CN115449052B - A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method - Google Patents
A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method Download PDFInfo
- Publication number
- CN115449052B CN115449052B CN202211156451.5A CN202211156451A CN115449052B CN 115449052 B CN115449052 B CN 115449052B CN 202211156451 A CN202211156451 A CN 202211156451A CN 115449052 B CN115449052 B CN 115449052B
- Authority
- CN
- China
- Prior art keywords
- compound
- mixture
- polymer material
- solution
- folding
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/74—Polyisocyanates or polyisothiocyanates cyclic
- C08G18/76—Polyisocyanates or polyisothiocyanates cyclic aromatic
- C08G18/7657—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings
- C08G18/7664—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings containing alkylene polyphenyl groups
- C08G18/7671—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings containing alkylene polyphenyl groups containing only one alkylene bisphenyl group
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C253/00—Preparation of carboxylic acid nitriles
- C07C253/30—Preparation of carboxylic acid nitriles by reactions not involving the formation of cyano groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/08—Processes
- C08G18/10—Prepolymer processes involving reaction of isocyanates or isothiocyanates with compounds having active hydrogen in a first reaction step
- C08G18/12—Prepolymer processes involving reaction of isocyanates or isothiocyanates with compounds having active hydrogen in a first reaction step using two or more compounds having active hydrogen in the first polymerisation step
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/38—Low-molecular-weight compounds having heteroatoms other than oxygen
- C08G18/3819—Low-molecular-weight compounds having heteroatoms other than oxygen having nitrogen
- C08G18/3838—Low-molecular-weight compounds having heteroatoms other than oxygen having nitrogen containing cyano groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/48—Polyethers
- C08G18/4854—Polyethers containing oxyalkylene groups having four carbon atoms in the alkylene group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/65—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen
- C08G18/66—Compounds of groups C08G18/42, C08G18/48, or C08G18/52
- C08G18/6666—Compounds of group C08G18/48 or C08G18/52
- C08G18/667—Compounds of group C08G18/48 or C08G18/52 with compounds of group C08G18/32 or polyamines of C08G18/38
- C08G18/6674—Compounds of group C08G18/48 or C08G18/52 with compounds of group C08G18/32 or polyamines of C08G18/38 with compounds of group C08G18/3203
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K9/00—Tenebrescent materials, i.e. materials for which the range of wavelengths for energy absorption is changed as a result of excitation by some form of energy
- C09K9/02—Organic tenebrescent materials
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/14—Macromolecular compounds
- C09K2211/1408—Carbocyclic compounds
- C09K2211/1425—Non-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Polyurethanes Or Polyureas (AREA)
Abstract
本发明涉及一种基于折叠‑解折叠效应的力致变色高分子材料及其制备方法,该力致变色高分子材料由二元醇、二异氰酸酯与氰基取代亚苯基乙烯二聚体DCOP聚合得到。当聚合物薄膜在UV照射下进行单轴拉伸(λ=365nm)时,激基缔合物解折叠,荧光从黄色变为绿色,随着应力的放松,薄膜又从绿色恢复到黄色,并且这个过程可以反复多次。本发明提出的策略易于实现,丰富了分子镊子型力致变色材料,进一步推动了力致变色材料的发展。
The invention relates to a mechanochromic polymer material based on the folding-unfolding effect and a preparation method thereof. The mechanochromic polymer material is polymerized by diol, diisocyanate and cyano-substituted phenylene vinylene dimer DCOP get. When the polymer film is uniaxially stretched (λ=365nm) under UV irradiation, the excimer is unfolded, and the fluorescence changes from yellow to green. With the relaxation of the stress, the film returns from green to yellow, and This process can be repeated many times. The strategy proposed in the present invention is easy to implement, enriches the molecular tweezers type mechanochromic materials, and further promotes the development of mechanochromic materials.
Description
技术领域technical field
本发明涉及力致变色智能材料领域,特别是涉及一种基于折叠-解折叠效应的力致变色高分子材料及其制备方法。The invention relates to the field of mechanochromic smart materials, in particular to a mechanochromic polymer material based on the folding-unfolding effect and a preparation method thereof.
背景技术Background technique
对聚合物施加过大的应力会导致分子链断裂,从而导致材料的宏观断裂行为,因此,了解这种损坏是如何以及在哪里发生的非常重要。近年来,具有光学力响应的力致变色聚合物的发展越来越受到人们的关注,这种聚合物应用广泛,包括压力传感材料以及结构材料中的损伤检测等等。最常使用的力响应荧光团通常具有特征弱键,它们通常以共价键连接到高分子链中,使聚合物材料具有力响应性。当分子链的应力超过一定阈值时,这些弱键就会发生均裂或异裂反应。这种结构变化虽然会导致分子吸收或荧光性质的变化,用于检测材料的应力分布。然而,这样的力敏基团中弱键的断裂是典型的不可逆过程,这直接破坏了材料本身的效用。Excessive stress on a polymer can cause the molecular chains to break, resulting in the macroscopic fracture behavior of the material, so it is important to understand how and where this damage occurs. In recent years, the development of mechanochromic polymers with optical force response has attracted increasing attention for a wide range of applications, including pressure sensing materials and damage detection in structural materials, etc. The most commonly used force-responsive fluorophores usually have characteristic weak bonds, which are often covalently linked into polymer chains, making polymeric materials force-responsive. When the stress of the molecular chain exceeds a certain threshold, these weak bonds will undergo homolytic or heterolytic reactions. Such structural changes though can lead to changes in molecular absorption or fluorescence properties, which are used to detect stress distributions in materials. However, the breaking of weak bonds in such force-sensitive groups is a typical irreversible process, which directly destroys the utility of the material itself.
力敏荧光团以非牺牲甚至可逆的方式来预警才是可取的,近年来,轮烷被开发为一种高效的机械致变色荧光力传感器,它是一个由携带荧光团的大环和包含匹配淬灭剂的哑铃状分子组成的机械互锁荧光团,其被整合到聚合物主链中,由于分子链的拉伸会导致荧光团和淬灭剂的空间分离,从而使荧光开启。通过选择不同的荧光团,可以直接调节轮烷的光学性质。同时,这种荧光团可用于观察细胞分裂时产生的超小机械力。但是漫长的合成路线和较低的合成产率对它的应用建立了巨大的障碍。此外,Weder等人报道了一种含有两个荧光基团的环状力致变色荧光团,其能够形成分子内激基缔合物,在受力变形后,含上述环状荧光团聚氨酯的荧光发射由激基缔合物变为单分散为主,这是由于分子内染料分子的相对错位造成。It is desirable for force-sensitive fluorophores to alert in a non-sacrificial or even reversible manner. In recent years, rotaxanes have been developed as an efficient mechanochromic fluorescent force sensor, which is a macrocycle carrying a fluorophore and contains a matching A mechanically interlocked fluorophore composed of dumbbell-shaped molecules of the quencher, which is integrated into the polymer backbone, causes the spatial separation of the fluorophore and the quencher due to molecular chain stretching, thereby enabling the fluorescence to be turned on. By choosing different fluorophores, the optical properties of rotaxanes can be directly tuned. At the same time, this fluorophore can be used to observe the ultra-small mechanical forces produced by cell division. But the long synthetic route and low synthetic yield have established huge obstacles to its application. In addition, Weder et al. reported a cyclic mechanochromic fluorophore containing two fluorophores, which can form intramolecular excimer associations. The emission changes from excimate to monodisperse dominance due to the relative dislocation of the dye molecules within the molecule.
