CN101511186A - Dihydropyridine compounds and compositions for headache - Google Patents
Dihydropyridine compounds and compositions for headache Download PDFInfo
- Publication number
- CN101511186A CN101511186A CNA2006800197719A CN200680019771A CN101511186A CN 101511186 A CN101511186 A CN 101511186A CN A2006800197719 A CNA2006800197719 A CN A2006800197719A CN 200680019771 A CN200680019771 A CN 200680019771A CN 101511186 A CN101511186 A CN 101511186A
- Authority
- CN
- China
- Prior art keywords
- migraine
- headache
- compound
- phenyl
- optional substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
相关申请related application
本申请根据35 U.S.C.§119要求享受于2005年6月13日提交的美国临时申请号60/689,519、于2005年4月4日提交的美国临时申请号60/667,665的优先权,在此将其全部内容引入作为参考。This application claims priority under 35 U.S.C. §119 to U.S. Provisional Application No. 60/689,519, filed June 13, 2005, and U.S. Provisional Application No. 60/667,665, filed April 4, 2005, which are hereby incorporated The entire contents are incorporated by reference.
发明领域 field of invention
本发明提供了含有二氢吡啶化合物的药物组合物以及使用二氢吡啶化合物治疗各种疾病和障碍的方法。所述二氢吡啶化合物可以任选与用于治疗各种疾病和障碍的其它药物组合使用,所述药物例如胆碱酯酶抑制剂或抗偏头痛剂。The present invention provides pharmaceutical compositions containing dihydropyridine compounds and methods of using the dihydropyridine compounds in the treatment of various diseases and disorders. The dihydropyridine compounds may optionally be used in combination with other drugs used in the treatment of various diseases and disorders, such as cholinesterase inhibitors or anti-migraine agents.
发明背景Background of the invention
已有论文对偏头痛的病理生理学进行过详细描述。参见Waeber等人,Neurology,61(Suppl 4):S9-S20(2003)。尽管引起偏头痛的机理目前仍然没有完全清楚,但是已报道过偏头痛的神经性理论。该理论认为,向脑膜突出的三叉神经脉管(trigeminovascular)纤维在偏头痛期间被激活,导致神经肽释出以及在硬脑膜出现神经性无菌炎症。据此建立了痛觉过敏症状。The pathophysiology of migraine has been described in detail. See Waeber et al., Neurology, 61 (Suppl 4): S9-S20 (2003). Although the mechanism causing migraine is still not fully understood, neurological theories of migraine have been reported. The theory is that trigeminovascular fibers projecting toward the meninges are activated during migraine, leading to release of neuropeptides and neurogenic sterile inflammation in the dura. Based on this established hyperalgesia symptoms.
有证据证实,至少存在10种受体(5-HT1B、5-HT1D、5-HT1F、5-HT2B、NK-1、GABAA、N-甲基-D-天冬氨酸(NMDA)、α-氨基-3-羟基-5-甲基异噁唑-4-丙酸(AMPA)、III类代谢型谷氨酸受体、阿片μ受体)代表了抗偏头痛剂的潜在靶标。舒马普坦影响5-HT1B/1D/1F受体。参见Mitsikostas等人,BrainResearch Reviews,35:20-35(2001)。已经研究过AMPA受体、红藻氨酸(kainate)受体和mGluR(代谢型谷氨酸受体)在头痛中的作用。据报道,阻断AMPA而不是红藻氨酸受体、或者激活mGluR4受体在头痛治疗中具有一定作用。参见Mitsikostas等人,British Journal of Pharmacology,127:623-630(1999)。将LY293558(α-氨基-3-羟基-5-甲基-4-异噁唑丙酸(AMPA)/红藻氨酸(KA)受体拮抗剂)与舒马普坦和安慰剂在偏头痛的临床前模型中进行了对比。对安慰剂而言,输注后2小时持续应答的有效率为25%(4患者/16患者);对LY293558而言,为69%(9患者/13患者);以及对舒马普坦而言为87%(13患者/15患者)。据此总结认为,AMPA/GLuR5拮抗作用为偏头痛治疗提供了靶标。参见Ramadan等人,Cephalalgia,21:267-272(2001);Sang等人,Cephalalgia,24:596-602(2004)。There is evidence for at least 10 receptors (5-HT 1B , 5-HT 1D , 5-HT 1F , 5-HT 2B , NK-1, GABA A , N-methyl-D-aspartate ( NMDA), α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA), class III metabotropic glutamate receptor, opioid μ receptor) represent potential anti-migraine agents target. Sumatriptan affects 5-HT 1B/1D/1F receptors. See Mitsikostas et al., BrainResearch Reviews, 35:20-35 (2001). The role of AMPA receptors, kainate receptors and mGluRs (metabotropic glutamate receptors) in headache has been studied. Blockade of AMPA but not kainate receptors, or activation of mGluR4 receptors, have been reported to have some role in headache treatment. See Mitsikostas et al., British Journal of Pharmacology, 127:623-630 (1999). Comparison of LY293558 (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainic acid (KA) receptor antagonist) with sumatriptan and placebo in migraine compared in preclinical models. For placebo, the effective rate of sustained
本领域仍然需要能够治疗可通过NMDA受体、AMPA受体和/或红藻氨酸受体以任何程度调节的疾病和障碍的新化合物和新方法。本发明目的即在于此以及其它的重要目的。There remains a need in the art for new compounds and new methods of treating diseases and disorders that are modulated to any degree by NMDA receptors, AMPA receptors and/or kainate receptors. It is with this and other important objectives that the present invention aims.
发明概述Summary of the invention
本发明提供了在有需要治疗头痛的患者中治疗和/或预防头痛(例如偏头痛)的方法,所述方法通过给药治疗有效量的至少一种1,2-二氢吡啶化合物,以及任选的治疗有效量的:(i)至少一种胆碱酯酶抑制剂、(ii)至少一种抗偏头痛剂或者(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。头痛可以是原发性头痛或继发性头痛。在一实施方案中,所述1,2-二氢吡啶化合物是3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮。在一实施方案中,所述胆碱酯酶抑制剂是多奈哌齐。1,2-二氢吡啶化合物和胆碱酯酶抑制剂的组合在头痛的治疗和/或预防中可以意料不到地产生协同效应。The present invention provides a method of treating and/or preventing headache (eg, migraine) in a patient in need thereof by administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound, and any A therapeutically effective amount of: (i) at least one cholinesterase inhibitor, (ii) at least one anti-migraine agent or (iii) at least one cholinesterase inhibitor and at least one anti-migraine agent agent. Headaches can be primary or secondary. In one embodiment, the 1,2-dihydropyridine compound is 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine- 2-keto. In one embodiment, the cholinesterase inhibitor is donepezil. The combination of a 1,2-dihydropyridine compound and a cholinesterase inhibitor can unexpectedly produce a synergistic effect in the treatment and/or prevention of headache.
在其它实施方案中,本发明提供了药物组合物,其中含有治疗有效量的至少一种1,2-二氢吡啶化合物(例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)。本发明提供了药物组合物,其中含有治疗有效量的:(i)至少一种胆碱酯酶抑制剂(例如多奈哌齐)和(ii)至少一种1,2-二氢吡啶化合物(例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)。本发明还提供了药物组合物,其中含有治疗有效量的:(i)至少一种抗偏头痛剂和(ii)至少一种1,2-二氢吡啶化合物(例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)。本发明提供了药物组合物,其中含有治疗有效量的:(i)至少一种胆碱酯酶抑制剂(例如多奈哌齐);(ii)至少一种1,2-二氢吡啶化合物(例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮);和(iii)至少一种抗偏头痛剂。In other embodiments, the present invention provides pharmaceutical compositions comprising a therapeutically effective amount of at least one 1,2-dihydropyridine compound (e.g., 3-(2-cyanophenyl)-5-(2-pyridine base)-1-phenyl-1,2-dihydropyridin-2-one). The present invention provides pharmaceutical compositions comprising a therapeutically effective amount of: (i) at least one cholinesterase inhibitor (such as donepezil) and (ii) at least one 1,2-dihydropyridine compound (such as 3- (2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one). The present invention also provides pharmaceutical compositions comprising a therapeutically effective amount of: (i) at least one anti-migraine agent and (ii) at least one 1,2-dihydropyridine compound (such as 3-(2-cyano phenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one). The present invention provides pharmaceutical compositions comprising a therapeutically effective amount of: (i) at least one cholinesterase inhibitor (such as donepezil); (ii) at least one 1,2-dihydropyridine compound (such as 3- (2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one); and (iii) at least one anti-migraine agent.
附图简述Brief description of the drawings
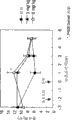
图1示出了化合物A(即3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)对同侧后爪退缩潜伏期(withdrawal latency)的影响。Figure 1 shows the reaction of compound A (i.e. 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one) on the same side Effect of paw withdrawal latency (withdrawal latency).
图2示出了化合物A(即3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)对对侧后爪退缩潜伏期的影响。Figure 2 shows the reaction of compound A (i.e. 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one) to the opposite side Effect of paw withdrawal latency.
发明详述Detailed description of the invention
"患者"是指动物,优选哺乳动物,更优选人。术语"患者"包括男人和女人;包括成年人、儿童和婴儿。在一实施方案中,患者可以是宠物,例如狗或猫。"Patient" refers to an animal, preferably a mammal, more preferably a human. The term "patient" includes both men and women; including adults, children and infants. In one embodiment, the patient may be a pet, such as a dog or cat.
按照在Cephalagia,24(Suppl.1):9-160(2004)(在此将其全部内容引入作为参考)中描述的The International Classification of Headache Disorders,"头痛"是指原发性头痛和继发性头痛。原发性头痛和继发性头痛可以是发作性的或慢性的。"发作性的头痛(Episodic headaches)"是指患者每月有1-14天感觉到头痛。"慢性头痛"是指患者每月有15天或者更多天感觉到头痛。治疗方法可以是急性或慢性的。"急性治疗"是指根据需要治疗头痛,例如一旦头痛发作则向患者给药治疗有效量的本文所述的本发明化合物或组合物。"慢性治疗"是指持续(例如每天)治疗头痛,不管患者在给药治疗有效量的本文所述的本发明化合物或组合物时是否感觉头痛。头痛可以引起一种或多种症状,例如眩晕、恶心、呕吐、疲劳、先兆(aura)、畏光、恐响(phonophobia)等。"Headache" refers to primary headache and secondary sexual headache. Primary and secondary headaches can be episodic or chronic. "Episodic headaches" means that patients feel headaches 1-14 days a month. "Chronic headache" means that the patient experiences headaches 15 or more days per month. Treatment can be acute or chronic. "Acute treatment"means treating headache as needed, eg, administering to the patient a therapeutically effective amount of a compound or composition of the invention described herein upon onset of headache. "Chronic treatment" refers to the ongoing (eg, daily) treatment of headache, whether or not the patient experiences headache when administered a therapeutically effective amount of a compound or composition of the invention described herein. Headaches may cause one or more symptoms such as dizziness, nausea, vomiting, fatigue, aura, photophobia, phonophobia, and the like.
"原发性头痛"包括偏头痛(migraine)、紧张性头痛(tension Headache)、丛集性头痛(cluster Headache)、突发性偏头痛(paroxysmal hemicrania)、因结膜充血和撕裂发作的短时间持续性单侧神经痛样头痛(SUNCT)、三叉神经自发性头痛(trigeminal autonomic cephalalgia)、刺痛性头痛(stabbing Headache)、咳嗽性头痛(cough Headache)、用力所致头痛(exertional headaches)、与性欲活动有关的头痛、催眠性头痛(hypnic Headache)、霹雳性头痛(thunderclapHeadache)、连续性偏头痛(hemicrania continua)或者新生的每天持续性头痛(new daily-persistent Headache)。"继发性头痛"包括归因于头和/或颈创伤的头痛;归因于颅和/或颈血管障碍的头痛;归因于非血管性颅内障碍的头痛;归因于药物的头痛;归因于药物停药的头痛;归因于感染的头痛;归因于体内稳态失衡的头痛;归因于面部结构和/或颅结构障碍的头痛或面部疼痛;或者归因于精神障碍的头痛。"Primary headache" includes migraine, tension headache, cluster headache, paroxysmal hemicrania, short-duration episodes due to conjunctival injection and tearing Unilateral neuralgia-like headache (SUNCT), trigeminal autonomic cephalalgia, stabbing headache, cough headache, exertional headaches, and libido Activity-related headache, hypnic headache, thunderclap headache, hemicrania continua, or new daily-persistent headache. "Secondary headache" includes headache attributable to head and/or neck trauma; headache attributable to cranial and/or carotid vascular disorders; headache attributable to nonvascular intracranial disorders; headache attributable to medications ; Headache due to drug discontinuation; Headache due to infection; Headache due to imbalance in homeostasis; Headache or facial pain due to disturbance of facial structure and/or cranial structure; or headache.
"偏头痛"是指周期性出现的综合症状,其特征在于头部出现疼痛,通常是头部出现单侧疼痛。偏头痛通常具有搏动性,且适度或严重影响或妨碍日常活动。由偏头痛引起的一种或多种症状包括眩晕、恶心、呕吐、疲劳、先兆、畏光、恐响等。出现偏头痛可以有也可以没有先兆。示例性偏头痛包括典型偏头痛(classic migraine)、普通型偏头痛(common migraine)、偏头痛型脑梗死(complicated migraine)、月经性偏头痛(menstrual migraine)、月经前偏头痛(premenstrual migraine)、眼型偏头痛(ophthalmic migraine)、眼肌麻痹性偏头痛(ophthalmoplegic migraine)、闪电状偏头痛(fulgurating migraine),Harris偏头痛(Harris migraine),和偏瘫性偏头痛(hemiplegic migraine)。可能出现由偏头痛引起的神经症状,但是后者并非在头部疼痛之后才发生。例如可能出现腹部疼痛和呕吐,因此头部疼痛并非偏头痛的唯一表现。"Migraine" means a recurrent syndrome characterized by pain in the head, usually on one side of the head. Migraines are usually throbbing and moderately or severely impair or interfere with daily activities. One or more symptoms caused by migraine include dizziness, nausea, vomiting, fatigue, aura, photophobia, phonophobia, etc. Migraines can occur with or without aura. Exemplary migraines include classic migraine, common migraine, complicated migraine, menstrual migraine, premenstrual migraine, Ophthalmic migraine, ophthalmoplegic migraine, fulgurating migraine, Harris migraine, and hemiplegic migraine. Neurologic symptoms due to migraine may occur, but the latter do not follow the head pain. For example, abdominal pain and vomiting may occur, so head pain is not the only manifestation of migraine.
针对给药两种或多种化合物以治疗和/或预防本文所述疾病和障碍而言的"单独给药"包括例如以任意顺序相继给药化合物或者同时给药化合物。同时给药化合物是指根据给药方式,将化合物基本上同时或者完全同一时间向患者给药。相继给药化合物可以以任意顺序进行,在给药化合物之间也可以没有任何等待时间。相继给药可以考虑各种影响因素:哪种化合物应该首先给药,哪种化合物应该随后给药,给药化合物之间应该等到多长时间。例如,当单独和相继给药两种或多种化合物时,当向患者给药化合物时的影响因素包括例如:(a)获得所给药化合物最大效力的时间,(b)获得所给药化合物最少副作用的时间,(c)化合物的剂量,(d)化合物的给药途径,(e)待治疗的疾病或障碍,(f)待治疗的患者,(g)所给药化合物的体内关系,以及本领域已知的其它类似因素。优选选择相继给药的时间间隔,使得在对比效果时,活性成分的组合使用对待治疗疾病或障碍所产生的效果大于仅仅使用一种活性成分所取得效果的加合。"Single administration" with respect to the administration of two or more compounds for the treatment and/or prevention of the diseases and disorders described herein includes, for example, sequential administration of the compounds in any order or simultaneous administration of the compounds. Simultaneous administration of the compounds means that the compounds are administered to the patient at substantially the same time or at exactly the same time, depending on the mode of administration. The sequential administration of the compounds may be in any order and without any waiting time between administrations of the compounds. Sequential administration can take into account various influencing factors: which compound should be administered first, which compound should be administered second, and how long there should be a wait between administering the compounds. For example, when two or more compounds are administered separately and sequentially, factors influencing when administering the compounds to a patient include, for example: (a) the time to achieve maximum efficacy of the administered compounds, (b) the time to achieve maximum efficacy of the administered compounds. time to least side effects, (c) dose of compound, (d) route of administration of compound, (e) disease or disorder to be treated, (f) patient to be treated, (g) in vivo relationship of administered compound, and other similar factors known in the art. The time interval between successive administrations is preferably selected such that, when comparing effects, the combination of active ingredients produces an effect greater than the sum of the effects obtained with only one active ingredient for the disease or disorder to be treated.
术语"组合"是指将1,2-二氢吡啶化合物和第二活性成分(例如胆碱酯酶抑制剂)以分离的药物组合物或制剂形式单独给药(例如第一药物组合物含有1,2-二氢吡啶化合物,第二药物组合物含有胆碱酯酶抑制剂)。该药物组合物或制剂可以具有相同或不同的给药方式。The term "combination" refers to the separate administration of a 1,2-dihydropyridine compound and a second active ingredient (eg, a cholinesterase inhibitor) in separate pharmaceutical compositions or preparations (eg, the first pharmaceutical composition contains 1 , 2-dihydropyridine compound, the second pharmaceutical composition contains a cholinesterase inhibitor). The pharmaceutical compositions or formulations may have the same or different modes of administration.
"活性成分"是指1,2-二氢吡啶、胆碱酯酶抑制剂、抗偏头痛剂以及本文所述的用于治疗和/或预防疾病或障碍的其它化合物。"Active ingredient" refers to 1,2-dihydropyridine, cholinesterase inhibitors, anti-migraine agents, and other compounds described herein that are useful in the treatment and/or prevention of a disease or disorder.
"单一疗法"是指仅仅使用一种活性成分治疗和/或预防疾病或障碍的疗法。"Monotherapy" refers to therapy that uses only one active ingredient to treat and/or prevent a disease or disorder.
"组合疗法"是指其中两种或多种活性成分被单独给药或者以药物组合物形式给药用于治疗和/或预防疾病的疗法。"Combination therapy" refers to a therapy in which two or more active ingredients are administered alone or in a pharmaceutical composition for the treatment and/or prevention of a disease.
"治疗有效量"是指活性成分治疗和/或预防疾病所需要的用量。当给药两种或多种活性成分用于组合疗法时,术语"治疗有效量"是指各种活性成分治疗和/或预防疾病所需要的用量,包括例如:(a)治疗有效量的第一活性成分和治疗有效量的第二活性成分(即将每种活性成分用于单一疗法治疗和/或预防疾病的用量用于组合疗法);(b)治疗有效量的第一活性成分和亚治疗用量(sub-therapeutic amount)的第二活性成分,将其有效组合用于治疗和/或预防疾病(例如可以在组合疗法中使用亚治疗用量的第二活性成分,以获得等于或者大于第二活性成分如果用于单一疗法所取得的结果);c)亚治疗用量的第一活性成分和治疗有效量的第二活性成分,将其有效组合用于治疗和/或预防疾病(例如可以在组合疗法中使用亚治疗用量的第一活性成分,以获得等于或者大于第一活性成分如果用于单一疗法所取得的结果);和(d)亚治疗用量的第一活性成分和亚治疗用量的第二活性成分,它们在组合疗法中用于治疗和/或预防疾病或障碍(例如可以在组合疗法中使用亚治疗用量的第一活性成分,以获得等于或者大于第一活性成分如果用于单一疗法所取得的结果;以及可以在组合疗法中使用亚治疗用量的第二活性成分,以获得等于或者大于第二活性成分如果用于单一疗法所取得的结果)。当三种或多种活性成分用于组合疗法时,可以使用相同的治疗/亚治疗用量。例如:(a)可以是治疗有效量的所有三种活性成分;(b)可以是治疗有效量的两种活性成分和亚治疗用量的的第三活性成分;(c)可以是治疗有效量的一种活性成分和亚治疗用量的另两种活性成分;或者(d)可以是亚治疗用量所有三种活性成分。"Therapeutically effective amount" refers to the amount of the active ingredient required for the treatment and/or prevention of diseases. When two or more active ingredients are administered for combination therapy, the term "therapeutically effective amount" refers to the amount required for each active ingredient to treat and/or prevent diseases, including, for example: (a) a therapeutically effective amount of the first an active ingredient and a therapeutically effective amount of a second active ingredient (i.e. the amount of each active ingredient used for monotherapy treatment and/or disease prevention for combination therapy); (b) a therapeutically effective amount of the first active ingredient and sub-therapeutic A second active ingredient in an amount (sub-therapeutic amount), which is effectively combined for treatment and/or prevention of disease (for example, a sub-therapeutic amount of the second active ingredient can be used in combination therapy to obtain equal to or greater than the second active ingredient ingredients if used in monotherapy); c) a sub-therapeutic amount of the first active ingredient and a therapeutically effective amount of the second active ingredient in effective combination for the treatment and/or prophylaxis of disease (e.g. may be used in combination therapy (d) subtherapeutic amounts of the first active ingredient and subtherapeutic amounts of the second Active ingredients, which are used in combination therapy for the treatment and/or prophylaxis of a disease or disorder (e.g. subtherapeutic amounts of the first active ingredient may be used in combination therapy to obtain an amount equal to or greater than that of the first active ingredient if used in monotherapy) results obtained; and sub-therapeutic amounts of the second active ingredient may be used in combination therapy to achieve results equal to or greater than those obtained with the second active ingredient if used in monotherapy). When three or more active ingredients are used in combination therapy, the same therapeutic/subtherapeutic amounts can be used. For example: (a) may be a therapeutically effective amount of all three active ingredients; (b) may be a therapeutically effective amount of two active ingredients and a subtherapeutic amount of a third active ingredient; (c) may be a therapeutically effective amount of one active ingredient and subtherapeutic amounts of the other two active ingredients; or (d) may be subtherapeutic amounts of all three active ingredients.
"商品化包装"也称作试剂盒,可以包括下述的组合:(i)第一药物组合物或制剂,其中含有1,2-二氢吡啶化合物;(ii)第二药物组合物或制剂,其中含有第二活性成分(例如胆碱酯酶抑制剂);(iii)经FDA批准用于使用该药物组合物或制剂治疗或预防疾病的说明;和(iv)任选的给药药物组合物或制剂的其它材料(例如注射器、稀释剂、医用手套、手用消毒剂等);监测体内药物水平的其它材料;支持患者顺应药物治疗的其它材料;或者监测疾病状况的其它材料。商品化包装可以供应数天、数周或数月的充足药物和材料。在另一实施方案中,"商品化包装"可以包括(i)药物组合物或制剂,其中同时含有1,2-二氢吡啶化合物和第二活性成分(例如胆碱酯酶抑制剂);(ii)经FDA批准用于使用该药物组合物或制剂治疗或预防疾病的说明;和(iii)任选的给药药物组合物或制剂的其它材料(例如注射器、稀释剂、医用手套、手用消毒剂等);监测体内药物水平的其它材料;支持患者顺应药物治疗的其它材料;或者监测疾病状况的其它材料。商品化包装可以供应数天、数周或数月的充足药物和材料。A "commercial package", also known as a kit, may include a combination of: (i) a first pharmaceutical composition or preparation containing a 1,2-dihydropyridine compound; (ii) a second pharmaceutical composition or preparation , which contains a second active ingredient (eg, a cholinesterase inhibitor); (iii) FDA-approved instructions for using the pharmaceutical composition or preparation to treat or prevent a disease; and (iv) optionally administering the drug combination Other materials to monitor drug levels in the body; to support patient compliance with drug therapy; or to monitor disease conditions. Commercially available packaging can supply sufficient drugs and materials for days, weeks or months. In another embodiment, a "commercial package" may include (i) a pharmaceutical composition or formulation containing both a 1,2-dihydropyridine compound and a second active ingredient (eg, a cholinesterase inhibitor); ( ii) FDA-approved instructions for using the pharmaceutical composition or preparation to treat or prevent disease; and (iii) optional other materials for administering the pharmaceutical composition or preparation (e.g., syringes, diluents, medical gloves, hand disinfectants, etc.); other materials that monitor drug levels in the body; other materials that support patient compliance with drug therapy; or other materials that monitor disease conditions. Commercially available packaging can supply sufficient drugs and materials for days, weeks or months.
"水合物"是指含有结晶水分子的化合物。结晶水的分子可以是整数1或更多,例如1-10;或者是大于0的任意分数或者整数1-10的分数。例如水合物可以表示为化合物;化合物;化合物;化合物·2H2O;化合物·;化合物·6H2O等。"化合物"可以是本文所述的任意化合物,例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮。"Hydrate"refers to a compound containing water molecules of crystallization. The molecule of crystal water may be an integer of 1 or more, such as 1-10; or any fraction greater than 0 or a fraction of an integer of 1-10. For example hydrates can be expressed as compounds ; compound ; compound ; Compound · 2H 2 O; Compound · ; Compound · 6H 2 O, etc. "Compound" may be any compound described herein, for example 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one.
"可药用盐"是本领域熟知的,包括无机酸的可药用盐例如盐酸盐、硫酸盐、氢溴酸盐和磷酸盐,以及有机酸的可药用盐例如甲酸盐、乙酸盐、三氟乙酸盐、甲磺酸盐、苯磺酸盐和甲苯磺酸盐。当某些取代基被指定时,本发明化合物可以形成例如碱金属盐如钠或钾盐;碱土金属盐入如钙或镁盐;有机胺盐如与三甲胺、三乙胺、吡啶、甲基吡啶、二环己胺或N,N′-二苄基乙二胺的盐。本领域技术人员应该理解,本发明化合物可以制备成任何其它的可药用盐形式。"Pharmaceutically acceptable salts" are well known in the art and include pharmaceutically acceptable salts of inorganic acids such as hydrochloride, sulfate, hydrobromide and phosphate, and pharmaceutically acceptable salts of organic acids such as formate, ethyl salt, trifluoroacetate, methanesulfonate, besylate and toluenesulfonate. When certain substituents are specified, the compounds of the present invention can form, for example, alkali metal salts such as sodium or potassium salts; alkaline earth metal salts such as calcium or magnesium salts; organic amine salts such as trimethylamine, triethylamine, pyridine, methyl Salts of pyridine, dicyclohexylamine or N,N'-dibenzylethylenediamine. It should be understood by those skilled in the art that the compounds of the present invention can be prepared in any other pharmaceutically acceptable salt form.
在一实施方案中,用于本文所述方法和组合物中的化合物是1,2-二氢吡啶化合物。所述1,2-二氢吡啶化合物可以是本领域已知的任何化合物。术语"1,2-二氢吡啶化合物"包括1,2-二氢吡啶化合物、1,2-二氢吡啶化合物的可药用盐、1,2-二氢吡啶化合物的立体异构体、1,2-二氢吡啶化合物的立体异构体的可药用盐、1,2-二氢吡啶化合物的水合物、1,2-二氢吡啶化合物的可药用盐的水合物、1,2-二氢吡啶化合物的水合物的立体异构体以及1,2-二氢吡啶化合物的可药用盐的水合物的立体异构体。In one embodiment, the compound used in the methods and compositions described herein is a 1,2-dihydropyridine compound. The 1,2-dihydropyridine compound may be any compound known in the art. The term "1,2-dihydropyridine compounds" includes 1,2-dihydropyridine compounds, pharmaceutically acceptable salts of 1,2-dihydropyridine compounds, stereoisomers of 1,2-dihydropyridine compounds, 1 , pharmaceutically acceptable salts of stereoisomers of 2-dihydropyridine compounds, hydrates of 1,2-dihydropyridine compounds, hydrates of pharmaceutically acceptable salts of 1,2-dihydropyridine compounds, 1,2 - Stereoisomers of hydrates of dihydropyridine compounds and stereoisomers of hydrates of pharmaceutically acceptable salts of 1,2-dihydropyridine compounds.
