CN110840892A - 酪氨酸激酶抑制剂与cdk4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途 - Google Patents
酪氨酸激酶抑制剂与cdk4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途 Download PDFInfo
- Publication number
- CN110840892A CN110840892A CN201910766740.9A CN201910766740A CN110840892A CN 110840892 A CN110840892 A CN 110840892A CN 201910766740 A CN201910766740 A CN 201910766740A CN 110840892 A CN110840892 A CN 110840892A
- Authority
- CN
- China
- Prior art keywords
- formula
- tumor
- compound
- inhibitor
- cancer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 229940124297 CDK 4/6 inhibitor Drugs 0.000 title claims abstract description 42
- 239000003814 drug Substances 0.000 title claims abstract description 26
- 238000011282 treatment Methods 0.000 title claims abstract description 18
- 201000010099 disease Diseases 0.000 title claims abstract description 11
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 title claims abstract description 11
- 238000002360 preparation method Methods 0.000 title claims abstract description 11
- 230000001613 neoplastic effect Effects 0.000 title claims abstract description 9
- 230000002265 prevention Effects 0.000 title claims abstract description 5
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 title abstract description 15
- 239000005483 tyrosine kinase inhibitor Substances 0.000 title abstract description 15
- 150000004917 tyrosine kinase inhibitor derivatives Chemical class 0.000 title abstract description 12
- 150000001875 compounds Chemical class 0.000 claims abstract description 86
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 46
- 150000003839 salts Chemical class 0.000 claims abstract description 34
- -1 tyrosine kinase inhibitor compound Chemical class 0.000 claims abstract description 6
- 229940125497 HER2 kinase inhibitor Drugs 0.000 claims description 13
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 13
- 206010017758 gastric cancer Diseases 0.000 claims description 13
- 201000011549 stomach cancer Diseases 0.000 claims description 13
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 claims description 10
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 claims description 10
- 206010006187 Breast cancer Diseases 0.000 claims description 9
- 208000026310 Breast neoplasm Diseases 0.000 claims description 9
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 claims description 9
- 239000002136 L01XE07 - Lapatinib Substances 0.000 claims description 7
- 229950001573 abemaciclib Drugs 0.000 claims description 7
- 229960004891 lapatinib Drugs 0.000 claims description 7
- BCFGMOOMADDAQU-UHFFFAOYSA-N lapatinib Chemical compound O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 BCFGMOOMADDAQU-UHFFFAOYSA-N 0.000 claims description 7
- UZWDCWONPYILKI-UHFFFAOYSA-N n-[5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl]-5-fluoro-4-(7-fluoro-2-methyl-3-propan-2-ylbenzimidazol-5-yl)pyrimidin-2-amine Chemical group C1CN(CC)CCN1CC(C=N1)=CC=C1NC1=NC=C(F)C(C=2C=C3N(C(C)C)C(C)=NC3=C(F)C=2)=N1 UZWDCWONPYILKI-UHFFFAOYSA-N 0.000 claims description 7
- 201000011510 cancer Diseases 0.000 claims description 6
- RHXHGRAEPCAFML-UHFFFAOYSA-N 7-cyclopentyl-n,n-dimethyl-2-[(5-piperazin-1-ylpyridin-2-yl)amino]pyrrolo[2,3-d]pyrimidine-6-carboxamide Chemical compound N1=C2N(C3CCCC3)C(C(=O)N(C)C)=CC2=CN=C1NC(N=C1)=CC=C1N1CCNCC1 RHXHGRAEPCAFML-UHFFFAOYSA-N 0.000 claims description 5
- 206010073095 invasive ductal breast carcinoma Diseases 0.000 claims description 5
- 201000010985 invasive ductal carcinoma Diseases 0.000 claims description 5
- 229960004390 palbociclib Drugs 0.000 claims description 5
- AHJRHEGDXFFMBM-UHFFFAOYSA-N palbociclib Chemical compound N1=C2N(C3CCCC3)C(=O)C(C(=O)C)=C(C)C2=CN=C1NC(N=C1)=CC=C1N1CCNCC1 AHJRHEGDXFFMBM-UHFFFAOYSA-N 0.000 claims description 5
- 239000008194 pharmaceutical composition Substances 0.000 claims description 5
- 229950003687 ribociclib Drugs 0.000 claims description 5
- 206010025323 Lymphomas Diseases 0.000 claims description 4
- 229950010817 alvocidib Drugs 0.000 claims description 4
- BIIVYFLTOXDAOV-YVEFUNNKSA-N alvocidib Chemical compound O[C@@H]1CN(C)CC[C@@H]1C1=C(O)C=C(O)C2=C1OC(C=1C(=CC=CC=1)Cl)=CC2=O BIIVYFLTOXDAOV-YVEFUNNKSA-N 0.000 claims description 4
- 206010073096 invasive lobular breast carcinoma Diseases 0.000 claims description 4
- 239000005461 Canertinib Substances 0.000 claims description 3
- 210000000481 breast Anatomy 0.000 claims description 3
- 229950002826 canertinib Drugs 0.000 claims description 3
- OMZCMEYTWSXEPZ-UHFFFAOYSA-N canertinib Chemical compound C1=C(Cl)C(F)=CC=C1NC1=NC=NC2=CC(OCCCN3CCOCC3)=C(NC(=O)C=C)C=C12 OMZCMEYTWSXEPZ-UHFFFAOYSA-N 0.000 claims description 3