发明内容Contents of the invention
基于此,本发明开发出了具有类似上述优点且易于制备的新一代镊子型力敏荧光团,并由此合成了具有折叠-解折叠效应的力致变色高分子材料。设计的镊子型力敏荧光分子是由一个短连接单元将两个荧光基团进行化学键合而形成。最初,力敏荧光团中的两个荧光基团通过非共价相互作用堆积在一起形成二聚体激基缔合物,当外力传递到镊子上时,二聚体被拉开,形成单分散主导荧光。因此,通过对π共轭染料分子的折叠-解折叠,可以获得可逆的力敏光响应特性。Based on this, the present invention has developed a new generation of tweezer-type force-sensitive fluorophores that have similar advantages to the above and are easy to prepare, and thus synthesized a mechanochromic polymer material with a folding-unfolding effect. The designed tweezer-type force-sensitive fluorescent molecule is formed by chemically bonding two fluorescent groups with a short linker unit. Initially, the two fluorophores in the force-sensitive fluorophore stack together through non-covalent interactions to form a dimer excimer, and when an external force is transmitted to the tweezers, the dimer is pulled apart to form a monodisperse dominant fluorescence. Therefore, reversible force-sensitive photoresponsive properties can be obtained through the folding-unfolding of π-conjugated dye molecules.
为实现上述目的,本发明提供的技术方案是:To achieve the above object, the technical solution provided by the invention is:
一种基于折叠-解折叠效应的力致变色高分子材料,该力致变色高分子材料由二元醇、二异氰酸酯与氰基取代亚苯基乙烯二聚体DCOP聚合得到,氰基取代亚苯基乙烯二聚体DCOP的结构如下:A mechanochromic polymer material based on folding-unfolding effect, the mechanochromic polymer material is obtained by polymerization of diol, diisocyanate and cyano-substituted phenylene vinylene dimer DCOP, cyano-substituted phenylene The structure of ethylene dimer DCOP is as follows:
所述二元醇为聚乙二醇、乙二醇、1,3-丙二醇、1,4-丁二醇、1,5-戊二醇、1,6-己二醇中的至少一种。The dihydric alcohol is at least one of polyethylene glycol, ethylene glycol, 1,3-propanediol, 1,4-butanediol, 1,5-pentanediol, and 1,6-hexanediol.
所述二异氰酸酯为六亚甲基二异氰酸酯(HDI)、二环己基甲烷二异氰酸酯、二苯基甲烷二异氰酸酯(MDI)、异佛尔酮二异氰酸酯、甲苯二异氰酸酯中的至少一种。The diisocyanate is at least one of hexamethylene diisocyanate (HDI), dicyclohexylmethane diisocyanate, diphenylmethane diisocyanate (MDI), isophorone diisocyanate and toluene diisocyanate.
基于折叠-解折叠效应的力致变色高分子材料的制备方法,包括以下步骤:将一定量氰基取代亚苯基乙烯二聚体DCOP、二元醇和二异氰酸酯溶于四氢呋喃中,向混合物中滴加二月桂酸二丁基锡,并在室温下搅拌3小时;然后再加入溶有丁二醇的THF溶液,在室温下混合物搅拌反应24小时;随后再在反应混合物中加入乙醇,搅拌30分钟后,将反应混合物倒入乙醇中;过滤收集黄色沉淀;固体再从THF中析出两次,得到黄色固体DPU-m,即为所述的力致变色高分子材料。The preparation method of the mechanochromic polymer material based on the folding-unfolding effect comprises the following steps: dissolving a certain amount of cyano-substituted phenylene vinylene dimer DCOP, dibasic alcohol and diisocyanate in tetrahydrofuran, and dropping into the mixture Add dibutyltin dilaurate, and stir at room temperature for 3 hours; then add a THF solution dissolved in butanediol, and stir the mixture at room temperature for 24 hours; then add ethanol to the reaction mixture, and stir for 30 minutes, The reaction mixture was poured into ethanol; the yellow precipitate was collected by filtration; the solid was precipitated twice from THF to obtain a yellow solid DPU-m, which was the mechanochromic polymer material.
进一步的,二异氰酸酯与二元醇的摩尔比为1:0.9~1:1.1。Further, the molar ratio of diisocyanate to diol is 1:0.9˜1:1.1.
进一步的,聚合物DPU中DCOP的质量分数为0.1%~0.5%。Further, the mass fraction of DCOP in the polymer DPU is 0.1%-0.5%.
