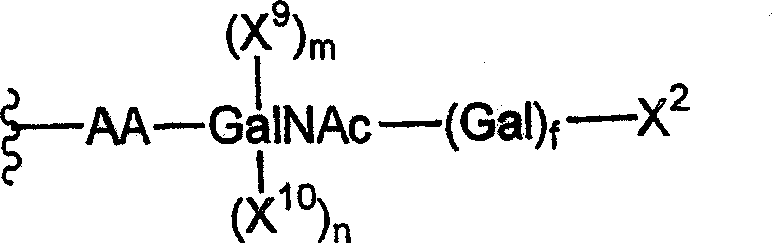CN100522988C - GlycoPEGylation method and protein/peptide produced by the method - Google Patents
GlycoPEGylation method and protein/peptide produced by the method Download PDFInfo
- Publication number
- CN100522988C CN100522988C CNB2004800159188A CN200480015918A CN100522988C CN 100522988 C CN100522988 C CN 100522988C CN B2004800159188 A CNB2004800159188 A CN B2004800159188A CN 200480015918 A CN200480015918 A CN 200480015918A CN 100522988 C CN100522988 C CN 100522988C
- Authority
- CN
- China
- Prior art keywords
- peptide
- polyethylene glycol
- conjugate
- sialic acid
- sialyltransferase
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- Y02A50/472—
Landscapes
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Enzymes And Modification Thereof (AREA)
Abstract
本发明包括用于重构肽分子的方法和组合物,包括向肽添加或删除一个或多个糖基基团,和/或向肽添加修饰基团。
The present invention includes methods and compositions for reconstructing peptide molecules, including adding or deleting one or more glycosyl groups to the peptide, and/or adding modifying groups to the peptide.
Description
Background
Most naturally occurring peptides contain a carbohydrate moiety attached to the peptide by specific bonding to a selected number of amino acids over the length of the primary peptide chain. Thus, many naturally occurring peptides are referred to as "glycopeptides". The variability of the glycosylation pattern on any given peptide is of great significance for the function of that peptide. For example, the structure of the N-linked glycans on the peptide can affect various characteristics of the peptide, including protease sensitivity, intracellular trafficking, secretion, tissue targeting, biological half-life, and antigenicity of the peptide in a cell or organism. Changes in one or more of these characteristics greatly affect the efficacy of the peptide in its natural environment, and also affect the efficacy of the peptide as a therapeutic agent in the conditions in which it was produced for that purpose.
Carbohydrate structures attached to peptide chains are known as "glycan" molecules. The presence of specific glycan structures on a peptide affects the solubility and aggregation characteristics of the peptide, the folding of the primary peptide chain and its function or enzymatic activity, the resistance of the peptide to proteolytic attack, and the control of proteolysis resulting in the conversion of the inactive form of the peptide to the active form. Importantly, the terminal sialic acid residues present on glycan molecules affect the length of the half-life of the peptide in the mammalian circulatory system. Peptides whose glycans do not contain terminal sialic acid residues will be rapidly removed from circulation by the liver, an event that negates any potential therapeutic benefit of the peptide.
Glycan structures found naturally on glycopeptides are generally classified into two classes, N-linked and O-linked.
Peptides expressed in eukaryotic cells are typically N-glycosylated at an asparagine residue at a site in the primary structure of the peptide that contains the sequence asparagine-X-serine/threonine, where X can be any amino acid except proline and aspartic acid. The carbohydrate moiety in such peptides is known as an N-linked glycan. The early events of N-glycosylation occur in the Endoplasmic Reticulum (ER) and are the same in mammals, plants, insects and other higher eukaryotes. First, an oligosaccharide chain comprising 14 sugar residues is constructed on a lipid carrier. When nascent peptides are translated and translocated into the ER, the entire oligosaccharide chain is then transferred to the amide group of an asparagine residue in a reaction catalyzed by membrane-bound glycosyltransferases. The N-linked glycans are further processed in both the ER and golgi. Further processing typically requires the removal of some sugar residues and the addition of other sugar residues in reactions catalyzed by glycosidases and glycosyltransferases, wherein the enzymes are specific for the removed and added sugar residues.
In general, the final structure of the N-linked glycan depends on the organism producing the peptide. For example, peptides produced by bacteria are typically completely aglycosylated. Peptides expressed in insect cells contain high mannose and paunci-mannose N-linked oligosaccharide chains, among others. Peptides produced in mammalian cell culture are typically differentially glycosylated depending on, for example, species and cell culture conditions. Even in the same species and under the same conditions, sometimes encounter some heterogeneity in glycosyl chains. Further, peptides produced in plant cells comprise glycan structures that are significantly different from those produced in animal cells. A dilemma in the field of producing recombinant peptides, particularly when the peptides are used as therapeutic agents, is the ability to produce correctly glycosylated peptides, i.e. to produce peptides having a glycan structure similar or identical to that present in the native form of the peptide. Most peptides produced by recombinant methods contain glycan structures that differ from naturally occurring glycans.
Various methods have been proposed in the art to tailor the glycosylation pattern of peptides, including those described in WO 99/22764, WO 98/58964, WO 99/54342, and U.S. Pat. No.5,047,335, among others. Basically, many enzymes necessary for the in vitro glycosylation of peptides have been cloned and sequenced. In some cases, these enzymes have been used in vitro to add specific sugars to incomplete glycan molecules on peptides. In other cases, the cell has been genetically engineered to express a combination of the enzyme and the desired peptide such that addition of the desired sugar moiety to the expressed peptide can occur in the cell.
Peptides can also be modified by the addition of O-linked glycans, also known as mucin-type glycans due to their ubiquitous presence on mucin glycopeptides. Unlike N-glycans, which are attached to asparagine residues and are formed by the integral transfer of oligosaccharides from lipid-bound intermediates, O-glycans are predominantly attached to serine and threonine residues and are formed by the stepwise addition of sugars from nucleotide sugars (Tanner et al, Biochim. Biophys. acta.906: 81-91 (1987); and Hounsell et al, Glycoconj.J.13: 19-26 (1996)). The function of the peptide may be influenced by the presence of an O-linked glycan structure thereon. For example, the activity of a P-selectin ligand is affected by the structure of the O-linked glycans present thereon. For a review of O-Linked glycan structures, see Schachter and Brockhausen, The biosyntheses of branched O-Linked Glycans, 1989, Society for Experimental biology, pp.1-26 (UK). Other glycosylation patterns are formed by attachment of glycosylphosphatidylinositol to the carboxy-terminal carboxyl group of proteins (Takeda et al, trends biochem. Sci.20: 367-.
Although various techniques currently exist for modifying the N-linked glycans of peptides, there is a need in the art for a general method of generating peptides having a desired, i.e., tailored, glycosylation pattern. There is a particular need in the art for in vitro glycosylation of peptides for customization (cusomize), wherein the resulting peptides can be produced on an industrial scale. The present invention fulfills this and other needs.
The administration of glycosylated and non-glycosylated peptides to produce a particular physiological response is well known in the medical arts. One of the best known peptides for this purpose is insulin, which is used to treat diabetes. Enzymes have also been used for their therapeutic benefits. The main factor limiting the use of therapeutic peptides is the immunogenic properties of most peptides. In a patient, an immunogenic response to the administered peptide can neutralize the peptide and/or lead to the development of an allergic response in the patient. Other deficiencies of therapeutic peptides include suboptimal efficacy and rapid clearance rates. Problems inherent to peptide therapeutics have been recognized in the art, and various approaches to eliminating the problems have been investigated. To provide soluble peptide therapeutics, synthetic polymers have been attached to the peptide backbone.
Poly (ethylene glycol) ("PEG") is an exemplary polymer that has been conjugated to a peptide. Derivatization of peptide therapeutics with PEG has been shown to reduce the immunogenicity of the peptide and prolong clearance time from circulation. For example, U.S. Pat. No.4,179,337(Davis et al) relates to non-immunogenic peptides such as enzymes and peptide hormones coupled to polyethylene glycol (PEG) or polypropylene glycol. 10 to 100 moles of polymer per mole of peptide is used and at least 15% of physiological activity is maintained.
WO 93/15189(Veronese et al) relates to a method of maintaining the activity of a polyethylene glycol modified proteolytic enzyme by linking the proteolytic enzyme to a macromolecular inhibitor. The conjugates are intended for medical applications.
The main mode of attachment of PEG and its derivatives to peptides is through nonspecific attachment of peptide amino acid residues. For example, U.S. patent No.4,088,538 discloses enzymatically active polymer-enzyme conjugates of enzymes covalently linked to PEG. Similarly, U.S. patent No.4,496,689 discloses complexes of an alpha-1 protease inhibitor covalently attached to a polymer such as PEG or methoxy poly (ethylene glycol) ("mPEG"). Abuchowski et al (J.biol. chem.252: 3578(1977)) disclose the covalent attachment of mPEG to the amino group of bovine serum albumin. U.S. patent No.4,414,147 discloses a method of making interferons less hydrophobic by conjugating them with anhydrides of dicarboxylic acids such as poly (ethylene succinic anhydride). PCT WO 87/00056 discloses the conjugation of PEG and polyoxyethylated polyols to proteins such as interferon-beta, interleukin-2 and immunotoxins. EP 154,316 discloses and claims chemically modified lymphokines such as IL-2 containing PEG directly attached to at least one primary amino group of the lymphokine. U.S. patent No.4,055,635 discloses pharmaceutical compositions of water-soluble complexes of proteolytic enzymes covalently linked to polymeric substances such as polysaccharides.
Another mode of attachment of PEG to peptides is through non-specific oxidation of glycosyl residues on the peptide. The oxidized saccharide can be used as a locus for attachment of the PEG moiety to the peptide. For example, M' Timkulu (WO94/05332) discloses the use of hydrazine-PEG or amino-PEG to add PEG to glycoproteins. The glycosyl moieties are randomly oxidized to the corresponding aldehyde and then coupled with amino-PEG. See also Bona et al (WO 96/40731) in which PEG is added to an immunoglobulin molecule by enzymatically oxidizing glycans on the immunoglobulin and then contacting the glycans with amino-PEG molecules.
In each of the above methods, poly (ethylene glycol) is added to the reactive residues of the peptide backbone in a random, non-specific manner. For the production of therapeutic peptides, it is clearly desirable to have a derivatization strategy that results in the formation of a specifically labeled, easily characterized and substantially homogeneous product.
Two major classes of enzymes are used in carbohydrate synthesis, glycosyltransferases (e.g., sialyltransferases, oligosaccharyltransferases, N-acetylglucosaminyltransferases) and glycosidases. Glycosidases are further classified as exoglycosidases (e.g., beta-mannosidase, beta-glucosidase) and endoglycosidases (e.g., Endo-A, Endo-M). Each of these classes of enzymes has been successfully synthesized for the production of sugars. For a general review, see Crout et al, curr, opin, chem, biol.2: 98-111(1998).
Glycosyltransferases modify the oligosaccharide structure on peptides. Glycosyltransferases are effective for producing specific products with good stereochemical and regiochemical (regiochemical) control. Glycosyltransferases have been used to prepare oligosaccharides and to modify terminal N-or O-linked carbohydrate structures, particularly on peptides produced by mammalian cells. For example, the terminal oligosaccharides of glycopeptides have been fully sialylated and/or fucosylated to provide a more robust sugar structure, which improves the pharmacokinetics and various other biological properties of glycopeptides. For example, β -1, 4-galactosyltransferases may be used in the synthesis of lactosamine, an example of a glycosyltransferase used in the synthesis of carbohydrates (see, e.g., Wong et al, J.org.chem.47: 5416-5418 (1982)). In addition, many synthetic procedures use α -sialyltransferase to transfer sialic acid from cytidine-5' -monophosphate-N-acetylneuraminic acid to the 3-OH or 6-OH of galactose (see, e.g., Kevin et al, chem. Eur. J.2: 1359-1362 (1996)). Fucosyltransferases can be used in the synthetic pathway to transfer fucose units from guanosine-5' -diphosphonite to specific hydroxyl groups of the sugar acceptor. Ichikawa, for example, prepared sialylic acid Lewis-X by a method involving fucosylation of sialylated lactosamine by cloned fucosyltransferases (Ichikawa et al, J.Am.chem.Soc.114: 9283-. For a discussion of recent advances in glycoconjugate synthesis for therapeutic use, see Koeller et al, nature bio technology 18: 835-841(2000). See also U.S. Pat. Nos. 5,876,980, 6,030,815, 5,728,554, 5,922,577 and WO/9831826.
Glycosidases may also be used to prepare sugars. Glycosidases generally catalyze the hydrolysis of glycosidic bonds. However, under appropriate conditions, they may be used to form the bond. Most glycosidases used for carbohydrate synthesis are exoglycosidases; glycosyl transfer occurs at the non-reducing end of the substrate. Glycosidases bind to glycosyl donors in glycosyl-enzyme intermediates that can be picked up by water to produce hydrolysates or by acceptors to produce new glycosides or oligosaccharides. An exemplary pathway for the use of exoglycosidases is the synthesis of the core trisaccharide of all N-linked glycopeptides, including the β -mannosidic bond formed by the action of β -mannosidase (Singh et al, chem. Commun.993-994 (1996)).
In another exemplary application of glycosidases to form glycosidic bonds, mutant glycosidases are made in which the normal nucleophilic amino acid of the active site is changed to a non-nucleophilic amino acid. The mutated enzyme does not hydrolyze the glycosidic bond, but can still form the bond. Such mutant glycosidases are useful for the preparation of oligosaccharides using alpha-glycosyl fluoride donor and glycoside acceptor molecules (Withers et al, U.S. Pat. No.5,716,812).
Although their use is less prevalent than exoglycosidases, endoglycosidases are also used to prepare sugars. The methods based on the use of endoglycosidases have the advantage that oligosaccharides can be transferred instead of monosaccharides. Oligosaccharide fragments have been added to substrates using endo-. beta. -N-acetylglucosamines such as endo-F, endo-M (Wang et al, Tetrahedron Lett.37: 1975-1978); and Haneda et al, carbohydr. res.292: 61-70(1996)).
In addition to their use in the preparation of saccharides, the enzymes described above may also be used in the synthesis of glycopeptides. The synthesis of the homogeneous sugar form of ribonuclease B (glycoform) has been published (Witte K. et al, J.Am.chem.Soc.119: 2114-2118 (1997)). The high mannose core of ribonuclease B can be cleaved by treating the glycopeptide with endoglycosidase H. This cleavage occurs specifically between the two core GlcNAc residues. Tetrasaccharide sialyl Lewis X can then be enzymatically reconstructed on the remaining GlcNAc anchor sites on the homogeneous protein by applying β -1, 4-galactosyltransferase, α -2, 3-sialyltransferase and α -1, 3-fucosyltransferase in that order. However, although each enzymatic step proceeds in excellent yields, this procedure is not suitable for the production of glycopeptides on an industrial scale.
Methods of combining chemical and enzymatic synthesis elements are also well known in the art. For example, Yamamoto and colleagues (carbohydrate. Res. 305: 415-422(1998)) reported the chemoenzymatic (chemoenzymatic) synthesis of glycopeptides, glycosylated peptide T, using endoglycosidases. N-acetylglucosamine peptides are synthesized entirely by chemical methods. The peptide was then enzymatically treated elaborately with the oligosaccharide of the human transferrin peptide. The sugar moiety is added to the peptide by treatment with endo-beta-N-acetylglucosaminidase (acetylglucosaminidase). The resulting glycosylated peptide is highly stable and resistant to proteolysis compared to peptide T and N-acetylglucosamine peptide T.
The use of glycosyltransferases for modifying peptide structures with reporter groups has also been explored. For example, Brossmer et al (U.S. Pat. No.5,405,753) disclose the formation of fluorescently labeled cytidine monophosphate ("CMP") derivatives of sialic acid and the use of fluorescent glycosides to determine sialyltransferase activity and fluorescent labeling of cell surfaces, glycoproteins and peptides. Similar assays are described by Gross et al (Analyt. biochem.186: 127 (1990)). Bean et al (U.S. Pat. No.5,432,059) disclose the determination of glycosylation deficient disorders using the re-glycosylation of insufficiently glycosylated proteins. The defective protein is re-glycosylated with a fluorescently labeled CMP glycoside. Each fluorescent sialic acid derivative is substituted with a fluorescent moiety at the 9-position or amine of sialic acid, which is usually acetylated. The method of using fluorescent sialic acid derivatives is the determination of the presence of glycosyltransferases or glycoproteins that are not glycosylated or are incorrectly glycosylated. The assay is performed on small amounts of enzymes or glycoproteins in a sample of biological origin. Enzymatic derivatization of glycosylated or non-glycosylated peptides on the preparative or industrial scale using modified sialic acids has not been disclosed or suggested in the prior art.
There has also been considerable effort directed to modifying the cell surface by altering the glycosyl residues presented by the cell surface. For example, Fukuda and coworkers have developed methods for attaching glycosides with defined structures to cell surfaces. The method utilizes the relaxed substrate specificity of fucosyltransferases, which can transfer fucose and fucose analogues with different glycosyl substrates (Tsuboi et al, J.biol.chem.271: 27213 (1996)).
Enzymatic methods have also been used to activate glycosyl residues on glycopeptides to facilitate subsequent chemical processing. Glycosyl residues are typically activated with galactose oxidase, which converts the terminal galactose residue to the corresponding aldehyde. The aldehyde is then coupled with a modifying group comprising ammonia. For example, Casares et al (Nature Biotech.19: 142(2001)) have attached doxorubicin to the oxidized galactose residue of recombinant MHCII-peptide chimeras.
Glycosyl residues have also been modified to contain ketone groups. For example, N-acetylpropionylmannosamine ("ManLev") has been prepared by Mahal and colleagues (Science 276: 1125(1997)) which has a ketone functionality in the position normally occupied by an acetyl group in natural substrates. Cells were treated with ManLev to integrate the ketone group into the cell surface. See also, Saxon et al, Science 287: 2007 (2000); hang et al, j.am.chem.soc.123: 1242 (2001); yarema et al, j.biol.chem.273: 31168 (1998); and Charter et al, Glycobiology 10: 1049(2000).
Methods of modifying the surface of cells have not been applied to modifying glycosylated or non-glycosylated peptides in the absence of cells. Further, cell surface modification methods have not been used to enzymatically incorporate modified glycosyl donor moieties into peptides. Furthermore, none of the cell surface modification methods is feasible in the production of glycosyl-modified peptides on an industrial scale.
Despite efforts directed to enzymatic treatment of carbohydrate structures, there remains a need for industrially feasible methods for modifying glycosylated and non-glycosylated peptides with modifying groups such as water-soluble polymers, therapeutic moieties, biomolecules, and the like. Of particular interest are methods in which the modified peptides have improved properties that enhance the use of the peptides as therapeutic or diagnostic agents. The present invention meets these and other needs.
Summary of The Invention
The present invention includes a number of methods for reconstituting peptides to have glycan structures attached thereto. Although specific glycan structures are described herein, the present invention should not be construed as limited to any one particular structure. Furthermore, although specific peptides are described herein, the invention should not be limited by the characteristics of the described peptides, but rather should encompass any or all suitable peptides and variants thereof.
The following description discloses preferred embodiments of the invention and provides a written description of the appended claims. The present invention includes variations of any and all of these embodiments that may be or become apparent upon reading the present specification.
The present invention includes a cell-free in vitro method of reconstituting a polyethylene glycol-containing peptide having the formula:
wherein
AA is a terminal or internal amino acid residue of the peptide;
X1-X2is a sugar covalently linked to AA, wherein
X1Is the first oneA glycosyl residue; while
X2Is a reaction of with X1A covalently linked second glycosyl residue wherein X1And X2Selected from the group consisting of monosaccharide and oligosaccharide residues, the method comprising:
(a) removal of X from peptides2Or a saccharide subunit thereof, thereby forming a truncated glycan.
In one aspect, the invention further comprises forming truncated glycans by removing Sia residues.
In one embodiment of the invention, the peptide has the formula:
wherein
X3、X4、X5、X6、X7And X17Is an independently selected monosaccharide or oligosaccharide residue; and
a. b, c, d, e and x are independently selected from the integers 0, 1 and 2.
In one aspect of the invention, the oligosaccharide residue is a member selected from GlcNAc-Gal-Sia and GlcNAc-Gal. In another aspect, at least one oligosaccharide member is selected from a, b, c, d, e and x is 1 or 2. In another aspect, the removing in step (a) produces truncated glycans in which at least one of a, b, c, e, and x is 0.
The invention includes methods of reconstituting peptides wherein X3、X5And X7Is independently selected from (mannose)zAnd (mannose)z-(X8) Members of (2)
Wherein
X8Is a glycosyl moiety selected from mono-and oligo-saccharides; and
z is an integer between 1 and 20, wherein
When z is 3 or more, each (mannose)zIndependently selected from linear and branched structures.
In one aspect, X4Selected from GlcNAc and xylose. In another aspect, X3、X5And X7Is (mannose)uWherein u is selected from an integer between 1 and 20, and when u is 3 or greater, each (mannose)uIndependently selected from linear and branched structures.
The invention also includes a method of reconstituting a peptide, wherein the peptide has the formula:
wherein
r, s and t are integers independently selected from 0 and 1.
In one embodiment of the invention, the peptide has the formula:
wherein
X9And X10Is an independently selected monosaccharide or oligosaccharide residue; and
m, n and f are integers independently selected from 0 and 1.
In one aspect, the peptide has the formula:
wherein
X16Is selected from the following formulasMember (b):
wherein,
s and i are integers independently selected from 0 and 1.
In another aspect, the peptide has the formula:
X13、X14And X15Independently selected from glycosyl residues; and
g. h, i, j, k, and p are independently selected from the integers 0 and 1.
In another aspect of the present invention, at least one of g, h, i, j, k, and p is 1. In another aspect, X14And X15Is a member independently selected from GlcNAc and Sia, and i and k are independently selected from the integers 0 and 1. In another aspect, at least one of i and k is 1, and if k is 1, then g, h, and j are 0.
The invention also includes a method of reconstituting a peptide, wherein the method comprises contacting a truncated glycan with at least one glycosyltransferase and at least one glycosyl donor under conditions suitable for transferring the at least one glycosyl donor to the truncated glycan, thereby reconstituting a peptide comprising polyethylene glycol.
In one aspect, the glycosyl donor comprises a modifying group covalently attached thereto.
The invention also includes a method of reconstituting a peptide comprising removing X1Thereby exposing AA. In one aspect, the method comprises contacting AA with at least one glycosyltransferase and at least one glycosyl donor in a suitable containerUnder conditions that transfer the at least one glycosyl donor to AA, thereby reconstituting the peptide comprising polyethylene glycol.
In one aspect, at least one glycosyl donor comprises a modifying group covalently attached thereto. In another aspect, the modifying group is polyethylene glycol. In one embodiment, the polyethylene glycol has a substantially monodisperse (homodispersee) molecular weight distribution.
The invention includes a method of reconstituting a peptide, wherein groups added to the saccharide during post-translational modification are removed prior to contacting the truncated glycan with at least one glycosyltransferase and at least one glycosyl donor under conditions suitable for transferring the at least one glycosyl donor to the truncated glycan, thereby reconstituting the peptide comprising polyethylene glycol.
In one aspect, the group removed is a member selected from the group consisting of phosphoric acid, sulfuric acid, carboxylic acid, and esters thereof.
The present invention includes a method of reconstituting a peptide, wherein the peptide has the formula:
wherein
Z is a member selected from O, S, NH and a crosslinker.
The invention also includes a method of reconstituting a peptide, wherein the peptide has the formula:
wherein
X11And X12Is an independently selected glycosyl moiety; and is
r and x are integers independently selected from 0 and 1.
In one aspect of the invention, X11And X12Is (mannose)qWherein q is an integer selected from 1 to 20, and when q is 3 or more, (mannose)qSelected from linear and branched structures.
The present invention includes a pharmaceutical composition comprising a pharmaceutically acceptable diluent and a peptide reconstituted according to a cell-free in vitro method of reconstituting a peptide comprising polyethylene glycol, said peptide having the formula:
Wherein
AA is a terminal or internal amino acid residue of the peptide;
X1-X2is a glycosyl residue covalently attached to AA, wherein
X1Is the first glycosyl residue; and is
X2Is a reaction of with X1A covalently linked second glycosyl residue wherein X1And X2Selected from the group consisting of monosaccharide and oligosaccharide residues;
the method comprises the following steps:
(a) removal of X from the peptide2Or a saccharide subunit thereof, thereby forming a truncated glycan.
The invention also includes a cell-free in vitro method of reconstituting a peptide comprising polyethylene glycol, said peptide having the formula:
wherein
AA is a terminal or internal amino acid residue of the peptide;
X1is a glycosyl residue covalently attached to AA selected from the group consisting of monosaccharide and oligosaccharide residues; and is
u is an integer selected from 0 and 1,
the method comprises the following steps:
the peptide is reconstituted by contacting the peptide with at least one glycosyltransferase and at least one glycosyl donor under conditions suitable for transfer of the at least one glycosyl donor to the truncated glycan.
In one aspect, at least one glycosyl donor comprises a modifying group covalently attached thereto. In another aspect, the modifying group is polyethylene glycol. In another aspect, the polyethylene glycol has a substantially monodisperse (homodispersee) molecular weight distribution.
The invention also includes a pharmaceutical composition comprising a pharmaceutically acceptable diluent and a peptide reconstituted according to a cell-free in vitro method of reconstituting a peptide comprising polyethylene glycol, said peptide having the formula:
Wherein
AA is a terminal or internal amino acid residue of the peptide;
X1is a glycosyl residue covalently attached to AA selected from the group consisting of monosaccharide and oligosaccharide residues; and is
u is an integer selected from 0 and 1,
the method comprises the following steps:
the peptide is reconstituted by contacting the peptide with at least one glycosyltransferase and at least one glycosyl donor under conditions suitable for transfer of the at least one glycosyl donor to the truncated glycan.
Brief Description of Drawings
For the purpose of illustrating the invention, certain embodiments of the invention are illustrated in the accompanying drawings. However, the invention is not limited to the precise arrangements and instrumentalities of the embodiments depicted in the drawings.
FIG. 1 is a schematic diagram depicting the trimannosyl decorin (left) and the enzymatic process for producing glycans with bisected (bisecting) GlcNAc (right).
FIG. 2 is a schematic diagram depicting the basic trimannosyl core structure and the complex chains of various degrees of completion. Methods for the in vitro enzymatic production of the basic trimannosyl core structure from complex carbohydrate glycan structures that do not contain an aliquot of GlcNAc residues and the production of glycan structures containing an aliquot of GlcNAc are shown. Symbol: square: GlcNAc; light circle: man; black circle: gal; triangle: NeuAc.
FIG. 3 is a schematic representation of the enzymatic production of sialylated glycan structures (right) starting with a trimannosyl core and an aliquot of GlcNAc (left).
FIG. 4 is a schematic of a typical glycan structure with high mannose content (left) and an enzymatic method to reduce the structure to the basic trimannosyl core structure. In this schematic, X is mannose as a monosaccharide, oligosaccharide or polysaccharide.
FIG. 5 is a schematic representation of the structure of N-linked glycans containing fucose and xylose, typically produced in plant cells.
FIG. 6 is a schematic representation of fucose-containing N-linked glycans typically produced in insect cells. Note that the glycan may not have core fucose, it may have mononuclear fucose of any one kind of bond, or it may have mononuclear fucose in which one kind of bond is dominant.
Fig. 7 is a schematic diagram depicting various routes of pruning high mannose structures and synthesizing complex sugar chains therefrom. Symbol: square: GlcNAc; circle: man; diamond shape: fucose; a pentagon: xylose.
FIG. 8 is a schematic depicting an in vitro strategy for the synthesis of complex structures from a basic trimannosyl core structure. Symbol: square: GlcNAc; light circle: man; black circle: gal; black triangle: NeuAc; GnT: n-acetylglucosaminyltransferase; GalT: a galactosyltransferase; ST: sialyltransferase.
Figure 9 is a schematic depicting two in vitro strategies for the synthesis of monoantennary glycans and optionally saccharide pegylation of such glycans. Black squares: GlcNAc; black circle: man; light circle: gal; black triangle: sialic acid.
Figure 10 is a schematic depicting two in vitro strategies for the synthesis of monoantennary glycans and optionally saccharide pegylation of such glycans. Black squares: GlcNAc; black circle: man; light circle: gal; black triangle: sialic acid.
FIG. 11 is a schematic diagram depicting various complex structures that can be synthesized from the basic trimannosyl core structure. Symbol: square: GlcNAc; light circle: man; black circle: gal; triangle: NeuAc; diamond shape: fucose; FT and FucT: a fucosyltransferase; GalT: a galactosyltransferase; ST: sialyltransferase; le: a Lewis antigen; SLe: sialylated Lewis antigens.
FIG. 12 is an exemplary schematic for the preparation of O-linked glycopeptides derived from serine or threonine. Optionally, a Water Soluble Polymer (WSP) such as polyethylene glycol is added to the final glycan structure.
FIG. 13 is a series of diagrams depicting the structure of class 4O-glycans, referred to as cores 1-4. The core structure is depicted with dashed lines.
FIG. 14, comprising FIGS. 14A and 14B, is a series of schematic diagrams representing exemplary embodiments of the present invention in which sugar residues comprising complex sugar structures and/or high mannose structures are trimmed to the structure of the first generation biantennary. Optionally, fucose is added only after reaction with the GnTI. The modified saccharide with the Water Soluble Polymer (WSP) is then conjugated to one or more saccharide residues exposed by the trimming process.
Fig. 15 is a schematic diagram similar to that shown in fig. 4, in which a high mannose structure (high mannose structure) or complex structure is "trimmed" to a mannose β -linked core, and then a modified saccharide having a water soluble polymer is conjugated to one or more saccharide residues exposed by the trimming process. The sugars are added sequentially with glycosyltransferase.
FIG. 16 is a schematic diagram similar to that shown in FIG. 4, in which the high mannose structure or complex is "trimmed" to the GlcNAc to which the first mannose is attached, and then a modified sugar having a water soluble polymer is conjugated to one or more sugar residues exposed by the trimming process. The sugars are added sequentially with glycosyltransferase.
Fig. 17 is a schematic diagram similar to that shown in fig. 4, in which the high mannose structure or complex is "trimmed" until the first GlcNAc attached to the Asn of the peptide, and then a water soluble polymer is conjugated to one or more sugar residues that have been subsequently added. The sugars are added sequentially with glycosyltransferase.
FIG. 18, comprising FIG. 18A and FIG. 18B, is a schematic representation of the optional trimming of N-linked sugars from a high mannose structure or complex and subsequent derivatization with a modified sugar moiety (Gal or GlcNAc) with a water soluble polymer.
Figure 19, comprising figures 19A and 19B, is a schematic of the trimming of N-linked saccharides from a high mannose structure or complex and subsequent derivatization with sialic acid moieties with water soluble polymers. The sugars are added sequentially with glycosyltransferase.
Figure 20 is a schematic of sialic acid termination with derivatization with water soluble polymers, optionally trimming of N-linked saccharides from high mannose structures or complex structures and subsequent derivatization with one or more sialic acid moieties. The sugars are added sequentially with glycosyltransferase.
FIG. 21 is a schematic of "trimming" of O-linked saccharides followed by conjugation with modified saccharides having water-soluble polymers. In an exemplary schematic, the sugar moiety is "trimmed" up to the first generation of the dual-antenna structure.
FIG. 22 illustrates the trimming of the saccharide moiety of an O-linked glycopeptide to yield mannose that can be used to conjugate with a modified saccharide having a water-soluble polymer attached thereto.
Fig. 23, which includes fig. 23A to 23C, is a series of exemplary schematic diagrams. Figure 23A is a schematic illustrating the addition of pegylated sugars and the subsequent addition of unmodified sugars. Figure 23B is a schematic illustrating the addition of more than one modified sugar to one glycan. FIG. 23C is a schematic illustrating the addition of different modified sugars to O-linked glycans and N-linked glycans.
Figure 24 is an illustration of various methods of improving the therapeutic function of peptides through glycan remodeling (including conjugation).
FIG. 25 is a schematic representation of glycan remodeling with a set of therapeutic peptides for treatment of Gaucher's disease.
FIG. 26 is a schematic of glycan remodeling to generate glycans with terminal mannose-6-phosphate moieties.
FIG. 27 is a graph illustrating CHO-produced glucocerebrosidase (Cerezyme) after sialylationTM) Schematic of the structural arrangement of glycans above.
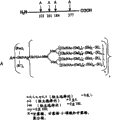
FIG. 28, comprising FIGS. 28A-28Z and FIGS. 28 AA-28 CC, is a listing of peptides used in the methods of the present invention.
FIG. 29, comprising FIGS. 29A-29G, provides exemplary diagrams for remodeling glycan structures on granulocyte colony stimulating factor (G-CSF). Fig. 29A is a diagram depicting the molecular formulae of G-CSF peptides and exemplary glycans attached thereto, showing amino acid residues to which the glycans are attached. FIGS. 29B-29G are prospective reconstitution steps performed on the glycans of the peptide in FIG. 29A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 30, comprising fig. 30A-30 EE, presents exemplary schematic diagrams of the reconstruction of glycan structures on interferon alpha. Fig. 30A is a diagram depicting the molecular formulae of an interferon alpha isoform 14c peptide and exemplary glycans attached thereto, showing the amino acid residues to which the glycans are attached. FIGS. 30B-30D are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 30A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 30E is a diagram depicting the molecular formulae of an interferon alpha isoform 14c peptide and exemplary glycans attached thereto, showing the amino acid residues to which the glycans are attached. FIGS. 30F-30N are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 30E based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 30O is a diagram depicting the molecular formulae of an interferon alpha isoform 2a or 2b peptide and exemplary glycans attached thereto, showing the amino acid residues to which the glycans are attached. FIGS. 30P-30W are graphical illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 30O based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 30X is a diagram depicting the molecular formulae of an interferon alpha-mucin fusion peptide and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 30Y-30 AA are illustrations of the expected reconstitution steps performed on the glycans of the peptides in FIG. 30X based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 30BB is a diagram depicting the molecular formulae of interferon alpha-mucin fusion peptides and interferon alpha peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 30 CC-30 EE are illustrations of the expected reconstitution steps performed on the glycans of the peptide in FIG. 30BB based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 31, comprising fig. 31A-31S, provides exemplary schematic diagrams of the reconstruction of glycan structures on interferon beta. Fig. 31A is a diagram depicting the molecular formulae of interferon-beta peptides and exemplary glycans attached thereto, showing the amino acid residues to which the glycans are attached. FIGS. 31B-31O are illustrations of the expected reconstitution steps performed on the glycans of the peptide in FIG. 31A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 31P is a diagram depicting the molecular formulae of interferon-beta peptides and exemplary glycans attached thereto, showing the amino acid residues to which the glycans are attached. FIGS. 31Q-31S are illustrations of the expected reconstitution steps performed on the glycans of the peptides in FIG. 31P based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 32, comprising fig. 32A-32D, presents exemplary schematic diagrams of the reconstruction of glycan structures on factor VII and factor VIIa. Fig. 32A is a graph depicting the formula of factor VII and factor VIIa peptides a (solid line) and B (dashed line) and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 32B-32D are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 32A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 33, comprising fig. 33A-33G, provides exemplary schematic illustrations of the reconstitution of glycan structures on factor IX. Figure 33A is a graph depicting the molecular formulae of coagulation factor IX peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 33B-33G are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 33A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 34, which comprises fig. 34A-34J, presents exemplary schematic diagrams of the reconstruction of glycan structures on Follicle Stimulating Hormone (FSH), including the alpha and beta subunits. Fig. 34A is a diagram depicting the molecular formulae of the follicle stimulating hormone peptides FSH α and FSH β and exemplary glycans attached thereto, showing residues attached to the glycan expected to be reconstituted. FIGS. 34B-34J are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 34A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Figure 35, comprising figures 35A-35 AA, provides exemplary schematic diagrams of the reconstitution of glycan structures on Erythropoietin (EPO). Fig. 35A is a graph depicting the molecular formulae of EPO peptides and glycans, showing residues attached to glycans expected to be reconstituted. FIGS. 35B-35S are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 35A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 35T is a graph depicting the molecular formulae of EPO peptides and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 35U-35W are illustrations of the expected reconstitution steps performed on the glycans of the peptides in FIG. 35T based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 35X is a graph depicting the molecular formulae of EPO peptides and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 35Y-35 AA are illustrations of the expected reconstitution steps performed on the glycans of the peptides in FIG. 35X based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 36, comprising FIGS. 36A-36K, provides an exemplary schematic of the reconstitution of glycan structures on granulocyte macrophage colony stimulating factor (GM-CSF). FIG. 36A is a graph depicting the molecular formulae of GM-CSF peptide and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 36B-36G are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 36A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 36H is a graph depicting the molecular formulae of GM-CSF peptide and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 36I-36K are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 36H based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 37, comprising fig. 37A-37N, provides an exemplary schematic of the reconstruction of glycan structures on an interferon gamma peptide. Fig. 37A is a graph depicting the molecular formulae of interferon gamma peptides and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 37B-37G are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 37A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 37H is a diagram depicting the molecular formulae of an interferon gamma peptide and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 37I-37N are graphical illustrations of the expected reconstitution procedure for the peptide of FIG. 37H based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 38 contains FIGS. 38A-38N, giving pairs α1Exemplary schematic representation of the reconstitution of glycan structures on antitrypsin (ATT or alpha-1 protease inhibitor). Fig. 38A is a graph depicting the molecular formulae of ATT peptides and exemplary glycans attached thereto, showing residues bound to glycans expected for reconstitution. FIGS. 38B-38F are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 38A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 38G is a graph depicting the molecular formulae of ATT peptides and exemplary glycans attached thereto, showing residues bound to glycans expected for reconstitution. FIGS. 38H-38J are illustrations of the expected reconstitution steps performed on the peptide of FIG. 38G based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 38K is a graph depicting the molecular formulae of ATT peptides and exemplary glycans attached thereto, showing residues bound to glycans expected for reconstitution. FIGS. 38L-38N are illustrations of the expected reconstitution steps performed on the peptide of FIG. 38K based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 39, comprising FIGS. 39A-39J, provides exemplary schematic diagrams of the reconstruction of glycan structures on glucocerebrosidase. FIG. 39A is a graph depicting the molecular formulae of glucocerebrosidase peptide and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 39B-39F are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 39A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 39G is a graph depicting the molecular formulae of glucocerebrosidase peptide and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 39H-39K are illustrations of expected reconstitution procedures for the glycans of the peptides in FIG. 39G based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 40, comprising FIGS. 40A-40W, shows an exemplary schematic of the reconstitution of glycan structures on tissue-Type Plasminogen Activator (TPA). Fig. 40A is a graph depicting the molecular formulae of TPA peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 40B-40G are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 40A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 40H is a graph depicting the molecular formulae of TPA peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 40I-40K are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 40H based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 40L is a graph depicting the molecular formulae of mutated TPA peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 40M-40O are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 40L based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 40P is a graph depicting the molecular formulae of mutated TPA peptides and glycans, showing residues bound to glycans expected to be reconstituted. FIGS. 40Q-40S are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 40P based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 40T is a graph depicting the molecular formulae of mutated TPA peptides and glycans, showing residues attached to glycans expected to be reconstituted. FIGS. 40U-40W are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 40T based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 41, comprising FIGS. 41A-41G, provides exemplary schematic diagrams of the reconstruction of glycan structures on interleukin-2 (IL-2). FIG. 41A is a diagram depicting the molecular formulae of interleukin-2 peptides and exemplary glycans attached thereto, showing amino acid residues to which the glycans are attached. FIGS. 41B-41G are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 41A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Fig. 42, comprising fig. 42A-42M, provides exemplary schematic diagrams of the reconstruction of glycan structures on factor VIII. FIG. 42A is a molecular formula of glycans bound to N-linked glycosylation sites (A and A') and O-linked sites (B) of a factor VIII peptide. FIGS. 42B-42F are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 42A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 42G is a molecular formula of glycans bound to N-linked glycosylation sites (A and A') and O-linked sites (B) of a factor VIII peptide. FIGS. 42H-42M are graphical illustrations of the expected reconstitution procedure for the peptide of FIG. 42G based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 43 comprises FIGS. 43A-43M, which provide exemplary schematic illustrations of the reconstitution of glycan structures on urokinase. Figure 43A is a graph depicting the molecular formulae of urokinase peptide and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 43B-43F are illustrations of expected reconstitution procedures for the glycans of the peptides in FIG. 43A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 43G is a graph depicting the molecular formulae of urokinase peptide and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 43H-43L are illustrations of expected reconstitution procedures performed on the glycans of the peptides in FIG. 43G based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 44 contains FIGS. 44A-44J, which present exemplary schematic diagrams of the reconstruction of glycan structures on human DNase (hDNase). Fig. 44A is a graph depicting the molecular formulae of human DNase peptides and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 44B-44F are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 44A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 44G is a diagram depicting the molecular formulae of human DNase peptides and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 44H-44J are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 44F based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 45, comprising FIGS. 45A-45L, provides an exemplary schematic of the reconstruction of glycan structures on insulin. FIG. 45A is a diagram depicting the molecular formulae of an insulin peptide containing an N-glycosylation site after mutation and exemplary glycans attached thereto. FIGS. 45B-45D are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 45A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 45E is a diagram depicting the molecular formulae of an insulin-mucin fusion peptide and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 45F-45H are illustrations of the expected reconstitution steps performed on the peptide of FIG. 45E based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 45I is a diagram depicting the molecular formulae of insulin-mucin fusion peptides and insulin peptides and glycans, showing residues attached to glycans expected to be reconstituted. FIGS. 45J-45L are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 45I based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 46, comprising FIGS. 46A-46K, provides exemplary schematic diagrams of the reconstitution of glycan structures on the M-antigen (preS and S) of hepatitis B surface protein (HbsAg). Fig. 46A is a graph depicting the molecular formulae of M-antigen peptides and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 46B-46G are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 46A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 46H is a diagram depicting the molecular formulae of M-antigen peptides and glycans, showing residues bound to glycans expected for reconstitution. FIGS. 46I-46K are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 46H based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 47, comprising FIGS. 47A-47K, presents exemplary schematic diagrams of the reconstruction of glycan structures on human growth hormone (including N, V and variants thereof). Figure 47A is a graph depicting the molecular formulae of human growth hormone peptides and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 47B-47D are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 47A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Fig. 47E is a diagram depicting the molecular formulae of 3 fusion peptides comprising part or all of human growth hormone peptide and mucin peptide and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 47F-47K are illustrations of expected reconstitution procedures for the glycans of the peptide in FIG. 47E based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 48 contains FIGS. 48A to 48G, which show fusion proteins to the Fc region of TNF receptor-IgG (Enbrel)TM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 48A is a diagram depicting the molecular formulae of TNF receptor-IgG Fc region fusion peptides and glycans that can be mutated to contain additional N-glycosylation sites, showing residues that bind to glycans expected for reconstitution. TNF receptor peptides are depicted in bold lines, while IgG Fc regions are depicted in plain lines. FIGS. 48B-48G are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 48A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 49, comprising FIGS. 49A-49D, shows an anti-HER 2 monoclonal antibody (Herceptin)TM) Exemplary schematic of the above glycan structures for reconstitution. Figure 49A is a graph depicting the molecular formulae of a mutated anti-HER 2 monoclonal antibody containing an N-glycosylation site and an exemplary glycan attached thereto, showing residues on the heavy chain of the antibody attached to the glycan expected to be reconfigured. FIGS. 49B-49D are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 49A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 50 contains FIGS. 50A-50D, which show monoclonal antibodies to respiratory syncytial virus protein F (Synagis)TM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 50A is a graph depicting the molecular formulae of monoclonal antibodies mutated to protein F peptides containing N-glycosylation sites and exemplary glycans attached thereto, showing attachment to expected peptidesResidues on the reconstructed glycans were aligned. FIGS. 50B-50D are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 50A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 51 contains FIGS. 51A-51D, which show monoclonal antibodies (Remicade) to TNF- α TM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 51A is a graph depicting the molecular formulae of a monoclonal antibody to TNF- α containing N-glycosylation sites and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 51B-51D are graphical illustrations of the expected reconstitution steps performed on the peptide of FIG. 51A based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 52 contains FIGS. 52A-52L, which show monoclonal antibodies (Reopro) to glycoprotein IIb/IIIaTM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 52A is a graph depicting the molecular formula of a monoclonal antibody mutated to a glycoprotein IIb/IIIa peptide containing an N-glycosylation site and exemplary glycans attached thereto, showing residues bound to the glycans expected for reconstitution. FIGS. 52B-52D are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 52E is a graph depicting the molecular formulae of monoclonal antibodies to glycoprotein IIb/IIIa-mucin fusion peptides and exemplary glycans attached thereto, showing residues bound to glycans expected to be reconstituted. FIGS. 52F-52H are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 52I is a graph depicting the molecular formulae of monoclonal antibodies to glycoprotein IIb/IIIa-mucin fusion peptides and glycoprotein IIb/IIIa peptides and exemplary glycans attached thereto, showing residues that bind to glycans expected to be reconstituted. FIGS. 52J-52L are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 53 comprises FIGS. 53A-53G, which show monoclonal antibodies to CD20 (Rituxan)TM) Exemplary schematic of the above glycan structures for reconstitution. Figure 53A is a graph depicting the molecular formulae of a monoclonal antibody mutated to CD20 containing an N-glycosylation site and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 53B-53D are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 53A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. Figure 53E is a graph depicting the molecular formulae of a monoclonal antibody mutated to CD20 containing an N-glycosylation site and exemplary glycans attached thereto, showing residues attached to glycans expected to be reconstituted. FIGS. 53F-53G are illustrations of expected reconstitution steps performed on the glycans of the peptides in FIG. 53E based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 54, comprising FIGS. 54A-54O, shows an exemplary schematic of the reconstruction of glycan structures on antithrombin III (AT III). FIG. 54A is a diagram depicting the formula of an antithrombin III peptide and exemplary glycans attached thereto, showing amino acid residues attached to N-linked glycans. FIGS. 54B-54G are illustrations of expected reconstitution steps performed on the glycans of the peptide in FIG. 54A based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 54H is a diagram depicting the formula of an antithrombin III peptide and exemplary glycans attached thereto, showing amino acid residues attached to N-linked glycans. FIGS. 54I-54K are illustrations of expected reconstitution procedures for the glycans of the peptide in FIG. 54H based on the type of cell expressing the peptide and the desired reconstituted glycan structure. FIG. 54L is a diagram depicting the formula of an antithrombin III peptide and exemplary glycans attached thereto, showing amino acid residues attached to N-linked glycans. FIGS. 54M-54O are illustrations of expected reconstitution procedures for the glycans of the peptides in FIG. 54L based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
Figure 55, comprising figures 55A-55J, provides exemplary schematic diagrams of the reconstruction of glycan structures on the alpha and beta subunits of human chorionic gonadotropin (hCG). Figure 55A is a diagram depicting hCG α and hCG β peptides showing the amino acid residues bound to N-linked glycan (a) and O-linked glycan (B) and the molecular formulae of the glycans that are expected to undergo reconstitution. FIGS. 55B-55J are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 56 contains FIGS. 56A-56J, which show para-alpha-galactosidase (Fabrazyme)TM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 56A is a diagram depicting an α -galactosidase A peptide, showing amino acid residues bound to N-linked glycans (A) expected to be reconstituted and the molecular formula of the glycans. FIGS. 56B-56J are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 57 contains FIGS. 57A-57J, which show para-alpha-iduronidase (Aldura zyme)TM) Exemplary schematic of the above glycan structures for reconstitution. FIG. 57A is a diagram depicting α -iduronidase peptides showing amino acid residues bound to N-linked glycans (A) expected to be reconstituted and the molecular formula of the glycans. FIGS. 57B-57J are graphical illustrations of expected reconstitution procedures based on the type of cell expressing the peptide and the desired reconstituted glycan structure.
FIG. 58 contains FIGS. 58A and 58B, are exemplary nucleotide and corresponding amino acid sequences of granulocyte colony stimulating factor (G-CSF) (SEQ ID NOS: 1 and 2, respectively).
FIG. 59, comprising FIGS. 59A and 59B, is an exemplary nucleotide and corresponding amino acid sequence of interferon alpha (IFN-alpha) (SEQ ID NOS: 3 and 4, respectively).
FIG. 60 contains FIGS. 60A and 60B, which are exemplary nucleotide and corresponding amino acid sequences of interferon beta (IFN- β) (SEQ ID NOS: 5 and 6, respectively).
FIG. 61 comprises FIGS. 61A and 61B, exemplary nucleotide and corresponding amino acid sequences of factor VIIa (SEQ ID NOS: 7 and 8, respectively).
FIG. 62 comprises FIGS. 62A and 62B, is an exemplary nucleotide and corresponding amino acid sequence of coagulation factor IX (SEQ ID NOS: 9 and 10, respectively).
FIG. 63 includes FIGS. 63A-63D, exemplary nucleotides and corresponding amino acid sequences of Follicle Stimulating Hormone (FSH) (SEQ ID NOs: 11-14, respectively).
FIG. 64, comprising FIGS. 64A and 64B, is an exemplary nucleotide and corresponding amino acid sequence of Erythropoietin (EPO) (SEQ ID NOS: 15 and 16, respectively).
FIG. 65 is the amino acid sequence of mature EPO, 165 amino acids (SEQ ID NO: 73).
FIG. 66 contains FIGS. 66A and 66B, is an exemplary nucleotide and corresponding amino acid sequence of granulocyte-macrophage colony-stimulating factor (GM-CSF) (SEQ ID NOs: 17 and 18, respectively).
FIG. 67 contains FIGS. 67A and 67B, is an exemplary nucleotide and corresponding amino acid sequence of interferon gamma (IFN-. gamma.) (SEQ ID NOS: 19 and 20, respectively).
FIG. 68 contains FIGS. 68A and 68B, are exemplary nucleotide and corresponding amino acid sequences (SEQ ID NOS: 21 and 22, respectively) of an alpha-1 protease inhibitor (A-1-PI or alpha-antitrypsin).
FIG. 69, comprising FIGS. 69A-1-69A-2 and 69B, is an exemplary nucleotide and corresponding amino acid sequence of glucocerebrosidase (SEQ ID NOS: 23 and 24, respectively).
FIG. 70, comprising FIGS. 70A and 70B, is an exemplary nucleotide and corresponding amino acid sequence of tissue Type Plasminogen Activator (TPA) (SEQ ID NOS: 25 and 26, respectively).
FIG. 71, comprising FIGS. 71A and 71B, is an exemplary nucleotide and corresponding amino acid sequence of interleukin-2 (IL-2) (SEQ ID NOS: 27 and 28, respectively).
FIG. 72 comprises FIGS. 72A-1-71A-4 and 72B-1-72B-4, exemplary nucleotide and corresponding amino acid sequences, respectively, of factor VIII (SEQ ID NOS: 29 and 30, respectively).
FIG. 73 contains FIGS. 73A and 73B, is an exemplary nucleotide and corresponding amino acid sequence of urokinase (SEQ ID NOS: 33 and 34, respectively).
FIG. 74, comprising FIGS. 74A and 74B, is an exemplary nucleotide and corresponding amino acid sequence of human recombinant DNase (hrDNase) (SEQ ID NOS: 39 and 40, respectively).
FIG. 75, comprising FIGS. 75A and 75B, is an exemplary nucleotide and corresponding amino acid sequence of an insulin molecule (SEQ ID NOS: 43 and 44, respectively).
FIG. 76 contains FIGS. 76A and 76B, is an exemplary nucleotide and corresponding amino acid sequence of the S-protein from hepatitis B virus (HBsAg) (SEQ ID NOS: 45 and 46, respectively).
FIG. 77 contains FIGS. 77A and 77B, is an exemplary nucleotide and corresponding amino acid sequence of human growth hormone (hGH) (SEQ ID NOS: 47 and 48, respectively).
FIG. 78, comprising FIGS. 78A and 78D, is an exemplary nucleotide and corresponding amino acid sequence of antithrombin III. 78A and 78B are exemplary nucleotides and corresponding amino acid sequences of "Wild Type (WT)" antithrombin III (SEQ ID NOS: 63 and 64, respectively).
FIG. 79 contains FIGS. 79A-79D, are exemplary nucleotides and corresponding amino acid sequences of the alpha and beta subunits of human chorionic gonadotropin (hCG). 79A and 79B are exemplary nucleotides and corresponding amino acid sequences of the alpha subunit of human chorionic gonadotropin (SEQ ID NOS: 69 and 70, respectively). 79C and 79D are exemplary nucleotides and corresponding amino acid sequences of the beta subunit of human chorionic gonadotropin (SEQ ID NOS: 71 and 72, respectively).
FIG. 80 contains FIGS. 80A and 80B, is an exemplary nucleotide and corresponding amino acid sequence of α -iduronidase. (SEQ ID NOS: 65 and 66, respectively).
FIG. 81, comprising FIGS. 81A and 81B, is an exemplary nucleotide and corresponding amino acid sequence for α -galactosidase. (SEQ ID NOS: 67 and 68, respectively).
FIG. 82, which includes FIGS. 82A and 82B, includes EnbrelTMExemplary nucleotide and corresponding amino acid sequences of 75kDa tumor necrosis factor (TNF-R) of the moiety of (tumor necrosis factor receptor (TNF-R)/IgG fusion) (SEQ ID NOS: 31 and 32, respectively).
FIG. 83 includes FIGS. 83A and 83B, Herceptin, respectivelyTMExemplary nucleotide and corresponding amino acid sequences of the light and heavy chains of (monoclonal antibodies (MAb) against human epidermal growth factor receptor Her-2) (SEQ ID NOS: 35 and 36, respectively).
FIG. 84 contains FIGS. 84A and 84B, Synagis, respectivelyTMExemplary nucleotide and corresponding amino acid sequences of the heavy and light chains (MAb of respiratory syncytial virus F peptide) (SEQ ID NOs: 37 and 38, respectively).
FIG. 85, containing FIGS. 85A and 85B, is RemicadeTM(anti-TNF. alpha. MAb) and the corresponding amino acid sequences (SEQ ID NOS: 41 and 42, respectively).
FIG. 86, comprising FIGS. 86A and 86B, is an exemplary nucleotide and corresponding amino acid sequence of the Fc portion of human IgG (SEQ ID NOS: 49 and 50, respectively).
FIG. 87 is an exemplary amino acid sequence of the light chain of the mature variable region of the murine antibody against glycoprotein IIb/IIIa (SEQ ID NO: 52).
FIG. 88 is an exemplary amino acid sequence of the murine antibody mature variable region heavy chain against glycoprotein IIb/IIIa (SEQ ID NO: 54).
FIG. 89 is an exemplary amino acid sequence of a human IgG variable region light chain (SEQ ID NO: 51).
FIG. 90 is an exemplary amino acid sequence of a human IgG variable region heavy chain (SEQ ID NO: 53).
FIG. 91 is an exemplary amino acid sequence of a human IgG light chain (SEQ ID NO: 55).
FIG. 92 is an exemplary amino acid sequence of a human IgG heavy chain (SEQ ID NO: 56).
FIG. 93 comprises FIGS. 93A and 93B, exemplary nucleotides and corresponding amino acid sequences of the mature variable region of the murine antibody light chain of anti-CD 20 (SEQ ID NOS: 59 and 60, respectively).
FIG. 94 contains FIGS. 94A and 94B, exemplary nucleotides and corresponding amino acid sequences (SEQ ID NOS: 61 and 62, respectively) of the mature variable region of the murine antibody heavy chain of anti-CD 20.
FIG. 95 contains the nucleotide sequences of tandem chimeric antibody expression vector TCAE8 (SEQ ID NO: 57) as shown in FIGS. 95A-95E.
FIG. 96, which contains FIGS. 96A-96E, is the nucleotide sequence of tandem chimeric antibody expression vector TCAE8 (SEQ ID NO: 58) containing the murine antibody light and heavy chain variable domains of anti-CD 20.
Fig. 97, comprising fig. 97A-97C, is a graph depicting 2-AA HPLC analysis of glycans released by PNGaseF from Cri-IgG1 antibodies expressed from a myeloma. The structure of glycans is determined by retention time: the G0 glycoform (glycoform) eluted at 30 minutes, the G1 glycoform eluted at 33 minutes, the G2 glycoform eluted at about 37 minutes, and the S1-G1 glycoform eluted at 70 minutes. Figure 97A depicts the analysis of DEAE antibody samples. Figure 97B depicts the analysis of SPA antibody samples. Fig. 97C depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 14.
Fig. 98, comprising fig. 98A-98C, is a graph depicting MALDI analysis of glycans released by PNGaseF from Cri-IgG1 antibodies expressed from myeloma. Glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 98A depicts the analysis of a sample of DEAE antibody. Figure 98B depicts the analysis of SPA antibody samples. Figure 98C depicts analysis of Fc antibody samples.
FIG. 99, comprising FIGS. 99A-99D, is a graph depicting capillary electrophoretic analysis of glycans released from a Cri-IgG1 antibody that has been glycoremodeled (glycoremodeled) to contain the M3N2 glycoform. A graph of capillary electrophoresis analysis of glycan standards derivatized with APTS is shown in fig. 99A. Figure 99B depicts the analysis of DEAE antibody samples. Figure 99C depicts analysis of SPA antibody samples. Figure 99D depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 15.
FIG. 101, comprising FIGS. 101A-101C, is a graph depicting 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted to contain a G0 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column. Figure 101A depicts the analysis of a sample of DEAE antibody. Figure 101B depicts the analysis of SPA antibody samples. Figure 101C depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 16.
FIG. 102, comprising FIGS. 102A-102C, is a graph depicting a MALDI analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted to contain the G0 glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 102A depicts the analysis of a sample of DEAE antibody. Figure 102B depicts the analysis of SPA antibody samples. Figure 102C depicts analysis of Fc antibody samples.
FIG. 103, comprising FIGS. 103A-103D, is a graph depicting capillary electrophoretic analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted to contain a G2 glycoform. A graph of capillary electrophoresis analysis of glycan standards derivatized with APTS is shown in figure 103A. Figure 103B depicts analysis of DEAE antibody samples. Figure 103C depicts analysis of SPA antibody samples. Figure 103D depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 17.
FIG. 104, comprising FIGS. 104A-104C, is a graph depicting a 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted to contain a G2 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column. Figure 104A depicts the analysis of a sample of DEAE antibody. Figure 104B depicts analysis of SPA antibody samples. Figure 104C depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 17.
FIG. 105, comprising FIGS. 105A-105C, is a graph depicting a MALDI analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted to contain the G2 glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 105A depicts the analysis of a sample of DEAE antibody. Figure 105B depicts the analysis of SPA antibody samples. Figure 105C depicts analysis of Fc antibody samples.
FIG. 106, comprising FIGS. 106A-106D, is a graph depicting capillary electrophoretic analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by GnT-I treatment of the M3N2 glycoform. A graph of capillary electrophoresis analysis of glycan standards derivatized with APTS is shown in figure 106A. Figure 106B depicts analysis of DEAE antibody samples. Figure 106C depicts analysis of SPA antibody samples. Figure 106D depicts analysis of Fc antibody samples.
FIG. 107, comprising FIGS. 107A-107C, is a graph depicting 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been reconstituted by GnT-I treatment of the M3N2 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column. Figure 107A depicts the analysis of a sample of DEAE antibody. Figure 107B depicts the analysis of SPA antibody samples. Figure 107C depicts analysis of Fc antibody samples.
FIG. 108, comprising FIGS. 108A-108C, is a graph depicting MALDI analysis of glycans released from a Cri-IgG1 antibody that has been glycoremodeled by GnT-I treatment of the M3N2 glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 108A depicts the analysis of a sample of DEAE antibody. Figure 108B depicts the analysis of SPA antibody samples. Figure 108C depicts analysis of Fc antibody samples.
FIG. 109, comprising FIGS. 109A-109D, is a graph depicting capillary electrophoretic analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by GnT-I, II and III treatment of the M3N2 glycoform. A graph of capillary electrophoresis analysis of glycan standards derivatized with APTS is shown in fig. 109A. Figure 109B depicts analysis of DEAE antibody samples. Figure 109C depicts analysis of SPA antibody samples. Figure 109D depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 18.
FIG. 110, comprising panels 110A-110C, is a graph depicting 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by GnT-I, II and III treatment of the M3N2 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column. Figure 110A depicts the analysis of a sample of DEAE antibody. Figure 110B depicts analysis of SPA antibody samples. Figure 110C depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 18.
FIG. 111 comprises panels 111A-111C, which are graphs depicting MALDI analysis of glycans released from Cri-IgG1 antibody that has been glycoremodeled by galactosyltransferase treatment of the NGA2F glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 111A depicts the analysis of DEAE antibody samples. Figure 111B depicts the analysis of SPA antibody samples. Figure 111C depicts analysis of Fc antibody samples.
FIG. 112, comprising panels 112A-112D, is a graph depicting 2-AA HPLC analysis of glycans released from Cri-IgG1 antibodies containing the NGA2F isotype before GalT1 treatment (panels 112A and 112C) and after GalT1 treatment (panels 112B and 112D). FIGS. 112A and 112B depict analysis of DEAE antibody samples. Fig. 112C and 112D depict analysis of Fc antibody samples. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column.
FIG. 113, comprising FIGS. 113A-113C, is a graph depicting 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by ST3Gal3 treatment of the G2 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH2P-504D amino column. Figure 113A depicts analysis of a sample of DEAE antibody. Figure 113B depicts analysis of SPA antibody samples. Figure 113C depicts analysis of Fc antibody samples. The percentage area under the peaks in these plots is summarized in table 19.
FIG. 114, comprising FIGS. 114A-114C, is a graph depicting MALDI analysis of glycans released from a Cri-IgG1 antibody that had been glycoreconstituted by ST3Gal3 treatment of the G2 glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 114A depicts the analysis of a sample of DEAE antibody. Figure 114B depicts the analysis of SPA antibody samples. Figure 114C depicts analysis of Fc antibody samples.
FIG. 115, comprising FIGS. 115A-115D, is a graph depicting capillary electrophoretic analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by ST6Gal1 treatment of the G2 glycoform. A graph of capillary electrophoresis analysis of glycan standards derivatized with APTS is shown in figure 115A. Figure 115B depicts the analysis of a sample of DEAE antibody. Figure 115C depicts the analysis of SPA antibody samples. Figure 115D depicts analysis of Fc antibody samples.
FIG. 116, comprising FIGS. 116A-116C, is a graph depicting 2-AA HPLC analysis of glycans released from a Cri-IgG1 antibody that has been glycoreconstituted by ST6Gal1 treatment of the G2 glycoform. The released glycans were labeled with 2AA and separated by HPLC on NH 2P-504D amino column. Figure 116A depicts the analysis of a sample of DEAE antibody. Figure 116B depicts the analysis of SPA antibody samples. Figure 116C depicts analysis of Fc antibody samples.
FIG. 117 comprises FIGS. 117A-117C, which are graphs depicting MALDI analysis of glycans released from Cri-IgG1 antibody that has been glycoreconstituted by ST6Gal1 treatment of the G2 glycoform. The released glycans were derivatized with 2-AA and then analyzed by MALDI. Figure 117A depicts the analysis of a sample of DEAE antibody. Figure 117B depicts the analysis of SPA antibody samples. Figure 117C depicts analysis of Fc antibody samples.
FIG. 118 contains FIGS. 118A-118E, which depict SDS-PAGE analysis of Cri-IgG1 antibodies sugar reconstituted with different sugar forms under non-reducing conditions. Bovine Serum Albumin (BSA) was run under non-reducing conditions as a quantitative standard. Protein molecular weight standards are shown and their sizes are shown in kDa. FIG. 118A depicts SDS-PAGE analysis of DEAE, SPA and Fc Cri-IgG1 antibodies with glycoremodeling to contain G0 and G2 glycoforms. FIG. 118B depicts SDS-PAGE analysis of DEAE, SPA and Fc Cri-IgG1 antibodies that were glycoreconstituted to contain NGA2F (bisected) and GnT-I-M3N2(GnT1) glycoforms. FIG. 118C depicts SDS-PAGE analysis of DEAE, SPA and Fc Cri-IgG1 antibodies with saccharide reconstitution to contain the S2G2(ST6Gal1) saccharide form. FIG. 118D depicts SDS-PAGE analysis of DEAE, SPA and Fc Cri-IgG1 antibodies in the form of M3N2 saccharide, and BSA with saccharide reconstitution. FIG. 118E depicts SDS-PAGE analysis of DEAE, SPA and Fc Cri-IgG1 antibodies, and BSA with glycoremodeling to contain the glycoform of Gal-NGA2F (Gal-bisected).
FIG. 119 is an acrylamide gel image depicting the results of a FACE analysis of TP10 before and after sialylation. BiNA0The species do not have sialic acid residues. BiNA1The species has 1 sialic acid residue. BiNA2The species has 2 sialic acid residues. Bi is dual-antenna; NA-neuraminic acid.
FIG. 120 is a graph of plasma concentration (. mu.g/ml) -time profile of TP10 before and after sialylation in rats.
FIG. 121 is a graph depicting the area under the plasma concentration-time curve (AUC) in μ g/hr/ml for TP10 before and after sialylation.
FIG. 122 is an acrylamide gel image depicting the results of FACE glycan analysis of TP10 and TP-20 produced by CHO cells before and after fucosylation. BiNA2F2Species have 2 Neuraminic Acid (NA) residues and 2 fucose residues (F).
FIG. 123 is a graph depicting TP20(sCR1 sLe) glycosylated in vitro (diamonds) and in vivo (squares) in Lec11 CHO cellsX) Graph of in vitro binding of (a).
FIG. 124 is a graph depicting 2-AA HPLC analysis of GlcNAc-derivatized glycoforms from EPO.
FIG. 125, comprising FIGS. 125A and 125B, is a graph depicting a 2-AA HPLC analysis of 2 batches of EPO to which N-acetylglucosamine has been added. FIG. 125A depicts the analysis of batch A and FIG. 125B depicts the analysis of batch B.
FIG. 126 is a graph depicting 2-AA HPLC analysis of reaction products of the introduction of a third glycan branch to EPO with GnT-V.
FIG. 127 is a MALDI-TOF spectrum plot depicting glycans of EPO preparations treated with GnT-I, GnT-II, GnT-III, GnT-V, and GalT1 and an appropriate donor.
FIG. 128 is a graph depicting glycan MALDI spectra of native EPO.
FIG. 129 is an SDS-PAGE gel image of PEGylation reaction products with CMP-SA-PEG (1kDa) and CMP-SA-PEG (10 kDa).
FIG. 130 is a graph depicting the results of PEGylated EPO in vitro bioassay. Diamonds represent data from sialylated EPO without PEG molecules. The squares represent data obtained with EPO with PEG (1 kDa). Triangles represent data obtained with EPO with PEG (10 kDa).
FIG. 131 is a schematic of CHO-expressed EPO. The EPO peptide is 165 amino acids in length and the unglycosylated molecular weight is 18 kDa. The glycosylated form of EPO produced in CHO cells has a molecular weight of about 33kDa to 39 kDa. The shapes of the sugars in the glycan chain are given in the box at the bottom of the figure.
FIG. 132 is a schematic of EPO expressed by insect cells. The shapes of the sugars in the glycan chain are given in the box at the bottom of fig. 131.
FIG. 133 is a bar graph depicting the molecular weights of EPO peptides expressed in insect cells, reconstituted to form complete mono-, di-, and triantennary glycans, and optionally glycoPEGylated with 1kDa, 10kDa, or 20kDa PEGs. Epoetin TMIs EPO expressed in mammalian cells without further glycan modification or pegylation. NESP (Aranesp)TMAmgen, thunder and Oaks, CA) is a form of EPO with 5N-linked glycan sites that is also expressed in mammalian cells without further glycan modification or pegylation.
FIG. 134 contains graphs 134A and 134B, which depict a protocol for the reconstitution and glycoPEGylation of insect cell-expressed EPO. Figure 134A depicts the reconstitution and saccharide pegylation steps for insect-expressed glycans reconstituted as single-touch saccharide pegylation glycans. Figure 134B depicts reconstituted EPO peptides with complete saccharide pegylated monokeratoglycan at each N-linked glycan site of the polypeptide. The shapes of the saccharides in the glycan chain are given in the boxes at the bottom of FIG. 131, except that the triangles represent sialic acids.
FIG. 135 is a graph depicting the in vitro biological activity of EPO-SA and EPO-SA-PEG constructs. In vitro assay measures the proliferation of TF-1 erythroleukemia cells maintained in RBMI + FBS 10% + GM-CSF (12ng/ml) for 48 hours after addition of 10.0, 5.0, 2.0, 1.0, 0.5 and 0 μ g/ml EPO construct. Tri-SA refers to an EPO construct with SA in which the glycan is three-antennary. Tri-SA1K PEG refers to the construction of EPO whose glycans are triantennary and have Gal and then PEGylated with SA-PEG1kDa saccharide And (3) a body. Di-SA 10K PEG refers to an EPO construct where the glycan is biantennary and has Gal and is then PEGylated with the SA-PEG 10kDa sugar. Di-SA 1K PEG refers to an EPO construct where the glycan is biantennary and has Gal and is then PEGylated with the SA-PEG 1kDa sugar. Di-SA refers to an EPO construct where the glycan is biantennary and is constructed to SA. EpogenTMIs EPO expressed in CHO cells with no further glycan modifications.
Figure 136 is a graph depicting the pharmacokinetics of EPO constructs in rats. Using the rat with [ I125]Labeled saccharide pegylated and non-saccharide pegylated EPO in bolus injections. The graph shows the concentration of radiolabeled EPO in the bloodstream of rats from 0 to about 72 minutes after injection. "Biant-10K" refers to EPO with a biantennary glycan structure with a terminal 10kDa PEG moiety. "Mono-20K" refers to EPO having a one-touch angle glycan structure with a terminal 20kDa PEG moiety. NESP refers to commercially available Aranesp. "Biant-1K" refers to EPO with a biantennary glycan structure with a terminal 1kDa PEG moiety. "Biant-SA" refers to EPO having a biantennary glycan structure with a terminal 1kDa moiety. The concentration of EPO construct in the bloodstream at 72 hours was as follows: Biant-10K, 5.1 cpm/ml; Mono-20K, 3.2 cpm/ml; NESP, 1 cpm/ml; and Biant-1K, 0.2 cpm/ml; Biant-SA, 0.1 cpm/ml. The relative area under the curve in the EPO construct is as follows: Biant-10K, 2.9; Mono-20K, 2.1; NESP, 1; Biant-1K, 0.5; and Biant-SA, 0.2.
FIG. 137 is a bar graph depicting the ability of EPO constructs to stimulate reticulocytosis (reticulocytosis) in vivo. Each treatment group consisted of 8 mice. Mice were given a single subcutaneous injection of 10 μ g protein/kg body weight. The percent reticulocyte proliferation was measured at 96 hours. The triantenna-SA 2, 3(6) construct has SA molecules conjugated with 2, 3 or 2, 6 bonds (see preparation example 18 herein), wherein the glycan on EPO is a triantenna glycan with SA-PEG 10K attached thereto. Similarly, biantennary-10K PEG is EPO with biantennary N-glycans of SA-PEG with 10K PEG attached thereto.
FIG. 138 is a graph depicting the in vivo hematocrit increasing capacity of mouse blood for EPO constructs. CD-1 female mice were injected intraperitoneally (i.p.) with 2.5 μ g protein/kg body weight. The hematocrit of the mice was measured on day 15 after EPO injection. Bi-1K refers to an EPO construct where the glycan is biantennary and is constructed to (build out to) Gal and then PEGylated with the SA-PEG 1kDa sugar. Mono-20K refers to an EPO construct where the glycan is monoantennary and is constructed to Gal and then PEGylated with the SA-PEG 20kDa sugar.
FIG. 139 contains FIGS. 139A and 139B, depicting analysis of glycans enzymatically released from EPO (Protein Sciences, Lot #060302) expressed in insect cells. Figure 139A depicts HPLC analysis of released glycans. FIG. 139B depicts MALDI analysis of released glycans. Diamonds represent fucose, squares represent GlcNAc, and circles represent mannose.
FIG. 140 depicts MALDI analysis of glycans released from EPO following a GnT-I/GalT-1 reaction. The structure of the glycans was determined by comparison of the peak spectra with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose, stars represent galactose.
FIG. 141 depicts SDS-PAGE analysis of EPO after GnT-I/GalT-1 reaction, Superdex 75 purification, ST3Gal3 reaction with SA-PEG (10kDa) and SA-PEG (20 kDa).
FIG. 142 depicts the results of a TF-1 cell in vitro bioassay of PEGylated monoantennary EPO.
FIG. 143 contains panels 143A and 143B, which depict an analysis of glycans released from EPO following a GnT-I/GnT-II response. FIG. 143A depicts HPLC analysis of released glycans, where peak 3 represents biantennary GlcNAc glycans. FIG. 143B depicts MALDI analysis of released glycans. The structure of the glycans was determined by comparison of the peak spectra with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose.
FIG. 144 contains graphs 144A and 144B, depicting HPLC analysis of glycans released from EPO following a GalT-1 reaction. FIG. 144A depicts glycans released after small-scale GalT-1 reaction. FIG. 144B depicts glycans released after a large-scale GalT-1 reaction. In both figures, peak 1 is a biantennary glycan with a terminal galactose moiety, while peak 2 is a biantennary glycan without a terminal galactose moiety.
FIG. 145 depicts Superdex 75 chromatographic separation of EPO species following GalT-1 response. Peak 2 contains EPO with biantennary glycans with terminal galactose moieties.
FIG. 146 depicts SDS-PAGE analysis of each product of the sugar reconstitution process to make biantennary glycans with terminal galactose moieties.
FIG. 147 depicts SDS-PAGE analysis of EPO after sialylation of ST3Gal3 or PEGylation with SA-PEG (1kDa) and SA-PEG (10 kDa).
FIG. 148 depicts HPLC analysis of glycans released from EPO following a GnT-I/GnT-II reaction. The structure of the glycans was determined by comparison of peak retention with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose.
FIG. 149 depicts HPLC analysis of glycans released from EPO following a GnT-V response. The structure of the glycans was determined by comparison of peak retention with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose.
FIG. 150 depicts HPLC analysis of glycans released from EPO following a GalT-1 reaction. The structure of the glycans was determined by comparison of peak retention with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose, open circles represent galactose, and triangles represent sialic acid.
Fig. 151 depicts HPLC analysis of glycans released from EPO following ST3Gal3 reaction. The structure of the glycans was determined by comparison of peak retention with standard glycans. The structure of the glycans is depicted next to the peak. Diamonds represent fucose, and squares represent GlcNAc, circles represent mannose, open circles represent galactose, and triangles represent sialic acid.
Fig. 152 depicts HPLC analysis of glycans released from EPO following ST6Gal1 reaction. The structure of the glycans was determined by comparison of peak retention with standard glycans. The structure of the glycans is depicted next to the peak.
FIG. 153 depicts the results of in vitro bioassay of TF-1 cells with EPO having biantennary and triantennary glycans. "Di-SA" refers to sialic acid terminated EPO with biantennary glycans. "Di-SA 10K PEG" refers to EPO with biantennary glycans terminated with sialic acid derivatized with PEG (10 kDa). "Di-SA 1K PEG" refers to EPO with biantennary glycans terminated with sialic acid derivatized with PEG (1 kDa). "Tri-SA ST6+ ST 3" refers to EPO with triantennary glycans terminating in 2, 6-SA capped with 2, 3-SA. "Tri-SA ST 3" refers to EPO with triantennary glycans terminating in 2, 3-SA.
FIG. 154 is an IEF gel image depicting the pI of the product of the desialylation procedure. Lanes 1 and 5 are IEF standards. Lane 2 is a coagulation factor IX protein. Lane 3 is rfactorIX protein. Lane 4 is an asialo reaction of rFactor IX protein at 20 hours.
FIG. 155 is an SDS-PAGE gel image depicting the molecular weight of factor IX conjugated with SA-PEG (1kDa) or SA-PEG (10kDa) after reaction with CMP-SA-PEG. Lanes 1 and 6 are the SeeBlue +2 molecular weight standards. Lane 2 is rF-IX. Lane 3 is desialylated rF-IX. Lane 4 is rFactor IX conjugated with SA-PEG (1 kDa). Lane 5 is rFactor IX conjugated with SA-PEG (10 kDa).
FIG. 156 is an SDS-PAGE gel image depicting direct sialylation of factor IX and the sialic acid capping reaction product of factor IX-SA-PEG. Lane 1 is a protein standard, lane 2 is blank; lane 3 is rFactor-IX; lane 4 is SA-capped rFactor-IX-SA-PEG (10 kDa); lane 5 is rFactor-IX-SA-PEG (10 kDa); lane 6 is ST3Gal 1; lane 7 is ST3Gal 3; lanes 8, 9, 10 are rFactor-IX-SA-PEG (10kDa) without sialidase pretreatment.
FIG. 157 is an image of an isoelectric focusing gel (pH3-7) of asialo (asialo) -factor VIIa. Lane 1 is rFactor VIIa; lanes 2-5 are asialo-coagulation factor VIIa.
FIG. 158 is a graph of MALDI spectra of factor VIIa.
FIG. 159 is a graph of MALDI spectra of factor VIIa-PEG (1 kDa).
FIG. 160 is a graph depicting MALDI spectra of factor VIIa-PEG (10 kDa).
FIG. 161 is an SDS-PAGE gel image of PEGylated factor VIIa. Lane 1 is asialo-coagulation factor VIIa. Lane 2 is the product of asialo-coagulation factor VIIa and CMP-SA-PEG (1kDa) reacted with ST3Gal3 after 48 hours. Lane 3 is the product of the reaction of asialo-coagulation factor VIIa and CMP-SA-PEG (1kDa) with ST3Gal3 after 48 hours. Lane 4 is the product of the reaction of asialo-coagulation factor VIIa and CMP-SA-PEG (10kDa) with ST3Gal3 after 96 hours.
FIG. 162 is an image depicting an isoelectric focusing (IEF) gel of human pituitary FSH desialylation reaction products. Lanes 1 and 4 are the standards for isoelectric focusing (IEF). Lane 2 is native FSH. Lane 3 is desialylated FSH.
FIG. 163 is an SDS-PAGE gel image of the products of the reaction to prepare PEG-sialylated rFSH. Lanes 1 and 8 are SeeBlue +2 molecular weight standards. Lane 2 is 15. mu.g of native FSH. Lane 3 is 15. mu.g asialo-FSH (AS-FSH). Lane 4 is 15. mu.g of the product of the reaction of AS-FSH with CMP-SA. Lane 5 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (1 kDa). Lane 6 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (5 kDa). Lane 7 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (10 kDa).
FIG. 164 is an isoelectric focusing gel image of the product of the reaction to prepare PEG-sialylated FSH. Lanes 1 and 8 are IEF standards. Lane 2 is 15. mu.g of native FSH. Lane 3 is 15. mu.g asialo-FSH (AS-FSH). Lane 4 is the product of the reaction of 15. mu.g AS-FSH with CMP-SA. Lane 5 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (1 kDa). Lane 6 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (5 kDa). Lane 7 is the product of a reaction of 15. mu.g AS-FSH with CMP-SA-PEG (10 kDa).
FIG. 165 is an SDS-PAGE gel image of native non-recombinant FSH produced in human pituitary cells. Lanes 1, 2 and 5 are SeeBlueTM+2 molecular weight standards. Lanes 3 and 4 are 5 μ g and 25 μ g of native FSH, respectively.
FIG. 166 is an image of an isoelectric focusing gel (pH3-7) depicting the products of the desialylation reaction of rFSH. Lanes 1 and 4 are IEF standards. Lane 2 is native rFSH. Lane 3 is asialo-rFSH.
FIG. 167 is an SDS-PAGE gel image depicting the results of PEG-sialylation of asialo-rFSH. Lane 1 is native rFSH. Lane 2 is asialo-FSH. Lane 3 is the product of the reaction of asialo-FSH with CMP-SA. Lanes 4-7 are the products of the reaction of asialo-FSH with 0.5mM CMP-SA-PEG (10kDa) at 2 hours, 5 hours, 24 hours and 48 hours, respectively. Lane 8 is the product of the reaction of asialo-FSH with 1.0mM CMP-SA-PEG (10kDa) at 48 hours. Lane 9 is the product of the reaction of asialo-FSH with 1.0mM CMP-SA-PEG (1kDa) at 48 hours.
FIG. 168 is an isoelectric focusing gel image showing the product of PEG-sialylation of asialo-rFSH using CMP-SA-PEG (1 kDa). Lane 1 is native rFSH. Lane 2 is asialo-rFSH. Lane 3 is the product of the 24-hour reaction of asialo-rFSH with CMP-SA. Lanes 4-7 are the products of the reaction of asialo-rFSH with 0.5mM CMP-SA-PEG (1kDa) at 2 hours, 5 hours, 24 hours and 48 hours, respectively. Lane 8 is blank. Lanes 9 and 10 are the products of the reaction of asialo-rFSH with 0.5mM and 1.0mM CMP-SA-PEG (10kDa), respectively, at 48 hours.
FIG. 169 is a graph of the pharmacokinetics of rFSH and rFSH-SA-PEG (1KDa and 10 KDa). The graph illustrates the relationship between time of rFSH compound in rat bloodstream and mean concentration of rFSH compound in blood compared to non-pegylated rFSH for saccharide-pegylated rFSH.
Fig. 170 is a graph of FSH bioassay results using support cells. The graph illustrates the relationship between the concentration of FSH in the support cell incubation medium and the amount of 17- β estradiol released from the support cells.
FIG. 171 is a graph depicting the results of a Steelman-Pohley bioassay for glycoPEGylated and non-glycoPEGylated FSH. Rats were injected subcutaneously with human chorionic gonadotropin and various amounts of FSH over 3 days and the mean ovarian weight of the treatment group was determined on day 4. rFSH-SA-PEG refers to recombinant FSH that has been glycopegylated with PEG (1 kDa). rFSH refers to non-saccharide pegylated FSH. Each treatment group contained 10 rats.
FIG. 172 contains panels 172A and 172B, which depict chromatograms of INF- β elution from Superdex-75 columns. FIG. 172A depicts the entire chromatogram. FIG. 172B depicts in more detail the box-shaped region containing peaks 4 and 5 in FIG. 172A.
Figure 173 comprises figures 173A and 173B, depicting MALDI analysis of glycans enzymatically released from INF- β. FIG. 173A depicts MALDI analysis of glycans released from native INF- β. FIG. 173B depicts MALDI analysis of glycans released from desialylated INF- β. The structure of the glycans was determined by comparison of the peak spectra with standard glycans. The structure of the glycans is depicted next to the peak. Squares represent GlcNAc, triangles represent fucose, circles represent mannose, diamonds represent galactose and stars represent sialic acid.
FIG. 174 depicts a lectin blot analysis performed on desialylated INF- β. The blot on the right was probed with the Digoxigenin (DIG) (RocheApplied Science, Indianapolis, IL) -labeled kui-hua (Maackia amurensis) lectin (MAA) to detect α 2, 3-sialylation. The left blot was probed with Erthrincristagalli lectin (ECL) labeled with biotin (Vector Laboratories, Burlingame, Calif.) to detect exposed galactose residues. FIG. 175 depicts SDS-PAGE analysis of PEG (10kDa) PEGylation reaction products of INF- β. "-PEG" refers to INF- β prior to the PEGylation reaction. "+ PEG" refers to INF- β after PEGylation reaction.
FIG. 176 depicts SDS-PAGE analysis of PEG (20kDa) PEGylation reaction products of INF- β. "unmodified" refers to INF- β prior to PEGylation. "PEGylated" refers to INF- β after the PEGylation reaction.
FIG. 177 depicts the chromatogram of PEG (10kDa) PEGylated INF-beta elution from a Superdex-200 column.
FIG. 178 depicts the results of bioassay of PEG (10kDa) PEGylated INF- β peak fractions shown in the INF-PEG6 chromatogram.
FIG. 179 depicts the chromatogram of PEG (20kDa) PEGylated INF-beta elution from a Superdex-200 column.
FIG. 181 is a schematic depicting the conversion of high mannose N-glycans to hybrid N-glycans. Enzyme 1 is an α 1, 2-mannosidase from trichoderma reesei (Trichodoma reesei) or Aspergillus saitoi. Enzyme 2 is GnT-I (. beta. -1, 2-N-acetylglucosaminyltransferase I). Enzyme 3 is GalT-I (. beta. -1, 4-galactosyltransferase 1). The enzyme 4 is an alpha 2, 3-sialyltransferase or an alpha 2, 6-sialyltransferase.
FIG. 182 contains panels 182A and 182B, which are two graphs depicting MALDI-TOF spectra of RNaseB treated with recombinant Trichoderma reesei α 1, 2-mannosidase (FIG. 182A) and HPLC profiles of oligosaccharides cleaved from modified RNaseB by N-glycanase (FIG. 182B).
Fig. 183 is a graph depicting MALDI-TOF spectra of rnase b treated with commercially available α 1, 2-mannosidase purified from a. saitoi (Glyko & CalBioChem).
FIG. 184 is a graph depicting MALDI-TOF spectra of modified RNaseB obtained by treating the product shown in FIG. 182 with recombinant GnT-I (GlcNAc transferase I).
FIG. 185 is a graph depicting MALDI-TOF spectra of modified RNaseB obtained by treating the product shown in FIG. 184 with recombinant GalT-1 (galactosyltransferase 1).
FIG. 186 is a graph depicting MALDI-TOF spectra of modified RNaseB obtained by treating the product shown in FIG. 185 with recombinant ST3Gal III (. alpha.2, 3-sialyltransferase III) using CMP-SA as a transferase donor.
FIG. 187 is a graph depicting MALDI-TOF spectra of modified RNaseB obtained by treating the product shown in FIG. 185 with recombinant ST3Gal III (. alpha.2, 3-sialyltransferase III) using CMP-SA-PEG (10kDa) as a transferase donor.
Figure 188 is a series of protocols for the conversion of high mannose N-glycans to complex N-glycans. Enzyme 1 is an α 1, 2-mannosidase from trichoderma reesei or Aspergillus saitoi. Enzyme 2 is GnT-I. Enzyme 3 is GalT 1. The enzyme 4 is an alpha 2, 3-sialyltransferase or an alpha 2, 6-sialyltransferase. Enzyme 5 is alpha mannosidase II. Enzyme 6 is an alpha mannosidase. The enzyme 7 is GnT-II. The enzyme 8 is an alpha 1, 6-mannosidase. Enzyme 9 is an alpha 1, 3-mannosidase.
FIG. 189 is a diagram showing ligation catalyzed by N-acetylglucosamine transferases I-VI (GnT-I-VI). R ═ GlcNAc β 1, 4 GlcNAc-Asn-X.
FIG. 190 is an image of an SDS-PAGE gel: standard (lane 1); native transferrin (lane 2); asialo-transferrin (lane 3); asialoglycotransferrin and CMP-SA (lane 4); lanes 5 and 6, asialo transferrin and CMP-SA-PEG (1kDa) at 0.5mM and 5mM, respectively; lanes 7 and 8, asialo transferrin and CMP-SA-PEG (5kDa) at 0.5mM and 5mM, respectively; lanes 9 and 10, asialo transferrin and CMP-SA-PEG (10kDa) at 0.5mM and 5mM, respectively.
Fig. 191 is an image of an IEF gel: native transferrin (lane 1); asialo transferrin (lane 2); asialo-transferrin and CMP-SA, 24 hours (lane 3); asialo transferrin and CMP-SA, 96 hours (lane 4); lanes 5 and 6, asialoglycotransferrin and CMP-SA-PEG (1kDa) at 24 and 96 hours, respectively; lanes 7 and 8, asialoglycotransferrin and CMP-SA-PEG (5kDa) at 24 and 96 hours, respectively; lanes 9 and 10, asialoglycotransferrin and CMP-SA-PEG (10kDa) at 24 and 96 hours, respectively.
Detailed Description
The present invention includes cell-free in vitro methods and compositions for adding and/or removing saccharides from peptide molecules in such a manner as to provide glycopeptide molecules having a particular customized or desired glycosylation pattern, wherein the glycopeptides are produced on an industrial scale. In a preferred embodiment of the invention, the glycopeptide thus prepared has attached to it a modified sugar which is added to the peptide by an enzymatic reaction. A key feature of the present invention is the acquisition of peptides produced by any cell type and the generation of a decorin structure on the peptide, followed by in vitro reconstitution of the glycan structure to generate a glycopeptide having a glycosylation pattern suitable for therapeutic use in mammals. More specifically, it is possible according to the present invention to prepare glycopeptide molecules having a modified sugar molecule or other compound conjugated thereto, such that the conjugated molecule confers a beneficial characteristic to the peptide. According to the present invention, the conjugate molecule is enzymatically added to the peptide, since the enzyme-based addition of the conjugate molecule to the peptide has the advantages of regioselectivity and stereoselectivity. The glycoconjugates can be added to the glycans on the peptides before or after glycosylation is complete. In other words, the order of glycoconjugation and glycosylation may be varied as described elsewhere herein. It is therefore possible to reconstitute a peptide using the methods and compositions provided herein to impart to the peptide a desired glycan, preferably having a modified sugar attached thereto. It is also possible to generate peptide molecules with desired and or modified glycan structures on an industrial scale using the methods and compositions of the invention, thus providing the art for the first time with practical solutions to efficiently generate improved therapeutic peptides.
Definition of
Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. Generally, the nomenclature used herein and the laboratory procedures in cell culture, molecular genetics, organic chemistry, and nucleic acid chemistry and hybridization are those well known and commonly employed in the art. Standard techniques are applied for nucleic acid and peptide synthesis. The techniques and procedures are generally performed according to conventional methods in the art and various general references (e.g., Sambrook et al, 1989, molecular cloning: A Laboratory Manual, 2 d. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.), which are provided throughout this document. The nomenclature used herein and the laboratory procedures for analytical chemistry and organic synthesis described below are those well known and commonly employed in the art. Standard techniques or modified methods thereof are used for chemical synthesis and chemical analysis.
The articles "a" and "an" are used herein to refer to one or to more than one (i.e., to at least one) of the grammatical object of the article. By way of example, "an element" refers to one element or more than one element.
As used herein, the term "antibody" refers to an immunoglobulin molecule that is capable of specifically binding to a particular epitope on an antigen. The antibody may be an intact immunoglobulin derived from a natural source or a recombinant source, and may also be an immunoreactive portion of an intact immunoglobulin. Antibodies are typically tetramers of immunoglobulin molecules. The antibodies of the invention may exist in a variety of forms, including, for example, polyclonal, monoclonal, Fv, Fab and F (ab)2And single chain Antibodies and humanized Antibodies (Harlow et al, 1999, Using Antibodies: antibody Manual, Cold Spring Harbor Laboratory Press NY; Harlow et al, 1989, Antibodies: A Laboratory Manual, Cold Spring Harbor, New York; houston et al, 1988, proc.natl.acad.sci.usa 85: 5879-5883; bird et al, 1988, Science 242: 423-426).
As used herein, the term "synthetic antibody" refers to an antibody produced using recombinant DNA techniques, such as an antibody expressed by a bacteriophage as described herein. The term may also be interpreted to refer to antibodies generated by synthesizing a DNA molecule encoding an antibody, which DNA molecule expresses an antibody protein, or an amino acid sequence encoding the antibody, which DNA or amino acid sequence has been obtained using techniques for synthesizing DNA or amino acid sequences available and well known in the art.
As used herein, the term "functional" biological molecule is a form of biological molecule in which the molecule exhibits the characteristic upon which it is characterized. For example, a functional enzyme is an enzyme that exhibits a characteristic catalytic activity on which the enzyme is characterized.
As used herein, structureIs the point in the peptide chain at which an amino acid or amino acid side chain is attached to the glycan structure.
"N-linked" oligosaccharides are those which are linked to the peptide backbone via asparagine in an asparagine-N-acetylglucosamine linkage. N-linked oligosaccharides are also known as "N-glycans". All N-linked oligosaccharides have a common pentasaccharide core Man3GlcNAc2. They differ in the presence or absence and number of peripheral sugar branches (also known as antennaries), such as N-acetylglucosamine, galactose, N-acetylgalactosamine, fucose and sialic acid. Alternatively, the structure may also contain a core fucose molecule and/or xylose molecule.
By "essential trimannose core structure" is meant a glycan moiety comprising only the trimannose core structure without additional sugar attached thereto. When the term "essential" is not included in the description of "trimannose core structure", then the glycan comprises the trimannose core structure with an additional sugar attached thereto. Alternatively, the structure may also contain a core fucose molecule and/or xylose molecule.
The term "basic trimannose core glycopeptide" is used herein to refer to glycopeptides having a glycan structure which predominantly comprises a basic trimannose core structure. Alternatively, the structure may also contain a core fucose molecule and/or xylose molecule.
"O-linked" oligosaccharides are those that are attached to the peptide backbone via threonine, serine, hydroxyproline, tyrosine, or other hydroxyl-containing amino acids.
All oligosaccharides described herein are identified by the name or abbreviation for non-reducing sugars (i.e., Gal), followed by the configuration of the glycosidic bond (α or β), the ring bond (1 or 2), the ring position of the reducing sugar referred to in the bond (2, 3, 4, 6 or 8) and then the name or abbreviation for reducing sugars (i.e., GlcNAc). Each sugar is preferably a pyranose. For a review of the standard Glycobiology nomenclature, see essences of Glycobiology Varki et al eds, 1999, CSHL Press.
The term "sialic acid" refers to any member of the nine-carbon carboxylated sugar family. The most common member of the sialic acid family is N-acetylneuraminic acid (2-keto-5-acetylamino-3, 5-dideoxy-D-glycero-D-lactonnolopyranos-1-oic acid (often abbreviated Neu5Ac, NeuAc or NANA)). The second member of the family is N-glycolyl-neuraminic acid (Neu5Gc or NeuGc), in which the N-acetyl group of NeuAc is hydroxylated. The third sialic acid family member is 2-keto-3-deoxy-nonulosonic acid (KDN) (Nadano et al, (1986) J.biol.chem.261: 11550-11557; Kanamori et al, J.biol.chem.265: 21811-21819 (1990)). Also included are 9-substituted sialic acids such as 9-O-C 1-C6acyl-Neu 5Ac, such as 9-O-lactyl-Neu 5Ac or 9-O-acetyl-Neu 5Ac, 9-deoxy-9-fluoro-Neu 5Ac, and 9-azido-9-deoxy-Neu 5 Ac. For a review of the sialic acid family, see Varki, Glycobiology 2: 25-40 (1992); sialic Acids: chemistry, Metabolism and function, R.Schauer, Ed. (Springer-Verlag, Ne.)wYork (1992)). The synthesis and use of sialic acid compounds in sialylation is disclosed in international application WO 92/16640 published on month 10 and 1 1992.
As used herein, a peptide having "desired glycosylation" is a peptide that comprises one or more oligosaccharide molecules necessary for effective biological activity of the peptide.
A "disease" is a health condition of an animal in which the animal is unable to maintain homeostasis, and in which the animal's health will continue to deteriorate if the disease is not ameliorated.
As used herein in administering a peptide drug to a patient, "area under the curve" or "AUC" is defined as the total area under the curve that describes the concentration of the drug in the systemic circulation of the patient as a function of time from 0 to infinity.
As used herein in administering a peptide drug to a patient, the term "half-life" or "t 1/2" is defined as the time required for the plasma concentration of the drug in the patient to decrease by half. Depending on various clearance mechanisms, redistribution and other mechanisms well known in the art, there may be more than one half-life associated with a peptide drug. Typically, an alpha half-life and a beta half-life are defined such that alpha phase is associated with redistribution and beta phase is associated with clearance. However, there are at least 2 clearance half-lives for the vast majority of protein drugs that are confined to the blood stream. For some glycosylated peptides, rapid β -phase clearance can be mediated by receptors on endothelial cells or macrophages that recognize terminal galactose, N-acetylgalactosamine, N-acetylglucosamine, mannose, or fucose. Slow β -phase clearance can be mediated by glomerular filtration and/or specific or non-specific uptake and metabolism in tissues of molecules with an effective radius <2nm (about 68 kD). Saccharide pegylation can cap terminal saccharides (e.g., galactose or N-acetylgalactosamine) thereby blocking rapid alpha-phase clearance through receptors that recognize these saccharides. It may also impart a larger effective radius, thereby reducing the dispensed volume and tissue uptake, thereby extending the late beta phase. Thus, the precise effect of glycopegylation on the alpha and beta phase half-lives will vary depending on size, glycosylation state, and other parameters, as is well known in the art. Further explanation of the "half-life" can be found in Pharmaceutical Biotechnology (1997, DFA Crommelin and RDSindalar, eds., Harwood publishers, Amsterdam, pp 101-120).
As used herein in the administration of a peptide drug to a patient, the term "retention time" is defined as the average time that the drug remains in the patient after administration.
An "isolated nucleic acid" refers to a nucleic acid segment or fragment that has been isolated from sequences that flank it in a naturally occurring state, such as a DNA fragment that has been removed from the sequence that normally flanks it, such as the sequence that flanks it in a naturally occurring genome. The term also applies to nucleic acids that have been substantially purified from other components that naturally accompany the nucleic acid, such as RNA or DNA or proteins that naturally accompany the nucleic acid in a cell. The term therefore also includes recombinant DNA such as that incorporated into a vector, or into an autonomously replicating plasmid or virus, or into the genomic DNA of a prokaryote or eukaryote, or that exists as a separate molecule (e.g., a cDNA or genomic or cDNA fragment produced by PCR or restriction enzyme digestion) independent of other sequences. It also includes recombinant DNA as part of a hybrid nucleic acid encoding additional peptide sequences.
"Polynucleotide" refers to a single-stranded or parallel and antiparallel-stranded nucleic acid. Thus, a polynucleotide may be a single-stranded or double-stranded nucleic acid.
The term "nucleic acid" generally refers to large polynucleotides. The term "oligonucleotide" generally refers to short polynucleotides, usually no more than about 50 nucleotides.