- KFAKESMKRPNZTM-UHFFFAOYSA-N 1,4-dimethoxy-10H-acridine-9-thione Chemical compound N1C2=CC=CC=C2C(=S)C2=C1C(OC)=CC=C2OC KFAKESMKRPNZTM-UHFFFAOYSA-N 0.000 claims description 2
- OVPNQJVDAFNBDN-UHFFFAOYSA-N 4-(2,6-dichlorobenzamido)-N-(piperidin-4-yl)-pyrazole-3-carboxamide Chemical compound ClC1=CC=CC(Cl)=C1C(=O)NC1=CNN=C1C(=O)NC1CCNCC1 OVPNQJVDAFNBDN-UHFFFAOYSA-N 0.000 claims description 2
- YVXCDLCJCIDFHE-UHFFFAOYSA-N 4-[[6-(ethylamino)-2-[[1-(phenylmethyl)-5-indolyl]amino]-4-pyrimidinyl]amino]-1-cyclohexanol Chemical compound N=1C(NC=2C=C3C=CN(CC=4C=CC=CC=4)C3=CC=2)=NC(NCC)=CC=1NC1CCC(O)CC1 YVXCDLCJCIDFHE-UHFFFAOYSA-N 0.000 claims description 2
- PETCVZZPKYJZAU-UHFFFAOYSA-N 8-(3-bicyclo[2.2.1]heptanyl)-2-[4-[4-(3-hydroxypropyl)piperidin-1-yl]anilino]pyrido[2,3-d]pyrimidin-7-one Chemical compound C1CC(CCCO)CCN1C(C=C1)=CC=C1NC1=NC=C(C=CC(=O)N2C3C4CCC(C4)C3)C2=N1 PETCVZZPKYJZAU-UHFFFAOYSA-N 0.000 claims description 2
- BBUVDDPUURMFOX-SAABIXHNSA-N AMG-925 Chemical compound C1C[C@@H](C)CC[C@@H]1N1C2=NC(NC=3N=C4CCN(CC4=CC=3)C(=O)CO)=NC=C2C2=CC=NC=C21 BBUVDDPUURMFOX-SAABIXHNSA-N 0.000 claims description 2
- 206010004593 Bile duct cancer Diseases 0.000 claims description 2
- 208000003174 Brain Neoplasms Diseases 0.000 claims description 2
- 201000009030 Carcinoma Diseases 0.000 claims description 2
- 206010009944 Colon cancer Diseases 0.000 claims description 2
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 2
- 208000000461 Esophageal Neoplasms Diseases 0.000 claims description 2
- 208000007842 Fibroepithelial Neoplasms Diseases 0.000 claims description 2
- 208000022072 Gallbladder Neoplasms Diseases 0.000 claims description 2
- SJRXVLUZMMDCNG-UHFFFAOYSA-N Gossypin Natural products OC1C(O)C(O)C(CO)OC1OC1=C(O)C=C(O)C2=C1OC(C=1C=C(O)C(O)=CC=1)=C(O)C2=O SJRXVLUZMMDCNG-UHFFFAOYSA-N 0.000 claims description 2
- 208000037396 Intraductal Noninfiltrating Carcinoma Diseases 0.000 claims description 2
- 208000008839 Kidney Neoplasms Diseases 0.000 claims description 2
- 206010073099 Lobular breast carcinoma in situ Diseases 0.000 claims description 2
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 2
- 208000004059 Male Breast Neoplasms Diseases 0.000 claims description 2
- 208000007054 Medullary Carcinoma Diseases 0.000 claims description 2
- 208000034578 Multiple myelomas Diseases 0.000 claims description 2
- 208000009287 Myoepithelioma Diseases 0.000 claims description 2
- 206010029260 Neuroblastoma Diseases 0.000 claims description 2
- 206010030155 Oesophageal carcinoma Diseases 0.000 claims description 2
- 208000000035 Osteochondroma Diseases 0.000 claims description 2
- 206010033128 Ovarian cancer Diseases 0.000 claims description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 2
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 2
- 206010060862 Prostate cancer Diseases 0.000 claims description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 2
- 206010038389 Renal cancer Diseases 0.000 claims description 2
- 206010039491 Sarcoma Diseases 0.000 claims description 2
- 201000010208 Seminoma Diseases 0.000 claims description 2
- 208000000453 Skin Neoplasms Diseases 0.000 claims description 2
- 208000024313 Testicular Neoplasms Diseases 0.000 claims description 2
- 208000024770 Thyroid neoplasm Diseases 0.000 claims description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 2
- 208000002495 Uterine Neoplasms Diseases 0.000 claims description 2
- 208000017733 acquired polycythemia vera Diseases 0.000 claims description 2
- 208000009956 adenocarcinoma Diseases 0.000 claims description 2
- 208000002517 adenoid cystic carcinoma Diseases 0.000 claims description 2
- 229960001686 afatinib Drugs 0.000 claims description 2
- ULXXDDBFHOBEHA-CWDCEQMOSA-N afatinib Chemical compound N1=CN=C2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC2=C1NC1=CC=C(F)C(Cl)=C1 ULXXDDBFHOBEHA-CWDCEQMOSA-N 0.000 claims description 2
- 208000026900 bile duct neoplasm Diseases 0.000 claims description 2
- 201000005389 breast carcinoma in situ Diseases 0.000 claims description 2
- 208000006990 cholangiocarcinoma Diseases 0.000 claims description 2
- 239000003085 diluting agent Substances 0.000 claims description 2
- 208000028715 ductal breast carcinoma in situ Diseases 0.000 claims description 2
- 210000001951 dura mater Anatomy 0.000 claims description 2
- HKSZLNNOFSGOKW-UHFFFAOYSA-N ent-staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(C)O1 HKSZLNNOFSGOKW-UHFFFAOYSA-N 0.000 claims description 2
- 201000004101 esophageal cancer Diseases 0.000 claims description 2