进一步的,DCOP的制备包括以下步骤:Further, the preparation of DCOP comprises the following steps:
1)将2,5-二羟基-1,4-对苯二甲醛、1-溴己烷、无水碳酸钾和DMF加入反应容器中,得到混合物;将混合物在80℃加热24h,反应完成后,倒入水中,用CH2Cl2提取若干次,无水Na2SO4干燥后,真空蒸发有机层,得到粗产物;通过在甲醇中重结晶得到化合物A;1) Add 2,5-dihydroxy-1,4-terephthalaldehyde, 1-bromohexane, anhydrous potassium carbonate and DMF into the reaction vessel to obtain a mixture; heat the mixture at 80°C for 24h, after the reaction is completed , poured into water, extracted several times with CH 2 Cl 2 , dried over anhydrous Na 2 SO 4 , and evaporated the organic layer in vacuo to obtain a crude product; compound A was obtained by recrystallization in methanol;
2)将三甘醇溶于干燥的CH2Cl2中,在氮气气氛下用冰浴将溶液冷却至0℃,加入三乙胺;随后,在冷却的反应混合物中加入对甲苯磺酰氯;完全加入后,将溶液加热至室温,搅拌24小时;用水洗涤混合物,并用Na2SO4将有机层干燥,过滤,真空浓缩,得到粗产物;柱色谱提纯得到化合物B;2) Dissolve triethylene glycol in dry CH 2 Cl 2 , cool the solution to 0° C. with an ice bath under a nitrogen atmosphere, and add triethylamine; subsequently, add p-toluenesulfonyl chloride to the cooled reaction mixture; completely After the addition, the solution was heated to room temperature and stirred for 24 hours; the mixture was washed with water, and the organic layer was dried with Na 2 SO 4 , filtered, and concentrated in vacuo to obtain a crude product; purified by column chromatography to obtain compound B;
3)将对羟基苯乙腈、化合物B和K2CO3加入到乙腈中;将混合物回流12h,冷却至室温后,过滤,用乙腈洗涤若干次,有机层用无水Na2SO4干燥,真空浓缩;柱色谱提纯得到化合物C;3) Add p-hydroxybenzonitrile, compound B and K 2 CO 3 into acetonitrile; reflux the mixture for 12 h, cool to room temperature, filter, wash with acetonitrile several times, dry the organic layer with anhydrous Na 2 SO 4 , vacuum Concentrate; Purify by column chromatography to obtain compound C;
4)在CH2Cl2中加入对羟基苯乙腈和无水对甲苯磺酸;将溶液冷却至0℃,然后滴加3,4-2H-二氢吡喃;完全加入后,将混合物加热至室温,搅拌6小时;用Na2CO3水溶液洗涤溶液;有机相用无水硫酸钠干燥,然后减压蒸馏;柱色谱提纯得到化合物D;4) Add p-hydroxybenzonitrile and anhydrous p-toluenesulfonic acid in CH 2 Cl 2 ; cool the solution to 0°C, then add 3,4-2H-dihydropyran dropwise; after complete addition, heat the mixture to Stir at room temperature for 6 hours; wash the solution with Na 2 CO 3 aqueous solution; dry the organic phase with anhydrous sodium sulfate, then distill under reduced pressure; purify by column chromatography to obtain compound D;
5)将化合物A、化合物C和化合物D在70℃溶于t-BuOH和THF的混合物中;快速加入t-BuOK和n-Bu4NOH,溶液立即转化为紫色;将混合物在70℃下搅拌15分钟,冷却至室温,倒入水中;用CH2Cl2溶液提取两次;有机层经无水Na2SO4干燥后,减压蒸馏;柱色谱提纯得到化合物E和MCOP;5) Dissolve compound A, compound C and compound D in a mixture of t-BuOH and THF at 70°C; quickly add t-BuOK and n-Bu 4 NOH, the solution turns purple immediately; stir the mixture at 70°C 15 minutes, cooled to room temperature, poured into water; extracted twice with CH 2 Cl 2 solution; dried the organic layer over anhydrous Na 2 SO 4 , and distilled under reduced pressure; purified by column chromatography to obtain compound E and MCOP;
6)在甲醇中加入化合物E和对甲苯磺酸;混合物在室温下搅拌一夜;在此期间形成了红色固体沉淀物;这种固体用甲醇过滤和洗涤;将固体真空干燥,得到红色固体化合物F;6) Compound E and p-toluenesulfonic acid were added in methanol; the mixture was stirred overnight at room temperature; a red solid precipitate was formed during this period; this solid was filtered and washed with methanol; the solid was dried in vacuo to give compound F as a red solid ;
7)将四乙二醇和三乙胺溶于CH2Cl2中,并将溶液冷却至0℃,滴加对甲苯磺酰氯溶液;将得到的混合物在室温下搅拌16小时;溶液依次用5%HCl(aq)、饱和NaHCO3、H2O洗涤;有机层经无水Na2SO4干燥,然后减压蒸馏;柱色谱提纯得到化合物G;7) Dissolve tetraethylene glycol and triethylamine in CH 2 Cl 2 , and cool the solution to 0°C, and add p-toluenesulfonyl chloride solution dropwise; stir the resulting mixture at room temperature for 16 hours; HCl(aq), saturated NaHCO 3 , H 2 O washed; the organic layer was dried over anhydrous Na 2 SO 4 , and then distilled under reduced pressure; column chromatography was purified to obtain compound G;
8)将化合物F、化合物G、K2CO3和乙腈加入烧瓶中;将混合物回流24h,冷却至室温后,过滤,用乙腈洗涤若干次,真空干燥得到红固体DCOP。8) Compound F, compound G, K 2 CO 3 and acetonitrile were added into the flask; the mixture was refluxed for 24 h, cooled to room temperature, filtered, washed several times with acetonitrile, and vacuum dried to obtain DCOP as a red solid.
进一步的,步骤1)中2,5-二羟基-1,4-对苯二甲醛、1-溴己烷、无水碳酸钾的反应摩尔比例为:1/2.5/2.5;步骤3)中对羟基苯乙腈、化合物B和K2CO3的反应摩尔比例为:1/1.2/1.5。Further, the reaction molar ratio of 2,5-dihydroxy-1,4-terephthalaldehyde, 1-bromohexane, and anhydrous potassium carbonate in step 1) is: 1/2.5/2.5; The reaction molar ratio of hydroxyphenylacetonitrile, compound B and K 2 CO 3 is: 1/1.2/1.5.
进一步的,步骤5)中化合物A、化合物C和化合物D的反应摩尔比例为:1/1.1/1.2;步骤6)中化合物E和对甲苯磺酸的反应摩尔比例为:100/0.5。Further, the reaction molar ratio of compound A, compound C and compound D in step 5) is: 1/1.1/1.2; the reaction molar ratio of compound E and p-toluenesulfonic acid in step 6) is: 100/0.5.
进一步的,步骤8)中化合物F、化合物G、K2CO3的反应摩尔比例为:2/1/2.5。Further, the reaction molar ratio of compound F, compound G and K 2 CO 3 in step 8) is: 2/1/2.5.
本发明还保护该基于折叠-解折叠效应的力致变色高分子材料在可逆力致变色材料中的应用。The present invention also protects the application of the mechanochromic polymer material based on the folding-unfolding effect in reversible mechanochromic materials.
与现有技术相比,本发明的有益效果是:Compared with prior art, the beneficial effect of the present invention is:
本发明设计的基于DCOP镊子分子的力致变色高分子材料表现出良好的可逆力致变色性能。所述高分子材料中的DCOP镊子分子的力响应特性主要来自于COP基团在外力刺激下的分子内折叠-解折叠效应。在基态下,COP基团之间存在一定程度的分子内相互作用,形成激基缔合物。当聚合物薄膜在UV照射下进行单轴拉伸(λ=365nm)时,激基缔合物解折叠,荧光从黄色变为绿色,随着应力的放松,薄膜又从绿色恢复到黄色,并且这个过程可以反复多次。本发明提出的策略易于实现,丰富了分子镊子型力致变色材料,进一步推动了力致变色材料的发展。The DCOP tweezers-based mechanochromic polymer material designed in the present invention exhibits good reversible mechanochromic performance. The force response characteristics of the DCOP tweezers in the polymer material mainly come from the intramolecular folding-unfolding effect of the COP group under external force stimulation. In the ground state, there is a certain degree of intramolecular interaction between the COP groups to form excimer associations. When the polymer film is uniaxially stretched (λ=365nm) under UV irradiation, the excimer is unfolded, and the fluorescence changes from yellow to green. With the relaxation of the stress, the film returns from green to yellow, and This process can be repeated many times. The strategy proposed in the present invention is easy to implement, enriches the molecular tweezers type mechanochromic materials, and further promotes the development of mechanochromic materials.