优选地,用于本文所述方法和组合物中的1,2-二氢吡啶化合物是式(I)化合物:Preferably, the 1,2-dihydropyridine compound used in the methods and compositions described herein is a compound of formula (I):
其中in
Q是NH、O或S;Q is NH, O or S;
R1、R2、R3、R4和R5各自独立地是氢、卤素、C1-6烷基或-X-A;R 1 , R 2 , R 3 , R 4 and R 5 are each independently hydrogen, halogen, C 1-6 alkyl or -XA;
X是单键、任选被取代的C1-6亚烷基(alkylene)、任选被取代的C2-6亚烯基(alkenylene)、任选被取代的C2-6亚炔基(alkynylene)、-O-、-S-、-CO-、-SO-、-SO2-、-N(R6)-、-N(R7)-CO-、-CO-N(R8)-、-N(R9)-CH2-、-CH2-N(R10)-、-CH2-CO-、-CO-CH2-、-N(R11)S(O)m-、-S(O)n-N(R12)-、-CH2-S(O)p-、-S(O)q-CH2-、-CH2-O-、-O-CH2-、-N(R13)-CO-N(R14)-或-N(R15)-CS-N(R16)-;X is a single bond, optionally substituted C 1-6 alkylene (alkylene), optionally substituted C 2-6 alkenylene (alkenylene), optionally substituted C 2-6 alkynylene ( alkynylene), -O-, -S-, -CO-, -SO-, -SO 2 -, -N(R 6 )-, -N(R 7 )-CO-, -CO-N(R 8 ) -, -N(R 9 )-CH 2 -, -CH 2 -N(R 10 )-, -CH 2 -CO-, -CO-CH 2 -, -N(R 11 )S(O) m - , -S(O) n -N(R 12 )-, -CH 2 -S(O) p -, -S(O) q -CH 2 -, -CH 2 -O-, -O-CH 2 - , -N(R 13 )-CO-N(R 14 )- or -N(R 15 )-CS-N(R 16 )-;
R6、R7、R8、R9、R10、R11、R12、R13、R14、R15和R16各自独立地是氢、C1-6烷基或C1-6烷基氧基;R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 and R 16 are each independently hydrogen, C 1-6 alkyl or C 1-6 alkane base oxygen;
m、n、p和q各自独立地是整数0、1或2;m, n, p and q are each independently integers 0, 1 or 2;
A是任选被取代的C3-8环烷基、任选被取代的C3-8环烯基、任选被取代的5-至14-元非芳香杂环、任选被取代的C6-14芳香烃环或任选被取代的5-至14-元芳香杂环;条件是R1、R2、R3、R4和R5中的3个基团是-X-A;以及R1、R2、R3、R4和R5中其余2个基团独立地是氢、卤素或C1-6烷基。A is optionally substituted C 3-8 cycloalkyl, optionally substituted C 3-8 cycloalkenyl, optionally substituted 5- to 14-membered non-aromatic heterocycle, optionally substituted C 6-14 aromatic hydrocarbon rings or optionally substituted 5- to 14-membered aromatic heterocyclic rings; provided that three of R 1 , R 2 , R 3 , R 4 and R 5 are -XA; and R 1 , the remaining 2 groups in R 2 , R 3 , R 4 and R 5 are independently hydrogen, halogen or C 1-6 alkyl.
在一实施方案中,下述化合物排除在式(I)化合物范围之外:(1)当Q是O时;R1和R5是氢;以及R2、R3和R4是苯基;(2)当Q是O时;R1和R4是氢;以及R2、R3和R5是苯基;以及(3)当Q是O时;R1和R2是氢;以及R3、R4和R5是苯基。In one embodiment, the following compounds are excluded from the scope of compounds of formula (I): (1) when Q is O; R 1 and R 5 are hydrogen; and R 2 , R 3 and R 4 are phenyl; (2) when Q is O; R1 and R4 are hydrogen; and R2 , R3 and R5 are phenyl; and (3) when Q is O; R1 and R2 are hydrogen; 3 , R 4 and R 5 are phenyl.
在另一优选实施方案中,用于本文所述方法和组合物中的1,2-二氢吡啶化合物是式(II)化合物:In another preferred embodiment, the 1,2-dihydropyridine compound used in the methods and compositions described herein is a compound of formula (II):
其中Q是NH、O或S;Wherein Q is NH, O or S;
X1、X2和X3各自独立地是单键、任选被取代的C1-6亚烷基、任选被取代的C2-6亚烯基、任选被取代的C2-6亚炔基、-O-、-S-、-CO-、-SO-、-SO2-、-N(R6)-、-N(R7)-CO-、-CO-N(R8)-、-N(R9)-CH2-、-CH2-N(R10)-、-CH2-CO-、-CO-CH2-、-N(R11)-S(O)m-、-S(O)n-N(R12)-、-CH2-S(O)p-、-S(O)q-CH2-、-CH2-O-、-O-CH2-、-N(R13)-CO-N(R14)-或-N(R15)-CS-N(R16);X 1 , X 2 and X 3 are each independently a single bond, an optionally substituted C 1-6 alkylene group, an optionally substituted C 2-6 alkenylene group, an optionally substituted C 2-6 Alkynylene, -O-, -S-, -CO-, -SO-, -SO 2 -, -N(R 6 )-, -N(R 7 )-CO-, -CO-N(R 8 )-, -N(R 9 )-CH 2 -, -CH 2 -N(R 10 )-, -CH 2 -CO-, -CO-CH 2 -, -N(R 11 )-S(O) m -, -S(O) n -N(R 12 )-, -CH 2 -S(O) p -, -S(O) q -CH 2 -, -CH 2 -O-, -O-CH 2 -, -N(R 13 )-CO-N(R 14 )- or -N(R 15 )-CS-N(R 16 );
R6、R7、R8、R9、R10、R11、R12、R13、R14、R15和R16各自独立地是氢、C1-6烷基或C1-6烷氧基;R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 and R 16 are each independently hydrogen, C 1-6 alkyl or C 1-6 alkane Oxygen;
m、n、p和q各自独立地是整数0、1或2;m, n, p and q are each independently integers 0, 1 or 2;
A1、A2和A3各自独立地是任选被取代的C3-8环烷基、任选被取代的C3-8环烯基、任选被取代的5-至14-元非芳香杂环、任选被取代的C6-14芳香烃环或任选被取代的5-至14-元芳香杂环;以及R17和R18各自独立地是氢、卤素或C1-6烷基。A 1 , A 2 and A 3 are each independently optionally substituted C 3-8 cycloalkyl, optionally substituted C 3-8 cycloalkenyl, optionally substituted 5- to 14-membered non- Aromatic heterocycle, optionally substituted C 6-14 aromatic hydrocarbon ring or optionally substituted 5- to 14-membered aromatic heterocycle; and R 17 and R 18 are each independently hydrogen, halogen or C 1-6 alkyl.
在另一实施方案中,本发明提供了式(II)化合物,其中X1、X2和X3各自独立地是单键、任选被取代的C1-6亚烷基、任选被取代的C2-6亚烯基或任选被取代的C2-6亚炔基。取代基可以是一个或多个-O-、-S-、-CO-、-SO-、-SO2-、-N(R6)-、-N(R7)-CO-、-CO-N(R8)-、-N(R9)-CH2-、-CH2-N(R10)-、-CH2-CO-、-CO-CH2-、-N(R11)-S(O)m-、-S(O)n-N(R12)-、-CH2-S(O)p-、-S(O)q-CH2-、-CH2-O-、-O-CH2-、-N(R13)-CO-N(R14)-和-N(R15)-CS-N(R16)-;In another embodiment, the present invention provides a compound of formula (II), wherein X 1 , X 2 and X 3 are each independently a single bond, optionally substituted C 1-6 alkylene, optionally substituted C 2-6 alkenylene or optionally substituted C 2-6 alkynylene. Substituents can be one or more -O-, -S-, -CO-, -SO-, -SO 2 -, -N(R 6 )-, -N(R 7 )-CO-, -CO- N(R 8 )-, -N(R 9 )-CH 2 -, -CH 2 -N(R 10 )-, -CH 2 -CO-, -CO-CH 2 -, -N(R 11 )- S(O) m -, -S(O) n -N(R 12 )-, -CH 2 -S(O) p -, -S(O) q -CH 2 -, -CH 2 -O-, -O-CH 2 -, -N(R 13 )-CO-N(R 14 )- and -N(R 15 )-CS-N(R 16 )-;
R6、R7、R8、R9、R10、R11、R12、R13、R14、R15和R16各自独立地是氢、C1-6烷基或C1-6烷基氧基(alkoxy);R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 and R 16 are each independently hydrogen, C 1-6 alkyl or C 1-6 alkane Alkoxy;
m、n、p和q各自独立地是整数0、1或2;m, n, p and q are each independently integers 0, 1 or 2;
A1、A2和A3各自独立地是任选被取代的C3-8环烷基、任选被取代的C3-8环烯基、任选被取代的5-至14-元非芳香杂环、任选被取代的C6-14芳香烃环或任选被取代的5-至14-元芳香杂环。A 1 , A 2 and A 3 are each independently optionally substituted C 3-8 cycloalkyl, optionally substituted C 3-8 cycloalkenyl, optionally substituted 5- to 14-membered non- An aromatic heterocycle, an optionally substituted C 6-14 aromatic hydrocarbon ring, or an optionally substituted 5- to 14-membered aromatic heterocycle.
本发明的1,2-二氢吡啶化合物的取代基可以是一个或多个羟基;卤素;腈;硝基;C1-6烷基;C2-6烯基;C2-6炔基[其中烷基、烯基和炔基可以独立地任选被一个或多个选自下述的基团取代:羟基、腈、卤素、C1-6烷基氨基、二(C1-6烷基)氨基、C2-6烯基氨基、二(C2-6烯基)氨基、C2-6炔基氨基、二(C2-6炔基)氨基、N-C1-6烷基-N-C2-6烯基氨基、N-C1-6烷基-N-C2-6炔基氨基、N-C2-6烯基-N-C2-6炔基氨基、芳烷基氧基、TBDMS氧基、C1-6烷基磺酰氨基、C1-6烷基羰基氧基、C2-6烯基羰基氧基、C2-6炔基羰基氧基、N-C1-6烷基氨甲酰基、N-C2-6烯基氨甲酰基和N-C1-6炔基氨甲酰基];C1-6烷氧基;C2-6烯基氧基;C2-6炔基氧基[其中烷基氧基、烯基氧基和炔基氧基可以独立地任选被一个或多个选自下述的基团取代:C1-6烷基氨基、芳烷基氧基和羟基];C1-6烷基硫基;C2-6烯基硫基;C2-6炔基硫基[其中烷基硫基、烯基硫基和炔基硫基可以独立地任选被一个或多个选自下述的基团取代:羟基、腈、卤素、C1-6烷基氨基、芳烷基氧基、TBDMS氧基、C1-6烷基磺酰氨基、C1-6烷基羰基氧基和C1-6烷基氨甲酰基];任选被取代的羰基[其可以被下述基团取代:C1-6烷氧基、氨基、C1-6烷基氨基、二(C1-6烷基)氨基、C2-6烯基氨基、二(C2-6烯基)氨基、C2-6炔基氨基、二(C2-6炔基)氨基、N-C1-6烷基-N-C2-6烯基氨基、N-C1-6烷基-N-C2-6炔基氨基和N-C2-6烯基-N-C2-6炔基氨基];任选被取代的氨基[其可以被一个或两个选自下述的基团取代:C1-6烷基、C2-6烯基、C2-6炔基、C1-6烷基磺酰基、C2-6烯基磺酰基、C2-6炔基磺酰基、C1-6烷基羰基、C2-6烯基羰基和C2-6炔基羰基];C1-6烷基磺酰基;C2-6烯基磺酰基;C2-6炔基磺酰基;C1-6烷基亚磺酰基;C2-6烯基亚磺酰基;C2-6炔基亚磺酰基;甲酰基;任选被取代的C3-8环烷基;任选被取代的C3-8环烯基[其中环烷基和/或环烯基可以独立地任选被一个或多个选自下述的基团取代:羟基、卤素、腈、C1-6烷基、C1-6烷氧基、C1-6烷氧基C1-6烷基和芳烷基];5-至14-元非芳香杂环[其可以任选被一个或多个选自下述的基团取代:羟基、卤素、腈、C1-6烷基、C1-6烷氧基、C1-6烷氧基C1-6烷基和芳烷基];C6-14芳香烃环[其可以任选被一个或多个选自下述的基团取代:羟基、卤素、腈、C1-6烷基、C1-6烷氧基、C1-6烷氧基C1-6烷基和芳烷基];以及5-至14-元芳香杂环[其可以任选被一个或多个选自下述的基团取代:羟基、卤素、腈、C1-6烷基、C1-6烷氧基、C1-6烷氧基C1-6烷基和芳烷基]。The substituting group of 1,2-dihydropyridine compound of the present invention can be one or more hydroxyl; Halogen; Nitrile; Nitro; C 1-6 alkyl; C 2-6 alkenyl; C 2-6 alkynyl[ Wherein the alkyl, alkenyl and alkynyl groups can be independently optionally substituted by one or more groups selected from the following groups: hydroxyl, nitrile, halogen, C 1-6 alkylamino, di(C 1-6 alkyl ) amino, C 2-6 alkenyl amino, two (C 2-6 alkenyl) amino, C 2-6 alkynyl amino, two (C 2-6 alkynyl) amino, NC 1-6 alkyl-NC 2 -6 alkenylamino, NC 1-6 alkyl-NC 2-6 alkynylamino, NC 2-6 alkenyl-NC 2-6 alkynylamino, aralkyloxy, TBDMSoxy, C 1-6 Alkylsulfonylamino, C 1-6 alkylcarbonyloxy, C 2-6 alkenylcarbonyloxy, C 2-6 alkynylcarbonyloxy, NC 1-6 alkylcarbamoyl, NC 2-6 Alkenylcarbamoyl and NC 1-6 alkynylcarbamoyl]; C 1-6 alkoxy; C 2-6 alkenyloxy; C 2-6 alkynyloxy [wherein alkyloxy, alkenyl C 1-6 alkylamino, aralkyloxy and hydroxy]; C 1-6 alkyl Thio; C 2-6 alkenylthio; C 2-6 alkynylthio [wherein alkylthio, alkenylthio and alkynylthio can be independently optionally selected from one or more of the following The group substitution: hydroxyl, nitrile, halogen, C 1-6 alkylamino, aralkyloxy, TBDMSoxy, C 1-6 alkylsulfonylamino, C 1-6 alkylcarbonyloxy and C 1-6 alkylcarbamoyl]; optionally substituted carbonyl [which may be substituted by the following groups: C 1-6 alkoxy, amino, C 1-6 alkylamino, di(C 1-6 Alkyl) amino, C 2-6 alkenyl amino, two (C 2-6 alkenyl) amino, C 2-6 alkynyl amino, two (C 2-6 alkynyl) amino, NC 1-6 alkyl- NC 2-6 alkenylamino, NC 1-6 alkyl-NC 2-6 alkynylamino and NC 2-6 alkenyl-NC 2-6 alkynylamino]; optionally substituted amino [which may be replaced by one Or two groups selected from the following substitutions: C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkylsulfonyl, C 2-6 alkenylsulfonyl , C 2-6 alkynylsulfonyl, C 1-6 alkylcarbonyl, C 2-6 alkenylcarbonyl and C 2-6 alkynylcarbonyl]; C 1-6 alkylsulfonyl; C 2-6 alkenyl Sulfonyl; C 2-6 alkynyl sulfinyl; C 1-6 alkyl sulfinyl; C 2-6 alkenyl sulfinyl; C 2-6 alkynyl sulfinyl; formyl; optionally substituted C 3-8 cycloalkyl; Optionally substituted C 3-8 cycloalkenyl [wherein the cycloalkyl and/or cycloalkenyl can be independently optionally substituted by one or more groups selected from the following groups: Hydroxy, halogen, nitrile, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkoxy C 1-6 alkyl and aralkyl]; 5- to 14-membered non-aromatic heterocycle [which may be optionally substituted by one or more groups selected from the group consisting of: hydroxyl, Halogen, nitrile, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkoxy C 1-6 alkyl and aralkyl]; C 6-14 aromatic hydrocarbon ring [it can be optionally Substituted by one or more groups selected from the group consisting of hydroxyl, halogen, nitrile, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkoxy C 1-6 alkyl and aryl Alkyl]; and a 5- to 14-membered aromatic heterocyclic ring [which may be optionally substituted by one or more groups selected from the group consisting of hydroxyl, halogen, nitrile, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkoxy C 1-6 alkyl and aralkyl].
在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地是任选被取代的C3-8环烷基、任选被取代的C3-8环烯基或任选被取代的5-至14-元非芳香杂环。在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地是任选被取代的C6-14芳香烃环或任选被取代的5-至14-元芳香杂环。在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地是苯基、吡咯基、吡啶基、哒嗪基、嘧啶基、吡嗪基、噻吩基、噻唑基、呋喃基、萘基、喹啉基、异喹啉基、吲哚基、苯并咪唑基、苯并噻唑基、苯并噁唑基、咪唑并吡啶基、咔唑基、环戊基、环己基、环己烯基、二噁英基(dioxinyl)、金刚烷基(adamantyl)、吡咯烷基、哌啶基、哌嗪基或吗啉基;其中任何一个任选具有取代基。在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地选自:In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently an optionally substituted C 3-8 cycloalkyl, an optionally substituted C 3 -8 cycloalkenyl or an optionally substituted 5- to 14-membered non-aromatic heterocyclic ring. In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently an optionally substituted C 6-14 aromatic hydrocarbon ring or an optionally substituted 5- to 14-membered aromatic heterocycle. In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently phenyl, pyrrolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, Thienyl, thiazolyl, furyl, naphthyl, quinolinyl, isoquinolyl, indolyl, benzimidazolyl, benzothiazolyl, benzoxazolyl, imidazopyridyl, carbazolyl, Cyclopentyl, cyclohexyl, cyclohexenyl, dioxinyl, adamantyl, pyrrolidinyl, piperidinyl, piperazinyl, or morpholinyl; any of which may optionally have a substituent . In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently selected from:
或 or
其中每一个可以任选被取代。在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地被羟基、卤素、氨基或腈取代。在另一实施方案中,本发明提供了式(II)化合物,其中A1、A2和A3各自独立地是羟基、卤素、氨基、腈或硝基。在另一实施方案中,本发明提供了式(II)化合物,其中Q是氧。Each of these can be optionally substituted. In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently substituted with hydroxyl, halogen, amino or nitrile. In another embodiment, the present invention provides a compound of formula (II), wherein A 1 , A 2 and A 3 are each independently hydroxyl, halo, amino, nitrile or nitro. In another embodiment, the invention provides compounds of formula (II), wherein Q is oxygen.
在另一实施方案中,本发明提供了式(I)或(II)化合物,其中X1、X2和X3各自独立地是单键、-CH2-、-CH(OH)-、-CH2-CH2-、-CH=CH-、-C≡C-、-O-或-CO-。在另一实施方案中,本发明提供了式(I)或(II)化合物,其中X1、X2和X3各自是单键。在另一实施方案中,本发明提供了式(I)或(II)化合物,其中R17和R18各自独立地是氢、氟、氯、溴、碘、甲基、乙基、正丙基或异丙基。在另一实施方案中,本发明提供了式(I)或(II)化合物,其中R17和R18各自是氢。In another embodiment, the present invention provides a compound of formula (I) or (II), wherein X 1 , X 2 and X 3 are each independently a single bond, -CH 2 -, -CH(OH)-, - CH2 - CH2- , -CH=CH-, -C≡C-, -O- or -CO-. In another embodiment, the invention provides a compound of formula (I) or (II), wherein each of X1 , X2 and X3 is a single bond. In another embodiment, the present invention provides compounds of formula (I) or (II), wherein R 17 and R 18 are each independently hydrogen, fluorine, chlorine, bromine, iodine, methyl, ethyl, n-propyl or isopropyl. In another embodiment, the invention provides compounds of formula (I) or (II), wherein R 17 and R 18 are each hydrogen.
对于本发明的1,2-二氢吡啶化合物,卤素原子是指氟、氯、溴、碘等,优选的原子包括氟、氯和溴。对于本发明的1,2-二氢吡啶化合物,C1-6烷基是指具有1-6个碳的烷基,其实例包括直链或支链烷基,例如甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基(1-甲基丙基)、叔丁基、异戊基、正戊基、叔戊基(1,1-二甲基丙基)、1,2-二甲基丙基、2,2-二甲基丙基(新戊基)、1-乙基丙基、2-甲基丁基、正己基、异己基、1,2-二甲基丁基、1-乙基-1-甲基丙基、1-乙基-2-甲基丙基、1,1,2-三甲基丙基、1,2,2-三甲基丙基、1-乙基丁基、1-甲基丁基、1,1-二甲基丁基、2,2-二甲基丁基、1,3-二甲基丁基、2,3-二甲基丁基、3,3-二甲基丁基、2-乙基丁基、1-甲基戊基、2-甲基戊基、3-甲基戊基等。For the 1,2-dihydropyridine compound of the present invention, the halogen atom means fluorine, chlorine, bromine, iodine, etc., and preferred atoms include fluorine, chlorine and bromine. For the 1,2-dihydropyridine compound of the present invention, C 1-6 alkyl refers to an alkyl group having 1-6 carbons, examples of which include linear or branched chain alkyl groups such as methyl, ethyl, normal Propyl, isopropyl, n-butyl, isobutyl, sec-butyl (1-methylpropyl), tert-butyl, isopentyl, n-pentyl, tert-amyl (1,1-dimethyl propyl), 1,2-dimethylpropyl, 2,2-dimethylpropyl (neopentyl), 1-ethylpropyl, 2-methylbutyl, n-hexyl, isohexyl, 1 , 2-dimethylbutyl, 1-ethyl-1-methylpropyl, 1-ethyl-2-methylpropyl, 1,1,2-trimethylpropyl, 1,2,2 -Trimethylpropyl, 1-ethylbutyl, 1-methylbutyl, 1,1-dimethylbutyl, 2,2-dimethylbutyl, 1,3-dimethylbutyl , 2,3-dimethylbutyl, 3,3-dimethylbutyl, 2-ethylbutyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, etc.
对于本发明的1,2-二氢吡啶化合物,C2-6烯基是指具有2-6个碳的烯基,其实例包括乙烯基、1-乙基乙烯基(1-丁烯-2-基)、烯丙基(2-丙烯基)、1-丙烯基、异丙烯基、2-甲基-1-丙烯基、1-甲基-1-丙烯基(2-丁烯-2-基)、2-甲基-2-丙烯基、1-甲基-2-丙烯基、1-丁烯基(1-丁烯-1-基)、2-丁烯基(2-丁烯-1-基)、3-丁烯基、1-戊烯基、1-己烯基、1,3-己二烯基、1,6-己二烯基等。For the 1,2-dihydropyridine compound of the present invention, C 2-6 alkenyl refers to alkenyl having 2-6 carbons, examples of which include vinyl, 1-ethylvinyl (1-butene-2 -yl), allyl (2-propenyl), 1-propenyl, isopropenyl, 2-methyl-1-propenyl, 1-methyl-1-propenyl (2-butene-2- base), 2-methyl-2-propenyl, 1-methyl-2-propenyl, 1-butenyl (1-buten-1-yl), 2-butenyl (2-butene- 1-yl), 3-butenyl, 1-pentenyl, 1-hexenyl, 1,3-hexadienyl, 1,6-hexadienyl and the like.
对于本发明的1,2-二氢吡啶化合物,C2-6炔基是指具有2-6个碳的炔基,其实例包括乙炔基、1-丙炔基、2-丙炔基、1-丁炔基、2-丁炔基、3-丁炔基、1-乙基-1-丙炔基、1-乙炔基-2-丙炔基、2-甲基-3-丁烯基、1-戊炔基、1-己炔基、1,3-己二炔基、1,6-己二炔基等。For the 1,2-dihydropyridine compound of the present invention, C 2-6 alkynyl refers to an alkynyl group having 2-6 carbons, examples of which include ethynyl, 1-propynyl, 2-propynyl, 1 -butynyl, 2-butynyl, 3-butynyl, 1-ethyl-1-propynyl, 1-ethynyl-2-propynyl, 2-methyl-3-butenyl, 1-pentynyl, 1-hexynyl, 1,3-hexadiynyl, 1,6-hexadiynyl and the like.
对于本发明的1,2-二氢吡啶化合物,C1-6烷基氧基是指具有1-6个碳的烷氧基,其实例包括甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、仲丁氧基、叔丁氧基、正戊氧基、异戊氧基、叔戊氧基、1,2-二甲基丙氧基、新戊氧基、1-乙基丙氧基、1-甲基丁氧基、2-甲基丁氧基、正己氧基、异己氧基、1-乙基-1-甲基丙氧基、1-乙基-2-甲基丙氧基、1,1,2-三甲基丙氧基、1,2,2-三甲基丙氧基、1,1-二甲基丁氧基、2,2-二甲基丁氧基、2,3-二甲基丁氧基、3,3-二甲基丁氧基、2-乙基丁氧基、1,3-二甲基丁氧基、1-乙基丁氧基、1-甲基丁氧基、1-甲基戊氧基、2-甲基戊氧基、3-甲基戊氧基等。For the 1,2-dihydropyridine compound of the present invention, C 1-6 alkyloxy refers to an alkoxy group having 1-6 carbons, examples of which include methoxy, ethoxy, n-propoxy, Isopropoxy, n-butoxy, isobutoxy, sec-butoxy, tert-butoxy, n-pentoxy, isopentoxy, tert-pentoxy, 1,2-dimethylpropoxy , neopentyloxy, 1-ethylpropoxy, 1-methylbutoxy, 2-methylbutoxy, n-hexyloxy, isohexyloxy, 1-ethyl-1-methylpropoxy , 1-ethyl-2-methylpropoxy, 1,1,2-trimethylpropoxy, 1,2,2-trimethylpropoxy, 1,1-dimethylbutoxy , 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 2-ethylbutoxy, 1,3-dimethylbutoxy oxy, 1-ethylbutoxy, 1-methylbutoxy, 1-methylpentyloxy, 2-methylpentyloxy, 3-methylpentyloxy, and the like.
对于本发明的1,2-二氢吡啶化合物,C2-6炔基氧基是指具有2-6个碳原子的炔基氧基,其实例包括乙炔氧基、1-丙炔氧基、2-丙炔氧基、1-丁炔氧基、2-丁炔氧基、3-丁炔氧基、1-甲基-2-丙炔氧基、1-乙基-2-丙炔氧基、1-乙炔基-2-丙炔氧基、1-戊炔氧基、1-己炔氧基、1,3-己二炔氧基、1,6-己二炔氧基等。For the 1,2-dihydropyridine compound of the present invention, C 2-6 alkynyloxy refers to an alkynyloxy group having 2-6 carbon atoms, examples of which include ethynyloxy, 1-propynyloxy, 2-propynyloxy, 1-butynyloxy, 2-butynyloxy, 3-butynyloxy, 1-methyl-2-propynyloxy, 1-ethyl-2-propynyloxy 1-ethynyl-2-propynyloxy, 1-pentynyloxy, 1-hexynyloxy, 1,3-hexadiynyloxy, 1,6-hexadiynyloxy, etc.
对于本发明的1,2-二氢吡啶化合物,C2-6烯基氧基是指具有2-6个碳的烯基氧基,其实例包括乙烯氧基、1-乙基乙烯氧基(1-丁烯-2-基氧基)、烯丙氧基(2-丙烯氧基)、1-丙烯氧基、异丙烯氧基、2-甲基-1-丙烯氧基、1-甲基-1-丙烯氧基(2-丁烯-2-基氧基)、2-甲基-2-丙烯氧基、1-甲基-2-丙烯氧基(1-丁烯-3-基氧基)、1-丁烯氧基(1-丁烯-1-基氧基)、2-丁烯氧基(2-丁烯-1-基氧基)、3-丁烯氧基、1-戊烯氧基、1-己烯氧基、1,3-己二烯氧基、1,6-己二烯氧基等。For the 1,2-dihydropyridine compound of the present invention, C 2-6 alkenyloxy refers to an alkenyloxy group having 2-6 carbons, examples of which include ethyleneoxy, 1-ethylvinyloxy ( 1-buten-2-yloxy), allyloxy (2-propenyloxy), 1-propenyloxy, isopropenyloxy, 2-methyl-1-propenyloxy, 1-methyl -1-propenyloxy (2-buten-2-yloxy), 2-methyl-2-propenyloxy, 1-methyl-2-propenyloxy (1-buten-3-yloxy base), 1-butenyloxy (1-buten-1-yloxy), 2-butenyloxy (2-buten-1-yloxy), 3-butenyloxy, 1- Pentenyloxy, 1-hexenyloxy, 1,3-hexadienyloxy, 1,6-hexadienyloxy and the like.
对于本发明的1,2-二氢吡啶化合物,C3-8环烷基是指由3-8个碳原子组成的环烷基,其实例包括环丙基、环丁基、环戊基、环己基、环庚基、环辛基等。For the 1,2-dihydropyridine compound of the present invention, C 3-8 cycloalkyl refers to a cycloalkyl group consisting of 3-8 carbon atoms, examples of which include cyclopropyl, cyclobutyl, cyclopentyl, Cyclohexyl, cycloheptyl, cyclooctyl, etc.
对于本发明的1,2-二氢吡啶化合物,C3-8环烯基是指由3-8个碳原子组成的环烯基,其实例包括环丙烯-1-基、2-环丙烯-1-基、环丁烯-1-基、2-环丁烯-1-基、1,3-环丁二烯-1-基、环戊烯-1-基、2-环戊烯-1-基、3-环戊烯-1-基、1,3-环戊二烯-1-基、1,4-环戊二烯-1-基、2,4-环戊二烯-1-基、环己烯-1-基、2-环己烯-1-基、3-环己烯-1-基、1,3-环己二烯-1-基、1,4-环己二烯-1-基、1,5-环己二烯-1-基、2,4-环己二烯-1-基、2,5-环己二烯-1-基、环戊烯-1-基、2-环戊烯-1-基、3-环戊烯-1-基、4-环戊烯-1-基、1,3-环戊二烯-1-基、1,4-环戊二烯-1-基、1,5-环戊二烯-1-基、1,6-环戊二烯-1-基、2,4-环戊二烯-1-基、2,5-环戊二烯-1-基、2,6-环戊二烯-1-基、3,5-环戊二烯-1-基、1,3,5-环庚三烯-1-基、1,3,6-环庚三烯-1-基、1,4,6-环庚三烯-1-基、2,4,6-环庚三烯-1-基、环辛烯-1-基、2-环辛烯-1-基、3-环辛烯-1-基、4-环辛烯-1-基、1,3-环辛二烯-1-基、1,4-环辛二烯-1-基、1,5-环辛二烯-1-基、1,6-环辛二烯-1-基、1,7-环辛二烯-1-基、2,4-环辛二烯-1-基、2,5-环辛二烯-1-基、2,6-环辛二烯-1-基、2,7-环辛二烯-1-基、3,5-环辛二烯-1-基、3,6-环辛二烯-1-基、1,3,5-环辛三烯-1-基、1,3,6-环辛三烯-1-基、1,3,7-环辛三烯-1-基、1,4,6-环辛三烯-1-基、1,4,7-环辛三烯-1-基、1,5,7-环辛三烯-1-基、2,4,6-环辛三烯-1-基、2,4,7-环辛三烯-1-基等。For the 1,2-dihydropyridine compound of the present invention, C 3-8 cycloalkenyl refers to a cycloalkenyl group consisting of 3-8 carbon atoms, examples of which include cyclopropen-1-yl, 2-cyclopropene- 1-yl, cyclobuten-1-yl, 2-cyclobuten-1-yl, 1,3-cyclobutadien-1-yl, cyclopenten-1-yl, 2-cyclopentene-1 -yl, 3-cyclopentadien-1-yl, 1,3-cyclopentadien-1-yl, 1,4-cyclopentadien-1-yl, 2,4-cyclopentadien-1- Base, cyclohexen-1-yl, 2-cyclohexen-1-yl, 3-cyclohexen-1-yl, 1,3-cyclohexadien-1-yl, 1,4-cyclohexyl En-1-yl, 1,5-cyclohexadien-1-yl, 2,4-cyclohexadien-1-yl, 2,5-cyclohexadien-1-yl, cyclopentene-1 - Base, 2-cyclopenten-1-yl, 3-cyclopenten-1-yl, 4-cyclopenten-1-yl, 1,3-cyclopentadien-1-yl, 1,4- Cyclopentadien-1-yl, 1,5-cyclopentadien-1-yl, 1,6-cyclopentadien-1-yl, 2,4-cyclopentadien-1-yl, 2, 5-cyclopentadien-1-yl, 2,6-cyclopentadien-1-yl, 3,5-cyclopentadien-1-yl, 1,3,5-cyclopentadien-1- Base, 1,3,6-cycloheptatrien-1-yl, 1,4,6-cycloheptatrien-1-yl, 2,4,6-cycloheptatrien-1-yl, cyclooctene -1-yl, 2-cycloocten-1-yl, 3-cycloocten-1-yl, 4-cycloocten-1-yl, 1,3-cyclooctadien-1-yl, 1, 4-cyclooctadien-1-yl, 1,5-cyclooctadien-1-yl, 1,6-cyclooctadien-1-yl, 1,7-cyclooctadien-1-yl, 2,4-cyclooctadien-1-yl, 2,5-cyclooctadien-1-yl, 2,6-cyclooctadien-1-yl, 2,7-cyclooctadiene-1- Base, 3,5-cyclooctadien-1-yl, 3,6-cyclooctadien-1-yl, 1,3,5-cyclooctatrien-1-yl, 1,3,6-ring Octatrien-1-yl, 1,3,7-cyclooctatrien-1-yl, 1,4,6-cyclooctatrien-1-yl, 1,4,7-cyclooctatriene-1 -yl, 1,5,7-cyclooctatrien-1-yl, 2,4,6-cyclooctatrien-1-yl, 2,4,7-cyclooctatrien-1-yl, etc.
对于本发明的1,2-二氢吡啶化合物,5-至14-元非芳香杂环是指含有一个或多个选自氮、硫和氧中的杂原子的单环、二环或三环5-至14-元非芳香杂环。其具体实例包括吡咯烷基、吡咯啉基、哌啶基、哌嗪基、吡唑烷基、咪唑烷基、吗啉基、四氢呋喃基、四氢吡喃基、二氢呋喃基、二氢吡喃基、咪唑烷基、噁唑烷基等。另外,非芳香杂环还包括由吡啶酮环和非芳香稠合环衍生的基团(例如由酞酰亚胺环、琥珀酰亚胺环等衍生的基团)。For the 1,2-dihydropyridine compounds of the present invention, the 5- to 14-membered non-aromatic heterocyclic ring refers to a monocyclic, bicyclic or tricyclic ring containing one or more heteroatoms selected from nitrogen, sulfur and oxygen 5- to 14-membered non-aromatic heterocycle. Specific examples thereof include pyrrolidinyl, pyrrolinyl, piperidinyl, piperazinyl, pyrazolidinyl, imidazolidinyl, morpholinyl, tetrahydrofuranyl, tetrahydropyranyl, dihydrofuranyl, dihydropyridine pyryl, imidazolidinyl, oxazolidinyl, etc. In addition, non-aromatic heterocyclic rings also include groups derived from pyridone rings and non-aromatic condensed rings (eg, groups derived from phthalimide rings, succinimide rings, etc.).
对于本发明的1,2-二氢吡啶化合物,C6-14芳香烃环和芳基是指由6-14个碳原子组成的芳香烃环、单环和稠合二环、三环等。其具体实例包括苯基、茚基、1-萘基、2-萘基、甘菊环烃基(azulenyl)、庚间三烯并庚间三烯(heptalenyl)、联苯基、indathenyl、苊基、芴基、phenalenyl、菲基、蒽基、环戊并环辛烯基、苯并环辛烯基等。For the 1,2-dihydropyridine compound of the present invention, C 6-14 aromatic hydrocarbon ring and aryl refer to aromatic hydrocarbon ring, monocyclic and fused bicyclic, tricyclic, etc. composed of 6-14 carbon atoms. Specific examples thereof include phenyl, indenyl, 1-naphthyl, 2-naphthyl, azulenyl, heptalenyl, biphenyl, indathenyl, acenaphthyl, fluorenyl , phenalenyl, phenanthrenyl, anthracenyl, cyclopentacyclooctenyl, benzocyclooctenyl, etc.
对于本发明的1,2-二氢吡啶化合物,5-至14-元芳香杂环和杂芳基环是指含有一个或多个选自氮、硫和氧中的杂原子的单环、二环或三环5-至14-元芳香杂环。其具体实例包括(1)含有氮的芳香杂环,例如吡咯基、吡啶基、哒嗪基、嘧啶基、吡嗪基、三唑基、四唑基、苯并三唑基、吡唑基、咪唑基、苯并咪唑基、吲哚基、异吲哚基、吲嗪基、异戊二烯基、吲唑基、喹啉基、异喹啉基、喹嗪基、酞嗪基(phthalazyl)、萘啶基(naphthylidinyl)、喹喔啉基(quinoxalyl)、喹唑啉基、肉啉基(cynnolinyl)、蝶啶基、咪唑并三嗪基、吡嗪并哒嗪基、吖啶基、菲啶基(phenanthridinyl)、咔唑基、咔唑啉基(carbazolinyl)、萘嵌间二氮杂苯基(perimidinyl)、菲咯啉基、吩嗪基、咪唑并吡啶基、咪唑并嘧啶基或吡唑并吡啶基;(2)含有硫的芳香杂环,例如噻吩基或苯并噻吩基;(3)含有氧的芳香杂环,例如呋喃基、吡喃基、环戊并吡喃基、苯并呋喃基或异苯并呋喃基;以及(4)含有两个或多个不同杂原子的芳香杂环,例如噻唑基、异噻唑基、苯并噻唑基、苯并噻二唑基、吩噻嗪基、异噁唑基、呋咱基、吩噁嗪基、噁唑基、异噁唑基、苯并噁唑基、噁二唑基、吡唑并噁二唑基、咪唑并噻唑基、噻吩并呋喃基、呋喃并吡咯基或pyridoxadinyl。For the 1,2-dihydropyridine compounds of the present invention, the 5- to 14-membered aromatic heterocyclic and heteroaryl rings refer to monocyclic, bicyclic or bicyclic rings containing one or more heteroatoms selected from nitrogen, sulfur and oxygen. Cyclic or tricyclic 5- to 14-membered aromatic heterocyclic ring. Specific examples thereof include (1) nitrogen-containing aromatic heterocycles such as pyrrolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazolyl, tetrazolyl, benzotriazolyl, pyrazolyl, Imidazolyl, benzimidazolyl, indolyl, isoindolyl, indolyl, isoprenyl, indazolyl, quinolinyl, isoquinolyl, quinazinyl, phthalazyl (phthalazyl) , naphthylidinyl (naphthylidinyl), quinoxalyl (quinoxalyl), quinazoline, cinnolinyl (cynnolinyl), pteridinyl, imidazotriazinyl, pyrazinopyridazinyl, acridyl, phenanthrene Phenanthridinyl, carbazolyl, carbazolinyl, perimidinyl, phenanthrolinyl, phenazinyl, imidazopyridinyl, imidazopyrimidinyl or pyrimidinyl Azolopyridyl; (2) sulfur-containing aromatic heterocycles, such as thienyl or benzothienyl; (3) oxygen-containing aromatic heterocycles, such as furyl, pyranyl, cyclopentapyranyl, benzene and (4) aromatic heterocycles containing two or more different heteroatoms, such as thiazolyl, isothiazolyl, benzothiazolyl, benzothiadiazolyl, phenothiazolyl, Azinyl, isoxazolyl, furazanyl, phenoxazinyl, oxazolyl, isoxazolyl, benzoxazolyl, oxadiazolyl, pyrazolooxadiazolyl, imidazothiazolyl, Thienofuryl, furopyrrolyl or pyridoxadinyl.
在另一实施方案中,用于本文所述方法和组合物中的1,2-二氢吡啶化合物优选为式(III)化合物:In another embodiment, the 1,2-dihydropyridine compound used in the methods and compositions described herein is preferably a compound of formula (III):
其中X1、X2、X3、A1、A2、A3、R17和R18具有与上述式(II)化合物中相同的含义。Wherein X 1 , X 2 , X 3 , A 1 , A 2 , A 3 , R 17 and R 18 have the same meanings as in the compound of formula (II).
在另一实施方案中,本发明提供了式(III)化合物,其中A1、A2和A3各自独立地是任选被取代的C6-14芳香烃环或5-至14-元芳香杂环。在另一实施方案中,本发明提供了式(III)化合物,其中A1、A2和A3各自独立地是苯基、吡咯基、吡啶基、哒嗪基、嘧啶基、吡嗪基、噻吩基、噻唑基、呋喃基、萘基、喹啉基、异喹啉基、吲哚基、苯并咪唑基、苯并噻唑基、苯并噁唑基、咪唑并吡啶基、咔唑基、环戊基、环己基、环己烯基、二噁英基(dioxinyl)、金刚烷基、吡咯烷基、哌啶基、哌嗪基或吗啉基;其中每个可以任选被取代。在另一实施方案中,本发明提供了式(III)化合物,其中A1、A2和A3各自独立地选自:In another embodiment, the present invention provides a compound of formula (III), wherein A 1 , A 2 and A 3 are each independently an optionally substituted C 6-14 aromatic hydrocarbon ring or a 5- to 14-membered aromatic heterocycle. In another embodiment, the present invention provides a compound of formula (III), wherein A 1 , A 2 and A 3 are each independently phenyl, pyrrolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, Thienyl, thiazolyl, furyl, naphthyl, quinolinyl, isoquinolyl, indolyl, benzimidazolyl, benzothiazolyl, benzoxazolyl, imidazopyridyl, carbazolyl, Cyclopentyl, cyclohexyl, cyclohexenyl, dioxinyl, adamantyl, pyrrolidinyl, piperidinyl, piperazinyl or morpholinyl; each of which may be optionally substituted. In another embodiment, the present invention provides a compound of formula (III), wherein A 1 , A 2 and A 3 are each independently selected from:
或 or
其中每一个可以任选被取代。在另一实施方案中,本发明提供了式(III)化合物,其中取代基在A1、A2和A3上的结合位置位于分别与基团X1、X2和X3相连的碳原子的α-位。在另一实施方案中,本发明提供了式(III)化合物,其中X1、X2和X3是单键。在另一实施方案中,本发明提供了式(III)化合物,其中R7和R18是氢。Each of these can be optionally substituted. In another embodiment, the present invention provides a compound of formula (III), wherein the binding positions of the substituents on A 1 , A 2 and A 3 are located at the carbon atoms connected to the groups X 1 , X 2 and X 3 respectively The α-bit. In another embodiment, the invention provides compounds of formula (III), wherein X 1 , X 2 and X 3 are single bonds. In another embodiment, the invention provides compounds of formula (III), wherein R 7 and R 18 are hydrogen.
在一实施方案中,用于本文所述方法和组合物中的1,2-二氢吡啶化合物优选为化合物A:In one embodiment, the 1,2-dihydropyridine compound used in the methods and compositions described herein is preferably Compound A:
化合物A的IUPAC名称是2-(2-氧代-1-苯基-5-吡啶-2-基-1,2-二氢吡啶-3-基)苯甲腈。化合物A还被称作3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮。The IUPAC name of compound A is 2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl)benzonitrile. Compound A is also known as 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one.
说明书上下文中,术语"化合物A"、"2-(2-氧代-1-苯基-5-吡啶-2-基-1,2-二氢吡啶-3-基)苯甲腈"和"3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮"意味着包括其可药用盐、其立体异构体、其立体异构体的可药用盐、其水合物、其可药用盐的水合物、其水合物的立体异构体和其可药用盐的水合物的立体异构体。在另一实施方案中,术语"化合物A"、"2-(2-氧代-1-苯基-5-吡啶-2-基-1,2-二氢吡啶-3-基)苯甲腈"和"3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮"意味着包括其可药用盐、其水合物和其可药用盐的水合物。In the context of the specification, the terms "compound A", "2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl)benzonitrile" and " 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one" means including its pharmaceutically acceptable salts, its stereoisomers hydrates, pharmaceutically acceptable salts of stereoisomers thereof, hydrates thereof, hydrates of pharmaceutically acceptable salts thereof, stereoisomers of hydrates thereof, and stereoisomers of hydrates of pharmaceutically acceptable salts thereof. In another embodiment, the term "compound A", "2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl)benzonitrile " and "3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one" are meant to include their pharmaceutically acceptable salts, their Hydrates and hydrates of their pharmaceutically acceptable salts.
在其它实施方案中,可用于本发明方法和组合物中的1,2-二氢吡啶化合物是3-(2-氰基苯基)-5-(2-甲基磺酰氨基苯基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氯-3-吡啶基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-硝基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-氨基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲基磺酰氨基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲基氨基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-二甲基氨基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-[3-(5-甲氧甲基-2-噁唑烷酮-3-基)-苯基]-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲氧羰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲基氨基羰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基-3-吡啶基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(4-羟苯基)-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(4-二甲基氨基乙氧基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲酰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-羟甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-氰基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-乙酰氨基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲基磺酰氨基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-乙酰氧基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-甲基噻吩基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-甲磺酰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-甲酰基噻吩-3-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-二乙氨基甲基噻吩-3-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-羟甲基噻吩-3-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-苄基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-苯基-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1,5-二苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-甲氧苯基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(3,4-二甲氧苯基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(噻吩-3-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-氟苯基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(噻吩-2-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(3-糠基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-糠基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-甲氧羰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-苯基-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氟苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(3-甲氧苯基)-1,2-二氢吡啶-2-酮;3-(2-氟-3-吡啶基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(4-甲氧基-3-吡啶基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氟-3-吡啶基)-5-(2-吡啶基)-1-(3-甲氧苯基)-1,2-二氢吡啶-2-酮;3-(2-氟-3-吡啶基)-5-(2-吡啶基)-1-(3-氟苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-氟苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-氟苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-甲氧苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲氧基-苯基)-1,2-二氢吡啶-2-酮;3-苯基-5-(2-吡啶基)-1-(3-氟苯基)-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(4-氟苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-甲酰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-甲酰基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-氯苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-三氟甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(噻吩-3-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-糠基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-甲苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-三氟甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-甲氧基吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(嘧啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-苄氧甲基吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-乙硫基吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(3-甲氧基吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-氯吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-氟吡啶-5-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-甲氧苯基)-1,2-二氢吡啶-2-酮;3-苯基-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(噻吩-3-基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2,6-二甲基苯基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基噻吩-3-基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氟-3-吡啶基)-5-(2-吡啶基)-1-(3-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(3-羟苯基)-1-,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(3-二甲基氨基乙氧基苯基)-1,2-二氢吡啶-2-酮;3-(2-氯苯基)-5-(2-吡啶基)-1-(3-二甲氨基丙氧苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-羟甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(4-氰基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-氰基甲基苯基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(6-二乙氨基甲基-2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-苯基-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-羟基吡啶-6-基)-1-苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;1-(2-氨基苯并噻唑-6-基)-3-(2-氰基苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(1-苄基-1,2,3,6-四氢吡啶-5-基)-1,2-二氢吡啶-2-酮;3-[2-(5-甲基-1,2,4-噁二唑-3-基)苯基]-1-苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(6-甲基吡啶-2-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(5-甲基吡啶-2-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(3-羟基吡啶-2-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-苯基-5-(2-噻唑基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-甲氧基吡啶-6-基)-1-苯基-1,2-二氢吡啶-2-酮;1-(4-氨基苯基)-3-(2-氰基苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;1-(3-氨基苯基)-3-(2-氰基苯基)-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-氨基甲苯-4-基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(二甲基氨基乙氧基)苯基]-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(哌啶乙氧基)苯基]-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(吡咯烷乙氧基)苯基]-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(二异丙基氨基乙氧基)苯基]-5-(-2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(4-哌啶丁基-1-氧基)苯基]-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-(4-硝基苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;1-苯基-5-(2-吡啶基)-3-(2-噻唑基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-(3-吡啶基)-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-氟吡啶-3-基)-1-苯基-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基吡啶-3-基)-1-苯基-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-(3-硝基苯基)-5-(2-嘧啶基)-1,2-二氢吡啶-2-酮;3-(2-硝基苯基)-1-苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-甲酰基噻吩-3-基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-萘基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(1-萘基)-1,2-二氢吡啶-2-酮;5-(2-氨基吡啶-6-基)-3-(2-氰基苯基)-1-苯基-1,2-二氢吡啶-2-酮;5-(2-溴吡啶-6-基)-3-(2-氰基苯基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吗啉并吡啶-6-基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-(3-羟苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-[3-(4-哌啶基氧基)]苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;1-[3-(N-乙酰基哌啶-4-基-氧基)苯基]-3-(2-氰基苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-1-{3-[1-(甲磺酰基)哌啶-4-基-氧基]苯基}-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;1-[3-(N-甲基哌啶-4-基-氧基)苯基]-3-(2-氰基苯基)-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-(6-氯-1H-苯并咪唑-2-基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-(2-氰基苯基)-5-(2-吡啶基)-1-(2-硝基甲苯(nitrotoluen)-4-基)-1,2-二氢吡啶-2-酮;3-(2-氰基噻吩-3-基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;3-[2-(5-噁唑基)苯基]-1-苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;3-[2-(5-噁唑基)噻吩-3-基]-1-苯基-5-(2-吡啶基)-1,2-二氢吡啶-2-酮;以及3-(2-乙氧羰基乙烯基噻吩-3-基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮。In other embodiments, the 1,2-dihydropyridine compound useful in the methods and compositions of the present invention is 3-(2-cyanophenyl)-5-(2-methylsulfonylaminophenyl)- 1-phenyl-1,2-dihydropyridin-2-one; 3-(2-chloro-3-pyridyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydro Pyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyano Phenyl)-5-(2-pyridyl)-1-(3-nitrophenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-( 2-pyridyl)-1-(3-aminophenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1- (3-methylsulfonylaminophenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-methyl ylaminophenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-dimethylaminophenyl) -1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-[3-(5-methoxymethyl-2-oxazole Alkanon-3-yl)-phenyl]-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-methyl Oxycarbonylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-methylaminocarbonylphenyl) -1,2-dihydropyridin-2-one; 3-(2-cyano-3-pyridyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2 -ketone; 3-(2-chlorophenyl)-5-(2-pyridyl)-1-(4-hydroxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-chloro Phenyl)-5-(2-pyridyl)-1-(4-dimethylaminoethoxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl )-5-(2-pyridyl)-1-(3-formylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2- Pyridyl)-1-(3-hydroxymethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1- (3-cyanomethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-acetamido Methylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-methylsulfonamidomethyl Phenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-acetoxymethylphenyl)- 1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1- (4-methylthienyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(4-methylsulfonylbenzene Base)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-formylthiophen-3-yl)-1-phenyl-1,2-two Hydropyridine-2-one; 3-(2-cyanophenyl)-5-(2-diethylaminomethylthiophen-3-yl)-1-phenyl-1,2-dihydropyridine-2- Ketone; 3-(2-cyanophenyl)-5-(2-hydroxymethylthiophen-3-yl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2- Cyanophenyl)-5-(2-pyridyl)-1-benzyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl )-1-(3-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-phenyl-(2-pyridyl)-1,2- Dihydropyridine-2-one; 3-(2-cyanophenyl)-1,5-diphenyl-1,2-dihydropyridine-2-one; 3-(2-cyanophenyl)- 5-(2-methoxyphenyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(3,4-dimethoxybenzene Base)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(thiophen-3-yl)-1-phenyl-1,2- Dihydropyridine-2-one; 3-(2-cyanophenyl)-5-(2-fluorophenyl)-1-phenyl-1,2-dihydropyridine-2-one; 3-(2 -cyanophenyl)-5-(thiophen-2-yl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(3- Furfuryl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-furfuryl)-1-phenyl-1,2- Dihydropyridine-2-one; 3-(2-chlorophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one; 3-(2-methyl Oxycarbonylphenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-phenyl-5-(2-pyridyl)-1-phenyl -1,2-dihydropyridin-2-one; 3-(2-fluorophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3 -(2-chlorophenyl)-5-(2-pyridyl)-1-(3-methoxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-fluoro-3- Pyridyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(4-methoxy-3-pyridyl)-5-(2- Pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-fluoro-3-pyridyl)-5-(2-pyridyl)-1-(3-methoxy Phenyl)-1,2-dihydropyridin-2-one; 3-(2-fluoro-3-pyridyl)-5-(2-pyridyl)-1-(3-fluorophenyl)- 1,2-dihydropyridine-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(4-fluorophenyl)-1,2-dihydropyridine- 2-keto; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-fluorophenyl)-1,2-dihydropyridin-2-one; 3-(2 -cyanophenyl)-5-(2-pyridyl)-1-(4-methoxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)- 5-(2-pyridyl)-1-(3-methoxy-phenyl)-1,2-dihydropyridin-2-one; 3-phenyl-5-(2-pyridyl)-1- (3-fluorophenyl)-1,2-dihydropyridin-2-one; 3-(2-chlorophenyl)-5-(2-pyridyl)-1-(4-fluorophenyl)-1 , 2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(4-formylphenyl)-1,2-dihydropyridine- 2-keto; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-formylphenyl)-1,2-dihydropyridin-2-one; 3-( 2-cyanophenyl)-5-(2-pyridyl)-1-(3-chlorophenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)- 5-(2-pyridyl)-1-(3-methylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)- 1-(3-trifluoromethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(thiophene- 3-yl)-1,2-dihydropyridine-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-furfuryl)-1,2- Dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(4-methylphenyl)-1,2-dihydropyridin-2-one; 3 -(2-cyanophenyl)-5-(2-pyridyl)-1-(4-trifluoromethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyano phenyl)-5-(2-pyridyl)-1-(2-methoxypyridin-5-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl )-5-(2-pyridyl)-1-(pyrimidin-5-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridine Base) -1-(3-benzyloxymethylpyridin-5-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl) -1-(2-Ethylthiopyridin-5-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1- (4-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(3-methoxypyridine-5 -yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-chloropyridine- 5-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-fluoropyridin-5-yl)- 1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-methoxyphenyl)-1,2-dihydropyridine -2-one; 3-phenyl-5-(2-pyridyl)-1-(3-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-chlorophenyl)- 5-(2-pyridyl)-1-(3-pyridyl)-1,2-dihydropyridin-2-one; 3-(thiophen-3-yl)-5-(2-pyridyl)-1 -(3-pyridyl)-1,2-dihydropyridin-2-one; 3-(2,6-dimethylphenyl)-5-(2-pyridyl)-1-(3-pyridyl )-1,2-dihydropyridin-2-one; 3-(2-cyanothiophen-3-yl)-5-(2-pyridyl)-1-(3-pyridyl)-1,2- Dihydropyridin-2-one; 3-(2-fluoro-3-pyridyl)-5-(2-pyridyl)-1-(3-pyridyl)-1,2-dihydropyridin-2-one ; 3-(2-chlorophenyl)-5-(2-pyridyl)-1-(3-hydroxyphenyl)-1-,2-dihydropyridin-2-one; 3-(2-chlorobenzene Base)-5-(2-pyridyl)-1-(3-dimethylaminoethoxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-chlorophenyl)- 5-(2-pyridyl)-1-(3-dimethylaminopropoxyphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2 -pyridyl)-1-(2-hydroxymethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1 -(4-cyanomethylphenyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-cyano 3-(2-cyanophenyl)-5-(6-diethylaminomethyl-2-pyridyl)-1-benzene Base-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-phenyl-5-(2-pyrimidinyl)-1,2-dihydropyridine-2-one ; 3-(2-hydroxypyridin-6-yl)-1-phenyl-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 1-(2-aminobenzothiazole- 6-yl)-3-(2-cyanophenyl)-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5- (2-pyridyl)-1-(1-benzyl-1,2,3,6-tetrahydropyridin-5-yl)-1,2-dihydropyridin-2-one; 3-[2-( 5-Methyl-1,2,4-oxadiazol-3-yl)phenyl]-1-phenyl-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3 -(2-cyanophenyl)-5-(6-methylpyridin-2-yl)-1-phenyl-1,2-dihydropyridine-2- Ketone; 3-(2-cyanophenyl)-5-(5-methylpyridin-2-yl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyano phenyl)-5-(3-hydroxypyridin-2-yl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-phenyl -5-(2-thiazolyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-methoxypyridin-6-yl)-1- Phenyl-1,2-dihydropyridin-2-one; 1-(4-aminophenyl)-3-(2-cyanophenyl)-5-(2-pyridyl)-1,2-di Hydropyridine-2-one; 1-(3-aminophenyl)-3-(2-cyanophenyl)-5-(2-pyrimidinyl)-1,2-dihydropyridin-2-one; 3 -(2-cyanophenyl)-5-(2-pyridyl)-1-(2-aminotoluene-4-yl)-1,2-dihydropyridin-2-one; 3-(2-cyano phenyl)-1-[3-(dimethylaminoethoxy)phenyl]-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyano phenyl)-1-[3-(piperidineethoxy)phenyl]-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanobenzene Base)-1-[3-(pyrrolidineethoxy)phenyl]-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl) -1-[3-(Diisopropylaminoethoxy)phenyl]-5-(-2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanobenzene Base)-1-[3-(4-piperidinyl-1-oxyl)phenyl]-5-(2-pyridinyl)-1,2-dihydropyridin-2-one; 3-(2 -cyanophenyl)-1-(4-nitrophenyl)-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 1-phenyl-5-(2-pyridine Base)-3-(2-thiazolyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-(3-pyridyl)-5-(2-pyrimidine Base)-1,2-dihydropyridin-2-one; 3-(2-fluoropyridin-3-yl)-1-phenyl-5-(2-pyrimidinyl)-1,2-dihydropyridine- 2-keto; 3-(2-cyanopyridin-3-yl)-1-phenyl-5-(2-pyrimidinyl)-1,2-dihydropyridin-2-one; 3-(2-cyano phenyl)-1-(3-nitrophenyl)-5-(2-pyrimidinyl)-1,2-dihydropyridin-2-one; 3-(2-nitrophenyl)-1- Phenyl-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-formylthiophen-3-yl)-5-(2-pyridyl)-1-benzene Base-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(2-naphthyl)-1,2-dihydropyridine -2-one; 3-(2-cyanophenyl)-5-(2-pyridyl)-1-(1-naphthyl)-1,2-dihydropyridine- 2-keto; 5-(2-aminopyridin-6-yl)-3-(2-cyanophenyl)-1-phenyl-1,2-dihydropyridin-2-one; 5-(2- Bromopyridin-6-yl)-3-(2-cyanophenyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-( 2-morpholinopyridin-6-yl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-(3-hydroxyphenyl)- 5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-[3-(4-piperidinyloxy)]phenyl- 5-(2-pyridinyl)-1,2-dihydropyridin-2-one; 1-[3-(N-acetylpiperidin-4-yl-oxyl)phenyl]-3-(2- Cyanophenyl)-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-1-{3-[1-(methylsulfonyl )piperidin-4-yl-oxyl]phenyl}-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 1-[3-(N-methylpiperidin-4 -yl-oxy)phenyl]-3-(2-cyanophenyl)-5-(2-pyridyl)-1,2-dihydropyridin-2-one; 3-(6-chloro-1H -benzimidazol-2-yl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-(2-cyanophenyl)-5-( 2-pyridyl)-1-(2-nitrotoluen (nitrotoluen)-4-yl)-1,2-dihydropyridin-2-one; 3-(2-cyanothiophen-3-yl)-5 -(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; 3-[2-(5-oxazolyl)phenyl]-1-phenyl-5-(2 -pyridyl)-1,2-dihydropyridin-2-one; 3-[2-(5-oxazolyl)thiophen-3-yl]-1-phenyl-5-(2-pyridyl)- 1,2-dihydropyridin-2-one; and 3-(2-ethoxycarbonylvinylthiophen-3-yl)-5-(2-pyridyl)-1-phenyl-1,2-dihydro Pyridin-2-one.
1,2-二氢吡啶化合物及制备1,2-二氢吡啶化合物的方法描述在US专利号6,949,571、US公开号2004/0023973和PCT公开号WO 03/047577、WO04/009553、WO 06/004100和WO 06/004107中,在此将其全部内容引入作为参考。1,2-Dihydropyridine compounds and methods of
在一实施方案中,用于本文所述方法和组合物中的化合物是胆碱酯酶抑制剂。所述胆碱酯酶抑制剂可以是本领域已知的任何一种。术语"胆碱酯酶抑制剂"包括胆碱酯酶抑制剂、胆碱酯酶抑制剂的可药用盐、碱酯酶抑制剂的立体异构体和胆碱酯酶抑制剂的立体异构体的可药用盐。示例性胆碱酯酶抑制剂包括多奈哌齐、他克林、毒扁豆碱、吡啶斯的明、新斯的明、利伐斯的明、加兰他敏、胞磷胆碱、维吖啶、石杉碱(例如石杉碱A)、美曲磷酯、heptastigmine、腾喜隆(edrophonium)、苯羟基丙氨酸(phenserine)、tolserine、phenethylnorcymserine、奎洛斯的明、更斯的明、epastigmine、upreazine、3-[1-(苯甲基)-4-哌啶基]-1-(2,3,4,5-四氢-1H-1-苯并氮杂-8-基)-1-丙酮、(2-[2-(1-苄基哌啶-4-基)乙基]-2,3-二氢-9-甲氧基-1H-吡咯并[3,4-b]喹啉-1-酮)等。在一实施方案中,所述胆碱酯酶抑制剂是多奈哌齐、他克林、加兰他敏或利伐斯的明。In one embodiment, the compounds used in the methods and compositions described herein are cholinesterase inhibitors. The cholinesterase inhibitor can be any one known in the art. The term "cholinesterase inhibitor" includes cholinesterase inhibitors, pharmaceutically acceptable salts of cholinesterase inhibitors, stereoisomers of alkaline esterase inhibitors and stereoisomers of cholinesterase inhibitors pharmaceutically acceptable salts. Exemplary cholinesterase inhibitors include donepezil, tacrine, physostigmine, pyridostigmine, neostigmine, rivastigmine, galantamine, citicoline, vinacridine, Perzine (such as huperzine A), metrifosate, heptastigmine, edrophonium, phenserine, tolserine, phenylnorcymserine, quinostigmine, ganstigmine, epastigmine, upreazine, 3-[1-(Benzyl)-4-piperidinyl]-1-(2,3,4,5-tetrahydro-1H-1-benzazepine -8-yl)-1-propanone, (2-[2-(1-benzylpiperidin-4-yl)ethyl]-2,3-dihydro-9-methoxy-1H-pyrrolo[ 3,4-b] quinolin-1-one) and the like. In one embodiment, the cholinesterase inhibitor is donepezil, tacrine, galantamine or rivastigmine.
在一实施方案中,用于本文所述方法和组合物中的优选胆碱酯酶抑制剂是式(IV)化合物:In one embodiment, a preferred cholinesterase inhibitor for use in the methods and compositions described herein is a compound of formula (IV):
其中J是(a)被取代或未被取代的选自下述的基团:(i)苯基、(ii)吡啶基、(iii)吡嗪基、(iv)喹啉基、(v)环己基、(vi)喹喔啉基(vii)呋喃基;(b)单价或二价基团,其中苯基可以具有一个或多个选自下述的取代基:(i)茚满基、(ii)茚满酮基、(iii)茚基、(iv)茚酮基、(v)茚满二酮基(indanedionyl)、(vi)四氢萘酮(tetralonyl)、(vii)苯并环庚酮基(benzosuberonyl)、(viii)茚满醇基(indanolyl)和(ix)C6H5-CO-CH(CH3)-;(c)由环酰胺化合物衍生的单价基团;(d)低级烷基;或(e)R21-CH=CH-,其中R21是氢或低级烷氧羰基;B是-(CHR22)r-、-CO-(CHR22)r-、-NR4-(CHR22)r-、-CO-NR5-(CHR22)r、-CH=CH-(CHR22)r-、-OCOO-(CHR22)r-、-OOC-NH-(CHR22)r-、-NH-CO-(CHR22)r-、-CH2-CO-NH-(CHR22)r-、-(CH2)2-NH-(CHR22)r、-CH(OH)-(CHR22)r-、=(CH-CH=CH)b-、=CH-(CH2)c-、=(CH-CH)d=、-CO-CH=CH-CH2-、-CO-CH2-CH(OH)-CH2-、-CH(CH3)-CO-NH-CH2-、-CH=CH=CO-NH-(CH2)2-、-NH-、-O-、-S-、二烷基氨基烷基-羰基或低级烷氧羰基;R4是氢、低级烷基、酰基、低级烷基磺酰基、苯基、被取代的苯基、苄基或被取代的苄基;R5是氢、低级烷基或苯基;r是0或者整数1-10;R22是氢或甲基,使得一个亚烷基可以没有甲基分支或者具有一个或多个甲基分支;b是整数1-3;c是0或者整数1-9;d是0或者整数1-5;T是氮或碳;Q是氮、碳或wherein J is (a) a substituted or unsubstituted group selected from (i) phenyl, (ii) pyridyl, (iii) pyrazinyl, (iv) quinolinyl, (v) Cyclohexyl, (vi) quinoxalinyl (vii) furyl; (b) monovalent or divalent groups, wherein phenyl may have one or more substituents selected from: (i) indanyl, (ii) indanonyl, (iii) indenyl, (iv) indanonyl, (v) indanedionyl (indanedionyl), (vi) tetralone (tetralonyl), (vii) benzo ring Heptanyl (benzosuberonyl), (viii) indanolyl (indanolyl) and (ix) C 6 H 5 -CO-CH(CH 3 )-; (c) a monovalent group derived from a cyclic amide compound; (d ) lower alkyl; or (e) R 21 -CH=CH-, wherein R 21 is hydrogen or lower alkoxycarbonyl; B is -(CHR 22 ) r -, -CO-(CHR 22 ) r -, -NR 4 -(CHR 22 ) r -, -CO-NR 5 -(CHR 22 ) r , -CH=CH-(CHR 22 ) r -, -OCOO-(CHR 22 ) r -, -OOC-NH-(CHR 22 ) r -, -NH-CO-(CHR 22 ) r -, -CH 2 -CO-NH-(CHR 22 ) r -, -(CH 2 ) 2 -NH-(CHR 22 ) r , -CH( OH)-(CHR 22 ) r -, =(CH-CH=CH) b -, =CH-(CH 2 ) c -, =(CH-CH) d =, -CO-CH=CH-CH 2 - , -CO-CH 2 -CH(OH)-CH 2 -, -CH(CH 3 )-CO-NH-CH 2 -, -CH=CH=CO-NH-(CH 2 ) 2 -, -NH- , -O-, -S-, dialkylaminoalkyl-carbonyl or lower alkoxycarbonyl; R4 is hydrogen, lower alkyl, acyl, lower alkylsulfonyl, phenyl, substituted phenyl, benzyl or substituted benzyl; R 5 is hydrogen, lower alkyl or phenyl; r is 0 or an integer of 1-10; R 22 is hydrogen or methyl, so that an alkylene can have no methyl branch or have one or multiple methyl branches; b is an integer of 1-3; c is 0 or an integer of 1-9; d is 0 or an integer of 1-5; T is nitrogen or carbon; Q is nitrogen, carbon or
q是整数1-3;K是氢、苯基、被取代的苯基、芳烷基,其中苯基可以具有取代基、肉桂酰基、低级烷基、吡啶基甲基、环烷基烷基、金刚烷甲基(adamantanemethyl)、呋喃基甲基、环烷基、低级烷氧羰基或者酰基;以及是单键或双键。 q is an integer of 1-3; K is hydrogen, phenyl, substituted phenyl, aralkyl, wherein phenyl may have a substituent, cinnamoyl, lower alkyl, pyridylmethyl, cycloalkylalkyl, adamantanemethyl, furylmethyl, cycloalkyl, lower alkoxycarbonyl or acyl; and is a single or double bond.
在式(IV)化合物中,J优选是(a)或(b),更优选(b)。在(b)的定义中,优选单价基团(2)、(3)和(5)和二价基团(2)。基团(b)优选包括下示结构式:In compounds of formula (IV), J is preferably (a) or (b), more preferably (b). In the definition of (b), monovalent groups (2), (3) and (5) and divalent groups (2) are preferred. Group (b) preferably comprises the following structural formula:
其中t是整数1-4;以及每个S独立地是氢或取代基,例如具有1-6个碳原子的低级烷基或具有1-6个碳原子的低级烷氧基。在这些取代基中,最优选甲氧基。苯基最优选在其上具有1-3个甲氧基。(S)t可以在与苯基两个相邻碳原子上形成亚甲二氧基或亚乙二氧基。在上述基团中,最优选茚满酮基、茚满二酮基和茚基,它们任选在苯基上具有取代基。wherein t is an integer of 1-4; and each S is independently hydrogen or a substituent, such as lower alkyl having 1-6 carbon atoms or lower alkoxy having 1-6 carbon atoms. Among these substituents, methoxy is most preferred. Phenyl most preferably has 1-3 methoxy groups on it. (S) t can form methylenedioxy or ethylenedioxy on two adjacent carbon atoms of phenyl. Among the above groups, indanonyl, indanedionyl and indenyl groups, which optionally have a substituent on the phenyl group, are most preferred.
在B的定义中,优选-(CHR22)r、-CO-(CHR22)r、=(CH-CH=CH)b-、=CH-(CH2)c-和=(CH-CH)d=(其中b是整数1-3;c是0或者整数1-9;d是0或者整数1-5)。最优选基团-(CHR22)r-,以及基团=CH-(CH2)c-,其中R22是氢以及r是整数1-3。B的优选基团可以与J的(b)、特别是(b)(2)相连。式(I)中含T和Q的环可以是5-、6-或7-元。优选Q是氮,T是碳或氮,以及q是2;或者Q是氮,T是碳,以及q是1或3;或者Q是碳,T是氮以及q是2。优选K是苯基、芳烷基、肉桂酰基、苯烷基或在苯基上具有取代基的苯烷基。In the definition of B, -(CHR 22 ) r , -CO-(CHR 22 ) r , =(CH-CH=CH) b -, =CH-(CH 2 ) c - and =(CH-CH) are preferred d = (wherein b is an integer of 1-3; c is 0 or an integer of 1-9; d is 0 or an integer of 1-5). Most preferred are the group -( CHR22 ) r- , and the group =CH-( CH2 ) c- , wherein R22 is hydrogen and r is an integer 1-3. Preferred groups of B may be attached to (b) of J, especially (b)(2). The ring containing T and Q in formula (I) may be 5-, 6- or 7-membered. Preferably Q is nitrogen, T is carbon or nitrogen, and q is 2; or Q is nitrogen, T is carbon, and q is 1 or 3; or Q is carbon, T is nitrogen and q is 2. Preferably K is phenyl, aralkyl, cinnamoyl, phenylalkyl or phenylalkyl having a substituent on the phenyl.
在一实施方案中,用于本文所述方法和组合物中的优选胆碱酯酶抑制剂是式(V)化合物:In one embodiment, a preferred cholinesterase inhibitor for use in the methods and compositions described herein is a compound of formula (V):
其中R1是(1)被取代或未被取代的苯基;(2)被取代或未被取代的吡啶基;(3)被取代或未被取代的吡嗪基(pyrazyl);(4)被取代或未被取代的喹啉基;(5)被取代或未被取代的茚满基;(6)被取代或未被取代的环己基;(7)被取代或未被取代的喹喔啉基;(8)被取代或未被取代的呋喃基;(9)由具有被取代或未被取代的苯基的茚满酮衍生的单价或二价基团(10)由环酰胺化合物衍生的单价基团;(11)低级烷基;或者(12)R3-CH=C-,其中R3是氢原子或低级烷氧羰基;X是-(CH2)n-、-C(O)-(CH2)n-、-N(R4)-(CH2)n-、-C(O)-N(R5)-(CH2)n-、-CH=CH-(CH2)n-、-O-C(O)-O-(CH2)n-、-O-C(O)-NH-(CH2)n-、-CH=CH-CH=CO-、-NH-C(O)-(CH2)n-、-CH2-C(O)-NH-(CH2)n-、-(CH2)2-C(O)-NH-(CH2)n-、-CH(OH)-(CH2)n-、-C(O)-CH=CH-CH2-、-C(O)-CH2-CH(OH)-CH2-、-CH(CH3)-C(O)-NH-CH2-、-CH=CH-C(O)-NH-(CH2)2-、二烷基氨基烷基羰基、低级烷氧羰基;n是整数0-6;R4是氢、低级烷基、酰基、低级烷基磺酰基、被取代或未被取代的苯基或被取代或未被取代的苄基;以及R5是氢、低级烷基或苯基;R2是被取代或未被取代的苯基;被取代或未被取代的芳烷基;肉桂酰基;低级烷基;吡啶基甲基;环烷基烷基;金刚烷甲基;或呋喃甲酰甲基;以及是单键或双键。Wherein R is (1) substituted or unsubstituted phenyl; (2) substituted or unsubstituted pyridyl; (3) substituted or unsubstituted pyrazyl (pyrazyl); (4) Substituted or unsubstituted quinolinyl; (5) substituted or unsubstituted indanyl; (6) substituted or unsubstituted cyclohexyl; (7) substituted or unsubstituted quinoxa (8) a substituted or unsubstituted furyl group; (9) a monovalent or divalent group derived from an indanone having a substituted or unsubstituted phenyl group (10) derived from a cyclic amide compound (11) lower alkyl; or (12) R 3 -CH=C-, wherein R 3 is a hydrogen atom or lower alkoxycarbonyl; X is -(CH 2 ) n -, -C(O )-(CH 2 ) n -, -N(R 4 )-(CH 2 ) n -, -C(O)-N(R 5 )-(CH 2 ) n -, -CH=CH-(CH 2 ) n -, -OC(O)-O-(CH 2 ) n -, -OC(O)-NH-(CH 2 ) n -, -CH=CH-CH=CO-, -NH-C(O )-(CH 2 ) n -, -CH 2 -C(O)-NH-(CH 2 ) n -, -(CH 2 ) 2 -C(O)-NH-(CH 2 ) n -, -CH (OH)-(CH 2 ) n -, -C(O)-CH=CH-CH 2 -, -C(O)-CH 2 -CH(OH)-CH 2 -, -CH(CH 3 )- C(O)-NH-CH 2 -, -CH=CH-C(O)-NH-(CH 2 ) 2 -, dialkylaminoalkylcarbonyl, lower alkoxycarbonyl; n is an integer 0-6; R is hydrogen, lower alkyl, acyl, lower alkylsulfonyl, substituted or unsubstituted phenyl or substituted or unsubstituted benzyl; and R is hydrogen, lower alkyl or phenyl; R2 is substituted or unsubstituted phenyl; substituted or unsubstituted aralkyl; cinnamoyl; lower alkyl; pyridylmethyl; cycloalkylalkyl; adamantylmethyl; Acylmethyl; and is a single or double bond.
对于本文所述的胆碱酯酶抑制剂,术语"低级烷基"是指具有1-6个碳原子的直链或支链烷基。示例性"低级烷基"包括甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、戊基(amyl)、异戊基、新戊基、叔戊基、1-甲基丁基、2-甲基丁基、1,2-二甲基丙基、己基、异己基、1-甲基戊基、2-甲基-戊基、3-甲基戊基、1,1-二甲基丁基、1,2-二甲基丁基、2,2-二甲基丁基、1,3-二甲基-丁基、2,3-二甲基丁基、3,3-二甲基丁基、1-乙基丁基、2-乙基丁基、1,1,2-三甲基丙基、1,2,2-三甲基丙基、1-乙基-1-甲基丙基、1-乙基-2-甲基丙基等。低级烷基优选是甲基、乙基、丙基或异丙基;更优选甲基。For the cholinesterase inhibitors described herein, the term "lower alkyl" refers to straight or branched chain alkyl having 1 to 6 carbon atoms. Exemplary "lower alkyl" includes methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, t-butyl, amyl, isopentyl, neopentyl, tert-amyl, 1-methylbutyl, 2-methylbutyl, 1,2-dimethylpropyl, hexyl, isohexyl, 1-methylpentyl, 2-methyl-pentyl, 3- Methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 2,2-dimethylbutyl, 1,3-dimethyl-butyl, 2,3- Dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1,1,2-trimethylpropyl, 1,2,2-trimethyl propyl, 1-ethyl-1-methylpropyl, 1-ethyl-2-methylpropyl, etc. Lower alkyl is preferably methyl, ethyl, propyl or isopropyl; more preferably methyl.
对于本文所述的胆碱酯酶抑制剂,R1定义中被取代或未被取代的苯基、吡啶基、吡嗪基、喹啉基、茚满基、环己基、喹喔啉基和呋喃基的取代基的实例包括具有1-6个碳原子的低级烷基,例如甲基、乙基、正丙基、异丙基、正丁基、异丁基、和叔丁基;相应于上述低级烷基的低级烷氧基,例如甲氧基和乙氧基;硝基;卤素,例如氯、氟和溴;羧基;相应于上述低级烷氧基的低级烷氧羰基,例如甲氧基羰基、乙氧基羰基、异丙氧基羰基、正丙氧基羰基和正丁氧基羰基;氨基;低级单烷基氨基;低级二烷基氨基;氨甲酰基;由具有1-6个碳原子的脂肪族饱和单羧酸衍生的酰氨基,例如乙酰氨基、丙酰氨基、丁酰氨基、异丁酰氨基、戊酰氨基和特戊酰氨基;环烷氧基羰基,例如环己基氧基羰基;低级烷基氨基羰基,例如甲基氨基羰基和乙基氨基羰基;相应于上述低级烷基的低级烷基羰基氧基,例如甲基羰基氧基、乙基羰基氧基和正丙基羰基氧基;卤代低级烷基,例如三氟甲基;羟基;甲酰基;和低级烷氧基低级烷基,例如乙氧甲基、甲氧甲基和甲氧基乙基。上述取代基中的"低级烷基"和"低级烷氧基"包括由上述基团衍生的所有基团。可以有1、2或3个可以相同或不同的取代基。For the cholinesterase inhibitors described herein, substituted or unsubstituted phenyl, pyridyl, pyrazinyl, quinolinyl, indanyl, cyclohexyl, quinoxalinyl and furan in the definition of R Examples of substituents of radicals include lower alkyl groups having 1 to 6 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, and tert-butyl; corresponding to the above-mentioned Lower alkoxy of lower alkyl such as methoxy and ethoxy; nitro; halogen such as chlorine, fluorine and bromine; carboxyl; lower alkoxycarbonyl corresponding to the above lower alkoxy such as methoxycarbonyl , ethoxycarbonyl, isopropoxycarbonyl, n-propoxycarbonyl and n-butoxycarbonyl; amino; lower monoalkylamino; lower dialkylamino; carbamoyl; amido groups derived from aliphatic saturated monocarboxylic acids, such as acetylamino, propionylamino, butyrylamino, isobutyrylamino, valerylamino and pivalylamino; cycloalkoxycarbonyl, such as cyclohexyloxycarbonyl; lower alkylaminocarbonyl such as methylaminocarbonyl and ethylaminocarbonyl; lower alkylcarbonyloxy corresponding to the above-mentioned lower alkyl such as methylcarbonyloxy, ethylcarbonyloxy and n-propylcarbonyloxy; Halogenated lower alkyl such as trifluoromethyl; hydroxy; formyl; and lower alkoxy lower alkyl such as ethoxymethyl, methoxymethyl and methoxyethyl. "Lower alkyl" and "lower alkoxy" in the above-mentioned substituents include all groups derived from the above-mentioned groups. There may be 1, 2 or 3 substituents which may be the same or different.
对于本文所述的胆碱酯酶抑制剂,当取代基是苯基时,下式落入被取代的苯基范围之内:For the cholinesterase inhibitors described herein, when the substituent is phenyl, the following formula falls within the range of substituted phenyl:
其中G是-C(O)-、-O-C(O)-、-O-、-CH2-NH-C(O)-、-CH2-O-、-CH2-SO2-、-CH(OH)-或-CH2-S(→O)-;E是碳或氮;以及D是取代基。where G is -C(O)-, -OC(O)-, -O-, -CH 2 -NH-C(O)-, -CH 2 -O-, -CH 2 -SO 2 -, -CH (OH)- or -CH2 -S(→O)-; E is carbon or nitrogen; and D is a substituent.
对于本文所述的胆碱酯酶抑制剂,苯基的取代基(即"D")的优选实例包括低级烷基、低级烷氧基、硝基、卤代低级烷基、低级烷氧基羰基、甲酰基、羟基和低级烷氧基低级烷基、卤素以及苯甲酰基和苄基磺酰基。可以有两个或更多个可以相同或不同的取代基。吡啶基的取代基的优选实例包括低级烷基和氨基以及卤素。吡嗪基的取代基的优选实例包括低级烷氧基羰基、羧基、酰氨基、氨甲酰基和环烷氧基羰基。For the cholinesterase inhibitors described herein, preferred examples of substituents for phenyl (i.e., "D") include lower alkyl, lower alkoxy, nitro, halogenated lower alkyl, lower alkoxycarbonyl , formyl, hydroxy and lower alkoxy lower alkyl, halogen and benzoyl and benzylsulfonyl. There may be two or more substituents which may be the same or different. Preferable examples of the substituent of pyridyl include lower alkyl and amino and halogen. Preferable examples of the substituent of the pyrazinyl group include lower alkoxycarbonyl, carboxyl, amido, carbamoyl and cycloalkoxycarbonyl.
对于本文所述的胆碱酯酶抑制剂,至于R1,所述吡啶基优选是2-吡啶基、3-吡啶基或4-吡啶基;所述吡嗪基优选是2-吡嗪基;所述喹啉基优选是2-喹啉基或3-喹啉基;所述喹喔啉基优选是2-喹喔啉基或3-喹喔啉基;以及所述呋喃基优选是2-呋喃基。For the cholinesterase inhibitors described herein, as for R 1 , said pyridyl is preferably 2-pyridyl, 3-pyridyl or 4-pyridyl; said pyrazinyl is preferably 2-pyrazinyl; The quinolinyl is preferably 2-quinolinyl or 3-quinolinyl; the quinoxalinyl is preferably 2-quinoxalinyl or 3-quinoxalinyl; and the furyl is preferably 2- furyl.
对于本文所述的胆碱酯酶抑制剂,单价或二价基团的实例衍生自具有(A)或(B)的未被取代或被取代的苯基的茚满酮:For the cholinesterase inhibitors described herein, examples of monovalent or divalent groups are derived from indanones with unsubstituted or substituted phenyl groups of (A) or (B):
其中m是整数1-4,每个A独立地是氢、低级烷基、低级烷氧基、硝基、卤素、羧基、低级烷氧基羰基、氨基、低级单烷基氨基、低级二烷基氨基、氨甲酰基、由具有1-6个碳原子的脂肪族饱和单羧酸衍生的酰氨基、环烷氧基羰基、低级烷基氨基羰基、低级烷基羰基氧基、卤代低级烷基、羟基、甲酰基或低级烷氧基低级烷基;优选氢、低级烷基或低级烷氧基;最优选茚满酮未被取代或者被1-3个甲氧基取代。Wherein m is an integer of 1-4, each A is independently hydrogen, lower alkyl, lower alkoxy, nitro, halogen, carboxyl, lower alkoxycarbonyl, amino, lower monoalkylamino, lower dialkyl Amino, carbamoyl, amido derived from aliphatic saturated monocarboxylic acid having 1-6 carbon atoms, cycloalkoxycarbonyl, lower alkylaminocarbonyl, lower alkylcarbonyloxy, halogenated lower alkyl , hydroxy, formyl or lower alkoxy lower alkyl; preferably hydrogen, lower alkyl or lower alkoxy; most preferably indanone is unsubstituted or substituted with 1-3 methoxy.
对于本文所述的胆碱酯酶抑制剂,由环酰胺化合物衍生的单价基团的实例包括喹唑啉酮、四氢异喹啉酮、四氢苯并二氮杂酮(tetrahydrobenzodiazepinone)和hexahydrobenzazocinone。单价基团可以是其结构式中具有环酰胺的任何一种,不限于上述具体实例。环酰胺可以是衍生自单环或稠合杂环的环酰胺。稠合杂环优选是通过与苯基稠合形成的杂环。在该情形中,苯基可以被具有1-6个碳原子的低级烷基优选甲基或者具有1-6个碳原子的低级烷氧基优选甲氧基取代。For the cholinesterase inhibitors described herein, examples of monovalent groups derived from cyclic amide compounds include quinazolinones, tetrahydroisoquinolones, tetrahydrobenzodiazepines, Ketones (tetrahydrobenzodiazepinone) and hexahydrobenzodiazepinone. The monovalent group may be any one having a cyclic amide in its structural formula, and is not limited to the above specific examples. The cyclic amide may be a cyclic amide derived from a single ring or a fused heterocycle. The fused heterocycle is preferably a heterocycle formed by fusion with a phenyl group. In this case, the phenyl group may be substituted with a lower alkyl group having 1 to 6 carbon atoms, preferably methyl group, or a lower alkoxy group having 1 to 6 carbon atoms, preferably methoxy group.
对于本文所述的胆碱酯酶抑制剂,单价基团的实例包括下述:For the cholinesterase inhibitors described herein, examples of monovalent groups include the following:
在上式中,Y是氢或低级烷基;V和U各自是氢或低级烷氧基(优选二甲氧基);W1和W2各自是氢、低级烷基或低级烷氧基;以及W3是氢或低级烷基。式(j)和(l)中的右手环是7-元环,式(k)中的右手环是8-元环。In the above formula, Y is hydrogen or lower alkyl; V and U are each hydrogen or lower alkoxy (preferably dimethoxy); W and W are each hydrogen, lower alkyl or lower alkoxy; and W 3 is hydrogen or lower alkyl. The right-hand rings in formulas (j) and (l) are 7-membered rings, and the right-hand rings in formula (k) are 8-membered rings.
对于本文所述的胆碱酯酶抑制剂,R1最优选的实例包括衍生自具有未被取代或被取代的苯基的茚满酮的单价基团和衍生自环酰胺化合物的单价基团。对于本文所述的胆碱酯酶抑制剂,X最优选的实例包括-(CH2)n-、酰胺或者上式所示基团,其中n是2。因此,最优选式所示基团的任何一部分具有羰基或酰胺。For the cholinesterase inhibitors described herein, most preferred examples of R 1 include monovalent groups derived from indanones having unsubstituted or substituted phenyl groups and monovalent groups derived from cyclic amide compounds. For the cholinesterase inhibitors described herein, the most preferred examples of X include -(CH 2 ) n -, amide, or a group represented by the above formula, wherein n is 2. Therefore, the optimal formula Any portion of the indicated groups has a carbonyl or amide.
对于本文所述的胆碱酯酶抑制剂,上述R2定义中的描述"被取代或未被取代的苯基"和"被取代或未被取代的芳烷基"中涉及的取代基是与前面对R1定义中苯基、吡啶基、吡嗪基、喹啉基、茚满基、环己基、喹喔啉基或呋喃基相同的取代基。术语"芳烷基"意思是指未被取代的苄基或苯乙基等。吡啶基甲基的具体实例包括2-吡啶基甲基、3-吡啶基甲基和4-吡啶基甲基。R2的优选实例包括苄基和苯乙基。符号表示双键或单键。该化学键只有当R1是衍生自具有未被取代或被取代的苯基的茚满酮的二价基团(B)时才是双键,在其它情形中均是单键。For the cholinesterase inhibitors described herein, the substituents involved in the description of "substituted or unsubstituted phenyl" and "substituted or unsubstituted aralkyl" in the definition of R above are the same as The same substituents as phenyl, pyridyl, pyrazinyl, quinolinyl, indanyl, cyclohexyl, quinoxalinyl or furyl in the above definition of R1 . The term "aralkyl" means unsubstituted benzyl or phenethyl or the like. Specific examples of pyridylmethyl include 2-pyridylmethyl, 3-pyridylmethyl and 4-pyridylmethyl. Preferable examples of R 2 include benzyl and phenethyl. symbol Indicates a double or single bond. This chemical bond is a double bond only if R 1 is a divalent group (B) derived from an indanone with unsubstituted or substituted phenyl, in all other cases a single bond.
在一实施方案中,用于本文所述方法和组合物中的优选胆碱酯酶抑制剂是式(VI)化合物:In one embodiment, a preferred cholinesterase inhibitor for use in the methods and compositions described herein is a compound of formula (VI):
其中r是整数1-10;每个R22独立地是氢或甲基;K是苯烷基或在苯基上具有取代基的苯烷基;每个S独立地是氢、C1-6低级烷基或C1-6低级烷氧基;t是整数1-4;q是整数1-3;条件是(S)t可以是与苯基上两个相邻碳原子相连的亚甲二氧基或亚乙二氧基。wherein r is an integer from 1 to 10; each R is independently hydrogen or methyl; K is phenylalkyl or phenylalkyl having a substituent on phenyl; each S is independently hydrogen, C 1-6 Lower alkyl or C 1-6 lower alkoxy; t is an integer of 1-4; q is an integer of 1-3; with the proviso that (S) t may be methylene dihydrogen connected to two adjacent carbon atoms on the phenyl group; Oxygen or ethylenedioxy.
在其它实施方案中,式(VI)化合物是1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶;1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基茚基(ylidenyl))甲基-哌啶;1-苄基-4-((5-甲氧基-1-茚满酮)-2-基)甲基哌啶;1-苄基-4-((5,6-二乙氧基-1-茚满酮)-2-基)甲基哌啶;1-苄基-4-((5,6-亚甲二氧基(methnylenedioxy)-1-茚满酮)-2-基)甲基哌啶;1-(间硝基苄基)-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶;1-环己基甲基-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶;1-(间氟苄基)-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶;1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基)丙基哌啶;1-苄基-4-((5-异丙氧基-6-甲氧基-1-茚满酮)-2-基)甲基哌啶;1-苄基-4-((5,6-二甲氧基-1-氧代茚满酮)-2-基)丙烯基哌啶;一种或多种前述化合物的可药用盐;一种或多种前述化合物的立体异构体;或者一种或多种前述化合物的立体异构体的可药用盐。In other embodiments, the compound of formula (VI) is 1-benzyl-4-((5,6-dimethoxy-1-indanone)-2-yl)methylpiperidine; 1-benzyl -4-((5,6-dimethoxy-1-indanone)-2-ylindenyl (ylidenyl))methyl-piperidine; 1-benzyl-4-((5-methoxy -1-indanone)-2-yl)methylpiperidine; 1-benzyl-4-((5,6-diethoxy-1-indanone)-2-yl)methylpiperidine ; 1-benzyl-4-((5,6-methylenedioxy)-1-indanone)-2-yl)methylpiperidine; 1-(m-nitrobenzyl)-4 -((5,6-dimethoxy-1-indanone)-2-yl)methylpiperidine; 1-cyclohexylmethyl-4-((5,6-dimethoxy-1- Indanone)-2-yl)methylpiperidine; 1-(m-fluorobenzyl)-4-((5,6-dimethoxy-1-indanone)-2-yl)methylpiperidine Pyridine; 1-benzyl-4-((5,6-dimethoxy-1-indanone)-2-yl)propylpiperidine; 1-benzyl-4-((5-isopropoxy Base-6-methoxy-1-indanone)-2-yl)methylpiperidine; 1-benzyl-4-((5,6-dimethoxy-1-oxoindanone) -2-yl)propenylpiperidine; a pharmaceutically acceptable salt of one or more of the aforementioned compounds; a stereoisomer of one or more of the aforementioned compounds; or a combination of one or more of the stereoisomers of the aforementioned compounds Pharmaceutically acceptable salt.
在其它实施方案中,用于本文所述方法和组合物中的式(VI)化合物优选是1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶;其如式(B)所示:In other embodiments, the compound of formula (VI) for use in the methods and compositions described herein is preferably 1-benzyl-4-((5,6-dimethoxy-1-indanone)-2 -yl) methyl piperidine; It is shown in formula (B):
式(B)化合物被称作多奈哌齐,可以是多晶型物或多晶型结晶形式。例如多奈哌齐可以是多晶型物(II)、(III)、(IV)或(V)形式;优选多晶型物(III)形式。多奈哌齐可以是多晶型结晶(A)、(B)或(C)形式。多晶型物、多晶型结晶以及制备多晶型物和多晶型结晶的方法描述在U.S专利号5,985,864、6,140,321和6,245,911中,在此将其全部内容引入作为参考。The compound of formula (B) is known as donepezil and may be in polymorphic or polymorphic crystalline forms. For example donepezil may be in the form of polymorph (II), (III), (IV) or (V); preferably the form of polymorph (III). Donepezil may be in polymorphic crystalline (A), (B) or (C) form. Polymorphs, polymorphic crystals, and methods of making polymorphs and polymorphic crystals are described in U.S. Patent Nos. 5,985,864, 6,140,321, and 6,245,911, the entire contents of which are incorporated herein by reference.
在另外的其它实施方案中,式(III)化合物是1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶盐酸盐,其还被称作多奈哌齐盐酸盐,其如式(B1)所示:In still other embodiments, the compound of formula (III) is 1-benzyl-4-((5,6-dimethoxy-1-indanone)-2-yl)methylpiperidine hydrochloride , which is also known as donepezil hydrochloride, as shown in formula (B1):
本发明化合物可能具有不对称碳原子,因此根据取代基可能具有立体异构体,后者落入本发明范围之内。例如多奈哌齐或其可药用盐可以是如日本专利申请号4-187674和4-21670中所述的形式,在此将其全部内容引入作为参考。The compounds of the present invention may have asymmetric carbon atoms and thus may have stereoisomers depending on substituents, the latter falling within the scope of the present invention. For example donepezil or a pharmaceutically acceptable salt thereof may be in the form as described in Japanese Patent Application Nos. 4-187674 and 4-21670, the entire contents of which are hereby incorporated by reference.
日本专利申请号4-187674描述了式(B2)化合物:Japanese Patent Application No. 4-187674 describes compounds of formula (B2):
其可以是可药用盐例如盐酸盐形式。日本专利申请号4-21670描述了式(B3)化合物:It may be in the form of a pharmaceutically acceptable salt such as hydrochloride. Japanese Patent Application No. 4-21670 describes compounds of formula (B3):
其可以是可药用盐例如盐酸盐形式;以及式(B4)化合物:It may be in the form of a pharmaceutically acceptable salt such as hydrochloride; and a compound of formula (B4):
其可以是可药用盐例如盐酸盐形式;以及式(B5)化合物:It may be in the form of a pharmaceutically acceptable salt such as hydrochloride; and a compound of formula (B5):
在说明书上下文中,术语"多奈哌齐"和"1-苄基-4-((5,6-二甲氧基-1-茚满酮)-2-基)甲基哌啶"意味着包括一种或多种下述物质(例如两种或多种的组合):可药用盐;立体异构体;多晶型物;和多晶型结晶。In the context of the specification, the terms "donepezil" and "1-benzyl-4-((5,6-dimethoxy-1-indanon)-2-yl)methylpiperidine" are meant to include a or more (eg, a combination of two or more) of the following: pharmaceutically acceptable salts; stereoisomers; polymorphs; and polymorphic crystals.
本发明的胆碱酯酶抑制剂可以商购得到,也可以由本领域已知的方法制备得到,例如描述在例如U.S专利号4,895,841、5,985,864、6,140,321和6,245,911;WO 98/39000以及日本专利申请号4-187674和4-21670中的方法,在此将其全部内容引入作为参考。The cholinesterase inhibitors of the present invention are commercially available or can be prepared by methods known in the art, such as described in, for example, U.S. Patent Nos. 4,895,841, 5,985,864, 6,140,321 and 6,245,911; WO 98/39000 and Japanese Patent Application No. 4 - the methods of 187674 and 4-21670, the entire contents of which are hereby incorporated by reference.
用于本文所述方法和组合物中的抗偏头痛剂可以是本领域已知的任何一种。抗偏头痛剂包括例如5-羟色胺拮抗剂、非甾类抗炎药(NSAIDs)(例如COX-1抑制剂和/或COX-2抑制剂)、钙通道阻断剂、β-肾上腺素阻断剂、抗惊厥剂和抗抑郁剂(例如三环抗抑郁剂、单胺氧化酶抑制剂和选择性5-羟色胺重摄取抑制剂)。其它示例性抗偏头痛剂包括PNU-142633、氨己烯酸、托吡酯、孟鲁司特(优选其钠盐)、加巴喷丁、吡罗昔康(优选吡罗昔康倍他环糊精)、丙戊酸盐(优选其半钠盐(semisodium salt))、双氯芬酸(优选钾盐)、噻加宾、肉毒杆菌毒素(botulinum)、奈比洛尔、赖洛普利、尼莫地平、替扎尼定、佐米曲坦、舒马普坦(优选其琥珀酸盐)、利扎曲坦(优选其苯甲酸盐)、苯噻啶、奥昔托隆、那拉曲坦、洛美利嗪(优选其盐酸盐)、吉培福林、氟桂利嗪、阿莫曲坦、阿吡必利、托芬那酸、migpriv、噻吗洛尔(优选其马来酸盐)、布克力嗪(优选其盐酸盐)、巴氯芬、美西麦角(优选其马来酸盐)、氟桂利嗪(优选其盐酸盐)、赛庚啶(优选其盐酸盐)、麦角胺(优选其酒石酸盐)、利多卡因(优选其盐酸盐)、吲哚拉明(优选其盐酸盐)、布托啡诺、KT 2962、BMS 181885、ADDS-麦角胺、NPS-1776、GW-468816、阿米替林、药物开发号6313、MT-500、多尼普曲坦(优选其甲磺酸盐)、ALX-0646、civamide、心得安、珠卡赛辛、CNS5161、沃氟匹坦、拉奈匹坦、达匹坦、加奈索酮、LY-53857、麦角克索(优选其马来酸盐)、舒马普坦、MT-400、氟西汀、(S)-氟西汀、二氢麦角胺(优选其甲磺酸盐)、托那博沙、IS-159、BIBN-4096、甲氧氯普胺、萘普生、MT-100(即甲氧氯普胺和萘普生的组合)、多他利嗪、夫罗曲普坦、依来曲普坦、阿司匹林、布洛芬、对乙酰氨基酚、阿米替林(amitryptiline)、多塞平、麦角制剂、咖啡因、咖啡角(例如咖啡因和麦角胺的组合)、可待因、哌替啶、异丙嗪、阿托品、苯巴比妥、硝苯地平、维拉帕米、氯丙嗪、锂、泼尼松、普萘洛尔、苯乙肼、甲灭酸、氟芬那酸、LY334370、吲哚美辛、氯醛比林、异美汀、布他比妥、酮咯酸、氯硝西泮、阿替洛尔、美托洛尔、纳多洛尔、丙米嗪、去甲替林(nortripyline)、地尔硫卓、丙戊酸、双丙戊酸钠或赛庚啶。抗偏头痛剂可以商购得到,也可以由本领域已知的方法制备得到。Anti-migraine agents for use in the methods and compositions described herein can be any known in the art. Anti-migraine agents include, for example, serotonin antagonists, non-steroidal anti-inflammatory drugs (NSAIDs) (eg, COX-1 inhibitors and/or COX-2 inhibitors), calcium channel blockers, beta-adrenergic blockers anticonvulsants, and antidepressants (such as tricyclic antidepressants, monoamine oxidase inhibitors, and selective serotonin reuptake inhibitors). Other exemplary anti-migraine agents include PNU-142633, vigabatrin, topiramate, montelukast (preferably its sodium salt), gabapentin, piroxicam (preferably piroxicam beta cyclodextrin), valproate ( Preferably its semisodium salt (semisodium salt)), diclofenac (preferably potassium salt), tiagabine, botulinum, nebivolol, lyslopril, nimodipine, tizanidine, adjuvant Mitriptan, sumatriptan (preferably its succinate), rizatriptan (preferably its benzoate), phenthiazine, oxytolone, naratriptan, lomerizine (preferably its hydrochloride), gilpephrine, flunarizine, almotriptan, alpiropride, tolfenamic acid, migpriv, timolol (preferably its maleate), bucrizine ( preferably its hydrochloride), baclofen, methysergide (preferably its maleate), flunarizine (preferably its hydrochloride), cyproheptadine (preferably its hydrochloride), ergotamine (preferably its tartrate), lidocaine (preferably its hydrochloride), indolamine (preferably its hydrochloride), butorphanol, KT 2962, BMS 181885, ADDS-ergotamine, NPS-1776, GW- 468816, Amitriptyline, Drug Development No. 6313, MT-500, Donipratriptan (preferably its mesylate), ALX-0646, civamide, Propranolol, Zurcasecin, CNS5161, Woflupitant , lanepitant, dapitant, ganaxolone, LY-53857, ergoxol (preferably its maleate), sumatriptan, MT-400, fluoxetine, (S)-fluoxetine , dihydroergotamine (preferably its mesylate), tonabersat, IS-159, BIBN-4096, metoclopramide, naproxen, MT-100 (i.e. metoclopramide and naproxen combination), dotazine, frovatriptan, eletriptan, aspirin, ibuprofen, acetaminophen, amitryptiline, doxepin, ergot preparations, caffeine , caffeine (such as a combination of caffeine and ergotamine), codeine, meperidine, promethazine, atropine, phenobarbital, nifedipine, verapamil, chlorpromazine, lithium, prednisolone Pine, propranolol, phenelzine, mefenamic acid, flufenamic acid, LY334370, indomethacin, chloralpyrine, Isometheptene, butalbital, ketorolac, clonazepam, Atenolol, metoprolol, nadolol, imipramine, nortriptyline, diltiazem, valproic acid, divalproex sodium, or cyproheptadine. Anti-migraine agents are commercially available or can be prepared by methods known in the art.
在其它实施方案中,本发明提供了药物组合物,其中含有治疗有效量的:(i)至少一种1,2-二氢吡啶化合物、(ii)至少一种胆碱酯酶抑制剂以及(iii)至少一种可药用赋型剂。本发明还提供了组合,其中含有治疗有效量的:(i)至少一种1,2-二氢吡啶化合物和(ii)至少一种胆碱酯酶抑制剂;其中所述化合物可以单独给药(例如同时、相继)至患者以治疗所述疾病或障碍。本发明提供了商品化包装(例如试剂盒),其中含有治疗有效量的:(i)至少一种1,2-二氢吡啶化合物,(ii)至少一种胆碱酯酶抑制剂;和(iii)用于同时、单独或相继使用(i)和(ii)治疗本文所述疾病和障碍的说明。所述1,2-二氢吡啶化合物可以是本文所述的任何一种。例如所述1,2-二氢吡啶化合物可以是式(I)化合物、式(II)化合物、式(III)化合物、化合物A。所述胆碱酯酶抑制剂可以是本文所述的任何一种。例如所述胆碱酯酶抑制剂可以是式(IV)化合物、式(V)化合物、式(VI)化合物、式(B)化合物、式(B1)化合物、式(B2)化合物、式(B3)化合物、式(B4)化合物或式(B5)化合物。在其它实施方案中,所述胆碱酯酶抑制剂可以是他克林、毒扁豆碱、吡啶斯的明、新斯的明、利伐斯的明,加兰他敏、胞磷胆碱、维吖啶、石杉碱(例如石杉碱A)、美曲磷酯、heptastigmine、腾喜隆、苯羟基丙氨酸(phenserine)、tolserine、phenethylnorcymserine、奎洛斯的明、更斯的明、epastigmine、upreazine、3-[1-(苯甲基)-4-哌啶基]-1-(2,3,4,5-四氢-1H-1-苯并氮杂-8-基)-1-丙酮或(2-[2-(1-苄基哌啶-4-基)乙基]-2,3-二氢-9-甲氧基-1H-吡咯并[3,4-b]喹啉-1-酮。在一实施方案中,本发明提供了药物组合物,其中含有治疗有效量的:(i)多奈哌齐;(ii)3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮;和(iii)至少一种可药用赋型剂。所述药物组合物可以任选进一步含有至少一种抗偏头痛剂。In other embodiments, the present invention provides pharmaceutical compositions comprising a therapeutically effective amount of: (i) at least one 1,2-dihydropyridine compound, (ii) at least one cholinesterase inhibitor, and ( iii) at least one pharmaceutically acceptable excipient. The present invention also provides combinations comprising therapeutically effective amounts of: (i) at least one 1,2-dihydropyridine compound and (ii) at least one cholinesterase inhibitor; wherein said compounds are administered alone (eg simultaneously, sequentially) to a patient to treat the disease or disorder. The invention provides a commercial package (eg, a kit) containing a therapeutically effective amount of: (i) at least one 1,2-dihydropyridine compound, (ii) at least one cholinesterase inhibitor; and ( iii) Instructions for the simultaneous, separate or sequential use of (i) and (ii) in the treatment of the diseases and disorders described herein. The 1,2-dihydropyridine compound may be any one described herein. For example, the 1,2-dihydropyridine compound may be a compound of formula (I), a compound of formula (II), a compound of formula (III), or compound A. The cholinesterase inhibitor can be any one described herein. Such as described cholinesterase inhibitor can be formula (IV) compound, formula (V) compound, formula (VI) compound, formula (B) compound, formula (B1) compound, formula (B2) compound, formula (B3 ) compound, compound of formula (B4) or compound of formula (B5). In other embodiments, the cholinesterase inhibitor may be tacrine, physostigmine, pyridostigmine, neostigmine, rivastigmine, galantamine, citicoline, Viacridine, huperzine (e.g. huperzine A), metrifos, heptastigmine, Tensiron, phenserine, tolserine, phenylnorcymserine, quinostigmine, ganstigmine, epastigmine, upreazine, 3-[1-(phenylmethyl)-4-piperidinyl]-1-(2,3,4,5-tetrahydro-1H-1-benzazepine -8-yl)-1-propanone or (2-[2-(1-benzylpiperidin-4-yl)ethyl]-2,3-dihydro-9-methoxy-1H-pyrrolo[ 3,4-b] quinolin-1-one. In one embodiment, the invention provides a pharmaceutical composition comprising a therapeutically effective amount of: (i) donepezil; (ii) 3-(2-cyanobenzene base)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one; and (iii) at least one pharmaceutically acceptable excipient. The pharmaceutical composition can be optionally further comprising at least one anti-migraine agent.
本发明提供了药物组合物,其中含有治疗有效量的:(i)至少一种1,2-二氢吡啶化合物、(ii)至少一种抗偏头痛剂以及(iii)至少一种可药用赋型剂。所述1,2-二氢吡啶化合物可以是本文所述的任何一种。例如所述1,2-二氢吡啶化合物可以是式(I)化合物、式(II)化合物、式(III)化合物、化合物A。所述抗偏头痛剂可以是本文所述的任何一种。例如所述抗偏头痛剂可以是阿司匹林、布洛芬、对乙酰氨基酚、双氯芬酸、非诺洛芬、ketaprofen、酮咯酸、氟比洛芬、甲氯芬那酸盐(meclofenamate)、萘普生、麦角胺、舒马普坦、佐米曲坦、利扎曲普坦、naratriptin、almotriptin、夫罗曲普坦、依立曲坦、阿米替林、地昔帕明、多塞平、丙米嗪、nortripytyline、氟西汀、帕罗西汀、舍曲林、venlafazine、曲唑酮、丁氨苯丙酮、阿替洛尔、美托洛尔、纳多洛尔、普萘洛尔、噻吗洛尔、地尔硫卓、尼卡地平、硝苯地平、尼莫地平、维拉帕米、双丙戊酸钠、加巴喷丁、丙戊酸或托吡酯。在一实施方案中,所述抗偏头痛剂是阿司匹林、布洛芬、扑热息痛或萘普生。在另一实施方案中,所述抗偏头痛剂是麦角胺、二氢麦角胺、舒马普坦,佐米曲坦、利扎曲普坦、那拉曲坦或阿莫曲坦。所述药物组合物可以任选进一步含有至少一种胆碱酯酶抑制剂。The present invention provides a pharmaceutical composition comprising a therapeutically effective amount of: (i) at least one 1,2-dihydropyridine compound, (ii) at least one anti-migraine agent, and (iii) at least one pharmaceutically acceptable Excipients. The 1,2-dihydropyridine compound may be any one described herein. For example, the 1,2-dihydropyridine compound may be a compound of formula (I), a compound of formula (II), a compound of formula (III), or compound A. The anti-migraine agent can be any of those described herein. For example the anti-migraine agent may be aspirin, ibuprofen, acetaminophen, diclofenac, fenoprofen, ketaprofen, ketorolac, flurbiprofen, meclofenamate, naproxen ergotamine, sumatriptan, zolmitriptan, rizatriptan, naratriptin, almotriptin, frovatriptan, eletriptan, amitriptyline, desipramine, doxepin, Imipramine, nortripytyline, fluoxetine, paroxetine, sertraline, venlafazine, trazodone, bupropion, atenolol, metoprolol, nadolol, propranolol, timolol Lore, diltiazem, nicardipine, nifedipine, nimodipine, verapamil, divalproex sodium, gabapentin, valproic acid, or topiramate. In one embodiment, the anti-migraine agent is aspirin, ibuprofen, paracetamol or naproxen. In another embodiment, the anti-migraine agent is ergotamine, dihydroergotamine, sumatriptan, zolmitriptan, rizatriptan, naratriptan or almotriptan. The pharmaceutical composition may optionally further contain at least one cholinesterase inhibitor.
本发明提供了治疗和/或预防有需要的患者中头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗头痛的方法包括(i)降低头痛频率的方法,(ii)降低头痛严重程度的方法,(iii)降低头痛持续时间的方法,(iv)降低头痛频率和严重程度的方法,(v)降低头痛频率和持续时间的方法,(vi)降低头痛严重程度和持续时间的方法,以及(vii)降低头痛频率、严重程度和持续时间的方法。治疗头痛的方法包括治疗由头痛引起的一种或多种症状。头痛可以是原发性头痛或继发性头痛。所述1,2-二氢吡啶化合物和所述胆碱酯酶抑制剂和/或抗偏头痛剂可以单独向患者给药,也可以以药物组合物形式给药。The present invention provides a method of treating and/or preventing headache in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b) ; wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and at least one An anti-migraine agent. Methods of treating headaches include (i) methods to reduce the frequency of headaches, (ii) methods to reduce the severity of headaches, (iii) methods to reduce the duration of headaches, (iv) methods to reduce the frequency and severity of headaches, (v) methods to reduce Methods of headache frequency and duration, (vi) methods of reducing headache severity and duration, and (vii) methods of reducing headache frequency, severity and duration. Methods of treating headaches include treating one or more symptoms caused by the headache. Headaches can be primary or secondary. The 1,2-dihydropyridine compound and the cholinesterase inhibitor and/or anti-migraine agent can be administered to the patient alone or in the form of a pharmaceutical composition.
本发明提供了治疗和/或预防有需要的患者中原发性头痛或继发性头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗原发性头痛或继发性头痛的方法包括(i)降低原发性头痛或继发性头痛频率的方法,(ii)降低原发性头痛或继发性头痛严重程度的方法,(iii)降低原发性头痛或继发性头痛持续时间的方法,(iv)降低原发性头痛或继发性头痛频率和严重程度的方法,(v)降低原发性头痛或继发性头痛频率和持续时间的方法,(vi)降低原发性头痛或继发性头痛严重程度和持续时间的方法,以及(vii)降低原发性头痛或继发性头痛频率、严重程度和持续时间的方法。治疗原发性头痛或继发性头痛的方法包括治疗由原发性头痛或继发性头痛引起的一种或多种症状。所述1,2-二氢吡啶化合物和所述胆碱酯酶抑制剂和/或抗偏头痛剂可以单独向患者给药,也可以以药物组合物形式给药。The present invention provides a method of treating and/or preventing primary or secondary headache in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinergic agent Alkaline esterase inhibitors and at least one anti-migraine agent. Methods of treating primary or secondary headaches include (i) methods of reducing the frequency of primary or secondary headaches, (ii) methods of reducing the severity of primary or secondary headaches, (iii) ) a method of reducing the duration of a primary or secondary headache, (iv) a method of reducing the frequency and severity of a primary or secondary headache, (v) reducing the frequency of a primary or secondary headache and duration, (vi) methods of reducing the severity and duration of primary or secondary headaches, and (vii) methods of reducing the frequency, severity, and duration of primary or secondary headaches . Methods of treating primary or secondary headaches include treating one or more symptoms caused by primary or secondary headaches. The 1,2-dihydropyridine compound and the cholinesterase inhibitor and/or anti-migraine agent can be administered to the patient alone or in the form of a pharmaceutical composition.
本发明提供了治疗和/或预防有需要的患者中偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗偏头痛的方法包括(i)降低偏头痛频率的方法,(ii)降低偏头痛严重程度的方法,(iii)降低偏头痛持续时间的方法,(iv)降低偏头痛频率和严重程度的方法,(v)降低偏头痛频率和持续时间的方法,(vi)降低偏头痛严重程度和持续时间的方法,以及(vii)降低偏头痛频率、严重程度和持续时间的方法。治疗偏头痛的方法包括治疗由偏头痛引起的一种或多种症状。所述1,2-二氢吡啶化合物和所述胆碱酯酶抑制剂和/或抗偏头痛剂可以向患者单独给药,也可以以药物组合物形式给药。The present invention provides a method of treating and/or preventing migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b ); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and at least An anti-migraine agent. Methods of treating migraines include (i) methods to reduce the frequency of migraines, (ii) methods to reduce the severity of migraines, (iii) methods to reduce the duration of migraines, (iv) methods to reduce the frequency and severity of migraines , (v) methods of reducing the frequency and duration of migraines, (vi) methods of reducing the severity and duration of migraines, and (vii) methods of reducing the frequency, severity and duration of migraines. Methods of treating migraines include treating one or more symptoms caused by migraines. The 1,2-dihydropyridine compound and the cholinesterase inhibitor and/or anti-migraine agent can be administered to the patient alone or in the form of a pharmaceutical composition.
本发明提供了治疗和/或预防有需要的患者中典型偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗典型偏头痛的方法包括(i)降低典型偏头痛频率的方法,(ii)降低典型偏头痛严重程度的方法,(iii)降低典型偏头痛持续时间的方法,(iv)降低典型偏头痛频率和严重程度的方法,(v)降低典型偏头痛频率和持续时间的方法,(vi)降低典型偏头痛严重程度和持续时间的方法,以及(vii)降低典型偏头痛频率、严重程度和持续时间的方法。治疗典型偏头痛的方法包括治疗由典型偏头痛引起的一种或多种症状。"典型偏头痛"通常由神经症状状开始,例如视觉闪烁(visual scintillation)、刺眼锯齿状光线(dazzling zigzag lines)、畏光和盲点扩大(spreading scotomas)或者眩晕和耳鸣。典型偏头痛可能具有先兆症状,例如感觉情绪高涨(feelings of elation)、精力过度(excessive energy)、口渴、嗜糖和/或困倦。在其它时候,典型偏头痛可能有先兆症状,例如精神活动迟缓、感觉被迫(impending doom)和/或抑郁。在其它时候,可能没有先兆症状。The present invention provides a method of treating and/or preventing typical migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally ( b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and At least one anti-migraine agent. Methods of treating typical migraines include (i) methods to reduce the frequency of typical migraines, (ii) methods to reduce the severity of typical migraines, (iii) methods to reduce the duration of typical migraines, (iv) methods to reduce the frequency of typical migraines and severity, (v) methods to reduce the frequency and duration of typical migraines, (vi) methods to reduce the severity and duration of typical migraines, and (vii) methods to reduce the frequency, severity and duration of typical migraines Methods. Methods of treating classic migraine include treating one or more symptoms caused by classic migraine. "Classic migraine" usually begins with neurological symptoms such as visual scintillation, dazzling zigzag lines, photophobia and spreading scotomas, or vertigo and tinnitus. A typical migraine may have aura symptoms such as feelings of elation, excessive energy, thirst, sugar cravings, and/or sleepiness. At other times, a typical migraine may have aura symptoms, such as mental retardation, feeling impending doom, and/or depression. At other times, there may be no aura symptoms.
本发明提供了治疗和/或预防有需要的患者中普通型偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗普通型偏头痛的方法包括(i)降低普通型偏头痛频率的方法,(ii)降低普通型偏头痛严重程度的方法,(iii)降低普通型偏头痛持续时间的方法,(iv)降低普通型偏头痛频率和严重程度的方法,(v)降低普通型偏头痛频率和持续时间的方法,(vi)降低普通型偏头痛严重程度和持续时间的方法,以及(vii)降低普通型偏头痛频率、严重程度和持续时间的方法。治疗普通型偏头痛的方法包括治疗由普通型偏头痛引起的一种或多种症状。"普通型偏头痛"通常是不受控制地头痛发作,可能伴随恶心和/或呕吐。与典型偏头痛不同的是,普通型偏头痛在头痛发作之前通常没有神经症状。The present invention provides a method of treating and/or preventing migraine vulgaris in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and at least one anti-migraine agent. Methods of treating migraine vulgaris include (i) methods of reducing the frequency of migraine vulgaris, (ii) methods of reducing the severity of migraine vulgaris, (iii) methods of reducing the duration of migraine vulgaris, (iv) reducing Methods of reducing frequency and duration of general migraine, (v) methods of reducing frequency and duration of general migraine, (vi) methods of reducing severity and duration of general migraine, and (vii) reducing frequency and duration of general migraine Methods for headache frequency, severity, and duration. Methods of treating migraine vulgaris include treating one or more symptoms caused by migraine vulgaris. A "common migraine" is usually an uncontrolled attack of headache that may be accompanied by nausea and/or vomiting. Unlike typical migraine, migraine vulgaris usually has no neurological symptoms before the onset of the headache.
本发明提供了治疗和/或预防有需要的患者中偏头痛型脑梗死(complicated migraines)的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗偏头痛型脑梗死的方法包括(i)降低偏头痛型脑梗死频率的方法,(ii)降低偏头痛型脑梗死严重程度的方法,(iii)降低偏头痛型脑梗死持续时间的方法,(iv)降低偏头痛型脑梗死频率和严重程度的方法,(v)降低偏头痛型脑梗死频率和持续时间的方法,(vi)降低偏头痛型脑梗死严重程度和持续时间的方法,以及(vii)降低偏头痛型脑梗死频率、严重程度和持续时间的方法。治疗偏头痛型脑梗死的方法包括治疗由偏头痛型脑梗死引起的一种或多种症状。"偏头痛型脑梗死"是指在头痛之前或同时伴随有神经症状(例如典型偏头痛所述的神经症状)的偏头痛。在偏头痛型脑梗死中,可能在身体一侧的嘴唇、脸、手、臂和/或腿出现麻木和麻感,有时还并发失语障碍。在一侧臂和/或腿可能变得虚弱或瘫痪,跟中风类似。这种麻木和虚弱可能在数分钟内从身体一部分缓慢蔓延至另一部分。"偏头痛型脑梗死"包括基底性偏头痛。在基底性(basilar)偏头痛中,视觉障碍和感觉异常出现在两侧,并且可能伴随着意识模糊、麻痹、昏迷、攻击性爆发(aggressive outburst)、眩晕、复视和/或发音困难。30%的偏头痛儿童出现基底性偏头痛。The present invention provides a method of treating and/or preventing complicated migraines in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinergic agent Alkaline esterase inhibitors and at least one anti-migraine agent. The method for treating migraineous cerebral infarction includes (i) a method for reducing the frequency of migraineous cerebral infarction, (ii) a method for reducing the severity of migraineous cerebral infarction, (iii) a method for reducing the duration of migraineous cerebral infarction, (iv) methods of reducing the frequency and severity of migrainous infarctions, (v) methods of reducing the frequency and duration of migrainous infarctions, (vi) methods of reducing the severity and duration of migrainous infarctions, and (vii) Methods of reducing the frequency, severity and duration of migraineous infarctions. The method of treating migraineous cerebral infarction includes treating one or more symptoms caused by migraineous cerebral infarction. "Migrainous cerebral infarction" refers to migraine headache preceded by or accompanied by neurological symptoms (such as neurological symptoms described in typical migraine). In a migraine infarction, numbness and tingling may occur on one side of the body in the lips, face, hand, arm, and/or leg, sometimes accompanied by aphasia. The arm and/or leg may become weak or paralyzed on one side, similar to a stroke. This numbness and weakness may slowly spread from one part of the body to another over a period of minutes. "Migrainous cerebral infarction" includes basal migraine. In basilar migraine, visual disturbances and paresthesias are bilateral and may be accompanied by confusion, paralysis, coma, aggressive outburst, vertigo, diplopia, and/or dysarthria. Basal migraine occurs in 30% of children with migraine.
本发明提供了治疗和/或预防有需要的患者中月经性偏头痛或月经前偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗月经性偏头痛或月经前偏头痛的方法包括(i)降低月经性偏头痛或月经前偏头痛频率的方法,(ii)降低月经性偏头痛或月经前偏头痛严重程度的方法,(iii)降低月经性偏头痛或月经前偏头痛持续时间的方法,(iv)降低月经性偏头痛或月经前偏头痛频率和严重程度的方法,(v)降低月经性偏头痛或月经前偏头痛频率和持续时间的方法,(vi)降低月经性偏头痛或月经前偏头痛严重程度和持续时间的方法,以及(vii)降低月经性偏头痛或月经前偏头痛频率、严重程度和持续时间的方法。治疗月经性偏头痛或月经前偏头痛的方法包括治疗由月经性偏头痛或月经前偏头痛引起的一种或多种症状。"月经性偏头痛"是指通常出现在妇女月经周期前大约两天直到妇女月经周期后大约三天结束的偏头痛。在另一实施方案中,月经性偏头痛是指通常出现在妇女月经周期前大约两天直到妇女月经周期最后一天结束的偏头痛。月经性偏头痛可能在月经周期期间任何时间出现或再次出现。"月经前偏头痛"是指通常出现在妇女月经周期前大约七天直到妇女月经周期前大约三天结束的偏头痛。月经前偏头痛可能出现或再次出现在月经周期期间的任何时间。The present invention provides a method of treating and/or preventing menstrual migraine or premenstrual migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinergic agent Alkaline esterase inhibitors and at least one anti-migraine agent. Methods of treating menstrual or premenstrual migraine include (i) methods of reducing the frequency of menstrual or premenstrual migraine, (ii) methods of reducing the severity of menstrual or premenstrual migraine, (iii) ) a method of reducing the duration of menstrual or premenstrual migraine, (iv) a method of reducing the frequency and severity of menstrual or premenstrual migraine, (v) reducing the frequency of menstrual or premenstrual migraine and duration, (vi) methods of reducing the severity and duration of menstrual or premenstrual migraine, and (vii) methods of reducing the frequency, severity, and duration of menstrual or premenstrual migraine . The method of treating menstrual migraine or premenstrual migraine includes treating one or more symptoms caused by menstrual migraine or premenstrual migraine. "Menstrual migraine"means a migraine that usually occurs about two days before a woman's menstrual cycle and ends about three days after a woman's menstrual cycle. In another embodiment, menstrual migraine refers to migraine that typically occurs about two days before a woman's menstrual cycle until the end of the last day of the woman's menstrual cycle. Menstrual migraines may occur or recur at any time during the menstrual cycle. "Premenstrual migraine"means a migraine that usually occurs about seven days before a woman's menstrual cycle and ends about three days before her menstrual cycle. Premenstrual migraines may occur or recur at any time during the menstrual cycle.
本发明提供了治疗和/或预防有需要的患者中眼型偏头痛或眼肌麻痹性偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗眼型偏头痛或眼肌麻痹性偏头痛的方法包括(i)降低眼型偏头痛或眼肌麻痹性偏头痛频率的方法,(ii)降低眼型偏头痛或眼肌麻痹性偏头痛严重程度的方法,(iii)降低眼型偏头痛或眼肌麻痹性偏头痛持续时间的方法,(iv)降低眼型偏头痛或眼肌麻痹性偏头痛频率和严重程度的方法,(v)降低眼型偏头痛或眼肌麻痹性偏头痛频率和持续时间的方法,(vi)降低眼型偏头痛或眼肌麻痹性偏头痛严重程度和持续时间的方法,以及(vii)降低眼型偏头痛或眼肌麻痹性偏头痛频率、严重程度和持续时间的方法。治疗眼型偏头痛或眼肌麻痹性偏头痛的方法包括治疗由眼型偏头痛或眼肌麻痹性偏头痛引起的一种或多种症状。"眼型偏头痛"是指通常伴随着明显视觉障碍的偏头痛。"眼肌麻痹性偏头痛"是指与眼部肌肉麻痹有关的偏头痛。The present invention provides a method of treating and/or preventing ocular migraine or ophthalmoplegic migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-bis A hydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one antimigraine agent; or (iii) at least one a cholinesterase inhibitor and at least one anti-migraine agent. Methods of treating ocular or ophthalmoplegic migraine include (i) methods of reducing the frequency of ocular or ophthalmoplegic migraine, (ii) reducing the severity of ocular or ophthalmoplegic migraine extent, (iii) methods of reducing the duration of ocular or ophthalmoplegic migraine, (iv) methods of reducing the frequency and severity of ocular or ophthalmoplegic migraine, (v) methods of reducing Methods of Ocular Migraine or Ophthalmoplegic Migraine Frequency and Duration, (vi) Methods of Reducing Ocular Migraine or Ophthalmoplegic Migraine Severity and Duration, and (vii) Reducing Ocular Migraine or ophthalmoplegic migraine frequency, severity, and duration. Methods of treating ocular migraine or ophthalmoplegic migraine include treating one or more symptoms caused by ocular migraine or ophthalmoplegic migraine. "Ocular migraine" refers to migraine that is usually accompanied by significant visual disturbance. "Ophthalmoplegic migraine" means a migraine associated with paralysis of the eye muscles.
本发明提供了治疗和/或预防有需要的患者中闪电状偏头痛(fulguratingmigraines)的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗闪电状偏头痛的方法包括(i)降低闪电状偏头痛频率的方法,(ii)降低闪电状偏头痛严重程度的方法,(iii)降低闪电状偏头痛持续时间的方法,(iv)降低闪电状偏头痛频率和严重程度的方法,(v)降低闪电状偏头痛频率和持续时间的方法,(vi)降低闪电状偏头痛严重程度和持续时间的方法,以及(vii)降低闪电状偏头痛频率、严重程度和持续时间的方法。治疗闪电状偏头痛的方法包括治疗由闪电状偏头痛引起的一种或多种症状。"闪电状偏头痛"是指其特征在于突然开始并且严重的偏头痛。The present invention provides a method of treating and/or preventing fulfilling migraines in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and Optional (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase Enzyme inhibitors and at least one anti-migraine agent. Methods of treating lightning migraines include (i) methods of reducing the frequency of lightning migraines, (ii) methods of reducing the severity of lightning migraines, (iii) methods of reducing the duration of lightning migraines, (iv) reducing Methods of Lightning Migraine Frequency and Severity, (v) Methods of Reducing Lightning Migraine Frequency and Duration, (vi) Methods of Reducing Lightning Migraine Severity and Duration, and (vii) Reducing Lightning Migraine Methods for headache frequency, severity, and duration. Treatments for lightning migraines include treating one or more of the symptoms caused by lightning migraines. "Lightning migraine" refers to a migraine characterized by sudden onset and severity.
本发明提供了治疗和/或预防有需要的患者中Harris偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗Harris偏头痛的方法包括(i)降低Harris偏头痛频率的方法,(ii)降低Harris偏头痛严重程度的方法,(iii)降低Harris偏头痛持续时间的方法,(iv)降低Harris偏头痛频率和严重程度的方法,(v)降低Harris偏头痛频率和持续时间的方法,(vi)降低Harris偏头痛严重程度和持续时间的方法,以及(vii)降低Harris偏头痛频率、严重程度和持续时间的方法。治疗Harris偏头痛的方法包括治疗由Harris偏头痛引起的一种或多种症状。"Harris偏头痛"还被称作周期性偏头痛神经痛。The present invention provides a method for treating and/or preventing Harris migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally ( b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and At least one anti-migraine agent. Methods of treating Harris migraines include (i) methods of reducing the frequency of Harris migraines, (ii) methods of reducing the severity of Harris migraines, (iii) methods of reducing the duration of Harris migraines, (iv) methods of reducing the frequency of Harris migraines and severity, (v) methods to reduce the frequency and duration of Harris migraines, (vi) methods to reduce the severity and duration of Harris migraines, and (vii) methods to reduce the frequency, severity and duration of Harris migraines Methods. Methods of treating Harris migraine include treating one or more symptoms caused by Harris migraine. "Harris migraine" is also known as periodic migraine neuralgia.
本发明提供了治疗和/或预防有需要的患者中偏瘫性偏头痛的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗偏瘫性偏头痛的方法包括(i)降低偏瘫性偏头痛频率的方法,(ii)降低偏瘫性偏头痛严重程度的方法,(iii)降低偏瘫性偏头痛持续时间的方法,(iv)降低偏瘫性偏头痛频率和严重程度的方法,(v)降低偏瘫性偏头痛频率和持续时间的方法,(vi)降低偏瘫性偏头痛严重程度和持续时间的方法,以及(vii)降低偏瘫性偏头痛频率、严重程度和持续时间的方法。治疗偏瘫性偏头痛的方法包括治疗由偏瘫性偏头痛引起的一种或多种症状。"偏瘫性偏头痛"是指与短暂性偏瘫有关的偏头痛形式。The present invention provides a method of treating and/or preventing hemiplegic migraine in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and optionally (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitor and at least one anti-migraine agent. Methods of treating hemiplegic migraines include (i) methods of reducing the frequency of hemiplegic migraines, (ii) methods of reducing the severity of hemiplegic migraines, (iii) methods of reducing the duration of hemiplegic migraines, (iv) methods of reducing Methods for the frequency and severity of hemiplegic migraines, (v) methods for reducing the frequency and duration of hemiplegic migraines, (vi) methods for reducing the severity and duration of hemiplegic migraines, and (vii) methods for reducing the frequency and duration of hemiplegic migraines Methods for headache frequency, severity, and duration. Methods of treating hemiplegic migraine include treating one or more symptoms caused by hemiplegic migraine. "Hemiplegic migraine" refers to a form of migraine associated with transient hemiplegia.
本发明提供了治疗和/或预防有需要的患者中腹型偏头痛(abdominalmigraines)的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗腹型偏头痛的方法包括(i)降低腹型偏头痛频率的方法,(ii)降低腹型偏头痛严重程度的方法,(iii)降低腹型偏头痛持续时间的方法,(iv)降低腹型偏头痛频率和严重程度的方法,(v)降低腹型偏头痛频率和持续时间的方法,(vi)降低腹型偏头痛严重程度和持续时间的方法,以及(vii)降低腹型偏头痛频率、严重程度和持续时间的方法。治疗腹型偏头痛的方法包括治疗由腹型偏头痛引起的一种或多种症状。"腹型偏头痛"其特征在于没有明显原因就出现阵发性腹部疼痛。The present invention provides a method of treating and/or preventing abdominal migraines in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and any Optional (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitors and at least one anti-migraine agent. Methods of treating abdominal migraine include (i) methods of reducing the frequency of abdominal migraine, (ii) methods of reducing the severity of abdominal migraine, (iii) methods of reducing the duration of abdominal migraine, (iv) methods of reducing Methods for reducing the frequency and duration of abdominal migraine, (v) methods for reducing the frequency and duration of abdominal migraine, (vi) methods for reducing the severity and duration of abdominal migraine, and (vii) methods for reducing the frequency and duration of abdominal migraine Methods for headache frequency, severity, and duration. Methods of treating abdominal migraine include treating one or more symptoms caused by abdominal migraine. "Abdominal migraine" is characterized by episodic abdominal pain with no apparent cause.
本发明提供了治疗和/或预防有需要的患者中丛集性头痛(clusterheadaches)的方法,所述方法包括给药治疗有效量的:(a)至少一种1,2-二氢吡啶化合物以及任选的(b);其中(b)是治疗有效量的(i)至少一种胆碱酯酶抑制剂;(ii)至少一种抗偏头痛剂;或(iii)至少一种胆碱酯酶抑制剂和至少一种抗偏头痛剂。治疗丛集性头痛的方法包括(i)降低丛集性头痛频率的方法,(ii)降低丛集性头痛严重程度的方法,(iii)降低丛集性头痛持续时间的方法,(iv)降低丛集性头痛频率和严重程度的方法,(v)降低丛集性头痛频率和持续时间的方法,(vi)降低丛集性头痛严重程度和持续时间的方法,以及(vii)降低丛集性头痛频率、严重程度和持续时间的方法。治疗丛集性头痛的方法包括治疗由丛集性头痛引起的一种或多种症状。"丛集性头痛"也被称作阵发性夜间头痛、偏头痛神经痛、组胺头痛和Horton综合征。丛集性头痛其特征在于通常在入睡后2或3小时内发作的持久性单侧眼眶痛。这种疼痛可能是剧烈和平稳的,同时伴随着流泪、鼻孔阻滞,随后出现鼻漏,有时还会瞳孔缩小、上睑下垂、面红和面颊水肿。The present invention provides a method of treating and/or preventing cluster headaches in a patient in need thereof, said method comprising administering a therapeutically effective amount of: (a) at least one 1,2-dihydropyridine compound and any Optional (b); wherein (b) is a therapeutically effective amount of (i) at least one cholinesterase inhibitor; (ii) at least one anti-migraine agent; or (iii) at least one cholinesterase inhibitors and at least one anti-migraine agent. Methods of treating cluster headaches include (i) methods of reducing the frequency of cluster headaches, (ii) methods of reducing the severity of cluster headaches, (iii) methods of reducing the duration of cluster headaches, (iv) methods of reducing the frequency of cluster headaches and severity, (v) methods to reduce the frequency and duration of cluster headaches, (vi) methods to reduce the severity and duration of cluster headaches, and (vii) methods to reduce the frequency, severity and duration of cluster headaches Methods. Methods of treating cluster headaches include treating one or more symptoms caused by cluster headaches. "Cluster headache" is also known as episodic nocturnal headache, migraine neuralgia, histamine headache, and Horton's syndrome. Cluster headaches are characterized by persistent unilateral orbital pain that usually begins within 2 or 3 hours of falling asleep. The pain may be sharp and steady, accompanied by tearing, nasal blockage, followed by rhinorrhea, and sometimes miosis, ptosis, flushing, and edema of the cheeks.
在其它实施方案中,本发明提供了治疗和/或预防恐慌发作(panic attack)和/或恐慌障碍的方法,所述方法包括给药治疗有效量的至少一种1,2-二氢吡啶化合物以及任选的至少一种抗焦虑剂。所述抗焦虑剂可以是本领域任何一种。示例性抗焦虑剂包括苯并二氮杂类、氮螺杂癸二酮类(azaspirodecanediones)或哌嗪衍生物。示例性苯并二氮杂类包括地西泮、阿普唑仑、氯氮、氯硝西泮、氯酸盐(clorazepate)、哈拉西泮、lorpam、奥沙西泮及其衍生物和其可药用盐。示例性氮螺杂癸二酮包括丁螺环酮及其衍生物和其可药用盐。示例性哌嗪衍生物包括双羟萘酸羟嗪和盐酸羟嗪及其衍生物和其可药用盐。所述1,2-二氢吡啶化合物和抗焦虑剂可以是单独给药,也可以以组合物形式给药。In other embodiments, the present invention provides methods of treating and/or preventing panic attacks and/or panic disorders comprising administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound and optionally at least one anxiolytic. The anxiolytic agent can be any one in the art. Exemplary anxiolytics include benzodiazepines Classes, azaspirodecanediones or piperazine derivatives. Exemplary benzodiazepines Classes include diazepam, alprazolam, chlordiazepoxide , clonazepam, chlorine Clorazepate, halazepam, lorpam, oxazepam and their derivatives and their pharmaceutically acceptable salts. Exemplary azaspirodecanediones include buspirone and its derivatives and pharmaceutically acceptable salts thereof. Exemplary piperazine derivatives include hydroxyzine pamoate and hydroxyzine hydrochloride and derivatives thereof and pharmaceutically acceptable salts thereof. The 1,2-dihydropyridine compound and the anti-anxiety agent can be administered alone or in combination.
本发明提供了治疗和/或预防急性应急障碍(acute stress disorder)和/或一般性焦虑障碍的方法,所述方法包括给药治疗有效量的至少一种1,2-二氢吡啶化合物以及任选的至少一种抗焦虑剂。所述抗焦虑剂可以是本领域任何一种。示例性抗焦虑剂包括苯并二氮杂、氮螺杂癸二酮(azaspirodecanedione)或哌嗪衍生物。示例性苯并二氮杂包括地西泮、阿普唑仑、氯氮、氯硝西泮、氯酸盐、哈拉西泮、lorpam、奥沙西泮及其衍生物和其可药用盐。示例性氮螺杂癸二酮包括丁螺环酮及其衍生物和其可药用盐。示例性哌嗪衍生物包括双羟萘酸羟嗪和羟嗪盐酸盐及其衍生物和其可药用盐。所述1,2-二氢吡啶化合物和抗焦虑剂可以是单独给药,也可以以组合物形式给药。The present invention provides a method of treating and/or preventing acute stress disorder and/or general anxiety disorder, said method comprising administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound and any at least one anxiolytic of choice. The anxiolytic agent can be any one in the art. Exemplary anxiolytics include benzodiazepines , azaspirodecanedione or piperazine derivatives. Exemplary benzodiazepines Including diazepam, alprazolam, chlordiazepoxide , clonazepam, chlorine salt, halazepam, lorpam, oxazepam and their derivatives and their pharmaceutically acceptable salts. Exemplary azaspirodecanediones include buspirone and its derivatives and pharmaceutically acceptable salts thereof. Exemplary piperazine derivatives include hydroxyzine pamoate and hydroxyzine hydrochloride and derivatives and pharmaceutically acceptable salts thereof. The 1,2-dihydropyridine compound and the anti-anxiety agent can be administered alone or in combination.
本发明提供了使患者镇静的方法,所述方法包括给药治疗有效量的至少一种1,2-二氢吡啶化合物。在一实施方案中,本发明提供了在手术前使患者镇静的方法,所述方法包括给药治疗有效量的至少一种1,2-二氢吡啶化合物。在另一实施方案中,本发明提供了在有需要的患者中用于急性镇静进行手术麻醉的方法,所述方法包括给药治疗有效量的至少一种1,2-二氢吡啶化合物。The present invention provides methods of sedating a patient comprising administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound. In one embodiment, the present invention provides a method of sedating a patient prior to surgery comprising administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound. In another embodiment, the present invention provides a method of surgical anesthesia for acute sedation in a patient in need thereof, the method comprising administering a therapeutically effective amount of at least one 1,2-dihydropyridine compound.
所述1,2-二氢吡啶化合物和其它的本发明化合物(例如胆碱酯酶抑制剂、抗偏头痛剂、抗焦虑剂)可以以含有常规无毒性可药用载体、佐剂和载体(视需要)的剂量单位制剂形式通过口服、局部、非肠道(parenteral)、吸入(鼻或口腔)或直肠给药。术语非肠道包括皮下、静脉内、肌内、鞘内、胸骨内注射或输注技术。The 1,2-dihydropyridine compounds and other compounds of the present invention (such as cholinesterase inhibitors, anti-migraine agents, anxiolytics) may contain conventional non-toxic pharmaceutically acceptable carriers, adjuvants and carriers ( Dosage unit formulations as required) for oral, topical, parenteral, inhalation (nasal or buccal) or rectal administration. The term parenteral includes subcutaneous, intravenous, intramuscular, intrathecal, intrasternal injection or infusion techniques.
本发明1,2-二氢吡啶化合物(例如3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)的日剂量通常为大约30μg-10g,优选100μg-5g,或者在口服给药情形中更优选为100μg-100mg。对于注射给药,日剂量通常为大约30μg-1g,优选100μg-500mg,或者更优选100μg-30mg。化合物每天给药一次,或者每天分成数份给药。当用于剂量的上下文中时,所述重量数目是指1,2-二氢吡啶的重量,不包括其任意盐、抗衡离子和水合物等。因此,为了获得500毫克3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮的等价物,可能需要使用超过500毫克的化合物的可药用盐和/或水合物,这是因为可药用盐和/或水合物具有另外的重量。1,2-dihydropyridine compounds (such as 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one) of the present invention The daily dosage is usually about 30 μg-10 g, preferably 100 μg-5 g, or more preferably 100 μg-100 mg in the case of oral administration. For injection administration, the daily dose is usually about 30 μg-1 g, preferably 100 μg-500 mg, or more preferably 100 μg-30 mg. The compound is administered once daily, or in divided doses per day. When used in the context of dosage, stated weight numbers refer to the weight of 1,2-dihydropyridine, excluding any salts, counterions, hydrates, etc. thereof. Therefore, to obtain the equivalent of 500 mg of 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one, it may be necessary to use more than 500 mg of the pharmaceutically acceptable salt and/or hydrate of the compound because the pharmaceutically acceptable salt and/or hydrate has an additional weight.
本发明所述胆碱酯酶抑制剂(例如多奈哌齐)的日剂量通常为大约0.1毫克-100毫克,优选1毫克-50毫克,更优选5毫克-25毫克。在其它实施方案中,日剂量为10毫克-20毫克;或5毫克-10毫克。化合物每天给药一次,或者每天分成数份给药。当用于剂量的上下文中时,所述重量数目是指本发明所述胆碱酯酶抑制剂的重量,不包括其任意盐、抗衡离子等。因此,为了获得10毫克多奈哌齐的等价物,可能需要使用超过10毫克的多奈哌齐盐酸盐,这是因为盐酸盐具有另外的重量。The daily dosage of the cholinesterase inhibitor (such as donepezil) of the present invention is generally about 0.1 mg-100 mg, preferably 1 mg-50 mg, more preferably 5 mg-25 mg. In other embodiments, the daily dosage is between 10 mg and 20 mg; or between 5 mg and 10 mg. The compound is administered once daily, or in divided doses per day. When used in the context of dosage, said weight numbers refer to the weight of the cholinesterase inhibitor of the invention, excluding any salts, counterions, etc. thereof. Thus, to obtain the equivalent of 10 mg of donepezil, it may be necessary to use more than 10 mg of donepezil hydrochloride due to the additional weight of the hydrochloride.
本文所述的其它化合物(例如抗偏头痛剂、抗焦虑剂)可以以本领域熟知的剂量给药,参见例如The Physician′s Desk Reference、阐述化合物剂量的专利以及阐述化合物剂量的杂志。Other compounds described herein (eg, anti-migraine agents, anxiolytics) can be administered at dosages well known in the art, see, eg, The Physician's Desk Reference, patents describing compound dosages, and journals describing compound dosages.
在一实施方案中,给药方式是注射,例如皮下注射、肌内注射、静脉内注射或动脉内注射。注射制剂例如无菌注射用水性或油性混悬剂可以按照现有技术使用适宜的分散或润湿剂、助悬剂(例如甲基纤维素、聚山梨酯80、羟乙基纤维素、阿拉伯胶、粉状黄蓍胶、羧甲基纤维素钠、聚氧乙烯去水山梨糖醇单月桂酸酯等)、pH调节剂、缓冲剂、增溶剂(例如聚氧乙烯氢化蓖麻油、聚山梨酯80、烟酰胺、聚氧乙烯去水山梨糖醇单月桂酸酯、聚乙二醇(Macrogol)、蓖麻油脂肪酸的乙酯等)和防腐剂制备。无菌注射制剂还可以是在无毒性非肠道可接受的稀释剂或溶剂中的无菌注射溶液剂或混悬剂,例如在1,3-丁二醇中的溶液剂。可以使用的可接受载体和溶剂有水、Ringer溶液和等渗氯化钠溶液。此外,无菌的不挥发油通常也被用作溶剂或助悬溶媒。为此,可以使用任何一种温和的不挥发油,包括合成的甘油单或二酯,此外脂肪酸例如油酸也可以用于制备注射剂。这些制剂可以通过本领域已知的方法冻干。In one embodiment, the administration is by injection, eg, subcutaneously, intramuscularly, intravenously or intraarterially. Injectable preparations such as sterile injectable aqueous or oily suspensions can use suitable dispersing or wetting agents, suspending agents (such as methylcellulose, polysorbate 80, hydroxyethylcellulose, gum arabic, etc.) according to the prior art. , powdered tragacanth gum, sodium carboxymethylcellulose, polyoxyethylene sorbitan monolaurate, etc.), pH regulators, buffers, solubilizers (such as polyoxyethylene hydrogenated castor oil, polysorbate 80. Niacinamide, polyoxyethylene sorbitan monolaurate, polyethylene glycol (Macrogol), ethyl ester of castor oil fatty acid, etc.) and preservatives. The sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, for example a solution in 1,3-butanediol. Among the acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending vehicle. For this purpose any bland fixed oil may be employed including synthetic mono- or diglycerides, in addition fatty acids such as oleic acid may be used in the preparation of injectables. These formulations can be lyophilized by methods known in the art.
用于口服给药的固体剂型可以包括香口胶(chewing gum)、胶囊剂、片剂、舌下片剂、散剂、颗粒剂和凝胶剂。在这类固体剂型中,可以将活性化合物与一种或多种惰性稀释剂混合,例如乳糖或淀粉。在正通实践中,这类剂型还可以含有其它物质,包括润滑剂例如硬脂酸镁。在胶囊剂、片剂和丸剂的情形中,这些剂型还可以含有缓冲剂。片剂可以使用肠或膜包衣制备,优选膜包衣。Solid dosage forms for oral administration may include chewing gum, capsules, tablets, sublingual tablets, powders, granules and gels. In such solid dosage forms, the active compound may be admixed with one or more inert diluents, such as lactose or starch. In normal practice, such dosage forms may also contain other substances including lubricating agents such as magnesium stearate. In the case of capsules, tablets and pills, these dosage forms may also contain buffering agents. Tablets may be prepared with enteric or film coatings, film coatings being preferred.
为了制备片剂,可以将化合物与本领域已知的可药用载体混合,例如辅料(vehicle)(例如乳糖、白糖、甘露醇、蔗糖、淀粉、碳酸钙、结晶纤维素、硅酸等)、粘合剂(例如水、乙醇、myranol、葡萄糖溶液、淀粉溶液、明胶溶液、聚乙烯吡咯烷酮等)、崩解剂(例如干淀粉、海藻酸钠、碳酸氢钠、碳酸钙、聚氧乙烯去水山梨糖醇脂肪酸酯、十二烷基硫酸钠、硬脂酰甘油单酯、淀粉、乳糖等)、吸收促进剂(例如季铵碱、十二烷基硫酸钠等)、润湿剂(例如甘油、淀粉等)、润滑剂(例如硬脂酸盐、聚乙二醇等)以及调味剂(例如甜味剂)。片剂可以是常规片剂、模制片剂、薄片等形式。To prepare tablets, the compound can be mixed with pharmaceutically acceptable carriers known in the art, such as excipients (vehicles) (such as lactose, white sugar, mannitol, sucrose, starch, calcium carbonate, crystalline cellulose, silicic acid, etc.), Binders (such as water, ethanol, myranol, glucose solution, starch solution, gelatin solution, polyvinylpyrrolidone, etc.), disintegrants (such as dry starch, sodium alginate, sodium bicarbonate, calcium carbonate, polyoxyethylene dehydrated Sorbitan fatty acid esters, sodium lauryl sulfate, stearyl monoglyceride, starch, lactose, etc.), absorption enhancers (such as quaternary ammonium bases, sodium lauryl sulfate, etc.), wetting agents (such as glycerin, starch, etc.), lubricants (such as stearates, polyethylene glycol, etc.), and flavoring agents (such as sweeteners). Tablets may be in the form of conventional tablets, molded tablets, flakes and the like.
舌下给药是指在嘴里给药(例如舌下、颊和牙龈之间、舌和口腔顶部之间)。嘴里丰富的血管粘膜内层可以方便化合物向体内给药。Sublingual administration refers to administration in the mouth (eg, under the tongue, between the cheek and gum, between the tongue and the roof of the mouth). The abundant vascular mucosal lining of the mouth facilitates the administration of compounds into the body.
在其它实施方案中,固体剂型可以在可药用载体中包装成颗粒剂或散剂,当这些颗粒剂或散剂脱离包装后,喷散在食物上或者与液体例如水或液体(juice)混合,也可以将这些颗粒剂插入胶囊。在该实施方案中,本文所述化合物可以与调味剂或甜味剂混合。包装材料可以是塑料的包衣纸或者防止水或湿度到达颗粒和/或粉末的其它任何材料。In other embodiments, the solid dosage forms can be packaged as granules or powders in pharmaceutically acceptable carriers, and when these granules or powders are out of the packaging, they can be sprayed on food or mixed with liquids such as water or liquid (juice), or Insert these granules into a capsule. In this embodiment, the compounds described herein may be admixed with flavoring or sweetening agents. The packaging material may be a plastic wrap or any other material that prevents water or humidity from reaching the granules and/or powder.
用于口服给药的液体剂型可以包括可药用乳剂、溶液剂、舌下溶液剂、混悬剂和糖浆剂,其中含有本领域常用的惰性稀释剂,例如水。这类组合物还可以含有佐剂,例如润湿剂、乳化剂和助悬剂、甜味剂、调味剂和芳香剂。为了制得舌下溶液剂,可以将化合物与各种载体、赋形剂、pH调节剂等混合(例如水、糖、乳酸、乙酸、果糖、葡萄糖、糖精、聚乙二醇、丙二醇、乙醇、皂土、黄蓍胶、明胶、阿司帕坦、山梨糖醇、对羟基苯甲酸甲酯、对羟基苯甲酸丙酯、苯甲酸钠、人造调味剂和着色剂)。Liquid dosage forms for oral administration may include pharmaceutically acceptable emulsions, solutions, sublingual solutions, suspensions and syrups containing inert diluents commonly used in the art, such as water. Such compositions may also contain adjuvants, such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents. To prepare a sublingual solution, the compound can be mixed with various carriers, excipients, pH adjusters, etc. (for example, water, sugar, lactic acid, acetic acid, fructose, glucose, saccharin, polyethylene glycol, propylene glycol, ethanol, bentonite, tragacanth, gelatin, aspartame, sorbitol, methylparaben, propylparaben, sodium benzoate, artificial flavor and color).
对于吸入给药,可以将化合物由吹入器、喷雾器或压力包或者用于递送气溶胶(aerosol spray)的其它常规模式递送。压力包可以含义适宜的推进剂。或者,对于吸入给药,可以将化合物以干燥粉末组合物或者液体喷雾剂的形式给药。For administration by inhalation, the compounds may be delivered from an insufflator, nebulizer, or pressure pack, or other conventional modes of delivery for aerosol sprays. The pressure pack may contain a suitable propellant. Alternatively, for administration by inhalation, the compounds may be administered as dry powder compositions or liquid sprays.
用于直肠给药的栓剂可以通过将活性化合物与适宜的无刺激性赋型剂混合制备,例如在室温下为固体、在体温下为液体的可可脂和聚乙二醇。或者,可以制备灌肠剂用于直肠给药本文所述的化合物。Suppositories for rectal administration can be prepared by mixing the active compound with suitable non-irritating excipients, such as cocoa butter and polyethylene glycols, which are solid at room temperature and liquid at body temperature. Alternatively, enemas may be prepared for rectal administration of a compound described herein.
为了向表皮局部给药,可以将化合物配制成软膏剂、霜剂或洗剂或者活性成分的经皮贴剂形式。化合物还可以通过离子电渗疗法或渗透泵给药。软膏剂、霜剂和洗剂可以使用水或油性基质,同时加入适宜的增稠剂和/或胶凝剂制备。或者,软膏剂、霜剂和洗剂还可以使用水或油基质制备,同时还可以含有一种或多种乳化剂、稳定剂、分散剂、助悬剂、增稠剂和/或着色剂。对于霜剂或洗剂,化合物可以与一种或多种防腐剂(例如苄醇1%或2%(wt/wt))、乳化蜡、甘油、棕榈酸异丙酯、乳酸、纯化水、山梨糖醇溶液混合形成光滑、均匀的霜剂或洗剂。这类局部给药组合物可以含有聚乙二醇400。为了形成软膏剂,可以将化合物与一种或多种防腐剂(例如苄醇2%(wt/wt))、凡士林、乳化蜡和Tenox(II)(例如丁基化羟基茴香醚、没食子酸丙酯、柠檬酸、丙二醇)混合。包扎材料的针织垫或滚例如纱布可以使用上述经皮给药组合物浸泡用于局部施用。For topical administration to the epidermis, the compounds may be formulated as ointments, creams or lotions, or as a transdermal patch of the active ingredient. Compounds can also be administered by iontophoresis or osmotic pumps. Ointments, creams and lotions may be formulated with an aqueous or oily base with the addition of suitable thickening and/or gelling agents. Alternatively, ointments, creams and lotions may be formulated with an aqueous or oily base and may contain one or more emulsifying, stabilizing, dispersing, suspending, thickening and/or coloring agents. For creams or lotions, the compound can be mixed with one or more preservatives (such as
化合物还可以使用经皮系统局部应用,例如一种使用本文所述化合物浸透的用树脂交联剂粘连的丙烯酸基质聚合物,然后将其层压成具有不可渗透性的衬垫。例如,化合物可以以经皮贴剂如持续释出经皮贴剂的形式给药。经皮贴剂可以包括任意的常规形式,例如粘着基质、聚合物基质、储库式贴剂、基质或单片(monolithic)类型的层压结构,它们通常由衬垫层、粘合剂、渗透促进剂和/或速率控制膜中的一种或多种组成。经皮贴剂通常具有释放衬里,在使用之前将其除去后,会曝露出粘性物/活性成分。经皮贴剂描述在例如U.S专利号5,262,165、5,948,433、6,010,715和6,071,531中,在此将其全部内容引入作为参考。The compounds may also be applied topically using a transdermal system, such as an acrylic matrix polymer impregnated with the compounds described herein, bonded with a resinous crosslinker, which is then laminated into an impermeable pad. For example, the compounds may be administered in the form of a transdermal patch, such as a sustained release transdermal patch. Transdermal patches may comprise any conventional form, such as adhesive matrices, polymer matrices, depot patches, matrices or monolithic type laminated structures, which typically consist of backing layers, adhesives, osmotic Accelerators and/or rate control one or more components of the membrane. Transdermal patches typically have a release liner which, when removed prior to use, exposes the adhesive/active ingredient. Transdermal patches are described, for example, in U.S. Patent Nos. 5,262,165, 5,948,433, 6,010,715, and 6,071,531, the entire contents of which are incorporated herein by reference.
本发明提供了将化合物向患者通过鼻给药以治疗本文所述疾病和障碍。"通过鼻给药"或"鼻给药"是指将至少一种化合物与适合穿过患者鼻粘膜吸收的递送体系组合。通常,当与例如口服给药所需要的剂量相比时,通过鼻给药可以需要更低剂量的化合物。The present invention provides nasal administration of compounds to a patient for the treatment of the diseases and disorders described herein. "Nasal administration" or "nasal administration" refers to combining at least one compound with a delivery system suitable for absorption across the nasal mucosa of a patient. In general, lower dosages of the compound may be required for nasal administration when compared to, for example, oral administration.
化合物可以以例如鼻喷雾剂、鼻滴剂、鼻混悬剂、鼻凝胶剂、鼻软膏剂、鼻霜剂或鼻粉末剂形式给药。化合物还可以使用鼻棉塞或鼻海绵给药。可以将化合物通过常用体系引入至粘性基底中,例如天然树胶、甲基纤维素和衍生物、丙烯酸聚合物(聚羧乙烯)和乙烯基聚合物(聚乙烯吡咯烷酮)。在该组合物中,可以加入本领域已知的多种其它赋形剂,例如水、防腐剂、表面活性剂、溶剂、粘合剂、抗氧化剂、缓冲剂、生物粘合剂、粘性增强剂以及调节pH和渗透性的赋形剂。The compounds can be administered in the form of, for example, nasal sprays, nasal drops, nasal suspensions, nasal gels, nasal ointments, nasal creams, or nasal powders. The compounds can also be administered using nasal tampons or nasal sponges. The compounds can be introduced into the adhesive base by conventional systems, such as natural gums, methylcellulose and derivatives, acrylic polymers (carboxyvinyl) and vinyl polymers (polyvinylpyrrolidone). In this composition, various other excipients known in the art may be added, such as water, preservatives, surfactants, solvents, binders, antioxidants, buffers, bioadhesives, viscosity enhancers and excipients to adjust pH and osmolarity.
鼻递送体系可以采取各种形式,包括水性溶液、非水性溶液及其组合物。水性溶液包括例如水性凝胶剂、水性混悬剂、水性脂质分散体、水性乳剂、水性微乳剂及其组合物。非水性溶液包括例如例如非水性凝胶剂、非水性混悬剂、非水性脂质分散体、非水性乳剂、非水性微乳剂及其组合物。Nasal delivery systems can take various forms, including aqueous solutions, non-aqueous solutions, and combinations thereof. Aqueous solutions include, for example, aqueous gels, aqueous suspensions, aqueous lipid dispersions, aqueous emulsions, aqueous microemulsions, and combinations thereof. Non-aqueous solutions include, for example, non-aqueous gels, non-aqueous suspensions, non-aqueous lipid dispersions, non-aqueous emulsions, non-aqueous microemulsions, and combinations thereof.
在其它实施方案中,鼻递送体系可以是粉末制剂。粉末制剂(powderformulation)包括例如粉末混合物、粉末微球、包衣粉末微球、脂质分散体、及其组合物。优选粉末制剂是粉末微球。粉末微球优选由选自淀粉、甲基纤维素、黄蓍胶、羧甲基纤维素、羟丙基纤维素、卡波姆(carbomer)、聚乙烯醇海藻酸酯、阿拉伯胶、壳聚糖以及其中两种或多种的混合物中的各种多糖和纤维素形成。In other embodiments, the nasal delivery system may be a powder formulation. Powder formulations include, for example, powder mixtures, powder microspheres, coated powder microspheres, lipid dispersions, and combinations thereof. Preferred powder formulations are powder microspheres. The powder microspheres are preferably made of starch, methylcellulose, gum tragacanth, carboxymethylcellulose, hydroxypropylcellulose, carbomer, polyvinyl alcohol alginate, gum arabic, chitosan and various polysaccharides and cellulose in mixtures of two or more thereof are formed.
在某些实施方案中,向鼻粘膜递送的水性和/或非水性溶液的小滴或粉末的粒径可以为例如大约0.1微米至大约100微米,大约1微米至大约70微米;大约5微米至大约50微米;大约10微米至大约20微米。上述粒径可以使用本领域已知的适合容器或计量装置获得。示例性装置包括其中通过活塞运动实现递送的机械泵;其中通过向容器中手动泵入空气实现递送的压缩其它装置;其中通过在密闭容器中控制释出压缩气体实现递送的压缩气体(例如氮气)技术;其中将低沸点液体烃(例如丁烷)蒸发产生气压并压迫组合物通过计量阀门的液化推进技术等。粉末可以以例如置于随后放置在吸入或吹入装置中的胶囊中的形式给药。使针刺过胶囊从而在胶囊的顶部和底部制得小孔,然后输送空气来向外吹出粉末颗粒。粉末制剂还可以以惰性气体的喷雾(jet-spray)形式或者悬浮于液体有机流体中的形式给药。In certain embodiments, droplets or powders of aqueous and/or non-aqueous solutions delivered to the nasal mucosa can have a particle size, for example, of about 0.1 microns to about 100 microns, about 1 micron to about 70 microns; about 5 microns to about 70 microns; about 50 microns; about 10 microns to about 20 microns. The above particle sizes can be obtained using suitable containers or metering devices known in the art. Exemplary devices include mechanical pumps, where delivery is achieved by piston movement; compressed other devices, where delivery is achieved by manually pumping air into a container; compressed gas (such as nitrogen) where delivery is achieved by controlled release of the compressed gas in a closed container technology; liquefaction propulsion technology in which vaporization of a low-boiling liquid hydrocarbon (such as butane) generates an air pressure and forces the composition through a metering valve, etc. The powder can be administered, for example, in the form of a capsule which is then placed in an inhalation or insufflation device. Needles are pierced through the capsule to make small holes in the top and bottom of the capsule, and air is then delivered to blow the powder particles outward. Powder formulations can also be administered as a jet-spray of an inert gas or suspended in a liquid organic fluid.
在一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种化合物,所述化合物分散于提高化合物的溶解性的鼻递送体系中。提高溶解性的鼻递送体系可以包括下面的一种或其组合物:(i)二醇衍生物(例如丙二醇、聚乙二醇或其混合物);(ii)糖醇(例如甘露醇、木糖醇或其混合物);(iii)甘油;(iv)二醇衍生物(例如丙二醇、聚乙二醇或其混合物)和甘油;(v)抗坏血酸和水;(vi)抗坏血酸钠和水;或者(vii)偏亚硫酸钠和水。In one embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound dispersed in a nasal delivery system that enhances the solubility of the compound. Nasal delivery systems that enhance solubility may include one or a combination of: (i) glycol derivatives (e.g., propylene glycol, polyethylene glycol, or mixtures thereof); (ii) sugar alcohols (e.g., mannitol, xylose alcohol or mixtures thereof); (iii) glycerol; (iv) glycol derivatives (such as propylene glycol, polyethylene glycol or mixtures thereof) and glycerin; (v) ascorbic acid and water; (vi) sodium ascorbate and water; or ( vii) Sodium metasulfite and water.
在另一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种本文所述的化合物和鼻递送体系,其中鼻递送体系含有至少一种维持本文所述化合物pH的缓冲剂、至少一种可药用增稠剂和至少一种保湿剂(humectant)。鼻递送体系可以任选进一步含有表面活性剂、防腐剂、抗氧化剂、生物粘合剂、pH调节剂、等渗剂、增溶剂和/或其它可药用赋形剂。本文所述化合物可以任选分散于能够提高其溶解性的鼻递送体系中。In another embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound described herein and a nasal delivery system, wherein the nasal delivery system comprises at least one agent that maintains the pH of the compound described herein. buffer, at least one pharmaceutically acceptable thickener and at least one humectant. The nasal delivery system may optionally further contain surfactants, preservatives, antioxidants, bioadhesives, pH regulators, isotonic agents, solubilizers and/or other pharmaceutically acceptable excipients. Compounds described herein can optionally be dispersed in nasal delivery systems to enhance their solubility.
在另一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种本文所述的化合物和鼻递送体系,其中鼻递送体系含有至少一种增溶剂、至少一种可药用增稠剂和至少一种保湿剂。鼻递送体系可以任选进一步含有缓冲剂、pH调节剂、等渗剂、表面活性剂、防腐剂、抗氧化剂、生物粘合剂和/或其它可药用赋形剂。本文所述化合物可以任选分散于能够提高其溶解性的鼻递送体系中。In another embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound described herein and a nasal delivery system, wherein the nasal delivery system comprises at least one solubilizer, at least one A medicinal thickener and at least one humectant. Nasal delivery systems may optionally further contain buffers, pH adjusters, isotonic agents, surfactants, preservatives, antioxidants, bioadhesives and/or other pharmaceutically acceptable excipients. Compounds described herein can optionally be dispersed in nasal delivery systems to enhance their solubility.