- 201000010175 gallbladder cancer Diseases 0.000 claims description 2
- 208000005017 glioblastoma Diseases 0.000 claims description 2
- SJRXVLUZMMDCNG-KKPQBLLMSA-N gossypin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=C(O)C=C(O)C2=C1OC(C=1C=C(O)C(O)=CC=1)=C(O)C2=O SJRXVLUZMMDCNG-KKPQBLLMSA-N 0.000 claims description 2
- 208000014829 head and neck neoplasm Diseases 0.000 claims description 2
- 201000010982 kidney cancer Diseases 0.000 claims description 2
- YPJRHEKCFKOVRT-UHFFFAOYSA-N lerociclib Chemical compound C1CN(C(C)C)CCN1C(C=N1)=CC=C1NC1=NC=C(C=C2N3C4(CCCCC4)CNC2=O)C3=N1 YPJRHEKCFKOVRT-UHFFFAOYSA-N 0.000 claims description 2
- 208000032839 leukemia Diseases 0.000 claims description 2
- 206010024627 liposarcoma Diseases 0.000 claims description 2
- 201000007270 liver cancer Diseases 0.000 claims description 2
- 208000014018 liver neoplasm Diseases 0.000 claims description 2
- 201000011059 lobular neoplasia Diseases 0.000 claims description 2
- 201000005202 lung cancer Diseases 0.000 claims description 2
- 208000020816 lung neoplasm Diseases 0.000 claims description 2
- 150000002688 maleic acid derivatives Chemical class 0.000 claims description 2
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 2
- 208000023356 medullary thyroid gland carcinoma Diseases 0.000 claims description 2
- 201000001441 melanoma Diseases 0.000 claims description 2
- 201000010879 mucinous adenocarcinoma Diseases 0.000 claims description 2
- 208000026226 myoepithelial tumor Diseases 0.000 claims description 2
- RBOKLZGCVRXGEP-XTQSDGFTSA-N n-[[5-[(3e)-3-(4,6-difluorobenzimidazol-2-ylidene)-1,2-dihydroindazol-5-yl]-4-methylpyridin-3-yl]methyl]ethanamine Chemical compound CCNCC1=CN=CC(C=2C=C3C(=C/4N=C5C(F)=CC(F)=CC5=N\4)/NNC3=CC=2)=C1C RBOKLZGCVRXGEP-XTQSDGFTSA-N 0.000 claims description 2
- 208000025189 neoplasm of testis Diseases 0.000 claims description 2
- 208000008798 osteoma Diseases 0.000 claims description 2
- 201000008968 osteosarcoma Diseases 0.000 claims description 2
- 201000002528 pancreatic cancer Diseases 0.000 claims description 2
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 2
- 201000010198 papillary carcinoma Diseases 0.000 claims description 2
- 229960002087 pertuzumab Drugs 0.000 claims description 2
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 2
- 208000037244 polycythemia vera Diseases 0.000 claims description 2
- 201000007416 salivary gland adenoid cystic carcinoma Diseases 0.000 claims description 2
- 201000000849 skin cancer Diseases 0.000 claims description 2
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 2
- HKSZLNNOFSGOKW-FYTWVXJKSA-N staurosporine Chemical compound C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1[C@H]1C[C@@H](NC)[C@@H](OC)[C@]4(C)O1 HKSZLNNOFSGOKW-FYTWVXJKSA-N 0.000 claims description 2
- CGPUWJWCVCFERF-UHFFFAOYSA-N staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(OC)O1 CGPUWJWCVCFERF-UHFFFAOYSA-N 0.000 claims description 2
- 201000003120 testicular cancer Diseases 0.000 claims description 2
- 208000013076 thyroid tumor Diseases 0.000 claims description 2
- 229960000575 trastuzumab Drugs 0.000 claims description 2
- 201000007423 tubular adenocarcinoma Diseases 0.000 claims description 2
- 208000026517 ureter neoplasm Diseases 0.000 claims description 2
- 201000011476 ureteral benign neoplasm Diseases 0.000 claims description 2
- 206010046766 uterine cancer Diseases 0.000 claims description 2
- MRPGRAKIAJJGMM-OCCSQVGLSA-N 2-[2-chloro-4-(trifluoromethyl)phenyl]-5,7-dihydroxy-8-[(2r,3s)-2-(hydroxymethyl)-1-methylpyrrolidin-3-yl]chromen-4-one Chemical compound OC[C@@H]1N(C)CC[C@H]1C1=C(O)C=C(O)C2=C1OC(C=1C(=CC(=CC=1)C(F)(F)F)Cl)=CC2=O MRPGRAKIAJJGMM-OCCSQVGLSA-N 0.000 claims 1
- 206010056286 Nipple neoplasm Diseases 0.000 claims 1
- 201000007436 apocrine adenocarcinoma Diseases 0.000 claims 1
- 239000000969 carrier Substances 0.000 claims 1
- 210000002445 nipple Anatomy 0.000 claims 1
- 229950003294 voruciclib Drugs 0.000 claims 1
- 210000001519 tissue Anatomy 0.000 description 19
- 229940079593 drug Drugs 0.000 description 14
- 238000000034 method Methods 0.000 description 13
- 206010059866 Drug resistance Diseases 0.000 description 7
- 241000699670 Mus sp. Species 0.000 description 7
- 230000022131 cell cycle Effects 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 238000003304 gavage Methods 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 230000003902 lesion Effects 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 101000715943 Caenorhabditis elegans Cyclin-dependent kinase 4 homolog Proteins 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 102000006311 Cyclin D1 Human genes 0.000 description 3