附图说明Description of drawings
图1.DCOP和MCOP在THF溶液中的紫外可见吸收光谱和荧光发射光谱(c=5um/L,λex=365nm)。Fig. 1. UV-Vis absorption spectrum and fluorescence emission spectrum (c=5um/L, λex=365nm) of DCOP and MCOP in THF solution.
图2.(a)DPU-0.2和MPU-0.2在UV光和可见光下的图像;DPU-0.2(b)和MPU-0.2(c)薄膜的荧光衰减谱。Figure 2. (a) Images of DPU-0.2 and MPU-0.2 under UV and visible light; fluorescence decay spectra of DPU-0.2(b) and MPU-0.2(c) films.
图3.DPU-0.2薄膜的归一化荧光光谱(应变逐渐增大)。Figure 3. Normalized fluorescence spectra of DPU-0.2 films (increasing strain).
具体实施方式Detailed ways
以下通过实施例的形式对本发明的上述内容再作进一步的详细说明,但不应将此理解为本发明上述主题的范围仅限于以下的实施例,凡基于本发明上述内容所实现的技术均属于本发明的范围。The above-mentioned content of the present invention will be described in further detail below by the form of the embodiment, but this should not be interpreted as the scope of the above-mentioned theme of the present invention being limited to the following embodiments, all technologies realized based on the above-mentioned content of the present invention belong to scope of the invention.
本发明制备得到的力致变色高分子材料,按照下述方法进行测试:The mechanochromic polymer material prepared by the present invention is tested according to the following method:
核磁共振氢谱和碳谱用Bruker AVANCEⅡ核磁共振仪测定,四甲基硅为内标,溶剂为氘代氯仿或氘代二甲亚砜。荧光光谱用Shimadzu RF-5301PC荧光分光光度计测得。The H NMR and C NMR spectra were measured with a Bruker AVANCE II NMR instrument, tetramethyl silicon was used as an internal standard, and the solvent was deuterated chloroform or deuterated dimethyl sulfoxide. Fluorescence spectra were measured with a Shimadzu RF-5301PC fluorescence spectrophotometer.
DCOP的制备包括以下步骤:The preparation of DCOP comprises the following steps:
1)将2,5-二羟基-1,4-对苯二甲醛、1-溴己烷、无水碳酸钾和DMF加入反应容器中,得到混合物;将混合物在80℃加热24h,反应完成后,倒入水中,用CH2Cl2提取若干次,无水Na2SO4干燥后,真空蒸发有机层,得到粗产物;通过在甲醇中重结晶得到化合物A;1) Add 2,5-dihydroxy-1,4-terephthalaldehyde, 1-bromohexane, anhydrous potassium carbonate and DMF into the reaction vessel to obtain a mixture; heat the mixture at 80°C for 24h, after the reaction is completed , poured into water, extracted several times with CH 2 Cl 2 , dried over anhydrous Na 2 SO 4 , and evaporated the organic layer in vacuo to obtain a crude product; compound A was obtained by recrystallization in methanol;
2)将三甘醇溶于干燥的CH2Cl2中,在氮气气氛下用冰浴将溶液冷却至0℃,加入三乙胺;随后,在冷却的反应混合物中加入对甲苯磺酰氯;完全加入后,将溶液加热至室温,搅拌24小时;用水洗涤混合物,并用Na2SO4将有机层干燥,过滤,真空浓缩,得到粗产物;柱色谱提纯得到化合物B;2) Dissolve triethylene glycol in dry CH 2 Cl 2 , cool the solution to 0° C. with an ice bath under a nitrogen atmosphere, and add triethylamine; subsequently, add p-toluenesulfonyl chloride to the cooled reaction mixture; completely After the addition, the solution was heated to room temperature and stirred for 24 hours; the mixture was washed with water, and the organic layer was dried with Na 2 SO 4 , filtered, and concentrated in vacuo to obtain a crude product; purified by column chromatography to obtain compound B;
3)将对羟基苯乙腈、化合物B和K2CO3加入到乙腈中;将混合物回流12h,冷却至室温后,过滤,用乙腈洗涤若干次,有机层用无水Na2SO4干燥,真空浓缩;柱色谱提纯得到化合物C;3) Add p-hydroxybenzonitrile, compound B and K 2 CO 3 into acetonitrile; reflux the mixture for 12 h, cool to room temperature, filter, wash with acetonitrile several times, dry the organic layer with anhydrous Na 2 SO 4 , vacuum Concentrate; Purify by column chromatography to obtain compound C;
4)在CH2Cl2中加入对羟基苯乙腈和无水对甲苯磺酸;将溶液冷却至0℃,然后滴加3,4-2H-二氢吡喃;完全加入后,将混合物加热至室温,搅拌6小时;用Na2CO3水溶液洗涤溶液;有机相用无水硫酸钠干燥,然后减压蒸馏;柱色谱提纯得到化合物D;4) Add p-hydroxybenzonitrile and anhydrous p-toluenesulfonic acid in CH 2 Cl 2 ; cool the solution to 0°C, then add 3,4-2H-dihydropyran dropwise; after complete addition, heat the mixture to Stir at room temperature for 6 hours; wash the solution with Na 2 CO 3 aqueous solution; dry the organic phase with anhydrous sodium sulfate, then distill under reduced pressure; purify by column chromatography to obtain compound D;
5)将化合物A、化合物C和化合物D在70℃溶于t-BuOH和THF的混合物中;快速加入t-BuOK和n-Bu4NOH,溶液立即转化为紫色;将混合物在70℃下搅拌15分钟,冷却至室温,倒入水中;用CH2Cl2溶液提取两次;有机层经无水Na2SO4干燥后,减压蒸馏;柱色谱提纯得到化合物E和MCOP;5) Dissolve compound A, compound C and compound D in a mixture of t-BuOH and THF at 70°C; quickly add t-BuOK and n-Bu 4 NOH, the solution turns purple immediately; stir the mixture at 70°C 15 minutes, cooled to room temperature, poured into water; extracted twice with CH 2 Cl 2 solution; dried the organic layer over anhydrous Na 2 SO 4 , and distilled under reduced pressure; purified by column chromatography to obtain compound E and MCOP;
6)在甲醇中加入化合物E和对甲苯磺酸;混合物在室温下搅拌一夜;在此期间形成了红色固体沉淀物;这种固体用甲醇过滤和洗涤;将固体真空干燥,得到红色固体化合物F;6) Compound E and p-toluenesulfonic acid were added in methanol; the mixture was stirred overnight at room temperature; a red solid precipitate was formed during this period; this solid was filtered and washed with methanol; the solid was dried in vacuo to give compound F as a red solid ;
7)将四乙二醇和三乙胺溶于CH2Cl2中,并将溶液冷却至0℃,滴加对甲苯磺酰氯溶液;将得到的混合物在室温下搅拌16小时;溶液依次用5%HCl(aq)、饱和NaHCO3、H2O洗涤;有机层经无水Na2SO4干燥,然后减压蒸馏;柱色谱提纯得到化合物G;7) Dissolve tetraethylene glycol and triethylamine in CH 2 Cl 2 , and cool the solution to 0°C, and add p-toluenesulfonyl chloride solution dropwise; stir the resulting mixture at room temperature for 16 hours; HCl(aq), saturated NaHCO 3 , H 2 O washed; the organic layer was dried over anhydrous Na 2 SO 4 , and then distilled under reduced pressure; column chromatography was purified to obtain compound G;
8)将化合物F、化合物G、K2CO3和乙腈加入烧瓶中;将混合物回流24h,冷却至室温后,过滤,用乙腈洗涤若干次,真空干燥得到红固体DCOP。8) Compound F, compound G, K 2 CO 3 and acetonitrile were added into the flask; the mixture was refluxed for 24 h, cooled to room temperature, filtered, washed several times with acetonitrile, and vacuum dried to obtain DCOP as a red solid.