Conventional notation is used herein to describe polynucleotide sequences: the left-hand end of the single-stranded polynucleotide sequence is the 5' -end; the left-hand direction of a double-stranded polynucleotide sequence is referred to as the 5' -direction. The direction of nucleotide addition from 5 'to 3' to the nascent RNA transcript is referred to as the direction of transcription. A DNA strand having the same sequence as mRNA is called "coding strand"; sequences on the DNA strand that are 5' to a reference site on the DNA are referred to as "upstream sequences"; sequences on the DNA strand that are 3' to a reference site on the DNA are referred to as "downstream sequences".
"encoding" refers to the inherent characteristic of a particular nucleotide sequence in a polynucleotide (e.g., a gene, cDNA, or mRNA) that serves as a template for the synthesis of other polymers and macromolecules in biological processes having defined nucleotide sequences (i.e., rRNA, tRNA, and mRNA) or defined amino acid sequences and biological characteristics resulting therefrom. Thus, a nucleic acid sequence encodes a protein if transcription and translation of the mRNA corresponding to the nucleic acid produces the protein in a cell or other biological system. Both the coding strand, whose nucleotide sequence is identical to that of the mRNA and is typically provided in the sequence listing, and the non-coding strand, which serves as a template for transcription of the gene or cDNA, may be referred to as a protein or other product that encodes the nucleic acid or cDNA.
Unless otherwise defined, "a nucleotide sequence encoding an amino acid sequence" includes all nucleotide sequences that encode the same amino acid sequence in degenerate form to each other. Nucleotide sequences encoding proteins and RNAs may include introns.
As used herein, "homology" refers to subunit sequence similarity between two polymer molecules, such as between two nucleic acid molecules, such as between two DNA molecules or two RNA molecules, or between two peptide molecules. When a subunit position in two molecules is occupied by the same monomeric subunit, e.g., if a position in both DNA molecules is occupied by adenine, then they are homologous at that position. Homology between two sequences is a direct function of the number of matches or positions of homology, e.g., if half of the positions (e.g., 5 positions in a polymer of 10 subunits in length) in two compound sequences are homologous then the two sequences are 50% homologous, and if 90% of the positions, e.g., 9 out of 10, are matched or homologous then the two sequences are 90% homologous. For example, the DNA sequences 3 ' ATTGCC5 ' and 3 ' TATGGC have 50% homology.
As used herein, "homology" is used synonymously with "identity".
The determination of percent identity between two nucleotide or amino acid sequences can be accomplished using a mathematical algorithm. For example, the mathematical algorithm used to compare the two sequences is that of Karlin and Altschul (1990, Proc. Natl. Acad. Sci. USA 87: 2264-. The algorithm is integrated into the NBLAST and XBLAST programs of Altschul et al (1990, J.mol.biol.215: 403-. BLAST nucleotide searches can be performed using the NBLAST program (named "blastn" on NCBI internet sites) using the following parameters: gap penalty of 5; gap extension penalty of 2; mismatch penalty of 3; 1 is the matching award; the expected value is 10.0; and string size 11 to obtain nucleotide sequences homologous to the nucleic acids described herein. BLAST protein searches can be performed using the XBLAST program (designated "blastn" on NCBI internet sites) or the NCBI "blastp" program, using the following parameters: the expected value is 10.0; BLOSUM62 scoring matrix to obtain amino acid sequences homologous to the protein molecules described herein. To obtain Gapped alignments for comparison purposes, Gapped BLAST as described in Altschul et al (1997, Nucleic Acids Res.25: 3389-. Alternatively, PSI-Blast or PHI-Blast can be used to perform repeated searches that probe distant relationships (Id.) between molecules and relationships between molecules with common patterns. When BLAST, Gapped BLAST, PSI-BLAST, and PHI-BLAST programs are used, default parameters for the respective programs (e.g., XBLAST and NBLAST) can be used. See http:// www.ncbi.nlm.nih.gov.
Percent identity between two sequences can be determined using techniques similar to those described above that allow or disallow gaps. In calculating percent identity, a typical exact match is calculated.
A "heterologous nucleic acid expression unit" encoding a peptide is defined as a nucleic acid having a peptide-coding sequence of interest operably linked to one or more expression control sequences, such as a promoter and/or inhibitor sequence, and wherein at least one of the sequences is heterologous, i.e., not normally found in a host cell.
Two polynucleotides are "operably linked" in the sense that the two polynucleotides are arranged in the nucleic acid portion in such a way that at least one of the two polynucleotides is capable of exerting its characteristic physiological effect on the other. For example, a promoter operably linked to a nucleic acid coding region is capable of initiating transcription of the coding region.
As used herein, the term "promoter/regulatory sequence" refers to a nucleic acid sequence necessary for expression of a gene product operably linked to the promoter/regulatory sequence. In some instances, the sequence may be a core promoter sequence, while in other instances, the sequence may also include enhancer sequences and other regulatory elements necessary for expression of the gene product. For example, the promoter/regulatory sequence may be a sequence that expresses the gene product in a tissue-specific manner.
A "constitutive" promoter is a promoter that promotes expression of a gene to which it is operably linked in a cell in an invariant manner. For example, a promoter that promotes expression of a housekeeping gene of a cell may be considered a constitutive promoter.
An "inducible" promoter is a nucleotide sequence that, when operably linked to a polynucleotide that encodes or defines a gene product, results in the production of the gene product in a living cell substantially only when an inducer corresponding to the promoter is present in the cell.
A "tissue-specific" promoter is a nucleotide sequence that, when operably linked to a polynucleotide that encodes or defines a gene product, results in the production of the gene product in a living cell substantially only when the cell is of the tissue type corresponding to the promoter.
A "vector" is a composition of matter that comprises an isolated nucleic acid and can be used to deliver the isolated nucleic acid into the interior of a cell. Many vectors are known in the art, including but not limited to linear polynucleotides, polynucleotides related to ionic or amphipathic compounds, plasmids, and viruses. Thus, the term "vector" includes an autonomously replicating plasmid or virus. The term can also be interpreted to include non-plasmid and non-viral compounds capable of facilitating the transfer of nucleic acids to cells, such as polylysine compounds, liposomes, and the like. Examples of viral vectors include, but are not limited to, adenoviral vectors, adeno-associated viral vectors, retroviral vectors, and the like.
An "expression vector" refers to a vector comprising a recombinant polynucleotide comprising an expression control sequence operably linked to a nucleotide sequence to be expressed. The expression vector contains sufficient cis-acting elements for expression; other elements for expression may be provided by the host cell or in an in vitro expression system. Expression vectors include all those known in the art, such as cosmids, plasmids (e.g., naked or contained in liposomes) and viruses, which incorporate the recombinant polynucleotide.
A "genetically engineered" or "recombinant" cell is a cell in which one or more modifications have been made to the genetic material of the cell. Such modifications may include, but are not limited to, insertion of genetic material, deletion of genetic material, and insertion of extrachromosomal genetic material, whether stably maintained or not.
A "peptide" is an oligopeptide, polypeptide, peptide, protein, or glycoprotein. When a sugar molecule is attached thereto, the use of the term "peptide" herein includes peptides having a sugar molecule attached thereto.
As used herein, "native form" refers to the form of a peptide produced by a cell and/or organism in which the peptide is found in its native state. When the peptide is produced by a plurality of cells and/or organisms, the peptide may have various native forms.
"peptide" refers to a polymer in which the monomers are amino acids and are linked together by amide linkages, which are alternatively referred to as peptide bonds. Optionally, unnatural amino acids such as beta-alanine, phenylglycine, and homoarginine are also included. Non-nucleic acid encoded amino acids may also be used in the present invention. In addition, amino acids that have been modified to include reactive groups, glycosylation sites, polymers, therapeutic moieties, biomolecules, and the like, may also be used in the present invention. All amino acids used in the present invention may be in their D-or L-isomers. The L-isomer is generally preferred. In addition, other peptidomimetics may also be used in the present invention. As used herein, "peptide" refers to both glycosylated and non-glycosylated peptides. Also included are peptides that are incompletely glycosylated by a system expressing the peptide. For a general review, see Spatola, AF, Chemistry and biochemistry f Amino Acids, Peptides and Proteins, B.Weinstein, eds., Marcel Dekker, New York, p.267 (1983).
The term "peptide conjugate" refers to the class of the present invention (species) in which a peptide is conjugated to a modified saccharide as set forth herein.
The term "amino acid" refers to naturally occurring and synthetic amino acids, as well as amino acid analogs and amino acid mimetics that function in a manner similar to the naturally occurring amino acids. Naturally occurring amino acids are those encoded by the genetic code, as well as those amino acids that are subsequently modified, such as hydroxyproline, gamma-carboxyglutamic acid, and O-phosphoserine. Amino acid analogs refer to compounds that have the same basic chemical structure as a natural amino acid, i.e., an alpha carbon, a carboxyl group, an amino group, and an R group attached to a hydrogen, such as homoserine, norleucine, methionine sulfoxide, methionine methyl sulfonium. Such analogs have modified R groups (e.g., norleucine) or modified peptide backbones, but retain the same basic chemical structure as a naturally occurring amino acid. Amino acid mimetics refer to chemical compounds that have a structure that is different from the general chemical structure of an amino acid, but which function in a manner similar to a naturally occurring amino acid.
As used herein, amino acids are identified by their full name, corresponding three letter code, or corresponding one letter code, as shown in Table 1 below:
TABLE 1 amino acids and three and one letter codes
Complete name three letter code single letter code
Aspartic acid Asp D
Glutamic acid Glu E
Lysine Lys K
Arginine Arg R
Histidine His H
Tyrosine Tyr Y
Cysteine Cys C
Asparagine Asn N
Glutamine Gln Q
Serine Ser S
Threonine Thr T
Glycine Gly G
Alanine Ala A
Val V of valine
Leucine Leu L
Ile I
Methionine Met M
Proline Pro P
Phenylalanine Phe F
Tryptophan Trp W
The invention also provides analogues of proteins or peptides comprising the above proteins. Analogs differ from naturally occurring proteins or peptides by conservative amino acid sequence differences or modifications that do not affect the sequence, or both. For example, conservative amino acid changes may be made that, while changing the primary sequence of a protein or peptide, do not normally alter its function. Conservative amino acid substitutions generally include substitutions within the following groups:
glycine, alanine;
valine, isoleucine, leucine;
aspartic acid, glutamic acid;
asparagine, glutamine;
serine, threonine;
lysine, arginine;
phenylalanine, tyrosine.
Modifications (which do not generally alter primary sequence) include in vivo or in vitro chemical derivatization of peptides, such as acetylation or carboxylation. Also included are glycosylation modifications, such as by modification of the glycosylation pattern of the peptide during its synthesis and processing or in further processing steps; such as by exposing the peptide to an enzyme that affects glycosylation, such as a mammalian glycosylation or deglycosylation enzyme. Also included are sequences having phosphorylated amino acid residues, such as phosphotyrosine, phosphoserine, or phosphothreonine.
Of course, it is understood that the peptide may incorporate amino acid residues that are modified without affecting activity. For example, the termini may be derivatized to include blocking groups, i.e., chemical substituents suitable to protect and/or stabilize the N-and C-termini against "unwanted degradation," which is any type of enzymatic, chemical or biochemical disruption of the compound involving the termini that may affect the function of the compound, i.e., degradation that occurs sequentially at the termini of the compound.
Blocking groups include protecting groups conventionally used in the field of peptide chemistry that do not adversely affect the in vivo activity of the peptide. For example, suitable N-terminal blocking groups may be introduced by alkylation or acylation of the N-terminus. Examples of suitable N-terminal blocking groups include C1-C5Branched or unbranched alkyl groups, acyl groups such as formyl and acetyl groups and substituted versions thereof such as acetamidomethyl (Acm), Fmoc or Boc groups. Deaminated analogs of amino acids are also useful N-terminal blocking groups and can be coupled to the N-terminus of a peptide or used to replace N-terminal residues. Suitable C-terminal blocking groups include esters, ketones and amides, with or without the C-terminal carboxyl group being incorporated. Alkyl groups forming esters or ketones, in particular lower alkyl groups such as methyl, ethyl and propyl, and amide-forming amino groups such as primary amines (-NH) 2) And mono-and di-alkylamino groups such as methylamino, ethylamino, dimethylamino, diethylamino, methylethylamino and the like are examples of C-terminal blocking groups. Decarboxylated amino acid analogs such as spermine are also useful C-terminal blocking groups and can be used to couple to or replace the C-terminus of a peptide. Further, it is understood that the terminal free amino and carboxyl groups may be removed together from the peptide to obtain its deaminated and decarboxylated form without affecting the peptide activity.
Other modifications may also be incorporated without adversely affecting activity and include, but are not limited to, substitution of one or more of the naturally occurring L-isomeric amino acids with a D-isomeric amino acid. Thus, a peptide may comprise one or more D-amino acid residues, or may comprise all of the amino acids in the D-form. Reverse-inversional (retro-inverso) versions of the peptides according to the invention may also be used, for example inverted (inversed) peptides (in which all amino acids are substituted with the D-amino acid form).
The acid addition salts of the present invention are also contemplated to be functional equivalents. Thus, treatment of the peptides of the invention with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid and the like, or organic acids such as acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, malic acid, succinic acid, maleic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid, sialic acid and the like, to provide water soluble salts of the peptides are suitable for use in the invention.
Also included are peptides that have been modified using common molecular biology techniques to improve their resistance to protease degradation or to optimize solubility properties or to make them more suitable for use as therapeutic agents. Analogs of such peptides include those containing residues other than naturally occurring L-amino acids, such as D-amino acids or non-naturally occurring synthetic amino acids. The peptides of the invention are not limited to the products of any particular exemplary method listed herein.
As used herein, the term "MALDI" is an abbreviation for matrix-assisted laser desorption ionization. During ionization, SA-PEG (sialic acid-poly (ethylene glycol)) can be partially removed from the N-glycan structure of the glycoprotein.
As used herein, the term "glycosyltransferase" refers to any enzyme/protein that has the ability to transfer a donor saccharide to an acceptor moiety.
As used herein, the term "modified sugar" refers to a naturally or non-naturally occurring sugar that is enzymatically added to the glycosyl residue of an amino acid or peptide in the methods of the invention. The modified sugar is selected from a number of enzyme substrates including, but not limited to, sugar nucleotides (mono-, di-, and tri-phosphates), activated sugars (e.g., glycosyl halides, glycosyl mesylates), and sugars that are neither activated nor nucleotides.
A "modified sugar" is covalently functionalized with a "modifying group". Useful modifying groups include, but are not limited to, water-soluble polymers, therapeutic moieties, diagnostic moieties, biomolecules, and the like. The position of functionalization with the modifying group is chosen so as not to prevent enzymatic addition of the "modified sugar" to the peptide.
The term "water-soluble" refers to moieties that have some detectable degree of solubility in water. Methods of detecting and/or quantifying water solubility are well known in the art. Exemplary water-soluble polymers include peptides, sugars, poly (ethers), poly (amines), poly (carboxylic acids), and the like. Peptides may have mixed sequences or consist of only a single amino acid, such as poly (lysine). Similarly, the saccharide may be a mixed sequence or consist of only a single saccharide subunit, such as dextran, amylose, chitosan and poly (sialic acid). An exemplary poly (ether) is poly (ethylene glycol). Poly (ethyleneimine) is an exemplary polyamine, and poly (aspartic) acid is a representative poly (carboxylic acid).
"polyalkylene oxide" refers to a class of compounds having a polyether backbone. The polyalkylene oxide species useful in the present invention include, for example, straight chain and branched chain species. Further, the ends of exemplary polyalkylene oxides can be one or more reactive, activatable, or inert moieties. For example, a polyalkylene oxide is a polyalkylene oxide composed of repeating ethylene oxide subunits, which may or may not include additional reactive, activatable, or inert moieties at each end. Useful polyalkylene oxide species include those that are "capped" at one end by an inert group, such as monomethoxy-polyalkylene oxide. When the molecule is a branched species, multiple reactive, activatable or inert groups may be included at the end of the alkylene oxide chain, and the reactive groups may be the same or different. Derivatives of heterobifunctional linear polyalkylene oxide species are also well known in the art.
As used herein, the term "glycosyl linking group" refers to a glycosyl residue to which an agent (e.g., water-soluble polymer, therapeutic moiety, biomolecule) is covalently attached. In the methods of the invention, a "glycosyl linking group" is covalently attached to a glycosylated or non-glycosylated peptide, thereby linking the agent to amino acids and/or glycosyl residues on the peptide. "glycosyl linking groups" are typically derived from "modified sugars" by the enzymatic attachment of the "modified sugar" to amino acids and/or glycosyl residues on the peptide. More specifically, as used herein, "glycosyl linking group" refers to a moiety that covalently binds a "modifying group" as discussed herein to an amino acid residue of a peptide. The glycosyl linking group-modifying group adduct has a structure that is a substrate for an enzyme. Enzymes having a glycosyl linker-modifier adduct as substrate are generally those capable of transferring a glycosyl moiety to an amino acid residue of a peptide, such as glycosyltransferases, amidases, glycosidases, trans-sialidases, and the like. A "glycosyl linking group" is inserted between and covalently bonded to a "modifying group" and an amino acid residue of a peptide.
An "intact glycosyl linking group" refers to a linking group derived from a glycosyl moiety wherein the individual saccharide monomers attached to the conjugate are not degraded, e.g., oxidized (e.g., by sodium metaperiodate). The "intact glycosyl linking group" of the invention can be derived from a naturally occurring oligosaccharide by the addition of a glycosyl unit or the removal of one or more glycosyl units from the parent sugar structure. Exemplary "intact glycosyl linking groups" include at least one intact (e.g., non-degraded) glycosyl moiety covalently attached to an amino acid residue on a peptide. The remainder of the "linking group" can have essentially any structure. For example, the modifying group is optionally directly attached to the entire glycosyl moiety. Alternatively, the modifying group is attached to the entire glycosyl moiety through the linker arm. The linker arm can have essentially any structure that can be used in selected embodiments. In an exemplary embodiment, the linker arm is one or more intact glycosyl moieties, i.e., "intact glycosyl linking group" resembles an oligosaccharide. Another exemplary intact glycosyl linking group is one in which the glycosyl moiety attached directly or indirectly to the intact glycosyl moiety is degraded or derivatized (e.g., periodate oxidation followed by reductive amination). The other linker arm includes a modifying group, such as those described herein and the like, attached directly or indirectly to the entire glycosyl moiety through a cross-linking agent.
As used herein, "degrade" refers to the removal of one or more carbon atoms from the glycosyl moiety.
As used herein, the terms "targeting moiety" and "targeting agent" refer to a species that selectively localizes to a particular tissue or region of the body. This localization is mediated by specific recognition of molecular determinants, molecular size of the targeting agent or conjugate, ionic interactions, hydrophobic interactions, and the like. Other mechanisms for directing an agent to a particular tissue or region are known to those skilled in the art.
As used herein, "therapeutic moiety" refers to any agent used in therapy, including, but not limited to, antibiotics, anti-inflammatory agents, antineoplastic agents, cytotoxins, and radioactive agents. "therapeutic moiety" includes prodrugs of biologically active agents, i.e., a construct in which more than one therapeutic moiety is linked to a carrier, e.g., a multivalent agent. Therapeutic moieties also include peptides and constructs comprising the peptides. Exemplary peptides include those disclosed herein in figure 28 and tables 6 and 7. Thus, "therapeutic moiety" means any agent used in therapy, including, but not limited to, antibiotics, anti-inflammatory agents, antineoplastic agents, cytotoxins, and radioactive agents. "therapeutic moiety" includes prodrugs of biologically active agents, i.e., constructs in which more than one therapeutic moiety is attached to a carrier, such as a multivalent agent.
As used herein, "antineoplastic agent" refers to any agent useful against cancer, including, but not limited to, cytotoxins and agents such as antimetabolites, alkylating agents, plectranthin, antibiotics, antimitotics, procarbazine, hydroxyurea, asparaginase, corticosteroids, interferons, and radioactive agents. Also included within the scope of the term "antineoplastic agent" are conjugates of peptides with antineoplastic actives such as TNF- α. Conjugates include, but are not limited to, those formed between a therapeutic protein and a glycoprotein of the invention. Representative conjugates are formed between PSGL-1 and TNF- α.
As used herein, "cytotoxin or cytotoxic agent" refers to any agent that is harmful to cells. Examples include paclitaxel, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, epipodophyllotoxin glucopyranoside, epipodophyllotoxin thiophenoside (tenoposide), vincristine, vinblastine, colchicine, doxorubicin, daunorubicin, dihydroxyanthrenedione, mitoxantrone, plicamycin, actinomycin D, 1-dehydrotestosterone, glucocorticoids, procaine, tetracaine, lidocaine, propranolol, and puromycin and analogs or homologs thereof. Other toxins include, for example, ricin, CC-1065, and the analog duocarmycin. Still other toxins include diphtheria toxin and snake venom (e.g., cobra venom).
As used herein, "radioactive agent" includes any radioisotope effective in diagnosing or destroying a tumor. Examples include, but are not limited to, indium-111, cobalt-60, and technetium. Furthermore, naturally occurring radioactive elements such as uranium, radium and thorium, which generally represent a mixture of radioisotopes, are suitable examples of radioactive agents. The metal ion is typically chelated to the organic chelating moiety.
Many useful chelating groups, crown ethers, cryptands, and the like are well known in the art and can be incorporated into the compounds of the invention (e.g., EDTA, DTPA, DOTA, NTA, HDTA, and the like, and phosphate analogs thereof such as DTPP, EDTP, HDTP, NTP, and the like). See, e.g., Pitt et al, "the design of chemical Agents for the Treatment of Iron Overload," organic Chemistry in Biology and Medicine; martell, ed.; american Chemistry Society, Washington, D.C., 1980, pp.279-312; lindoy, The Chemistry of multicyclic Ligand Complexes; cambridge university Press, Cambridge, 1989; dugas, Bioorganic Chemistry; Springer-Verlag, New York, 1989, and references contained therein.
Furthermore, a variety of routes to attach chelators, crown ethers, and cyclodextrins to other molecules are available to those skilled in the art. See, e.g., Meares et al, "Properties of InVivo Chemate-Tagged Proteins and Polypeptides", Modification of Proteins: food, Nutritional and pharmaceutical accessories; feeney et al, eds., American Chemical Society, Washington, D.C., 1982, pp.370-387; kasina et al, Bioconjugate chem, 9: 108-117 (1998); song et al, Bioconjugate chem, 8: 249-255(1997).
As used herein, "pharmaceutically acceptable carrier" includes any material that retains the activity of the conjugate when bound thereto and is non-reactive with the immune system of the subject. Examples include, but are not limited to, any standard pharmaceutical carrier such as phosphate buffered saline, water, emulsions such as oil/water emulsions, and various types of wetting agents. Other carriers may also include sterile solutions, tablets (including coated tablets) and capsules. Typically such carriers contain excipients such as starch, milk, sugar, certain types of clays, gelatin, stearic acid and its salts, magnesium or calcium stearate, talc, vegetable fats or oils, gums, glycols or other known excipients. Such carriers may also include fragrance and color additives or other ingredients. Compositions containing such carriers are prepared by well-known conventional methods.
As used herein, "administration" refers to oral administration to a subject, administration as a suppository, topical contact, intravenous, intraperitoneal, intramuscular, intralesional, intranasal or subcutaneous administration, intrathecal administration or implantation of a slow release device, such as a mini-osmotic pump.
The term "isolated" refers to a material that is substantially free of the components used to make the material. For the peptide conjugates of the present invention, the term "isolated" refers to a material that is substantially free of components that are normally associated with the material in the mixture used to prepare the peptide conjugate. "isolated" and "pure" are used interchangeably-generally, the isolated peptide conjugates of the present invention have a purity level that is preferably expressed as a range. The lower limit of the purity range is about 60%, about 70%, or about 80%, while the upper limit of the purity range is about 70%, about 80%, about 90%, or more than about 90%.
When the peptide conjugate is more than about 90% pure, its purity is also preferably expressed as a range. The lower limit of the purity range is about 90%, about 92%, about 94%, about 96%, or about 98%, and the upper limit of the purity range is about 92%, about 94%, about 96%, about 98%, or about 100%.
Purity is determined by any art-recognized analytical method (e.g., band brightness on silver stained gel, polyacrylamide gel electrophoresis, HPLC, or the like).
As used herein, "commercial scale" means that about 1 gram or several grams of the final product is produced in the process.
As used herein, "substantially each member of a population" refers to a characteristic of a population of peptide conjugates of the invention, wherein a selected percentage of modified sugars added to a peptide are added to multiple identical receptor sites on the peptide. "substantially every member of the population" refers to "homogeneity" with a site on the modified glycoconjugated peptide and to conjugates of the invention that are at least about 80%, preferably at least about 90% and more preferably at least about 95% homogeneous.
"homogeneity" refers to structural identity in the population of receptor moieties to which the modified saccharide is conjugated. Thus, in a peptide conjugate of the invention in which each modified saccharide moiety is conjugated to an acceptor site, and the acceptor site has the same structure as the acceptor site to which each other modified saccharide is conjugated, the peptide conjugate is said to be about 100% homogeneous. Homogeneity is generally expressed as a range. The lower limit of the homogeneity range of the peptide conjugate is about 60%, about 70%, or about 80%, while the upper limit of the purity range is about 70%, about 80%, about 90%, or more than about 90%.
When the peptide conjugate is more than or equal to about 90% homogeneous, its homogeneity is also preferably expressed as a range. The lower limit of the homogeneity range is about 90%, about 92%, about 94%, about 96%, or about 98%, while the upper limit of the purity range is about 92%, about 94%, about 96%, about 98%, or about 100%. The purity of the peptide conjugate is generally determined by one or more methods known to those skilled in the art, such as liquid chromatography-mass spectrometry (LC-MS), matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI-TOF), capillary electrophoresis, and the like.
When referring to glycopeptides, a "substantially uniform glycoform" or "substantially uniform glycosylation pattern" refers to the percentage of acceptor moieties that are glycosylated by a glycosyltransferase of interest (e.g., a fucosyltransferase). For example, in the case of α 1, 2-fucosyltransferase, a substantially uniform fucosylation pattern exists if substantially all (as defined below) of Gal β 1, 4-GlcNAc-R and sialylated analogs thereof in the peptide conjugates of the invention are fucosylated. It will be appreciated by those skilled in the art that the starting material may contain a glycosylated acceptor moiety (e.g., a fucosylated Gal β 1, 4-GlcNAc-R moiety). Thus, the calculated percent glycosylation will include acceptor moieties glycosylated by the methods of the invention as well as those acceptor moieties that have been glycosylated in the starting material.
The term "substantially" in the above definition of "substantially uniform" generally means that at least about 40%, at least about 70%, at least about 80%, or more preferably at least about 90% and still more preferably at least about 95% of the acceptor moiety of a particular glycosyltransferase is glycosylated.
Description of the invention
I. Method for reconstructing glycan chains
The present invention includes methods and compositions for the in vitro addition of saccharides to and/or deletion of saccharides from glycopeptide molecules in such a way as to provide peptide molecules having a particular tailored or desired glycosylation pattern, preferably including the addition of modified saccharides thereto. It is therefore a key feature of the present invention to take a peptide produced by any cell type and generate a decorin structure on the peptide, which is subsequently reconstituted in vitro to generate a peptide having a glycosylation pattern suitable for therapeutic use in mammals.
The importance of glycosylation patterns of peptides is well known in the art, as are the limitations of existing in vivo methods for producing properly glycosylated peptides, particularly when these peptides are produced by recombinant DNA methods. Furthermore, until the present invention, it has not been possible to generate glycopeptides having a desired glycan structure thereon, wherein the peptides can be produced on an industrial scale.
In the present invention, peptides produced by cells are enzymatically treated in vitro by systematically adding appropriate enzymes and their substrates so that the carbohydrate moieties that should not be present on the peptides can be removed, and the carbohydrate moieties that should be added to the peptides (optionally including modified carbohydrates) are added to the peptides in such a way as to provide glycopeptides having a "desired glycosylation" as defined elsewhere.
A. Method for reconstituting N-linked glycans
In one aspect, the invention takes advantage of the fact that most peptides of commercial or pharmaceutical interest contain a common pentasaccharide structure, referred to herein as the trimannose core, which is N-linked to asparagine in the Asn-X-Ser/Thr sequence of the peptide chain. The basic trimannose core consists essentially of 2N-acetylglucosamine (GlcNAc) and 3 mannose (Man) residues attached to the peptide, i.e. it contains these 5 sugar residues without additional sugars, with the exception that it may optionally include a fucose residue. The first GlcNAc is attached to the amide group of asparagine and the second GlcNAc is attached to the first by a β 1, 4-linkage. 1 mannose residue is attached to the second GlcNAc by a β 1, 4-linkage, and 2 mannose residues are attached to the mannose by α 1, 3 and α 1, 6 linkages, respectively. A schematic depiction of the trimannose core structure is shown on the left of figure 1. While the glycan structures on most peptides contain other sugars besides the trimannose core, the trimannose core structure represents an essential feature of N-linked glycans on mammalian peptides.
The present invention includes the generation of peptides having a trimannose core structure as a basic structural element of a glycan molecule contained thereon. Given the variety of cellular systems available for the production of peptides, whether naturally occurring per se or involving recombinant DNA methodology, the present invention provides a means by which glycan structures on peptides produced in any cell type can be reduced to the basic trimannose core structure. Once the basic trimannose core structure is generated, it is possible to generate in vitro the desired glycan structure on the peptide using the methods described herein, which confers one or more properties on the peptide that enhance its therapeutic efficacy.
From the discussion herein it can be seen that the term "trimannose core" is used to describe the glycan structure shown on the left side of figure 1. Glycopeptides having a trimannose core structure also have additional sugars added thereto, and for the most part, additional structures added thereto, regardless of whether the sugars impart the desired glycan structure to the peptide. The term "essential trimannose core structure" is defined elsewhere. When the term "essential" is not included in the description of "trimannose core structure", then the glycan comprises the trimannose core structure with an additional sugar attached to the mannose.
The term "basic trimannose core glycopeptide" is used herein to refer to glycopeptides having a glycan structure which predominantly comprises a basic trimannose core structure. However, it may alternatively contain fucose residues attached thereto. As discussed herein, the basic trimannose core glycopeptide is the most suitable and therefore the preferred starting material for the glycan reconstitution process of the present invention.
Another most suitable starting material for the glycan remodeling process of the present invention is a glycan structure with a trimannose core in which one or more additional GlcNAc residues are added to each of α 1, 3 and α 1, 6 of the mannose residues (see the structure on the second line in fig. 2, the second structure from the left of the figure). This structure is referred to herein as "Man 3GlcNAc 4". When the structure is a single-touch angle, the structure is referred to herein as "Man 3GlcNAc 3" alternatively, the structure may also contain a core fucose molecule. Once the Man3GlcNAc3 or Man3GlcNAc4 structure is generated, it is possible to generate in vitro the desired glycan structures on a peptide using the methods described herein, which confer one or more properties on the peptide that enhance its therapeutic efficacy.
In its native form, the N-linked glycopeptides of the present invention, particularly mammalian and human glycopeptides useful in the present invention, are N-linked glycosylated with a trimannose core structure to which one or more sugars are attached.
The terms "glycopeptide" and "glycopolypeptide" are used synonymously herein to refer to a peptide chain having a carbohydrate moiety attached thereto. Small glycopolypeptides or glycopeptides are not distinguished herein from large glycopolypeptides or glycopeptides. Thus, hormone molecules and other very large peptides having very few amino acids (e.g., as few as 3 amino acids) in their peptide chain are encompassed by the general terms "glycopolypeptide" and "glycopeptide" so long as they have a carbohydrate moiety attached thereto. However, the use of the term "peptide" does not exclude that the peptide is a glycopeptide.
An example of an N-linked glycopeptide having a desired glycosylation is a peptide having an N-linked glycan with a trimannose core having at least one GlcNAc residue attached thereto. This residue was added to the trimannose core using N-acetylglucosaminyltransferase I (GnT-I). If a second GlcNAc is added, N-acetylglucosaminyltransferase II (GnT-II) is used. Alternatively, additional GlcNAc residues can be added by GnT-IV and/or GnT-V, and the third bisecting GlcNAc residue can be attached to the β 1, 4 mannose of the trimannose core by N-acetylglucosaminyltransferase III (GnT-III). Alternatively, the structure may be extended with β 1, 4 galactosyltransferase treatment to add a galactose residue to each non-bisected GlcNAc, and further alternatively, sialic acid residues may be added to each galactose residue using α 2, 3 or α 2, 6-sialyltransferase. The addition of aliquots of GlcNAc to glycans is not necessary for the subsequent addition of galactose and sialic acid residues; however, the presence of one or more galactose residues on glycans excludes the addition of bisecting GlcNAc for substrate affinity of rat and human GnT-III enzymes, because galactose-containing glycans are not substrates for these forms of GnT-III. Thus, in the case where aliquots of GlcNAc are intended to exist and use these forms of GnT-III, it is important that if glycans contain added galactose and/or sialic acid, they should be removed before aliquots of GlcNAc are added. Other forms of GnT-III activity may not require this particular substrate order. In a more preferred reaction, a mixture of GnT-I, GnT-II and GnT-III is added to the reaction mixture so that the GlcNAc residues can be added in any order.
Examples of glycan structures representing various aspects of peptides with "desired glycosylation" are shown in the figures provided herein. The precise process for the in vitro production of peptides with "desired glycosylation" is described elsewhere. However, the present invention should in no way be construed as being limited to only any one of the glycan structures disclosed herein. The invention should more suitably be construed to include any or all of the glycan structures that can be made using the methods provided herein.
In some cases, the basic trimannose core alone may constitute the desired glycosylation of the peptide. For example, peptides having only a trimannose core have been shown to be useful components of enzymes for treating gaucher's disease (Mistry et al, 1966, Lancet 348: 1555-1559; Bijsterbosch et al, 1966, Eur.J.biochem.237: 344-349).
According to the present invention, the procedure for generating peptides with the desired glycosylation is described below.
a) Starting from a glycopeptide having one or more glycan molecules having a common feature of at least one or more heterogeneous or homogeneous mixtures having a trimannose core structure and one or more sugars added thereto, it is possible to increase the proportion of glycopeptides having the basic trimannose core structure as the only glycan structure or having Man3GlcNAc3 or Man3GlcNAc4 as the only glycan structure. This is achieved in vitro by systematically adding an appropriate number of enzymes to the glycopeptide in the appropriate order that can cleave heterogeneous or homogeneous mixtures of sugars on the glycan structure until they are reduced to the basic trimannose core or Man3GlcNAc3 or Man3GlcNAc4 structure. The particular examples of how this is achieved will depend on a variety of factors, including in large part the type of cell producing the peptide and therefore the degree of complexity of the glycan structures present on the peptide originally produced by the cell. Examples of how complex glycan structures can be reduced to the basic trimannose core or Man3GlcNAc3 or Man3GlcNAc4 structures are shown in fig. 2, or described in detail elsewhere.
b) It is possible to generate peptides with the basic trimannose core structure as the only glycan structure by isolating naturally occurring cells whose glycosylation machinery produces such peptides. The DNA encoding the peptide of interest is then transfected into cells where it is transcribed, translated and glycosylated, such that the peptide of interest has the basic trimannose core structure as the only glycan structure. For example, cells lacking a functional GnT-I enzyme will produce several types of glycopeptides. In some cases, these will be glycopeptides that do not have an additional sugar attached to the trimannose core. However, in other cases, the resulting peptide may have 2 additional mannose residues attached to the trimannose core, resulting in Man5 glycans. This is also the desired starting material for the reconstitution process of the present invention. Specific examples of generating such glycan structures are described herein.
c) Alternatively, it is possible to genetically engineer a cell to confer a particular glycosylation machinery on the cell, thereby producing a peptide having a substantially trimannose core or a Man3GlcNAc3 or Man3GlcNAc4 structure as the only glycan structure on the peptide. The DNA encoding the peptide of interest is then transfected into a cell where it is transcribed, translated, and glycosylated, such that the peptide of interest has an increased number of glycans that individually comprise the basic trimannose core structure. For example, certain types of cells genetically engineered to lack GnT-I will produce glycans with a basic trimannose core structure, or depending on the cell, can produce glycans with a trimannose core and 2 additional mannose residues attached thereto (Man 5). When the cell produces Man5 glycan structures, the cell can be further genetically engineered to express mannosidase 3, which cleaves 2 additional mannose residues to generate a trimannose core. Alternatively, Man5 glycans can be incubated with mannosidase 3 in vitro to achieve the same effect.
d) When the peptide is expressed in insect cells, the glycans on the peptide comprise partially complexed chains. Insect cells also express hexosaminidase in the cell, which cleaves a partially complexed strand into a trimannose core structure, which is then reconstituted as described herein.
e) It is apparent from the discussion in b), c) and d) that the cell need not produce only peptides having a substantially trimannose core or Man3GlcNAc3 or Man3GlcNAc4 structure attached thereto. It is more appropriate that unless the cells described in b) and c) produce a polypeptide having either 100% of the basic trimannose core structure (i.e. no additional sugar attached thereto) or 100% of the Man3GlcNAc3 or Man3GlcNAc4 structure, the cells in fact produce a heterogeneous mixture of peptides having these structures with other sugars attached thereto in addition to the basic trimannose core structure or a combination of the Man3GlcNAc3 or Man3GlcNAc4 structures as the only glycan structures. The ratio of peptides having a trimannose core or Man3GlcNAc3 or Man3GlcNAc4 structure with additional sugars attached thereto to those having only one structure will vary depending on the cell in which they are produced. The complexity of the glycans (i.e. what and how much of the sugar is attached to the trimannose core) will also depend on the cell that produces them.
f) Once a glycopeptide having a basic trimannose core or a trimannose core having one or more GlcNAc residues attached thereto is produced by a), b) or c) according to the present invention, additional sugar molecules can be added to the trimannose core structure in vitro to generate a peptide having the desired glycosylation (i.e. a peptide having a glycan structure tailored in vitro).
g) However, when a peptide is produced having an essentially trimannose core or Man3GlcNAc4 structure with some, but not all, of the desired sugars attached thereto, then only the remaining desired sugars need be added, without reducing the glycan structure to an essentially trimannose core or Man3GlcNAc4 structure. Thus, in some cases, a peptide with a glycan structure having a trimannose core structure with additional sugars attached thereto will be a suitable substrate for reconstitution.
Isolation of basic trimannose core glycopeptides
If desired, the basic trimannose core or Man3GlcNAc3 or Man3GlcNAc4 glycopeptides of the present invention can be isolated and purified using techniques well known in the art of peptide purification. Suitable techniques include chromatographic techniques, isoelectric focusing techniques, ultrafiltration techniques, and the like. The compositions of the invention can be prepared using any such technique, wherein the glycopeptides of the invention are separated from other peptides and from other components typically found in cell culture media. The degree of purification may be, for example, 90%, or 95% or higher, such as 98%, relative to other peptides. See, e.g., Deutscher et al (ed., 1990, Guide to Protein Purification, Harbourt Brace Jovanovich, San Diego).
The heterogeneity of N-linked glycans present in glycopeptides produced by prior art methods generally allows isolation of only a small fraction of the glycopeptide of interest, which can be modified to produce the desired glycopeptide. In the methods of the invention, substantial trimannose core glycopeptides and other desired glycopeptides, including Man3GlcNAc3 or Man3GlcNAc4 glycopeptides, can be produced in large quantities, which can then be further modified to produce large quantities of peptides having the desired glycosylation.
Specific enrichment of any particular type of glycan attached to a peptide can be achieved with lectins that have an affinity for the desired glycan. Such techniques are well known in the art of glycobiology.
A key feature of the invention, described in more detail below, is that once a glycan structure is generated on any peptide, it can be reconstituted in vitro to generate a peptide with the desired glycosylation, which has improved therapeutic applications in mammals. The mammal may be any type of suitable mammal, and is preferably a human.
Various situations and precise methods and compositions for producing peptides with the desired glycosylation will become apparent in the ensuing disclosure.
The ultimate goal in producing a peptide for therapeutic use in mammals is that the peptide should contain glycan structures that promote, but do not negate, the therapeutic benefit of the peptide. As disclosed throughout the specification, peptides produced in cells can be treated in vitro with various enzymes that catalyze the cleavage of sugars that should not be present on glycans and the addition of sugars that should be present on glycans, thereby producing peptides having the desired glycosylation and thus suitable for therapeutic use in mammals. The production of different glycoforms of peptides in cells is described above. Various mechanisms for producing peptides with the desired glycosylation are now described, wherein the starting material, i.e., the peptide produced by the cell, varies depending on the cell type. As will be apparent from the disclosure of the present invention, the starting materials need not be uniform in their glycan composition. However, it is preferred to enrich the starting material for certain glycoforms to produce large amounts of the final product, i.e. the correctly glycosylated peptide.
In a preferred embodiment according to the present invention, the resulting degradation and synthesis events of a peptide having a desired glycosylation sometimes involve the generation of a substantially trimannose core structure or a Man3GlcNAc3 or a Man3GlcNAc4 structure on the peptide.
The invention also provides a method of adding one or more selected glycosyl residues to a peptide, followed by conjugating a modified saccharide to at least one selected glycosyl residue on the peptide. This embodiment is useful, for example, when it is desired to conjugate a modified sugar to a selected glycosyl residue, wherein the selected glycosyl residue is not present on the peptide or is not present in the desired amount. Thus, prior to coupling the modified saccharide to the peptide, a selected glycosyl residue is conjugated to the peptide either by enzymatic or chemical coupling. In another embodiment, the glycosylation pattern of the peptide is altered by removing saccharide residues from the peptide prior to conjugation of the modified saccharide. See, e.g., WO 98/31826.
The addition or removal of any sugar moieties present on the peptide is effected chemically or enzymatically. Chemical deglycosylation preferably occurs by exposing the peptide variant to the compound triflic acid or equivalent compound. This treatment results in cleavage of most or all of the sugars except the linked sugar (N-acetylglucosamine or N-acetylgalactosamine) leaving the intact peptide. Chemical degradation is described in Hakimuddin et al, 1987, arch, biochem, biophysis, 259: 52 and Edge et al, 1981, anal. biochem.118: 131. enzymatic cleavage of the sugar moiety on peptide variants can be achieved by the application of various endo-and exo-glycosidases, as described by Thotakura et al, 1987, meth.enzymol.138: 350.
Chemical addition of the glycosyl moiety is carried out by any art-recognized method. Enzymatic addition of the glycosyl moiety is preferably accomplished using modifications to the methods presented herein, i.e., the natural glycosyl unit is substituted for the modified sugar used in the present invention. Other methods of adding sugar moieties are described in U.S. patent nos. 5,876,980, 6,030,815, 5,728,554 and 5,922,577.
Exemplary attachment sites for selected glycosyl residues include, but are not limited to: (a) n-and O-glycosylation sites; (b) a terminal glycosyl moiety as a glycosyltransferase acceptor; (c) arginine, asparagine, and histidine; (d) a free carboxyl group; (e) free sulfhydryl groups such as cysteine; (f) free hydroxyl groups such as serine, threonine or hydroxyproline; (g) aromatic residues such as those of phenylalanine, tyrosine or tryptophan; or (h) an amide group of glutamine. Exemplary methods for use in the present invention are described in WO 87/05330 and Aplin and Wriston, CRC crit. rev. biochem., pp.259-306(1981), published 11/9/1987.
With specific reference to the examples shown in the several figures provided herein, a description of an in vitro enzymatic reaction sequence to produce a desired glycan structure on a peptide is now provided. The precise reaction conditions for each of the enzymatic conversions described below are well known to those skilled in the art of glycobiology and are therefore not repeated here. For a review of the reaction conditions for these types of reactions, see Sadler et al, 1982, Methods in Enzymology 83: 458-514 and the references cited therein.
The basic trimannose core glycan structure is shown on the left in figure 1. It is possible to convert the basic trimannose core structure to an intact glycan structure with bisected GlcNAc by incubating the structure in the presence of GnT-I followed by GnT-II and further followed by GnT-III and a sugar donor comprising UDP-GlcNAc, wherein GlcNAc is sequentially added to the basic trimannose core structure to produce a trimannose core with bisected GlcNAc. In some cases, for example when the Fc glycans are reconstituted as described herein, the order of addition of GnT-I, GnT-II and GnT-III may be reversed from that reported in the literature. Aliquots of GlcNAc structures can be produced by adding GnT-I, GnT-II and GnT-III and UDP-GlcNAc to the reaction mixture.
The conversion of trimannose decorin containing bisecting GlcNAc to a complex glycan structure containing galactose and N-acetylneuraminic acid is shown in fig. 3. The trimannose core glycan containing an aliquot of GlcNAc is first incubated with galactosyltransferase and UDP-Gal as a donor molecule, in which two galactose residues are added to the peripheral GlcNAc residues on the molecule. NeuAc-transferase was then used to add one NeuAc per galactose residue for a total of two NeuAc.
The conversion of the high mannose glycan structure to the basic trimannose core glycan is shown in figure 4. High mannose glycans (Man9) were sequentially incubated in the presence of mannosidase 1 to generate a Man5 structure, followed by incubation in the presence of mannosidase 3, where all mannose residues except 3 would be removed from the glycans. Alternatively, the incubation of the Man9 structure can trim to the trimannose core structure by only incubation in the presence of mannosidase 3. The conversion of the basic trimannose core glycan to a complex glycan molecule is then possible according to the scheme provided in figures 1 and 3 above.
Typical complex N-linked glycan structures produced in plant cells are shown in FIG. 5. It is important to note that xylose and fucose cannot be added to glycans when the plant cells lack GnT-I enzymatic activity. Thus, the use of GnT-I knockout cells provides a particular advantage of the present invention in that these cells can produce peptides with a basic trimannose core to which additional sugars can be added without any "trimming" reactions. Similarly, in the case of Man5 variants whose structures are glycans produced in plant cells, xylose and fucose cannot be added to the structure if GnT-I is absent in these cells. In this case, the Man5 structure can be trimmed with mannosidase 3 into the basic trimannose core (Man 3). According to the methods provided herein, it is currently possible to add sugar moieties to the trimannose core to generate the desired glycan structures.
Typical complex N-linked glycan structures produced in insect cells are shown in FIG. 6. It is obvious that additional sugars such as fucose may also be present. Although not shown further here, insect cells can produce high mannose glycans with up to 9 mannose residues and onto which additional sugars can be attached. It is also the case that in insect cells, GnT-I knockout cells prevent the addition of fucose to glycans. Thus, the production of peptides in insect cells can preferably be effected in GnT-I gene knock-out cells. Thus, if desired, the glycans so produced can then be trimmed in vitro using any of the methods and protocols described herein, and additional sugars can also be added thereto in vitro using the methods and protocols described herein.
The glycan structures for various degrees of completion are shown in figure 2. Specifically, the basic trimannose core structure is shown to be enzymatically produced in vitro from complex glycan structures that do not contain bisecting GlcNAc residues. Also shown are glycan structures thus produced containing bisecting GlcNAc. Several intermediate glycan structures that can be produced are shown. These structures may be produced by cells, or may be produced in an in vitro trimming reaction as described herein. The sugar moiety can be added to the basic trimannose core structure in vitro, or to any suitable intermediate structure to produce the desired glycan.
A series of possible in vitro reactions that can perform the trimming and addition of glycans starting from high mannose structures is shown in figure 7. For example, Man9 glycan can be trimmed with mannosidase 1 to produce Man5 glycan, or it can be trimmed to the trimannose core with mannosidase 3 or one or more microbial mannosidases. GnT-I and or GnT-II can then be used to transfer additional GlcNAc residues to the glycans. Further, a situation is shown that does not occur when glycan molecules are produced in cells without GnT-I (see shaded box). For example, fucose and xylose can be added to glycans only when GnT-I is active and facilitates transfer of GlcNAc to the molecule.
Figure 8 depicts a well-known strategy for synthesizing bi-antennary, tri-antennary and even tetra-antennary glycan structures starting from a trimannose core structure. According to the method of the present invention, it is possible to synthesize each of these structures in vitro using appropriate enzymes and reaction conditions well known in the art of glycobiology.
FIG. 9 depicts two methods for synthesizing mono-antennary glycan structures starting from high mannose (6-9 mannose moieties) glycan structures. In accordance with the saccharide pegylation methods described herein, terminal sialic acid-PEG moieties can be added in place of sialic acid moieties. In the first method, endo-H is used to cleave the glycan structure on the peptide to the first GlcNAc residue. Galactose was then added with galactosyltransferase, while sialylated PEG was added as described elsewhere herein. In the second method, mannosidase I is used to cleave mannose residues from glycan structures in peptides. The galactose residue was added to one arm of the remaining mannose residue, which was cleaved from the glycan with canavalin a-mannosidase. Sialylated PEG was then added to the structure as described.
FIG. 10 depicts two additional methods for synthesizing mono-antennary glycan structures starting from high mannose (6-9 mannose moieties) glycan structures. As in fig. 9, a terminal sialic acid-PEG moiety was added in place of the sialic acid moiety according to the glycopegylation method described herein. In the case described here, some mannose residues were removed from the arm where no sialylated PEG was added.
The scheme for the synthesis of more complex sugar structures starting from the trimannose core structure is shown in FIG. 11. For example, protocols for the in vitro production of sialylated or non-sialylated Lewis x and Lewis a antigen structures are shown. Such structures, when present on a peptide, may confer immunological advantages to the peptide for up-or down-regulating immune responses. Furthermore, such structures may be useful for targeting of peptides to specific cells, as such structures are involved in binding to cell attachment peptides and the like.
FIG. 12 is an exemplary scheme for preparing O-linked peptide arrays derived from serine or threonine.
FIG. 13 is a series of diagrams depicting the structure of 4 types of O-linked glycans, referred to as cores 1-4. The core structure is depicted in dashed lines. Sugars that may also be included in the structure include sialic acid residues added to galactose residues and fucose residues added to GlcNAc residues.
Thus, in a preferred embodiment, the present invention provides a method for preparing an N-linked glycosylated glycopeptide, which method comprises: providing an isolated and purified glycopeptide having attached thereto a basic trimannose core or a Man3GlcNAc4 structure, contacting the glycopeptide with a glycosyltransferase and a donor molecule having a glycosyl moiety under conditions suitable for transfer of the glycosyl moiety into the glycopeptide. The basic trimannose core or Man3GlcNAc4 structure is then customized to produce a peptide with the desired glycosylation pattern, which can be achieved by sequential addition of the desired sugar moieties using techniques well known in the art.
Determination of glycan primary Structure
When an N-linked glycopeptide is produced by a cell, as noted elsewhere, it may comprise a heterogeneous mixture of glycan structures that must be reduced to a common and common basic trimannose core or Man3GlcNAc4 structure before additional sugar moieties are added thereto. In order to determine exactly which saccharide should be removed from a particular glycan structure, it is sometimes necessary to identify the primary glycan structure. Techniques for determining the primary Structure of glycans are well known in the art and are described in detail, for example, in Montreuil, "Structure and Biosynthesis of Polysaccharides," Polysaccharides in Medicinal Applications, pp.273-327, 1996, eds. Severian Damitriu, Marcel Dekker, N.Y.. It is therefore a simple matter for a person skilled in the art of glycobiology to isolate the peptide population produced by the cell and to determine the glycan structures attached thereto. For example, there are available effective methods for the following: (i) cleavage of glycosidic bonds by chemical cleavage such as hydrolysis, acetic anhydride decomposition, hydrazinolysis or deamination by nitrous acid; (ii) complete methylation followed by hydrolysis or methanolysis and then gas-liquid chromatography and mass spectrometry analysis of partially methylated monosaccharides; and (iii) the determination of anomeric bonds between monosaccharides by exoglycosidases, which also provides insight into the structure of primary glycans by sequential degradation. In particular, mass spectrometry and Nuclear Magnetic Resonance (NMR) spectrometry, in particular high field NMR, can be successfully used to determine glycan primary structures.
Kits and devices for sugar analysis are also commercially available. Fluorophore-assisted saccharide electrophoresis () Commercially available from Glyko, Inc (Novato, CA). In the FACE assay, glycoconjugates are released from peptides with endo-H or N-glycanases against N-linked glycans (PNGase F) or hydrazine against Ser/Thr linked glycans. The glycans are then labeled with fluorophores at the reducing end in a non-structurally differentiated manner. Fluorophore-labeled glycans were then separated on polyacrylamide gels based on the charge/mass ratio of the saccharide and the hydrodynamic volume. Gel images were taken under UV light and the composition of the glycans was determined by migration distance compared to standards. Oligosaccharides can be sequenced in this way by analyzing the migratory changes due to the sequential removal of sugars by digestion with exoglycosidases.
Exemplary embodiments
Reconstitution of N-linked glycosylation is best illustrated with reference to formula 1 below
Wherein X3、X4、X5、X6、X7And X17Is a (independently selected) monosaccharide or oligosaccharide residue; and is
a. b, c, d, e and x are (independently selected) 0, 1 or 2, provided that at least one member selected from a, b, c, d, e and x is 1 or 2.
In a preferred embodiment, the glycan structure is reconfigured such that the structure depicted in formula 1 has a specific determinant. The structure of the glycans can be selected to enhance the biological activity of the peptide, to confer new biological activity to the peptide, to remove the biological activity of the peptide or to better approximate (aproximate) the glycosylation pattern of the native peptide, among others.
In a first preferred embodiment, the peptide N-linked glycans are reconfigured to better approximate the glycosylation pattern of native human proteins. In this embodiment, the glycan structures described in formula 1 were reconstructed to have the following moieties:
X3and X5=|-GlcNAc-Gal-SA;
a and c are 1;
d is 0 or 1;
b. e and x are 0.
This embodiment is particularly advantageous for human peptides expressed in heterologous cell expression systems. By reconfiguring the N-linked glycan structure to this configuration, the peptide can be less immunogenic and/or more stable in human patients, as well as having other properties.
In a second preferred embodiment, the peptide N-linked glycans are reconfigured to have bisecting GlcNAc on the trimannose core. In this embodiment, the glycan structures described in formula 1 were reconstructed to have the following moieties:
X3And X5Is | -GlcNAc-Gal-SA;
a and c are 1;
X4is GlcNAc;
b=1;
d is 0 or 1;
e and x are 0.
This embodiment is particularly advantageous for recombinant antibodies expressed in heterologous cell systems. When antibody molecules include Fc-mediated cytotoxicity, it is well known that the presence of aliquots of oligosaccharides linked to the Fc domain dramatically increases antibody-dependent cellular cytotoxicity.
In a third preferred embodiment, the peptide N-linked glycans are reconstituted to have sialylated Lewis X moieties. In this embodiment, the glycan structures described in formula 1 were reconstructed to have the following moieties:
X3and X5Is that
a. c and d are 1;
b. e and x are 0;
X6is fucose.
This embodiment is particularly advantageous when the peptide to be reconstituted is intended to be directed to a selectin molecule and a cell displaying the same.
In a fourth preferred embodiment, the peptide N-linked glycans are reconstituted to have conjugated moieties. The conjugated moiety can be a PEG molecule, another peptide, a small molecule such as a drug, among others. In this embodiment, the glycan structures described in formula 1 were reconstructed to have the following moieties:
X3And X5Is | -GlcNAc-Gal-SA-R;
a and c are 1 or 2;
d is 0 or 1;
b. d, e and x are 0.
Wherein R ═ conjugate group.
The conjugated moiety can be a PEG molecule, another peptide, a small molecule such as a drug, among others. This embodiment can therefore be used to conjugate a peptide to a PEG molecule that slows the clearance of the peptide from the patient's bloodstream, to a peptide that directs both peptides into a particular tissue or cell, or to another peptide with a complementary therapeutic application.
It will be apparent to those skilled in the art that the present invention is not limited to the preferred glycan molecules described above. Preferred embodiments are only a few of the many useful glycan molecules that can be made using the reconstitution methods of the present invention. Those skilled in the art know how to design other useful glycans.
In a first exemplary embodiment, the peptide is expressed in CHO (chinese hamster ovary cells) according to methods well known in the art. When a peptide with an N-linked glycan consensus site is expressed and secreted in CHO cells, the N-linked glycan will have the structure depicted in the top row of fig. 2 but also contains core fucose. Although all of these structures may be present, the most common structures to date are the two on the right. In the term of the formula 1,
X3And X5Is | -GlcNAc-Gal- (SA);
a and c are 1;
b. e and x are 0, and
d is 0 or 1.
Thus, in an exemplary embodiment, N-linked glycans of a peptide expressed in CHO cells are reconstituted into preferred humanized glycans by contacting the peptide with a glycosyltransferase specific for a galactose acceptor molecule and a sialic acid donor molecule. This process is illustrated in figure 2 and example 17. In another exemplary embodiment, the N-linked glycans of peptides expressed and secreted in CHO cells are reconstituted as preferred pegylated structures. The peptide is first contacted with a glycosidase specific for sialic acid to remove the terminal SA moiety, and then contacted with a glycosyltransferase specific for the galactose acceptor moiety and the sialic acid acceptor moiety in the presence of a PEG-sialic acid-nucleotide donor molecule. Alternatively, the peptide may then be contacted with a glycosyltransferase specific for both the galactose acceptor moiety and the sialic acid acceptor moiety in the presence of the sialic acid-nucleotide donor molecule to ensure that SA capping of all glycan molecules is complete.
In another exemplary embodiment, the peptide is expressed in insect cells according to methods well known in the art, such as the sf9 cell line. When peptides with N-linked glycan consensus sites are expressed and secreted in sf9 cells, the N-linked glycans often have the structures depicted in the top row of fig. 6. In the term of formula 1:
X3And X5Is | -GlcNAc;
a and c are 0 or 1;
b=0;
X6is a fucose;
d is 0, 1 or 2; and is
e and x are 0.
The trimannose core is present in most N-linked glycans produced by insect cells, and sometimes also antennary GlcNAc and/or fucose residues. Note that the glycan may not have core fucose, it may have mononuclear fucose of any one bond, or it may have mononuclear fucose of one bond predominance. In an exemplary embodiment, the N-linked glycans of peptides expressed and secreted in insect cells are reconstituted into preferably humanized glycans by: contacting glycans with a glycosidase specific for fucose and then with a glycosyltransferase specific for a mannose acceptor molecule, a GlcNAc donor molecule, on each antenna of the trimannose core in the presence of a nucleotide-GlcNAc molecule; (ii) then contacting the glycans with a glycosyltransferase specific for a GlcNAc acceptor molecule, a Gal donor molecule, in the presence of a nucleotide-Gal molecule; the glycans are then contacted with a glycosyltransferase specific for the galactose acceptor molecule, the sialic acid donor molecule, in the presence of the nucleotide-SA molecule. One skilled in the art will appreciate that if some fucose molecules are present, they can be removed at any time in the process, and that if the core fucose has the same α 1, 6 linkages as found in human glycans, it can be left intact. In another exemplary embodiment, the humanized glycans of the previous examples are further reconstituted into sialylated Lewis X glycans by further contacting the glycans with a glycosyltransferase specific for a GlcNAc acceptor molecule, a fucose donor molecule, in the presence of a nucleotide-fucose molecule. This process is illustrated in figure 11 and example 39.
In another exemplary embodiment, the peptide is expressed in yeast according to methods well known in the art, such as Saccharomyces cerevisiae. When a peptide having an N-linked glycan consensus site is expressed and secreted in s.cerevisiae cells, the N-linked glycan will have the structure depicted on the left in FIG. 4. N-linked glycans will often have a trimannose core, which will typically be specified by mannose or related polysaccharides of up to 1000 residues. In the term of formula 1:
X3and X5Is | -Man-Man- (Man)0-1000;
a and c are 1 or 2;
b. d, e and x are 0.
In an exemplary embodiment, the N-linked glycans of the peptides expressed and secreted in the yeast cells are reconstituted into a basic trimannose core by: the glycans are first contacted with a glycosidase specific for the α 2 mannose molecule and then with a glycosidase specific for the α 6 mannose molecule. This process is illustrated in figure 4 and example 38.
In another exemplary embodiment, the N-linked glycans are further reconstituted to produce glycans suitable for use with recombinant antibodies having Fc-mediated cytotoxic function by: contacting the essential trimannose decorin with a glycosyltransferase in the presence of a nucleotide-GlcNAc molecule, wherein the glycosyltransferase is specific for a mannose acceptor molecule, a GlcNAc donor molecule, on each antenna of the trimannose core. Then contacting the glycans with a glycosyltransferase specific for an acceptor mannose molecule, a GlcNAc donor molecule, in the middle of the trimannose core in the presence of a nucleotide-GlcNAc molecule, and further contacting the glycans with a glycosyltransferase specific for a GlcNAc acceptor molecule, a Gal donor molecule in the presence of a nucleotide-Gal molecule; the glycan is then optionally contacted with a glycosyltransferase specific for the galactose acceptor molecule, and further optionally for the sialic acid donor molecule, in the presence of the nucleotide-SA molecule. This process is illustrated in fig. 1, 2 and 3.
In another exemplary embodiment, the peptide is expressed in bacteria, particularly in e. When a peptide having an N-linked glycan consensus site is expressed in E.coli cells, the N-linked consensus site will not be glycosylated. In an exemplary embodiment, the humanized glycan molecule is constructed from a peptide backbone by: contacting the peptide with a glycosyltransferase specific for the N-linked consensus site and the GlcNAc donor molecule in the presence of the nucleotide-GlcNAc; and further sequentially contacting the growing glycan with a glycosyltransferase specific for the acceptor and donor moieties in the presence of the necessary donor moieties until the desired glycan structure is completed. When peptides with N-linked glycans are expressed in eukaryotic cells, but do not have an appropriate leader peptide that directs nascent peptides into the golgi apparatus, the mature peptide may not be glycosylated. Also in this case, the peptide can be glycosylated by constructing an N-linked glycosylation from the consensus site of the peptide N-linkage as described above. When the protein is chemically modified with a sugar moiety, it can be constructed as described above.
These examples are intended to illustrate the invention and are not intended to be limiting. Those skilled in the art will appreciate that the steps performed in each example can, in some cases, be performed in a different order to achieve the same results. One skilled in the art will also appreciate that different steps can also produce the same glycans. Preferably the reconstituted glycans are never specific for the expression system expressing the peptide. The reconstituted glycans are illustrative only, and one skilled in the art would know how to take the principles from these examples and apply them to peptides produced in different expression systems to make glycans not specifically described herein.
B. Method for reconstituting O-linked glycans
O-glycosylation is characterized by the attachment of various monosaccharides to a hydroxyamino acid by an O-glycosidic bond. O-glycosylation is a common post-translational modification in the animal and plant kingdom. The structural complexity of protein O-linked glycans far exceeds N-linked glycans. The serine or threonine residues of the newly translated peptide are essentially modified by peptidyl GalNAc transferase in the medial to lateral compartment of the golgi apparatus. The site of O-glycosylation is not only determined by the sequence specificity of glycosyltransferases, but also by competition between different substrate sites and by competition-mediated epigenetic regulation (epigenetic regulation) with other glycosyltransferases responsible for glycan formation.
O-linked glycans have been artificially defined as having 3 regions: a core, a backbone, and a peripheral region. The "core" region of the O-linked glycan is the innermost 2 or 3 sugars of the glycan chain closest to the peptide. The backbone region is composed mainly of the length of the glycan chain formed by uniform extension. The peripheral region exhibits a high degree of structural complexity. The structural complexity of O-linked glycans begins with the core structure. In most cases, the first sugar residue added at the consensus site of the O-linked glycan is GalNAc; however, the sugar may also be GlcNAc, glucose, mannose, galactose, fucose or the like. FIG. 12 is a diagram of some known O-linked glycan core structures and enzymes responsible for their synthesis in vivo.
In mammalian cells, at least 8 different O-linked core structures are found, all based on core- α -GalNAc residues. The 4 core architecture depicted in fig. 13 is the most common. Core 1 and core 2 are the most abundant structures in mammalian cells, while core 3 and core 4 are found in more restricted and organ-characteristic expression systems. Reviews of O-linked glycans are available from Montreuil, Structure and Synthesis of Polysaccharides, Polysaccharides in Medicinal Applications, pp.273-327, 1996, eds. Severian Damitriu, Marcel Dekker, NY, and Schachter and Brockhausen, The Biosynthesis of branched O-LinkedGlycans, 1989, Society for Experimental Biology, pp.1-26 (UK).
It will be apparent from the disclosure of the present invention that the glycan structure of the O-glycosylated peptide can be reconstructed using similar techniques as described for the N-linked glycans. O-glycans differ from N-glycans in that they are attached to serine or threonine instead of asparagine residues. As described herein with respect to N-glycan reconstitution, hydrolytic enzymes can be used to cleave the unwanted sugar moieties on the O-linked glycans, and additional desired sugars can then be added thereto, thereby building customized O-glycan structures on the peptides (see fig. 12 and 13).
The initial step in O-glycosylation in mammalian cells is the attachment of N-acetylgalactosamine (GalNAc) using any one of at least 11 known families of α -N-acetylgalactosamine transferase, each of which has strict receptor peptide specificity. Typically, the receptor peptide recognized by each enzyme constitutes a sequence of at least 10 amino acids. Peptides containing the amino acid sequence recognized by a particular GalNAc-transferase become O-glycosylated if they are expressed in cells expressing the enzyme and if they are located approximately on the Golgi apparatus where UDP-GalNAc is also present.
However, in the case of recombinant proteins, initial attachment of GalNAc may not occur. The native alpha-N-acetylgalactosamine transferase of the expressing cell can have a consistent sequence specificity that is different from that of the recombinant peptide expressed.
The desired recombinant peptide can be expressed in bacterial cells that do not synthesize glycan chains, such as E.coli. In these cases, it is advantageous to add the initial GalNAc moiety in vitro. Once the recombinant peptide is recovered in a soluble form, the GalNAc moiety can be introduced onto the peptide in vitro by contacting the peptide with an appropriate GalNAc transferase in the presence of UDP-GalNAc.
In one embodiment, there may be additional amino acid sequences that constitute efficient receptors for the transfer of O-linked sugars. Such amino acid sequences are encoded by DNA sequences fused in frame to the coding sequence for the peptide, or alternatively may be introduced chemically. The peptide may additionally lack glycan chains. Alternatively, the peptide may have N-and/or O-linked glycan chains, but require additional glycosylation sites, such as when additional glycan substituents are required.
In an exemplary embodiment, the amino acid sequence PTTTK-COOH, which is the native GalNAc receptor sequence in the human mucin MUC-1, is added as a fusion tag. The fusion protein was then expressed in E.coli and purified. The peptide is then contacted with recombinant human GalNAc-transferase T3 or T6 in the presence of UDP-GalNAc to transfer GalNAc residues into the peptide in vitro.
The glycan chain on the peptide can then be further extended using the methods described herein for N-linked or O-linked glycans. Alternatively, the GalNAc transferase reaction can be carried out in the presence of UDP-GalNAc, where PEG covalently replaces the O-3, 4 or 6 position or the N-2 position. Glycoconjugation is described in detail elsewhere. Any antigenicity introduced by the novel peptide sequence may conveniently be masked by pegylation of the relevant glycan. Receptor site fusion techniques can be used to introduce not only PEG moieties, but also other glycan and non-glycan moieties, including but not limited to toxins, anti-infectives, cytotoxic agents, chelators of radionucleotides, and glycans with other functions such as tissue targeting.
Exemplary embodiments
The remodeling of O-linked glycosylation is best illustrated by the following formula 2:
In a preferred embodiment, the glycan structure is reconfigured so that the structure depicted in formula 2 has specific portions. The structure of the glycans can be selected to enhance the biological activity of the peptide, to confer a new biological activity to the peptide, to remove or alter the biological activity of the peptide or to better approximate the glycosylation pattern of the native peptide, among others.
In a first preferred embodiment, the peptide O-linked glycans are reconfigured to better approximate the glycosylation pattern of native human proteins. In this embodiment, the glycan structures described in formula 2 were reconstructed to have the following moieties:
X2is | -SA; or | -SA;
f and n are 0 or 1;
X10is SA;
m=0。
this embodiment is particularly advantageous for human peptides expressed in heterologous cell expression systems. By reconfiguring the O-linked glycan structure to have this configuration, the peptide can be made less immunogenic and/or more stable in human patients.
In another preferred embodiment, the peptide O-linked glycans are reconstituted to display sialylated Lewis X antigens. In this embodiment, the glycan structures described in formula 2 were reconstructed to have the following moieties:
X2Is | -SA;
X10is Fuc or | -glcnac (Fuc) -Gal-SA;
f and n are 1
m=0。
This embodiment is particularly advantageous when the peptide to be reconstituted is most effective in targeting to a selectin molecule and cells displaying the same.
In another preferred embodiment, the peptide O-linked glycans are reconstituted to have conjugated moieties. The conjugated moiety can be a PEG molecule, another peptide, a small molecule such as a drug, among others. In this embodiment, the glycan structures described in formula 2 were reconstructed to have the following moieties:
X2is | -SA-R;
f=1;
n and m are 0.
Wherein R ═ conjugate group.
This embodiment can be used to conjugate a peptide to a PEG molecule that slows clearance of the peptide from the patient's bloodstream, to a peptide that directs both peptides into a particular tissue or cell, or to another peptide with a complementary therapeutic application.
It will be apparent to those skilled in the art that the present invention is not limited to the preferred glycan molecules described above. Preferred embodiments are only a few of the many useful glycan molecules that can be made using the reconstitution methods of the present invention. One skilled in the art, once reading the present invention, would know how to design other useful glycans.
In a first exemplary embodiment, the peptide is expressed in CHO (chinese hamster ovary cells) according to methods well known in the art. When peptides with O-linked glycan consensus sites are expressed and secreted in CHO cells, most of the O-linked glycans will generally have the following structure, i.e., in the terminology of formula 2,
X2is | -SA;
f=1;
m and n are 0.
Therefore, glycans in most CHO cells do not require reconstitution to be suitable for use in human patients. In an exemplary embodiment, the O-linked glycans of peptides expressed and secreted in CHO cells are reconstituted to contain sialylated Lewis X structures by contacting the glycans with a glycosyltransferase specific for GalNAc acceptor moieties and fucose donor moieties in the presence of nucleotide-fucose. This process is illustrated in figure 11 and the N-linked glycans of example 39.
In another exemplary embodiment, the peptide is a line expressed in insect cells such as sf9 according to methods well known in the art. When a peptide having an O-linked glycan consensus site is expressed and secreted in most sf9 cells, the majority of the O-linked glycans have a structure of formula 2, wherein:
X2=H;
f is 0 or 1;
n and m are 0.
See, e.g., Marchal et al, (2001, biol. chem.382: 151-. In an exemplary embodiment, O-linked glycans to peptides expressed in insect cells are reconstituted as humanized glycans by: first contacting glycans with a glycosyltransferase specific for a GlcNAc acceptor molecule and a galactose donor molecule in the presence of nucleotide-Gal; the glycans are then contacted with a glycosyltransferase specific for the Gal acceptor molecule and the SA donor molecule in the presence of nucleotide-SA. In another exemplary embodiment, the O-linked glycans are further reconstituted from a humanized form into a sialylated Lewis X form by contacting the glycans with a glycosyltransferase in the presence of nucleotide-fucose, wherein the glycosyltransferase is specific for a GalNAc acceptor molecule and a fucose donor molecule.
In another exemplary embodiment, the peptide is expressed in fungi, particularly in s.cerevisiae cells, according to methods well known in the art. When a peptide having an O-linked glycan consensus site is expressed and secreted in s.cerevisiae cells, most of the O-linked glycans have the following structure:
|-AA-Man-Man1-2.
See Gemmill and Trimble (1999, Biochim. Biophys. acta 1426: 227-237). To reconstitute these O-linked glycans for use in humans, it is preferred that the glycans be cleaved at the amino acid level and reconstituted therefrom.
In an exemplary embodiment, the glycans are O-linked glycans of peptides expressed in the fungal cell and are reconstituted as humanized glycans by: contacting the glycan with an endoglycosylase specific for an amino acid-GalNAc bond; then contacting the glycan with a glycosyltransferase specific for the O-linked consensus site and the GalNAc donor molecule in the presence of the nucleotide-GalNAc; then contacting the glycan with a glycosyltransferase specific for the GalNAc acceptor molecule and the galactose donor molecule in the presence of the nucleotide-Gal; the glycans are then contacted with a glycosyltransferase specific for the Gal acceptor molecule and the SA donor molecule in the presence of nucleotide-SA.
Alternatively, in another exemplary embodiment, the glycans are O-linked glycans of peptides expressed in the fungal cell and are reconstituted into humanized glycans by: contacting glycans with the protein O-mannose beta-1, 2-N-acetylglucosaminyltransferase (POMGnTI) in the presence of GlcNac-nucleotides; then contacting the glycan with a galactosyltransferase in the presence of nucleotide-Gal; the glycans are then contacted with sialyltransferase in the presence of nucleotide-SA.
In another exemplary embodiment, the peptide is expressed in bacteria, particularly in E.coli cells, according to methods well known in the art. When a peptide having an O-linked glycan consensus site is expressed in E.coli cells, the O-linked consensus site will not be glycosylated. In this case, the desired glycan molecule must be constructed from the peptide backbone in a manner similar to that described above for s.cerevisiae expression. Further, when peptides with O-linked glycans are expressed in eukaryotic cells, but do not have an appropriate leader peptide that directs nascent peptides into the golgi apparatus, the mature peptide may not be glycosylated. Also in this case, O-linked glycosyl structures can be added to the peptide by constructing glycans directly from the peptide O-linked consensus sites. Further, when the protein is chemically modified with a sugar moiety, it can also be reconstituted as described above.
These examples are intended to illustrate the invention and are in no way limiting. Those skilled in the art will appreciate that the steps performed in each example can, in some cases, be performed in a different order to achieve the same results. One skilled in the art will also appreciate that different steps can also produce the same glycans. Further, it is preferred that the reconstituted glycans are never specific for the expression system expressing the peptide. The reconstituted glycans are illustrative only, and one skilled in the art would know how to take the principles from these examples and apply them to peptides produced in different expression systems to make glycans not specifically described herein.
C. Glycoconjugation, described generally
The present invention provides methods of making conjugates of glycosylated or non-glycosylated peptides. The conjugates of the invention are formed between a peptide and a different species such as a water-soluble polymer, a therapeutic moiety, a diagnostic moiety, a targeting moiety, and the like. Also provided are conjugates comprising two or more peptides linked together by a linker arm, i.e., multifunctional conjugates. The multifunctional conjugates of the present invention may comprise two or more copies of the same peptide or a collection of different peptides having different structures and/or properties.
The conjugates of the invention may also be formed by enzymatic attachment of a modified sugar to a glycosylated or non-glycosylated peptide. When inserted between the peptide and the modifying group on the sugar, the modified sugar becomes what is referred to herein as an "intact glycosyl linking group". The methods of the invention provide peptides having a desired group at one or more specific positions, taking advantage of the precise selectivity of enzymes such as glycosyltransferases. Thus, according to the present invention, the modified sugar is attached directly to a selected position on the peptide chain, or alternatively, the modified sugar is appended to the sugar moiety of the peptide. Peptides in which the modified sugar is linked to both the sugar of the peptide and directly to the amino acid residue of the peptide backbone are also within the scope of the invention.
Contrary to known chemical and enzymatic peptide processing strategies, the method of the invention makes it possible to assemble peptides and glycopeptides having a substantially homogeneous derivatization pattern; the enzymes used in the present invention are typically selective for a particular amino acid residue or combination of amino acid residues on a peptide or a particular glycan structure. This method is also feasible for large scale production of modified peptides and glycopeptides. Thus, the method of the present invention provides a practical method for large scale production of peptides having a preselected substantially uniform derivatization pattern. The method is particularly suited for the modification of therapeutic peptides, including but not limited to peptides that are not fully glycosylated during production in cell culture cells (e.g., mammalian cells, insect cells, fungal cells, plant cells, yeast cells, or prokaryotic cells) or transgenic plants or animal cells.
The methods of the invention also provide glycosylated and unglycosylated peptide conjugates having increased half-life due to, for example, reduced clearance rates or reduced uptake rates by the immune or reticuloendothelial system (RES). In addition, the methods of the present invention provide a means of masking antigenic determinants on a peptide, thereby reducing or eliminating the host immune response to the peptide. Selective attachment of targeting agents can also be used to target peptides to specific tissue or cell surface receptors specific for that particular targeting agent. In addition, a class of peptides specifically modified with a therapeutic moiety is provided.
1. Conjugates
In a first aspect, the invention provides a conjugate between a peptide and a selected moiety. The linkage between the peptide and the selected moiety includes an intact glycosyl linking group interposed between the peptide and the selected moiety. As discussed herein, the selected moiety is essentially any species that can be attached to a saccharide unit, resulting in a "modified saccharide" that can be recognized by an appropriate transferase, which attaches the modified saccharide to the peptide. The sugar component of the modified sugar becomes an "intact glycosyl linking group" when inserted between the peptide and the selected moiety. The glycosyl linking group is formed from any mono-or oligosaccharide which, after modification with the selected moiety, is the substrate of the appropriate transferase.
The conjugates of the invention will generally correspond to the following general structure:
wherein the symbols a, b, c, d and s represent positive integers other than 0; and t is 0 or a positive integer. An "agent" is a therapeutic agent, a bioactive agent, a detectable label, a water soluble moiety, and the like. The "agent" may be a peptide, such as an enzyme, antibody, antigen, or the like. The linker may be any of a wide variety of linking groups hereinafter. Alternatively, the linker may be a single bond or a "zero order linker". The characteristics of the peptide are not limited. Exemplary peptides are provided in fig. 28.
In an exemplary embodiment, the selected moiety is a water-soluble polymer. The water-soluble polymer is covalently attached to the peptide through an intact glycosyl linking group. The glycosyl linking group is covalently attached to an amino acid residue or a glycosyl residue of the peptide. Alternatively, the glycosyl linking group is attached to one or more glycosyl units of the glycopeptide. The invention also provides conjugates in which the glycosyl linking group is attached to both the amino acid residue and the glycosyl residue.
In addition to providing conjugates formed by enzymatically added intact glycosyl linking groups, the present invention provides conjugates whose substitution patterns are highly homogeneous. Using the methods of the invention, it is possible to form peptide conjugates in which substantially all of the modified sugar moieties in the population of conjugates of the invention are attached to multiple copies of structurally identical amino acids or sugar residues. Thus, in a second aspect, the present invention provides a peptide conjugate having a population of water-soluble polymer moieties covalently linked to a peptide through an intact glycosyl linking group. In preferred conjugates of the invention, substantially each member of the population is linked to a glycosyl residue of the peptide via a glycosyl linking group, and each glycosyl residue of the peptide to which the glycosyl linking group is attached has the same structure.
Also provided are peptide conjugates having thereon a population of water-soluble polymer moieties covalently linked by an intact glycosyl linking group. In a preferred embodiment, substantially each member of the population of water-soluble polymer moieties is linked to an amino acid residue of the peptide via an intact glycosyl linking group, and each amino acid residue having an intact glycosyl linking group attached thereto has the same structure.
The present invention also provides conjugates similar to those described above, wherein the peptide is conjugated to the therapeutic moiety, diagnostic moiety, targeting moiety, toxin moiety, etc. via an intact glycosyl linking group. Each of the above moieties may be a small molecule, a natural polymer (e.g., a peptide), or a synthetic polymer.
In an exemplary embodiment, interleukin-2 (IL-2) is conjugated to transferrin via a bifunctional linker that includes an intact glycosyl linking group at each end of the PEG moiety (scheme 1). For example, one end of the PEG linker is functionalized with an intact sialic acid linker attached to transferrin, while the other end is functionalized with an intact GalNAc linker attached to IL-2.
In another exemplary embodiment, EPO is conjugated to transferrin. In another exemplary embodiment, EPO is conjugated to glial-derived neurotrophic growth factor (GDNF). In these embodiments, each conjugation is achieved by the aforementioned bifunctional linker comprising an intact glycosyl linking group at each end of the PEG moiety. Transferrin transport proteins pass through the blood-brain barrier.
As set forth in the figures, the conjugates of the invention can include a mono-or multivalent (e.g., antennary structure) intact glycosyl linking group, see fig. 14-22. The conjugates of the invention also include a glycosyl linking group derived from an O-linked glycan of serine or threonine (FIG. 11). Thus, the conjugates of the present invention include two classes in which selected moieties are attached to a peptide via a monovalent glycosyl linking group. Also included in the invention are conjugates in which more than one selected moiety is attached to the peptide via a multivalent linking group. One or more proteins may be conjugated together to take advantage of their biophysical and biological properties.
In yet a further embodiment, the invention provides conjugates that selectively localize in a particular tissue due to the presence of a targeting agent that is a component of the conjugate. In an exemplary embodiment, the targeting agent is a protein. Exemplary proteins include transferrin (brain, blood pool), Human Serum (HS) -glycoprotein (bone, brain, blood pool), antibodies (brain, tissue with antibody-specific antigens, blood pool), coagulation factor V-XII (damaged tissue, clot, cancer, blood pool), serum proteins such as alpha-acid glycoprotein, fetuin, alpha-fetal protein (brain, blood pool), beta 2-glycoprotein (liver, atherosclerotic plaque, brain, blood pool), G-CSF, GM-CSF, M-CSF and EPO (immune stimulation, cancer, blood pool, red blood cell overproduction, neuroprotection) and albumin (increase in half-life).
In addition to the conjugates discussed above, the present invention provides methods of preparing these and other conjugates. Thus, in a further aspect, the invention provides a method of forming a covalent conjugate between a selected moiety and a peptide. In addition, the invention provides methods of directing the conjugates of the invention to specific tissues or regions of the body.
In exemplary embodiments, the conjugate is formed between a water-soluble polymer, therapeutic moiety, targeting moiety or biomolecule and a glycosylated or non-glycosylated peptide. The polymer, therapeutic moiety, or biomolecule is conjugated to the peptide through an intact glycosyl linking group interposed between and covalently attached to both the peptide and a modifying group (e.g., a water-soluble polymer). The method comprises contacting the peptide with a mixture comprising a modified sugar and a glycosyltransferase that has the modified sugar as a substrate. The reaction is carried out under conditions sufficient to form a covalent bond between the modified sugar and the peptide. The sugar moiety of the modified sugar is preferably selected from the group consisting of nucleotide sugars, activated sugars and sugars that are neither nucleotide sugars nor activated sugars.
In one embodiment, the present invention provides a method of linking two or more peptides through a linking group. The linking group has any useful structure and can be selected from linear or branched structures. Preferably, each end of the linker attached to the peptide includes a modified sugar (i.e., a nascent intact glycosyl linking group).
In an exemplary method of the invention, two peptides are linked together by a linker moiety comprising a PEG linker. This construct is consistent with the general structure set forth in the figures above. As described herein, the constructs of the invention include two intact glycosyl linking groups (i.e., s + t ═ 1). The emphasis on the description of a PEG linker comprising two glycosyl groups is for the sake of brevity and should not be construed as limiting the features of the linker arm used in this embodiment of the invention.
Thus, the PEG moiety is functionalized with a first glycosyl unit at a first end and a second glycosyl unit at a second end. The first and second glycosyl units are preferably substrates for different transferases, such that the first peptide and the second peptide can be attached at right angles to the first and second peptide, respectively. In practice, let (glycosyl)1-PEG- (glycosyl)2The linker is contacted with a first peptide and a first transferase having a first glycosyl unit as substrate, thereby forming a (peptide)1- (sugar base)1-PEG- (glycosyl)2. The first transferase and/or unreacted peptide can then optionally be removed from the reaction mixture. Adding a second peptide and a second transferase having a second glycosyl unit as substrate to the mixture 1- (sugar base)1-PEG- (glycosyl)2In a conjugate, thereby forming a (peptide)1- (sugar base)1-PEG- (glycosyl)2- (peptide)2. One skilled in the art will appreciate that the above methods may also be applied to form conjugates between more than two peptides by using, for example, branched PEG, dendrimers, poly (amino acids), polysaccharides, and the like.
As previously described, in one exemplary embodiment, interleukin-2 (IL-2) is conjugated to transferrin via a bifunctional linker that includes an intact glycosyl linking group at each terminus of the PEG moiety (scheme 1). The IL-2 conjugates have an increased half-life in vivo over IL-2 itself, taking advantage of the larger molecular size of the conjugate. In addition, conjugation of IL-2 to transferrin selectively directs the conjugate to the brain. For example, one end of the PEG linker is functionalized with CMP-sialic acid, and the other is functionalized with UDP-GalNAc. The linker is allowed to combine with IL-2 in the presence of GalNAc transferase, resulting in attachment of GalNAc on the linker arm to a serine and/or threonine residue on IL-2.
In another exemplary embodiment, transferrin is conjugated to nucleic acids for gene therapy
The above process can be performed in multiple cycles and is not limited to the formation of conjugates between two peptides with a single linker. Furthermore, one skilled in the art will appreciate that the reaction to functionalize the intact glycosyl linking group located at the end of the PEG (or other) linker of the peptide can occur simultaneously in the same reaction vessel, or can be performed in a stepwise manner. When the reaction is carried out in a stepwise manner, the conjugate produced in each step is optionally purified from one or more reaction components (e.g., enzyme, peptide).
A further exemplary embodiment is set forth in scheme 2. Scheme 2 shows a method for preparing conjugates that direct a selected protein, such as EPO, to the bone and increase the circulating half-life of the selected protein.
It is within the scope of the invention to attach one or more peptide moieties to the linker using a reactive derivative of PEG (or other linker). The present invention is not limited by the characteristics of the reactive PEG analogs. Many activated derivatives of poly (ethylene glycol) are commercially and publicly available. It is also within the ability of the skilled person to select and, if desired, synthesize suitable PEG derivatives with which substrates for use in the present invention can be prepared. See Abuchowski et al, Cancer biochem. biophysis, 7: 175-186 (1984); abuchowski et al, j.biol.chem., 252: 3582-; jackson et al, anal. biochem., 165: 114-127 (1987); koide et al, biochem. biophysis. res. commun., 111: 659-: 56-69 (1984); delgado et al, biotechnol.appl.biochem, 12: 119-; n-hydroxysuccinimide-derived active esters (Buckmann et al, Makromol. chem., 182: 1379-1384 (1981); Joppich et al, Makromol. chem., 180: 1381-1384 (1979); Absuchowski et al, Cancer biochem. Biophys., 7: 175-186 (1984); Katre et al, Proc. Natl. Acad. Sci.U.S.A., 84: 1487-propan 1491 (1987); Kitamura et al, Cancer Res., 51: 4310-propan 4315 (1991); Boccu et al, Z.Nature formsch., 38C: 94-99(1983)), carbonates (Zaldiphy et al, Poly (ethylene) Chemistry, 38C: 94-99, 1983), pyridine (Biophyr et al, Biophysomnic., 19811, Biophyr. 164141, Biophysomnic., 1992., 99, Biophysomnic), Biophysomnic, 99, and 29-imidazole, 99, 23-imidazole, Biophysomnic, Biophyk et al, 1984, Biophysomnic, 1984, Biophym et al, Biophysomnic, Biophym et al, Biophym, 1984, Biophysomnic, 1992 20, 1984, 2000, Biophysomnic, 1992, 2000, Bio, bioconjugugat chem, 4: 314, 318(1993)), isocyanates (Byun et al, ASAIO Journal, M649-M-653(1992)) and epoxides (U.S. Pat. No.4,806,595 to Noishiki et al (1989)). Other linking groups include a urethane linkage between the amino group and the activated PEG. See Veronese et al, appl.biochem.biotechnol, 11: 141-152(1985).
In another exemplary embodiment utilizing reactive PEG derivatives, the present invention provides a method of extending the blood circulation half-life of a selected peptide and substantially directing the peptide to the blood pool by conjugating the peptide to a synthetic or natural polymer of sufficient size to impede the filtration of proteins (such as albumin) by the glomeruli. An embodiment of the invention is illustrated in scheme 3, wherein Erythropoietin (EPO) is conjugated to albumin via a PEG linker using chemical and enzymatic modification.
Thus, as shown in scheme 3, the amino acid residues of albumin are modified with reactive PEG derivatives, such as X-PEG- (CMP-sialic acid)), where X is an activating group (e.g., active ester, isothiocyanate, etc.). The PEG derivative and EPO were combined and contacted with a transferase with CMP-sialic acid as substrate. In a further illustrative embodiment, the epsilon amino group of lysine is reacted with the N-hydroxysuccinimide ester of the PEG-linker to form an albumin conjugate. The CMP-sialic acid of the linker is enzymatically conjugated to an appropriate residue on EPO, such as Gal, to form a conjugate. The skilled person will appreciate that the above methods are not limited to the reactants presented. In addition, the method may actually form conjugates comprising more than two proteins using, for example, branched linkers having more than two ends.
2. Modified sugar
The modified glycosyl donor species ("modified sugar") is preferably selected from the group consisting of modified sugar nucleotides, activated modified sugars, and sugars that are modified monosaccharides that are neither nucleotides nor activated. Any desired sugar structure can be added to the peptide using the methods of the invention. Typically, the structure is a monosaccharide, but the invention is not limited to the use of modified monosaccharides; oligo-and polysaccharides may also be used.
The modified group is attached to the sugar moiety by enzymatic means, chemical means, or a combination thereof, thereby producing a modified sugar. The sugar is substituted at any position that enables attachment of the modified moiety and this still enables the sugar to serve as a substrate for the enzyme used to attach the modified sugar to the peptide. In a preferred embodiment, when the sialic acid is the saccharide, the sialic acid is substituted with a modifying group at the 9-position on the Pyruvyl side chain or at the 5-position on an amine moiety, which is typically acetylated in sialic acid.
In certain embodiments of the invention, modified sugar nucleotides are used to add modified sugars to peptides. Exemplary sugar nucleotides for use in the invention in their modified form include mono-, di-or triphosphate nucleotides or analogs thereof. In a preferred embodiment, the modified sugar nucleotide is selected from the group consisting of UDP-glycoside, CMP-glycoside or GDP-glycoside. Even more preferably, the modified sugar nucleotide is selected from the group consisting of UDP-galactose, UDP-galactosamine, UDP-glucose, UDP-glucosamine, GDP-mannose, GDP-fucose, CMP-sialic acid or CMP-NeuAc. N-acetamide derivatives of sugar nucleotides are also useful in the methods of the invention.
The invention also provides methods of synthesizing modified peptides using modified sugars, such as modified galactose, modified fucose, and modified sialic acid. When modified sialic acids are used, sialyltransferases or trans-sialidases (directed only to α 2, 3-linked sialic acids) may be used in these methods.
In another embodiment, the modified sugar is an activated sugar. Activated modified sugars for use in the present invention are typically glycosides that have been synthetically altered to include an activated leaving group. As used herein, the term "activated leaving group" refers to those moieties that are readily displaced in an enzymatically mediated nucleophilic substitution reaction. Many activated sugars are well known in the art. See, e.g., Vocadlo et al, Carbohydrate Chemistry and Biology, Vol.2, Ernst et al, Wiley-VCH Verlag: weinheim, germany, 2000; kodama et al, tetrahedron Lett.34: 6419 (1993); loughed et al, j.biol.chem.274: 37717(1999)).
Examples of activating groups (leaving groups) include fluorine, chlorine, bromine, tosylate (tosylate), mesylate (mesylate), triflate and the like. Preferred activated leaving groups for use in the present invention are those that do not sterically hinder enzymatic transfer of the glycoside to the acceptor. Thus, preferred embodiments of activated glycoside derivatives include glycosyl fluorides and glycosyl mesylates, with glycosyl fluorides being particularly preferred. Among glycosyl fluorides, most preferred are α -galactosyl fluoride, α -mannosyl fluoride, α -glucosyl fluoride, α -fucosyl fluoride, α -xylosyl fluoride, α -sialic acid fluoride, α -N-acetylglucosamine fluoride, α -N-acetylgalactosamine fluoride, β -galactosyl fluoride, β -mannosyl fluoride, β -glucosyl fluoride, β -fucosyl fluoride, β -xylosyl fluoride, β -sialic acid fluoride, β -N-acetylglucosamine fluoride, β -N-acetylgalactosamine fluoride.
For example, glycosyl fluorides can be prepared from the free sugars by first acetylating the sugars and then treating them with HF/pyridine. This produces the thermodynamically most stable anomer of protected (acetylated) glycosyl fluoride (i.e., alpha glycosyl fluoride). If a less stable anomer (i.e., β -glycosyl fluoride) is desired, it can be prepared by converting the peracetylated saccharide to the anomeric bromide or chloride with HBr/HOAc or with HCl. The intermediate is reacted with a fluoride salt, such as silver fluoride, to form glycosyl fluoride. Acetylated glycosyl fluorides can be deprotected by reaction with a mild (catalytic) base such as NaOMe/MeOH in methanol. In addition, many glycosyl fluorides are commercially available.
Other activated glycosyl derivatives can be prepared by conventional methods well known to those skilled in the art. For example, glycosyl mesylates can be prepared by treatment of the fully benzylated hemiacetal form of the sugar with methanesulfonyl chloride followed by removal of the benzyl group by catalytic hydrogenation.
In a further exemplary embodiment, the modified sugar is an oligosaccharide having an antennary structure. In a preferred embodiment, one or more of the ends of the antenna has a modified portion. When one or more modifications are attached to an oligosaccharide having an antennary structure, the oligosaccharide may be used to "amplify" the modification; each oligosaccharide unit conjugated to a peptide attaches multiple copies of a modifying group to the peptide. The general structure of the chelates of the invention as set forth in the above figures comprises the multivalent species that result from the preparation of the conjugates of the invention with an antennal structure. Many antennary sugar structures are known in the art, and the methods of the invention can be practiced on them without limitation.
Exemplary modifying groups are discussed below. The modifying group may be selected based on one or more desired properties. Exemplary properties include, but are not limited to, enhanced pharmacokinetics, improved biodistribution, providing multivalent species, improved water solubility, enhanced or reduced lipophilicity, and tissue targeting.
D. Peptide conjugates
a) Water-soluble polymers
The hydrophilicity of the selected peptide is enhanced by conjugation to polar molecules such as molecules containing amines, esters, hydroxyl and polyhydroxy. Representative examples include, but are not limited to, polylysine, polyethyleneimine, poly (ethylene glycol), and poly (propylene glycol). Preferred water-soluble polymers are substantially non-fluorescent, or emit only a small amount of fluorescence, such that they are not suitable for use as fluorescent labels in assays. Polymers that are sugars not naturally occurring can be used. In addition, other naturally occurring sugars modified by covalent attachment of other entities (e.g., poly (ethylene glycol), poly (propylene glycol), poly (aspartic acid), biomolecules, therapeutic moieties, diagnostic moieties, etc.) can also be used. In another exemplary embodiment, the therapeutic saccharide moiety is conjugated to a linker arm, and the saccharide-linker arm is subsequently conjugated to the peptide by the methods of the invention.
Methods and chemicals for activating water soluble polymers and saccharides and methods for conjugating saccharides and polymers to various species are described in the literature. Commonly used methods for activating polymers include activation of functional groups with cyanogen bromide, periodate, glutaraldehyde, diepoxides (epoxides), epichlorohydrin, divinyl sulfone, carbodiimides, sulfonyl halides, trichlorotriazines, and the like (see R.F. Taylor, (1991), Protein immunization: Fundamentals and applications, Marcel Dekker, N.Y.; S.S.Wong, (1992) chemistry Protein Conjugation and Crosslinking, CRC Press, Boca Raton; G.T.Herson et al, (1993), Immobilized Affinity technologies, Academic Press, N.Y.; Dunn, R.L. et al, eds. polymeric Drug Delivery Systems, S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S. Press, S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.S.
Routes for preparing reactive PEG molecules and forming conjugates with reactive molecules are well known in the art. For example, U.S. Pat. No.5,672,662 discloses water-soluble and isolatable conjugates of active esters of polymeric acids selected from linear or branched polyalkylene oxides, poly (oxyethylated polyols), poly (enol), poly (propylenmorphol), wherein the polymer has about 44 or more repeating units.
U.S. Pat. No.6,376,604 proposes a method for preparing water-soluble 1-benzotriazolyl carbonates of water-soluble and non-peptidic polymers by reacting the terminal hydroxyl groups of the polymer with bis (1-benzotriazolyl) carbonate in an organic solvent. The active esters are useful for forming conjugates with biologically active agents such as proteins or peptides.
WO 99/45964 describes a conjugate comprising a biologically active agent and an activated water soluble polymer, the polymer comprising a polymeric backbone having at least one terminus attached to the polymeric backbone by a stabilizing bond, wherein at least one terminus comprises a branching moiety having a proximal reactive group attached to the branching moiety, wherein the biologically active agent is attached to the at least one proximal reactive group. Other branched poly (ethylene glycols) are described in WO 96/21469, and U.S. patent No.5,932,462 describes conjugates with branched PEG molecules that include a branched terminus that includes a reactive functional group. The free reactive groups are capable of reacting with a biologically active species, such as a protein or peptide, to form a conjugate between the poly (ethylene glycol) and the biologically active species. U.S. Pat. No.5,446,090 describes bifunctional PEG linkers and their use in forming conjugates having a peptide at the end of each PEG linker.
Conjugates comprising degradable PEG linkages are described in WO 99/34833 and WO99/14259 and in U.S. Pat. No.6,348,558. Such degradable linkages are useful in the present invention.
Although both reactive PEG derivatives and conjugates formed with such derivatives are well known in the art, until the present invention, it has not been recognized that conjugates can be formed between PEG (or other polymers) and other species such as peptides or glycopeptides via an intact glycosyl linking group.
Many water-soluble polymers are well known to those skilled in the art and can be used in the practice of the present invention. The term water-soluble polymer encompasses species such as sugars (e.g., dextran, amylose, hyaluronic acid, poly (sialic acid), heparinoids, heparin, etc.); poly (amino acids), such as poly (glutamic acid); a nucleic acid; synthetic polymers (such as poly (acrylic acid)); poly (ethers) such as poly (ethylene glycol); a peptide; proteins, and the like. The invention can be carried out with any water-soluble polymer, the only limitation being that the polymer must include sites to which the remainder of the conjugate can be attached.