- 108010058546 Cyclin D1 Proteins 0.000 description 3
- 208000031448 Genomic Instability Diseases 0.000 description 3
- 241000699660 Mus musculus Species 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 238000010171 animal model Methods 0.000 description 3
- 239000002246 antineoplastic agent Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 238000011580 nude mouse model Methods 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- 238000011729 BALB/c nude mouse Methods 0.000 description 2
- 108091007914 CDKs Proteins 0.000 description 2
- 102000003910 Cyclin D Human genes 0.000 description 2
- 108090000259 Cyclin D Proteins 0.000 description 2
- 108010024986 Cyclin-Dependent Kinase 2 Proteins 0.000 description 2
- 102100036239 Cyclin-dependent kinase 2 Human genes 0.000 description 2
- 102000003903 Cyclin-dependent kinases Human genes 0.000 description 2
- 108090000266 Cyclin-dependent kinases Proteins 0.000 description 2
- 206010061818 Disease progression Diseases 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 108700020796 Oncogene Proteins 0.000 description 2
- 102000043276 Oncogene Human genes 0.000 description 2
- 102000052575 Proto-Oncogene Human genes 0.000 description 2
- 108700020978 Proto-Oncogene Proteins 0.000 description 2
- 101710124357 Retinoblastoma-associated protein Proteins 0.000 description 2
- 230000005856 abnormality Effects 0.000 description 2
- 229940041181 antineoplastic drug Drugs 0.000 description 2
- 238000009395 breeding Methods 0.000 description 2
- 230000001488 breeding effect Effects 0.000 description 2
- 238000011260 co-administration Methods 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- 230000005750 disease progression Effects 0.000 description 2
- 238000001647 drug administration Methods 0.000 description 2
- 230000000857 drug effect Effects 0.000 description 2
- 210000003236 esophagogastric junction Anatomy 0.000 description 2
- 238000005286 illumination Methods 0.000 description 2
- 238000011532 immunohistochemical staining Methods 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 238000011081 inoculation Methods 0.000 description 2
- 229950008835 neratinib Drugs 0.000 description 2
- ZNHPZUKZSNBOSQ-BQYQJAHWSA-N neratinib Chemical compound C=12C=C(NC\C=C\CN(C)C)C(OCC)=CC2=NC=C(C#N)C=1NC(C=C1Cl)=CC=C1OCC1=CC=CC=N1 ZNHPZUKZSNBOSQ-BQYQJAHWSA-N 0.000 description 2
- 238000007911 parenteral administration Methods 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000012163 sequencing technique Methods 0.000 description 2
- 102000034285 signal transducing proteins Human genes 0.000 description 2
- 108091006024 signal transducing proteins Proteins 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- LDMOEFOXLIZJOW-UHFFFAOYSA-N 1-dodecanesulfonic acid Chemical class CCCCCCCCCCCCS(O)(=O)=O LDMOEFOXLIZJOW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000321096 Adenoides Species 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 102000002554 Cyclin A Human genes 0.000 description 1
- 108010068192 Cyclin A Proteins 0.000 description 1
- 102000002427 Cyclin B Human genes 0.000 description 1
- 108010068150 Cyclin B Proteins 0.000 description 1
- 102000003909 Cyclin E Human genes 0.000 description 1
- 108090000257 Cyclin E Proteins 0.000 description 1
- 108010025464 Cyclin-Dependent Kinase 4 Proteins 0.000 description 1
- 108010025468 Cyclin-Dependent Kinase 6 Proteins 0.000 description 1
- 102100032857 Cyclin-dependent kinase 1 Human genes 0.000 description 1
- 101710106279 Cyclin-dependent kinase 1 Proteins 0.000 description 1
- 102100036329 Cyclin-dependent kinase 3 Human genes 0.000 description 1
- 102100036252 Cyclin-dependent kinase 4 Human genes 0.000 description 1
- 102100026804 Cyclin-dependent kinase 6 Human genes 0.000 description 1
- 102100026810 Cyclin-dependent kinase 7 Human genes 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 208000017891 HER2 positive breast carcinoma Diseases 0.000 description 1
- 238000011460 HER2-targeted therapy Methods 0.000 description 1
- 101000715946 Homo sapiens Cyclin-dependent kinase 3 Proteins 0.000 description 1
- 101000911952 Homo sapiens Cyclin-dependent kinase 7 Proteins 0.000 description 1
- 101000583175 Homo sapiens Prolactin-inducible protein Proteins 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 206010029113 Neovascularisation Diseases 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-L Phosphate ion(2-) Chemical compound OP([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-L 0.000 description 1
- 102100030350 Prolactin-inducible protein Human genes 0.000 description 1