步骤1)中2,5-二羟基-1,4-对苯二甲醛、1-溴己烷、无水碳酸钾的反应摩尔比例为:1/2.5/2.5;步骤3)中对羟基苯乙腈、化合物B和K2CO3的反应摩尔比例为:1/1.2/1.5。In step 1), the reaction molar ratio of 2,5-dihydroxy-1,4-terephthalaldehyde, 1-bromohexane, and anhydrous potassium carbonate is: 1/2.5/2.5; in step 3), p-hydroxyphenylacetonitrile , The reaction molar ratio of compound B and K 2 CO 3 is: 1/1.2/1.5.
步骤5)中化合物A、化合物C和化合物D的反应摩尔比例为:1/1.1/1.2;步骤6)中化合物E和对甲苯磺酸的反应摩尔比例为:100/0.5。The reaction molar ratio of compound A, compound C and compound D in step 5) is: 1/1.1/1.2; the reaction molar ratio of compound E and p-toluenesulfonic acid in step 6) is: 100/0.5.
步骤8)中化合物F、化合物G、K2CO3的反应摩尔比例为:2/1/2.5。The reaction molar ratio of compound F, compound G and K 2 CO 3 in step 8) is: 2/1/2.5.
实施例1:力致变色高分子材料DPU-0.1的制备Example 1: Preparation of Mechanochromic Polymer Material DPU-0.1
制备路线如下:The preparation route is as follows:
将5mg DCOP、端羟基聚四氢呋喃(Mn=2000g/mol,3.20g,1.60mmol)和MDI(1.23g,4.90mmol)溶于30mL四氢呋喃中,向混合物中滴加二月桂酸二丁基锡(2滴),并在室温下搅拌3小时。然后再加入10mL溶有丁二醇(288mg,3.20mmol)的THF溶液,在室温下混合物搅拌反应24小时。随后再在反应混合物中加入5mL乙醇,搅拌30分钟后,将反应混合物倒入乙醇(200mL)中。过滤收集黄色沉淀。固体再从THF中析出两次,得到黄色固体DPU-0.1。Dissolve 5mg of DCOP, hydroxyl-terminated polytetrahydrofuran (Mn=2000g/mol, 3.20g, 1.60mmol) and MDI (1.23g, 4.90mmol) in 30mL of tetrahydrofuran, and add dibutyltin dilaurate (2 drops) dropwise to the mixture , and stirred at room temperature for 3 hours. Then 10 mL of THF solution dissolved in butanediol (288 mg, 3.20 mmol) was added, and the mixture was stirred and reacted at room temperature for 24 hours. Then 5 mL of ethanol was added to the reaction mixture, and after stirring for 30 minutes, the reaction mixture was poured into ethanol (200 mL). The yellow precipitate was collected by filtration. The solid was precipitated from THF two more times to obtain DPU-0.1 as a yellow solid.
实施例2:力致变色高分子材料DPU-0.2的制备Example 2: Preparation of Mechanochromic Polymer Material DPU-0.2
具体的制备步骤如下:其与实施例1的相同部分不再赘述,与实施例1的不同在于,DCOP的添加量为10mg。The specific preparation steps are as follows: the same parts as in Example 1 will not be repeated, and the difference from Example 1 is that the amount of DCOP added is 10 mg.
实施例3:力致变色高分子材料DPU-0.4的制备Example 3: Preparation of Mechanochromic Polymer DPU-0.4
具体的制备步骤如下:其与实施例1的相同部分不再赘述,与实施例1的不同在于,DCOP的添加量为20mg。The specific preparation steps are as follows: the same part as in Example 1 will not be repeated, and the difference from Example 1 is that the amount of DCOP added is 20 mg.
对照例1:对照高分子材料MPU-0.2的制备Comparative example 1: Preparation of the reference polymer material MPU-0.2
制备路线如下:The preparation route is as follows:
将10mg MCOP、端羟基聚四氢呋喃(Mn=2000g/mol,3.20g,1.60mmol)和MDI(1.23g,4.90mmol)溶于30mL四氢呋喃中,向混合物中滴加二月桂酸二丁基锡(2滴),并在室温下搅拌3小时。然后再加入10mL溶有丁二醇(288mg,3.20mmol)的THF溶液,在室温下混合物搅拌反应24小时。随后再在反应混合物中加入5mL乙醇,搅拌30分钟后,将反应混合物倒入乙醇(200mL)中。过滤收集黄色沉淀。固体再从THF中析出两次,得到黄色固体MPU-0.2。Dissolve 10mg of MCOP, hydroxyl-terminated polytetrahydrofuran (Mn=2000g/mol, 3.20g, 1.60mmol) and MDI (1.23g, 4.90mmol) in 30mL of tetrahydrofuran, and add dibutyltin dilaurate (2 drops) dropwise to the mixture , and stirred at room temperature for 3 hours. Then 10 mL of THF solution dissolved in butanediol (288 mg, 3.20 mmol) was added, and the mixture was stirred and reacted at room temperature for 24 hours. Then 5 mL of ethanol was added to the reaction mixture, and after stirring for 30 minutes, the reaction mixture was poured into ethanol (200 mL). The yellow precipitate was collected by filtration. The solid was precipitated from THF two more times to afford MPU-0.2 as a yellow solid.