Methods for activating polymers are also found in WO 94/17039, U.S. Pat. No.5,324,844, WO 94/18247, WO 94/04193, U.S. Pat. No.5,219,564, U.S. Pat. No.5,122,614, WO 90/13540, U.S. Pat. No.5,281,698 and more in WO 93/15189, and also methods directed to conjugates between activated polymers and peptides, such as coagulation factor VIII (WO 94/15625), hemoglobin (WO94/09027), oxygen-carrying molecules (U.S. Pat. No.4,412,989), ribonucleases and superoxide dismutase (Veronese et al, appl.biochem. Appth.11: 141: 145 (1985)).
Preferred water-soluble polymers are those in which a substantial proportion of the polymer molecules in a polymer sample have about the same molecular weight; such polymers are "monodisperse".
The invention is further illustrated with poly (ethylene glycol) conjugates. Several reviews and monographs on the functionalization and conjugation of PEG are available. See, e.g., Harris, macronol, chem, phys, c 25: 325-373 (1985); scouten, Methods in Enzymology 135: 30-65 (1987); wong et al, Enzyme micro b.technol.14: 866-874 (1992); delgado et al, clinical Reviews in Therapeutic drug carriers Systems 9: 249-304 (1992); zalipsky, Bioconjugate chem.6: 150-165 (1995); and bhandra et al, Pharmazie, 57: 5-29(2002).
Suitable poly (ethylene glycols) for use in the present invention include, but are not limited to, those described by formula 3 below:
R ═ H, alkyl, benzyl, aryl, acetal, OHC-, H2N-CH2CH2-、HS-CH2CH2-、-sugar-nucleotides, proteins, methyl, ethyl;
x, Y, W, U (independently selected) ═ O, S, NH, H — R';
r ', R' "(independently selected) ═ alkyl, benzyl, aryl, alkylaryl, pyridyl, substituted aryl, arylalkyl, acylalkyl;
n=1~2000;
m, q, and p (independently selected) are 0-20;
o=0~20;
Z=HO、NH2halogen, S-R' ", activated esters,
-sugar-nucleotides, proteins, imidazoles, HOBT, tetrazoles, halides; and
V=HO、NH2halogen, S-R' ", activated esters, activated amides, -sugar-nucleotides, proteins.
In a preferred embodiment, the poly (ethylene glycol) molecule is selected from the following:
the poly (ethylene glycol) used to form the conjugates of the invention is linear or branched. Branched poly (ethylene glycols) suitable for use in the present invention include, but are not limited to, those described by the following formula:
R ', R ", R'" (independently selected) ═ H, alkyl, benzyl, aryl, acetal, OHC-, H2N-CH2CH2-、HS-CH2CH2-、-(CH2)qCY-Z, -sugar-nucleotides, proteins, methyl, ethyl, heteroaryl, acylalkyl, acylaryl, acylalkylaryl;
x, Y, W, A, B (independently selected) ═ O, S, NH, H-R', (CH)2)1;
n and p (independently selected) are 1-2000;
m, q, and o (independently selected) are 0-20;
Z=HO、NH2halogen, S-R' ", activated esters,
-sugar-nucleotides, proteins;
V=HO、NH2halogen, S-R' ", activated esters, activated amides, -sugar-nucleotides, proteins.
The in vivo half-life, area under the curve, and/or retention time of the therapeutic agent may also be enhanced with water soluble polymers such as polyethylene glycol (PEG) and polypropylene glycol (PPG). For example, chemical modification of proteins with PEG (pegylation) increases their molecular size and decreases their surface accessibility and functional group accessibility, both depending on the size of the PEG attached to the protein. This results in improved plasma half-life and proteolytic stability and reduced immunogenicity and liver uptake (Chaffee et al, J.Clin.invest.89: 1643-1651 (1992); Pyratak et al, Res.Commun.chem.Pathol Pharmacol.29: 113-127 (1980)). It has been reported that PEGylation of interleukin-2 increases the antitumor ability in vivo (Katre et al, Proc. Natl. Acad. Sci. USA.84: 1487-one 1491(1987)), while F (ab') derived from monoclonal antibody A7) 2PEGylation of (A) can improve tumor localization (Kitamura et al, biochem. Biophys. Res. Commun.28: 1387-1394 (1990)).
In a preferred embodiment, the in vivo half-life of a peptide derivatized with a water-soluble polymer by the method of the invention is increased relative to the in vivo half-life of a non-derivatized peptide. In another preferred embodiment, the area under the curve of a peptide derivatized with a water-soluble polymer by the method of the invention is increased relative to the area under the curve of a non-derivatized peptide. In another preferred embodiment, the retention time of a peptide derivatized with a water-soluble polymer by the method of the invention is increased relative to the retention time of a non-derivatized peptide. Techniques for determining in vivo half-life, area under the curve, and retention time are well known in the art. This technique is described in j.g. wagner, 1993, pharmacopoetics for the pharmaceutical Scientist, technical Publishing Company, inc.
The increase in the in vivo half-life of the peptide is best expressed as a percentage increase in the amount. The lower end of the range of this percentage increase is about 40%, about 60%, about 80%, about 100%, about 150%, or about 200%. The upper end of the range is about 60%, about 80%, about 100%, about 150%, or more than about 250%.
In an exemplary embodiment, the present invention provides pegylated follicle stimulating hormone (examples 23 and 24). In a further preferred embodiment, the present invention provides pegylated transferrin (example 42).
Other examples of water-soluble polymers useful in the present invention include, but are not limited to, linear or branched polyalkylene oxides, (polyoxyethylated polyols), poly (enol), poly (propyleneomorph), dextran, starch, poly (amino acids), and the like.
b) Water-insoluble polymer
The conjugates of the invention may also include one or more water-insoluble polymers. Embodiments of the invention are illustrated by the use of conjugates as vehicles for the delivery of therapeutic peptides in a controlled manner. Polymeric drug delivery systems are well known in the art. See, e.g., Dunn et al, eds. polymeric Drugs and Drug Delivery Systems, ACS symposium series Vol.469, American Chemistry Society, Washington, D.C. 1991. Those skilled in the art will appreciate that essentially any known drug delivery system can be applied to the conjugates of the invention.
Representative water insoluble polymers include, but are not limited to, polyphosphazine, polyvinyl alcohol, polyamides, polycarbonates, polyalkylenes, polyacrylamides, polyalkylene glycols, polyalkylene oxides, polyalkylene terephthalates, polyvinyl ethers, polyvinyl esters, polyvinyl halides, polyvinylpyrrolidone, polyglycolides, polysiloxanes, polyurethanes, polymethyl methacrylates, polyethyl methacrylates, polybutyl methacrylates, polyisobutyl methacrylates, polyhexamethyl methacrylates, polyisodecyl methacrylates, polylauryl methacrylates, polyphenyl methacrylates, polymethyl acrylates, isopropyl polyacrylates, polyisobutyl acrylates, stearyl polyacrylates, polyethylenes, polypropylenes, polyethylene glycols, polyethylene oxides, polyethylene terephthalates, polyvinyl acetates, polyvinyl chlorides, polyvinyl alcohol, polyvinyl glycolides, polyvinyl alcohol, Polystyrene, polyvinylpyrrolidone, pluronics and polyvinylphenol and copolymers thereof.
Synthetically modified natural polymers useful in the conjugates of the invention include, but are not limited to, alkyl celluloses, hydroxyalkyl celluloses, cellulose ethers, cellulose esters, and nitrocellulose. Particularly preferred members of the broad class of synthetically modified natural polymers include, but are not limited to, methyl cellulose, ethyl cellulose, hydroxypropyl methyl cellulose, hydroxybutyl methyl cellulose, cellulose acetate, cellulose propionate, cellulose acetate butyrate, cellulose acetate phthalate, carboxymethyl cellulose, cellulose triacetate, cellulose sulfate sodium salt, and polymers of acrylic and methacrylic esters and alginic acid.
These and other polymers discussed herein are readily commercially available from commercial sources such as Sigma chemical Co. (St. Louis, Mo.), Polysci ends (Warrenton, PA.), Aldrich (Milwaukee, Wis.), Fluka (Ronkonkoma, NY) and BioRad (Richmond, CA), or can be synthesized using standard techniques from monomers obtained from these suppliers.
Representative biodegradable polymers for use in the conjugates of the present invention include, but are not limited to, polylactides, polyglycolides and copolymers thereof, polyethylene terephthalate, polybutanoic acid, polypentanoic acid, poly (lactide-co-caprolactone), polyanhydrides, polyorthoesters, and mixtures and copolymers thereof. Of particular utility are gel-forming compositions such as those comprising collagen, pluronics, and the like.
The polymers useful in the present invention include "hybrid" polymers having a water-insoluble material with bioresorbable molecules in at least a portion of its structure. An example of such a polymer is as follows: comprising a water-insoluble copolymer having a bioresorbable region, a hydrophilic region, and each polymer chain having a plurality of crosslinkable functional groups.
For purposes of the present invention, "water-insoluble material" includes materials that are substantially insoluble in water or in an environment containing water. Thus, while certain regions or segments of the copolymer may be hydrophilic or even water-soluble, the polymer molecule as a whole may not be soluble to any substantially measurable degree in water.
For the purposes of the present invention, the term "bioresorbable material" includes regions capable of undergoing metabolism or breakdown and being resorbed and/or eliminated by the body through normal excretory pathways. Such metabolites or breakdown products are preferably substantially non-toxic to the body.
The bioresorbable region may be hydrophobic or hydrophilic so long as the polymer as a whole is not water soluble. Thus, the bioresorbable region is preferably selected based on the overall retention of water insolubility of the polymer. Thus, the relevant properties (i.e., the type of functional group contained in the bioresorbable region and the relative proportion of that region and the hydrophilic region) are selected to ensure that the useful bioresorbable composition remains water-insoluble.
Exemplary resorbable polymers include resorbable poly α -hydroxy-carboxylic acid/polyoxyalkylene block copolymers as synthetically produced (see Cohn et al, U.S. patent No.4,826,945). These copolymers are not crosslinked and are water soluble so that the body can excrete the degraded block copolymer composition. See Younes et al, j.biomed.mater.res.21: 1301 + 1316 (1987); and Cohn et al, j.biomed.mater.res.22: 993-1009(1988).
Presently preferred bioresorbable polymers include one or more components selected from the group consisting of polyesters, polyhydroxy acids, polylactones, polyamides, polyesteramides, polyaminoacids, polyanhydrides, polyorthoesters, polycarbonates, polyphosphazines, polyphosphoesters, polythioesters, polysaccharides, and mixtures thereof. The bioresorbable polymer more preferably includes a polyhydroxy acid component. Polyhydroxy acids, polylactic acid, polyglycolic acid, polycaproic acid, polybutanoic acid, polyvaleric acid, and copolymers and mixtures thereof are preferred.
In addition to forming fragments that are absorbed in the body ("bioresorbable"), preferred polymeric coatings for use in the methods of the present invention may also form excretable and/or metabolizable fragments.
Higher order copolymers may also be used in the present invention. For example, U.S. Pat. No.4,438,253 issued to Casey et al at 20.3.1984 discloses tri-block copolymers produced from the transesterification of polyglycolic acid and a hydroxyl-terminated polyalkylene glycol. Such compositions are disclosed for use as resorbable monofilament sutures. The flexibility of such compositions is controlled by incorporating aromatic orthocarbonates, such as tetra-p-tolyl orthocarbonate, into the copolymer structure.
Other coatings based on lactic acid and/or glycolic acid may also be used. For example, U.S. patent No.5,202,413 issued by Spinu on 4.13.1993 discloses biodegradable multiblock copolymers with sequential order of polylactide and/or polyglycolide blocks produced by ring opening polymerization of lactide and/or glycolide on oligomeric diols or diamines followed by chain extension with bifunctional compounds such as diisocyanates, diacylchlorides or dichlorosilanes.
The bioresorbable region of the coating for use in the present invention may be designed to be hydrolytically and/or enzymatically cleavable. For the purposes of the present invention, "water-cleavable" refers to the susceptibility of the copolymer, and in particular the bioresorbable region, to hydrolysis in water or in an aqueous environment. Similarly, "enzymatically cleavable" is used herein to refer to the susceptibility of a copolymer, particularly a bioresorbable region, to cleavage by an endogenous or exogenous enzyme.
The hydrophilic region may be processed into excretable and/or metabolizable fragments when in the body. Thus, the hydrophilic region can include, for example, polyethers, polyalkylene oxides, polyols, poly (vinylpyrrolidine), polyvinyl alcohols, poly (alkyloxazolines), polysaccharides, sugars, peptides, proteins, and copolymers and mixtures thereof. Furthermore, the hydrophilic region can also be, for example, a polyalkylene oxide. Such polyalkylene oxides may include, for example, polyethylene oxide, polypropylene oxide, and mixtures and copolymers thereof.
Polymers that are components of hydrogels may also be used in the present invention. Hydrogels are polymeric materials that are capable of absorbing relatively large amounts of water. Examples of hydrogel-forming compounds include, but are not limited to, polyacrylic acid, sodium carboxymethylcellulose, polyvinyl alcohol, polyvinyl pyrrolidine, gelatin, carrageenan and other polysaccharides, hydroxyethylidene methacrylic acid (HEMA), and derivatives thereof, and the like. Stable, biodegradable and bioresorbable hydrogels can be produced. In addition, the hydrogel composition may include subunits that exhibit one or more of these properties.
Biocompatible hydrogel compositions whose integrity can be controlled by crosslinking are well known and are presently preferred for use in the methods of the present invention. For example, U.S. Pat. No.5,410,016 issued at 25.4.1995 and 5,529,914 issued at 25.6.1996 to Hubbell et al disclose water-soluble systems that are cross-linked block copolymers with a water-soluble central block segment sandwiched between two hydrolytically unstable extensions. This copolymer is further end-capped with photopolymerizable acrylate functionality. When crosslinked, these systems become hydrogels. The water-soluble center block of such copolymers may include polyethylene glycol; however, the hydrolytically unstable extensions may be poly-alpha-hydroxy acids, such as polyglycolic acid or polylactic acid. See, Sawhney et al, Macromolecules 26: 581-587(1993).
In another preferred embodiment, the gel is a thermoreversible gel. Thermally reversible gels including, for example, pluronics, collagen, gelatin, hyaluronic acid, polysaccharides, polyurethane hydrogels, polyurethane-urea hydrogels, and combinations thereof are currently preferred.
In another exemplary embodiment, the conjugates of the invention comprise a liposomal composition. Liposomes can be prepared according to methods well known to those skilled in the art, as described in U.S. Pat. No.4,522,811 to Eppstein et al, 6/11 1985. For example, liposomes can be prepared by dissolving the appropriate lipid (e.g., stearoyl phosphatidylethanolamine, stearoyl phosphatidylcholine, arachidoyl phosphatidylcholine, and cholesterol) in an inorganic solvent and then evaporating, leaving a thin layer of dried lipid on the surface of the container. An aqueous solution of the active compound or a pharmaceutically acceptable salt thereof is then introduced into the container. The container is then rotated by hand to release the lipid material from the sides of the container and disperse the lipid aggregates, thereby forming a liposome suspension.
The microparticles described above and the methods of making the microparticles are provided by way of illustration and are not intended to limit the scope of microparticles useful in the present invention. It will be apparent to those skilled in the art that a range of microparticles made by different methods can be used in the present invention.
c) Biological molecules
In another preferred embodiment, the modified sugar has a biomolecule. In yet further preferred embodiments, the biomolecule is a functional protein, enzyme, antigen, antibody, peptide, nucleic acid (such as single or nucleoside, oligonucleotide, polynucleotide and single and more than one strand nucleic acid), lectin, receptor or a combination thereof.
Some preferred biomolecules are substantially non-fluorescent, or emit only minimal fluorescence, such that they are not suitable for use as fluorescent labels in assays. Other biomolecules may be fluorescent. It is appropriate to use other naturally occurring sugars that are modified by covalent attachment of another entity (e.g., PEG, biomolecules, therapeutic moieties, diagnostic moieties, etc.). In an exemplary embodiment, a saccharide moiety as a biomolecule is conjugated to a linker arm, and the saccharide-linker arm cassette is subsequently conjugated to a peptide by the methods of the invention.
Biomolecules used in the practice of the present invention may be derived from any source. The biomolecules may be isolated from natural sources or may be produced by synthetic methods. The peptide may be a native peptide or a mutated peptide. Mutagenesis may be accomplished by chemical mutagenesis, site-directed mutagenesis, or other methods of inducing mutations known to those skilled in the art. Peptides useful in the practice of the present invention include, for example, enzymes, antigens, antibodies, and receptors. The antibody may be polyclonal or monoclonal; whole or fragmented. The peptide may alternatively be the product of an directed evolution program.
Both naturally-derived and synthetic peptides and nucleic acids may be used in the present invention; these molecules can be attached to the glycosyl residue composition or crosslinker through any available reactive group. For example, peptides may be attached through reactive amine, carboxyl, sulfhydryl, or hydroxyl groups. The reactive group may be located at the end of the peptide or at an internal site in the peptide chain. Nucleic acids can be attached through reactive groups on the base (e.g., exocyclic amines) or available hydroxyl groups on the sugar moiety (e.g., 3 '-or 5' -hydroxyl groups). Peptide and nucleic acid strands may be further derivatized at one or more sites to enable attachment of appropriate reactive groups to the strands. See Chrisey et al, Nucleic acids sRs.24: 3031-3039(1996).
In a further preferred embodiment, the biomolecule is selected to target a peptide modified by the methods of the invention to a specific tissue, thereby enhancing delivery of the peptide to the tissue relative to the amount of underivatized peptide delivered to the tissue. In yet a further preferred embodiment, the amount of derivatized peptide delivered to a particular tissue over a selected period of time is enhanced by derivatization by at least about 20%, more preferably at least about 40%, and even more preferably at least about 100%. Presently, preferred biomolecules for targeted applications include antibodies, hormones, and ligands for cell surface receptors. Exemplary targeting biomolecules include, but are not limited to, antibodies specific for transferrin receptors used to deliver the molecule to the brain (Penichet et al, 1999, J.Immunol.163: 4421-.
In a presently preferred embodiment, the modifying group is a protein. In an exemplary embodiment, the protein is an interferon. The interferon is an antiviral glycoprotein that is secreted in humans by human primary fibroblasts following induction with viruses or double-stranded RNA. Interferons are useful as therapeutic agents, such as antiviral agents and in the treatment of multiple sclerosis. For references discussing interferon- β, see, e.g., Yu et al, j.neuro imitunal, 64 (1): 91-100 (1996); schmidt, j.neurosci.res., 65 (1): 59-67 (2001); wender et al, Folia neuropathohol, 39 (2): 91-93 (2001); martin et al, Springer seminin. 1-24 (1996); takane et al, j.pharmacol.exp.ther., 294 (2): 746-752 (2000); sburlati et al, biotechnol.prog., 14: 189-; dodd et al, biochimicet Biophysica Acta, 787: 183-187 (1984); edelbaum et al, j. interferon res, 12: 449-453 (1992); conradt et al, j.biol.chem., 262 (30): 14600-14605 (1987); civas et al, eur.j.biochem., 173: 311-316 (1988); demoder et al, j.biotechnol, 32: 179-189 (1994); sedmak et al, j. interferon res, 9(Suppl 1): S61-S65 (1989); kagawa et al, j.biol.chem., 263 (33): 17508-17515 (1988); hershenson et al, U.S. Pat. No.4,894,330; jaya ram et al, j. interferon res, 3 (2): 177-180 (1983); menge et al, develop.biol.standard, 66: 391-401 (1987); von et al, j. interferon res, 3 (2): 169-175 (1983); and Adolf et al, j. interferon res, 10: 255-267(1990). For reference to interferon- α, see Asano et al, eur.j. cancer, 27(Suppl 4): S21-S25 (1991); nagy et al, anticancer research, 8 (3): 467-; dron et al, j.biol.regul.homeost.Agents, 3 (1): 13-19 (1989); habib et al, am. Surg., 67 (3): 257-260 (3/2001); and sugyimaa et al, eur.j. biochem., 217: 921-927(1993).
In an exemplary interferon conjugate, interferon beta is conjugated to a second peptide through a linker arm. The linker arm includes an intact glycosyl linking group through which the linker arm is attached to the second peptide by the methods of the invention. The linker arm may also optionally include a second intact glycosyl linking group through which the interferon may be attached.
In another exemplary embodiment, the present invention provides conjugates of Follicle Stimulating Hormone (FSH). FSH is a glycoprotein hormone. See, e.g., Saneyoshi et al, biol. reprod., 65: 1686-1690 (2001); hakola et al, j.endocrinol, 158: 441-448 (1998); stanton et al, mol, cell, endocrinol, 125: 133-141 (1996); walton et al, j.clin.endocrinol.meta.86 (8): 3675-3685 (08/2001); Ulloa-Aguirre et al, Endocrine, 11 (3): 205-215 (12/1999); Castro-Fern ndez et al, i.j.clin.endocrinol.meta, 85 (12): 4603-4610 (2000); prevost, Rebecca r., Pharmacotherapy, 18 (5): 1001-1010 (1998); linskens et al, The FASEBJournal, 13: 639-645 (04/1999); butnev et al, biol. reprod., 58: 458-469 (1998); muyan et al, mol. endo, 12 (5): 766 772 (1998); min et al, endo.j., 43 (5): 585 and 593 (1996); bo ime et al, Recentrepressure in Hormoneresearch, 34: 271-289 (1999); and Rafferty et al, j.endo, 145: 527-533(1995). FSH conjugates may be formed in a similar manner to that described for interferons.
In another exemplary embodiment, the conjugate comprises Erythropoietin (EPO). EPO is known to mediate the hypoxic response and stimulate the production of red blood cells. For relevant references, see Cerami et al, Semins in Oncology, 28(2) (Suppl 8): 66-70(04/2001). Exemplary conjugates of EPO are formed in a manner similar to conjugates of interferon.
In a further exemplary embodiment, the present invention provides conjugates of human granulocyte colony-stimulating factor (G-CSF). G-CSF is a glycoprotein that stimulates the proliferation, differentiation and activation of a neurogenic (neurogenic) progenitor cell to form a functionally mature neutrophil. It is known that injected G-CSF will be cleared from the body very quickly. See, e.g., Nohynek et al, cancer chemither, pharmacol, 39: 259-266 (1997); lord et al, clinical cancer Research, 7 (7): 2085-2090 (07/2001); rotondaro et al, Molecular Biotechnology, 11 (2): 117-Et al, Bone Marrow translation, 28: 259-264(2001). Exemplary G-CSF conjugates are prepared as described above for conjugates of interferon. One skilled in the art will appreciate that many other proteins can be conjugated to interferon using the methods and compositions of the present invention, including, but not limited to, the peptides listed in tables 7 and 8 (provided elsewhere) and fig. 28 and fig. 29-57, where separate modification schemes are provided.
In yet further exemplary embodiments, conjugates with biotin are provided. Thus, for example, a selectively biotinylated peptide can be made by attaching avidin or streptavidin having one or more modifying groups.
In a further preferred embodiment, the biomolecule is selected to direct a peptide modified by the method of the invention to a specific intracellular compartment, thereby enhancing delivery of the peptide to the intracellular compartment relative to the amount of underivatized peptide delivered to the tissue. In yet a further preferred embodiment, the amount of derivatized peptide delivered to a particular intracellular compartment over a selected period of time is enhanced by derivatization by at least about 20%, more preferably at least about 40%, and even more preferably at least about 100%. In another particularly preferred embodiment, the biomolecule is linked to the peptide via a cleavable linker which, once internalized, is hydrolysable. Currently, preferred biomolecules for intracellular targeting applications include transferrin, lactotransferrin (lactoferrin), melanoma transferrin (melanotropin) (p97), ceruloplasmin and divalent cation transporters, as well as antibodies against specific vascular targets. Contemplated linkages include, but are not limited to, protein-sugar-linker-sugar-protein, protein-sugar-linker-protein and multivalent forms thereof, and protein-sugar-linker-drug, wherein the drug includes small molecules, peptides, lipids, and the like.
Site-specific and target-directed delivery of therapeutic agents is desirable for the purpose of treating a variety of human diseases, such as various types of malignancies and certain neurological disorders. This procedure is accompanied by lower side effects and higher efficacy of the drug. Various principles are relied upon in designing these delivery systems. For a review see Garnett, Advanced drug delivery re views 53: 171-216(2001).
An important consideration in designing drug delivery systems is target tissue specificity. The discovery of tumor surface antigens makes it possible to develop therapeutic methods in which tumor cells displaying defined surface antigens are specifically targeted and killed. There are mainly 3 classes of therapeutic monoclonal antibodies (antibodies) that have proven effective in human clinical trials for the treatment of malignancies: (1) unconjugated MAbs that can directly induce growth inhibition and/or apoptosis, or indirectly activate host defense mechanisms to mediate antitumor cytotoxicity; (2) drug-conjugated mabs that preferentially deliver potent cytotoxins into tumor cells thus minimizing systemic cytotoxicity commonly associated with conventional therapies; and (3) radioisotope conjugated MAbs, which deliver a sterile dose of radiation into the tumor. See Reff et al, cancer control 9: 152, 166 (2002).
To confer the ability to kill malignant cells, the MAb may be linked to a toxin, which may be obtained from a plant, bacterial or fungal source, to form a chimeric protein known as an immunotoxin. Commonly used phytotoxins fall into two categories: (1) holotoxins (or class II ribosome inactivating proteins), such as ricin, abrin, mistletoe lectin and modecin, and (2) hemitoxins (class I ribosome inactivating proteins), such as Pokeweed Antiviral Protein (PAP), saporin, bryodin, bouganin and gelonin. Commonly used bacterial toxins include Diphtheria Toxin (DT) and Pseudomonas Exotoxin (PE). Kreitman, Current pharmaceutical Biotechnology 2: 313-325(2001). Other toxins contemplated for use in the present invention include, but are not limited to, the toxins shown in table 2.
TABLE 2 toxins.
Conventional immunotoxins contain mabs chemically conjugated to the toxin, which has been mutated or chemically modified to minimize binding to normal cells. Examples include anti-B4-blocked ricin targeting CD5 and RFB 4-deglycosylated ricin A chain targeting CD 22. Recently developed recombinant immunotoxins are chimeric proteins consisting of the variable regions of antibodies against tumor antigens fused to a protein toxin using recombinant DNA technology. The toxin is also often genetically engineered to remove binding sites of normal tissue but retain its cytotoxicity. A number of differentiation antigens, overexpressed receptors or cancer-specific antigens have been identified as targets for immunotoxins, such as CD19, CD22, CD20, IL-2 receptor (CD25), CD33, IL-4 receptor, EGF receptor and mutants thereof, Er β 2, Lewis sugars, mesothelin, transferrin receptor, GM-CSF receptor, Ras, Bcr-Ab1 and c-Kit, for the treatment of various malignancies, including hematopoietic cell carcinomas, gliomas and carcinomas, colon, uterus, bladder and gastrointestinal cancers. See, e.g., Brinkmann et al, expertopin, biol, ther, 1: 693-702 (2001); perentesis and Sievers, Hematology/Oncology Clinics of North America 15: 677-701(2001).
MAbs conjugated with radioisotopes are used as an alternative method of treating human malignancies, particularly hematopoietic malignancies, with a high level of specificity and efficacy. The most commonly used isotopes for therapy are high energy emitters, such as131I and90and Y. In the recent past, it has been desired to develop,213bi-labeled anti-CD 33 humanized MAbs have also been tested in phase I human clinical trials. Reff et al, supra.
Many mabs have been used for therapeutic purposes. For example, the recombinant chimeric anti-CD 20MAb, rituximab (Rituxan), is used to treat certain hematopoietic malignanciesTM) Has been approved by the FDA in 1997. From then onOther mabs that have been approved for the treatment of human cancer include: humanized rat anti-CD 52 antibody alemtuzumab (Campath-1H)TM) (ii) a And a calicheamicin conjugated humanized mouse anti-CD 33 MAb gemtuzumab ozogamicin (Mylotarg)TM). The FDA has also currently examined the safety and efficacy of several other mabs for the purpose of site-specific delivery of cytotoxic agents or radiation, such as radiolabeled ZevalinTMAnd BexxarTM. Reff et al, supra.
A second important consideration in designing drug delivery systems is the accessibility of the target tissue to the therapeutic agent. This is a particularly contemplated situation in the treatment of diseases of the Central Nervous System (CNS), since the blood-brain barrier prevents the diffusion of macromolecules. Several approaches have been developed to effectively deliver therapeutic agents to the CNS by bypassing the blood-brain barrier.
Understanding the iron transport mechanism from plasma to brain provides a useful tool to circumvent the blood-brain barrier (BBB). The iron transported in plasma by transferrin is an essential component of almost all types of cells. The brain requires iron for metabolic processes and accepts iron via receptor-mediated transcytosis and endocytosis via transferrin receptors located on brain capillary endothelial cells. Moos and Morgan, Cellular and Molecular Neurobiology 20: 77-95(2000). Transferrin-transferrin receptor interaction based delivery systems have been established to efficiently deliver peptides, proteins and liposomes into the brain. For example, peptides can be coupled to anti-transferrin receptor mabs to achieve greater brain uptake, Moos and Morgan, supra. Similarly, the transport of basic fibroblast growth factor (bFGF) across the blood-brain barrier is enhanced when coupled to an anti-transferrin receptor MAb. Song et al, The Journal of pharmacy and Experimental Therapeutics 301: 605- > 610 (2002); wu et al, Journal of Drug Targeting 10: 239-245(2002). In addition, a liposomal delivery system has been reported to effectively transport the chemotherapeutic drug doxorubicin into the C6 glioma, where transferrin is attached to the distal end on the liposomal PEG chain. Eavarone et al, J.biomed.Mater.Res.51: 10-14(2000). Many U.S. patents also relate to delivery methods based on transferrin-transferrin receptor interactions that bypass the blood-brain barrier. See, e.g., U.S. patent nos. 5,154,924, 5,182,107, 5,527,527, 5,833,988, 6,015,555.
There are other suitable conjugate partners for pharmaceutical agents that bypass the blood-brain barrier. For example, U.S. patent nos. 5,672,683, 5,977,307 and WO95/02421 are directed to methods of delivering a neuropharmaceutical agent across the blood-brain barrier, wherein the agent is administered as a fusion protein with a ligand that reacts with a capillary endothelial cell receptor of the brain; WO99/00150 describes a drug delivery system in which transport of the drug across the blood-brain barrier is facilitated by conjugation to mabs to the human insulin receptor; WO 89/10134 describes a chimeric protein comprising a peptide capable of crossing the blood-brain barrier at a relatively high rate and a hydrophilic neuropeptide incapable of transcytosis as a means of introducing the hydrophilic neuropeptide into the brain; WO 01/60411 provides a pharmaceutical composition which can easily transport a pharmaceutically active ingredient into the brain. The active ingredient is bound to a hibernating-specific protein that acts as a conjugate and is administered with a thyroid hormone or a substance that promotes thyroid hormone production. In addition, alternative drug delivery routes for bypassing the blood-brain barrier have been explored. For example, intranasal administration of therapeutic agents without conjugation has been shown to be a promising alternative Delivery method (Frey, 2002, Drug Delivery Technology, 2 (5): 46-49).
In addition to facilitating transport of drugs across the blood-brain barrier, transferrin-transferrin receptor interactions can also be used for specific targeting of certain tumor cells, since many tumor cells overexpress transferrin at their surface. This strategy has been used for The delivery of biologically active macromolecules into K562 cells by transferrin conjugates (Wellhoner et al, The Journal of biological Chemistry 266: 4309-.
Furthermore, as a result of the increased understanding of the function of iron transport proteins and their expression patterns, such as the lactoferrin receptor, melanoma transferrin, ceruloplasmin and divalent cation transporters, it has been found that some proteins involved in the iron transport mechanism (such as melanoma transferrin) or fragments thereof have similar efficiency in assisting the transport of therapeutic agents across the blood-brain barrier or targeting to specific tissues (WO 02/13843 a2, WO 02/13873 a 2). For a review of the use of transferrin and related proteins as conjugates in drug delivery, which are involved in iron uptake, see Li and Qian, Medical Research Reviews 22: 225-250(2002).
The concept of tissue-specific delivery of therapeutic agents is not limited to interactions between transferrin and transferrin receptor or related proteins thereof. For example, bone-specific delivery systems have been described in which proteins are conjugated to osteogenic amino diphosphates to improve delivery of the proteins to mineralized tissues. Uludag and Yang, Biotechnol.prog.18: 604-611(2002). For a review of this subject, see Vyas et al, clinical Reviews in therapeutic Drug Carrier System 18: 1-76(2001).
Various linkers can be used in methods of generating bioconjugates for the purpose of specific delivery of therapeutic agents. Suitable linkers include homobifunctional and heterobifunctional crosslinking reagents that are cleavable or non-cleavable by cleavage, e.g., catalyzed by an acid (see, e.g., Srinivasachar and Neville, Biochemistry 28: 2501-2509 (1989); Wellhoner et al, The Journal of Biological Chemistry 266: 4309-4314 (1991)). Any known interaction between binding partners such as biotin and avidin/streptavidin may also be used in the method of binding the therapeutic agent to the conjugate partner, thereby ensuring specific and efficient delivery of the therapeutic agent. Using the methods of the invention, proteins can be used to deliver molecules and conjugates to intracellular compartments. Proteins, peptides, hormones, cytokines, small molecules, etc., that bind to specific cell surface receptors that are internalized upon ligand binding can be used for intracellular targeting of conjugated therapeutic compounds. Generally, the receptor-ligand complex will internalize into an intracellular vesicle delivered into a particular cellular compartment, including but not limited to the nucleus, mitochondria, golgi, ER, lysosome, and endosome, depending on the intracellular location targeted by the receptor. By conjugating the receptor ligand to the desired molecule, the drug can be carried in the receptor-ligand complex and delivered to the intracellular compartment to which the receptor is normally directed. Thus, the drug may be delivered to a specific intracellular location of the cell where it is needed to treat the disease.
Many proteins are useful for targeting therapeutic agents to specific tissues and organs. Targeting proteins include, but are not limited to, growth factors (EPO, HGH, EGF, nerve growth factor, FGF, etc.), cytokines (GM-CSF, G-CSF, interferon family, interleukins, etc.), hormones (FSH, LH, steroid family, estrogens, corticosteroids, insulin, etc.), serum proteins (albumin, lipoproteins, fetoprotein, human serum proteins, antibodies, and antibody fragments, etc.), and vitamins (folic acid, vitamin C, vitamin A, etc.). Targeting agents specific for receptors on most cell types are commercially available.
Contemplated linkage configurations include, but are not limited to, protein-sugar-linker-sugar-protein and multivalent forms thereof, protein-sugar-linker-therapeutic agents, wherein the therapeutic agents include, but are not limited to, small molecules, peptides, and lipids. In some embodiments, a hydrolyzable linker is employed that hydrolyzes upon internalization. When the protein conjugate is internalized into an endosomal or lysosomal pair having an acidic pH, an acid-labile linker can be employed to advantage. Once internalized into the endosome or lysosome, the linker is hydrolyzed and the therapeutic agent is released from the targeting agent.
In an exemplary embodiment, transferrin is conjugated via a linker to an enzyme or nucleic acid vector encoding the enzyme intended to be directed into cells of a patient that display the transferrin receptor. The patient may require enzyme replacement therapy for that particular enzyme. In a particularly preferred embodiment, the enzyme is deficient in a patient with a lysosomal storage disease (see table 5). Once in circulation, transferrin-enzyme conjugates can attach to transferrin receptors and internalize in early endosomes (Xing et al, 1998, biochem. J.336: 667; Li et al, 2002, Trends in Pharmacol. Sci.23: 206; Suhaila et al, 1998, J.biol. chem.273: 14355). Other contemplated targeting agents related to transferrin include, but are not limited to, lactoferrin (lactoferrin), melanoma transferrin (p97), ceruloplasmin, and divalent cation transporters.
In another exemplary embodiment, the transferrin-dystrophin conjugate will enter the endosome through the transferrin pathway. Once in the endosome, dystrophin proteins are released due to the hydrolysable linker and can then be brought into the intracellular compartment where they are needed. This embodiment can be used to treat a patient suffering from muscular dystrophy by supplementing the genetically deficient dystrophin gene and/or protein with a functional dystrophin protein linked to transferrin.
E. Treatment section
In another preferred embodiment, the modified saccharide comprises a therapeutic moiety. One skilled in the art will appreciate that there is an overlap between the therapeutic moiety and the category of biomolecules; many biomolecules have therapeutic properties or potential.
Therapeutic moieties may be agents that have been accepted for clinical use, or they may be drugs in experiments, or their activity or mechanism of action is under investigation. The therapeutic moiety may have a proven effect in a given disease state, or may simply be assumed to exhibit a desired effect in a given disease state. In a preferred embodiment, the therapeutic moiety is a compound selected for its ability to interact with a selected tissue. The therapeutic moieties used in the practice of the present invention include drugs from a wide variety of drug classes with a variety of pharmacological activities. In some embodiments, it is preferred to use a non-sugar therapeutic moiety. One exception to this preference is the use of saccharides that are covalently attached to another entity, such as PEG, biomolecules, therapeutic moieties, diagnostic moieties, etc. In an exemplary embodiment, the antisense nucleic acid moiety is conjugated to a linker arm attached to the targeting moiety. In another exemplary embodiment, the therapeutic saccharide moiety is conjugated to a linker arm, and the saccharide-linker arm cassette is subsequently conjugated to the peptide by the methods of the invention.
Methods of conjugating therapeutic and diagnostic agents to various other species are well known to those skilled in the art. See, e.g., Hermanson, Bioconjugate Techniques, academic Press, San Diego, 1996; and Dunn et al, eds. polymeric Drugs and drug Delivery Systems, ACS Symposium Series Vol.469, American chemical Society, Washington, D.C. 1991.
In an exemplary embodiment, the therapeutic moiety is attached to the modified sugar through a bond that is cleavable under selected conditions. Exemplary conditions include, but are not limited to, a selected pH (e.g., stomach, intestine, endocytophaga), the presence of active enzymes (e.g., esterases, proteases, reductases, oxidases), light, heat, and the like. Many cleavable groups are known in the art. See, e.g., Jung et al, biochem. biophysis. acta, 761: 152-162 (1983); joshi et al, j.biol.chem.265: 14518-14525 (1990); zarling et al, J.Immunol., 124: 913-920 (1980); bouizar et al, eur.j. biochem., 155: 141-147 (1986); park et al, j.biol.chem.261: 205-210 (1986); browning et al, j.immunol., 143: 1859-1867(1989).
A useful class of therapeutic moieties include anti-inflammatory drugs such as non-steroids (NSAIDS). NSAIDS may be selected from the following classes: (e.g., propionic acid derivatives, acetic acid derivatives, fenamic acid derivatives, diphenylcarboxylic acid derivatives, and oxicams); steroidal anti-inflammatory drugs including hydrocortisone and the like; an adjuvant; antihistamine (such as chlorpyrifos, Triprolidine); antitussives (e.g., dextromethorphan, codeine, caramiphene, and pentoxyverine); anti-pruritus agents (such as methdilazine and alimemazine); anticholinergic agents (such as hyoscyamine, atropine, homatropine, levodopa); anti-emetic and anti-cachectic agents (such as cyclizine, meclizine, chlorpromazine, buclizine); anorexic drugs (e.g., benzphetamine, phentermine, p-chlorobenzene butyl amine, fenfluramine); central stimulant drugs (e.g., amphetamine, methamphetamine, dextroamphetamine, and methylphenidate); anti-dysrhythmic drugs (e.g., propranolol, procainamide, propiram, quinidine, emcamide); beta-adrenergic blocking drugs (e.g., metoprolol, acebutolol, betaxolol, labetalol, and timolol); cardiotonics (e.g., milrinone, amrinone, and dobutamine); antihypertensive agents (e.g., enalapril, cloidine, hydrazine trozine, minoxidil, guanadrel, guanethidine); diuretic drugs (such as amiloride and hydrochlorothiazide); vasodilating drugs (such as diltiazem, amiodarone, isoxsuprine, buphenin, tolazolin and verapamil); vasoconstrictors (such as dihydroergotamine, ergotamine and merergot); antiulcer drugs (such as ranitidine and cimetidine); narcotic drugs (such as lidocaine, bupivacaine, chloroprocaine, dibucaine); antidepressants (e.g., imipramine, desipramine, amitriptyline (amitriptiline), nortriptyline (nortriptiline)); tranquilizers and sedatives (e.g. chlorazines) Benazetyzine, benzquinamine, flurazepam, hydroxyzine, loxapine, and promazine; antipsychotic drugs (such as chlorprothixene, fluphenazine, haloperidol, molindone, thioridazine, and trifluoperazine); antimicrobial drugs (e.g., antibacterial, antifungal, antiprotozoal, and antiviral drugs).
Useful therapeutic moiety classes include adjuvants. The adjuvant can be selected from keyhole limpet hemocyanin conjugate, monophosphoryl lipid A, lipopeptide MALP-2 from mycoplasma, cholera toxin B subunit, Escherichia coli heat-labile toxin, and universal T helper cell antigen determining part from tetanus toxoidPosition, interleukin-12, CpG oligodeoxynucleotide, dimethyl dioctadecyl ammonium bromide, cyclodextrine, squalene, aluminium salts, meningococcal Outer Membrane Vesicles (OMVs), montanideiSA, TiterMaxTM(purchased from Sigma, St. Louis MO), nitrocellulose absorbents, immunostimulatory complexes such as Quil A, GerbuTMAdjuvants (Gerbu Biotechnik, Kirchwald, Germany), threonyl muramyl dipeptide, thymosin alpha, bupivacaine, GM-CSF, incomplete Freund's adjuvant, MTP-PE/MF59(Ciba/Geigy, Basel, Switzerland), polyphosphazenes, saponins from the Quillaja saponaria Quillaja, and Syntex adjuvant formulations (Biocine, Emeryville, Calif.), among others well known in the art.
Antimicrobial drugs that are preferably incorporated into the compositions of the present invention include, for example, pharmaceutically acceptable salts of β -lactam drugs, quinolone drugs, ciprofloxacin, norfloxacin, tetracycline, erythromycin, amikacin, triflurodocin, doxycycline, capreomycin, chlorhexidine, chlortetracycline, oxytetracycline, clindamycin, ethambutol, hexamidine isothionate (hexamideinioshionate), metronidazole, pentamidine, gentamycin, kanamycin, lineomycin, methacycline, hexamine, minocycline, neomycin, netilmicin (netilmicin), paromomycin, streptomycin, tobramycin, imizole, and amantadine. Other drug moieties useful in the practice of the present invention include antineoplastic agents (e.g., antiandrogens such as leuprolide or flutamide), cytocidal agents (e.g., andriamycin, doxorubicin, taxol, cyclophosphamide, busulfan, cisplatin, beta-2-interferon), antiestrogens such as tamoxifen, antimetabolites such as fluorouracil, methotrexate, mercaptopurine, thioguanine) also included in this class are radioisotope-based agents and conjugated toxins such as ricin, geldanamycin, mytansin, CC-1065, C-1027, duocarmycin, spinosad, and related structures and analogs thereof, as well as the toxins listed in table 2.
The therapeutic moiety may also be a hormone (e.g., 6 α -methyl-17 α -progesterone, estradiol, leuprolide, megestrol, somatostatin octapeptide, or somatostatin); muscle relaxants (such as cinnamylin, cyclobenzaprine, flavoxate, oxyphenamid, paraferine, mebeverine, isodaverin, ritodrine, diphenoxylate, nifedipine, and azumoplene)); antispasmodic drugs; bone activating drugs (such as diphosphates and phosphonoalkylphosphinates) drug compounds); endocrine regulating drugs (e.g. contraceptives (e.g. ethiodiol, ethinyl estradiol, norethindrone, mestranol, desogestrel, 6 α -methyl-17 α -pregnenone), diabetes regulators (e.g. glibenclamide or chlorpropamide), anabolics such as testolactone or stanozolol, androgens (e.g. methyltestosterone, testosterone or fluoxymesterone), antidiuretic agents (e.g. desmopressin) and calcitonin).
Also useful herein are estrogens (such as diethylstilbestrol), glucocorticoids (such as hydroxyfludroxydermosterol, betamethasone, and the like), and progestins, such as norethindrone, norethindrone (ethynodiol), norethindrone, levonorgestrel; thyroid agents (such as liothyronine or levothyroxine) or antithyroid agents (such as methimazole); anti-hyperprolactinemia drugs (e.g., cabergoline); hormonal suppressors (such as danazol or goserelin), oxytocin (such as ergonovine or oxytocin) and prostaglandins such as mioprostol, alprostadil or dinoprostone.
Other useful modifying groups include immunomodulatory drugs (e.g., antihistamines, mast cell stabilizers such as lodoxylamine and/or cromolyn), steroids (e.g., hydroxydehydrocortexolone, beclomethazone, cortisone, dexamethasone, prednisolone, methylprednisolone, beclomethasone or clobetasone), histamine H2 antagonists (e.g., famotidine, cimetidine, ranitidine), immunosuppressive agents (e.g., azathioprine, cyclosporine), and the like.
Kit of parts useful for therapyIncluding antisense drugs, and also including naked DNA. Antisense drugs can be selected from, for example, Affinitak (ISIS, Carlsbad, Calif.) and GenasenseTM(available from Genta, Berkeley Heights, NJ). Naked DNA may be delivered, for example, as a gene therapy agent together with DNA encoding, for example, blood coagulation factors VIII and IX for treatment of hemophilia.
F. Preparation of modified sugars
The modified saccharides used to form the conjugates of the invention are discussed herein. For clarity of illustration, the discussion focuses on the preparation of sugars modified with water-soluble polymers. In particular, the discussion focuses on the preparation of modified sugars that include a poly (ethylene glycol) moiety. The skilled artisan will appreciate that the methods set forth herein are broadly applicable to the preparation of modified sugars, and therefore, such discussion should not be considered as limiting the scope of the invention.
Typically, the sugar moiety and modifying group are linked together through the use of a reactive group that is typically converted to a new organofunctional group or non-reactive species by the linking method. The saccharide reactive functional group is located anywhere on the saccharide moiety. Reactive groups and the types of reactions used to practice the invention are generally well known in those areas of bioconjugate chemistry. The currently prevailing types of reactions available for reactive sugar moieties are those conducted under relatively mild conditions. These include, but are not limited to, nucleophilic substitutions (such as the reaction of amines and alcohols with acyl halides, active esters), electrophilic substitutions (such as enamine reactions), and additions to carbon-carbon and carbon-heteroatom multiple bonds (such as Michael's reaction, Diels-Alder addition). A discussion of these and other useful reactions can be found, for example, in Smith and March, Advanced organic chemistry, 5thEd.,John Wiley &Sons, New York, 2001; hermanson, Bioconjugate Techniques, Academic Press, San Diego, 1996; and Feeney et al, Modification of Proteins; advances in chemistry series, Vol.198, American Chemical Society, Washington, D.C., 1982.
Useful reactive functional groups that extend (pendant) into the sugar core or modifying group include, but are not limited to:
(a) carboxyl groups and their various derivatives, including but not limited to N-hydroxysuccinimide esters, N-hydroxybenzotriazole esters, acid halides, acylimidazoles, thioesters, p-nitrophenyl esters, alkyl, alkenyl, alkynyl, and aryl esters;
(b) hydroxyl groups, which can be converted to, for example, esters, ethers, aldehydes, and the like.
(c) Haloalkyl groups wherein the halide can later be replaced with a nucleophilic group, such as an amine, carboxylate anion, thiol anion, carbanion, or alkoxide ion, resulting in the covalent attachment of a new group to the halogen atom functional group;
(d) dienophile groups capable of participating in Diels-Alder reactions, such as maleimido (maleimido) groups;
(e) an aldehyde or ketone group, whereby subsequent derivatization by formation of a carbonyl derivative, such as an imine, hydrazone, semicarbazone or oxime, or by a mechanism such as Grignard addition or alkyllithium addition is possible;
(f) sulfonyl halide groups for subsequent reaction with amines to form, for example, sulfonamides;
(g) thiol groups which can be converted, for example, to disulfides or reacted with alkyl and acyl halides;
(h) Amine or sulfhydryl groups which may be, for example, acylated, alkylated or oxidized;
(i) an alkene which can be subjected to, for example, cycloaddition, acylation, Michael addition, etc.; and
(j) epoxides which can be reacted with, for example, amine and hydroxyl compounds.
The functional groups may be selected such that they do not participate in or interfere with the reactions necessary to assemble the reactive sugar core or modifying group. Alternatively, the reactive functional group may be protected from participating in the reaction by protecting the group in the presence of a protecting group. One skilled in the art understands how to protect a particular functional group so that it does not interfere with a selected set of reaction conditions. For examples of useful protecting groups, see, e.g., Greene et al, Protective group in Organic Synthesis, John Wiley & Sons, New York, 1991.
In the following discussion, specific examples of a number of modified sugars are presented that can be used in the practice of the present invention. In an exemplary embodiment, sialic acid derivatives are used as the sugar core to which the modifying group is attached. The discussion of sialic acid derivatives is focused on for clarity purposes only and should not be construed as limiting the scope of the invention. Those skilled in the art will appreciate that various other saccharide moieties may be activated and derivatized in a manner similar to that set forth with sialic acid as an example. For example, many methods of modifying galactose, glucose, N-acetylgalactosamine and fucose are available, these sugars are only a few of the mentioned substrates, and they can be easily modified by art-recognized methods. See, e.g., Elhalabi et al, curr.med.chem.6: 93 (1999); and Schafer et al, j.org.chem.65: 24(2000).
In an exemplary embodiment, the peptides modified by the methods of the invention are peptides produced in mammalian cells (e.g., CHO cells) or in transgenic animals, and thus contain incompletely sialylated N-and/or O-linked oligosaccharide chains. The glycopeptide may be pegylated, PPG-modified or modified with modified sialic acid in the absence of sialic acid and oligosaccharide chains containing terminal galactose residues.
In scheme 4, mannosamine glycoside 1 is treated with an active ester of a protected amino acid (e.g., glycine) derivative to convert the sugar amine residue to the corresponding protected amino acid amide adduct. The adduct is treated with aldolase to form sialic acid 2. Compound 2 is converted to the corresponding CMP derivative by the action of CMP-SA synthetase, followed by catalytic hydrogenation of the CMP derivative to produce compound 3. The amine introduced by the formation of the glycine adduct serves as a site for attachment of the PEG or PPG such that Compound 3 reacts with an activated PEG or PPG derivative (e.g., PEG-C (O) NHS, PPG-C (O) NHS) to form 4 or 5, respectively.
Table 3 presents representative examples of sugar monophosphates derivatized with PEG or PPG moieties. Certain of the compounds in table 3 were prepared by the method of scheme 1. Other derivatives are prepared by art-recognized methods. See, e.g., Keppler et al, Glycobiology 11: 11R (2001); and Charter et al, Glycobiology 10: 1049(2000)). Other amine-reactive PEG and PPG analogs are commercially available or they can be prepared by methods readily available to those skilled in the art.
TABLE 3 examples of sugar monophosphates derivatized with PEG or PPG moieties
The modified sugar phosphates used in the practice of the present invention may be substituted at other positions as well as those set forth above. "i" can be Na or another salt, and "i" can be exchanged for Na. Presently preferred substitutions for sialic acid are set forth in formula 5.
Molecular formula 5:
wherein X is preferably selected from-O-, -N (H) -, -S, CH2-and N (R)2Wherein each R is independently selected from R1-R5Is a member of (1). "i" may be Na or another salt, and Na may be interchanged with "i". The symbols Y, Z, A and B each represent a group selected from those described above for X. X, Y, Z, A and B are each independently selected so they may be the same or different. Symbol R1、R2、R3、R4And R5Represents H, a polymer, a water-soluble polymer, a therapeutic moiety, a biomolecule or other moiety. The symbol R6 represents H, OH or a polymer. Alternatively, the symbols represent linkers attached to polymers, water soluble polymers, therapeutic moieties, biomolecules or other moieties.
In another exemplary embodiment, mannosamine is acylated and activated by simultaneous nucleophilic substitution using chloroacetic anhydride as set forth in scheme 5. In each of the schemes provided in this section, i +Or Na+Interchangeably, wherein the salt may be sodium, or may be any other suitable salt.
Scheme(s)
The resulting chloro-derivatized glycan is contacted with pyruvate in the presence of aldolase to form chloro-derivatized sialic acid. The corresponding nucleotide sugar is prepared by contacting the sialic acid derivative with the appropriate nucleotide triphosphate and synthetase. The chloro group on the sialic acid moiety can then be substituted with a nucleophilic PEG derivative such as thio-PEG.
In a further exemplary embodiment, as shown in scheme 6, mannosamine is acylated with bis-HOBT dicarboxylate to produce the corresponding amide-alkyl-carboxylic acid, which is subsequently converted to a sialic acid derivative. The sialic acid derivative is converted to a nucleotide sugar and the carboxylic acid is activated and reacted with a nucleophilic PEG derivative such as amino-PEG.
In another exemplary embodiment, the amine-and carboxy-protected neuraminic acids are activated by conversion of the primary hydroxyl group to the corresponding p-toluenesulfonate, and the methyl ester is cleaved as set forth in scheme 7. The activated neuraminic acid is converted to the corresponding nucleotide sugar and the activating group is replaced with a nucleophilic PEG species such as thio-PEG.
In yet a further exemplary embodiment, the primary hydroxyl moieties of the amine-and carboxy-protected neuraminic acid derivatives are alkylated with a nucleophilic PEG, such as chloro-PEG, as set forth in scheme 8. The methyl ester was then cleaved and the PEG-sugar was converted to the nucleotide sugar.
Glycans other than sialic acid can be derivatized with PEG using the methods set forth herein. Derivatized glycans are also within the scope of the invention per se. Thus, scheme 9 provides an exemplary synthetic route to pegylated galactose nucleotide sugars. The primary hydroxyl group of galactose was activated to the corresponding p-toluenesulfonate and subsequently converted to the nucleotide sugar.
Exemplary moieties attached to the conjugates disclosed herein include, but are not limited to, PEG derivatives (e.g., acyl-PEG, acyl-alkyl-PEG, alkyl-acyl-PEG carbamoyl-PEG, aryl-PEG, alkyl-PEG), PPG derivatives (e.g., acyl-PPG, acyl-alkyl-PPG, alkyl-acyl-PPG carbamoyl-PPG, aryl-PPG), polyaspartic acid, polyglutamic acid, polylysine, therapeutic moieties, diagnostic moieties, mannose-6-phosphate, heparin, heparinoids, SLexMannose, mannose-6-phosphate, Sialyl Lewis X, FGF, VFGF, proteins (such as transferrin), chondroitin, keratin, dermatan, dextran, modified dextran, amylose, diphosphate, poly-SA, hyaluronic acid, keritan, albumin, integrin, antennary oligosaccharide, peptide, and the like. Methods for conjugating various modifying groups to sugar moieties are readily available to those skilled in the art (Poly (ethylene glycol) Chemistry: biotechnical biological Applications, J.Milton Harris, ed., Plenum pub. corp., 1992; poly (ethylene glycol) Chemical and biological applications, J.Milton Harris, Ed., ACS Symposium Series No.680, American Chemical Society, 1997; hermanson, bioconjugateTechniques, Academic Press, San Diego, 1996; and Dunn et al, eds. polymeric Drugs and Drug Delivery Systems, ACS Symposium series Vol.469, American Chemical Society, Washington, D.C. 1991).
Purification of sugars, nucleotide sugars and derivatives
The nucleotide sugars and derivatives produced by the above methods can be used without purification. However, it is generally preferred to recover the product. Standard well-known techniques for recovering glycosylated sugars can be applied, such as thin or thick layer chromatography, column chromatography, ion exchange chromatography or membrane filtration. Preferably, membrane filtration is applied, more preferably reverse osmosis membranes or one or more column chromatography techniques are used for recovery, as discussed below and in the references cited herein. For example, membrane filtration may be used to remove proteins from reagents having a molecular weight of less than 10,000Da, the membrane having a molecular weight cut-off of about 3000 to about 10,000. Membrane filtration or reverse osmosis can then be used to remove salts and/or purify the product sugars (see e.g. WO 98/15581). Nanofilter membranes are a class of reverse osmosis membranes that can pass monovalent salts while retaining multivalent salts and uncharged solutes greater than about 100 to about 2,000 daltons (depending on the membrane used). Thus, in general use, the sugars produced by the process of the invention will remain on the membrane, while the contaminating salts will pass through the membrane.
G. Crosslinking groups
The preparation of modified sugars for use in the methods of the invention involves the attachment of a modifying group to a sugar residue and the formation of a stable adduct that is a substrate for a glycosyltransferase. Thus, it is often preferred to conjugate the modifying group to the saccharide using a cross-linking agent. Exemplary bifunctional compounds that can be used to attach a modifying group to a sugar moiety include, but are not limited to, bifunctional poly (ethylene glycols), polyamides, polyethers, polyesters, and the like. General methods for linking carbohydrates to other molecules are well known in the literature. See, e.g., Lee et al, Biochemistry 28: 1856 (1989); bhatia et al, anal. biochem.178: 408 (1989); janda et al, j.am.chem.soc.112: 8886(1990) and Bednarski et al, WO 92/18135. In the discussion that follows, the reactive groups of the sugar moiety of the nascent modified sugar are subjected to mild treatment. The emphasis of this discussion is for clarity. One skilled in the art will understand that this discussion also applies to the reactive groups on the modifying group.
One exemplary strategy involves incorporating sulfhydryl groups protected with the heterobifunctional crosslinker SPDP (n-succinimide-3- (2-pyridyldithio) propionic acid) into the saccharide, followed by deprotection of the sulfhydryl groups to form disulfide bonds with other sulfhydryl groups on the modifying group.
If SPDP adversely affects the ability of the modified sugar to act as a substrate for glycosyltransferases, then some other crosslinker can be used to form disulfide bonds, such as 2-iminotetrahydrothiophene or N-succinimide S-acetylthioacetic acid (SATA). 2-iminothiolanes can be reacted with primary amines and the unprotected sulfhydryl groups are immediately incorporated into the amine-containing molecule. SATA also reacts with primary amines, but incorporates a protected sulfhydryl group which is subsequently deacetylated with hydroxylamine to produce a free sulfhydryl group. In each case, the integrated sulfhydryl group is free to react with other sulfhydryl groups or protected sulfhydryl groups, such as SPDP, to form the desired disulfide bond.
The above strategies are exemplary, but not limiting, of the linkers used in the present invention. Other cross-linking agents are available that can be used in different strategies for cross-linking modifying groups to peptides. For example, TPCH (S- (2-thiopyridyl) -L-cysteine hydrazide and TPMPH ((S- (2-thiopyridyl) mercapto-propionyl hydrazide) react with sugar moieties oxidized by mild treatment with periodate beforehand, thereby forming a hydrazone bond between the hydrazide moiety of the crosslinker and the aldehyde generated by periodate TPCH and TPMPH introduce a 2-pyridylthioketone protected sulfhydryl group on the sugar, which can be deprotected with DTT and subsequently used for conjugation, such as forming a disulfide bond between the components.
If disulfide bonds were found to be inappropriate for generating stable modified sugars, other cross-linking agents that incorporate more stable bonds between the components could be used. The heterobifunctional crosslinkers GMBS (N-gama-maleimidoyloxy) succinimide) and SMCC (succinimidyl 4- (N-maleimide-methyl) cyclohexane) are reacted with a primary amine to introduce a maleimide group onto the composition. The maleimide groups may then react with sulfhydryl groups on the other components which may be introduced by the crosslinking agents described hereinbefore, to form stable thioether linkages between the components. If steric hindrance between the components interferes with the activity of the components or the ability of the modified sugar to act as a substrate for the glycosyltransferase, a cross-linking agent capable of introducing a longer spacer between the components may be used and may include some of the cross-linking agent (i.e., SPDP) derivatives described above. Thus, there are a number of useful suitable crosslinking agents; each of them is selected depending on its effect on optimal peptide conjugates and modified sugar production.
A number of reagents are available for modifying the composition of modified sugars with intramolecular chemical crosslinking (for a review of crosslinking agents and crosslinking procedures, see: Wold, F., meth.enzymol.25: 623. sup. 651, 1972; Weetall, H.H. and Cooney, D.A., Enzymes as Drugs (Holcenberg and Roberts, eds.) pp.395-442, Wiley, New York, 1981; Ji, T.H., meth.enzymol.91: 580. sup. 609, 1983; Mattson et al, mol.biol.Rep.17: 167. sup. 183, 1993, all incorporated herein by reference). Preferred crosslinkers can be derived from a variety of zero length, homo-bifunctional and hetero-bifunctional crosslinkers. Zero-length crosslinkers involve direct conjugation of two internal chemical groups without introducing external materials. Agents that catalyze disulfide bond formation are of this type. Another example is a reagent that induces condensation of a carboxyl group and a primary amino group to form an amide bond, such as carbodiimide, ethylchloroformate, Woodward reagent K (2-ethyl-5-phenylisoxazolium-3' -sulfonate), and carbonyldiimidazole. In addition to these chemical agents, transglutaminase (glutamyl-peptide gamma-glutamyltransferase; EC 2.3.2.13) can be used as a zero-length cross-linker. The enzyme catalyzes the transacylation of the carbamoyl group of a protein-linked glutamine residue, usually with a primary amino group as substrate. Preferred homo-and hetero-bifunctional reagents contain two identical or different sites, respectively, which are reactive towards amino, sulfhydryl, guanidino, indole or unspecified groups.
2. Preferred specific sites on the crosslinker
a. Amino-reactive groups
In a preferred embodiment, the sites on the crosslinker are amino-reactive groups. Useful non-limiting examples of amino-reactive groups include N-hydroxysuccinimide (NHS) esters, urethanes, isocyanates, acyl halides, aryl nitrides, p-nitrophenyl esters, aldehydes, and sulfonyl chlorides.
The NHS ester is preferably reacted with a primary (including aromatic) amino group of the modified sugar moiety. The imidazole group of histidine is known to compete with primary amines for reaction, but the reaction product is unstable and susceptible to hydrolysis. This reaction involves a nucleophilic attack of the amine on the acidic carboxyl group of the NHS ester to form an amide, thereby releasing the N-hydroxysuccinimide. The positive charge of the original amino group is thus lost.
The urethanene is the most specific acylating agent that reacts with the amine group of the modified sugar moiety. At a pH of 7 to 10, the imide reacts only with the primary amine. The primary amine nucleophilically attacks the imidate to yield an intermediate that decomposes to an amidine at high pH and a new imidate at low pH. The novel imidates can be reacted with another primary amine to crosslink the two amino groups, which is the case for the putative monofunctional imidates to undergo a bifunctional reaction. The main product of the reaction with the primary amine is an amidine, which is a more basic base than the original amine. The positive charge on the original amino group is retained.
Isocyanates (and isothiocyanates) react with the primary amines of the modified sugar moieties to form stable linkages. Their reaction with sulfhydryl, imidazole and tyrosyl groups produces relatively unstable products.
Acyl nitrides are also used as amino-specific reagents, where the nucleophilic amine of the affinity component attacks the acidic carboxyl group under slightly basic conditions, such as ph 8.5.
Aryl halides such as 1, 5-difluoro-2, 4-dinitrobenzene react preferably with the amino group of the modified sugar moiety and the phenol group of tyrosine, but also with sulfhydryl groups and imidazole groups.
P-nitrophenyl esters of mono-and dicarboxylic acids are also useful amino-reactive groups. Although the specificity of the reagent is not particularly high, the reaction of the alpha-and epsilon-amino groups is the fastest.
Aldehydes such as glutaraldehyde are reacted with the primary amines of the modified sugars. While the labile schiff base is formed in the reaction of the amino group with the aldehyde, glutaraldehyde is capable of modifying the modified sugar with stable cross-linking. At typical cross-linking pH conditions of pH6-8, the cyclic polymer undergoes dehydration to form an alpha-beta unsaturated aldehyde polymer. Whereas schiff bases are stable when conjugated to another double bond. The resonance interaction of the two double bonds prevents the hydrolysis of the schiff bond. In addition, high local concentrations of amines can attack the ethylene double bond to form stable Michael addition products.
Aromatic sulfonyl chlorides react with multiple sites of the modified sugar moiety, but the reaction with the amino group is most important, resulting in the formation of stable sulfonamide linkages.
b. Sulfhydryl reactive groups
In another preferred embodiment, the site is a sulfhydryl-reactive group. Useful non-limiting sulfhydryl-reactive groups include maleimides, alkyl halides, pyridyl disulfides, and thiophthalimides.
The maleimide reacts with the sulfhydryl group of the modified sugar moiety to form a stable thioether bond. They also react at a very low rate with the primary amino groups and the imidazole groups of histidine. However, maleimide can be considered a sulfhydryl-specific group at pH7, since the reaction rate of simple thiols is 1000 times higher than the corresponding amines at pH 7.
The alkyl halides react with sulfhydryl, sulfide, imidazole and amino groups. However, at neutral to slightly basic pH, the alkyl halides react primarily with sulfhydryl groups to form stable thioether bonds. At higher pH, the reaction with amino groups is predominant.
The pyridyl disulfide reacts with the free sulfhydryl groups by disulfide exchange to form a mixed disulfide. As a result, pyridyl disulfide is the most specific sulfhydryl-reactive group.
The thiophthalimide reacts with a free sulfhydryl group to form a disulfide.
c. Carboxyl reactive residue
In another embodiment, a carbodiimide that is soluble in water and organic solvents is used as the carboxyl-reactive reagent. These compounds react with free carboxyl groups to form pseudoureas, which can then be coupled with available amines to form amide bonds. Procedures for modifying carboxyl groups with carbodiimides are well known in the art (see Yamada et al, Biochemistry 20: 4836-4842, 1981).
3. Preferred non-specific sites in the crosslinker
In addition to the use of site-specific reactive moieties, the present invention also relates to the use of non-specific reactive groups to attach sugars to modifying groups.
Exemplary non-specific cross-linkers include photoactive groups that are completely inert in the dark, and which can be converted to a reactive species upon absorption of a photon of appropriate energy. In a preferred embodiment, the photoactive group is selected from nitrene precursors generated by heating or photolysis of nitrides. Electron deficient nitrenes are extremely reactive and can react with a variety of chemical bonds, including N-H, O-H, C-H and C ═ C. While class 3 nitrides (aryl, alkyl and acyl derivatives) may be employed, aryl nitrides are presently preferred. Aryl nitrides are more reactive with N-H and O-H than with C-H after photolysis. Electron deficient arylnitrenes rapidly undergo ring expansion to form dehydroazepines that tend to react with nucleophiles rather than forming C-H insertions. The reactivity of aryl nitrides can be enhanced by the presence of electron-withdrawing substituents such as nitro or hydroxyl groups in the ring. Such substituents push the maximum absorption of the aryl nitride to a greater wavelength. Unsubstituted arylnitrides have absorption maxima in the range of 260-280nm, while hydroxyl and nitroarylnitrides absorb light significantly above 305 nm. Therefore, hydroxyl and nitroaryl nitrides are most preferred because they enable the application of photolytic conditions less detrimental to the affinity component than unsubstituted aryl nitrides.
In another preferred embodiment, the photoactive group is selected from fluorinated aryl nitrides. The photolysis products of fluorinated aryl nitrides are aryl nitrenes, which all undergo highly efficient and characteristic reactions with this group, including C-H bond insertions (Keana et al, J.org.chem.55: 3640-3647, 1990).
In another embodiment, the photoactive group is selected from the group consisting of benzophenone residues. The benzophenone reagent generally gives higher crosslinking results than the aryl nitride reagent.
In another embodiment, the photoactive group is selected from diazo compounds that form an electron-deficient carbene upon photolysis. These carbenes can undergo a variety of reactions including insertion into a C-H bond, addition to double bonds (including aromatic systems), hydrogen attraction, and coordination to nucleophilic centers to give a carbon ion.
In another embodiment, the photoactive group is selected from diazopyruvate. For example, the p-nitrophenyl ester of p-nitrophenyldiazopyruvate reacts with an aliphatic amine to form diazopyruvate amide, which undergoes ultraviolet photolysis to form an aldehyde. The photolyzed diazopyruvate modified affinity component will react similarly to formaldehyde or glutaraldehyde to form crosslinks.
4. Homobifunctional reagents
a. Homobifunctional reagents with primary amines
The synthesis, nature and use of amine-reactive crosslinkers are commercially described in the literature (for a review of crosslinking procedures and reagents, see above). Many reagents are commercially available (e.g., Pierce Chemical Company, Rockford, I11.; Sigma Chemical Company, St. Louis, Mo.; Molecular Probes, Inc., Eugene, OR).
Preferred non-limiting examples of homobifunctional NHS esters include disuccinimidyl glutarate (DSG), disuccinimidyl suberate (DSS), bis (sulfosuccinimidyl) suberate (BS), disuccinimidyl tartrate (DST), disuccinimidyl tartrate (sulfo-DST), bis-2- (succinimidyloxycarbonyloxy) ethyl sulfone (BSOCOES), bis-2- (sulfosuccinimidyloxycarbonyloxy) ethyl sulfone (sulfo-BSOCOES), ethylene glycol di (succinimidyl succinate) (EGS), ethylene glycol di (sulfosuccinimidyl succinate) (sulfo-EGS), dithiobis (succinimidyl propionate) (DSP), and dithiobis (sulfosuccinimidyl propionate) (sulfo-DSP). Preferred non-limiting examples of homobifunctional imidoesters include Dimethylpropanimidate (DMM), dimethylsuccinimidyl ester (DMSC), Dimethyladiimidate (DMA), Dimethylpimidate (DMP), Dimethyloctanimidate (DMS), dimethyl-3, 3 ' -oxydiprimidate (DODP), dimethyl-3, 3 ' - (methylenedioxy) dipropimidate (DMDP), dimethyl-3 ' - (dimethylenedioxy) dipropimidate (DDDP), dimethyl-3, 3 ' - (tetramethylenedioxy) dipropimidate (DTDP), and dimethyl-3, 3 ' -Dithiodipropimidate (DTBP).
Preferred non-limiting examples of homobifunctional isothiocyanates include: terephthalocyanurate (DITC) and 4, 4 '-diisothiocyanato-2, 2' -disulphonate stilbene (DIDS).
Preferred non-limiting examples of homobifunctional isocyanates include xylene-diisocyanate, toluene-2, 4-diisocyanate, toluene-2-isocyanate-4-isothiocyanate, 3-methoxydiphenylmethane-4, 4 ' -diisocyanate, 2 ' -dicarboxy-4, 4 ' -azophenyl diisocyanate and hexamethylene diisocyanate.
Preferred non-limiting examples of homobifunctional aryl halides include 1, 5-difluoro-2, 4-dinitrobenzene (DFDNB) and 4, 4 '-difluoro-3, 3' -dinitrophenylsulfone.
Preferred non-limiting examples of homobifunctional aliphatic aldehyde reagents include glyoxal, malondialdehyde (malondialdehyde), and glutaraldehyde.
Preferred non-limiting examples of homobifunctional acylating agents include nitrophenyl esters of dicarboxylic acids.
Preferred non-limiting examples of homobifunctional aromatic sulfonyl chlorides include phenol-2, 4-disulfonyl chloride and alpha-naphthol-2, 4-disulfonyl chloride.
Preferred, non-limiting examples of additional amino-reactive homobifunctional reagents include erythritol dicarboxylic acid (erythro lbiscarbonate) which reacts with an amine to form a dicarbamate (bisscarbamate).
b. Homobifunctional crosslinkers reactive with free sulfhydryl groups
The synthesis, properties and use of such reagents are described in the literature (for a review of the crosslinking procedures and reagents, see above). Many reagents are commercially available (e.g., Pierce Chemical Company, Rockford, i 11.; Sigma Chemical Company, st. louis, mo.; Molecular Probes, inc., Eugene, OR).
Preferred non-limiting examples of homobifunctional maleimides include bismaleimide hexane (BMH), N '- (1, 3-phenylene) bismaleimide, N' - (1, 2-phenylene) bismaleimide, azophenyl bismaleimide, and bis (N-maleimidomethyl) ether.
A preferred non-limiting example of a homobifunctional pyridyl disulfide includes 1, 4-bis-3 '- (2' -pyridyldithio) propionamidobutane (DPDPPB).
Preferred non-limiting examples of homobifunctional alkyl halides include 2, 2 '-dicarboxy-4, 4' -diiodoacetamidoazobenzene, α '-diiodo-p-xylenesulfonic acid, α' -dibromo-p-xylenesulfonic acid, N '-bis (b-bromoethyl) benzylamine, N' -bis (bromoacetyl) phenylhydrazine, and 1, 2-bis (bromoacetyl) amino-3-phenylpropane.
c. Homobifunctional photoactivated cross-linking agents
The synthesis, properties and use of such reagents are described in the literature (for a review of the crosslinking procedures and reagents, see above). Many reagents are commercially available (e.g., Pierce Chemical Company, Rockford, i 11.; Sigma Chemical Company, st. louis, mo.; Molecular Probes, inc., Eugene, OR).
Preferred non-limiting examples of homobifunctional photoactivatable crosslinkers include bis- β - (4-azidosalicylamido) ethyl disulfide (BASED), bis-N- (2-nitro-4-azidophenyl) -cystamine-S, S '-Dioxide (DNCO), and 4, 4' -dithiobisphenyl nitride.
5. Heterobifunctional reagents
a. Amino reactive heterobifunctional reagents with pyridyl disulfide moieties
The synthesis, properties and use of such reagents are described in the literature (for a review of the crosslinking procedures and reagents, see above). Many reagents are commercially available (e.g., Pierce Chemical Company, Rockford, i 11.; Sigma Chemical Company, st. louis, mo.; Molecular Probes, inc., Eugene, OR).
Preferred non-limiting examples of heterobifunctional reagents having a pyridyl disulfide moiety and an amino-reactive NHS ester include N-succinimidyl-3- (2-pyridyldithio) propionic acid (SPDP), succinimidyl 6-3- (2-pyridyldithio) propionamidohexanoic acid (LC-SPDP), sulfosuccinimidyl 6-3- (2-pyridyldithio) propionamidohexanoic acid (sulfo-LCSPP), 4-succinimidyloxycarbonyl-alpha-methyl-alpha- (2-pyridyldithio) toluene (SMPT), and sulfosuccinimidyl 6-alpha-methyl-alpha- (2-pyridyldithio) toluamide hexanoic acid (sulfo-LC-SMPT).
b. Amino reactive heterobifunctional reagents with maleimide moieties
The synthesis, properties and use of such agents are described in the literature. Preferred non-limiting examples of heterobifunctional reagents having a maleimide moiety and an amino-reactive NHS ester include succinimide maleimide acetyl ester (AMAS), succinimide 3-maleimide propionate (BMPS), N-gamma-maleimide butyryloxysuccinimide ester (GMBS), N-gamma-maleimide butyryloxysulfosuccinimide ester (sulfo-GMBS), succinimide 6-maleimide hexanoate (EMCS), succinimide 3-maleimide benzoate (SMB), m-maleimide benzoyl-N-hydroxysuccinimide ester (MBS), m-maleimide benzoyl-N-hydroxysulfosuccinimide ester (sulfo-MBS), succinimide 4- (N-maleimidomethyl) -cyclohexane-1-carboxylate (SMCC), Sulfosuccinimide 4- (N-maleimidomethyl) -cyclohexane-1-carboxylate (sulfo-SMCC), succinimide 4- (p-maleimidophenyl) butyrate (SMPB), and sulfosuccinimide 4- (p-maleimidophenyl) butyrate (sulfo-SMPB).
c. Amino reactive heterobifunctional reagents with alkyl halide moieties
The synthesis, properties and use of such agents are described in the literature. Preferred non-limiting examples of heterobifunctional reagents having an alkyl halide moiety and an amino-reactive NHS ester include N-succinimide- (4-iodoacetyl) aminobenzoate (SIAB), sulfosuccinimide- (4-iodoacetyl) aminobenzoate (sulfo-SIAB), succinimide-6- (iodoacetyl) aminocaproate (SIAX), succinimide-6- (6- ((iodoacetyl) -amino) hexanoylamino) hexanoate (SIAXX), succinimide-6- (((4-iodoacetyl) -amino) -methyl) -cyclohexane-1-carbonyl) aminocaproate (SIACX) and succinimide-4 ((iodoacetyl) -amino) methylcyclohexane-1-carboxylate (SIAC).
A preferred example of a heterobifunctional reagent having an amino-reactive NHS ester and an alkyl dihalide moiety is N-hydroxysuccinimide 2, 3-dibromopropionate (SDBP). SDBP introduces intramolecular cross-linking to the affinity component by conjugation to its amino group. The reactivity of the dibromopropionyl moiety with primary amines can be controlled by the reaction temperature (McKenzie et al, Protein chem.7: 581-592 (1988)).
Preferred non-limiting examples of heterobifunctional reagents having an alkyl halide moiety and an amino-reactive p-nitrophenyl ester include p-nitrophenyliodoacetate.
Other crosslinking agents are well known to those skilled in the art. See, e.g., Pomato et al, U.S. Pat. No.5,965,106. It is within the ability of the person skilled in the art to select a suitable cross-linking agent for a particular application.
d. Cleavable linker group
In yet further embodiments, the linker group has a group that can be cleaved to release the modifying group from the sugar residue. Many cleavable groups are known in the art. See, e.g., Jung et al, biochem. biophysis. acta 761: 152-162 (1983); joshi et al, j.biol.chem.265: 14518-14525 (1990); zarling et al, J.Immunol.124: 913-920 (1980); bouizar et al, eur.j. biochem.155: 141-147 (1986); park et al, j.biol.chem.261: 205-210 (1986); browning et al, j.immunol.143: 1859-1867(1989). In addition, a wide range of cleavable bifunctional linker groups (homo-or heterobifunctional) are commercially available from suppliers such as Pierce.
Exemplary cleavable moieties can be cleaved with light, heat, or a reagent such as thiol, hydroxylamine, a base, periodate, and the like. In addition, certain preferred groups are cleaved in vivo in response to endocytosis (e.g., cis-aconityl; see Shen et al, biochem. Biophys. Res. Commun.102: 1048 (1991)). Preferred cleavable groups comprise a cleavable moiety selected from the group consisting of a member of the disulfide, ester, imide, carbonate, nitrobenzyl, acetophenone, and benzoin groups.
e. Conjugation of modified saccharides to peptides
The modified saccharide is conjugated to the glycosylated or non-glycosylated peptide by an appropriate enzyme that mediates conjugation. Preferably, the concentrations of the modified donor sugar, enzyme and acceptor peptide are selected so that glycosylation can proceed until the acceptor is used up. Although suggested with respect to sialyltransferases, the factors discussed below are generally applicable to other glycosyltransferase reactions.
Many methods of synthesizing the desired oligosaccharide structures using glycosyltransferases are well known and are commonly employed in the present invention. Some documents describe exemplary methods, such as WO 96/32491, Ito et al, Pure appl.chem.65: 753(1993) and U.S. Pat. No.5,352,670, 5,374,541 and 5,545,553.
The present invention is practiced with a single glycosyltransferase or a combination of glycosyltransferases. For example, one may use a combination of sialyltransferase and galactosyltransferase. In embodiments where more than one enzyme is employed, the enzyme and substrate are preferably combined in the initial reaction compound, or the enzyme and reagent of the second enzymatic reaction are added to the reaction medium at or near the completion of the first enzymatic reaction. By performing the two enzymatic reactions in sequence in a single vessel, the overall yield is improved over procedures in which intermediate species are isolated. In addition, purging and disposal of additional solvents and by-products is reduced.
In a preferred embodiment, both the first and second enzymes are glycosyltransferases. In another preferred embodiment, one enzyme is an endoglycosidase. In another preferred embodiment, one enzyme is an exoglycosidase. In additional preferred embodiments, more than two enzymes are used to assemble the modified glycoproteins of the present invention. The enzyme is used to alter the carbohydrate structure on a peptide at any site, either before or after the addition of a modified carbohydrate to the peptide.
In another embodiment, the at least two enzymes are glycosyltransferases and the last saccharide added to the saccharide structure on the peptide is an unmodified saccharide. Alternatively, the modified saccharide is located within the glycan structure and thus need not be the last saccharide on the glycan. In an exemplary embodiment, the galactosyltransferase enzyme can catalyze the transfer of Gal-PEG from UDP-Gal-PEG to glycans, followed by incubation in the presence of ST3Gal3 and CMP-SA, which will add unmodified "capped" sialic acid to the glycans (FIG. 23A).
In another embodiment, the at least two enzymes used are glycosyltransferases and at least two modified sugars are added to the glycan structure of the peptide. In this way, two or more different peptide conjugates can be added to one or more glycans on the peptide. This process results in glycan structures with two or more functionally different modified sugars. In an exemplary embodiment, incubation of the peptide with GnT-I, II and UDP-GlcNAc-PEG can add GlcNAc-PEG molecules to the glycans; incubation with galactosyltransferase and UDP-Gal can then add Gal residues thereto; and incubation with ST3Gal3 and CMP-SA-Man-6-phosphate can add SA-mannose-6-phosphate molecules to the glycans. The result of this series of reactions produced glycan chains with the functional characteristics of pegylated glycans and mannose-6-phosphate targeting activity (fig. 23B).
In another embodiment, the at least two enzymes used in the reaction are glycosyltransferases and different modified sugars are added to the N-linked and O-linked glycans on the peptide. This embodiment is useful when two different modified saccharides are added to the glycan of the peptide and when it is important to spatially separate the modified saccharides on the peptide from each other. For example, this method is preferred if the modified sugar comprises bulky molecules including, but not limited to, PEG and other molecules such as linker molecules. The modified sugars may be added simultaneously to the glycans of the peptide, or they may be added sequentially. In an exemplary embodiment, incubation with ST3Gal3 and CMP-SA-PEG can add sialic acid-PEG to the N-linked glycans, while incubation with ST3Gal1 and CMP-SA-diphosphate can add sialic acid diphosphate to the O-linked glycans (fig. 23C).
In another embodiment, the method employs one or more exo-or endoglycosidases. The glycosidase is typically a mutant that has been processed to form a glycosyl bond rather than disrupting the bond. Mutant glycanases, sometimes referred to as sugar synthases, typically include substitution of an acidic amino acid residue at the active site with an amino acid residue. For example, when the endo-glycanase is endo-H, the active site of substitution is typically Asp at position 130, Glu at position 132 or a combination thereof. The amino acid is typically substituted with serine, alanine, asparagine, or glutamine. Exoglycosidases such as transsialidase (transalylidase) are also useful.
The mutated enzyme usually catalyzes the reaction in a synthetic step, which is similar to the reverse reaction of the hydrolysis step of the endoglucanase. In these embodiments, the glycosyl donor molecule (e.g., the desired oligo-or mono-saccharide structure) contains a leaving group and the reaction proceeds with the addition of the donor molecule to the GlcNAc residues on the protein. For example, the leaving group may be a halogen, such as fluoride. In other embodiments, the leaving group is an Asn or Asn-peptide moiety. In yet further embodiments, the GlcNAc residue on the glycosyl donor molecule is modified. For example, a GlcNAc residue may comprise a 1, 2 oxazoline moiety.
In a preferred embodiment, each of the enzymes used to produce the conjugates of the invention is present in a catalytic amount. The catalytic amount of a particular enzyme will vary depending on the concentration of the enzyme substrate and the reaction conditions, such as temperature, time and pH. Methods for determining the catalytic amount of a given enzyme at a preselected substrate concentration and reaction conditions are well known to those skilled in the art.
The temperature range at which the above process is carried out is from the freezing point to the temperature at which the most sensitive enzymes denature. The preferred temperature range is from about 0 ℃ to about 55 ℃, and more preferably from about 20 ℃ to about 37 ℃. In another exemplary embodiment, one or more of the components of the method of the invention is performed with a thermophilic enzyme at elevated temperatures.
The reaction mixture is maintained for a sufficient time to allow glycosylation of the acceptor, thereby forming the desired conjugate. Some conjugates are often detectable in a few hours, while recoverable amounts are typically obtained in 24 hours or less. Those skilled in the art understand that the reaction rate depends on many variable factors (e.g., enzyme concentration, donor concentration, acceptor concentration, temperature, solvent volume) that are optimized depending on the system chosen.
The invention also provides for the production of modified peptides on an industrial scale. As used herein, at least one gram of the final purified conjugate is typically produced on an industrial scale.
In the discussion that follows, the present invention is exemplified by the conjugation of modified sialic acid moieties to glycosylated peptides. The exemplary modified sialic acid is labeled with PEG. The following discussion of the use of PEG-modified sialic acid and glycosylated peptides is for clarity of illustration and is not meant to limit the invention to conjugation of these two partners. The skilled artisan will appreciate that this discussion is generally applicable to the addition of modified glycosyl moieties other than sialic acid. In addition, the discussion is also applicable to the modification of glycosyl units with agents other than PEG, including other water-soluble polymers, therapeutic moieties, and biomolecules.
An enzymatic method can be used to selectively introduce pegylated or PPG-ized saccharides into peptides or glycopeptides. The method utilizes a modified saccharide containing PEG, PPG, or masked reactive functional groups, in combination with a suitable glycosyltransferase or saccharide synthase. By selecting a glycosyltransferase that can form the desired saccharide linkage and utilize the modified saccharide as a donor substrate, the PEG or PPG can be introduced directly onto the peptide backbone, onto an existing saccharide residue on the glycopeptide, or onto a saccharide residue that has been added to the peptide.
The receptor for sialyltransferase is present on the peptide modified by the method of the invention either as a naturally occurring structure or recombinantly, enzymatically or chemically placed thereon. Suitable receptors include, for example, galactosyl receptors such as Gal β 1, 4GlcNAc, Gal β 1, 4GalNAc, Gal β 1, 3GalNAc, lacto-N-tetraose, Gal β 1, 3GlcNAc, Gal β 1, 3Ara, Gal β 1, 6GlcNAc, Gal β 1, 4Glc (lactose) and others known to those skilled in the art (see, for example, Paulson et al, J.biol.chem.253: 5617-5624 (1978)).
In one embodiment, the receptor for sialyltransferase is present on the peptide to be modified after its synthesis in vivo. Such peptides can be sialylated by the method of the invention without prior modification of the glycosylation pattern of the peptide. Alternatively, the methods of the invention may be used to sialylate peptides that do not include the appropriate receptor; one first modifies the peptide to include the receptor by methods well known to those skilled in the art. In an exemplary embodiment, GalNAc residues are added by the action of GalNAc transferase.
In an exemplary embodiment, the galactosyl acceptor is assembled by adding galactose residues to an appropriate acceptor, such as GlcNAc, attached to the peptide. The method comprises incubating the peptide to be modified with a reaction mixture containing an appropriate amount of a galactosyltransferase (e.g., gal β 1, 3 or gal β 1, 4) and an appropriate galactosyl donor (e.g., UDP-galactose). Allowing the reaction to proceed substantially to completion or alternatively allowing the reaction to terminate when a preselected amount of galactose residues has been added. Other methods of assembling selected sugar receptors will be apparent to those skilled in the art.
In another embodiment, the peptide-linked oligosaccharide is first "trimmed" in whole or in part to expose the sialyltransferase receptor or a moiety to which one or more appropriate residues may be added to obtain the appropriate receptor. For example, glycosyltransferases and endoglycosidases (see, e.g., U.S. Pat. No.5,716,812) are useful for attachment and trimming reactions. A detailed discussion of "pruning" and reconstituting N-linked and O-linked glycans is provided elsewhere.
In the discussion that follows, the method of the present invention is exemplified by the application of a modified sugar having a water-soluble polymer attached thereto. The emphasis in the discussion is for clarity. The skilled person will understand that the discussion is equally applicable to embodiments in which the modified sugar carries a therapeutic moiety, a biomolecule, etc.
An exemplary embodiment of the invention is set forth in fig. 14, wherein sugar residues are "trimmed" prior to addition of the modified sugar, which presents a scheme for trimming high mannose to the first generation of the dual-antennary structure. The modified saccharide carrying the water soluble polymer is then conjugated to one or more saccharide residues exposed by the "trimming". In one example, the water soluble polymer is added through a GlcNAc moiety conjugated to the water soluble polymer. The modified GlcNAc is attached to one or both terminal mannose residues of the biantennary structure. Alternatively, unmodified GlcNAc may be added to one or both ends of the branching species.
In another exemplary embodiment, the water soluble polymer is added to one or both terminal mannose residues of the biantennary structure through a modified sugar having a galactose residue, the modified sugar being conjugated to a GlcNAc residue added to the terminal mannose residue. Alternatively, unmodified Gal may be added to one or both terminal GlcNAc residues.
In yet a further example, the water soluble polymer is a modified sialic acid added to a Gal residue.
Another exemplary embodiment is set forth in fig. 15, which illustrates a similar scheme to that shown in fig. 14, wherein the high mannose structure is "clipped" to mannose (from which the biantennary structure branches). In one example, the water soluble polymer is added by GlcNAc modified with a polymer. Alternatively, unmodified GlcNAc is added to mannose, followed by the addition of Gal with the attached water-soluble polymer. In another embodiment, unmodified GlcNAc and Gal residues are added sequentially to mannose followed by the addition of a sialic acid moiety modified with a water-soluble polymer.
Fig. 16 presents a further exemplary embodiment utilizing a similar approach as shown in fig. 14, wherein the high mannose is "clipped" to the GlcNAc to which the first mannose is attached. The GlcNAc is conjugated to a Gal residue carrying a water soluble polymer. Alternatively, unmodified Gal is added to GlcNAc followed by sialic acid modified with a water soluble sugar. In yet further examples, the terminal GlcNAc is conjugated to Gal and the GlcNAc is subsequently fucosylated with a modified fucose carrying a water soluble polymer.
Fig. 17 is a scheme similar to that shown in fig. 14, wherein the high mannose is trimmed to the first GlcNAc attached to the Asn of the peptide. In one example, GlcNAc- (Fuc)aThe GlcNAc of the residue is conjugated to GlcNAc carrying a water soluble polymer. In another example, GlcNAc- (Fuc)aThe GlcNAc of the residue is modified with Gal carrying a water soluble polymer. In yet further embodiments, the GlcNAc is modified with Gal followed by Gal conjugation to sialic acid modified with a water soluble polymer.
Other exemplary embodiments are set forth in fig. 18-22. In each of the foregoing figures, an illustration of a series of reaction types in which the invention may be practiced is provided.
The examples given above provide a strength clarification of the approach presented herein. With the method of the invention, it is possible to "trim" and construct sugar residues with almost any desired structure. The modified sugar may be added to the end of the sugar moiety as set forth above, or it may be intermediate between the peptide core and the sugar end.
In an exemplary embodiment, the sialic acid present is removed from the glycopeptide with a sialidase, thereby exposing all or most of the galactosyl residues underneath. Alternatively, the peptide or glycopeptide is labelled with a galactose residue or an oligosaccharide residue ending with a galactose unit. After exposure or addition of galactose residues, an appropriate sialyltransferase is used to add the modified sialic acid. The method is summarized in scheme 12.
In a further method summarized in scheme 13, masked reactive functional groups are present on sialic acid. The masked reactive group is preferably unaffected by the conditions used to attach the modified sialic acid to the peptide. Following covalent attachment of the modified sialic acid to the peptide, the mask is removed and the peptide is conjugated to, for example, PEG, PPG, a therapeutic moiety, a biomolecule or other agent. The agent is conjugated to the peptide in a specific manner by its reaction with exposed reactive groups on the modified sugar residue.
Depending on the terminal saccharide of the oligosaccharide side chain of the glycopeptide, any modified saccharide with its appropriate glycosyltransferase can be used (table 4). As described above, the terminal sugar of the glycopeptide necessary for introducing the PEGylated or PPG-based structure may be introduced naturally during its expression, or it may be produced after expression using an appropriate glycosidase, glycosyltransferase or a mixture of glycosidase and glycosyltransferase.
TABLE 4 modified sugars
In a further exemplary embodiment, UDP-galactose-PEG is reacted with milk β 1, 4-galactosyltransferase to transfer the modified galactose to the appropriate terminal N-acetylglucosamine structure. The terminal GlcNAc residue on the glycopeptide may be produced during expression, as occurs in mammalian, insect, plant or fungal expression systems, but may also be produced by treating the glycopeptide with a desired sialidase and/or glycosidase and/or glycosyltransferase.
In another exemplary embodiment, a GlcNAc transferase, such as GnT-I-IV, is used to transfer PEGylated GlcNAc to a mannose residue of a glycopeptide. In still further exemplary embodiments, the N-and/or O-linked glycan structures are enzymatically removed from the glycopeptide to expose amino acids or terminal glycosyl residues that can then be conjugated to the modified saccharide. For example, an endoglucanase is used to remove the N-linked structure of a glycopeptide to expose the terminal GlcNAc, which appears as a GlcNAc-linked Asn on the glycopeptide. UDP-Gal-PEG and a suitable galactosyltransferase can be used to introduce a PEG-or PPG-galactose functional group onto the exposed GlcNAc.
In an alternative embodiment, the modified sugar is added directly to the peptide backbone using a glycosyltransferase, which is known to transfer sugar residues to the peptide backbone. This exemplary embodiment is set forth in scheme 14. Exemplary glycosyltransferases useful in practicing the invention include, but are not limited to, GalNAc transferase (GalNAc T1-14), GlcNAc transferase, fucosyltransferase, glucosyltransferase, xylosyltransferase, mannosyltransferase, and the like. Application of the present method enables the direct addition of modified saccharides to peptides lacking any saccharide or to existing glycopeptides. In both cases, the addition of the modified sugar occurs at a specific position on the peptide backbone as defined by the substrate specificity of the glycosyltransferase, rather than in a random manner as occurs during chemical modification of the peptide backbone of a protein. By processing the appropriate amino acid sequence into the peptide chain, a range of reagents can be introduced into a protein or glycopeptide that otherwise lacks the substrate peptide sequence for the glycosyltransferase.
In each of the exemplary embodiments set forth above, one or more additional chemical or enzymatic modification steps may be applied after conjugation of the modified saccharide to the peptide. In an exemplary embodiment, an enzyme (e.g., fucosyltransferase) can be used to attach a glycosyl unit (e.g., fucose) to a terminally modified sugar attached to a peptide. In another example, enzymatic reactions can be used to "cap" sites to which modified sugars cannot be conjugated. Alternatively, a chemical reaction is used to alter the structure of the conjugated modified saccharide. For example, the conjugated modified saccharide is reacted with an agent that stabilizes or destabilizes its linkage to the peptide component to which the modified saccharide is attached. In another example, the modified sugar moiety is deprotected after conjugation to the peptide. The skilled person will appreciate that there is a range of enzymatic and chemical procedures that can be used in the methods of the invention at a stage after the modified saccharide is conjugated to the peptide. Further modifications to the modified saccharide-peptide conjugates are within the scope of the invention.
Mannose-6-phosphate directed peptides
In an exemplary embodiment, the peptide is derivatized with at least one mannose-6-phosphate moiety. The mannose-6-phosphate moiety can direct the peptide to the lysosome of the cell, and can be used, for example, to direct a therapeutic protein to the lysosome for the treatment of lysosomal storage diseases.
Lysosomal storage diseases are a population of more than 40 disorders that are the result of a genetic defect encoding an enzyme that breaks down glycolipid or polysaccharide waste products in the lysosomes of cells. The products of the enzyme, such as sugars and lipids, can be recycled into new products. Each of these disorders results from inherited autosomal or X-linked recessive traits that affect the levels of enzymes in lysosomes. Typically, the affected enzymes in the cells and tissues of an affected individual have no biological or functional activity. Table 5 provides a list of representative storage disorders and enzyme deficiencies associated with the disease. In this disease, a defect in enzyme function results in a gradual systemic deposition of lipid or carbohydrate substrates in the lysosomes of body cells, ultimately leading to loss of organ function and death. The genetic etiology, clinical presentation, Molecular biology and incidence of lysosomal storage diseases are described in detail in Scriver et al, eds, The metabolic Molecular Basis of Inherited diseases, 7.sup.th ed., vol.ii, McGraw Hill, (1995).
TABLE 5 lysosomal storage diseases and associated enzyme deficiencies
MPS ═ mucopolysaccharide
De Duve first proposed that replacement of a missing lysosomal enzyme with an exogenous biologically active enzyme might be a viable approach to the treatment of lysosomal storage diseases (De Duve, Fed. Proc.23: 1045 (1964)). Since then, various studies have suggested that enzyme replacement therapy is beneficial for treating various lysosomal storage diseases. Has shown greatest success in individuals with type I gaucher disease, who have With exogenous enzyme (beta-glucocerebrosidase) prepared from placentaTM) Or recently recombinantly produced enzymes (Cerezymes)TM) And (6) carrying out treatment. Enzyme replacement has also been proposed to be beneficial for the treatment of bruise and other lysosomal storage diseases. See, e.g., Dawson et al, ped. res.7 (8): 684 (1973) (in vitro) and Mapes et al, Science 169: 987(1970) (in vivo). Clinical trials for enzyme replacement therapy have been reported in patients with infusion Fabry's disease with normal plasma (Mapes et al, Science 169: 987-. These studies show the potential to eliminate or significantly reduce pathological glycolipid storage through repeated enzyme substitutions. For example, in one study (Desnick et al, supra), intravenous injection of purified enzyme resulted in a temporary decrease in plasma levels of the stored lipid substrate globotriasylceramide.
Thus, there is a need in the art for methods of providing sufficient amounts of biologically active lysosomal enzymes, such as human α -galactosidase a, to defective cells. More recently, attempts have been made to meet these needs using recombinant methods, see e.g., u.s.pat. nos. 5,658,567, 5,580,757; bishop et al, proc.natl.acad.sci., USA, 83: 4859 4863 (1986); medin et al, proc.natl.acad.sci., USA 93: 7917 7922 (1996); novo, f.j., Gene Therapy 4: 488-492 (1997); ohshima et al, proc.natl.acad.sci., USA 94: 2540 2544 (1997); and Sugimoto et al, Human Gene Therapy 6: 905-915(1995). The present invention provides compositions and methods for delivering sufficient amounts of biologically active lysosomal peptides into defective cells through mannose-6-phosphate mediated targeting of therapeutic peptides into lysosomes.
Thus, in an exemplary embodiment, the present invention provides the peptides in table 7 derivatized with mannose-6-phosphate (fig. 24 and 25). The peptides may be recombinantly or chemically prepared. In addition, theThe peptide may be the entire native sequence, or may be modified, e.g., by truncation, extension, or it may include substitutions or deletions. Exemplary proteins that are reconstituted using the methods of the invention include glucocerebrosidase, beta-glucosidase, alpha-galactosidase a, acid-alpha-glucosidase (acid maltase). Representative modified peptides for clinical use include, but are not limited to CeredaseTM、CerezymeTMAnd FabryzymeTM. The glycosyl groups on the modified and clinically equivalent peptides can also be altered by the methods of the invention. Mannose-6-phosphate is attached to the peptide through a glycosyl linking group. In an exemplary embodiment, the glycosyl linking group is derived from sialic acid. Exemplary sialic acid derived glycosyl linking groups are set forth in table 3, wherein one or more "R" moieties are mannose-6-phosphate or a spacer group having one or more mannose-6-phosphate moieties attached thereto. The modified sialic acid moiety is preferably a terminal residue of an oligosaccharide attached to the peptide surface (figure 26).
In addition to mannose-6-phosphate, the peptides of the invention may be further derivatized with moieties such as water-soluble polymers, therapeutic moieties, or additional targeting moieties. Methods for attaching these and other groups are presented herein. In an exemplary embodiment, groups other than mannose-6-phosphate are attached to the peptide by derivatized sialic acid derivatives according to table 3, wherein one or more "R" moieties are groups other than mannose-6-phosphate.
In an exemplary embodiment, sialic acid moieties modified with Cbz-protected glycine-based linker arms are prepared. The corresponding nucleotide sugar is prepared and the Cbz group is removed by catalytic hydrogenation. The resulting nucleotide sugar has available reactive amines for contact with the activated mannose-6-phosphate derivative, thereby providing a mannose-6-phosphate derivatized nucleotide sugar useful in practicing the invention.
As shown in the following scheme (scheme 15), exemplary activated mannose-6-phosphate derivatives are formed as follows: the 2-bromo-benzyl-protected phosphotriester is converted in situ to the corresponding triflate and the triflate is reacted with a linker having a reactive oxygen-containing moiety, thereby forming an ether linkage between the sugar and the linker. The benzyl protecting group is removed by catalytic hydrogenation and the methyl ester of the linker is hydrolyzed to provide the corresponding carboxylic acid. The carboxylic acid may be activated by any method known in the art. One exemplary activation procedure relies on the conversion of a carboxylic acid to an N-hydroxysuccinimide ester.
In another exemplary embodiment, as shown in the following scheme (scheme 16), the N-acetylated sialic acid is converted to an amine by treatment of the pyruvyl moiety. Thus, the primary hydroxyl group is converted to a sulfonate ester and reacted with sodium azide. The azide is catalytically reduced to the corresponding amine. The sugar is then converted to its nucleotide analog and coupled via an amine group to mannose-6-phosphate derived from the linker arm prepared as described above.
Peptides for the treatment of lysosomal storage diseases can be derivatized with other targeting moieties including, but not limited to, transferrin (delivery of the peptide across the blood-brain barrier and to the endosome), carnitine (delivery of the peptide into muscle cells), and phosphates, such as diphosphate (targeting the peptide to bone and other calcified tissues). The targeting moiety and therapeutic peptide are conjugated by any of the methods discussed herein or otherwise known in the art.
In an exemplary embodiment, the targeting agent and the therapeutic peptide are coupled through a linker moiety. In this embodiment, at least one therapeutic peptide or targeting agent is coupled to the linker moiety through an intact glycosyl linking group according to the methods of the invention. In an exemplary embodiment, the linker moiety comprises a poly (ether) such as poly (ethylene glycol). In another exemplary embodiment, the linker moiety includes at least one bond that degrades in vivo, thereby releasing the therapeutic peptide from the targeting agent upon delivery of the conjugate to a target tissue or region of the body.
In another exemplary embodiment, the in vivo partitioning of the therapeutic moiety is altered by altering the glycoform on the therapeutic moiety without conjugating the therapeutic peptide to a targeting moiety. For example, uptake of therapeutic peptides by the reticuloendothelial system can be circumvented by capping the terminal galactose moiety of the glycosyl group with sialic acid (or derivatives thereof) (fig. 24 and 27). Sialylation of the capped Gal avoids uptake of the peptide by the hepatic Asialoglycoprotein (ASGP) receptor and can extend the half-life of the peptide relative to peptides that have no sialylation but only complex glycan chains.
II. peptides/glycopeptides of the invention
In one embodiment, the invention provides a composition comprising multiple copies of a single peptide having a primary trimannose core as the predominant glycan structure attached thereto. In a preferred embodiment, the peptide may be a therapeutic molecule. The native form of the peptide may comprise complex N-linked glycans or may be high mannose glycans. The peptide may be a mammalian peptide, and preferably a human peptide. In some embodiments, the peptide is selected from the group consisting of an immunoglobulin, erythropoietin, a tissue-type activating factor peptide, and the like (see fig. 28).
Exemplary peptides whose glycans can be reconstituted using the methods of the invention are set forth in fig. 28.
TABLE 6 preferred peptides for glycan reconstitution
TABLE 7 most preferred peptides for glycan reconstitution
A more detailed list of peptides and their sources for use in the present invention is set forth in figure 28.
Other exemplary peptides modified by the methods of the invention include members of the immunoglobulin family (e.g., antibodies, MHC molecules, T cell receptors, etc.), intercellular receptors (e.g., integrins, hormone or growth factor receptors, etc.), lectins, and cytokines (e.g., interleukins). Additional examples include tissue Type Plasminogen Activator (TPA), renin, blood coagulation factors such as factor VIII and factor IX, bombesin, thrombin, hematopoietic growth factors, colony stimulating factors, viral antigens, complement peptides, alpha 1-antitrypsin, erythropoietin, P-selectin glycopeptide ligand-1 (PSGL-1), granulocyte macrophage colony stimulating factor, antithrombin III, interleukins, interferons, peptides A and C, fibrinogen, herceptinTMLeptin, glycosidase, and the like. This list of peptides is exemplary and should not be considered exclusive. Rather, as is apparent from the disclosure provided herein, the methods of the present invention are applicable to any peptide in which any desired glycan structure can be formed.
The methods of the invention are also useful for modifying chimeric peptides, including but not limited to chimeric peptides comprising portions derived from immunoglobulins such as IgG.
The peptides modified by the methods of the invention may be synthetic or wild-type peptides, or they may be mutant peptides generated by methods well known in the art, such as site-directed mutagenesis. Glycosylation of peptides is typically N-linked or O-linked. An exemplary N-linkage is attachment of a modified sugar to the side chain of an asparagine residue. The tripeptide structures asparagine-X-serine and asparagine-X-threonine, where X is any amino acid except proline, are recognition sequences for the enzymatic attachment of a sugar moiety to an asparagine side chain. Thus, the presence of any of these tripeptide sequences in a peptide creates a potential glycosylation site. As described elsewhere, O-linked glycosylation refers to the attachment of a sugar (such as N-acetylgalactosamine, galactose, mannose, GlcNAc, glucose, fucose or xylose) to the hydroxyl side chain of a hydroxyl amino acid, preferably serine or threonine, although 5-hydroxyproline or 5-hydroxylysine may also be used.
Several exemplary embodiments of the invention are discussed below. Although several of these embodiments employ peptides having trade names and other specific peptides as exemplary peptides, these examples are not limited to any particular peptide. The exemplary embodiments below include all peptide equivalents and variants of any peptide. Such variants include, but are not limited to, the addition and deletion of N-linked and O-linked glycosylation sites and fusion proteins with added glycosylation sites. Those skilled in the art will appreciate that the following embodiments and the basic methods disclosed herein can be applied with equal success to many peptides.
In an exemplary embodiment, the present invention provides a method for modifying granulocyte colony stimulating factor (G-CSF). FIGS. 29A-29G set forth some examples of how this modification can be achieved using the methods disclosed herein. In FIG. 29B, the G-CSF peptide expressed in mammalian cells was trimmed with sialidase. The residues thus exposed can be modified by the addition of a sialic acid-poly (ethylene glycol) moiety (PEG moiety) with its appropriate donor and ST3Gal 1. Fig. 29C presents an exemplary scheme for modifying G-CSF peptides expressed on insect cells. The peptide is modified by the addition of a galactose moiety with its appropriate donor and galactosyltransferase. The galactose residues were functionalized with PEG by the action of ST3Gal1 and sialic acid-PEG derivatives. In FIG. 29D, the bacterially expressed G-CSF is contacted with an N-acetylgalactosamine donor and N-acetylgalactosamine transferase. The peptide is functionalized with PEG by a pegylated sialic acid donor and sialyltransferase. In FIG. 29E, G-CSF expressed by mammalian cells was contacted with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the glycans on the peptides, the ketones are derivatized with moieties such as hydrazine-or amine-PEG. In FIG. 29F, bacterially expressed G-CSF was reconstituted by contacting the peptide with an endo-GalNAc enzyme under conditions where it exerts a synthetic function rather than a hydrolytic function, thereby adding a PEG-Gal-GalNAc molecule from its activated derivative. FIG. 29G provides another pathway for reconstituting bacterially expressed G-CSF. The polypeptide is derivatized with PEGylated N-acetylgalactosamine residues by contacting the polypeptide with N-acetylgalactosamine transferase and an appropriate PEGylated N-acetylgalactosamine donor.
In another exemplary embodiment, the present invention provides a method for modifying interferon alpha-14C (IFN alpha 14C), as shown in FIGS. 30A-30N. Various forms of IFN α are disclosed elsewhere. In figure 30B, in mammalian cells expressed in IFN alpha 14C first with sialidase treatment to trim its sialic acid units, then with ST3Gal3 and PEG sialic acid donor PEG to the molecule. In figure 30C, so that N-acetyl glucosamine is first added in insect or fungal cells in expression of IFN alpha 14C, the reaction is through the use of N-acetyl glucosamine donor GnT-I and/or II effect. The polypeptide is then pegylated with a galactosyltransferase and a PEG-galactose donor. In figure 30D, in yeast expression of IFN alpha 14C first with Endo-H treatment to trim its glycosyl unit. The molecule was galactosylated with galactosyltransferase and galactose donor and then pegylated with ST3Gal3 and PEG-sialic acid donor. In figure 30F, ST3Gal3 and PEG-sialic acid donor to mammalian cell produced IFN alpha 14C modified to have (embedded) PEG part. In figure 30G, first using one or more GnT-I, II, IV and V and N-acetyl glucosamine donors will be N-acetyl glucosamine added to in insect or fungal cells in the expression of IFN alpha 14C. The protein is subsequently galactosylated with the appropriate donor and galactosyltransferase. IFN alpha 14C was then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In figure 30H, first with mannosidase treatment yeast produced IFN alpha 14C to trim the mannose group. N-acetylglucosamine is then added with an N-acetylglucosamine donor and one or more of GnT-I, II, IV and V. Further IFN alpha 14C with the appropriate donor and galactosyl transferase galactosylation. The polypeptide was then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In figure 30I, IFN α 14C expressed by NSO cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In figure 30J, the expression of mammalian cells by IFN alpha 14C PEG-sialylation with PEG-sialic acid donor and alpha 2, 8-sialyltransferase. In figure 30K, IFN α 14C produced by mammalian cells was first treated with sialidase to trim terminal sialic acid residues, and the molecule was then pegylated with trans-sialidase and pegylated sialic acid-lactose complex. In figure 30L, by sialic acid donor and alpha 2, 8-sialyltransferase by mammalian system expression of IFN alpha 14C sialylation. In figure 30M, first with the appropriate donor and GnT-I and/or II N-acetyl glucosamine is added to the insect or fungal cells in the expression of IFN alpha 14C. The molecule is then contacted with a galactosyltransferase and a galactose donor, the galactose donor being derivatized with reactive sialic acid through a linker, such that the polypeptide can be attached to the reactive sialic acid through the linker and a galactose residue. The polypeptide was then contacted with ST3Gal1 and transferrin, thereby linking it to transferrin via sialic acid residues. In figure 30N, IFN α 14C expressed in insect or fungal cells was first treated with an endoglucanase to trim glycosyl groups and then contacted with a galactosyltransferase and a galactose donor derivatized with reactive sialic acid through a linker such that the polypeptide could be attached to the reactive sialic acid through the linker and galactose residues. This molecule was then contacted with ST3Gal3 and transferrin, thereby linking it to transferrin via sialic acid residues.
In another exemplary embodiment, the present invention provides a method for modifying interferon alpha-2 a or 2b (IFN alpha), as shown in FIGS. 30O-30 EE. In figure 30P, the mammalian cell expression of IFN α first with sialidase treatment to trim glycosyl units, then with ST3Gal3 and pegylated sialic acid donor on its pegylation. In FIG. 30Q, IFN α expressed in insect cells was first galactosylated with the appropriate donor and galactosyltransferase, and then pegylated with ST3Gal1 and a pegylated sialic acid donor. Figure 30R provides another method of reconstituting IFN α in bacteria: PEGylated N-acetylgalactosamine is added to the protein using an appropriate donor and N-acetylgalactosamine transferase. In figure 30S, IFN α expressed in mammalian cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In figure 30T, used to synthesis but not hydrolysis function of the modified enzyme of N-acetylgalactosamine enzyme and PEG moiety derivatization of N-acetylgalactosamine donor on bacterial expression of IFN alpha PEG. In FIG. 30U, N-acetylgalactosamine is first added to IFN using an appropriate donor and N-acetylgalactosamine transferase, which is then PEGylated with sialyltransferase and a PEGylated sialic acid donor. In figure 30V, IFN α expressed in mammalian systems was first treated with sialidase to trim sialic acid residues and then pegylated with the appropriate donor and ST3Gal1 and/or ST3Gal 3. In figure 30W, IFN α expressed in mammalian systems was first treated with sialidase to trim sialic acid residues. The polypeptide was then contacted with ST3Gal1 and two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and a second sialic acid residue. The polypeptide was then contacted with ST3Gal3 and transferrin, thereby linking it to transferrin via sialic acid residues. In figure 30Y, IFN α expressed in mammalian systems was first treated with sialidase to trim sialic acid residues, which was then pegylated with ST3Gal1 and a PEG-sialic acid donor. In fig. 30Z, IFN α produced by insect cells was pegylated with galactosyltransferase and pegylated galactose donor. In figure 30AA, first with the appropriate donor and N-acetylgalactosamine transferase N-acetylgalactosamine to bacterial expression of IFN alpha. The protein was then pegylated with sialyltransferase and a PEG-sialic acid donor. In figure 30CC, IFN α expressed in bacteria was modified in another procedure: that is, PEGylated N-acetylgalactosamine donor is added to the protein by N-acetylgalactosamine transferase. In figure 30DD, IFN α expressed in bacteria was reconstituted in another protocol. The polypeptide is first contacted with N-acetylgalactosamine transferase and an N-acetylgalactosamine donor derivatized with reactive sialic acid via a linker, such that IFN α is attached to the reactive sialic acid via the linker and N-acetylgalactosamine. IFN α was then contacted with ST3Gal3 and asialoglycotransferrin, thereby linking it to transferrin via sialic acid residues. IFN alpha was then capped with sialic acid residues using ST3Gal3 and a sialic acid donor. An additional method of modifying bacterially expressed IFN α is disclosed in fig. 30EE, where IFN α is first exposed to NHS-CO-linker-SA-CMP and then linked to reactive sialic acid via a linker. It was subsequently conjugated with transferrin using ST3Gal3 and transferrin.
The method of reconstituting IFN ω is essentially the same as that provided herein for IFN, except that attachment of the glycan to the IFN peptide occurs at SEQ ID NO: 75 amino acid residue 101. The nucleotide and amino acid sequences of IFN ω are provided herein as SEQ ID NO: 74 and 75. Methods for the preparation and use of IFN ω are described in U.S. Pat. Nos. 4,917,887 and 5,317,089, and European patent No. 0170204-A.
In another exemplary embodiment, the present invention provides a method for modifying interferon beta (IFN β), as shown in FIGS. 31A-31S. In figure 31B, IFN β expressed in mammalian systems was first treated with sialidase to trim terminal sialic acid residues. The protein was then pegylated with ST3Gal3 and a pegylated sialic acid donor. Figure 31C is a modified by insect cells produced by IFN beta scheme. First, N-acetylglucosamine is added to IFN β using an appropriate donor and GnT-I and/or-II. The protein is then galactosylated with a galactose donor and a galactosyltransferase. Finally, IFN β was PEGylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 31D, IFN β expressed in yeast was first treated with Endo-H to trim its glycosyl chains, then galactosylated with a galactose donor and galactosyltransferase, and then PEGylated with ST3Gal3 and a PEGylated sialic acid donor. In figure 31E, mammalian cell produced IFN beta using ST3Gal3 and has PEG moiety derivatization of sialic acid donor PEG modified. In FIG. 31F, N-acetylglucosamine is first added to IFN β expressed in insect cells via one or more of GnT-I, II, IV and V using an N-acetylglucosamine donor, then galactosylated using a galactose donor and galactosyltransferase, and then PEGylated using ST3Gal3 and a PEG-sialic acid donor. In FIG. 31G, IFN β expressed in yeast was first treated with mannosidase to trim mannose units and then N-acetylglucosamine was added by one or more GnT-I, II, IV and V with an N-acetylglucosamine donor. The protein was further galactosylated with a galactose donor and galactosyltransferase, and then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 31H, IFN β expressed by mammalian cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In figure 31I, IFN beta expressed in mammalian systems was PEGylated with a PEG-sialic acid donor and alpha 2, 8-sialyltransferase. In fig. 31J, IFN β expressed by mammalian cells was first treated with sialidase to trim terminal sialic acid residues, and then pegylated with trans-sialidase and pegylated sialic acid donor. In fig. 31K, IFN β expressed in mammalian cells was first treated with sialidase to trim terminal sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. In fig. 31L, IFN β expressed in mammalian cells was first treated with sialidase and galactosidase to trim glycosyl chains, then galactosylated with a galactose donor and α -galactosyltransferase, and then pegylated with ST3Gal3 or sialyltransferase and PEG-sialic acid donor. In figure 31M, the IFN β expressed in mammalian cells was first treated with sialidase to trim the glycosyl units. It was then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. In figure 31N, IFN β expressed by mammalian cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In FIG. 31O, IFN β expressed in mammalian cells was sialylated using a sialic acid donor and α 2, 8-sialyltransferase. In FIG. 31Q, N-acetylglucosamine is first added to IFN β expressed in insect cells by one or more of GnT-I, II, IV and V with an N-acetylglucosamine donor, and is further PEGylated with a PEG-galactose donor and a galactosyltransferase. In FIG. 31R, IFN β expressed in yeast was first treated with an endo-glycanase to trim the glycosyl groups, then galactosylated with a galactose donor and galactosyltransferase, and then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 31S, IFN β expressed in mammalian systems is first contacted with ST3Gal3 and two reactive sialic acids linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and a second sialic acid residue. The polypeptide was then contacted with ST3Gal3 and desialylated transferrin, thereby linking it to transferrin via sialic acid residues. Then, further with sialic acid donor and ST3Gal3 IFN beta sialylation.
In another exemplary embodiment, the present invention provides a method for modifying factor VII or VIIa, as shown in fig. 32A-32D. In fig. 32B, factor VII or VIIa produced in a mammalian system was first treated with sialidase to trim terminal sialic acid residues, which were then pegylated with ST3Gal3 and a pegylated sialic acid donor. In fig. 32C, factor VII or VIIa expressed in mammalian cells was first treated with sialidase to trim terminal sialic acid residues, which were then pegylated with ST3Gal3 and a pegylated sialic acid donor. Further, the polypeptide was sialylated using ST3Gal3 and a sialic acid donor. Fig. 32D provides another modification to factor VII or VIIa produced by mammalian cells: that is, the polypeptide is first treated with sialidase and galactosidase to trim its sialic acid and galactose residues, then galactosylated with galactosyltransferase and a galactose donor, and then pegylated with ST3Gal3 and a pegylated sialic acid donor.
In another exemplary embodiment, the present invention provides methods for modifying coagulation factor IX, some examples of which are included in fig. 33A-33G. In fig. 33B, coagulation factor IX produced in mammalian cells was first treated with sialidase to trim terminal sialic acid residues and then pegylated with ST3Gal3 using PEG-sialic acid as donor. In fig. 33C, coagulation factor IX expressed in mammalian cells was first treated with sialidase to trim terminal sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and further sialylated with ST3Gal1 and a sialic acid donor. Another approach to reconstitute clotting factor IX produced by mammalian cells can be found in fig. 33D. The polypeptide was first treated with sialidase to trim terminal sialic acid residues, then galactosylated with galactose donor and galactosyltransferase, further sialylated with sialic acid donor and ST3Gal3, and then pegylated with pegylated sialic acid donor and ST3Gal 1. In fig. 33E, the coagulation factor IX expressed in the mammalian system was pegylated by sialylation catalyzed by ST3Gal3 with PEG-sialic acid donor. In fig. 33F, clotting factor IX expressed by mammalian cells was modified by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to which a reactive ketone was added. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. Fig. 33G provides additional methods of modifying coagulation factor IX. The polypeptide produced by mammalian cells is pegylated with a PEG-sialic acid donor and an alpha 2, 8-sialyltransferase.
In another exemplary embodiment, the present invention provides a method for modifying Follicle Stimulating Hormone (FSH). Fig. 34A-34J provide some examples. In fig. 34B, FSH was expressed in mammalian systems and modified by sialidase treatment to trim terminal sialic acid residues, which were subsequently pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 34C, FSH expressed in mammalian cells was first treated with sialidase to trim terminal sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. Figure 34D provides a scheme for modifying FSH expressed in a mammalian system. The polypeptide was treated with sialidase and galactosidase to trim its sialic acid and galactose residues, then galactosylated with a galactose donor and galactosyltransferase, and then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 34E, FSH expressed in mammalian cells was modified using the following procedure: i.e. the FSH was first treated with a sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. Fig. 34F provides another example of modifying FSH produced by mammalian cells: that is, the polypeptide is modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to which a reactive ketone has been added. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 34G, FSH expressed in the mammalian system was modified using another procedure: i.e. the polypeptide is reconstituted by addition of sialic acid with a sialic acid donor and an alpha 2, 8-sialyltransferase. In fig. 34H, FSH is expressed in insect cells and modified by the following procedure: i.e. adding N-acetylglucosamine to FSH via one or more GnT-I, II, IV and V, using an appropriate N-acetylglucosamine donor; the FSH is then pegylated with a PEG-galactose donor and galactosyltransferase. Figure 34I depicts a scheme for modifying FSH produced by yeast. According to this protocol, FSH is first treated with an endo-glycanase to trim glycosyl groups, then galactosylated with a galactose donor and galactosyltransferase, and then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 34J, FSH expressed in mammalian cells was first contacted with ST3Gal3 and two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to the reactive sialic acid via the linker and a second sialic acid residue. The polypeptide was then contacted with ST3Gal1 and desialylated Chorionic Gonadotropin (CG) produced in CHO, such that it was attached to CG via sialic acid residues. The FSH is then sialylated with a sialic acid donor and ST3Gal3 and/or ST3Gal 1.
In another exemplary embodiment, the present invention provides a method for modifying Erythropoietin (EPO). FIGS. 35A-35 AA set forth some examples relating to reconstituting wild-type or mutated EPO peptides. In fig. 35B, EPO is expressed in various mammalian systems and reconstituted by contacting the expressed protein with a sialidase to remove terminal sialic acid residues. The resulting peptide was contacted with sialyltransferase and CMP-sialic acid derivatized with PEG moieties. In FIG. 35C, EPO expressed in insect cells was reconstituted with GnT-I and/or-II as N-acetylglucosamine. Galactose was then added to the peptide using galactosyltransferase. PEG groups are added to the reconstituted peptide by contacting the peptide with sialyltransferase and CMP-sialic acid derivatized with PEG moieties. In FIG. 35D, EPO expressed in mammalian cell systems was reconstituted by the removal of terminal sialic acid residues by the action of sialyltransferase. The terminal galactose residue of the N-linked glycosyl group was "capped" with sialic acid using ST3Gal3 and a sialic acid donor. The terminal galactose residues of the O-linked glycans were functionalized with sialic acid carrying PEG moieties using appropriate sialic acid donors and ST3Gal 1. In fig. 35E, EPO expressed in mammalian cell systems was reconstituted by functionalization of N-linked glycosyl residues with PEG-derivatized sialic acid moieties. The peptide was contacted with ST3Gal3 and an appropriately modified sialic acid donor. In FIG. 35F, EPO expressed in an insect cell system, yeast or fungus is reconstituted by the addition of at least one N-acetylglucosamine residue added by contacting the peptide with an N-acetylglucosamine donor and one or more of GnT-I, GnT-II and GnT-V. The peptide is then pegylated by contacting it with a pegylated galactose donor and galactosyltransferase. In FIG. 35G, EPO expressed in an insect cell system, yeast or fungus is reconstituted by the addition of at least one N-acetylglucosamine residue with an appropriate N-acetylglucosamine donor and one or more of GnT-I, GnT-II and GnT-V. A galactosidase that was altered to function synthetically rather than hydrolytically was used to add an activated PEGylated galactose donor to the N-acetylglucosamine residue. In FIG. 35H, EPO expressed in insect cell systems, yeast or fungi was reconstituted by addition of at least one terminal N-acetylglucosamine-PEG residue. The peptide is contacted with GnT-I and an appropriate N-acetylglucosamine donor derivatized with a PEG moiety. In fig. 35I, EPO expressed in insect cell systems, yeast or fungi was reconstituted by addition of one or more terminal galactose-PEG residues. The peptide is contacted with GnT-I and an appropriate N-acetylglucosamine donor derivatized with a PEG moiety. The peptide is then contacted with a galactosyltransferase and an appropriate galactose donor modified with a PEG moiety. In FIG. 35J, EPO expressed in insect cell systems, yeast or fungi was reconstituted by the addition of a further terminal sialic acid-PEG residue. The peptide is contacted with an appropriate N-acetylglucosamine donor and GnT-I. The peptide is further contacted with a galactosyltransferase and an appropriate galactose donor. The peptide was then contacted with ST3Gal3 and an appropriate sialic acid donor derivatized with PEG moieties. In fig. 35K, EPO expressed in insect cell systems, yeast or fungi was reconstituted by addition of terminal sialic acid-PEG residues. The peptide is contacted with an appropriate N-acetylglucosamine donor and one or more of GnT-I, GnT-II and GnT-V. The peptide is then contacted with a galactosyltransferase and an appropriate galactose donor. The peptide was further contacted with ST3Gal3 and an appropriate sialic acid donor derivatized with PEG moieties. In fig. 35L, EPO expressed in insect cell systems, yeast or fungi was reconstituted by the addition of one or more terminal α 2, 6-sialic acid-PEG residues. The peptide is contacted with an appropriate N-acetylglucosamine donor and one or more of GnT-I, GnT-II and GnT-V. The peptide is further contacted with a galactosyltransferase and an appropriate galactose donor. The peptide is then contacted with an alpha 2, 6-sialyltransferase and an appropriately modified sialic acid donor. In fig. 35M, EPO expressed in mammalian cell systems was reconstituted by addition of one or more terminal sialic acid-PEG residues. The peptide is contacted with a sialidase to remove terminal sialic acid residues. The peptide is further contacted with a sialyltransferase and an appropriate sialic acid donor. The peptide is further contacted with a sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35N, EPO expressed in mammalian cell systems was reconstituted by addition of one or more terminal sialic acid-PEG residues. The peptide is contacted with a sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35O, EPO expressed in mammalian cell systems was reconstituted by adding one or more terminal α 2, 8-sialic acid-PEG residues to the main O-linked glycans. The peptide is contacted with an alpha 2, 8-sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35P, EPO expressed in mammalian cell systems was reconstituted by adding one or more terminal α 2, 8-sialic acid-PEG residues to O-linked and N-linked glycans. The peptide is contacted with an alpha 2, 8-sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35Q, EPO expressed in yeast or fungi was reconstituted by the addition of one or more terminal sialic acid-PEG residues. The peptides were contacted with mannosidase to remove terminal mannose residues. Next, the peptide is contacted with GnT-I and an appropriate N-acetylglucosamine donor. The peptide is further contacted with a galactosyltransferase and an appropriate galactose donor. The peptide is then contacted with a sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In FIG. 35R, EPO expressed in yeast or fungi was reconstituted by the addition of at least one terminal N-acetylglucosamine-PEG residue. The peptides were contacted with mannosidase to remove terminal mannose residues. The peptide is then contacted with GnT-I and an appropriate N-acetylglucosamine donor derivatized with a PEG moiety. In fig. 35S, EPO expressed in yeast or fungi was reconstituted by the addition of one or more terminal sialic acid-PEG residues. The peptide is contacted with mannosidase-I to remove the alpha 2 mannose residue. The peptide is further contacted with GnT-I and an appropriate N-acetylglucosamine donor. The peptide is then contacted with a galactosyltransferase and an appropriate galactose donor. The peptide is then contacted with a sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35U, EPO expressed in yeast or fungi was reconstituted by the addition of one or more galactose-PEG residues. The peptide was contacted with endo-H to trim to glycosyl groups. The peptide is then contacted with a galactosyltransferase and an appropriate galactose donor derivatized with a PEG moiety. In fig. 35V, EPO expressed in yeast or fungi was reconstituted by the addition of one or more terminal sialic acid-PEG residues. The peptide was contacted with endo-H to trim to glycosyl groups. The peptide is further contacted with a galactosyltransferase and an appropriate galactose donor. The peptide is then contacted with a sialyltransferase and an appropriate sialic acid donor derivatized with a PEG moiety. In fig. 35W, EPO expressed in insect cell systems was reconstituted by addition of terminal galactose-PEG residues. The peptides were contacted with mannosidase to remove terminal mannose residues. The peptide is then contacted with a galactosyltransferase and an appropriate galactose donor derivatized with a PEG moiety. In fig. 35Y, a mutated BPO, called a "novel erythropoiesis stimulating protein" or NESP, expressed in NSO murine myeloma cells is reconstituted by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 35Z, the mutant EPO, NESP, expressed in mammalian cell systems was reconstituted by the addition of one or more terminal sialic acid-PEG residues. PEG is added to glycosyl residues on glycans using PEG-modified sialic acid and α 2, 8-sialyltransferase. In fig. 35AA, NESP expressed in mammalian cell systems was reconstituted by addition of terminal sialic acid residues. Sialic acid is added to glycosyl residues using a sialic acid donor and an alpha 2, 8-sialyltransferase.
In another exemplary embodiment, the present invention provides a method of reconstituting granulocyte macrophage colony stimulating factor (GM-CSF), as shown in FIGS. 36A-36K. In FIG. 36B, GM-CSF expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, which were then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 36C, GM-CSF expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then further sialylated with a sialic acid donor and ST3Gal3 and/or ST3Gal 1. In fig. 36D, GM-CSF expressed in NSO cells was first treated with sialidase and α -galactosidase to trim glycosyl groups, then sialylated with the sialic acid donor and ST3Gal3, and then pegylated with ST3Gal1 and PEG-sialic acid donor. In fig. 36E, GM-CSF expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. In fig. 36F, GM-CSF expressed in mammalian cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In FIG. 36G, GM-CSF expressed in mammalian cells was sialylated using a sialic acid donor and α 2, 8-sialyltransferase. In FIG. 36I, modification of GM-CSF expressed in insect cells was performed by addition of N-acetylglucosamine with the appropriate donor and one or more of GnT-I, II, IV and V, and subsequent addition of PEGylated galactose with the appropriate donor and galactosyltransferase. In FIG. 36J, yeast expressed GM-CSF is first treated with an endo-glycanase and/or mannosidase to trim the glycosyl units, which are then PEGylated with a galactosyltransferase and a PEG-galactose donor. In FIG. 36K, GM-CSF expressed in mammalian cells was first treated with sialidase to trim sialic acid residues and then sialylated with ST3Gal1 and a sialic acid donor. The polypeptide was then contacted with ST3Gal1 and two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and a second sialic acid residue. The polypeptide was further contacted with ST3Gal3 and transferrin, thereby linking it to transferrin.
In another exemplary embodiment, the present invention provides a method for modifying interferon gamma (IFN γ). Fig. 37A to 37N contain some examples. In fig. 37B, IFN γ expressed in various mammalian cells was first treated with sialidase to trim terminal sialic acid residues, which were then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 37C, IFN γ expressed in mammalian systems was first treated with sialidase to trim terminal sialic acid residues. The polypeptide was then pegylated with ST3Gal3 and a PEG-sialic acid donor and further sialylated with ST3Gal3 and a sialic acid donor. In fig. 37D, mammalian cell-expressed IFN γ was first treated with sialidase and α -galactosidase to trim sialic acid and galactose residues. The polypeptide is then galactosylated with a galactose donor and a galactosyltransferase. IFN γ was then PEGylated with a PEG-sialic acid donor and ST3Gal 3. In fig. 37E, IFN γ expressed in mammalian systems was first treated with sialidase to trim terminal sialic acid residues. The polypeptide was then pegylated with ST3Gal3 and a PEG-sialic acid donor and further sialylated with ST3Gal3 and a sialic acid donor. Figure 37F depicts a modified in mammalian system expression of IFN gamma another method. The protein is modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 37G, IFN γ expressed in mammalian cells was reconstituted by addition of sialic acid with a sialic acid donor and α 2, 8-sialyltransferase. In FIG. 37I, IFN γ expression in insect or fungal cells was modified by addition of N-acetylglucosamine with the appropriate donor and one or more of GnT-I, II, IV and V. The protein is further modified by the addition of a PEG moiety with a pegylated galactose donor and galactosyltransferase. Figure 37J provides a method of modifying IFN γ expressed in yeast. The polypeptide is first treated with an endoglucanase to trim the sugar chain, which is then galactosylated with a galactose donor and a galactosyltransferase. Then IFN gamma was PEGylated with a PEGylated sialic acid donor and ST3Gal 3. In fig. 37K, IFN γ production by mammalian cells was modified as follows: the polypeptide is first contacted with ST3Gal3 and a sialic acid donor derivatized with a reactive galactose via a linker, such that the polypeptide can be attached to the reactive galactose via the linker and sialic acid residues. The polypeptide is then contacted with a galactosyltransferase and transferrin pre-treated with an endo-glycanase, thereby linking it to transferrin via a galactose residue. In the protocol depicted by fig. 37L, IFN γ expressed in the mammalian system was modified by the action of ST3Gal 3: even though pegylated sialic acid was transferred from the appropriate donor to IFN γ. FIG. 37M is an example of a modification of IFN γ expressed in insect or fungal cells, wherein PEGylation of the polypeptide is achieved by transfer of PEGylated N-acetylglucosamine from a donor onto IFN γ with GnT-I and/or II. In figure 37N, IFN γ expressed in mammalian systems was reconstituted by addition of pegylated sialic acid with the appropriate donor and α 2, 8-sialyltransferase.
In another exemplary embodiment, the present invention provides methods for modifying alpha1Methods of anti-trypsin (alpha 1-protease inhibitors). Some such examples can be found in FIGS. 38A-38O. In FIG. 38B, α expressed in various mammalian cells was first treated with sialidase1Antitrypsin to trim sialic acid residues. The pegylated sialic acid residue is then added with an appropriate donor such as CMP-SA-PEG and a sialyltransferase such as ST3Gal 3. α is exemplified in FIG. 38C1Another approach to antitrypsin modification. First, alpha expressed in mammalian systems is treated with sialidase1Antitrypsin to trim sialic acid residues. Sialic acid residues derivatized with PEG are then added with an appropriate donor and sialyltransferase such as ST3Gal 3. Subsequently, the molecule was further modified by the addition of sialic acid residues with a sialic acid donor and ST3Gal 3. Optionally, the alpha expressed by the mammalian cells is first treated with sialidase and alpha-galactosidase1Antitrypsin to trim sialic acid and alpha-linked galactose residues. The polypeptide is then galactosylated with galactosyltransferase and an appropriate galactose donor. Further, sialic acid derivatized with PEG was added by ST3Gal3 action using a pegylated sialic acid donor. In FIG. 38D, α expressed in mammalian systems was first treated with sialidase 1Antitrypsin, whereby the terminal sialic acid residues are trimmed. PEG is then added to the N-linked glycosyl residues by the action of ST3Gal3, which ST3Gal3 mediates PEGylated sialic acid from a donor such as CMP-SA-PEG to α1Anti-trypsin metastasis. Subsequently more sialic acid residues were attached with sialic acid donors and ST3Gal 3. FIG. 38E depicts reconstructing α1Another method of antitrypsin is provided. By passingModification of alpha expressed in mammalian cells by capping appropriate terminal residues with sialic acid donors1Antitrypsin, the sialic acid donor is modified with levulinic acid, thereby adding a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In FIG. 38F, α is disclosed1Another method of antitrypsin modification. Alpha from mammalian expression systems1Antitrypsin is reconstituted by addition of sialic acid with a sialic acid donor and an alpha 2, 8-sialyltransferase. In FIG. 38H, α1Antitrypsin is expressed in insect or yeast cells and is reconstituted by contacting the polypeptide with UDP-N-acetylglucosamine and one or more GnT-I, II, IV or V to add a terminal N-acetylglucosamine residue. The polypeptide is then modified with a PEG moiety using a pegylated galactose donor and galactosyltransferase. In FIG. 38I, alpha expressed in yeast cells was first treated with an endoglucanase 1Antitrypsin to trim the sugar chains. It is then galactosylated with galactosyltransferase and a galactose donor. The polypeptide was then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 38J, α1Antitrypsin is expressed in mammalian systems. The polypeptide is first contacted with ST3Gal3 and a sialic acid donor derivatized with a reactive galactose via a linker, such that the polypeptide can be attached to the reactive galactose via the linker and sialic acid residues. The polypeptide is then contacted with a galactosyltransferase and transferrin pre-treated with an endo-glycanase, thereby linking it to transferrin via a galactose residue. In FIG. 38L, alpha expressed in yeast was first treated with endoglucanase1Antitrypsin to trim the glycosyl groups. The protein is then pegylated with galactosyltransferase and a galactosyl donor with a PEG moiety. In FIG. 38M, alpha expressed in plant cells was treated with hexosaminidase, mannosidase, and xylosidase1Antitrypsin to trim the glycosyl chain and subsequently N-acetylglucosaminyltransferase and appropriate donor Modification with N-acetylglucosamine derivatized with a PEG moiety. In FIG. 38N, α expressed in mammalian cells1Antitrypsin was modified by the addition of pegylated sialic acid residues with ST3Gal3 and a sialic acid donor derivatized with PEG.
In another exemplary embodiment, the present invention provides methods for modifying glucocerebrosidase (beta-glucosidase, Cerezyme)TMOr CeredaseTM) The method (2) is as shown in FIGS. 39A to 39K. In FIG. 39B, Cerezyme expressed in mammalian systems was first treated with sialidaseTMTo trim the terminal sialic acid residues, which were then pegylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 39C, Cerezyme expressed in mammalian cells was first treated with sialidaseTMTo trim sialic acid residues, mannose-6-phosphate was then attached with ST3Gal3 and reactive sialic acid derivatized with mannose-6-phosphate, and then sialylated with ST3Gal3 and a sialic acid donor. Optionally, NSO cell-expressed Cerezyme is first treated with sialidase and galactosidaseTMTo trim the glycosyl groups, which are then galactosylated with the galactose donor and alpha-galactosyltransferase. The mannose-6-phosphate moiety can then be added to the molecule with ST3Gal3 and a reactive sialic acid derivatized with mannose-6-phosphate. In FIG. 39D, Cerezyme expressed in mammalian cells was first treated with sialidase TMTo trim sialic acid residues, which were then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and sialic acid. In FIG. 39E, Cerezymes expressed in mammalian cells were modified by capping the appropriate terminal residues with sialic acid donorsTMThe sialic acid donor is modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with moieties such as, for example, one or more mannose-6-phosphate groups. In FIG. 39F, Cerezyme expressed in mammalian cellsTMIs sialylated using a sialic acid donor and an alpha 2, 8-sialyltransferase. In the case of the graph of figure 39H,cerezymes expressed in insect cells using appropriate donors and one or more of GnT-I, II, IV and VTMTo which N-acetylglucosamine is added, and then PEGylated with a galactosyltransferase and a PEG-galactose donor. In FIG. 39I, Cerezyme expressed in yeast cells was first treated with an endoglucanaseTMTo trim the glycosyl groups, then galactosylate them with a galactose donor and galactosyltransferase, and then PEGylate the polypeptide with ST3Gal3 and a PEG-sialic acid donor. In FIG. 39JK, Cerezyme expressed in mammalian cells was first allowed to express TMContacting ST3Gal3 with two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and a second sialic acid residue. The polypeptide was then contacted with ST3Gal3 and desialylated transferrin, thereby linking it to transferrin. The polypeptide was then sialylated using a sialic acid donor and ST3Gal 3.
In another exemplary embodiment, the present invention provides a modification method for tissue Type Plasminogen Activator (TPA) and mutants thereof. Several specific modifications are given in FIGS. 40A-40W. FIG. 40B illustrates a modification procedure: after TPA is expressed in mammalian cells, it is treated with one or more mannosidases and sialidases to trim mannosyl and/or sialic acid residues. Terminal N-acetylglucosamine is then added by contacting the polypeptide with an appropriate N-acetylglucosamine donor and one or more of GnT-I, II, IV and V. TPA was further galactosylated with a galactose donor and galactosyltransferase. PEG was then attached to the molecule by sialylation of the sialic acid donor catalyzed by ST3Gal3 and derivatized with PEG moieties. In FIG. 40C, TPA is expressed in insect or fungal cells. The modification comprises the steps of adding N-acetylglucosamine with an appropriate N-acetylglucosamine donor and GnT-I and/or II; galactosylation with a galactose donor and a galactosyltransferase; and attachment of PEG by sialylation of sialic acid donors derivatized with ST3Gal3 and PEG moieties. In FIG. 40D, TPA was expressed in yeast and subsequently treated with endoglucanase to trim sugar chains. The polypeptide is further pegylated by the action of galactosyltransferase, which catalyzes the transfer of PEG-galactose from the donor to TPA. In FIG. 40E, TPA is expressed in insect or yeast cells. The polypeptide is then treated with alpha-and beta-mannosidase enzymes to trim the terminal mannose residues. Further, the PEG moiety is attached to the molecule by transfer of PEG-galactose from a suitable donor to TPA, the transfer being mediated by galactosyltransferase. Figure 40F provides different methods of modifying TPA obtained from insect or yeast systems: the polypeptide is reconstituted by addition of N-acetylglucosamine with an N-acetylglucosamine donor and GnT-I and/or II, followed by PEGylation with a galactosyltransferase and a PEGylated galactose donor. FIG. 40G provides an alternative to reconstituting TPA expressed in insect or yeast cells. The terminal N-acetylglucosamine is added with an N-acetylglucosamine donor and GnT-I and/or II. A galactosidase modified to function synthetically rather than hydrolytically is used to add PEGylated galactose from the appropriate donor to the N-acetylglucosamine residue. In fig. 40I, TPA expressed in mammalian systems was first treated with sialidase and galactosidase to modify sialic acid and galactose residues. The polypeptide is further modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid, thereby adding a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 40J, TPA expressed in mammalian systems was reconstituted according to the following protocol: first, the polypeptide is treated with α -and β -mannosidase enzymes to trim the terminal mannose residues; sialic acid residues were then attached to the terminal galactose residues with a sialic acid donor and ST3Gal 3; further, TPA is pegylated by the galactosylation-catalyzed transfer of galactose from the donor to the N-acetylglucosamine residue by galactosyltransferase. In fig. 40K, TPA was expressed in the plant system. The modification procedure in this example is as follows: first treating TPA with hexosaminidase, mannosidase and xylosidase to trim its glycosyl groups; the PEGylated N-acetylglucosamine is then added to TPA using an appropriate donor and N-acetylglucosamine transferase. In fig. 40M, TPA mutants expressed in mammalian cells (TNK TPA) were reconstituted. First trimming the terminal sialic acid residues with sialidase; ST3Gal3 was then used to transfer the pegylated sialic acid from the donor to TNK TPA, thereby pegylating the polypeptide. In fig. 40N, TNK TPA expressed in mammalian systems was first treated with sialidase to trim terminal sialic acid residues. The protein was then PEGylated with CMP-SA-PEG as donor and ST3Gal3 and further sialylated with sialic acid donor and ST3Gal 3. In fig. 40O, NSO cells expressing TNK TPA were first treated with sialidase and α -galactosidase to trim terminal sialic acid and galactose residues. TNK TPA was then galactosylated with a galactose donor and galactosyltransferase. The final step in this reconstitution protocol is the transfer of PEG-partially derivatized sialic acid from the donor to TNK TPA with a sialyltransferase such as ST3Gal 3. In fig. 40Q, TNK TPA was expressed in mammalian systems and was first treated with sialidase to trim terminal sialic acid residues. The protein was then pegylated with ST3Gal3 and a pegylated sialic acid donor. The protein was then sialylated with a sialic acid donor and ST3Gal 3. In fig. 40R, TNK TPA expressed in mammalian systems was modified by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 40S, TNK TPA expressed in mammalian cells was modified by different methods: the polypeptide is reconstituted by addition of sialic acid with a sialic acid donor and an alpha 2, 8-sialyltransferase. In FIG. 40U, TNK TPA expressed in insect cells was reconstituted by addition of N-acetylglucosamine with the appropriate donor and one or more of GnT-I, II, IV and V. The protein is further modified by the addition of a PEG moiety with a pegylated galactose donor and galactosyltransferase. In FIG. 40V, TNK TPA is expressed in yeast. The polypeptide is first treated with an endo-glycanase to trim its glycosyl chains and then pegylated with PEG-derivatized galactose donor and galactosyltransferase. In fig. 40W, TNK TPA was produced in a mammalian system. The polypeptide is first contacted with ST3Gal3 and a sialic acid donor derivatized with a reactive galactose via a linker, such that the polypeptide can be attached to the reactive galactose via the linker and sialic acid residues. The polypeptide was then contacted with galactosyltransferase and an anti-tnfaig chimera produced in CHO, linking it to the chimera through a galactose residue.
In another exemplary embodiment, the present invention provides a method for modifying interleukin-2 (IL-2). Fig. 41A to 41G provide some examples. FIG. 41B provides a two-step modification: IL-2 produced by mammalian cells was first treated with sialidase to trim terminal sialic acid residues, which were then PEGylated with ST3Gal3 and a PEGylated sialic acid donor. In FIG. 41C, insect cells expressing IL-2 were first modified by galactosylation with a galactose donor and galactosyltransferase. IL-2 was subsequently PEGylated with ST3Gal3 and a PEGylated sialic acid donor. In FIG. 41D, IL-2 expressed in bacteria was modified with N-acetylglucosamine using an appropriate donor and N-acetylglucosaminyltransferase, followed by a PEGylation step with a PEG-sialic acid donor and sialyltransferase. FIG. 41E provides another approach to modifying IL-2 produced by mammalian systems. The polypeptide is modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. FIG. 41F illustrates an example of reconstitution of IL-2 expressed by E.coli. The polypeptide is pegylated with a reactive N-acetylglucosamine complex derivatized with a PEG group and an enzyme, wherein the enzyme is modified such that it functions as a synthetase rather than a hydrolase. In FIG. 41G, the bacterial expression of IL-2 is through the use of the appropriate donor and N-acetylgalactosamine transferase add PEG N-acetylgalactosamine modification.
In another exemplary embodiment, the present invention provides a method for modifying factor VIII, as shown in figures 42A-42N. In fig. 42B, factor VIII expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, which was then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 42C, coagulation factor VIII expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and an appropriate donor, and further sialylated with ST3Gal1 and a sialic acid donor.
In fig. 42E, mammalian cell-produced factor VIII was modified by one-step pegylation with ST3Gal3 and a pegylated sialic acid donor. Figure 42F provides another example of modifying coagulation factor VIII expressed in mammalian cells. The protein was pegylated with ST3Gal1 and a pegylated sialic acid donor. In fig. 42G, the mammalian cell expressed coagulation factor VIII was reconstituted according to the following protocol: i.e.by PEGylation with alpha 2, 8-sialyltransferase and a PEG-sialic acid donor. In fig. 42I, factor VIII expressed by mammalian cells is modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to which a reactive ketone has been added. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 42J, mammalian cell-expressed factor VIII was first treated with Endo-H to trim to glycosyl groups. PEGylation was then performed with galactosyltransferase and a PEG-galactose donor. In fig. 42K, coagulation factor VIII expressed in mammalian systems was first sialylated with ST3Gal3 and a sialic acid donor, then treated with Endo-H to trim the glycosyl groups, and then pegylated with galactosyltransferase and a PEG-galactose donor. In fig. 42L, factor VIII expressed in mammalian systems is first treated with mannosidase to trim terminal mannose residues, then an N-acetylglucosamine group is added with the appropriate donor and GnT-I and/or II, and then pegylated with galactosyltransferase and a PEG-galactose donor. In FIG. 42M, factor VIII expressed in mammalian cells was first treated with mannosidase to trim mannose units and then N-acetylglucosaminyl transferase and the appropriate donor was used to add N-acetylglucosamine groups. Further galactosylation was performed with galactosyltransferase and galactose donor, followed by sialylation with ST3Gal3 and sialic acid donor. In fig. 42N, factor VIII was produced by mammalian cells and modified as follows: first treated with mannosidase to trim the terminal mannose residues. The PEGylated N-acetylglucosamine group is then added with GnT-I and an appropriate PEGylated N-acetylglucosamine donor.
In another exemplary embodiment, the present invention provides a method for modifying urokinase, as shown in FIGS. 43A-43M. In fig. 43B, urokinase expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, which were then pegylated with ST3Gal3 and a pegylated sialic acid donor. In fig. 43C, urokinase expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a pegylated sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. Optionally, urokinase expressed in mammalian systems is first treated with sialidase and galactosidase to trim glycosyl chains, then galactosylated with a galactose donor and a-galactosyltransferase, and then pegylated with ST3Gal3 or sialyltransferase and PEG-sialic acid donor. In fig. 43D, urokinase expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and further sialylated with ST3Gal3 and a sialic acid donor. In fig. 43E urokinase expressed in mammalian cells was modified by capping the appropriate terminal residue with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In FIG. 43F, urokinase expressed in mammalian cells was sialylated using a sialic acid donor and α 2, 8-sialyltransferase. In FIG. 43H, urokinase expressed in insect cells was modified by the following procedure: first, adding N-acetylglucosamine to the polypeptide using an appropriate N-acetylglucosamine donor and one or more of GnT-I, II, IV and V; the pegylated galactose was then added with galactosyltransferase and a PEG-galactose donor. In FIG. 43I, urokinase expressed in yeast was first treated with an endo-glycanase to trim glycosyl groups, then galactosylated with a galactose donor and galactosyltransferase, and then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 41J, urokinase expressed in mammalian cells was first contacted with ST3Gal3 and two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and a second sialic acid residue. The polypeptide is then contacted with ST3Gal1 and a desialylated urokinase produced in a mammalian cell, thereby linking it to a second urokinase molecule. The whole molecule was then further sialylated with sialic acid donors and ST3Gal1 and/or ST3Gal 3. In fig. 43K, isolated urokinase was first treated with sulfohydrolase (sulfohydrolase) to remove sulfate groups, and then pegylated with sialyltransferase and PEG-sialic acid donor. In FIG. 43LM, isolated urokinase was first treated with sulfohydrolase and hexosaminidase to remove sulfate groups and hexosamine groups, and then PEGylated with galactosyltransferase and PEG-galactose donor.
In another exemplary embodiment, the present invention provides a method for modifying DNase I as shown in fig. 44A-44J. In fig. 44B DNase I is expressed in mammalian systems and modified with the following steps: first, the protein is treated with sialidase to trim sialic acid residues; it was then pegylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 44C, dnase i expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. Optionally, DNase I expressed in mammalian systems is first exposed to sialidase and galactosidase to trim glycosyl groups, then galactosylated with a galactose donor and a-galactosyltransferase, and then pegylated with ST3Gal3 or sialyltransferase and PEG-sialic acid donor. In fig. 44D, dnase i expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, then pegylated with ST3Gal3 and a PEG-sialic acid donor, and then sialylated with ST3Gal3 and a sialic acid donor. In fig. 44E DNase I expressed in mammalian cells was modified by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 44F DNase I expressed in mammalian cells was sialylated with sialic acid donor and α 2, 8-sialyltransferase. In FIG. 44H, N-acetylglucosamine is first added to DNase I expressed in insect cells using an appropriate donor and one or more of GnT-I, II, IV and V. The protein was then pegylated with galactosyltransferase and a PEG-galactose donor. In FIG. 44I, DNase I expressed in yeast was first treated with an endo-glycanase to trim glycosyl units, then galactosylated with a galactose donor and galactosyltransferase, and then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In fig. 44JK DNase I expressed in mammalian cells was first contacted with ST3Gal3 and two reactive sialic acid residues linked by a linker, thereby attaching the polypeptide to one reactive sialic acid via the linker and the second sialic acid residue. The polypeptide is then contacted with ST3Gal1 and a desialylated alpha-1-protease inhibitor such that it is linked to the inhibitor via a sialic acid residue. The polypeptide is then further sialylated with an appropriate donor and ST3Gal1 and/or ST3Gal 3.
In another exemplary embodiment, the present invention provides a method for modifying post-mutation insulin containing an N-glycosylation site, as shown in FIGS. 45A-45L. In FIG. 45B, insulin expressed in a mammalian system was first treated with sialidase to trim sialic acid residues, which were then PEGylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 45C, insulin expressed in insect cells was modified by the addition of PEGylated N-acetylglucosamine with the appropriate donor and GnT-I and/or II. In FIG. 45D, insulin expressed in yeast was first treated with Endo-H to trim glycosyl groups, then PEGylated with galactosyltransferase and a PEG-galactose donor. In fig. 45F, insulin expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, which was then pegylated with ST3Gal1 and a PEG-sialic acid donor. In fig. 45G, insulin expressed in insect cells was modified by the addition of pegylated galactose with the appropriate donor and galactosyltransferase. In FIG. 45H, N-acetylgalactosamine is first added to insulin expressed in bacteria using an appropriate donor and N-acetylgalactosamine transferase. The polypeptide is then pegylated with sialyltransferase and a PEG-sialic acid donor. In fig. 45J, insulin expressed in bacteria was modified by different methods: that is, the appropriate donor and N-acetylgalactosamine transferase to protein adding PEG N-acetylgalactosamine. In fig. 45K, insulin expressed in bacteria was modified according to another scheme: the polypeptide is first contacted with N-acetylgalactosamine transferase and reactive N-acetylgalactosamine, which is derivatized with reactive sialic acid via a linker, such that the polypeptide can be attached to the reactive sialic acid via the linker and the N-acetylgalactosamine. The polypeptide is then contacted with ST3Gal3 and desialylated transferrin and thus can be linked to transferrin, and the polypeptide is then sialylated using ST3Gal3 and a sialic acid donor. In FIG. 45L, insulin expressed in bacteria was modified by another method: the polypeptide is first exposed to NHS-CO-linker-SA-CMP and linked to reactive sialic acid residues via a linker. The polypeptide was then conjugated to transferrin with ST3Gal3 and desialylated transferrin. The polypeptide was then further sialylated with ST3Gal3 and a sialic acid donor.
In another exemplary embodiment, the present invention provides a method for modifying hepatitis B antigens (M antigens-PRES and S), as shown in FIGS. 46A-46K. In fig. 46B, the M-antigen was expressed in a mammalian system and modified by initial treatment with sialidase to trim the terminal sialic acid residues and subsequent conjugation to lipid a with ST3Gal3 and reactive sialic acid linked to lipid a by a linker. In fig. 46C, M-antigen expressed in mammalian cells was first treated with sialidase to trim terminal sialic acid residues, then conjugated to tetanus toxin with ST3Gal1 and reactive sialic acid residues linked to the toxin by a linker, and then sialylated with ST3Gal3 and a sialic acid donor. In FIG. 46D, M-antigen expressed in mammalian systems was first treated with galactosidase to trim galactosyl residues, which were then sialylated with ST3Gal3 and a sialic acid donor. Sialic acid derivatized with KLH was then added to the polypeptide using ST3Gal1 and an appropriate donor. In fig. 46E, the yeast expressed M-antigen was first treated with mannosidase to trim the mannosyl residues and then conjugated to diphtheria toxin with GnT-I and an N-acetylglucosamine donor linked to diphtheria toxin. In fig. 46F, M-antigen expressed by mammalian cells was modified by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to add a reactive ketone to the sialic acid donor. After addition to the glycosyl residues of the peptide, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 46G, M-antigen obtained from mammalian systems was reconstituted by sialylation with sialic acid donor and poly α 2, 8-sialyltransferase. In FIG. 46I, M-antigen expressed in insect cells was conjugated to Neisseria proteins using GnT-II and an appropriate N-acetylglucosamine donor linked to Neisseria (Neisseria) proteins. In FIG. 46J, M-antigen expressed in yeast was first treated with an endo-glycanase to trim glycosyl chains and then conjugated to a Neisseria protein with a galactosyltransferase and appropriate galactose donor linked to the Neisseria protein. FIG. 46K is another example of modification of M-antigen expressed in yeast. The polypeptide is first treated with mannosidase to trim the terminal mannosyl residues and then N-acetylglucosamine is added thereto using GnT-I and/or II. Subsequently, the polypeptide is galactosylated with a galactose donor and a galactosyltransferase, which is then capped with sialic acid residues with a sialyltransferase and a sialic acid donor.
In another exemplary embodiment, the present invention provides a method for modifying human growth hormone (N, V and variants thereof) as shown in figures 47A-47K. In FIG. 47B, human growth hormone produced by mammalian cells, either containing an N-attachment site after mutation or a naturally occurring isoform with an N-attachment site (i.e., placental enzyme), was first treated with sialidase to trim terminal sialic acid residues and then PEGylated with ST3Gal3 and a PEGylated sialic acid donor. In FIG. 47C, human growth hormone expressed in insect cells was modified by the addition of PEGylated N-acetylglucosamine with GnT-I and/or II and an appropriate PEGylated N-acetylglucosamine donor. In FIG. 47D, human growth hormone was expressed in yeast, treated with Endo-H to trim glycosyl groups, and further PEGylated with galactosyltransferase and a PEGylated galactose donor. In fig. 47F, the human growth hormone-mucin fusion protein expressed in the mammalian system was modified by initial treatment with sialidase to trim sialic acid residues and subsequent pegylation thereof with PEG-sialic acid donor and ST3Gal 1. In fig. 47G, the human growth hormone-mucin fusion protein expressed in insect cells was reconstituted by pegylation with galactosyltransferase and a pegylated galactose donor. In FIG. 47H, human growth hormone-mucin fusion proteins were produced in bacteria. N-acetylgalactosamine is first added to a fusion protein by the action of N-acetylgalactosamine transferase using an N-acetylgalactosamine donor, and then the fusion protein is PEGylated with a PEG-sialic acid donor and sialyltransferase. Figure 47I depicts another scheme for modifying bacterially expressed human growth hormone-mucin fusion proteins: the fusion protein is PEGylated with the PEGylated N-acetylgalactosamine donor by the action of N-acetylgalactosamine transferase. Figure 47J provides a further reconstitution protocol for human growth hormone-mucin fusion proteins. The fusion protein is first contacted with N-acetylgalactosamine transferase and an N-acetylgalactosamine donor derivatized with reactive sialic acid via a linker, such that the fusion protein can be attached to the reactive sialic acid via the linker and N-acetylgalactosamine. The fusion protein is then contacted with sialyltransferase and desialylated transferrin, thus linking it to transferrin via sialic acid residues. The fusion protein was then capped with sialic acid residues using ST3Gal3 and a sialic acid donor. In fig. 47K, another scheme for modifying human growth hormone (N) produced in bacteria is provided. The polypeptide is first contacted with NHS-CO-linker-SA-CMP and coupled to reactive sialic acids via a linker. The polypeptide was then contacted with ST3Gal3 and desialylated transferrin and conjugated to transferrin via sialic acid residues. The polypeptide was then sialylated using ST3Gal3 and a sialic acid donor.
In another exemplary embodiment, the invention provides methods for modifying TNF receptor IgG fusion proteins (TNFR-IgG or Enbrel)TM) The method (5) is shown in FIGS. 48A to 48G. FIG. 48B illustrates a modification procedure in which TNFR-IgG expressed in a mammalian system is first sialylated with a sialic acid donor and sialyltransferase ST3Gal 1; then galactosylating the fusion protein with galactose donor and galactosyltransferase; the fusion protein was then pegylated by ST3Gal3 and a sialic acid donor derivatized with PEG. In FIG. 48C, TNFR-IgG expressed in mammalian cells was first treated with sialidase to trim sialic acid residues. The PEG moiety was subsequently attached to the TNFR-IgG by transferring the pegylated sialic acid from the donor to the fusion protein in a reaction catalyzed by ST3Gal 1. In FIG. 48D, TNFR-IgG is expressed in mammalian systems and modified by the addition of PEG by a galactosylation procedure mediated by galactosyltransferases with a PEG-galactose donor. In FIG. 48E, TNFR-IgG was expressed in a mammalian system. The first step in the reconstitution of the fusion protein is the addition of an O-linked sialic acid residue with a sialic acid donor and the sialyltransferase ST3Gal 1. Subsequently, pegylated galactose was added to the fusion protein with galactosyltransferase and an appropriate galactose donor with PEG moiety. In fig. 48F, TNFR-IgG expressed in mammalian cells was modified by capping the appropriate terminal residues with a sialic acid donor modified with levulinic acid to which a reactive ketone was added. After addition to the glycosyl residues of the fusion protein, the ketone is derivatized with a moiety such as hydrazine-or amine-PEG. In fig. 48G, TNFR-IgG expressed in mammalian cells was reconstituted with α 2, 8-sialyltransferase, which catalyzes a reaction that transfers pegylated sialic acid from a sialic acid donor with a PEG moiety to a fusion protein.
In another exemplary embodiment, the present invention provides a method for generating HerceptinTMThe method of conjugation is shown in FIGS. 49A-49D. In FIG. 49B, HerceptinTMIs expressed in a mammalian system and is first galactosylated with a galactose donor and a galactosyltransferase. Herceptin was then induced by the action of ST3Gal3 using a reactive sialic acid-toxin complexTMConjugation to toxins via sialic acid. In FIG. 49C, Herceptin produced in mammalian cells or fungiTMConjugation to toxins occurs through a galactosylation process that employs a galactosyltransferase and a reactive galactose-toxin complex. FIG. 49D contains the preparation HerceptinTMAnother embodiment of the conjugate: herceptin produced in fungi was first treated with Endo-HTMTo trim the glycosyl group, then galactosylate it with a galactose donor and galactosyltransferase, and then conjugate it with a radioisotope by sialylation using ST3Gal3And a reactive sialic acid-radioisotope complex. Alternatively, the reactive sialic acid moiety may be attached only to the chelating moiety, which may then be loaded with the radioisotope at a later stage.
In another exemplary embodiment, the present invention provides a method for preparing SynagisTMThe method of conjugation is shown in FIGS. 50A-50D. In FIG. 50B, Synagis expressed in mammalian cells was first paired with a galactose donor and a galactosyltransferaseTMGalactosylation was performed and then it was pegylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 50C, Synagis expressed in mammalian cells or fungiTMPEGylation was performed with galactosyltransferase and a PEG-galactose donor. In FIG. 50D, expressed Synagis was first treated with Endo-HTMTo trim the glycosyl groups, then galactosylate them with a galactose donor and galactosyltransferase, and then PEGylate them with ST3Gal3 and a PEG-sialic acid donor.
In another exemplary embodiment, the present invention provides a method for generating a RemicadeTMThe method of conjugation is shown in FIGS. 51A-51D. In FIG. 51B, Remicade expressed in mammalian cells was first paired with a galactose donor and a galactosyltransferaseTMGalactosylation was performed and then it was pegylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 51C, Remicade expressed in mammalian systems TMIs modified by the addition of pegylated galactose with the appropriate donor and galactosyltransferase. In FIG. 51D, Remicade expressed in fungi was first treated with Endo-HTMTo trim the glycosyl group, then galactosylate it with a galactose donor and galactosyltransferase, and then conjugate it with a radioisotope with ST3Gal3 and a reactive sialic acid derivatized with a radioisotope.
In another exemplary embodiment, the present invention provides a method for modifying a post-mutation Reopro containing an N-glycosylation site. Fig. 52A to 52L include such an example. In fig. 52B, Reopro expressed in a mammalian system was first treated with sialidase to trim sialic acid residues, which were then pegylated with ST3Gal3 and a PEG-sialic acid donor. In FIG. 52C, Reopro expressed in insect cells was modified by the addition of PEGylated N-acetylglucosamine with the appropriate donor and GnT-I and/or II. In FIG. 52D, Reopro expressed in yeast was first treated with Endo-H to trim glycosyl groups. Subsequently, the protein was pegylated with galactosyltransferase and a PEG-galactose donor. In fig. 52F, Reopro expressed in mammalian cells was first treated with sialidase to trim sialic acid residues, which was then pegylated with ST3Gal1 and a PEG-sialic acid donor. In fig. 52G, Reopro expressed in insect cells was modified by pegylation with galactosyltransferase and PEG-galactose donor. In FIG. 52H, N-acetylgalactosamine was first added to Reopro expressed in the bacteria using N-acetylgalactosamine transferase and an appropriate donor. The protein was then pegylated with sialyltransferase and a PEG-sialic acid donor. In fig. 52J, Reopro expressed in bacteria was modified by different schemes: that is, the PEGylated N-acetylgalactosamine donor is PEGylated by the action of N-acetylgalactosamine transferase. In FIG. 52K, the bacterially expressed Reopro was modified by another method: first, the polypeptide is contacted with N-acetylgalactosamine transferase and an N-acetylgalactosamine donor derivatized with reactive sialic acid via a linker, such that the polypeptide can be attached to the reactive sialic acid via the linker and N-acetylgalactosamine. The polypeptide was then contacted with ST3Gal3 and desialylated transferrin, thereby linking it to transferrin via sialic acid residues. The polypeptide was then capped with sialic acid residues using the appropriate donor and ST3Gal 3. Fig. 52L provides additional schemes for modifying bacterially expressed Reopro. The polypeptide is first exposed to NHS-CO-linker-SA-CMP and linked to reactive sialic acids via a linker. The polypeptide was then contacted with ST3Gal3 and desialylated transferrin, thereby conjugating it to transferrin via sialic acid residues. The polypeptide was then capped with sialic acid residues using the appropriate donor and ST3Gal 3.
In another exemplary embodiment, the present invention provides a method for generating RituxanTMMethods of conjugates. Fig. 53A-53G provide some examples. In FIG. 53B, Rituxan expressed in various mammalian systems was first paired with the appropriate galactose donor and galactosyltransferaseTMGalactosylation was performed. The peptide was then functionalized with sialic acid donors and ST3Gal3 with sialic acids derivatized with toxin moieties. In FIG. 53C, Rituxan expressed in mammalian cells or fungal cellsTMGalactosylation is performed with galactosyltransferase and a galactose donor, which provides the peptide with galactose containing drug moiety. FIG. 53D provides a representation of the reconstitution of Rituxan expressed in a fungal systemTMAs another example. The glycosyl groups of the polypeptide are first trimmed with Endo-H. Galactose was then added with galactosyltransferase and galactose donor. Subsequently, the radioisotope was conjugated to the molecule by means of a radioisotope complexed sialic acid donor and sialyltransferase ST3Gal 3. In FIG. 53F, RituxanTMIs expressed in a mammalian system and is first galactosylated with a galactosyltransferase and an appropriate galactose donor; sialic acid with PEG moieties was then attached using ST3Gal3 and a pegylated sialic acid donor. Rituxan expressed in fungal, yeast or mammalian cells as shown in FIG. 53G TMThe following method can also be used for modification: first, the polypeptide is treated with α -and β -mannosidase to remove terminal mannose residues; GlcNAc is then attached to the molecule with GnT-I and II and a GlcNAc donor, and then the radioisotope is attached thereto by galactosylation using a galactosyltransferase and a galactose donor coupled to a chelating moiety capable of binding the radioisotope.
In another exemplary embodiment, the invention provides a method for modifying antithrombin iii (at iii). Fig. 54A-54O provide some examples. In fig. 54B, antithrombin III expressed in various mammalian systems was reconstituted by addition of one or more terminal sialic acid-PEG moieties. The AT III molecule is first contacted with a sialidase to remove the terminal sialic acid moiety. The molecule is then contacted with a sialyltransferase and an appropriate sialic acid donor which has been derivatized with a PEG moiety. In fig. 54C, AT III expressed in various mammalian systems was reconstituted by addition of sialic acid-PEG moieties. The AT III molecule is contacted with a sialidase to remove the terminal sialic acid moiety. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with 1.2mol equivalents of PEG moieties. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor to cap the remaining terminal galactose moiety. In fig. 54D, AT III expressed in NSO murine myeloma cells was reconstituted to have a complex glycan molecule with a terminal sialic acid-PEG moiety. The AT III molecule is contacted with a sialidase and an alpha-galactosidase to remove terminal sialic acid and galactose moieties. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG moieties. In fig. 54E, AT III expressed in various mammalian systems was reconstituted to have a substantially complete terminal sialic acid-PEG moiety. The AT III molecule is contacted with a sialidase to remove the terminal sialic acid moiety. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with 16mol equivalents of PEG moieties. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor to cap the remaining terminal galactose moiety. In fig. 54F, AT III expressed in various mammalian systems was reconstituted by addition of one or more terminal sialic acid-PEG moieties. The AT III molecule was contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with a levulinic acid moiety. The molecule is then contacted with hydrazine-PEG. In fig. 54G, AT III expressed in various mammalian systems was reconstituted by addition of one or more terminal poly α 2, 8-linked sialic acid moieties. The AT III molecule is contacted with poly α 2, 8-sialyltransferase and an appropriate sialic acid donor. In FIG. 54I, AT III expressed in insect, yeast or fungal cells was reconstituted by the addition of a branched N-N-acetylglucosamine-PEG moiety. The AT III molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor, which has been derivatized with PEG. In fig. 54J, AT III expressed in yeast was reconstituted by removal of the high mannose glycan structure and addition of a terminal sialic acid-PEG moiety. The AT III molecule is contacted with an endo-glycanase to trim the glycosyl groups. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG moieties. In figure 54K, AT III expressed in various mammalian systems was reconstituted by addition of glycoconjugated transferrin. The AT III molecule was contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with a linker-galactose donor moiety. The molecule is then contacted with the galactosyltransferase and endoglucanase treated transferrin. In fig. 54M, AT III expressed in yeast was reconstituted by removing the high mannose glycan structure and adding a terminal galactose-PEG moiety. The molecule is contacted with an endo-glycanase to trim the glycosyl groups. The molecule is further contacted with a galactosyltransferase and an appropriate galactose donor that has been derivatized with a PEG moiety. In fig. 54N, AT III expressed in plant cells was reconstituted by converting glycan structures to mammalian-type complex glycans and then adding one or more terminal galactose-PEG moieties. The AT III molecule is contacted with xylosidase (xylosidase) to remove xylose residues. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor that has been derivatized with a PEG moiety. In fig 54O, ATIII expressed in various mammalian systems was reconstituted by adding one or more terminal sialic acid-PEG moieties to the terminal galactose moiety. The AT III molecule was contacted with ST3Gal3 and an appropriate sialic acid PEG donor that had been derivatized with PEG.
In another exemplary embodiment, the present invention provides a method for modifying the alpha and beta subunits of human chorionic gonadotropin (hCG). FIGS. 55A-55J provide some examples. In figure 55B, hCG expressed in various mammalian and insect systems was reconstituted by addition of a terminal sialic acid-PEG moiety. The hCG molecule is contacted with a sialidase to remove the terminal sialic acid moiety. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG moieties. In figure 55C, hCG expressed in insect cells, yeast or fungal systems was reconstituted by construction of N-linked glycans and addition of terminal sialic acid-PEG moieties. The hCG molecules are contacted with GnT-I and GnT-II and an appropriate N-acetylglucosamine donor. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecule was further contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG moieties. In figure 55D, hCG expressed in various mammalian and insect systems was reconstituted by the addition of one or more terminal sialic acid-PEG moieties to the O-linked glycan structures. The hCG molecule is contacted with a sialidase to remove terminal sialic acid. The molecule was then contacted with ST3Gal3 and an appropriate sialic acid donor to cap the glycan structure with sialic acid moieties. The molecule was then contacted with ST3Gal1 and an appropriate sialic acid donor that had been derivatized with PEG moieties. In figure 55E, hCG expressed in various mammalian and insect systems was reconstituted by addition of sialic acid-PEG moieties to the N-linked glycan structures. The hCG molecules were contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG. In figure 55F, hCG expressed in insect cells, yeast or fungi was reconstituted by addition of a terminal N-acetylglucosamine-PEG molecule. The hCG molecules are contacted with GnT-I and GnT-II and an appropriate N-acetylglucosamine donor which has been derivatized with PEG. In figure 55G, hCG expressed in insect cells, yeast or fungi was reconstituted by the addition of no more than one N-acetylglucosamine-PEG moiety per N-linked glycan structure. The hCG molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor which has been derivatized with a PEG moiety. In figure 55H, hCG expressed in various mammalian systems was reconstituted by adding one or more terminal sialic acid-PEG moieties to the O-linked glycan structures. The hCG molecules were contacted with ST3Gal3 and an appropriate sialic acid donor that had been derivatized with PEG. In figure 55I, hCG expressed in various mammalian systems was reconstituted by addition of a terminal sialic acid-PEG moiety. The hCG molecule is contacted with α 2, 8-SA and an appropriate sialic acid donor which has been derivatized with a PEG moiety. In figure 55J, hCG expressed in various mammalian systems was reconstituted by addition of a terminal sialic acid moiety. The hCG molecule is contacted with poly-alpha 2, 8-ST and an appropriate sialic acid donor which has been derivatized with a PEG moiety.
In another exemplary embodiment, the present invention provides methods for modifying alpha-galactosidase A (Fabrazyme)TM) The method of (1). Fig. 56A-56J provide some examples. In fig. 56B, α -galactosidase a expressed in and secreted from various mammalian and insect systems was reconstituted by the addition of one or more terminal galactose-PEG-transferrin moieties. The α -galactosidase a molecule is contacted with Endo-H to trim the glycosyl groups. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor that has been derivatized with PEG and transferrin. In fig. 56C, α 0-galactosidase a expressed in and secreted from various mammalian and insect cell systems was reconstituted by the addition of one or more terminal sialic acid-linker-mannose-6-phosphate moieties. Contacting the a-galactosidase a molecule with a sialidase to remove terminal sialic acid moieties. The molecule was further contacted with ST3Gal3 and an appropriate sialic acid donor which had been conjugated to mannose-6-phosphate via a linker. In fig. 56D, α -galactosidase a expressed in NSO murine myeloma cells was reconstituted by addition of a terminal sialic acid-linker-mannose-6-phosphate moiety. The α -galactosidase a molecule is contacted with a sialidase and an α -galactosidase to remove terminal sialic acid and galactose moieties. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecule is then contacted with a sialyltransferase and an appropriate sialic acid donor which has been conjugated to mannose-6-phosphate via a linker. In fig. 56E, α -galactosidase a expressed in and secreted from various mammalian and insect cell systems was reconstituted by the addition of one or more terminal sialic acid-PEG moieties. Contacting the a-galactosidase a molecule with a sialidase to remove terminal sialic acid moieties. The molecule is then contacted with a sialyltransferase and an appropriate sialic acid donor which has been derivatized with a PEG moiety. In fig. 56F, α -galactosidase a expressed in mammalian, insect, yeast, or fungal systems was reconstituted by the addition of one or more terminal mannose-linker-ApoE moieties. Allowing alpha-galactosidase to divide into The seeds were contacted with mannosyltransferase and a suitable mannose donor which had been conjugated to ApoE via a linker. In fig. 56G, α -galactosidase a expressed in mammalian, insect, yeast, or fungal systems was reconstituted by the addition of a galactose-linker- α 2-macroglobulin moiety. The α -galactosidase a molecule is contacted with Endo-H to trim the glycosyl groups. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor which has been conjugated to α -2-macroglobulin via a linker. In FIG. 56H, alpha-galactosidase A expressed in insect, yeast, and fungal systems was reconstituted by the addition of one or more N-acetylglucosamine-PEG-mannose-6-phosphate moieties. The α -galactosidase a molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor which has been derivatized with PEG and mannose-6-phosphate. In fig. 56I, α -galactosidase a expressed in insect, yeast or fungal systems was reconstituted by the addition of one or more terminal galactose-PEG-transferrin moieties. An alpha-galactosidase a molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor which has been derivatized with PEG and transferrin. In fig. 56J, α -galactosidase a expressed in insect, yeast or fungal systems was reconstituted by the addition of one or more terminal sialic acid-PEG-black transferrin (melantotrasferrin) moieties. An alpha-galactosidase A molecule is contacted with GnT-I and GnT-II and an appropriate N-acetylglucosamine donor. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecules are then contacted with sialyltransferase and an appropriate sialic acid donor which has been derivatized with PEG and black transferrin.
In another exemplary embodiment, the present invention provides a method for modifying alpha-iduronidase (Aldura zyme)TM) The method of (1). FIGS. 57A-57J provide some examples. In fig. 57B, α -iduronidase expressed in and secreted from various mammalian and insect systems was reconstituted by addition of one or more terminal galactose-PEG-transferrin moieties. The α -iduronidase molecule is contacted with Endo-H to trim the glycosyl groups. Then, the molecule is contacted with galactosyltransferase and usedPEG was contacted with transferrin derivatized appropriate galactose donor. In fig. 57C, α -iduronidase expressed in and secreted from various mammalian and insect cell systems was reconstituted by addition of a terminal sialic acid-linker-mannose-6-phosphate moiety. Contacting the alpha-iduronidase molecule with a sialidase to remove terminal sialic acid moieties. The molecule was further contacted with ST3Gal3 and an appropriate sialic acid donor which had been conjugated to mannose-6-phosphate via a linker. In fig. 57D, α 0-iduronidase expressed in NSO murine myeloma cells was reconstituted by addition of one or more terminal sialic acid-linker-mannose-6-phosphate moieties. The α 1-iduronidase molecule is contacted with a sialidase and an α 2-galactosidase to remove terminal sialic acid and galactose moieties. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecule is then contacted with a sialyltransferase and an appropriate sialic acid donor which has been conjugated to mannose-6-phosphate via a linker. In fig. 57E, α 3-iduronidase expressed in and secreted from various mammalian and insect cell systems was reconstituted by addition of one or more terminal sialic acid-PEG moieties. Contacting the alpha-iduronidase molecule with a sialidase to remove terminal sialic acid moieties. The molecule is further contacted with a sialyltransferase and an appropriate sialic acid donor which has been derivatized with a PEG moiety. In fig. 57F, α -iduronidase expressed in mammalian, insect, yeast or fungal systems is reconstituted by addition of one or more terminal mannose-linker-ApoE moieties. Contacting an alpha-iduronidase molecule with a mannosyltransferase and a suitable mannose donor which has been conjugated to ApoE via a linker. In fig. 57G, α -iduronidase expressed in mammalian, insect, yeast or fungal systems is reconstituted by addition of one or more galactose-linker- α 2-macroglobulin moieties. The α -iduronidase molecule is contacted with Endo-H to trim the glycosyl groups. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor which has been conjugated to α -2-macroglobulin via a linker. In FIG. 57H, α -iduronidase expressed in insect, yeast, and fungal systems by addition of one or more N-acetylglucosamine-PEG-mannose-6-phosphate moieties And (4) carrying out reconstruction. The α -iduronidase molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor that has been derivatized with PEG and mannose-6-phosphate. In fig. 57I, α -iduronidase expressed in insect, yeast or fungal systems is reconstituted by addition of one or more terminal galactose-PEG-transferrin moieties. An alpha-iduronidase molecule is contacted with GnT-I and an appropriate N-acetylglucosamine donor. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor which has been derivatized with PEG and transferrin. In fig. 57J, α -iduronidase expressed in insect, yeast, or fungal systems is reconstituted by addition of one or more terminal sialic acid-PEG-ferrous protein moieties. Contacting an alpha-iduronidase molecule with GnT-I and GnT-II and an appropriate N-acetylglucosamine donor. The molecule is then contacted with a galactosyltransferase and an appropriate galactose donor. The molecules are further contacted with sialyltransferase and an appropriate sialic acid donor which has been derivatized with PEG and black transferrin.
A. Generation or elimination of N-linked glycosylation sites
The present invention relates to the use of peptides in which the glycan chain site on the peptide has been altered relative to that of the native peptide. Typically, the N-linked glycan chain is linked to the primary peptide structure at an asparagine residue located in an amino acid sequence recognized by a membrane-bound glycosyltransferase on the Endoplasmic Reticulum (ER). Typically, the recognition site on the primary peptide structure is the sequence asparagine-X-serine/threonine, where X can be any amino acid except proline and aspartic acid. Although this recognition site is typical, the invention further comprises peptides having N-linked glycans at other recognition sites, wherein the N-linked chains are added with native or recombinant glycosyltransferases.
Since recognition sites for N-linked glycosylation are known, it is within the skill of the person skilled in the art to generate a mutated primary peptide sequence in which the native N-linked glycosylation recognition site is removed, orOptionally, one or more additional N-glycosylation recognition sites are additionally generated. Most simply, asparagine residues can be removed from the primary sequence of the peptide, thereby removing glycan attachment sites, and thus removing a glycan from the mature peptide. For example, genetic engineering methods can be used to haveAsparagineProcessing of the recognition native site of the serine-serine sequence to haveLeucine-serine sequence, thereby eliminating the N-linked glycosylation site at this position.
Further, the N-linked glycosylation site can be removed by altering the residues in the recognition site such that, even if an asparagine residue is present, one or more additional recognition residues are not present. For example, the native sequence asparagine-serine-SerineMutation to asparagine-serine-LysineThereby eliminating the N-glycosylation site at that location. In the case where the N-linked glycosylation site comprises a residue other than the typical recognition site described above, the skilled artisan can determine the sequence and residues necessary for recognition by the appropriate glycosyltransferase, and then mutate at least one residue so that the appropriate glycosyltransferase no longer recognizes the site. In other words, it is within the skill of the artisan to treat the primary sequence of a peptide to generate or remove, or both generate and remove, glycosylation sites, such that peptides with altered glycosylation patterns can be generated. Thus, the present invention should not be construed as limited to any of the primary peptide sequences provided herein as the only sequence for glycan remodeling, but rather should be construed to include any or all peptide sequences suitable for glycan remodeling.
To generate a mutated peptide, the nucleic acid sequence encoding the primary sequence of the peptide is altered such that the native codons encoding the native amino acid residues are mutated to generate codons encoding other amino acid residues. Techniques for altering nucleic acid sequences are common in the art and are described, for example, in any well-known handbook of molecular biology.
In addition, nucleic acids encoding primary peptide structures can be synthesized in vitro using standard techniques. For example, nucleic acid molecules can be synthesized in a "gene machine" using procedures such as the phosphoramidite method. If chemically synthesized double-stranded DNA is desired in applications such as the synthesis of nucleic acids or fragments thereof, each complementary strand may be synthesized separately. The production of short nucleic acids (60-80 base pairs) is technically simple and can be achieved by synthesizing complementary strands and then annealing them. For the production of longer nucleic acids (>300 base pairs), specific techniques are required, since the coupling efficiency in each cycle of chemical DNA synthesis is rarely 100%. To solve this problem, synthetic genes (double-stranded) are assembled in a modular fashion from single-stranded fragments of 20-100 nucleotides in length. For an overview of polynucleotide synthesis, see, e.g., Glick and Paternak (Molecular Biotechnology, Principles and Applications of Recombinant DNA, 1994, ASM Press), Itakura et al (1984, Annu. Rev. biochem. 53: 323) and Climie et al (1990, Proc. Nat' 1.Acad. Sci. USA 87: 633).
Furthermore, changes in the nucleic acid sequence encoding the peptide can be made by site-directed mutagenesis. As will be appreciated, this technique generally employs phage vectors that exist in both single-stranded and double-stranded form. Typical vectors for use in site-directed mutagenesis include vectors such as the M13 phage. These bacteriophages are readily available and their use is generally well known to those skilled in the art. Double-stranded plasmids are also routinely used in site-directed mutagenesis, which eliminates the step of transferring the target nucleic acid from the plasmid to the phage.
In general, site-directed mutagenesis is performed by first obtaining a single-stranded vector or melting both strands of a double-stranded vector that includes in its sequence a DNA sequence encoding the desired peptide. Oligonucleotide primers having the desired mutated sequence are typically prepared synthetically. The primer is then annealed to the single stranded vector and then treated with a DNA polymerase such as the Klenow fragment of E.coli polymerase I to complete the synthesis of the strand with the mutation. Thus, a heteroduplex is formed in which one strand encodes the original non-mutated sequence and the other strand has the desired mutation. The heteroduplex is then used to transform or transfect appropriate cells, e.g. Coli cells and clones comprising the recombinant vector with the mutated sequence were selected. Kunkel et al (1987, Kunkel et al, Methods Enzymol.154: 367- > 382) designed a genetic selection scheme to enrich for clones that incorporated the mutagenized oligonucleotide. Alternatively, commercially available thermostable enzymes such as Taq polymerase and PCR can be usedTMOligonucleotide primers for mutagenesis are incorporated into the amplified DNA fragment, which can then be cloned into an appropriate cloning or expression vector. PCR of Tomic et al (1990, Nucl. acids Res., 12: 1656) and Uupender et al (1995, Biotechniques, 18: 29-31)TMTwo examples of such procedures are provided by the-mediated mutagenesis method. PCR using thermostable ligase in addition to thermostable polymeraseTMIt can also be used to incorporate phosphorylated mutagenic oligonucleotides into amplified DNA fragments, which can then be cloned into an appropriate cloning or expression vector. An example of such a procedure is provided by the mutagenesis method described by Michael (1994, Biotechniques 16: 410-412).
Not all Asn-X-Ser/Thr sequences are N-glycosylated, suggesting that the context in which the motif is located is important. In another approach, libraries of mutant peptides with novel N-linked consensus sites are generated to identify novel N-linked sites that are glycosylated in vivo and are beneficial for the activity, stability or other characteristics of the peptide.
As indicated previously, the consensus sequence for adding an N-linked glycan chain to a glycoprotein is Asn-X-Ser/Thr, where X can be any amino acid. The nucleotide sequence encoding the amino acid two positions on the carboxy-terminal side of an Asn can be mutated to encode Ser and/or Thr using standard methods well known to those skilled in the art. As stated above, not all Asn-X-Ser/Thr sites are modified by addition of glycans. Thus, each recombinant mutated glycoprotein must be expressed in a fungal, yeast or animal or mammalian expression system and analyzed for the addition of N-linked glycan chains. Techniques for characterizing glycosylation sites are well known to those skilled in the art. Further, the biological function of a mutated recombinant glycoprotein can be determined using assays that are standard for the particular protein being examined. Thus, it becomes a simple matter to process the primary sequence of a peptide and identify the new glycosylation site contained therein, and further determine the effect of the new site on the biological activity of the peptide.
In an alternative embodiment, the nucleotide sequence encoding the amino acid two positions flanking the amino terminus of Ser and/or Thr may be mutated using standard procedures well known to those skilled in the art to encode Asn. Methods for determining whether a new glycosylation site has been generated and the effect of that site on the biological activity of the peptide are described above.
B. Generation or elimination of O-linked glycosylation sites
The addition of an O-linked glycosylation site to a peptide can be conveniently accomplished by altering the primary amino acid sequence of the peptide such that it contains one or more additional O-linked glycosylation sites compared to the starting peptide primary amino acid sequence. The addition of O-linked glycosylation sites to a peptide can also be achieved by incorporating one or more amino acid species into a peptide that contains an-OH group (preferably a serine or threonine residue) in its peptide sequence, such that the OH group is accessible and available for O-linked glycosylation. Similar to the alteration of N-linked glycosylation sites in peptides, the primary amino acid sequence of a peptide is preferably altered at the nucleotide level. Specific nucleotides in the DNA sequence encoding the peptide may be altered so that the sequence encodes the desired amino acid. Mutations in the DNA are preferably accomplished by methods well known in the art, such as the phosphoramidite DNA synthesis methods described above and techniques of site-directed mutagenesis.
Alternatively, the nucleotide sequence encoding the putative site for addition of an O-linked glycan may be added to the DNA molecule in one or several copies at the 5 'or 3' end of the molecule. The altered DNA sequence can then be expressed in a fungal, yeast or animal or mammalian expression system and analyzed for the sequence added to the peptide and whether the sequence is a functional O-linked glycosylation site. Briefly, synthetic peptide receptor sequences are introduced at the 5 'or 3' end of a nucleotide molecule. In principle, the addition of such sequences is less disruptive to the resulting glycoprotein when expressed in an appropriate expression system. The altered DNA is then expressed in CHO cells or other suitable expression system and the protein expressed therein is examined to determine the presence or absence of O-linked glycosylation sites. Further, the presence or absence of a glycan chain can be determined.
In another approach, favorable sites for new O-attachment sites can be found in peptides by generating libraries of peptides containing various new O-attachment sites. For example, the amino acid sequence of N-acetylgalactosamine addition by N-acetylgalactosamine transferase depends on the particular transferase used. The amino acid sequence of the peptide can be scanned to identify a contiguous set of amino acids that can be mutated to generate potential sites for the addition of O-linked glycan chains. These mutations can be generated using standard procedures known to those skilled in the art as described previously. To determine whether any of the glycosylation sites found are actually glycosylated, each of the recombinant mutated peptides is expressed in an appropriate expression system and then analyzed for the addition of sites and/or for the presence or absence of O-linked glycan chains.
C. Chemical synthesis of peptides
Although the primary structure of the peptides used in the present invention may be most efficiently produced in a cell-based expression system, it is within the scope of the present invention that the peptides may be produced synthetically. Chemical synthesis of peptides is well known in the art and includes, without limitation, stepwise solid phase synthesis and condensation of fragments in solution or solid phase. Typical stepwise solid phase synthesis involves covalently attaching an amino acid corresponding to the carboxy terminal amino acid of the desired peptide chain to a solid support, and then extending the peptide chain to the amino terminus by stepwise binding of an activated amino acid derivative having an activated carboxy group. After assembly of the fully protected solid phase-bound peptide chain is complete, the peptide-solid phase covalent attachment is cleaved by an appropriate chemical reaction and the protecting group is removed to give the product peptide. See r.merrifield, Solid Phase Peptide Synthesis: the Synthesis of aTetrapeptide, j.am.chem.soc., 85: 2149-2154(1963). The longer the peptide chain, the more challenging it is to obtain a defined product of high purity. Stepwise solid phase synthesis methods have size limitations due to the generation of complex mixtures. Generally, defined peptides having 100 consecutive or more amino acid residues cannot be routinely prepared by stepwise solid phase synthesis.
The segment condensation method involves the preparation of several peptide segments by a solid phase stepwise method followed by cleavage and purification of these maximally protected segments from the solid phase. The protected segments are condensed one after the other with the first segment bound to the solid phase.
The peptides used in the present invention can be synthesized by complete solid phase synthesis, partial solid phase methods, fragment condensation or classical solution synthesis. These synthetic Methods are well known to those skilled in the art (see, e.g., Merrifield, J.Am.chem.Soc., 85: 2149 (1963)), Stewart et al, "Solid Phase Peptide Synthesis" (second edition), (Pierce Chemical Co.1984), Bayer and Rapp, chem.Pept.Prot.3: 3(1986), Atherton et al, Solid Phase Peptide Synthesis: A practical apparatus (IRL Press1989), Fields and Colowck, "Solid-Phase Peptide Synthesis", Methods in Enzymology volume 289 (Academic Press1997) and Lloyd-Williams et al, Chemical application to Synthesis of Peptides (CRC, Inc.). Methods of altering the overall chemical synthesis strategy such as "native chemical ligation" and "expressed peptide ligation" are also standard (see, e.g., Dawson et al, Science 266: 776(1994), Hackeng et al, Proc. Nat '1. Acad. Sci. USA 94: 7845(1997), Dawson, Methods enzymol. 287: 34(1997), Muir et al, Proc. Nat' 1.Acad. Sci. USA 95: 6705(1998) and Severinov and Muir, J.biol. chem. 273: 16205 (1998)). Also useful is the solid phase peptide synthesis method developed by Gryphon Sciences, South san Francisco, Calif. See U.S. patent nos. 6,326,468, 6,217,873, 6,174,530 and 6,001,364, which are incorporated herein by reference in their entirety.
D. Post-translational modification
One skilled in the art will appreciate that peptides may be post-translationally modified in addition to having N-linked and/or O-linked glycans added thereto. It is contemplated that peptides having post-translational modifications other than glycosylation can be used as the peptides of the invention, so long as the desired biological activity or function of the peptide is maintained or improved. Such post-translational modifications may be natural modifications, typically performed in vivo, or processing modifications of the peptide, performed in vitro. Contemplated known modifications include, but are not limited to, acetylation, acylation, ADP-ribosylation, amidation, covalent attachment of flavin, covalent attachment of a heme moiety, covalent attachment of a nucleotide or nucleotide derivative, covalent attachment of a lipid or lipid derivative, covalent attachment of phosphatidylinositol, cross-linking, cyclization, disulfide bond formation, demethylation, formation of covalent cross-links, formation of cysteine, formation of pyroglutamate, formylation, gamma carboxylation, glycosylation, GPI anchor formation, hydroxylation, iodination, methylation, myristoylation, oxidation, proteolytic processing, phosphorylation, prenylation, racemization, selenoylation, sulfation, transfer RNA mediated addition of amino acids to peptides such as arginylation (and ubiquitination, enzymes useful for making many of these modifications are well known in the art, and are available from companies such as Boehringer Mannheim (Indianapolis, IN) and Sigma Chemical Company (St. Louis, Mo.).
Such modifications are well known to those skilled in the art and have been described in detail in the scientific literature. Several particularly common modifications such as glycosylation, lipid attachment, sulfation, gamma carboxylation of glutamic acid residues, hydroxylation and ADP-ribosylation are described in the most basic texts, such as Peptides-structures and Molecular Properties, 2ndEd., T.E.Creighton, W.H.Freeman and Company, New York (1993). There are many detailed reviews on this topic, such as Wold, f., Post-translational evaluation of Peptides, b.c. johnson, EdAcademic Press, New York 1-12 (1983); seifter et al (meth.enzymol.182: 626-646(1990)) and Rattan et al (Ann.N.Y.Acad.Sci.663: 48-62 (1992)).
Covalent modification of peptides can also be introduced into molecules in vitro by reacting reactive target amino acid residues of the peptide with an organic derivatizing agent capable of reacting with selected side chains or terminal amino acid residues. The most commonly derivatized residues are cysteinyl, histidyl, lysyl, arginyl, tyrosyl, glutaminyl, asparaginyl and the amino-terminal residues. Hydroxylation of proline and lysine, phosphorylation of the hydroxyl groups of seryl and threonyl residues, methylation of the alpha-amino groups of lysine, histidine and histidine side chains, acetylation of the N-terminal amine and amidation of the C-terminal carboxyl group. Such derivatized moieties may improve solubility, absorption, biological half-life, and the like. The moiety may also eliminate or reduce any unwanted side effects of the peptide, etc.
In addition, derivatization with bifunctional reagents can be used to crosslink the peptide with a water-insoluble support matrix or other macromolecular carrier. Commonly used cross-linking agents include glutaraldehyde, N-hydroxysuccinimide esters, homobifunctional imidoesters, 1-bis (-diazoleacetyl) -2-phenylethane and bifunctional maleimides. Derivatizing agents such as methyl-3- [ 9-p-azidophenyl) ] dithiopropionimidate produce photoactivated intermediates capable of crosslinking in the presence of light. Alternatively, reactive water-insoluble substrates such as cyanogen bromide activated sugars and reactive substrates described in U.S. Pat. No.3,969,287 and 3,691,016 can be used for peptide immobilization.
E. Fusion peptides/peptides
The peptide for use in the present invention may comprise a fusion peptide. Fusion peptides are particularly advantageous when it is desired to combine the biological and/or functional characteristics of two peptides in one peptide molecule. Such fusion peptides can exhibit a naturally non-existing combination of biological activity and function to generate new useful molecules for therapeutic and industrial applications. Biological activities of interest include, but are not limited to, enzymatic activity, receptor and/or ligand activity, immunogenic motifs and structural domains.
Such fusion peptides are well known in the art, and methods for their production are well known to those skilled in the art. For example, human interferon-alpha-human albumin fusion peptides have been prepared, wherein the resulting peptide has the therapeutic benefits of interferon-alpha combined with the long circulating life of albumin, thereby generating a therapeutic composition that enables a reduction in dosing frequency and potential side effects to the patient. See Albuferon from Human Genome Sciences, Inc. and U.S. Pat. No.5,766,883 TM. Other fusion peptides include antibody molecules described elsewhere.
F. Production of smaller "biologically active" molecules
The peptides used in the present invention may be variants of the native peptide, in which a fragment of the native peptide is substituted for the full-length native peptide. Furthermore, the use of a prepropeptide (pre-pro-peptide) and a propeptide (pre-peptide) is contemplated. The variant peptide may be smaller in size than the native peptide, and may comprise one or more domains of a larger peptide. The selection of a particular peptide domain is advantageous when the biological activity of certain domains in the peptide is desired but the biological activity of other domains in the peptide is not. Also included are truncation and internal deletions of the peptide which enhance the desired therapeutic effect of the peptide. Any such form of peptide can be used in the present invention provided that the desired biological activity of the peptide is retained.
Shorter forms of the peptide may have unique advantages not found in the native peptide. In the case of human albumin, it has been found to be advantageous to include a truncated form of as little as 63% of the native albumin peptide as a bulking agent for plasma volume. Truncated albumin peptides are believed to be superior to native peptides for this therapeutic purpose because the peptides achieve equivalent colloidal penetration at doses of 1/2 to 2/3 relative to native human serum albumin doses or recombinant human serum albumin doses. See U.S. patent No.5,380,712, which is incorporated herein by reference in its entirety.
Smaller "biologically active" peptides were also found to have enhanced therapeutic activity compared to the native peptide. The therapeutic potential of IL-2 is limited by side effects that predominate in vascular leak syndrome. It was found that smaller chemically synthesized forms of the peptide consisting of residues 1-30 corresponding to the entire alpha-helix fold correctly and contain the natural IL-2 biological activity without major side effects.
G. Generation of novel peptides
The peptides of the invention may be derived from the primary sequence of a native peptide or may be processed by a number of methods well known to those skilled in the art. Such processed peptides may be designed and/or selected for their enhanced or new properties compared to the native peptide. For example, the peptide may be processed to have an increased rate of enzymatic reaction, increased or decreased affinity for a substrate or ligand, increased or decreased binding affinity for a receptor, altered specificity for a substrate, ligand, receptor, or other binding partner, increased or decreased in vivo and/or in vitro stability, or increased or decreased immunogenicity in an animal.
H. Mutations
1. Rational design mutations
Peptides used in the methods of the invention may be mutated to enhance a desired biological activity or function, to reduce an undesired property of the peptide, and/or to add a new activity or function to the peptide. "rational peptide design" can be used to generate such altered peptides. Once the amino acid sequence and structure of the peptide are known and the desired mutation is planned, the mutation can be most conveniently made at the corresponding nucleic acid codon that encodes the amino acid residue that is desired to be mutated. One skilled in the art can readily determine how changes to the nucleic acid sequence should be made based on the general genetic code and knowledge of the codon preferences of the expression system selected. Mutations can be made in codons to alter the amino acid residues that will be polymerized into the peptide during translation. Alternatively, the codons may be mutated such that the corresponding encoded amino acid residues are unchanged, but the choice of codon is more suitable for the desired peptide expression system. For example, cys-residues may be replaced with other amino acids to remove disulfide bonds in the native peptide, catalytic domains may be mutated to alter biological activity, and isoforms of the peptide may be processed in general. Such mutations may be point mutations, deletions, insertions, truncations, and the like.
Techniques for mutating specific amino acids in peptides are well known in the art. The above-described site-directed mutagenesis techniques are well suited for codon-directed mutagenesis. Oligonucleotide-mediated mutagenesis methods are also discussed in detail in Sambrook et al (2001, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, New York, starting at page 15.51). Systematic deletions, insertions and truncations can be performed using linker insertion mutagenesis, nuclease Bal31 digestion and linker partition mutagenesis, as well as other methods well known in the art (Sambrook et al, 2001, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, New York).
Rational peptide design has been successfully used to increase enzyme stability to heat inactivation and oxidation. For example, enzyme stability can be improved by removing the asparagine residue in alpha-amylase (Declerck et al, 2000, J.Mol.biol.301: 1041-minus 1057), introducing more rigid structural elements such as proline into alpha-amylase (Igarashi et al, 1999, biosci.Biotechnol.Bichem.63: 1535-minus 1540) and D-xylose isomerase (Zhu et al, 1999, Peptide Eng.12: minus 638). Further, the introduction of additional hydrophobic contacts stabilizes the 3-isopropylmalate dehydrogenase (Akanuma et al, 1999, Eur. J. biochem. 260: 499-. The mechanism of stabilization of these mutations is generally applicable to many peptides. These and similar mutations are expected to be useful for the reconstituted peptides in the methods of the invention.
2. Followed byMachine mutation technology
The novel peptides used in the methods of the invention can be generated using techniques that introduce random mutations in the coding sequence of the nucleic acid. The nucleic acid is then expressed in the desired expression system and the resulting peptide is evaluated for the property of interest. Techniques for introducing random mutations into DNA sequences are well known in the art and include PCR mutagenesis, saturation mutagenesis, and degenerate oligonucleotide methods. See, Sambrook and Russell (2001, Molecular Cloning, A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, NY) and Ausubel et al (2002, Current Protocols in Molecular Biology, John Wiley & Sons, N.Y.).
In PCR mutagenesis, reduced fidelity of Taq polymerase is used to introduce random mutations into cloned DNA fragments (Leung et al, 1989, Technique 1: 11-15). This is a very powerful and relatively fast method of introducing random mutations into a DNA sequence. The DNA region to be mutagenized is amplified by Polymerase Chain Reaction (PCR) under conditions which reduce the fidelity of the DNA synthesized by Taq DNA polymerase, e.g. by using a modified dGTP/dATP ratio and by adding Mn to the PCR reaction2+. The amplified collection of DNA fragments is inserted into an appropriate cloning vector to provide a random library of mutations.
Saturation mutagenesis allows rapid introduction of large numbers of single base substitutions into cloned DNA fragments (Mayers et al, 1985, Science 229: 242). This technique involves, for example, chemical treatment of single-stranded DNA or irradiation in vitro to generate mutations and synthesize complementary DNA strands. The frequency of mutations can be adjusted by adjusting the severity of the treatment and essentially all possible base substitutions can be obtained. Because the procedure does not involve genetic selection of the mutated segment, neutral substitutions as well as those that alter function may be obtained. The distribution of point mutations is not biased towards conserved sequence elements.
Libraries of nucleic acid homologues may also be generated from a degenerate set of oligonucleotide sequences. Chemical synthesis of degenerate oligonucleotide sequences can be performed on an automated DNA synthesizer, and the synthesized can then beThe gene is ligated into an appropriate expression vector. The synthesis of degenerate oligonucleotides is well known in the art (see e.g.Narang, SA (1983) Tetrahedron 39: 3; Itakura et al (1981) Recombinant DNA, Proc 3rdMacromolecules, ed.ag Walton, Amsterdam: elsevier pp.273-289; itakura et al (1984) annu, rev, biochem.53: 323; itakura et al (1984) Science 198: 1056; ike et al (1983) Nucleic Acid Res.11: 477). This technique has been applied to directed evolution of other peptides (see, e.g., Scott et al (1990) Science 249: 386-390; Roberts et al (1992) PNAS 89: 2429-2433; Devrin et al (1990) Science 249: 404-406; Cwirla et al (1990) PNAS 87: 6378-6382 and U.S. Pat. No.5,223,409, 5,198,346 and 5,096,815).
a. Directed evolution
Peptides for use in the methods of the invention may also be generated using "directed evolution" techniques. In contrast to site-directed mutagenesis techniques, where knowledge of the structure of the peptide is essential, strategies currently exist to generate libraries of mutations from which peptides with improved properties can be obtained without knowledge of the structural characteristics of the peptide. These strategies are commonly referred to as "directed evolution" techniques and differ from traditional random mutagenesis procedures in that they perform multiple rounds of repeated mutagenesis, screening and amplification of a nucleic acid sequence encoding a peptide of interest.
In some "directed evolution" techniques, diversity in the nucleic acid obtained is generated by mutation methods that randomly generate point mutations in the nucleic acid sequence. Point mutation techniques include, but are not limited to, "error-prone PCRTM"(Caldwell and Joyce, 1994; PCR Methods appl.2: 28-33; and Ke and Madison, 1997, Nucleic Acids Res.25: 3371-3372), repetitive oligonucleotide-directed mutagenesis (Reidhaar-Olson et al, 1991, Methods enzymol.208: 564-586) and any of the foregoing Methods of random mutagenesis.
Another method of generating diversity is the use of mutator (mutator) genes, on which diversity directed evolution can act. The nucleic acid of interest is cultured in an expanded cell line whose genome typically encodes a defective DNA repair gene (U.S. Pat. No.6,365,410; Selifonova et al, 2001, appl. environ. Microbiol. 67: 3645-.
Diversity acquisition by directed evolution techniques can also be achieved using degenerate primers and Saturation Mutagenesis (Gene Site validation Mutagenesis)TMSan Diego, CA). In such saturation mutagenesis, degenerate primers designed to cover the full length of the nucleic acid sequence to be diversified are used to prime the polymerase in the PCR reaction. In this manner, each codon of the amino acid coding sequence may be mutated so that it encodes each of the remaining 19 common amino acids. This technique can also be used to introduce mutations, deletions and insertions into specific regions of a nucleic acid coding sequence, while leaving the remainder of the nucleic acid molecule unchanged. Procedures for gene saturation techniques are well known in the art and may be found in U.S. Pat. No. 6,171,820.
DNA rearrangement (shuffling)
The novel peptides used in the methods of the invention can also be generated using gene rearrangement, motif rearrangement, exon rearrangement, and/or codon rearrangement (collectively, "DNA rearrangement") techniques. DNA rearrangement techniques may be applied to modulate the activity of the peptides used in the present invention or may be used to generate peptides with altered activity. See generally U.S. Pat. Nos.5,605,793, 5,811,238, 5,830,721, 5,834,252 and 5,837,458 and Stemmer et al (1994, Nature 370 (6488): 389-391); crameri et al (1998, Nature 391 (6664): 288-291); zhang et al (1997, Proc. Natl. Acad. Sci. USA94 (9): 4504-; stemmer et al (1994, Proc. Natl. Acad. Sci. USA 91 (22): 10747-10751), Patten et al (1997, curr. opinion Biotechnol.8: 724-33); harayama (1998, Trends Biotechnol.16 (2): 76-82); hansson et al (1999, J.mol.biol.287: 265-76) and Lorenzo and Blasco (1998, Biotechniques 24 (2): 308-13) (each of which is incorporated herein by reference in its entirety),
DNA rearrangement involves the assembly of two or more DNA fragments by homologous or site-specific recombination to generate variations in the polynucleotide sequence. DNA rearrangements have been used to produce human immunodeficiency virus type 1 proteins (Pekrun et al, 2002, J.Virol.76 (6): 2924-35), triazine hydrolases (Rallard et al, 2001, Chem Biol 8 (9): 891-.
DNA shuffling techniques have also been developed to mimic natural recombination by homologous recombination of DNA in vitro to generate molecular diversity (Stemmler, 1994, Nature 370: 389-107391; and Stemmler, 1994, PNAS 91: 10747-10751). Typically, in this method a population of related genes is fragmented and subjected to multiple denaturation, rehybridization and concomitant extension of the 5' overhang by Taq polymerase. In each cycle, the length of the fragments increases and recombination can occur when fragments derived from different genes hybridize to each other. Initial DNA fragmentation can be achieved by nuclease digestion, typically using DNase (see Stemmler reference, supra), but can also be achieved by interrupted PCR synthesis (U.S. patent 5,965,408, incorporated herein by reference in its entirety; see Diversa corp., San Diego, CA). The advantage of the DNA shuffling approach over the random point mutation approach is that direct recombination of the favorable mutations generated by each round of shuffling can be achieved, thus allowing autonomous selection for improved peptide phenotypes.
DNA rearrangement techniques are well known in the art. A detailed explanation of this technology can be found in Stemmler, 1994, Nature 370: 389-391 and Stemmler, 1994, PNAS 91: 10747 and 10751. DNA rearrangement techniques are also described in U.S. patents 6,180,406, 6,165,793, 6,132,970, 6,117,679, 6,096,548, 5,837,458, 5,834,252, 5,830,721, 5,811,238, and 5,605,793 (all of which are incorporated herein by reference in their entirety).
Recent modifications to the basic techniques of DNA rearrangement are also provided in the art. In one example, a combination of exon rearrangements, exons, or exons encoding specific domains of peptides are amplified using chimeric oligonucleotides. The amplified molecules were then recombined by self-directed PCR assembly (Kolkman and Stemmler, 2001, nat. Biotech.19: 423-428). In another example, single-stranded parent DNA fragments are annealed to full-length single-stranded template using random chimera generation (RACHITT) techniques in transient template library constructs (Coco et al, 2001, Nat Biotechnol.19: 354-. In another example (staggered extension procedure (StEP)) thermal cycling with greatly reduced annealing/extension cycles was used to repeatedly interrupt the polymerization of DNA from the flanking primers (Zhao et al, 1998, NatBiotechnol.16: 258-261). In a technique known as CLERY, serial rearrangement in vitro is combined with in vivo homologous recombination in yeast (Abecassis et al, 2000, Nucleic Acids Res.28: E88). To maximize the intergenic recombination, single-stranded DNA from the complementary strand of each nucleic acid was digested with DNase and annealed (Kikuchi et al, 2000, Gene 243: 133-137). The blunt ends of two truncated Nucleic Acids of variable length joined by a cleavable sequence are then ligated to generate a gene fusion without the need for homologous recombination (Sieber et al, 2001, Nat Biotechnol.19: 456-460; Lutz et al, 2001, Nucleic Acids Res.29: E16; Ostermeier et al, 1999, Nat Biotechnol.17: 1205-1209; Lutz and Benkovic, 2000, curr. Opin. Biotechnol.11: 319-324). Exonuclease-mediated blunting of DNA fragments and ligating the fragments together to allow recombination can also be used to enhance recombination between nucleic acids with little consensus sequence homology (U.S. patent No.6,361,974, incorporated herein by reference in its entirety). The present invention contemplates the use of each and all of the variant methods described above as a method of enhancing the biological properties of any peptide and/or enzyme used in the methods of the present invention.
In addition to published protocols that detail directed evolution and gene rearrangement techniques, commercial services are currently available that perform gene rearrangement and selection procedures on selected peptides. Maxygen (Redwood City, Calif.) provides a commercial service for generating custom libraries of DNA rearrangements. In addition, the company will perform a customized evolutionary program that includes gene rearrangement and selection of selected peptide species.
Optigenix, Inc (Newark, DE) provides services related to plasmid rearrangement. Optigenix utilizes gene families to obtain mutations with novel properties therein. The target nucleic acid was cloned into a plasmid of the Aspergillus expression system. The DNA of the relevant family is then introduced into an expression system and recombination in conserved regions of the family can occur in the host. The resulting mutant DNA is then expressed and the peptides produced therein are screened for the presence and absence of the desired property.
c. Screening program
After each "evolution" cycle, the desired peptide expressed from the mutant gene is screened for the desired characteristic. The "candidate" genes are then amplified and pooled for the next round of DNA rearrangement. The screening procedure used is highly dependent on the peptide undergoing "evolution" and the target characteristics. Characteristics such as peptide stability, biological activity, antigenicity, and the like can be selected using procedures well known in the art. A single assay for the biological activity of preferred peptides for use in the methods of the invention is described elsewhere.
d. Combination of techniques
The skilled person will appreciate that the above mutation and selection techniques can be combined with each other and with additional procedures to generate the most likely peptide molecule for use in the methods of the invention. Thus, the present invention is not limited to any one method of producing a peptide, but should be construed to encompass any or all of the methods described herein. For example, a procedure may be initiated to introduce point mutations into a nucleic acid sequence, followed by cycles of DNA rearrangement, selection, and amplification. The initial introduction of point mutations can be used to introduce diversity into a population of genes lacking this diversity, and subsequent cycles of DNA rearrangement and screening will select and recombine favorable point mutations.
Glycosidases and glycosyltransferases
A. Glycosidases
Glycosidases are glycosyltransferases that use water as an acceptor molecule, and are likewise typically glycoside hydrolases. Glycosidases can be used to form glycosidic bonds in vitro by controlling the thermodynamics and kinetics of the reaction mixture. However, even under modified reaction conditions, glycosidase reactions are still difficult to handle and tend to give low synthesis results as a result of reversible transglycosylase reactions and competitive hydrolysis reactions.
Glycosidases can function by either maintaining their stereochemistry at the bond positions cleaved during hydrolysis or by reversing their stereochemistry at the bond positions cleaved during hydrolysis, thereby classifying glycosidases as "maintenance" glycosidases or "inversion" glycosidases, respectively. The retentive glycosidase has two key carboxylic moieties present at the active site, one carboxylic acid acting as an acid/base catalyst and the other as a nucleophile, while for the inverse glycosidase, one carboxylic acid functions as an acid and the other as a base.
Methods for determining the activity and ligation specificity of any glycosidase are well known in the art, including simplified HPLC procedures (Jacob and Scuder, 1994, Methods in enzymol.230: 280-300). A general discussion of glycosidases and glycosidase treatments can be found in Glycobiology, A Practical Approach, (1993, Fukuda and Kobataeds, Oxford University Press Inc., New York).
Glycosidases useful in the invention include, but are not limited to, sialidases, galactosidases, endoglucanases, mannosidases (i.e., alpha and beta Man I, Man II, and Man III), xylosidases, fucosidases, Agrobacterium species (Agrobacterium sp.) beta-glucosidase, Cellulomonas fimbriae mannosidase 2A, Humicolainsolens glycosidase, Sulfolobus solfataricus (Sulfolobus solfataricus) glycosidase, and Bacillus licheniformis (Bacillus licheniformiformis) glycosidase.
The selection of fucosidase for use in the present invention depends on the attachment of fucose to other molecules. The specificity of many alpha-fucosidases for use in the methods of the invention is well known to those skilled in the art, and many fucosidase variants are commercially available (Glyko, Novato, CA; PROzyme, San Leandro, CA; Calbiochem-Novabiochem, San Diego, CA, etc.). The alpha-fucosidases of interest include, but are not limited to, alpha-fucosidase from Turbo cornutus, Charonia lampas, Torulopsis flexneri (Bacillus fumnans), Aspergillus niger (Aspergillus niger), Clostridium perfringens (Clostridium perfringens), bovine kidney (Glyko), chicken liver (Tyagarajan et al, 1996, Glycobiology 6: 83-93), and alpha-fucosidase II from Pseudomonas manihot (Glyko, PROzyme). Chicken liver fucosidases are particularly useful for removing core fucose from N-linked glycans.
B. Glycosyltransferases
Glycosyltransferases catalyze the addition of an activated saccharide (donor NDP-saccharide) to the non-reducing end of a protein, glycopeptide, lipid or glycolipid or growing oligosaccharide in a stepwise manner. The N-linked glycopeptide is an oligosaccharide donor Dol-PP-NAG linked via transferase and lipid 2Glc3Man9Synthesized by performing an ensemble (enblock) transfer and then pruning the kernel. In this case, the "core" sugar has properties that are somewhat different from the subsequent attachment. A large number of glycosyltransferases are known in the art.
The glycosyltransferase used in the present invention may be any glycosyltransferase as long as it can utilize a modified sugar as a sugar donor. Examples of such enzymes include glycosyltransferases of the Leloir's pathway, such as galactosyltransferases, N-acetylglucosaminyltransferases, N-acetylgalactosamine transferases, fucosyltransferases, sialyltransferases, mannosyltransferases, xylosyltransferases, glucuronyltransferases, and the like.
For enzymatic sugar synthesis involving a glycosyltransferase reaction, the glycosyltransferase can be cloned or isolated from any source. Many cloned glycosyltransferases and their polynucleotide sequences are known. See, e.g., Taniguchi et al, 2002, Handbook of glycerol transferases and dried genes, Springer, Tokyo.
Glycosyltransferase amino acid sequences and nucleotide sequences encoding glycosyltransferases have also been found in various public databases including GenBank, Swiss-Prot, EMBL, etc., from which amino acid sequences can be deduced.
Glycosyltransferases useful in the methods of the present invention include, but are not limited to, galactosyltransferases, fucosyltransferases, glucosyltransferases, N-acetylgalactosaminyltransferases, N-acetylglucosaminyltransferases, glucuronyltransferases, sialyltransferases, mannosyltransferases, glucuronyltransferases, galacturonyltransferases, and oligosaccharyltransferases. Suitable glycosyltransferases include those obtained from eukaryotes as well as prokaryotes.
DNA encoding glycosyltransferases may be obtained by chemical synthesis, by screening reverse transcripts of mRNA from cultures of the appropriate cell or cell line, by screening genomic libraries from the appropriate cell, or by a combination of these procedures. Screening for mRNA or genomic DNA may be performed using oligonucleotide probes derived from the glycosyltransferase nucleic acid sequence. The probes may be labeled with a detectable label such as, but not limited to, a fluorophore, a radioactive atom, or a chemiluminescent group according to known procedures and used in conventional hybridization assays. Alternatively, the glycosyltransferase nucleic acid sequence can be obtained by applying the Polymerase Chain Reaction (PCR) procedure, wherein the oligonucleotide primers of the PCR are generated from the glycosyltransferase nucleic acid sequence. See Mullis et al, U.S. Pat. No.4,683,195, and Mullis, U.S. Pat. No.4,683,202.
Glycosyltransferases can be synthesized in host cells transformed with a vector containing DNA encoding the glycosyltransferase. The vector is a replicable DNA construct. The vector may be used to amplify and/or express DNA encoding a glycosyltransferase. An expression vector is a replicable DNA construct in which a DNA sequence encoding a glycosyltransferase is operably linked to appropriate control sequences capable of effecting expression of the glycosyltransferase in an appropriate host cell. The need for such control sequences will vary depending on the host chosen and the transformation method chosen. Typically, control sequences include a transcription promoter, an optional operator sequence to control transcription, a sequence encoding an appropriate mRNA ribosome binding site, and sequences which control termination of transcription and translation. The amplification vector does not require an expression control domain. All that is required is the ability to replicate in the host, usually conferred by the origin of replication, and the selection gene which facilitates recognition by the transformants.
1. Fucosyltransferase
In some embodiments, the glycosyltransferase used in the methods of the invention is a fucosyltransferase. Fucosyltransferases are well known to those skilled in the art. Exemplary fucosyltransferases include enzymes that transfer L-fucose from GDP-fucose to a hydroxyl position of an acceptor sugar. Fucosyltransferases that transfer non-nucleotide sugars into acceptors are also useful in the present invention.
In some embodiments, the acceptor saccharide is, for example, GlcNAc in the Gal β (1 → 3, 4) GlcNAc β -group in an oligosaccharide glycoside. Suitable fucosyltransferases for this reaction include Gal β (1 → 3, 4) GlcNAc β 1- α (1 → 3, 4) fucosyltransferase (FTIIIE.C. No.2.4.1.65) first characterized from human milk (see Palcic et al, Carbohydrate Res.190: 1-11 (1989); Prieels et al, J.biol.chem.256: 10456-. Sialyl α (2 → 3) Gal β (1 → 3) GlcNAc β fucosyltransferase FTVII (e.c. No.2.4.1.65) was also characterized. Recombinant forms of Gal β (1 → 3, 4) GlcNAc β 1- α (1 → 3, 4) fucosyltransferases have also been characterized (see Dumas et al, bioorg. Med. letters 1: 425-428(1991) and Kukowska-Latallo et al, Genes and Development 4: 1288-1303 (1990)). Other exemplary fucosyltransferases include, e.g., alpha 1, 2 fucosyltransferases (e.c. No. 2.4.1.69). Enzymatic fucosylation can be achieved by the methods described in Mollicone et al, eur.j. biochem.191: 169, 176(1990) or in U.S. Pat. No.5,374,655.
2. Galactosyltransferase enzymes
In another set of embodiments, the glycosyltransferase is a galactosyltransferase. Exemplary galactosyltransferases include α (1, 3) galactosyltransferases (E.C. No.2.4.1.151, see e.g. Dabkowski et al, Transpl ant Proc.25: 2921(1993) and Yamamoto et al, Nature 345: 229- & 233(1990), bovine (GenBank j04989, Joziass et al, J.biol. chem.264: 14290- & 14297(1989)), murine (GenBank m 26925; Larsen et al, Proc. Nat' 1.Acad. Sci. USA 86: 8227 828231 (1989)), porcine (GenBank L36152; Strahan et al, Immunogenetics 41: 101- & 105 (1995)). Another suitable α 1, 3 galactosyltransferase is involved in blood group B antigen formation (E.C.2.4.1.37, Yamamoto et al, J. 1986) (E.C. 2.4.1: 1156).
Also suitable for use in the methods of the invention are β (1, 4) galactosyltransferases including, for example, E.C.2.4.1.90(LacNAc synthetase) and E.C.2.4.1.22 (lactose synthetase) (bovine (D' Agostaro et al, Eur.J.biochem.183: 211-217(1989)), human (Masri et al, biochem.Biophys.Res.Commun.157: 657-663(1988)), murine (Nakawa et al, J.biochem.104: 165-168(1988)), and E.C.2.4.1.38 and ceramide galactosyltransferases (E.2001.2.4.1.45, Stahl et al, J.Neurospora.38: 234 (1994), other suitable galactosyltransferases including, Saccharomyces cerevisiae alpha.38: 234 (Schlegungsz.242, Schizoyltransferases et al (Biophys. J.10, J.11: 78, 1994), see further Saccharomyces cerevisiae et al (Saccharomyces cerevisiae et al, Schizogenes; Schizogenes et al, j biochem.123: 1000-1009).
Production of e.g. the enzyme GalNAc T from cloned genes by genetic engineeringI-XIVAre well known. See, e.g., U.S. Pat. No.4,761,371. One method involves pooling sufficient samples and then determining the amino acid sequence of the enzyme by N-terminal sequencing. This information was then used to isolate a cDNA clone encoding the full-length (membrane-bound) transferase, which resulted in the synthesis of the fully active enzyme upon expression in the insect cell line Sf 9. The receptor specificity of the enzyme was then determined by semi-quantitative analysis of the amino acids surrounding known glycosylation sites in 16 different proteins, with in vitro glycosylation studies on synthetic peptides. This study has demonstrated that certain amino acid residues are present with high frequency in glycosylated peptide segments (overpressent), and residues at specific positions around glycosylated serine and threonine residues have a more significant effect on receptor efficacy than other amino acid moieties.
3. Sialyltransferases
Sialyltransferases are another class of glycosyltransferases useful in the recombinant cells and reaction mixtures of the present invention. Examples of sialyltransferases suitable for use in the present invention include ST3GalIII (e.g., rat or human ST3GalIII), ST3GalIV, ST3Gal I, ST6Gal I, ST3Gal V, ST6Gal II, ST6GalNAc I, ST6GalNAc II, and ST6GalNAc III (the nomenclature for sialyltransferases herein is described in Tsuji et al, Glycobiology 6: - (1996)). An exemplary α (2, 3) sialyltransferase, referred to as α (2, 3) sialyltransferase (e.c.2.4.99.6), can transfer sialic acid to the non-reducing terminal Gal of Gal β 1 → 3Glc disaccharide or glycoside. See Van den Eijnden et al, j.biol.chem.256: 3159(1981), Weinstein et al, J.biol.chem.257: 13845(1982) and Wen et al, J.biol.chem.267: 21011(1992). Another exemplary α 2, 3-sialyltransferase (e.c.2.4.99.4) can transfer sialic acid to the non-reducing terminal Gal of a disaccharide or glycoside. See realick et al, j.biol.chem.254: 4444(1979) and Gillespie et al, J.biol.chem.267: 21004(1992). Further exemplary enzymes include Gal- β -1, 4-GlcNAc-2, 6 sialyltransferase (see Kurosawa et al, Eur. J. biochem. 219: 375-381 (1994)).
Preferably, for glycosylation of the saccharide of the glycopeptide, sialyltransferase is capable of transferring sialic acid to the sequence Gal β 1, 4GlcNAc-, Gal β 1, 3 GlcNAc-or Gal β 1, 3GalNAc-, i.e. the most common penultimate sequence of terminal sialic acids on a fully sialylated saccharide structure (see Table 8). Alpha 2, 8-sialyltransferases capable of transferring sialic acid to alpha 2, 3Gal beta 1, 4GlcNAc may also be used in the method of the invention.
TABLE 8 sialyltransferases using Gal β 1, 4GlcNAc sequences as acceptor substrates
1)Goochee et al.,Bio/Technology 9:1347-1355(1991)
2)Yamarmoto et al.,J.Biochem.120:104-110(1996)
3)Gilbert et al.,J.Biol.Chem.271:28171-28276(1996)
An example of a sialyltransferase for use in the method of the invention is ST3Gal III, which is also known as α (2, 3) sialyltransferase (e.c. 2.4.99.6). This enzyme catalyzes the transfer of sialic acid to Gal β 1, 3GlcNAc or Gal β 1, 4 GlcNAc-glycoside Gal (see, e.g., Wen et al, J.biol. chem.267: 21011 (1992); Van den Eijnden et al, J.biol. chem.256: 3159(1991)), and is responsible for the sialylation of asparagine-linked oligosaccharides in glycopeptides. Sialic acid is linked to Gal, thereby forming an a-linkage between the two sugars. The bond (linkage) between the sugars is between the 2-position of NeuAc and the 3-position of Gal. This particular enzyme can be isolated from rat liver (Weinstein et al, J.biol.chem.257: 13845 (1982)); the DNA sequences of human cDNA (Sasaki et al (1993) J.biol.chem.268: 22782. 22787; Kitagawa & Paulson (1994) J.biol.chem.269: 1394. 1401) and genome (Kitagawa et al (1996) J.biol.chem.271: 931. 938) are known, thereby facilitating the production of the enzyme by recombinant expression. In a preferred embodiment, the sialylation method of the invention employs rat ST3 GalIII.
Examples of sialyltransferases for use in the claimed method are CST-I from Campylobacter (Campylobacter) (see, e.g., U.S. Pat. Nos. 6,503,744, 6,096,529 and 6,210,933 and WO99/49051, and published U.S. patent application 2002/2,042,369). The enzyme catalyzes the transfer of sialic acid to Gal β 1, 4Glc or Gal β 1, 3 GalNAc. Other exemplary sialyltransferases for use in the present invention include those isolated from Campylobacter jejuni subspecies jejuni (Campylobacter jejuni), including alpha (2, 3) sialyltransferase. See, e.g., WO 99/49051.
Other sialyltransferases (including those listed in table 8) are also used in economical and efficient large scale processes to sialylate commercially important glycopeptides. As a simple test to find the utility of these other enzymes, various amounts of each enzyme (1-100mU/mg protein) were combined with desialylated-. alpha.1AGP (1-10mg/ml) was reacted to compare the ability of the target sialyltransferase to sialylate glycopeptides relative to either or both of bovine ST6Gal I, ST3Gal III. Alternatively, other glycopeptides or N-linked oligosaccharides enzymatically released from the peptide backbone may be substituted for the desialylated-alpha 1AGP is used to make this estimate. Sialyltransferases with the ability to sialylate the N-linked oligosaccharides of glycopeptides more efficiently than ST6Gal I were used in large scale practice of peptide sialylation (as demonstrated for ST3Gal III in this disclosure).
4. Other glycosyltransferases
Those skilled in the art will appreciate that other glycosyltransferases may be substituted into similar transferase cycles, as described in detail for sialyltransferases. In particular, the glycosyltransferase can also be, for example, a glucosyltransferase such as Alg8(Stagljov et al, Proc. Natl. Acad. Sci. USA 91: 5977(1994)) or Alg5(Heesen et al, Eur. J. biochem. 224: 71 (1994)).
N-acetylgalactosamine transferase can also be used in the practice of the present invention. Suitable N-acetylgalactosamine transferases include, but are not limited to, α (1, 3) N-acetylgalactosamine transferase, β (1, 4) N-acetylgalactosamine transferase (Nagata et al, J.biol. chem.267: 12082-12089(1992) and Smith et al, J.biol. chem.269: 15162(1994)), and peptide N-acetylgalactosamine transferase (Homa et al, J.biol. chem.268: 12609 (1993)). Suitable N-acetylglucosamine transferases include GnT-I (2.4.1.101, Hull et al, BBRC 176: 608(1991)), GnT-II, GnT-III (Ihara et al, J.biochem.113: 692(1993)), GnT-IV, GnT-V (Shoreibah et al, J.biol.chem.268: 15381(1993)), and GnT-VI, O-linked N-acetylglucosamine transferase (Bierhuiren et al, Proc.Natl.Acad.Sci.USA 89: 9326(1992)), N-acetylglucosamine-1-phosphotransferase (Rajput et al, biom J.285: 985(1992)), and hyaluronan synthase (hyaluronan synthase).
Mannosyltransferases can be used to transfer modified mannose moieties. Suitable mannosyltransferases include alpha (1, 2) mannosyltransferase, alpha (1, 3) mannosyltransferase, alpha (1, 6) mannosyltransferase, beta (1, 4) mannosyltransferase, Dol-P-Man synthetase, Och1 and Pmt1 (see Kornfeld et al, Annu. Rev. biochem. 54: 631-Asn 664 (1985)).
Xylosyltransferases may also be used in the present invention. See, e.g., Rodgers et al, biochem.j., 288: 817-822 (1992); and Elbain et al, U.S. patent No. 6,168,937.
Other suitable glycosyltransferase cycles are described in Ichikawa et al, JACS 114: 9283(1992), Wong et al, j. org. chem.57: 4343(1992) and Ichikawa et al Carbohydrate and Carbohydrate Polymers, Yaltami, ed. (ATLPress, 1993).
Prokaryotic glycosyltransferases may also be used in the practice of the present invention. Such glycosyltransferases include enzymes involved in the synthesis of Lipooligosaccharides (LOS), which can be produced by many gram-negative bacteria. LOS generally has a terminal glycan sequence that mimics the glycoconjugate found on the surface of human epithelial cells or in host secretions (Preston et al, clinical Reviews in Microbiology 23 (3): 139-180 (1996)). Such enzymes include, but are not limited to, proteins such as the rfa operon of the species Escherichia coli and Salmonella typhimurium (Salmonella typhimurium), including β 1, 6 galactosyltransferases and β 1, 3 galactosyltransferases (see, e.g., EMBL Access Nos. M80599 and M86935 (Escherichia coli); EMBL Access No. S5661 (Salmonella typhimurium)), glucosyltransferases (Swiss-Process Access No. P25740 (Escherichia coli)), β 1, 2-glucosyltransferase (rfaJ) (Swiss-Process Access No. P27129 (Escherichia coli)) and Swiss-Process Access No. P19817 (Salmonella typhimurium)), and β 1, 2-N-acetylglucosaminyltransferase (rfaK) (EMBL No. U00039 (Escherichia coli)). other amino acid sequence-known glycosyltransferases include those encoded by an operon such as rfaB, which have been characterized in the following organisms, e.g. Klebsiella pneumoniae (Klebsiella pneumoniae) species, The rh1 operon of Escherichia coli, Salmonella typhimurium, Salmonella enterica, Yersinia enterocolitica (Yersinia enterocolitica), Mycobacterium leprosum, and Pseudomonas aeruginosa (Pseudomonas aeruginosa).
Also suitable for use in the present invention are glycosyltransferases involved in the production of structures containing lacto-N-neotetraose (lacto-N-neotetraose) D-galactosyl-beta-1, 4-N-acetyl-D-glucosaminyl-beta-1, 3-D-galactosyl-beta-1, 4-D-glucose and the Pk blood group trisaccharide sequence D-galactosyl-alpha-1, 4-D-galactosyl-beta-1, 4-D-glucose, which have been identified in the LOS of the mucosal pathogens Neisseria gonorrhoeae (Neisseria gononorhoea) and Neisseria meningitidis (N.meningitis) (Scholten et al, J.Med.Microbiol.41: 236. sup. 243 (1994)). Genes encoding glycosyltransferases involved in the biosynthesis of these structures from Neisseria meningitidis and Neisseria gonorrhoeae have been carried out from N.meningitidis immunotypes L3 and L1(Jennings et al, mol. Microbiol. 18: 729-2740 (1995)) and N.gonorrhoeae mutant F62(Gotshlich, J.exp. Med.180: 2181-2190(1994))And (5) identifying. In Neisseria meningitidis, the locus consisting of 3 genes lgTA, lgTB and lgE encodes the glycosyltransferase necessary to add the last 3 sugars in lacto-N-neotetraose (Wakarhuk et al, J.biol. chem.271: 19166-73 (1996)). Enzymatic activity of the lgTB and lgTA gene products was recently demonstrated, providing the first direct evidence for their glycosyltransferase function (Wakarchuk et al, J.biol. chem.271 (45): 28271-276 (1996)). In N.gonorrhoeae, there are 2 additional genes, i.e., lgD where β -D-GalNAc is added to the terminal galactose 3 position of the lacto-N-neotetraose structure and lgC where the terminal α -D-Gal is added to the lactose element of the truncated LOS, resulting in the generation of Pk blood group antigen structures (Gotshlich (1994), supra). In Neisseria meningitidis, L1, immunized alone, also expressed P kBlood group antigens and have been shown to have the lgTC gene (Jennings et al (1995), supra). Neisseria glycosyltransferases and related genes are also described in USPN5,545,553 (Gotshlich). Alpha 1, 2-fucosyltransferase and alpha 1, 3-fucosyltransferase from helicobacter pylori (Helicobacterpylori) have also been characterized (Martin et al, J.biol.chem.272: 21349-21356 (1997)). Also useful in the present invention are glycosyltransferases from Campylobacter jejuni subspecies jejuni (see Taniguchi et al, 2002, handbook of Glycosyltransferases and related genes, Springer, Tokyo).
B. Sulfotransferases
The invention also provides methods of producing peptides comprising sulfated molecules, including, for example, sulfated polysaccharides such as heparin, heparan sulfate, carrageenan, and related compounds. Suitable sulfotransferases include, for example, chondroitin-6-sulfotransferase (chicken cDNA described by Fukuta et al, J.biol. chem.270: 18575-18580 (1995); GenBank Accession No. D49915), glycosaminoglycan N-acetylglucosamine N-deacetylase/N-sulfotransferase 1(Dixon et al, Genomics 26: 239-241 (1995); UL18918), and glycosaminoglycan N-acetylglucosamine N-deacetylase/N-sulfotransferase 2 (murine cDNA described in Orellana et al, J.biol. chem.269: 2270-2276(1994) and Erssikon et al, J.biol. chem.269: 10438-10443 (1994); human cDNA described in GenBank Accession No. U2304).
C. Cell-bound glycosyltransferases
In another embodiment, the enzyme used in the methods of the invention is a cell-bound glycosyltransferase. Although many soluble glycosyltransferases are known (see, e.g., U.S. Pat. No.5,032,519), glycosyltransferases are typically in membrane-bound form when bound to cells. Many of the membrane-bound enzymes studied to date can be considered intrinsic proteins; i.e. they are not releasable from the membrane by sonication and require detergents to solubilize them. Surface glycosyltransferases have been identified on the surface of vertebrate and invertebrate cells and it has been recognized that these surface transferases retain catalytic activity under physiological conditions. However, a more recognized function of cell surface glycosyltransferases is intercellular recognition (Roth, 1990, Molecular implants to Supercellular Phenomena).
Methods have been developed to alter glycosyltransferases expressed by cells. For example, Larsen et al, proc.natl.acad.sci.usa86: 8227-8231(1989) reports a genetic method for isolating cloned cDNA sequences which determine the expression of cell surface oligosaccharide structures and their homologous glycosyltransferases. A cDNA library generated from mRNA obtained from a gene known to express UDP-galactose was transfected into COS-1 cells: β. -D-galactosyl-1, 4-N-acetyl-D-glucosaminide α -1, 3-galactosyltransferase. The transfected cells are then cultured and assayed for α 1-3 galactosyltransferase activity.
Francisco et al, proc.natl.acad.sci.usa 89: 2713-2717(1992) discloses a method of anchoring beta-lactamases to the outer surface of E.coli. A triple fusion is generated resulting in a surface-bound active beta-lactamase molecule, which consists of (i) the signal sequence of the outer membrane protein, (ii) the transmembrane portion of the outer membrane protein and (iii) the complete mature beta-lactamase sequence. However, the Francisco method is limited to prokaryotic cell systems and, as the authors acknowledge, requires a complete triplet structure to function properly.
D. Fusion enzyme
In another exemplary embodiment, the methods of the invention utilize fusion peptides having more than one enzymatic activity involved in the synthesis of the desired glycopeptide conjugate. The fusion protein may comprise, for example, a catalytically active domain of a glycosyltransferase in combination with a catalytically active domain of a helper enzyme. The catalytic domain of the helper enzyme may catalyze a step in the formation of, for example, a nucleotide sugar that is a donor for the glycosyltransferase or that catalyzes a reaction involved in the glycosyltransferase cycle. For example, a polynucleotide encoding a glycosyltransferase can be combined in-frame with a polynucleotide encoding an enzyme involved in nucleotide sugar synthesis. The resulting fusion peptide can then catalyze not only the synthesis of the nucleotide sugar, but also the transfer of the sugar moiety to an acceptor molecule. The fusion peptide can be two or more circulating enzymes linked to one expressible nucleotide sequence. In other embodiments, the fusion peptide comprises two or more catalytically active domains of a glycosyltransferase. See, e.g., U.S. patent No.5,641,668. The modified glycopeptides of the present invention may be readily designed and produced using a variety of suitable fusion peptides (see, e.g., PCT patent application PCT/CA98/01180, published as WO 99/31224 at 24.6.1999).
E. Immobilized enzymes
In addition to cell-bound enzymes, the invention also provides the use of enzymes immobilized on a solid and/or soluble support. In an exemplary embodiment, glycosyltransferases conjugated to PEG via an intact glycosyl linker according to the methods of the present invention are provided. The PEG-linker-enzyme conjugate may alternatively be attached to a solid support. The use of a solid-supported enzyme in the process of the invention simplifies the handling of the reaction mixture and the purification of the reaction product and also makes the enzyme easy to recover. The glycosyltransferase conjugates are used in the methods of the invention. Other combinations of enzymes and supports will be apparent to those skilled in the art.
F. Mutagenesis of glycosyltransferases
The novel forms of glycosyltransferases, sialyltransferases, sulfotransferases and any other enzymes used in the methods of the invention may be generated by any of the methods described hereinbefore and by other methods well known to those skilled in the art. Of particular interest are transferases with altered acceptor specificity and/or donor specificity. Also of interest are enzymes with higher conversion and higher stability, etc.
Inferential design mutagenesis techniques can be applied when the sequence of the peptide is known. Since the sequences and many tertiary structures of the transferases and glucosidases used in the present invention are known, these enzymes are ideal for rational mutation design. For example, the catalytic site of an enzyme may be mutated to alter the donor and/or acceptor specificity of the enzyme.
The extensive tertiary structural data of glycosyltransferases and glycosidase hydrolases also make these enzymes ideal for mutations involving domain swapping. Glycosyltransferases and glycosidase hydrolases are modular enzymes (see Bourn and Henrissat, 2001, Current Opinion in structural Biology 11: 593-600). Glycosyltransferases can be classified into two classes based on their structure: GT-A and GT-B. The GT-a class of glycosyltransferases contain 2 distinct domains, one involved in nucleotide binding and the other in receptor binding. Thus, one can conveniently fuse a DNA sequence from the coding domain of one gene in frame with a domain from a second gene to generate a new gene that encodes a protein with a new acceptor/donor specificity. Such domain exchanges may additionally include sugar modules and other ancillary domains.
Random mutagenesis and/or directed evolution techniques as described above may also be used to generate novel forms of glycosyltransferases and glycosidases for use in the present invention.
In vitro and in vivo expression systems
A. Glycopeptide-producing cells
The role of glycosyltransferases is critical for glycosylation of peptides, and thus, differences in the expression of a set of glycosyltransferases in any given cell type can affect the glycosylation pattern of any given peptide produced in that cell. For a review of host Cell-dependent peptide glycosylation see Kabata and Takasaki, "Structure and Biosynthesis of Cell Surface Carbohydrates", Cell Surface Carbohydrates and Cell development, 1991, pp.1-24, eds.Minoru Fukuda, CRC Press, Boca Raton, FL.
In accordance with the present disclosure, the cell type producing the peptide is only associated with the degree of reconstitution necessary to produce the peptide with the desired glycosylation. For example, the number and sequence of enzymatic digestion reactions and the number and sequence of enzymatic synthesis reactions necessary to produce a peptide with the desired glycosylation in vitro will vary depending on the structure of glycans on the peptide produced by a particular cell type. Although the present invention should in no way be construed as being limited to the production of peptides from any particular cell type, including those disclosed herein, a discussion of several cell systems is now provided that establishes the efficacy of the present invention and the independence of the cell type producing the peptide.
Generally, in order to express a peptide from a nucleic acid encoding the peptide, the nucleic acid must be incorporated into an expression cassette comprising a promoter element, a terminator element, and a peptide coding sequence operably linked therebetween. The expression cassette is then operably linked to a vector. For this purpose, linkers or linkers may be used to join nucleotide fragments, or other manipulations may be included to provide convenient restriction sites, remove excess nucleotides, remove restriction sites, and the like. For this purpose, in vitro mutagenesis, primer repair, restriction cleavage, annealing, re-substitution such as transitions and transversions may be included. The shuttle vector has genetic elements necessary for replication in the cell. Some vectors can replicate only in prokaryotes, or can replicate in both prokaryotes and eukaryotes. Such plasmid expression vectors will be maintained in one or more, preferably two, replication systems, which enable stable maintenance in yeast host cells for expression purposes and in prokaryotic hosts for cloning purposes. Many vectors with different characteristics are commercially available. The vector is typically a plasmid or phage, but may also be a cosmid or a minichromosome. Conveniently, many commercially available vectors will have promoters and terminators of the existing expression cassettes and polylinker sites that can be used to insert the coding sequence for the peptide of interest. The shuttle vector containing the expression cassette is then transformed in E.coli, where the vector replicates during cell division to produce a vector preparation sufficient to transform a host cell with the selected expression system. The above methods are well known in the art and the protocols implemented can be found in Sambrook et al (2001, Molecular Cloning: organic Manual, Cold Spring Harbor Laboratory, New York).
Once purified from the cells that have been expanded, the vector is transformed into cells of an expression system. Transformation protocols depend on the cell type and the nature of the vector. Transformants are grown on appropriate nutrient media and maintained under selective pressure as appropriate to ensure the maintenance of endogenous DNA. When expression is inducible, the yeast host can be grown to obtain a high density of cells, followed by induction of expression. The secreted mature heterologous peptide can be harvested by any conventional method and can be purified by chromatography, electrophoresis, dialysis, solvent-solvent extraction, and the like.
Molecular cloning techniques are well known in the art. Further, techniques for Molecular Cloning procedures can be found in Sambrook et al (2001, Molecular Cloning: ALaboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.); glover et al (1985, DNA Cloning: A practical approach, volumes I and II); gait et al (1985), Oligonucleotide eSeynthesis); hames and Higgins (1985, Nucleic Acid Hybridization); hames And Higgins (1984, Transcription And transformation); freshney et al (1986, Animal Cell Culture); perbal (1986, Immobilized cell and Enzymes, IRL Press); perbal (1984, A Practical Guide to molecular Cloning); ausubel et al (2002, Current Protocols in molecular Biology, John Wiley & Sons, Inc.).
B. Fungi and yeasts
The peptides produced in yeast are glycosylated and the glycan structures present on them are predominantly high mannose structures. In the case of N-glycans, glycan structures produced in yeast may contain as many as 9 or more mannose residues, with or without additional sugars. Examples of the types of glycans on peptides produced by yeast cells are shown on the left side of FIG. 4. Regardless of the number of mannose residues and the type and complexity of the additional sugars added thereto, the N-glycans, which are components of peptides produced in yeast cells, comprise a trimannose core structure as shown in fig. 4. When the glycan structure on the peptide produced by the yeast cell is a high mannose structure, it is a simple matter for the skilled person to remove all mannose from the molecule (except the molecule comprising the trimannose core of the glycan) in vitro with an appropriate mannosidase, thereby generating a peptide having the basic trimannose core structure attached thereto. Currently, it is a simple matter to enzymatically add additional sugar moieties to the basic trimannose core structure in vitro to generate peptides having the desired glycan structure attached thereto, using techniques available in the art and using the present disclosure. Similarly, when the peptide produced by the yeast cell contains a high mannose structure in addition to other complex sugars attached thereto, it is a simple matter to enzymatically remove all additional sugars (including additional mannose residues) to give the basic trimannose core structure. Once the basic trimannose core structure is produced, it is possible to generate peptides with the desired glycosylation according to the description provided herein.
"Yeast" means ascosporogenous yeasts (Endomycetales), basidiosporogenous yeasts and yeasts belonging to the Fungi Impfections (Blastomycetes) group. Ascogenous yeasts can be divided into two families, namely the family of the argillaceae and the family of the glycolytic. The latter family comprises 4 subfamilies, namely, subfamily Schizosaccharomyces (e.g., Schizosaccharomyces), subfamily Naxosonii, subfamily Lipomyces, and subfamily Saccharomyces (e.g., Pichia, Kluyveromyces, and Saccharomyces). Basidiosporangium yeasts include Leucosporium, Rhodosporidium, Spodophyllum, Filobasidium and Neurospora. Yeasts belonging to the Fungi Imperfecti family can be divided into two families, namely the Sporobolomycetaceae (e.g.Sporobolomyces, Marasmius) and Cryptococcus (e.g.Candida). Of particular interest for the present invention are species of the genera Saccharomyces, Pichia, Aspergillus, Trichoderma, Kluyveromyces, especially Kluyveromyces lactis (K.lactis) and Kluyveromyces drosophilus (K.drosophilum), Candida, Hansenula, Schizosaccharomyces, Yarrowia and Chrysosporium. Since the classification of yeasts may change in the future, for the purposes of the present invention, the definition of Yeast should be as described by Skinner et al, eds. (1980) Biology and Activities of Yeast (Soc. App. bacteriol. Symp. series No. 9).
In addition to the foregoing, one skilled in the art may be familiar with the manipulation of yeast biology and yeast genetics. See, e.g., Bacila et al, eds. (1978, biochemistry and Genetics of Yeast, Academic Press, New York); and Rose and Harrison (1987, The Yeast (2)nded.) Academic Press, London). Methods for introducing exogenous DNA into a yeast host are well known in the art. There are numerous methods available for transforming yeast. Spheroplast transformations were described by Hinnen et al (1978, Proc. Natl. Acad. Sci. USA 75: 1919-. Electroporation is described by Becker and Gaurante (1991, Methods Enzymol.194: 182-. Transformation of non-SaccharomycesFor a review of the chemosynthesis system, see Wang et al (Crit Rev Biotechnol.2001; 21 (3): 177-218). For a general procedure for Yeast genetic engineering, see Barr et al (1989, Yeast geneticingineeering, Butterworks, Boston).
In addition to wild-type yeast and fungal cells, there are also yeast and fungal strains that have been mutated and/or selected to enhance the expression level of the foreign gene, resulting in purity, post-translational processing of the resulting peptide, and recovery and purity of the mature peptide. Expression of the foreign peptide may also be directed to the cell secretory pathway as demonstrated by expression of insulin (see Kjeldsen, 2000, appl. Microbiol. Biotechnol. 54: 277-286 and references cited therein). In general, in order to cause secretion of the foreign peptide from the Yeast Cell, secretion signals derived from Yeast genes such as those of killer toxins (Stark and Boyd, 1986, EMBO J.5: 1995-.
With respect to Filamentous Fungi in general, methods of genetic manipulation can be found in Kinghorn and Turner (1992, Applied Molecular Genetics of Filamentous Fungi, Blackie Academic and Professional, New York). A guide to suitable vectors can be found in Martinelli and Kinghorn (1994, Aspergillus: 50 years, Elsevier, Amsterdam).
1. Sugar yeast
Suitable yeast vectors for the production of peptides in Saccharomyces include YRp7(Struhl et al, Proc. Natl. Acad. Sci. USA 76: 1035 + 1039, 1978), YEp13(Broach et al, Gene 8: 121 + 133, 1979), POT vectors (Kawasaki et al, U.S. Pat. No.4,931,373, incorporated herein by reference), pJDB249 and pJDB219(Beggs, Nature 275: 104 + 108, 1978) and derivatives thereof. Preferred promoters for use in yeast include yeast glycolytic gene expression (Hitzeman et al, J.biol.chem.255: 12073-12080, 1980; Alber and Kawasaki, J.mol.appl.Genet.1: 419-434, 1982; Kawasaki, U.S.Pat.No.4,599,311) or alcohol dehydrogenase groupsThus (Young et al, Genetic Engineering of Microorganisms for Chemicals, Hollaender et al, (eds.), the promoter of p.355, Plenum, New York, 1982; Ammerer, meth.Enzymol.101: 192-cPromoters (Russell et al, Nature 304: 652-654, 1983; Irani and Kilgore, U.S. patent application Ser. No.07/784,653, CA 1,304,020 and EP 284044, incorporated herein by reference). The expression unit may also include a transcription terminator. A preferred transcription terminator is the TPI1 terminator (Alber and Kawasaki, supra).
Examples of such yeast-bacterial shuttle vectors include Yep24(Botstein et al (1979) Gene 8: 17-24), pC1(Brake et al (1984) Proc. Natl. Acad. Sci. USA 81: 4642-4646), and YRp17(Stnich et al (1982) J. mol. biol. 158: 157). In addition, plasmid expression vectors can be high or low copy number plasmids, which typically range from about 1 to about 200. In the case of high copy number yeast vectors, there will generally be at least 10, preferably at least 20 and usually no more than about 150 copies of the vector in a single host. Depending on the heterologous peptide chosen, high or low copy number of the vector may be desirable depending on the vector and the effect of the recombinant peptide on the host. See, e.g., Brake et al (1984) proc.natl.acad.sci.usa 81: 4642-4646. The DNA construct of the invention may also be integrated into the yeast genome by an integrating vector. Examples of such carriers are well known in the art. See, e.g., Botstein et al (1979) Gene 8: 17-24.
It is within the skill in the art to select appropriate yeast and other microbial hosts to practice the invention. Of particular interest are the species saccharomyces: s.cerevisiae (s.cerevisae), s.carlsbergensis (s.carlsbergensis), s.saccharified (s.diastaticus), s.douglasii, s.kluyveri, s.norbensis, and s.oviformis. When selecting yeast host cells to express the desired peptide, suitable host cells may include those with good secretion capacity, low proteolytic activity, overall viability, and the like. Yeast and other microorganisms are commonly available from a variety of sources, including Yeastgenetic Stock Center, Department of Biophysics and medical physics, University of California, Berkeley, Calif.; and American Type Culture Collection, ManassasVA. For a review, see Stratel et al, eds. (1981, The molecular biology of The Yeast Saccharomyces, Cold Spring Harbor laboratory, Cold Spring Harbor, N.Y.).
Methods for introducing exogenous DNA into a yeast host are well known in the art.
2. Pichia pastoris
The production of recombinant peptides using Pichia methanolica as host cell is disclosed in PCT applications WO 97/17450, WO 97/17451, WO 98/02536 and WO 98/02565. The DNA molecule used for transformation of p.methanolica is usually prepared as a double-stranded circular plasmid, which is preferably linearized prior to transformation. For peptide production in p.methanolica, it is preferred that the promoter and terminator of the plasmid are of the p.methanolica gene, such as of the p.methanolica alcohol utilization gene (AUG1 or AUG 2). Other useful promoters include those of the dihydroxyacetone synthase (DHAS), formate dehydrogenase (FMD) and Catalase (CAT) genes and those disclosed in U.S. Pat. No.5,252,726. To facilitate integration of the DNA into the host chromosome, it is preferred that the entire expression segment of the plasmid be flanked on both ends by host DNA sequences. A preferred selectable marker for use in Pichia pastoris is the P.methanolica ADE2 gene, which encodes phosphoribosyl-5-aminoimidazole carboxylase (AIRC; EC 4.1.1.21), which allows ADE2 host cells to grow in the absence of adenine. For large-scale industrial processes where it is desirable to minimize the use of methanol, host cells with both methanol utilization genes (AUG1 and AUG2) deleted are preferred. For the production of secreted peptides, host cells deficient in vacuolar protease genes (PEP4 and PRB1) are preferred. Electroporation can be used to facilitate the introduction of plasmids containing DNA encoding a peptide of interest into p. It is preferred to transform p.methanolica cells with electroporation using a pulsed electric field with an exponential decay electric field strength of 2.5 to 4.5kV/cm, preferably about 3.75kV/cm, and a time constant (t) of 1 to 40 milliseconds, most preferably about 20 milliseconds. For a review of the application of Pichia pastoris (Pichia pastoris) for large scale antibody fragment production, see Fischer et al (1999, Biotechnol Appl biochem.30(Pt 2): 117-120).
3. Aspergillus genus
Methods for expressing peptides in Aspergillus species are well known in the art, including but not limited to those described in Carrez et al, 1990, Gene 94: 147-; contreras, 1991, Bio/Technology 9: 378-381; yelton et al, 1984, proc.natl.acad.sci.usa 81: 1470-; tilbourn et al, 1983, Gene 26: 205-221; kelly and Hynes, 1985, EMBO j.4: 475-; ballance et al, 1983, biochem. biophysis. res. comm.112: 284-289; buxton et al, 1985, Gene 37: 207-. An example of a promoter for use in Aspergillus can be found in U.S. Pat. No.5,252,726. Strains of Aspergillus for peptide expression can be found in U.S. Pat. No.4,935,349. Commercial products of exogenous peptides from Aspergillus niger and Aspergillus oryzae (Aspergillus oryzae) are available from Novoenzymes.
4. Trichoderma reesei
Trichoderma has advantages over other recombinant host cell species for expression of the desired peptide. The organism is susceptible to mass growth and has the ability to glycosylate and efficiently secrete high yields of recombinant mammalian peptides into the culture medium, thereby making isolation of the peptides relatively easy. Furthermore, the glycosylation pattern on the expressed peptides is more similar to that on human peptides than in many other systems. However, there are still differences in glycan structures on the peptides expressed in these cells. For example, terminal sialic acid residues are important for the therapeutic function of peptides in mammalian systems because the presence of these moieties at the termini of the glycan structure prevents clearance of the peptide from the mammalian blood. The mechanism by which sialylated molecules increase biological half-life is thought to be due to a decrease in their recognition by lectins (Drickamer, 1988, J.biol. chem.263: 9557-9560). However, typically the fungal cell does not add additional terminal sialic acid residues to the glycans on the peptide, and therefore the peptide synthesized in the fungal cell is desialylated. According to the present invention, this drawback can be compensated for by the in vitro glycan reconstitution method of the present invention described in detail elsewhere.
Trichoderma species used as hosts to produce peptides to be reconstituted include T.reesei, such as QM6a, ALKO2442 or CBS383.78 (Central braalbureau voor Schimmelcultures, Oosterstraat 1, PO Box 273, 3740 AG Baarn, the Netherlands) or ATCC13631 (American type culture Collection, Manassas VA, 10852, USA, model)); trichoderma viride (t.viride) (e.g. CBS189.79 (det.w.gams)); longibachiatum, such as CBS816.68 (mode); pseudokonningii (e.g., MUCL 19358; Mycotheque de l' Universal Cathoique de Louvain); saturnisporum CBS330.70 (schema); t.harzianum CBS316.31 (det.w.gams); t.virgatum (t.pseudokinngii) ATCC 24961. Most preferably, the host is T.reesei, and more preferably T.reesei strain QM9414(ATCC26921), RUT-C-30(ATCC56765) and highly productive mutants derived from QM9414 such as VTT-D-79125(Nevalainen, Technical Research Centre of Finlandpublications 26, (1985), Espoo, Finland).
Transformation of Trichoderma with DNA is carried out by any technique known in the art, including those described in European patent No. EP0244234, Harkki (1989, Bio/Technology 7: 596-. The cultivation of trichoderma is supported by a large amount of experience with the aforementioned industrial scale fermentation techniques; see, for example, Finkelstein, 1992, Biotechnology of Filamentous Fungi: technologies and Products, Butterworth-Heinemann, publishers, Stoneham, Mass.
5. Kluyveromyces yeast
Yeasts belonging to the genus Kluyveromyces have been used as host organisms for the production of recombinant peptides. The peptides produced by yeasts of this genus are in particular chymosin (European patent 96430), thaumatin (European patent 96910), albumin, interleukin-1 beta, TPA, TIMP (European patent 361991) and albumin derivatives with therapeutic function (European patent 413622). Species of particular interest in the genus Kluyveromyces include Kluyveromyces lactis.
Methods for expressing recombinant peptides in Kluyveromyces species are well known in the art. Vectors for expression and secretion of human recombinant peptides in the genus Kluyveromyces are well known in the art (Yeh, J.cell.biochem.suppl.14C: 68, Abs.H402; Fleer, 1990, Yeast 6 (extra term): S449), as are procedures for transformation and expression of recombinant peptides (Ito et al, 1983, J.Bacteriol.153: 163. 168; van den Berg, 1990, Bio/Technology 8: 135. beta. 139; U.S. Pat. Nos. 5,633,146, WO8304050A1, EP0096910, EP0241435, EP0301670, EP0361991, all of which are incorporated herein by reference in their entirety). For an overview of the genetic manipulation of the linear DNA plasmid of Kluyveromyces lactis by gene targeting and plasmid shuttle, see Schaffarath et al (1999, FEMS Microbiol Lett.178 (2): 201-210).
6. Chrysosporium bacteria
Recently the fungus chrysosporium has been used for the expression of exogenous recombinant peptides. A description of the procedure by which a person skilled in the art can utilise Chrysosporium to express foreign peptides can be found in WO 00/20555 (incorporated herein by reference in its entirety). Species particularly suitable for expression systems include, but are not limited to, c.botryoids, c.carmichaelii, c.crassiticus, c.europae, c.evolcennuli, f.fandidium, c.filiforme, c.gergiae, c.globiferum, c.crisperium, c.kresoiseli, c.hispidium, c.lignorum, c.lobatum, c.lucknowense, c.lute, c.garten, c.k.garden, c.sub.c.sub.c.c.sub.sub.c.sub.c.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.c.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.c.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.sp.
7. Others
A process for the transformation of Schwanniomyces (Schwanniomyces) is disclosed in European patent 394538. A method for transforming Acremonium chrysogenum is disclosed by u.s.pat. No.5,162,228. A process for transforming neurospora is disclosed by u.s.pat. No.4,486,533. Also known is an expression system specific for Schizosaccharomyces pombe (European patent 385391). Conventional methods for expressing peptides in the fission yeast Schizosaccharomyces pombe can be found in Giga-Hama and Kumagai (1977, Foreign genexpression in fission year: Schizosaccharomyces pombe, Springer, Berlin).
C. Mammalian system
As noted above, mammalian cells typically produce a heterogeneous mixture of N-glycan structures that vary in the number and arrangement of additional sugars attached to the trimannose core. Generally, mammalian cells produce peptides with complex glycan structures, as shown on the right side of fig. 3. Using the methods of the invention, peptides produced in mammalian cells can be reconstituted to produce peptides with the desired glycosylation by first identifying the primary glycan structure and then determining which sugars must be removed in order to reconstitute the glycan structure. As discussed herein, the sugar to be removed will determine which cleaving enzyme will be used, and thus the precise steps of the reconstitution process will vary depending on the primary glycan structure used as the initial substrate. An exemplary protocol for the reconstitution of glycan structures commonly produced in mammalian cells is shown in figure 2. The N-glycan biosynthetic pathway in mammalian cells has been well characterized (for review see Moremen, 1994, Glycobiology 4: 113-. Many enzymes essential for glycan synthesis have been identified, and mutant cell lines defective in this enzymatic pathway have been isolated, including the Chinese Hamster Ovary (CHO) cell line Lec23 (alpha-glucosidase I deficient) and Lec18 (novel GlcNAc-TVIII). The glycosylation pattern of the peptides produced by these mutant cells was altered relative to normal CHO cells. As discussed herein, glycosylation defects in these and other mutant cells can be exploited for the purpose of generating peptides that lack complex glycan structures. For example, peptides produced by Lec23 cells lack sialic acid residues and thus require fewer enzymatic manipulations in order to reduce glycan structures to the basic trimannose core or Man3GlcNAc 4. Thus, peptides produced by these cells may serve as preferred substrates for glycan reconstitution. Other glycosylation-deficient Cell lines can be isolated or identified by those skilled in the art based on well-known methods, such as those described in Stanley et al, 1990, Somatic Cell mol. 211- > 223. For the purpose of generating preferred peptide substrates for the reconstitution process described herein, the use of those identified and yet unidentified glycosylation-deficient cell lines can be included in the present invention.
Expression vectors for expressing exogenous peptides in mammalian cells are numerous and well known in the art. Many mammalian expression vectors are currently commercially available from companies, including Novagen, Inc (Madison, Wis.), Gene Therapy Systems (SanDiego, Calif.), Promega (Madison, Wis.), ClonTech Inc. (Palo Alto, Calif.), and Stratagene (La Jolla, Calif.), among others.
There are several mammalian cell lines that are particularly adept at expressing exogenous peptides. Mammalian cell lines are typically derived from tumor cells extracted from mammals that have been immortalized, i.e., they replicate essentially indefinitely in culture. These cell lines include, but are not limited to, CHO (Chinese hamster ovary, e.g., CHO-K1; ATCC No. CCL61) and variants thereof, NS0 (mouse myeloma), BNK, BHK570(ATCC No. CRL 10314), BHK (ATCC No. CRL 1632), Per.C6TM(immortalized human cells, Crucell N.V., Leiden, the Netherlands), COS-1(ATCC No. CRL 1650), COS-7(ATCC No. CRL 1651), HEK 293, mouse L cells, T lymphoid cellsLines, BW5147 cells and MDCK (Madin-Darby canine kidney), HeLa (human), A549 (human lung carcinoma), 293(ATCC No. CRL 1573; Graham et al, 1977, Gen. Virol.36: 59-72), BGMK (Buffalo Green Monkey kidney), Hep-2 (human epidermoid laryngeal carcinoma), LLC-MK 2(African green monkey kidney, McCoy, NCI-H292 (human lung mucoepidermoid tumor tubes), RD (rhabdomyosarcoma), Vero (African green monkey kidney), HEL (human embryonic lung), human fetal lung-Chang, MRC5 (embryonic lung), MRHF (human foreskin) and WI-38 (human embryonic lung.) in some cases, cells expressing a therapeutic peptide can be cells derived from the patient to be treated, or can be derived from another related or unrelated mammal.
Mammalian cells can be transformed with DNA using any of several techniques well known in the art. Such techniques include, but are not limited to, calcium phosphate transformation (Chen and Okayama, 1988; Graham and van der Eb, 1973; Corsaro and Pearson, 1981; Somatocell Genetics 7: 603; Diethylaminoethyl (DEAE) -dextran transfection (Fujita et al, 1986; Lopata et al, 1984; Selden et al, 1986), electroporation (Neumann et al, 1982; Potter, 1988; Potter et al, 1984; Wong and Neuman, 1982), cationic lipid reagent transfection (Elroy-Stein and Moss, 1990; Feigner et al, 1987; Rose et al, 1991; Whitt et al, 1990; Hawley-Nelson et al, 1993, Focus 15: 73; Ciccarone et al, 1993, 15: 80), retrovirus (Cepkopkoler et al, 1990; Boakware et al, 1986; Wilkinson et al, 1984; Wilkinson, 1986; and Fabrek et al, 1996, Nature med.2: 714-6), 1, 5-dimethyl-1, 5-diaza-undecamethylene polymethylenebromide (polynene) (Chaney et al, 1986; kawai and Nishizawa, 1984), microinjection (Capecchi, 1980), and protoplast fusion (Rassoulzadegan et al, 1982; Sandri-Goldin et al, 1981; schafer, 1980), etc. In general, transformation techniques are described in Sambrook et al (2001, molecular cloning, A Laboratory Manual, Cold Spring Harbor Laboratory, New York) and Ausubel et al (2002, Current Protocols in molecular biology, John Wiley & Sons, New York).
The baculovirus system commonly used for insect cell transformation has recently been adapted for stable transformation of mammalian cells (see review Koat and Condreay, 2002, Trends Biotechnol.20: 173-180 and references cited therein). Production of recombinant peptides in cultured mammalian cells is disclosed in, e.g., U.S. Pat. nos. 4,713,339, 4,784,950, 4,579,821, and 4,656,134. Several companies offer services for transforming and culturing mammalian cells, including Cell Trends, Inc. Techniques for culturing mammalian cells are well known in the art and further can be found in Hauser et al (1997,Mammalian Cell Biotechnologywalter de Gruyer, inc., Hawthorne, NY) and Sambrook et al (2001,Molecular Cloning,A Laboratory Manualcold Spring Harbor) and references cited therein.
D. Insect pest
Insect cells, particularly cultured insect cells, can express peptides with N-linked glycan structures that are poorly sialylated and typically contain mannose residues with or without additional fucose attached thereto. Examples of such glycan structures and their mannans present on peptides produced in cultured insect cells are shown in fig. 6. In this case, core fucose, which may be linked to the glycans through several different bonds, if present, may or may not be present.
Baculovirus-mediated expression in insect cells has become particularly suitable for recombinant peptide production (Altmann et al, 1999, Glycoconjugate J.16: 109-123). Insect cells are inferior to mammalian cells in terms of peptide folding and post-translational processing. However, as noted above, N-glycosylation of peptides in insect cells differs from N-glycosylation in mammalian cells in many ways, particularly insect cells often produce truncated glycan structures comprising oligosaccharides containing only 3 or sometimes only 2 mannose residues. These structures may additionally be replaced by fucose residues.
According to the present invention, peptides produced in insect cells can first be reconstituted in vitro by selectively removing any substituted fucose residues with an appropriate fucosidase to produce peptides with the desired glycosylation. In the case where the peptide contains a basic trimannose core structure after removal of fucose residues, then all that is required is to add the appropriate sugar to the trimannose core structure in vitro to produce a peptide with the desired glycosylation. In the case where the peptide may contain only 2 mannose residues in the glycan structure after removal of any fucose residues, a third mannose residue may be added with a mannosyltransferase and an appropriate donor molecule such as GDP-mannose to add the appropriate residue to produce a peptide with the desired glycosylation. Optionally, monokeratoglycans can also be generated from these species.
Procedures for transforming insect cells with baculoviruses are well known in the art. Several books have been published that provide procedures for expressing peptides in insect cells using baculovirus systems. These books include, but are not limited to, Richardson (Baculovirus Expression Protocols, 1998, methods in Molecular Biology, Vol.39, Humana Pr), O' Reilly et al (1994, Baculovirus Expression Vectors: A Laboratory Manual, Oxford Univ Press) and King and Possee (1992, The Baculovirus Expression System: A Laboratory Guide, Chapman & Hall). Furthermore, there are publications such as Lucklow (1993, curr. Opin. Biotechnol.4: 564-572) and Miller (1993, curr. Opin. Genet.Dev.3: 97-101).
Many patents have been granted relating to baculovirus expression systems for foreign proteins. These patents include, but are not limited to, U.S. Pat. No.6,210,966 (insect cell culture medium lacking glutamine but containing ammonium salt), U.S. Pat. No.6,090,584 (application of BVAC (baculovirus artificial chromosome) to produce recombinant peptides), U.S. Pat. No.5,871,986 (application of baculovirus to express recombinant nucleic acids in mammalian cells), U.S. Pat. No.5,759,809 (method of expressing peptides in insect cells and method of killing insects), U.S. Pat. No.5,753,220 (cysteine protease gene-deficient baculovirus, process for producing same, and process for producing economical peptides using same), U.S. Pat. No.5,750,383 (baculovirus cloning system), U.S. Pat. No.5,731,182 (non-mammalian DNA virus expressing recombinant nucleic acids in mammalian cells), U.S. Pat. No.5,728,580 (method and medium for inducing suspension in insect cell lines), U.S. Pat. No.5,583,023 (modified single cell virus, process for producing same, and application thereof as gene expression vector), U.S. Pat. No.5,571,709 (modified baculoviruses and baculoviral expression vectors), U.S. Pat. No.5,521,299 (oligonucleotides for detecting baculoviral infection), U.S. Pat. No.5,516,657 (baculoviral vectors for expressing secretory and membrane-bound peptides), U.S. Pat. No.5,475,090 (genes encoding peptides that enhance viral infection of host insects), U.S. Pat. No.5,472,858 (production of recombinant peptides in insect larvae), U.S. Pat. No.5,348,886 (method for producing recombinant eukaryotic viruses in bacteria), U.S. Pat. No.5,322,774 (prokaryotic leader sequence in recombinant baculovirus expression systems), U.S. Pat. No.5,278,050 (method for improving the processing and secretion efficiency of recombinant genes in insect systems), U.S. Pat. No.5,244,805 (baculoviral expression vectors), U.S. Pat. No.5,229,293 (baculoviral), U.S. Pat. No.5,194,376 (baculovirus expression systems capable of producing recombinant peptides at high levels), U.S. Pat. No.5,179,007 (methods and vectors for purifying recombinant peptides), U.S. Pat. No.5,169,784 (baculovirus dual promoter expression vector), U.S. Pat. No.5,162,222 (use of baculovirus early promoters to express recombinant nucleic acids in stably transformed insect cells or recombinant baculoviruses), U.S. Pat. No.5,155,037 (insect signal sequence for improving efficiency of processing and secretion of recombinant nucleic acids in insect systems), U.S. Pat. No.5,147,788 (baculovirus vectors and methods of use), U.S. Pat. No.5,110,729 (method of producing peptides in cultured cells using baculovirus vectors), U.S. Pat. No.5,077,214 (use of baculovirus early promoters to express recombinant genes in stably transformed insect cells), U.S. Pat. No.5,023,328 (Lepidopteran) AKH signal sequence, and U.S. Pat. Nos. 4,879,236 and 4,745,051 (methods of producing recombinant baculovirus expression vectors). The above patents are incorporated herein by reference in their entirety.
Several insect cell lines of different species origin are currently used for the expression of peptides and these cell lines are well known in the art. Insect cell lines of interest include, but are not limited to, typical dipteran and lepidopteran insect cells, Sf9 and variants thereof (fall armyworm) Spodoptera frugiperda), Estimene acre, Trichoplusiani, Bombyx mori (Bombyx mori), Malacosa distri, Drosophila strains Kc1 and SL2, and the like, and mosquitoes.
E. Plant and method for producing the same
Plant cells that are producers of peptides provide different scenarios. Although the N-linked glycans produced in plants comprise a trimannose core structure, the pentasaccharide backbone may comprise several different additional sugars as shown in figure 5. For example, in one instance, the trimannose core structure is substituted with β 1, 2 linked xylose residues and α 1, 3 linked fucose residues. In addition, plant cells can also produce Man5GlcNAc2 structures. Peptides produced in plant cells are often highly antigenic due to the presence of core α 1, 3 xylose and fucose on glycan structures and will be rapidly cleared from the bloodstream when introduced into mammals due to the absence of terminal sialic acid residues. Thus, unless these peptides are reconstituted using the methods provided herein, they are generally considered unsuitable for use as therapeutic agents in mammals. Although some monoclonal antibodies expressed in plants were found to be non-immunogenic in mice, it is possible that the glycan chain was non-immunogenic due to its buried Fc region in these antibodies (Chargelegue et al, 2000, Transgenic Res.9 (3): 187-.
In accordance with the description provided herein, it is now possible to generate peptides produced in plant cells in which an increased number of glycan structures present thereon comprise the basic trimannose core structure or the Man3GlcNAc4 structure. This is achieved by cleaving additional sugars in vitro with a combination of appropriate glycosidases, including fucosidases, until the basic trimannose core structure or Man3GlcNAc4 structure is reached. These cleavage reactions should also include the removal of any fucose or xylose residues from the structure to eliminate antigenicity of the final peptide when introduced into a mammal. Plant cells with mutations that inhibit the addition of fucose and xylose residues to the trimannose core structure are well known in the art (von Schaewen et al, 1993, plantaphysiology 102: 1109-. The invention contemplates the use of these cells in the production of peptides having glycans lacking fucose and xylose. After the basic trimannose core or Man3GlcNAc4 structure is produced, additional sugars can then be added thereto to give a peptide with the desired glycosylation, which is thus suitable for therapeutic use in mammals.
Many believe that transgenic plants are the expression system of choice for pharmaceutical peptides. Potentially, plants can provide an inexpensive source of recombinant peptides. It has been estimated that the production cost of recombinant peptides in plants can be 10-50 times lower than the cost of producing the same peptides in E.coli. This can be compensated by adjusting the recombinant DNA sequence despite minor differences in codon usage in plants compared to animals (see Kusnadi et al 1997 Biotechnol. Bioeng.56: 473-484; Khoudi et al 1999 Biotechnol. Bioeng.135-143; Hood et al 1999 Adv. exp. Med. biol. 464: 127-147). Furthermore, peptide synthesis, secretion and post-translational modifications are very similar in plants and animals, with only minor differences in plant glycosylation (see Fischer et al, 2000, J.biol.Regul.Homest.Agents 14: 83-92). Products from transgenic plants are then also less likely to be contaminated with animal pathogens, microbial toxins and oncogenic sequences.
Expression of recombinant peptides in plant cells is well known in the art. In addition to transgenic plants, peptides can also be produced in transgenic plant cell cultures (Lee et al, 1997, mol. cell.7: 783-787) and in non-transgenic plants inoculated with recombinant plant viruses. Several books describing the genetic transformation process of plant cells have been published: potrykus (1995, Gene transfer to plants, Springer, New York), Nickoloff (1995, Plant cell electrophoresis and electrofusion protocols, Humana Press, Totowa, New York) and Draper (1988, Plant genetic transformation, Oxford Press, Boston).
Several methods have been used to stably transform plant cells with recombinant genetic material. These methods include, but are not limited to, Agrobacterium transformation (Bechtold and Pelletier, 1998; Escudero and Hohn, 1997; Hansen and Chilton, 1999; Touraev et al, 1997), biolistics (microprojectile bombardment) (Finer et al, 1999; Hansen and Chilton, 1999; Shilito, 1999), protoplast electroporation (Fromm et al, 1985; Ou-Lee et al, 1986; Rhodes et al, 1988; Saunders et al, 1989; Trick et al, 1997), polyethylene glycol treatment (Shilito, 1999; Trick et al, 1997), in planta microinjection (Leduc et al, 1996; Zhou et al, 1983), seed aspiration (Trick et al, 1997), laser beam (1996) and silicon carbide (Thompson et al, 1995; App. 201S.20077, herein incorporated by reference in its entirety).
Many types of plants can be transformed and expressed with exogenous peptides. Plants of particular interest for expressing the peptides for use in the reconstruction method of the invention include, but are not limited to, Arabidopsis (Arabidopsis thaliana), rapeseed (Brassica spp.; Ruiz and Blumwald, 2002, Planta 214: 965-; deng et al, 2001, cell. res.11: 156-; spencer and Snow, 2001, Heredity 86(Pt 6): 694-; lamblin et al, 2001, Physiol Plant 112: 223-; hood et al, 1999, adv.exp.med.biol.464: 127-147; hood et al, 1997, mol. breed.3: 291-306; petolino et al, 2000, Transgenic Research 9: 1-9), alfalfa (Khoudi et al, 1999, biotechnol.bioeng.64: 135-; wright et al, Transgenic Res.10: 177-181; frigerio et al, 2000, plantariol.123: 1483 + 1493; cramer et al, 1996, ann.new York acad.sci.792: 62-8-71; Cabanes-Macheteau et al, 1999, Glycobiology 9: 365-; ruggiero et al, 2000, FEBS lett.469: 132-: 168-; zhang et al, 2000, j.anim.sci.78: 2868-: 302-305; richter et al, 2000, nat. biotechnol.18: 1167-1171; chong et al, 2000, Transgenic Res.9: 71-78), alfalfa (Wigdorovitz et al, 1999, Virology 255: 347-; perrin et al, 2000, mol. breed.6: 345-352), rice (Oryza sativa; stoger et al, 2001; plant mol.biol.42: 583-; kornyeyeyeye et al, 2001, Physiolplant 113: 323-; petersen et al, 2002, Plant Mol Biol 49: 45-58); wheat (Triticumspp.; Pellegrineschi et al, 2002, Genome 45: 421-.
If it is desired to express the recombinant nucleic acid in a whole plant, rather than in cultured cells, the plant cells can first be transformed with DNA encoding the peptide, followed by regeneration of the plant. This includes tissue culture procedures that are generally optimized for each plant species. Plant regeneration procedures are well known in the art for many species. In addition, one skilled in the art can develop protocols for other species using routine experimentation. A number of laboratory manuals are available which describe Plant regeneration programs, including but not limited to Smith (2000, Plant tissue culture: techniques and Experiments, Academic Press, San Diego), Bhojwai and Razdan (1996, Plant tissue culture: the organism and practice, Elsevier Science Pub., Amsterdam), Islam (1996, Plant tissue culture, Oxford & IBH Pub.Co., New Delhi, India), Dodds and Roberts (1995, Experiments in Plant tissue culture, New York: Cambriverse Press, Cambridge UK), Bhowai (Plant tissue culture: applications, and engineering, Plant tissue culture, and culture, Plant tissue culture, Inc., Boswervey, Bosweelware, Bosweelkulture, Plant culture, and grade), and gradient (2000, Plant tissue culture: applications, engineering Press, and Plant tissue culture, Plant culture, Inc., Bosweelware, Plant culture, and culture).
Although purification of recombinant peptides from plants can be expensive, several systems have been developed to minimize this cost. One approach directs synthetic peptides to the endosperm of seeds where they are readily extracted (Wright et al, 2001, Transgenic Res.10: 177-. An alternative approach is to co-extract the recombinant peptide with conventional plant products such as starch, meal or oil. In oilseed rape, the oleosin-hurudin fusion peptide, when expressed in plants, attaches to the oil bodies of the seeds and can be extracted from the Plant seeds together with the oil (Parmenter, 1995, Plant mol. biol. 29: 1167-1180; U.S. Pat. Nos. 5,650,554, 5,792,922, 5,948,682 and 6,288,304 and U.S. application 2002/0037303, all of which are incorporated herein by reference in their entirety). In one variant of this method, oleosins are fused to peptides having affinity for the target peptide that is co-expressed exogenously (U.S. Pat. No.5,856,452, incorporated herein by reference in its entirety).
Expression of recombinant peptides in plant plastids, such as chloroplasts, produces peptides that do not have glycan structures thereon, similar to that found in prokaryotes. However, the yield of such peptides when expressed in such plant organelles is extremely large and such expression systems may have advantages over other systems. For a general review of the expression techniques of exogenous peptides in higher plant plastids, see Hager and Beck (2000, appl. microbiol. biotechnol. 54: 302-310 and references cited therein). Plastid expression is particularly successful in tobacco (see e.g., Staub et al, 2000, nat. Biotechnol.18: 333-338).
F. Transgenic animals
Introduction of the recombinant DNA into a fertilized egg of an animal (e.g., a mammal) can be accomplished using any number of standard techniques in transgenic animal technology. See, e.g., Hogan et al, Manipulating the Mouse Embryo: a Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., 1986; and U.S. Pat. No.5,811,634, which is incorporated herein by reference in its entirety. More generally, recombinant DNA can be introduced into embryos by pronuclear microinjection (Gordon et al, 1980, PNAS 77: 7380-. Microinjection has the advantage of being applicable to a large number of species. Pre-implantation embryos can also be transformed with retroviruses (Jaenisch and Mintz, 1974, Proc. Natl. Acad. Sci. U.S.A.71: 1250-. Retrovirus-mediated transformation has the advantage that a single copy of the recombinant nucleic acid can be added to the cell, but it produces a high level of mosaicism. Recently, embryonic stem Cell-mediated techniques (Gossler et al, 1986, Proc. Natl. Acad. Sci. U.S.A.83: 9065-9069), transfer of whole chromosome fragments (Lavitrano et al, 1989, Cell 57: 717-723), and gamete transfection combined with in vitro fertilization (Lavitrano et al, 1989, Cell 57: 717-723) have been applied. Several books disclosing laboratory procedures for these techniques have been published: Cid-Arregui and Garc i a-Carranc (1998, Microinjection and Protocols, Springer, Berlin), Clarke (2002, Transgenesis technologies: Principles and Protocols, Humana Press, Totowa, NJ) and Pinkert (1994, Transgenic animal technology: A Laboratory Handbook, Academic Press, San Diego).
Once the recombinant DNA is introduced into the egg, the egg is incubated for a short period of time and then transferred to a pseudopregnant animal of the same species from which the egg was obtained (Hogan et al, supra). In the case of mammals, generally 125 eggs are injected per experiment, of which approximately two thirds will survive the procedure. 20 viable eggs were transferred to pseudopregnant mammals, 4-10 of which would develop into live offspring. Typically, 10-30% of the progeny (in the case of mice) carry recombinant DNA.
Although whole animals can be used as expression systems for the peptides of the invention, in a preferred embodiment, the exogenous peptide accumulates in the product of the animal from which it can be harvested without harming the animal. In a preferred embodiment, the exogenous peptide accumulates in milk, eggs, hair, blood and urine.
Suitable mammals are ruminants, ungulates, domesticated mammals and milk producing animals if the recombinant peptide accumulates in the milk of the animal. Particularly preferred animals are goats, sheep, camels, cows, pigs, horses, bulls and llamas (llamas). Techniques for generating transgenic cows that accumulate recombinant peptides in their milk are well known: see Newton (1999, J.Immunol. methods 231: 159-. The production of transgenic animals that produce the desired recombinant peptides is commercially available from GTCBiotherapeutics, Framingham, MA.
If the recombinant peptide is intended to accumulate in eggs, suitable birds include, but are not limited to, chickens, geese and turkeys. Other target animals include, but are not limited to, birds, other species, fish, reptiles, and amphibians. The introduction of recombinant DNA into chickens by retroviral transformation is well known in the art: thoraval et al (1995, Transgenic Research 4: 369-. Successful transformation of chickens with recombinant DNA was also achieved by introducing DNA into blastodermal cells and introducing the thus transfected blastodermal cells into embryos: brazolot et al (1991, mol. reprod. Dev.30: 304-. High throughput techniques have been developed to assess whether transgenic chickens express the desired peptide (Harvey et al, 2002, Poult. Sci.81: 202-212, U.S. Pat. No.6,423,488, incorporated herein by reference in its entirety). The exogenous beta-lactamase accumulated in the egg white of chickens upon retroviral transformation of chickens with recombinant DNA (Harvey et al, 2002, nat. Biotechnol.20 (4): 396-399). Production of chickens that produce exogenous peptides in eggs is commercially available from AviGenics, inc.
G. Bacteria
Recombinantly expressed peptides produced in bacteria are typically not glycosylated. However, bacterial systems capable of glycosylating peptides have become apparent and therefore glycosylated recombinant peptides may be produced in bacteria in the future.
Many bacterial expression systems are known in the art. Preferred bacterial species include, but are not limited to, Escherichia coli and Bacillus species.
Expression of recombinant peptides in E.coli is well known in the art. Procedures for e.coli based expression systems can be found in u.s.appln No.20020064835, U.S. patent nos. 6,245,539, 5,606,031, 5,420,027, 5,151,511, RE33,653, and the like. Methods for transforming bacteria include, but are not limited to, calcium chloride (Cohen et al, 1972, Proc. Natl. Acad. Sci. U.S.A.69: 2110-2114; Hanahan, 1983, J.mol. biol. 166: 557-580; Mandel and Higa, 1970, J.mol. biol. 53: 159-162) and electroporation (Shigekawa and Dow, 1988, Biotechniques 6: 742-580), and those described in Sambrook et al, 2001 (supra). For a review of the laboratory procedures for Microbial transformation and expression systems, see Saunders and Saunders (1987, microbiological Genetics Applied to Biotechnology: Principles and technologies of Gene Transfer and management, Croom Helm, London), Suhler (1993, Genetic Engineering of Microorganisms, Weinheim, New York), Lee et al (1999, Metabolic Engineering, Marcel Dekker, New York), Adolph (1996, microbiological genom Methods, CRC Press, Boca Ras), and Birren and Lai (1996, Nonmarian genomic analysis: A practical chemical guide, Academic Press, Academic digital).
For a general review of the literature on peptide expression in E.coli, see Balbas (2001, mol. Biotechnol.19: 251-. Several companies currently offer the option of selecting bacterial strains for expression of mammalian peptides, such as Rosetta of E.coliTMStrains (Novagen, inc., Madison, Wis.; with enhanced expression of eukaryotic codons and enhanced disulfide bond formation not normally used in bacterial cells).
H. Cell engineering
It will be apparent from the disclosure of the present invention that the more uniform the starting material produced by the cell, the more efficient it is to produce large amounts of peptide with the desired glycosylation in vitro. Thus, engineering host cells to produce uniformly glycosylated peptides as disclosed herein as starting materials for in vitro enzymatic reactions provides significant advantages over using peptide starting materials having heterogeneous glycan structures attached thereto. One preferred peptide starting material for use in the present invention is a peptide having predominantly glycan molecules consisting exclusively of a basic trimannose core structure. Another preferred starting material is Man3GlcNAc 4. After the reconstitution process, the preferred peptides will yield the maximum amount of peptides with the desired glycosylation, thus improving clinical efficacy. However, other glycan starting materials are also suitable for use in the methods described herein, e.g., high mannose can be easily tailored in vitro to the basic trimannose core structure with a range of mannosidases. As described elsewhere, other glycan starting materials can also be used, provided that all extraneous sugar moieties are cleaved to yield the basic trimannose core structure or Man3GlcNAc 4. Thus, the purpose of using genetically engineered cells to produce the peptides of the invention is to produce peptides with glycan structures attached thereto that are as uniform as possible, wherein the glycan structures can be reconstituted in vitro to produce peptides with the desired glycosylation. This will result in a drastic reduction in the production costs of these peptides. Since glycopeptides produced by this method have predominantly the same N-linked glycan structure, post-production modification protocols can be standardized and optimized to produce greater batch-to-batch consistency of the final product. As a result, the heterogeneity of the final finished chain product may be lower than those currently available. The product will have improved biological half-life and biological activity compared to prior art products. Alternatively, the present invention can be used to introduce limited and specific heterogeneity, if desired, such as by selecting reaction conditions that result in differential addition of sugar moieties.
Preferably (and not strictly necessary), the genetically engineered cell is one that produces a peptide having a glycan structure consisting essentially of the basic trimannose core structure or Man3GlcNAc 4. The minimum requirements are: the ratio of these preferred structures produced by the genetically engineered cells must be sufficient to produce a peptide with the desired glycosylation after reconstitution procedures.
In general, any eukaryotic cell may be modified to become a host cell of the invention. First, the glycosylation pattern of endogenous and biologically produced recombinant glycopeptides is determined to identify additions/deletions of the enzymatic activity that result in the production of the basic trimannose core glycopeptide or the Man3GlcNAc4 glycopeptide. This generally requires the deletion of glycosyltransferase activity using trimannoglycopeptides as substrates and the increased degradation of more complex N-linked glycans to produce shorter chain enzyme activities. In addition, genetically engineered cells can produce high mannose glycans that can be cleaved with mannosidase enzymes to produce the desired starting glycan structures. The mannosidases may be active in vivo in a cell (i.e., the cell is genetically engineered to produce the enzyme), or they may be used in reactions after in vitro production.
Techniques for genetically modifying host cells to alter the glycosylation properties of expressed peptides are well known. See, e.g., Altmann et al (1999, Glycoconjugate J.16: 109-.
Techniques also exist to genetically alter the glycosylation profile of peptides expressed in E.coli. Coli has been processed to produce oligosaccharides in vivo using various glycosyltransferases from the bacteria Neisseria meningitidis and Rhizobium azotobacterium (Bettler et al, 1999, Glycoconj. J.16: 205-212). Coli genetically engineered to overexpress the β 1, 3N acetylglucosaminyltransferase lgTA gene of Neisseria meningitidis will effectively glycosylate exogenous lactose (Priem et al, 2002, Glycobiology 12: 235-240).
Fungal cells have also been genetically modified to produce exogenous glycosyltransferases (Yoshida et al, 1999, Glycobiology 9 (1): 53-58; Kalsner et al, 1995, glycon j.J.12: 360-.
Thus, in one aspect, the invention provides a cell that glycosylates a population of glycopeptides such that a portion of the glycopeptides produced have a basic trimannose core or Man3GlcNAc4 structure. Preferably, the cell produces a peptide having a glycan structure consisting exclusively of a trimannose core. The minimum requirements are: the proportion of peptides having a basic trimannose core or Man3GlcNAc4 structure must be sufficient to produce peptides with the desired glycosylation after the reconstitution process. One or more heterologous nucleic acid expression units are introduced into the cell, each of which expression units may comprise one or more nucleic acid sequences encoding one or more peptides of interest. The native form of the glycopeptide of interest may comprise one or more complex N-linked glycans or may simply be high mannose glycans.
The cell may be any type of cell, and is preferably a eukaryotic cell. The cell may be a mammalian cell, such as a human, mouse, rat, rabbit, hamster, or other type of mammalian cell. When the cell is a mammalian cell, the mammalian cell can be derived from or contained in a non-human transgenic mammal, wherein the cells in the mammal encode the desired glycopeptide and the various glycosylation and glycosidase enzymes necessary for production of the desired glycopeptide molecule. Furthermore, the cell may be a fungal cell, preferably a yeast cell, or the cell may be an insect or plant cell. Similarly, when the cell is a plant cell, the plant cell may be derived from or contained in a transgenic plant, wherein the plant encodes the desired glycopeptide and the various glycosylation and glycosidase enzymes necessary for production of the desired glycopeptide molecule.
In some embodiments, the host cell may be a eukaryotic cell expressing one or more heterologous glycosyltransferases and/or one or more heterologous glycosidases, wherein expression of the recombinant glycopeptide in the host cell results in the production of a recombinant glycopeptide having a substantially trimannose core as the primary glycan structure attached thereto.
In some embodiments, the heterologous glycosyltransferase used in the cell may be selected from any known glycosyltransferase included in the list of glycosyltransferase families of, for example, Taniguchi et al (2002, Handbook of Glycosyltransferases and Related Genes, Springer, New York).
In other embodiments, the heterologous glycosylase may be selected from the group consisting of mannosidase 1, mannosidase 2, mannosidase 3, and other mannosidase enzymes, including but not limited to microbial mannosidase. Additional disclosure regarding enzymes for use in the present invention is provided elsewhere.
In yet other embodiments, the host cell may be a eukaryotic cell in which one or more endogenous glycosyltransferases and/or one or more endogenous glycosidases have been inactivated, such that expression of the recombinant glycopeptide in the host cell results in production of a recombinant glycopeptide having a substantially trimannose core as the predominant glycan structure attached thereto.
In additional embodiments, the host cell may express a heterologous glycosyltransferase and/or glycosidase while one or more endogenous glycosyltransferases and/or glycosidases are inactivated. Endogenous glycosyltransferases and/or glycosidases may be inactivated by any technique known to those skilled in the art, including, but not limited to, antisense techniques and techniques involving insertion of nucleic acids into the genome of a host cell. In some embodiments, the endogenous enzyme may be selected from GnT-I, mannosidase, xylosyltransferase, core α 1, 3 fucosyltransferase, serine/threonine O-mannosyltransferase, and the like.
Alternatively, expression systems can be used that naturally glycosylate the peptide so that the N-linked glycans are predominantly of the trimannose core type or of the Man3GlcNAc4 type. An example of a cell type that produces a trimannose core is Sf9 cells. Other such systems can be identified by analyzing glycopeptides that are naturally or recombinantly expressed in cells and selecting those that exhibit the desired glycosylation characteristics. The invention should be construed to include any or all of these cells for producing the peptides of the invention.
Glycan reconstituted and/or glycoconjugated peptides Purification of (2)
If the modified glycoprotein is produced or secreted intracellularly, the first step may be to remove particulate debris or fragments of host cell lysis, for example by centrifugation or ultrafiltration; alternatively, the protein may be concentrated using a commercially available protein concentration filter, followed by separation of the peptide variant from other impurities by one or more steps selected from immunoaffinity chromatography, ion exchange column fractionation (such as Diethylaminoethyl (DEAE) or matrices containing carboxymethyl or sulfopropyl groups), chromatography on Blue-Sepharose, CM Blue-Sepharose, MONO-Q, MONO-S, Lectins-Sepharose, WGA-Sepharose, Con A-Sepharose, Ether Toyopearl, butyl Toyopearl, phenyl Toyopearl or protein A Sepharose, SDS-PAGE chromatography, silica chromatography, chromatofocusing, reverse phase HPLC (RP-HPLC), gel filtration using e.g.Sephadex molecular sieves or size exclusion chromatography, chromatography on columns selectively binding the peptide, and precipitation with ethanol, pH or ammonium sulfate, membrane filtration and various techniques.
The modified peptides produced in culture are typically isolated by initial extraction from cells, enzymes, etc., and subsequent separation by one or more of concentration, salting out, aqueous phase ion exchange, or size exclusion chromatography steps. In addition, the modified glycoprotein can be purified by affinity chromatography. HPLC may then be used for the final purification step.
A protease inhibitor such as phenylmethylsulfonyl fluoride (PMSF) may be included in any of the foregoing steps to inhibit proteolysis, and an antibiotic may be included to prevent the growth of adventitious contaminants.
In another embodiment, the supernatant from the system producing the modified peptide of the invention is first concentrated using a commercially available protein concentration filter, such as an Amicon or Millipore Pellicon ultrafiltration unit. After the concentration step, the concentrate can be applied to a suitable purification matrix. For example, a suitable affinity matrix may comprise a ligand for a peptide, a lectin or an antibody molecule bound to a suitable support. Alternatively, an anion exchange resin may be used, such as a matrix or substrate having extended DEAE groups. Suitable matrices include acrylamide, agarose, dextran, cellulose or other types commonly used in protein purification. Alternatively, a cation exchange step may be applied. Suitable cation exchangers include various insoluble matrices containing sulfopropyl or carboxymethyl groups. Sulfopropyl groups are particularly preferred.
One or more RP-HPLC steps using hydrophobic RP-HPLC media such as silica gel with extended methyl or other aliphatic groups can then be applied to further purify the peptide variant composition. Various combinations of some or all of the foregoing purification steps may also be used to provide a homogeneous modified glycoprotein.
The modified peptides of the invention obtained from large scale fermentation can be prepared by reaction with a peptide prepared by Urdal et al, j.chromatography.296: 171(1984) was purified by a similar method. This reference describes two sequential RP-HPLC steps for purification of recombinant human IL-2 on a preparative HPLC column. Alternatively, techniques such as affinity chromatography may be applied to purify the modified glycoprotein.
Preferred peptides and nucleic acids encoding the same
The present invention includes isolated nucleic acids and similar molecules or fragments thereof that encode various peptides and proteins. The present invention should not be construed as in any way limited to the use of these peptides in the methods of the present invention only, but should be construed to include any and all peptides that are or will become available to those of skill in the art. Furthermore, the present invention should not be construed as including only one particular nucleic acid or amino acid sequence of a peptide listed herein, but rather should be construed to include any and all variants, homologs, mutants, and the like of each peptide. It should be noted that when a peptide is identified as having a mutation or other alteration in the peptide sequence, unless otherwise indicated herein, the numbering of the altered or mutated amino acid is determined such that the first amino acid in the mature peptide sequence is amino acid 1.
Preferred peptides include, but are not limited to, human granulocyte colony stimulating factor (G-CSF), human interferon alpha (IFN-alpha), human interferon beta (IFN-beta), human coagulation factor VII (FactorVII), human coagulation factor IX (FactorIX), human Follicle Stimulating Hormone (FSH), human Erythropoietin (EPO), human granulocyte/macrophage colony stimulating factor (GM-CSF), human interferon gamma (IFN-gamma), human alpha-1-protease inhibitors (also known as alpha-1-antitrypsin or alpha-1-trypsin inhibitors; A-1-PI), glucocerebrosidase, human tissue-type activating factor (TPA), human interleukin-2 (IL-2), human coagulation factor VIII (FactorVIII), tumor necrosis factor receptor 75kDa fused to the Fc portion of human IgG immunoglobulins, known commercially as ENBRELTMOr ETANERCEPTTM(chimeric TNFR), human urokinase (urokinase), specific binding glycoprotein IIb/IIIa and vitronectin alphavβ3Fab fragments of human/murine chimeric monoclonal antibodies to the receptor, commercially known as REOPROTMOr ABCIXIMAB (chimeric anti-glycoprotein IIb/IIIa), a human/murine chimeric monoclonal antibody that specifically binds human HER2, known commercially as HERCEPTINTM(chimeric anti-HER 2), human/murine chimeric antibody that specifically binds to the F protein or A antigenic site of respiratory syncytial virus, commercially known as SYNAGIS TMOr PALIVIZUMAB (chimeric anti-RSV), a chimeric human/murine monoclonal antibody that specifically binds CD20 on human B-cells, known commercially as RITUXANTMOr RITUXAMAB (chimeric anti-CD 20), human recombinant dnase (dnase), chimeric human/mouse monoclonal antibody that specifically binds human tumor necrosis factor, known commercially as REMICADETMOr INFLIXIMAB (chimeric anti-TNF), human insulin, surface antigen of hepatitis B virus (adw subtype; HBsAg), and Human Growth Hormone (HGH), alpha-galactosidase A (Fabryzyme)TM) Alpha-iduronidase (Aldurazyme)TM) Antithrombin (antithrombin III, AT-III), human chorionic gonadotropin (hCG), interferon omega, and the like.
The isolated nucleic acids of the present invention should be construed to include RNA or DNA sequences encoding any of the above-described peptides of the present invention and modified forms thereof, including chemical modifications to the DNA or RNA to render the nucleotide sequence more stable when free of or associated with a cell. As a non-limiting example, oligonucleotides containing at least one phosphorothioate modification are known to confer enhanced resistance to nucleases. Specific examples of modified oligonucleotides include those containing phosphorothioate, phosphotriester, methylphosphonate, short chain alkyl or cycloalkyl intersugar linkages or short chain heteroatom or heterocyclic intersugar ("backbone") linkages. Furthermore, oligonucleotides having a morpholine backbone structure (U.S. Pat. No. 5,034,506) or a polyamide backbone structure (Nielsen et al, 1991, Science 254: 1497) can also be used.
Chemical modifications of nucleotides can be used to enhance the efficiency with which a cell takes up a nucleotide sequence and the efficiency with which the nucleotide sequence is expressed in the cell. The present invention relates to any and all combinations of modifications to nucleotide sequences.
The present invention should not be construed as limited to only the nucleic acid and amino acid sequences disclosed herein. As described in more detail elsewhere, once read the present invention, it will be apparent to those skilled in the art that other nucleic acids encoding the peptides of the present invention can be obtained by the procedures described herein (i.e., site-directed mutagenesis, frameshift mutagenesis, etc.) and techniques well known in the art.
Also included are isolated nucleic acids encoding peptide fragments wherein the peptide fragments retain the desired biological activity of the peptide. Furthermore, although exemplary nucleic acids encoding peptides are disclosed herein with particular SEQ ID NOS, the present invention should in no way be construed as being limited to any particular nucleic acid disclosed herein. Rather, the invention should be construed to include any and all nucleic acid molecules having a sufficient percent identity to the sequences disclosed herein such that the nucleic acids also encode peptides having the desired biological activities disclosed herein. Also contemplated are isolated nucleic acids that are shorter than the full-length nucleic acid, which retain the biological activity of the peptide encoded thereby. Methods for determining percent identity between one nucleic acid and another are disclosed elsewhere herein as are assays for determining the biological activity of any particular preferred peptide.
Also as disclosed elsewhere herein, any other procedure can be used to produce derivatives, mutants or variant forms of the peptides of the invention using recombinant DNA methods well known in the art, such as those described in Sambrook et al (1989, molecular cloning, A Laboratory Manual, Cold Spring Harbor Laboratory Press, New York) and Ausubel et al (1997, Current Protocols in molecular Biology, Green & Wiley, New York). Procedures for introducing amino acid changes in peptides or polypeptides by altering the DNA sequence encoding the peptide are well known in the art and are also described in Sambrook et al (1989, supra); ausubel et al (1997, supra).
The invention includes nucleic acids encoding G-CSF, IFN-alpha, IFN-beta, factor VII, factor IX, FSH, EPO, GM-CSF, IFN-gamma, A-1-PI, glucocerebrosidase, TPA, IL-2, factor VIII, chimeric TNFR, urokinase, chimeric anti-glycoprotein IIb/IIIa, chimeric anti-HER 2, chimeric anti-RSV, chimeric anti-CD 20, DNase, chimeric anti-TNF, human insulin, HBsAg, and HGH, wherein a nucleic acid encoding a marker peptide is covalently linked thereto. That is, the present invention encompasses chimeric nucleic acids in which a nucleic acid sequence encoding a marker peptide is covalently linked to a nucleic acid encoding a peptide of the present invention. Such tag peptides are well known in the art and include, for example, Green Fluorescent Protein (GFP), myc-pyruvate kinase (myc-PK), His 6Maltose Binding Protein (MBP), influenza virus hemagglutinin tag polypeptide, FLAG (FLAG) tag polypeptide (FLAG), and glutathione-S-transferase (GST) tag polypeptide. However, the present invention should in no way be construed as being limited to nucleic acids encoding the above-listed marker peptides. Rather, any nucleic acid sequence encoding a peptide that can function in a substantially similar manner to these marker peptides should be construed as being included in the present invention.
Nucleic acids comprising nucleic acids encoding a marker peptide can be used to localize the peptides of the invention in cells, tissues and/or whole organisms (e.g., mammalian embryos), detect the secretion of the peptides of the invention from cells and study the effect of the peptides in cells. Further, the addition of a labeled peptide facilitates the isolation and purification of the "labeled" peptide, so that the peptide of the present invention can be easily produced and purified.
The present invention includes the following preferred isolated peptides: G-CSF, IFN-alpha, IFN-beta, factor VII, factor IX, FSH, EPO, GM-CSF, IFN-gamma, A-1-PI, glucocerebrosidase, TPA, IL-2, factor VIII, chimeric TNFR, urokinase, chimeric anti-glycoprotein IIb/IIIa, chimeric anti-HER 2, chimeric anti-RSV, chimeric anti-CD 20, DNase, chimeric anti-TNF, human insulin, HBsAg, HGH, alpha-galactosidase A, alpha-iduronidase, antithrombin III, hCG, interferon omega and the like.
The invention should also be construed as encompassing "derivatives," "mutants," and "variants" of the peptides (or DNA encoding the peptides) of the invention that are altered at one or more amino acids (or at one or more base pairs when referring to the nucleotide sequence encoding the peptide), such that the resulting peptides (or DNA) differ from the sequences enumerated herein, but have the same biological properties as the peptides disclosed herein, i.e., the peptides have G-CSF, IFN- α, IFN- β, factor VII, factor IX, FSH, EPO, GM-CSF, IFN- γ, A-1-PI, glucocerebrosidase, TPA, IL-2, factor VIII, chimeric TNFR, urokinase, chimeric anti-glycoprotein Ilb/IIIa, chimeric anti-HER 2, amino acid residues, and amino acid residues, Biological/biochemical properties of chimeric anti-RSV, chimeric anti-CD 20, DNase, chimeric anti-TNF, human insulin, HBsAg and HGH.
Further included are peptide fragments that retain the desired biological activity of the peptide, regardless of their length. It is well within the skill of the artisan to isolate peptides that are shorter than the full-length form of any of the peptides used in the present invention, and to determine which isolated fragments retain the desired biological activity using the assays provided herein and are thus peptides that can be used in the present invention.
The biological properties of the proteins of the present invention should be interpreted to include, but are not limited to, the ability of the peptides to function in biological assays and in the context described herein, such as reducing inflammation, eliminating immune responses, blood coagulation, increased hematopoietic production, protease inhibition, immune system regulation, binding of antigens, growth, alleviation or treatment of disease, DNA cleavage, and the like.
A.G-CSF
The invention comprises methods of modifying glycan structures on G-CSF. G-CSF is a cytokine produced by activated T-cells, macrophages, endothelial cells and stromal fibroblasts, as is well known in the art. G-CSF acts primarily in the bone marrow to increase the production of inflammatory leukocytes and further functions as an endocrine hormone to initiate the recruitment of neutrophils that are consumed during inflammatory functions. G-CSF also has clinical application in bone marrow replacement following chemotherapy.
The reconstituted G-CSF peptide may be administered to a patient selected from a non-myeloid cancer patient receiving myelosuppressive chemotherapy, an Acute Myeloid Leukemia (AML) patient receiving induced or potentiated chemotherapy, a non-myeloid cancer patient receiving a bone marrow transplant, a patient undergoing collection of peripheral blood progenitor cells, a patient suffering from severe chronic neutropenia, and a patient suffering from persistent neutropenia and also suffering from advanced HIV infection. Preferably, the patient is a human patient.
Although G-CSF is important and useful for therapeutic applications in mammals, particularly humans, existing methods of producing G-CSF from recombinant cells result in products with relatively short biological lifetimes, incorrect glycosylation patterns that can potentially lead to immunogenicity, loss of function, and the need for greater amounts and more frequent dosing to achieve the same effect, among other things.
G-CSF has been isolated and cloned and its nucleic and amino acid sequences are set forth in SEQ ID NO: 1 and SEQ ID NO: 2 (fig. 58A and 58B, respectively). The present invention comprises methods of modifying G-CSF, particularly as it relates to the ability of G-CSF to function as a potent and functional biological molecule. While the present disclosure and description herein have been made in the light of the present disclosure, it will be readily understood by those skilled in the art that the present invention provides compositions and methods for modifying G-CSF.
The invention further comprises G-CSF variants, as are well known in the art. By way of example, but not meant to limit the invention in any way, a G-CSF variant has been described in U.S. Pat. No.6,166,183, which describes a G-CSF comprising a natural complement of lysine residues (complement) further linked to one or two polyethylene glycol molecules. Furthermore, U.S. Pat. nos. 6,004,548, 5,580,755, 5,582,823 and 5,676,941 describe G-CSF variants in which one or more cysteine residues at positions 17, 36, 42, 64 and 74 are replaced by alanine or serine. U.S. patent No.5,416,195 describes a G-CSF molecule in which the cysteine at position 17, the aspartic acid at position 27 and the serine at positions 65 and 66 are replaced by serine, proline and proline, respectively. Other variations are well known in the art and are described, for example, in U.S. patent No.5,399,345.
The expression and activity of the modified G-CSF molecules of the invention can be determined using methods well known in the art and described, for example, in U.S. Pat. No.4,810,643. By way of example, activity may be measured using a radiolabeled thymidine uptake assay. Briefly, human bone marrow from healthy donors was density separated (cut) using Ficoll-Hypaque (1.077g/ml, Pharmacia, Piscataway, N.J.) and the low density cells were suspended in Iscove's medium (GIBCO, Jolla, Calif.) containing 10% fetal bovine serum, glutamine and antibiotics. To about 2X 104Human bone marrow cells of (A) with control Medium or G-CSF of the invention on 96-well flat-bottom plates in air 5% CO2Incubate at about 37 ℃ for about 2 days. Then with 0.5. mu. Ci/well3The cultures were pulsed for about 4 hours with H-thymine (New England Nuclear, Boston, Mass.) and uptake was measured as described in, for example, Ventua et al (1983, Blood 61: 781). Compared to bone marrow cells treated with control Compounds3The increase of integration of H-thymine into human bone marrow cells is active and livingAn indication of the force of the G-CSF compound.
IFN alpha, IFN beta and IFN omega
The invention further comprises the reconstitution and modification of IFN alpha, IFN beta And IFN omegaThe method of (1). IFN alpha weight of about 18kDa about 20 peptides family part. IFN omega in structure and function is very similar to IFN alpha. When the host immune response to IFN alpha increases, thereby rendering the treatment ineffective, IFN omega can be used to treat hepatitis C virus infection. anti-IFN alpha antibody and IFN omega cross-reaction. Thus, when IFN α therapy is no longer possible, IFN ω can be used to continue the treatment of hepatitis c.
IFN α, ω and IFN β, collectively known as type I interferons, bind to the same cellular receptor and elicit similar responses. Type I IFNs inhibit viral replication, increase the lytic potential of NK cells, regulate expression of MHC molecules and inhibit cell proliferation, and the like. Type I IFNs have been used as therapeutic agents against viral infections, particularly hepatitis viral infections, and against multiple sclerosis.
The existing compositions of type I IFNs as described above are compounds useful for modulating aberrant immunological responses and as therapeutic agents for various diseases. However, they are hampered by reduced capacity and function and limited in vivo half-life compared to native cytokines comprising glycosylated native complement.
The reconstituted IFN α peptide can be administered to a patient selected from the group consisting of a patient with hairy cell leukemia, a patient with malignant melanoma, a patient with follicular lymphoma, a patient with condyloma acuminatum, a patient with AIDS-related kaposi's sarcoma, a patient with hepatitis c, a patient with hepatitis b, a patient with human papillomavirus infection, a patient with Chronic Myelogenous Leukemia (CML), a patient with chronic philadelphia chromosome (Ph) positive chronic myelogenous leukemia, a patient with non-hodgkin's lymphoma (NHL), a patient with lymphoma, a patient with bladder cancer, and a patient with renal cancer. Preferably, the patient is a human patient.
The reconstituted IFN beta peptide may be administered to a patient selected from the group consisting of a patient having Multiple Sclerosis (MS), a patient having hepatitis b, a patient having hepatitis c, a patient having a human papillomavirus infection, a patient having breast cancer, a patient having brain cancer, a patient having colorectal cancer, a patient having pulmonary fibrosis, and a patient having rheumatoid arthritis. Preferably, the patient is a human patient.
Reconstituted IFNωThe peptide can be administered to a patient selected from the group consisting of a patient with hairy cell leukemia, a patient with malignant melanoma, a patient with follicular lymphoma, a patient with condyloma acuminatum, a patient with AIDS-related Kaposi's sarcoma, a patient with hepatitis C, a patient with hepatitis B, a patient with a human papilloma virus infection, a patient with Chronic Myelogenous Leukemia (CML), a patient with chronic Philadelphia chromosome (Ph) positive chronic myelogenous leukemia, a patient with non-Hodgkin's lymphoma (NHL), a patient with lymphoma, a patient with bladder cancer, and a patient with renal cancer. Preferably, the patient is a human patient.
The prototype nucleotide and amino acid sequences of IFN α are herein represented by SEQ ID NOs: 3 and SEQ ID NO: 4 (fig. 59A and 59B, respectively). The prototype nucleotide and amino acid sequences of IFN ω are herein represented by SEQ ID NO: 74 and SEQ ID NO: 75 (fig. 84A and 84B, respectively). IFN β comprises a single gene product of about 20kDa whose nucleic and amino acid sequences are represented herein by SEQ ID NOs: 5 and SEQ ID NO: 6 (FIGS. 60A and 60B, respectively). The present invention is not limited to the nucleotide and amino acid sequences herein. Those skilled in the art will readily appreciate that many variants of IFN exist, either naturally or as processed derivatives. Similarly, IFN beta has been modified to achieve more favorable therapeutic properties. Examples of modified type I IFNs are well known in the art (see table 9) and are described, for example, in U.S. patent 6,323,006, wherein cysteine-60 is replaced by tyrosine, and also in U.S. patent nos. 4,737,462, 4,588,585, 4,545,723 and 6,127,332, wherein IFN β is described with various amino acid substitutions. In addition, U.S. Pat. Nos. 4,966,843, 5,376,567, 5,795,779 describe IFN α -61 and IFN-76. U.S. Pat. Nos. 4,748,233 and 4,695,543 describe IFN α gx-1, while U.S. Pat. No.4,975,276 describes IFN α -54. Furthermore, U.S. patent nos. 4,695,623, 4,897,471, 5,661,009 and 5,541,293 all describe consensus IFN α sequences representing all variants known at the filing date. Although this list of type I IFNs and variants thereof is by no means exhaustive, it will be readily understood by those skilled in the art that the present invention encompasses known or future discovered IFN β and IFN α molecules, derivatives and variants.
TABLE 9 Interferon-alpha isoforms
Methods for expressing IFN in recombinant cells are well known in the art and are readily accomplished using techniques described in, for example, U.S. Pat. No.4,966,843 and Sambrook et al (2001, Molecular Cloning, A Laboratory Manual, Cold spring harbor Laboratory Press, New York) and Ausubel et al (1997, Current protocols in Molecular Biology, Green & Wiley, New York).
Assays for determining the biological activity of type I IFN modified by the invention are well known to those skilled in the art. For example, the assays described in Rubinstein et al (1981, journal of Virology 37: 755-. This method is only one of many methods known in the art for determining the biological function of type I IFNs.
C. Blood coagulation factor VIIa
The invention further comprises methods of reconstituting and modifying factor VII. The blood coagulation pathway is a complex reaction involving many events. One intermediate event in the pathway is factor VII, a zymogen (factor VIIa after activation) that participates in the extrinsic pathway of blood coagulation by converting factor X to Xa in the presence of tissue factor and calcium ions. Factor Xa then converts prothrombin to thrombin in the presence of factor Va, calcium ions and phospholipids. Factor X activation to factor Xa is a common event in both the intrinsic and extrinsic blood coagulation pathways, and factor VIIa is therefore useful in treating patients with factor VIII deficiency or inhibition. There is also evidence that factor VIIa may also be involved in the intrinsic pathway, thus increasing the prominence and importance of factor VII's role in blood coagulation.
Factor VII is a single-chain glycoprotein with a molecular weight of about 50 kDa. In this form, the factor circulates in the blood as an inactive zymogen. The activation of factor VII to factor VIIa can be catalyzed by several different plasma proteases, such as factor XIIa. Activation of factor VII results in the heavy and light chains being held together by at least one disulfide bond. Further, modified factor VII molecules are described which are not convertible to factor VIIa and which are useful as anti-coagulation agents, e.g. in case of blood clots, thrombi, etc. In view of the importance of factor VII in the blood coagulation pathway and its use as a treatment for increased and decreased levels of coagulation, molecules with longer biological half-lives, increased capacity, and generally more similar therapeutic properties to wild-type factor VII synthesized and secreted in healthy humans would be advantageous and useful in the treatment of blood coagulation disorders.
The reconstituted factor VII peptide may be administered to a patient selected from hemophiliacs with bleeding episodes; patients with hemophilia a; patients with hemophilia B; a patient suffering from hemophilia a, wherein the patient also has antibodies against factor VIII; a patient suffering from hemophilia B, wherein the patient also has antibodies against factor IX; patients with cirrhosis of the liver; cirrhosis patients with orthotopic liver transplantation; sclerosant patients with upper gastrointestinal bleeding; patients with bone marrow transplants and patients with liver resections. Preferably, the patient is a human patient.
Factor VII has been cloned and sequenced, and its nucleic and amino acid sequences are herein identified as SEQ ID NO: 7 and SEQ ID NO: 8 (fig. 61A and 61B, respectively). The present invention should in no way be construed as being limited to the factor VII nucleic acid and amino acid sequences set forth herein. Variants of factor VII are described, for example, in U.S. patent nos. 4,784,950 and 5,580,560, wherein lysine-38, lysine-32, arginine-290, arginine-341, isoleucine-42, tyrosine-278, and tyrosine-332 are replaced by various amino acids. Further, U.S. patent nos. 5,861,374, 6,039,944, 5,833,982, 5,788,965, 6,183,743, 5,997,864, and 5,817,788 describe factor VII variants that cannot be cleaved to form factor VIIa. The skilled artisan will recognize that the blood coagulation pathway and the role of factor VII therein is well known and thus many naturally occurring and processed variants as described above are encompassed within the present invention.
Methods for expressing and determining factor VII activity are well known in the art and are described, for example, in U.S. patent No.4,784,950. Briefly, expression of factor VII or variants thereof can be achieved in a variety of prokaryotic and eukaryotic systems, including e.coli, CHO cells, BHK cells, insect cells employing baculovirus expression systems, all of which are well known in the art.
Assays for the activity of modified factor VII prepared according to the methods of the present invention are well known in the art. As a non-limiting example, Quick et al (HemorragicDisase and Thrombosis, 2)nded., Leat Febiger, Philadelphia, 1966) describes a one-stage clotting assay for determining the biological activity of factor VII molecules prepared according to the methods of the present invention.
D. Blood coagulation factor IX
The invention further comprises methods of reconstituting and/or modifying coagulation factor IX. As mentioned above, coagulation factor IX is crucial in the blood coagulation cascade. Deficiencies in the body of clotting factor IX are characteristic of a class of hemophilias (type B). Treatment of this disease is generally limited to human plasma protein concentrates infused with factor IX. However, in addition to the disadvantages of time and expense of the procedure, the infusion of blood concentrates involves the risk of delivering viral hepatitis, acquired immunodeficiency syndrome or thromboembolic disease to the recipient.
While factor IX itself has proven to be an important and useful compound for therapeutic applications, current methods of producing factor IX from recombinant cells (U.S. patent No.4,770,999) result in products with rather short biological lifetimes, incorrect glycosylation patterns that can potentially lead to immunogenicity, loss of function, and the need for greater amounts and more frequent dosing to achieve the same effect, among other things.
The reconstituted factor IX peptide may be administered to a patient selected from a hemophilia patient having bleeding episodes and also having hemophilia B, a patient having hemophilia B and also having antibodies to anti-coagulation factor IX, a patient having liver cirrhosis, a cirrhosis patient having an orthotopic liver transplant, a cirrhosis patient having upper gastrointestinal bleeding, a patient having a bone marrow transplant, and a patient having a liver resection. The reconstituted factor IX peptide may also be administered to control and/or prevent bleeding episodes in patients with hemophilia B, congenital factor IX deficiency, or creasmase disease. The reconstituted factor IX peptide may also be administered to a patient to control and/or prevent bleeding episodes in the patient during surgery. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of coagulation factor IX are herein represented by SEQ ID NO: 9 and SEQ ID NO: 10 (fig. 62A and 62B, respectively). The present invention is in no way limited to the sequences set forth herein. Factor IX variants are well known in the art, as described in U.S. patent No.4,770,999, 5,521,070 in which the tyrosine at the first position is replaced by an alanine, U.S. patent No.6,037,452 in which factor XI is linked to an alkylene oxide group, and U.S. patent No.6,046,380 in which the DNA encoding factor IX is modified at least one splice site. As demonstrated herein, variants of factor IX are well known in the art, and the present disclosure encompasses those variants that are known or that may be developed or discovered in the future.
The method of determining modified Coagulation factor IX prepared according to the methods of the present invention may be accomplished as described above or alternatively by methods well known in the art, such as the one-step activated topical tissue thromboplastin time assay, as described in Biggs (1972, Humanblood Cooperation Haemostasis and Thrombosins (Ed.1), Oxford, Blackwell, Scientific, pg.614). Briefly, to determine the biological activity of a factor IX molecule developed according to the methods of the present invention, assays may be performed using equal volumes of activated topical tissue thromboplastin reagent, factor IX deficient plasma isolated from patients with hemophilia B using sterile phlebotomy techniques well known in the art, and normally collected plasma as a standard or sample. In this assay, 1 unit of activity is defined as the amount present in 1 ml of normally collected plasma. Further, assays based on the ability of factor IX to reduce plasma clotting time from factor IX deficient patients to normal time can be performed, as described in Proctor and Rapaport (1961, Amer.J.Clin.Path.36: 212).
E.FSH
The invention further includes methods of reconstituting and/or modifying FSH. The human reproductive function is controlled in part by a heterodimeric human glycoprotein hormone family that has a common 92 amino acid glycoprotein alpha subunit, but differs in its hormone-specific beta subunit. This family includes Follicle Stimulating Hormone (FSH), Luteinizing Hormone (LH), thyrotropin or Thyroid Stimulating Hormone (TSH) and human chorionic gonadotropin (hCG). Human FSH and LH are therapeutically useful for regulating various aspects of reproduction-related metabolism in human females. For example, partially purified FSH from urine can be used clinically to stimulate follicle maturation in ovarieless women with anovulatory syndrome or luteal phase deficiency. Luteinizing Hormone (LH) and FSH may be used in combination to stimulate the development of follicles for in vitro fertilization. The role of FSH in the reproductive cycle is well known to allow its therapeutic use, but difficulties have been encountered due in part to the heterogeneity and impure nature of preparations from natural sources. This heterogeneity is due to changes in the glycosylation pattern.
FSH is a valuable tool in both in vitro fertilization and in vivo fertilization stimulation, but as mentioned above, its clinical efficacy is hampered by inconsistencies in protein glycosylation. It is therefore evident that a method of reconstituting FSH would have great benefit to the reproductive sciences.
The reconstituted FSH peptide may be administered to a patient selected from the group consisting of a patient undergoing intrauterine insemination (IUI), a patient undergoing In Vitro Fertilization (IVF) and a sterile patient. The reconstituted FSH peptide may also be administered to induce or increase ovulation in a patient; stimulating the development of follicles in the patient; inducing follicle growth for gametogenesis in the patient; stimulating, inducing or increasing follicular development and subsequent ovulation in a patient or treating infertility in a patient. Preferably, the patient is a human female patient. The reconstituted FSH peptide may also be administered to a patient suffering from pituitary defects or a patient in puberty. Preferably, the patient is a human male patient.
FSH has been cloned and sequenced, and its nucleic and amino acid sequences are herein represented by seq id no: 11. SEQ ID NO: 12(α subunit) and the amino acid sequences set forth in SEQ ID NOs: 13 and SEQ ID NO: 14(β subunit) was proposed (FIGS. 63A, 63B, 63C and 63D, respectively). The skilled person will readily appreciate that the present invention is not limited to the sequences described herein, as variants of FSH are well known in the art. As a non-limiting example, U.S. patent No.5,639,640 describes a β subunit comprising two different amino acid sequences, while U.S. patent No.5,338,835 describes a β subunit comprising an additional amino acid sequence derived from the β subunit of human chorionic gonadotropin and being about 27 amino acids in length. Thus, the present invention encompasses natural and human engineered FSH variants, all of which are well known in the art.
Methods of expressing FSH in both prokaryotic and eukaryotic cells are well known in the art and are largely described in the literature (U.S. patent nos. 4,840,896, 4,923,805, 5,156,957). Further, methods of estimating the biological activity of the reconstituted FSH molecules of the present invention are well known in the art and are described, for example, in U.S. patent No.4,589,402, which describes methods of determining the effect of FSH on fertility, egg production, and pregnancy rate in non-human primate and human subjects.
F.EPO
The invention further comprises methods of reconstituting and/or modifying EPO. EPO is an acid glycoprotein of about 34kDa and can exist in 3 natural forms: alpha, beta and desialylated. The α and β forms differ only slightly in sugar composition, but have the same ability, biological activity and molecular weight. The desialylated form is an alpha or beta form with terminal sialic acid removed. EPO is present in plasma at very low concentrations in the healthy state, where the tissue receives sufficient oxidation from the existing number of erythrocytes. The normal concentration is sufficient to stimulate replacement of red blood cells lost through normal senescence. The amount of erythropoietin in the circulation is increased under hypoxic conditions when the oxygen transported in the circulation by the blood cells is reduced. Hypoxia can be caused by massive blood loss through hemorrhage, destruction of red blood cells due to excessive exposure to radiation, reduction in oxygen intake due to high altitude or prolonged unconsciousness, or various forms of anemia. EPO is therefore a useful compound for the supplementation of red blood cells following radiation therapy, anemia and other life threatening conditions.
The reconstituted EPO peptide can be administered to a patient selected from a patient having anemia, an anemia patient having chronic renal insufficiency, an anemia patient having end-stage renal disease, an anemia patient undergoing dialysis, an anemia patient having chronic renal failure, an HIV-infected patient treated with anemic zidovudine, an anemia patient having non-myeloid cancer and undergoing chemotherapy, and an anemia patient scheduled for non-cardiac non-vascular surgery. The reconstituted EPO peptide can also be administered to a patient undergoing surgery to reduce the need for allogeneic blood transfusion. Reconstituted EPO peptides can also be administered to patients who are expected to have significant blood loss with increased risk of transfusion during surgery. Preferably, the patient is a human patient.
In light of the importance of EPO in assisting recovery from various diseases and conditions, the present invention can be used to produce EPO having a natural and thus more potent sugar component. EPO synthesized to date lacks complete glycosylation and therefore must be administered more frequently and at higher doses due to its short life in vivo. The present invention also provides for the production of pegylated EPO molecules having a greatly improved half-life compared to EPO molecules that can be obtained by maximizing the desired glycoform.
EPO has been cloned and sequenced and its nucleotide and amino acid sequences are herein shown as SEQ ID NOs: 15 and SEQ ID NO: 16 (fig. 64A and 64B, respectively). Those skilled in the art will readily appreciate that the sequences presented herein are merely examples of sequences that encode and contain EPO. By way of example, U.S. patent No.6,187,564 describes fusion proteins comprising the amino acid sequence of two or more EPO peptides, and U.S. patent nos. 6,048,971 and 5,614,184 describe mutant EPO molecules having amino acid substitutions at positions 101, 103, 104 and 108. U.S. patent No.5,106,954 describes truncated EPO molecules, and U.S. patent No.5,888,772 describes EPO analogs with substitutions at positions 33, 139, and 166. Thus, the skilled person will recognize that the present invention encompasses EPO and EPO derivatives and variants, which are well documented in the literature and in the art as a whole.
In addition, methods for expressing EPO in cells are well known in the art. EPO can be expressed in prokaryotic and eukaryotic expression systems, as exemplified in U.S. patent nos. 4,703,008, 5,688,679, 6,376,218, and the like. Methods for determining the biological activity of EPO are also well known in the art. Making By way of example, the Krystal assay (Krystal, 1983, exp. Hematol.11: 649-. Briefly, the assay measures the effect of erythropoietin on intact mouse spleen cells. Mice were treated with phenylhydrazine to stimulate the production of erythropoietin-responsive red blood cell progenitors. After treatment, the spleen is harvested, intact spleen cells are isolated and incubated with various amounts of wild-type erythropoietin or erythropoietin protein as described herein. After overnight incubation, add3H-thymine and its integration into the cellular DNA was measured.3The amount of H-thymidine incorporation is indicative of the production of erythropoietin-stimulated red blood cells by interaction of erythropoietin with its cellular receptor. The concentration of the erythropoietin protein of the invention as well as the concentration of wild-type erythropoietin is quantified by competitive radioimmunoassay methods well known in the art. The specific activity was calculated as the international units measured in the Krystal assay divided by the microgram of immunoprecipitated protein measured by radioimmunoassay.
Several different mutant EPO's with different glycosylation patterns have been reported. Many have been stimulated with improved reticulocytosis activity without affecting the half-life of the peptide in the animal's bloodstream. It is contemplated that the mutated EPO peptides can be used in place of the native EPO peptides in any of the glycan remodeling, glycopegylation, and/or glycoconjugation embodiments described herein. Preferred EPO mutations are listed in, but not limited to, the following Table (see, e.g., Chern et al, 1991, Eur. J. biochem.202: 225-229; Grodberg et al, 1993, Eur. J. biochem.218: 597-601; Burns et al, 2002, Blood 99: 4400-4405; U.S. Pat. No.5,614,184; GenBank Accession No. AAN76993; O' Connell et al, 1992, J. biol. chem.267: 25010-25018; Elliott et al, 1984, Proc. Natl. Acad. Sci.U.S.A.81: 2708-2712; Biossel et al, 1993, J. biol. chem.268: 83-15993). The most preferred EPO mutation is Arg139To Ala139,Arg143To Ala143And Lys154To Ala154. The preferred native EPO form for making these mutations is the 165aa form, which is depicted in fig. 65; however, other natural forms of EPO may also be used. Finally, the mutations described in table 10 can be combined with each other or with other mutations to prepare EPO peptides for use in the present invention.
TABLE 10 EPO mutations
G.GM-CSF
The invention encompasses methods of modifying GM-CSF. GM-CSF is a cytokine well known in the art that is produced by activated T-cells, macrophages, endothelial cells, and stromal fibroblasts. GM-CSF acts primarily in the bone marrow to increase the production of inflammatory leukocytes and further functions as an endocrine hormone to initiate the recruitment of neutrophils that are consumed during inflammatory functions. GM-CSF is further a macrophage stimulating factor and promotes differentiation of Lagerhans cells into dendritic cells. Similar to G-CSF, GM-CSF also has clinical applications in bone marrow replacement following chemotherapy.
Although G-CSF itself has proven to be an important and useful compound for therapeutic applications, existing methods of producing G-CSF from recombinant cells result in products with a relatively short biological life span, incorrect glycosylation patterns that could potentially lead to immunogenicity, loss of function and the need for larger and more frequent administrations to achieve the same effect, etc.
The reconstituted GM-CSF peptide may be administered to a patient selected from patients with Acute Myeloid Leukemia (AML) or acute non-lymphocytic leukemia (ANLL), patients undergoing leukapheresis to collect hematopoietic progenitor cells from peripheral blood, patients undergoing autologous peripheral blood progenitor cell transplantation, non-hodgkin's lymphoma (NHL) patients undergoing autologous bone marrow transplantation, hodgkin's patients undergoing autologous bone marrow transplantation, and Acute Lymphoblastic Leukemia (ALL) patients undergoing autologous bone marrow transplantation. Reconstituted GM-CSF peptides may also be administered to patients to speed bone marrow engraftment, to shorten the time for neutrophil recovery following chemotherapy, to mobilize hematopoietic progenitor cells into peripheral blood for collection by leukapheresis, or to facilitate bone marrow reconstitution following autologous or allogeneic Bone Marrow Transplantation (BMT). Reconstituted GM-CSF peptide may also be administered to patients who have failed a bone marrow transplant or who have delayed bone marrow engraftment. Preferably, the patient is a human patient.
GM-CSF has been isolated and cloned and its nucleic and amino acid sequences are set forth in SEQ ID NO: 17 and SEQ ID NO: 18 (fig. 66A and 66B, respectively). The present invention encompasses methods of modifying GM-CSF, particularly when it concerns the ability of GM-CSF to function as a potent and functional biological molecule. While utilizing the disclosure of the present invention and the description herein, the skilled artisan will readily appreciate that the present invention provides compositions and methods for modifying GM-CSF.
The invention further comprises GM-CSF variants, as are well known in the art. By way of example, but in no way meant to limit the invention, variants of GM-CSF have been described in WO 86/06358 in which the protein is modified to an alternative quaternary structure. Further, U.S. Pat. No.6,287,557 describes a GM-CSF nucleic acid sequence linked to the genome of herpes virus for gene therapy applications. Furthermore, European patent publication No.0288809 (corresponding to PCT patent publication No. WO 87/02060) reports a fusion protein comprising IL-2 and GM-CSF. The IL-2 sequence may be at the N-or C-terminus of GM-CSF, such that upon acidic cleavage of the fusion protein, GM-CSF may be generated with modifications of the N-or C-terminal sequence. Accordingly, derivatives, mutants and variants of GM-CSF are well known in the art and are encompassed within the methods of the invention.
Expression and activity of the modified GM-CSF molecules of the invention can be determined using methods well known in the art and described, for example, in U.S. Pat. No.4,810,643. By way of example, activity may be measured using a radiolabeled thymidine uptake assay. In short, from a healthy supplyHuman bone marrow of the body was density-separated using Ficoll-Hypaque (1.077g/ml, Pharmacia, Piscataway, NJ) and the low density cells were suspended in Iscove's medium (GIBCO, La Jolla, Calif.) containing 10% fetal bovine serum, glutamine and antibiotics. To about 2X 104Human bone marrow cells of (A) with control Medium or G-CSF of the invention on 96-well flat-bottom plates in air 5% CO2Incubate at about 37 ℃ for about 2 days. Then with 0.5. mu. Ci/well3Cultures were pulsed for approximately 4 hours with H-thymine (New England Nuclear, Boston, Mass.) and uptake was measured as described in Ventua et al (1983, Blood 61: 781). Compared to bone marrow cells treated with control Compounds3An increase in the integration of H-thymidine into human bone marrow cells is indicative of an active and viable GM-CSF compound.
H.IFN-γ
One object of the present invention is a method comprising the modification and/or reconstitution of IFN- γ. IFN-gamma, also known as type II interferon, is a homodimeric glycoprotein comprising two subunits of about 21-24kDa, in contrast to IFN alpha and IFN beta. This size variation is due to a varying glycosylation pattern that is not generally reproducible when recombinantly produced in various expression systems known in the art. IFN- γ is a potent activator of macrophages, increases the expression of MHC class I molecules, and to a lesser extent is a stimulator of MHC class II molecules. Further, IFN- γ promotes differentiation of T-cells and isotype switching in B-cells. It is also well documented that IFN- γ is a stimulator of neutrophil, NK cell, and antibody responses leading to macrophage-mediated clearance. IFN- γ has been proposed as a therapeutic drug for use in infections with intracellular pathogens, such as tuberculosis and leishmaniasis, or as an antiproliferative therapeutic for conditions characterized by abnormal cell proliferation, such as various cancers and other neoplasias.
IFN- γ has proven to have potent immunological activity, but its therapeutic capacity, efficacy, biological half-life and other important factors are variable due to differences in glycosylation from the systems currently used to express IFN- γ. The present invention includes a method of correcting this critical defect.
The reconstituted IFN- γ peptide may be administered to a patient selected from the group consisting of a patient with chronic granulomatous disease, a patient with malignant osteopetrosis, a patient with pulmonary fibrosis, a patient with tuberculosis, a patient with cryptococcal meningitis, and a patient with a Mycobacterium Avium Complex (MAC) infection. Preferably, the patient is a human patient.
The nucleotide and amino acid sequences of IFN- γ are represented herein as SEQ ID NO: 19 and SEQ ID NO: 20 (fig. 67A and 67B, respectively). It is to be readily understood that the sequences presented herein are in no way limiting of the invention. In contrast, variants, derivatives and mutants of IFN- γ are well known to the skilled person. By way of example, U.S. Pat. No.6,083,724 describes recombinant avian IFN- γ, while U.S. Pat. No.5,770,191 describes C-terminal variants of human IFN- γ. In addition, U.S. Pat. No.4,758,656 describes novel IFN- γ derivatives and methods for synthesizing the same in various expression systems. The invention is thus not limited to IFN- γ sequences disclosed elsewhere herein, but encompasses all derivatives, variants, mutants, etc. well known in the art.
Expression systems for IFN- γ are also well known in the art and include prokaryotic and eukaryotic systems as well as plant and insect preparations, methods of which are well known to the skilled artisan. By way of example, U.S. Pat. No.4,758,656 describes a system for expressing IFN-. gamma.derivatives in E.coli, while U.S. Pat. No.4,889,803 describes an expression system using Chinese hamster ovary cells and the SV40 promoter.
Assays for the biological activity of reconstituted IFN- γ prepared according to the methods disclosed herein are well known to those skilled in the art. Biological assays for IFN- γ expression may be found, for example, in U.S. Pat. No.5,807,744. Briefly, IFN- γ was added to CD34++CD38-Cell (100 cells per well) cultures stimulated with a combination of cytokines to induce CD34++CD38-Proliferation of cells in which the cytokines are e.g.IL-3, c-kit ligand and IL-1, IL-6 or G-CSF. Cell proliferation and the production of second colony forming cells will be greatly inhibited in a dose dependent manner and near complete inhibition at 5000U/ml IFN- γ. As a confirmatory test for IFN- γ inhibition, an IFN- γ antibody may be added as a control.
I. Alpha-protease inhibitors (alpha-antitrypsin)
The invention further includes methods of reconstituting alpha-protease inhibitors (A-1-PI, alpha-1-antitrypsin or alpha-1-trypsin inhibitors), also known as alpha-antitrypsin. A-1-PI is a glycoprotein with a molecular weight of 53 kDa. A-1-PI plays a role in controlling tissue destruction by endogenous serine proteases and is the most significant serine protease inhibitor in plasma. In particular, A-1-PI inhibits various elastase enzymes, including neutrophil elastase. Elastase is a tissue-degrading protease and can be particularly problematic when its activity is not regulated in lung tissue. The protease functions by degrading foreign proteins. However, when the API is not present in an amount sufficient to modulate elastase activity, elastase can degrade lung tissue. Thus, this imbalance can lead to chronic lung tissue damage and emphysema. In fact, genetic defects in A-1-PI have been shown to be associated with the premature development of emphysema. A-1-PI supplementation has been successfully used in the treatment of this type of emphysema. Further, defects in A-1-PI will also lead to the exacerbation of other diseases, such as cystic fibrosis and arthritis, where leukocytes migrate into the lungs or joints to combat infection.
Thus, a-1-PI is convincingly useful in the treatment of diseases in which an imbalance between inhibitors and proteases, especially neutrophil elastase, is responsible for the development of the disease condition. A-1-PI also has antiviral activity. From these, it can be logically concluded that the present invention can be used to generate A-1-PI, which is safe, effective and powerful in the ever changing environment of the lung.
The reconstituted A-1-PI peptide may be administered to a patient selected from the group consisting of patients with congenital alpha-1-antitrypsin deficiency and emphysema, patients with cystic fibrosis, and patients with pulmonary fibrosis. Preferably, the patient is a human patient.
A-1-PI has been cloned and sequenced and is shown in SEQ ID NO: 21 and SEQ ID NO: 22 (fig. 68A and 68B, respectively). As understood by those skilled in the art, natural and processed variants of A-1-PI exist and are encompassed by the present invention. By way of example, U.S. Pat. No.5,723,316 describes A-1-PI derivatives having an amino acid substitution at position 356-361 and further comprising an N-terminal extension of about 3 amino acids. U.S. Pat. No.5,674,708 describes A-1-PI analogs having an amino acid substitution at position 358 of the primary amino acid sequence. The skilled artisan will readily recognize that the invention encompasses known and yet to be discovered variants, derivatives and mutants of A-1-PI.
Methods for determining the expression and activity of the reconstituted A-1-PI produced according to the methods of the invention are well known in the art and are described, for example, in U.S. Pat. No.5,674,708 and U.S. Pat. No.5,723,316. Briefly, biological activity can be determined in an assay for chymotrypsin activity by measuring inhibition of chymotrypsin-catalyzed hydrolysis of the substrate N-suc-Ala-Ala-Pro-Phe-p-nitroanilide (0.1ml of a 10mM solution in 90% DMSO), as described in DelMar et al (1979, anal. biochem. 99: 316). A typical 1.0 ml chymotrypsin assay comprises: 100mM Tris-Cl buffer, pH8.3, 0.005% (v/v) Triton X-100, bovine chymotrypsin (18kmmol) and A-1-PI of the invention. The assay mixture was preincubated at room temperature for about 5 minutes, substrate (0.01ml of a 10mM solution in 90% DMSO) was added, and the remaining chymotrypsin activity was determined by the range of change in absorbance at 410nm resulting from p-nitroanilide release. The measurement of light absorption was carried out at 25 ℃ using a spectrophotometer equipped with a temperature-controlled sample cell.
J. Glucocerebrosidase
The invention described herein further includes methods of modifying glucocerebrosidase. Glucocerebrosidase is a lysosomal glycosidase enzyme that catalyzes the hydrolysis of the glycolipid glucocerebroside to glucose and ceramides. The variant of glucocerebrosidase is Cerezyme TMAnd CeredaseTMAre commercially available and are approved therapeutics for the treatment of gaucher disease. CeredaseTMIs a placenta-derived form of glucocerebrosidase having a fully N-linked structure. CerezymeTMIs a recombinant variant of glucocerebrosidase 497 amino acids in length and expressed in CHO cells. The 4N-linked glycans of Cerezyme have been modified to terminate in a trimannose core.
Glucocerebrosidase is currently produced in recombinant mammalian cell cultures and therefore reflects the glycosylation pattern of those cells, usually rodent cells such as chinese hamster ovary cells or baby hamster kidney cells, which is vastly different from human glycosylation patterns, leading to loss of immunogenicity and potency, among other things.
The reconstituted glucocerebrosidase peptide can be administered to a patient selected from the group consisting of patients with lysosomal storage diseases, patients with glucocerebrosidase deficiency, and patients with gaucher's disease. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of glucocerebrosidase are referred to herein as SEQ ID NO: 23 and 24 (fig. 69A and 69B, respectively). However, as will be appreciated by the skilled artisan, the sequences represented herein are prototype sequences and do not limit the invention. In fact, variants of glucocerebrosidase are well known and are described, for example, in U.S. Pat. No.6,015,703, which describes enhanced production of glucocerebrosidase analogs and variants thereof. Further, U.S. Pat. No.6,087,131 describes the cloning and sequencing of another variant of glucocerebrosidase. The present invention is intended to encompass these and other derivatives, variants and mutants that are known or that may be discovered in the future.
Methods for expressing glucocerebrosidase using standard techniques are well known in the art and are described in detail, for example, in U.S. Pat. No.6,015,703. Assays for the biological efficacy of glucocerebrosidase molecules prepared according to the methods of the invention are also well known in the art, and mouse models of gaucher disease for the estimation and use of glucocerebrosidase therapeutics are described, for example, in Marshall et al (2002, mol. ther. 6: 179).
K.TPA
The invention further comprises a method of reconstituting a tissue-type activating factor (TPA). TPA activates plasminogen to form plasmin, which dissolves fibrin, which is a major component of the substrate for thrombocytes. TPA preparations have been developed as thrombolytic agents in thrombolytic therapy, which have high selectivity for thrombus formation leading to myocardial infarction and cerebral infarction.
Further, various modified TPAs have been produced by genetic engineering for the purpose of obtaining higher affinity for fibrin and longer blood half-life than native TPA. TPA is a protein that is generally very poorly soluble in water. In particular, modified TPA is more difficult to dissolve in water than natural TPA, thus making the production of modified TPA very difficult. Thus modified TPA is difficult to dissolve in water when administered to a patient. However, modified TPA has various advantages, such as increased affinity for fibrin and longer half-life in blood. It is an object of the present invention to increase the solubility of modified TPA.
The reconstituted TPA peptide can be administered to a patient selected from the group consisting of patients with acute myocardial infarction and patients with acute ischemic attacks. Reconstituted TPA peptide can also be administered to a patient to improve ventricular function following acute myocardial infarction, reduce the incidence of congestive heart failure following acute myocardial infarction, or reduce mortality associated with acute myocardial infarction. Reconstituted TPA peptide can also be administered to patients to improve neurological recovery after acute ischemic attack or to reduce the incidence of disability and paralysis after acute ischemic attack. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of TPA are set forth herein as SEQ ID NOs: 25 and SEQ ID NO: 26 (fig. 70A and 70B, respectively). As described above, TPA variants have been constructed and used in therapeutic applications. For example, U.S. patent No.5,770,425 describes TPA variants in which some or all of the fibrin domains are deleted. Further, U.S. patent No.5,736,134 describes TPA modified with an amino acid at position 276. The inclusion of the TPA sequences set forth herein, as well as those variants well known to those skilled in the art, will be readily apparent to the skilled artisan using the disclosure of the present invention and the description herein.
The expression of TPA from TPA-encoding nucleic acid sequences is well known and is described in detail, for example, in U.S. patent No.5,753,486. Assays for determining the biological properties of TPA molecules produced according to the methods of the invention are well known in the art. Briefly, TPA molecules synthesized by methods disclosed elsewhere herein can be assayed for their ability to cleave fibrin in the presence of saturating concentrations of plasminogen according to the method of Carlsen et al (1988, anal. biochem.168: 428). In vitro clot lysis assay the activity of the tissue-type activating factor was measured by turbidity assay with a microcentrifuge analyzer. The mixture of thrombin and TPA was centrifuged into the mixture of fibrinogen and plasminogen to initiate clot formation and subsequent clot lysis. The resulting absorbance-time relationship is analyzed to determine the assay endpoint. The activity of the TPA variants was compared to a standard curve of TPA. The buffer used during the assay was 0.06M sodium phosphate, pH7.4, containing 0.01% (v/v) TWEEN80 and 0.01% (w/v) sodium azide. The human thrombin concentration was about 33 units/ml. Fibrinogen (2.0mg/ml clumpable protein) was frozen on wet ice to precipitate fibronectin, which was then gravity filtered. Glu-plasminogen concentration was 1 mg/ml. The analyzer compartment temperature was set at 37 ℃. The charger was set to dispense 20 microliters of TPA (about 500 nanograms/milliliter to about 1.5 micrograms/milliliter) as a sample of the standard curve, or a variant of 20 microliters of TPA at a concentration that would result in cleavage within the range of the standard curve. 20 microliters of thrombin was used as a secondary reagent, and 200 microliters of 50:1(v/v) fibrinogen: the plasminogen mixture serves as a primary reagent. The absorption/time program applies an incubation time of 5 minutes, a 340-nm-filter and readings at 90 second intervals.
L.IL-2
The invention further comprises methods of reconstructing and modifying IL-2. IL-2 is a major growth factor for T lymphocytes and can increase both humoral and cellular immune responses by stimulating cytotoxic CD8T cells and NK cells. IL-2 is therefore critical in the defense mechanism against tumors and viral infections. IL-2 is also used in the treatment of anti-metastatic melanoma and renal adenocarcinoma, and has been used in clinical trials for many forms of cancer. Further, IL-2 has also been used in HIV infected patients, where it results in a significant increase in the number of CD 4.
Due to the success of IL-2 in the management and treatment of life-threatening diseases such as various cancers and AIDS, the methods of the present invention are useful for developing IL-2 molecules having longer biological half-lives, increased capacity, and therapeutic properties generally more similar to those of wild-type IL-2 that is synthetically secreted in healthy humans.
The reconstituted IL-2 peptide may be administered to a patient selected from the group consisting of patients with metastatic renal cell carcinoma, patients with metastatic melanoma, patients with ovarian cancer, patients with Acute Myeloid Leukemia (AML), patients with non-hodgkin's lymphoma (NHL), patients infected with HIV, and patients infected with hepatitis c. The reconstituted IL-2 peptide may also be administered to a patient as a cancer vaccine adjuvant. Preferably, the patient is a human patient.
IL-2 has been cloned and sequenced, and the nucleic acid and amino acid sequences are herein identified as SEQ ID NO: 27 and SEQ ID NO: 28 (fig. 71A and 71B, respectively). The present invention should in no way be construed as being limited to the IL-2 nucleic acid and amino acid sequences set forth herein. Variants of IL-2 are described, for example, in U.S. Pat. No.6,348,193, in which the asparagine at position 88 is replaced by arginine, and in U.S. Pat. No.5,206,344, in which multimers comprising IL-2 variants with various amino acid substitutions are described. The present invention encompasses these and other IL-2 variants well known in the art.
Methods for determining the expression and activity of IL-2 are well known in the art and are described, for example, in U.S. Pat. No.5,417,970. Briefly, expression of IL-2 or variants thereof can be achieved in a variety of prokaryotic and eukaryotic systems, including E.coli, CHO cells, BHK cells, insect cells employing baculovirus expression systems, all of which are well known in the art.
The determination of the activity of the modified IL-2 prepared according to the method of the invention can be carried out as follows. Peripheral blood lymphocytes are separated from erythrocytes and granulocytes by centrifugation on a Ficoll-Hypaque (Pharmacia, Piscataway, NJ) gradient by the method described, for example, in A.Boyum et al (Methods in Enzymology, 1984, Vol.108, p.88, Academic Press, Inc.). The lymphocytes were subsequently washed about 3 times in a medium consisting of RPMI 1640(Gibco-BRL, La Jolla, Calif.) and heat (1 hour at 56 ℃) inactivated 10% AB human serum (CTS Purpan, Toulose, France), 2mM sodium pyruvate, 5mM HEPES, 4mM L-glutamine, 100U/ml penicillin, 100. mu.g/ml streptomycin and 0.25. mu.g/ml amphotericin (complete medium). Removing adherent cells (monocytes and macrophages) by attachment to plastic, and removing the remaining cells at about 5-10X 10 per ml 5The concentration of cells is resuspended in complete medium and about 1-2X 10 per square centimeter5The density of cells was seeded in culture flasks. The bottles were then placed in 5% CO2Incubated at 37 ℃ for about 1 hour, after which non-adherent lymphocytes were recovered by aspiration after gently agitating the flask.
Non-adherent lymphocytes were washed once and at about 10 per ml5The concentration of cells in the presence of IL-2 of the invention was cultured in complete medium in an incubator as described above for about 48 hours. The cells were then washed once.
The cytotoxic activity of the cells is in the order of the human T-lymphoid cell line C8166-45/C63(HT1 cells) against NK cell cytotoxicityThe target cells were estimated after about 4 hours of contact, as described by Salahuddin et al (1983, Virology 129: 51-64; 1984, Science 223: 703-707). Make 6X 105HT1 cells were cultured at 37 ℃ in complete serum-free medium with a concentration of about 200. mu. Ci51Cr (sodium chromate, Amersham, Arlington Heights, IL) was radiolabeled for about 1 hour, and then washed several times. Target and valid cells were dispensed on round-bottom microtiter plates at different ratios of valid to target cells (50:1, 10:1, 1: 1). The microtiter plates were centrifuged and after incubation as described above, the supernatant was recovered from each well and radioactivity was measured with a gamma counter. The cytotoxicity being released from dead target cells 51The amount of Cr is determined. Non-specific cytotoxicity is determined from the amount of radioactivity that is spontaneously released from the target cells in the absence of effective cells.
The method of the present invention is only one of many methods for measuring cytotoxicity of effector cells that are well known in the art, and should not be construed as limiting the present invention.
M. blood coagulation factor VIII
The invention further comprises methods of reconstituting and modifying factor VIII. As described above for factor VII and factor IX, factor VIII is a key component in the blood coagulation pathway. Human coagulation factor VIII (antihemophilic factor; FVIII: C) is a human plasma protein consisting of two peptides (a light chain with a molecular weight of 80kDa and a heavy chain with a molecular weight of 90-220 kDa depending on the glycosylation state). It is an essential cofactor in the coagulation pathway and is required for the conversion of factor X to its active form (factor Xa). Coagulation factor VIII circulates in plasma as a non-covalent complex (akaVIII: RP) with Von willebrand (Von willebrand) factor, a 2050 aa peptide (see us patent No.6,307,032). Blood concentrations of factor VIII that are 20% lower than normal result in a bleeding disorder known as hemophilia a. Blood levels of factor VIII below 1% result in severe bleeding disorders, with spontaneous joint bleeding being the most common symptom.
Similar to other blood coagulation factors, coagulation factor VIII is a therapeutic agent with great potential for the treatment of various bleeding disorders, such as hemophilia a and hemophilia B. Existing methods for preparing factor VIII from recombinant cells result in products that are less efficient than native factor VIII due to glycosylation of the heavy chain. Purification processes from human plasma result in crude compositions that are less efficient and more difficult to prepare than recombinant coagulation factor VIII. The present invention seeks to improve this situation.
The reconstituted factor VIII peptide may be administered to a patient selected from the group consisting of patients with von willebrand disease, patients with haemophilia a, patients with factor VIII: patients with C deficiency, patients with fibrinogen deficiency, patients with factor XIII deficiency and patients with acquired factor VIII inhibitors (acquired hemophilia). The reconstituted factor VIII peptide may also be administered to a patient to prevent, treat or control bleeding or bleeding episodes. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of factor VIII are represented herein as SEQ ID NO: 29 and SEQ ID NO: 30 (fig. 72A and 72B, respectively). Various coagulation factor VIII are common in the art, as described in us patent No.5,668,108, in which the aspartic acid at position 1241 is replaced by glutamic acid, with the same change in nucleic acid. U.S. patent No.5,149,637 describes factor VIII variants comprising a glycosylated or non-glycosylated C-terminal fragment, and U.S. patent No.5,661,008 describes factor VIII variants comprising amino acids 1-740 linked to amino acids 1649-2332 by at least 3 amino acid residues. Thus, variants, derivatives, modifications and complexes of factor VIII are well known in the art and encompassed by the present invention.
Expression systems for producing factor VIII are well known in the art and include prokaryotic and eukaryotic cells, as exemplified in U.S. patent nos. 5,633,150, 5,804,420, and 5,422,250.
In order to determine the biological activity of factor VIII molecules synthesized according to the methods of the present invention, the skilled person will recognize that the assays described herein for the estimation of factor VII and factor IX may be applied to factor VIII.
Urokinase N
The invention also includes methods of reconstituting and/or modifying urokinase. Urokinase is a serine protease that activates plasminogen to plasmin. The protein is synthesized in various tissues, including endothelium and kidney, and is excreted in trace amounts into urine. Purified urokinase exists in two active forms, a high molecular weight form (HUK; about 50kDa) and a low molecular weight form (LUK; about 30 kDa). LUK has been shown to be derived from HUK by proteolysis after lysine 135, releasing the first 135 amino acids from HUK. It is believed that HUK or LUK must be converted to a proteolytically active form by proteolysis of a single-chain precursor called prourokinase between lysine 158 and isoleucine 159 to produce the two-chain active form (which continues to correspond to HUK or LUK). The proteolytically active urokinase species resulting from this proteolytic cleavage contains two amino acid chains linked together by a single disulfide bond. The two chains formed by active trimming are referred to as A or A 1Chain (HUK or LUK, respectively), and the B chain comprising the protease domain of the molecule.
Urokinase has been shown to be an effective thrombolytic agent. However, since it is naturally produced in trace amounts, the cost of effective doses of enzyme is high. Urokinase has been produced in recombinant cell culture and DNA encoding urokinase is known as well as appropriate vectors and host microorganisms. Existing compositions comprising urokinase and methods of recombinantly producing urokinase are hampered by products with defective glycosylation patterns, and this aberrant glycosylation results in inefficient and low-capacity products due to complex proteolytic cleavage events upon urokinase activation.
The reconstituted urokinase peptide can be administered to a patient selected from the group consisting of patients with embolism, patients with acute massive pulmonary embolism, and patients with coronary thrombosis. Preferably, the patient is a human patient. Reconstituted urokinase peptide may also be used to restore patency to intravenous catheters, including central venous catheters that are blocked by clotted blood or fibrin.
The nucleotide sequence encoding the primary amino acid chain of urokinase is depicted in SEQ ID NO: 33 and seq id NO: 34 (fig. 73A and 73B, respectively). Variants of urokinase are well known in the art and therefore the present invention is not limited to the sequences set forth herein. Indeed, the skilled artisan readily recognizes that the urokinase variants described in U.S. Pat. Nos. 5,219,569, 5,648,253, and 4,892,826 exist as functional parts and are therefore encompassed by the present invention.
The expression and evaluation of urokinase molecules prepared according to the methods of the present invention are also well known in the art. As a non-limiting example, the expression of urokinase in various systems is described in detail in U.S. Pat. No.5,219,569. Assays to determine the activity and functionality of urokinase prepared according to the methods provided herein are described in this document and are similar to other plasminogen and fibrin related assays described elsewhere. An assay for determining the activity of urokinase molecules synthesized by the methods described herein is described, for example, in Ploug et al (1957, Biochim. Biophys. acta 24: 278- > 282) using fibrin plates containing 1.25% agarose, 4.1mg/ml human plasminogen, 0.3 units/ml thrombin and 0.5. mu.g/ml soybean trypsin inhibitor.
Human DNase
The invention further comprises methods for reconstituting and/or modifying a recombinant human DNase. Human DNase I has been validated as a therapeutic agent and has been shown to reduce the viscosity of cystic fibrosis mucus in vitro. It has been determined that purulent mucus contains about 10-13mg/ml of DNA, an ionic polymer that affects the rheological properties of respiratory fluids. Thus, the enzyme bovine pancreatic DNase I, which degrades DNA, was validated as a mucolytic agent many years ago, but did not enter clinical practice because of side effects caused by antigenic and/or contaminating proteases. Recombinant human dnases are currently available as therapeutic agents to alleviate symptoms of diseases such as cystic fibrosis.
The reconstituted rDNase peptide can be administered to a patient suffering from cystic fibrosis. Reconstituted rDNase peptides can also be administered to cystic fibrosis patients to improve lung function. Preferably, the patient is a human patient.
Similar to dnases derived from bovine sources, recombinant human dnases also pose some problems, mostly due to reduced efficacy, due to inappropriate glycosylation caused by the mammalian expression systems currently used. The present invention describes methods of reconstituting dnases which may lead to increased efficacy and better therapeutic outcomes.
The nucleotide and amino acid sequences of human dnases are herein represented by SEQ ID NO: 39 and SEQ ID NO: 40 (fig. 74A and 74B, respectively). Peptide variants comprising dnases are well known in the art. As an example, us patent No.6,348,343 describes a human DNase with multiple amino acid substitutions in the overall primary structure. Furthermore, us patent No.6,391,607 describes hyperactive DNase variants with multiple amino acid substitutions at positions 9, 14, 74, 75 and 205. This embodiment and others that are well known in the art or that may be discovered in the future are encompassed by the present invention.
Expression systems for the production of DNase peptides are well known to the skilled person and have been described in prokaryotic and eukaryotic systems. For example, PCT patent publication No. wo 90/07572 describes these processes in considerable detail.
Assays for determining the biological activity of DNase molecules developed according to the methods of the present invention are well known in the art. By way of example, but in no way meant to limit the invention, assays for determining the DNA-hydrolyzing activity of human DNase I are provided herein. Briefly, two different plasmid digestion assays were applied. The first assay ("supercoiled DNA digestion assay") measures the relaxation of supercoiled double-stranded plasmid DNA to relaxed (with a deletion)Oral), linear, and degraded forms. The second assay ("linear DNA digestion assay") measures the conversion of linear double-stranded plasmid DNA to a degraded form. Specifically, DNase prepared according to the method of the present invention was added to 160. mu.l of a solution containing 25. mu.g per ml of supercoiled plasmid DNA or EcoR I-digested linearized plasmid DNA in 25mM HEPES (pH7.1), 100. mu.g/ml bovine serum albumin, 1mM MgCl2、2.5mM CaCl2150mM NaCl, and the sample was incubated at room temperature. Aliquots of the reaction mixture were taken at various times and the reaction was stopped by the addition of 25mM EDTA and xylene nitrile blue, bromophenol blue and glycerol. The integrity of plasmid DNA in the terminated samples can be analyzed by running the samples on agarose gels. After electrophoresis, the gel was stained with ethyl bromide bonds, and the DNA in the gel was visualized by ultraviolet light. The relative amounts of supercoiled, relaxed and linear forms in plasmid DNA are determined by scanning the gel with a fluorescence imager (e.g., molecular dynamics Model 575 FluorImager) and quantifying the amount of DNA in bands on the gel corresponding to the different forms.
P. insulin
The invention further includes methods of reconstituting insulin. Insulin is well known as the most effective therapeutic agent for type I diabetes, in which the beta islet cells of the pancreas do not produce insulin for regulating blood glucose levels. The consequences of diabetes and uncontrolled blood glucose include circulatory and foot problems, and blindness, as well as various other complications resulting from or exacerbated by diabetes.
Porcine insulin was used as a therapeutic for diabetes prior to cloning and sequencing of human insulin. Insulin is currently produced recombinantly, but the short 51 amino acid sequence of the mature molecule is a complex structure containing multiple disulfide bonds. Existing methods for recombinant production of insulin result in products that lack similarity to the native protein produced in healthy non-insulin subjects. The present invention seeks to remedy this disadvantage.
The reconstituted insulin peptide can be administered to a patient selected from the group consisting of patients with type I diabetes (diabetes) and patients with type 2 diabetes who require basal (long-acting) insulin to control hyperglycemia. Reconstituted insulin peptides can also be administered to diabetic patients to control hyperglycemia. Preferably, the patient is a human patient.
The nucleotide and amino acid sequences of human insulin are set forth in SEQ ID NO: 43 and SEQ ID NO: 44 (fig. 75A and 75B, respectively). Insulin variants are abundant in the art. U.S. patent No.6,337,194 describes insulin fusion protein analogs, U.S. patent No.6,323,311 describes insulin derivatives comprising cyclic anhydrides of dicarboxylic acids, and U.S. patent No.6,251,856 describes insulin derivatives comprising multiple amino acid substitutions and lipophilic groups. The skilled person will appreciate that the following examples of insulin derivatives are by no means exclusive and represent only a small part of those well known in the art. Thus, the present invention encompasses known or to be discovered insulin derivatives.
Expression systems for the production of insulin are well known in the art and can be achieved using Molecular biology techniques, as described in Sambrook et al (1989, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, New York).
Assays for determining the functionality of insulin molecules prepared according to the methods of the invention are also well known in the art. For example, an in vitro model of glucose inhibition can be used to estimate the biological activity of insulin synthesized by the methods of the invention. A rat model may be used for this purpose. Animals are fasted overnight (16 hours) prior to the experiment and then anesthetized with intraperitoneal administration of sodium pentobarbital or other suitable anesthetic such as ketamine. Each animal received an intravenous injection (tail vein) of a specific insulin derivative (20. mu.g/ml/kg). Blood samples were taken from the jugular vein 15 and 5 minutes before injection and 15, 30, 60, 90, 120, 180 and 240 minutes after injection. Blood glucose levels were measured using blood glucose monitors, which were available from various commercial suppliers.
Hepatitis B vaccine (HBsAg)
The invention further comprises a method of reconstituting an antigen (HBsAg or hepatitis B surface antigen) for a hepatitis B vaccine. HBsAg is a surface antigen of the recombinantly produced hepatitis B S-protein and is used to elicit an immune response to the hepatitis B virus, an increasingly dangerous virus that can cause liver diseases such as cirrhosis and cancer, etc., and death of over 1 million annually worldwide. The HBsAg vaccine is currently administered 3 times at 6 month intervals to elicit protective and neutralizing immune responses.
HBsAg is currently produced in yeast strains and thus reflects the natural glycosylation pattern of fungi. The present invention provides methods for reconstituting hbsags that also result in improved immunogenicity, antibodies with improved affinity for the virus, and the like.
The reconstituted HBsAg peptide can be administered to a patient to immunize the patient against disease caused by hepatitis B virus. Reconstituted HBsAg peptides can also be administered to a pre-dialysis (predialysis) or dialysis patient for immunization of the patient against disease caused by hepatitis B virus. Preferably, the patient is a human patient.
The sequence of the nucleic acid and primary amino acid chain from the hepatitis b virus S-protein (HBsAg) is identified herein with SEQ ID NO: 45 and SEQ ID NO: 46 (fig. 76A and 76B, respectively). The nucleotide length is 1203 bases. Amino acids are 400 residues long. The last 226 amino acid residues are small S-antigens that can be used in GlaxoSmithKline vaccines and Merck vaccines. The 55 amino acids upstream of the small S-antigen are the Pre-S start codon. The Pre-S plus S region is the S-antigen, which is useful in the Aventis Pasteur vaccine. The remainder of the S-protein is contained from the first codon to the start codon of Pre-S and is called the large S-protein. This is only one example of an HBsAg for use in a vaccine and other subtypes are well known, such as those described by GenBank Acc No.: AF415222, AF415221, AF415220 and AF 415219. The sequences presented herein are only examples of hbsags known in the art. Similar antigens have been isolated from other strains of hepatitis b virus and have or have not been estimated for their antigenicity and potential as vaccine candidates. The present invention thus encompasses hepatitis B virus vaccine S-protein surface antigens that are known or to be discovered.
Expression of HBsAg in an expression system is a routine procedure for those skilled in the art and is described, for example, in U.S. Pat. No.5,851,823. Assays for vaccine immunogenicity are well known in the art and include various assays for the production of neutralizing antibodies and employ techniques such as ELISA, neutralization assays, western blotting, immunoprecipitation, and the like. Briefly, a sandwich ELISA for detecting potent anti-HBsAg antibodies is described. The Enzygnost HBsAg assay (Aventis Behring, King of Prussia, Pa.) can be used in this method. The wells were covered with anti-HB. Serum plasma or purified protein and appropriate controls were added to the wells and incubated. After washing, peroxidase-labeled antibodies against HBsAg were reacted with the remaining antigenic determinants. Unbound enzyme-linked antibody is removed by washing and the enzyme activity on the solid phase is determined by methods well known in the art. The enzyme-catalyzed reaction of hydrogen peroxide and chromogen is terminated by addition of dilute sulfuric acid. The color intensity is proportional to the concentration of HBsAg in the sample and is obtained by photometric comparison of the color intensity of the unknown sample with the color intensities of the accompanying negative and positive control sera.
R. human growth hormone
The invention further comprises a method for reconstituting Human Growth Hormone (HGH). The HGH isoform secreted in the human pituitary gland consists of 191 amino acids and has a molecular weight of about 21,500. The HGH isoforms prepared in the placenta are glycosylated forms. HGH is involved in many regulators of normal human growth and development, including linear growth (somatogenesis), lactation, macrophage activation, and insulin-like and diabetogenic effects, among others.
HGH is a complex hormone, and its action varies as a result of interactions with various cellular receptors. Although compositions comprising HGH have been used in clinical settings, particularly in the treatment of dwarfism, their efficacy is limited by the absence of glycosylation in recombinantly produced HGH.
The reconstituted HGH peptide may be administered to a patient selected from the group consisting of a patient suffering from growth hormone deficiency, a patient suffering from turner's syndrome, a patient suffering from growth deficiency due to lack of sufficient endogenous growth hormone secretion, a patient suffering from growth deficiency due to pleckstrin-weirs syndrome (PWS), a patient suffering from growth deficiency associated with chronic renal insufficiency, and a patient suffering from AIDS-related wasting or cachexia. Reconstituted HGH peptides may also be administered to patients of small stature. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of HGH are herein represented by SEQ ID NO: 47 and SEQ ID NO: 48 (fig. 77A and 77B, respectively). The skilled artisan will recognize that variants, derivatives and mutants of HGH are well known. Examples can be found in U.S. Pat. No.6,143,523, in which the amino acid residues at positions 10, 14, 18, 21, 167, 171, 174, 176 and 179 are substituted, and in U.S. Pat. No.5,962,411, which describes splice variants of HGH. The present invention encompasses these HGH variants and variants that will be found, which are well known in the art.
Methods for expressing HGH in recombinant cells are described, for example, in U.S. Pat. No.5,795,745. Methods for expressing HGH in prokaryotes, eukaryotes, insect cell systems, plants, and in vitro translation systems, among other factors, are well known in the art.
The HGH molecules produced by the methods of the present invention can be assayed for activity by a variety of methods well known to the skilled artisan. For example, U.S. patent No.5,734,024 describes a method of determining the biological function of the expressed HGH.
S. antithrombin III
Antithrombin (antithrombin III, AT-III) is a potent inhibitor of the coagulation cascade in the blood. It is a non-vitamin K-dependent protease that inhibits thrombin and other procoagulant factors (such as factor Xa). Deficient congenital antithrombin III is a condition of autosomal dominant nature in which an individual inherits one copy of a defective gene. This condition leads to an increased risk of venous and arterial thrombosis, the onset of clinical manifestations generally occurring in young adults. A severe congenital deficiency of antithrombin III in individuals inheriting two defective genes is an autosomal recessive condition associated with increased thrombosis, which is generally pronounced at early childhood. Insufficient acquired antithrombin III is most often seen in situations where the coagulation system is improperly activated. General conditions leading to a deficiency in acquired antithrombin III include disseminated intravascular coagulation, microangiopathic hemolytic anemia due to endothelial injury (i.e. hemolytic uremic syndrome), and Venous Occlusive Disease (VOD) seen in patients undergoing bone marrow transplantation. AT-III deficiency can be corrected for short term by infusion of AT-III concentrates.
The reconstituted AT-III peptide can be administered to a patient selected from a patient having a genetic AT-III deficiency associated with surgery or obstetrics and a genetic AT-III deficiency patient having thromboembolism. Preferably, the patient is a human patient.
Antithrombin III (AT-III) is an alpha 2-glycoprotein with a molecular weight of 58,000. It is expressed as Thrombate IIITMCommercially available (Bayer corp., West Haven, CT). The nucleic acid and amino acid sequences of human antithrombin III are shown in FIGS. 78A (SEQ ID NO: 63) and 78B (SEQ ID NO: 64), respectively.
Methods for preparing antithrombin III are well known to those skilled in the art. For example, published nucleic acid and amino acid sequences are available for human antithrombin III (see U.S. patent No.4,517,294) and mutants of human antithrombin III (see U.S. patent nos. 5,420,252, 5,618,713, 5,700,663). The method of the present invention may be applied to any of these amino acid sequences and any of their coding sequences, but is not limited to these sequences. Exemplary methods for producing recombinant antithrombin III are well known in the art, and several methods are described in U.S. patent nos. 5,420,252, 5,843,705, 6,441,145 and 5,994,628. Exemplary methods for purifying recombinant antithrombin III are described in U.S. patent nos. 5,989,593, 6,268,487, 6,395,888, 6,395,881, 6,451,978 and 6,518,406.
Recombinant antithrombin III has many well-known uses. Antithrombin III can be used as an anticoagulant in surgery (U.S. patent nos. 5,252,557, 5,182,259), as part of a pharmaceutical formulation or method of inhibiting thrombosis (U.S. patent nos. 5,565,471, 6,001,820), and to reduce the deleterious side effects of cell transplantation (U.S. patent No.6,387,366). In addition, antithrombin III formulations can be used to increase placental blood flow (U.S. patent No.5,888,964), inhibit fertilization (U.S. patent No.5,545,615), treat asthma (U.S. patent No.6,355,626), and treat arthritis (U.S. patent No.5,252,557) and other inflammatory conditions (U.S. patent No.6,399,572). Antithrombin III may also be used in the manufacture of replacement plasma (U.S. patent No.4,900,720) or in the preparation of stable cellular blood products for transfusion (U.S. patent No.6,139,878). Antithrombin III can be administered as a pharmaceutical formulation (U.S. patent nos. 5,084,273, 5,866,122, 6,399,572, 6,156,731 and 6,514,940) or using gene therapy methods (U.S. patent No.6,410,015). Compositions comprising antithrombin III may be used as tissue adhesives (U.S. patent No.6,500,427) or lubricants for medical devices introduced into patients (U.S. patent No.6,391,832). Antithrombin III is also commonly used to coat intravascular stents (U.S. patent nos. 6,355,055, 6,240,616, 5,985,307, 5,685,847, and 5,222,971), ocular implants (U.S. patent No.5,944,753), and prosthetic organs (U.S. patent nos. 6,503,556, 6,491,965, and 6,451,373). Antithrombin III may also be used in methods for locating an internal bleeding site in a patient (U.S. patent No.6,314,314) and for determining cessation of blood dysfunction in a patient (U.S. patent No.6,429,017).
Human chorionic gonadotropin
Human chorionic gonadotropin (hCG) is a glycoprotein composed of an alpha subunit and a beta subunit. HCG is closely related to the other two gonadotropins Luteinizing Hormone (LH) and Follicle Stimulating Hormone (FSH) and Thyroid Stimulating Hormone (TSH), all of which are glycoprotein hormones. The alpha subunits of these different glycoprotein hormones are very similar in structure, but the beta subunits differ in amino acid sequence.
The nucleic acid and amino acid sequences of the alpha subunit of human chorionic gonadotropin are shown in FIGS. 79A (SEQ ID NO: 69) and 79B (SEQ ID NO: 70), respectively. The nucleic acid and amino acid sequences of the beta subunit of human chorionic gonadotropin are shown in FIGS. 79C (SEQ ID NO: 71) and 79D (SEQ ID NO: 72), respectively.
Human chorionic gonadotropin is used in infertility treatment to promote ovulation or the release of ova from ovaries in women who are unable to ovulate themselves. Human chorionic gonadotropin is also administered to young males to treat uninhabited or hypoplastic testes. It is used in men to stimulate testosterone production. Some physicians also prescribe human chorionic gonadotropin for patients with erectile dysfunction with loss of libido and for the treatment of male "menopause".
The reconstituted hCG peptide may be administered to a patient selected from the group consisting of patients undergoing Assisted Reproductive Technologies (ART), patients undergoing In Vitro Fertilization (IVF), patients undergoing embryo transfer, patients with infertility, male patients with pre-pubertal cryptorchidism that is not due to anatomical obstruction, and male patients with hypogonadotropic hypogonadism. Reconstituted hCG peptide can also be administered to induce eventual follicular maturation in sterile female patients who have undergone pituitary desensitization and pretreatment with follicle stimulating hormone. Reconstituted hCG peptides can also be administered to anovulatory infertile patients to induce ovulation and pregnancy. Preferably, the patient is a human patient.
Methods for preparing human chorionic gonadotropin are well known to those skilled in the art. Heterodimeric hCG can be recombinantly produced in any of the many expression systems currently used for the industrial production of recombinant proteins. One method of preparing recombinant hCG is described in U.S. patent No.5,639,639. Methods for preparing recombinant heterodimeric proteins by expressing both subunits in the same cell are generally well known in the art, and several methods are described in U.S. Pat. No.5,643,745 (expressed in filamentous fungi), 5,985,611 and 6,087,129 (expressed in secretory cells). Alternatively, each subunit can be expressed separately in a cell, and the two subunits are later brought together in vitro to assemble a heterodimer.
Methods of applying human chorionic gonadotropin are numerous and well known in the art. Typically, hCG is used to induce or synchronize ovulation in mammals (see U.S. patent nos. 6,489,288, 5,589,457, 5,532,155, 4,196,123, 4,062,942 and 4,845,077). In addition, hCG can be used in pregnancy tests, particularly in agglutination based tests (see U.S. patent nos. 3,991,175, 4,003,988, 4,071,314 and 4,088,749). hCG is also used in contraceptive vaccines (see U.S. patent nos. 4,161,519 and 4,966,888). In addition, hCG can be used to treat conditions associated with aging and altered hormone balance such as benign prostatic hypertrophy (see us patent No.5,610,136) and central nervous system disorders commonly found in the elderly (see us patent No.4,791,099).
Alternatively, hCG can be used to detect and treat cancers that express hCG or one of its subunits. hCG-expressing tumors include, but are not limited to, breast, prostate, ovarian and stomach cancers, and neuroblastoma such as kaposi's sarcoma. Antibodies against hCG that have been glycoreconstructed to have a glycan structure similar to that found on tumor-expressing hCG can be generated and used to detect hCG-expressing tumors in patients according to methods well known in the art (see U.S. patent nos. 4,311,688, 4,478,815, and 4,323,546). In addition, reconstituted hCG can be used to elicit an immune response to hCG-expressing tumors (see U.S. patent nos. 5,677,275, 5,762,931, 5,877,148, 4,970,071, and 4,966,753).
hCG can also be used in methods of immunomodulation in animals in general, such as the methods described in U.S. patent nos. 5,554,595, 5,851,997 and 5,700,781. In addition, hCG can be used as an inhibitor of matrix metalloproteinases in conditions benefiting from such treatment, such as chronic inflammation, multiple sclerosis and angiogenesis-dependent diseases (see us patent No.6,444,639).
U, alpha-iduronidase
Alpha-iduronidase from Aldura zymeTMCommercially available (BioMarin and Genzyme). It is used for alternative treatment of MPS I, a lysosomal storage disorder. MPS I (also known as huler's disease) is a genetic disease caused by a deficiency in α -L-iduronidase, an enzyme that is generally required for the breakdown of certain complex carbohydrates known as glycosaminoglycans (GAGs). If the enzyme is not present in sufficient quantity, then the normal breakdown of GAGs is incomplete or blocked. The cells are thus unable to shed sugar residues, whereupon they accumulate in the lysosomes of the cells and cause MPS I.
The reconstituted α -iduronidase peptide can be administered to a patient selected from a patient having a lysosomal storage disorder, a patient having a deficiency in α -L-iduronidase, a patient having mucopolysaccharidosis i (mps i), and a patient having huler disease. Preferably, the patient is a human patient.
Methods for producing and purifying alpha-iduronidase and methods for treating certain genetic conditions, including alpha-L-iduronidase deficiency and mucopolysaccharidosis i (mps i), are described in U.S. patent No.6,426,208. The nucleic acid and amino acid sequences of human α -iduronidase are shown in FIGS. 80A (SEQ ID NO: 65) and 80B (SEQ ID NO: 66), respectively.
V. alpha-galactosidase A
Alpha-galactosidase A (also known as agalsidase beta) by FabrazymeTMFor commercial sale (Genzyme). Alpha-galactosidase A is used for treating Fabry disease. Fabry disease is a rare genetic sign caused by a key alpha-galactosidase a deficiency. Without this enzyme, fabry patients are unable to break down their in vivo fatty acid material called globotriasylceramide (GL-3), which accumulates in cells in the heart, kidneys, brain and other critical organ vessels. The gradual accumulation of the substance can cause the patient to have apoplexy and heartRisk of seizure, renal injury, and disabling pain. Many patients develop renal failure during adulthood, and serious organ complications lead to death at about the age of forty.
The reconstituted α -galactosidase a peptide can be administered to a patient selected from the group consisting of a patient having a lysosomal storage disorder, a patient having a deficiency in α -galactosidase a, and a patient having fabry's disease. Preferably, the patient is a human patient.
Alpha-galactosidase A is a lysosomal enzyme that hydrolyzes globotriaosylceramide and related glycolipids with terminal alpha-galactosidase linkages. It is a 45kDa N-glycosylated protein encoded on the long arm of the X chromosome. The initial glycosylated form (Mr. 55,000-58,000) synthesized in human fibroblasts or Chang hepatocytes is processed to the mature glycosylated form (Mr. 50,000). The mature active enzyme purified from human tissues and plasma is a homodimer (Bishop et al, 1986, Proc. Natl. Acad. Sci. USA 83: 4859-4863). The nucleic acid and amino acid sequences of alpha-galactosidase A are seen in FIGS. 81A (SEQ ID NO: 67) and 81B (SEQ ID NO: 68), respectively. Other useful nucleic acid and amino acid sequences of alpha-galactosidase A are found in U.S. Pat. No.6,329,191.
References teaching how to prepare alpha-galactosidase a are found in U.S. patent nos. 5,179,023 and 5,658,567 (expressed in insect cells), U.S. patent No.5,356,804 (expressed and secreted in mammalian cells, including CHO cells), U.S. patent No.5,401,451 (expressed in mammalian cells), U.S. patent No.5,580,757 (expressed in mammalian cells as a fusion protein), and U.S. patent No.5,929,304 (expressed in plant cells). Methods for purifying recombinant α -galactosidase a are described in U.S. Pat. No.6,395,884.
References teaching how to apply alpha-galactosidase a to treat patients include, but are not limited to, U.S. patent No.6,066,626 (gene therapy) and U.S. patent No.6,461,609 (protein therapy). Alpha-galactosidase A mutants useful in the methods of the invention include, but are not limited to, those described in U.S. Pat. No.6,210,666.
W. antibody
The invention further comprises methods of reconstituting various antibody formulations including chimeric antibody formulations, including chimeric TNFR, chimeric anti-glycoprotein IIb/IIIa, chimeric anti-HER 2, chimeric anti-RSV, chimeric anti-CD 20, and chimeric anti-TNF. The chimeric antibody preparation comprises a human Fc portion from an IgG antibody and a variable region from a monoclonal antibody specific for the antigen. Other formulations comprise a receptor, such as the 75kDa TNF receptor, fused to the Fc portion of a human IgG. These molecules further include Fab fragments comprising light and heavy chains from human and mouse. The chimeric TNFR are useful for the treatment of inflammatory diseases, such as rheumatoid arthritis. The chimeric anti-glycoprotein IIb/IIIa is useful for treating cardiac abnormalities, blood coagulation, and platelet dysfunction. Chimeric anti-HER 2 is useful as a therapeutic agent for breast cancer, chimeric anti-RSV is useful for the treatment of respiratory syncytial virus, chimeric anti-CD 20 is useful for the treatment of non-hodgkin's lymphoma, and chimeric anti-TNF is useful for the treatment of Crohn's disease.
Although these chimeric antibodies have proven useful in the management of various diseases, they need to be administered quite frequently and at quite high doses due to the relatively short half-life of the recombinant protein produced in rodent cells. Although most chimeric antibodies are human and therefore recognized as "self by the immune system, they will be degraded and destroyed due to the non-natural glycosylation pattern. The present invention addresses this problem, thereby greatly increasing the efficacy of these new drugs.
Antibodies and methods for producing same
As used herein, the term "antibody" refers to an immunoglobulin molecule that is capable of specifically binding to a particular epitope on an antigen. The antibody may be an intact immunoglobulin derived from a natural source or derived from a recombinant source, or an immunoreactive portion of an intact immunoglobulin. Antibodies are typically tetramers of immunoglobulin molecules. The antibody of the present invention may be stored in various formsIn this context, such forms include, for example, polyclonal antibodies, monoclonal antibodies, Fv, Fab and F (ab)2And single chain Antibodies and humanized Antibodies (Harlow et al, 1999, Using Antibodies: antibody Manual, Cold Spring Harbor Laboratory Press, NY; Harlow et al, 1989, Antibodies: A Laboratory Manual, Cold Spring Harbor, New York; Houston et al, 1988, Proc. Natl. Acad. Sci. USA 85: 5879-.
As used herein, the term "synthetic antibody" refers to an antibody produced using recombinant DNA techniques, such as an antibody expressed by a bacteriophage as described herein. The term should also be construed as an antibody produced by synthesizing a DNA encoding and expressing the antibody or an amino acid sequence encoding the antibody, wherein the DNA or amino acid sequence is obtained using synthetic DNA or amino acid sequence techniques available and well known in the art.
Monoclonal Antibodies directed against full length peptides or peptide fragments can be prepared using any of the well known monoclonal antibody preparation procedures, such as those described in Harlow et al (1988, Antibodies, laboratory Manual, Cold Spring Harbor, NY) and Tuszynski et al (1988, Blood, 72: 109-. The desired peptide may also be synthesized using chemical synthesis techniques. Alternatively, the DNA encoding the desired peptide may be cloned and expressed from an appropriate promoter sequence in a cell suitable for the production of large quantities of the peptide. Monoclonal antibodies directed against the peptide were generated from mice immunized with the peptide using standard procedures as referenced herein.
Nucleic acids encoding monoclonal antibodies obtained using the procedures described herein can be cloned and sequenced using techniques available in the art and are described, for example, in Wright et al (1992, Critical Rev. in Immunol.12(3, 4): 125- & 168) and in the references cited therein. Further, the antibodies of the invention can be "humanized" using techniques described in Wright et al (supra) and references cited therein and in Gu et al (1997, Thrombosis and Hematocyst 77 (4): 755 759).
To generate a phage antibody library, a cDNA library is first obtained from mRNA isolated from cells (e.g., hybridomas) that express the desired peptide, e.g., the desired antibody, to be expressed on the phage surface. cDNA copies of mRNA are generated using reverse transcriptase. The cDNA encoding the immunoglobulin fragments is obtained by PCR, and the resulting DNA is cloned into an appropriate phage vector to generate a phage DNA library comprising DNA-defined immunoglobulin genes. Procedures for preparing phage libraries comprising heterologous DNA are well known in the art and are described, for example, in Sambrook and Russell (2001, Molecular Cloning: A laboratory Manual, Cold Spring Harbor, N.Y.).
The phage encoding the desired antibody can be processed so that the peptides are displayed on their surface in such a way that they are capable of binding to their corresponding binding peptide, such as the antigen against which the antibody is directed. Thus, when a phage expressing a particular antibody is incubated in the presence of cells expressing the corresponding antigen, the phage will bind to the cells. Phage that do not express antibodies will not bind to cells. Such panning techniques are well known in the art and are described, for example, in Wright et al (supra).
Methods for producing human antibodies using M13 phage display have been developed, such as those described above (Burton et al, 1994, adv. Immunol.57: 191-280). Basically, cDNA libraries are generated from mRNA obtained from a population of antibody-producing cells. The mRNA encodes a rearranged immunoglobulin gene, and the cDNA thus encodes the same rearranged immunoglobulin gene. The amplified cDNA was cloned into an M13 expression vector, thereby generating a phage library expressing human antibody fragments on its surface. Phage displaying the antibody of interest are selected by antigen binding and propagated in bacteria to produce soluble human immunoglobulins. Thus, in contrast to conventional monoclonal antibody synthesis, this procedure immortalizes DNA encoding human immunoglobulin rather than cells expressing human immunoglobulin.
For antibody molecule glycanStructure of the organization
The specific glycosylation of a class of peptides, i.e. immunoglobulins, has a particularly important role for the biological activity of these peptides. The present invention should not be construed as limited to immunoglobulins of the IgG class only, but rather as immunoglobulins comprising antibodies of the IgA, IgE and IgM classes.
Further, the present invention should not be construed as limited to any type of conventional antibody structure. Rather, the invention should be construed to include all types of antibody molecules, including, for example, antibody fragments, chimeric antibodies, human antibodies, humanized antibodies, and the like.
A typical immunoglobulin molecule comprises an effector moiety and an antigen-binding moiety. For a review of immunoglobulins, see Harlow et al, 1988, Antibodies: a Laboratory Manual, Cold Spring Harbor, New York; and Harlow et al, 1999, Using Antibodies: a Laboratory Manual, Cold Spring Harbor Laboratory Press, NY. The effector portion of an immunoglobulin is located in the Fc portion of the molecule and is responsible, in part, for the efficient binding of the immunoglobulin to its associated cellular receptor. Incorrect glycosylation of an immunoglobulin molecule, particularly in the CH2 domain of the Fc portion of the molecule, can affect the biological activity of an immunoglobulin.
With respect to immunoglobulin IgG, more specifically, IgG effector function is determined in large part by whether IgG contains an N-acetylglucosamine (GlcNAc) residue attached at the 4-0 position of the branching mannose of the trimannose core of an N-linked glycan at asparagine (Asn)297 in the CH2 domain of the IgG molecule. This residue is known as "bisected GlcNAc". The purpose of adding aliquots of GlcNAc to the N-glycan chains of a native or recombinant IgG molecule or chimeric construct containing IgG-Fc is to optimize the Fc immune effector function of the Fc portion of the molecule. Such effector functions may include Antibody Dependent Cellular Cytotoxicity (ADCC) and any other biological effects that require effective binding to the Fc γ R receptor and binding to the C1 component of complement. The importance of bisecting GlcNAc for achieving the maximum immune effector function of IgG molecules has been described (Lifely et al, 1995, Glycobiology5 (8): 813-822; Jeffris et al, 1990, biochem. J.268 (3): 529-537).
Glycans found at the glycosylation site Asn297 of the CH2 domain of IgG molecules are structurally characteristic for the discovery of IgG molecules circulating in the plasma of human and animal blood, IgG produced by myeloma cells, hybridoma cells, and various transfected immortalized mammalian and insect cell lines. In all cases, N-glycans are either high mannose chains or complete (Man3, GlcNAc4, Gal2, NeuAc2, Fuc1) or variable incomplete bipartite chains with or without bisecting GlcNAc (Raju et al, 2000, Glycobiology 10 (5): 477-.
The present invention provides glycosylated immunoglobulin molecules that are customized in vitro. The immunoglobulin molecule may be any immunoglobulin molecule, including but not limited to monoclonal antibodies, synthetic antibodies, chimeric antibodies, humanized antibodies, and the like. Specific methods of generating antibodies and characterizing them are described elsewhere. Preferably, the immunoglobulin is an IgG, and more preferably the IgG is a humanized or human IgG, most preferably IgG 1.
The present invention specifically relates to the use of β 1, 4-mannosyl-glycopeptide β 1, 4-N-acetylglucosaminyltransferase, GnT-III: EC2.4.1.144 as an in vitro reagent to allow N-acetylglucosamine (GlcNAc) to be linked by glycosidic linkages to the 4-0 position of the branching mannose of the trimannose core of an N-glycan at Asn 297 in the CH2 domain of an IgG molecule. However, as will be understood from the disclosure provided herein, the invention should not be construed as merely including the application of the enzyme to provide an aliquot of GlcNAc to an immunoglobulin molecule. In contrast, it has been found possible to modulate the glycosylation pattern of an antibody molecule such that the antibody molecule has enhanced biological activity, i.e. effector function, in addition to potential enhancements in other properties such as stability and the like.
In the present invention, a general method is provided for the removal of fucose molecules from Asn (297) N-linked glycans for the purpose of enhancing binding to Fc- γ RIIIA and enhancing antibody-dependent cytotoxicity (see Shiels et al, 2002, J.biol. chem.277: 26733-26740). The method entails contacting the antibody molecule with a fucosidase suitable for the attachment of a fucose molecule on an antibody glycan. Alternatively, the recombinant antibody may be expressed in a cell expressing fucosyltransferase, such as the Lec13 variant of CHO cells. The removal of fucose from the antibody glycans can be done alone or in combination with other methods of reconstituting the glycans, such as adding aliquots of GlcNAc. Expression of antibodies in cells lacking GnT-I also produces Fc glycans lacking core fucose, which glycans can be further modified by the present invention.
In the present invention, a general method is provided for introducing an aliquot of GlcNAc for the purpose of enhancing Fc immune effector function in a preparation of IgG molecules containing N-linked oligosaccharides in the CH2 domain, particularly at Asn 297. This method requires that the population of IgG molecules be subjected to a glycosylation state such that the glycan chain is a receptor for GnT-III. This is achieved by any of the following 3 approaches: 1) by selection or genetic manipulation of a host expression system that secretes IgG having an N-glycan chain as a substrate of GnT-III; 2) treating the population of IgG glycoforms with an exoglycosidase such that the glycan structures remaining after exoglycosidase treatment are receptors for GnT-III; 3) some combination of host selection and exoglycosidase treatment as in 1) and 2) above, plus the subsequent addition of GlcNAc by GnT-I and GnT-II to generate receptors for GnT-III.
For example, IgG obtained from chicken plasma contains predominantly high mannose chains and requires digestion with one or more alpha-mannosidases to produce substrates to add GlcNAc to the alpha 1, 3 mannose branches of the mannose core by GnT-I. The substrate may be the basic trimannose core, Man3GlcNAc 2. Treatment of this core structure with a combination of GnT-I, GnT-II and GnT-III with UDP-GlcNAc as the sugar donor produced Man3GlcNAc5 as shown in FIG. 1. The order of action of these glycosyltransferases may be altered to optimize production of the desired product. Alternatively, the structure may then be extended by treatment with β 1, 4 galactosyltransferase. If desired, the galactosylated oligosaccharides may be further extended with α 2, 3-or α 2, 6-sialyltransferase to give the complete biantennary structure. Using this approach, biantennary glycan chains can be reconfigured as necessary for optimal Fc immune effector function of any of the therapeutic IgGs under development (FIG. 3).
Alternatively, IgG molecules found in the plasma of most animals or IgG secreted as recombinant products by most animal cells or by transgenic animals generally comprise a series of bi-antennary glycoforms including the complete form with or without bisecting GlcNAc (NeuAc2, Gal2, GlcNAc4, Man3,. + -. Fuc1) (FIG. 3) or the variable incomplete form (Raju et al, 2000, Glycobiology 10 (5): 477-. To ensure that an aliquot of GlcNAc is present in the entire immunoglobulin population thus produced, the mixture of molecules may be treated with the following exoglycosidases either sequentially or in mixture: neuraminidase, beta-galactosidase, beta-hexosaminidase, alpha-fucosidase. The resulting trimannose core can then be reconstituted with a glycosyltransferase as described above.
In some cases, it is desirable to eliminate effector functions from existing antibody molecules. The invention also includes modification of Fc glycans with appropriate glycosidases and glycosyltransferases to eliminate effector functions. It is also contemplated to add a saccharide modified with PEG or other polymer for blocking or eliminating Fc receptor or complement binding to the antibody.
Furthermore, IgG secreted by transgenic animals and stored by transgenic plants as "plant bodies" (were characterized. IgG molecules produced in transgenic plants having N-glycans with β 1, 2-linked xylose and/or α 1, 3-linked fucose can also be treated with exoglycosidases other than the exoglycosidases described above to remove these residues, thereby producing a trimannose core or Man3GlcNAc4 structure, and then treated with glycosyltransferases to reconstitute the above-described N-glycans.
The main innovative aspect of the present invention is the use of a suitable glycosyltransferase, with or without prior exoglycosidase treatment, and applied in the correct sequence to optimize the effector function of the antibody. In an exemplary embodiment, an aliquot of GlcNAc is introduced into the glycans of an IgG molecule or other IgG-Fc-chimeric construct that require the aliquot of GlcNAc. In another exemplary embodiment, core fucose is removed from glycans of an IgG molecule or other IgG-Fc-chimeric construct.
TNF receptor-IgG Fc fusion protein
The nucleotide and amino acid sequences of the 75kDa human TNF receptor are herein represented by SEQ ID NOs: 31 and SEQ ID NO: 32 (fig. 82A and 82B, respectively). The amino acid sequences of the light and heavy chain variable regions of chimeric anti-HER 2 are set forth in SEQ ID NOs: 35 and SEQ ID NO: 36 (fig. 83A and 83B, respectively). The amino acid sequences of the heavy and light chain variable regions of the chimeric anti-RSV are represented by SEQ ID NOs: 38 and SEQ ID NO: 37 (fig. 84A and 84B, respectively). The amino acid sequences of the non-human variable regions of anti-TNF are represented herein as SEQ ID NOs: 41 and SEQ ID NO: 42 (fig. 85A and 85B, respectively). The nucleotide and amino acid sequences of the Fc portion of human IgG are represented by SEQ ID NOs: 49 and SEQ ID NO: 50 (fig. 86A and 86B, respectively).
Reconstituted chimeric ENBRELTMCan be administered to a patient selected from the group consisting of patients with rheumatoid arthritis and patients with juvenile arthritis of a polyarticular course. Reconstituted chimeric ENBRELTMCan also be administered to an arthritic subject to reduce signs, symptoms or structural damage in the subject. Preferably, the patient is a human patient.
Reconstituted SynagisTMThe antibodies can be administered to a patient to immunize the patient against Respiratory Syncytial Virus (RSV) infection. Reconstituted SynagisTMThe antibodies may also be administered to a patient for prophylaxis orReducing the severity of lower respiratory tract disease caused by RSV. Preferably, the patient is a human patient.
MAb anti-glycoprotein IIb/IIIa
The amino acid sequences of the variable regions of the murine anti-glycoprotein IIb/IIIa antibodies are represented by SEQ ID NOs: 52 (murine mature variable light chain, fig. 87) and SEQ ID NO: 54 (murine mature variable heavy chain, FIG. 88). These murine sequences can be compared to the human IgG amino acid sequence SEQ ID NO: 51 (human mature variable light chain, fig. 89), SEQ ID NO: 53 (human mature variable heavy chain, FIG. 90), SEQ ID NO: 55 (human light chain, fig. 91) and SEQ ID NO: 56 (human heavy chain, FIG. 92) were combined to generate chimeric humanized murine anti-glycoprotein IIb/IIIa antibodies. Other anti-glycoprotein IIb/IIIa humanized antibodies can be found in U.S. Pat. No.5,877,006. Cell lines expressing anti-glycoprotein IIb/IIIa MAb7E3 are commercially available from ATCC (Manassas, VA) under catalog No. HB-8832.
Indications for selected antibodies
Reconstituted ReoproTMCan be administered to a patient selected from the group consisting of a patient undergoing a percutaneous coronary intervention and a patient having unstable angina (wherein the patient is scheduled to undergo a percutaneous coronary intervention within 24 hoursTMCan also be administered to a patient undergoing percutaneous coronary intervention to reduce or prevent cardiac ischemic complications in the patient. Preferably, the patient is a human patient.
Reconstituted HerceptinTMCan be administered to a patient having metastatic breast cancer overexpressing HER2 protein. Preferably, the patient is a human patient.
Reconstructed RemicadeTMCan be administered to a patient selected from the group consisting of a patient with rheumatoid arthritis, a patient with Crohn's disease, and a patient with fistulized Crohn's disease. Reconstructed RemicadeTMCan also be used for treating rheumatoid arthritis to relieve rheumatoid arthritisSigns and symptoms of wet arthritis. RemicadeTMCan also be administered to a patient with Crohn's disease to reduce the signs and symptoms of Crohn's disease in the patient. Preferably, the patient is a human patient.
The nucleic acid and amino acid sequences of the chimeric anti-CD 20 antibody are set forth in SEQ ID NO: 59 (nucleic acid sequence of murine variable region light chain, FIG. 93A), SEQ ID NO: 60 (amino acid sequence of murine variable region light chain, FIG. 93B), SEQ ID NO: 61 (nucleic acid sequence of murine variable region heavy chain, FIG. 94A) and SEQ ID NO: 62 (amino acid sequence of murine variable region heavy chain, FIG. 94B). To humanize murine antibodies, TCAE 8(SEQ ID NO: 57, FIGS. 95A-95E) containing human IgG heavy and light chain constant domains can be conveniently used. Cloning of the murine variable region encoding DNA described above into the TCAE 8 vector according to the teachings given in U.S. Pat. No.5,736,137A vector (SEQ ID NO: 58, FIGS. 96A-96E) was constructed which, when transformed into a mammalian cell line, expresses the chimeric anti-CD 20 antibody. Other humanized anti-CD 20 antibodies may be found in U.S. patent No.6,120,767. Cell lines expressing anti-CD 20 antibody MAb C273 are commercially available from ATCC (Manassas, VA) under catalog No. hb-9303.
The skilled artisan will readily appreciate that the sequences presented herein are not exclusive, but are examples of the variable regions, receptors, and other binding portions of chimeric antibodies. Further, methods of constructing chimeric or "humanized" antibodies are well known in the art and are described, for example, in U.S. Pat. No.6,329,511 and U.S. Pat. No.6,210,671. In connection with the present disclosure and methods well known in the art, the skilled artisan will recognize that the invention is not limited to the sequences disclosed herein.
Expression of chimeric antibodies is well known in the art and is described in detail, for example, in U.S. patent No.6,329,511. The expression system may be prokaryotic, eukaryotic, and the like. Further, expression of the chimeric antibody in insect cells using a baculovirus expression system is described in Putlitz et al (1990, Bio/Technology 8: 651-654). Furthermore, methods of expressing nucleic acids encoding fusion or chimeric proteins are well known in the art and are described, for example, in Sambrook et al (2001, Molecular Cloning, A Laboratory Manual, Cold Spring harbor Laboratory Press, New York) and Ausubel et al (1997, Current protocols Molecular Biology, Green & Wiley, New York).
Determination of the function and biological activity of the chimeric antibodies produced according to the methods of the invention is also an essential operation for those skilled in the art. Methods for determining antibody affinity by competition assays are described in detail in Berzofsky (j.a. Berzofsky and i.j. berkower, 1984, Fundamental Immunology (ed.w.e.paul), Raven Press (new york), 595). Briefly, the affinity of a chimeric antibody is compared to that of a monoclonal antibody from which the chimeric antibody was derived using a radioiodinated monoclonal antibody.
Reconstituted anti-CD 20 antibody can be administered to a patient with relapsed or refractory low-grade or follicular, CD 20-positive, B-cell non-hodgkin's lymphoma. Preferably, the patient is a human patient.
VII pharmaceutical compositions
In another aspect, the present invention provides a pharmaceutical composition. The pharmaceutical composition includes a pharmaceutically acceptable diluent and a covalent conjugate between a non-naturally occurring water-soluble polymer, a therapeutic moiety or biomolecule and a glycosylated or non-glycosylated peptide. The polymer, therapeutic moiety or biomolecule is conjugated to the peptide through an intact glycosyl linking group interposed between and covalently attached to the peptide and the polymer, therapeutic moiety or biomolecule.
The pharmaceutical compositions of the present invention are suitable for use in a variety of drug delivery systems. Suitable formulations for use in the present invention may be found in Remington's Pharmaceutical Sciences, MacePublizing Company, Philadelphia, PA, 17thed. (1985). For drug delivery formulationsFor a brief description of the method, see Langer, Science 249: 1527-1533(1990).
The pharmaceutical composition can be prepared for any suitable mode of administration, including, e.g., topical, oral, nasal, intravenous, intracranial, intraperitoneal, subcutaneous, or intramuscular administration. For parenteral administration, such as subcutaneous injection, the carrier preferably comprises water, saline, alcohol, fat, wax or buffer. For oral administration, any of the above carriers or solid carriers can be employed, such as mannitol, lactose, starch, magnesium stearate, sodium saccharine, talcum, cellulose, glucose, sucrose, and magnesium carbonate. Biodegradable microspheres (e.g., polylactate polyglycolite) may also be used as carriers for the pharmaceutical compositions of the present invention. Suitable biodegradable microspheres are disclosed, for example, in U.S. Pat. Nos. 4,897,268 and 5,075,109.
Typically, the pharmaceutical composition is administered parenterally, such as intravenously. Thus, the present invention provides compositions for parenteral administration comprising the compound dissolved or suspended in a suitable carrier, preferably an aqueous carrier, such as water, buffered water, saline, PBS and the like. The composition may contain pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents, wetting agents, detergents and the like.
These compositions may be sterilized by conventional sterilization techniques, or may be filter sterilized. The resulting aqueous solution may be packaged for use as is, or may be lyophilized, the lyophilized formulation being combined with a sterile aqueous carrier prior to administration. The pH of the formulation is generally 3 to 11, more preferably 5 to 9, and most preferably 7 to 8.
In some embodiments, the peptides of the invention may be incorporated into liposomes formed from standard vesicle-forming lipids. Various methods of preparing liposomes are available, as described in Szoka et al, ann.rev.biophysis.bioeng.9: 467(1980), U.S. Pat. Nos. 4,235,871, 4,501,728 and 4,837,028. Targeting of liposomes using various targeting agents, such as the sialylgalactosides of the present invention, is well known in the art (see U.S. Pat. Nos. 4,957,773 and 4,603,044).
Standard methods of coupling targeting agents to liposomes can be used. These methods generally involve incorporating a lipid component into the liposome, such as phosphatidylethanolamine, which can be activated to attach a targeting agent, or a derivatized lipophilic compound, such as a lipid-derived peptide of the invention.
The targeting mechanism generally requires that the targeting agent be localized to the surface of the liposome in such a way that the targeting moiety is capable of interacting with a target, such as a cell surface receptor. The saccharides of the invention may be attached to the lipid molecules prior to formation of the liposomes by methods well known to those skilled in the art, such as alkylation or acylation of the hydroxyl groups present on the saccharides with long chain alkyl halides or fatty acids, respectively. Alternatively, the liposomes can be formed in such a way that the linker moiety can be first incorporated into the membrane at the time of membrane formation. The linker moiety must have a lipophilic portion that can be firmly embedded and anchored in the membrane. It must also have a reactive moiety that is chemically available at the aqueous surface of the liposome. The reaction moiety is selected such that it is chemically suitable to form a stable chemical bond with a subsequently added directing agent or sugar. In some cases it is possible to attach the targeting agent directly to the linker molecule, but in most cases it is more appropriate to use a third molecule as a chemical bridge, connecting the linker molecule in the membrane and the targeting agent or sugar, which is three-dimensionally extended from the vesicle surface. The peptides of the invention are administered in a dosage range large enough to produce the desired effect, wherein the symptoms of the immune response show some degree of suppression. The dosage should not be too large to cause side effects. Generally, the dosage will vary depending on the age, condition, sex, and extent of disease in the animal, and can be determined by one of skill in the art. The dosage can be adjusted by a single physician in the presence of any contraindications.
Additional pharmaceutical methods may be applied to control the duration of action. Controlled release formulations can be achieved by conjugating, complexing, or adsorbing the peptide using a polymer. Controlled release delivery can be practiced by selecting the appropriate macromolecules (e.g., polyesters, polyaminocarboxymethyl cellulose, and protamine sulfate) and the concentration of macromolecules and integration methods to control release. Another possible way to control the duration of action of a controlled release formulation is to incorporate the peptide into particles of a polymeric material such as a polyester, polyamino acid, hydrogel, poly (lactic acid) or ethylene vinyl acetate copolymer.
To prevent binding of the peptide to the plasma protein, it is preferred to entrap the peptide in microcapsules prepared by coacervation techniques or interfacial polymerization, such as hydroxymethylcellulose or gelatin-microcapsules and poly (methylmethacylate) microcapsules, respectively, or in colloidal drug delivery systems, such as liposomes, albumin microspheres, microemulsions, nanoparticles and nanocapsules or macroemulsions. Such an introduction is disclosed in Remington's Pharmaceutical Sciences (16)thEd.,A.Oslo,ed.,Mark,Easton,Pa.,1980)。
The peptides of the invention are well suited for use in a targeted drug delivery system such as synthetic or natural polymers in the form of macromolecular complexes, nanocapsules, microspheres or beads, and in lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, liposomes and re-encapsulated red blood cells. These systems are collectively known as colloidal drug delivery systems. Typically, such colloidal particles containing dispersed peptides are about 50nm to 2 μm in diameter. The colloidal particles are of a size such that they can be administered intravenously by injection or as an aerosol. Materials used in the formulation of colloidal systems are generally sterilized by filter sterilization, non-toxic and biodegradable, such as albumin, ethylcellulose, casein, gelatin, lecithin, phospholipids and soybean oil. The polymerized colloid system is prepared by a process similar to coacervation microencapsulation.
In an exemplary embodiment, the peptide is used as a component of a liposome for a targeted delivery system. When phospholipids are gently dispersed in an aqueous medium, they swell, hydrate and spontaneously form multiple concentric bilayer vesicles in which the aqueous medium layer separates the lipid bilayers. Such systems are commonly referred to as multilamellar liposomes or multilamellar vesicles (MLVs) and range in diameter from about 100nm to about 4 μm. When MLVs are sonicated, Small Unilamellar Vesicles (SUVS) are formed having diameters ranging from about 20 to about 50nm, which contain an aqueous solution in the core of the SUV.
Examples of lipids used in liposome production include phosphatidyl compounds such as phosphatidylglycerol, phosphatidylcholine, phosphatidylserine, and phosphatidylethanolamine. Particularly useful are diacylphosphatidylglycerols, wherein the lipid moiety contains 14-18 carbon atoms, particularly 16-18 carbon atoms, and is saturated. Exemplary phospholipids include egg phosphatidylcholine, dipalmitoylphosphatidylcholine and distearoylphosphatidylcholine.
In preparing liposomes containing the peptides of the invention, such variables as the efficiency with which the peptides are encapsulated, peptide instability, homogeneity and size of the resulting liposome population, peptide-lipid ratio, osmotic instability of the formulation and pharmaceutical acceptability of the formulation should be considered. Szoka et al, annual lView of Biophysics and Bioengineering, 9: 467 (1980); deamer et al, Liposomes, Marcel Dekker, New York, 1983, 27; hope et al, chem.phys.lipids, 40: 89(1986)).
The targeted delivery systems containing the peptides of the invention can be administered to a host, particularly a mammalian host, in a variety of ways, such as intravenous, intramuscular, subcutaneous, intraperitoneal, intravascular, topical, intracavity, transdermal, intranasal, or inhalation. The concentration of the peptide will vary depending on the particular application, the nature of the disease, the frequency of administration, and the like. The encapsulated peptide of the targeted delivery system may be provided in a formulation comprising other suitable compounds and an aqueous physiologically acceptable medium, such as saline, phosphate buffered saline, and the like.
The compounds prepared by the methods of the invention are also useful as diagnostic reagents. For example, labeled compounds may be used in the treatment of a patient suspected of having inflammationTo locate areas of inflammation or tumor metastasis in a patient. For this use, the compounds are useful125I、14C or tritium.
Experimental examples
The invention will now be described in connection with the following examples. These examples are provided for illustrative purposes only, and the present invention should in no way be construed as limited to these examples, but rather should be construed to encompass any and all variations which become apparent as a result of the description provided herein.
The materials and methods used in the experiments presented in this example are now described.
A. General procedure
Preparation of CMP-SA-PEG
This example presents the preparation of CMP-SA-PEG.
Preparation of 2- (benzyloxyamide) -glycylamide-2-deoxy-D-mannopyranose.N-benzyloxycarbonyl-glycyl-N-hydroxysuccinimide ester (3.125g, 10.2mmol) was added to a solution containing MeOH (10mL) and H2D-mannosamine-HCl (2g, 9.3mmol) in O (6mL) and triethylamine (1.42mL, 10.2 mmol). The reaction was stirred at room temperature for 16 hours and concentrated by rotary evaporation (rotoevaporation). Chromatography (silica, 10% MeOH/CH)2Cl2) 1.71g (50% yield) of white solid product are obtained: rf0.62 (silicon, CHCl)3:MeOH:H2O,6/4/1);1H NMR(CD3OD,500MHz)δ3.24-3.27(m,2H),3.44(t,1H),3.55(t,1H),3.63-3.66(m,1H),3.76-3.90(m,6H),3.91(s,2H),4.0(dd,2H),4.28(d,1H,J=4.4),4.41(d,1H,J=3.2),5.03(s,1H),5.10(m,3H),7.29-7.38(m,10H)。
5- (N-Benzyloxyamide) glycylamide-3, 5-dideoxy-D-glycero-D-galactose Preparation of 2-nonylpyranosuronate.2- (N-benzyl)Oxyamide) glycylamide-2-deoxy-D-mannopyranose (1.59g, 4.3mmol) is dissolved in a solution of 0.1M HEPES (12mL, pH7.5) and sodium pyruvate (4.73g, 43 mmol). Neuraminic acid aldolase (540U of the enzyme in 45mL 10mM phosphate buffer pH6.9 containing 0.1M NaCl) and the reaction mixture were heated to 37 ℃ for 24 hours. The reaction mixture was then centrifuged and the supernatant was subjected to chromatography (C18 silica, gradient from H20 (100%) to 30% MeoH/water). The appropriate fractions were collected, concentrated and the residue was chromatographed (C18 silica, from 10% MeOH/CH) 2Cl2To CH2Cl2/MeOH/H2A gradient of O6/4/1). The appropriate fractions were collected, concentrated and the residue resuspended in water. After lyophilization, the product was obtained as a white solid (1.67g, 87% yield): rf0.26 (silicon, CHCl)3:MeOH:H2O is 6/4/1);1H NMR(D2O,500MHz)δ 1.82(t,1H),2.20(m,1H),3.49(d,1H),3.59(dd,1H),3.67-3.86(m,2H),3.87(s,2H),8.89-4.05(m,3H),5.16(s,2H),7.45(m,5H)。
5-glycylamide-3, 5-dideoxy-D-glycero-D-galactose Preparation of 2-nonylpyranosuronate.5- (N-Benzyloxyamide) glycylamide-3, 5-dideoxy-D-glycero-D-galactose-nonylpyranosuronate (1.66g, 3.6mmol) was dissolved in 20mL of 50% water/methanol. The flask was repeatedly evacuated and placed in argon, then 10% Pd/C (0.225g) was added. After repeated evacuation, hydrogen (about 1atm) was added to the flask and the reaction mixture was stirred for 18 hours. The reaction mixture was filtered through celite, concentrated on a rotary evaporator and lyophilized to give 1.24g (100% yield) of the product as a white solid: rf0.25 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O,500MHz)δ 1.83(t,1H,J=9.9),2.23(dd,1H,J=12.9,4.69),3.51-3.70(m,2H),3.61(s,2H),3.75-3.84(m,2H),3.95-4.06(m,3H)。
Cytidine-5' -monophosphoryl- [5- (N-fluorenylmethoxy-amide) glycylamide-3, 5-dideoxy -beta-D-Glycerol-D-galactose-2-nonylpyranosuronate]And (4) preparing.Dissolving the solution in 20mL of H2A solution of 5-glycylamide-3, 5-dideoxy-D-glycero-D-galactose-2-nonulopyranosyuronate (0.55g, 1.70mmol) in O was added to Tris (1.38g, 11.4mmol), 1M MgCl 2(1.1mL) and BSA (55 mg). The pH of the solution was adjusted to 8.8 with 1M NaOH (2mL) and CTP-2Na was added+(2.23g, 4.2 mmol). The pH of the reaction mixture is controlled by a pH controller which releases the 1M NaOH required to maintain pH 8.8. The fusion protein (sialyltransferase/CMP-neuraminidase) was added to the solution and the reaction mixture was stirred at room temperature. After 2 days, an additional amount of fusion protein was added and the reaction was stirred for an additional 40 hours. The reaction mixture was precipitated in EtOH and the precipitate was washed 5 times with cold EtOH to give 2.3 g of a white solid. About 1.0g of the crude product was dissolved in 1, 4 dioxane (4mL), H2O (4mL) and saturated NaHCO3(3mL) and a solution of FMOC-Cl (308mg, 1.2mmol) dissolved in 2mL dioxane was added dropwise. After stirring at room temperature for 16H, the reaction mixture was concentrated to about 6mL by rotary evaporation and chromatographed (C18 Si, from 100% H)2O to 30% MeOH/H2Gradient of O) was purified. The appropriate fractions were combined and concentrated. The residue was dissolved in water and lyophilized to obtain 253mg of a white solid: rf0.50 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) δ 1.64(dt, 1H, J is 12.0, 6.0), 2.50(dd, 1H, J is 13.2, 4.9), 3.38(d, J is 9.67, 1H), 3.60(dd, J is 11.65, 6.64, 1H), 3.79(d, J is 4.11, 1H), 3.87(dd, J is 12.24, 1.0, 1H), 3.97(m, 2H), 4.07(td, J is 10.75, 4.84, 1H), 4.17(dd, J is 10.68, 1.0, 1H), 4.25(s, 2H), 4.32(t, J is 4.4, 1H), 4.37(t, J is 5, 1.8, 4.6, 7.7, 7H), 7.7H, 7. Ms (es); to C 35H42N5O18P([M-H]-) Calculated value of 851.7; found 850.0.
Cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycerol-D-galacto-galactan Sugar-2-nonylpyranosuronate).Diisopropylamine (83 μ L, 0.587 μmol) was added to cytidine 5' -monophosphoryl- [5- (N-fluorenylmethoxy-amide) glycylamide-3, 5-dideoxy-. beta. -D-glycerol-D-galactose-2-nonylpyranosuronate dissolved in water (3mL) and methanol (1mL)](100mg, 0.117 mmol). The reaction mixture was stirred at room temperature for 16 hours and the reactant methanol was removed from the reaction mixture by rotary evaporation. The crude reaction mixture was filtered through a C18 silica gel column using water, and the eluent was collected and lyophilized to give the product as a white solid (87mg, 100%): rf0.21 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) δ 1.66(td, 1H, J is 5.3), 2.50(dd, 1H, J is 13.2, 4.6), 3.43(d, J is 9.58, 1H), 3.63(dd, J is 11.9, 6.44, 1H), 3.88(dd, J is 11.8, 1.0, 1H), 3.95(td, J is 9.0, 2.3, 1H), 4.10(t, J is 10.42, 1H), 4.12(td, J is 10.34, 4.66, 1H), 4.18(d, J is 10.36, 1H), 4.24(m, 2H), 4.31(t, J is 4.64, 1H), 4.35(t, 6.00), 1H, 7.7, 7H), 7.7, 7H, 13H, 7H). Ms (es); to C 21H32N5O11P([M-H]-) Calculated value of 629.47; found 627.9.
Cytidine-5' -monophosphoryl- [5- (N-methoxy-polyoxyethylene- (1kDa) -3-oxopropanoyl) AMINE) -GLYCAMIDE-3, 5-DIDEOXY-BETA-D-GLYCERIN-D-GALACTOSE -2-nonulopyranosuronate]And (4) preparing.Benzyltriazol-1-yloxy-tris (dimethylamino) -phosphonium hexafluorophosphate (BOP, 21mg, 48. mu. mol) was added to a solution of methoxypolyoxyethylene- (1kDa average molecular weight) -3-oxopropanoic acid (48mg, 48. mu. mol) dissolved in anhydrous DMF (700. mu.L) and triethylamine (13. mu.L, 95. mu. mol). After 30 minutes, a solution containing cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonylpyranosuronate) (30mg, 48. mu. mol), water (400. mu.L) and triethylamine (13. mu.L, 95. mu. mol) was added. The solution was stirred at room temperature for 20 minutes, thenThis was followed by chromatography (C18 silica, methanol/water gradient). The appropriate fractions were collected, concentrated and the residue resuspended in water and lyophilized to obtain 40mg (50% yield) of a white solid: rf0.36 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) δ 1.66(td, 1H, J5.3), 2.50(dd, 1H, J13.2, 4.6), 2.64(t, J5.99, 3H), 3.43(d, J9.58, 1H), 3.63(m, 1H), 3.71(s, 70H), 3.79(m, blurring caused by the peak of 3.71), 3.82(t, J6.19, 1H), 3.88(dd, J11.8, 1.0, 1H), 3.95(td, J9.0, 2.3, 1H), 3.98(t, J5.06, 1H), 4.12(td, J10.34, 4.66, 1H), 4.18(d, 18, 4.18, 7, 4.35, 4H), 4.6, 4H, 4, 7, 4H, 4, 7, 6, 4.6, 4H, 7, 4H, 7, 6, 4H, 7, 4H, 1H, 6H, 1H, 3H, and 3H. MS (MALDI), Observation of [ M-H ] ];1594.5,1638.5,1682.4,1726.4,1770.3,1814.4,1858.2,1881.5,1903.5,1947.3。
Cytidine-5' -monophosphoryl- [5- (N-methoxy-polyoxyethylene- (10kDa) -oxoamide) -glycylamide-3, 5-dideoxy-beta-D-glycero-D-galactose -2-nonulopyranosuronate]And (4) preparing.Cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonulopyranosyronate) (2.5mg, 4. mu. mol) and water (180. mu.L) were added to a solution of (methoxypolyoxyethylene- (10kDa, average molecular weight) -oxycarbonyl- (N-oxobenzotriazole) ester (40mg, 4. mu. mol) in anhydrous DMF (800. mu.L) containing triethylamine (1.1. mu.L, 8. mu. mol), the reaction mixture was then diluted with water (8mL) and purified by reverse phase flash chromatography (C18 silica, methanol/water gradient.) the appropriate fractions were combined, concentrated and the residue dissolved in water and lyophilized to give 20mg (46% yield) of the product as a white solid: and R is shown in the specification.f0.35 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) delta 1.66(td, 1H), 2.50(dd, 1H), 2.64(t, 3H), 3.55-3.7(m, blurring caused by the peak of 3.71), 3.71(s, 488H), 3.72-4.0(m, blurring caused by the peak of 3.71), 4.23(m, 3H)) 4.31(t, 1H), 4.35(t, 1H), 6.00(d, J ═ 4.77, 1H), 6.12(d, J ═ 7.52, 1H), 7.98(d, J ═ 7.89, 1H). MS (MALDI), Observation of [ M-CMP + Na ];10780。
2.Preparation of CMP-SA-PEG II
This example presents a general method for the preparation of CMP-SA-PEG, and in particular a method for the preparation of CMP-SA-PEG (1kDa) and CMP-SA-PEG (20 kDa).
Preparation of cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-beta-D-glycerol-D- Galactose-2-nonylpyranosuronate).Will be provided withCytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-beta-D-glycero-D-galactose -2-nonylpyranosuronate) (870mg, 1.02mmol) was dissolved in 25mL of water, and 5.5mL of a 40 wt% aqueous dimethylamine solution was added.The reaction was stirred for 1 hour, and then excess dimethylamine was removed by rotary evaporation. The aqueous solution was filtered through a C-18 silica gel column and the column was washed with water. The eluates were combined and lyophilized to give 638mg (93%) of the product as a white solid: rf0.10 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) δ 1.66(td, 1H, J is 5.3), 2.50(dd, 1H, J is 13.2, 4.6), 3.43(d, J is 9.58, 1H), 3.63(dd, J is 11.9, 6.44, 1H), 3.88(dd, J is 11.8, 1.0, 1H), 3.95(td, J is 9.0, 2.3, 1H), 4.10(t, J is 10.42, 1H), 4.12(td, J is 10.34, 4.66, 1H), 4.18(d, J is 10.36, 1H), 4.24(m, 2H), 4.31(t, J is 4.64, 1H), 4.35(t, 6.00), 1H, 7.7, 7H), 7.7, 7H, 13H, 7H). Ms (es); to C 21H32N5O11P([M-H]-) Calculated value of 629.47; found 627.9.
General procedure for the preparation of CMP-SA-PEG using mPEG- (p-nitrophenol) carbonate.
Cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonulopyranosyronate) (175 mg)0.259mMol) was dissolved in a mixture of water and DMF or THF at pH8.5 (ratio 1: 2). mPEG-nitrophenol carbonate (2-20 kDa mPEG) (0.519mMole) was added in portions over 8 hours at room temperature, and the reaction mixture was stirred at room temperature for 3 days. When complete, add water (40ml) and 1.5ml NH4OH (29% aqueous solution). The yellow reaction mixture was stirred for a further 2 hours and then concentrated by rotary evaporation. The reaction mixture was then diluted with water (ph8.5) to a volume of about 500ml and purified by reverse phase flash chromatography (Biotage 40M, C18 silica column) with a methanol/water gradient. The appropriate fractions were combined and concentrated to give the product as a white solid. Rf(silicon; 1-propanol/water/29% NH)4OH; 7/2/1); (2kDa PEG) ═ 0.31; (5kDa PEG) ═ 0.33; (10kDaPEG) ═ 0.36; (20kDa PEG) ═ 0.38(TLC silicon, IPA/H)2O/NH4OH 7/2/1);MS(MALDI),[M-CMP+Na](iii) an observed value; (2kDa) ═ 2460; (5kDa) ═ 5250; (10kDa) ═ 10700; (20kDa) ═ 22500.
Preparation of Cytidine-5' -monophosphoryl- [5- (N-fluorenylmethoxy-carboxamide) -glycylamide-3, 5-
dideoxy-beta-D-glycero-D-galactose-2-nonulopyranosyuronate]General method (4).
Sodium pyruvate (2.4g, 218mmol), HEPES buffer (0.25M, pH7.34) and 1.0g (22mmol) Fmoc-glycylmannosamide were mixed in a 150mL polycarbonate vial. Neuraminic acid aldolase solution (19mL, 600U) was then added and the reaction mixture incubated on an orbital shaker at 30 ℃. After 23 hours, Thin Layer Chromatography (TLC) showed that about 75% conversion to the product had occurred. CTP (1.72g, 33mmol) and 0.1M MnCl were then added to the reaction mixture2(6 mL). The pH was adjusted to 7.5 with 1M NaOH (5.5mL) and a solution (25mL, 386U) containing CMP-neuraminic acid synthetase (Neisseria) was added. The reaction was complete after 24 hours and the reaction mixture was chromatographed (C-18 Si, H)2O (100%) to 10% MeOH/H2Gradient of O). The appropriate fractions were combined, concentrated and lyophilized to give a white solid. Rf(IPA/H2O/NH4OH,7/2/1)=0.52.1H NMR(D2O, 500MHz) δ 1.64(dt, 1H, J is 12.0, 6.0), 2.50(dd, 1H, J is 13.2, 4.9), 3.38(d, J is 9.67, 1H), 3.60(dd, J is 11.65, 6.64, 1H), 3.79(d, J is 4.11, 1H), 3.87(dd, J is 12.24, 1.0, 1H), 3.97(m, 2H), 4.07(td, J is 10.75, 4.84, 1H), 4.17(dd, J is 10.68, 1.0, 1H), 4.25(s, 2H), 4.32(t, J is 4.4, 1H), 4.37(t, J is 5), 4.81, 6.7, 7.7, 7H), 7.7H, 7H; to C 35H42N5O18P([M-H]-) Calculated value of 850.7; found 850.8.
Preparation of Cytidine-5' -monophosphoryl- [5- (N-methoxy-polyoxyethylene- (1kDa) -3-oxo Propionamide) -glycylamide-3, 5-dideoxy-beta-D-glycerol-D-galactose -2-nonulopyranosuronate]General method (4).Methoxy-polyoxyethylene- (1kDa average molecular weight) -3-oxopropanoic acid-N-succinimidyl ester (52mg, 52. mu. mol) was dissolved in anhydrous DMF (450. mu.L) and triethylamine (33. mu.L, 238. mu. mol). Solid cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonulopyranosyuronate) (30mg, 48. mu. mol) was added. Water (330. mu.L) pH8 was added, and after 30 minutes, another 28mg NHS-activated PEG was added. After a further 5 minutes, the reaction mixture was chromatographed (C-18 silica, methanol/water gradient) and the appropriate fractions concentrated to give 32mg (40% yield) of a white solid. Rf0.31 (Si, IPA/H)2O/NH4OH 7/2/1);1H NMR(D2O, 500MHz) δ 1.66(td, 1H, J5.3), 2.50(dd, 1H, J13.2, 4.6), 2.64(t, J5.99, 3H)3.43(d, J9.58, 1H), 3.63(m, 1H), 3.71(s, 70H), 3.79(m, blurring caused by the peak of 3.71), 3.82(t, J6.19, 1H)3.88(dd, J11.8, 1.0, 1H), 3.95(td, J9.0, 2.3, 1H), 3.98(t, J5.06, 1H), 4.12(td, J10.34, 4.66, 1H, 4.18(d, 18, 1H), 4.35, 4H, 4, 6, 4.6, 1H), 4.6, 1H, 4H, 6, 1H, 4H, 6, 1H, 4H, 6, 1H, 4H, 1H, 6, 1H, 4H, 1H, and 3H, 6H, 1H 8(d,J=7.54,1H).MS(MALDI),[(M-CMP)-H](iii) an observed value; 1506.4, 1550.4, 1594.5, 1638.5, 1682.4, 1726.4, 1770.3, 1814.4, 1858.2.
General procedure for the preparation of Cytidine-5' -monophosphoryl- {5- [ N- (2, 6-dimethoxy-polyoxyethylene- (20kDa) -3-oxopropanamide-lysylamide-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonylpyranosuronate }. 2, 6-bis [ methoxy-polyoxyethylene- (20kDa average molecular weight) -3-oxopropanamide ]]-lysylamide-N-succinimidyl ester (367mg, 9. mu. mol) was dissolved in anhydrous THF (7mL) and triethylamine (5. mu.L, 36. mu. mol). Cytidine-5' -monophosphoryl- (5-glycylamide-3, 5-dideoxy-. beta. -D-glycero-D-galactose-2-nonylpyranosuronate) (30mg, 48. mu. mol) was dissolved in 1.0mL of water and added to the reaction mixture. The reaction was stirred at room temperature for 4 hours and then chromatographed (HPLC, Waters Xterra RP8, Water/NH)4OH, 100% to 20% methanol/water/NH4Gradient of OH, 1 mL/min) to obtain a white solid, R thereoft22.8 minutes. MS (MALDI), [ (M-CMP) -H](iii) an observed value; 43027.01(40,000-45,500).
Preparation of UDP-Gal-PEG
This example presents a general procedure for the preparation of UDP-Gal-PEG.
348mg of N-hydroxysuccinimide methoxy-polyoxyethylene propionate (mPEG-SPA, MW1,000) in THF (0.5ml) was added to a solution of 25mg of galactosamine-1-phosphate in 1ml of water followed by 67. mu.L of triethylamine. The resulting mixture was stirred at room temperature for 17 hours. Concentration under reduced pressure afforded a crude reaction mixture that was purified by chromatography (C-18 silica, using a fractional gradient of 10%, 20%, 30%, 40% aqueous MeOH) to afford, after combining the appropriate fractions and concentrating to dryness, 90mg (74%) of the product. R f0.5 (Si, propanol/H)2O/NH4OH 30/20/2); ms (maldi), observed values 1356, 1400, 1444, 1488, 1532, 1576, 1620.
[ alpha-1- (uridine-5' -diphosphoryl)]-2-deoxy-2-, (Methoxy-polyoxyethylene-propionyl Amine-1 kDa) -alpha-D-galactosamine.2-deoxy-2- (methoxy-polyoxyethylene-propionamide-1 kDa) - α -1-monophosphate-D-galactosamine (58mg) was dissolved in 6mL of DMF and 1.2mL of pyridine. UMP-morpholidate (60mg) was then added and the resulting mixture was stirred at 70 ℃ for 48 hours. After concentration, the residue was chromatographed (C-18 silica, using a fractional gradient of 10%, 20%, 30%, 40%, 50%, 80% MeOH) to give 50mg of product after concentration of the appropriate fractions. Rf0.54 (Si, propanol/H)2O/NH4OH 30/20/2). Ms (maldi); observations 1485, 1529, 1618, 1706.
[ alpha-1- (uridine-5' -diphosphoryl)]-6-deoxy-6- (methoxy-polyoxyethylene-amino) -2kDa) - α -D-galactose.Will [ alpha-1- (uridine-5' -diphosphoryl)]-6-Carboxaldehyde (. alpha. -D-galactose (10mg) was dissolved in 2mL of 25mM sodium phosphate buffer (pH6.0) and treated with methoxy-polyethylene glycol amine (MW 2,000, 70mg), followed by 25uL of 1M NaBH3The CN solution was treated at 0 ℃. The resulting mixture was frozen at-20 ℃ for 3 days. The reaction mixture was chromatographed (HPLC, Water Xterra P8) using 0.015M NH 4OH as mobile phase A and MeOH as mobile phase B, at a flow rate of 1.0 mL/min. The product was collected and concentrated to give a solid: rt9.4 min. Rf0.27 (Si, EtOH/H)2O7/3)。
[ alpha-1- (uridine-5' -diphosphoryl)]-6-amino-6-deoxy-alpha-D-galactose.15mg of ammonium acetate was added to [ alpha-1- (uridine-5' -diphosphoryl)]-6-Carboxaldehyde- α -D-galactopyranose (10mg) in sodium phosphate buffer (pH 6.0). Then 1M NaBH was added3CN solution (25 μ L) and the mixture was stirred for 24 hours. The solution was concentrated and the residue was chromatographed (Sephadex G)10) To obtain 10mg of a white solid. Rf0.62 (Si, EtOH/0.1M NH)4Ac)。
[ alpha-1- (uridine-5' -diphosphoryl)]-6-deoxy-6- (methoxy-polyoxyethylene-propionyl) Amine, -2 kDa) -alpha-D-galactopyranose.Will [ alpha-1- (uridine-5' -diphosphoryl)]-6-amino-6-deoxy-alpha-D-galactopyranose (5mg) dissolved in 1mL H2And (4) in O. Methoxy-polyethylene glycol propionyl-NHS ester (MW 2,000, 66mg) was then added followed by 4.6. mu.L triethylamine. The resulting mixture was stirred at room temperature overnight and then purified on HPLC (C-8 silicon) to give the product, Rt9.0 min.
[ alpha-1- (uridine-5' -diphosphoryl)]-6-deoxy-6- (methoxy-polyoxyethylene-formyl) Amine, -2 kDa) -alpha-D-galactopyranose.Will [ alpha-1- (uridine-5' -diphosphoryl)]-6-amino-6-deoxy-alpha-D-galactopyranose (10mg) with methoxy-polyethyleneglycol carboxy-HOBT (MW2,000, 67mg) in 1mL H2O, followed by addition of 6.4mg of EDC (1-ethyl-3- (3-dimethylaminopropyl) carbodiimide hydrochloride and 4.6. mu.L of triethylamine, the resulting mixture was stirred at room temperature for 24 hours, and the mixture was chromatographed (C-8 silica) to give the product.
Preparation of UDP-GlcNAc-PEG
This example presents a general method for the preparation of UDP-GlcNAc-PEG. On the left side of scheme 17, the protected amino sugar diphosphate-nucleotide is oxidized to form an aldehyde at the 6-position of the sugar. The aldehyde is converted to the corresponding primary amine by the formation and reduction of the schiff base. The resulting adduct is contacted with p-nitrophenol carbonate of m-PEG, and the ester is reacted with an amine, thereby binding m-PEG to the sugar core via an amide bond. On the right side of the top of scheme 17, the protected amino sugar diphosphate-nucleotide is treated with a chemical oxidizing agent to form a carboxyl group on the 6-carbon of the sugar core. The carboxyl group is activated and reacted with the m-PEG amine, thereby binding the m-PEG to the sugar core via an amide bond. On the right side of the lower part of scheme 17, the reaction is essentially the same as on the upper part of the right side, except that the starting sugar nucleotide is contacted with an oxidizing enzyme, such as a dehydrogenase, rather than a chemical oxidizing agent.
Preparation of UDP-GalNAc-PEG
This example (scheme 18) presents a general method for the preparation of UDP-GalNAc-PEG. The reaction set forth above begins with a sugar diphosphate-nucleotide, where R is either a hydroxyl group 1 or a protected amine 2. In step a, the starting sugar is treated with a mixture of oxidase and catalase, thereby converting the 6-position of the sugar to the aldehyde moiety (3 and 4). In step c, the aldehyde is converted to the corresponding amine (7 and 8) by the formation and reduction of the schiff base. In step e, the amine is optionally treated with an activated m-PEG derivative, thereby acylating the amine to yield the corresponding m-PEG amide (11 and 13). Alternatively, in step f, the amine is contacted with an activated m-PEG species, such as an m-PEG active ester, to form the corresponding m-PEG amides (12 and 14). In step b, the starting material is also treated with catalase and oxidase, completely oxidizing the hydroxymethyl moiety, thereby forming a carboxyl group at the 6-position. In step d, the carboxyl moiety is activated and subsequently converted to m-PEG adduct (9 and 10) by reaction with m-PEG amine intermediate. This is shown in scheme 18.
a and b): galactose oxidase and catalase dissolved in 25mM sodium phosphate buffer (pH 6.0); c) NH (NH) 4Ac,NaBH3CN dissolved in 25mM sodium phosphate buffer (pH 6.0);
d)CH3(OCH2CH2)NH2,EDC,H2O;e).CH3(OCH2CH2)nNH2,NaBH3CN,H2O
for 15 and 16; f) CH (CH)3O(CH2CH2O)nCH2CH2CONHS,H2O,Et3N
The amino-sugar phosphate is contacted with m-PEG N-hydroxysuccinimide active ester to form the corresponding sugar-PEG-amide. The amide is contacted with UMP-morpholinoate to form the corresponding active sugar diphosphate-nucleotide.
Synthesis of CMP-SA-levulinic acid
This example presents the procedure for the synthesis of CMP-SA-levulinic acid.
Preparation of 2-acetyl propionamide-2-deoxy-D-mannopyranose.Isobutyl chloroformate (100. mu.L, 0.77mmol) was added dropwise to a solution of levulinic acid (86. mu.L, 0.84mmol), anhydrous THF (3mL), and triethylamine (127. mu.L, 0.91 mmol). The solution was stirred at room temperature for 3 hours, and then added dropwise to a solution containing D-mannosamine hydrochloric acid (151mg, 0.7mmol), triethylamine (127. mu.L, 0.91mmol), THF (2mL) and water (2 mL). The reaction mixture was stirred for 15 hours and then concentrated to dryness by rotary evaporation. Chromatography (silica, 5-15% MeOH/CH)2Cl2Step gradient) was used to isolate the product, yielding 0.156g (73% yield) of a white solid: rf0.41 (silicon, CHCl)3MeOH/water 6/4/1);1H NMR(D2O,500MHz)δ2.23(s,3H),2.24(s,3H),2.57(td,J=6.54,3.68,2H),2.63(t,J=6.71,2H),2.86-2.90(m,4H),3.42(m,1H),3.53(t,J=9.76,1H),3.64(t,J=9.43,1H),3.80-3.91(m,4H),4.04(dd,J=9.79,4,71,1H),4.31(dd,J=4.63,1.14,1H),4.45(dd,J=4.16,1.13,1H),5.02(d,J=1.29,1H),5.11(s,J=1.30,1H),MS(ES);C11H19NO7277.27; measured value [ M +1]277.9。
5-acetyl propionamide-3, 5-dideoxy-D-glycero-D-galactose Preparation of 2-nonylpyranosuronate.Sodium pyruvate (0.616g, 5.6mmol) and N-acetylneuraminic acid aldolase (50U) were added to a solution of 2-acetylpropionamide-2-deoxy-D-mannopyranose (0.156g, 0,56mmol) in 0.1M HEPES (pH 7.5). The reaction mixture was heated to 37 ℃ for 20 hours and then frozen. The reaction mixture was then filtered through C18 silicon, frozen and lyophilized. The crude solid was purified by flash chromatography (silica, first using 10-40% MeOH/CH)2Cl2Then is CH2Cl2/MeOH/H2O6/4/0.5). The appropriate fractions were combined and concentrated to give 45mg (80% yield) of a white solid: rf0.15 (silicon, CHCl)3MeOH/water 6/4/1);1H NMR(D2o, 500MHz) δ 1.82(t, J ═ 11.9, 1H), 2.21(dd, J ═ 13.76, 4.84, 1H), 2.23(s, 3H), 2.57(appq, J ═ 6.6, 2H), 2.86-2.95(m, 2H), 3.15-3.18(m, 1H), 3.28-3.61 (composite, 1H), 3.60(dd, J ═ 11.91, 6.66, 1H), 3.75(td, J ═ 6.65, 2.62, 1H), 3.84(dd, J ═ 11.89, 2.65, 1H), 3.88-4.01 (composite, 2H), 4.04(td, J ═ 11.18, 4.67, 1H), ms (es), (1H); to C14H23NO10365.33; measured value ([ M-1 ]]-),363.97。
Cytidine-5' -monophosphoryl- (5-acetyl propionamide-3, 5-dideoxy-beta-D-glycerol-D-galactose Sugar-2-nonylpyranosuronate).5-Acetylpropionamide-3, 5-dideoxy-D-glycero-D-galactose-2-nonylpyranosuronate (50mg, 137. mu. mol) was dissolved in 2mL of a buffer solution of 100mM HEPES (pH7.5), and 1M MnCl was added2(300. mu.L, 300. mu. mol). CTP-2Na+(79mg, 1.5. mu. mol) was dissolved in 5mL of HEPES buffer and added to the sugar. sialyltransferase/CMP-neuraminidase fusion enzyme (11U) was added and the reaction mixture was stirred at room temperature for 45 hours. The reaction mixture was filtered through a 10,000MWCO filter and the filtrate containing the reaction product was used without further purification: rf0.35 (silicon, IPA/water/NH)4OH7/2/1)。
B. Glycoconjugation and glycoPEGylation of peptides
Alpha-protease inhibitors (alpha-antitrypsin)
7. Sialylation of recombinant glycoprotein antithrombin III, fetuin and alpha 1-antitrypsin
This example presents the preparation of sialylated forms of several recombinant peptides.
Sialylation of recombinant glycoproteins with ST3Gal III.Several glycoproteins were examined for their ability to sialylate from recombinant rat ST3Gal III. For each of these glycoproteins, sialylation would be a valuable processing step in the development of the individual glycoprotein as a commercial product.
And (3) reaction conditions.The reaction conditions are summarized in table 11. The sialyltransferase reaction was carried out at room temperature and 37 ℃ for 24 hours. The degree of sialylation is determined by the incorporation into glycoprotein-linked oligosaccharides14The amount of C-NeuAc. See table 11 for reaction conditions for each protein.
TABLE 11 reaction conditions
1"cycling" refers to the "in situ" enzymatic generation of CMP-NeuAc using standard conditions (20mM NeuAc and 2mM CMP) as described in the specification. The buffer was 0.1M HEPES, pH 7.5.
The results presented in table 12 demonstrate that despite the low levels of enzyme applied, a significant degree of sialylation was achieved in each case. Essentially, complete sialylation was obtained based on an estimate of the available terminal galactose. Table 12 shows the results of the sialylation reaction. The amount of enzyme used per mg of protein (mU/mg) was used as a reference for comparison in each study. In several of the examples shown, only 7-13mU of ST3Gal III per mg of protein are required to achieve essentially complete sialylation after 24 hours.
TABLE 12 analytical results
| Protein | Source | Terminal Gal<sup>1</sup>Mole/mole | NeuAc integration<sup>2</sup>Mole/mole | %Rxn<sup>3</sup> | Other features |
| AT III<sup>4</sup> | GenzymeTransgenics | 102 | 104 | 117 | Is free of |
| AT III<sup>4</sup> | GenzymeTransgenics | 102 | 1.3 | 108 | SDS-gel: protein purity FACs: saccharide sugar forms |
| Desialylated fetuin | Sigma | 802 | 905 | 116 | Is free of |
| Desialylation of-AAAT<sup>5</sup> | |
7 | 7.0 | 100 | SDS-gel: purity of protein |
1Determined by the supplier or from the value of the documentThe amount of Gal (exposed) at the upper end of the N-linked oligosaccharide (fetuin, desialylated-AAAT).
2Integrated NeuAc determined by integration of 14C-NeuAc after separation from free radiolabeled precursor by gel filtration.
3% Rxn refers to the percentage of reaction completion based on the amount of terminal Gal at the theoretical maximum.
4Antithrombin III.
5 Alpha 1 antitrypsin.
These results are significantly in contrast to those reported in the detailed study with bovine ST6Gal I, in which less than 50mU/mg protein caused sialylation by less than 50% and 1070mU/mg protein caused sialylation by about 85-90% in 24 hours. Paulson et al (1977), j.biol.chem.252: 2363-; paulson et al (1978), j.biol.chem.253: 5617-5624. Studies of rat α 2, 3 and α 2, 6 sialyltransferases by another group revealed that complete sialylation of desialylated-AGP requires an enzyme concentration of 150-250mU/mg protein (Weinstein et al (1982) J.biol.chem.257: 13845-13853). Together, these early studies suggest that ST6Gal I sialyltransferase requires more than 50mU/mg and as much as 150mU/mg to achieve complete sialylation.
This example demonstrates that sialylation of recombinant glycoproteins with ST3Gal III sialyltransferase requires less enzyme than expected. For a one kilogram reaction, about 7,000 units of ST3GalIII sialyltransferase are required, rather than the 100,000-150,000 units shown in earlier studies. Purification of these enzymes from natural sources is impractical and yields of only 1-10 units are obtained after 1-2 months in large scale preparations. Assuming that both ST6Gal I and ST3Gal III sialyltransferases are produced as recombinant sialyltransferases and achieve the same expression levels of both enzymes, a 14-21 fold higher (or higher) fermentation scale is required for ST6Gal I sialyltransferase relative to ST3Gal III sialyltransferase. For ST6Gal I sialyltransferase, an expression level of 0.3U/1 in yeast has been reported (Borsig et al (1995) biochem. Biophys. Res. Commun.210: 14-20). Expression levels of 1000U/l of ST3Gal III sialyltransferase have been achieved in A.niger. At the present expression level, 300-450,000 liters of yeast fermentate would be required to produce enough enzymes to sialylate 1kg of glycoprotein with ST6Gal I sialyltransferase. In contrast, sialylation of 1kg of glycoprotein with ST3Gal III sialyltransferase requires less than 10 l of a. niger fermentate. Thus, the fermentation capacity required to produce ST3Gal III sialyltransferases for large scale sialylation reactions is 10-100 times lower than that required to produce ST6Gal I; the cost of producing sialyltransferase will be proportionally reduced.
Cri-IgG antibodies
Glycoremodeling of Cri-IgG1 antibody
This example presents a method for in vitro reconstitution of Cri-IgG1 antibody.
N-glycosylation at a conserved site of Asn297 in the Fc domain of monoclonal antibodies can modulate its pharmacokinetic properties and effector function (Dwek et al, 1995, J.Anat.187: 279-292; Boyd et al, 1995, mol.Immunol.32: 1311-1318; Lund et al, 1995, FASEB J.1995, 9: 115-119; Lund et al, 1996, J.Immunol.157: 4963-4969; Wright & Morrison, 1998, J.Immunol.160: 3393-3402; Flynn & Byrd, 2000, Curr. Opin. Oncol.12: 574-581). During cell culture fermentation or certain pathological conditions, significant heterogeneity is created in the glycosylation pattern at this site. The resulting differential glycosylation patterns on the Fc domain are characterized by complex biantennary structures with 0, 1 and 2 terminal galactose residues (G0, G1 and G2, respectively, see table 13). Observed glycoform variations, such as those in terminal galactosylation, truncated N-glycoforms, and modifications of aliquots, have been shown to affect the therapeutic properties of antibodies, particularly their ability to modulate target cell killing through complement binding and activation (Boyd et al, 1995, supra; Wrght & Morrison, 1998, supra; Mimura et al, 2000, molecular. Immunol.37: 697-one 706; Davies et al, 2001, Biotechnol.Bioeng.74: 288-one 294).
To obtain the different glycoforms of Cri-IgG1 antibody and to examine its Fc effector function, the Cri-IgG1 antibody was gradually trimmed using exoglycosidases to generate a glycoform lacking sialic acid (G2, G1), a glycoform lacking sialic acid and galactose (G0), and a glycoform lacking sialic acid, galactose and N-acetylglucosamine (M3N2F), as shown in table 13. These molecules are subsequently modified with different glycosyltransferases and appropriate sugars. The modification conditions were developed to result in the conversion of the original antibody glycan structure to a different glycoform: M3N2, GnT-I-M3N2 (using the M3M2 form of GnT-I with the GlcNAc moiety added), G0, aliquot-G0 (the G0 form of GnT-III with the GlcNAc moiety added in aliquot), galactosylated aliquot-G0 (the G0 glycoform with the terminal galactose moiety added), G2, monosialylated S1(α 2, 6) -G2 (the G2 glycoform with a terminal sialic acid moiety added with α 2, 6-sialyltransferase), S1(α 2, 3) -G2 (the G2 glycoform with a terminal sialic acid moiety added with α 2, 3-sialyltransferase), and desialylated S2(α 2, 3) -G2 (the G2 glycoform). After each sugar reconstitution step, glycan structures are enzymatically released from the antibody protein and analyzed by various methods including separation by capillary electrophoresis, 2-AA HPLC characterization, and MALDI-TOF mass spectrometry.
TABLE 13 abbreviations for the saccharide form structures
Diamond-fucose □ GlcNAc, mannose ● galactose
The materials and methods used in these experiments are now described.
The Cri-IgG1 monoclonal antibody.Cri-IgG1 antibody was obtained from R.Jefferies (MRCCenter for Immune Regulation, TheMedical School, University of birmingham, uk). The antibody is a non-recombinant antibody and is isolated from human myeloma. Antibodies were prepared using three methods. In the first method, which is called "DEAE", antibodies are separated under relatively mild conditions using a DEAE ion exchange column. In the second method, referred to as "SPA", the antibody is purified on a protein a column (Staphylococcus aureus protein a) with an elution step at low pH. In a third method, referred to as "Fc", the antibody is treated with a protease, thereby retaining only the Fc portion of the antibody, while removing the antibody binding domain. These antibody purification methods are well known to those skilled in the art and are not repeated in detail herein.
Affinity purification of reconstituted antibody.Antibodies modified by exoglycosidase or glycosyltransferase were purified on a ProA-Sepharose 4-fast flow column (Amersham Bioscience, Arlington Heights, IL), eluted with 0.1M glycine-HCl buffer (pH2.7), and immediately neutralized with 1M Tris pH 9.5. The eluate was subjected to buffer exchange using NAP-10 column (Amersham Bioscience, Arlington Heights, IL) and replaced with an appropriate buffer for the next glycosylation, such as 100mM MES pH6.5 or 50mM Tris-HCl pH 7.2. At 4 ℃ in Tube-O-diamizers TMThe reconstituted final product was extensively dialyzed against PBS (Chemicon International, Temecula, Calif.) at a MWCO of 8 kDa.
In vitro glycosidase treatment of Cri-antibodies.The antibody was buffer exchanged with 50mM sodium phosphate/citrate pH6.0 using a NAP-10 column (Amersham bioscience, Arlington Heights, IL). The antibody (5mg/mL) was gradually in vivo 2 by contacting 20mU/mg protein neuraminidase overnight at 37 ℃ (to remove terminal sialic acid moieties), 20mU/mg protein beta-galactosidase overnight at 37 ℃ (to remove terminal galactose moieties, resulting in the G0 saccharide form), and/or 2mU/mg beta-N-acetylhexosaminidase (from Canava, Seikagaku, Tokyo, Japan) overnight at 37 ℃ (to remove terminal N-acetylglucosamine, resulting in the M3N2 saccharide form) to gradually in vivoThe sugar portion was trimmed externally. The samples were affinity purified as described above.
In vitro glycosylation of Cri-antibodies.In vitro GnT1 modification was performed using 1mg/ml M3N2 glycoform antibody as substrate, 25mU/mg recombinant human β 1, 2-mannosyl-UDP-N-acetylglucosaminyltransferase in 100mM MES, pH6.5, 5mM MnCl25mM UDP-GlcNAc and 0.02% NaN 3At 32 ℃ for 24 hours. Aliquots were taken for glycan analysis and the resulting product was affinity purified as described above.
In vitro modification of the aliquot-sugar form was performed using 1mg/ml M3N2 sugar form antibody as substrate, 25mU/mg β 1, 2-recombinant human mannosyl-UDP-N-acetylglucosaminyltransferase I, 25mU/mg β 1, 2-recombinant human mannosyl-UDP-N-acetylglucosaminyltransferase II, and 3.5mU/mg β 1, 4-recombinant mouse mannosyl-UDP-N-acetylglucosaminyltransferase III, MES at 100mM pH6.5, 10mM MnCl25mMUDP-GlcNAc and 0.02% NaN3At 32 ℃ for 24 hours. Aliquots were taken for glycan analysis and the remaining product was affinity purified as described above.
In vitro galactosylation was performed using G0 glycoform antibody or an aliquot of the glycoform antibody by contacting the antibody with 0.6U/mg recombinant milk beta 1, 4-galactosyltransferase in Tris-HCl, 150mM NaCl, 5mM UDP-galactose, 5mM MnCl at 50mM pH7.42At 32 ℃ for 24 hours. Aliquots were taken for glycan analysis and the remaining product was affinity purified as described above.
In vitro sialylation using the G2 glycoform antibody (1mg/ml) was carried out by contacting the antibody with 0.1U/mg ST3Gal3 or 0.1U/mg ST6Gal1, 5mM CMP-sialic acid in 50mM Tris pH7.4, 150mM NaCl and 3mM CMP-SA buffer at 32 ℃ for 24 hours. Aliquots were taken for glycan analysis and the remaining product was affinity purified as described above.
Glycan analysis:
capillary electrophoresis with laser-induced fluorescence detection.By dilution and mixing in MicroconTMConcentration on YM-30 microconcentrators (Millipore, Bedford, Mass.) removes buffer components and nucleotide sugars from aliquots of sugar reconstituted antibodies. The N-linked oligosaccharides were released from the protein by contacting the protein with PNGase F (Prozyme, San Leandro, Calif.) using the method provided by the manufacturer. Briefly, samples were denatured at 100 ℃ for 10 minutes in a buffer of 50mM sodium phosphate pH7.5, 0.1% SDS, and 50mM β -mercaptoethanol. TX100 to 0.75% (v/v) and 10U PNGase F/200. mu.g protein were then added. After 3 hours incubation at 37 ℃, the proteins were ethanol precipitated and the supernatant was dried. The released free oligosaccharides were then labeled with 8-aminopyrene-1, 3, 6-trisulfonic acid and analyzed by capillary electrophoresis using a carbohydrate labeling and analysis kit available from Beckman-Coulter, Inc. (Fullerton, Calif.), as shown by the manufacturer (see also Ma and Nashabeh, 1999, anal. chem.71: 5185-.
Capillary Electrophoresis (CE) on eCAPTMN-CHO coated capillaries (50 μm I.D., length to detector 40 cm; Beckman-Coulter, Inc., Fullerton, Calif.) were carried out using P/ACE with laser induced fluorescence detector (Beckman-Coulter, Inc. Fullerton, Calif.)TMThe MDQ glycoprotein system (Beckman-Coulter, Inc. Fullerton, Calif.). Through 20 lb/in2The sample was introduced into the column under pressure for 10 seconds and then separated with reverse polarity at 25kV for 20 minutes. The column temperature was controlled at 20 ℃. The electropherograms were generated by laser-induced fluorescence detection at an excitation wavelength of 488nm and an emission wavelength of 520 nm.
Saccharide standards (a)EMD Biosciences, Inc., San Diego, Calif.) includes M3N2 (an N-linked trimannose core of coreless fucose),G0 (asialo, biantennary N-linked oligosaccharides with core fucose), G2 (asialo, biantennary N-linked oligosaccharides with core fucose) and afucose G2, S1-G2 (monosialylated, galactosylated biantennary oligosaccharide with no core fucose) and S2-G2 (disialylated, galactosylated biantennary oligosaccharide with no core fucose) (available from Glyko, see Prozyme, San leindro, CA), M3N2F (N-linked trimannose core with core fucose) and NGA2F (desialylate, degalactose, biantennary with core fucose and N-linked oligosaccharides with equally divided GlcNAc) these carbohydrate standards were labeled with 1-aminopyrene-3, 6, 8-trisulfonate (APTS, Beckman-Coulter, inc., Fullerton, CA) and used to identify the distribution of glycans released from the antibody.
2-AA HPLC.PNGase F-released glycans were labeled with 2-AA (2-anthranilic acid) according to a slightly modified method described by Anumula and Dhume (1998, Glycobiology 8: 685-694). Reductively aminated N-glycans were analyzed on a Shodex Asahipak NH 2P-504D amino column (4.6mm x 150mm) (Showa Denko K.K., Tokyo, Japan). Two solvents used for the separation were a) 2% acetic acid and 1% tetrahydrofuran in acetonitrile, and B) 5% acetic acid, 3% triethylamine and 1% tetrahydrofuran in water.
To isolate 2-AA labeled neutral glycans, the column was isocratically eluted with 70% a for 5 minutes, followed by a linear gradient from 70% to 50% B over a 60 minute period, followed by a steep gradient from 50% to 5% B over a 5 minute period, and finally isocratically eluted with 5% B for 10 minutes. The eluted peak was detected by fluorescence detection at 230nm excitation and a detection wavelength of 420 nm. Under this gradient condition, the G0 saccharide form will elute at about 30.5 minutes, the G1 saccharide form will elute at about 34.0 minutes, and the G2 saccharide form will elute at about 37.0 minutes. Under these conditions, the presence of fucose did not alter the elution time.
To isolate the 2-AA labeled anionic glycans, the column was eluted isocratically with 70% a for 2.5 minutes, followed by a linear gradient from 70% to 5% a over a 97.5 minute period, and finally eluted isocratically with 5% a for 15 minutes. The eluted peak was detected by fluorescence detection with excitation at 230nm and detection at 420 nm. Under this gradient condition, neutral glycans are expected to elute between 18.00-29.00 minutes, glycans with one charge are expected to elute between 30.00-40.00 minutes, glycans with two charges are expected to elute between 43.00-52.00 minutes, glycans with three charges are expected to elute between 54.00-63.00 minutes, and glycans with four charges are expected to elute between 65.00-74.00 minutes.
MALDI analysis of reductively aminated N-glycans.A small aliquot of PNGase-released N-glycans labeled with 2-anthranilic acid (2AA) was dialyzed for 45 minutes on an MF-Millipore membrane filter (0.025 μm pores, 47mm diameter) which was floated on water. Dialyzed aliquots were placed in a SpeedvacTM(ThermoSavant, Holbrook, NY), redissolved in a small amount of water and mixed with a solution of 2, 5-dihydroxybenzoic acid (10g/L) dissolved in water/acetonitrile (50: 50).
The mixture was dried on a MALDI target and analyzed using a Biosystems DE-Pro mass spectrometer (Applied Biosystems, inc., foster city, CA) operating in linear/negative ion mode. The structure of the oligosaccharides was determined based on the observed mass-to-charge ratios and previous literature. No attempt was made to adequately characterize the isobaric structure.
SDS-PAGE.To determine the stability of the glycoreconstituted antibodies, all samples were analyzed by SDS-PAGE. The final product of the sample was electrophoresed using 8-16% Tris-glycine gel (Invitrogen, Carlsbad, Calif.) under non-reducing conditions. Bovine serum albumin was electrophoresed under reducing conditions as a quantitative standard. The gel was stained with GelCode blue stain Reagent (Pierce Chemical co., Rockford, IL) for color development.
The results of the experiment are now described.
A native glycoform of Cri expressed in human myeloma cells.From suffering fromThe purified Cri-IgG1 antibody contained a variable sugar form in serum from patients with multiple myeloma. FIGS. 97A-97C show HPLC profiles of enzymatically released glycans from Cri-IgG1 antibody. FIGS. 98A-98C show MALDI curves of glycans enzymatically released from Cri-IgG1 antibody expressed in human myeloma cells. The major forms are G0, G1 that are under galactosylated, whereas G2 and sialylation structures are relatively minor (table 14 and fig. 97C). To examine the effect of modified glycans on the therapeutic properties of monoclonal antibodies, the Cri-IgG1 antibody was modified by performing in vitro exoglycosidase trimming and in vitro glycosylation reconstruction to generate different glycoforms of the antibody.
Table 14. the relative amounts of the different glycoforms of Cri-IgG1 expressed by human myeloma cells isolated by HPLC were calculated from the area of the respective peaks.
Initially, the optimization of each step in exoglycosidase trimming and glycosylation was performed on a small scale (100. mu.g each).
The trimannose core sugar form of the Cri-IgG1 antibody (M3N2).M3N2 was generated by stepwise treatment with glycosidases including neuraminidase, β 1, 4-galactosidase, and β 1-2, 3, 4, 6N-acetylhexosaminidase. To estimate the removal of terminal galactose and GlcNAc on the sugar reconstituted Cri-IgG1 antibody sample, a quantitative Capillary Electrophoresis (CE) method was applied. Glycans were enzymatically released from glycoreconstituted antibodies with PNGase F and derivatized at the reducing end with 8-aminopyrene-1, 3, 6-trisulfonic Acid (APTS). Results the resulting product was analyzed by CE with laser induced fluorescence detection (LIF) on column (Ma) &Nashabeh, 1999, supra). Since separation of glycans is based on differences in hydrodynamic size, APTS-labeled glycans migrate in order of increasing size (M3N 2)<M3N2F<G0<G1<G2)。
FIGS. 99A-99D show electropherograms of glycans released from saccharide reconstituted Cri-IgG1 antibody and glycan standards derivatized with APTS (FIG. 99A). The glycoform is identified by comparing its electrophoretic mobility to a standard. The relative amounts of each glycan species were calculated from the relative area percentages of each peak shown, and the results are shown in table 15. The M3N2F glycoform represents 91% DEAE-Cri glycan, 80% SPA-Cri glycan and 100% Fc-Cri glycan. Incomplete removal of the GlcNAc moiety, which leads to the sugar form of GnT-I-M3N2F, was observed in glycan structures from DEAE-Cri (8.6%) and SPA-Cri (. about.20%) (see Table 15). The glycoform GnT-I-M3N2F is a M3N2F glycoform with an additional GlcNAc, as can be added by GnT-I.
Table 15. the area of the individual peaks of the CE curve in FIG. 99 was calculated and the relative amounts of the M3N2F and GnT-I-M3N2F glycoforms were determined.
A degalactosylated saccharide form (G0).The Cri-IgG1 antibody with the G0 glycoform was obtained by stepwise treatment of the native Cri-IgG1 antibody with neuraminidase and β 1, 4-galactosidase, each reaction was performed for 24 hours. Glycans released from the antibody reconstituted from the saccharide were analyzed by CE, HPLC and MALDI. Fig. 100A shows the CE curve of released glycans. In all three samples, only one peak was observed, which was designated as the G0 glycoform based on comparison to the standard (fig. 100A and table 16).
TABLE 16 relative amounts of Cri-IgG 1G 0 glycoforms as determined by CE and HPLC.
In addition to glycan analysis provided by CE, quantitative HPLC methods were also used to determine the percentage of G0 glycoforms represented by the reconstituted glycans of Cri-IgG1 antibody. Glycan distribution on glycoreconstructive antibodies was monitored by enzymatic release of glycans with PNGase F and derivatization of the released products at the reducing end with 2-anthranilic acid (2-AA). The derivatized mixture was separated by HPLC on a Shodex Asahipak NH 2P-504D column with fluorescent detection. FIGS. 101A-101C show chromatograms obtained from released glycans. Since only one major peak was found in all three samples, the HPLC results confirmed the CE analysis. Consistent with the CE and HPLC data, MALDI analysis also showed almost complete reconstitution of the G0 saccharide form (fig. 102A-102C).
The fully galactosylated form of G2 sugar (G2).Treatment of Cri-IgG antibodies with neuraminidase to obtain the asialo glycoform, which is also under galactosylated. These asialo glycoforms were then treated with 0.6U/ml bovine β 1, 4 galactosyltransferase and galactose donor molecules to reconstitute the antibody glycoforms to have the G2 glycoform.
The degree of terminal galactosylation was determined by glycan analysis. Only one major peak was observed in the CE and HPLC curves (FIGS. 103A-103C and 104A-104C). This peak corresponds to the G2 saccharide form in each case. Calculation of the percent total peak area showed almost complete (-90%) conversion from the galactosylated under-saccharide form of the original sample to G2 (see table 14). These results are summarized in table 17. MALDI analysis of glycans further supported the almost complete reconstitution of the saccharide to the G2 saccharide form in all samples (fig. 105A-105C).
TABLE 17 relative amounts of the glycoforms of the reconstituted Cri-IgI1 antibody G2 as determined by percentage total peak area in CE and HPLC analysis.
GnT-I-sugar form (GnT-I-M3N2).The M3N2 glycoform Cri-IgG antibody glycoform is reconstituted into the GnT-I-M3N2 glycoform by adding a GlcNAc moiety to the molecule. This molecule is then conjugated with 25mU GnT-I/mg antibody and appropriateA GlcNAc donor molecule. CE, HPLC, and MALDI analysis of the released glycans (FIGS. 106A-106D, 107A-107C, and 108A-108C, respectively) showed that the original M3N2F glycoform was completely reconstituted. However, only 40-60% of the modified structures are the GnT-I-M3N2 glycoforms, and about 30% are the G0 glycoforms. The presence of the G0 sugar form may be the result of incomplete GlcNAc trimming when preparing the original M3N2 form.
The saccharide form (NGA2F) was aliquoted.The M3N2 glycoform Cri-IgG antibody is reconstituted into the NGA2F glycoform by contacting its glycoform with a combination of three transferases GnT-I, GnT-II and GnT-III and an appropriate N-acetylglucosamine donor molecule. The reaction was completed within 24 hours. To determine the extent of addition of the aliquot-GlcNAc moieties to the glycans, CE analysis was used to determine the glycoforms present on the glycoreshaped antibody.
FIGS. 109A-109D show electrophoretograms obtained from CB analysis of glycans released from saccharide reconstituted Cri-IgG1 antibody. After reconstruction 4 peaks appear. One major peak migrates with the same retention time as the NGA2F standard sugar form. The 3 other minor peaks may be incompletely reconstructed glycans. For comparison, a quantitative HPLC method was also applied, in which 2-AA labeled glycans were eluted in order of increasing size (Gn1< G0< NGA2F). Similar results were obtained from CE analysis of glycans, as shown in FIGS. 110A-110C. Neither CE nor HPLC analysis revealed M3N 2F. NGA2F glycan was the major peak for CE and HPLC analysis. The Gn1 and G0 glycans that remain in the sample may be incompletely modified results. Most of the original M3N2F sugar forms were reconstituted as the NGA2F sugar form by 3 GlcNAc moieties (60-70%), about 15-18% were reconstituted as the G0 sugar form by the addition of 2 GlcNAc moieties, and only a small amount (-7%) were reconstituted by the addition of only one GlcNAc moiety. MALDI analysis of the released glycans (fig. 111A-111C) showed peaks of glycoforms with 1, 2, or 3 terminal GlcNAc moieties, consistent with CE and HPLC analysis (fig. 109 and 110). The relative amounts of each glycan species were calculated from the relative area percentages of each peak shown and are summarized in table 18.
TABLE 18 relative amounts of different glycoforms of Cri-IgG1 from GnT-I, II and III reconstitution, as determined by CE and HPLC.
Galactosylated aliquot (Gal-NGA2F) of the sugar form.The NGA2F glycoform of the Cri-IgG1 antibody was glycoreconstituted with bovine β 1, 4-galactosyltransferase and an appropriate galactose donor. The terminal galactose moiety was added using 0.6U/ml β 1, 4-galactosyltransferase. FIGS. 112A-112D show electrophoretograms obtained using the 2-AA HPLC method. Briefly, the sugar forms terminating in GalNAc were almost 100% galactosylated. Comparing FIGS. 112A-112B of DEAE Cri-IgG1 with FIGS. 112C-112D of Fc Cri-IgG1, the 2-AA HPLC profiles (FIGS. 112A and 112C) of GnT-I, II and III modified glycans were modified with GalT1, so that the peaks of all glycans became eluted later due to the increase in size after the addition of the galactose moiety (FIGS. 112B and 112D). These results were further confirmed by MALDI-MS analysis.
Sialylated (S2G2) saccharide form of Cri-IgG 1.Sugar reconstitution of Cri-IgG1 antibody G2 the sugar form was further reconstituted using ST3Gal3 and ST6Gal 1. FIGS. 113A-113C show HPLC profiles of the G2 glycoform reconstituted with ST3Gal 3. Most of the G2 saccharide forms were converted to the S2G2 saccharide form (G2 saccharide form with 2 additional terminal sialic acid moieties; -70%, see Table 19), with only a minor amount being the S1G2 saccharide form (G2 saccharide form with 1 additional terminal sialic acid moiety; <25%, see table 19). These results were further confirmed by MALDI analysis shown in FIGS. 114A-114C. MALDI data also showed that all G2 glycoforms were sialylated to the S2G2 or S1G2 glycoforms.
TABLE 19 relative amounts of different glycoforms of Cri-IgG1 reconstituted from ST3Gal3 as determined by HPLC.
By comparison, ST6Gal1 reconstitution of the G0 glycoform failed to reach the full level found in ST3Gal3 reconstitution. FIGS. 115A-115D and FIGS. 116A-116C show results obtained from CE and HPLC analysis, respectively. The S2G2 saccharide form was not found in any of the saccharide reconstituted samples. However, all of the G2 saccharide forms were converted to S1-G2. MALDI-MS analysis also supported these data (FIGS. 117A-117C).
Stability of the reconstituted glycan of Cri-IgG 1.Finally, the stability of Cri-IgG1 glycans, reconstituted by exoglycosidase treatment and glycosylation, was investigated. The Cri-IgG1 antibody reconstituted for each saccharide was stored at 4 ℃ and checked for degradation by SDS-PAGE 2 weeks after reconstitution. As shown in fig. 118A-118E, both the reconstituted DEAE and SPA antibodies retained a molecular weight of about 150kDa, showing little to no degradation, regardless of the type of sugar reconstitution performed. The Fc Cri-IgG1 antibody retained a molecular weight of about 38kDa, and also showed little to no degradation, regardless of the type of reconstitution performed.
Biological assay of effector function of reconstituted Cri-IgG1 antibody.The effector function bioassay was derived from the method of Mimura et al (2000, Molecular Immunology 37: 697-706). IC of the glycoform of the Cri-IgG1 antibody50Is determined by inhibiting the superoxide response of U937 cells by erythrocytes sensitized by natural anti-NIP antibodies.
U937 cells of single cells were cultured for 2 days in the presence of 1000 units/mL interferon γ to induce cell differentiation and their capacity to produce superoxide. Cells were then washed and washed at 2 x106cells/mL were resuspended in Hanks balanced salt solution without phenol red, containing 20mM HEPES pH7.4 and 0.15mM BSA. Erythrocytes were sensitized with anti-NIP (5-iodo-4-hydroxy-3-nitrophenylacetyl) antibody at 37 ℃ for 30 minutes in the absence or presence of the various glycoforms of Cri-IgG1 antibody. Cells were then washed 3 times with PBS and washed 2.5 x107cells/mL were resuspended in HBSS-BSA. U937 cells (100. mu.l, 2X 10)6CellsmL) was added to a plastic tube, and bis-N-methylacridine nitrate (20 μ l, 2.5mM) was added to the tube. The tube was warmed in a 37 ℃ water bath for 5 minutes. Sensitized erythrocytes (80. mu.l, 2.5X 10) 7cells/mL) was added to the tube. Superoxide anion production was measured by enhanced chemiluminescence with bis-N-methylacridine nitrate using a Berthold LV953 luminometer (Berthold Australia Pty Ltd, Bungiora, Australia) at 37 ℃ over a 30 minute time period.
The G0 and M3N2 glycoform Cri-IgG1 antibodies had relative inhibition values of 92% and 85%, respectively, compared to the native antibody. However, the native Cri-IgG1 antibody lacks core fucose. Shield et al (2002, J.biol.chem.277: 26733-26740) suggested that the lack of core fucose improved the inhibition value by 10-fold. Based on these results, it is expected that the inhibition value of galactosylated-bisected-G0 saccharide forms will be higher than for bisected-G0 saccharide forms, bisected-G0 saccharide forms will be much higher than for G2 saccharide forms, G2 saccharide forms will be approximately equal to the disialylated-G2 saccharide forms and monosialylated-G2 saccharide forms, disialylated-G2 saccharide forms and monosialylated-G2 saccharide forms will be higher than for native antibody saccharide forms, native antibody saccharide forms will be higher than for G0 saccharide forms, and G0 saccharide forms will be higher than for M3N2 saccharide forms.
Complement receptor-1
9.Sialylation and fucosylation of TP10
This example presents the preparation of TP10 with a sialic acid Lewis X moiety and analysis of enhanced biological activity.
Even a short interruption of blood flow into the brain can trigger inflammatory events in the brain microvascular structures, which exacerbate brain tissue damage. The resulting tissue damage can be augmented by activation of the inflammation and coagulation cascade. In a murine model of stroke, increased expression of P-selectin and ICAM-1 can promote leukocyte recruitment. sCR-1 is a recombinant form of the extracellular domain of complement receptor-1 (CR-1). sCR-1 is a potent inhibitor of complement activation. sCR1sLeX(CD20) is an alternative to sCR1(ii) a form of sexual glycosylation alternatively glycosylated to exhibit sialylated LewisXAn antigen. Previously, sCR-1sLeX, expressed and glycosylated in engineered Lec11 CHO cells in vivo, was found to be located in ischemic cerebral microvasculature and in neurons expressing C1q, thereby inhibiting neutrophil and platelet accumulation and reducing cerebral infarct volume (Huang et al, 1999, Science 285: 595-599). In this example, sCR1sLe prepared in vitro by glycan reconstitutionXsCRsLe exhibiting glycosylation in vivoXSimilar enhanced biological activity.
The TP10 peptide was expressed in BUK B11 CHO cells. The CHO cell line produced TP10 peptide with typical CHO cell glycosylation, which has many, but not all, glycans capped with sialic acid.
Sialylation of 66mg TP 10.TP10(2.5mg/mL), CMPSA (5mM), and ST3Gal3(0.1U/mL) were incubated in 50mM Tris, 0.15M NaCl, 0.05% sodium azide, pH7.2 at 32 ℃ for 48 hours. Radiolabeled CMP sialic acid was added to a small aliquot to monitor integration. TP10 was isolated from the nucleotide sugar by SEC HPLC. Samples analyzed at 24 hours and 48 hours demonstrated that the reaction was complete after 24 hours. The reaction mixture was then frozen. Subjecting the reaction product to Fluorophore-assisted saccharide Electrophoresis (Fluorophore assay carbohydrate Electrophoresis) (II)Glyko, Inc, Novato CA) (FIG. 119).
Pharmacokinetic studies.Rats were purchased with jugular vein cannulae. 10mg/kg of the pre-sialylated or post-sialylated TP10 peptide was administered by tail vein injection to 3 rats (n-3) per group treated. 14 blood samples were obtained from 0-50 hours. After 0 hours at each time point, the concentration of TP10 peptide was higher in the blood after sialylation than TP10 before sialylation (fig. 120). Sialic acid addition doubled the area under the plasma concentration-time curve (AUC) of the pharmacokinetic profile compared to the starting material (FIG. 1) 21)。
Fucosylation of sialylated TP 10.10mL (25mgTP10) of the above sialylated mixture was thawed and GDP-fucose was added to 5mM, MnCl2To 5mM, and FTVI (fucosyltransferase VI) to 0.05U/mL. The reaction was incubated at 32 ℃ for 48 hours. Subjecting the reaction product to Fluorophore-Assisted saccharide electrophoresis (Fluorophore Assisted carbohydrate electrophoresis) (II)Glyko, Inc, Novato CA) (FIG. 122). Radiolabelled GDP-fucose was added to a small aliquot to monitor integration. TP10 was isolated from the nucleotide sugar by SEC HPLC. Samples analyzed at 24 hours and 48 hours demonstrated that the reaction was complete at 24 hours. In vitro assays measuring binding to E-selectin show: addition of fucose produced a biologically active E-selectin ligand (fig. 123).
Enbrel
TM
10.AntibodiesEnbrelTMIs/are as followsPEGylation of sugars
This example presents a PEGylation procedure for O-linked glycans of antibody molecules. Here, Enbrel is usedTMAs an example, however, one skilled in the art will appreciate that the present procedure is applicable to a wide variety of antibody molecules.
Enbrel TM Preparation of SA-PEG (10 kDa).The O-linked glycans will be sialylated or unsialylated Enbrel prior to PEGylationTM(TNF-receptor-IgG)1Chimera) was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 5mM MnCl 2、0.05% NaN3pH 7.2. The solution was incubated with 5mM UDP-galactose and 0.1U/mL galactosyltransferase at 32 ℃ for 2 days to cap the hypolactosylated glycans with galactose. To monitor the incorporation of galactose, small aliquots of the reactants were added14C-galactose-a UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration in methanol and water using a Toso Haas G2000SW analytical column. The radiolabel incorporated into the peptide was quantified using an in-line radioactive detector.
When the reaction was complete, the solution was incubated with 1mM CMP-sialic acid-linker-PEG (10kDa) and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor the integration of sialic acid-linker-PEG, peptides were isolated by gel filtration on Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). When the reaction was completed, the reaction mixture was purified with a Toso Haas TSK-Gel-3000 preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The fractions containing the product were combined, concentrated, buffer exchanged, and then lyophilized. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Erythropoietin (EPO)
11.Addition of GlcNAc to EPO
This example shows the addition of a GlcNAc residue to the trimannose core.
Addition of GlcNAc to EPO.EPO is expressed and purified in SF-9 insect cells (Protein Sciences, Meriden, CT). 100% conversion from the trimannose form of Epo to "trimannose core +2 GlcNAc" (Peak 1, P1 in FIG. 124) was achieved by incubation with 100mU/ml GlcNAcT-I and 100mU/ml GlcNAcT-II at the following final reaction concentrations for 24 hours at 32 ℃:
100mM MES pH6.5, or 100mM Tris pH7.5
5mM UDP-GlcNAc
20mM MnCl2
100mU/ml GlcNAcT-I
100mU/ml GlcNAcT-II
1mg/ml EPO (purified, expressed in Sf9 cells, purchased from Protein Sciences).
Analysis of the sugar form.This assay is a minor modification of K-R Anumula and ST Dhume, Glycobiolgy8(1998) 685-69. The N-glycans released by the N-glycanase (PNGase) are reductively labeled with anthranilic acid. Reductively aminated N-glycans were injected onto ShodexAsahipak NH 2P-504D amino columns (4.6 mm. times.150 mm). Two solvents were used for the separation: A) 5% (v/v) acetic acid, 1% tetrahydrofuran and 3% triethylamine dissolved in water; and B) 2% acetic acid and 1% tetrahydrofuran in acetonitrile. The column was then isocratically eluted with 70% B for 2.5 minutes, followed by a linear gradient from 70% to 5% B over a 97.5 minute period, and finally isocratically eluted with 5% B for 15 minutes. The eluted peak was detected with a fluorescence detector with an excitation wavelength of 230nm and an emission wavelength of 420 nm.
Under these conditions, the trimannose core had a retention time of 22.3 minutes and the product of the GnT reaction had a retention time of 30 minutes. The starting materials were all trimannose cores with a core GlcNAc (fig. 124).
12. Preparation of EPO with multiantennary complex glycans
This example presents the preparation of pegylated, biantennary EPO and triantennary sialylated EPO from insect cell-expressed EPO.
Glycan analysis was performed on recombinant human erythropoietin (rhEPO) from the baculovirus/Sf 9 expression system (Protein Sciences corp., Meriden, CT) and the resulting glycans showed a predominantly trimannose core with a core fucose, with only a small percentage of glycans also having a single GlcNAc.
N-acetylglucosamine was added using GnT-I and GnT-II.Two batches of rhEPO (1mg/ml) were combined with GnT-I and GnT-II, 5mM UDP-GlcNAc, 20mM MnCl2And 0.02% sodium azide in 100mM MES pH6.5 at 32 deg.CIncubate for 24 hours. Batch A contained 20mg EPO and 100mU/mL GnT-I and 60mU/mL GnT-II. Batch B contained 41mg EPO and 41mU/mL GnT-I +50mU/mL GnT-II. After the reaction, the sample was desalted by gel filtration (PD10 column, Pharmacia LKB Biotechnology inc., Piscataway, NJ).
EPO glycans analyzed by 2-AA HPLC profiling.The assay is a minor modification to Anumula and Dhume, Glycobiology 8(1998) 685-69. The reductively aminated N-glycans were injected onto a Shodex Asahipak NH 2P-504D amino column (4.6 mm. times.150 mm). Two solvents were used for the separation: A) 5% (v/v) acetic acid, 1% tetrahydrofuran and 3% triethylamine dissolved in water; and B) 2% acetic acid and 1% tetrahydrofuran in acetonitrile. The column was then isocratically eluted with 70% B for 2.5 minutes, followed by a linear gradient from 70% to 5% B over a 100 minute period, and finally isocratically eluted with 5% B for 20 minutes. The eluted peak was detected by fluorescence with an excitation wavelength of 230nm and an emission wavelength of 420 nm. The non-sialylated N-linked glycans were in the LC range of 23-34 minutes, the mono-sialylated ones were in 34-42 minutes, the di-sialylated ones were in 42-52 minutes, the tri-sialylated ones were in 55-65 minutes, and the tetra-sialylated ones were in 68-78 minutes.
Glycan characterization by 2AA HPLC revealed 92% conversion in lot a to a bi-antennary structure with two GlcNAc (the remaining (balance) has a single GlcNAc). Batch B showed 97% conversion to the desired product (fig. 125A and 125B).
A third antenna branch was introduced with GnT-V.Product EPO (1mg/mL batch B) from the reaction of GnT-I and GnT-II was desalted on a PD-10 column and subsequently concentrated and incubated with 10mU/mLGnT-V and 5mM UDP-GlcNAc in 100mM MES pH6.5 containing 5mM MnCl for 24 hours at 32 deg.C2And 0.02% sodium azide. 2AA HPLC analysis demonstrated that the transition occurred at 92% efficiency (FIG. 126).
After desalting (PD-10) and concentration, galactose was added with rGalTI: namely EPO (1mg/ml) and 0.1U/ml GalT1, 5mM UDP-galactose, 5mM MnCl2Incubation at 32 deg.CFor 24 hours.
MALDI analysis of reductively aminated N-glycans from EPO.A small aliquot of the N-glycan released from EPO with PNGase, which had been reductively labeled with anthranilic acid, was dialyzed for 45 minutes on MF-Millipore membrane filters (0.025 μm pores, 47mm diameter) floating on water. The dialyzed aliquot was dried in a vacuum centrifugal evaporator concentrator, redissolved in a small amount of water, and mixed with a solution of 2, 5-dihydroxybenzoic acid (10g/L) dissolved in water/acetonitrile (50: 50). The mixture was dried on the target and analyzed with an Applied Biosystems DE-Pro MALDI-TOF mass spectrometer operating in linear/negative ion mode. Oligosaccharides were determined based on the observed mass-to-charge ratios and previous literature.
Analysis of the released glycans by MALDI showed that galactose was quantitatively added to all available sites (fig. 127). The EPO derived from the galactosylation described above was then purified by gel filtration on a Superdex1.6/60 column in 50mM Tris, 0.15M NaCl, pH 6.
And (4) sialylation.After concentration and desalting (PD-10), 10mg galactosylated EPO (1mg/mL) was incubated with ST3Gal3(0.05U/mL) and CMP-SA (3mM) in 50mM Tris, 150mM NaCl, pH7.2 containing 0.02% sodium azide. Separate aliquots contained radiolabeled CMP-SA. The resulting integrated and free labels were separated by isocratic size exclusion chromatography/HPLC in 45% MeOH, 0.1% TFA at 0.5 mL/min (7.8mm X30 cm column, particle size 5 μm, TSK G2000SWXLToso Haas, Ansys Technologies, Lake Forest, CA). Using this procedure, 12% of the population (360 micromolar at 33 micromolar EPO, or about 10.9 moles/mole) was integrated. The theoretical value (3N-linked sites, triantennary) is about 9 moles/mole integration. These are in accordance with the limitations of the present method. In the same reaction with ST6Gal1 instead of ST3Gal3, 5.7% of the radiolabel was integrated into galactosylated EPO, or about 48% compared to ST3Gal 3.
13. In insect cellsGlycopegylation of the produced EPO
This example presents the preparation of pegylated dual-antennal EPO from insect cell-expressed EPO.
Recombinant human erythropoietin (rhEPO) (Protein Sciences corp., Meriden, CT) from the baculovirus/Sf 9 expression system was subjected to glycan analysis and showed that glycans were predominantly trimannose cores with core fucose, while a small percentage of glycans also had single GlcNAc (figure 128).
N-acetylglucosamine was added using GnT-I and GnT-II.Two batches of rhEPO (1mg/ml) were combined with GnT-I and GnT-II, 5mM UDP-GlcNAc, 20mM MnCl2And 0.02% sodium azide in 100mM MES pH6.5 at 32 ℃ for 24 hours. Batch A contained 20mg EPO and 100mU/mL GnT-I and 60mU/mL GnT-II. Batch B contained 41mg EPO and 41mU/mL GnT-I +50mU/mL GnT-II. After the reaction, the sample was desalted by gel filtration (PD10 column, Pharmacia LKB Biotechnology inc., Pi scataway, NJ).
Glycan characterization by 2AA HPLC revealed 92% conversion of lot a to a bi-antennary structure with two GlcNAc (the remainder with a single GlcNAc). Batch B showed 97% conversion to the desired product (fig. 125A and 125B).
Galactosylation of batch A EPO.EPO (16 mg batch A) was treated with GnT-II to complete the addition of GlcNAc. The reaction was carried out in the presence of 150mM NaCl, EPO mg/mL, 1mM ADP-GlcNAc, 5mM MnCl 20.02% sodium azide and 0.02U/ml GnT-II in 50mM Tris, pH7.2, at 32 ℃ for 4 hours. Then the galactosylation of EPO was performed by adding UDP-galactose to 3mM and GalT1 to 0.5U/ml and incubating at 32 ℃ for 48 hours.
The galactosylated EPO was then purified by gel filtration on a Superdex751.6/60 column in 50mM Tris, 0.15M NaCl, pH 6. The EPO-containing peaks were then analyzed by 2 AAHPLC. Based on HPLC data, after the galactosylation reaction-85% of the glycans contained two galactose and-15% of the glycans did not have any galactose.
Sialylation of galactosylated EPO.Sialylation of galactosyl EPO was performed in 100mM Tris containing 150mM NaCl, 0.5mg/ml EPO, 200U/ml ST3Gal3 with 0.5mM CMP-SA or CMP-SA-PEG (1kDa) or CMP-SA-PEG (10kPa) at 32 ℃ for 48 hours. Almost all glycans with two galactose residues were fully sialylated (2 sialic acids/glycan) after sialylation reaction with CMP-SA. MALDI-TOF analysis confirmed the HPLC data.
Pegylation of galactosylated EPO.For PEGylation reactions using CMP-SA-PEG (1kDa) and CMP-SA-PEG (10kDa), aliquots of the reaction mixtures were analyzed by SDS-PAGE (FIG. 129). The molecular weight of the EPO peptide increased with the addition of each sugar, and there was a more dramatic increase in molecular weight after the pegylation reaction.
In vitro bioassay of EPO.The in vitro EPO assay (adapted from Hammerling et al, 1996, J.Pharm.Biomed.anal.14: 1455-1469) is based on the responsiveness of TF-1 cell lines to various concentrations of EPO. TF-1 cells provide a good system for studying proliferation and differentiation of myeloid progenitor cells. The cell line was established by t.kitamura et al in 10 months 1987 from heparinized bone marrow aspirate from a 35 year old japanese male with severe pancytopenia. These cells are completely dependent on interleukin 3 or granulocyte macrophage colony stimulating factor (GM-CSF).
TF-1 cell line (ATCC, Cat. No. CRL-2003) was grown in RPMI + FBS 10% + GM-CSF (12ng/ml) and 5% CO at 37 deg.C2And (4) carrying out incubation. The cells were present in suspension at a concentration of 5000 cells/ml medium and 200. mu.l were placed in 96-well plates. The cells were incubated with various concentrations of EPO (0.1. mu.g/ml-10. mu.g/ml) for 48 hours. MTT viability assays were then performed as follows: add 25. mu.l of 5mg/ml MTT (SIGMA M5655), incubate the plates at 37 ℃ for 20 minutes to 4 hours, add 100. mu.l isopropanol/HCl solution (100ml isopropanol + 333. mu.l HCl6N), read the OD at 570nm and at 630nm or 690nm, and subtract the reading at 630nm or 690nm from the reading at 570nm 。
FIG. 130 contains the results obtained when sialylated EPO and EPO glycopegylated with 1kDa or 10kDa PEG were tested for EPO biological activity in vitro. EPO glycoPEGylated with 1kDa PEG has almost the same activity as EPO not glycoPEGylated, when both are at a concentration of about 5. mu.g/ml. EPO glycoPEGylated with 10kDa PEG has about half the activity of EPO that is not glycoPEGylated, when both are at a concentration of about 5. mu.g/ml.
PEGylation of saccharides of O-linked glycans of EPO produced in CHO cells
Preparation of O-linked EPO-SA-PEG (10 kDa).The desialylated-EPO originally produced by CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 5mM CMP-SA and 0.1U/mL ST3Gal3 for 2 days at 32 ℃. To monitor the incorporation of sialic acid into N-linked glycans, CMP-SA-14C; the peptides were isolated by gel filtration on a Toso Haas G3000SW analytical column using methanol and water, and the product was probed with a radiation detector. When the reaction was completed, the solution was concentrated using a Centricon-20 filter. The remaining solution was mixed with 0.05M Tris (pH7.2), 0.15M NaCl, 0.05% NaN 3Buffer was exchanged to a final volume of 7.2mL until no more CMP-SA was detected. The retentate was then resuspended in 0.05M Tris (pH7.2), 0.15M NaCl, 0.05% NaN at 2.5mg/mL protein3In (1). To glycosylate the O-attachment sites, the solution was incubated with 1mM CMP-SA-PEG (10kDa) and ST3Gal1 at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, small aliquots of the reaction were separated by gel filtration and analyzed by UV probing on a Toso Haas TSK-gel-3000 analytical column eluted with PBS ph 7.0. When the reaction was complete, the reaction mixture was purified with a Toso Haas TSK-gel-3000 preparative column using PBS buffer (ph7.0) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. Dialyzing the sample against water andanalysis was performed by MALDI-TOF MS.
EPO-transporting iron egg
This example presents the procedure for glycoconjugation of proteins to O-linked glycans, in particular transferrin to EPO. Sialic acid residues were removed from the O-linked glycans of EPO and EPO-SA-linker-SA-CMP was prepared. EPO-SA-linker-SA-CMP was glycoconjugated with desialylated transferrin with ST3Gal 3.
Preparation of O-linked desialylated-EPO.The EPo (erythropoietin) produced in CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl pH7.4, 0.15M NaCl and incubated with 300mU/mL sialidase (Vibrio cholerae) -agarose conjugate for 16 hours at 32 ℃. To monitor the reaction, small aliquots of the reaction were diluted with the appropriate buffer and the IEF gel was run according to the Invitrogen procedure. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The supernatant was concentrated to 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3And an EPO concentration of about 2.5mg/mL in pH7.2. The solution was incubated with 5mM CMP-sialic acid and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor sialic acid incorporation, a small aliquot of the reaction was added with CMP-SA-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). When the reaction was complete, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of EPO-SA-linker-SA-CMP.O-linked desialylated-EPO was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-linker-SA-CMP and 0.1U/mL ST3Gal1 at 32 ℃ for 2 days. To monitor and controlIntegration of sialic acid-linker-SA-CMP peptides were isolated by gel filtration on Toso Haas G3000SW analytical column using PBS buffer (ph 7.1).
After 2 days, the reaction mixture was purified with a Toso HaasG3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of transferrin-SA-linker-SA-EPO.The EPO-SA-adaptor-SA-CMP was dissolved in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN at a concentration of 2.5mg/mL3pH 7.2. The solution was incubated with 2.5mg/mL desialylated transferrin and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor the integration of transferrin, peptides were isolated by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph7.1) and the product was probed by UV absorption. When the reaction was complete, the solution was incubated with 5mM CMP-SA and 0.1U/mL ST3Gal3 (to cap any unreacted transferrin glycan) for 2 days at 32 ℃. The reaction mixture was purified with a Toso HaasG3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
16.EPO-GDNF
This example presents the procedure for glycoconjugation of proteins, in particular the preparation of EPO-SA-linker-SA-GDNF.
Preparation of EPO-SA-linker-SA-GDNF.The EPO-SA-adaptor-SA-CMP was dissolved in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN at a concentration of 2.5mg/mL3pH 7.2. This solution was incubated with 2.5mg/mL GDNF (produced in NSO cells) and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor the integration of GDNF, the peptides were applied in PBS buffer (p)H7.1) on a Toso Haas G3000SW analytical column by gel filtration and the product is detected by UV absorption. When the reaction was complete, the solution was incubated with 5mM CMP-SA and 0.1U/mL ST3Gal3 (to cap any unreacted GDNF glycans) for 2 days at 32 ℃. The reaction mixture was purified with a Toso HaasG3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
PEGylation of the one-touch sugar of EPO
This example presents methods for the preparation of glycopegylated single-contact Erythropoietin (EPO), and its in vitro and in vivo biological activities.
When EPO (GenBank Accession No. P01588) is expressed in CHO cells, N-linked glycans are formed at amino acid residues 24, 38 and 83, and O-linked glycans are formed at amino acid residue 126 (FIG. 131; Lai et al, 1986, J.biol.chem.261: 3116-3121). The biological activity of the glycoprotein is directly related to the content of NeuAc. Increased sialic acid reduces EPO binding to its receptor in vitro; however, increased sialic acid increases the in vivo biological activity of EPO. O-linked glycans have no effect on the in vitro or in vivo activity of EPO or the pharmacokinetics of this molecule (Wasley et al, 1991, Blood 77: 2624-.
When EPO is expressed in insect cells, N-linked glycans are formed at amino acid residues 24, 38 and 83, but O-linked glycans are not formed at amino acid residue 126, as is achieved using the baculovirus/Sf 9 expression system (see also Wojchowshi et al, 1987, biochem. Biophys. acta 910: 224-232; Quelle et al, 1989, Blood 74: 652-657) (FIG. 132). This is because the insect cells do not have glycosyltransferase recognizing the amino acid sequence around amino acid residue 126 of EPO. Most N-linked glycans are GlcNAc2Man3Fuc composition. In this example, EPO expressed in insect cells was efficiently reconstituted To generate complex glycans SA by contacting proteins sequentially with GnT1, 2, GalT-1, and ST in the presence of an appropriate donor molecule2Gal2GlcNAc2Man3FucGlcNAc2. These enzymatic reactions were performed on insect cell-expressed EPO using the reaction conditions described herein, resulting in a total efficiency of 92% in the production of the complex glycans herein (table 21). Optionally, O-linked glycans can also be added (O' Connell and Tabak, 1993, J.Dent.Res.72: 1554-1558; Wang et al, 1993, J.biol.chem.268: 22979-22983).
