 108,7 (d, C5), 116,0 (d, C2', C6'), 123,8 (s), 131 ,4 (d, C3\ C5'), 138,3 (d), 144,5 (s), 164,3 (s), 166,2 (s, C=0). 13 C-NMR (50 MHz, DMSO-d 6 ) δ: 14.4 (q, -CH 3 ), 60.8 (t, -OCH 2 ), 108.7 (d, C5), 116.0 ( d, C2 ', C6'), 123.8 (s), 131, 4 (d, C3 \ C5 '), 138.3 (d), 144.5 (s), 164.3 (s), 166 , 2 (s, C = 0).
MS m/z (Cl, NH3): 248 (100, M+), 202 (62), 175 (19), 131 (8), 102 (20), 58 (11). MS m / z (Cl, NH 3): 248 (100, M +), 202 (62), 175 (19), 131 (8), 102 (20), 58 (11).
Svnthesebeispiel 7 Synthesis example 7
Herstellun von N-(2-Chlorphenyl)-2-thiazolamin 14 Preparation of N- (2-chlorophenyl) -2-thiazolamine 14
4 18 14 4 18 14
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 2-Chloranilin 18 umgesetzt. Nach 2 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 14 (100 mg, 95 % d.Th.) als farbloser Feststoff erhalten. According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 2-chloroaniline 18 in the presence of p-TsOH. After 2 d stirring and Kugelrohr distillation, the title compound 14 (100 mg, 95% of theory) was obtained as a colorless solid.
Fp.: 87-90 °C (Lit.: 88-90 °C) Mp .: 87-90 ° C (Lit .: 88-90 ° C)
Rf (PE/EtOAc 2:1): 0,85 R f (PE / EtOAc 2: 1): 0.85
1H-NMR (200 MHz, CDCI3) δ: 6,61 (d, J=3,6Hz, 1 H, H5), 7,20-7,26 (m, 6H, PhH', H4). 1H-NMR (200 MHz, CDCl3) δ: 6.61 (d, J = 3.6Hz, 1H, H5), 7.20-7.26 (m, 6H, PhH ', H4).
MS m/z (Cl, NH3): 210 (100), 183 (12), 138 (17), 87 (18). MS m / z (CI, NH 3 ): 210 (100), 183 (12), 138 (17), 87 (18).
Svnthesebeispiel 8 Synthetic Example 8
Herstellung von N-(4-Chlorphenyl)-2-thiazolamin 15 Preparation of N- (4-chlorophenyl) -2-thiazolamine 15
4 19 15 4 19 15
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 4-Chloranilin 19 umgesetzt. Nach 2 d Rühren und Kugelrohrdestillation wurde die Titelverbindung 15 (80 mg, 76 % d.Th.) als farbloser Feststoff erhalten. According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 4-chloroaniline 19 in the presence of p-TsOH. After stirring for 2 h and kugelrohr distillation, the title compound 15 (80 mg, 76% of theory) was obtained as a colorless solid.
Fp.: 166-168 °C (Lit.: 167-168 °C) Mp .: 166-168 ° C (Lit .: 167-168 ° C)
1H-NMR (200 MHz, CDCI3) δ: 6,67 (d, J=3,6Hz, H5), 7,26-7,32 (m, 6H, ArH, H4), 8,14 (s, 1 H, -NH). 1H-NMR (200 MHz, CDCl 3 ) δ: 6.67 (d, J = 3.6Hz, H5), 7.26-7.32 (m, 6H, ArH, H4), 8.14 (s, 1H, -NH).
13C-NMR (50 MHz, DMSO-d6) δ: 108,3 (d, ArC), 118,1 (d, ArC), 123,8 (d, ArC), 127,7 (d, ArC), 136,4 (s), 138,0 (d, ArC), 140,5 (d, ArC), 162,9 (s), 163,5 (s). 13 C-NMR (50 MHz, DMSO-d 6) δ: 108.3 (d, ArC), 118.1 (d, ArC), 123.8 (d, ArC), 127.7 (d, ArC) , 136.4 (s), 138.0 (d, ArC), 140.5 (d, ArC), 162.9 (s), 163.5 (s).
MS m/z (CI, NH3): 210 (32, M+), 175 (100), 131 (11), 87 (12). MS m / z (CI, NH 3): 210 (32, M +), 175 (100), 131 (11), 87 (12).
Svnthesebeispiel 9 Synthetic Example 9
Herstellung von N-(4-Nitrophenyl)-2-thiazolamin 16 Preparation of N- (4-nitrophenyl) -2-thiazolamine 16
4 20 16 4 20 16
Gemäß dem obigen allgemeinen Verfahren A wurden 60 mg (0,50 mmol) 2-Chlor- thiazol 4 in Gegenwart von p-TsOH mit 4-Nitroanilin 20 umgesetzt. Nach 12 h Rühren und Kugelrohrdestillation wurde die Titelverbindung 16 (104 mg, 98 % d.Th.) als hellorangefarbener Feststoff erhalten. According to general procedure A above, 60 mg (0.50 mmol) of 2-chlorothiazole 4 were reacted with 4-nitroaniline 20 in the presence of p-TsOH. After stirring for 12 h and bulb tube distillation, the title compound 16 (104 mg, 98% of theory) was obtained as a pale orange solid.
Fp.: 164-165 °C Mp .: 164-165 ° C
1H-NMR (200 MHz, CDCI3) δ: 6,79 (d, J=3,6Hz, H5), 7,40 (d, J=3,6Hz, H4), 7,50 (t, J=8,2Hz, 1 H, H5'), 7,74 (dd, J=7,0Hz, J=2,3Hz, 1 H, H6'), 7,87 (dd, J=7,1 Hz, J=2,0Hz, 1 H, H4'), 8,31 (s, 1 H, -NH), 8,36-8,38 (m, 1 H, H2'). 3C-NMR (50 MHz, DMSO-d6/Aceton-d6) δ: 110,3 (d, ArC), 111 ,8 (d, ArC), 116,3 (d, ArC), 123,5 (d, ArC), 130,7 (d, ArC), 140,0 (d, ArC), 143,4 (s), 149,8 (s), 164,2 (s). MS m/z (Cl, NH3): 221 (100, M+), 174 (34), 131 (15), 90 (6), 58 (10). 1 H NMR (200 MHz, CDCl 3 ) δ: 6.79 (d, J = 3.6Hz, H5), 7.40 (d, J = 3.6Hz, H4), 7.50 (t, J = 8.2Hz, 1H, H5 '), 7.74 (dd, J = 7.0Hz, J = 2.3Hz, 1H, H6'), 7.87 (dd, J = 7.1Hz, J = 2.0Hz, 1H, H4 '), 8.31 (s, 1H, -NH), 8.36-8.38 (m, 1H, H2'). 3 C-NMR (50 MHz, DMSO-d 6 / acetone-d 6) δ: 110.3 (d, ArC), 111, 8 (d, ArC), 116.3 (d, ArC), 123.5 (d, ArC), 130.7 (d, ArC), 140.0 (d, ArC), 143.4 (s), 149.8 (s), 164.2 (s). MS m / z (Cl, NH 3): 221 (100, M +), 174 (34), 131 (15), 90 (6), 58 (10).
HRMS (Cl, NH3): MH+, gef.: 222,0332; ber. für C9H7N302S: 222,0338. HRMS (Cl, NH 3): MH +, Found .: 222.0332; calcd for C 9 H 7 N 3 O 2 S: 222.0338.
Svnthesebeispiele 10 und 11 - Einführung von N-Benzylgruppen Synthesebeispiel 10 Synthetic Examples 10 and 11 - Introduction of N-Benzyl Groups Synthesis Example 10
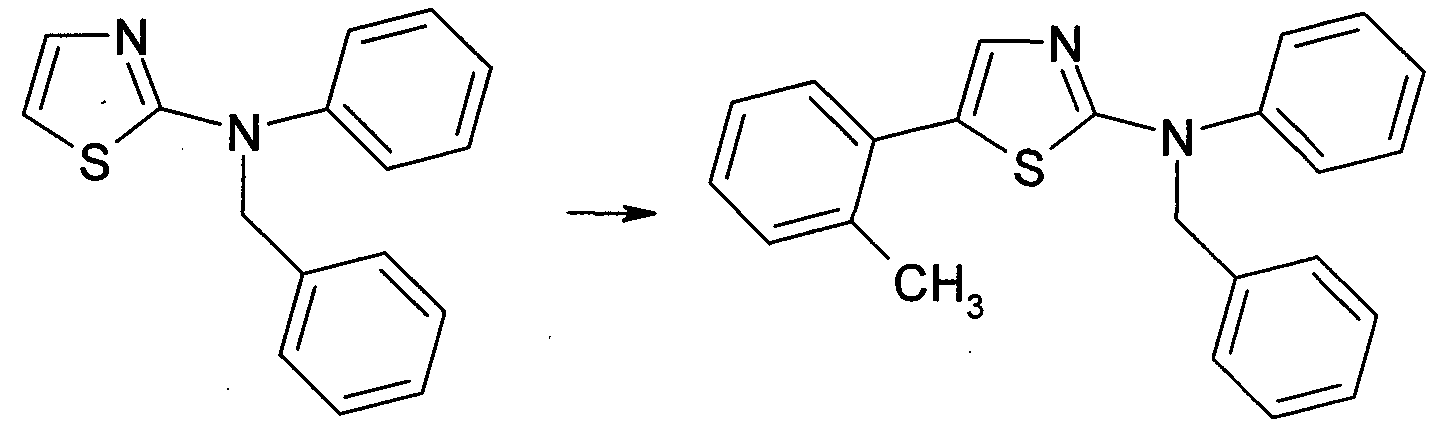
Herstellung von N-Benzyl-N-phenyl-2-thiazolamin 21 Preparation of N-benzyl-N-phenyl-2-thiazolamine 21
3 21 3 21
N-Phenyl-2-thiazolamin 3 (30 mg, 0,17 mmol, 1 ,0 Äquiv.), Benzylbromid (38 mg, 0,22 mmol, 1 ,3 Äquiv.) und NaH (4,1 mg, 0,17 mmol, 1 ,0 Äquiv.) wurden in trockenem DMF (2 ml) gelöst und bei Raumtemperatur 5 h lang gerührt. Anschließend wurde das Reaktionsgemisch mit EtOAc verdünnt, am Rotationsverdampfer über Kieselgel eingeengt und mittels MPLC gereinigt (LP:EtOAc 85:15 + 5 % TEA). Die Titelverbindung 21 wurde als beigefarbener Feststoff erhalten (30 mg, 66 % d.Th.). N-phenyl-2-thiazolamine 3 (30 mg, 0.17 mmol, 1.0 equiv.), Benzylbromide (38 mg, 0.22 mmol, 1.3 equiv.) And NaH (4.1 mg, 0). 17 mmol, 1.0 equiv) were dissolved in dry DMF (2 ml) and stirred at room temperature for 5 h. The reaction mixture was then diluted with EtOAc, concentrated on silica gel on a rotary evaporator and purified by MPLC (LP: EtOAc 85:15 + 5% TEA). The title compound 21 was obtained as a beige solid (30 mg, 66% of theory).
Fp.: 79-80 °C Mp: 79-80 ° C
Rf (PE/EtOAc 5:2): 0,20 R f (PE / EtOAc 5: 2): 0.20
1H-NMR (200 MHz, CDCI3) δ: 5,12 (s, 2H, -CH2), 6,40 (d, J=3,7Hz, H4), 7,15-7,33 (m, 10H, ArH).1H-NMR (200 MHz, CDCl 3 ) δ: 5.12 (s, 2H, -CH 2 ), 6.40 (d, J = 3.7Hz, H 4), 7.15-7.33 (m, 10H, ArH).
3C-NMR (50 MHz, CDCI3) δ: 56,6 (t, -CH2), 107,7 (d), 126,2 (d, ArC), 126,8 (d, ArC),3C-NMR (50 MHz, CDCI 3) δ: 56.6 (t, CH 2), 107.7 (d), 126.2 (d, ArC), 126.8 (d, ArC),
127.2 (d, ArC), 127,7 (d, ArC); 128,5 (d, ArC), 129,8 (d, ArC), 137,7 (s), 139,3 (d),127.2 (d, ArC), 127.7 (d, ArC); 128.5 (d, ArC), 129.8 (d, ArC), 137.7 (s), 139.3 (d),
145.3 (s), 170,9 (s). 145.3 (s), 170.9 (s).
MS m/z (CI, NH3): 266 (56, M+), 174 (100), 167 (30), 131 (11), 91 (16). MS m / z (CI, NH 3 ): 266 (56, M + ), 174 (100), 167 (30), 131 (11), 91 (16).