对照例2:对照高分子材料MPU-0.4的制备Comparative example 2: Preparation of the reference polymer material MPU-0.4
具体的制备步骤如下:其与对照例1的相同部分不再赘述,与对照例1的不同在于,MCOP的添加量为20mg。The specific preparation steps are as follows: the same part as Comparative Example 1 will not be repeated, and the difference from Comparative Example 1 is that the amount of MCOP added is 20 mg.
以下,将结合部分实施例对本发明的基于折叠-解折叠效应的力致变色高分子材料及其制备方法进一步说明。Hereinafter, the mechanochromic polymer material based on the folding-unfolding effect and the preparation method thereof of the present invention will be further described with reference to some examples.
首先,通过紫外-可见吸收光谱和荧光光谱研究了MCOP和DCOP(c=5um/L)在THF溶液中的光学性质(图1)。在吸收光谱中,MCOP表现出S0-S1跃迁振动带,在~365和431nm处有两个吸收峰,分别属于A0-1和A0-0振动带(图1中的(a));在发射光谱中~505nm处有明显的发射带,并发出明显的亮绿色荧光(图1中的(b))。DCOP的最大振动带(~366,431nm)与MCOP的波长相近,但A0-1的振动带要强于MCOP。这表明,DCOP中的COP部分在基态具有一定程度的相互作用,有利于激基缔合物的形成。此外,DCOP在~550nm处的发射带更强,进一步验证了激基缔合物的形成(图1中的(b))。A0-1和A0-0振动带的强度比(A0-1/A0-0)可以用来估计MCOP或DCOP中COP基团分子间或分子内相互作用的强度,A0-1/A0-0值越高,说明COP基团间或分子内的非共价相互作用越强。由吸收光谱计算,MCOP和DCOP的A0-1/A0-0值分别为0.66和0.75。First, the optical properties of MCOP and DCOP (c = 5um/L) in THF solution were studied by UV-Vis absorption and fluorescence spectroscopy (Fig. 1). In the absorption spectrum, MCOP exhibits the S 0 -S 1 transition vibration band, and there are two absorption peaks at ~365 and 431 nm, which belong to the A 0-1 and A 0-0 vibration bands (Fig. 1(a) ); there is an obvious emission band at ~505nm in the emission spectrum, and it emits obvious bright green fluorescence ((b) in Figure 1). The maximum vibration band of DCOP (~366,431nm) is similar to the wavelength of MCOP, but the vibration band of A 0-1 is stronger than that of MCOP. This suggests that the COP moiety in DCOP has a certain degree of interaction in the ground state, which is beneficial to the formation of excimates. In addition, the stronger emission band of DCOP at ∼550 nm further validates the formation of excimates ((b) in Figure 1). The intensity ratio of A 0-1 and A 0-0 vibrational bands (A 0-1 /A 0-0 ) can be used to estimate the strength of the intermolecular or intramolecular interaction of the COP group in MCOP or DCOP, A 0-1 / The higher the A 0-0 value, the stronger the non-covalent interaction between COP groups or within the molecule. Calculated from the absorption spectrum, the A 0-1 /A 0-0 values of MCOP and DCOP were 0.66 and 0.75, respectively.
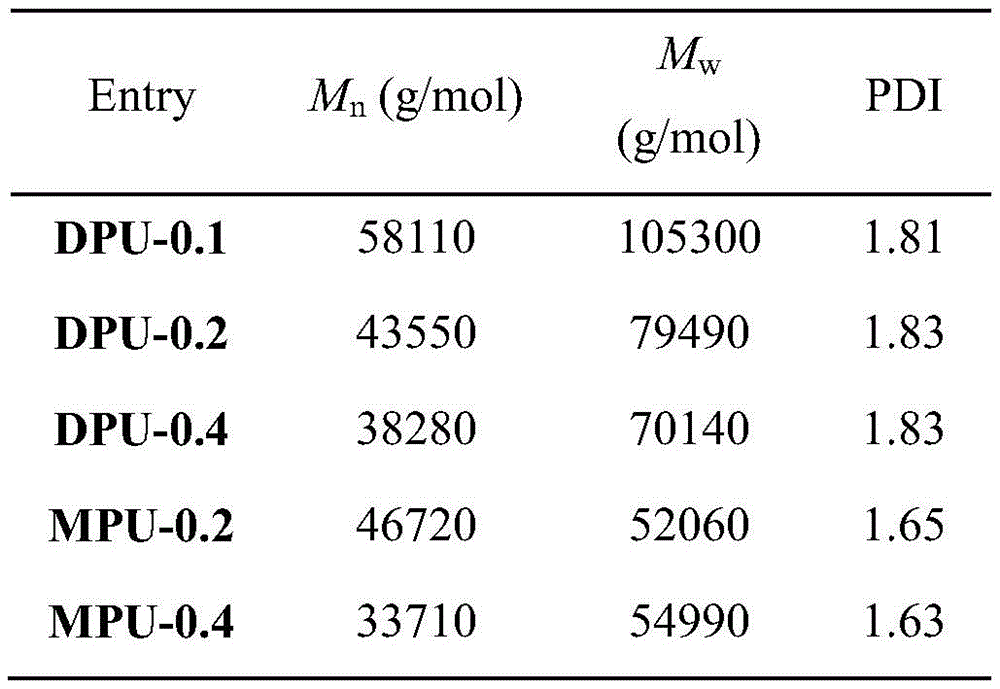
随后由聚四氢呋喃、亚甲基二苯基二异氰酸酯(MDI)、MCOP或DCOP之间的聚加成反应制备了DPU-m和MPU-n,即含有DCOP和MCOP基团的聚氨酯力致变色高分子材料,其中m和n分别表示DCOP和MCOP在聚氨酯中的质量百分比。合成的所有聚氨酯的数均分子量在31000到58100g/mol之间,聚合度在1.6到2之间,这与通过聚合反应得到的聚合物的预期结果一致(表1)。通过将相应浓度聚合物的THF溶液直接涂覆在聚四氟乙烯膜上,溶剂蒸发之后得到DPU-0.4、DPU-0.2、DPU-0.1、MPU-0.4和MPU-0.2薄膜。结果表明DPU和MPU薄膜的光学性质与染料含量无关(较低浓度下),因此我们以DPU-0.2和MPU-0.2为例,进一步证实DCOP镊子分子对于材料力致变色性能的决定性作用。(图2中的(a))显示DPU-0.2和MPU-0.2薄膜在可见光下分别呈现黄色、黄绿色,在365nm UV下分别呈现绿色、黄色荧光。薄膜的荧光寿命测量结果显示(图2中的(b),图2中的(c)),MPU-0.2薄膜的衰减曲线呈现单指数函数,验证了只有一个寿命为3.2ns的物种存在,该物种可能属于COP基团的单分散状态。不同的是,DPU-0.2膜的衰减曲线呈现出双指数函数,表明存在两个不同的物种,寿命分别为2.8ns和14.2ns,认为这两个物种与单分散COP(2.8ns)和弱偶联激基缔合物(14.2ns)有关。这一结论说明,DPU-0.2薄膜中COP基团之间形成了二聚体激基缔合物。DPU-m and MPU-n, that is, polyurethane mechanochromic high Molecular materials, where m and n represent the mass percentages of DCOP and MCOP in polyurethane, respectively. All polyurethanes synthesized had number-average molecular weights between 31,000 and 58,100 g/mol and a degree of polymerization between 1.6 and 2, which is consistent with the expected results for polymers obtained by polymerization (Table 1). DPU-0.4, DPU-0.2, DPU-0.1, MPU-0.4 and MPU-0.2 films were obtained by directly coating the THF solution of the corresponding concentration polymer on the polytetrafluoroethylene film, and the solvent was evaporated. The results show that the optical properties of DPU and MPU films have nothing to do with the dye content (lower concentration), so