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 1
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- 206010064390 Tumour invasion Diseases 0.000 description 1
- 210000002534 adenoid Anatomy 0.000 description 1
- 230000002927 anti-mitotic effect Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- 230000009400 cancer invasion Effects 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 230000006369 cell cycle progression Effects 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 229940043378 cyclin-dependent kinase inhibitor Drugs 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 230000003831 deregulation Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- POULHZVOKOAJMA-UHFFFAOYSA-M dodecanoate Chemical compound CCCCCCCCCCCC([O-])=O POULHZVOKOAJMA-UHFFFAOYSA-M 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 238000001861 endoscopic biopsy Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000010201 enrichment analysis Methods 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 229940049906 glutamate Drugs 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 1
- IPCSVZSSVZVIGE-UHFFFAOYSA-M hexadecanoate Chemical compound CCCCCCCCCCCCCCCC([O-])=O IPCSVZSSVZVIGE-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 238000007901 in situ hybridization Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 229940099584 lactobionate Drugs 0.000 description 1
- JYTUSYBCFIZPBE-AMTLMPIISA-N lactobionic acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O JYTUSYBCFIZPBE-AMTLMPIISA-N 0.000 description 1
- 229940070765 laurate Drugs 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-M mandelate Chemical compound [O-]C(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-M 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- 230000000394 mitotic effect Effects 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 229940049964 oleate Drugs 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 206010033675 panniculitis Diseases 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 102000027426 receptor tyrosine kinases Human genes 0.000 description 1
- 108091008598 receptor tyrosine kinases Proteins 0.000 description 1
- 230000022983 regulation of cell cycle Effects 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 1
- 229960001860 salicylate Drugs 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000010008 shearing Methods 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 210000004304 subcutaneous tissue Anatomy 0.000 description 1
- 229940086735 succinate Drugs 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 102000035160 transmembrane proteins Human genes 0.000 description 1
- 108091005703 transmembrane proteins Proteins 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 230000005747 tumor angiogenesis Effects 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 230000005760 tumorsuppression Effects 0.000 description 1
- 229940070710 valerate Drugs 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- WKOLLVMJNQIZCI-UHFFFAOYSA-M vanillate Chemical compound COC1=CC(C([O-])=O)=CC=C1O WKOLLVMJNQIZCI-UHFFFAOYSA-M 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/4709—Non-condensed quinolines and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/472—Non-condensed isoquinolines, e.g. papaverine
- A61K31/4725—Non-condensed isoquinolines, e.g. papaverine containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
本发明涉及酪氨酸激酶抑制剂与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途。具体而言,本发明涉及酪氨酸激酶抑制剂式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途。
Description
技术领域
本发明属于医药领域,涉及酪氨酸激酶抑制剂与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途。
背景技术
恶性肿瘤是危害人们生命健康的重大疾病。近年来,随着肿瘤生物学及相关学科的飞速发展,针对肿瘤细胞内异常信号系统靶点的特异性抗肿瘤药物是新药研发的焦点。同时,多种抗肿瘤药物联合用于治疗肿瘤疾病也是科学研究的热点。
受体酪氨酸激酶是一类参与信号转导的跨膜蛋白,在多种细胞中表达,调节细胞的生长、分化和新生血管生成。研究表明,超过50%的原癌基因和癌基因产物都具有酪氨酸激酶活性,它们的异常表达将导致肿瘤发生,另外还与肿瘤的侵袭和转移、肿瘤新生血管的生成、肿瘤的化疗抗性密切相关。目前已公开了一些酪氨酸激酶抑制剂,例如拉帕替尼(Lapatinib)、来那替尼(Neratinib)等。WO2011029265公开了一种有效的酪氨酸激酶抑制剂及其制备方法,其结构如式I所示,
该化合物具有明显的药效优势。CN102933574A中描述了该化合物的二马来酸盐形式,其具有改善的理化性质、药代动力学性质以及生物利用度。
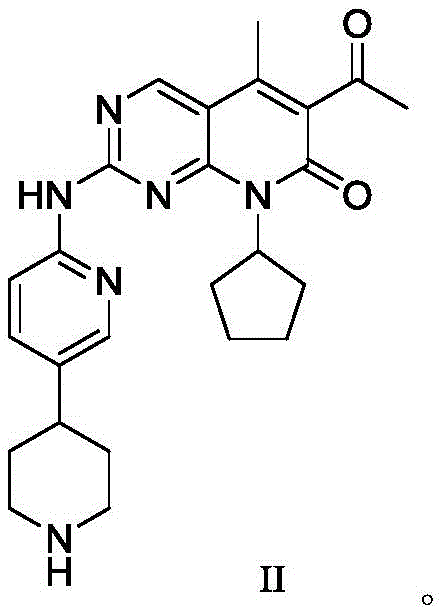
另外,已有大量研究发现肿瘤与细胞周期反常相关,大部分肿瘤都存在有丝分裂信号蛋白的大量突变/抗有丝分裂信号蛋白缺陷,基因组不稳定性(GIN)和染色体组不稳定性(CIN),这三种基本的细胞周期缺陷都直接或间接由细胞周期蛋白依赖性激酶(CDK)的失控引起。细胞周期蛋白Cyclin B/CDK1、Cyclin A/CDK2、Cyclin E/CDK2、Cyclin D/CDK4、Cyclin D/CDK6和其它杂二聚物(包括CDK3和CDK7)是细胞周期进展的重要调节剂。目前已公开了一些CDK抑制剂,其中CDK4/6抑制剂有abemaciclib、ribociclib、palbociclib等等。W02014183520提供了一种有效的CDK4/6抑制剂,其结构如式II所示,
WO2016124067公开了上述新的CDK4/6抑制剂的羟乙基磺酸盐。
现有技术公开了一些酪氨酸激酶抑制剂与CDK4/6抑制剂联合用于肿瘤疾病治疗的方案。Shom Goel等(Cancer Cell 29,255–269)公开了拉帕替尼和abemaciclib用于治疗拉帕替尼耐药的HER-2阳性乳腺癌细胞,结果显示两者联合治疗后能够有效的协同抑制乳腺癌细胞的活力。
本发明提供了一种新的酪氨酸激酶抑制剂与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途,并显示了良好的抑瘤效果。
发明内容
本发明提供了酪氨酸激酶抑制剂与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途,其中,所述酪氨酸激酶抑制剂选自式(I)所示化合物或其复合物或其可药用盐或其立体异构体,
在某些实施方式中,所述的CDK4/6抑制剂可以是abemaciclib、ribociclib、palbociclib、alvocidib、trilaciclib、voruciclib、AT-7519、G1T-38、FLX-925、INOC-005、G1T28-1、BPI-1178、gossypin、G1T30-1、GZ-38-1、P-276-00、staurosporine、R-547、PAN-1215、PD-0183812、AG-024322、NSC-625987、CGP-82996、PD-171851或式(II)所示化合物,优选abemaciclib、ribociclib、palbociclib、alvocidib或式(II)所示化合物,更优选式(II)所示化合物或其复合物或其可药用盐,
在某些实施方式中,所述肿瘤疾病可以是乳腺肿瘤、卵巢癌、前列腺癌、黑色素瘤、脑瘤、食管癌、胃癌、肝癌、胰腺癌、结肠直肠癌、肺癌、肾癌、皮肤癌、成胶质细胞瘤、神经母细胞瘤、肉瘤、脂肪肉瘤、骨软骨瘤、骨瘤、骨肉瘤、精原细胞瘤、睾丸肿瘤、子宫癌、头颈肿瘤、多发性骨髓瘤、恶性淋巴瘤、真性红细胞增多症、白血病、甲状腺肿瘤、输尿管肿瘤、膀胱肿瘤、胆囊癌、胆管癌或绒毛膜上皮癌。
在某些实施方式中,所述肿瘤疾病选自HER2阳性或HER2突变肿瘤。
在某些实施方式中,所述肿瘤疾病选自HER2阳性或HER2突变的乳腺癌或胃癌。
在某些实施方式中,所述的胃癌包括胃食管结合部癌。
在某些实施方式中,所述的胃癌为HER2抑制剂耐药的胃癌。在某些实施方式中,所述的HER2抑制剂选自曲妥珠单抗、帕妥珠单抗、拉帕替尼、阿法替尼、卡奈替尼(Canertinib)、来那替尼和式(I)所示化合物中的一种或几种,优选式(I)所示化合物。
在某些实施方式中,所述的乳腺肿瘤选自乳头肿瘤、男性乳腺肿瘤、乳腺恶性淋巴瘤、纤维上皮性肿瘤、上皮-肌上皮性肿瘤、导管内癌、小叶原位癌、乳头湿疹样乳腺癌、早期浸润性导管癌、早期浸润性小叶癌、乳头状癌、髓样癌、小管癌、腺样囊性癌、粘液腺癌、大汗腺样癌、鳞状细胞癌、浸润性小叶癌、浸润性导管癌、硬癌。
本发明优选的实施方式中,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂的重量比例选自0.01:1-100:1,优选1:0.1、1:0.125、1:0.14、1:0.15、1:0.175、1:0.1875、1:0.2、1:0.25、1:0.28、1:0.3、1:0.35、1:0.4、1:0.5、1:0.7、1:0.75、1:1、1:1.25、1:1.75、1:2、1:2.5、1:3.5、1:4、1:5、1:8、1:10、1:15、2:15、1:20、1:25、3:1、3:2、6:1、6:5、6:7、8:5、8:7、12:1、15:7、16:3、16:5、16:7、16:15、16:25、16:35、24:5、24:7、60:7。
本发明优选的另一个实施方式中,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的剂量范围选自100-1000mg;所述CDK4/6抑制剂的剂量范围选自1-1000mg。
在某些实施方案中,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的剂量选自100mg、125mg、150mg、175mg、200mg、250mg、300mg、350mg、400mg、450mg、500mg,600mg、700mg、750mg、800mg、900mg、1000mg。
本发明所述CDK4/6抑制剂的剂量选自5mg、10mg、15mg、20mg、25mg、30mg、35mg、40mg、45mg、50mg、55mg、60mg、65mg、70mg、75mg、80mg、85mg、90mg、100mg、125mg、150mg、175mg、200mg、250mg、300mg、350mg、400mg、450mg、500mg,600mg、700mg、750mg、800mg、900mg、1000mg。
本发明所述药物的可药用盐可以是盐酸盐、磷酸盐、磷酸氢盐、硫酸盐、硫酸氢盐、亚硫酸盐、乙酸盐、草酸盐、丙二酸盐、戊酸盐、谷氨酸盐、油酸盐、棕榈酸盐、硬脂酸盐、月桂酸盐、硼酸盐、对甲苯磺酸盐、甲磺酸盐、羟乙基磺酸盐、马来酸盐。苹果酸盐、酒石酸盐、苯甲酸盐、双羟萘酸盐、水杨酸盐、香草酸盐、扁桃酸盐、琥珀酸盐、葡萄糖酸盐、乳糖酸盐或月桂基磺酸盐等。
在某些实施方式中,所述式(I)所示化合物的可药用盐为马来酸盐,优选二马来酸盐。
在某些实施方式中,所述式(II)所示化合物的可药用盐为羟乙基磺酸盐。
本发明所述联合的给药方式选自同时给药、独立地配制并共给药或独立地配制并相继给药。
本发明所述联合的给药途径选自经口给药、胃肠外给药、经皮给药,所述胃肠外给药包括但不限于静脉注射、皮下注射、肌肉注射。
本发明进一步涉及式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途,其中式(I)所示化合物或其复合物或其可药用盐或其立体异构体的给药频次可以是一日一次、一日二次、一日三次、一周一次、二周一次、三周一次或一月一次,CDK4/6抑制剂的给药频次可以是一日一次、一日二次、一日三次、一周一次、二周一次、三周一次或一月一次。
在某些实施方式中,式(I)所示化合物或其复合物或其可药用盐或其立体异构体的给药频次是一日一次,CDK4/6抑制剂的给药频次是一日一次。
在某些实施方式中,给药周期是21天,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的给药频次为一日一次,连续给药,所述CDK4/6抑制剂的给药频次是一日一次,连续给药。
在某些实施方式中,给药周期是28天,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的给药频次为一日一次,连续给药四周,所述CDK4/6抑制剂的给药频次是一日一次,连续给药三周,停药一周。
在某些实施方式中,给药周期是28天,所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的给药频次为一日一次,连续给药四周,给药剂量为每天400mg或320mg,所述CDK4/6抑制剂的给药频次是一日一次,连续给药三周,停药一周,给药剂量为每天100mg、125mg、150mg或175mg。
本发明所述的方案中,所述的联合任选的还包含其他组分,所述其他组分包括但不限于其他抗肿瘤药等。
本发明还提供了式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途,其中,所述的CDK4/6抑制剂为式(II)所示化合物或其复合物或其可药用盐。
本发明还提供了一种治疗肿瘤疾病的方法,包括向患者施用本发明所述的式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂。
本发明还涉及一种包含式(I)所示化合物或其复合物或其可药用盐或其立体异构体、CDK4/6抑制剂以及一种或多种药用载体、赋形剂、稀释剂的药物组合物。所述药物组合物可以制成药学上可接受的任一剂型。例如,可以配制为片剂、胶囊剂、丸剂、颗粒剂、溶液剂、混悬剂、糖浆剂、注射剂(包括注射液、注射用无菌粉末与注射用浓溶液)、栓剂、吸入剂或喷雾剂。
本发明所述的含式(I)所示化合物或其复合物或其可药用盐或其立体异构体以及CDK4/6抑制剂的药物组合物,可以单独给药,或者与一种或多种治疗剂联合使用。
待组合的各成分(例如,式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂,式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂与其他任意组分药物)可同时给药或依次顺序地分开用药。此外,待组合的各成分还可以以同一制剂形式或以分开的不同制剂的形式联合给药。
本发明还提供了一种药物包装盒,其中包装有本发明所述的酪氨酸激酶抑制剂与CDK4/6抑制剂的药物组合物,其中酪氨酸激酶抑制剂选自式(I)所示化合物或其复合物或其可药用盐或其立体异构体。
本发明所述的“联合”是一种给药方式,是指一定时间期限内给予至少一种剂量的式(I)所示化合物以及至少一种剂量的CDK4/6抑制剂,其中给予的药物都显示药理学作用。所述的时间期限可以是一个给药周期内,优选4周内,3周内,2周内,1周内,或24小时以内,更优选12小时以内。可以同时或依次给予式(I)所示化合物以及CDK4/6抑制剂。这种期限包括这样的治疗,其中通过相同给药途径或不同给药途径给予式(I)所示化合物以及CDK4/6抑制剂。本发明所述联合的给药方式选自同时给药、独立地配制并共给药或独立地配制并相继给药。
本发明将式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂联合给药,从而增强了抗肿瘤活性,以及改善了肿瘤疾病的治疗效果。
附图说明
图1为HER2抑制剂继发耐药胃癌PDX模型的鉴定。
图2为耐药前后PDX组织的病理形态学一致性鉴定。
图3为western blot方法检测耐药前后组织中细胞周期几个关键分子的表达变化。
图4为耐药PDX模型中运用单药或联合用药后的肿瘤生长曲线(A),以及westernblot方法检测用药前后细胞周期几个关键分子的表达变化(B)。
具体实施方式
实施例1:式(I)所示化合物和式(II)所示化合物联合给药与各自单独给药对乳腺癌PDX(ER-,HER2+)BALB/c裸小鼠的药效作用研究。
1.受试药物
药物名称:式(I)所示化合物的二马来酸盐,根据CN102933574A公开的方法制备;式(II)所示化合物的羟乙基磺酸盐,根据WO2016124067公开的方法制备。
配制方法:式(I)所示化合物的二马来酸盐用蒸馏水配制;式(II)所示化合物的羟乙基磺酸盐用0.05M柠檬酸/0.5%CMC-Na/1%Tween 80(w/v)的去离子水配制。
2.实验动物
BALB/c裸小鼠,6-8周龄,雌性,购自北京维通利华实验动物技术有限公司。实验动物使用许可证号:SCXK(沪)2015-0022。饲养环境:SPF级。
饲养环境:温度:控制温度20~26℃;相对湿度:控制相对湿度40%~70%;光照:自动光照,每12h明暗交替。
3.实验步骤
将病人乳腺癌肿瘤(浸润性导管癌III级,上皮细胞来源)于1640培养液中剪成为15-30mm3的小块接种到裸鼠的皮下,待肿瘤长至600-700mm3后在裸鼠身上进行传代。待第八代(P8)肿瘤长至600-700mm3时,将肿瘤在1640培养液中剪成为15-30mm3的小块用于实验裸鼠皮下接种。待肿瘤生长至200-250mm3后,将动物随机分组并给药。试验组分为对照组,式(I)所示化合物的二马来酸盐单药组(给药剂量5mg/kg、10mg/kg)、式(II)所示化合物的羟乙基磺酸盐单药组(给药剂量150mg/kg)、式(I)所示化合物的二马来酸盐+式(II)所示化合物的羟乙基磺酸盐联合组(给药剂量5mg/kg+150mg/kg、10mg/kg+150mg/kg),每组6只。每组给药体积为10ml/kg。每天灌胃给药1次,共3周。每周测2-3次瘤体积,称鼠重,记录数据。肿瘤体积(V)计算公式为:
V=1/2×a×b2,其中a、b分别表示长、宽。
T/C(%)=(T-T0)/(C-C0)×100%,其中T、C为实验结束时的肿瘤体积;T0、C0为实验开始时的肿瘤体积。
实施例2:式(I)所示化合物和式(II)所示化合物联合给药与各自单独给药对HER2抑制剂耐药的胃癌PDX小鼠的药效作用研究
HER2抑制剂敏感的PDX模型以及HER2抑制剂耐药PDX模型的构建流程如下(式(I)所示化合物为HER2抑制剂):
用实时性内镜活检微量晚期胃癌组织构建大规模基于患者肿瘤组织的小鼠皮下移植瘤(patient-derived tumor xenograft,PDX)模型,建立PDX组织库。从PDX组织库中复苏PDX组织,接种至小鼠皮下,待肿瘤体积长至约150-200mm3大小(历时约1个月),开始给予式(I)所示化合物并历时约3个月时间(灌胃,每天一次,单次剂量为20mg/kg),肿瘤表现出对20mg/kg的式(I)所示化合物耐药;分离肿瘤组织,重新接种到新小鼠中,待肿瘤体积长至约150-200mm3大小(历时约1个月),开始给予式(I)所示化合物并历时约3个月时间(灌胃,每天一次,单次剂量为40mg/kg),肿瘤表现出对40mg/kg的式(I)所示化合物完全耐药。C019代表皮下接种复苏PDX组织1个月后的小鼠,S019代表以20mg/kg单次剂量给予式(I)所示化合物2周后的小鼠,R019代表以40mg/kg单次剂量给予式(I)所示化合物3个月后的小鼠。
耐药试验的方法:抑制剂组给予式(I)所示化合物(灌胃,每天一次,单次剂量为40mg/kg),对照组给予等体积生理盐水,每隔3天测量肿瘤体积。C019和R019模型的药物敏感性结果见图1(每组均为5只小鼠的平均值)。C019对式(I)所示化合物高度敏感,式(I)所示化合物的抑瘤率>100%。R019显示出对式(I)所示化合物的高度耐药。
C019、S019和R019的PDX组织进行HE染色,结果见图2。PDX组织历经长时间药物诱导后病理形态学保持一致。
对耐药前后的PDX组织(C019的PDX组织、S019的PDX组织和R019的PDX组织)进行转录组测序,对测序结果进行信号通路富集分析发现,相对于耐药前组织,耐药后组织中细胞周期的异常调控富集最显著,用western blot对耐药前后组织进行验证,发现参与细胞周期调控的关键分子(cyclin D1、CDK4/6、pRb等)表达水平在耐药后组织中显著上调(图3),尤其是CDK4/6及cyclin D1的下游效应分子Rb磷酸化显著升高。因此发明人推测,cyclinD1-CDK4/6通路异常可能参与胃癌HER2靶向治疗耐药,联合CDK4/6抑制剂或可成为耐药后治疗策略,该假说在耐药PDX模型中进行了验证。
联合给药方法:
对HER2抑制剂耐药的PDX模型(R019小鼠,每组5只小鼠)分组给药:
第一组(对照组):每天一次灌胃(口服)给予生理盐水,单只单次体积为100μl,连续3周;
第二组(HER2抑制剂组):每天一次灌胃(口服)给予式(I)所示化合物,单只单次体积为100μl,单次剂量为“式(I)所示化合物40mg/kg”,连续3周;
第三组(CDK4/6抑制剂组):每天一次灌胃(口服)给予式(II)所示化合物,单只单次体积为100μl,单次剂量为“式(II)所示化合物100mg/kg”,连续3周;
第四组(CDK4/6抑制剂联合HER2抑制剂组):每天一次灌胃(口服)给予式(I)所示化合物和式(II)所示化合物,单只单次体积为100μl,单次剂量为“式(I)所示化合物40mg/kg”和“式(II)所示化合物100mg/kg”,连续3周。
每隔3天测量肿瘤体积,结果见图4A,CDK4/6抑制剂联合HER2抑制剂具有显著的协同抑瘤作用。
联合用药后,组织中参与细胞周期的关键分子(cyclin D1、CDK4/6、pRb等)表达下调,尤其pRb显著下调,细胞停滞于G1期,而发挥抑瘤作用(图4B)。
实施例3:式(I)所示化合物和式(II)所示化合物联合给药用于治疗HER2表达阳性晚期胃癌
1、试验药物
式(I)所示化合物的二马来酸盐片剂,规格为40mg、60mg、200mg、160mg和80mg;
式(II)所示化合物的羟乙基磺酸盐片剂,规格为125mg和25mg。
2、入组受试者
(1)年龄为18~75岁;
(2)ECOG评分0~1级;
(3)经病理学确诊的HER2表达阳性,临床分期为III、IV期的胃癌(包括胃食管结合部癌)患者(HER2表达阳性指免疫组化染色为2+且经荧光原位杂交技术[FISH]确认阳性者,或免疫组化染色为3+);
(4)既往接受过针对转移性疾病的系统治疗并出现疾病进展。
3、给药方法
筛选合格的受试者,给予式(I)所示化合物及式(II)所示化合物。21天或28天为一个给药周期,式(I)所示化合物的给药方法为:口服,每日一次,给药剂量为每天400mg或320mg;式(II)所示化合物的给药方法为:口服,每日一次,给药剂量为每天100mg、125mg、150mg或175mg。持续给药至疾病进展,不能耐受毒性或患者意愿拒绝继续治疗,评估其病理缓解情况。
4、试验结果
共评价了3例经治疗的受试者,给药方法为28天为一个给药周期,式(I)所示化合物400mg,口服,每天一次,连续4周;式(II)所示化合物100mg,口服,每天一次,连续3周,停药一周。
患者A经治疗两个周期后,患者病灶缩小SD(stable disease),肿瘤标志物由治疗前的CEA 121.4ng/ml,CA199 60U/ml,CA72.4 5.4U/ml变成治疗后的CEA 15.8ng/ml,CA19945.3U/ml,CA72.4 4.1U/ml,从治疗中获益明显。
患者B经治疗两个周期后,患者病灶缩小特别明显,肿瘤标志物CEA、CA199、CA72.4也分别由入组前的122.2ng/ml、1871U/ml、23.54U/ml降低到26.97ng/ml、86.1U/ml、7.92U/ml,评效PR(partial response),病灶显著缩小,从治疗中获益明显。
患者C经治疗两个周期后,患者病灶缩小特别明显(肿瘤缩小约20%),肿瘤标志物CEA、CA199、CA72.4也分别由入组前的5.49ng/ml、250.1U/ml、78.46U/ml降低到1.92ng/ml、14.32U/ml、12.74U/ml,评效缩小SD,病灶显著缩小,从治疗中获益明显。
Claims (12)
2.根据权利要求1所述的用途,其中所述CDK4/6抑制剂选自abemaciclib、ribociclib、palbociclib、alvocidib、trilaciclib、voruciclib、AT-7519、G1T-38、FLX-925、INOC-005、G1T28-1、BPI-1178、gossypin、G1T30-1、GZ-38-1、P-276-00、staurosporine、R-547、PAN-1215、PD-0183812、AG-024322、NSC-625987、CGP-82996、PD-171851或式(II)所示化合物,优选abemaciclib、ribociclib、palbociclib、alvocidib或式(II)所示化合物,更优选式(II)所示化合物或其复合物或其可药用盐,
3.根据权利要求1所述的用途,其中所述肿瘤疾病选自HER2阳性或HER2突变肿瘤。
4.根据权利要求1所述的用途,其中所述肿瘤疾病选自乳腺肿瘤、卵巢癌、前列腺癌、黑色素瘤、脑瘤、食管癌、胃癌、肝癌、胰腺癌、结肠直肠癌、肺癌、肾癌、皮肤癌、成胶质细胞瘤、神经母细胞瘤、肉瘤、脂肪肉瘤、骨软骨瘤、骨瘤、骨肉瘤、精原细胞瘤、睾丸肿瘤、子宫癌、头颈肿瘤、多发性骨髓瘤、恶性淋巴瘤、真性红细胞增多症、白血病、甲状腺肿瘤、输尿管肿瘤、膀胱肿瘤、胆囊癌、胆管癌或绒毛膜上皮癌。
5.根据权利要求4所述的用途,其中所述的乳腺肿瘤选自乳头肿瘤、男性乳腺肿瘤、乳腺恶性淋巴瘤、纤维上皮性肿瘤、上皮-肌上皮性肿瘤、导管内癌、小叶原位癌、乳头湿疹样乳腺癌、早期浸润性导管癌、早期浸润性小叶癌、乳头状癌、髓样癌、小管癌、腺样囊性癌、粘液腺癌、大汗腺样癌、鳞状细胞癌、浸润性小叶癌、浸润性导管癌、硬癌。
6.根据权利要求1-4任意一项所述的用途,其中所述肿瘤疾病选自HER2抑制剂耐药的胃癌,所述的HER2抑制剂优选自曲妥珠单抗、帕妥珠单抗、拉帕替尼、阿法替尼、卡奈替尼、来那替尼和式(I)所示化合物中的一种或几种,更优选式(I)所示化合物。
7.根据权利要求1-6任意一项所述的用途,其中所述式(I)所示化合物的可药用盐为马来酸盐,优选二马来酸盐。
8.根据权利要求1-6任意一项所述的用途,其中所述式(II)所示化合物的可药用盐为羟乙基磺酸盐。
9.根据权利要求1-6任意一项所述的用途,其中所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂重量比为0.01:1-100:1,优选1:0.1、1:0.125、1:0.14、1:0.15、1:0.175、1:0.1875、1:0.2、1:0.25、1:0.28、1:0.3、1:0.35、1:0.4、1:0.5、1:0.7、1:0.75、1:1、1:1.25、1:1.75、1:2、1:2.5、1:3.5、1:4、1:5、1:8、1:10、1:15、2:15、1:20、1:25、3:1、3:2、6:1、6:5、6:7、8:5、8:7、12:1、15:7、16:3、16:5、16:7、16:15、16:25、16:35、24:5、24:7、60:7。
10.根据权利要求1-6任意一项所述的用途,其中所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体的剂量范围选自100-1000mg。
11.根据权利要求1-6任意一项所述的用途,其中所述CDK4/6抑制剂的剂量范围选自1-1000mg。