TABLE 21 percentage of each glycan structure species in the population of glycan structures on insect cell-expressed EPO ("starting material") and on EPO after each successive enzymatic reconstitution step.
Also in this example, EPO expressed in insect cells was reconstituted to form mono-, di-and tri-antennary glycans, which were subsequently sugar pegylated with 1kDa, 10kDa and 20kDa PEG molecules using the methods described elsewhere herein. The molecular weights of these EPO forms were determined and then conjugated to Epoetin having 3N-linked glycansTMAnd NESP (Aranesp) with 5N-linked glycans TM) Comparison was made (fig. 133). Examples of the preparation of biantennary and triantennary glycan structures are given in example 7 herein.
EPO having a mono-antennary pegylated glycan structure is prepared by expressing an EPO peptide in insect cells and then contacting the EPO peptide with GnTI only (or alternatively with GnTII only) in the presence of a GlcNAc donor. The EPO peptide is then contacted with GalT-I in the presence of a galactose donor. The EPO peptide was then contacted with ST in the presence of SA-PEG donor molecules (fig. 134A) to generate EPO peptide with 3N-linked single-tentacle pegylated glycan structures (fig. 134B).
The in vitro biological activity of EPO-SA and EPO-SA-PEG produced from EPO expressed by insect cells was estimated by measuring the stimulation of the molecule to the proliferation of TF-1 erythroleukemia cells. The triantennary EPO-SA-PEG1kDa showed the biological activity of almost all triantennary EPO-SA, while the biantennary EPO-SA-PEG10kDa showed the biological activity of almost all biantennary EPO-SA in some EPO concentration ranges (FIG. 135). Reconstituted and glycoPEGylated EPO produced in insect cells exhibits Epogen up to 94%TMThe Epogen, the in vitro biological activity ofTMIs EPO expressed in CHO without further glycan reconstitution or pegylation (table 22).
TABLE 22 comparison of Epogen at 2. mu.g/ml protein and 48 hoursTMIn vitro activity of the compared EPO constructs.
1The triantennary- SA 2, 3 construct has SA molecules bound with 2, 3 bonds.
The pharmacokinetics in vivo of glycopegylated and non-glycopegylated EPO were determined. PEGylation of sugars and non-sugar PEGylation [ I125]The labelled EPO bolus was injected into rats and the pharmacokinetics of the molecule was determined. The AUC of the 1kDa of biantennary EPO-PEG was 1.8 times higher and the AUC of the 10kDa of biantennary EPO-PEG was 11 times higher than that of the biantennary EPO (FIG. 136). The AUC of 1kDa for biantennary EPO-PEG was 1.6 times higher, and 46 times higher for 10kDa for biantennary EPO compared to biantennary EPO (FIG. 136). Thus, glycopegylation greatly improves the pharmacokinetics of EPO.
The in vivo biological activity of glycopegylated and non-glycopegylated EPO was also determined by measuring the extent to which the EPO construct stimulates reticulocytosis. Reticulocytosis is erythrocyte precursorMeasurement of the rate at which somatic cells mature into mature erythrocytes (erythrocytes). A single subcutaneous injection of 10 μ g protein/Kg was performed on 8 mice per treatment group and the percentage of reticulocytes was measured at 96 hours (fig. 137). Triante and biantennary PEGylated EPO over non-PEGylated EPO forms (including Epogen) TM) Shows higher in vivo biological activity.
Further determination of the in vivo bioactivity of the EPO constructs was estimated by measuring the hematocrit (percentage of whole blood consisting of red blood cells) of CD-1 female mice 15 days after three intraperitoneal injections per week of EPO constructs at 2.5. mu.g peptide/Kg body weight. The increase in hematocrit increases with the size of the EPO form, with a monoleptic angle of 82.7kDa EPO-PEG20kDa with a NESP (Ananesp) slightly greater than 35.6kDaTM) And its activity is Epogen at 28.5kDaTMAbout 2-fold higher biological activity (fig. 138).
This example demonstrates that it is feasible to produce sugar pegylated EPO with longer duration of action. The pharmacokinetic profile of glycopegylated EPO can be tailored by varying the number of glycopegylated sites and the size of the PEG molecule added to alter the half-life of the peptide in the bloodstream. Finally, glycopegylated EPO retains in vitro and in vivo biological activity.
18. Preparation and biological activity of sialylated and PEGylated mono-, di-and tri-antennary EPO
This example illustrates the production of saccharide pegylated EPO, particularly pegylated EPO having mono-antennary and bi-antennary glycans with PEG attached thereto. The following EPO variants were produced: single antenna PEG (1kDa) and PEG (20 kDa); biantennary 2, 3-Sialic Acid (SA), biantennary SA-PEG (1kDa), biantennary SA-PEG (10 kDa); three-antenna 2, 3-SA and three-antenna 2, 6-SA capped with 2, 3-SA.
Recombinant erythropoietin (rEPO) expressed in insect cells was obtained from ProteinSciences (Lot #060302, Meridan CT). The glycan composition of this batch of EPO had a trimannose core structure of about 98%. Fig. 139A depicts an HPLC analysis of glycans released from the EPO, wherein peak "P2" indicates a trimannose core. FIG. 139B shows MALDI analysis of released glycans, where the structures of the released glycans are shown next to the peaks they represent.
Single antenna branch
Several steps are performed to create a single antenna branched structure. Briefly, the first step is a GnT-I/GalT-1 reaction, followed by purification by Superdex-75 chromatography. This reaction adds a GlcNAc moiety to one branch of the trimannose core and a galactose moiety to the GlcNAc moiety. The branches were extended by adding a SA-PEG (10kDa) moiety or a SA-PEG (20kDa) moiety on the terminal galactose moiety by an ST3Gal3 reaction. Final purification was achieved by Superdex-200 chromatography (Amersham Biosciences, Arlington Heights, IL).
GnT-I/GalT-1 reactionGnT-I and GalT-1 reactants were combined and incubated at 32 ℃ for 36 hours. The reaction contained 1mg/mL EPO, 100mM Tris-Cl pH7.2, 150mM NaCl, 5mM MnCl2、0.02% NaN33mM UDP-GlcNAc, 50mU/mg GnT-I, 3mM UDP-Gal and 200mU/mg GalT-1. FIG. 140 depicts MALDI analysis of glycans released from EPO following a GnT-I/GalT-1 reaction. Glycan analysis showed that about 90% of the glycans had the expected one-touch angle branching structure with terminal galactose moieties.
Superdex-75 purificationEPO was purified from enzyme protein contaminants and nucleotide sugars in PBS (Sigma-Aldrich Corp., St.Louis, Mo.) with 0.02% Tween 20 using 1.6cm x 60cm Superdex-75 gel filtration chromatography (Amersham Biosciences, Arlington heights, IL) after the GnT-I/GalT-1 reaction.
ST3Gal3 reactionThe ST3Gal3 pegylation reaction was incubated at 32 ℃ for 24 hours. The reaction contained 1mg/mL EPO, 100mM Tris-Cl pH7.2, 150mM NaCl, 0.02% NaN3200mU/mg ST3Gal3 and 0.5mM CMP-SA-PEG (10kDa) or 0.5mM CMP-SA-PEG (20 kDa). FIG. 141 depicts SDS-PAGE analysis of EPO after this reaction. The corresponding molecular weights of the protein bands are shown by the GnT-I/GalT-1 reactionThe formed EPO glycans were completely sialylated by the PEG derivatives.
Superdex-200 purificationEPO was then purified from the ST3Gal3 reaction contaminant in PBS containing 0.02% Tween 20 using 1.6cm x 60cm Superdex-200 gel filtration chromatography (Amersham Biosciences, Arlington Heights, IL).
TF-1 cell in vitro bioassay of Single antennal PEGylated EPOThe TF-1 cell line was used to evaluate in vitro EPO activity. The TF-1 cell line is a bone marrow progenitor cell line available from the American type culture Collection (Catalogue No. CRL-2003, Rockyile, Md.). The viability of this cell line is completely dependent on interleukin-3 or granulocyte-macrophage colony stimulating factor. TF-1 cells provide a good system for studying the effects of EPO on proliferation and differentiation.
TF-1 cells at 37 ℃ in 5% CO2In RPMI medium with 10% FBS and 12ng/ml GM-CSF. Cells were suspended at a concentration of 10,000 cells/ml medium. A200. mu.l aliquot of cells was dispensed into 96-well plates. Cells were incubated with 0.1-10. mu.g/ml EPO for 48 hours.
MTT viability assay was then performed by first adding 25. mu.l of 5. mu.g/ml MTT (3- [4, 5-dimethylthiazol-2-yl ] -2, 5-diphenyltetrazolium bromide or thiazolyl blue (thiazolyl blue); Sigma chemical Co., St.Louis, Mo., Catalogue No. M5655). The plates were incubated at 37 ℃ for 4 hours. 100. mu.l of isopropanol/HCl solution (100ml of isopropanol and 333. mu.l of HCl 6N) was added. The absorbance of the plate was read at 570nm and at 630 or 690nm and the reading at 630 or 690nm was subtracted from the 570nm reading.
FIG. 142 depicts the bioassay of EPO activity following PEGylation of its monokeratoglycans. In this bioassay, monotact pegylated EPO is much less active than non-pegylated EPO (epogen).
Double antenna branch
Several reactions were performed to achieve the double antennary branching of EPO. Briefly, the first reaction is a combination of GnT-I and GnT-II reactions to add GlcNAc moieties to the two trimannose core branches. The second reaction, the GalT-1 reaction, adds a galactose moiety to each GlcNAc moiety. Superdex-75 chromatography (Amersham Biosciences, Arlington Heights, IL) was performed prior to ST3Gal3 reaction. The biantennary branches were further extended by an ST3Gal3 reaction to add 2, 3-SA or SA-PEG (1kDa), SA-PEG (10 kDa). Final purification was achieved using Superdex-200 chromatography (Amersham Biosciences, Arlington Heights, IL).
GnT-I/GnT-II reactionGnT-I and GnT-II reactants were combined and incubated at 32 ℃ for 48 hours. The reaction contained 1mg/mL EPO, 100mM MES pH6.5, 150mM NaCl, 20mM MnCl2、0.02% NaN35mM UDP-GlcNAc, 100mU/mg GnT-I, 60mU/mg GnT-II. The reaction achieves 92% completion of the addition of the bi-antennary GlcNAc moiety, while 8% is the mono-antennary GlcNAc moiety. FIG. 143A shows HPLC analysis of released glycans, with peak "P3" representing biantennary GlcNAc glycans. FIG. 143B shows MALDI analysis of released glycans, in which the structures of the glycans are shown next to the peaks they represent.
For further reaction, 1mM UDP-GlcNAc, 5mM MnCl were added2、0.02% NaN3An additional 20mU/mg GnT-II were added together and the mixture was incubated at 32 ℃ for 4 hours. Greater than 99% of the reaction achieves completion of biantennary GlcNAc glycans.
GalT-1 reactionAfter completion of the second GnT-II reaction, the GalT-1 reaction was started immediately. To the completed GnT-II reaction were added the enzyme and nucleotide sugar at a concentration of 0.5mU/mg GalT-1 and 3mM UDP-Gal.
When the GalT-1 reaction is carried out on a small scale with about 100. mu.g of EPO per reaction, about 95% of the reactions produce EPO with a biantennary terminal galactose moiety. Figure 144A shows HPLC analysis of released glycans in which peak "P2" is a biantennary glycan with a terminal galactose moiety (85% of glycans) and peak "P1" is a biantennary glycan without a terminal galactose moiety (15% of glycans).
The GalT-1 reaction was carried out on a large scale with about 16mg EPO per reaction. FIG. 144B shows HPLC analysis of glycans released from a large-scale GalT-1 reaction, where peak "P2" is a biantennary glycan with a terminal galactose moiety and peak "P1" is a biantennary glycan without a terminal galactose moiety.
Superdex-75 purificationEPO was purified from enzyme protein contaminants and nucleotide sugars in PBS containing 0.02% Tween 20 after the GnT-1/GalT1 reaction using 1.6cm x 60cm Superdex-75 gel filtration chromatography (Amersham Biosciences, Arlington heights, IL). FIG. 145 depicts a Superdex 75 gel filtered chromatogram in which peak 2 is a biantennary glycan with a terminal galactose moiety. Figure 146 shows SDS-PAGE analysis of the product from each reconstitution step, showing an increase in EPO molecular weight with each reconstitution step.
ST3Gal3 reactionThe ST3Gal3 reaction was incubated at 32 ℃ for 24 hours. The reaction contained 0.5mg/mL EPO, 100mM Tris-Cl pH7.2, 150mM NaCl, 0.02% NaN3100mU/mg ST3Gal3 and 0.5mM CMP-SA, 0.5mM CMP-SA-PEG (1kDa) or 0.5mM CMP-SA-PEG (10 kDa). FIG. 147 depicts SDS-PAGE analysis of EPO before and after ST3Gal3 response. Based on this SDS-PAGE analysis, the biantennary EPO containing the terminal Gal was no longer visibly detectable after each ST3Gal3 response. All sialylated EPO variants showed an increase in size compared to non-sialylated EPO at the start of the reaction.
Superdex-200 purificationEPO was then purified from ST3Ga l3 reaction contaminants in PBS containing 0.02% Tween 20 using 1.6cm x 60cm Superdex-200 gel filtration chromatography (Amersham Biosciences, Arlington Heights, IL). Table 23 summarizes the distribution of glycan structures at each reconstitution step.
Table 23 summary of glycan structures on EPO after each reconstitution step.
Diamonds represent fucose and squares represent GlcNAc, circles represent mannose and open circles represent galactose.
Three-antenna branch
Several reactions were performed to achieve the three tentacle branching of EPO. Briefly, the first reaction is a combination of GnT-I and GnT-II reactions to add GlcNAc moieties to the two outer trimannose core branches of glycans. The second reaction, the GnT-V reaction, adds a second GlcNAc moiety to one of the two outer trimannose core branches, so that there are now 3 GlcNAc moieties. The third reaction, the GalT-1 reaction, adds a galactose moiety to each terminal GlcNAc moiety. The EPO product was then isolated by Superdex-75 chromatography. The triantennary branches were further extended with an ST3Gal3 reaction to add 2, 3-SA or 2, 6-SA moieties and capped with 2, 3-SA moieties. Final purification was achieved using Superdex-75 chromatography.
GnT-I/GnT-II reactionGnT-I and GnT-II reactants were combined and incubated at 32 ℃ for 24 hours. The reaction contained 1mg/mL EPO, 100mM MES pH6.5, 150mM NaCl, 20mM MnCl2、0.02% NaN35mM UDP-GlcNAc, 50mU/mg GnT-I and 41mU/mg GnT-II. The reaction achieved 97% completion of the addition of the biantennary GlcNAc moiety, with the remaining 3% trimannose core. FIG. 148 depicts HPLC analysis of glycans released from EPO following a GnT-I/GnT-II reaction.
GnT-V reactionThe GnT-V reaction contained 100mM MES pH6.5, 5mM UDP-GlcNAc, 5mM MnCl2、0.02% NaN310mU/mg GnT-V and 1mg/mL EPO, and incubated at 32 ℃ for 24 hours. This reaction adds a GlcNAc moiety to the outer mannose moiety that already contains a GlcNAc moiety. FIG. 149 depicts HPLC analysis of glycans released from EPO following a GnT-V response. Based on glycan and MALDI analysis, approximately 92% of the glycans released from EPO are expected products, i.e., with terminal GlcNAcPart of the three-antennary branched EPO. The remaining 8% of the glycans are structures containing bi-antennary branches with terminal GlcNAc moieties.
GalT-1 reactionGalT-1 reaction containing 100mM Tris pH7.2, 150mM NaCl, 5mM UDP-Gal, 100mU/mg GalT-1, 5mM MnCl2、0.02% NaN3And 1mg/mL EPO, and incubated at 32 ℃ for 24 hours. Figure 150 depicts HPLC analysis of glycans released from EPO after this reaction. Glycan and MALDI analysis showed that 97% of the released glycans had terminal galactose moieties on the triantennary branch structure. The remaining 3% are biantennary structures containing terminal galactose.
Superdex-75 purificationEPO was purified from enzyme protein contaminants and nucleotide sugars in PBS containing 0.02% Tween 20 using 1.6cm x 60cm Superdex-75 gel filtration chromatography (Amersham Biosciences, Arlington heights, IL) following GnT-I/GalTl reaction. The purified material was divided into two batches to produce triantennary glycans with terminal 2, 6-SA moieties and triantennary glycans with terminal 2, 6-SA moieties capped with 2, 6-SA moieties.
ST3Gal3 reactionThe ST3Gal3 reaction was incubated at 32 ℃ for 24 hours. The reaction contained 1mg/mL galactosylated BPO, 100mM Tris-Cl pH7.2, 150mM NaCl, 0.02% NaN350mU/mg ST3Gal3 and 3mM CMP-SA. Figure 151 depicts HPLC analysis of glycans released from EPO after this step. Based on glycan and MALDI analysis, approximately 80% of the released glycans are structures with three antennary branches with terminal 2, 3-SA moieties. The remaining 20% of the released glycans are bi-antennary structures containing terminal 2, 3-SA moieties.
ST6Gal1 sialylation reaction after ST3Gal3 reactionThe ST6Gal1 reaction was incubated at 32 ℃ for 24 hours. The reaction contained 1mg/mL sialylated and galactosylated EPO, 100mM Tris-Cl pH7.2, 150mM NaCl, 0.02% NaN 350mU/mg ST6Gal1 and 3mM CMP-SA. Fig. 152 depicts HPLC analysis of glycans released from EPO following ST6Gal1 reaction. Approximately 80% of the three antennae based on glycan and MALDI analysisBranched glycans contain terminal 2, 3-SA moieties. The remaining 20% of the glycans are biantennary containing terminal 2, 3-SA moieties.
Superdex-75 purificationEPO was purified from the ST3Gal3 reaction contaminant in PBS containing 0.02% Tween20 using 1.6cm x 60cm Superdex-75-brene filtration chromatography (Amersham Biosciences, Arlington Heights, IL).
Bioassay of triantennary and biantennary sialylated or PEGylated EPOThe activity of the triantennary and biantennary sialylated EPO glycoform and the PEG 10kDa and 1kDa biantennary glycoforms was determined using the TF-1 cell line and the MTT viability assay as described above. Figure 153 depicts the results of MTT cell proliferation assay. At 2 μ g/ml EDP, the biantennary sialylated EPO had almost the activity of the control Epogen, whereas the triantennary sialylated EPO had significantly lower activity.
Blood coagulation factor IX
19.Glycopegylation of coagulation factor IX produced by CHO cells
This example presents the preparation of desialylated coagulation factor IX and its sialylation with CMP-sialic acid-PEG.
Desialylation of rFactor IX.The recombinant form of coagulation factor ix (r facorix) was produced in CHO cells. 6000IU of rFactor IX were dissolved in a total of 12mL of USP H2And (4) in O. This solution was mixed with another 6mL USP H2O was transferred to a Centricon Plus 20, PL-10 centrifugal filter. The solution was concentrated to 2mL and then 15mL of 50M Tris-HClpH7.4, 0.15M NaCl, 5mM CaCl was used2、0.05% NaN3Diluted and concentrated again. The dilution/concentration was repeated 4 times to effectively change the buffer to a final volume of 3.0 mL. 2.9mL of this solution (ca. 29mg rFactor IX) were transferred to a small plastic tube and 530 mU α 2-3, 6, 8-neuraminidase-agarose conjugate (Vibrio cholerae, Calbiochem, 450 μ L) were added thereto. The reaction mixture was gently swirled at 32 ℃ for 26.5 hoursThen (c) is performed. The mixture was centrifuged at 10,000rpm for 2 minutes and the supernatant was collected. Agarose beads (containing neuraminidase) were washed with 0.5mL of 50M Tris-HCl pH7.12, 1M NaCl, 0.05% NaN3Washing is carried out 6 times. The collected wash and supernatant were again centrifuged at 10,000rpm for 2 minutes to remove any remaining agarose resin. The collected desialylated protein solution was diluted to 19mL with the same buffer and concentrated to 2mL in a Centricon Plus 20 PL-10 centrifuge filter. The solution was washed with 15mL of 50M Tris-HCl pH7.4, 0.15M NaCl, 0.05% NaN 3Diluted two-fold and concentrated to 2 mL. The final desialylated rFactor IX solution was diluted to a final volume of 3mL (. about.10 mg/mL) with Tris buffer. Native and desialylated rFactor IX samples were analyzed by IEF-electrophoresis. Isoelectric bathing Gels (pH3-7) were run with 1.5. mu.L (15. mu.g) samples that were first diluted with 10. mu.L Tris buffer and mixed with 12. mu.L sample loading buffer. Gels were loaded, run and fixed using standard procedures. The gel was stained with Colloidal Blue Stain (FIG. 154), showing bands of desialylated coagulation factor IX.
Preparation of PEG (1kDa and 10kDa) -SA-coagulation factor IX.The desialylated rFactor-IX (29mg, 3mL) was split into two 1.5mL (14.5mg) samples in two 1.5mL centrifuge tubes. Each solution was treated with 12.67mL of 50mM Tris-HCl pH7.4, 0.15M NaCl, 0.05% NaN3Diluted and CMP-SA-PEG-1k or 10k (7.25. mu. mol) was added. The tube was gently inverted for mixing and 2.9U of ST3Gal3 (326. mu.L) was added (14.5mL total volume). The tube was inverted again and gently rotated at 32 ℃ for 65 hours. The reaction was stopped by freezing at-20 ℃. Mu.g of the reaction sample was analyzed by SDS-PAGE. The PEGylated protein was purified on a Toso Haas Biosep G3000SW (21.5X 30cm, 13um) HPLC column using Dulbecco's phosphate buffered saline (Gibco), pH7.1, 6 mL/min. Reactions and purifications were monitored using SDS Page and IEF gels. After the sample was washed with 2. mu.L of 50mM Tris-HCl pH7.4, 150mM NaCl, 0.05% NaN 3Buffer dilution, mixing with 12. mu.L sample loading buffer and 1. mu.L of 0.5M DTT, andafter heating at 85 ℃ for 6 minutes, 10. mu.L (10. mu.g) of this sample was loaded onto a Novex Tris-glycine 4-20% 1mm gel. The gel was stained with Colloidal Blue Stain (FIG. 155), showing bands of PEG (1kDa and 10kDa) -SA-coagulation factor IX.
20. Direct sialic acid-sugar PEGylation of coagulation factor IX
This example proposes sialic acid-pegylation of coagulation factor IX without prior sialidase treatment.
Sialic acid-PEGylation of factor IX was performed using CMP-SA-PEG (10 kDa).Coagulation factor IX (1100 IU) was dissolved in 5mL of 20mM histidine, 520mM glycine, 2% sucrose, 0.05% NaN3And 0.01% polysorbate 80, ph5.0, said coagulation factor IX being expressed in CHO cells and being fully sialylated. CMP-SA-PEG- (10kDa) (27mg, 2.5. mu. mol) was then dissolved in the solution and 1U ST3Gal3 was added. The reaction was complete after gentle mixing at 32 ℃ for 28 hours. The reaction was analyzed using SDS-pAGE described by Invitrogen. The product protein was purified on an Amersham Superdex 200 (10X 300nm, 13 μm) HPLC column using Phosphate Buffered Saline (PBS) pH7.0, 1 mL/min. R t9.5 min.
Sialic acid-PEGylation of factor IX was performed using CMP-SA-PEG (20 kDa).Coagulation factor IX (1100 IU) was dissolved in 5mL of 20mM histidine, 520mM glycine, 2% sucrose, 0.05% NaN3And 0.01% polysorbate 80, ph5.0, said coagulation factor IX being expressed in CHO cells and being fully sialylated. CMP-SA-PEG- (20kDa) (50mg, 2.3. mu. mol) was then dissolved in the solution and added to CST-II. After gentle mixing at 32 ℃ for 42 hours the reaction mixture was complete. The reactions were analyzed by SDS-PAGE as described by Invitrogen.
The product protein was purified on an Amersham Superdex 200 (10X 300nm, 13 μm) HPLC column using phosphate buffered saline (Fisher) pH7.0, 1 mL/min. Rt8.6 min.
21. Sialic acid capping of glycoPEGylated coagulation factor IX
This example presents the procedure for sialic acid capping of sialic acid-sugar pegylated peptides. Herein, coagulation factor-IX is an exemplary peptide.
N-linked and O-linked glycans of coagulation factor-IX-SA-PEG (10kDa) sialic acid plus Cap (hat)Purified rFactor-IX-PEG (10kDa) (2.4mg) was added toThe concentrate was performed on Plus20PL-10(Millipore Corp., Bedford, Mass.) centrifugal filters and the buffer was changed to 1.85mL of 50mM Tris-HCl pH7.2, 0.15M NaCl, 0.05% NaN in final volume 3. The protein solution was diluted with 372. mu.L of the same Tris buffer and 7.4mg of CMP-SA (12. mu. mol) solid was added. The solution was gently inverted to mix and 0.1U ST3Gal1 and 0.1U ST3Gal3 were added. The reaction mixture was gently swirled at 32 ℃ for 42 hours.
A10. mu.g reaction sample was analyzed by SDS-PAGE. Novex Tris-glycine 4-12% 1mm gels were run as described by Invitrogen and stained with Colloid Blue. Briefly, 10. mu.L (10. mu.g) of the sample was mixed with 12. mu.L of sample loading buffer and 1. mu.L of 0.5M DTT and heated at 85 ℃ for 6 minutes (FIG. 156, lane 4).
Blood coagulation factor VIIa
22.Glycopegylation of recombinant coagulation factor VIIa produced in BHK cells
This example proposes pegylation of recombinant factor VIIa produced in BHK cells.
Preparation of desialylated factor VIIa.Recombinant factor VIIa is produced in BHK cells (baby hamster kidney cells). Factor VIIa (14.2mg) was dissolved at 1mg/ml in a buffer (pH7.4, 0.05M Tris, 0.15M NaCl, 0.001M CaCl)2、0.05% NaN3) And 3 with00mU/mL sialidase (Vibrio cholerae) -agarose conjugate was incubated at 32 ℃ for 3 days. To monitor the reaction, small aliquots of the reaction were diluted with the appropriate buffer and analyzed on an IEF gel (fig. 157) according to the procedure of Invitrogen. The mixture was centrifuged at 3,500rpm and the supernatant was collected. The resin was washed with the above buffer (pH7.4, 0.05M Tris, 0.15M NaCl, 0.05% NaN) 3) Wash 3 times (3X 2mL) and concentrate the combined washes in Centricon-Plus-20. The remaining solution was mixed with 0.05M Tris (pH7.4), 0.15M NaCl, 0.05% NaN3Buffer exchange was performed to obtain a final volume of 14.4 mL.
Preparation of factor VIIa-SA-PEG (1kDa and 10 kDa).The desialylated rFactorVIIa solution was divided into two equal 7.2ml samples. To each sample was added CMP-SA-5-PEG (1kDa) (7.4mg) or CMP-SA-5-PEG (10kDa) (7.4 mg). ST3Gal3(1.58U) was added to both tubes and the reaction mixture was incubated at 32 ℃ for 96 hours. The reaction was monitored by SDS-PAGE gels using the reagents and conditions described by Invitrogen. When the reaction was complete, the reaction mixture was purified using a Toso Haas TSK-Gel-3000 preparative column using PBS buffer (pH7.1) and fractions were collected based on UV absorption. The combined product-containing fractions were concentrated at 4 ℃ in a Centricon-Plus-20 centrifugal filter (Millipore, Bedford, Mass.), and the concentrated solution was reconstituted to give 1.97mg (bicinchoninic acid protein assay, BCA assay, Sigma-Aldrich, St. LouisMO) of coagulation factor VIIa-PEG. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF. FIG. 158 shows MALDI results obtained from native factor VIIa. FIG. 159 contains MALDI results for factor VIIa PEGylated with 1kDa PEG, where the peak for factor VIIa PEGylated with 1kDa PEG is evident. FIG. 160 contains MALDI results for factor VIIa PEGylated with 10kDa PEG, where the peak of factor VIIa PEGylated with 10kDa PEG is evident. FIG. 161 depicts SDS-PAGE analysis of all reaction products, factor VIIa-SA-PEG (10-kD) a) The band of (a) is evident.
Follicle Stimulating Hormone (FSH)
23.glycoPEGylation of human pituitary-derived FSH
This example illustrates the assembly of a conjugate of the invention. Follicle Stimulating Hormone (FSH) was desialylated and then conjugated with CMP- (sialic acid) -PEG.
Desialylation of follicle stimulating hormone.1mg of Follicle Stimulating Hormone (FSH) (HumanPituitary, Calbiochem Cat No.869001) was dissolved in 500. mu.L of 50mM Tris-HCl, 0.15M NaCl, 5mM CaCl, pH7.42In (1). 375. mu.L of this solution was transferred to a small plastic tube, and 263mU of neuraminidase II (Vibrio cholerae) was added thereto. The reaction mixture was gently shaken at 32 ℃ for 15 hours. The reaction mixture was added to 600. mu. L N- (p-aminophenyl) oxamic acid-agarose conjugate prepared in advance with 50mM Tris-HCl pH7.4, 150mM NaCl and 0.05% NaN3Equilibration was performed and gentle rotation at 4 ℃ for 6.5 hours. The suspension was centrifuged at 14,000rpm for 2 minutes and the supernatant was collected. The beads were washed 5 times with 0.5mL buffer and all supernatants were collected. The enzyme solution was diluted with 2L of 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3The solution was dialyzed (7000MWCO) at 4 ℃ for 15 hours and then at 4 ℃ for 4 hours against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN 3Dialyzed twice. The solution was concentrated to 2. mu.g/. mu.L with a Speed Vac and stored at-20 ℃. Reaction samples were analyzed by IEF gel (pH3-7) (Invitrogen) (FIG. 162).
Preparation of human pituitary-derived SA-FSH and PEG-SA-follicle stimulating hormone.Disialylated FSH (100. mu.g, 50. mu.L) and CMP-sialic acid or CMP-SA-PEG (1kDa or 10kDa) (0.05. mu. mol) were dissolved in 13.5. mu. L H in 0.5mL plastic tubes2O (adjusted to pH8 with NaOH). Tubes were briefly vortexed and 40mU ST3Gal3 (36.5. mu.L) was added (100. mu.L total volume). The tube was vortexed again and gently shaken at 32 ℃ for 24 hours. The reaction was stopped by freezing at-80 ℃. SDS-assisted assay for 15. mu.g of reaction samplespAGE (FIG. 163), IEF gel (FIG. 164) and MALDI-TOF. Native FSH was also analyzed by SDS-PAGE (fig. 165).
SDS PAGE and IEF gel analysis of the reaction products.Novex Tris-glycine 8-16% 1mm gels for SDS PAGE analysis were purchased from Invitrogen. mu.L (15. mu.g) of FSH reaction sample was incubated with 5. mu.L of 50mM Tris-HCl pH7.4, 150mM NaCl, 0.05% NaN3Buffer dilution, mixing with 15. mu.L sample loading buffer and 1. mu.L 9M. mu.mercaptoethanol and heating at 85 ℃ for 6 minutes. Gels were run as indicated by Invitrogen and stained with coloidalblue Stain (Invitrogen).
The FSH sample (15 μ g) was diluted with 5 μ L Tris buffer and mixed with 15 μ L sample loading buffer (FIG. 162). The samples were then applied to Isoelectric Focusing Gels (pH3-7) (Invitrogen) (FIG. 165). Gels were run and fixed as indicated by Invitrogen and then stained with Colloidal Blue Stain.
24glycoPEGylation of recombinantly produced recombinant FSH in CHO cells
This example illustrates the assembly of a conjugate of the invention. Desialylated FSH is conjugated to CMP- (sialic acid) -PEG.
Preparation of recombinant desialylated follicle stimulating hormone.Recombinant follicle stimulating hormone (rFSH) produced from CHO was used in these studies. 7,500IU of rFSH was dissolved in 8mL of water. FSH solution was incubated at 50mM Tris-HCl pH7.4, 0.15M NaCl, 5mM CaCl2Dialyzed and concentrated to 500. mu.L on a Centricon Plus 20 centrifugal filter. A portion of this solution (400. mu.L) (. about.0.8 mg FSH) was transferred to a small plastic tube and 275mU of neuraminidase II (Vibrio cholerae) was added thereto. The reaction mixture was allowed to mix for 16 hours at 32 ℃. The reaction mixture was added to the previously washed N- (p-aminophenyl) oxamic acid-agarose conjugate (800 μ L) and gently rotated at 4 ℃ for 24 hours. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The beads were washed 3 times with 0.6mL Tris-EDTA buffer, 1 time with 0.4mL Tris-EDTA buffer and Wash 1 time with 0.2mL Tris-EDTA buffer and collect all supernatants. The supernatant was incubated at 4 ℃ with 2L of 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialyzed, and then against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialysis was performed twice. The dialyzed solution was then concentrated to 420. mu.L in a Centricon Plus 20 centrifugal filter and stored at-20 ℃.
Native and desialylated rFSH samples were analyzed by SDS-PAGE and IEF (fig. 166). Novex Tris-glycine 8-16% 1mm gels were purchased from Invitrogen. Samples (7.5. mu.L, 15. mu.g) were incubated with 5. mu.L of 50mM Tris-HCl pH7.4, 150mM NaCl, 0.05% NaN3Buffer dilution, mixing with 15. mu.L sample loading buffer and 1. mu.L 9M beta-mercaptoethanol and heating at 85 ℃ for 6 minutes. Gels were run as indicated by Invitrogen and stained with coloraldlue blue Stain (Invitrogen). Isoelectric Focusing Gels (pH3-7) were purchased from Invitrogen. Samples (7.5. mu.L, 15. mu.g) were diluted with 5. mu.L Tris buffer and mixed with 15. mu.L sample loading buffer. The gel was loaded, run and fixed as indicated by Invitrogen. The gel was stained with Colloidal Blue Stain. The native and desialylated FSH samples were also dialyzed against water and analyzed by MALDI-TOF.
Sialic acid-pegylation of recombinant follicle stimulating hormone.Disialylated FSH (100. mu.g, 54. mu.L) and CMP-SA-PEG (1kDa or 10kDa) (0.05. mu. mol) were dissolved in 28. mu.L of 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN in a 0.5mL plastic tube3pH 7.2. Tubes were briefly vortexed and 20mU of ST3Gal3 (100. mu.L total volume) added. The tube was vortexed again and gently mixed at 32 ℃ for 24 hours, and the reaction was stopped by freezing at-80 ℃. Samples of this reaction were analyzed by SDS-PAGE (FIG. 167), IEF gel (FIG. 168) and MALDI-TOF MS as described above.
MALDI was also performed on pegylated rFSH. During the ionization process, SA-PEG is removed from the N-glycan structure of the glycoprotein. Native FSH gave a peak at 13928; AS-rFSH (13282); resialylated r-FSH (13332); PEG1000-rFSH (13515; 14960 (1); 16455 (2); 17796 (3); 19321 (4)); and PEG10000(23560 (1); 34790 (2); 45670(3) and 56760 (4)).
25. Pharmacokinetic study of glycopegylated FSH
This example presents an in vivo test of the pharmacokinetic properties of glycopegylated Follicle Stimulating Hormone (FSH) prepared according to the method of the invention compared to non-pegylated FSH.
FSH, FSH-SA-PEG (1kDa) and FSH-SA-PEG (10kDa) were radioiodinated using standard conditions (Amersham Biosciences, Arlington Heights, IL) and prepared in phosphate buffered saline containing 0.1% BSA. After dilution to the appropriate concentration in phosphate buffer, each of the FSH proteins tested (0.4. mu.g each) was injected intravenously into female Sprague Dawley rats (body weight 250-300g) and blood was drawn at time points from 0 to 80 hours. Radioactivity in blood samples was analyzed by gamma counter and pharmacokinetics were analyzed by standard methods (fig. 169). FSH is cleared more rapidly from the blood than FSH-PEG (1kDa), whereas FSH-PEG (1kDa) is cleared slightly more rapidly than FSH-PEG (10 kDa).
26. Support cell bioassay of glycopegylated FSH in vitro Activity
This example presents a culture-based bioassay of Follicle Stimulating Hormone (FSH) by supporting cells. This assay can be used to determine the biological activity of FSH after glycan reconstitution (including glycoconjugation).
The bioassay is based on the dose-response relationship that exists between the amounts of estradiol that is produced when FSH, rather than Luteinizing Hormone (LH), is added to cultured support cells obtained from immature, aged rats. Exogenous testosterone may be converted to 17 β -estradiol in the presence of FSH.
Old Sprague-Dawley rats 7-10 days old were used to obtain the supporting cells. After sacrifice, the testes were decapsulated and the tissues dispersed by incubation in collagenase (1mg/ml), trypsin (1mg/ml), hyaluronidase (1mg/ml) and DNase (5. mu.g/ml) for 5-10 minutes. The vial fragment was placed at the bottom of the flask and washed in PBS (1 ×). The tubule fragments were again incubated for 20 minutes with medium containing the same enzyme: collagenase (1mg/ml), trypsin (1mg/ml), hyaluronidase (1mg/ml) and DNase (5. mu.g/ml).
The tubule fragments were homogenized and placed in serum-free medium on 24-well plates. Place 5x10 per well5And (4) cells. At 37 ℃ and 5% CO2After 48 hours of incubation, fresh medium was added to the cells. Composition of serum-free medium: DMEM (1 vol), Ham's F10 nutrient mixture (1 vol), insulin 1. mu.g/ml, transferrin 5. mu.g/ml, EGF10ng/ml, T420pg/ml, hydrocortisone 10-8M retinoic acid 10-6M。
The stimulation experiment was: with standard FSH or sample at 37 ℃ and 5% CO2Incubated for 24 hours. The mean coefficient of variation within the assay was 9%, and the mean coefficient of variation between assays was 11%.
The 17B-estradiol Elisa kit DE2000(R & D Systems, Minneapolis, MN) was used to quantify the level of estradiol after incubation with FSH, FSH-SA-PEG (1kDa) and FSH-SA-PEG (10 kDa).
The procedure was as follows: pipette 100 μ l estradiol standards (provided by the kit and prepared according to the kit instructions) or samples onto 17B-estradiol Elisa plates; add 50 μ Ι of 17B-estradiol conjugate (provided by the kit and prepared according to the instructions of the kit) to each well; add 50 μ Ι of 17B-estradiol antibody solution (provided by the kit and prepared according to the kit instructions) to each well; the plate was incubated at 200rpm for 2 hours at room temperature; aspirating liquid from each well; the wells were washed four times with wash solution; removing all liquid from the well; add 200 μ Ι of pNPP substrate (provided by the kit and prepared according to the kit instructions) to all wells and incubate for 45 minutes; add 50. mu.l of stop solution (provided by and prepared according to the kit instructions) and read the plate at 405nm (FIG. 170). Although FSH-PEG (10kDa) showed moderate stimulation of the supporting cells at 1. mu.l/ml, FSH-PEG (1kDa) stimulated the supporting cells by 50% more than the non-PEGylated FSH.
27. Steelman-Pohley bioassay of in vivo Activity of glycoPEGylated FSH
In this example, the Steelman-Pohley bioassay (Steelman and Pohley, 1953, Endocrinology 53: 604-. The Steelman-Pohley bioassay uses changes in rat ovarian weight to measure the in vivo activity of FSH co-injected with human chorionic gonadotropin.
The Steelman-Pohley bioassay was performed according to the protocol described by Christin-Maitre et al (2000, Methods 21: 51-57). 70 female Sprague-Dawley rats (Charles River Laboratories, Wilmington, Mass.) 21-22 days old were placed in the testing apparatus for at least 5 days before starting the assay procedure. During the whole procedure, the climate of the animal room is controlled to 18-26 ℃, 30-70% relative humidity and 12 hours of artificial light/12 hours of darkness. All animals were fed Certified Rodent Chow (Harlan Teklad, Madison WI) or equivalent and water, and were obtained ad libitum. Animal procedures were performed in calvert clinical Services, Inc (Olyphant, PA).
Recombinant FSH was expressed in CHO cells, purified by standard techniques and glycopegylated with PEG (1 kDa). Rats were divided into 7 experimental groups of 10 animals each. Animals of all groups were injected subcutaneously with 0.5ml of 20i.u. Human Chorionic Gonadotropin (HCG) in 0.9% NaCl on days-1 and 0. On days 1, 2 and 3, control animals were injected subcutaneously with 0.5ml doses containing 20i.u. HCG in 0.9% NaCl, whereas in other groups HCG doses increased with 0.14 μ g, 0.4 μ g or 1.2 μ g rFSH or rFSH-SA-PEG (1kDa) per dose. At 4 days, by CO 2Animals were euthanized by inhalation. Ovaries were harvested, trimmed and weighed. The average ovarian weight for each group was determined.
FIG. 171 shows the mean ovarian weights of the experimental groups at day 4. The group receiving HCG alone (control) or the group receiving a low dose (0.14. mu.g) of rFSH or rFSH-SA-PEG (1kDa) had approximately equal ovarian weights. The group receiving medium (0.4. mu.g) or high (1.2. mu.g) doses of rFSH or rFSH-SA-PEG (1kDa) had approximately twice the ovarian weight of the control group. At moderate doses (0.4 μ g), the glycopegylated rFSH had approximately the same in vivo activity as non-pegylated rFSH (as determined by ovarian weight). At high doses (1.2 μ g), the glycopegylated rFSH had slightly higher in vivo activity than the non-pegylated rFSH.
G-CSF
28.Glycopegylation of G-CSF produced in CHO cells
Desialylation-preparation of granulocyte colony stimulating factor (G-CSF).G-CSF produced in CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl pH7.4, 0.15M NaCl, 5mM CaCl2And concentrated to 500. mu.L in a Centricon Plus 20 centrifugal filter. The solution was incubated with 300mU/ml neuraminidase II (Vibrio cholerae) for 16 hours at 32 ℃. To monitor the reaction, a small aliquot of the reaction was diluted with the appropriate buffer and an IEF gel analysis was run. The reaction mixture was then added to a pre-washed N- (p-aminophenyl) oxamic acid-agarose conjugate (800. mu.L/mL reaction volume) and the washed beads were gently rotated at 4 ℃ for 24 hours. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The beads were washed 3 times with Tris-EDTA buffer, once with 0.4mL Tris-EDTA buffer and once with 0.2mL Tris-EDTA buffer, and all supernatants were collected. The supernatant was treated with 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN at 4 deg.C 3Dialysis was performed, and then 50mM Tris-HClpH7.4, 1M NaCl, 0.05% NaN were added3Dialysis was performed twice. The dialyzed solution was then concentrated with a Centricon plus 20 centrifugal filter and stored at-20 ℃. The conditions of the IEF gel were run according to the procedures and reagents provided by Invitrogen. The native and desialylated G-CSF samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of G-CSF- (. alpha.2, 3) -sialic acid-PEG.The desialylated G-CSF is diluted with 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-PEG and 0.1U/mL ST3Gal1 at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The native and PEGylated G-CSF samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of G-CSF- (. alpha.2, 8) -sialic acid-PEG.G-CSF containing α 2, 3-sialylated O-linked glycan produced in CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-PEG and 0.1U/mL CST-II at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The native and PEGylated G-CSF samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of G-CSF- (. alpha.2, 6) -sialic acid-PEG.G-CSF containing only O-linked GalNAc was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN 3pH 7.2. Allowing the solution to standIncubated with 1mM CMP-sialic acid-PEG and 0.1U/mL ST6GalNAcI or II at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso HaasG3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The native and PEGylated G-CSF samples were dialyzed against water and analyzed by MALDI-TOF MS.
G-CSF produced in CHO cells was treated with Arthrobacter (Arthrobacter) sialidase and purified by size exclusion on Superdex 75, treated with ST3Gal1 or ST3Gal2, and then treated with CMP-SA-PEG 20 kDa. The resulting molecules were purified by ion exchange and gel filtration, and analysis by SDS PAGE demonstrated that pegylation was complete. This is the first demonstration of glycoPEGylation of O-linked glycans.
Glucocerebrosidase
29.Glucocerebrosidase-mannose-6-phosphate produced in CHO cells
This example presents the procedure for glycoconjugation of mannose-6-phosphate to peptides produced in CHO cells, such as glucocerebrosidase.
Preparation of desialylated glucocerebrosidase.Glucocerebrosidase produced in CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl pH7.4, 0.15M NaCl and incubated with 300mU/mL sialidase-agarose conjugate for 16 hours at 32 ℃. To monitor the reaction, small aliquots of the reaction were diluted with the appropriate buffer and IEF gels and SDS-PAGE were run according to the Invitrogen program. The mixture was centrifuged at 10,000rpm and the supernatant was collected. Will be provided withThe beads were washed 3 times with Tris-EDTA buffer, once with 0.4mL Tris-EDTA buffer and once with 0.2mL Tris-EDTA buffer. All supernatants were collected. The supernatant was incubated at 4 ℃ with 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialyzed, and then against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialysis was performed twice. The dialyzed solution was then concentrated using a Centricon Plus 20 centrifugal filter. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of glucocerebrosidase-SA-linker-mannose-6-phosphate (procedure 1).The desialylated glucocerebrosidase was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. This solution was incubated with 1mM CMP-sialic acid-linker-Man-6-phosphate and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-linker-Man-6-phosphate, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas TSK-gel-3000 analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. When the reaction was completed, the reaction mixture was purified with a Toso Haas TSK-gel-3000 preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of glucocerebrosidase-SA-linker-mannose-6-phosphate (procedure 2).The CHO-produced, but incompletely sialylated glucocerebrosidase was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN 3pH 7.2. This solution was incubated with 1mM CMP-sialic acid-linker-Man-6-phosphate and 0.1U/mL ST3Gal3 at 32 ℃ for 2 days. To monitor the integration of sialic acid-linker-Man-6-phosphate, small to the reactionAliquots were added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas TSK-gel-3000 analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. When the reaction was completed, the reaction mixture was purified with a Toso HaasTSK-gel-3000 preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
30. Glucocerebrosidase-transferrin
This example presents the procedure for glycoconjugation of proteins, in particular transferrin to glucocerebrosidase. A GlcNAc-ASN structure is generated on glucocerebrosidase and transferrin-SA-linker-Gal-UDP is conjugated to the GlcNAc-ASN structure of GDNF using galactosyltransferase.
GlcNAc-glucocerebrosidase (Cerezyme) TM ) And (4) preparing.Cerezyme produced in CHO cellsTM(glucocerebrosidase) 2.5mg/mL dissolved in 50mM Tris-HClpH7.4, 0.15M NaCl, and 300mU/mL Endo-H-agarose conjugate at 32 degrees C temperature in 16 hours. To monitor the reaction, small aliquots of the reaction were diluted with the appropriate buffer and IEF gels and SDS-PAGE were run according to the Invitrogen program. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The beads were washed 3 times with Tris-EDTA buffer, once with 0.4mL Tris-EDTA buffer and once with 0.2mL Tris-EDTA buffer, and all supernatants were collected. The supernatant was incubated at 4 ℃ with 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialyzed, and then against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialysis was performed twice. The dialyzed solution was then concentrated using a Centricon Plus 20 centrifugal filter. The reaction product was according to the procedure provided by Invitrogen andreagents were analyzed by SDS-PAGE and IEF analysis. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of transferrin-SA-linker-Gal-glucocerebrosidase.The transferrin-SA-adaptor-Gal-UDP was dissolved in 50mM Tris-HCl, 0.15M NaCl, 5mM MnCl at 2.5mg/mL 2、0.05% NaN3pH 7.2. The solution was incubated with 2.5mg/mL GlcNAc-glucocerebrosidase and 0.1U/mL galactosyltransferase for 2 days at 32 ℃. To monitor the integration of glucocerebrosidase, the peptides were isolated by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph7.1) and the product probed by UV absorption. The reaction mixture was then purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
GM-CSF
31.Generation and PEGylation of GlcNAc-ASN structure: saccharomyces (Saccharomyces) GM-CSF produced in (1)
This example presents the preparation of a tissue-type activator with a PEGylated GlcNAc-Asn structure.
Recombinant GM-CSF expressed in yeast is expected to contain 2N-linked and 2O-linked glycans. The N-linked glycans should be of the branched mannose type. The recombinant glycoprotein was treated with an endoglycosidase selected from endoglycosidase H, endoglycosidase-F1, endoglycosidase-F2, endoglycosidase-F3, endoglycosidase-M, either alone or in combination with mannosidase I, II and III to produce GlcNAc nodules at asparagine (Asn) residues on the peptide/protein backbone.
The GlcNAc-Asn structure on the peptide/protein backbone can then be modified with UDP-galactose or UDP-galactose-6-PEG and a galactosyltransferase such as GalT1, respectively, for galactose or galactose-PEG. In one instance, galactose-PEG is a terminal residue. In the second case, galactose can be further modified with SA-PEG using a CMP-SA-PEG donor and a sialyltransferase such as ST3 GalIII. In another embodiment, the GlcNAc-Asn structure on the peptide/protein backbone can be galactosylated and sialylated as described above, followed by further sialylation with CMP-SA-PEG and α 2, 8-sialyltransferase, such as the enzyme encoded by the cst-II gene of Campylobacter jejuni subspecies jejuni.
HerceptinTM
32.Mithramycin andHerceptinTM glycoconjugation of
This example presents the procedure for glycoconjugation of small molecules such as mithramycin to the glycans of the Fc region of antibody molecules produced in mammalian cells. Here, the antibody Herceptin is usedTMHowever, one skilled in the art will appreciate that the method is applicable to many other antibodies.
Herceptin TM -preparation of Gal-linker-mithramycin.HerceptinTM2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 5nM MnCl2、0.05% NaN3pH 7.2. This solution was incubated with 1mM UDP-galactose-linker-mithramycin and 0.1U/mL galactosyltransferase at 32 ℃ for 2 days to introduce mithramycin in the glycans of the Fc region. To monitor the incorporation of galactose, small aliquots of the reactants were added 14A C-galactose-UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The radiolabel incorporated into the peptide was quantified using a built-in radioactivity detector.
When the reaction was completed, the reaction mixture was purified with a TosoHaas TSK-gel-3000 preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The fractions containing the product were combined, concentrated, buffer exchanged, and then lyophilized. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Interferon alpha and interferon beta
33.Glycopegylation of proteins expressed in mammalian or insect systems: EPO and interferon Alpha and interferon beta
This example presents the preparation of pegylated peptides expressed in mammalian and insect systems.
Preparation of receptors from mammalian expression systems.Peptides that are glycoPEGylated with CMP-sialic acid PEG need to have glycans that terminate with galactose. Most peptides from mammalian expression systems will have terminal sialic acids that need to be removed first.
And (4) digesting with sialidase.The peptides were desialylated with sialidase. The general procedure includes: the peptide solution at 1mg/mL was supplemented with 5mM CaCl2Is incubated with 0.2U/mL of immobilized sialidase (Calbiochem) from Vibrio cholerae for 24 hours at 32 ℃. Microbial growth can be stopped by sterile filtration or by inclusion of 0.02% sodium azide. The resin is then removed by centrifugation or filtration and washed to recover the captured peptide. At this point, EDTA may be added to the solution to inhibit any sialidase that leaks out of the resin.
A preparation from an insect expression system.EPO, interferon alpha and interferon beta may also be expressed in stem non-mammalian systems, such as yeast, plant or insect cells. Peptides that are glycoPEGylated with CMP-sialic acid PEG need to have glycans that terminate with galactose. Most glycans expressed on peptides in insect cells are, for example, the trimannose core. These glycans were first constructed as galactose-terminated glycans before becoming receptors for sialyltransferases.
From the mannose coreReceptor glycans were constructed.Peptide (1mg/mL) was dissolved in a solution containing 5mM MnCl25mM UDP-GlcNAc, 0.05U/mL GLCNACTI, 0.05U/mL GLCNACTII in Tris-buffered salt pH7.2 at 32 ℃ for 24 hours or until the reaction is substantially complete. Microbial growth can be stopped by sterile filtration or by inclusion of 0.02% sodium azide. After buffer exchange to remove UDP and other small molecules, UDP-galactose and MnCl were added 2Each to 5mM, galactosyltransferase was added to 0.05U/mL and incubated at 32 ℃ for 24 hours or until the reaction was substantially complete. Microbial growth can be stopped by sterile filtration or by inclusion of 0.02% sodium azide. The peptide can then be used to perform glycoPEGylation.
O-linked glycans were constructed.Similar strategies can be applied to interferon alpha to enzymatically produce the desired O-glycan Gal-GalNAc. If necessary, GalNAc linked to serine or threonine can be added to the peptide using an appropriate peptide GalNAc transferase (e.g., GalNAc Tl, GalNAc T2, T3, T4, etc.) and UDP-GalNAc. Likewise, galactose may be added using galactosyltransferase and UDP-galactose, if desired.
The sugar was PEGylated with sialyltransferase.Tris buffered saline + glycopeptides with a terminal galactose (1mg/mL) in 0.02% sodium azide were incubated with CMP-SA-PEG (0.75mM) and 0.4U/mL sialyltransferase (ST 3Gal3 or ST3Gal4 for EPO and N-glycans on interferon beta and ST3Gal4 or ST3Gal1 for O-glycans on interferon alpha) at 32 ℃ for 24 hours. Other useful transferases include α 2, 6 sialyltransferase from photobacterium mermaisonii (photobacterium damsella). The receptor peptide concentration is most preferably 0.1mg/mL to the solubility limit of the peptide. The concentration of CMP-SA-PEG should be sufficient to exceed available sites, but should not be so high as to cause peptide solubility problems (due to PEG), and can be 50. mu.M to 5mM, and the temperature can be 2 ℃ to 40 ℃. The time required to complete the reaction will depend on the temperature, the relative amount of enzyme to acceptor substrate, the concentration of donor substrate, and the pH.
34.Glycopegylation of interferon alpha produced in CHO cells
Preparation of desialylated-interferon alpha.Interferon alpha produced from CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl pH7.4, 0.15M NaCl, 5mM CaCl2And concentrated to 500. mu.L in a Centricon Plus 20 centrifugal filter. The solution was incubated with 300mU/mL neuraminidase II (Vibrio cholerae) for 16 hours at 32 ℃. To monitor the reaction, a small aliquot of the reaction was diluted with the appropriate buffer and an IEF gel analysis was run. The reaction mixture was then added to a pre-washed N- (p-aminophenyl) oxamic acid-agarose conjugate (800. mu.L/mL reaction volume) and the washed beads were gently rotated at 4 ℃ for 24 hours. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The beads were washed 3 times with Tris-EDTA buffer, once with 0.4mL Tris-EDTA buffer and once with 0.2mL Tris-EDTA buffer, and all supernatants were collected. The supernatant was incubated at 4 ℃ with 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialyzed, and then against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialysis was performed twice. The dialyzed solution was then concentrated with a Centricon Plus 20 centrifugal filter and stored at-20 ℃. The conditions of the IEF gel were run according to the procedures and reagents provided by Invitrogen. The native and desialylated G-CSF samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of interferon alpha- (. alpha.2, 3) -sialic acid-PEG.The desialylated interferon alpha was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-PEG and 0.1U/mL ST3Gal1 at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction product was according to the program provided by InvitrogenSequences and reagents were analyzed by SDS-PAGE and IEF analysis. Native and desialylated interferon alpha samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of interferon alpha- (. alpha.2, 8) -sialic acid-PEG.Interferon alpha containing alpha 2, 3-sialylated O-linked glycan produced in CHO cells was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN 3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-PEG and 0.1U/mL CST-II at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso HaasG3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The native and pegylated interferon alfa samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of interferon alpha- (. alpha.2, 6) -sialic acid-PEG.Interferon alpha containing only O-linked GalNAc was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with 1mM CMP-sialic acid-PEG and 0.1U/mL ST6GalNAcI or II at 32 ℃ for 2 days. To monitor the incorporation of sialic acid-PEG, a small aliquot of the reaction was added with CMP-SA-PEG-fluorescent ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The fluorescent label incorporated into the peptide is quantified using a built-in fluorescence detector. After 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were fractionated by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen And (4) performing precipitation. The native and pegylated interferon alfa samples were dialyzed against water and analyzed by MALDI-TOFMS.
35.PEGylation of interferon-beta-1 a saccharides with PEG (10kDa) and PEG (20kDa)
This example illustrates the procedure for PEGylation of interferon-beta with PEG (10kDa) or PEG (20 kDa).
Briefly, interferon-beta-1 a (INF-. beta.) was purchased from Biogen (Avonex)TM). INF-. beta.was first purified by Superdex-75. INF- β is then desialylated with Vibrio cholerae sialidase. INF-. beta.was then PEGylated with SA-PEG (10kDa) and SA-PEG (20kDa) and purified with Superdex-200.
Superdex-75 chromatographic purificationINF-. beta.was applied (150. mu.g) to a Superdex-75 column (Amersham Biosciences, Arlington Heights, IL) and eluted with PBS with 0.5M NaCl, 0.02 Tween-20, 20mM histidine and 10% glycerol. The absorbance of the eluate at 280nm was monitored (FIGS. 172A and 172B), and fractions were collected. Peaks 4 and 5 were pooled, concentrated on Amicon Ultra 15 spin filters (Millipore, Billerica, MA) and buffer exchanged with 0.5M CaCl2TBS of 0.02% Tween-20, 20mM histidine and 10% glycerol.
Sialidase reactionThen in the presence of 5mM CaCl20.02% Tween-20, 20mM histidine and 10% glycerol in TBS was purified by treatment with Vibrio cholerae sialidase (70mU/ml, EMD Biosciences, inc., San Diego, CA) desialyzed INF-beta. The reaction was carried out at 32 ℃ for 18 hours. With 0.22 μm Spin-XTMFilters (Corning technology, Inc., Norcross, GA) obtained INF-. beta.from agarose. FIG. 173A depicts MALDI analysis of glycans released from native INF- β. Native INF- β has many glycoforms with terminal sialic acid moieties. FIG. 173B depicts MALDI analysis of glycans released from asialo INF- β. asialo-INF-beta mainly has oneA biantennary saccharide form containing a terminal sialic acid moiety.
Sialylated lectin dot blot analysisDot blots of INF-. beta.samples from the desialylase reaction on nitrocellulose and blocking with Tris buffered saline (TBS: 0.05M Tris, 0.15M NaCl, pH7.5) and DIG kit (glycan discrimination kit from Roche # 1210238) blocking buffer. Some blots were incubated with Huai-Huai agglutinin (MAA) labeled with Digoxigenin (DIG) (Roche Applied Science, Indianapolis, IL) to detect α 2, 3-sialylation of INF- β. These blots were washed with TBS, then incubated with digitonin labeled with alkaline phosphatase, then washed again with TBS and developed with NBT/X-phosphate solution, where NBT is 4-nitro blue tetrazolium chloride and X-phosphate is 5-bromo-4-chloro-3 indolyl phosphate. The left side of FIG. 174 depicts the results of MAA blotting of INF-. beta.after the desialylation reaction. INF- β is partially desialylated, as shown by the reduction in blot development compared to native INF- β in desialylated samples.
Other blots were incubated with Erthrina Cristagalli Lectin (ECL) labeled with biotin (Vector Laboratories, Burlingame, Calif.) to detect exposed galactose residues on INF- β. After incubation with 2.5. mu.g/ml ECL, the blot was washed in TBS and incubated with streptavidin labeled with alkaline phosphatase. The blot was then washed again and developed. The right side of figure 174 depicts ECL blots after development. The imprinted increased intensity of desialylated INF-beta compared to native INF-beta indicates more exposed galactose moieties and thus sufficient desialylation.
PEGylation of desialylated INF-beta with SA-PEG (10kDa)PEGylation of desialylated INF-beta (0.05mg/ml) with ST3Gal3(50mU/ml) and CMP-SA-PEG (10kDa) (250. mu.M) in an appropriate buffer TBS +5mM CaCl for 50 h at 32 ℃20.02% Tween 20, 20mM histidine, 10% glycerol. FIG. 175 depicts the reverseSDS-PAGE analysis of the reaction product showed PEGylated INF- β of approximately 98 kDa.
PEGylation of desialylated INF-beta with SA-PEG (20kDa)PEGylation of desialylated INF-beta (0.5mg/ml) with ST3Gal3(l70mU/ml) and CMP-SA-PEG (20kDa) in an appropriate buffer TBS +5mM CaCl at 32 ℃ for 50 h 20.02% Tween 20, 20mM histidine, 10% glycerol. FIG. 176 depicts SDS-PAGE analysis of PEGylation reaction products. PEGylated INF- β has many higher molecular weight bands not seen in unmodified INF- β, indicating that it is more fully PEGylated.
Superdex-200 purification of INF-. beta.PEGylated with PEG (10kDa)The PEGylated reaction product was isolated on a Superdex-200 column (Amersham Biosciences, Arlington heights, IL) using PBS with 0.5M NaCl, 0.02 Tween-20, 20mM histidine and 10% glycerol at a flow rate of 1 ml/min and 30 cm/h. The absorbance of the eluate at 280nm was monitored (FIG. 177) and fractions were collected. Peaks 3 and 4 were pooled and concentrated on AmiconUltra 15 rotary filters.
Bioassay of INF- β PEGylated with PEG (10 kDa).
This assay is the inhibition of the proliferation of the lung cancer cell line a 549. The A549 cell line was 5% CO at 37 ℃2Lung cancer adherent cells grown in RPMI + 10% FBS. They are available from ATCC # CCL-185. Cells were washed with 10ml PBS and PBS was removed. 5ml trypsin was added and incubated at room temperature for 5 minutes or at 37 ℃ for 2 minutes. When the cells were detached from the wall, they were resuspended in 25ml of medium and the cells were counted. Cells were diluted at a concentration of 10000 cells/ml and added at 200. mu.l/well (96 well plate). 5% CO at 37 ℃ 2Incubate for 4 hours. 1ml of INF-. beta.was prepared at a concentration of 0.1. mu.g/ml. Filtration was performed under a fume hood using a 0.2 μm filter. Add 100 μ l per well (8 replicates to 1 lane). Incubate for 3 days (without confluency of cells). 200. mu.l of medium was removed (100. mu.l of medium remained in each well). Mu.l MTT (Sigma) (5mg/ml, 0.22 μm filtration) was added. At 37 ℃ and 5% CO2Incubate for 4 hours. The medium was gently aspirated and a mixture of 100. mu.l isopropanol (100ml) and 6N HCl was added. Pump up and down to homogenize the crystal violet. Read 570
OD at nm (background at 630 or 690nm removed).
FIG. 178 depicts the bioassay of the peak eluted from Superdex-200 column containing INF- β PEGylated with PEG (10 kDa).
Superdex-200 purification of INF-. beta.PEGylated with PEG (20kDa)PEG (20kDa) PEGylation reaction product was isolated on a Superdex-200 column (Amersham Biosciences, Arlington Heights, IL) using PBS with 0.5NaCl, 0.02Tween-20, 20mM histidine and 10% glycerol at a flow rate of 1 ml/min. The absorbance of the eluent at 280nm was monitored (FIG. 179) and fractions were collected. Peak 3 contains most of the INF- β PEGylated with PEG (20 kDa).
Endotoxin assay with PEG (20kDa) PEGylated INF-beta.
Limulus lysate assay, Bio Whittaker #50-647U
TABLE 24 endotoxin assay results for INF- β PEGylated with PEG (20 kDa).
Remicade
TM
36.Remicade
TM
Sugar PEGylation of antibodies
This example presents the procedure for glycopegylation of recombinant antibody molecules by introducing PEG molecules into the glycans of the Fc region. Here, RemicadeTMI.e., TNF-R: IgG Fc region fusion proteins, are exemplary peptides.
Remicade TM Preparation of Gal-PEG (10 kDa).Will RemicadeTM2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 5mM MnCl2、0.05% NaN3pH 7.2. This solution was incubated with 1mM UDP-galactose-PEG (10kDa) and 0.1U/mL galactosyltransferase at 32 ℃ for 2 days to introduce PEG in the glycans of the Fc region. To monitor the incorporation of galactose, small aliquots of the reactants were added14A C-galactose-UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The radiolabel incorporated into the peptide was quantified using a built-in radioactivity detector.
When the reaction was complete, the reaction mixture was purified with a TosoHaas TSK-gel-3000 preparative column using PBS buffer (ph7.l) and fractions were collected based on UV absorption. The fractions containing the product were combined, concentrated, buffer exchanged, and then lyophilized. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Rituxan
TM
37.Geldanamycin and Rituxan TM Glycoconjugation of
This example presents the use of small molecules such as geldanamycin with antibodies produced in CHO cells such as RituxanTMThe Fc region glycans of (a) are subjected to a glycoconjugation procedure. Here, the antibody Rituxan was usedTMHowever, one skilled in the art will appreciate that the method is applicable to many other antibodies.
Rituxan TM Preparation of Gal-linker-geldanamycin.RituxanTM2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 5mM MnCl2、0.05% NaN3pH 7.2. The solution was incubated with 1mM UDP-galactose-linker-geldanamycin and 0.1U/mL galactosyltransferase at 32 ℃ for 2 days to introduce geldanamycin into the glycans of the Fc region. To monitor the incorporation of galactose, small aliquots of the reactants were added14A C-galactose-UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso HaasG3000SW analytical column using PBS buffer (ph 7.1). The radiolabel incorporated into the peptide was quantified using a built-in radioactivity detector.
When the reaction was completed, the reaction mixture was purified with a TosoHaas TSK-gel-3000 preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The fractions containing the product were combined, concentrated, buffer exchanged, and then lyophilized. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Rnase
38.Reconstitution of high mannose N-glycans as hybrid and complex N-glycans: bovine pancreas RNase
This example presents the preparation of bovine pancreatic RNase with heterozygous or complex N-glycans. The high mannose N-linked glycans of the RNase are enzymatically digested and processed to generate hybrid N-linked glycans. In addition, RNase high mannose N-linked glycans can be enzymatically digested and processed to generate complex N-linked glycans.
The high mannose structure of N-linked oligosaccharides in glycopeptides can be modified to a hybrid or complex form using a combination of alpha-mannosidase and glycosyltransferase. This example summarizes the results of such efforts using simple N-glycans as model substrates.
Ribonuclease B (RNaseB) (Sigma) purified from bovine pancreas was a glycopeptide consisting of 124 amino acid residues. It has a single potential N-glycosylation site modified by a high mannose structure. Because of its simplicity and low molecular weight (13.7-15.5 kDa), ribonuclease B is a good candidate demonstrating the possibility of remodeling from a high mannose structure to N-glycans of hybrid or complex N-linked oligosaccharides. MALDI-TOF spectra of RNaseB (FIG. 180A) and HPLC plots of oligosaccharides cleaved from RNaseB by N-glycanase (FIG. 180B) show that: most of the N-glycosylation sites of the peptide, except for a small fraction of unmodified peptide, were modified with high mannose oligosaccharides consisting of 5-9 mannose residues.
Conversion of high mannose N-glycans to hybrid N-glycans.High mannose N-glycans were converted to hybrid N-glycans with a combination of α 1, 2-mannosidase, glcnactt-I (β -1, 2-N-acetylglucosaminyltransferase), GalT-I (β 1, 4-galactosyltransferase), and α 2, 3-sialyltransferase/or α 2, 6-sialyltransferase, as shown in figure 181.
As an example, the high mannose structure in rnase b was successfully transformed into a heterozygous structure.
Man5GlcNAc2-R is the catalytic Man of a single alpha 1, 2-mannosidase cloned from Trichoderma reesei5-9GlcNAc2-R obtained (fig. 182). RNaseB (1g, about 67. mU. mol) was incubated with 15mU of recombinant Trichoderma reesei α 1, 2-mannosidase in MES buffer (50mM, pH6.5) in a total volume of 10mL at 30 ℃ for 45 hours. Man6-9GlcNAc2The protein structure has been successfully converted to Man with high efficiency using recombinant mannosidase5GlcNAc2-a protein.
Alternatively, Man5GlcNAc2R is the catalytic Man with a single alpha 1, 2-mannosidase purified from Aspergillus saitoi5-9GlcNAc2-R (fig. 183). RNaseB (40. mu.g, ca. 2.7nmol) was incubated with 25. mu.U of a 1, 2-mannosidase from commercial A.saitoi (Glyko or CalBiochem) in NaOAC buffer (100mM, pH5.0) in a total volume of 20. mu.L for 42.5 hours at 37 ℃. Man 6-9GlcNAc2Protein Structure has been successfully converted to Man using commercially available mannosidase5GlcNAc2-a protein.However, a new peak corresponding to the GlcNAc-protein appeared in the spectrum, indicating possible contamination of endoglycosidase H in the preparation. Although several mammalian alpha-mannosidases are required to achieve this step, fungal alpha 1, 2-mannosidases are very effective at removing all alpha 1, 2-linked mannose residues.
GlcNAcT-I then adds a GlcNAc residue to Man5GlcNAc2-R (FIG. 184). After the alpha 1, 2-mannosidase reaction of Trichoderma reesei, the reaction mixture containing RNaseB (600. mu.g, about 40nmol) and non-purified recombinant GlcNAcT-I (34mU) was incubated at 37 ℃ for 42 hours in a total volume of 400. mu.L in MES buffer (50mM, pH6.5) containing MnCl2(20mM) and UDP-GlcNAc (5 mM). GlcNAc residues are quantitatively added to Man by recombinant GlcNAcT-I5GlcNAc2-in proteins.
Gal residues were then added with GalT1 (fig. 185). After the GnT-I reaction, the reaction mixture containing RNaseB (120. mu.g, about 8nmol) was incubated with 3.3mU of recombinant GalT-1 in Tris-HCl buffer (100mM, pH7.3) containing UDP-Gal (7.5mM) and MnCl in a total volume of 100. mu.L at 37 ℃ for 20 hours 2(20 mM). Gal residues are GlcNAc-Man added by recombinant GalT1 to about 98%5GlcNAc2-in proteins.
The next step is the addition of sialic acid with either α 2, 3-sialyltransferase or α 2, 6-sialyltransferase (fig. 186). By way of example, ST3GalIII, an α 2, 3-sialyltransferase, is used. After the GalT-1 reaction, the reaction mixture containing RNaseB (13. mu.g, about 0.87nmol) was incubated with 8.9mU of recombinant ST3GalIII in Tris-HCl buffer (100mM, pH7.3) containing CMP-sialic acid (5mM) and MnCl for 16 hours at 37 ℃ in a total volume of 20. mu.L2(20 mM). Sialic acid residues were added from recombinant ST3GalIII to about 90% of Gal-GlcNAc-Man using CMP-SA as donor5GlcNAc2-in proteins. The yield can be further improved by adjusting the reaction conditions.
For convenience, in each of the aboveNo purification or dialysis is required after the reaction. More interestingly, GnT-I and ST3GalIII can be combined in one (one pot) reaction. Similar yields can be obtained compared to isolated reactions. After the GlcNAcT-I reaction, the reaction mixture containing RNaseB (60. mu.g, about 4nmol) was incubated with 1.7mU of recombinant GalT1, 9.8mU of recombinant ST3Gal III in Tris-HCl buffer (100mM, pH7.3) containing UDP-Gal (7.5mM), CMP-sialic acid (5mM) and MnCl in a total volume of 60. mu.L at 37 ℃ for 20 hours 2(20mM)。
As shown in FIG. 187, SA-PEG (10kDa) was successfully added to RNaseB. Reaction mixture containing RNaseB (6.7. mu.g, ca. 0.45nmol) was reacted with H at room temperature after GalT-1 reaction2O dialysis for 1 hour, and incubation with 55mU of recombinant ST3GalIII in Tris-HCl buffer (50mM, pH7.3) containing CMP-SA-PEG (10kDa) (0.25mM) and MnCl at 37 ℃ for 15.5 hours in a total volume of 20. mu.L2(20 mM). PEG-modified sialic acid residues were successfully added to Gal-GlcNAc-Man by recombinant ST3GalIII5GlcNAc2-in a peptide. The yield can be further improved by adjusting the reaction conditions.
Conversion of high mannose N-glycans to complex N-glycans.To achieve this transition, GlcNAc β 1, 2Man is obtained3GlcNAc2-a peptide intermediate. As shown in FIG. 188, at least 4 possible routes are available from Man5GlcNAc2-reaction of the peptide to the intermediate:
route I: man produced by fungal alpha 1, 2 mannosidase5GlcNAc2The peptide is a substrate for GlcNAc transferase I (GlcNAcT-I, enzyme 2) which adds a GlcNAc. GlcNAcMan5GlcNAc2The terminal α 1, 3-and α 1, 6-linked mannose residues of the peptide can be removed by golgi α mannosidase II (ManII, enzyme 5). This route is part of the natural pathway for processing N-linked oligosaccharides that occurs in higher organisms.
Route II: first 2 mannose residues are removed with alpha mannosidase (enzyme 6) and then GlcNAc is added with GlcNAcT-I (enzyme 2). Except for its natural receptorMan5GlcNAc2besides-R, GlcNAcT-I can also recognize Man3GlcNAc2-R as its substrate and adding a GlcNAc to the mannose core structure to form GlcNAcMan3GlcNAc2-a peptide.
Route III: the α 1, 6-linked mannose is removed by α 1, 6-mannosidase, followed by GlcNAc addition by GlcNAcT-I and removal of the terminal α 1, 3-linked mannose by α 1, 3-mannosidase. From the experimental data obtained, GlcNAcT-I can identify the Man4GlcNAc2-peptide as acceptor and addition of a GlcNAc residue to form GlcNAcMan4GlcNAc2-a peptide.
Route IV: similar to scheme III, the α 1, 3-linked mannose is removed by α 1, 3-mannosidase followed by a GlcNAcT-I reaction. The terminal α 1, 6-linked mannose can then be removed by α 1, 6-mannosidase.
GlcNAc following interaction of GlcNAcT-I (responsible for adding GlcNAc linked to α 1, 3- mannose β 1, 2 on the mannose core) and GlcNAcT-II (responsible for adding GlcNAc linked to α 1, 6- mannose β 1, 2 on the mannose core) GlcNAc2Man3GlcNAc2The peptides can be processed by GalT1 and sialyltransferase to form bi-antennary complex N-glycans. Other GlcNAc transferases such as GlcNAcT-IV, GlcNAcT-V, and/or GlcNAcT-VI (FIGS. 188 and 189) can also be directed against GlcNAc 2Man3GlcNAc2-glycosylation of the peptide. Additional glycosylation by GalT1 and sialyltransferase will form multi-antennary complex N-glycans. The enzyme GlcNAcT-III catalyzes the insertion of bisecting GlcNAc, thereby preventing the action of ManII and the subsequent action of the transferases GlcNAcT-II, GlcNAcT-IV and GlcNAcT-V.
tissue-Type Plasminogen Activator (TPA)
39.Fucosylation of TPA to produce sialylated Lewis X
This example presents the preparation of Tissue Plasminogen Activator (TPA) with N-linked sialylated Lewis X antigen.
TPA expressed in mammalian cells often contains most of the glycans that end with sialic acid, but in order to ensure complete sialylation, it is advantageous to first perform in vitro sialylation. TPA in a suitable buffer (most preferably at pH 5.5-9, e.g., Tris buffered salt, pH7.2) is incubated with CMP sialic acid and sialyltransferase for a sufficient time to convert any sialic acid deficient glycans to sialylated species. General conditions were 1mg/mL TPA, 3mM CMP sialic acid, 0.02U/mL ST3Gal3, at 32 ℃ for 24 hours. Microbial growth can be inhibited by sterile filtration or by inclusion of 0.02% sodium azide. The TPA concentration is most preferably from 0.1mg/mL to the solubility limit of the peptide. The concentration of CMP-SA should be sufficient to exceed the available sites, and may be between 50. mu.M and 50mM, and at a temperature of 2 ℃ to 40 ℃. The time required to complete the reaction will depend on the temperature, the relative amount of enzyme to acceptor substrate, the concentration of donor substrate, and the pH. Other sialyltransferases capable of adding sialic acid in 2, 3 linkages include ST3Gal 4; microbial transferases may also be used.
Fucosylation.The general conditions for fucosylation were 1mg/mL TPA, 3mM GDP-fucose, 0.02U/mL FTVI, 5mM MnCl2And in Tris buffered salt at 32 ℃ for 24 hours. Microbial growth can be inhibited by sterile filtration or by inclusion of 0.02% sodium azide. The TPA concentration is most preferably from 0.1mg/mL to the solubility limit of the peptide. The concentration of GDP-fucose should be sufficient to exceed the available sites and may be between 50. mu.M and 50mM, and the temperature is between 2 ℃ and 40 ℃. The time required to complete the reaction will depend on the temperature, the relative amount of enzyme to acceptor substrate, the concentration of donor substrate, and the pH. Other fucosyltransferases that can produce sialylated Lewis X include FTVII, FTV, FTIII, and microbial transferases may also be used.
40.Pruning high mannose to a trimannose core structure: tissue produced in CHOModel (III)Plasmin A pro-activator.
This example proposes the preparation of tissue plasminogen activator with a trimannose core by trimming high mannose glycans.
Preparation of Tissue Plasminogen Activator (TPA) is currently produced in Chinese Hamster Ovary (CHO) cells and contains low amounts of high mannose N-linked oligosaccharides. The mannose may be trimmed with various specific mannosidases. The first step is to generate Man5GlcNAc2(Fuc0-1) from Man9GlcNAc2(Fuc 0-1). This can be done with mannosidase I. GlcNAcT1(GlcNAc transferase I) was then used to make GlcNAc1Man5GlcNAc2(Fuc0-1) or mannosidase III was used to make Man3GlcNAc2(Fuc 0-1). GlcNAc1Man3GlcNAc2(Fuc0-1) can be produced from Man3GlcNAc2(Fuc0-1) using GlcNAcT1, or GlcNAc1Man3GlcNAc2(Fuc0-1) can be produced from GlcNAc1Man5GlcNAc2(Fuc0-1) using mannosidase II. GlcNAc1Man3GlcNAc2(Fuc0-1) was then converted to GlcNAc2Man3GlcNAc2(Fuc0-1) by GlcNAc transferase II (GlcNAcTII). The two terminal GlcNAc residues can then be galactosylated with GalTI and then sialylated with SA-PEG with ST3Gal III.
Conversely, TPA can be produced in yeast or fungal systems. A similar process would be required for fungal derived materials.
41.Generation and PEGylation of GlcNAc-ASN structure: TPA produced in yeast
This example presents the preparation of a PEGylated GlcNAc-Asn structure on a peptide such as TPA expressed in yeast.
Yeast expression can yield TPA containing a single N-linked mannose-type structure. The recombinant glycoprotein is first treated with endoglycosidase H to produce a GlcNAc structure at an asparagine (Asn) residue on the peptide.
The GlcNAc-Asn structure on the peptide/protein backbone is then modified with UDP-galactose or UDP-galactose-6-PEG and a galactosyltransferase such as GalT1 for galactose or galactose-PEG, respectively. In one instance, galactose-PEG is a terminal residue. In the second case, galactose can be further modified with SA-PEG using a CMP-SA-PEG donor and a sialyltransferase such as ST3Gal III. In another embodiment, the GlcNAc-Asn structure on the peptide/protein backbone can be galactosylated and sialylated as described above, followed by further sialylation with CMP-SA-PEG and α 2, 8-sialyltransferase, such as the enzyme encoded by the cst-II gene of Campylobacter jejuni subspecies jejuni.
Transferrin
42.Glycopegylation of transferrin
This example presents the preparation of desialylated transferrin and sialylation with PEG-CMP-sialic acid.
Preparation of desialylated transferrin.Intact transferrin (10mg) of human origin was dissolved in 500. mu.L of 50mM NaOAc, 5mM CaCl2pH 5.5. To this solution was added 500mU of neuraminidase II (Vibrio cholerae), and the reaction mixture was gently shaken at 37 ℃ for 20.5 hours. The reaction mixture was added to the previously washed N- (p-aminophenyl) oxamic acid-agarose conjugate (600. mu.L) and the washed beads were gently rotated at 4 ℃ for 24 hours. The mixture was centrifuged at 10,000rpm and the supernatant was collected. The reaction mixture was adjusted to 5mM EDTA by adding 100. mu.l of 30mM EDTA to the washed beads, which were then gently spun at 4 ℃ for 20 hours. The suspension was centrifuged at 10,000rpm for 2 minutes, and the supernatant was collected. The beads were incubated with 0.35mL of 50mM NaOAc, 5mM CaCl25mM EDTA, pH5.5 washing 5 times and collecting all supernatant. The enzyme solution was dialyzed twice at 4 ℃ against 15mM Tris-HCl, 1M NaCl, pH 7.4. 0.3mL of transferrin solution (3.3 mL total) was taken and dialyzed against water twice. The residue was dialyzed against phosphate buffered saline twice again at 4 ℃. The dialyzed solution was stored at-20 ℃. Protein samples were analyzed by IEF electrophoresis. Samples (9. mu.L, 25. mu.g) were diluted with 16. mu.L Tris buffer and mixed with 25. mu.L sample loading buffer and applied to Isoelectric Focusing Gels (pH 3-7). The gel was run and fixed using standard procedures. The gel was stained with Colloidal Blue Stain.
Saliva removalSialic acid-pegylation of acidified transferrin.Desialylated transferrin (250. mu.g) and CMP-sialic acid or CMP-SA-PEG (1kDa or 10kDa) (0.05. mu. mol) were dissolved in 69. mu.L of 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN in a 1.5mL plastic tube3pH 7.2. Tubes were briefly vortexed and 100mU ST3Gal3 (90. mu.L) was added (250. mu.L total volume). The tube was vortexed again and gently mixed at 32 ℃ for 24 hours. The reaction was stopped by freezing at-80 ℃. Novex Tris-glycine 8-16% 1mm gels were used for SDS-PAGE analysis (FIG. 190). Samples (25. mu.L, 25. mu.g) were mixed with 25. mu.L of sample loading buffer and 0.4. mu.L of beta-mercaptoethanol and heated at 85 ℃ for 6 minutes. Gels were run using standard conditions and stained with Colloidal Blue Stain. IEF gels were also performed as described above (fig. 191). The samples were also dialyzed against water and analyzed by MALDI-TOF.
And (6) obtaining the result.MALDI was also performed. Natural transferrin (78729); desialylated transferrin (78197); resialylated transferrin (79626/80703); has SA-PEG1k (79037 (1); 80961 (2); 82535 (3); 84778 (4)); has SA-PEG5k (90003 (2); 96117 (3); 96117 (4)); has SA-PEG10k (100336 (2); 111421 (3); 122510 (4)).
43.transferrin-GDNF
This example presents procedures for glycoconjugation of proteins, in particular transferrin to GDNF. transferrin-SA-linker-Gal-UDP is prepared from transferrin. Galactose residues were removed from GDNF glycans, and transferrin-SA-linker-Gal-UDP was conjugated to GDNF glycans with galactosyltransferases.
Preparation of degalactosylated (agalacto) -GDNF.GDNF produced in NSO cells (NSO murine myeloma cells) was dissolved at 2.5mg/mL in 50mM Tris-HCl pH7.4, 0.15M NaCl and incubated with 300mU/mL beta-galactosidase-agarose conjugate for 16 hours at 32 ℃. To monitor the reaction, small aliquots of the reaction were diluted with the appropriate buffer and IEF gel analysis was run according to the Invitrogen program. Mixing the mixtureCentrifuged at 10,000rpm and the supernatant was collected. The supernatant was incubated at 4 ℃ with 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialyzed, and then against 50mM Tris-HCl pH7.4, 1M NaCl, 0.05% NaN3Dialysis was performed twice. The dialyzed solution was then concentrated with a Centricon Plus 20 centrifugal filter and stored at-20 ℃. The conditions of the IEF gel were run according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of transferrin-SA-linker-Gal-UDP.The desialylated transferrin was dissolved at 2.5mg/mL in 50mM Tris-HCl, 0.15M NaCl, 0.05% NaN3pH 7.2. The solution was incubated with CMP-sialic acid-linker-Gal-UDP (moles total 1 mole equivalent of nucleotide sugar added to transferrin) and 0.1U/mL of ST3Gal3 at 32 ℃ for 2 days. To monitor sialic acid incorporation, small aliquots of the reaction were added14A C-SA-UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso HaasG3000SW analytical column using PBS buffer (ph 7.1). The radiolabel incorporated into the peptide was quantified using a built-in radioactivity detector.
The solution was incubated with 5mM CMP-sialic acid and 0.1U/mL ST3Gal3 (to cap any unreacted transferrin glycan) for 2 days at 32 ℃. Integration into the peptide was quantified using a built-in UV detector. After 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
Preparation of transferrin-SA-linker-Gal-GDNF.transferrin-SA-adaptor-Gal-UDP prepared as above was dissolved in 50mM Tris-HCl, 0.15M NaCl, 5mM MnCl at 2.5mg/mL2、0.05% NaN3pH 7.2. This solution was transesterified with 2.5mg/mL degalactosylated-GDNF and 0.1U/mL galactosylThe transferases were incubated at 32 ℃ for 2 days. To monitor the incorporation of galactose, small aliquots of the reactants were added14C-galactose-UDP ligand; the label incorporated into the peptide was separated from the free label by gel filtration on a Toso Haas G3000SW analytical column using PBS buffer (ph 7.1). The radiolabel incorporated into the peptide was quantified using a built-in radioactivity detector.
When the reaction was complete, the solution was incubated with 5mM UDP-Gal and 0.1U/mL galactosyltransferase (to cap any unreacted transferrin glycan) for 2 days at 32 ℃ followed by the addition of 5mM CMP-SA and 0.1U/mL ST3Gal 3. After an additional 2 days, the reaction mixture was purified with a Toso Haas G3000SW preparative column using PBS buffer (ph7.1) and fractions were collected based on UV absorption. The reaction products were analyzed by SDS-PAGE and IEF analysis according to the procedures and reagents provided by Invitrogen. The samples were dialyzed against water and analyzed by MALDI-TOF MS.
The disclosures of each and every patent, patent application, and publication cited herein are hereby incorporated by reference in their entirety.
While the invention has been disclosed with respect to certain specific embodiments, it will be apparent that other embodiments and modifications of the invention can be devised by those skilled in the art without departing from the true spirit and scope of the invention. It is intended that the appended claims be construed to include all such embodiments and equivalent modifications.
Sequence listing
<110>Neose Technologies,Inc.
DeFrees,Shawn
Zopf,David
Bayer,Robert
Hakes,David
Chen,Xi
Bowe,Caryne
<120> sugar PEGylation method and protein/peptide produced by the same
<130>040853-01-5051WO
<150>US 60/328,523
<151>2001-10-10
<150>US 60/334,233
<151>2001-11-28
<150>US 60/334,301
<151>2001-11-28
<150>US 60/344,692
<151>2001-10-19
<150>US 60/387,292
<151>2002-06-07
<150>US 60/391,777
<151>2002-06-25
<150>US 60/396,594
<151>2002-07-17
<150>US 60/404,249
<151>2002-08-16
<150>US 60/407,527
<151>2002-08-28
<150>PCT/US02/32263
<151>2002-10-09
<150>US 10/360,779
<151>2003-02-19
<150>US 10/360,770
<151>2003-01-06
<150>US 10/287,994
<151>2002-11-05
<160>75
<170>PatentIn version 3.2
<210>1
<211>525
<212>DNA
<213>Homo sapiens
<400>1
<210>2
<211>174
<212>PRT
<213> Intelligent people
<400>2
<210>3
<211>1733
<212>DNA
<213> Intelligent people
<400>3
<210>4
<211>188
<212>PRT
<213> Intelligent people
<400>4
<210>5
<211>757
<212>DNA
<213> Intelligent people
<400>5
<210>6
<211>187
<212>PRT
<213> Intelligent people
<400>6
<210>7
<211>1332
<212>DNA
<213> Intelligent people
<400>7
<210>8
<211>444
<212>PRT
<213> Intelligent people
<400>8
<210>9
<211>1437
<212>DNA
<213> Intelligent people
<400>9
<210>10
<211>462
<212>PRT
<213> Intelligent people
<400>10
<210>11
<211>603
<212>DNA
<213> Intelligent people
<400>11
<210>12
<211>116
<212>PRT
<213> Intelligent people
<400>12
<210>13
<211>390
<212>DNA
<213> Intelligent people
<400>13
<210>14
<211>129
<212>PRT
<213> Intelligent people
<400>14
<210>15
<211>1342
<212>DNA
<213> Intelligent people
<400>15
<210>16
<211>193
<212>PRT
<213> Intelligent people
<400>16
<210>17
<211>435
<212>DNA
<213> Intelligent people
<400>17
<210>18
<211>144
<212>PRT
<213> Intelligent people
<400>18
<210>19
<211>501
<212>DNA
<213> Intelligent people
<400>19
<210>20
<211>166
<212>PRT
<213> Intelligent people
<400>20
<210>21
<211>1352
<212>DNA
<213> Intelligent people
<400>21
<210>22
<211>418
<212>PRT
<213> Intelligent people
<400>22
<210>23
<211>2004
<212>DNA
<213> Intelligent people
<400>23
<210>24
<211>536
<212>PRT
<213> Intelligent people
<400>24
<210>25
<211>1726
<212>DNA
<213> Intelligent people
<400>25
<210>26
<211>562
<212>PRT
<213> Intelligent people
<400>26
<210>27
<211>825
<212>DNA
<213> Intelligent people
<400>27
<210>28
<211>156
<212>PRT
<213> Intelligent people
<400>28
<210>29
<211>7931
<212>DNA
<213> Intelligent people
<400>29
<210>30
<211>2351
<212>PRT
<213> Intelligent people
<400>30
<210>31
<211>1471
<212>DNA
<213> Intelligent people
<400>31
<210>32
<211>461
<212>PRT
<213> Intelligent people
<400>32
<210>33
<211>1475
<212>DNA
<213> Intelligent people
<400>33
<210>34
<211>431
<212>PRT
<213> Intelligent people
<400>34
<210>35
<211>107
<212>PRT
<213>Mus musculus
<400>35
<210>36
<211>120
<212>PRT
<213>Mus musculus
<400>36
<210>37
<211>120
<212>PRT
<213>Mus musculus
<400>37
<210>38
<211>106
<212>PRT
<213>Mus musculus
<400>38
<210>39
<211>1039
<212>DNA
<213> Intelligent people
<400>39
<210>40
<211>282
<212>PRT
<213> Intelligent people
<400>40
<210>41
<211>678
<212>DNA
<213>Mus musculus
<400>41
<210>42
<211>226
<212>PRT
<213>Mus musculus
<400>42
<210>43
<211>450
<212>DNA
<213> Intelligent people
<400>43
<210>44
<211>110
<212>PRT
<213> Intelligent people
<400>44
<210>45
<211>1203
<212>DNA
<213> hepatitis B Virus
<400>45
<210>46
<211>400
<212>PRT
<213> hepatitis B Virus
<400>46
<210>47
<211>799
<212>DNA
<213> Intelligent people
<400>47
<210>48
<211>217
<212>PRT
<213> Intelligent people
<400>48
<210>49
<211>963
<212>DNA
<213> Intelligent people
<400>49
<210>50
<211>320
<212>PRT
<213> Intelligent people
<400>50
<210>51
<211>107
<212>PRT
<213> Intelligent people
<400>51
<210>52
<211>107
<212>PRT
<213>Mus musculus
<400>52
<210>53
<211>119
<212>PRT
<213> Intelligent people
<400>53
<210>54
<211>119
<212>PRT
<213>Mus musculus
<400>54
<210>55
<211>214
<212>PRT
<213> Intelligent people
<400>55
<210>56
<211>448
<212>PRT
<213> Intelligent people
<400>56
<210>57
<211>8540
<212>DNA
<213> Intelligent people
<400>57
<210>58
<211>9209
<212>DNA
<213>Mus musculus
<400>58
<210>59
<211>384
<212>DNA
<213>Mus musculus
<400>59
<210>60
<211>128
<212>PRT
<213>Mus musculus
<400>60
<210>61
<211>420
<212>DNA
<213>Mus musculus
<400>61
<210>62
<211>140
<212>PRT
<213>Mus musculus
<400>62
<210>63
<211>1395
<212>DNA
<213> Intelligent people
<400>63
<210>64
<211>464
<212>PRT
<213> Intelligent people
<400>64
<210>65
<211>1962
<212>DNA
<213> Intelligent people
<400>65
<210>66
<211>653
<212>PRT
<213> Intelligent people
<400>66
<210>67
<211>1290
<212>DNA
<213> Intelligent people
<400>67
<210>68
<211>429
<212>PRT
<213> Intelligent people
<400>68
<210>69
<211>351
<212>DNA
<213> Intelligent people
<400>69
<210>70
<211>116
<212>PRT
<213> Intelligent people
<400>70
<210>71
<211>498
<212>DNA
<213> Intelligent people
<400>71
<210>72
<211>165
<212>PRT
<213> Intelligent people
<400>72
<210>73
<211>165
<212>PRT
<213> Intelligent people
<400>73
<210>74
<211>588
<212>DNA
<213> Intelligent people
<400>74
<210>75
<211>195
<212>PRT
<213> Intelligent people
<400>75
Claims (146)
Applications Claiming Priority (15)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/411,043 | 2003-04-09 | ||
| US10/410,997 | 2003-04-09 | ||
| US10/411,026 | 2003-04-09 | ||
| US10/410,980 | 2003-04-09 | ||
| US10/411,037 | 2003-04-09 | ||
| US10/410,945 | 2003-04-09 | ||
| US10/410,897 | 2003-04-09 | ||
| US10/411,044 | 2003-04-09 | ||
| US10/411,012 | 2003-04-09 | ||
| US10/410,026 | 2003-04-09 | ||
| US10/410,913 | 2003-04-09 | ||
| US10/411,012 US7265084B2 (en) | 2001-10-10 | 2003-04-09 | Glycopegylation methods and proteins/peptides produced by the methods |
| US10/410,930 | 2003-04-09 | ||
| US10/411,049 | 2003-04-09 | ||
| US10/410,962 | 2003-04-09 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1863458A CN1863458A (en) | 2006-11-15 |
| CN100522988C true CN100522988C (en) | 2009-08-05 |
Family
ID=37390748
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB2004800159188A Expired - Fee Related CN100522988C (en) | 2003-04-09 | 2004-04-09 | GlycoPEGylation method and protein/peptide produced by the method |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN100522988C (en) |
| ZA (1) | ZA200508028B (en) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HRP20120522T1 (en) * | 2007-03-30 | 2012-08-31 | Ambrx | Modified fgf-21 polypeptides and their uses |
| DK2342223T3 (en) * | 2008-09-26 | 2017-07-24 | Ambrx Inc | Modified animal erythropoietin polypeptides and their uses |
| TWI793159B (en) * | 2013-10-23 | 2023-02-21 | 美商健臻公司 | Recombinant glycoproteins and uses thereof |
| CN105372214B (en) * | 2014-08-18 | 2019-06-28 | 中国科学院上海有机化学研究所 | A method for identifying N-linked oligosaccharide structures of new erythropoiesis-stimulating proteins |
| SMT202100388T1 (en) * | 2014-10-24 | 2021-09-14 | Bristol Myers Squibb Co | Modified fgf-21 polypeptides and uses thereof |
| KR102619071B1 (en) * | 2017-10-11 | 2023-12-27 | 엘랑코 유에스 인코포레이티드 | Porcine g-csf variants and their uses |
| CN111208284B (en) * | 2018-11-22 | 2021-08-24 | 北京大学 | Glycometabolism labeling probe, kit containing same and application thereof |
| CN113599503A (en) * | 2021-07-28 | 2021-11-05 | 安徽中起生物科技有限公司 | Biological agent for regulating ovarian function and preparation method and application thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4414147A (en) * | 1981-04-17 | 1983-11-08 | Massachusetts Institute Of Technology | Methods of decreasing the hydrophobicity of fibroblast and other interferons |
| CN1255944A (en) * | 1997-05-12 | 2000-06-07 | 凤凰药理学公司 | Modified arginine deiminase |
-
2004
- 2004-04-09 CN CNB2004800159188A patent/CN100522988C/en not_active Expired - Fee Related
-
2005
- 2005-10-04 ZA ZA200508028A patent/ZA200508028B/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4414147A (en) * | 1981-04-17 | 1983-11-08 | Massachusetts Institute Of Technology | Methods of decreasing the hydrophobicity of fibroblast and other interferons |
| CN1255944A (en) * | 1997-05-12 | 2000-06-07 | 凤凰药理学公司 | Modified arginine deiminase |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1863458A (en) | 2006-11-15 |
| ZA200508028B (en) | 2009-01-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7157277B2 (en) | Factor VIII remodeling and glycoconjugation of Factor VIII | |
| US7214660B2 (en) | Erythropoietin: remodeling and glycoconjugation of erythropoietin | |
| US7795210B2 (en) | Protein remodeling methods and proteins/peptides produced by the methods | |
| US7265085B2 (en) | Glycoconjugation methods and proteins/peptides produced by the methods | |
| US8853161B2 (en) | Glycopegylation methods and proteins/peptides produced by the methods | |
| US8716239B2 (en) | Granulocyte colony stimulating factor: remodeling and glycoconjugation G-CSF | |
| US7297511B2 (en) | Interferon alpha: remodeling and glycoconjugation of interferon alpha | |
| US7696163B2 (en) | Erythropoietin: remodeling and glycoconjugation of erythropoietin | |
| US8008252B2 (en) | Factor VII: remodeling and glycoconjugation of Factor VII | |
| US7265084B2 (en) | Glycopegylation methods and proteins/peptides produced by the methods | |
| US7179617B2 (en) | Factor IX: remolding and glycoconjugation of Factor IX | |
| US7399613B2 (en) | Sialic acid nucleotide sugars | |
| US7125843B2 (en) | Glycoconjugates including more than one peptide | |
| US7226903B2 (en) | Interferon beta: remodeling and glycoconjugation of interferon beta | |
| US7439043B2 (en) | Galactosyl nucleotide sugars | |
| HK1080090A1 (en) | Method for remodeling and glycoconjugation of peptides | |
| CN100522988C (en) | GlycoPEGylation method and protein/peptide produced by the method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20090805 Termination date: 20190409 |