we take DPU-0.2 and MPU-0.2 as examples to further confirm the decisive role of DCOP tweezers on the mechanochromic properties of materials. ((a) in Figure 2) shows that the DPU-0.2 and MPU-0.2 films exhibit yellow and yellow-green fluorescence under visible light, respectively, and green and yellow fluorescence under 365nm UV. The fluorescence lifetime measurement results of the film show ((b) in Figure 2, (c) in Figure 2), the decay curve of the MPU-0.2 film presents a single exponential function, which verifies that only one species with a lifetime of 3.2ns exists, the Species may belong to a monodisperse state of COP groups. The difference is that the decay curve of the DPU-0.2 film presents a double-exponential function, indicating the existence of two different species with lifetimes of 2.8ns and 14.2ns, which are considered to be related to monodisperse COP (2.8ns) and weakly coupled Associated bikinyl associations (14.2 ns). This conclusion indicates that dimer excimer associations are formed between the COP groups in the DPU-0.2 film.
在对MCOP、DCOP及其聚合物薄膜的光物理性质有了一定了解后,我们进一步研究了MPU-0.2和DPU-0.2薄膜的力致变色性质。初始DPU-0.2薄膜表现出典型的橡胶聚合物的应力-应变行为。在UV照射下(λ=365nm)进行单轴拉伸时,由于COP镊子的展开,检测到由黄色到绿色的荧光变化,当外力放松时,绿色荧光可以恢复到原始状态,这个过程可以反复多次。然而,MPU-0.2薄膜在拉伸过程中没有出现类似的现象,当应变达到300%时,薄膜的荧光仍保持绿色。利用光纤将荧光信号传输到探测器,分析薄膜在原位单轴拉伸过程中的荧光响应。随着应变的不断增大,DPU-0.2的荧光光谱出现了明显的蓝移,550nm处的发射带强度逐渐减弱,510nm处的发射带强度逐渐增强(图3)。发现在此过程中,I550/I510值与应变近似成线性关系,说明应力被有效地转移到了COP二聚体力学团上。应力松弛后,荧光光谱恢复到初始状态,这表明COD的折叠展开在机理上是可逆的。这些结果表明,DCOP荧光团中的连接单元有利于激基缔合物的形成,最终实现折叠-展开行为。After understanding the photophysical properties of MCOP, DCOP and their polymer films, we further studied the mechanochromic properties of MPU-0.2 and DPU-0.2 films. The pristine DPU-0.2 films exhibited the typical stress-strain behavior of rubbery polymers. When uniaxially stretched under UV irradiation (λ=365nm), due to the unfolding of the COP tweezers, a fluorescence change from yellow to green is detected. When the external force is relaxed, the green fluorescence can return to the original state. This process can be repeated many times. Second-rate. However, the MPU-0.2 film did not show a similar phenomenon during stretching, and the fluorescence of the film remained green when the strain reached 300%. The fluorescence signal is transmitted to the detector by optical fiber, and the fluorescence response of the film during in-situ uniaxial stretching is analyzed. With the continuous increase of the strain, the fluorescence spectrum of DPU-0.2 showed an obvious blue shift, the intensity of the emission band at 550nm gradually weakened, and the intensity of the emission band at 510nm gradually increased (Figure 3). It was found that during this process, the I 550 /I 510 value was approximately linear with the strain, indicating that the stress was effectively transferred to the COP dimer mechanogroup. After stress relaxation, the fluorescence spectrum returns to the initial state, which indicates that the folding and unfolding of COD is mechanistically reversible. These results suggest that the linker unit in the DCOP fluorophore facilitates the formation of excimers and ultimately enables the folding-unfolding behavior.
下表为聚合物DPU-m和MPU-n的数均分子量(Mn)、重均分子量(Mw)和多分散指数(PDI)。The following table shows the number average molecular weight (Mn), weight average molecular weight (Mw) and polydispersity index (PDI) of the polymers DPU-m and MPU-n.
表1Table 1
由此可以看出本发明提供的基于折叠-解折叠效应的聚氨酯高分子材料具有良好的可逆力致变色性能。该技术路线易于实现,丰富了分子镊子型力致变色材料,进一步推动了力致变色材料的发展。It can be seen that the polyurethane polymer material based on the folding-unfolding effect provided by the present invention has good reversible mechanochromic properties. This technical route is easy to realize, enriches the molecular tweezers type mechanochromic materials, and further promotes the development of mechanochromic materials.