HRMS (Cl, NH3): MH\ gef.: 267,0955; ber. für Ci6H14N2S: 267,0950. Svnthesebeispiel 11 HRMS (Cl, NH 3): MH \ Found .: 267.0955; Calc. for Ci 6 H 14 N 2 S: 267.0950. Synthetic Example 11
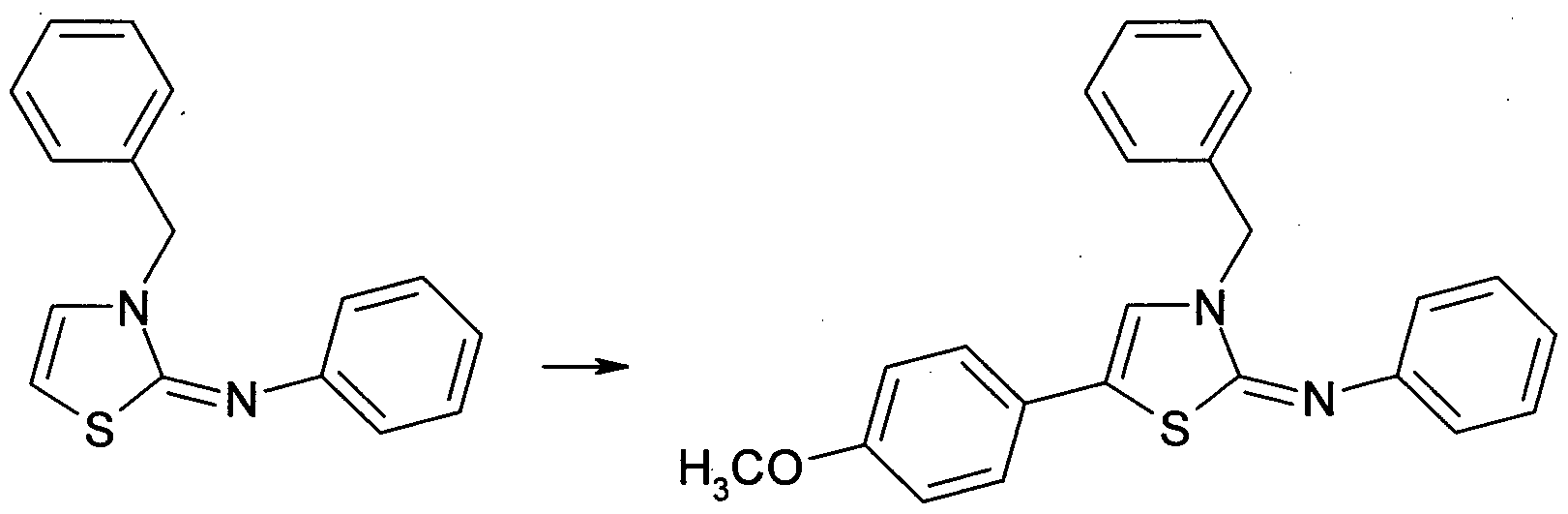
Herstellung von N-(3-Benzylthiazol-2(3H)-yliden)anilin 22 Preparation of N- (3-benzylthiazol-2 (3H) -ylidene) aniline 22
3 22 3 22
N-Phenyl-2-thiazolamin 3 (264 mg, 1 ,50 mmol, 1 ,0 Äquiv.), Benzylbromid (334 mg, 1 ,96 mmol, 1 ,3 Äquiv.) und Triethylamin (152 mg, 1 ,50 mmol, 1 ,0 Äquiv.) wurden bei 0 °C in trockenem Dioxan (4 ml) gelöst, langsam erwärmen gelassen und bei 120 °C auf einem Heizblock 3 d lang gerührt. Anschließend wurde das Reaktionsgemisch mit EtOAc verdünnt, am Rotationsverdampfer über Kieselgel eingeengt und mittels MPLC gereinigt (LP: EtOAc 85:15 + 4 % TEA). Die Titelverbindung 22 wurde als hellgelber Feststoff erhalten (287 mg, 72 % d.Th.). N-phenyl-2-thiazolamine 3 (264 mg, 1.50 mmol, 1.0 equiv.), Benzyl bromide (334 mg, 1.96 mmol, 1.3 equiv.) And triethylamine (152 mg, 1.50 mmol , 1.0 equiv) were dissolved at 0 ° C in dry dioxane (4 ml), allowed to warm slowly and stirred at 120 ° C on a heating block for 3 days. The reaction mixture was then diluted with EtOAc, concentrated on silica gel on a rotary evaporator and purified by MPLC (LP: EtOAc 85:15 + 4% TEA). The title compound 22 was obtained as a pale yellow solid (287 mg, 72% of theory).
Fp.: 94-95 °C Mp .: 94-95 ° C
Rf (PE/EtOAc 5:2): 0,20 R f (PE / EtOAc 5: 2): 0.20
1H-NMR (200 MHz, CDCI3) δ: 5,06 (s, 2H, -CH2), 5,85 (d, J=4,9Hz, 1 H, H5), 6,50 (d, J=4,9Hz, 1 H, H4), 7,00-7,10 (m, 3H, ArH), 7,33-7,37 (m, 7H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.06 (s, 2H, -CH 2 ), 5.85 (d, J = 4.9Hz, 1H, H5), 6.50 (d, J = 4.9Hz, 1H, H4), 7.00-7.10 (m, 3H, ArH), 7.33-7.37 (m, 7H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 49,8 (t, -CH2), 97,6 (d, 1 H, C5), 121 ,4 (d, ArC), 123,2 (d), 126,3 (s), 128,1 (s), 128,6 (d, ArC), 129,4 (d, ArC), 136,8 (s). 13 C-NMR (50 MHz, CDCI 3) δ: 49.8 (t, CH 2), 97.6 (d, 1 H, C5), 121, 4 (d, ArC), 123.2 (d ), 126.3 (s), 128.1 (s), 128.6 (d, ArC), 129.4 (d, ArC), 136.8 (s).
MS m/z (Cl, NH3): 266 (42, M+), 167 (100), 131 (10), 91 (18), 15. (8). MS m / z (Cl, NH 3): 266 (42, M +), 167 (100), 131 (10), 91 (18), 15 (8).
HRMS (Cl, NH3): MH\ gef.: 267,0954; ber. für C 6H14N2S: 267,0950. HRMS (Cl, NH 3): MH \ Found .: 267.0954; calcd for C 6 H 14 N 2 S: 267.0950.
Synthesebeispiel 12 Synthetic Example 12
Herstellung von N-(3-Benzylthiazol-2(3H)-yliden)anilin 33 Preparation of N- (3-benzylthiazol-2 (3H) -ylidene) aniline 33
13 33 13 33
4-(Thiazol-2-ylamino)benzoesäureethylester 13 (120 mg, 0,48 mmol, 1 ,0 Äquiv.), Benzylbromid (106 mg, 0,62 mmol, 1 ,3 Äquiv.) und Triethylamin (48 mg, 0,48 mmol, 1 ,0 Äquiv.) wurden bei 0 °C in trockenem Dioxan (4 ml) gelöst, langsam erwärmen gelassen und bei 120 °C auf einem Heizblock 3 d lang gerührt. Anschließend wurde das Reaktionsgemisch mit EtOAc verdünnt, am Rotationsverdampfer über Kieselgel eingeengt und mittels MPLC gereinigt (LP:EtOAc 85:15 + 4 % TEA). Die Titelverbindung 33 wurde als gelblicher Feststoff erhalten (138 mg, 85 % d.Th.). Ethyl 4- (thiazol-2-ylamino) benzoate 13 (120 mg, 0.48 mmol, 1.0 equiv.), Benzyl bromide (106 mg, 0.62 mmol, 1.3 equiv.) And triethylamine (48 mg, 0 , 48 mmol, 1, 0 equiv.) Were dissolved at 0 ° C in dry dioxane (4 ml), allowed to warm slowly and stirred at 120 ° C on a heating block for 3 d. The reaction mixture was then diluted with EtOAc, concentrated on silica gel on a rotary evaporator and purified by MPLC (LP: EtOAc 85:15 + 4% TEA). The title compound 33 was obtained as a yellowish solid (138 mg, 85% of theory).
Beispiele 1 bis 10 - Arylierungen von Thiazolamin 21 Examples 1 to 10 - Arylations of Thiazolamine 21
Allgemeines Verfahren B General procedure B
1 ,0 Äquivalent des Thiazolamin-Derivats 21., 2,0 Äquivalente des entsprechenden Halogenaryls, 2,0 Äquivalente Kaliumacetat (KOAc) und 0,01 Äquivalente Pd(OAc)2 wurden in dieser Reihenfolge in ein Reaktionsgefäß gefüllt. N,N-Dimethylacetamid (DMAc) wurde zugesetzt, wonach das Reaktionsgefäß evakuiert und dreimal mit Ar- gon gespült wurde. Das Reaktionsgemisch wurde anschließend bei 120 °C gerührt, bis vollständiger Umsatz erzielt war. Danach wurde das Reaktionsgemisch mit EtOAc verdünnt und am Rotationsverdampfer direkt über Kieselgel eingeengt. Die anschließende Reinigung erfolgte mittels MPLC (sofern nichts anderes angegeben ist mit LP:EtOAc 95:5- 0:100 + 3 % Triethylamin). Beispiel 1 1.0 equivalents of thiazolamine derivative 21, 2.0 equivalents of the corresponding haloaryl, 2.0 equivalents of potassium acetate (KOAc) and 0.01 equivalents of Pd (OAc) 2 were charged in this order into a reaction vessel. N, N-dimethylacetamide (DMAc) was added, after which the reaction vessel was evacuated and purged three times with argon. The reaction mixture was then stirred at 120 ° C until complete conversion was achieved. Thereafter, the reaction mixture was diluted with EtOAc and concentrated on silica gel directly on a rotary evaporator. Subsequent purification was by MPLC (unless otherwise indicated by LP: EtOAc 95: 5-0: 100 + 3% triethylamine). example 1
Herstellung von N-Benzyl-N,5-diphenylthiazolamin 23 Preparation of N-benzyl-N, 5-diphenylthiazolamine 23
21 23 21 23
Gemäß dem obigen Allgemeinen Verfahren B wurden 40 mg 2J. mit lodbenzol 48 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 23 als hellbrauner Feststoff erhalten (43 mg, 85 % d.Th.). According to the above General Procedure B, 40 mg of 2J. reacted with iodobenzene for 48 h. Using MPLC, the title compound 23 was obtained as a tan solid (43 mg, 85% of theory).
Fp.: 65-66 °C Mp .: 65-66 ° C
Rf (PE/EtOAc 5:2): 0,45 R f (PE / EtOAc 5: 2): 0.45
1H-NMR (200 MHz, CDCI3) δ: 5,12 (s, 2H, -CH2), 7,06-7,29 (m, 15H, ArH), 7,37 (s, 1H, H4).1 H-NMR (200 MHz, CDCl 3 ) δ: 5.12 (s, 2H, -CH 2 ), 7.06-7.29 (m, 15H, ArH), 7.37 (s, 1H, H4) ,
3C-NMR (50 MHz, CDCI3) δ: 56,7 (t, -CH2), 125,4 (d, ArC), 126,4 (d, ArC), 126,7 (d, ArC), 127,1 (d, ArC), 127,3 (d, ArC), 127,9 (d, ArC), 128,5 (d, ArC), 128,8 (d, ArC), 129,9 (d, ArC), 132,5 (s), 134,7 (d, CI'"), 137,6 (s), 145,0 (s), 169,6 (s). 3C-NMR (50 MHz, CDCI 3) δ: 56.7 (t, CH 2), 125.4 (d, ArC), 126.4 (d, ArC), 126.7 (d, ArC), 127.1 (d, ArC), 127.3 (d, ArC), 127.9 (d, ArC), 128.5 (d, ArC), 128.8 (d, ArC), 129.9 (i.e. , ArC), 132.5 (s), 134.7 (d, Cl '"), 137.6 (s), 145.0 (s), 169.6 (s).
MS m/z (CI, NH3): 343 (16), 342 (62, M+), 252 (20), 251 (100), 250 (79), 219 (19), 134 (20), 91 (73). MS m / z (CI, NH 3 ): 343 (16), 342 (62, M + ), 252 (20), 251 (100), 250 (79), 219 (19), 134 (20), 91 (73).
HRMS (CI, NH3): MH\ gef.: 343,1281 ; ber. für C22H18N2S: 343,1263. HRMS (CI, NH 3 ): MH \ gef .: 343.1281; calcd for C22H18N2S: 343.1263.
Beispiel 2 Example 2
Herstellung von N-Benzyl-N-phenyl-5-(2-fluorpyridin-3-yl)thiazolamin 24 Preparation of N-benzyl-N-phenyl-5- (2-fluoropyridin-3-yl) thiazolamine 24
21 24 21 24
Gemäß dem obigen Allgemeinen Verfahren B wurden 60 mg 2J. mit 2-Fluor-3-iod- pyridin 4 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 24 als beigefarbener Feststoff erhalten (78 mg, 94 % d.Th.). According to General Procedure B above, 60 mg of 2J. reacted with 2-fluoro-3-iodo-pyridine for 4 h. Using MPLC, the title compound 24 was obtained as a beige solid (78 mg, 94% of theory).
Fp.: 117-118 °C Mp .: 117-118 ° C
Rf (PE/EtOAc 10:1): 0,33 R f (PE / EtOAc 10: 1): 0.33
1H-NMR (200 MHz, CDCI3) δ: 5,13 (s, 2H, -CH2), 7,03 (dt, J=5,4Hz, JHF=2,1 HZ, 1 H, H5m), 7,18-7,32 (m, 10H, ArH), 7,59-7,64 (m, 2H, ArH), 7,90 (d, J=3,6Hz, ArH). 1 H NMR (200 MHz, CDCl 3 ) δ: 5.13 (s, 2H, -CH 2 ), 7.03 (dt, J = 5.4Hz, J H F = 2.1HZ, 1H, H5 m ), 7.18-7.32 (m, 10H, ArH), 7.59-7.64 (m, 2H, ArH), 7.90 (d, J = 3.6Hz, ArH).
13C-NMR (50 MHz, CDCI3) δ: 55,9 (t, -CH2), 116,1 (dd, J=29,2Hz, C6,M), 118,8 (ds, J=9,1 Hz, C1,M), 121 ,7 (ds, JCF=4,3HZ, C5), 126,7 (d, ArC), 127,5 (d, ArC), 127,6 (d, ArC), 127,9 (d, ArC), 128,5 (d, ArC), 130,1 (d, ArC), 137,1 (dd, J=4,8Hz, C5'"), 137,2 (s, Ar), 139,8 (dd, JCF=10,0HZ, C4), 144,3 (dd, JCF=14,7HZ, C4'"), 144,6 (s, Ar), 158,6 (ds, JCF=241 HZ, C2,m), 170,9 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 55.9 (t, -CH 2 ), 116.1 (dd, J = 29.2Hz, C6 , M ), 118.8 (ds, J = 9 , 1 Hz, C1 , M ), 121, 7 (ds, J C F = 4,3HZ, C5), 126,7 (d, ArC), 127,5 (d, ArC), 127,6 (d, ArC), 127.9 (d, ArC), 128.5 (d, ArC), 130.1 (d, ArC), 137.1 (dd, J = 4.8Hz, C5 '''), 137.2 (s, Ar), 139.8 (dd, J C F = 10.0HZ, C4), 144.3 (dd, J C F = 14.7HZ, C4 '"), 144.6 (s, Ar) , 158.6 (ds, J C F = 241 HZ, C2 , m ), 170.9 (s).
MS m/z (CI, NH3): 361 (45, M+), 269 (52), 167 (64), 140 (10), 110 (12), 91 (100). HRMS (Cl, NH3): MH+, gef.: 362,1130; ber. für C2iHi6FN3S: 362,1122. MS m / z (CI, NH 3): 361 (45, M +), 269 (52), 167 (64), 140 (10), 110 (12), 91 (100). HRMS (Cl, NH 3): MH +, Found .: 362.1130; calcd for C 2 iHi 6 FN 3 S: 362.1122.
Beispiel 3 Example 3
Herstellung vön 4-(2-(N-Benzyl-N-phenylamino)thiazol-5-yl)benzoesäureethylester 25 Preparation of 4- (2- (N-benzyl-N-phenylamino) thiazol-5-yl) -benzoic acid ethyl ester 25
21 25 21 25
Gemäß dem obigen Allgemeinen Verfahren B wurden 32 mg 2Λ mit 4-lodbenzoesäu- reethylester 24 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 25 als gelbes Öl erhalten (30 mg, 60 % d.Th.). According to General Procedure B above, 32 mg of 2Λ were reacted with 4-iodobenzoic acid ethyl ester for 24 hours. Using MPLC, the title compound 25 was obtained as a yellow oil (30 mg, 60% of theory).
Rf (PE/EtOAc 10: 1 ): 0,48R f (PE / EtOAc 10: 1): 0.48
H-NMR (200 MHz, CDCI3) δ: 1 ,28 (t, J=7,1 Hz, 3H, -CH3), 4,24 (q, J=7, 1 Hz, 2H, -OCH2), 5,10 (s, 2H, -CH2), 7, 14-7,32 (m, 13H, ArH), 7,47 (s, 1 H, H4), 7,84 (d, J=8,4Hz, H3"\ H5,M). 3C-NMR (50 MHz, CDCI3) δ: 14,1 (q, -CH3), 55,8 (t, -CH2), 61 ,2 (t, -OCH2), 124,6 (d, ArC), 126,4 (s), 126,6 (d, ArC), 127,4 (d, ArC), 127,5 (d, ArC), 127,8 (d, ArC), 128,1 (s), 128,5 (d, ArC), 130,0 (d, ArC), 130,2 (d, ArC), 136,8 (d, ArC), 137,4 (s), 144,6 (s), 166,6 (s), 170,4 (s, C=O)* (*ein Singulett fällt mit einem anderen Peak zusammen).H-NMR (200 MHz, CDCl 3 ) δ: 1.28 (t, J = 7.1 Hz, 3H, -CH 3 ), 4.24 (q, J = 7, 1 Hz, 2H, -OCH 2 ), 5.10 (s, 2H, CH 2), 7, 14 to 7.32 (m, 13H, ArH), 7.47 (s, 1 H, H4), 7.84 (d, J = . 8.4 Hz, H3 "\ H5, M) 3C-NMR (50 MHz, CDCI 3) δ: 14.1 (q, -CH 3), 55.8 (t, CH 2), 61, 2 ( t, -OCH 2 ), 124.6 (d, ArC), 126.4 (s), 126.6 (d, ArC), 127.4 (d, ArC), 127.5 (d, ArC), 127.8 (d, ArC), 128.1 (s), 128.5 (d, ArC), 130.0 (d, ArC), 130.2 (d, ArC), 136.8 (d, ArC ), 137.4 (s), 144.6 (s), 166.6 (s), 170.4 (s, C = O) * (* one singlet coincides with another peak).
HRMS (Cl, NH3): MH+, gef.: 415,1478; ber. für C25H22N202S: 414, 1475. HRMS (Cl, NH 3): MH +, Found .: 415.1478; calcd for C 25 H 22 N 2 O 2 S: 414, 1475.
Beispiel 4 - Verqleichsbeispiel Example 4 - Comparative Example
Herstellung von N-Benzyl-N-phenyl-5-(o-tolyl)thiazolam Preparation of N-benzyl-N-phenyl-5- (o-tolyl) thiazolam
21 26 21 26
Gemäß dem obigen Allgemeinen Verfahren B wurden 40 mg 2Λ mit 2-lodtoluol 20 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 26 als beigefarbener Feststoff erhalten (43 mg, 80 % d.Th.). According to General Procedure B above, 40 mg of 2Λ were reacted with 2-iodotoluene for 20 hours. Using MPLC, the title compound 26 was obtained as a beige solid (43 mg, 80% of theory).
Fp.: 71 -73 °C Mp .: 71-73 ° C
Rf (PE/EtOAc 10:1 ): 0,44 R f (PE / EtOAc 10: 1): 0.44
1H-NMR (200 MHz, CDCI3) δ: 2,31 (s, 3H, -CH3), 5,12 (s, 2H, -CH2), 7,1 1 -7,28 (m,1H-NMR (200 MHz, CDCl 3 ) δ: 2.31 (s, 3H, -CH 3 ), 5.12 (s, 2H, -CH 2 ), 7.1 1 -7.28 (m,
15H, ArH). 15H, ArH).
C-NMR (50 MHz, CDCI3) δ: 21 ,4 (q, -CH3), 56,2 (t, -CH2), 126,0 (d, ArC), 126,1 (s), 126,3 (d, ArC), 126,9 (d, ArC), 127,3 (d, ArC), 127,4 (d, ArC), 127,8 (d, ArC), 128,5 (d, ArC), 129,8 (d, ArC), 130,2 (d, ArC), 130,8 (d, ArC), 131 ,4 (s), 136,0 (s), 137,4 (d, ArC), 137,6 (s), 145, 1 (s), 170,1 (s). C-NMR (50 MHz, CDCl 3 ) δ: 21, 4 (q, -CH 3 ), 56.2 (t, -CH 2 ), 126.0 (d, ArC), 126.1 (s), 126.3 (d, ArC), 126.9 (d, ArC), 127.3 (d, ArC), 127.4 (d, ArC), 127.8 (d, ArC), 128.5 (i.e. , ArC), 129.8 (d, ArC), 130.2 (d, ArC), 130.8 (d, ArC), 131, 4 (s), 136.0 (s), 137.4 (i.e. , ArC), 137.6 (s), 145, 1 (s), 170.1 (s).
MS m/z (Cl, NH3): 356 (65, M+), 167 (88), 147 (38), 1 15 (88), 91 (100), 65 (26). MS m / z (Cl, NH 3): 356 (65, M +), 167 (88), 147 (38), 1 15 (88), 91 (100), 65 (26).
HRMS (Cl, NH3): MH+, gef.: 357,1427; ber. für C23H2oN2S: 357, 1420. HRMS (Cl, NH 3): MH +, Found .: 357.1427; calc. for C 23 H2oN 2 S: 357, 1420.
Beispiel 5 - Vergleichsbeispiel Example 5 - Comparative Example
Herstellung von N-Benzyl-N-phenyl-5-(p-tolyl)thiazolamin 27 Preparation of N-benzyl-N-phenyl-5- (p-tolyl) thiazolamine 27
21 27 21 27
Gemäß dem obigen Allgemeinen Verfahren B wurden 75 mg 2± mit 4-lodtoluol 20 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 27 als beigefarbener Feststoff erhalten (77 mg, 77 % d.Th.). According to the above General Procedure B, 75 mg of 2 ± was reacted with 4-iodotoluene for 20 hours. The title compound 27 was obtained as a beige solid by MPLC (77 mg, 77% of theory).
Fp.: 105-108 °C Mp: 105-108 ° C
Rf (PE/EtOAc 10:1): 0,48 R f (PE / EtOAc 10: 1): 0.48
1H-NMR (200 MHz, CDCI3) δ: 2,22 (s, 3H, -CH3), 5,1 1 (s, 2H, -CH2), 6,98-7,02 (m, 3H, ArH), 7, 15-7,26 (m, 12H, ArH), 7,32 (s, H, H4). 1 H-NMR (200 MHz, CDCl 3 ) δ: 2.22 (s, 3H, -CH 3 ), 5.1 1 (s, 2H, -CH 2 ), 6.98-7.02 (m, 3H , ArH), 7, 15-7.26 (m, 12H, ArH), 7.32 (s, H, H4).
13C-NMR (50 MHz, CDCI3) δ: 21 ,2 (q, -CH3), 56,1 (t, -CH2), 125,4 (d, ArC), 126,3 (d, ArC), 127,0 (d, ArC), 127,3 (d, ArC), 127,8 (d, ArC), 127,9 (s), 128,5 (d, ArC), 129,5 (d, ArC), 129,9 (d, ArC), 134,3 (d, ArC), 136,5 (s), 137,6 (s), 145,0 (s), 169,3 (s)* (*ein Singulett fällt mit einem anderen Peak zusammen). 13 C-NMR (50 MHz, CDCl 3 ) δ: 21, 2 (q, -CH 3 ), 56.1 (t, -CH 2 ), 125.4 (d, ArC), 126.3 (d, ArC), 127.0 (d, ArC), 127.3 (d, ArC), 127.8 (d, ArC), 127.9 (s), 128.5 (d, ArC), 129.5 ( d, ArC), 129.9 (d, ArC), 134.3 (d, ArC), 136.5 (s), 137.6 (s), 145.0 (s), 169.3 (s) * (* one singlet coincides with another peak).
MS m/z (Cl, NH3): 356 (48, M+), 265 (100), 233 (10), 167 (44), 105 (10), 91 (58).MS m / z (Cl, NH 3): 356 (48, M +), 265 (100), 233 (10), 167 (44), 105 (10), 91 (58).
HRMS (Cl, NH3): MH\ gef.: 357,1427; ber. für C23H2oN2S: 357,1420. HRMS (Cl, NH 3): MH \ Found .: 357.1427; calcd for C 23 H 2 oN 2 S: 357.1420.
Beispiel 6 Example 6
Herstellung von N-Benzyl-N-phenyl-5-(m-nitrophenyl)thiazolamin 28 Preparation of N-benzyl-N-phenyl-5- (m-nitrophenyl) thiazole amine 28
21 28 21 28
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 2J. mit 3-lodnitrobenzol 20 h lang umgesetzt. Mittels MPLC (LP:EtOAc 75:25^-0:100 + 3 % Triethylamin) wurde die Titelverbindung 28 als hellorangefarbener Feststoff erhalten (41 mg, 51 % d.Th.). According to the above General Procedure B, 30 mg of 2J . reacted with 3-iodonitrobenzene for 20 h. MPLC (LP: EtOAc 75: 25 → -0: 100 + 3% triethylamine) gave the title compound 28 as a light orange solid (41 mg, 51% of theory).
Fp.: 109-110 °C Mp .: 109-110 ° C
Rf (PE/EtOAc 5:2): 0,69R f (PE / EtOAc 5: 2): 0.69
H-NMR (200 MHz, DMSO-d6) δ: 5,23 (s, 2H, -CH2), 7,24-7,35 (m, 6H, ArH), 7,45- 7,50 (m, 3H, ArH), 7,58 (t, J=8,0Hz, 1 H, H5,M), 7,92 (s, 1 H, H4), 8,00 (dd, J=8,0Hz, J=2,0Hz, 1 H, H4m), 8,16 (s, 1 H, H2,M), 8,28 (dt, J=7,3Hz, J=2,1 Hz, 1 H, H4,M), 8,84 (d, J=8,0Hz, 1 H, ArH). H-NMR (200 MHz, DMSO-d 6 ) δ: 5.23 (s, 2H, -CH 2 ), 7.24-7.35 (m, 6H, ArH), 7.45-7.50 ( m, 3H, ArH), 7.58 (t, J = 8.0Hz, 1H, H5 , M ), 7.92 (s, 1H, H4), 8.00 (dd, J = 8.0Hz , J = 2.0 Hz, 1 H, H4 m), 8.16 (s, 1 H, H2, m), 8.28 (dt, J = 7.3 Hz, J = 2.1 Hz, 1 H, H4 , M ), 8.84 (d, J = 8.0Hz, 1H, ArH).
13C-NMR (50 MHz, DMSO-d6) δ: 55,2 (t, -CH2), 118,6 (d, ArC), 120,8 (d, ArC), 123,2 (s), 123,9 (d, ArC), 127,2 (d, ArC), 127,3 (d, ArC), 127,4 (d, ArC), 128,4 (d, ArC), 130,0 (d, ArC), 130,5 (d, ArC), 130,9 (d, ArC), 133,5 (d, ArC), 133,7 (s), 137,3 (d, ArC), 138,1 (s), 144,1 (s), 148,3 (s), 169,7 (s). 1 3 C-NMR (50 MHz, DMSO-d 6 ) δ: 55.2 (t, -CH 2 ), 118.6 (d, ArC), 120.8 (d, ArC), 123.2 (s ), 123.9 (d, ArC), 127.2 (d, ArC), 127.3 (d, ArC), 127.4 (d, ArC), 128.4 (d, ArC), 130.0 (d, ArC), 130.5 (d, ArC), 130.9 (d, ArC), 133.5 (d, ArC), 133.7 (s), 137.3 (d, ArC), 138 , 1 (s), 144.1 (s), 148.3 (s), 169.7 (s).
HRMS (Cl, NH3): MH+, gef.: 388,1118; ber. für C22H17N3O2S: 388,1114. HRMS (Cl, NH 3): MH +, Found .: 388.1118; calcd for C22H17N3O2S: 388.1114.
Beispiel 7 - Verqleichsbeispiel Example 7 - Comparative Example
Herstellung von N-Benzyl-N-phenyl-5-(p-nitrophenyl)thiazolamin 29 Preparation of N-benzyl-N-phenyl-5- (p-nitrophenyl) -thiazolamine 29
21 29 21 29
Gemäß dem obigen Allgemeinen Verfahren B wurden 69 mg 2_1 mit 4-lodnitrobenzol 20 h lang umgesetzt. Mittels MPLC (LP:EtOAc 70:30^-0:100 + 3 % Triethylamin) wurde die Titelverbindung 29 als dunkelorangefarbener Feststoff erhalten (20 mg, 20 % d.Th.). According to General Procedure B above, 69 mg of 2_1 were reacted with 4-iodonitrobenzene for 20 hours. MPLC (LP: EtOAc 70:30 -0: 100 + 3% triethylamine) gave the title compound 29 as a dark orange solid (20 mg, 20% of theory).
Fp.: 197-200 °C Mp: 197-200 ° C
Rf (PE/EtOAc 5:2): 0,66 R f (PE / EtOAc 5: 2): 0.66
1H-NMR (200 MHz, CDCI3) δ: 5,25 (s, 2H, -CH2), 7,30-7,47 (m, 6H, ArH), 7,46-7,48 (m, 4H, ArH), 7,66 (d, J=8,8Hz, 2H, H2,M, H4"'), 8,00 (s, 1 H, H4), 8,13 (d, J=8,7Hz, Η3'", H5'"). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.25 (s, 2H, -CH 2 ), 7.30-7.47 (m, 6H, ArH), 7.46-7.48 (m , 4H, ArH), 7.66 (d, J = 8.8Hz, 2H, H2 , M , H4 "'), 8.00 (s, 1H, H4), 8.13 (d, J = 8 , 7Hz, Η3 '", H5'").
3C-NMR (50 MHz, Aceton-d6) δ: 55,9 (t, -CH2), 124,3 (d, ArC), 124,8 (s), 125,0 (d, ArC), 126,9 (d, ArC), 127,3 (d, ArC), 127,8 (d, ArC), 128,0 (d, ArC), 128,4 (d, ArC), 130,1 (d, ArC), 137,6 (s), 139,3 (s), 139,4 (d, ArC), 144,5 (s), 145,6 (s), 171 ,4 (s).3C-NMR (50 MHz, acetone-d 6 ) δ: 55.9 (t, -CH 2 ), 124.3 (d, ArC), 124.8 (s), 125.0 (d, ArC), 126.9 (d, ArC), 127.3 (d, ArC), 127.8 (d, ArC), 128.0 (d, ArC), 128.4 (d, ArC), 130.1 (i.e. , ArC), 137.6 (s), 139.3 (s), 139.4 (d, ArC), 144.5 (s), 145.6 (s), 171, 4 (s).
HRMS (Cl, NH3): MH+, gef.: 388,1118; ber. für C22H17N302S: 388,1114. HRMS (Cl, NH 3): MH +, Found .: 388.1118; Calcd for C22 H17 N30 2 S:. 388.1114.
Beispiel 8 Example 8
Herstellung von N-Benzyl-N-phenyl-5-(p-methoxyphenyl)thiazolamin 30 Preparation of N-benzyl-N-phenyl-5- (p-methoxyphenyl) thiazolamine 30
21 30 21 30
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 2± mit 4-lodanisol 20 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 30 als beigefarbenes Öl erhalten (21 mg, 47 % d.Th.). According to the above General Procedure B, 30 mg of 2 ± was reacted with 4-iodoanisole for 20 hours. MPLC gave the title compound 30 as a beige oil (21 mg, 47% of theory).
Rf (PE/EtOAc 5:2): 0,66R f (PE / EtOAc 5: 2): 0.66
H-NMR (200 MHz, CDCI3) δ: 3,72 (s, 3H, -OCH3), 5,12 (s, 2H, -CH2), 6,74-6,79 (m, 2H, ArH).H-NMR (200 MHz, CDCl 3 ) δ: 3.72 (s, 3H, -OCH 3 ), 5.12 (s, 2H, -CH 2 ), 6.74-6.79 (m, 2H, Ar H).
3C-NMR (50 MHz, CDCI3) δ: 54,5 (q, -OCH3), 56,4 (t, -CH2), 1 14,3 (d, ArC), 125,1 (s), 126,2 (d, ArC), 126,8 (d, ArC), 126,9 (d, ArC), 127,2 (d, ArC), 127,6 (s), 127,7 (d, ArC), 128,5 (d, ArC), 129,8 (d, ArC), 133,6 (d, ArC), 137,5 (s), 145,1 (s), 158,6 (s), 169,0 (s). 3C-NMR (50 MHz, CDCl 3 ) δ: 54.5 (q, -OCH 3 ), 56.4 (t, -CH 2 ), 1 14.3 (d, ArC), 125.1 (s) , 126.2 (d, ArC), 126.8 (d, ArC), 126.9 (d, ArC), 127.2 (d, ArC), 127.6 (s), 127.7 (d, ArC), 128.5 (d, ArC), 129.8 (d, ArC), 133.6 (d, ArC), 137.5 (s), 145.1 (s), 158.6 (s) , 169.0 (s).
HRMS (Cl, NH3): MH+, gef.: 373,1374; ber. für C23H2oN2OS: 373,1369. HRMS (Cl, NH 3): MH +, Found .: 373.1374; calc. for C 23 H 2 O 2 OS: 373.1369.
Beispiel 9 Example 9
Herstellung von N-Benzyl-N-phenyl-5-(p-fluorphenyl)thiazolamin 31a und Preparation of N-benzyl-N-phenyl-5- (p -fluorophenyl) thiazolamine 31a and
N-Benzyl-N-phenyl-4,5-bis(p-fluorphenyl)thiazolamin 31b N-benzyl-N-phenyl-4,5-bis (p-fluorophenyl) thiazolamine 31b
Gemäß dem obigen Allgemeinen Verfahren B wurden 40 mg 2J. mit 4-lodfluorbenzol 20 h lang umgesetzt. Mittels MPLC wurden die Titelverbindung 31a als brauner Feststoff (39 mg, 71 % d.Th.) und die Titelverbindung 31b als gelber Feststoff erhalten (8 mg, 12 % d.Th.). According to the above General Procedure B, 40 mg of 2J . reacted with 4-iodofluorobenzene for 20 h. MPLC gave the title compound 31a as a brown solid (39 mg, 71% of theory) and the title compound 31b as a yellow solid (8 mg, 12% of theory).
31a: 31a:
Rf (PE/EtOAc 10:1): 0,40 R f (PE / EtOAc 10: 1): 0.40
1H-NMR (200 MHz, CDCI3) δ: 5,05 (s, 2H, -CH2), 6,85-6,95 (m, 2H, ArH), 7,17-7,30 (m, 13H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.05 (s, 2H, -CH 2 ), 6.85-6.95 (m, 2H, ArH), 7.17-7.30 (m , 13H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 55,5 (t, -CH2), 114,6 (dd, JCF=22HZ, C2"\ C6*"), 125,3 (d, ArC), 125,5 (s, Ar), 125,9 (dd, JCF=8HZ, C3m, C6'"), 126,1 (d, ArC), 126,3 (d, ArC), 126,7 (d, ArC), 127,5 (d, ArC), 127,6 (s, Ar), 128,9 (d, ArC), 133,7 (d, ArC), 136,4 (s, Ar), 144,0 (s, Ar), 160,1 (ds, JCF=247Hz, C4,M), 168,5 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 55.5 (t, -CH 2 ), 114.6 (dd, J C F = 22HZ, C2 "\ C6 * "), 125.3 (d, ArC), 125.5 (s, Ar), 125.9 (dd, J C F = 8HZ, C3 m , C6 '''), 126.1 (d, ArC), 126.3 (d, ArC), 126.7 (d, ArC), 127.5 (d, ArC), 127.6 (s, Ar), 128.9 (d, ArC), 133.7 (d, ArC), 136.4 (s , Ar), 144.0 (s, Ar), 160.1 (ds, J CF = 247Hz, C4 , M ), 168.5 (s).
MS m/z (Cl, NH3): 360 (49, M+), 269 (100), 91 (94), 167 (72), 152 (24). MS m / z (CI, NH 3 ): 360 (49, M + ), 269 (100), 91 (94), 167 (72), 152 (24).
HRMS (Cl, NH3): MH\ gef.: 361 ,1171 ; ber. für C22H17FN2S: 362,1169. 31b: HRMS (Cl, NH 3): MH \ Found .: 361, 1171; calcd for C22H17FN2S: 362.1169. 31b:
1H-NMR (200 MHz, CDCI3) δ: 5,24 (s, 2H, -CH2), 6,87-7,02 (m, 4H, ArH), 7,14-7,52 (m, 14H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.24 (s, 2H, -CH 2 ), 6.87-7.02 (m, 4H, ArH), 7.14-7.52 (m , 14H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 55,9 (t, -CH2), 115,1 (d, JCF=29Hz), 115,5 (d, JCF=29Hz), 126,7 (d), 127,2 (d), 127,3 (d), 128,3 (d), 128,4 (d), 129,9 (d, ArC), 130,8 (d, JCF=28Hz), 130,9 (d, JCF=28Hz). 13 C-NMR (50 MHz, CDCl 3 ) δ: 55.9 (t, -CH 2 ), 115.1 (d, J CF = 29Hz), 115.5 (d, J CF = 29Hz), 126, 7 (d), 127.2 (d), 127.3 (d), 128.3 (d), 128.4 (d), 129.9 (d, ArC), 130.8 (d, J CF = 28Hz), 130.9 (d, J CF = 28Hz).
Beispiel 10 Example 10
Herstellung von N-Benzyl-N-phenyl-5-(thiophen-2-yl)thiazolamin 32 Preparation of N-benzyl-N-phenyl-5- (thiophen-2-yl) thiazolamine 32
21 32 21 32
Gemäß dem obigen Allgemeinen Verfahren B wurden 40 mg 21 mit 2-lodthiophen 20 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 32 als braunes Öl erhalten (41 mg, 82 % d.Th.). According to General Procedure B above, 40 mg of 21 were reacted with 2-iodothiophene for 20 hours. Using MPLC, the title compound 32 was obtained as a brown oil (41 mg, 82% of theory).
Rf (PE/EtOAc 10:1): 0,40 R f (PE / EtOAc 10: 1): 0.40
1H-NMR (200 MHz, CDCI3) δ: 5,19 (s, 2H, -CH2), 6,90-6,96 (m, 2H, ArH), 7,10-7,11 (dd, J=4,0Hz, J=1 ,2Hz, 1 H, H3,M), 7,13-7,44 (m, 11 H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.19 (s, 2H, -CH 2 ), 6.90-6.96 (m, 2H, ArH), 7.10-7.11 (dd , J = 4.0Hz, J = 1, 2Hz, 1H, H3 , M ), 7.13-7.44 (m, 11H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 56,2 (t, -CH2), 120,9 (s), 123,4 (d, ArC), 126,4 (d, ArC), 127,3 (d, ArC), 127,5 (d, ArC), 127,8 (d, ArC), 128,5 (d, ArC), 129,9 (d, ArC), 134,6 (s), 135,4 (d, ArC), 137,4 (s), 144,8 (s), 169,5 (s). 13 C-NMR (50 MHz, CDCI 3) δ: 56.2 (t, CH 2), 120.9 (s), 123.4 (d, ArC), 126.4 (d, ArC), 127 , 3 (d, ArC), 127.5 (d, ArC), 127.8 (d, ArC), 128.5 (d, ArC), 129.9 (d, ArC), 134.6 (s) , 135.4 (d, ArC), 137.4 (s), 144.8 (s), 169.5 (s).
MS m/z (Cl, NH3): 348 (89, M+), 257 (100), 207 (48), 167 (40), 225 (34), 91 (10).MS m / z (CI, NH 3 ): 348 (89, M + ), 257 (100), 207 (48), 167 (40), 225 (34), 91 (10).
HRMS (Cl, NH3): MH+, gef.: 349,0832; ber. für C20Hi6N2S2: 349,0828. Beispiele 11 bis 21 - Arylierungen von Thiazolimin 22 HRMS (Cl, NH 3): MH +, Found .: 349.0832; calc. for C 20 Hi 6 N 2 S 2 : 349.0828. Examples 11 to 21 - Arylations of Thiazolimine 22
Für diese Arylierungen wurde im Wesentlichen ebenfalls das obige Allgemeine Verfahren B angewandt. Beispiel 11 For these arylations, the above General Method B was also used essentially. Example 11
Herstellung von N-(3-Benzyl-5-phenylthiazol-2(3H)-yliden)anilin 34 Preparation of N- (3-benzyl-5-phenylthiazol-2 (3H) -ylidene) aniline 34
22 34 22 34
Gemäß dem obigen Allgemeinen Verfahren B wurden 100 mg 22 mit lodbenzol 48 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 34 als hellbrauner Feststoff erhalten (99 mg, 79 % d.Th.). According to the above General Procedure B, 100 mg of 22 was reacted with iodobenzene for 48 hours. Using MPLC, the title compound 34 was obtained as a tan solid (99 mg, 79% of theory).
Fp.: 65-66 °C Mp .: 65-66 ° C
Rf (PE/EtOAc 5:2): 0,52 R f (PE / EtOAc 5: 2): 0.52
1H-NMR (200 MHz, CDCI3) δ: 5,02 (s, 1 H, -CH2), 6,71 (s, 1 H, H4), 6,95-7,29 (m, 15H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.02 (s, 1 H, -CH 2 ), 6.71 (s, 1 H, H 4), 6.95-7.29 (m, 15H , ArH).
13C-NMR (50 MHz, CDCI3) δ: 50,2 (t, -CH2), 115,3 (s), 121 ,7 (d, ArC), 121 ,7 (d), 123,1 (d, ArC), 124,4 (d, ArC), 127,0 (d, ArC), 128,0 (d, ArC), 128,8 (d, ArC), 129,3 (d, ArC), 131 ,2 (s), 136,2 (s), 151 ,1 (s)* (*ein Singulett fällt mit einem anderen Peak zusammen). 13 C-NMR (50 MHz, CDCl 3 ) δ: 50.2 (t, -CH 2 ), 115.3 (s), 121, 7 (d, ArC), 121, 7 (d), 123.1 (d, ArC), 124.4 (d, ArC), 127.0 (d, ArC), 128.0 (d, ArC), 128.8 (d, ArC), 129.3 (d, ArC) , 131, 2 (s), 136,2 (s), 151, 1 (s) * ( * one singlet coincides with another peak).
MS m/z (Cl, NH3): 342 (36, M+), 281 (18), 251 (32), 207 (68), 167 (100), 91 (10). HRMS (Cl, NH3): MH+, gef.: 343,1271 ; ber. für C22Hi8N2S: 343,1263. Beispiel 12 MS m / z (Cl, NH 3): 342 (36, M +), 281 (18), 251 (32), 207 (68), 167 (100), 91 (10). HRMS (Cl, NH 3): MH +, Found .: 343.1271; Calc. for C 2 2Hi 8 N 2 S: 343.1263. Example 12
Herstellung von N-(3-Benzyl-5-(2-fluorpyridin-3-yl)thiazol-2(3H)-yliden)anilin 35 Preparation of N- (3-Benzyl-5- (2-fluoropyridin-3-yl) thiazole-2 (3H) -ylidene) aniline 35
22 35 22 35
Gemäß dem obigen Allgemeinen Verfahren B wurden 70 mg 22 mit 2-Fluor-3-iod- pyridin 20 h lang umgesetzt. Mittels MPLC (LP:EtOAc 80:20-^-0:100 + 3 % Triethyl- amin) wurde die Titelverbindung 35 als hellbrauner Feststoff erhalten (48 mg, 51 % d.Th.). According to General Procedure B above, 70 mg of 22 was reacted with 2-fluoro-3-iodo-pyridine for 20 hours. MPLC (LP: EtOAc 80:20 -> - 0: 100 + 3% triethylamine) gave the title compound 35 as a tan solid (48 mg, 51% of theory).
Fp.: 139-140 °C Mp: 139-140 ° C
Rf (PE/EtOAc 10:2): 0,33 R f (PE / EtOAc 10: 2): 0.33
1H-NMR (200 MHz, CDCI3) δ: 7,06-7,13 (m, 4H, ArH), 7,24 (ds, JHF=1 ,0HZ, 1 H, H4), 7,34-7,49 (m, 8H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 7.06-7.13 (m, 4H, ArH), 7.24 (ds, J H F = 1, 0HZ, 1 H, H4), 7, 34-7.49 (m, 8H, ArH).
13C-NMR (50 MHz, CDCI3): 49,8 (t, -CH2), 106,9 (dd, JCF=8,5HZ, C4), 1 15,6 (dd, JCF=28,0Hz, C6,M), 121 ,5 (d, ArC), 121 ,8 (dd, JCF=4,3HZ, C5'"), 123,4 (d, ArC), 127,8 (dd, JCF=15,9Hz, ArC, C4,M), 127,9 (s, Ar), 128,0 (d, ArC), 128,9 (d, ArC), 129,6 (d, ArC), 136,1 (s, Ar), 136,3 (s, Ar), 137,2 (d, ArC), 143,7 (ds, JCF=15,2HZ, CT"), 151 ,1 (s, Ar), 156,2 (s), 158,7 (ds, J=240Hz, C2m). 13 C-NMR (50 MHz, CDCl 3 ): 49.8 (t, -CH 2 ), 106.9 (dd, J C F = 8.5HZ, C4), 1.15.6 (dd, J CF = 28.0Hz, C6 , M ), 121, 5 (d, ArC), 121, 8 (dd, J C F = 4.3HZ, C5 '"), 123.4 (d, ArC), 127.8 ( dd, J CF = 15.9Hz, ArC, C4 , M ), 127.9 (s, Ar), 128.0 (d, ArC), 128.9 (d, ArC), 129.6 (d, ArC ), 136.1 (s, Ar), 136.3 (s, Ar), 137.2 (d, ArC), 143.7 (ds, J C F = 15.2 Hz, CT "), 151, 1 (s, Ar), 156.2 (s), 158.7 (ds, J = 240Hz, C2 m).
MS m/z (Cl, NH3): 361 (26, M+), 269 (30), 167 (100), 1 10 (8), 91 (75). MS m / z (Cl, NH 3): 361 (26, M +), 269 (30), 167 (100), 1 10 (8), 91 (75).
HRMS (Cl, NH3): MH+, gef.: 362,1 131 ; ber. für C2iH16FN3S: 362,1122. Beispiel 13 HRMS (Cl, NH 3): MH +, 362.1 Found .: 131; calcd for C 2 iH 16 FN 3 S: 362.1122. Example 13
Herstellung von N-(3-Benzyl-5-(2-fluorpyridin-4-yl)thiazol-2(3H)-yliden)anilin 36 Preparation of N- (3-Benzyl-5- (2-fluoropyridin-4-yl) thiazole-2 (3H) -ylidene) aniline 36
22 36 22 36
Gemäß dem obigen Allgemeinen Verfahren B wurden 60 mg 22 mit 2-Fluor-4-iod- pyridin 10 h lang umgesetzt. Mittels MPLC (LP:EtOAc 80:20->0:100 + 3 % Triethyl- amin) wurde die Titelverbindung 36 als hellbrauner Feststoff erhalten (80 mg, 96 % d.Th.). According to General Procedure B above, 60 mg of 22 were reacted with 2-fluoro-4-iodo-pyridine for 10 hours. MPLC (LP: EtOAc 80: 20 → 0: 100 + 3% triethylamine) gave the title compound 36 as a tan solid (80 mg, 96% of theory).
Fp.: 111-112 °C Mp: 111-112 ° C
Rf (PE/EtOAc 10:2): 0,33R f (PE / EtOAc 10: 2): 0.33
H-NMR (200 MHz, CDCI3) δ: 5,17 (s, 2H, -CH2), 6,73 (s, 1 H, H4), 7,06 (td, J= 5,4Hz, JHF=1 ,7HZ, 1 H, H6m), 7,21-7,42 (m, 10H, ArH), 7,67 (s, 1 H, H4), 8,00 (d, J=8,0Hz, 1 H, H5'"). H-NMR (200 MHz, CDCI 3) δ: 5.17 (s, 2H, CH 2), 6.73 (s, 1 H, H4), 7.06 (td, J = 5.4 Hz, J HF = 1, 7Hz, 1 H, H6 m), 7.21 to 7.42 (m, 10H, ArH), 7.67 (s, 1 H, H4), 8.00 (d, J = 8.0Hz , 1 H, H5 '").
13C-NMR (50 MHz, CDCI3) δ: 56,3 (t, -CH2), 104,0 (dd, JCF=39,4Hz, C5,M), 117,0 (dd, JCF=3,7HZ, C6'"), 123,4 (ds, JCF=4,1 HZ, C5), 126,8 (d, ArC), 127,8 (dd, JCF=22HZ, C2'"), 127,9 (d, ArC), 128,6 (d, ArC), 138,2 (d, ArC), 136,8 (s, Ar), 139,4 (d, ArC), 144,3 (s, Ar), 145,2 (ds, JCF=8,9Hz, CT'), 147,8 (dd, JCF=16,1 HZ, C5,m), 164,5 (ds, JCF=250HZ, C3'"), 171 ,6 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 56.3 (t, -CH 2 ), 104.0 (dd, J CF = 39.4Hz, C5 , M ), 117.0 (dd, JC F = 3.7HZ, C6 '"), 123.4 (ds, J C F = 4.1 HZ, C5), 126.8 (d, ArC), 127.8 (dd, J C F = 22HZ, C2 '"), 127.9 (d, ArC), 128.6 (d, ArC), 138.2 (d, ArC), 136.8 (s, Ar), 139.4 (d, ArC), 144 , 3 (s, Ar), 145.2 (ds, J CF = 8.9Hz, CT '), 147.8 (dd, J C F = 16.1 HZ, C5 , m ), 164.5 (ds , J C F = 250HZ, C3 '"), 171, 6 (s).
MS m/z (Cl, NH3): 361 (58, M+), 281 (28), 269 (100), 253 (26), 207 (76), 167 (42), 91 (14). MS m / z (CI, NH 3 ): 361 (58, M + ), 281 (28), 269 (100), 253 (26), 207 (76), 167 (42), 91 (14).
HRMS (Cl, NH3): MH+, gef.: 362,1129; ber. für C2iHi6FN3S: 362,1122. Beispiel 14 HRMS (Cl, NH 3): MH +, Found .: 362.1129; calcd for C 2 iHi 6 FN 3 S: 362.1122. Example 14
Herstellung von 4-(3-Benzyl-2-(phenylimino)-2,3-dihydrothiazol-5-yl)benzoesäure- ethylester 37 Preparation of 4- (3-Benzyl-2- (phenylimino) -2,3-dihydrothiazol-5-yl) benzoic acid ethyl ester 37
22 37 22 37
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 22 mit lodbenzol 24 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 37 als gelbes Öl erhalten (29 mg, 58 % d.Th.). According to the above General Procedure B, 30 mg of 22 was reacted with iodobenzene for 24 hours. Using MPLC, the title compound 37 was obtained as a yellow oil (29 mg, 58% of theory).