在另一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种本文所述的化合物和鼻递送体系,其中鼻递送体系含有至少一种维持化合物pH的缓冲剂、至少一种可药用增稠剂、至少一种保湿剂和至少一种表面活性剂。鼻递送体系可以任选进一步含有pH调节剂、等渗剂、增溶剂、防腐剂、抗氧化剂、生物粘合剂和/或其它可药用赋形剂。本文所述化合物可以任选分散于能够提高其溶解性的鼻递送体系中。In another embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound described herein and a nasal delivery system, wherein the nasal delivery system comprises at least one buffer to maintain the pH of the compound, at least one pharmaceutically acceptable thickener, at least one humectant and at least one surfactant. The nasal delivery system may optionally further contain pH adjusters, isotonic agents, solubilizers, preservatives, antioxidants, bioadhesives and/or other pharmaceutically acceptable excipients. Compounds described herein can optionally be dispersed in nasal delivery systems to enhance their solubility.
在又一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种本文所述的化合物和鼻递送体系,其中鼻递送体系含有至少一种可药用增稠剂、至少一种保湿剂、至少一种表面活性剂和至少一种增溶剂。鼻递送体系可以任选进一步含有缓冲剂、pH调节剂、等渗剂、防腐剂、抗氧化剂、生物粘合剂和/或其它可药用赋形剂。化合物可以任选分散于能够提高其溶解性的鼻递送体系中。In yet another embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound described herein and a nasal delivery system, wherein the nasal delivery system comprises at least one pharmaceutically acceptable thickener, at least one humectant, at least one surfactant and at least one solubilizer. Nasal delivery systems may optionally further contain buffers, pH adjusters, isotonic agents, preservatives, antioxidants, bioadhesives and/or other pharmaceutically acceptable excipients. The compound can optionally be dispersed in a nasal delivery system to enhance its solubility.
在又一实施方案中,本发明提供了通过鼻给药的药物组合物,其中含有至少一种本文所述的化合物和鼻递送体系,其中鼻递送体系含有至少一种维持化合物pH的缓冲剂、至少一种可药用增稠剂、至少一种保湿剂、至少一种表面活性剂和至少一种增溶剂。鼻递送体系可以任选进一步含有缓冲剂、pH调节剂、等渗剂、防腐剂、抗氧化剂、生物粘合剂和/或其它可药用赋形剂。本文所述化合物可以任选分散于能够提高其溶解性的鼻递送体系中。In yet another embodiment, the present invention provides a pharmaceutical composition for nasal administration comprising at least one compound described herein and a nasal delivery system, wherein the nasal delivery system comprises at least one buffer to maintain the pH of the compound, at least one pharmaceutically acceptable thickener, at least one humectant, at least one surfactant, and at least one solubilizer. Nasal delivery systems may optionally further contain buffers, pH adjusters, isotonic agents, preservatives, antioxidants, bioadhesives and/or other pharmaceutically acceptable excipients. Compounds described herein can optionally be dispersed in nasal delivery systems to enhance their solubility.
选择具有可以使通过鼻粘膜的1,2-二氢吡啶化合物的吸收最大化pH的缓冲剂。缓冲剂的特定pH可以随特定的鼻递送制剂以及所选择的具体化合物改变而改变。适合用于本发明的缓冲剂包括乙酸盐(例如乙酸钠)、柠檬酸盐(例如柠檬酸钠二水合物)、邻苯二甲酸盐、硼酸盐、醇溶蛋白、三乙醇胺、碳酸盐、磷酸盐(例如磷酸二氢钾、磷酸二钠)以及其中的两种或多种的混合物。The buffer is selected to have a pH that maximizes the absorption of the 1,2-dihydropyridine compound through the nasal mucosa. The particular pH of the buffer may vary with the particular nasal delivery formulation and the particular compound chosen. Buffers suitable for use in the present invention include acetates (e.g. sodium acetate), citrates (e.g. sodium citrate dihydrate), phthalates, borates, prolamins, triethanolamine, carbon salts, phosphates (such as potassium dihydrogen phosphate, disodium phosphate) and mixtures of two or more thereof.
组合物的pH可以维持在大约3.0至大约10.0。具有pH小于大约3.0或者大于大约10.0的组合物可以增加刺激患者鼻粘膜的可能性。另外,优选组合物的pH维持在大约3.0至大约9.0。对于非水性鼻制剂而言,可以选择缓冲剂的适宜形式使得当该制剂被递送入哺乳动物的鼻腔时,在与例如鼻粘膜接触后可以获得所选择的pH范围。The pH of the composition can be maintained from about 3.0 to about 10.0. Compositions having a pH of less than about 3.0 or greater than about 10.0 may increase the likelihood of irritation of the patient's nasal mucosa. Additionally, it is preferred that the pH of the composition be maintained from about 3.0 to about 9.0. For non-aqueous nasal formulations, the appropriate form of buffering agent can be chosen such that when the formulation is delivered into the nasal cavity of a mammal, a selected pH range is obtained upon contact with, for example, the nasal mucosa.
用于本发明组合物中的增溶剂可以是本领域已知的任何一种,例如羧酸及其盐。示例性羧酸盐包括乙酸盐、葡萄糖酸盐、抗坏血酸盐、柠檬酸盐、富马酸盐、乳酸盐、酒石酸盐、马来酸盐、琥珀酸盐或者其中的两种或多种的混合物。The solubilizing agent used in the composition of the present invention may be any known in the art, such as carboxylic acids and their salts. Exemplary carboxylates include acetate, gluconate, ascorbate, citrate, fumarate, lactate, tartrate, maleate, succinate, or combinations of two or more thereof mixture.
本发明组合物的粘度可以使用可药用增稠剂维持在所希望的水平。例如,粘度可以至少1000cps;大约1000至大约10,000cps;大约2000cps至大约6500cps;或者大约2500cps至大约5000cps。可以用于本发明中的增稠剂包括例如甲基纤维素、黄蓍胶、羧甲基纤维素、羟丙基纤维素、卡波姆、聚乙烯醇、海藻酸盐、阿拉伯胶、壳聚糖以及其中的两种或多种的混合物。增稠剂的浓度取决于所选择的药物和所希望的粘度。这类增稠剂还可以用于粉末制剂中。The viscosity of the compositions of the present invention can be maintained at a desired level using pharmaceutically acceptable thickeners. For example, the viscosity can be at least 1000 cps; about 1000 to about 10,000 cps; about 2000 cps to about 6500 cps; or about 2500 cps to about 5000 cps. Thickeners that can be used in the present invention include, for example, methyl cellulose, tragacanth, carboxymethyl cellulose, hydroxypropyl cellulose, carbomer, polyvinyl alcohol, alginate, acacia, chitosan Sugar and mixtures of two or more thereof. The concentration of thickener depends on the drug chosen and the desired viscosity. Such thickeners can also be used in powder formulations.
通过鼻给药的组合物还可以含有保湿剂以减轻或防止粘膜干燥,从而防止对粘膜的刺激。可以使用的适宜保湿剂包括例如山梨糖醇、矿物油、植物油和甘油;安慰剂(soothing agent);膜调节剂;甜味剂以及其中的两种或多种的混合物。保湿剂的浓度取决于所选择的药物。在一实施方案中,保湿剂可以以占组合物大约0.01%至大约20%重量的浓度范围出现在鼻递送体系中。Compositions for nasal administration may also contain humectants to reduce or prevent drying of the mucous membranes, thereby preventing irritation to the mucous membranes. Suitable humectants that may be used include, for example, sorbitol, mineral oil, vegetable oil, and glycerin; soothing agents; membrane regulators; sweeteners and mixtures of two or more thereof. The concentration of humectant depends on the drug chosen. In one embodiment, the humectant may be present in the nasal delivery system at a concentration ranging from about 0.01% to about 20% by weight of the composition.
在其它实施方案中,鼻递送体系可以进一步含有促进化合物吸收的表面活性剂。适宜的表面活性剂包括非离子、阳离子和阴离子表面活性剂。示例性表面活性剂包括油酸、脂肪酸的聚氧乙烯衍生物、山梨糖醇酐的部分酯,例如Tweens(例如Tween 80、Tween 40、Tween 20)、Spans(例如Span 40、Span 80、Span 20)、聚乙二醇40硬脂酸酯、聚氧乙烯50硬脂酸酯、fusieates、胆汁盐、辛苯聚醇以及其中的两种或多种的混合物。示例性阳离子表面活性剂包括具有一种或多种下述官能基团的长链烃的盐(例如C6-30或C10-20):羧酸盐、磺酸盐和硫酸盐。优选具有硫酸盐官能基团的长链烃的盐,例如十六十八烷基硫酸钠(sodium cetostearyl sulfate)、十二烷基硫酸钠和十四烷基硫酸钠。特别优选的阴离子表面活性剂是月桂基硫酸钠(即十二烷基硫酸钠)。表面活性剂可以以大约0.001%至大约50%重量,例如大约0.001%至大约20%重量的含量存在。In other embodiments, nasal delivery systems may further contain surfactants to facilitate absorption of the compound. Suitable surfactants include nonionic, cationic and anionic surfactants. Exemplary surfactants include oleic acid, polyoxyethylene derivatives of fatty acids, partial esters of sorbitan, such as Tweens (e.g. Tween 80, Tween 40, Tween 20), Spans (e.g. Span 40, Span 80, Span 20 ), macrogol 40 stearate, polyoxyethylene 50 stearate, fusieates, bile salts, octoxynol, and mixtures of two or more thereof. Exemplary cationic surfactants include salts of long chain hydrocarbons (eg, C6-30 or C10-20) having one or more of the following functional groups: carboxylate, sulfonate, and sulfate. Preference is given to salts of long-chain hydrocarbons having a sulfate functional group, such as sodium cetostearyl sulfate, sodium lauryl sulfate and sodium tetradecyl sulfate. A particularly preferred anionic surfactant is sodium lauryl sulfate (ie sodium lauryl sulfate). The surfactant may be present in an amount from about 0.001% to about 50% by weight, for example from about 0.001% to about 20% by weight.
本发明药物组合物可以进一步含有等渗剂,例如氯化钠、右旋糖、硼酸、酒石酸钠或者其它无机或有机溶质。The pharmaceutical composition of the present invention may further contain isotonic agents such as sodium chloride, dextrose, boric acid, sodium tartrate or other inorganic or organic solutes.
本发明的鼻药物组合物可以任选组合pH调节剂使用。示例性pH调节剂包括硫酸、氢氧化钠、盐酸等。The nasal pharmaceutical compositions of the present invention may optionally be used in combination with pH adjusting agents. Exemplary pH adjusters include sulfuric acid, sodium hydroxide, hydrochloric acid, and the like.
为了延长贮存期限,可以向通过鼻给药的组合物中加入防腐剂。可以使用的适宜防腐剂包括苄醇、对羟苯甲酸酯、柳硫汞、三氯叔丁醇、苯扎氯铵(benzalkonium chloride)或者其中的两种或多种的混合物。优选使用苯扎氯铵。通常防腐剂以至多大约2重量%的浓度存在。然而,防腐剂的精确浓度取决于预期用途,这可以由本领域技术人员方便地加以确认。To prolong shelf life, preservatives can be added to compositions for nasal administration. Suitable preservatives that may be used include benzyl alcohol, parabens, thimerosal, chlorobutanol, benzalkonium chloride or mixtures of two or more thereof. Preference is given to using benzalkonium chloride. Typically preservatives are present in concentrations of up to about 2% by weight. However, the precise concentration of preservative depends on the intended use and can be readily ascertained by one skilled in the art.
可以加入延长贮存期限的其它成分,例如抗氧化剂。抗氧化剂的某些实例包括偏亚硫酸氢钠、偏亚硫酸氢钾、抗坏血酸基棕榈酸酯等。通常抗氧化剂以占总组合物大约0.001%至大约5%重量的浓度存在于组合物中。Other ingredients, such as antioxidants, may be added to extend shelf life. Some examples of antioxidants include sodium metabisulfite, potassium metabisulfite, ascorbyl palmitate, and the like. Antioxidants are generally present in the composition at a concentration of from about 0.001% to about 5% by weight of the total composition.
鼻递送体系可以按照描述在例如U.S专利号6,451,848、6,436,950和5,874,450及WO 00/00199中的方法制备,在此将其全部内容引入作为参考。Nasal delivery systems can be prepared as described in, for example, U.S. Patent Nos. 6,451,848, 6,436,950, and 5,874,450 and WO 00/00199, the entire contents of which are incorporated herein by reference.
实施例 Example
下面的实施例仅仅是用于示例性说明本发明的目的,而并不意味着对本发明主旨或范围构成限制。The following examples are only for the purpose of illustrating the present invention, and are not meant to limit the spirit or scope of the present invention.
实施例1Example 1
抗偏头痛剂通常使用角叉菜胶诱导的热痛觉过敏模型评价(Bingham等人,Experimental Neurology,167:65-73(2001);Daher等人,Life Sciences,76:2349-2359(2005))。化合物A(即3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮)对热痛觉过敏的抗偏头痛特性在下面的大鼠角叉菜胶诱导的炎性疼痛模型中评价。Anti-migraine agents are commonly evaluated using the carrageenan-induced thermal hyperalgesia model (Bingham et al., Experimental Neurology, 167:65-73 (2001); Daher et al., Life Sciences, 76:2349-2359 (2005)) . Antimigraine properties of compound A (i.e., 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one) in response to thermal hyperalgesia Evaluated in the following rat carrageenan-induced inflammatory pain model.
实验使用雄性Wistar大鼠(5/组)。使用TAIL FLICK 7360(Ugo Basile,Italy)测定大鼠两只后爪的避开热有害性刺激的退缩潜伏期。将1%角叉菜胶注射入大鼠的右后爪足底。在角叉菜胶注射2小时后,向大鼠口服给药3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮或溶媒。在给药1、3和5小时后测定热刺激大鼠两只后爪的退缩潜伏期。Male Wistar rats (5/group) were used in the experiments. Using TAIL FLICK 7360 (Ugo Basile, Italy) to measure the withdrawal latency of the two hind paws of rats to avoid thermal noxious stimuli. 1% carrageenan was injected into the sole of the right hind paw of the rat. 2 hours after carrageenan injection, rats were orally administered 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2 - ketone or vehicle. The withdrawal latencies of the two hind paws of the heat-stimulated rats were measured at 1, 3 and 5 hours after administration.
如图1所示,大鼠注射角叉菜胶的右后爪的退缩潜伏期有所缩短。如图2所示,大鼠左后爪的退缩潜伏期没有变化。结果表明,角叉菜胶在注射后爪中引起了炎性痛觉过敏反应,而在大鼠对侧后爪中没有引起痛觉过敏反应。在整个实验期间观察了角叉菜胶的作用。As shown in Figure 1, withdrawal latencies were shortened in the right hind paw of rats injected with carrageenan. As shown in Figure 2, there was no change in the withdrawal latency of the left hind paw of the rats. The results showed that carrageenan elicited an inflammatory hyperalgesic response in the injected hind paw but not in the contralateral hind paw of the rats. The effect of carrageenan was observed throughout the experimental period.
图1还表明,3和6mg/kg的3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮在给药1小时后抑制了所诱导的痛觉过敏,在给药3和5小时后效果下降。图2还表明,3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮没有改变大鼠对侧后爪的潜伏期,因此这说明3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮没有影响运动功能。上述动物模型结果预期了3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮可以治疗人的偏头痛和头痛。Figure 1 also shows that 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one at 3 and 6 mg/kg Induced hyperalgesia was suppressed after 1 hour, with a decline in effect after 3 and 5 hours of administration. Figure 2 also shows that 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one did not alter the contralateral hindpaw Latency, thus indicating that 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one did not affect motor function. The above animal model results predict that 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one can treat migraine and headache in humans .
实施例2Example 2
为了评价3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物在偏头痛预防和治疗中的效果和安全性,进行了随机双盲的安慰剂-对照、多中心平行组研究。To evaluate the efficacy and For safety, a randomized double-blind placebo-controlled, multicenter parallel group study was conducted.
主要效力端点是评价23-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物在减轻偏头痛方面的效果,这是基于该化合物在治疗期间相对于基线期而言对每28天内偏头痛期出现频率的改变。将偏头痛期定义为偏头痛发作、结束或者在24小时内复发。如果偏头痛持续长于24小时,则认为是新的偏头痛期。The primary efficacy endpoint was to evaluate the effect of 23-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one hydrate in reducing migraine , based on the compound's change in frequency of migraine sessions per 28 days during treatment relative to baseline. A migraine period was defined as the onset, end, or recurrence of migraine within 24 hours. If the migraine lasted longer than 24 hours, it was considered a new migraine period.
本研究的第二个目标在于评价23-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物在偏头痛患者中的安全性和耐受性;表征3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物在患者群中的药代动力学;确定3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物血浆浓度-应答关系;以及评价偏头痛严重程度的变化。第二效力是根据相对于1期而言一种或多种下述指标的降低来确定的:(a)在28天偏头痛期频率方面,降低至少50%的患者比例;(b)在3期期间每28天偏头痛期频率的变化;(c)偏头痛发作频率的变化;(d)每28天症状复原治疗所需天数的变化;(e)每28内出现偏头痛的总天数;(f)偏头痛发作的严重程度的变化;(g)偏头痛发作持续时间的变化;(h)患者的总体印象改变(Global Impression of Change)变化;以及(i)偏头痛失能评估(Migraine Disability Assessment(MIDAS))调查表评分的变化。The second objective of this study was to evaluate the role of 23-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one hydrate in migraine Safety and tolerability in patients; characterizing 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one hydrate in Pharmacokinetics in a patient population; determination of plasma concentrations of 3-(2-cyanophenyl)-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridin-2-one hydrate - Response relationship; and evaluation of changes in migraine severity. Secondary efficacy was determined based on a reduction in one or more of the following indicators relative to Phase 1: (a) the proportion of patients with at least a 50% reduction in the frequency of migraine sessions at 28 days; (b) at 3 Change in frequency of migraine sessions per 28-day period; (c) change in frequency of migraine attacks; (d) change in days required for symptom-reversal treatment per 28-day period; (e) total number of days with migraine episodes per 28-day period; (f) changes in the severity of migraine attacks; (g) changes in the duration of migraine attacks; (h) changes in the patient's Global Impression of Change; and (i) migraine disability assessment (Migraine Disability Assessment (MIDAS) questionnaire score changes.
对于血浆浓度研究,通过建立群体药动学模型(计算了CMinSS、CMAXSS和AUC0-TauSS。还测定了适宜个体的药代动力学参数的后验估计。还评价了患者之间和访问者之间这些参数的变化。对接触-反应(每28天内偏头痛频率降低)关系的研究使用混合效应模型进行。For the plasma concentration study, C MinSS , C MAXSS , and AUC 0 -TauSS were calculated by building a population pharmacokinetic model. Posterior estimates of appropriate individual pharmacokinetic parameters were also determined. Interpatient and visit Changes in these parameters between patients.The study of the exposure-response (reduction in migraine frequency within each 28-day period) relationship was performed using a mixed-effects model.
安全性通过不良事件的发生率以及物理学和神经学检查、生命体征、临床实验室测试、体重和心电图结果评价。Safety was assessed by the incidence of adverse events and the results of physical and neurological examination, vital signs, clinical laboratory tests, body weight and electrocardiogram.
本研究为14周预期的随机双盲、安慰剂-对照多中心平行组研究,具有4周基线期以及研究结束时的4周单盲安慰剂安全期。本研究具有下述各期。1期是4周基线期,在该期间偏头痛频率的预期确定借助患者的偏头痛日记来确定。2期为至多1-10周的逐步增加剂量期,该期间由0.5-2.0mg/天的剂量开始,每天晚间给药一次。1-5周后,剂量升高至1.0-2.0mg/天,再1-5周后剂量升高至1.5-4.0mg/天。出现不能忍受的不良事件的患者服用降低一个剂量级的剂量。在该期期间,获得血液样本以测定血浆浓度。在2期,每隔2周进行一次临床访问。3期为4-12周的维持期,此间剂量由2期过程中服用的最后一个剂量不再发生变化。在每次访问时获得血浆浓度样本。在3期过程中,每隔4周进行一次临床访问。4期为4周,此间所有患者以单盲方式服用安慰剂。继续记录患者的偏头痛频率和特征。在3期结束后大约4周进行最后一次访问,同时获得血浆浓度样本。This study was a 14-week prospective randomized double-blind, placebo-controlled multicenter parallel-group study with a 4-week baseline period and a 4-week single-blind placebo safety period at the end of the study. This study has the following phases.
患者是否合格按照Informed Consent的信号在筛选过程中确定。筛选评价包括病史、综合性物理学和神经学测验、生命体征、12-导联ECG、临床实验室测试、尿药筛选、竞争筛选(rival screen)以及MIDAS的给药。在筛选访问者的过程中,适合的患者被指导使用电子日记并被希望以每天为单位记录下偏头痛的出现和其它研究相关信息。此时,患者进入1期。Patient eligibility was determined during the screening process based on Informed Consent signals. Screening evaluations included medical history, comprehensive physical and neurological testing, vital signs, 12-lead ECG, clinical laboratory tests, urine drug screen, rival screen, and administration of MIDAS. During interview screening, eligible patients were instructed to use an electronic diary and were expected to record the onset of migraine and other study-related information on a daily basis. At this point, the patient enters
在成功进入1期并被证实符合所包括的所有标准且无排斥标准之后,允许患者开始本研究。出现少于4次或多于12次偏头痛、不能充分完成电子日记或者在1期不能顺从实验步骤的患者不被随机化。Patients were allowed to start the study after successfully entering
随机患者作为具有任意种族的18-65岁大的男性或女性,其偏头痛历史(根据the Headache Classification Committee of the International HeadacheSociety(HIS,2004指南),有或没有先兆)为至少12个月且在50岁前发作,在筛选和1期之前3个月期间每个月出现4-12次偏头痛发作。患者的体重指数(BMI)在筛选时应该为19-40kg/m2(包括端值)。Patients were randomized as males or females aged 18-65 years of any race with a history of migraine (with or without aura according to the Headache Classification Committee of the International Headache Society (HIS, 2004 guidelines)) for at least 12 months and in Onset before age 50, with 4-12 migraine attacks per month during the 3-month period prior to Screening and
在180名患者被募集进入本研究之后,以1:1的比例随机分组,安慰剂组90名患者,90名患者服用3-(2-氰基苯基)-5-(2-吡啶基)-1-苯基-1,2-二氢吡啶-2-酮水合物。预期164患者将完成研究。本研究在美国的大约25个调查点进行。After 180 patients were recruited into the study, they were randomized 1:1 to 90 patients in the placebo group and 90 patients in the 3-(2-cyanophenyl)-5-(2-pyridyl) -1-Phenyl-1,2-dihydropyridin-2-one hydrate. It is expected that 164 patients will complete the study. This study was conducted at approximately 25 survey sites in the United States.
在此将本文中引用的每篇专利、专利申请和出版物的全部内容引入作为参考。The entire contents of each patent, patent application, and publication cited herein are hereby incorporated by reference.
本领域技术人员应该理解,只要不偏离附录权利要求书的主旨或范围,可以对本发明进行各种变型。Those skilled in the art will appreciate that various modifications can be made to the present invention without departing from the spirit or scope of the appended claims.
Claims (38)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US66766505P | 2005-04-04 | 2005-04-04 | |
| US60/667,665 | 2005-04-04 | ||
| US60/689,519 | 2005-06-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101511186A true CN101511186A (en) | 2009-08-19 |
Family
ID=41003378
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA2006800197719A Pending CN101511186A (en) | 2005-04-04 | 2006-04-04 | Dihydropyridine compounds and compositions for headache |
Country Status (3)
| Country | Link |
|---|---|
| CN (1) | CN101511186A (en) |
| ES (1) | ES2365409T3 (en) |
| SI (1) | SI1871368T1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104649962A (en) * | 2013-11-25 | 2015-05-27 | 天津市汉康医药生物技术有限公司 | Perampanel sesquihydrate compound |
| CN109528670A (en) * | 2018-12-28 | 2019-03-29 | 正大制药(青岛)有限公司 | A kind of succinic acid SB 209509 piece and preparation method thereof |
| CN115279371A (en) * | 2020-01-14 | 2022-11-01 | 英倍尔制药公司 | Repeated administration of dihydroergotamine for the treatment of frequent migraine |
-
2006
- 2006-04-04 CN CNA2006800197719A patent/CN101511186A/en active Pending
- 2006-04-04 ES ES06740387T patent/ES2365409T3/en not_active Expired - Lifetime
- 2006-04-04 SI SI200631098T patent/SI1871368T1/en unknown
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104649962A (en) * | 2013-11-25 | 2015-05-27 | 天津市汉康医药生物技术有限公司 | Perampanel sesquihydrate compound |
| CN109528670A (en) * | 2018-12-28 | 2019-03-29 | 正大制药(青岛)有限公司 | A kind of succinic acid SB 209509 piece and preparation method thereof |
| CN109528670B (en) * | 2018-12-28 | 2021-05-07 | 正大制药(青岛)有限公司 | Frovatriptan succinate tablet and preparation method thereof |
| CN115279371A (en) * | 2020-01-14 | 2022-11-01 | 英倍尔制药公司 | Repeated administration of dihydroergotamine for the treatment of frequent migraine |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2365409T3 (en) | 2011-10-04 |
| SI1871368T1 (en) | 2011-10-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20060276510A1 (en) | Dihydropyridine compounds and compositions for headaches | |
| WO2005074535A2 (en) | Cholinesterase inhibitors for spinal cord disorders | |
| US20100168174A1 (en) | Combination of AMPA Receptor Antagonists and Acetylcholinesterase Inhibitors for the Treatment of Neuropathic Pain | |
| WO2003024456A1 (en) | Methods for treating and preventing migraines | |
| EP1875912A1 (en) | Agent for treating involuntary movement | |
| RS59002B1 (en) | Pyrimido[4,5-b]indole derivatives and use thereof in the expansion of hematopoietic stem cells | |
| WO2005087226A1 (en) | Cadasil treatment with cholinesterase inhibitors | |
| WO2009082039A1 (en) | Ampa receptor antagonists for epilepsy, mental disorders or deficits in sensory organ | |
| RS50404B (en) | USE OF CENTRAL CANABINOID RECEPTOR ANTAGONISTS TO OBTAIN MEDICAL PRODUCTS WHICH HELP COME FROM SMOKING | |
| JP2002527392A (en) | Peripherally acting antipruritic opiates | |
| EP2148673A1 (en) | Cinnamide compounds for dementia | |
| JPH10508029A (en) | How to treat anxiety | |
| JP2010520154A (en) | AMPA receptor antagonists and NMDA receptor antagonists for neurodegenerative diseases | |
| HU229150B1 (en) | Methods of using rapid-onset selective serotonin reuptake inhibitors for treating sxual dysfunction | |
| EP0709094A2 (en) | Method for treating anxiety using a tetrahydropyridine oxadiazole or thiadiazole compound | |
| JP2004503601A (en) | Use of COX-2 inhibitors in the treatment and prevention of ocular COX-2-mediated diseases | |
| HRP20040150A2 (en) | Use of bibn4096 u combination with other antimigraine drugs for the treatment of migraine | |
| JP2001510157A (en) | Peripherally acting anti-pruritus opiates | |
| EP1040829A2 (en) | Prokinetic agents for treating gastric hypomotility and related disorders | |
| CN101511186A (en) | Dihydropyridine compounds and compositions for headache | |
| JP2006096665A (en) | Treatment for spinal stenosis | |
| CA2690086A1 (en) | Ampa receptor antagonists and aldose reductase inhibitors for neuropathic pain | |
| JP2010533127A6 (en) | AMPA receptor antagonists and aldose reductase inhibitors for neuropathic pain | |
| MXPA99011155A (en) | Peripherally acting anti-pruritic opiates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Open date: 20090819 |