以上所述,仅是本发明的较佳实施例,并非对本发明作任何形式上的限制,任何熟悉本专业的技术人员,在不脱离本发明技术方案范围内,依据本发明的技术实质,对以上实施例所作的任何简单的修改、等同替换与改进等,均仍属于本发明技术方案的保护范围之内。The above are only preferred embodiments of the present invention, and are not intended to limit the present invention in any form. Any skilled person who is familiar with the profession, without departing from the scope of the technical solutions of the present invention, according to the technical essence of the present invention, Any simple modifications, equivalent replacements and improvements made in the above embodiments still fall within the protection scope of the technical solution of the present invention.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202211156451.5A CN115449052B (en) | 2022-09-22 | 2022-09-22 | A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202211156451.5A CN115449052B (en) | 2022-09-22 | 2022-09-22 | A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN115449052A CN115449052A (en) | 2022-12-09 |
| CN115449052B true CN115449052B (en) | 2023-07-04 |
Family
ID=84307686
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202211156451.5A Active CN115449052B (en) | 2022-09-22 | 2022-09-22 | A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115449052B (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002023696A (en) * | 2000-04-18 | 2002-01-23 | Semiconductor Energy Lab Co Ltd | Light emitting device |
| JP2003264086A (en) * | 2002-03-08 | 2003-09-19 | Tdk Corp | Organic electroluminescence device |
| CN102977341A (en) * | 2012-11-15 | 2013-03-20 | 南京邮电大学 | Preparation method and application of multiblock conjugated polymer containing phenylene ethylene skeleton |
| CN109020805A (en) * | 2018-08-03 | 2018-12-18 | 北京师范大学 | Fluorescence discoloration eutectic material and preparation method thereof under a kind of ultra-high pressure condition |
| CN109651587A (en) * | 2018-12-07 | 2019-04-19 | 南京工程学院 | It is a kind of to derive material resources mutagens color high molecular material and preparation method thereof containing phenolphthalein with fast self-recovery property |
| CN112778178A (en) * | 2021-01-21 | 2021-05-11 | 贵州民族大学 | Fluorescent material with piezochromism and lyotropic chromotropic effect and preparation method thereof |
-
2022
- 2022-09-22 CN CN202211156451.5A patent/CN115449052B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002023696A (en) * | 2000-04-18 | 2002-01-23 | Semiconductor Energy Lab Co Ltd | Light emitting device |
| JP2003264086A (en) * | 2002-03-08 | 2003-09-19 | Tdk Corp | Organic electroluminescence device |
| CN102977341A (en) * | 2012-11-15 | 2013-03-20 | 南京邮电大学 | Preparation method and application of multiblock conjugated polymer containing phenylene ethylene skeleton |
| CN109020805A (en) * | 2018-08-03 | 2018-12-18 | 北京师范大学 | Fluorescence discoloration eutectic material and preparation method thereof under a kind of ultra-high pressure condition |
| CN109651587A (en) * | 2018-12-07 | 2019-04-19 | 南京工程学院 | It is a kind of to derive material resources mutagens color high molecular material and preparation method thereof containing phenolphthalein with fast self-recovery property |
| CN112778178A (en) * | 2021-01-21 | 2021-05-11 | 贵州民族大学 | Fluorescent material with piezochromism and lyotropic chromotropic effect and preparation method thereof |
Non-Patent Citations (4)
| Title |
|---|
| Self-Assembly of Diacetylene-Bridged Phenylenevinylene Oligomers in Water and Organic Solvents;Dr. Miguel García-Iglesias,等;《Chem. Plus. Chem.》;第84卷(第5期);第488-492页 * |
| Sequential Energy and Electron Transfer in Aggregates of Tetrakis[oligo(p-phenylene vinylene)] Porphyrins and C60 in Water;Martin Wolffs,等;《Journal of the American Chemical Society》;第127卷(第39期);第13484–13485页 * |
| 多功能荧光化学传感器、荧光力传感器的设计合成;汪太生;《中国博士学位论文全文数据库工程科技Ⅰ辑》(第11期);第B014-232页 * |
| 对苯撑乙烯撑寡聚物-b-聚乙二醇的合成及结晶驱动自组装行为;陶大燎,等;《功能高分子学报》;第32卷(第5期);第617-625页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN115449052A (en) | 2022-12-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Zhao et al. | Self-healing metallo-supramolecular polymers showing luminescence off/on switching based on lanthanide ions and terpyridine moieties | |
| JP6444429B2 (en) | Mechanochromic luminescent material, mechanochromic resin crosslinked with the mechanochromic luminescent material, method for producing mechanochromic luminescent material, and method for producing mechanochromic resin | |
| Liu et al. | Novel triphenylamine polyamides bearing carbazole and aniline substituents for multi-colored electrochromic applications | |
| Meng et al. | Optically transparent polyamides bearing phenoxyl, diphenylamine and fluorene units with high-contrast of electrochromic and electrofluorescent behaviors | |
| Zhang et al. | 1, 4-Diketo-pyrrolo [3, 4-c] pyrroles (DPPs) based insoluble polymer films with lactam hydrogens as renewable fluoride anion chemosensor | |
| Ji et al. | A fluorescent supramolecular crosslinked polymer gel formed by crown ether based host-guest interactions and aggregation induced emission | |
| Wang et al. | Waterborne polyurethanes prepared from benzophenone derivatives with delayed fluorescence and room-temperature phosphorescence | |
| CN117285689B (en) | Self-repairing type light-response polyurea material and preparation method thereof | |
| CN110903202B (en) | A kind of diamine monomer and its preparation method, a kind of polyamide and its preparation method and application | |
| WO2015128884A1 (en) | Poly-benzimidazole with pyrene and anthracene flourophore | |
| CN115449052B (en) | A kind of mechanochromic polymer material based on folding-unfolding effect and its preparation method | |
| Zhang et al. | Incorporation of chromophores into dendrigraft polybutadiene: effect of dendrigraft matrix on the fluorescent properties | |
| US12338328B2 (en) | Reversible stress-responsive material, preparation method, and use thereof | |
| Liou et al. | Synthesis and photoluminescence properties of novel polyarylates bearing pendent naphthylamine chromophores | |
| CN118388953B (en) | Diphenylamine molecule and polyurethane composite material with force stimulus response phosphorescence function, and preparation method and application thereof | |
| Anannarukan et al. | Soluble polyimides containing trans-diaminotetraphenylporphyrin: Synthesis and photoinduced electron transfer | |
| Li et al. | A photoluminescent elastomer with high toughness and fast self-healing behavior enabled by crosslinking polyurethane with reactive lanthanide complex | |
| TWI852767B (en) | Polyurethane elastomer materials containing polyrotaxane tougheners with mechanochromic fluorescence properties | |
| Yamauchi et al. | Synthesis of acrylonitrile side chain-appended π-conjugated polymers by a Suzuki cross-coupling polycondensation and a Knoevenagel condensation, and their optical properties | |
| JP5629873B2 (en) | Light emitting material and light emitting element | |
| Sivakumar et al. | Hydroxyl‐and amine‐terminated hyperbranched polyurethanes using AB2‐type azide monomers: Synthesis, characterization, fluorescence, and charge‐transfer complexation studies | |
| CN108863823B (en) | Diphenol monomer containing asymmetric fluorophore structure and preparation method and application thereof | |
| Zhang et al. | Mechanochromism of polyurethane based on folding—unfolding of cyano-substituted oligo (p-phenylene) vinylene dimer | |
| CN118388954B (en) | Fused ring compound and polyurethane composite material with mechanical force reversible response phosphorescence function, and preparation method and application thereof | |
| CN107629184A (en) | A kind of S dinaphthols polyurethane and preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |