Disclosure of Invention
The invention aims to provide a preparation method of a sawtooth-like Prussian blue nano material aiming at the defects of the prior art. According to the invention, nickel-cobalt-iron Prussian blue (NiCoFe-PBA) is used as a template, and sawtooth-like Prussian blue (Z' -PBA) is obtained through ammonia corrosion, so that the complex morphological structure design of nano MOFs is enriched; the mixed oxide nano material obtained by heat treatment can well keep the structure of Z' -PBA after corrosion and can effectively catalyze and degrade bisphenol A, thereby solving the problem of pollution caused by bisphenol A.
In order to achieve the purpose, the invention adopts the following technical scheme:
a preparation method of a sawtooth-like nickel-cobalt-iron Prussian blue sintered oxide nano material comprises the following steps:
(1) preparing a cubic-structure nano nickel-cobalt-iron Prussian blue (NiCoFe-PBA) precursor: the method comprises the following steps of preparing nano nickel cobalt iron Prussian blue (NiCoFe-PBA) with a cubic structure by using potassium ferricyanide, potassium cobaltcyanide, trisodium citrate trihydrate and nickel nitrate hexahydrate as raw materials through a hydrothermal synthesis method;
(2) preparing a sawtooth-like nickel-cobalt-iron Prussian blue ('Z' -PBA) nano material: uniformly dispersing the cubic-structure nano nickel-cobalt-iron Prussian blue (NiCoFe-PBA) precursor obtained in the step (1) into 10 ml of ethanol, and stirring until the precursor is completely dispersed to obtain a mixed solution; then slowly pouring the ammonia water solution into the mixed solution, and stirring for 1 h; then carrying out centrifugal separation on the reaction liquid, and washing; putting the washed sample into an oven, and keeping the temperature for 12 h at 70 ℃ for drying to obtain a zigzag nickel-cobalt-iron Prussian blue ('Z' -PBA) nano material;
(3) sawtooth-like nickel cobalt iron Prussian blue sintered oxide ('Z' -O)x) Preparing a nano material: putting the sawtooth-like nickel-cobalt-iron Prussian blue (' Z ' -PBA) nano material obtained in the step (2) into a muffle furnace, preserving the heat for 1.5 h at 350 ℃, and increasing the temperature at the rate of 2 ℃/min to finally obtain the sawtooth-like nickel-cobalt-iron Prussian blue sintered oxide (' Z ' -O ')x) And (3) nano materials.
In the preparation method, in the ammonia water solution in the step (2), the volume ratio of deionized water to ammonia water is 1: 2.
in the preparation method, the step (1) of preparing the nanometer nickel-cobalt-iron Prussian blue (NiCoFe-PBA) precursor with the cubic structure comprises the following specific steps:
1) putting a certain amount of trisodium citrate trihydrate and nickel nitrate hexahydrate into a beaker, adding deionized water to obtain a mixed system, and stirring the mixed system until the raw materials are completely dissolved to obtain a mixed solution A;
2) putting a certain amount of potassium cobalt cyanide into a beaker, and adding deionized water to prepare a solution;
3) adding the potassium cobalt cyanide solution into the mixed solution 1, and magnetically stirring for 1 h to obtain a mixed solution B;
4) putting a certain amount of potassium ferricyanide into a beaker, and adding deionized water to prepare a solution;
5) adding the potassium ferricyanide solution into the mixed solution 2, and magnetically stirring for 1 h to obtain a mixed solution C;
6) pouring the mixed solution C into a reaction kettle, and reacting for 20 hours at 80 ℃; and (3) centrifugally washing the reaction product, and drying at 70 ℃ for 12 h to obtain the cubic nano nickel-cobalt-iron Prussian blue (NiCoFe-PBA).
Further, the dosage of trisodium citrate trihydrate, nickel nitrate hexahydrate and deionized water in the step 1) is 0.264 g, 0.174 g and 20 mL;
further, the dosage of the potassium cobalt cyanide in the step 2) is 0.026 g, and the dosage of the deionized water is 10 mL; further, the using amount of the potassium ferricyanide in the step 4) is 0.105 g, and the using amount of the deionized water is 10 mL;
a sawtooth-like nickel-cobalt-iron Prussian blue sintered oxide nano material is applied to catalyzing the degradation of bisphenol A.
The invention has the beneficial effects that:
1) the method adopts NiCoFe-PBA as a precursor, and then uses ammonia water for corrosion to obtain the zigzag NiCoFe Prussian blue (Z' -PBA). The complex morphological structure design of the nano MOF is enriched, and more development opportunities are brought for realizing the versatility of the MOF. It also provides a new idea for anisotropic etching of MOFs.
2) The obtained zigzag nickel-cobalt-iron Prussian blue is subjected to heat treatment to obtain zigzag nickel-cobalt-iron Prussian blue sintered oxide (' Z ' -O ')x) Due to the sawtooth shape, the surface area is increased, more catalytic active sites are provided, the Peroxymonosulfate (PMS) is catalyzed to generate more sulfate radicals and hydroxyl radicals, and the efficiency of degrading the bisphenol A is improved.
3) The preparation method disclosed by the invention has the advantages of easiness in obtaining required equipment and materials, simplicity in process operation, concise process conditions, low cost, safety, high efficiency and easiness in popularization and application.
Detailed Description
In order to make the objects, technical solutions and advantages of the present invention more apparent, the present invention will be described in further detail below with reference to the accompanying drawings, which are examples. It should be understood that the specific embodiments described herein are merely illustrative of the invention and are not intended to limit the invention. In addition, the technical features mentioned in the embodiments of the present invention described below may be combined as long as they do not conflict with each other.
Example 1
A preparation method of a sawtooth-like nickel-cobalt-iron Prussian blue sintered oxide nano material comprises the following steps:
(1) preparation of NiCoFe-PBA precursor:
taking potassium ferricyanide, potassium cobaltcyanide, trisodium citrate trihydrate and nickel nitrate hexahydrate as raw materials, and preparing NiCoFe-PBA by a hydrothermal synthesis method;
(2) preparation of "Z" -PBA nanomaterial:
uniformly dispersing NiCoFe-PBA obtained in the step (1) into 10 ml of ethanol, and stirring until the NiCoFe-PBA is completely dispersed to obtain a mixed solution; then slowly pouring the ammonia water solution into the mixed solution, and stirring for 1 h; then carrying out centrifugal separation on the reaction liquid, and washing; and (3) putting the washed sample into an oven, and keeping the temperature for 12 h at 70 ℃ for drying to obtain the Z' -PBA nano material.
(3) Preparation of "Z" -Ox nanomaterial:
and (3) putting the Z '-PBA nano material sample obtained in the step (2) into a muffle furnace, and preserving the heat for 1.5 h at 350 ℃ at the heating rate of 2 ℃/min to finally obtain the Z' -Ox nano material.
In the preparation method, in the ammonia water solution in the step (2), the volume ratio of deionized water to ammonia water is 1: 2.
in the preparation method, the step (1) of preparing the NiCoFe-PBA precursor comprises the following specific steps:
1) putting 0.264 g of trisodium citrate trihydrate and 0.174 g of nickel nitrate hexahydrate in a beaker, then adding 20 mL of deionized water to obtain a mixed system, and stirring the mixed system until the raw materials are completely dissolved to obtain a mixed solution A;
2) putting a certain amount of 0.026 g potassium cobalt cyanide into a beaker, and adding 10 mL deionized water to prepare a solution;
3) adding the potassium cobalt cyanide solution into the mixed solution 1, and magnetically stirring for 1 h to obtain a mixed solution B;
4) putting 0.105 g of potassium ferricyanide into a beaker, and adding 10 mL of deionized water to prepare a solution;
5) adding the potassium ferricyanide solution into the mixed solution 2, and magnetically stirring for 1 h to obtain a mixed solution C;
6) pouring the mixed solution C into a reaction kettle, and reacting for 20 hours at 80 ℃; and (3) centrifugally washing the reaction product, and drying at 70 ℃ for 12 h to obtain NiCoFe-PBA.
Comparative example 1
(1) Preparation of NiCoFe-PBA precursor:
taking potassium ferricyanide, potassium cobaltcyanide, trisodium citrate trihydrate and nickel nitrate hexahydrate as raw materials, and preparing NiCoFe-PBA by a hydrothermal synthesis method;
(2) synthesis of nickel-cobalt-iron Prussian blue sintered oxide (NiCoFe-Ox) nano material
Loading the NiCoFe-PBA sample obtained in the step (1) by using a small crucible, covering, putting the sample into a muffle furnace, preserving the temperature for 1.5 h at 350 ℃, and obtaining the NiCoFe-O at the heating rate of 2 ℃/minxAnd (3) nano materials.
The NiCoFe-O is obtainedxThe scanning microscopic image of the nanomaterial is shown in fig. 6.
Bisphenol A degradation experiment
Application example 1
"Z" -O obtained in example 1xThe nanometer material is used for the catalytic degradation of bisphenol A, and the specific steps are as follows:
(1) preparing a 30 ppm bisphenol A solution;
(2) 50 mL of the prepared bisphenol A solution (30 ppm) was weighed, and 5mg of "Z" -O was weighedxPutting the nano material into a bisphenol A solution, performing ultrasonic treatment for 10 min, performing magnetic stirring for 30 min to obtain a mixed solution, and establishing desorption-adsorption balance;
(3) weighing 10 mg of Peroxymonosulfate (PMS) and putting into the mixed solution obtained in the step (2), and magnetically stirring; sampling at 0 min, 10 min, 20 min, 30 min, 40 min and 50 min after the start of the degradation experiment, and quenching 1 mL of solution with 0.5 mL of methanol each time; bisphenol a was measured by liquid chromatography.
Application comparative example 1
NiCoFe-O obtained in comparative example 1xThe nanometer material is used for the catalytic degradation of bisphenol A, and the specific steps are as follows:
(1) preparing a 30 ppm bisphenol A solution;
(2) 50 mL of the prepared bisphenol A solution (30 ppm) was weighed and 5mg of NiCoFe-O was weighedxPutting the nano material into a bisphenol A solution, performing ultrasonic treatment for 10 min, performing magnetic stirring for 30 min to obtain a mixed solution, and establishing desorption-adsorption balance;
(3) weighing 10 mg of Peroxymonosulfate (PMS) and putting into the mixed solution obtained in the step (2), and magnetically stirring; sampling at 0 min, 10 min, 20 min, 30 min, 40 min and 50 min after the start of the degradation experiment, and quenching 1 mL of solution with 0.5 mL of methanol each time; bisphenol a was measured by liquid chromatography.
Analysis of results
The micro-morphology of the NiCoFe-PBA obtained in example 1 is shown in FIG. 1. The resulting "Z" -PBA has a microscopic morphology as shown in FIG. 2. The X-ray diffraction (XRD) results for NiCoFe-PBA and "Z" -PBA are shown in FIG. 3. The X-ray photoelectron spectroscopy (XPS) results for NiCoFe-PBA and "Z" -PBA are shown in FIG. 4. Analysis shows that in the process of synthesizing NiCoFe-PBA, the valence states of the cobalt and nickel elements are not changed, and the added ferric iron is subjected to oxidation-reduction reaction and is partially reduced into ferrous iron. When the 'Z' -PBA is obtained by corroding the 'Z' -PBA with ammonia water, the peak of the binding energy corresponding to the ferric iron disappears, which indicates that the ferric iron reacts with the ammonia water, but the ferrous iron does not react with the ammonia water, so that anisotropic corrosion occurs, and the sawtooth-like Prussian blue is obtained.
"Z" -O obtained by sintering "Z" -PBA "in example 1 at 350 ℃xFig. 5; the scanning microscopic image of NiCoFe-Ox obtained by sintering NiCoFe-PBA at 350 ℃ in comparative example 1 is shown in FIG. 6;
it can be seen from FIGS. 5 and 6 that the Z '-Ox and NiCoFe-Ox obtained from the heat treatment of Z' -PBA and NiCoFe-PBA can still maintain the original morphology.
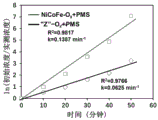
"Z" -O obtained by sintering in application example 1 and application comparative example 1xNanomaterial and NiCoFe-OxThe point diagram of the catalyst of the nanomaterial for catalyzing the degradation of bisphenol A by PMS is shown in figure 7. "Z" -O obtained by sintering in application example 1 and application comparative example 1xNanomaterial and NiCoFe-OxThe schematic diagram of the efficiency value K of the nanomaterial as the catalyst for catalyzing the degradation of the bisphenol A by PMS is shown in FIG. 8. Comparing the catalytic degradation efficiencies of application example 1 and application comparative example 1, it can be found that "Z" -PBA obtained by etching NiCoFe-PBA and "Z" -O obtained by sintering thereofxNano materialCan complete the catalytic degradation of 30 ppm of bisphenol A within 50 minutes, the efficiency reaches 99.9 percent, and the catalytic performance is higher than that of NiCoFe-OxThe apparent reaction rate constant k of the nano material is improved by 1.22 times. This is due to "Z" -OxThe sawtooth shape of the nano material provides more catalytic active sites so as to catalyze PMS to generate more sulfate radicals and hydroxyl radicals, and the catalytic performance is greatly improved.
It will be understood by those skilled in the art that the foregoing is merely a preferred embodiment of the invention, and is not intended to limit the invention, and that any modification, equivalent replacement or improvement made within the spirit and principle of the invention should be included within the scope of protection of the invention.