Rf (PE/EtOAc 10:1): 0,51 R f (PE / EtOAc 10: 1): 0.51
1H-NMR (200 MHz, CDCI3) δ: 1 ,38 (t, J=7,1 Hz, -CH3), 4,37 (q, J=7,1Hz, 2H, -OCH2), 5,14 (s, 2H, -CH2), 7,00 (s, 1 H, H4), 7,12 (d, J=8,5Hz, 2H, H2"\ H6,M), 7,24-7,28 (m, 3H, ArH), 7,37-7,40 (m, 7H, ArH), 7,94 (d, J=8,5Hz, 2H, H3"\ H5m). 1 H NMR (200 MHz, CDCl 3 ) δ: 1, 38 (t, J = 7.1 Hz, -CH 3 ), 4.37 (q, J = 7.1Hz, 2H, -OCH 2 ), 5 , 14 (s, 2H, -CH 2 ), 7.00 (s, 1 H, H4), 7.12 (d, J = 8.5Hz, 2H, H2 "\ H6 , M ), 7.24- 7.28 (m, 3H, ArH), 7.37-7.40 (m, 7H, ArH), 7.94 (d, J = 8.5 Hz, 2H, H3 "\ H5 m).
13C-NMR (50 MHz, CDCI3) δ: 14,5 (q, CH3), 49,6 (t, -OCH2), 60,3 (t, -CH2), 113,6 (d), 121 ,4 (d, ArC), 123,4 (d, ArC), 123,7 (s), 123,8 (d, ArC), 128,0 (d, ArC), 128,1 (s, Ar), 128,9 (d, ArC), 129,5 (d, ArC), 130,1 (d, ArC), 136,0 (s), 136,4 (s), 151 ,2 (s), 156,9 (s), 166,1 (s, C=0). 13 C-NMR (50 MHz, CDCl 3 ) δ: 14.5 (q, CH 3 ), 49.6 (t, -OCH 2 ), 60.3 (t, -CH 2 ), 113.6 (i.e. ), 121, 4 (d, ArC), 123.4 (d, ArC), 123.7 (s), 123.8 (d, ArC), 128.0 (d, ArC), 128.1 (s , Ar), 128.9 (d, ArC), 129.5 (d, ArC), 130.1 (d, ArC), 136.0 (s), 136.4 (s), 151, 2 (s ), 156.9 (s), 166.1 (s, C = 0).
HRMS (Cl, NH3): MH\ gef.: 415,1475; ber. für C25H22N202S: 415,1485. HRMS (Cl, NH 3): MH \ Found .: 415.1475; calcd for C 25 H 22 N 2 O 2 S: 415.1485.
Beispiel 15 Example 15
Herstellun von N-(3-Benzyl-5-(o-tolyl)thiazol-2(3H)-yliden)anilin 38 Preparation of N- (3-Benzyl-5- (o-tolyl) thiazole-2 (3H) -ylidene) aniline 38
22 38 22 38
Gemäß dem obigen Allgemeinen Verfahren B wurden 60 mg 22 mit 2-lodtoluol 12 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 38 als braunes Öl erhalten (60 mg, 73 % d.Th.). According to General Procedure B above, 60 mg of 22 were reacted with 2-iodotoluene for 12 hours. Using MPLC, the title compound 38 was obtained as a brown oil (60 mg, 73% of theory).
Rf (PE/EtOAc 4:1): 0,40 R f (PE / EtOAc 4: 1): 0.40
1H-NMR (200 MHz, CDCI3) δ: 2,25 (s, 3H, -CH3), 5,04 (s, 2H, -CH2), 6,42 (s, 1 H, H4), 6,93-7,30 (m, 15H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 2.25 (s, 3H, -CH 3 ), 5.04 (s, 2H, -CH 2 ), 6.42 (s, 1 H, H 4) , 6.93-7.30 (m, 15H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 21 ,1 (q, -CH3), 49,7 (t, -CH2), 115,2 (s), 121 ,1 (d, ArC), 121 ,5 (d, ArC), 123,1 (d, ArC), 124,4 (d, ArC), 127,9 (d, ArC), 128,0 (d, ArC), 128,8 (d, ArC), 128,9 (s), 129,4 (d, ArC), 136,7 (s), 136,9 (s), 151 ,5 (s), 157,5 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 21, 1 (q, -CH 3 ), 49.7 (t, -CH 2 ), 115.2 (s), 121, 1 (d, ArC) , 121, 5 (d, ArC), 123.1 (d, ArC), 124.4 (d, ArC), 127.9 (d, ArC), 128.0 (d, ArC), 128.8 ( d, ArC), 128.9 (s), 129.4 (d, ArC), 136.7 (s), 136.9 (s), 151, 5 (s), 157.5 (s).
MS m/z (Cl, NH3): 356 (50, M+), 281 (6), 265 (26), 207 (21), 115 (20), 167 (100). HRMS (Cl, NH3): MH\ gef.: 357,1428; ber. für C23H2oN2S: 357,1420. MS m / z (Cl, NH 3): 356 (50, M +), 281 (6), 265 (26), 207 (21), 115 (20), 167 (100). HRMS (Cl, NH 3): MH \ Found .: 357.1428; calc. for C 23 H2oN 2 S: 357.1420.
Beispiel 16 Example 16
Herstellung von N-(3-Benzyl-5-(p-tolyl)thiazol-2(3H)-yliden)anilin 39 Preparation of N- (3-Benzyl-5- (p-tolyl) thiazole-2 (3H) -ylidene) aniline 39
22 39 22 39
Gemäß dem obigen Allgemeinen Verfahren B wurden 60 mg 22 mit 4-lodtoluol 18 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 38 als brauner Feststoff erhalten (56 mg, 68 % d.Th.). According to General Method B above, 60 mg of 22 was reacted with 4-iodotoluene for 18 hours. Using MPLC, the title compound 38 was obtained as a brown solid (56 mg, 68% of theory).
Fp.: 59-60 °C Mp .: 59-60 ° C
Rf (PE/EtOAc 4:1): 0,41R f (PE / EtOAc 4: 1): 0.41
H-NMR (200 MHz, CDCI3) δ: 2,49 (s, 3H, -CH3), 5,26 (s, 2H, -CH2), 6,90 (s, 1 H, H4), 7,20-7,29 (m, 6H, ArH), 7,40-7,55 (m, 8H, ArH). H-NMR (200 MHz, CDCI 3) δ: 2.49 (s, 3H, -CH3), 5.26 (s, 2H, CH 2), 6.90 (s, 1 H, H4), 7.20-7.29 (m, 6H, ArH), 7.40-7.55 (m, 8H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 21 ,7 (q, -CH3), 50,3 (t, -CH2), 114,5 (s), 122,0 (d, ArC), 123,5 (d, ArC), 124,7 (d, ArC), 126,6 (d, ArC), 128,2 (d, ArC), 128,3 (d, ArC), 128,6 (d, ArC), 129,3 (d, ArC), 129,9 (d, ArC), 131 ,4 (d, ArC), 131 ,5 (s), 136,4 (s), 137,0 (s), 151 ,8 (s), 158,5 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 21, 7 (q, -CH 3 ), 50.3 (t, -CH 2 ), 114.5 (s), 122.0 (d, ArC) , 123.5 (d, ArC), 124.7 (d, ArC), 126.6 (d, ArC), 128.2 (d, ArC), 128.3 (d, ArC), 128.6 ( d, ArC), 129.3 (d, ArC), 129.9 (d, ArC), 131, 4 (d, ArC), 131, 5 (s), 136.4 (s), 137.0 ( s), 151, 8 (s), 158.5 (s).
MS m/z (Cl, NH3): 356 (90, M+), 281 (12), 264 (100), 233 (16), 207 (26), 167 (20), 91 (6)-MS m / z (Cl, NH 3): 356 (90, M +), 281 (12), 264 (100), 233 (16), 207 (26), 167 (20), 91 (6) -
HRMS (Cl, NH3): MH+, gef.: 357,1426; ber. für C23H2oN2S: 357,1420. HRMS (Cl, NH 3): MH +, Found .: 357.1426; calcd for C 23 H 2 oN 2 S: 357.1420.
Beispiel 17 Example 17
Herstellung von N-(3-Benzyl-5-(3-nitrophenyl)thiazol-2(3H)-yliden)anilin 40 Preparation of N- (3-Benzyl-5- (3-nitrophenyl) thiazole-2 (3H) -ylidene) aniline 40
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 22 mit 3-lodnitrobenzol 3 h lang umgesetzt. Mittels MPLC (LP:EtOAc 80:20^-0:100 + 3 % Triethylamin) wurde die Titelverbindung 40 als orangefarbener Feststoff erhalten (40 mg, 87 % d.Th.).According to General Procedure B above, 30 mg of 22 was reacted with 3-iodonitrobenzene for 3 hours. MPLC (LP: EtOAc 80: 20 → -0: 100 + 3% triethylamine) gave the title compound 40 as an orange solid (40 mg, 87% of theory).
Fp.: 134-146 °C Mp .: 134-146 ° C
Rf (PE/EtOAc 5:2): 0,68 R f (PE / EtOAc 5: 2): 0.68
1H-NMR (200 MHz, DMSO-d6) δ: 6,99-7,04 (m, 3H, ArH, H4), 7,31-7,45 (m, 7H, ArH), 7,49-7,75 (m, 2H, ArH), 8,00 (d, J=8,7Hz, 1 H, ArH), 8,15-8,23 (m, 2H, H3"\ H5'").1H-NMR (200 MHz, DMSO-d 6) δ: 6.99-7.04 (m, 3H, Ar H, H4), 7.31-7.45 (m, 7H, ArH), 7,49- 7.75 (m, 2H, ArH), 8.00 (d, J = 8.7Hz, 1H, ArH), 8.15-8.23 (m, 2H, H3 "\ H5 '").
13C-NMR (50 MHz, DMSO-d6) δ: 49,4 (t, -CH2), 110,5 (s), 117,9 (d, ArC), 120,9 (d, ArC), 123,2 (d, ArC), 127,6 (d, ArC), 127,8 (d, ArC), 128,6 (d, ArC), 129,5 (d, ArC), 130,0 (d, ArC), 130,5 (d, ArC), 133,1 (s), 136,9 (s), 148,1 (s), 150,4 (s), 155,4 (s). HRMS (Cl, NH3): MH+, gef.: 388,1118; ber. für C22Hi7N3O2S: 388,1114. 13 C-NMR (50 MHz, DMSO-d 6 ) δ: 49.4 (t, -CH 2 ), 110.5 (s), 117.9 (d, ArC), 120.9 (d, ArC) , 123.2 (d, ArC), 127.6 (d, ArC), 127.8 (d, ArC), 128.6 (d, ArC), 129.5 (d, ArC), 130.0 ( d, ArC), 130.5 (d, ArC), 133.1 (s), 136.9 (s), 148.1 (s), 150.4 (s), 155.4 (s). HRMS (Cl, NH 3): MH +, Found .: 388.1118; calc. for C 22 Hi7N 3 O 2 S: 388,1114.
Beispiel 18 Example 18
Herstellung von N-(3-Benzyl-5-(4-nitrophenyl)thiazol-2(3H)-yliden)anilin 4J. Preparation of N- (3-Benzyl-5- (4-nitrophenyl) thiazole-2 (3H) -ylidene) aniline 4J .
22 41 22 41
Gemäß dem obigen Allgemeinen Verfahren B wurden 23 mg 22 mit 4-lodnitrobenzol 2 h lang umgesetzt. Mittels MPLC (LP:EtOAc 80:20^-0:100 + 3 % Triethylamin) wurde die Titelverbindung 4J. als rotes Öl erhalten (30 mg, 86 % d.Th.). According to the above General Procedure B, 23 mg of 22 were reacted with 4-iodonitrobenzene for 2 hours. MPLC (LP: EtOAc 80: 20 ^ -0: 100 + 3% triethylamine) gave the title compound 4J. as a red oil (30 mg, 86% of theory).
Rf (PE/EtOAc 4:1): 0,40 R f (PE / EtOAc 4: 1): 0.40
1H-NMR (200 MHz, DMSO-d6) δ: 5,14 (s, 2H, -CH2), 7,00 (d, J=7,4Hz, 2H, ArH), 7,09-7,39 (m, 8H, ArH), 7,57 (d, 2H), 8,15 (d, J=8,8Hz, H3"\ H5'"), 8,26 (s, 1 H, H4). 3C-NMR (50 MHz, Aceton-d6) δ: 50,5 (t, -CH2), 112,6 (s), 121 ,9 (d, ArC), 124,2 (d, ArC), 125,0 (s), 125,1 (d, ArC), 125,2 (d, ArC), 128,7 (d, ArC), 129,0 (d, ArC), 129,1 (d, ArC), 129,6 (d, ArC), 130,3 (d, ArC), 138,0 (s), 139,4 (s), 146,5 (s), 151 ,9 (s). 1 H-NMR (200 MHz, DMSO-d 6 ) δ: 5.14 (s, 2H, -CH 2 ), 7.00 (d, J = 7.4Hz, 2H, ArH), 7.09-7 , 39 (m, 8H, ArH), 7.57 (d, 2H), 8.15 (d, J = 8.8Hz, H3 "\ H5 '"), 8.26 (s, 1H, H4) , 3C-NMR (50 MHz, acetone-d 6 ) δ: 50.5 (t, -CH 2 ), 112.6 (s), 121, 9 (d, ArC), 124.2 (d, ArC), 125.0 (s), 125.1 (d, ArC), 125.2 (d, ArC), 128.7 (d, ArC), 129.0 (d, ArC), 129.1 (d, ArC ), 129.6 (d, ArC), 130.3 (d, ArC), 138.0 (s), 139.4 (s), 146.5 (s), 151, 9 (s).
HRMS (Cl, NH3): MH\ gef.: 388,1118; ber. für C22H17N3O2S: 388,1114. HRMS (Cl, NH 3): MH \ Found .: 388.1118; calcd for C22H17N3O2S: 388.1114.
Beispiel 19 Example 19
Herstellun von N-(3-Benzyl-5-(4-methoxyphenyl)thiazol-2(3H)-yliden)anilin 42 Preparation of N- (3-Benzyl-5- (4-methoxyphenyl) thiazole-2 (3H) -ylidene) aniline 42
22 42 22 42
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 22 mit 4-lodanisol 18 h lang umgesetzt. Mittels MPLC (LP:EtOAc 12:1-^0:100 + 3 % Triethylamin) wurde die Titelverbindung 42 als braunes Öl erhalten (19 mg, 42 % d.Th.). According to General Procedure B above, 30 mg of 22 was reacted with 4-iodoanisole for 18 hours. MPLC (LP: EtOAc 12: 1- ^ 0: 100 + 3% triethylamine) gave the title compound 42 as a brown oil (19 mg, 42% of theory).
Rf (PE/EtOAc 4:1): 0,40 R f (PE / EtOAc 4: 1): 0.40
1H-NMR (200 MHz, DMSO-d6) δ: 3,74 (s, 3H, -OCH3), 5,21 (s, 2H, -CH2), 6,89 (d, J=8,7Hz, 2H, ArH), 7,29-7,49 (m, 13H, ArH). 1 H-NMR (200 MHz, DMSO-d 6 ) δ: 3.74 (s, 3H, -OCH 3 ), 5.21 (s, 2H, -CH 2 ), 6.89 (d, J = 8 , 7Hz, 2H, ArH), 7.29-7.49 (m, 13H, ArH).
13C-NMR (50 MHz, CDCI3) δ: 55,1 (q, -OCH3), 55,8 (t, -CH2), 114,3 (d, ArC), 125,1 (s), 126,2 (d, ArC), 126,8 (d, ArC), 126,9 (d, ArC), 127,2 (d, ArC), 127,7 (d, ArC), 128,4 (d, ArC), 129,8 (d, ArC), 133,6 (d, ArC), 137,6 (s), 145,1 (s), 158,6 (s), 168,8 (s). 13 C-NMR (50 MHz, CDCl 3 ) δ: 55.1 (q, -OCH 3 ), 55.8 (t, -CH 2 ), 114.3 (d, ArC), 125.1 (s) , 126.2 (d, ArC), 126.8 (d, ArC), 126.9 (d, ArC), 127.2 (d, ArC), 127.7 (d, ArC), 128.4 ( d, ArC), 129.8 (d, ArC), 133.6 (d, ArC), 137.6 (s), 145.1 (s), 158.6 (s), 168.8 (s) ,
HRMS (Cl, NH3): MH+, gef.: 373,1373; ber. für C23H2oN2OS: 373,1369. HRMS (Cl, NH 3): MH +, Found .: 373.1373; calc. for C 23 H 2 O 2 OS: 373.1369.
Beispiel 20 Example 20
Herstellung von N-(3-Benzyl-5-(4-fluorphenyl)thiazol-2(3H)-yliden)anilin 43 Preparation of N- (3-benzyl-5- (4-fluorophenyl) thiazole-2 (3H) -ylidene) aniline 43
22 43 22 43
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 22 mit 4-lodfluorbenzol 24 h lang umgesetzt. Mittels MPLC (LP:EtOAc 12:1 ^-0:100 + 3 % Triethylamin) wurde die Titelverbindung 43 als braunes öl erhalten (19 mg, 46 % d.Th.). According to General Procedure B above, 30 mg of 22 was reacted with 4-iodofluorobenzene for 24 hours. MPLC (LP: EtOAc 12: 1 → -0: 100 + 3% triethylamine) gave the title compound 43 as a brown oil (19 mg, 46% of theory).
Rf (PE/EtOAc 10:1): 0,48R f (PE / EtOAc 10: 1): 0.48
H-NMR (200 MHz, CDCI3) δ: 5,12 (s, 2H, -CH2), 6,73 (s, 1 H, H4), 6,93-7,22 (m, 7H, ArH), 7,33-7,39 (m, 7H, ArH).H-NMR (200 MHz, CDCI 3) δ: 5.12 (s, 2H, CH 2), 6.73 (s, 1 H, H4), 6.93 to 7.22 (m, 7H, ArH ), 7.33-7.39 (m, 7H, ArH).
3C-NMR (50 MHz, CDCI3) δ: 50,48 (t, -CH2), 1 15,7 (dd, J=22,0Hz, C2m, C6m), 121 ,4 (d, ArC), 123,2 (d, ArC), 126,2 (dd, JCF=8,0HZ, C3"', C5'"), 127,3 (d, ArC), 128,0 (d, ArC), 128,1 (d, ArC), 128,5 (d, ArC), 128,7 (d, ArC), 129,5 (d, ArC), 129,9 (d, ArC), 136,8 (s, Ar ), 151 ,9 (s), 162,1 (ds, JCF=250HZ, 1 C, C4,m). 3C-NMR (50 MHz, CDCI 3) δ: 50.48 (t, CH 2), 1 15.7 (dd, J = 22,0Hz, C2 m, C6 m), 121 4 (d, ArC ), 123.2 (d, ArC), 126.2 (dd, J C F = 8.0HZ, C3 "', C5'"), 127.3 (d, ArC), 128.0 (d, ArC ), 128.1 (d, ArC), 128.5 (d, ArC), 128.7 (d, ArC), 129.5 (d, ArC), 129.9 (d, ArC), 136.8 (s, Ar), 151, 9 (s), 162.1 (ds, J C F = 250HZ, 1 C, C4 , m ).
MS m/z (Cl, NH3): 360 (28, M+), 269 (51), 237 (1 1 ), 167 (100), 139 (12), 91 (52), 65 (12). MS m / z (Cl, NH 3): 360 (28, M +), 269 (51), 237 (1 1), 167 (100), 139 (12), 91 (52), 65 (12).
HRMS (Cl, NH3): MH+, gef.: 361 ,1 171 ; ber. für C22H17FN2S: 362,1169. HRMS (Cl, NH 3): MH +, Found .: 361, 1 171; calcd for C22H17FN2S: 362.1169.
-2-yl)thiazol-2(3H)-yliden)anilin 44 -2-yl) thiazole-2 (3H) -ylidene) aniline 44
22 44 22 44
Gemäß dem obigen Allgemeinen Verfahren B wurden 30 mg 22 mit 2-lodthiophen 24 h lang umgesetzt. Mittels MPLC (LP:EtOAc 12:1- 0:100 + 5 % Triethylamin) wurde die Titelverbindung 44 als braunes Öl erhalten (24 mg, 59 % d.Th.). According to General Procedure B above, 30 mg of 22 was reacted with 2-iodothiophene for 24 hours. MPLC (LP: EtOAc 12: 1-0: 100 + 5% triethylamine) gave the title compound 44 as a brown oil (24 mg, 59% of theory).
Rf (PE/EtOAc 9:1): 0,51 R f (PE / EtOAc 9: 1): 0.51
1H-NMR (200 MHz, CDCI3) δ: 5,09 (s, 2H, -CH2), 6,69 (s, 1 H, H4), 6,82 (d, J=3,7Hz, 1 H, H5,M), 6,92 (t, J=3,6Hz, H4'"), 7,10 (d, J=7,3Hz, 4H, ArH), 7,32-7,39 (m, 8H, ArH). 1 H-NMR (200 MHz, CDCl 3 ) δ: 5.09 (s, 2H, -CH 2 ), 6.69 (s, 1 H, H 4), 6.82 (d, J = 3.7Hz, 1 H, H5 , M ), 6.92 (t, J = 3.6Hz, H4 '''), 7.10 (d, J = 7.3Hz, 4H, ArH), 7.32-7.39 ( m, 8H, ArH).
MS m/z (Cl, NH3): 347 (43, M+), 257 (100), 225 (14), 213 (10), 167 (88), 127 (20), 91 (44). MS m / z (Cl, NH 3): 347 (43, M +), 257 (100), 225 (14), 213 (10), 167 (88), 127 (20), 91 (44).
HRMS (Cl, NH3): MH+, gef.: 349,0832, ber. für C20H16N2S2: 349,0828. HRMS (Cl, NH 3): MH +, Found .: 349.0832, Calcd for C20H16N2S2:. 349.0828.
Beispiel 22 - Arylierung von Thiazolimin 33 Example 22 - Arylation of thiazolimine 33
Herstellung von N-(3-Benzyl-5-(2-fluorpyridin-3-yl)thiazol-2(3H)-yliden)anilin-4-carbon- säureethylester 46 Preparation of N- (3-Benzyl-5- (2-fluoropyridin-3-yl) thiazol-2 (3H) -ylidene) aniline-4-carboxylic acid ethyl ester 46
33 46 33 46
Gemäß dem obigen Allgemeinen Verfahren B wurden 60 mg 33 mit 2-Fluor-3-iod- pyridin 20 h lang umgesetzt. Mittels MPLC wurde die Titelverbindung 46 als bräunliches Öl erhalten (60 mg, 77 % d.Th.). According to General Procedure B above, 60 mg of 33 was reacted with 2-fluoro-3-iodo-pyridine for 20 hours. MPLC gave the title compound 46 as a brownish oil (60 mg, 77% of theory).
Aktivitätstests activity tests
Präparierung von Zellproben Preparation of cell samples
Die Herstellung des Zellmaterials erfolgte am Institut für Pharmakologie der Medizinischen Universität Wien. C2C12-Mäusezellen wurden als Modell zur Bestimmung der differenzierungsfördernden Eigenschaften der wie oben beschrieben hergestellten Verbindungen eingesetzt. Dazu wurden die C2C12-Zellen zwei Tage lang in Kulturschalen mit Wachstumsmedium (Dulbecco's Modified Eagle's Medium, DMEM, mit 4,5 g/l Glucose, 4 mM L-Glutamin, 50 U/ml Penicillin, 50 pg/ml Streptomycin und 20 % fötalem Kälberserum) inkubiert, bevor die Testverbindungen in Differenzierungsmedium (im Wesentlichen identisch mit dem Wachstumsmedium, außer dass es anstelle von fötalem Kälberserum 2 % Pferdeserum enthielt) zugesetzt wurden. The cell material was produced at the Institute of Pharmacology of the Medical University of Vienna. C2C12 mouse cells were used as a model to determine the differentiation-promoting properties of the compounds prepared as described above. For this purpose, the C2C12 cells were incubated for two days in culture dishes with growth medium (Dulbecco's modified Eagle's medium, DMEM, with 4.5 g / l glucose, 4 mM L-glutamine, 50 U / ml penicillin, 50 pg / ml streptomycin and 20% fetal calf serum) were incubated before the test compounds were added in differentiation medium (substantially identical to the growth medium, except that it contained 2% horse serum instead of fetal calf serum).
Dazu wurden die Verbindungen zuerst in DMSO gelöst, und diese Lösungen wurden zu Aliquoten des Differenzierungsmediums zugesetzt. Als Vergleich wurde reines DMSO in Differenzierungsmedium gelöst. Zunächst wurden 5 μΜ Lösungen der Verbindungen eingesetzt, wobei jeder Versuch in Doppelansätzen durchgeführt wurde. For this purpose, the compounds were first dissolved in DMSO, and these solutions were added to aliquots of the differentiation medium. As a comparison was pure DMSO dissolved in differentiation medium. First, 5 μΜ solutions of the compounds were used, each experiment being carried out in duplicate.
Nach der zweitägigen Inkubation der Zellen in Wachstumsmedium wurde Letzteres entfernt, wonach 3 ml der obigen Lösungen der jeweiligen Verbindung und von DMSO in Differenzierungsmedium zu jeder Zellkultur zugesetzt wurden. Anschließend wurden die Zellen 7 Tage lang bei 37 °C inkubiert. Das Medium wurde währenddessen zweimal, nämlich nach 3 und nach 5 Tagen, ausgetauscht. Nach 7 Tagen wurden die Zellen geerntet, das Medium wurde entfernt, und die Zellen wurden mit PBS (phosphatgepufferter Salzlösung) gewaschen, um jegliche Reste an Differenzierungsmedium zu entfernen. Dies ist von essentieller Bedeutung, da das Medium Proteine enthält, die anschließende Proteomanalysen beeinflussen könnten. After the two-day incubation of the cells in growth medium, the latter was removed, after which 3 ml of the above solutions of the respective compound and DMSO in differentiation medium were added to each cell culture. Subsequently, the cells were incubated at 37 ° C for 7 days. Meanwhile, the medium was exchanged twice, after 3 and 5 days. After 7 days, the cells were harvested, the medium was removed, and the cells were washed with PBS (phosphate buffered saline) to remove any residual differentiation medium. This is of essential importance as the medium contains proteins that could influence subsequent proteome analyzes.