12.药物组合物,包含权利要求1所述式(I)所示化合物或其复合物或其可药用盐或其立体异构体与CDK4/6抑制剂,以及一种或多种可药用的赋形剂、稀释剂或载体。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2018109555189 | 2018-08-21 | ||
| CN201810955518 | 2018-08-21 | ||
| CN2019105370154 | 2019-06-20 | ||
| CN201910537015 | 2019-06-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN110840892A true CN110840892A (zh) | 2020-02-28 |
Family
ID=69584143
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910766740.9A Pending CN110840892A (zh) | 2018-08-21 | 2019-08-20 | 酪氨酸激酶抑制剂与cdk4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途 |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US10857146B2 (zh) |
| CN (1) | CN110840892A (zh) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021023204A1 (zh) * | 2019-08-06 | 2021-02-11 | 江苏恒瑞医药股份有限公司 | Cdk4/6抑制剂与多靶点酪氨酸激酶抑制剂联合在制备治疗肿瘤的药物中的用途 |
| WO2021037185A1 (zh) * | 2019-08-30 | 2021-03-04 | 江苏恒瑞医药股份有限公司 | 一种低杂质含量的酪氨酸激酶抑制剂 |
| WO2021190637A1 (en) * | 2020-03-27 | 2021-09-30 | Jiangsu Alphamab Biopharmaceuticals Co., Ltd. | Combination of anti-her2 antibody and cdk inhibitior for tumor treatment |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102933574A (zh) * | 2011-03-11 | 2013-02-13 | 上海恒瑞医药有限公司 | (e)-n-[4-[[3-氯-4-(2-吡啶基甲氧基)苯基]氨基]-3-氰基-7-乙氧基-6-喹啉基]-3-[(2r)-1-甲基吡咯烷-2-基]丙-2-烯酰胺的可药用的盐、其制备方法及其在医药上的应用 |
| WO2016124067A1 (zh) * | 2015-02-03 | 2016-08-11 | 江苏恒瑞医药股份有限公司 | 一种周期素依赖性蛋白激酶抑制剂的羟乙基磺酸盐、其结晶形式及制备方法 |
| CN109481687A (zh) * | 2018-11-12 | 2019-03-19 | 北京市肿瘤防治研究所 | 用于胃癌治疗的cdk4/6抑制剂联合her2抑制剂的组合 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102020639A (zh) * | 2009-09-14 | 2011-04-20 | 上海恒瑞医药有限公司 | 6-氨基喹唑啉或3-氰基喹啉类衍生物、其制备方法及其在医药上的应用 |
| WO2014183520A1 (zh) | 2013-05-17 | 2014-11-20 | 上海恒瑞医药有限公司 | 吡啶并嘧啶类衍生物、其制备方法及其在医药上的应用 |
-
2019
- 2019-08-20 CN CN201910766740.9A patent/CN110840892A/zh active Pending
- 2019-08-20 US US16/545,004 patent/US10857146B2/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102933574A (zh) * | 2011-03-11 | 2013-02-13 | 上海恒瑞医药有限公司 | (e)-n-[4-[[3-氯-4-(2-吡啶基甲氧基)苯基]氨基]-3-氰基-7-乙氧基-6-喹啉基]-3-[(2r)-1-甲基吡咯烷-2-基]丙-2-烯酰胺的可药用的盐、其制备方法及其在医药上的应用 |
| WO2016124067A1 (zh) * | 2015-02-03 | 2016-08-11 | 江苏恒瑞医药股份有限公司 | 一种周期素依赖性蛋白激酶抑制剂的羟乙基磺酸盐、其结晶形式及制备方法 |
| CN109481687A (zh) * | 2018-11-12 | 2019-03-19 | 北京市肿瘤防治研究所 | 用于胃癌治疗的cdk4/6抑制剂联合her2抑制剂的组合 |
| CN109481687B (zh) * | 2018-11-12 | 2020-06-16 | 北京市肿瘤防治研究所 | 用于胃癌治疗的cdk4/6抑制剂联合her2抑制剂的组合 |
Non-Patent Citations (1)
| Title |
|---|
| BEIJING CANCER HOSPITAL: "Study to Evaluate SHR6390 Combined With Pyrotinib in Patients With HER2 Positive Gastric Cancer.", 《CLINICALTRIALS.GOV》 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021023204A1 (zh) * | 2019-08-06 | 2021-02-11 | 江苏恒瑞医药股份有限公司 | Cdk4/6抑制剂与多靶点酪氨酸激酶抑制剂联合在制备治疗肿瘤的药物中的用途 |
| WO2021037185A1 (zh) * | 2019-08-30 | 2021-03-04 | 江苏恒瑞医药股份有限公司 | 一种低杂质含量的酪氨酸激酶抑制剂 |
| WO2021190637A1 (en) * | 2020-03-27 | 2021-09-30 | Jiangsu Alphamab Biopharmaceuticals Co., Ltd. | Combination of anti-her2 antibody and cdk inhibitior for tumor treatment |
Also Published As
| Publication number | Publication date |
|---|---|
| US20200061049A1 (en) | 2020-02-27 |
| US10857146B2 (en) | 2020-12-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10888559B2 (en) | Quinoline derivatives for non-small cell lung cancer | |
| JP6761852B2 (ja) | がん治療 | |
| WO2025036475A1 (en) | A pharmaceutical combination and use thereof | |
| US20230398119A1 (en) | Combination therapy involving diaryl macrocyclic compounds | |
| TWI762784B (zh) | Cdk4/6抑制劑與egfr抑制劑聯合在製備治療腫瘤疾病的藥物中的用途 | |
| WO2019057141A1 (zh) | 阿帕替尼和c-Met抑制剂联合在制备治疗肿瘤的药物中的用途 | |
| CN111184863B (zh) | 酪氨酸激酶抑制剂、cdk4/6抑制剂、serd联合在制备治疗肿瘤的药物中的用途 | |
| CN114831988A (zh) | 治疗非小细胞肺癌的喹啉衍生物 | |
| WO2017129094A1 (zh) | 一种egfr/her2受体酪氨酸激酶抑制剂在制备治疗her2突变癌症药物中的用途 | |
| WO2023035223A1 (zh) | 药物组合物及其用途 | |
| CN110840892A (zh) | 酪氨酸激酶抑制剂与cdk4/6抑制剂联合在制备预防或治疗肿瘤疾病的药物中的用途 | |
| JP7783217B2 (ja) | アズブジンを含む抗腫瘍医薬組成物 | |
| CN107949401A (zh) | 联合施用酪氨酸激酶抑制剂来抑制表皮生长因子/表皮生长因子受体途径的方法及组合物 | |
| CN111643502A (zh) | 喹啉衍生物联合化疗药物用于治疗非小细胞肺癌 | |
| CN105476996A (zh) | 姜黄素与阿法替尼联合治疗非小细胞肺癌的用途 | |
| WO2021018310A1 (zh) | 用于治疗非小细胞肺癌的氨基吡啶衍生物 | |
| CN113840608A (zh) | Cdk4/6抑制剂与vegfr抑制剂联合在制备治疗肿瘤的药物中的用途 | |
| CN120154611B (zh) | 阿昔替尼在制备丛状神经纤维瘤治疗药物中的应用 | |
| JP6810763B2 (ja) | がん治療 | |
| WO2021219137A1 (zh) | 用于治疗met基因异常疾病的氨基吡啶衍生物 | |
| KR20260048300A (ko) | 종양 질환을 치료하기 위한 EGFR 키나제 억제제와 c-Met 키나제 억제제의 약물 조합 | |
| CN111821304A (zh) | 酪氨酸激酶抑制剂联合长春碱类药物在制备预防或治疗肿瘤疾病的药物中的用途 | |
| JP2025528490A (ja) | ヘテロアリールオキシナフタレン系化合物の使用 | |
| WO2023011415A1 (zh) | Egfr抑制剂的药物组合及其应用 | |
| CN121041265A (zh) | 化合物在制备抗结直肠癌腹膜转移药物中的应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20200228 |