Nach dem Waschschritt wurden 800 μΙ PBS zu jeder Zellkultur zugesetzt, und die Zellen wurden von den Platten abgeschabt und in PBS suspendiert. Diese Suspensionen wurden in Eppendorf-Röhrchen übertragen und zentrifugiert. Der PBS-Über- stand wurde entfernt, und die Zellpellets wurden bei -80 °C eingefroren. Der Wasch- und der Zentrifugierschritt erfolgten bei 4 °C. After the washing step, 800 μΙ PBS was added to each cell culture and the cells were scraped from the plates and suspended in PBS. These suspensions were transferred to Eppendorf tubes and centrifuged. The PBS supernatant was removed and the cell pellets were frozen at -80 ° C. The washing and centrifuging steps were carried out at 4 ° C.
Beobachtung morphologischer Veränderungen Observation of morphological changes
Morphologische Veränderungen der Zellen wurden mittels Mikrofotografien der Zellkulturen nach 3, 5 und 7 Tagen dokumentiert. Normalerweise differenzieren C2C12- Zellen im Differenzierungsmedium zu reifen Skelettmuskelzellen (langen, vielkernigen "Myotuben"). Bereits nach 3 Tagen schienen manche der Verbindungen die Ge- schwindigkeit der Differenzierung im Vergleich zu den Kontrollen, die nur Differenzierungsmedium und DMSO enthielten, mitunter beträchtlich zu erhöhen. Morphological changes of the cells were documented by photomicrographs of the cell cultures after 3, 5 and 7 days. Normally, C2C12 cells in the differentiation medium differentiate into mature skeletal muscle cells (long, multinucleated "myotubes"). Already after 3 days, some of the compounds sometimes appeared to increase the rate of differentiation considerably compared to controls containing only differentiation medium and DMSO.
Unglücklicherweise entwickelten manche der Testverbindungen auch eine mehr oder weniger starke Toxizität für die Zellen, was sich anhand von abgestorbenem Zell- material in den Proben zeigte. Beide Effekte, d.h. Beschleunigung der Differenzierung und Toxizität, wurden während der folgenden Tage verstärkt. Unfortunately, some of the test compounds also developed more or less toxicity to the cells, as evidenced by dead cells. showed material in the samples. Both effects, ie acceleration of differentiation and toxicity, were enhanced during the following days.
Die Ergebnisse der Untersuchungen sind in nachstehender Tabelle 1 aufgelistet, wo- bei sowohl bezüglich Differenzierungsbeschleunigung als auch bezüglich Toxizität "-" jeweils für "keine Wirkung", "+" für "schwache Wirkung" und "++" für "starke Wirkung" stehen. Verbindungen, die keine beschleunigte Differenzierung bewirkten, sind als Vergleichsbeispiele "V" gekennzeichnet, während alle übrigen als "erfindungsgemäß" "E" bezeichnet werden. Davon werden jene mit möglichst geringer oder keinerlei Toxizität natürlich bevorzugt. The results of the tests are listed in Table 1 below, both for differentiation acceleration and for toxicity "-" in each case for "no effect", "+" for "weak effect" and "++" for "strong effect" , Compounds which did not effect accelerated differentiation are designated as Comparative Examples "V", while all others are designated "Inventive" "E". Of these, those with as little or no toxicity as possible are naturally preferred.
Tabelle 1 Table 1
Verbindung DifferenzierungsToxizität beschleunigung Connection differentiation toxicity acceleration
-V -V
28 -E ++ 28 -E ++
+ +
++ ++ ++ ++
31b-E ++ + 31b-E ++ +
-E ++ + Verbindung DifferenzierungsToxizität -E ++ + Compound differentiation toxicity
beschleunigung acceleration
39 - E ++ - 39 - E ++ -
Q> Q >
41 - E ++ + 41 - E ++ +
42 - E + - 42 - E + -
43 - E ++ - 43 - E ++ -
46 - E ++ ++ 46 - E ++ ++
Wie der Tabelle 1 zu entnehmen ist, weisen die zehn hergestellten Thiazolimine durchwegs differenzierungsbeschleunigende Wirkung auf, während von den zehn Thiazolaminen drei Verbindungen keine solche Wirkung zeigen. Aus der Tabelle geht außerdem klar hervor, dass sowohl die Art der Arylgruppen am Thiazolring, deren Substituenten als auch die Position dieser Substituenten eine wesentliche Rolle - sowohl für die Wirksamkeit als auch für die Toxizität - spielen. Stärker Elektronen anziehende Substituenten wie Nitro oder Fluor scheinen weiters einen entsprechend stärkeren aktivierenden Einfluss auf das aromatische Ringsystem auszuüben. Entsprechende Tests mit weiteren neuen Verbindungen sowie mit anderen Zelltypen werden von den Erfindern zurzeit durchgeführt. As can be seen from Table 1, the ten thiazolimines prepared show consistently differentiation-accelerating effect, while of the ten Thiazolamines three compounds show no such effect. It is also clear from the table that both the nature of the aryl groups on the thiazole ring, their substituents, and the position of these substituents play an essential role - both in terms of efficacy and toxicity. More electron-attractive substituents, such as nitro or fluorine, also appear to exert a correspondingly stronger activating influence on the aromatic ring system. Corresponding tests with other new compounds as well as with other cell types are currently being conducted by the inventors.
Somit stellt die vorliegende Erfindung eine Gruppe von Verbindungen bereit, die geeignet sind, die Differenzierung von Zellen zu beschleunigen, die auf relativ einfache und kostengünstige Weise zugänglich sind und die geringe bis keine Toxizität zeigen. Thus, the present invention provides a group of compounds capable of accelerating the differentiation of cells which are accessible in a relatively simple and inexpensive manner and which show little to no toxicity.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ATA1614/2010 | 2010-09-27 | ||
| AT16142010A AT510456B1 (en) | 2010-09-27 | 2010-09-27 | THIAZOLAMINE DERIVATIVES AS CELL DIFFERENTIATOR ACCUMULATORS |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012040754A2 true WO2012040754A2 (en) | 2012-04-05 |
| WO2012040754A3 WO2012040754A3 (en) | 2012-06-21 |
Family
ID=45841132
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/AT2011/000393 Ceased WO2012040754A2 (en) | 2010-09-27 | 2011-09-26 | Novel thiazolamine derivates as differentiation accelerators |
Country Status (2)
| Country | Link |
|---|---|
| AT (1) | AT510456B1 (en) |
| WO (1) | WO2012040754A2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12030875B2 (en) | 2018-09-07 | 2024-07-09 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
| US12157731B2 (en) | 2020-03-03 | 2024-12-03 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
| US12157732B2 (en) | 2021-08-25 | 2024-12-03 | PIC Therapeutics, Inc. | eIF4E inhibitors and uses thereof |
| US12234231B2 (en) | 2021-08-25 | 2025-02-25 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005047524A2 (en) | 2003-11-10 | 2005-05-26 | The Scripps Research Institute | Compositions and methods for inducing cell dedifferentiation |
| WO2007064891A1 (en) | 2005-12-01 | 2007-06-07 | The Scripps Research Institute | Compositions and methods for inducing neuronal differentiation |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5369108A (en) * | 1991-10-04 | 1994-11-29 | Sloan-Kettering Institute For Cancer Research | Potent inducers of terminal differentiation and methods of use thereof |
| WO2010051223A1 (en) * | 2008-10-31 | 2010-05-06 | Centocor Ortho Biotech Inc. | Differentiation of human embryonic stem cells to the pancreatic endocrine lineage |
| EP2251010A1 (en) * | 2009-05-08 | 2010-11-17 | Sygnis Bioscience GmbH & Co. KG | Use of thiabendazole and derivatives thereof for the therapy of neurological conditions |
-
2010
- 2010-09-27 AT AT16142010A patent/AT510456B1/en not_active IP Right Cessation
-
2011
- 2011-09-26 WO PCT/AT2011/000393 patent/WO2012040754A2/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005047524A2 (en) | 2003-11-10 | 2005-05-26 | The Scripps Research Institute | Compositions and methods for inducing cell dedifferentiation |
| WO2007064891A1 (en) | 2005-12-01 | 2007-06-07 | The Scripps Research Institute | Compositions and methods for inducing neuronal differentiation |
Non-Patent Citations (6)
| Title |
|---|
| C.B. ANDERSEN ET AL., ACS CHEMICAL BIOLOGY, vol. 3, no. 3, 2008, pages 180 - 192 |
| D. JUNG ET AL., J. AM. CHEM. SOC., vol. 131, 2009, pages 4774 - 4782 |
| M. HOSHINO ET AL., BIOCHEMICAL JOURNAL, vol. 427, 2010, pages 297 |
| M.P. HAY ET AL., J. MED. CHEM., vol. 53, no. 2, 2010, pages 787 - 797 |
| N. BOUQUIER ET AL., CHEM. BIOL., vol. 16, 2009, pages 657 - 666 |
| S. MAHBOOBI ET AL., J. MED. CHEM., vol. 52, no. 8, 2009, pages 2265 - 2279 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12030875B2 (en) | 2018-09-07 | 2024-07-09 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
| US12157731B2 (en) | 2020-03-03 | 2024-12-03 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
| US12157732B2 (en) | 2021-08-25 | 2024-12-03 | PIC Therapeutics, Inc. | eIF4E inhibitors and uses thereof |
| US12234231B2 (en) | 2021-08-25 | 2025-02-25 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| AT510456B1 (en) | 2012-11-15 |
| AT510456A1 (en) | 2012-04-15 |
| WO2012040754A3 (en) | 2012-06-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE60214392T2 (en) | 1,4-Disubstituted benzo-fused cycloalkyl urea compounds | |
| DE69503769T2 (en) | ISOXAZOLINE COMPOUND TO INHIBIT TNF RELEASE | |
| DE602004010829T2 (en) | ADAMANTAN AND AZABICYCLOOCTANESE AND NONANDERIVATES, PROCESS FOR THEIR PREPARATION AND THEIR USE AS DPP IV INHIBITORS | |
| EP1119557B1 (en) | Indeno-, naphto- and benzocyclohepta dihydrothiazole derivatives, the production thereof and their use as anorectic medicaments | |
| Bailey et al. | Pyrrole antibacterial agents. 2. 4, 5-Dihalopyrrole-2-carboxylic acid derivatives | |
| DE102004008141A1 (en) | Guanidine compounds and their use as binding partners for 5-HT5 receptors | |
| DE69500597T2 (en) | N-pyridyl carboxamide derivatives, process for their preparation and pharmaceutical compositions containing them | |
| JP2006517199A5 (en) | ||
| DD265899A5 (en) | PROCESS FOR THE PREPARATION OF NEW THIENO-TRIAZOLO-1,4-DIAZEPINO-2-CARBONSAEURES | |
| US20080306107A1 (en) | Compounds | |
| EP0183159A2 (en) | 1-Heteroaryl-4-aryl-pyrazolin-5-ones for use as medicaments | |
| CZ323094A3 (en) | Branched amino derivatives of thiazole, process of their preparation and pharmaceutical compositions containing thereof | |
| DE69006872T2 (en) | 1H, 3H, 7-pyrrolo [1,2-c] thiazolecarboxamide derivatives, their preparation and pharmaceutical preparations containing them. | |
| AT510456B1 (en) | THIAZOLAMINE DERIVATIVES AS CELL DIFFERENTIATOR ACCUMULATORS | |
| DE602004006379T2 (en) | BENZTHIAZOLE-3 OXIDE FOR THE TREATMENT OF PROLIFERATIVE DISORDER | |
| WO2004089881A1 (en) | New sulfonyl derivatives of aminonaphtols | |
| CH625526A5 (en) | ||
| DE69103630T2 (en) | OXAZOLE DERIVATIVES, THEIR PRODUCTION AND THE PHARMACEUTICAL PREPARATIONS CONTAINING THEM. | |
| CH396923A (en) | Process for the preparation of 4-mercapto-pyrazolo (3,4-d) pyrimidines | |
| DE3532279A1 (en) | 1,4-benzoxathiin derivatives | |
| DE102005005395A1 (en) | New thiazolidinone compounds are polo-like kinase inhibitors, useful for treating e.g. cancer, autoimmune diseases, cardiovascular diseases, infectious diseases, nephrological diseases and viral diseases | |
| US20080070946A1 (en) | Hydroxymethylbenzothiazoles Amides | |
| DE69607890T2 (en) | Aromatic hydroxamic acid compounds, their production and use | |
| US3523122A (en) | Novel 5-nitro-4-thiazolin-2-ylidene compounds | |
| DE69702433T2 (en) | Use of certain amides as samples for the detection of antitubulin activity and for resistance monitoring |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 11826129 Country of ref document: EP Kind code of ref document: A2 |